Abstract
Baclofen is a γ-aminobutyric acid-B receptor agonist used for control of spastic muscle activity and as a treatment for alcohol abuse. The review of the nonclinical database suggested a data gap for potential carcinogenicity following long-term use. Regulatory requirements for pharmaceutical safety testing of cancer-causing potential have historically included 2-year rodent studies in rats and mice. The availability of transgenic models with greater specificity and sensitivity to carcinogens provides safety testing alternatives that align with the 3Rs. The carcinogenicity of baclofen was evaluated in CB6F1-TgrasH2 transgenic mice following daily oral administration at 45, 90, and 180 mg/kg/d for 26 weeks, preceded by a 2-week drug-conditioning period. There were no treatment-related palpable masses or neoplastic findings, and survival rates were not affected by the baclofen treatment. In conclusion, baclofen was considered as noncarcinogenic in CB6F1-TgrasH2 mice, which is consistent with results previously obtained in a 2-year rat study.
Baclofen (γ-amino-β-[p-chlorophenyl]-butyric acid) is a centrally acting selective γ-aminobutyric acid-B (GABA-B) receptor agonist, and has emerged as a promising drug for the treatment of alcohol dependence. 1,2 Baclofen was originally approved by regulatory agencies in the early 1970s for therapeutic use in muscle spasticity associated with neurologic conditions. A previously conducted 2-year carcinogenicity study of orally administered baclofen in rats found no evidence of carcinogenesis up to the high dose of 100 mg/kg/d. Findings were limited to an apparent dose-related increase in the incidence of ovarian cysts and enlarged and/or hemorrhagic adrenals at 50 and 100 mg/kg in female rats (taken from publicly available baclofen product information documents: product labels, regulatory reviews, and monographs).
Although the safety of baclofen is well characterized, 3 gaps in existing nonclinical data were identified and additional carcinogenicity safety data were considered necessary to support the long-term therapeutic use of baclofen in alcohol-related disorders.
Over the last 10 years, a growing consensus has developed around the scientific merits and regulatory acceptability of replacing one of the required 2-year rodent carcinogenicity studies with a 6 month transgenic model study for assessment of carcinogenic potential of pharmaceuticals. A new standard was set for carcinogenicity testing with the ICH S1B (1997) guideline, which revised the testing requirement to include a 2-year carcinogenicity study in rodents and an optional shorter term study in an alternative model such as transgenic (Tg) rasH2 mice. The TgrasH2 mouse model was developed as an alternative to the 2-year mouse bioassay for prediction of carcinogenic potential of pharmaceuticals. 4,5 It is generally accepted that the TgrasH2 model has been adequately evaluated for consideration in carcinogenicity testing of pharmaceutical candidates and that a 6-month TgrasH2 mouse study can replace a 2-year mouse carcinogenicity study in support of pharmaceutical registration 5,6 as the TgrasH2 model can predict known or suspected human carcinogens just as well as the traditional mouse bioassay. 7 –9
The advantages of the TgrasH2 model include a low incidence of spontaneous tumors and responsiveness to genotoxic and nongenotoxic carcinogens. This model is considered to predict neoplastic findings that are relevant to human cancers 9 and is not prone to false-positives or rodent-specific neoplasms. 7 The TgrasH2 model conforms to the 3Rs (reduction, replacement, refinement) principles, as it enables the experimental group size to be reduced to ∼25/sex, and euthanasia occurs long before the senescence-related morbidity that occurs in 2-year studies, thus constituting a refinement with added biological relevance.
The aim of the study was to supplement the available data for evaluating the carcinogenic potential of baclofen using the TgrasH2 mouse model. The test item was baclofen (batch number: M1002831, CAS number: 1134-47-0) administered as a suspension in the vehicle (0.5% methylcellulose aqueous solution) under a dosage volume of 10 mL/kg. The positive control was N-methyl-N-nitrosurea—Isopac (MNU) (batch number: MKBQ7120 V, CAS number: 684-93-5), a known carcinogen, at 75 mg/kg intraperitoneal injection in citrate-buffered saline (dosage volume 10 mL/kg). Due to the physicochemical properties of MNU, special care was taken to ensure minimal dispersion into the environment and staff exposure. An important element in the design of this study was the choice of dose levels for a 15-day conditioning period to baclofen. This was needed, similar to dose titration required at initiation of baclofen treatment in humans, to avoid adverse clinical effects from baclofen and in order to permit administration at higher dose levels during the main 26-week treatment period (Table 1).
