Abstract
Animal models are commonly used to investigate the developmental basis of human birth defects. Such models may be used for safety assessment studies designed to reveal xenobiotic-related alterations in juvenile animals, or to investigate gene function or generate models of human disease, as with transgenics. Therefore, the evaluation of rodent embryos and placentas can be used to provide insight into various postnatal abnormalities such as structural or cellular abnormalities and early death. Depending on the defect, pups may be born dead, survive for only a short period of time, survive but with poor growth, or survive and be clinically normal. Mice are generally used to generate genetic alterations that can help in identifying genes involved in embryogenesis. Rats are more commonly used for toxicology studies. This article aims to share information on the importance of, and strategies for, mouse embryo, placenta, and metrial gland evaluations. Information on early postnatal development is also provided as well as select examples of developmental information on organ systems needed for postnatal evaluations. A list of additional studies that can aid in the evaluation of prenatal and postnatal phenotypes is also provided.
Introduction
Mice have historically been used to investigate mechanisms of disease, to screen agents for efficacy and/or toxicity, to evaluate potential new therapeutic targets, and to discover biological roles for novel genes. Mice are small, have a large litter size, and short generation times, all features that are practical for laboratory work. The mouse also possesses a hemochorial placenta, which is similar to the human placenta. For these reasons, they are a popular model for studies of human pregnancy and placentation. Genetically altered mice have historically been used for the identification of genes involved in embryogenesis. Complete or partial inactivation of a gene may lead to loss or modification of gene function in cells and tissues during any stage of gestation. Since the mouse is a common model for transgenic and other types of treatment evaluations, the mouse embryo and placenta are the focus of this article. For other species, one should know differences in terms of study design, staging, maturation, normal histology, and so on.
The terms embryo and fetus are sometimes used interchangeably in the literature when describing rodent embryos. In humans, the distinction between embryo and fetus is based on gestational age. An embryo is the early stage of development in which the organs are formed. After the 11th week of pregnancy, the correct term would be fetus. Since the mouse has a much shorter gestation period and organ development continues after birth, the designation of embryo versus fetus is less important, and the term embryo will be used throughout gestation here.
The purpose of this article is to convey strategies for the phenotypic evaluation of embryos and postnatal pups with a focus on critical information that is needed before study evaluations, strategies for study design, and an overview of placenta and metrial gland anatomy. When there are postnatal abnormalities such as low litter size, early postnatal death, or structural or cellular abnormalities, it may be advantageous to evaluate embryos to determine the time and sometimes the cause of the postnatal abnormality. Hematopoiesis, heart, and liver are used as examples of how normal developmental biology after birth may impact the postnatal evaluation. Select resources for the evaluation of mouse embryo organ development are also provided.
Methods
CD-1 [IGS/Crl:CD1(ICR)] mice (Charles River Laboratories) were used, and all animal procedures were approved by the National Institute of Environmental Health Sciences (NIEHS) Animal Care and Use Committee. Pregnant mice were euthanized in the morning by carbon dioxide inhalation according to the NIEHS standard operating procedure for euthanasia of rodents. Using a dissecting stereomicroscope (Leica MZ16; Leica), the uterus and individual conceptuses (where the “conceptus” is an embryo and its placenta collected as a single unit) and metrial glands were isolated and immersed in ice-cold 0.1 M phosphate-buffered saline (1×, pH 7.4). The conceptus or isolated placentas were then fixed by immersion at room temperature in a commercially available solution of methanol-free 4% formaldehyde, pH 7.4 (colloquially termed 4% paraformaldehyde; Affymetrix). Fixation time was 48 hours for placentas with metrial glands alone and 72 hours for both individual conceptuses and conceptuses fixed in situ. Following fixation, placentas with metrial glands were processed routinely into paraffin. For early time points (embryonic age [E] 9.5 and earlier), conceptuses or isolated placentas with metrial glands were embedded while intact to avoid traumatizing the fragile placental (and embryonic) tissues. For later time points (≥E10.5) isolated, fixed placentas were placed on a hard surface and trimmed with a sharp razor blade on the axial plane to produce two asymmetric halves; the larger piece—comprising approximately two-thirds of the placental mass—was embedded with the cut surface oriented “down” in the cassette to permit acquisition of full-thickness cross sections. Serial 5-μm-thick sections were acquired and placed on charged slides (A. Daigger & Co) and routinely stained with hematoxylin and eosin for histopathologic review.
Strategies for Phenotypic Evaluation of Prenatal Embryos and Postnatal Pups
Important Considerations
There are a variety of topics that should be considered when performing embryo and perinatal evaluations. These include, but are not limited to, the use of the appropriate tissue fixation protocol for the specific embryonic age (E9.5-E11.5—2 hours, E12.5-E16.5—4 hours, and E17.5-E18.5—72 hours), the availability of appropriate control embryos, and comparison to published data when there may be species and/or strain differences. For an accurate and effective histopathology review, one must consider embryo age, developmental stage, and morphological appearance. This is because there can be considerable variation in the timing of ovulation and conception (up to 24 hours) and, therefore, in the developmental status of individual embryos within the same litter (Figure 1). Another important consideration for the evaluation of embryos and placentas is that it may not be possible to determine the cause of postnatal abnormalities, as the developmental irregularities in cellular, tissue, or organ structure and function that underlie congenital defects may have complex genetic, epigenetic, and/or environmental causes that are independent of the test variable. Finally, if death occurs shortly after birth or if defective pups are born, the dam may remove the pups before the litter is collected for screening.
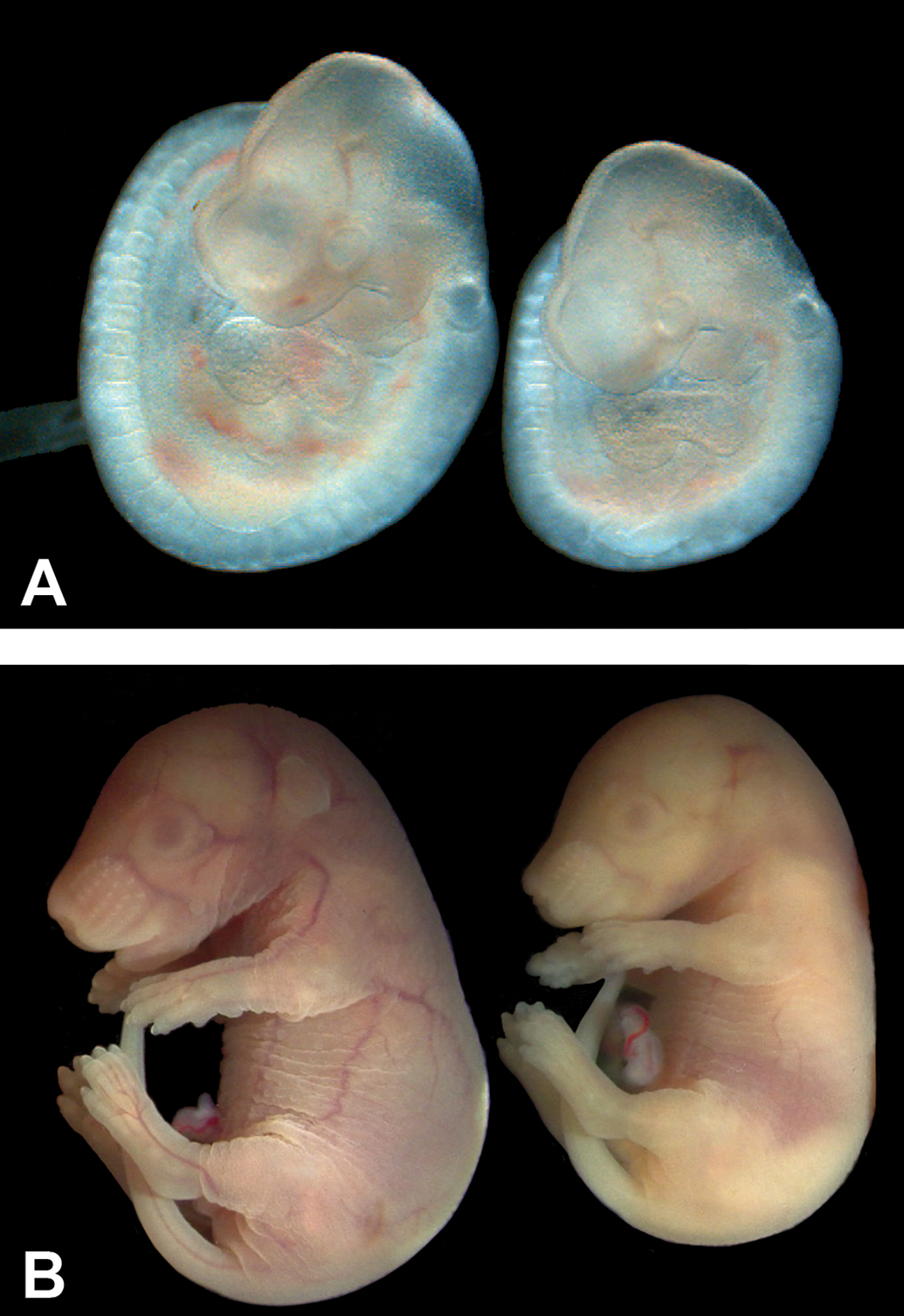
Morphological variation can occur among embryos of the same age. Gross embryo images (A) at E9.5 taken with a stereomicroscope to illustrate the size variation within a litter. Littermates at E18.5 (B) show a similar size variation. This size variation will most likely correspond to morphology variation when examined histologically. ([B] Reproduced from Elmore SA, et al. Histology atlas of the developing mouse placenta. Toxicol Pathol. 2021. In press.). E indicates embryonic age.
