Abstract
Toxicologic Pathology is the official journal of the Society of Toxicologic Pathology (STP), the British Society of Toxicological Pathology, and the European STP (ESTP). Toxicologic Pathology publishes articles related to topics in various aspects of toxicologic pathology such as anatomic pathology, clinical pathology, experimental pathology, and biomarker research. Publications include society-endorsed Best Practice/Position and Points to Consider publications and ESTP Expert Workshop articles that are relevant to toxicologic pathology and scientific regulatory processes, Opinion articles under the banner of the STP Toxicologic Pathology Forum, Original Articles, Review Articles (unsolicited/contributed, mini, and invited), Brief Communications, Letters to the Editor, Meeting Reports, and Book Reviews. This article provides details on the various publication categories in Toxicologic Pathology and will serve as a reference for authors and readers.
Keywords
Introduction
Toxicologic Pathology is the official journal of the Society of Toxicologic Pathology (STP), the British Society of Toxicological Pathology (BSTP), and the European STP (ESTP). The journal encourages submission of manuscripts relevant to toxicologic pathology including those on safety assessment of pharmaceuticals, chemicals, food additives, pesticides, environmentally relevant chemicals, and medical devices; risk characterization of xenobiotics; investigative studies that contribute to refinement of the safety assessment process; mechanistic studies that provide a basis for interpreting and predicting toxicologic outcome; emerging approaches and methods in toxicologic pathology; spontaneous diseases that may influence toxicology studies, and animal and alternative models relevant to toxicologic pathology.
This journal is a member of the Committee on Publication Ethics and follows the International Committee of Medical Journal Editors Recommendations for the Conduct, Reporting, Editing, and Publication of Scholarly work in Medical Journals (http://www.icmje.org/, last accessed October 28, 2020).
Toxicologic Pathology publishes original and innovative submissions in English with the understanding that the work is unpublished, except in abstract form, and is not being considered for publication elsewhere. However, manuscripts previously posted to preprint servers are acceptable. The aim of this review is to characterize the publication categories in Toxicologic Pathology and to serve as a reference for authors and readers.
General Principles for Best Practice/Position and Points to Consider Articles
Best Practice/Position (BP) and Points to Consider (PTC) articles are published under the banner of the STP Scientific and Regulatory Policy Committee (SRPC), the BSTP, and/or the ESTP. Best Practice/Position articles are generally a definitive statement on a topic that aligns the international toxicologic pathology community on standards of practice. As such, all 3 societies seek to collaborate on the generation, review, and endorsement of these articles, which often includes subject matter experts to ensure accuracy and integrity of scientific and regulatory content. Prior to publication of BP manuscripts, there is an additional review and comment period by the full membership of the sponsoring and endorsing societies to ensure broad input on the proposed best practice. After consideration of all input, the article is submitted for publication and then considered the official position of all endorsing societies.
In contrast, PTC articles do not represent a definitive statement on a particular topic but are intended to raise awareness of and provide guidance on key points and contemporary topics, such as improved nonclinical study designs, emerging technology, or new perspectives on data interpretation from nonclinical toxicity studies. Prior to publication of PTC manuscripts, there is a rigorous review conducted by the relevant body of the sponsoring society (STP SRPC and Executive Committee [EC], BSTP Council, or ESTP EC), which is dictated by the existing policies of that society and may include review by subject matter experts. Additional review by the governing bodies or related committees from each endorsing society is conducted prior to endorsement. However, unlike the BP articles, PTC articles do not include a review by the general membership. After the review period and consideration of all input, the article may be endorsed by the governing bodies of each society and submitted for publication.
Best Practice/Position and PTC publications are impactful to the toxicologic pathology profession and the global community. As such, STP, BSTP, and ESTP have established a common set of guidelines to (1) ensure awareness of new publication initiatives between the 3 organizations and the journal editor, and (2) foster collaboration from inception through publication which involves scrutiny by societal leadership to determine endorsement decisions. Importantly, the sponsoring society is highly encouraged to request and coordinate endorsement of BP and PTC manuscripts from the other 2 societies. The review and endorsement process generally follows each society’s normal endorsement process; however, these processes are similar and often occur in parallel in order to meet reasonable timelines. In addition to endorsement from the STP, BSTP, and ESTP, there may be collaboration with and/or endorsement from other relevant societies such as the American Society for Veterinary Clinical Pathology, American College of Veterinary Pathologists, or Japanese STP (JSTP).