Summary of Study Design.
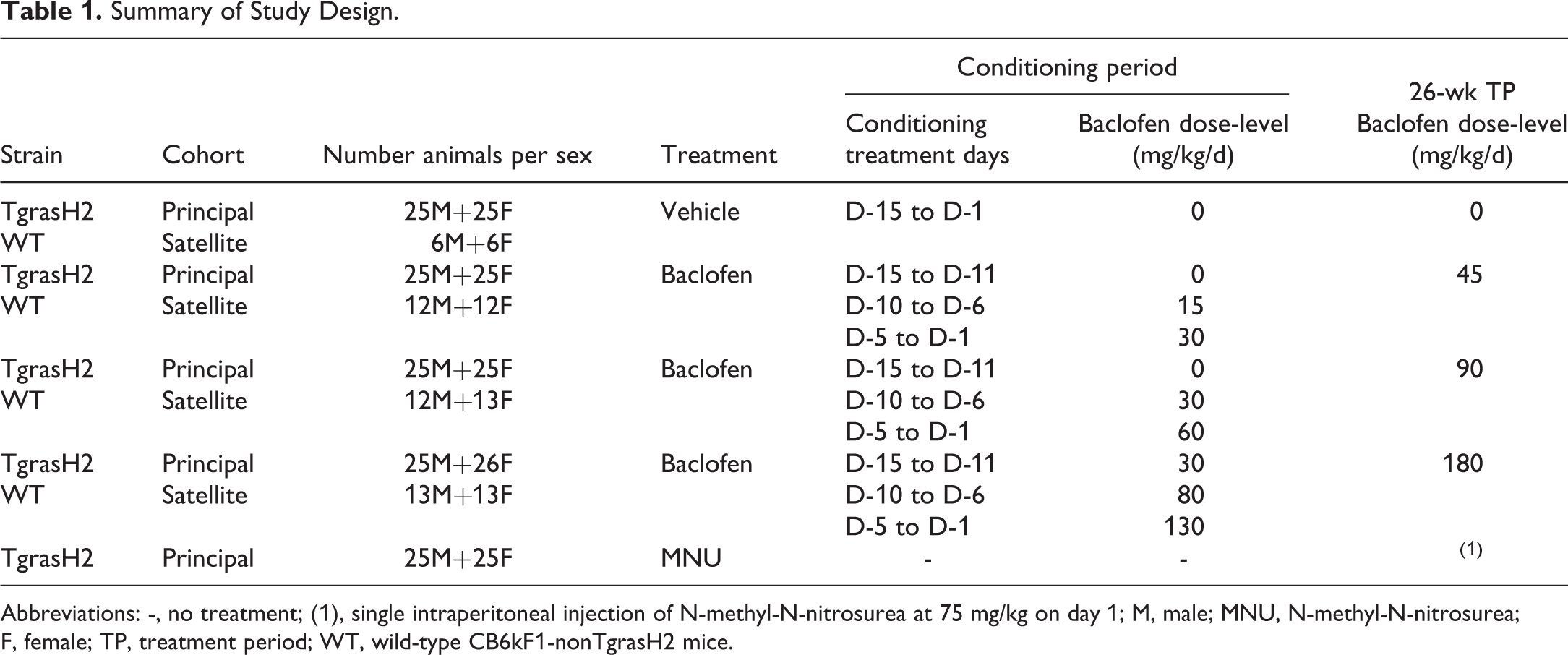
Abbreviations: -, no treatment; (1), single intraperitoneal injection of N-methyl-N-nitrosurea at 75 mg/kg on day 1; M, male; MNU, N-methyl-N-nitrosurea; F, female; TP, treatment period; WT, wild-type CB6kF1-nonTgrasH2 mice.