Essential Information Needed Prior to Evaluation
Before any embryonic or postnatal evaluation is performed, the reviewing pathologist should be apprised of basic information such as the manipulation of the dam (genetic experiments, chemical dosing, etc), the genetic background (strain, stock), prior knowledge of any defects due to genotype/phenotype or treatment, and finally any prior observations by the investigator of prenatal or postnatal deaths or morphological abnormalities. Also critical to the proper evaluation of embryos and pups is a working knowledge of age-specific normal anatomy and cellular morphology as well as common pathologies.
Study Design
The study design should be specific for each evaluation depending on known prior information, as discussed above, and the focus or goals of the overall study. This will determine what age and how many embryos to evaluate and how best to embed and section. The experimental units should first be determined (individual embryos or whole litters). Multiple litters may be needed to obtain the needed number of transgenics if too few embryos are obtained within a given litter due to early postnatal death or embryo resorption. A similar number of wild type or vehicle controls should always be included. Importantly, each species and strain have a normal background incidence of congenital defects that vary depending on the defect. Historical control data for that species and strain under similar conditions as the test condition are helpful for understanding the differences introduced by genetic manipulation and/or treatment.
How best to embed and section the embryos depends on the structures that are of interest. For an initial screening with no prior knowledge of defects, sagittal or frontal sections may be used (Figures 2 -4). Coronal sections are preferred for evaluation of brain development. For other specific organ system development, transverse or frontal sections may be a good choice.
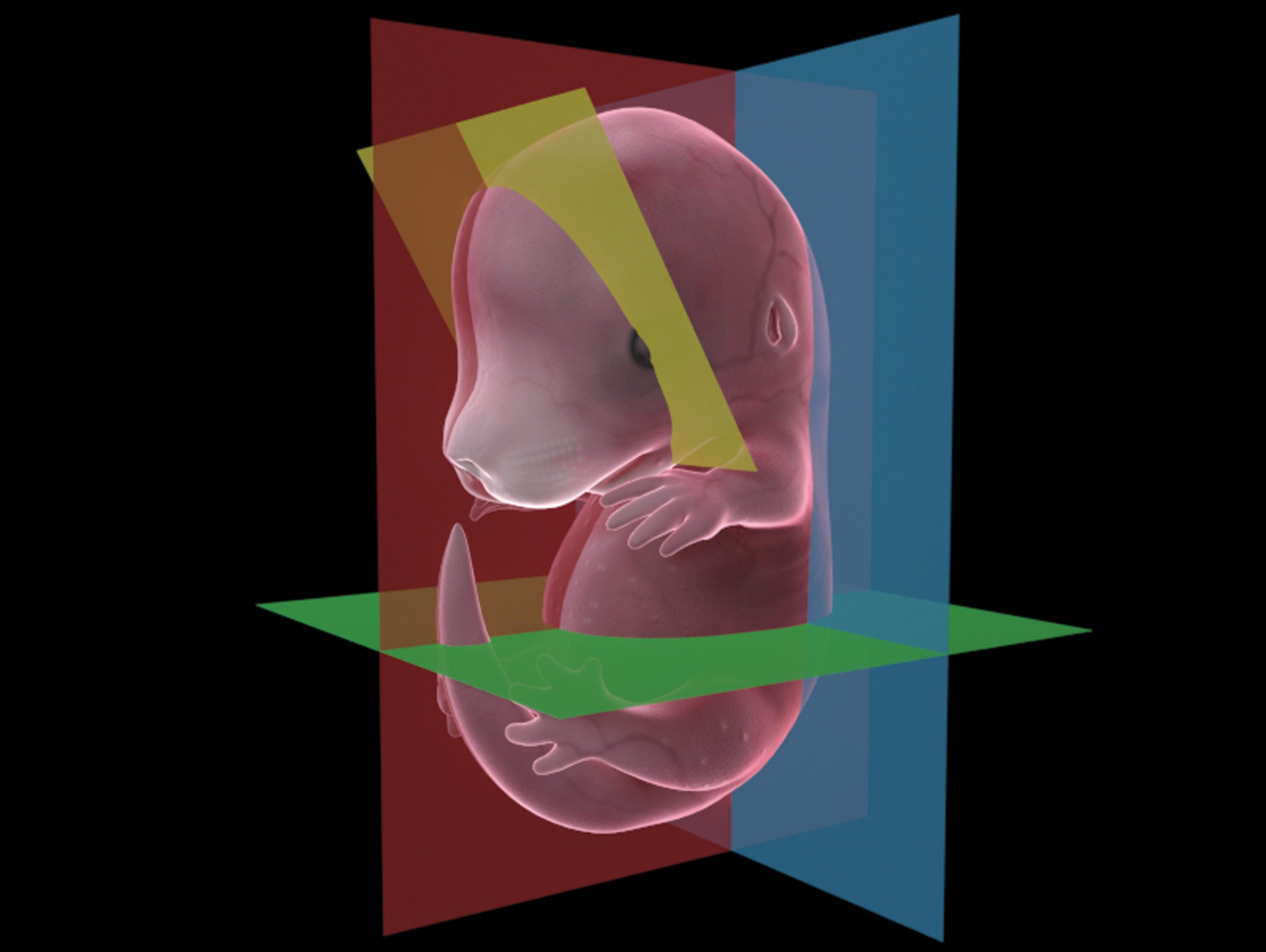
Three-dimensional graphic that shows the various options for embryo sectioning. Yellow is coronal, green is transverse, red is sagittal, and blue is frontal.
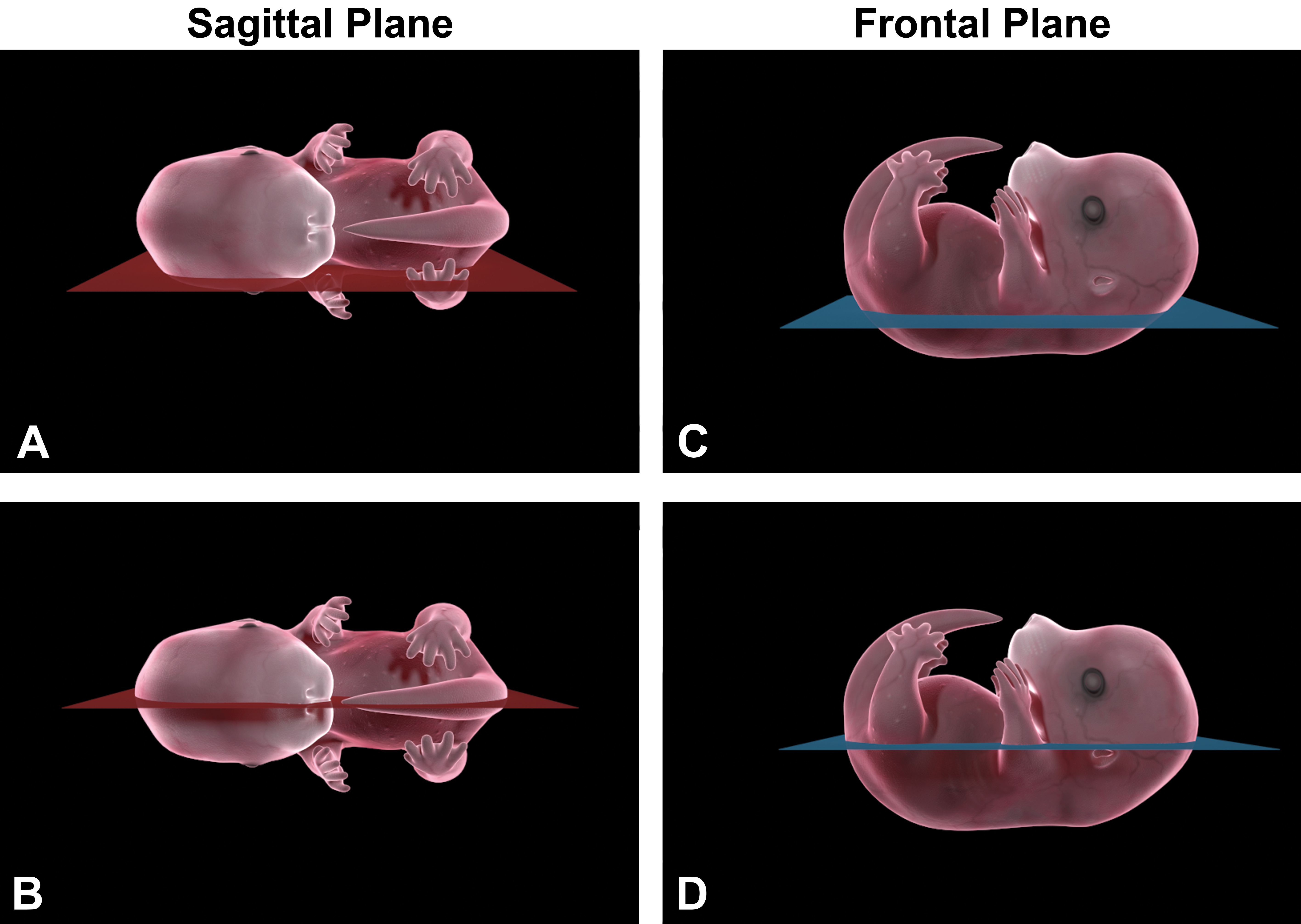
Sagittal sections are illustrated in (A) and (B). The embryo is embedded on its side and sectioned until the organs of interest are obtained. (C) and (D), Illustrate the frontal plane of sectioning where the embryo is embedded on its back and sectioned until the organs of interest are obtained.