Best Practice/Position and PTC articles have their category incorporated into the title and a respective disclaimer. In addition, all endorsing societies are reflected in the manuscript or disclaimer. Further details on the unique processes and principals for BP and PTC articles for each society are provided in the following paragraphs. Table 1 provides a general guide for review and endorsement procedures as described; however, exceptions to the process could occur if agreed to by the respective organizations.
STP, BSTP, and ESTP Best Practice, Points to Consider, ESTP Expert Workshop, and Opinion Publication Endorsement and Review.a
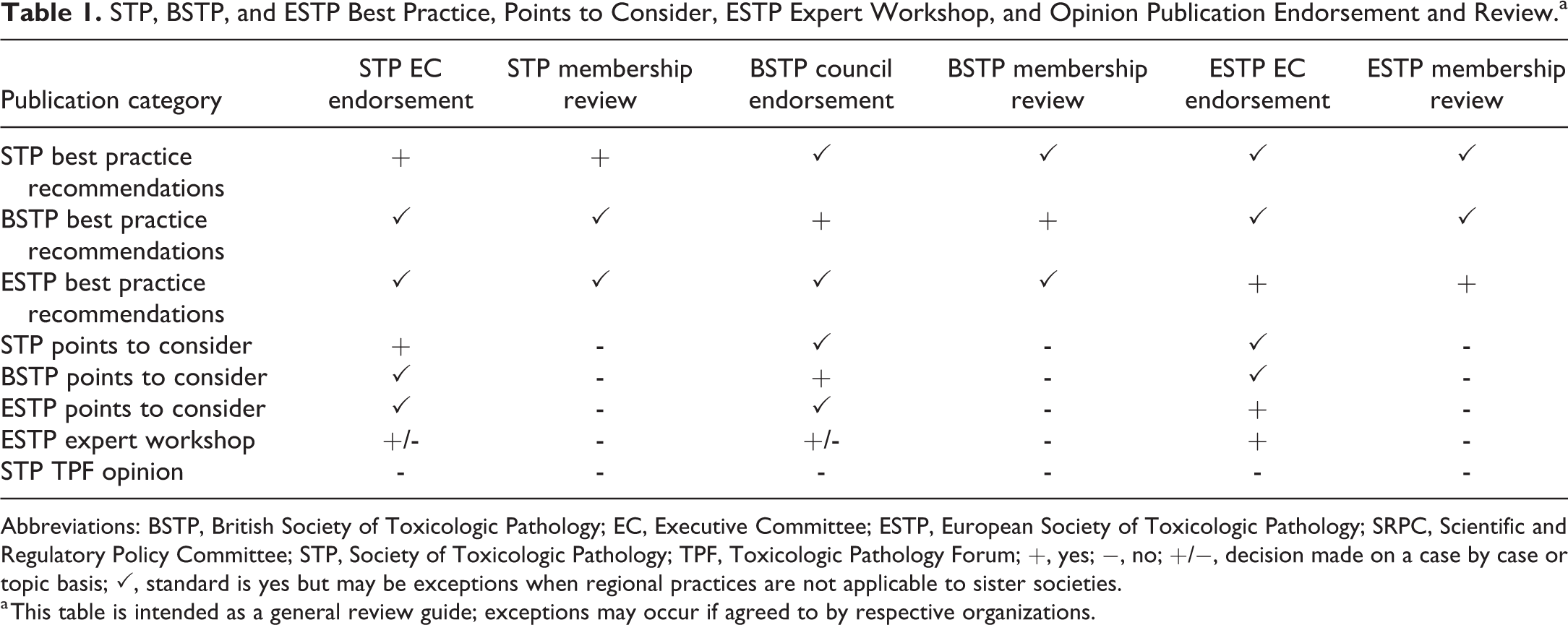
Abbreviations: BSTP, British Society of Toxicologic Pathology; EC, Executive Committee; ESTP, European Society of Toxicologic Pathology; SRPC, Scientific and Regulatory Policy Committee; STP, Society of Toxicologic Pathology; TPF, Toxicologic Pathology Forum; +, yes; −, no; +/−, decision made on a case by case or topic basis; ✓, standard is yes but may be exceptions when regional practices are not applicable to sister societies.
a This table is intended as a general review guide; exceptions may occur if agreed to by respective organizations.