Wild-type nontransgenic CB6F1-nonTgrasH2 (WT) mice (for toxicokinetics) and transgenic CB6F1-TgrasH2 (TgrasH2) mice were supplied by Taconic Farms Inc (7 weeks old on arrival). They were housed in a barrier-protected rodent unit. Each cage contained elements for environmental enrichment. Humane animal care measures were followed in accordance with EU Directive 2010/63 (on the protection of animals used for scientific purposes), and the animal laboratory facility ethics committee reviewed the study plan before the start of the study.
The health status of each animal was checked daily. All principal and positive control animals were palpated from week 5 to detect the time of onset, location, size, appearance, and progression of any palpable masses. Body weight and food consumption were recorded regularly. Plasma baclofen concentrations were determined using a validated LC-MS/MS method. Surviving principal and positive control mice were killed at the end of the treatment period. A macroscopic postmortem examination was performed, designated organs were weighed, and a microscopic examination was performed. WHO/IARC/RITA criteria 10 were used for the nomenclature and classification of tumors. Surviving satellite animals were killed after blood sampling in week 4 without further examination. Prematurely killed and found dead animals were macroscopically examined.
Statistical analysis was performed using PathData software, version 6.2d2. Survival probability functions were estimated by the Kaplan-Meier technique. Survival curves were compared by the log-rank procedure, according to Peto’s method. 11 Tumor incidence analysis was performed using SAS Enterprise Guide software, separately for each sex and tumor type, by comparison between vehicle and positive control-treated groups using a Fisher exact test, and by comparison between vehicle and baclofen-treated groups using a one tailed Fisher exact test and a Cochran-Armitage trend test. Interpretation criteria were based on the ILSI ACT program. 12
The study was designed to comply with the note for guidance on carcinogenic potential (CPMP/SWP/2877/00, 2002), ICH Guideline S1B on testing for carcinogenicity of pharmaceuticals (CPMP/ICH/299/95, 1997) and recommendations on the use of genetically modified animal models for carcinogenicity assessment (CPMP/SWP/2592/02 Rev1, 2004).
Mortality during the study (Supplemental Table 1) was similar in the principal vehicle control and baclofen-treated groups. Mortality rates were in agreement with the literature 13 –15 and in-house reference values. A high mortality rate and clinical signs of poor health were observed in positive control animals (main cause of death: malignant lymphoma in the thymus or tumors in the forestomach, consistent with known, MNU-induced effects 14 ). Survival rates at study termination (Supplemental Figure 1) were similar in the control and baclofen groups. There were no clinical signs at 0/15/30, 0/30/60, 45, or 90 mg/kg/d. At 30/80/130 and 180 mg/kg/d, notable treatment-related signs (hypoactivity, staggering gait, dyspnea, abdominal breathing, hunched posture, and ventral recumbency) appeared transiently and were largely limited to the first 2 weeks of dosing. Based on the nature of the clinical signs, it was concluded that the maximum tolerated dose (MTD) had been reached. In surviving positive control animals, clinical signs were associated with poor health.
No palpable masses (Supplemental Table 2) were detected in baclofen-treated females or vehicle control groups. Palpable masses were detected in the testis in 4 baclofen-treated males (1 high dose from week 24 and 3 low dose from week 14). Histological correlates were observed in 3 of these individuals at study termination, and in each case the finding was unilateral. In the high-dose male, the enlarged mass (left) presented with red discoloration was nonmalignant and correlated with severe testicular atrophy/degeneration. In 1 low-dose male, an enlarged cyst (left) with red discoloration was observed and correlated with testicular hemangioma and moderate testicular atrophy/degeneration. Tubular atrophy/degeneration in the testes is a common finding in rodents, with reported spontaneous occurrence of 28% in TgrasH2 mice. 15 It may be associated with edema, especially in high severity cases due to an associated inflammatory process and subsequent filling of the testicular cavity with liquid. These observations were consistent with other reports and with the time of onset of the observed palpable masses. In the other low-dose male, enlarged cyst (left) with red discoloration correlated with testicular hemangiosarcoma, which has been reported as an uncommon tumor seen grossly, among others of this type accounting for up to 28% of all background tumors in males of this strain. 16 Due to the very-low incidence, previous reporting of these findings in this mouse strain, lack of dose-relationship and nonmalignant mass found in the high-dose baclofen male, a relationship to baclofen could not be established. Palpable masses were also found in 3 male and 4 female positive control animals (time of onset and incidence consistent with reference data 14 ).