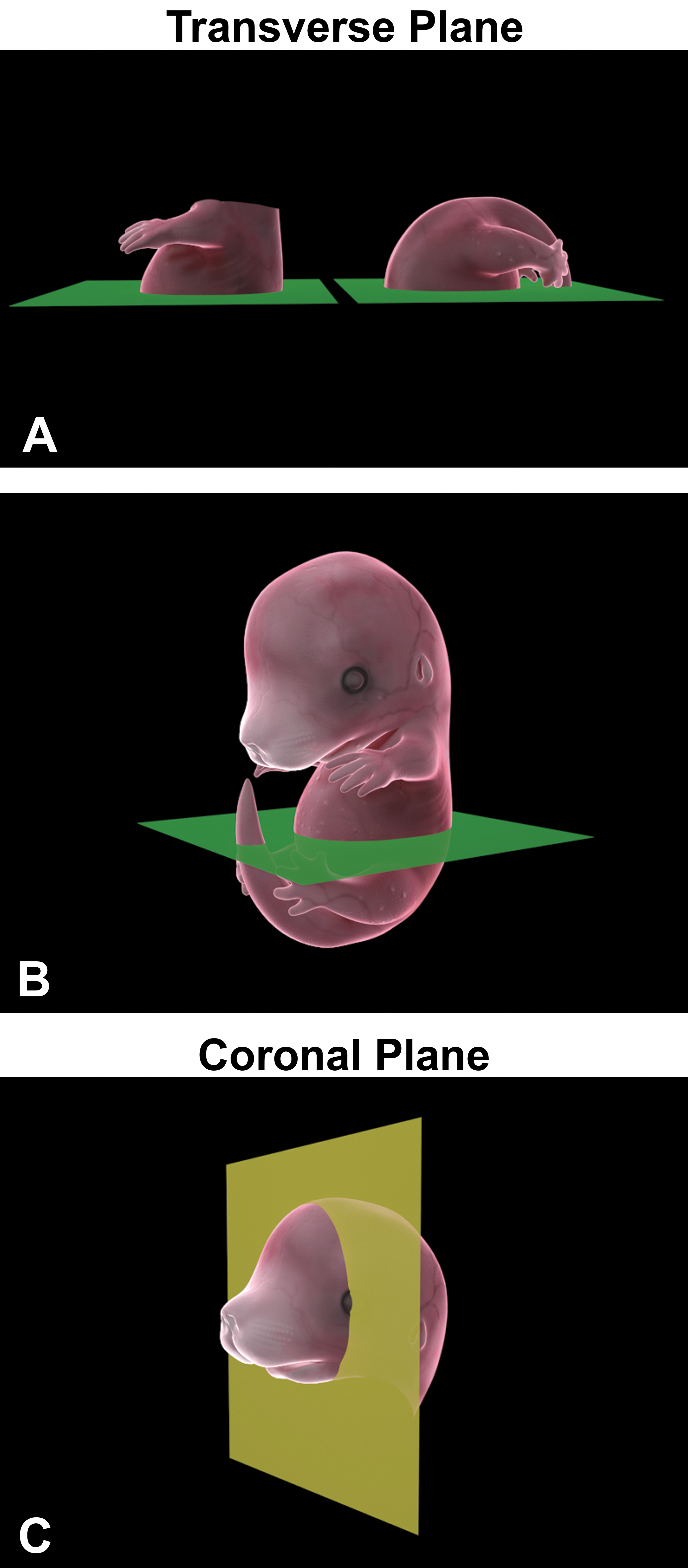
Transverse sectioning is illustrated in (A) and (B). The embryo is cut in half and one or both sides are embedded with the cut side down and sectioned until the organs of interest are obtained. (C) The coronal plane for sectioning the head with a specific focus on the brain, nasal cavity, ocular, or auricular tissues.
Mouse Stages of Gestation
When evaluating mice, knowledge of the stages of gestation provides important developmental information and may aid the pathologist in choosing the appropriate stages to review. Ages are approximate as there can be normal slight variations. The morning of the vaginal plug is designated embryonic day (E) 0.5. Pre- and postimplantation occurs between E0 and E4.5. That is, the blastocyst is free within the uterine lumen and then implants in the uterine wall at about E4.5 (range 4-5.5 days postconception). Differentiation of germ layers occurs between E6.5 and E9.5. The development of the placenta and cardiovascular system occurs between E9.5 and E12.5 and tissue/organ differentiation occurs between E12.5 and E18.5 with different critical periods for each organ. Finally, birth occurs between E19 and E21. 1
Tiered Strategy
The operational workflow for embryo analysis may be organized into primary, secondary, and tertiary “tiers.” This approach favors flexibility in experimental design and will provide a more cost-effective breeding scheme. One suggested tiered strategy, as depicted in Table 1, provides a purpose, subjects to evaluate, and end points for each tier. The purpose may be a basic description of the phenotype (tier 1), to evaluate younger ages for phenotypic abnormalities (tier 2), or to provide a detailed characterization of the pathogenesis of the phenotype (tier 3). Which tier to choose depends on what prior knowledge is available or possibly the end goal for the project. Importantly, each study project is different so applying a “cookbook” approach is not advised.
Tiered Strategy for Study Design and Evaluation of Mouse Embryos.

Abbreviation: E, embryonic age.
Mouse Placenta and Metrial Gland Evaluation
The placenta and metrial gland are two organs that are imperative for the normal growth and functioning of the developing embryos (Figure 5). The placenta is a multifunctional organ that serves a pivotal role in embryonic growth as it ensures an optimal environment for offspring survival. It is unique in that it acts as the lungs, kidneys, liver, as well as the gastrointestinal, endocrine, and immune systems for the developing embryo. Placental dysfunction and injury can be associated with both pre- and postnatal toxicity and/or death. Although gestation is short and much of organ development is completed postnatally, rodents are still a popular model for studies of human pregnancy and placentation. Moreover, both the human and mouse placentas are similar in that they are both hemochorial and discoid. 2 Discoid indicates that a single placenta is formed and is discoid in shape. Hemochorial indicates the number of cell layers in the interhemal area (ie, area of separation between maternal and fetal circulations). The hemochorial placenta has the trophoblast layer in direct contact with the maternal blood and not separated by endothelium and/or epithelium. Thus, the chorioallantoic placentas of mice belong to the same group as human placentas.
