Abstract
Polychlorinated biphenyls (PCBs) are fat-soluble environmental pollutants that can accumulate in adipose tissue or be secreted in milk. N-butyl-4-(hydroxy butyl) (BBN), a rat bladder carcinogen, recruits the host metabolism to yield its ultimate carcinogenic form via CYP1s. Since estrogen receptors (ERs) mediate biological responses important for the growth of bladder carcinoma, we investigated PCNA, Cyclin D1, ERs, CYP1s, and AhR expression in BBN rat bladder carcinomas with prenatal PCB exposure. Female SD rats were treated with 7.5 μg, 250 ng, and 2.5 ng of 3,3′,4,4′,5-pentachlorobiphenyl (PCB126)/kg or vehicle on days 13 to 19 post-pregnancy. Six-week-old male offspring were treated with 0.05% BBN for 10 weeks before being anesthetized and the urinary bladder wall incised to expose the bladder carcinomas. N-butyl-4-(hydroxybutyl) bladder carcinoma incidence increased with prenatal PCB exposure dose-dependently. In bladder carcinoma, PCB126 exposure significantly increased PCNA, D1, ERα, CYPIA1, CYP1B1, and AhR expression dose-dependently, and increased ERα expression was particularly prominent. However, the expression of ERβ was low, independent of the volume of PCB126 given, indicating similarity to the Vehicle group. We conclude that prenatal PCB126 exposure in rats can induce PCB126 to dose-dependently metabolize BBN via CYP1A1, and contribute to bladder carcinogenesis with upregulation of ERα expression.
Introduction
Urinary bladder carcinoma is one of the most frequently diagnosed neoplasms, it is the fifth most common carcinoma in men and ninth most common in women in the United States, and accounts for significant morbidity and mortality. 1 There is a significant gender disparity in bladder cancer incidence with 3 to 4 times more diagnoses in men than women, and the antiestrogenic character of bladder carcinoma has been thought to be responsible for its male-dominant induction. 2,3 It has been thought that estrogen receptors (ERs) are important mediators of bladder neoplasms, 1,4,5 but detailed expression of ERs in chemically induced bladder carcinogenesis in animals is unclear.
Estrogens, particularly 17b-estradiol (E2), are potent regulators of cell growth in various tissues. These effects are mediated primarily through binding to the two major ER subtypes (ERα, ERβ) that are thought to be ligand-activated transcription factors. Ligand binding induces a conformational change with ERs that may stimulate high-affinity binding to specific estrogen response elements or regulate transcription at promoter elements by directly binding heterologous transcription factors through protein–protein interaction. 6,7 ERα and ERβ are encoded by unique genes located in different chromosomes and exhibit overlapping tissue distribution and function. 8 Moreover, ERα and ERβ have different transcriptional activities in certain ligands are promoter context dependent, and exhibit distinct tissue- and cell-type-specific expression patterns, with ERα being found predominantly in normal pituitary, breast, uterus, vagina, testis, liver, and kidney, whereas ERβ is mainly expressed in normal thyroid, ovary, prostate, skin, urinary bladder, lung gastrointestinal tract, bone, and cartilage tissue. 9 –13
A recent report on analysis of 141 male and female patients showed that approximately 50% of benign urothelium and approximately 27% of bladder carcinomas were positive for ERα. 14 Another study has reported that 60% of human bladder carcinoma were positive for ERα. 7 In contrast, postmenopausal women show bladder carcinomas which are negative for ERα expression. 15 In vitro, high ERα expression was found to be upregulated in bladder cancer cell lines. 7 ERα knockdown has resulted in remarkable inhibition of tumor growth and tumor metastasis in nude mice. 7 However, it has been reported that exposure of N-butyl-4-(hydroxybutyl) (BBN) for bladder carcinogenesis suppresses ERα. 16 Moreover, ERβ has been detected in human bladder cancers in multiple studies, with up to 81% of tumors expressing ERβ. 16,17 There are various studies involving immunohistochemical staining investigations of ERα and ERβ protein expression in bladder carcinoma, which have reported inconsistent results. 1,14,18 The reason for these discrepancies remains unclear, but Goto and Miyamoto 1 suggested that the discrepancies occurred because of the antibodies used for analyses in terms of the specifically differing specificities of the antibodies used in previous studies. While studies concerning the ERs’ messenger RNA (mRNA) in bladder carcinoma are limited, two studies have described their expression in human urinary bladder 4 and in bladder cell lines. 8 In the present study, we applied antibodies that revealed a significant homology between the protein expression and mRNA expressions in our previous studies. 19 –25
Polychlorinated biphenyls (PCBs), dioxin-like environmental pollutant compounds, are a heterogeneous group of human-made organic compounds that are widely present in the environment. 26 The chemical stability and lipophilicity of PCBs and their resistance to degradation results in their persistence and concentration in food-chains, 27 as well as their bioaccumulation in human adipose tissue, blood, and breast milk. 28 –30 Furthermore, transplacental and lactational transfers of PCBs to a developing fetus and infant, at the perinatal stage, have the potential to cause adverse effects. 20 –25,31 –33
One of the most important factors for the sensitivity to bladder carcinogenesis is metabolism of the administered carcinogenic agent. N-butyl-4-(hydroxybutyl) is a procarcinogen that recruits the host metabolism to yield its ultimate carcinogenic form, N-butyl-N-(3-carboxy-propyl) nitrosamine via cytochrome CYP1A1, an extrahepatic phase I metabolizing enzyme, that is well known to play a decisive role in BBN urinary tumor induction. 34 –36 Moreover, it is well-known that PCBs are high inducers of CYP1 including CYP1A1. 20,21,23 –25,33 The present study investigated the expression of PCNA, cyclin D1, ERα, ERβ, CYP1A1, CYP1B1, and AhR in BBN-induced rat bladder carcinoma with in utero and lactational PCB126 exposure, using immunohistochemistry, Western blot, comparative real-time polymerase chain reaction (RT-PCR), and real-time quantitative polymerase chain reaction (RT-qPCR) analyses.
PCB126 has been shown to be transferred not only to the fetus during embryonic life but also to the next generation in large quantities after birth via breastfeeding and affect perinatal stages. 31,32 Hence, we have previously reported findings using rats that prenatal exposure to a relatively low dose and high dose of PCB126 affected subsequent next generations. 19 –25 However, the effect of prenatal exposure to PCB126 on bladder carcinogenesis is unclear. In the present study, we investigated the prenatal PCB126 effect on BBN-induced bladder carcinogenesis.
Material and Methods
Animals, Chemicals and Treatments
Sixty 8-week-old female Sprague-Dawley rats on gestation day 0 were procured from SRL Co. Upon arrival, the animals were distributed into 4 dose groups using body weight randomization. Rats were housed, 1 per plastic cage for pregnant animals and 3 per plastic cage for their offspring, on hardwood-chip bedding in an environment-controlled room on a 12-hour light/12-hour dark cycle at 22 °C ± 2 °C and 55% ± 5% relative humidity, with a conventional diet (MF, Oriental Yeast Co, Ltd). All experimental procedures were conducted following approval of the Animal Care and Use Committee of the Azabu University School of Veterinary Medicine. Guidelines set by the National Institute of Heath and Public Health Service Policy on the Humane Use and Care of Laboratory Animals were followed at all times.
PCB126 was obtained from AccuStandard Inc, and BBN was obtained from Tokyo Kasei Kogyo Co. A lifetime tolerable daily intake (TDI) of PCB126 has been described as ranging from 10 to 100 pg/kg/d. 32 In this study, the dose of BBN was selected in line with previous studies. 20 –23,25,33,37 Here, 3 doses of PCB126 were selected: 25 pg/kg/d as the TDI dose, 102 the TDI dose, 104 the TDI dose, and 3 × 105 the TDI dose. During pregnancy, 15 female rats each were treated with 2.5 ng, 250 ng, or 7.5 µg PCB126/kg body/d (∼0.5 mL/animal, intragastric injection [ig]), and 15 female rats were treated with an equivalent volume of corn oil (∼0.5 mL/animal, ig), on days 12 through 21 postconceptio)n. The offspring were sexed at birth, and litters were reduced so that each dam was left with 8 offspring (4 males/dam). Weaning was carried out to day 21 postpartum. For experiments, 6-week-old male rats were given water with 0.05% BBN for 10 weeks. Each PCB126-treated group (2.5 ng, 250 ng, or 7.5 µg group) was composed of 51 males in each group treated with 0.05% BBN for 10 weeks and 9 males per group treated with 0% BBN for 10 weeks. The rats were observed daily for abnormality and were weighed weekly. Food and water consumption was measured every day. At 16 weeks of age, all animals were deprived of food, but not water, overnight, and sacrificed under isoflurane anesthesia by exsanguination from the posterior aorta. Subsequently, the skin wall was incised to surgically expose the urinary bladder. The urinary bladder was then expanded by 1 mL physiological saline solution and then removed and immersed in Davidson/Hartmann fixed solutions. After urinary bladders were fixed, they were cut into semisplits. Then, two pathologists (SW and HT) counted urinary bladder tumor number and took photographs. The urinary bladder was then cut into 8 strips and photographs were taken of 21 rats of each PCB group and the Vehicle group, and 4 rats of each 0% BBN group. While 30 rat urinary bladders of each PCB group and the Vehicle group were aseptically cut, any bladder tumors were individually removed, and urinary bladder epithelia from the 0% BBN group were immediately frozen at −80 °C. Then, the Davidson/Hartmann-fixed tissue and part of the frozen urinary bladder tissues were histologically diagnosed by hematoxylin and eosin (H&E) and Periodic acid–Schiff staining. Therefore, the number of urinary bladder carcinomas developed in each rat was determined by comparing the gross bladder photographs and histologically diagnosed tissue sections by 2 pathologists (SW and HT).
Chemical Analysis
Analysis for PCB126 was carried out following the alkaline alcohol digestion method. 20 –23,33,38 Aliquots of homogenized samples were refluxed in 1N KOH-ethanol solution for 1 hour. PCB126 thus extracted into ethanol was transferred to 100 mL of hexane by shaking in a separating funnel. Subsequently, the hexane layer was concentrated and purified by passing it through 1.5 g of silica gel (Wako gel S-1, Wako Co, Ltd) packed in a glass column (10 mm inside diameter × 200 mm length). PCB126 was eluted with 200 mL of hexane at an elution rate of one drop per second. The eluate was concentrated to 5 mL in a Kuderna-Danish concentrator and further purified with 5% fuming sulfuric acid. All samples were injected into a gas chromatograph-mass spectrometer (GC-MS: Shimadzu 9020 DF with an SCAP-1123 data system, Shimadzu Co. Ltd) equipped with an electron-impact ion-source and moving needle-type injection system for the determination and identification of PCB126. The column consisted of a 0.23 mm inner diameter × 30 m length glass capillary coated with silicone. Operating conditions of the GC-MS were as follows: column oven temperature was programmed from 190 °C to 250 °C at 0.5 °C min−1; injector and ion-source temperatures were kept at 250 °C and 280 °C, respectively. PCB126 was determined by selected ion monitoring at m/z 326. The carrier flow of helium was controlled at 0.6 mL min−1.
Immunohistochemistry
Representative sections of each bladder tumor were fixed in 10% phosphate-buffered formalin and routinely processed for immunohistochemistry. Immunohistochemical expressions of PCNA, cyclin D1, ERα, and ERβ were analyzed using the avidin-biotin complex (ABC) method. After deparaffinization, 4-μm-thick sections were treated sequentially with 0.3% H2O2 for 10 minutes, then blocked with 10% goat serum or horse serum in phosphate-buffered saline (PBS) for 20 minutes. Frozen sections were thawed, rinsed in PBS, and treated with primary antibodies of rabbit anti-rat-PCNA (Novocastra Co Ltd diluted 1:50); cyclin D1, rabbit polyclonal antibody (Santa Cruz Biotech; dilution 1:100) 19 ; ERα, a mouse anti-ER antibody, MC-20 antiserum (Santa Cruz Biotech) 19,23,25 that has no affinity for ERβ, 39 and ERβ, a rabbit anti-ER antibody, PA1-310 antiserum (Affinity BioReagents; dilution 1:100) 19,24 that was generated against amino acids in the C-terminal region of rat ERβ corresponding to amino acids 467-485, and has only 6% amino acid homology with rat ERα 39 –41 followed by avidin-biotin 1 complex peroxidase (Vector Lab.) and diaminobenzidine (Sigma). Sections were then counterstained with hematoxylin. As a negative control, nonimmunized rabbit serum was substituted for the primary antibody.