Some statistically significant weight losses were observed in individual mid- and high-dose males and females (Supplemental Figure 2), but overall body weight gains in the baclofen groups were similar to those of vehicle controls. Food consumption (Supplemental Figure 3) during the conditioning period was dose-dependently lower in baclofen groups. During the treatment period, food consumption values were similar to controls from Week 2 or 3. These results indicated that the animals became conditioned to the baclofen treatment.
All plasma samples from vehicle control animals were below the limit of quantification (<5.00 ng/mL) for baclofen. On days 1 and 25, baclofen was quantifiable from 0.25 to 24 hours in plasma samples from baclofen-treated animals at all dose-levels (Supplemental Figure 4 and Supplemental Table 3).
Among the organ weights (Supplemental Table 4), group mean absolute and relative spleen weights were statistically significantly lower in males and females at 180 mg/kg/d, but in the absence of microscopic correlates, a treatment relationship was considered equivocal. No other organ weight differences were considered as baclofen-related, because there were no microscopic correlates and the differences were of low magnitude and/or poorly dose-related. A very-low incidence of small thymus was observed (1/25 males at 45 and 90 mg/kg/d and 1 female per baclofen-treated group). As there was no dose-related trend, a test item relationship could not be established. All other observations were considered as common spontaneous findings were not dose-related and/or were also seen in vehicle controls.
Most of the mice treated with MNU had lymphoma, enlarged spleen, liver, thymus and lymph nodes, and/or a forestomach tumor correlating with a white mass or thickening, which is consistent with MNU treatment-related neoplastic lesions in TgrasH2 transgenic mice. 14 The incidence of forestomach squamous cell papilloma (20/25 males and 18/25 females) was similar to that observed in TgrasH2 mice receiving MNU in other published studies (incidence: 76.0% in males [range: 53.3%-86.7%] and 76.2% in females [range: 53.3%-100%] 14 ). Malignant lymphoma was observed in the thymus, stomach, forestomach, skin, and Harderian glands of 11/25 males and 17/25 females (vs an incidence of ∼91% in both sexes in the literature 14 ). The diagnosis was established according to the massive lymphoid infiltration, rather than at the primary affected organ. Forestomach squamous cell carcinoma and Harderian gland adenoma were also observed with similar incidences to the literature. 14 Malignant thymoma in the thymus was observed in 4% of males. This positive response confirmed the sensitivity to tumor induction of the transgenic mice used in this study.
Minimally increased severity of lymphoid atrophy in the thymus was observed in male mice treated at 180 mg/kg/d and in all baclofen-treated female mice (considered as treatment-related, but nonadverse as it was low in magnitude and severity, poorly dose-related, found in controls with severities up to marked, and associated with no systemic deleterious effects 17 ). Increased severity and incidence of vaginal mucification was observed in control and 180 mg/kg/d baclofen-treated females (Supplemental Table 5). These findings were of low magnitude and severity and considered to be stress related as discussed elsewhere. 18 All other nonneoplastic microscopic findings were consistent with normal spontaneous background findings, distributed randomly among the groups, observed with a very-low incidence and severity, and/or similar to changes found in vehicle controls.
The incidence of primary tumors observed in baclofen-treated and vehicle control groups are presented in Table 2. The overall findings were consistent with previously reported spontaneous occurrences of the observed tumor types found in historical control data of this strain. 5,14 –16,19 The incidence of forestomach squamous cell papilloma, which is among the most common tumors in this strain, minimally exceeded reported spontaneous occurrence (group ranges in males up to 4%-6.7% 14,19 and 3% incidence in males 15 ) in high-dose baclofen-treated males (2/25 males [8%] at 180 mg/kg/d) and was statistically significantly different from the vehicle control by a Cochran-Armitage trend test (P ≤ .05). However, there were no predominant or differentiating preneoplastic changes such as dose-related squamous cell hyperplasia to support a carcinogenic mechanism of induction, and minimal or slight squamous cell hyperplasia in the forestomach was observed in 1 control male, 2 control females, and 4 baclofen-treated animals (1/25 males at 45 mg/kg/d, 1/25 females at 90 mg/kg/d, and 1/25 males and 1/26 females at 180 mg/kg/d). As the incidence was low and there was no dose–response relationship for this tumor type, known for its predisposition in TgrasH2 mice, a relationship to baclofen treatment could not be established.