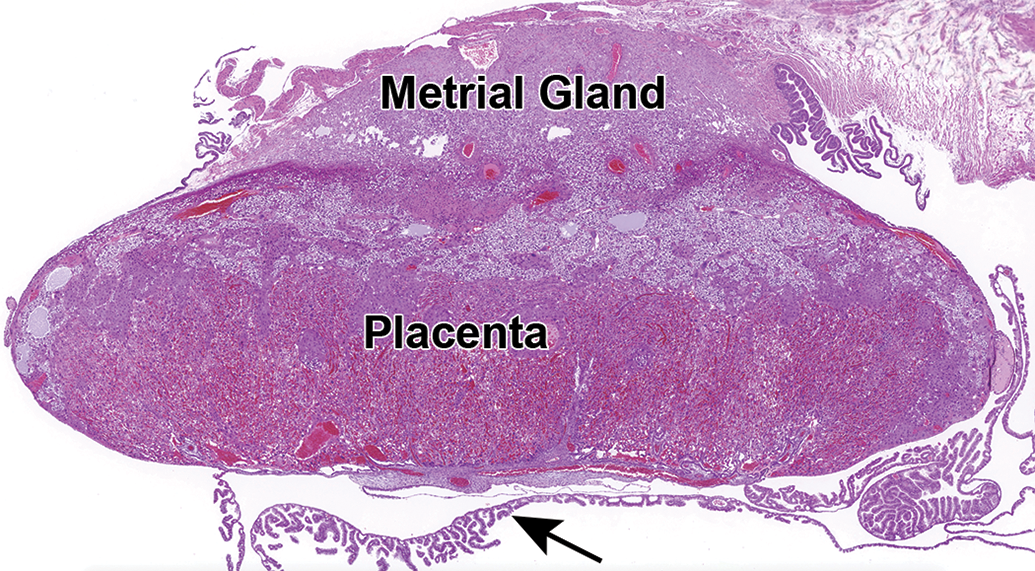
A low magnification view of the CD-1 mouse placenta and metrial gland at E17.5. Both tissues should be collected and evaluated along with the corresponding embryo. A section of the yolk sac can be seen below the placenta (arrow). Hematoxylin and eosin. E indicates embryonic age.
Mice, in addition to the chorioallantoic placenta, have a choriovitelline placenta—the inverted yolk sac placenta—which becomes active early in pregnancy and persists until term. This kind of placenta, which is typical for rodents, is absent in humans. For this type of placenta, the outer parietal layer of the yolk sac membrane and the overlying chorion disappears, and the visceral wall of the yolk sac is then in contact with the uterine lumen. 3 This type of a placenta allows for histiotrophic nutrition, which is the movement of nutritive substances to the embryo whereby local macromolecules are mainly responsible for the maintenance of the embryo. 4 Evaluation of this tissue in mice should not be overlooked as xenobiotic influence on the yolk sac placenta has been documented as a form of toxicity, and the mode of action has been shown to be interference of histotrophic trafficking. 5 There are other structural and molecular differences between mouse and human placentas that have been reviewed by Schmidt and colleagues. 6
The mouse chorioallantoic placenta has 4 anatomic divisions: decidua, junctional zone, labyrinth, and chorionic plate (Figure 6). The decidua is the maternal part of the placenta, and its functions include gas and nutrient exchange, hormone secretion, and protection of the embryo from the maternal immune system (Figure 7A). The junctional zone is located between the labyrinth and decidua and provides glycogen for energy, hormonal support, and physical support to ensure correct placentation and pregnancy progression (Figure 7B). The junctional zone is also considered the main endocrine compartment of the placenta. It produces hormones, growth factors, and cytokines that are important for the normal progression of pregnancy, acting on both the maternal and embryo physiology. 7,8 It contains specialized cells called glycogen cells, trophoblast giant cells, and spongiotrophoblasts (Figure 7C). Glycogen cells are believed to serve as an energy store that can provide additional nutrition to the placenta and/or embryo. The trophoblast giant cells help the embryo penetrate the uterine epithelium and implant into the endometrium. The function of the spongiotrophoblasts is poorly understood but may provide structural support for the labyrinth. 9 The labyrinth is the interface for gas and nutrient exchange between maternal and embryo blood circulations with an irregular and twisting vascular network (Figure 7D). It is composed of maternal sinusoids, trophoblastic septa, and embryo capillaries. The blood spaces are separated by vascular endothelium and 3 layers of specialized trophoblast cell types (cytotrophoblasts on maternal side and syncytiotrophoblast I and II cells on embryo side). Although this anatomical scheme provides an effective placental barrier, there are gap junctions that connect the syncytiotrophoblast layers, allowing for the transfer of small molecules between the maternal and embryo circulations. The chorionic plate is a layer of vascularized chorioallantois (trophoblast and mesoderm) that comprises the embryo side of the placental disc (Figure 7E). It is the attachment site for the umbilical cord and is involved in waste management, gas exchange, and nutrient transport.
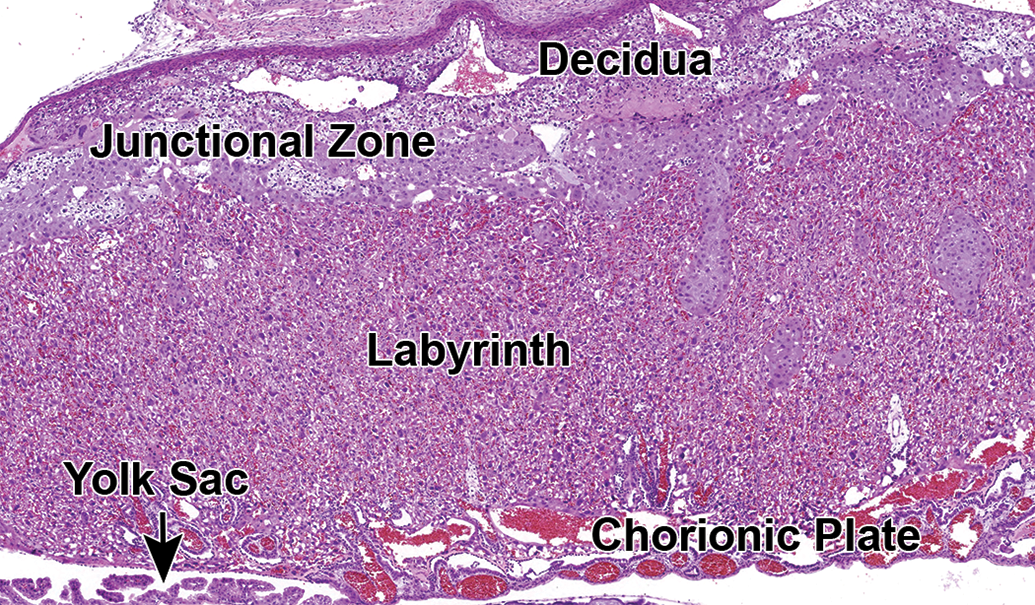
Overview of the CD-1 mouse placenta at E16.5 showing the anatomic locations of the decidua, junctional zone, labyrinth, and chorionic plate. Hematoxylin and eosin. E indicates embryonic age.
