Abstract
Malignant neuroendocrine tumors were diagnosed in the stomach of two out of sixty female Sprague-Dawley rats treated for 89 weeks with a high dose of a novel, small molecule, cannabinoid-1 antagonist. The tumors were associated with parietal cell atrophy accompanied by foveolar hyperplasia of the glandular stomach mucosa. Parietal cell atrophy/foveolar hyperplasia was considered test article related at the high dose, given the higher incidence and severity relative to untreated controls, although the precise mechanism of the parietal cell atrophy was undetermined. Spontaneous gastric neuroendocrine tumors are very rare in rats, and the current cases were considered secondary to parietal cell atrophy causing reduced gastric acid secretion and subsequent overstimulation of gastrin release through a feedback loop.
Keywords
Gastric neuroendocrine (NE) tumors are extremely rare spontaneous tumors in rodents with two notable exceptions. Striped field mice (Apodemus agrarius) are reported to exhibit a relatively high incidence of gastric NE tumors, with an incidence of 5% among aged mice. 1 Mastomys (Praomys natalensis) are reported to be particularly prone to the development of spontaneous gastric NE tumors, with an incidence of 40% to 60% observed between the ages of 12 to 24 months. 2 However, despite the rare spontaneous occurrence of these tumors in most rodent species, gastric NE tumors have been induced in common laboratory rats and mice following long-term treatment with various agents that block gastric acid secretion for prolonged periods. This effect on gastric acid secretion activates a positive feedback mechanism that results in prolonged increases in circulating levels of gastrin, a known trophic factor for both epithelial and enterochromaffin-like (ECL) cells of the gastric mucosa.3,4 Gastric NE tumors have also been reported later in the life of rats that have undergone partial gastric corpectomy, which also results in markedly reduced gastric acid secretion with associated increases in plasma gastrin levels. 5
Cannabinoid (CB) receptors have been attractive targets for the pharmaceutical industry because they play a critical role in numerous physiological and pathological conditions. Since CB-1 and CB-2 receptors were cloned in the 1990s, considerable efforts have been made to develop ligands for these receptors. However, only a few of these ligands are currently being used clinically. 6
Here, we present the appearance of gastric NE tumors and parietal cell atrophy that coincided in a long-term rat carcinogenicity study with a novel CB-1 receptor antagonist. The relationship between these gastric lesions is discussed.
Sprague-Dawley (Crl: [SD]) rats (300 males and 300 females), 6 weeks of age, were divided into 5 groups of 60 males and 60 females. Three groups of 60 rats/sex were given a novel CB-1 antagonist once daily by oral gavage at three different doses (low, mid, and high) for 622 consecutive days. Two separate groups of rats (60/sex) were given 0.5% methylcellulose aqueous suspension and served as controls. The test article was not mutagenic or clastogenic in a series of in vitro and in vivo assays. Plasma drug concentrations were measured during weeks 26 and 73. At the end of the dosing period, all surviving rats were euthanized and necropsied. The number of scheduled and unscheduled animals that were submitted for necropsy are shown in Table 1. Survival was similar across control and test article administered groups with no significant trend or differences among groups, suggesting a test article effect on survival/mortality for either gender. A wide range of organs and tissues were processed and microscopically examined (routine hematoxylin and eosin [H&E] stain) from all rats. Microscopic lesions were graded on a scale of 1 to 5 as minimal, mild, moderate, marked, or severe, based on the extent of change detected in the microscopic sections. A Sevier-Munger stain was performed on sections of the tumors as well as normal stomach to aid diagnosis. Briefly, sections were incubated in 20% silver nitrate in distilled water for 15 minutes at 60°C. After being rinsed in water, slides were placed in ammoniacal silver solution for 30 minutes. Slides were then rinsed in water and placed in 5% sodium thiosulfate for 2 minutes, dehydrated through 95% ethanol, absolute ethanol, and xylene, and then mounted with Permount. All procedures performed on these animals were in accordance with regulations and established guidelines reviewed and approved by an Institutional Animal Care and Use Committee.
Summary of relevant microscopic findings in the stomach in the long-term carcinogenicity study in rats.