Hemangiomas or hemangiosarcomas were observed mutually exclusively in baclofen-treated and vehicle control groups, and they occurred sporadically across 6 organs with the majority found in the spleen (Table 2). The incidences of these 2 vascular tumors were statistically analyzed separately by sex and by organ (and across organs, as total incidence of hemangiomas and total incidence of hemangiosarcomas). A slightly higher incidence (2/25-26 or 8%) of splenic hemangioma or splenic hemangiosarcoma occurred in baclofen-treated females at 180 mg/kg/d and baclofen-treated males at 45 or 90 mg/kg/d, respectively, compared to other baclofen-treated groups or vehicle controls, but the incidence remained within published reference data ranges in control TgrasH2 mice (0%-20% in males and 0%-13.3% in females 14 ; 0%-8% in males and 0%-17% in females 19 ; 8% in males and 7% in females 15 ). Hemangiosarcoma in the spleen is reported as the second most common spontaneous neoplastic lesion in TgrasH2 mice, 20 and historical control data in this strain demonstrate a predisposition to vascular tumors in the spleen and other organs with spontaneous incidences similar to those observed in this study. 14,15,19 –21 Due to a lack of dose relationship or statistically significant differences when comparing the incidence of hemangioma or hemangiosarcoma in baclofen-treated groups with vehicle controls either by organ or combined across organs, it was considered that a relationship to baclofen treatment could not be established.
Incidence of Primary Neoplastic Microscopic Findings in TgrasH2 Mice in the 26-Week Baclofen Carcinogenicity Study.a
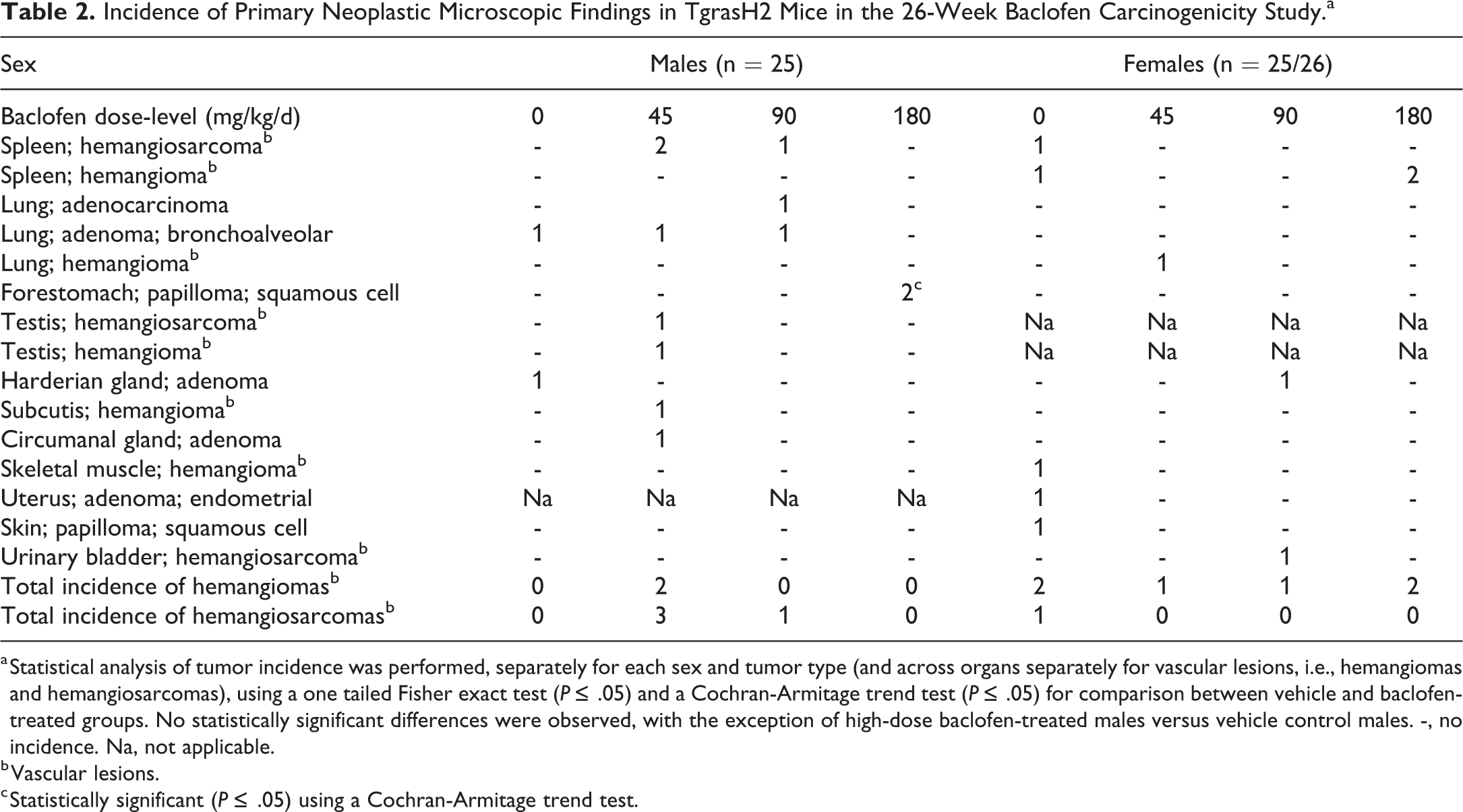
a Statistical analysis of tumor incidence was performed, separately for each sex and tumor type (and across organs separately for vascular lesions, i.e., hemangiomas and hemangiosarcomas), using a one tailed Fisher exact test (P ≤ .05) and a Cochran-Armitage trend test (P ≤ .05) for comparison between vehicle and baclofen-treated groups. No statistically significant differences were observed, with the exception of high-dose baclofen-treated males versus vehicle control males. -, no incidence. Na, not applicable.
b Vascular lesions.
c Statistically significant (P ≤ .05) using a Cochran-Armitage trend test.
Circumanal gland adenoma was seen in one baclofen-treated male (4%) at 45 mg/kg/d. This rare tumor was considered unrelated to baclofen-treatment in view of the low occurrence, the absence of a dose relationship and because statistical significance was not achieved.