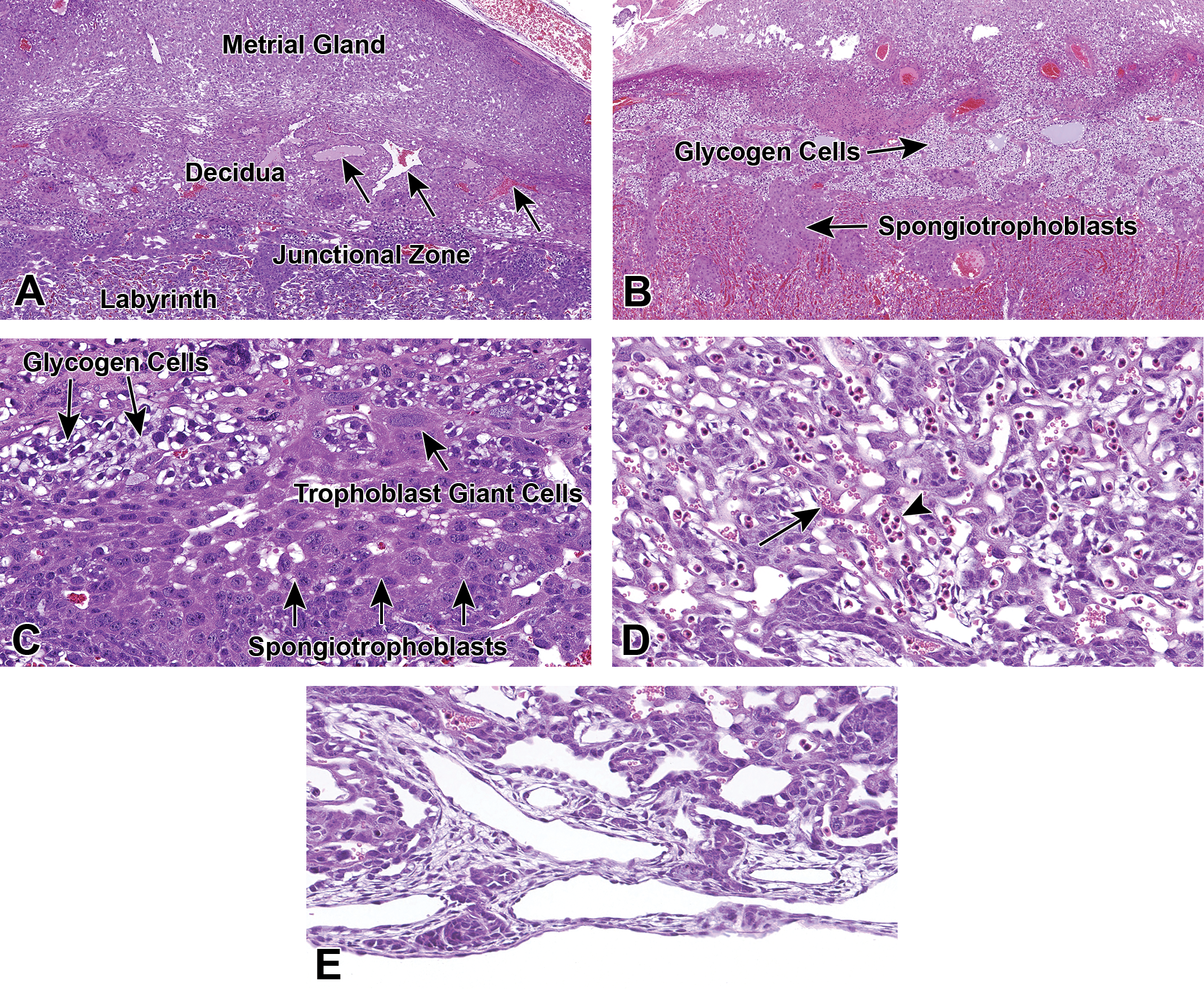
Higher magnification showing detail of the various regions of the placenta. The maternal derived decidua (A) lies between the metrial gland and junctional zone. The maternal spiral arteries (arrows) arise from the uterus and lack smooth muscle and elastica, allowing for the increasing maternal blood supply to the placenta. Important functions of the decidua include gas and nutrient exchange, hormone secretion, and protection of the embryo from the maternal immune system. The junctional zone (B and C) is positioned between the labyrinth and decidua and is composed of glycogen cells, spongiotrophoblasts, and trophoblast giant cells. Functions include glycogen for energy, hormonal support, and physical support to ensure correct placentation and pregnancy progression. The labyrinth (D) lies beneath the junctional zone and above the chorionic plate and is composed of maternal sinusoids, trophoblastic septa, and embryo capillaries. The blood spaces are separated by vascular endothelium and 3 layers of specialized trophoblast cell types. In embryos younger than ∼E14.5, the maternal blood spaces can be identified by anucleated RBCs (arrow), whereas the embryo blood spaces contain nucleated RBCs (arrowhead). The chorionic plate (E) is a layer of vascularized chorioallantois (trophoblast and mesoderm) that comprises the embryo side of the placental disc. This is where the umbilical cord attaches and its functions include waste management, gas exchange, and nutrient transport. All images are from CD-1 mice. (A) and (C) are from E11.5, (D) and (E) are from E12.5, (B) is from E17.5. E indicates embryonic age; Hematoxylin and Eosin; RBCs, red blood cells.
The labyrinth is a common toxicological target. Congestion, thrombi, degeneration, necrosis, and atrophy are some of the most common treatment-related lesions observed in this region (Figure 8A-E). Similar lesions may be seen in other areas of the placenta. However, as these lesions are sometimes observed in the normal regression process of the decidua as the gestation period ends, it is necessary to distinguish them from true toxicological lesions. The exact timing of these lesions may vary between mouse strains. Additionally, although the gestational (apparent) age may be the same within a litter, developmental (actual) ages may differ by as much as 24 hours. Comparison to concurrent controls is therefore critically important.
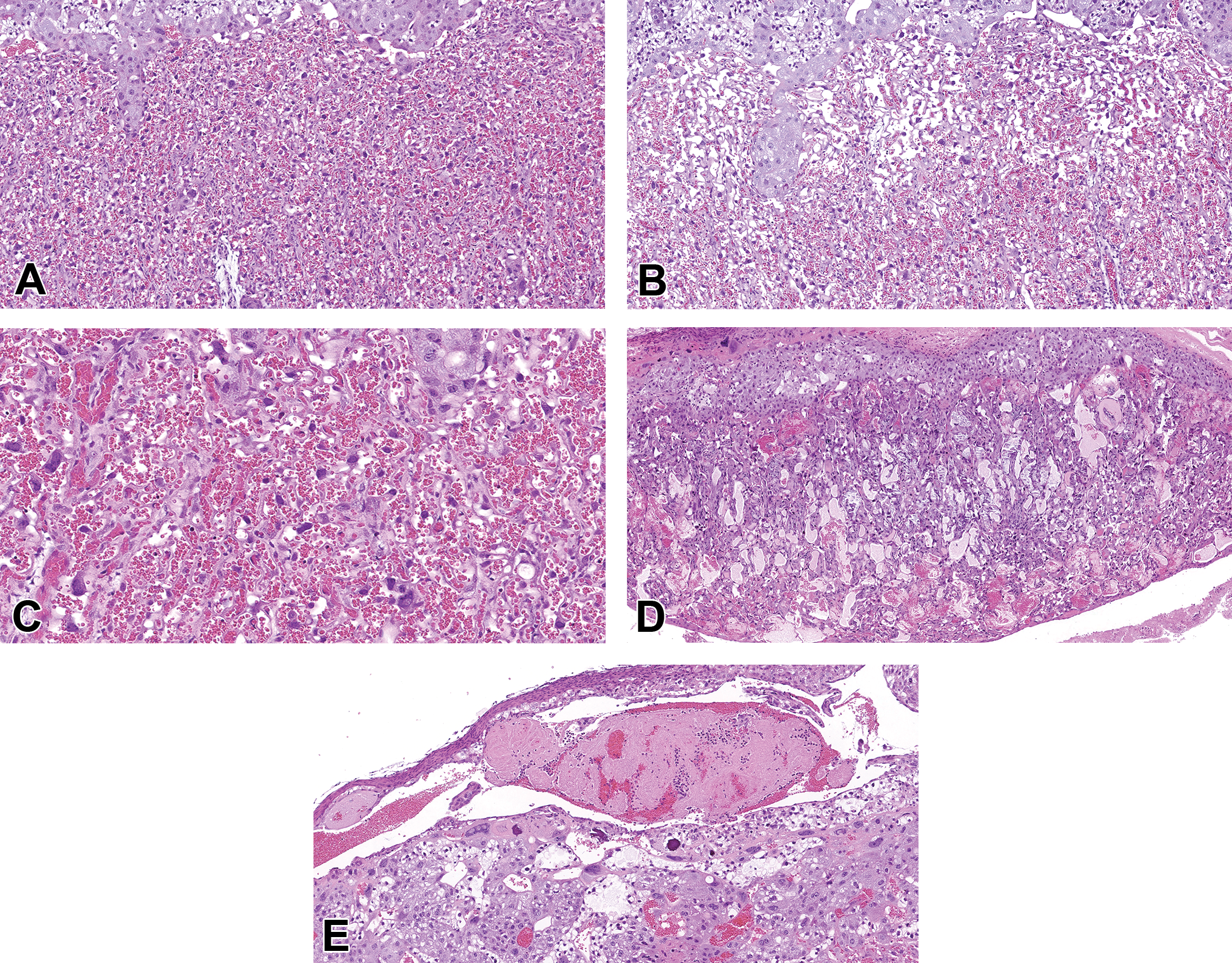
Common labyrinth pathologies include atrophy (B), congestion (C), necrosis (D), and intravascular thrombi (E). All examples from CD-1 mice are compared to a concurrent control with normal vascular morphology at E17.5 (A). Atrophy (B) is characterized by decreased labyrinth trophoblasts and other cellular contents. Congestion (C) is characterized by higher blood volumes expanding the vasculature. Necrosis (D) may represent a sign of primary placental damage but may also occur as a consequence of primary injury to the embryo. (E) Early thrombus formation in a maternal vessel following experimental treatment. Hematoxylin and eosin. E indicates embryonic age.
The metrial gland is another tissue that is important for embryo growth and successful placentation (Figure 9A). It is a pregnancy-specific modification of a segment of the uterine mesometrium that overlies the developing discoid chorioallantoic placenta of the mouse. It is the uterine entry point of blood vessels supplying each placenta and embryo and thus its biology and pathology are relevant to studying mechanisms underlying diseases such as preeclampsia and intrauterine growth restriction. It contains a dynamic mixture of blood vessels, endometrial stromal cells, trophoblasts, and fibroblasts. One important cell type to recognize is the granulated metrial gland (GMG) cell. 10 These large round to ovoid cells (up to 50 microns in diameter in mice) are easy to detect due to their numerous eosinophilic cytoplasmic granules (Figure 9B and C). They may be easily detected with Periodic acid–Schiff staining, with or without diastase digestion. They are derived from bone marrow precursors and are considered a subset of lymphocytes belonging to the natural killer (NK) cell lineage. 11 Importantly, because of their NK cell lineage, it is possible for GMG cells to play a primary or secondary role in pregnancy failure, so review of the metrial gland and GMG cells is always warranted.