The positive ratio of each immunohistochemistry of PCNA, cyclin D1, ERα, and ERβ was analyzed to evaluate, at random >50 sections of 16 × 104 µm square areas per section for a total of, and total number of immunostained urinary bladder carcinoma nuclei were measured at random in >104 cellular nuclei per each section, and the indices were expressed as percentage values using the quantitative analysis software Patholoscope ver.1.5 and WinRoof ver.7 (Mitani Co.).
Western Blot Analysis
Frozen rat bladder samples were homogenized in 50 mM Tris-HCl, 150 mM KCl (pH 7.4), 1% Triton X-100, and 0.25 mM phenylmethylsulphonyl fluoride (PMSF) and centrifuged at 8000 × g for 30 minutes at 4 °C. The supernatant obtained was centrifuged at 100,000 × g for 90 minutes at 4 °C. The pellet was suspended in 50 mM Tris-HCl (pH 7.4), 1% Triton X-100, and 1 mM PMSF, and the protein concentrations were determined with bicinchoninic acid protein assay reagent kit (Pierce) with bovine serum albumin as a standard. Microsomal samples were subjected to electrophoresis on a 10% SDS polyacrylamide gel using 10 µg of microsomes. The proteins were transferred for 2 hours to a nitrocellulose membrane that was blocked by immersing it in 5% nonfat dried milk in PBS with 0.1% (vol/vol) Tween 20 (PBS-T). Western bot analysis was performed using anti-PCNA monoclonal (Santa Cruz Biotech; dilution 1:1000) 24 ; anti-ERα antibody, MC-20 (Santa Cruz Biotech; dilution 1:500) 23,24 anti-ER antibody, PA1-310 (Affinity BioReagents; dilution 1:1000), anti-rat-CYP1A1 (Affinity Research Products; dilution 1:1000) 21 and anti-rat-CYP1B1 (BD Gentest; dilution 1:500), 21 anti-AhR antibody, H-211 (Santa Cruz Biotech; dilution 1:1000). 21 These antibodies were diluted by PBS-T and incubated for 1 hour at room temperature on an orbital shaker. After being washed 3e times with PBS-T, they were incubated with a 1:2500 dilution of horseradish peroxidase-conjugated anti-rabbit antibody (Amersham Biosciences) for 1 hour on an orbital shaker. After being washed 3 times with PBS-T, the membranes were detected using the ECL Plus Western Blotting Detection System (Amersham Biosciences). 21,23,33
Real-Time Quantitative RT-PCR
Measurement of RNA levels were performed by applying RT-qPCR. RNAs were prepared for PCNA, cyclin D1, ERα, ERβ, CYP1A1, CYP1B1, AhR, and β-actin. Polymerase chain reaction primer pairs used to amplify these specific RNAs are identified in Table 1. Total RNA was isolated from frozen bladder samples using an acid guanidinium thiocyanate-phenol-chloroform method. 20,21,23,24,42 Total bladder RNA (1 μg) was reverse transcribed using Reverse Transcriptase Superscript II (Life Technologies, Inc) in a total volume of 20 µg following the manufacture’s protocol. Controls included for each reaction were the RNA sample without reverse transcriptase (RNA-RT) and no RNA with reverse transcriptase (no RNA+RT). Polymerase chain reactions were performed by first-strand complementary DNA (cDNA) synthesis (2 μL) from each sample with 1× reaction buffer, 2 mM MgCl2, 200 μM deoxynucleotide triphosphate, and 0.5 U/µL Taq Gold (PE Biosystems), including PCNA, cyclin D1, ERα, ERβ, CYP1A1, CYP1B1, AhR, and β-actin with 0.5 M each forward and reverse primer. Amplifications were at 94 °C for 30 s, 57 °C for 30 s, and 72 °C for 30 s for 30 cycles using the GeneAmp PCR system 9700 (PE Biosystems). RNA-RT, no RNA+RT, and no DNA controls were included with each PCR run. Polymerase chain reaction products were separated by 2% agarose gel electrophoresis in 1× tris-borate-EDTA buffer and stained with 1 μg/mL ethidium bromide, and exposed to film, and normalized to β-actin expression for each sample.
Primer Sequences for Comparative RT-PCR.

For each RNA sample, 100 ng was then used as template for first-strand cDNA synthesis using a TaqMan Reverse Transcription kit, following a 2-step RT-PCR manufacturer’s protocol (PE Biosystem). 19,21,23,24,33 Controls included for each reaction were the RNA sample without reverse transcriptase (RNA-RT) and no RNA with reverse transcriptase (no RNA+RT). Final reaction conditions for reverse-transcription were as follows: 1× TaqMan RT buffer; 5.5 mM MgCl2; 500 uM dATP, dGTP, and dCTP; 1 mM dTTP; Random Hexamers 0.25 μM; 1.25 U/µL MuLV reverse transcriptase and 0.4 U (PE Biosystem). Quantitative analysis of target gene (PCNA, cyclin D1, CYP1A1, CYP1B1, AhR, ERα, and ERβ) mRNA expression was performed by real-time quantitative PCR using the ABI Prism 7700 Sequence Detection System (PE Biosystems) with TaqMan chemistry and probe. The TaqMan probes and primers for target genes were Assay-on-demand gene expression products (Applied Biosystems), and these were oligonucleotides with fluorescent reporter and quencher dyes attached, designed from the mRNA sequence to hybridize to a region between the forward and reverse PCR primers (Table 2). Optimal primer, probe, and cDNA concentrations were determined in a separate set of experiments to ensure that both target gene and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) fragments were amplified with equal efficiency. Polymerase chain reactions were performed with first-strand cDNA synthesis (2 μL) from each sample, Universal PCR Master Mix kit (PE Biosystems), 250 nM TaqMan probe, 0.16 U of AmpErase uracil N-glycosylase, and 900 nM forward-reverse primers of target gene and GAPDH. Three measurements per sample were performed in each of two independent experiments. Results were analyzed with the ABI Sequence Detector software version 1.7 (PE Biosystems). For relative quantification of target genes expression, the standard curve method was applied. The calibrated standard curves for each target gene cDNA and GAPDH amplification plot were examined at 5 different dilutions (containing 100, 50, 25, 10, or 5 ng) of total RNA samples that were obtained from each PCR product using the TOPO II TA Cloning Kit (Invitrogen) following the manufacturer’s recommendations. The target gene normalized values were determined by dividing the average target gene value by the average GAPDH value. The standard deviation (SD) of the quotient was calculated from the SD of the target gene and GAPDH using the following formula 19,21,23,24,33 :
SD = (CV) (mean of target gene value/mean of GAPDH value)
CV2 = CV1 2+CV2 2
CV1 = SD of target gene value/mean of target gene value
CV2 = SD of GAPDH value/mean of GAPDH.
ID Number of TaqMan Probes and Primers (Assay-on-Demand Gene Expression Products) Used for Real-Time Quantitative RT-PCR.a

a Assay-on-demand gene expression was supplied by Thermo Fisher Scientific, Tokyo, Japan.
The normalized amount of target gene value was a unitless number that can be used to compare the relative amount of target gene in different samples. In this study, the liver of rats 16 weeks of age in the Vehicle group without BBN induction was designated as the calibrator, and we divided the average target gene value by the average calibrator valueaccording to the manufacturer’s instructions for quantification of relative gene expression (User Bulletin #2; P/N 4303859).
Statistical Analysis
For each set, the mean value, standard deviation, and standard error of the mean were calculated and compared using Scheffé’s F-tests or Chi-square tests with the computer statistical analysis system Stat View-J 5.0 (Abacus Concepts).
Results
The body weights were similar between the PCB group and the Vehicle group (Figure 1). The bladder weight/body weight ratio were significantly gradually increased with prenatal exposure to PCB in a dose dependent manner (Figure 2). Macroscopical and pathological analyses showed that bladder tumors were observed in all rats of the BBN-treated groups, and the pathological analysis revealed the bladder carcinomas were papillary and protruded into the lumen. The shape of these bladder carcinomas was similar in the PCB group and the Vehicle group (Figures 3 and 4). The number of bladder carcinomas per urinary bladder were significantly increased with prenatal exposure to PCB in a dose dependent manner (Figures 4 and 5).
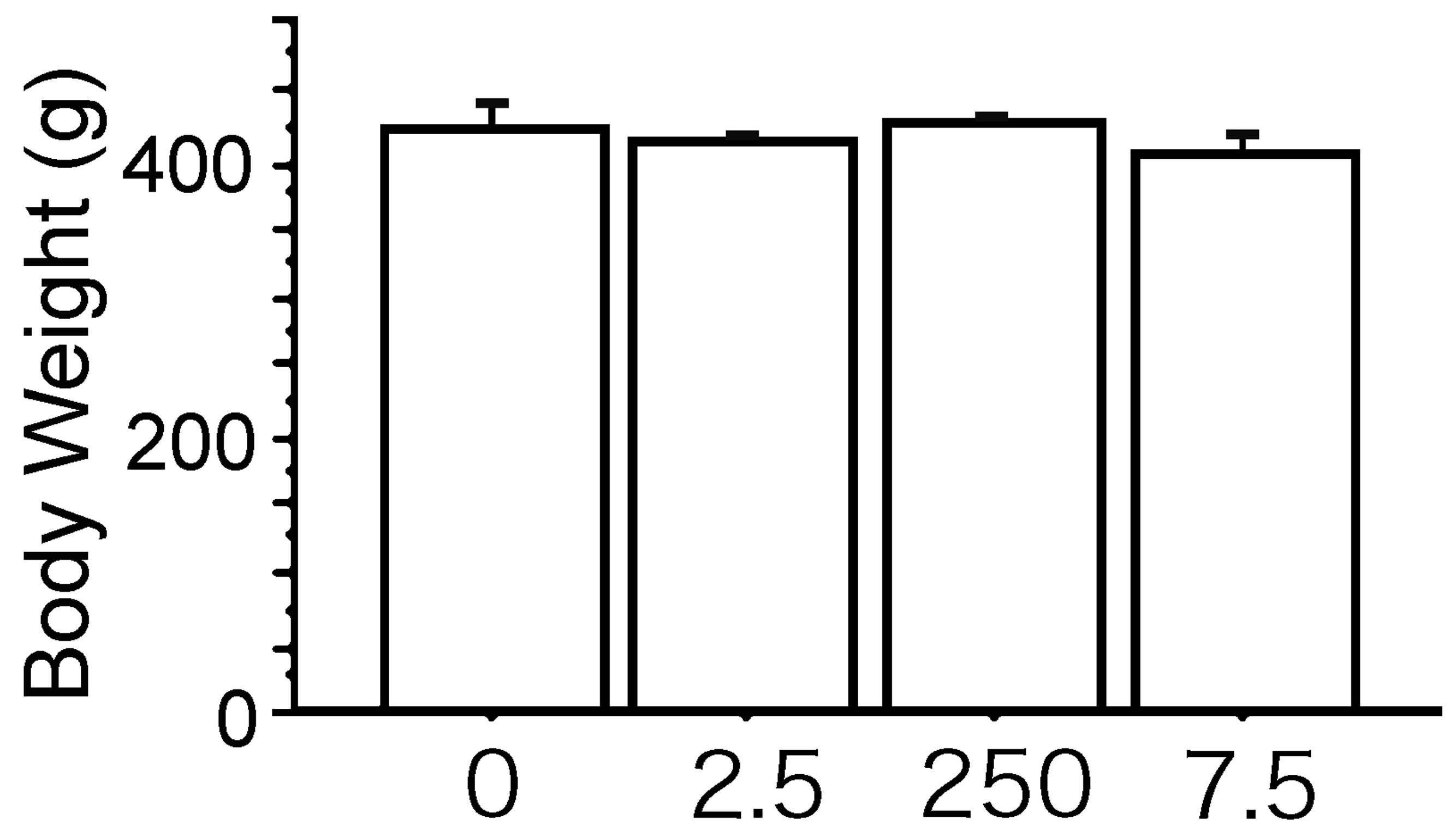
The body weights of 16-week-old male rats in the polychlorinated biphenyl (PCB) groups are similar to those in the Vehicle group. 0, The Vehicle group; 2.5, the PCB 2.5 ng group; 250, the PCB 250 ng group; 7.5, the PCB 7.5 µg group. Data are mean ± standard error of the mean. Scheffé’s F test. All results are nonsignificant.