Pulmonary adenocarcinoma was seen in 1/25 baclofen-treated males (4%) at 90 mg/kg/d (reference data range: 0%-6.7% 14 ; 0%-5% 18 ) and harderian gland adenoma was seen in one baclofen-treated female at 90 mg/kg/d (4%; reference data range: 0%-13.3% 14 ; 0%-4% 18 ; also seen in a control male). These rare tumors were considered unrelated to the treatment with baclofen as there was no dose relationship, the incidence was low and statistical significance was not achieved. Other neoplasms were considered unrelated to the baclofen treatment because they were seen with a similar incidence in treated animals and vehicle controls, or only in vehicle controls.
Overall, although some increases in tumor incidence were observed among baclofen-treated mice compared to vehicle controls following the administration of baclofen up to 180 mg/kg/d for 26 weeks, there were no occasions in which increased incidence presented with compelling preneoplastic correlates or a dose-response. In addition, all findings and the incidence of neoplastic lesions were within or near the reported historical control ranges in this strain. Based on our interpretation of these results and published historical control data on spontaneous lesions in this strain of mouse, baclofen was considered to be noncarcinogenic in CB6F1-TgrasH2 mice.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_01926233211054767 - Carcinogenicity Evaluation of Baclofen in TgrasH2 Mice
Supplemental Material, sj-docx-1-tpx-10.1177_01926233211054767 for Carcinogenicity Evaluation of Baclofen in TgrasH2 Mice by Guillaume Chevalier, Nicolas Aubert, Catherine Thirion-Delalande, Bernard Palate and Pramila Singh in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-docx-2-tpx-10.1177_01926233211054767 - Carcinogenicity Evaluation of Baclofen in TgrasH2 Mice
Supplemental Material, sj-docx-2-tpx-10.1177_01926233211054767 for Carcinogenicity Evaluation of Baclofen in TgrasH2 Mice by Guillaume Chevalier, Nicolas Aubert, Catherine Thirion-Delalande, Bernard Palate and Pramila Singh in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-docx-3-tpx-10.1177_01926233211054767 - Carcinogenicity Evaluation of Baclofen in TgrasH2 Mice
Supplemental Material, sj-docx-3-tpx-10.1177_01926233211054767 for Carcinogenicity Evaluation of Baclofen in TgrasH2 Mice by Guillaume Chevalier, Nicolas Aubert, Catherine Thirion-Delalande, Bernard Palate and Pramila Singh in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-docx-4-tpx-10.1177_01926233211054767 - Carcinogenicity Evaluation of Baclofen in TgrasH2 Mice
Supplemental Material, sj-docx-4-tpx-10.1177_01926233211054767 for Carcinogenicity Evaluation of Baclofen in TgrasH2 Mice by Guillaume Chevalier, Nicolas Aubert, Catherine Thirion-Delalande, Bernard Palate and Pramila Singh in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-docx-5-tpx-10.1177_01926233211054767 - Carcinogenicity Evaluation of Baclofen in TgrasH2 Mice
