Abstract
The Society of Toxicologic Pathology (STP, https://www.toxpath.org/) was founded in North America in 1971 as a nonprofit scientific and educational association to promote the professional practice of pathology as applied to pharmaceutical and environmental safety assessment. In the ensuing 50 years, the STP has become a principal global leader in the field. Society membership has expanded to include toxicologic pathologists and allied scientists (eg, toxicologists, regulatory reviewers) from many nations. In addition to serving membership needs for professional development and networking, major STP outreach activities include production of articles and presentations designed to optimize toxicologic pathology procedures (“best practice” recommendations), communicate core principles of pathology evaluation and interpretation (“points to consider” and “opinion” pieces), and participation in international efforts to harmonize diagnostic nomenclature. The STP has evolved into an essential resource for academic, government, and industrial organizations that employ and educate toxicologic pathologists as well as use toxicologic pathology data across a range of applications from assessing product safety (therapies, foods, etc) to monitoring and maintaining environmental and occupational health. This article recapitulates the important milestones and accomplishments of the STP during its first 50 years.
Introduction
Toxicologic pathology is a medical discipline that applies the principles and practices of pathology (the study of diseases) to toxicology (the study of poisonous agents and their effects on humans, animals, and/or the environment). Toxicologic pathologists work in many settings: academic institutions, government laboratories and regulatory agencies, industrial companies (agrochemical, biopharmaceutical, medical device, etc), contract research organizations (CROs), or as consultants. Depending on cultural and geographic preferences, toxicologic pathologists may utilize many different techniques and tools in their daily practice. Common options include traditional anatomic pathology end points (eg, macroscopic observations, organ weights, microscopic and ultrastructural evaluation) and clinical pathology data (eg, hematology, clinical chemistry, coagulation, urinalysis, and cytology). In recent decades, toxicologic pathologists also have spent an increasing amount of time on innovative pathology end points such as biomarker development and validation, molecular pathology (eg, immunohistochemistry, in situ hybridization, and various “–omics” tools), and quantitative microscopy (eg, digital imaging and digital image analysis, morphometry, and stereology). In the end, toxicologic pathologists integrate their comprehensive biomedical knowledge with study-specific pathology data to identify and characterize potential hazards. Toxicologic pathologists thus are instrumental in the risk assessment discussions required to safeguard humans, animals, and the environment from potentially toxic substances.
The Society of Toxicologic Pathology (STP) is a professional organization founded in North America with a focus on the application of pathology within the field of toxicology. October 2021 marks the 50th anniversary of the Society’s founding. This article provides a brief historical record of the purpose for the organization, major past and present contributions to the field, and current and future involvement as a global leader in using toxicologic pathology to protect human, animal, and ecosystem health.
The STP Mission
At its inception, the STP began as the Society of Pharmacological and Environmental Pathologists (SPEP or more recently SOPEP). The name reflects the greater emphasis of toxicologic pathologists on environmental toxicology challenges in the 1970s than what is typical today. 1 Like other impactful organizations, the STP began with motivated individuals who had a vision and pursued it with dedication. The specific objectives of SOPEP were (a) to foster the discipline of pharmacologic and environmental pathology; (b) to contribute to the development of criteria and standards for the evaluation of pathologic changes induced by drugs, chemicals, or environmental agents; (c) to promote the training of pathologists involved in all ancillary fields; and (d) to organize a registry of naturally occurring and experimentally induced diseases in all species of laboratory animals. 2,3 Fifty years later, the STP still participates in activities that address these objectives while having expanded its mission to serve additional purposes.
The STP of today strives to “be an international leader for improvement of human, animal, and environmental health using an interdisciplinary scientific approach based in pathology and toxicology” (https://www.toxpath.org/aboutstp.asp#mission). This mission of global leadership is pursued through 4 strategic goals:
The same mission and goals are shared to a variable extent by other global societies of toxicologic pathology. However, the STP is particularly well suited to address such goals due to its status as the longest standing such Society, its large international membership, and its proven procedures (administered by experienced and enthusiastic volunteer committees) for identifying and responding to issues as well as crafting guidance documents and opinion pieces that define consistent practices for generating and interpreting toxicologic pathology data.
Historical Highlights of the STP
The Society has evolved substantially in terms of organization and activities during its 50 years of existence. In many respects, this evolution mirrors the cultural shifts that have influenced the scientific community as a multidisciplinary collaborative enterprise as well as the many social currents affecting national and global interests of the public at large. A useful means of evaluating the implications of such shifts is to examine the history of the STP as the Society has changed over time (Figure 1).