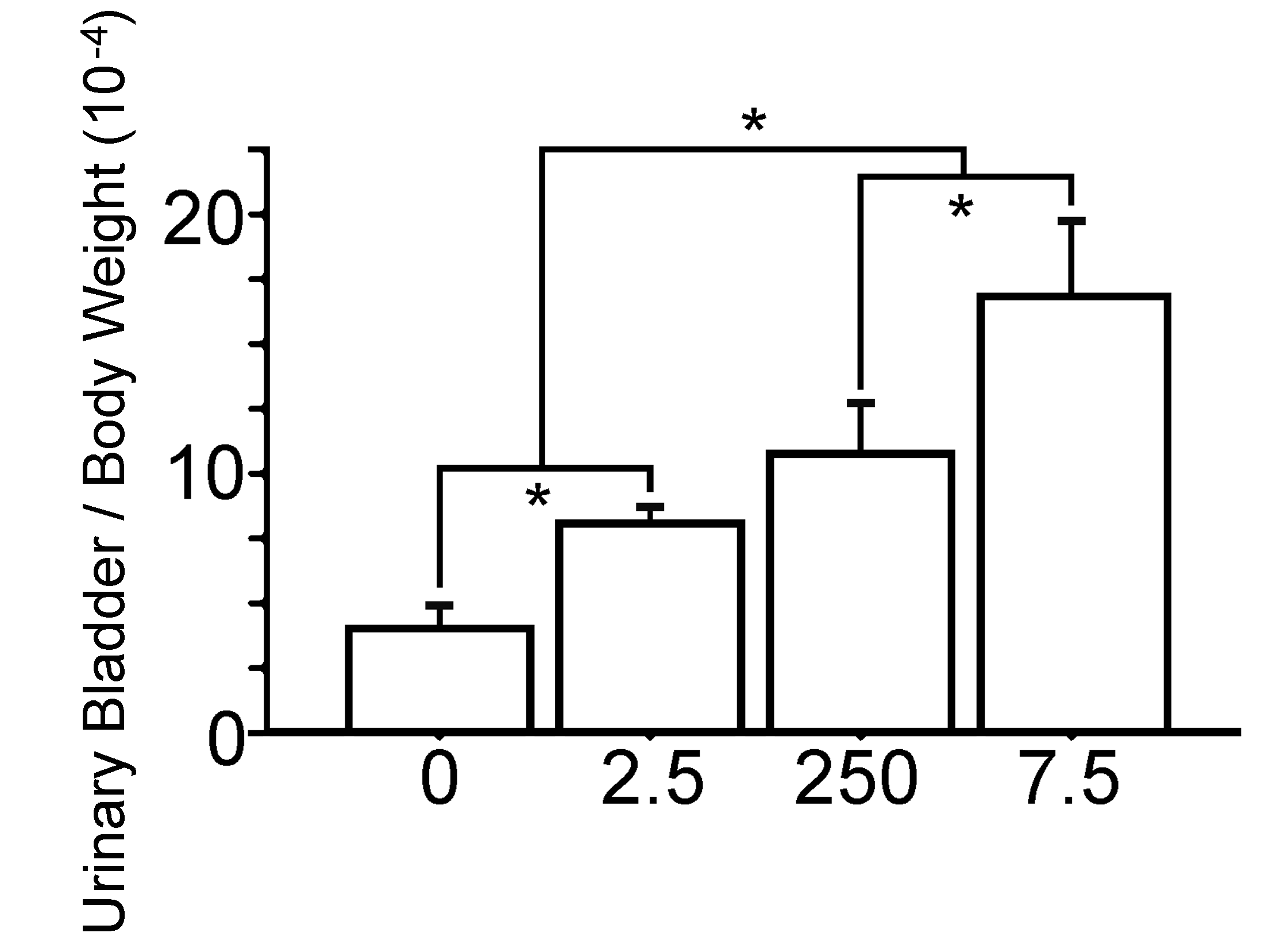
The bladder weight/body weight ratio of 16-week-old male rats are significantly increase with prenatal exposure to polychlorinated biphenyl (PCB) in a dose-dependent manner. 0, the Vehicle group; 2.5, the PCB 2.5 ng group; 250, the PCB 250 ng group; 7.5, the PCB 7.5 µg group. Data are mean ± standard error of the mean. *Scheffé’s F test, P < .01.
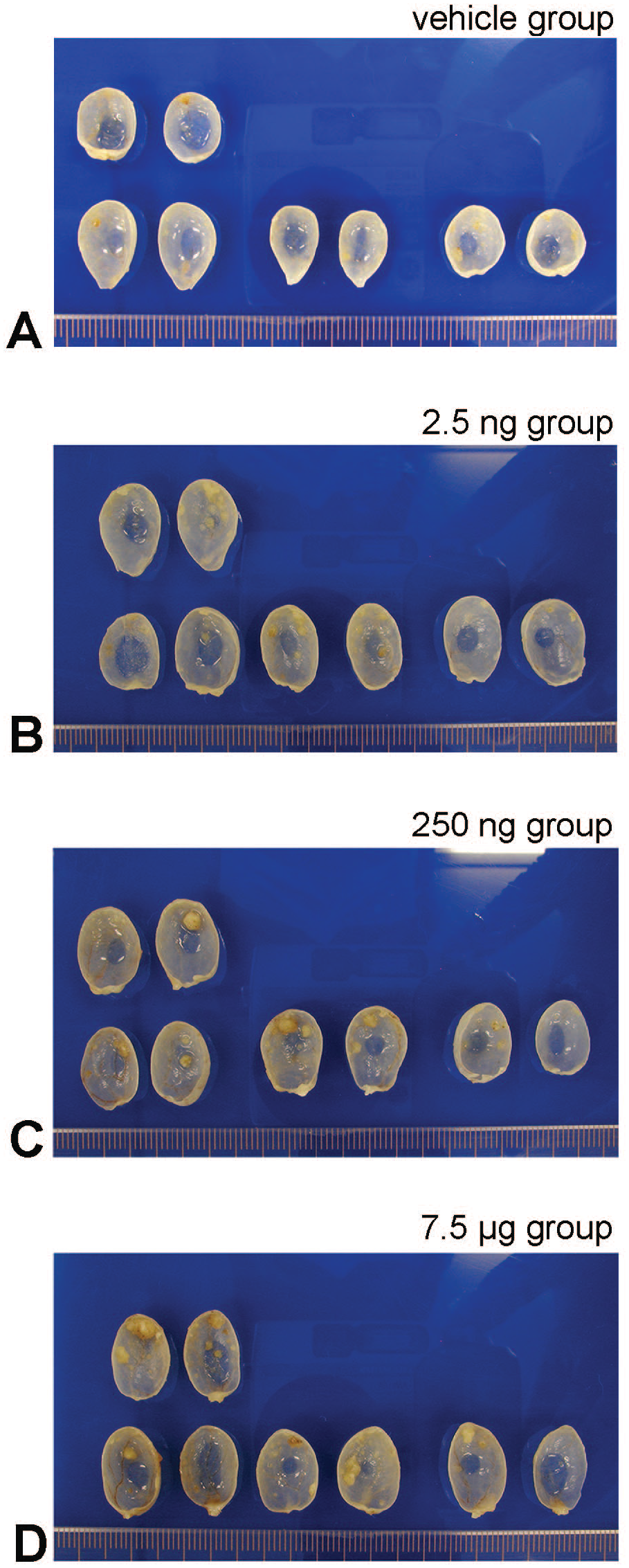
Macroscopical features of the split halves of 16-week-old, the split halves of the rat urinary bladders with 10 weeks BBN administrated, after prenatal treatment (ig) PCB126. The bladder carcinomas are papillary and protrude into the lumen. (A) Shows the Vehicle group urinary bladder with few bladder carcinomas. (B) The 2.5 ng group, (C) the 250 ng group, and (D) the 7.5 µg group are prenatal treated polychlorinated biphenyl (PCB) groups show PCB dose-dependent increase number of bladder carcinoma. Scale 1 mm.
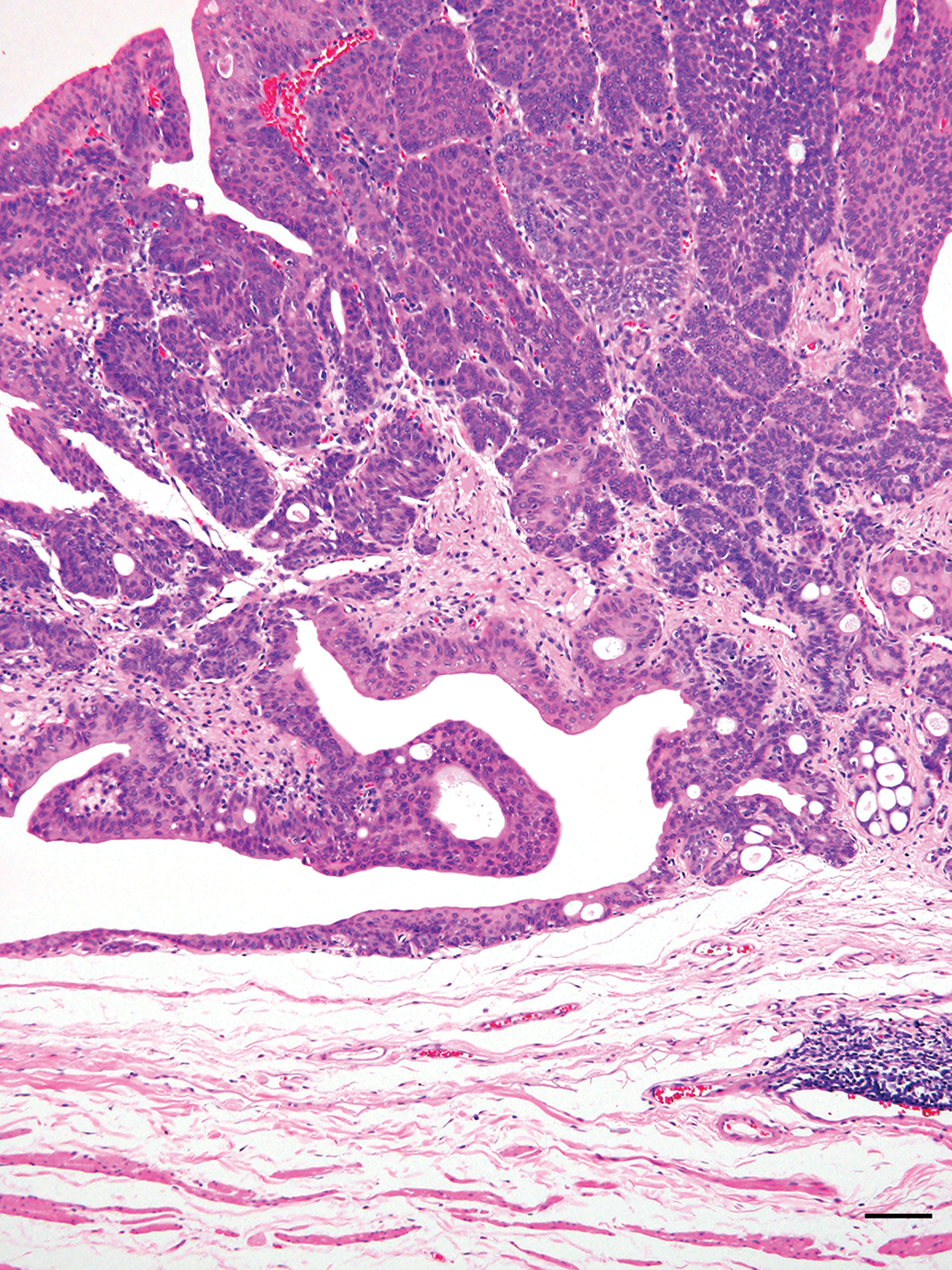
Representative image, low power view of the BBN-induced bladder carcinoma protruded into the bladder lumen. Bladder carcinoma of the 7.5 µg group. Hematoxylin and eosin stain, Scale 100 μm.