Supplemental Material, sj-docx-5-tpx-10.1177_01926233211054767 for Carcinogenicity Evaluation of Baclofen in TgrasH2 Mice by Guillaume Chevalier, Nicolas Aubert, Catherine Thirion-Delalande, Bernard Palate and Pramila Singh in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-docx-6-tpx-10.1177_01926233211054767 - Carcinogenicity Evaluation of Baclofen in TgrasH2 Mice
Supplemental Material, sj-docx-6-tpx-10.1177_01926233211054767 for Carcinogenicity Evaluation of Baclofen in TgrasH2 Mice by Guillaume Chevalier, Nicolas Aubert, Catherine Thirion-Delalande, Bernard Palate and Pramila Singh in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-tif-1-tpx-10.1177_01926233211054767 - Carcinogenicity Evaluation of Baclofen in TgrasH2 Mice
Supplemental Material, sj-tif-1-tpx-10.1177_01926233211054767 for Carcinogenicity Evaluation of Baclofen in TgrasH2 Mice by Guillaume Chevalier, Nicolas Aubert, Catherine Thirion-Delalande, Bernard Palate and Pramila Singh in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-tif-2-tpx-10.1177_01926233211054767 - Carcinogenicity Evaluation of Baclofen in TgrasH2 Mice
Supplemental Material, sj-tif-2-tpx-10.1177_01926233211054767 for Carcinogenicity Evaluation of Baclofen in TgrasH2 Mice by Guillaume Chevalier, Nicolas Aubert, Catherine Thirion-Delalande, Bernard Palate and Pramila Singh in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-tif-3-tpx-10.1177_01926233211054767 - Carcinogenicity Evaluation of Baclofen in TgrasH2 Mice
Supplemental Material, sj-tif-3-tpx-10.1177_01926233211054767 for Carcinogenicity Evaluation of Baclofen in TgrasH2 Mice by Guillaume Chevalier, Nicolas Aubert, Catherine Thirion-Delalande, Bernard Palate and Pramila Singh in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-tif-4-tpx-10.1177_01926233211054767 - Carcinogenicity Evaluation of Baclofen in TgrasH2 Mice
Supplemental Material, sj-tif-4-tpx-10.1177_01926233211054767 for Carcinogenicity Evaluation of Baclofen in TgrasH2 Mice by Guillaume Chevalier, Nicolas Aubert, Catherine Thirion-Delalande, Bernard Palate and Pramila Singh in Toxicologic Pathology
Footnotes
Acknowledgments
We extend our thanks to Joanna Moore, ELS (Charles River Evreux, France), for help in the preparation of this manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Guillaume Chevalier, Catherine Thirion-Delalande, Bernard Palate, and Pramila Singh are employees of Charles River Laboratories, France, which received payment for the conduct of this research from Ethypharm SA. Nicolas Aubert was employed by Ethypharm SA, the sponsor of this study at the time when it was performed, but he has since left the company.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Guillaume Chevalier, Catherine Thirion-Delalande, Bernard Palate, and Pramila Singh are employees of Charles River Laboratories, France, which received payment for the conduct of this research from Ethypharm SA.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