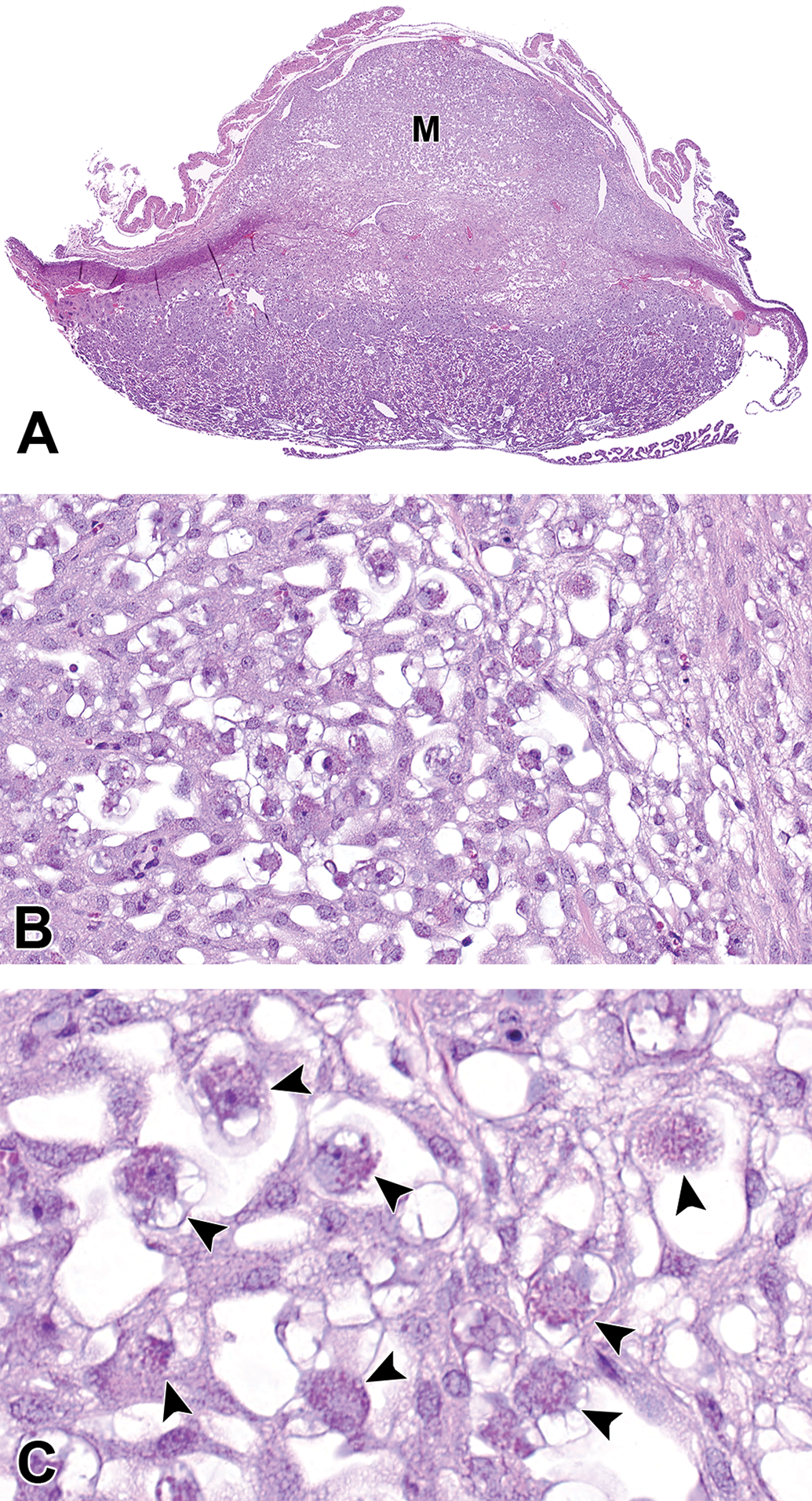
The metrial gland (M) from a CD-1 mouse at E12.5 is very robust (A and higher magnification B). This is a maternally derived tissue and is the uterine entry point of blood vessels supplying the placenta and embryo. At higher magnification (C), the granulated metrial gland cells (arrowheads) can be identified by their numerous eosinophilic granules. These cells are likely a subset of lymphocytes belonging to the natural killer cell lineage. Hematoxylin and eosin. E indicates embryonic age.
Mouse Embryo Evaluation
Before any evaluation of embryos or pups is undertaken, one should be able to recognize the normal anatomic and cellular features at each stage of embryonic gestation and each age of pup development. A working knowledge of major embryonic developmental time points and unique features of postnatal organ development is therefore required. This is important because one must be able to identify normal before abnormal phenotypes can be recognized. Normal may include a background incidence of abnormal and recognizing treatment or genetically induced changes over the background incidence of congenital defects is important. In addition, there are species and strain variations as well as litter effects that should be considered. Evaluation of, and comparison to, concurrent controls is a must. Similar embryo orientations (transverse, frontal, sagittal, coronal) should be compared. Additionally, a slight twist in an embryo during the embedding process may provide sections that cannot be compared.
There are a variety of published and online resources to aid the pathologist and biomedical scientist in the evaluation of embryos. The Mouse Embryo Phenotyping Core at the National Institutes of Environmental Health Sciences has published a series of mouse embryo atlases that each provide an overview of early development, H&E color sections from each embryonic stage, high and low magnifications, different planes of section, labeled structures, special stains, illustrative diagrams, common abnormalities, early postnatal development, and online digital images. To date, embryo atlases for the heart, hepatobiliary system, hepatobiliary hemolymphatic vascular system, central nervous system, and urinary atlas have been published; and the placenta, gastrointestinal tract, and respiratory system atlases are underway. 12 –16 Examples of additional published resources are provided. 17 –25 There is also an online tutorial of normal mammalian development using scanning electron micrographs, 26 an online high-resolution magnetic resonance histology atlas of the embryonic and neonatal mouse, 27 and the online Emouseatlas. 28
Mouse Pup Evaluation
In terms of postnatal development, knowledge of early postnatal development is important in terms of evaluating early postnatal abnormalities. Examples provided here are heart, hematopoiesis, and liver development.
Defects in embryo heart development may result in prenatal or postnatal lethality, so evaluation of this organ should be given careful consideration. 12 As part of normal development, there is some condensation, elongation, nodular thickening, and remodeling of the developing valve leaflets, resulting in thin, fibrous leaflets resembling the mature valve structures. This remodeling continues postnatally until around postnatal day (PND) 30. The entire heart undergoes “developmental hypertrophy” until around 3 months of age. Some of the common heart pathologies include hypocellular myocardial wall defect, persistent truncus arteriosus, double-outlet right ventricle, ventricular septal defect, and atrioventricular canal defects. 12
The mouse serves as an excellent model for mammalian hematopoiesis. In adult mice, hematopoiesis occurs mainly in the bone marrow. However, the site of hematopoiesis changes several times during ontogeny in the embryo. During embryonic development, hematopoiesis occurs in sequential waves to give rise to blood-forming cells with diverse lineage potentials and self-renewal properties. 29 –32 This transition of hematopoiesis is due to the migration and relocation of hematopoietic stem cells (HSCs) that are likely regulated by chemokines as well as adhesion molecules. In the first wave, hematopoiesis occurs as the appearance of blood islands in the developing yolk sac at about E7.5 and this is known as “primitive hematopoiesis” where mainly nucleated primitive erythrocytes are generated (Figure 10). These primitive hematopoietic cells enter the bloodstream and continue to mature while in circulation.