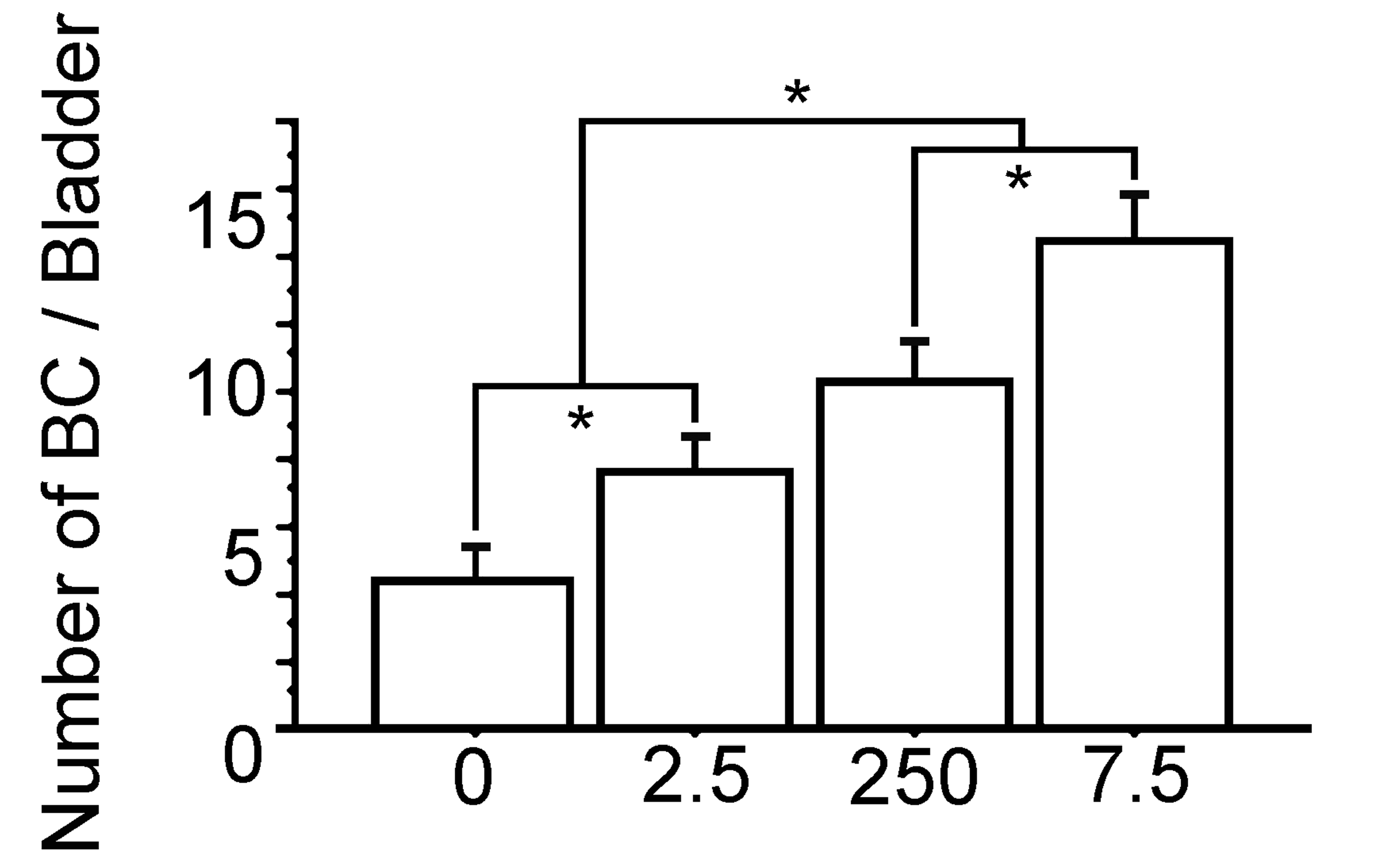
The number of bladder carcinomas per each urinary bladder of 16-week-old male rat are significantly increased with prenatal exposure to polychlorinated biphenyl (PCB) in a dose-dependent manner. 0, the Vehicle group; 2.5, the PCB 2.5 ng group; 250, the PCB 250 ng group; 7.5, the PCB 7.5 µg group. Data are mean ± standard error of the mean. *Scheffé’s F test, P < .01.
Concentration of PCB in Bladder Carcinoma
The concentration of PCB126 in the bladder carcinoma compared to the Vehicle group was 3.0-fold higher in the PCB 7.5 µg group, about 1.2-fold in the PCB 2.5 ng group, and about 1.7-fold higher in the PCB 250 ng group (Table 3).
Concentration of PCB126 in N-butyl-4-(hydroxy butyl) (BBN) Induced Bladder Carcinomas of Rats After Prenatal Exposure, at 16 Weeks of Age.a
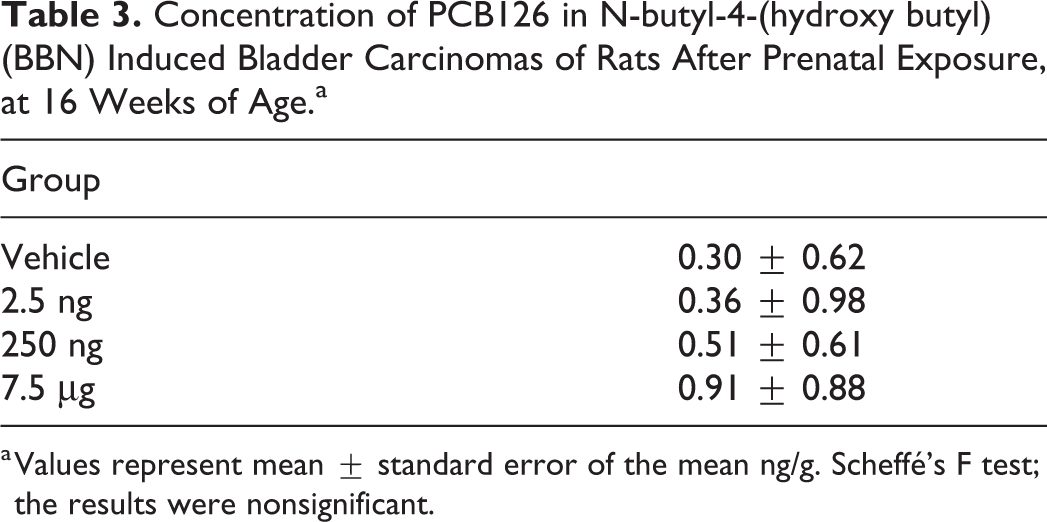
a Values represent mean ± standard error of the mean ng/g. Scheffé’s F test; the results were nonsignificant.
Histopathological and Immunohistochemical Analysis of Bladder Carcinoma
Histopathological morphology of the bladder carcinomas in the PCB groups was similar to that in the Vehicle group (Figures 6 –13). The histopathological investigations revealed that all bladder carcinomas were noninvasive papillary transitional cell carcinomas. The bladder carcinoma showed a number of papillary growth tumors with little cellular/nuclear dysplasia, and tumor cell mitoses were quite few. Moreover, the bladder carcinomas did not show any muscular or intravascular invasions or metastasis (Figures 6 –13). While the urinary bladder mucosa revealed increased urothelium proliferation including simple hyperplasia, papillary/nodular hyperplasia, and papilloma were also observed. It was very difficult to determine the exact number of these lesions by histopathological analysis, so these histological changes were not considered in the present study.
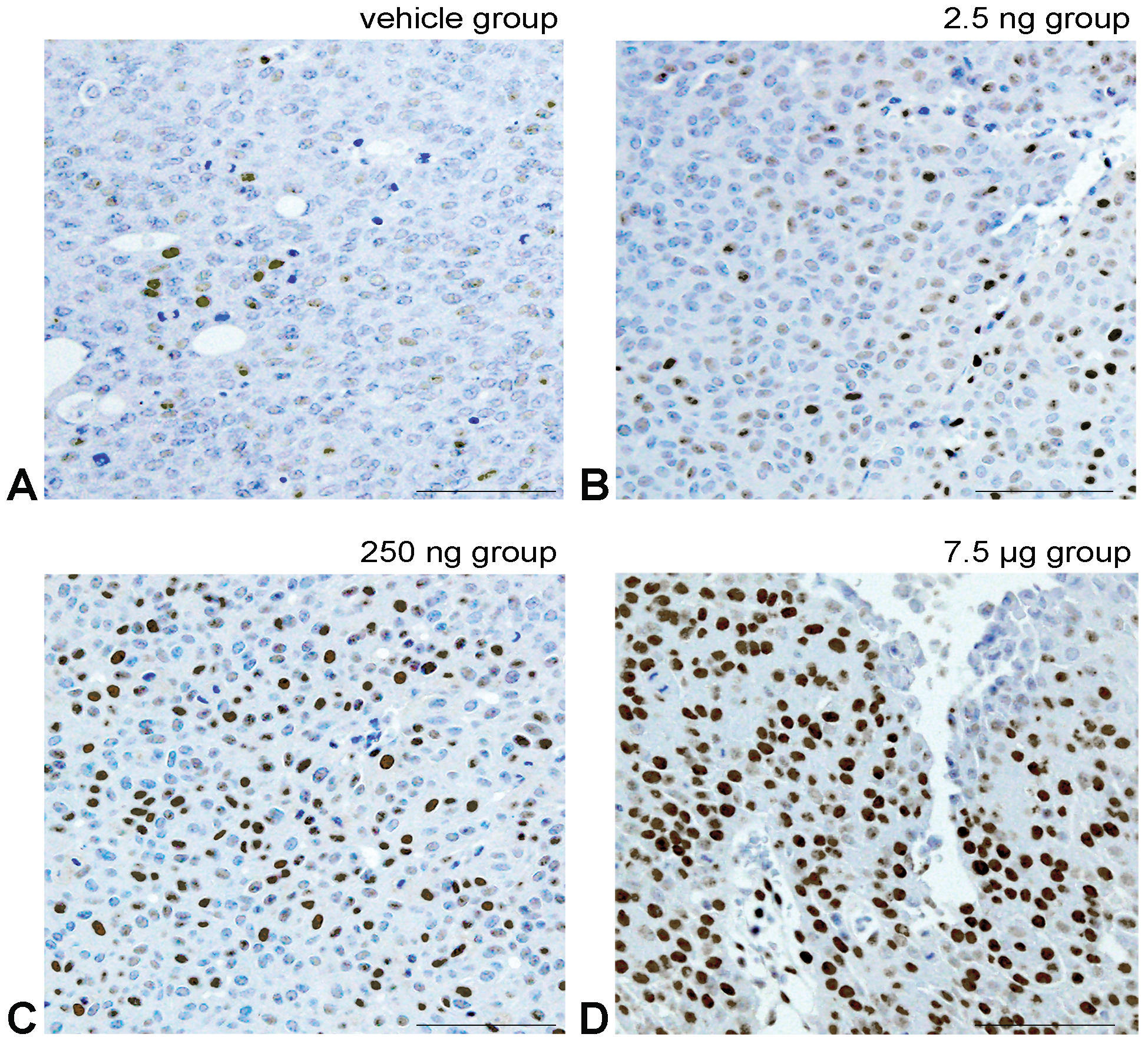
Representative images of the immunohistochemical analysis for PCNA. PCNA positive cell have light brown to black nuclear staining by diaminobenzidine. A, The Vehicle group shows few positive tumor cells. B-D, Show prenatal-treated polychlorinated biphenyl groups with a dose-dependent increase in the number of positive tumor cell nuclei. Bar shows 30 μm.