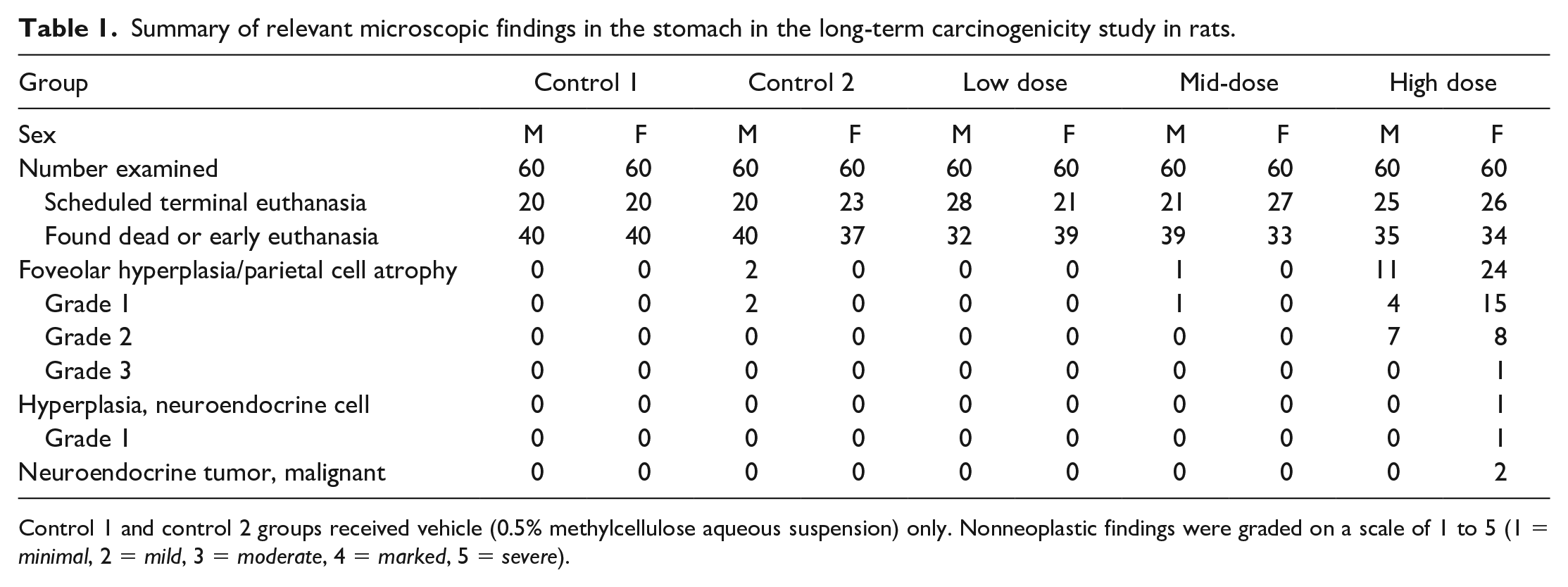
Control 1 and control 2 groups received vehicle (0.5% methylcellulose aqueous suspension) only. Nonneoplastic findings were graded on a scale of 1 to 5 (1 = minimal, 2 = mild, 3 = moderate, 4 = marked, 5 = severe).
An incidence summary of microscopic findings in the stomach is presented in Table 1. Malignant gastric NE tumors occurred in 2 high-dose females and focal NE cell hyperplasia was observed in another high-dose female. All three of these females were from the scheduled termination interval. Microscopically, these tumors were characterized by expansive masses that obliterated the normal gastric mucosal architecture with local infiltration into the underlying submucosa and morphological features consisting of densely packed nests of uniform round to polygonal cells delineated by a fine fibrovascular stroma (Figure 1). Based on the expansile invasive growth pattern, both were considered malignant tumors. There was no evidence of distant metastases. Mitotic figures were rare. Sevier-Munger staining revealed intracytoplasmic fine dark granules in NE cells scattered randomly throughout the fundic mucosa of a normal stomach. In tumors, the presence of similar argyrophilic granules was demonstrated in individual tumor cells confirming their NE origin (Figure 2). The Sevier-Munger argyrophil technique selectively stains ECL cells and a small subset of enterochromaffin (EC) and D cells, and the argyrophilic reaction of Sevier-Munger has been reported to be almost discriminatory when used on fundic ECL cell tumors.7,8 The NE cell hyperplasia noted in one female consisted of small aggregates of monomorphic cells between parietal and chief cells lining the fundic glands (Figure 3).