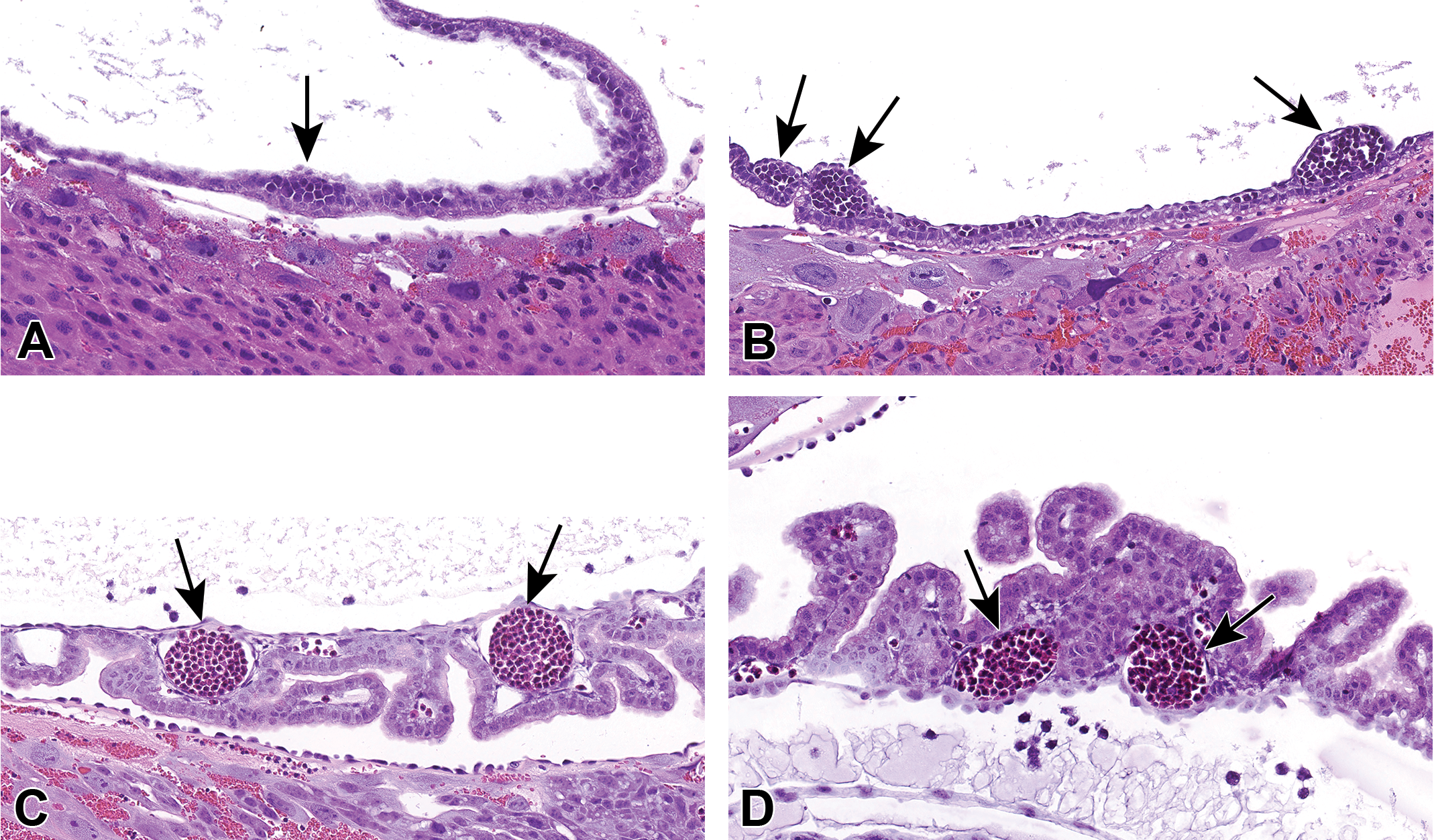
Examples of primitive hematopoiesis in blood islands in the developing yolk sac from embryonic ages E8.5 (A), E9.5 (B), E11.5 (C), and E13.5 (D) where mainly nucleated primitive erythrocytes are generated (arrows). CD-1 mouse. Hematoxylin and eosin. E indicates embryonic age.
The next hematopoietic wave occurs at around E9.0 to E11.5 in the aorta-gonad-mesonephros (AGM) region and lasts until around E13.5 (Figure 11). This is known as “definitive hematopoiesis” and is the first autonomous intraembryonic site of hematopoiesis. These are the first adult definitive HSCs and originate from the ventral endothelial wall of the embryonic dorsal aorta. These cells leave the bloodstream and enter multiple secondary sites (liver, then bone marrow) to occupy niches suitable for the expansion of HSCs. 33 They can differentiate into any of the blood cell lineages of the adult hematopoietic hierarchy and have long-term repopulating activity. The HSC activity is also detected in the placenta after E11.5.
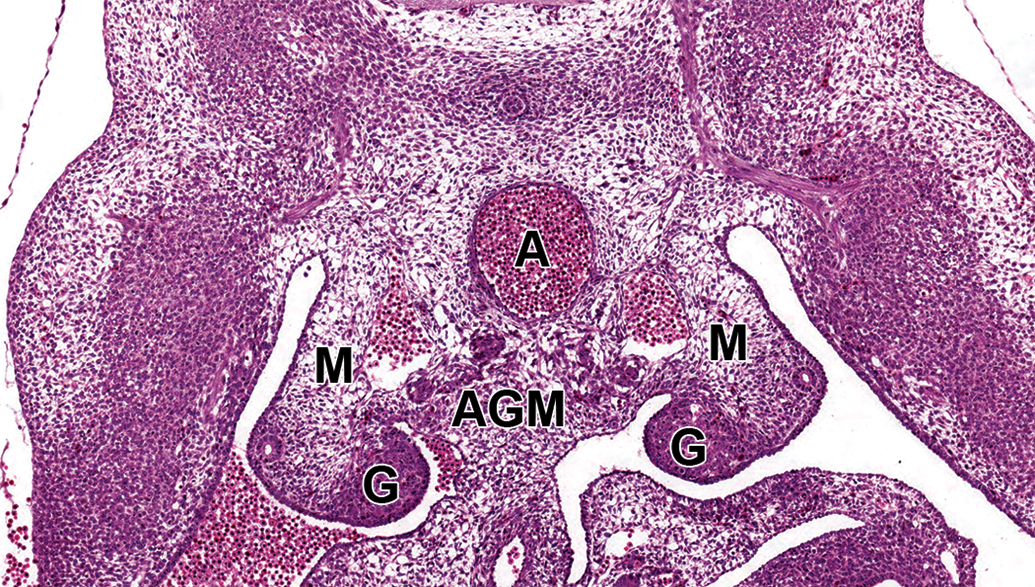
Section through the aorta-gonad-mesonephros (AGM) region of an E11.5 embryo. This is where the first autonomous intraembryonic hematopoiesis occurs. Hematopoietic stem cells originate from the ventral endothelial wall of the embryonic dorsal aorta, enter the bloodstream, and populate the embryo liver and finally the bone marrow. A indicates aorta; G, gonad; M, mesonephros. CD-1 mouse. Hematoxylin and eosin. E indicates embryonic age.
At around E12.0, the embryo liver takes over as the major source of hematopoietic cells during gestation (Figure 12) and serves as one of the sources for HSCs that eventually reach the thymus and spleen. Hematopoiesis in the mouse liver has been divided into 4 discrete stages. 34 Stage 1 occurs at E10.5 with expansion occurring during stage 2, between E11.0 and E12.0. Stage 3 is between E13.0 and E14.0, representing the peak volume of the hematopoietic compartment of the liver. At this stage, the hematopoietic colonies form the structural units of ellipsoidal foci with central macrophages surrounded by a ring of erythroblastic cells, eventually forming cords. 35 Stage 4 occurs at E15.0 when the cord-shaped hematopoietic foci become disrupted and round, solitary foci of hematopoietic cells form within the hepatic cell cords. The size of the hematopoietic compartment declines at this stage and the hepatocytes return to close contact with one another. Finally, at around E17.0, the embryo bone marrow begins producing hematopoietic cells and this is sustained throughout the life of the animal. The HSCs seeding the bone marrow come from secondary sites like the liver and spleen as the AGM has regressed by this stage of development.