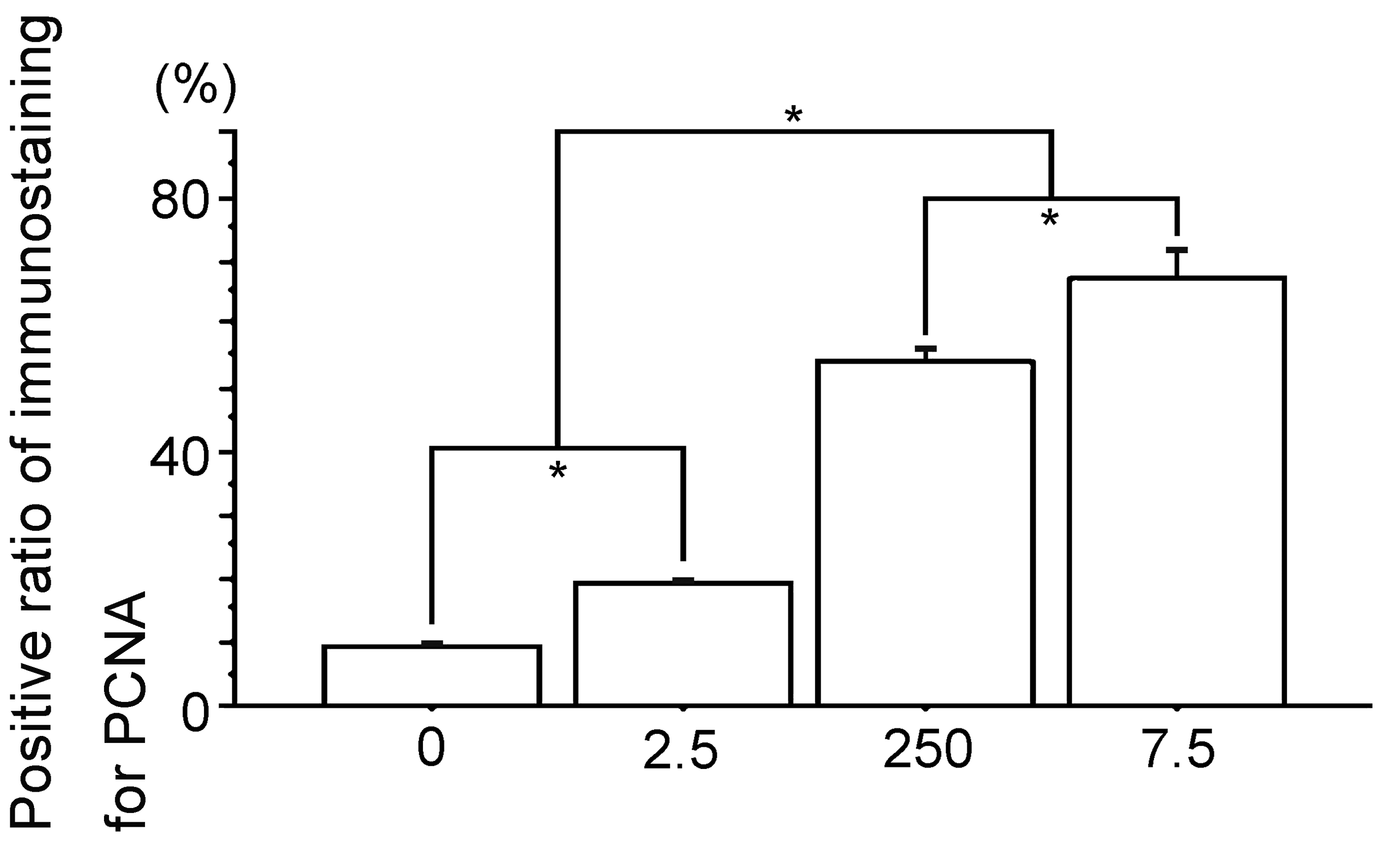
Percent positive cells of immunostaining for PCNA. 0, the Vehicle group; 2.5, the polychlorinated biphenyl (PCB) 2.5 ng group; 250, the PCB 250 ng group; 7.5, the PCB 7.5 µg group. Data are mean ± standard error of the mean. *Scheffé’s F test, P < .01.
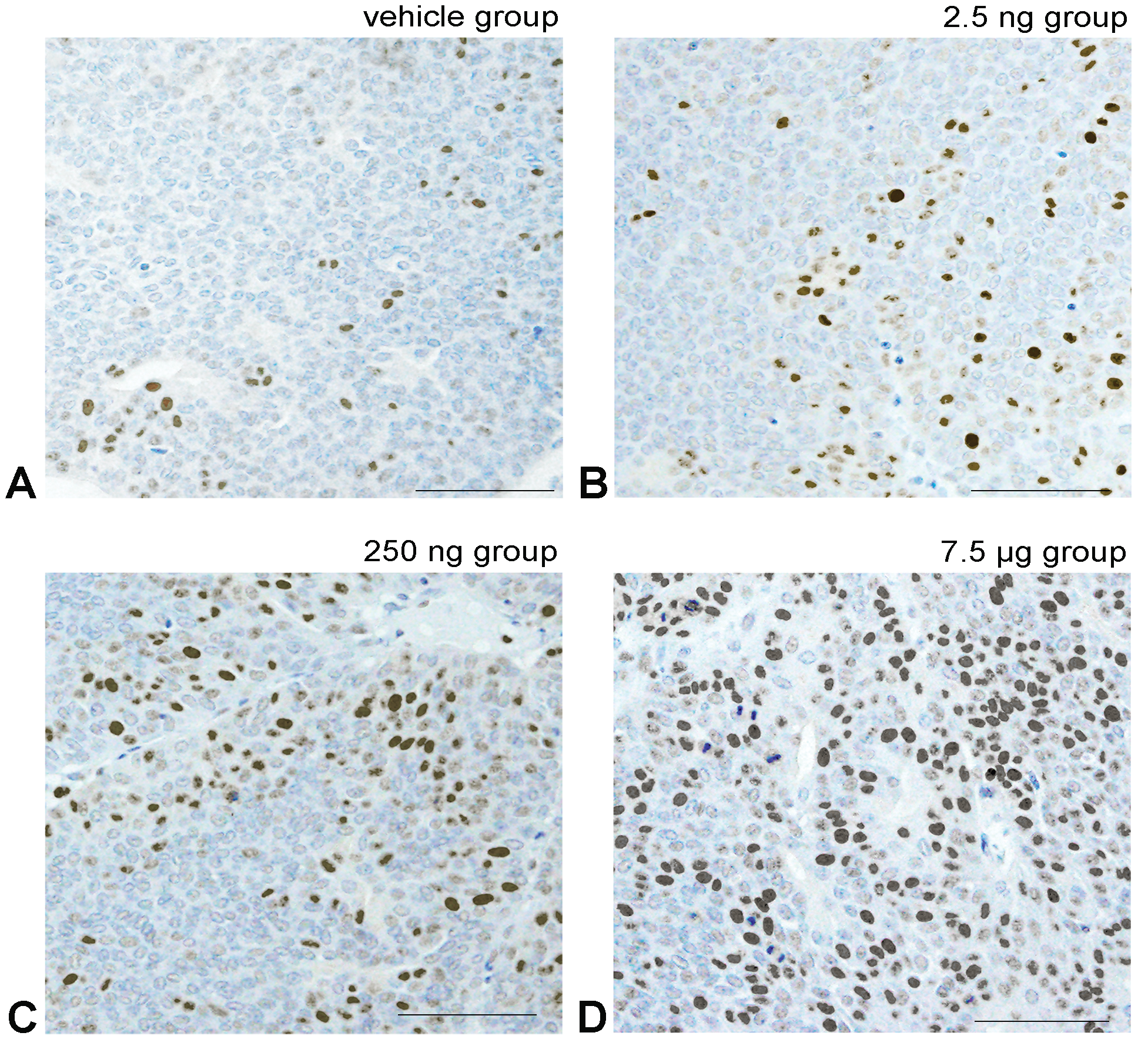
Representative images of the immunohistochemical analysis for cyclin D1. Cyclin D1 positive cell shows light brown to black staining in the cell nucleus due to diaminobenzidine. A, The Vehicle group shows few positive tumor cells. B-D, Show prenatal polychlorinated biphenyl (PCB) groups with a PCB dose-dependent increase in the number of positive tumor cell nuclei. Bar shows 30 μm.
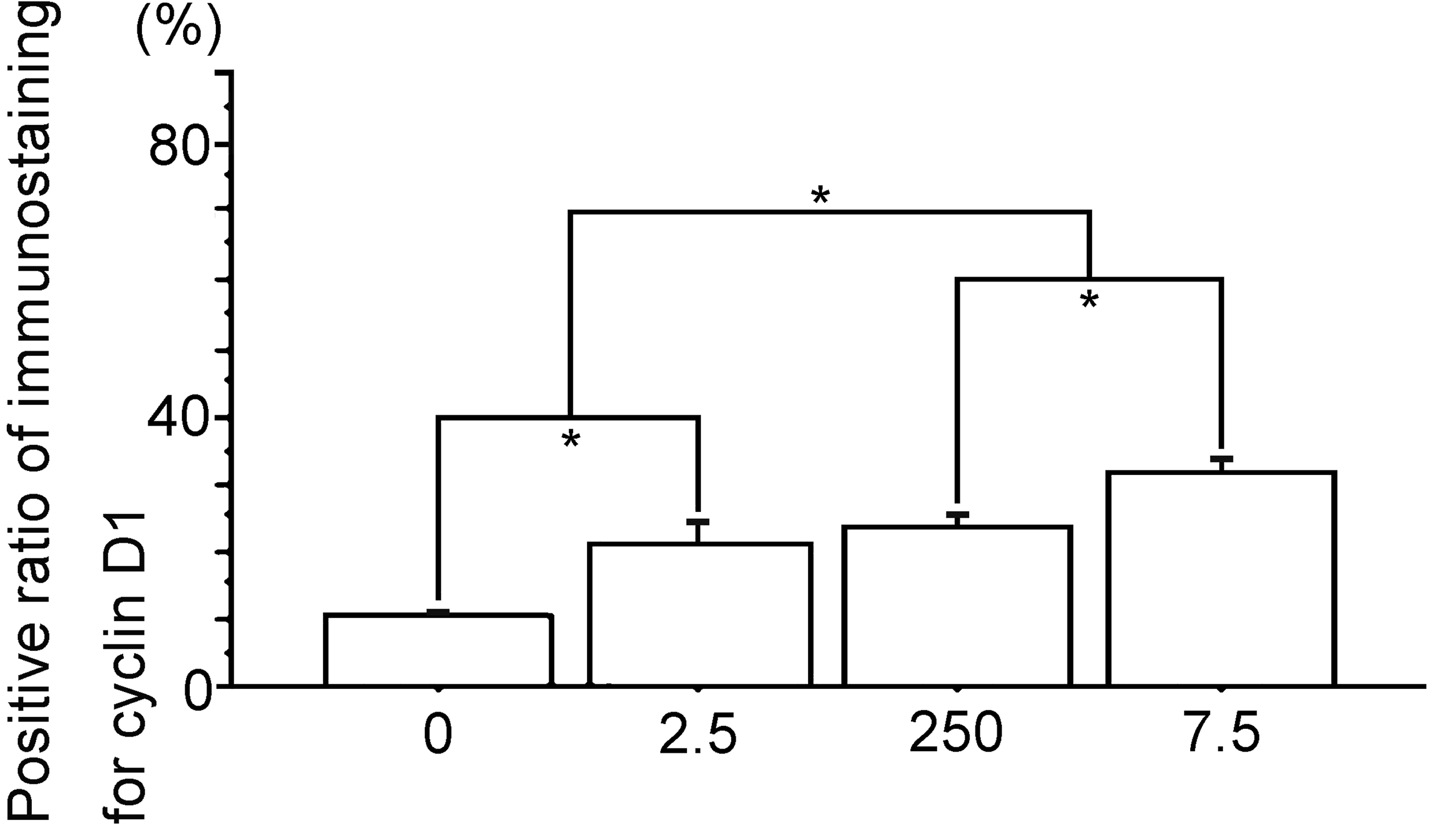
Percent positive cells of immunostaining for cyclin D1. 0, The Vehicle group; 2.5, the polychlorinated biphenyl (PCB) 2.5 ng group; 250, the PCB 250 ng group; 7.5, the PCB 7.5 µg group. Data are mean ± standard error of the mean. *Scheffé’s F test, P < .01.