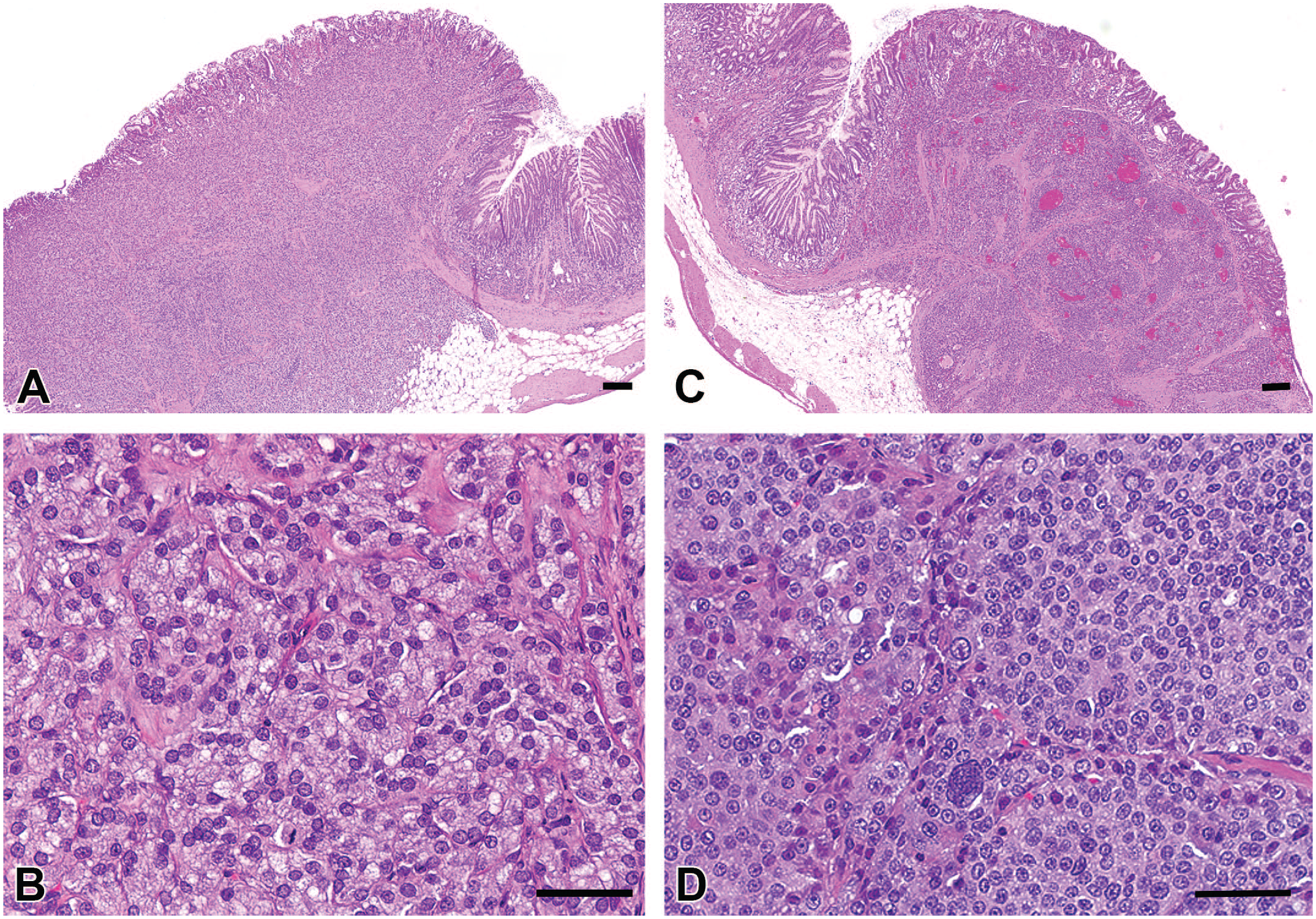
Histopathologic features of gastric NE tumors. (A) and (C) Sections through stomach fundus, showing NE tumors in 2 high-dose rats that exhibit expansive growth, obliterating normal mucosal architecture and infiltrating into the submucosa. Both tumors consist of densely packed nests of round to polygonal cells delineated by fine fibrovascular stroma. Bar = 200 μm. (B) Higher magnification of the NE tumor in (A). Bar = 50 μm. (D) Higher magnification of the NE tumor in (C). Bar = 50 μm. H&E staining. NE, neuroendocrine; H&E, hematoxylin and eosin.