Society of Toxicologic Pathology Best Practice and PTC Publications
The SRPC of the STP is tasked with providing focus and perspective on scientific and regulatory issues that are pivotal to the practice of toxicologic pathology. The SRPC provides scientific leadership on common or emerging global toxicologic pathology issues within the drug, chemical, and medical device industries in order to promote appropriate industry practices and good regulatory policies. One of the ways the SRPC accomplishes this task is by facilitating the publication of PTC articles on scientific and regulatory topics. SRPC products undergo a rigorous vetting process, with review, approval, and oversight by the SRPC and the EC of the STP, from initiation (including topic selection, charter approval, author selection) and continuing through to endorsement and publication. SRPC membership and authors of BP and PTC publications strive to include global representation with members from diverse backgrounds including pharmaceutical, chemical, biotechnical, contract research organization (CRO), academic, and government organizations.
Best Practice/Position and PTC articles cover a wide range of topics including, but not limited to, study design, data interpretation/management, study reports/peer review, system/organ specific pathology, and training of toxicologic pathologists. These topics help serve the interests of the membership by providing concrete recommendations and consensus principles to guide the profession, influencing various issue resolutions, and positioning the STP as a resource for establishing guidelines and policy. This process often involves collaboration with other societies and regulatory officials to harmonize best practices and to seek alignment among key scientific and regulatory stakeholders, and subject matter experts.
The process by which SRPC manuscripts are published has been refined and optimized to enable a successful submission of these important articles. SRPC manuscripts are drafted by SRPC Working Groups that include a lead, an SRPC committee liaison, and individuals from the general STP membership with relevant expertise. Working group membership is reviewed and approved by the EC to ensure broad representation and inclusion of subject matter experts on the topic. Before the manuscript is written, the lead author writes a charter that defines the scope and output of the working group and submits it for review and approval by the SRPC and EC.
The choice between a BP or PTC publication is made by the SRPC and EC, after considering the recommendation from the working group. Although the subsequent review and endorsement processes for BP and PTC publications are similar, there are a few important differences which impact timelines. When a draft PTC or BP manuscript is produced, it is first reviewed by the SRPC and the EC. The time frame for this initial review period is variable and dependent on reviewer and committee schedules, but generally the review occurs within 30 days. For PTC articles, this review phase often culminates in endorsement followed by publication. In contrast, BP manuscripts are subsequently circulated to the full STP membership for an additional review and comment period, which typically is completed in 30 days. After the membership review period, the updated BP manuscript is reviewed one final time by the SRPC and EC in order to receive endorsement and then submitted for publication. As noted previously, the review and endorsement process may occur in parallel with similar processes by other relevant societies (eg, BSTP, ESTP).
SRPC publications will have the article type incorporated into the title and generally include the following disclaimers (edited as needed):
Best Practice Disclaimer: This recommended Best Practice paper is the product of an STP Working Group commissioned by the Scientific and Regulatory Policy Committee (SRPC) of the STP. It has been reviewed by the entire STP membership, including the SRPC, and endorsed by the EC of the STP. These recommendations were also reviewed and endorsed by multiple other societies of toxicologic pathology (list ESTP, BSTP, LASTP, JSTP, etc) representing nations in Asia, Europe, and Latin America.
Points to Consider Disclaimer: This PTC article is the product of an STP Working Group commissioned by the Scientific and Regulatory Policy Committee (SRPC) of the STP. It has been reviewed and approved by the SRPC and EC of the STP but it does not represent a formal Best Practice recommendation of the Society; rather, it is intended to provide key “points to consider” for the toxicologic pathology community. The points expressed in this document are those of the authors and do not reflect views or policies of the employing institutions. Readers of Toxicologic Pathology are encouraged to send their thoughts on these articles or ideas for new topics to the Editor.