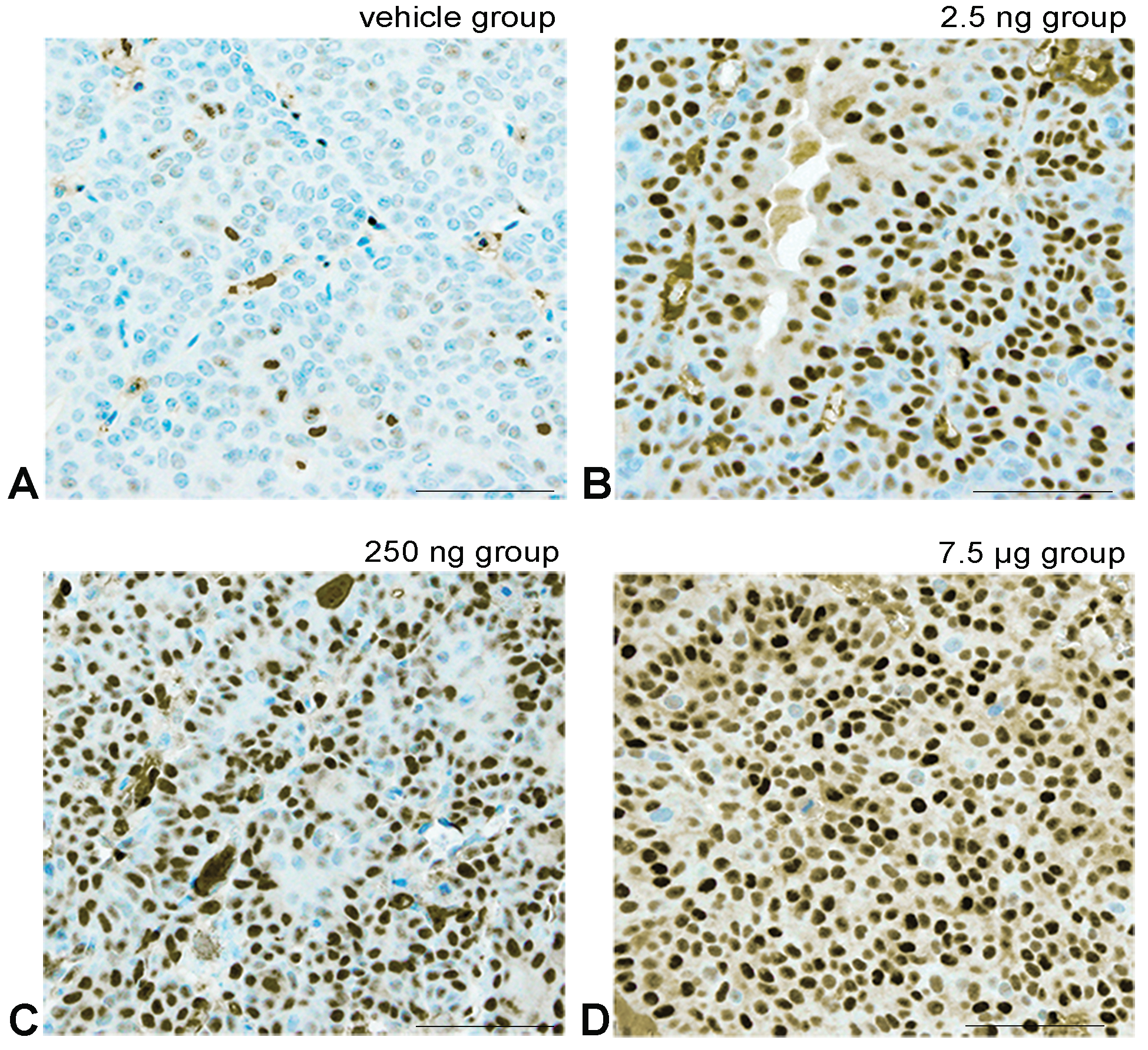
Representative images of the immunohistochemical analysis for ERα. ERα positive cell shows light brown to black staining in the cell nucleus due to diaminobenzidine. A, The Vehicle group shows a few positive tumor cells. B-D, Show prenatal polychlorinated biphenyl (PCB) groups showing a PCB dose-dependent increase in the number of positive tumor cell nuclei. Bar show 30 μm.
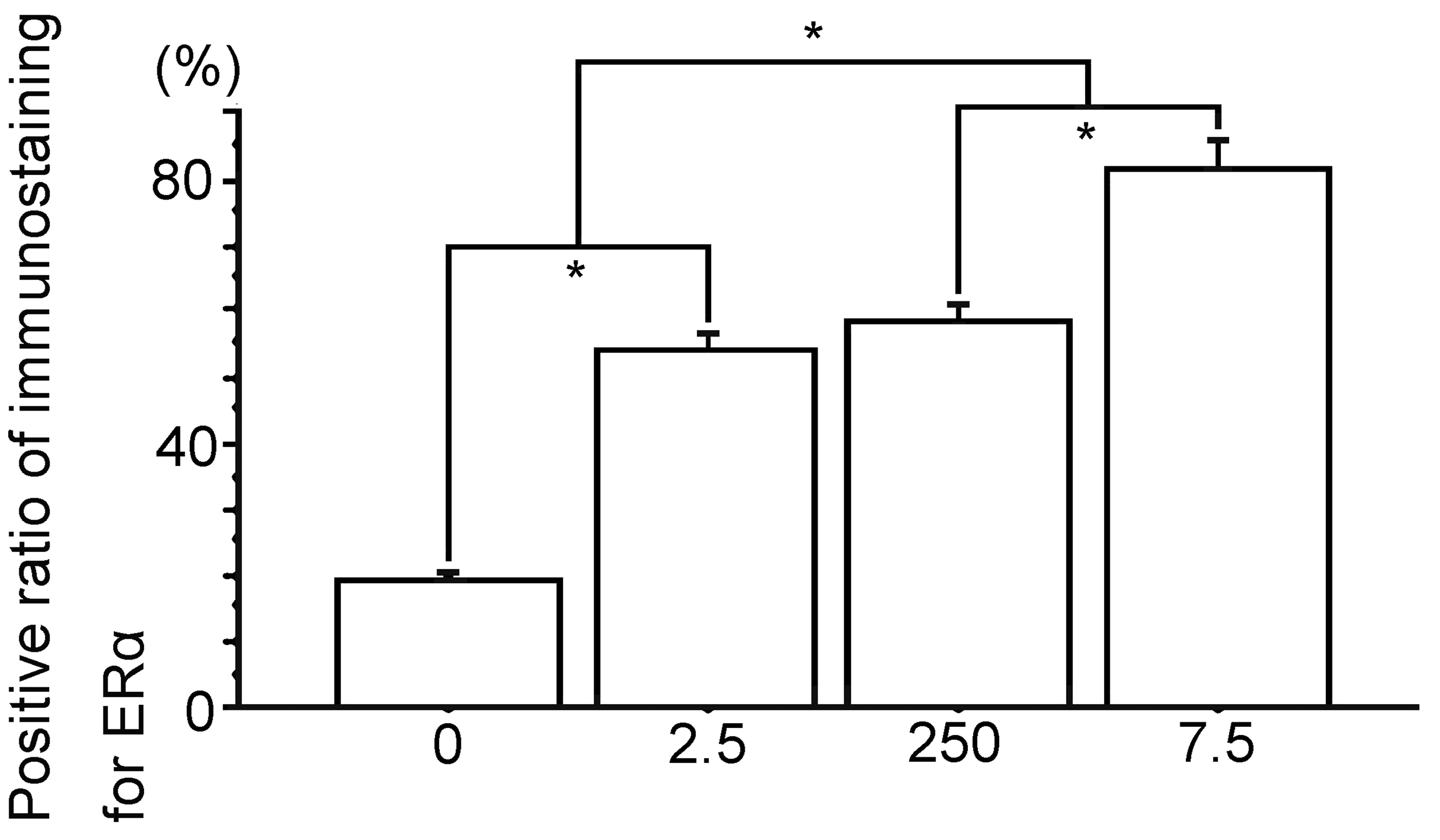
Percent positive cells of immunostaining for ERα. 0, The Vehicle group; 2.5, the polychlorinated biphenyl (PCB) 2.5 ng group; 250, the PCB 250 ng group; 7.5, the PCB 7.5 µg group. Data are mean ± standard error of the mean. *Scheffé’s F test, P < .01.