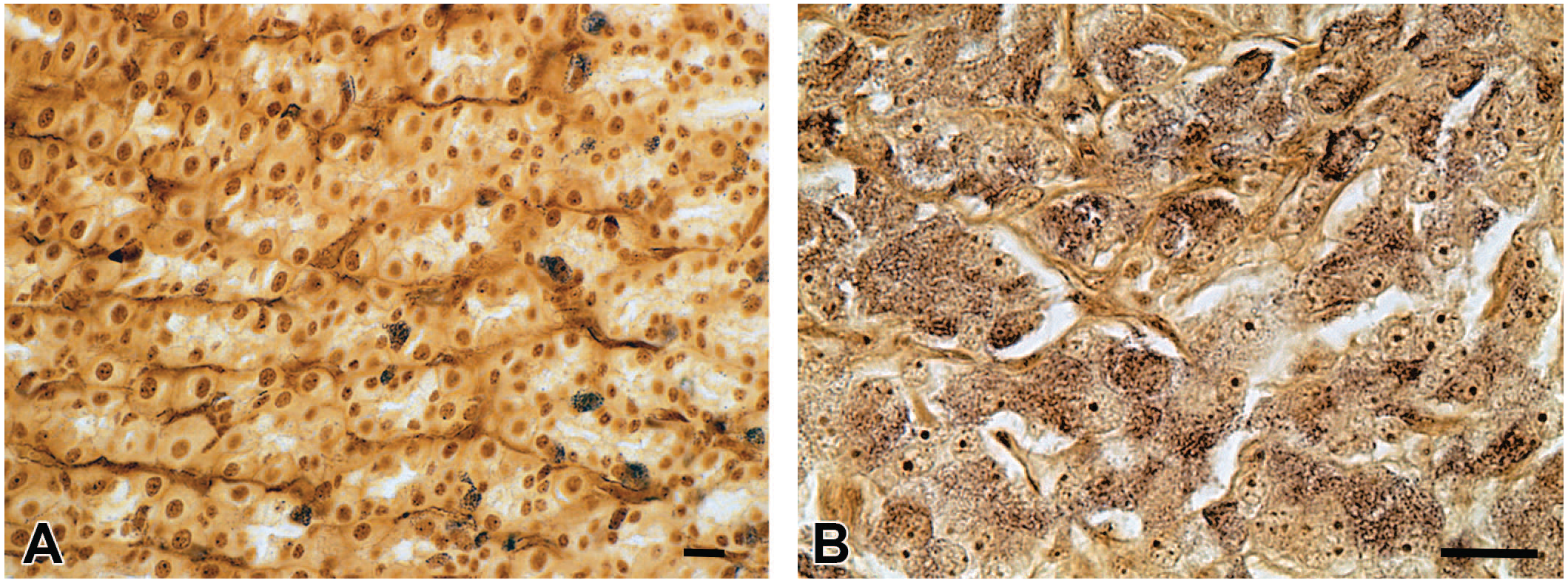
Sevier-Munger staining of gastric NE tumors. (A) Fine black-brown, granular, cytoplasmic staining is evident in NE cells scattered randomly throughout the fundic mucosa of a control rat. Bar = 20 μm. (B) Fine black-brown, granular, cytoplasmic staining is evident in tumor cells. Bar = 20 μm. Sevier-Munger staining. NE, neuroendocrine.