British Society of Toxicological Pathology Best Practice and PTC Publications
British Society of Toxicological Pathology Council is the governing body equivalent to the STP and ESTP ECs. Any BSTP member (including Council members), or a regulatory agency, may submit a proposal to BSTP Council for a BSTP-led publication. Council discusses new initiatives, and, if deemed suitable for a publication with BSTP endorsement (generally BP or PTC), a lead author group is formed to execute the new initiative, including outlining the intended scope and output. As with STP and ESTP, the BSTP Council is involved from inception through review, to ensure the highest quality publications. In addition, all BSTP Best Practice manuscripts are reviewed by the BSTP membership prior to Council endorsement or approval. BSTP may request endorsement of BP and PTC from the other 2 societies, especially when the topics are relevant to the global community.
British Society of Toxicological Pathology BP articles will have the article type incorporated into the title and will generally have the following disclaimer (edited as needed):
Best Practice Disclaimer: *This recommended Best Practice paper is a product of a BSTP Working Group commissioned by the Council of the BSTP. It has been reviewed and approved by the Council of the BSTP as well as the entire BSTP membership. These recommendations were also reviewed and endorsed by multiple other societies of toxicologic pathology (list STP, ESTP, LASTP, JSTP, etc) representing nations in Asia, Europe, North America, and Latin America.
European Society of Toxicologic Pathology Best Practice and PTC Publications
The ESTP EC is the governing body equivalent to the STP EC and BSTP Council. The ESTP solicits, supports, and endorses BP and PTC publications. These manuscripts are authored by ESTP Committees, or other ESTP working groups. Similar to the STP and BSTP practices, the ESTP EC and Committee for Scientific Standards (CSS) are involved from inception through review, to ensure the highest quality of these publications. The CSS is composed of toxicologic pathologists from international pharmaceutical and chemical industries, as well as CROs. For BP manuscripts, there is also review and comment from the ESTP membership. In line with the established common set of guidelines among the 3 societies, BP and PTC manuscripts on topics of relevance to the global toxicologic pathology community may also be shared by the ESTP with the other societies for review and comment from inception through publication, which may include endorsement.
European Society of Toxicological Pathology BP articles will have the article type incorporated into the title and will generally have the following disclaimer (edited as needed):
Best Practice Disclaimer: This recommended Best Practice paper is the product of an ESTP Working Group commissioned by the EC of the ESTP. It has been reviewed and approved by the EC of the ESTP as well as the entire ESTP membership. These recommendations were also reviewed and endorsed by multiple other societies of toxicologic pathology (list STP, BSTP, LASTP, JSTP, etc) representing nations in Asia, Europe, North America, and Latin America.
European Society of Toxicological Pathology Expert Workshop Articles: ESTP international expert workshops are invite-only meetings which convene regularly and focus on common or emerging global toxicologic pathology issues while promoting development of appropriate industry practices and good regulatory policies. These workshops are overseen by the ESTP EC and are composed of a diverse international panel of members with particular expertise on a specific topic. Summarizing manuscripts are published in Toxicologic Pathology or another relevant journal. The Expert Workshop manuscripts on topics of relevance to the global toxicologic pathology community may be shared with other societies for review and comments before publication, which may include endorsement.
European Society of Toxicological Pathology international expert workshop publications generally have the article type incorporated into the title and include the following disclaimer:
European Society of Toxicological Pathology Workshop Article Disclaimer: This ESTP expert workshop article is the product of an ESTP Expert Workshop commissioned by the ESTP. It has been reviewed and approved by the ESTP EC, but it does not represent a formal Best Practice recommendation of the Society; rather, it is intended to highlight expert perspective on emerging toxicologic pathology issues that may be relevant to the development of appropriate industry practices and good regulatory policies. The points expressed in this document are those of the authors and do not reflect views or policies of the employing institutions. Readers of Toxicologic Pathology are encouraged to send their thoughts on these articles to the Editor.