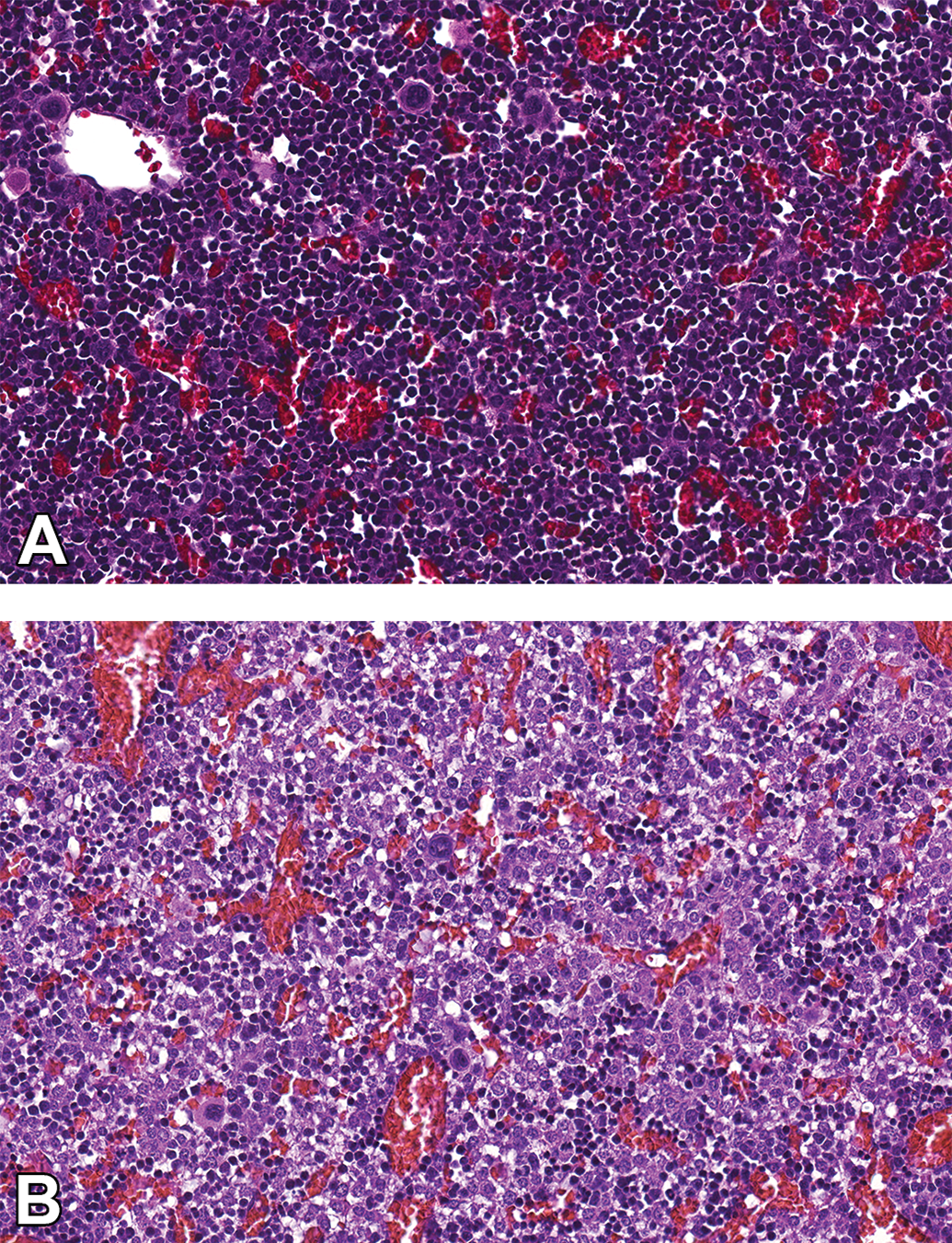
Hematopoietic cells in the mouse embryonic liver. At E14.5 (A) when the liver is at stage 3, peak hematopoietic production occurs. Compare that to E16.5 (B) during stage 4 when the cord-shaped hematopoietic foci become disrupted and round, solitary foci of hematopoietic cells form within the hepatic cell cords. The size of the hematopoietic compartment declines at this stage and the hepatocytes come into close contact with one another. CD-1 mouse. Hematoxylin and eosin. E indicates embryonic age.
In terms of postnatal hematopoietic events, the first week is the same as the near-term embryo. A developmental switch then occurs about 1 to 2 weeks after birth. Small foci remain in the liver until 2 weeks after birth where they can be seen surrounding a central macrophage. Megakaryocytes can be seen as isolated cells surrounded by hepatocytes. At 1 to 2 weeks after birth, embryo HSCs acquire adult characteristics in the mouse bone marrow in that they are capable of producing all classes of B cells. 36
Significant changes also occur in liver anatomy post birth. Liver mass increases by several fold in the first 3 weeks of postnatal life but declines in the fourth postnatal week when the liver:body weight ratio approaches adult levels. 37,38 Beginning around postnatal day 7, liver parenchymal cells (hepatocytes) begin to organize into well-defined hepatic plates. 39 Vascular development is dependent upon normal vasculogenesis, angiogenesis, and the transformation of vessels into their adult counterparts. Any alteration in vascular development has the potential to cause pre- and postnatal deformities or embryonic death. Proliferation of biliary cells, along with an increase in the formation of portal triads, can be observed between 10 and 20 days after birth. 37 Significant vascular events also occur during the postnatal period. The umbilical vessels are closed, terminating blood flow to the ductus venosus, which closes at around PND2, becoming the thin fibrous ligamentum venosum (Figure 13). The portal sinus with its tributaries now forms the distal aspects of the left portal vein and continues to supply the cranial portions of the left side of the liver. There is continued vascular remodeling. Intrahepatic bile ducts are established within the portal mesenchyme and, once this happens, hepatic artery formation is observed around PND3. The definitive portal triad with a branch of the portal vein, hepatic artery, and a lymphatic can be seen at around PND21.
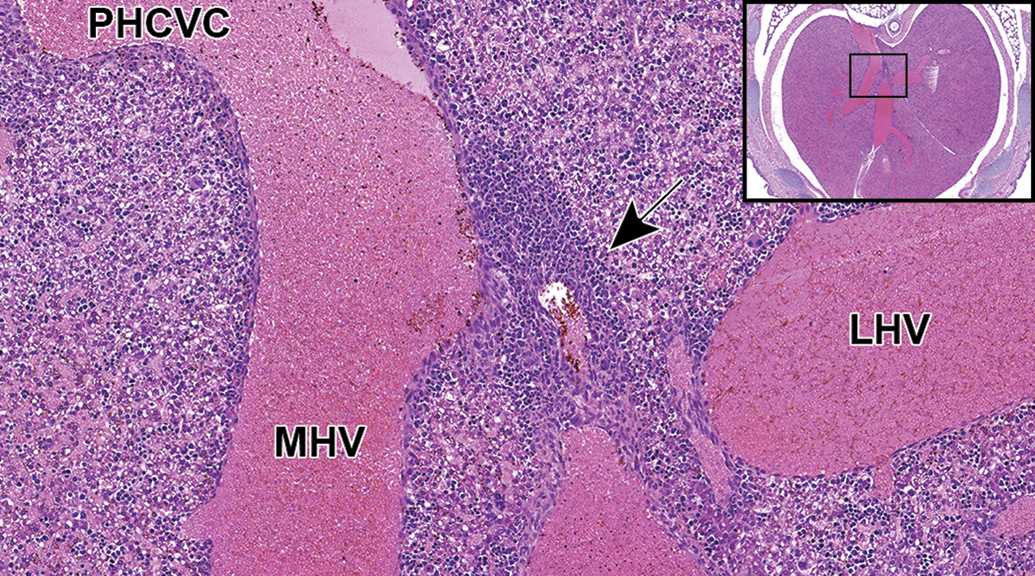
Soon after birth, the ductus venosus constricts into a fibrous cord, the ligamentum venosum, that is identified as a thin, linear band of fibrous tissue (arrow) positioned between the posthepatic caudal vena cava (PHCVC) and the portal sinus. Transverse sections of PND3 mice are best for observing the newly transformed ligamentum venosum. LHV indicates left hepatic vein; MHV, middle hepatic vein. (Reproduced with permission, Swartley et al, 2016). CD-1 mouse. Hematoxylin and eosin.
Additional Studies
There are a host of additional studies that can aid in the evaluation of pre- and postnatal phenotypes. Examples are whole embryo culture, whole mount in situ hybridization, clinical pathology, blastocyst evaluation, and a variety of noninvasive imaging modalities such as ultrasound, optical coherence tomography, microcomputed tomography, scanning electron microscopy, magnetic resonance imaging, and high resolution episcopic microscopy. There are also a variety of methods to assess gene expression such as special stains/immunohistochemistry, laser capture microdissection, RNA or DNA in situ hybridization, real-time qualitative polymerase chain reaction, DNA microarray analysis, and RNA sequencing. Most of these research methods would be done in the research setting and in conjunction with phenotypic evaluations.
Conclusions
The cause of early postnatal pathologies, whether death or structural abnormalities, can sometimes be understood by embryo phenotyping. It is possible for mice to survive the gestation period with an organ or system developmental defect, especially defects of the skin, skeleton, lungs, kidneys, liver, and nervous, immune, endocrine, and reproductive systems. Defects involving the placenta, erythroid, or platelet development and the heart often result in intrauterine mortality. Because of the complexity of intrauterine or postnatal death/pathologies, there is no “one size fits all” when it comes to embryo phenotyping. Each study is unique and will require a thorough understanding of the genetic manipulation, dam treatments, organs of interest, or any other potential confounding factors. But once designed, all evaluations are done in a systematic manner with a complete understanding of normal physiology and anatomy at all stages of development. As development of some organ systems, such as central nervous system, liver, heart, urinary tract, and so on, can continue into the postnatal period, a working knowledge of those developmental changes is needed as well.
Footnotes
Acknowledgments
This work was supported by the NIH, National Institute of Environmental Health Sciences. Appreciation to Beth Mahler (EPL) for figure preparation, David Sabio for the illustrations in Figures 2-4, and Drs Cynthia Willson (ILS) and Beth Lubeck (NIEHS) for critical manuscript review.
Declaration of Conflicting Interests
The author declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