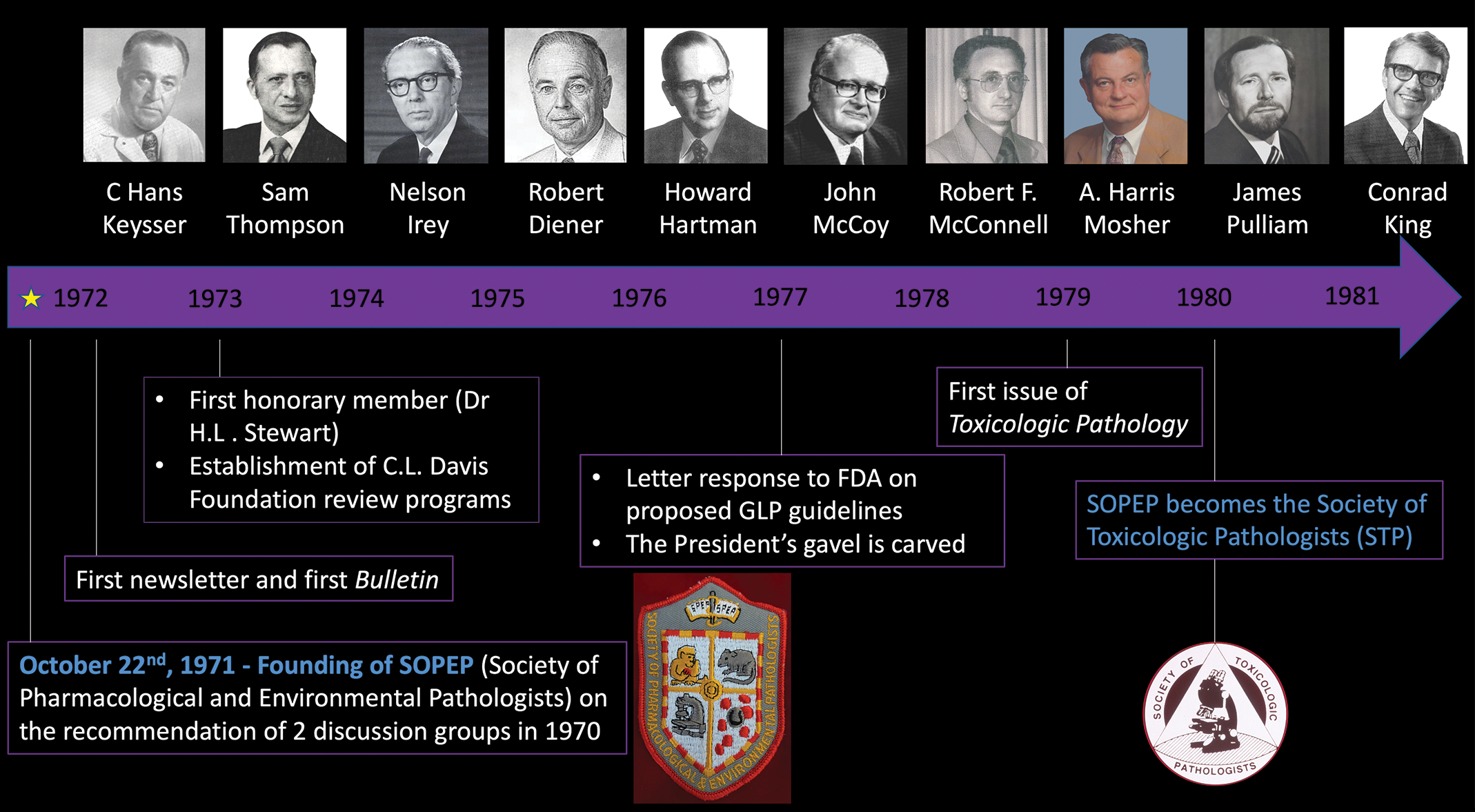
A-E, Historical highlights of the society over the first 50 years.
Learning to Fly—1971 to 1980
The Society was incorporated in New Jersey as a nonprofit scientific and educational organization on October 22, 1971. The rise of SOPEP began with 2 discussion groups, both originating in 1970, regarding the need for improved standardization and training of pathologists engaged in the discovery and the development of chemical and pharmaceutical agents. One group was steered by the team of Claude Biava, MD; Nelson Irey, MD; Hans Keysser, MD; and Daniel Sasmore, DVM, while the other was led by Howard Hartman, DVM and Samuel Thompson, DVM. The leadership of these groups reflected a central involvement of medical pathologists in toxicologic pathology during the 1970s. The consolidation of these groups under the SOPEP banner was followed by designation of an “Organizing Committee,” members of which elected the inaugural Executive Committee (EC). The first EC officers were Dr Keysser as President; Dr Thompson as President-elect; Dr Hartman as Secretary–Treasurer; and Dr Robert Diener, Dr J.R.M. Innes, Dr John McCoy, Dr Benjamin Sparano, and Dr Robert Van Ryzin as Councilors. The EC members elected a Chair to assume primary responsibility for leading activities of the Society, while the President fulfilled a more limited figurehead role; Dr Diener was the first Chair. Later, Chairs were required to serve on the EC for at least 2 years prior to seeking the position. This organizational structure was deliberately designed to ensure that the top STP leader (Chair) had substantial experience with societal activities and that the next key leader (President) would be selected from a broader candidate pool regardless of past STP experience. The EC Chair position was retired in 1999, and the STP leadership activities were transferred to the President. The symbol of presidential authority and responsibility, the gavel (Figure 2), was created during these early years.