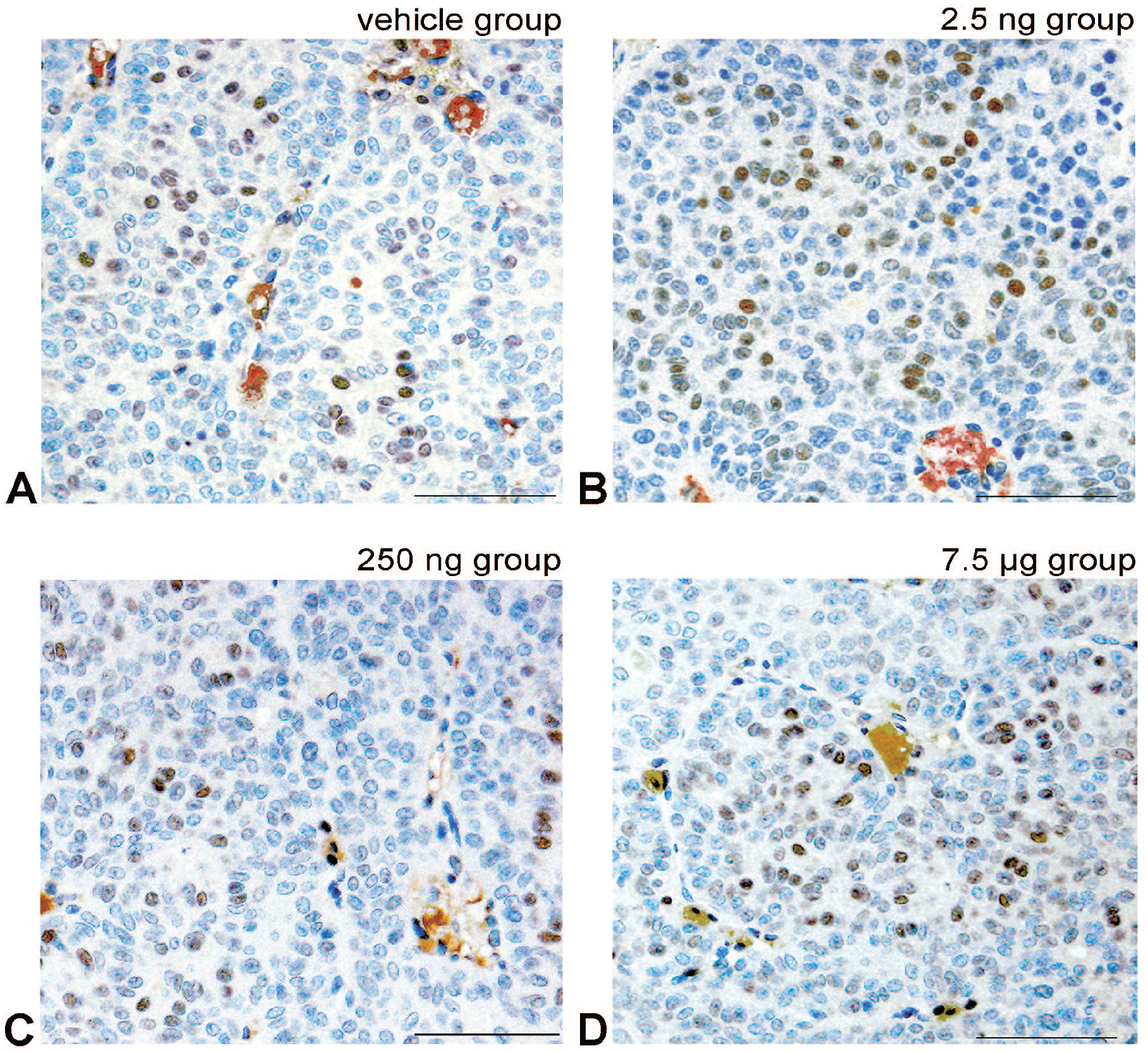
Representative images of the immunohistochemical analysis for ERβ. ERβ positive cell show cell nucleus staining in light brown to black colors by diaminobenzidine. A, The Vehicle group showing a few positive tumor cells. B-D, Images are the prenatal-treated polychlorinated biphenyl (PCB) groups showing a PCB non-dose-dependent number of positive tumor cell nuclei. 0, the Vehicle group; 2.5, the PCB 2.5 ng group; 250, the PCB 250 ng group; 7.5, the PCB 7.5 µg group. Data are mean ± standard error of the mean. All results are nonsignificant. Bar shows 30 μm.
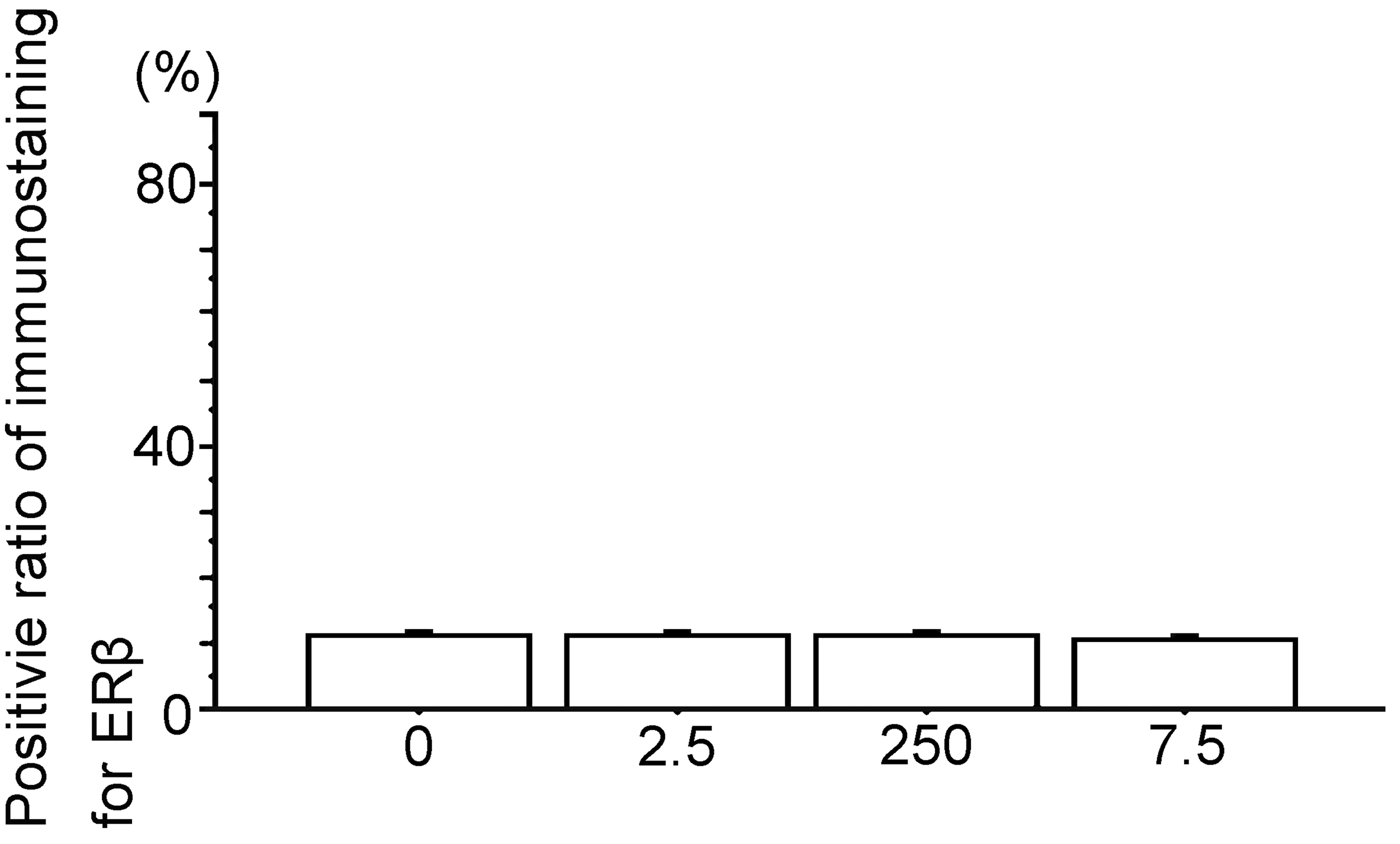
Percent positive cells of immunostaining for ERβ. 0, the Vehicle group; 2.5, the polychlorinated biphenyl (PCB) 2.5 ng group; 250, the PCB 250 ng group; 7.5, the PCB 7.5 µg group. Data are mean ± standard error of the mean. *Scheffé’s F test. All results are nonsignificant.
Immunohistochemical analyses of PCNA and cyclin D1 expression showed them to be nearly similar; bladder carcinoma in the Vehicle group had a few tumor cell nuclei, and the number of low stainable tumor cell nuclei increased with PCB in a dose-response manner (Figures 6 –9). Meanwhile, a few ERα expression stainable cell nuclei were shown in the Vehicle group, and highly stainable tumor cell nuclei increased in number with PCB in a dose-response manner (Figures 10 and 11). For ERβ, on the other hand, immunohistochemistry showed that the numbers of stainable tumor cell nuclei observed were similar between the Vehicle group and all PCB groups (Figures 12 and 13).
Western Blot Analysis in the Bladder Carcinoma
The relative protein expression of PCNA, cyclin D1, ERα, and ERβ in the bladder carcinomas was examined by Western blot analyses. Protein expressions of PCNA and cyclin D1 showed similar patterns; the expressions in each Vehicle group were significantly lower compared to those of the PCB groups, and the expressions in the PCB groups showed significant and pronounced increases with PCB in a dose-response manner. Moreover, ERα protein expression was almost similar to that of PCNA and cyclin D1, while ERα expression levels were higher than those of PCNA and cyclin D1 (Figure 14). ERβ protein expression in the Vehicle group was low and similar to that in the PCB groups, and that of all PCB groups was low and of a similar level (Figure 14).
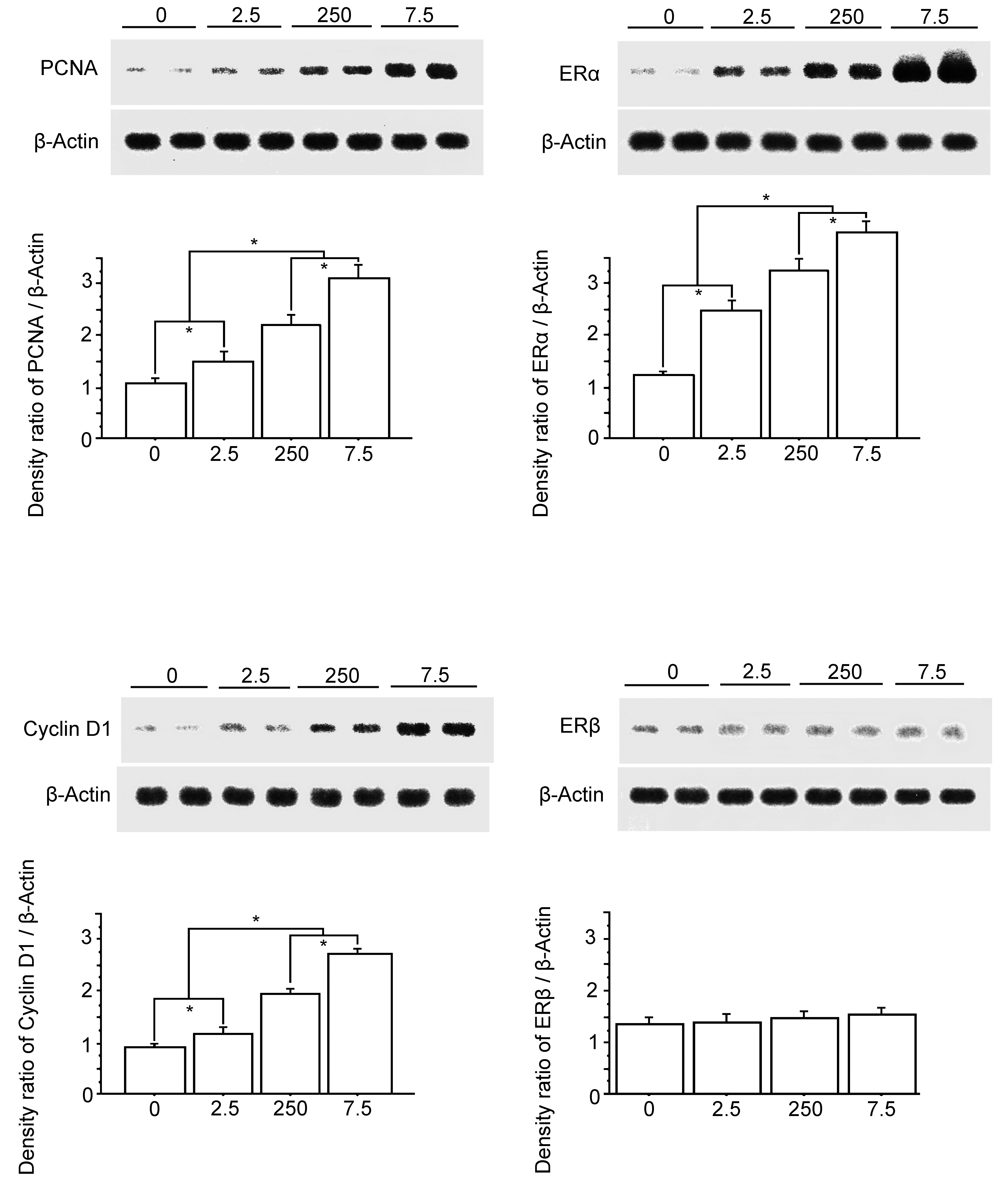
Representative western blot results are shown at the top. Bottom shows the density ratio of each protein expression band/β actin expression band of the membranes, detected using the ECL Plus Western Blotting Detection System. 0, the Vehicle group; 2.5, the polychlorinated biphenyl (PCB) 2.5 ng group; 250, the PCB 250 ng group; 7.5, the PCB 7.5 µg group. Data are mean ± standard error of the mean. *Scheffé’s F test, P < .01.