Toxicologic Pathology Forum Opinion Articles
Opinion articles are overseen by the Toxicologic Pathology Forum (TPF) committee of the STP which includes representation from the BSTP and ESTP. Toxicologic Pathology Forum provides a venue for the presentation of topics with potential relevance to toxicologic pathology, for the sole purpose of initiating discussion or presenting perspectives not previously considered. The format and subject matter of TPF articles vary, but generally these are short opinion pieces that provide author perspectives on regulatory issues and strategies related to nonclinical toxicology testing and risk assessment. Toxicologic Pathology Forum occasionally publishes full or short topical articles or reviews (including technical reviews), and commentaries on draft and final regulatory guidances, or announcements and events related to regulatory or nonregulatory issues of interest to toxicologic pathologists.
Toxicologic Pathology Forum articles can originate in a number of ways: (1) via request from the TPF committee to the potential authors or (2) unsolicited through author(s) submission directly to the TPF committee or (3) by direct submission to the Toxicologic Pathology journal.
The TPF review timeline and process is expedited compared to other types of articles. The review is generally limited to 5 or 6 TPF committee members and occasionally other STP members or content experts, if deemed necessary by the TPF committee. The review typically focuses on readability and relevance to the readership, with feedback generally offered as primarily discretionary with limited impact on acceptability.
To avoid confusion with STP-endorsed publications such as Best Practice articles, TPF articles have the article type incorporated into the title and a clear disclaimer (see below) indicating that these are personal opinions of the authors that are not endorsed by the STP or the authors’ respective organizations.
Opinion Article Disclaimer: This is an opinion article submitted to the TPF. It represents the views of the authors. It does not constitute an official position of the STP, BSTP, or ESTP, and the views expressed might not reflect the best practices recommended by these Societies. This article should not be construed to represent the policies, positions, or opinions of their respective organizations, employers, or regulatory agencies.
Special Interest Group Articles
The STP supports Special Interest Groups (SIGs) composed of STP members. SIGs enhance professional networking opportunities and scientific exchange among STP members and contribute to the mission and strategic plan of the STP. In addition, SIGs provide scientific expertise for the STP, including serving as a resource for opinion pieces and toxicologic pathology standards, which may lead to publications in Toxicologic Pathology. To facilitate the review and publication of these manuscripts, the SIGs collaborate with standing STP committees and the editors of Toxicologic Pathology. Draft opinion pieces are submitted to the TPF committee, which will review the article and solicit expert external reviews, as needed. As mentioned above, the TPF review is generally limited to TPF committee members or other STP members with relevant expertise, as necessary. For BP or PTC papers, the SIG members collaborate with the SRPC, which will facilitate the formation of a charter describing the rationale and scope of the publication, and the proposed list of authors. Early engagement with the SRPC and the formation of a diverse and balanced working group are critical for a successful outcome. Processes for each type of paper are described in relevant subsections above and in committee standard operating procedures, including the steps required for committee review, endorsement by the respective leadership from the STP, BSTP, and ESTP, and submission to the journal. For manuscripts that do not require committee review or society endorsement (eg, scientific review article), the SIG may submit directly to the journal.
Original Articles
Original articles are reports from hypothesis-driven experiments with clear objectives. This category has the largest number of articles published in Toxicologic Pathology. These articles contain an Abstract, Introduction, Materials and Methods, Results, Discussion, and References. The Abstract is one paragraph consisting of a 150- to 200-word concise description of the objectives, methods, findings, and significance of the research. The Introduction contains brief background information, a clear statement of purpose, and the significance of the research being reported. The Materials and Methods section provides a brief description of the methods and experimental design used, which allows for reproducibility by other investigators. The Discussion should include interpretation of the results, study shortcomings, conclusions, and possible implications of the findings. A compliance statement is included regarding rights of subjects in human experimentation and/or humane treatment and care of animals in research. If the manuscript is about in vivo experiments, then procedures employed for animal care and handling and anesthesia are included. Tables and figures are used to illustrate important points. These articles do not always have to be about positive results in which the hypothesis was proven, as there is value in sharing negative results.
Review Articles
Review Articles may be unsolicited or invited and summarize the current state of understanding on a topic by surveying and encompassing previously published studies and addressing how the topics are evolving. They are usually written by leaders in a particular discipline and have the same structure as Original Research Articles (Abstract, Introduction, Materials & Methods, Results, Discussion, Acknowledgements, References). Authors of a Review Article may be unsolicited or invited by the editors. Reviews are often widely read (e.g., by researchers looking for a robust introduction into a field) and highly referenced by citing many primary research articles.