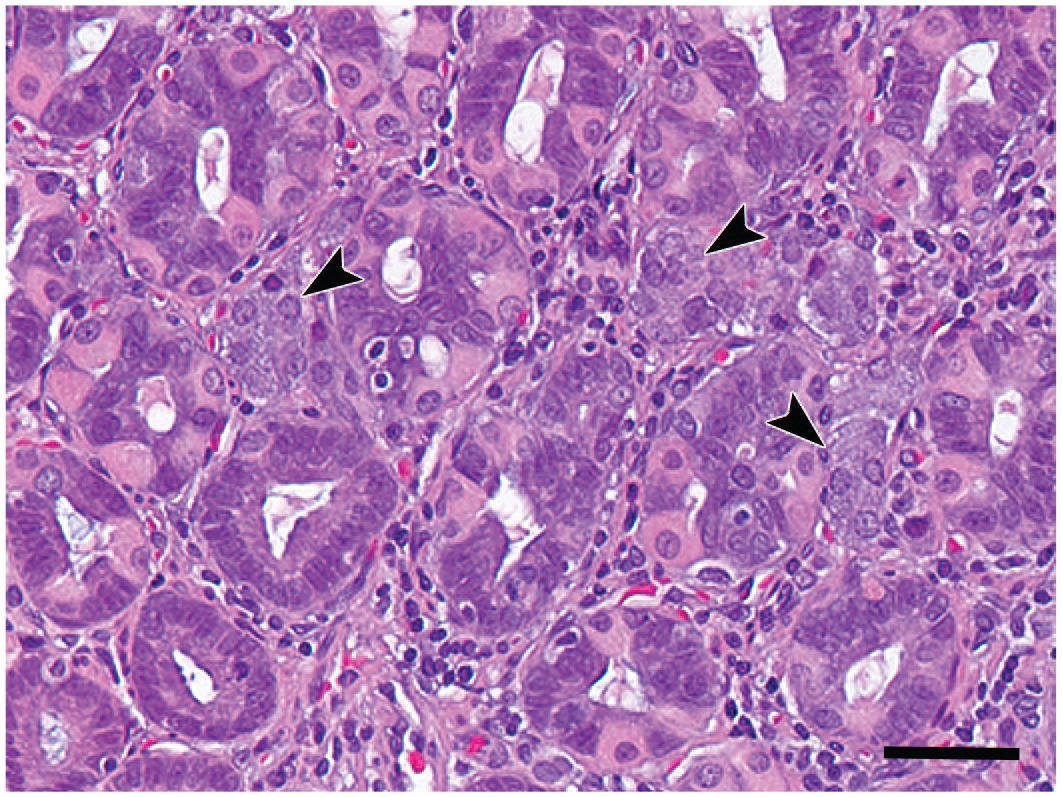
Histopathologic features of gastric ECL cell hyperplasia. Section through stomach fundus containing ECL cell hyperplasia, which consist of focal aggregates of ECL cells (arrowheads). Bar = 20 μm. H&E staining. ECL, enterochromaffin-like; H&E, hematoxylin and eosin.
Parietal cell atrophy accompanied by foveolar hyperplasia was also observed in the glandular mucosa of the stomach with increased incidence and severity in the high-dose group in both sexes. This was characterized by a decrease (atrophy) in the thickness of the parietal cell layer of the fundic mucosa and an increased thickness (hyperplasia) of the superficial layer (foveolar epithelium; Figure 4). The hyperplastic epithelium had a prominent mucous cell component. This finding occurred in animals during the second half of the study and, in females, it was most common in animals from the scheduled termination interval. Foveolar hyperplasia/parietal cell atrophy (8 females) and NE neoplasms (2 females) in the high-dose female group had correlated macroscopic findings in the glandular stomach (raised area, rough surface, or thickened).