Quantitative RT-PCR in Bladder Carcinoma
The expression patterns of PCNA, cyclin D1, ERα, ERβ, CYP1A1, CYP1B1, and AhR mRNA expression in the bladder carcinomas were analyzed by real-time quantitative RT-PCR. The mRNA expression patterns of PCNA and cyclin D1 were almost similar, while the expression levels of ERα were higher than those of PCNA and cyclin D1. This may be due to PCB exposure. Moreover, the mRNA expression of ERα in the Vehicle group showed significantly low expression compared to that in the PCB groups, and the PCB group significantly increased following PCB in a dose-response manner. The expression patterns of ERβ were low and similar to those of the Vehicle group and all PCB groups (Figures 4 and 15). The mRNA expression patterns of CYP1A1, CYP1B1, and AhR were similar. The Vehicle group showed significantly low expression, which gradually increased following the PCB 2.5 ng and 250 ng groups. No significant difference, however, was revealed between Vehicle, PCB 2.5 ng, and 250 ng groups. Moreover, the PCB 7.5 µg group was significantly higher than other groups (Figures 4 and 15).
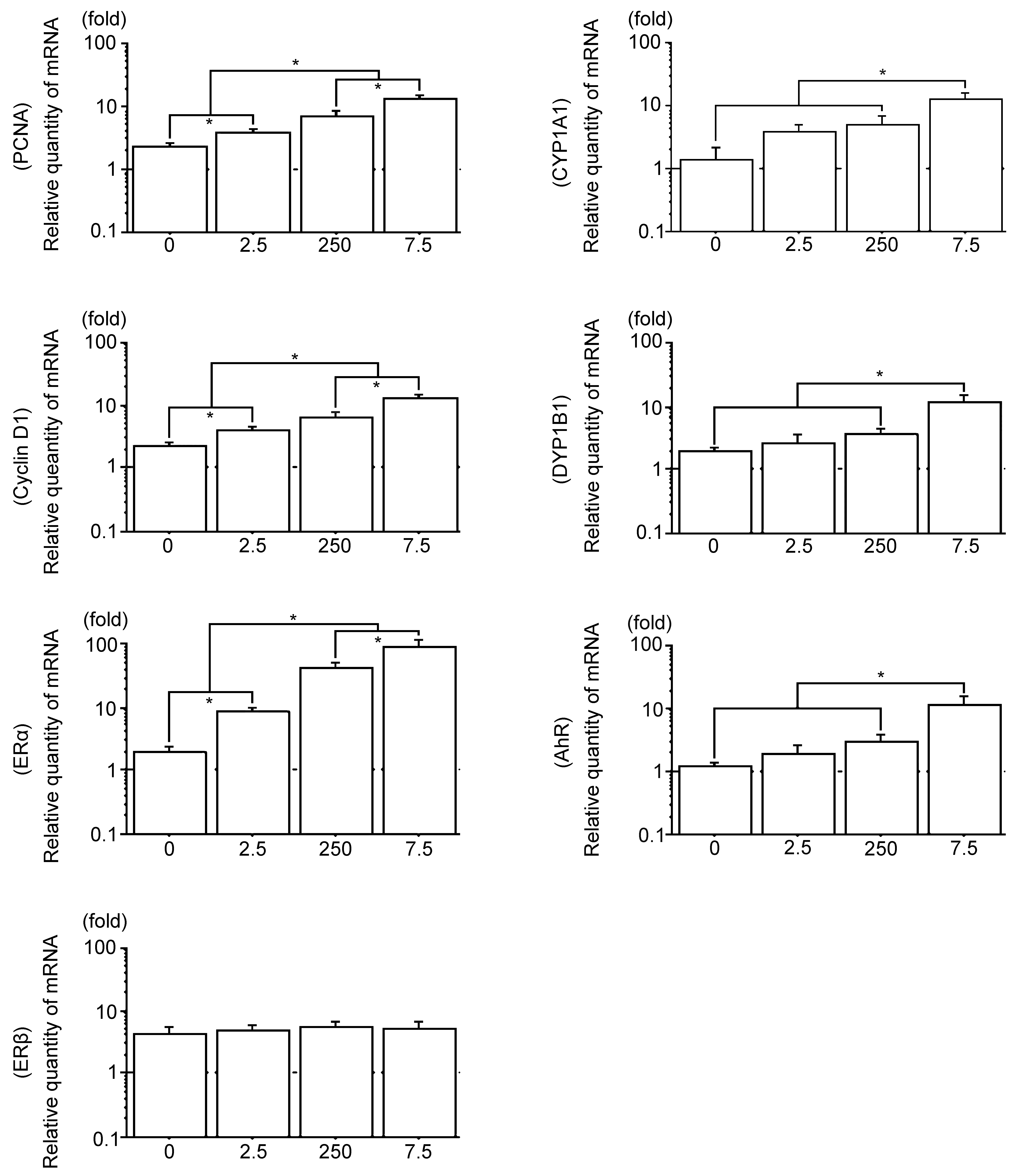
Real-time quantitative RT-PCR analysis of PCNA mRNA, cyclin D1 mRNA, ERα mRNA, ERβ mRNA, CYP1A1 mRNA, and CYP1B1 mRNA in N-butyl-4-(hydroxy butyl) (BBN) rat bladder carcinomas. The blots used to detect the above genes were subsequently stripped and rehybridized with GAPDH to serve as internal controls. The radioactive bands were quantified and normalized with those derived from GAPDH mRNA. 0, the Vehicle group; 2.5, the polychlorinated biphenyl (PCB) 2.5 ng group; 250, the PCB 250 ng group; 7.5, the PCB 7.5 µg group. Data are mean ± standard error of the mean. *Scheffé’s F test, P < .01.
Discussion
Polychlorinated biphenyls are ubiquitous environmental contaminants that produce a spectrum of adverse biochemical and biological effects including liver carcinoma induction in people and a wide variety of animals. 26 However, PCBs are considered nongenotoxic carcinogens that do not produce DNA adducts and are negative for genotoxic tests. 43 PCB126 binds and activates the aryl hydrocarbon receptor (AhR), which is a basic helix-loop-helix (b-HLH) protein. 44 Ligand binding results activation of AhR and subsequent nuclear translocation, where it heterodimerizes with another bHLH partner, the AhR nuclear translocator protein (ARNT). 45 The AhR-ARNT dimer binds to specific regulatory elements and xenobiotic responsive elements upstream of the responsive genes, and enhances their transcripts, the cytochrome P4501A1 (CYP1A1) enzyme family. 46 –51
N-butyl-4-(hydroxybutyl) is a procarcinogen and requires metabolic conversion through the α-hydroxylation pathway by CYP1A1 to its ultimate carcinogen metabolite BP-7,8-diol-9,10-epoxide, and binds to DNA and progresses into the initiation-promotion stages of bladder carcinogenesis. 1,52,53
Moreover, several studies have described PCB126 as being anti-estrogenic via crosstalk between the AhR and ERs signaling pathway in human and rodents. 31,54 –56 Bladder carcinogenesis has been considered to be enhanced in anti-estrogenic situations. 2,3 We therefore hypothesized that postnatal PCB126 exposure directly enhances BBN bladder carcinogenesis. However, in the 16-week-old rats, the end point in the present study, prenatally exposed PCB126 residue level in bladder carcinoma was low and not statistically different to that of the Vehicle group, and thus the low level PCB126 residue due to a direct progress effect for the BBN bladder carcinogenesis is unlikely. However, surprisingly, the present study revealed that prenatal PCB126 exposure dose-dependently increased BBN bladder carcinomas with PCB126 dose-dependent increases in PCNA, Cyclic D1, CYP1A1, CYP1B1, ERα, and AhR mRNA expressions, and especially highly expressed ERα mRNA. We suggest that prenatal exposure to PCB126 affects the perinatal stage of rats to obtain the biological-biochemical characters, upregulation for the progression and proliferation of BBN bladder carcinogenesis.
The ER subtype, which is now called ERα, was first reported in 1958, 57 whereas ERβ in rats 58 and humans 59 was cloned in 1996 and 1997, respectively. ERα and ERβ are physiologically expressed in human organs and, on binding with estrogen such as 17β-estradiol, possess various actions in various tissues by different pathways. 60 Several previous studies have shown that ERα is a carcinoma promotion factor, 4,7,61,62 while ERβ exerts an inhibitory effect on tumor cell proliferation. 61,62
Many studies have observed that ERβ is expressed at high levels in human bladder carcinoma. Most of these studies support the emerging consensus that ERβ is the dominant ER present in human bladder epithelium and cancer, 4,14,17,18,63 –66 and it has been reported that ERα and ERβ play suppressive roles in BBN mice bladder carcinoma. 1,16 However, in this study, the BBN bladder carcinoma Vehicle group showed high ERα levels and low levels of ERβ. There are various immunohistochemical studies describing the expression of ERα and ERβ proteins in bladder carcinoma that have produced inconsistent results. 1,14,18 We could not elucidate a mechanism for these conflicting conclusions at the time of our study. Therefore, a further detailed study is necessary.
Cyclin D1 is an important target gene through which ERα appears to play a key role in estrogen-provoked cellular proliferation. 67 We found high expression levels of cyclin D1 in the PCB group bladder carcinomas treated with PCB126 that were dose-dependent, as compared to the Vehicle group. While PCBs have been suggested in animal experiments to inhibit estrogen-induced responses from antiestrogenic activity via crosstalk of the AhR and ER signaling pathways. 31,54,56 Nevertheless, it would not be unreasonable to assume that prenatal exposure to PCB126 occurs through different effects and/or metabolic pathway by different kinds of carcinogenic chemical treatments. Previously, we reported that prenatal exposure to high level PCB126 decreases the incidence of DMBA-induced rat mammary carcinoma. 33 These carcinomas were estrogen-dependent and showed high expression of Ahr and CYP1B1 mRNA, 20,33 while antiestrogenic bladder carcinomas in the PCB group in the present study show high expression of ERα.
In summary, the present study suggests that prenatal PCB126 exposure induced PCB126 dose-dependent BBN rat bladder carcinogenesis with upregulation of ERα and low ERβ expression. Abundant cross-group expression of ERα mRNA in bladder carcinoma was similar by immunohistochemical analysis. Our results also suggest that prenatal PCB126 exposure dose-dependently metabolizes BBN via CYP1A1, and by inducing high ERα, also contributes to estrogen induced G1/S phase progression and cell proliferation in rat bladder carcinogenesis. Moreover, upregulated ERα may induce PCNA and cyclin D1 expression, resulting in dysregulated cell-proliferation in bladder carcinogenesis, although ERβ expression played no part in this cascade.
However, further investigations are warranted to determine whether the levels of PCB126 given during prenatal periods in this study will induce the changes determined in the present animal investigations in humans.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_01926233211064180 - In Utero Exposure to 3,3′,4,4′, 5-Pentachlorobiphenyl Dose-Dependently Induces N-butyl-4-(hydroxybutyl) Nitrosamine in Rats With Urinary Bladder Carcinoma
Supplemental Material, sj-docx-1-tpx-10.1177_01926233211064180 for In Utero Exposure to 3,3′,4,4′, 5-Pentachlorobiphenyl Dose-Dependently Induces N-butyl-4-(hydroxybutyl) Nitrosamine in Rats With Urinary Bladder Carcinoma by Shin Wakui, Hiroyuki Takahashi and Tomoko Muto in Toxicologic Pathology
Footnotes
Acknowledgments
The authors thank webshop_support@elsevier.com for editing a draft of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Grant-in-Aid for High-Tech Research Center Project from the Ministry of Education, Science and Culture, Japan.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