Mini Reviews are succinct articles that have the same structure as full-length Reviews and Original Research Articles, but in a shorter form. Mini Reviews are prepared using the same formatting instructions as Original Research Articles, except for the use of section headings only for the Abstract, Acknowledgements, and References. Mini Reviews may describe any topic relevant to the practice of toxicologic pathology that does not warrant a full-length manuscript. Mini reviews are summaries of recent insights or advances in areas within the scope of the journal and are usually from experts in a particular specialty area. Authors provide a balanced overview of the field without emphasis on their own work or those of close colleagues. Mini Reviews generally do not exceed 7 double-spaced, typed pages. In addition to the text, there may be 1 to 2 figures and 1 to 2 tables. Abstracts should not exceed 150 words and the number of citations should be limited to 15. Such parameters are subject to change over time, so review of current author instructions, located on the journal website, is strongly recommended prior to composition and submission of any type of article.
Brief Communications
Brief Communications are succinct articles which have the same structure as full-length Original Research Articles but in much shorter form. Section headings are used only for the Abstract, Acknowledgements, and References. A Brief Communication may describe any topic relevant to the practice of toxicologic pathology that does not warrant a full-length manuscript, including diagnostic or special reports. Diagnostic Reports present a clinical, gross, and/or histopathologic abnormality followed by expository text defining the morphologic and etiologic diagnoses. Special Reports describe scientific activities of the STP or affiliated societies. Brief Communications generally do not exceed 3 printed pages (usually 6 manuscript pages), including illustrations, tables, and references. Abstracts for Brief Communications are limited to 150 words and the number of citations is limited to 15.
Letters to the Editor
Letters to the Editor (LTEs) should have a purpose and convey the message in a short and definitive fashion. Letters to the Editors may be written on any topic that may be of interest to the readers. Letters to the Editors may also be in response to an issue in a recently published article; however, these interpretations should be objective and provide constructive comments. Although articles are always vetted by at least 2 scientific reviewers, the Illustrations Editor and an Associate Editor, LTEs can serve an important function in post-publication peer review and sharing experiences with fellow readers. Typically, one LTE is allowed in response to a published article and the author of the article will be given an opportunity to prepare a response, also in the form of an LTE. Any additional communications between the parties would be outside of the journal. All LTEs are reviewed and approved by the Editor-in-Chief.
Meeting Reports
Meeting Reports provide a summary of research presented in scientific forums with a focus on toxicologic pathology. Given the wide breadth of topics covered during meetings, authors generally focus on areas of research which are innovative and thought-provoking. Additionally, if the Meeting Report is for a large symposium, the authors typically focus on select topics which would be of interest in the field of toxicologic pathology, rather than providing an overview of the entire meeting. The intent is to inform on key topics relevant to the journal readership and provide details (date, location) so that additional material or collaboration can be attained as appropriate. Meeting Reports have a single unstructured short Abstract limited to 50 words. The report itself should be limited to 1000 to 1500 words and generally does not include tables or figures.
Book Reviews
Book Reviews are accepted based on the journals aim, scope, and readership. The goal of a Book Review is to critically evaluate the content, quality, and significance of the reference while providing a thoughtful perspective on similar works and informing the audience of any inaccurate information therein. Book Reviews should be written in a professional manner and be a succinct yet comprehensive overview of the topic. Book Reviews typically do not have an Abstract and are a short synopsis of the central idea, with information on the contributing authors and the applicable industry/government/academia sector, if any. The review should address the interests of the journal readership and provide insight on the book’s utility, uniqueness, and how it contributes to the existing repertoire of information on the field. Book reviews begin with a brief introduction, followed by scope, and an informed, engaging, and constructively opinionated discussion on the topics aforementioned.
Footnotes
Acknowledgments
The authors would like to thank Stephanie Dickinson (Managing Editor, Toxicologic Pathology Editorial Office) and Drs. Frank Geoly, Sean Troth, A. Eric Schultze, Béatrice Gauthier, Gabriele Pohlmeyer-Esch, Adam Hargreaves, and Kevin Keane for their valuable reviews and edits to this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the Intramural Research Program of the NIH, National Institute of Environmental Health Sciences.