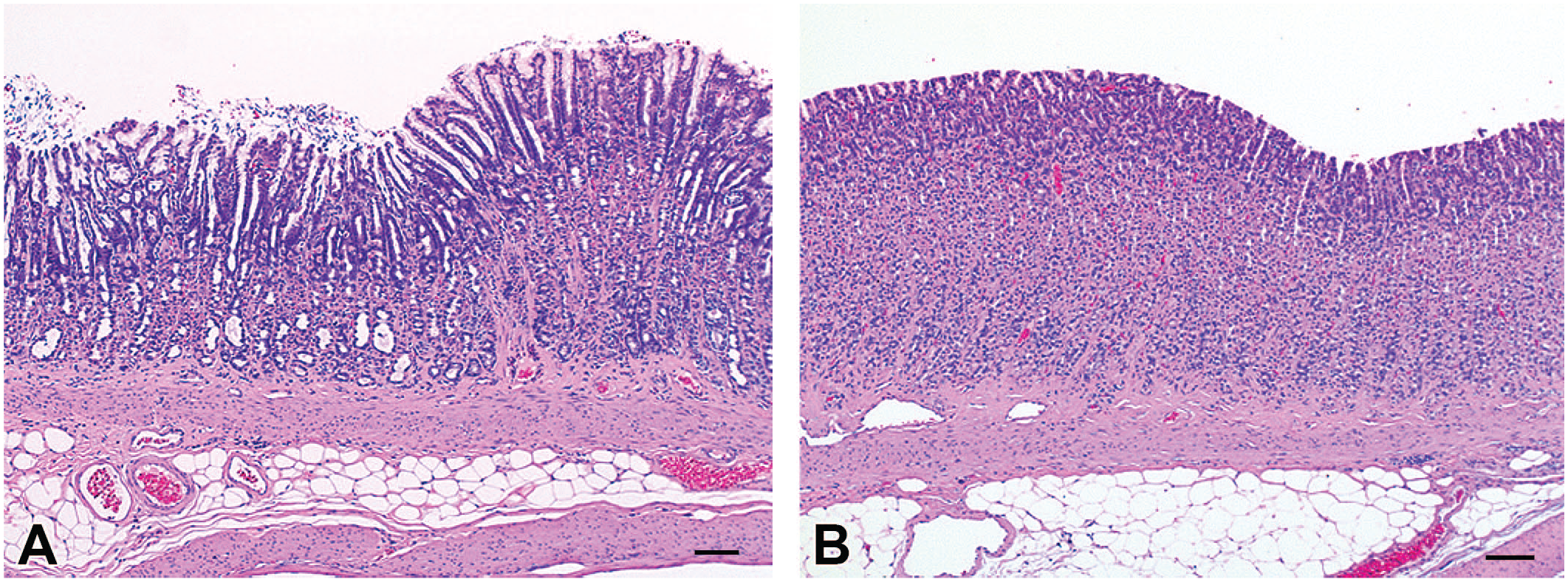
Histopathologic features of parietal cell atrophy/foveolar hyperplasia. (A) Stomach section from a high-dose rat showing parietal cell atrophy/foveolar hyperplasia, characterized by a decrease (atrophy) in the thickness of the parietal cell layer of the fundus and an increased thickness (hyperplasia) of the superficial layer (foveolar epithelium), with the hyperplastic epithelium having a prominent mucous cell component. (B) Stomach section from a control animal showing normal gastric mucosa. Bar = 100 μm. H&E staining. H&E, hematoxylin and eosin.
The gastric NE tumors and hyperplasia in the three high-dose females were considered test article–related effects, given the overall incidence relative to that which would be anticipated spontaneously in rats. As noted previously, gastric NE tumors are extremely rare lesions in this rodent species. Parietal cell atrophy/foveolar hyperplasia was also considered test article–related at the high dose, given the higher incidence and severity relative to controls. The concurrence of all these gastric lesions indicated that they were likely related, with the proliferative lesions probably occurring secondary to the parietal cell atrophy. Although not directly measured, phenotypically this parietal cell effect could be consistent with a functional change characterized by reduced gastric acid secretion, which would subsequently stimulate release of gastrin (from G cell: enteroendocrine cell), a known trophic factor for both epithelial and ECL cells of the gastric mucosa. Prolonged stimulation of fundic ECL cells by hypergastrinemia would, in turn, predispose these cells to hyperplasia/neoplasia. Prolonged hypergastrinemia may also account for the foveolar hyperplasia that was observed within the setting of an atrophic parietal cell layer. Experimental loss of parietal cells has been shown to result in foveolar hyperplasia in association with hypochlorhydria and subsequent hypergastrinemia. 9 Gastrin simultaneously stimulates ECL cells through the cholecystokinin 2 (gastrin) receptor on these cells, resulting in the secretion of histamine, which directly activates parietal cell acid secretion. Parietal cells have an acid stimulatory H2 receptor for histamine, but not toward gastrin because gastrin receptors are distributed mainly in ECL cells and to a lesser extent on parietal cells. 10 The nature of the specific trophic effect of gastrin on the ECL cell is a key component in the understanding of how prolonged increases in plasma gastrin levels can drive the development of ECL hyperplasia and neoplasia.
It was notable that female rats in this carcinogenicity study exhibited higher incidences of test article–related microscopic effects in the stomach when compared with males; however, these differences were ascribed to gender differences in exposure. Test article exposure (Cmax and area under the curve [AUC]0-24) was approximately up to 4- to 5-fold higher in females as compared with males at the high dose.
In previous rat general toxicity studies, oral administration of equivalent doses of this compound for up to 6 months did not result in parietal cell atrophy/foveolar hyperplasia or proliferative NE changes in the stomach. This is consistent with the fact that the gastric changes in this carcinogenicity study only occurred in animals during the second half of the study (earliest identified parietal cell atrophy/foveolar hyperplasia was in a rat euthanized on day 460), with the proliferative NE changes being limited to animals from the scheduled termination interval. Collectively, these results show that long-term administration of the test article is a causative factor for the development of these gastric effects.
Prolonged administration of long-acting inhibitors of gastric secretion has been associated with the development of gastric ECL cell tumors in rodents. In rats treated with the histamine H2-receptor blocker ranitidine for 2 years, gastric acid inhibition was associated with increased plasma gastrin, with subsequent development of pronounced gastric ECL hyperplasia and NE tumors. 4 Similarly, oral administration of ciprofibrate, a potent hypolipidemic compound, to rats produced hypertrophy and increased eosinophilia of parietal cells in the gastric mucosa. Electron microscopic evaluation of parietal cells in this instance revealed small secretory canaliculi with small microvilli, which are changes consistent with the inhibition of acid secretion. After lifetime (2-year) administration of ciprofibrate, ECL cell hyperplasia and malignant NE tumors developed in the fundus of the stomach. 3 Another example is the long-term administration of chloroacetanilide herbicides to rats, which resulted in a combination of mucosal atrophy and ECL tumors in the fundus of the stomach as well as hypergastrinemia, a chain of events induced through activation of a positive feedback loop. 11 Another example is methyleugenol, which was shown to induce ECL tumors along with parietal cell atrophy in a 2-year rat study. 12 In contrast to ECL cells, gastrin-expressing G cells do not seem to be prone to proliferate in response to long-term gastric hypoacidity, given that G cell hyperplasia or tumors have not been reported in rats treated with agents that block gastric acid secretion for prolonged periods. It has also been reported that, whereas the proliferation labeling index of ECL cells increases shortly after drug application and remains high, the labeling index of G cell only increases during the first week then decreases and returns to below control values within a month despite continuous drug-induced achlorhydria in rats. 13 Although not definitively proven, the microscopic features and the plausible underlying mechanism suggested by the data, the neoplasms, and hyperplasia noted in this study were consistent with an ECL origin.
The CB-1 receptor is a member of G protein-coupled receptor family, which is mainly expressed in the central and peripheral nervous systems including the enteric nervous system. 6 In the stomach, CB-1 receptors mediate inhibition of gastric acid secretions and also play a role in gastric motor activity and emptying. 14 CB-1 receptors also appear to play a role on gastric mucosal defense with increasing evidence, suggesting that activation of CB-1 is protective against experimentally induced–mucosal damage in the stomach of rats. 15 Although a definitive mechanism for the parietal cell atrophy in this study was not determined, the known roles of CB-1 in gastric physiology and pathophysiology suggest that lifelong CB-1 antagonism would be a plausible mechanism for the atrophic mucosal change, with associated long-term effects on gastrin secretion accounting for subsequent development of ECL hyperplasia and NE tumor formation.
In conclusion, gastric NE tumors and focal hyperplasia occurred in a long-term carcinogenicity study of a novel small molecule CB-1 antagonist in rats. These proliferative changes were associated with parietal cell atrophy (also known as oxyntic cells) accompanied by foveolar hyperplasia of the gastric fundic mucosa. Collectively, these gastric findings were consistent with alteration of a gastrin feedback loop, resulting in prolonged hypergastrinemia.
Footnotes
Acknowledgements
The authors thank Dr Michael R. Elwell (Apex ToxPath LLC, Apex, NC) for providing his scientific contributions to the investigations.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors are employees of Pfizer Inc. and own Pfizer’s stock. The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Pfizer is the sponsor of the study.