History of the Society’s presidential gavel. In 1976 to 1977, Dr Howard Hartman contacted Nelson Irey, MD, at the US Armed Forces Institute of Pathology (AFIP), a fellow Society Founder and third President of the Society. Dr Irey secured a piece of black walnut from the leg of a specimen display case from the first Army Medical Museum, which had originated in Ford’s Theater shortly after it was closed following the assassination of Abraham Lincoln in April 1865. The gavel was presented to the Society and used when the next annual meeting was called to order at Hoffman La Roche in Nutley, New Jersey (1977). A striking block for the gavel was crafted in 2003 from 2 pieces of wood. One is of black walnut from North Carolina, and the second is a cross section of a teakwood rail from the original trans-Atlantic liner the RMS Queen Elizabeth. The teakwood insert was used to symbolize the growing international character of the Society. At each Annual Meeting, the gavel is passed from the outgoing president to the incoming president, a tradition that continues to this day.
The first SOPEP committees were assembled to undertake administrative tasks and/or address important scientific questions. Most committees fulfilled administrative functions typical of any professional society. Key groups that were formed to benefit members included the Continuing Education and Special Education Committees, to devise training opportunities to benefit all members or individual members with specialized educational needs, respectively; an Elections Committee to oversee periodic selection of new officers; a Discussion Groups Committee to stimulate formation of local SOPEP “chapters”; a Publications Committee to edit and publish a newsletter and assess the eventual feasibility of a societal journal; and a Finance Committee to develop procedures for soliciting corporate and individual support for various societal initiatives. These standing committees were essential in both perpetuating the Society and enhancing its value to its members.
However, the more important contributions of the first SOPEP committees were the noteworthy efforts to improve the professional practice of toxicologic pathology and the relevance of pathology data sets in risk assessment of pharmaceutical, chemical, and other test materials. Two standing committees were established for this purpose in 1971 to explore means of refining carcinogenicity risk assessment; indeed, the vital importance of this issue was a key driver for establishing SOPEP. The Carcinogenicity Testing Committee was launched to review current philosophical considerations of programs (and specifically pathology end points) designed to investigate the carcinogenic potential of chemicals. The Control Data Sharing Committee was begun to define procedures for assembling, storing, retrieving, analyzing, and distributing historical control data (especially from lifetime carcinogenicity bioassays in rodents). In 1972, the Control Data Sharing Committee met with representatives of the US National Cancer Institute, US National Library of Medicine, and Pharmaceutical Manufacturers Association (PhRMA) to seek funding for the project. These multilateral discussions led to a decision that neither government agencies nor private industry should provide funding in order to avoid conflicts of interest. The inability to gain funding from private foundations despite numerous appeals by the committee resulted in the Society abandoning the effort in 1978. Despite the lack of success, the leading role that the Control Data Sharing Committee played in orchestrating these multidisciplinary discussions was instrumental in cementing the relevance of the Society as a leading resource for subject matter expertise in toxicologic pathology. The SOPEP pivoted in 1978 to discussions of establishing a reference library of histopathologic lesions (spontaneous and chemically induced) in laboratory animals. This initiative represented an early effort to generate a harmonized lexicon for diagnostic nomenclature in toxicologic pathology.
Several ad hoc STP committees were formed in this same time frame to help improve the relevance of animal-derived pathology data. A Committee on Good Laboratory Practice (GLP) was approved in 1976 due to interest by Howard Richardson, MD, the Chief of Pathology for the Office of Pesticides Programs at the US Environmental Protection Agency (EPA), who had served as a SOPEP EC member in 1973. Dr Richardson impressed upon the EC members that toxicologic pathologists must be aware of federal agency regulations guiding the practice of pathology in safety testing, resulting in a commitment to make SOPEP members aware of items appearing in the Federal Register that might affect the profession. 3 A natural extension of such increased awareness was preparation, in 1977, of a SOPEP letter to the US Food and Drug Administration (FDA) to provide a detailed response to proposed GLP guidelines for pharmaceutical research. This letter also acknowledged that the Society was considering means for certifying the proficiency of toxicologic pathologists. An ad hoc Committee of Recognition was added to explore the creation of a SOPEP certification process versus accreditation mechanisms available through allied scientific societies (eg, the American Board of Toxicology, the American College of Veterinary Pathologists [ACVP], and the College of American Pathologists). A lengthy debate by the EC concerned the feasibility of developing a certifying process suited to comparative pathologists (ie, PhD or an equivalent graduate degree) and diagnostic pathologists (ie, MD, DVM, or a similar medical degree). Ultimately, the EC decided that the Society (1) recognized a need for additional toxicologic pathologists to fill the anticipated needs of industrial firms and governmental agencies; (2) should establish recommendations for training toxicologic pathologists; (3) consider approaches to create a certification mechanism for toxicologic pathologists; and (4) develop liaisons with other professional organizations to coordinate any similar initiatives. Accordingly, a Committee on Professional Standards was established to address these new propositions, resulting in a recommendation that 5 to 8 years of postgraduate experience is necessary before an individual might be considered “qualified.” The Committee suggested that SOPEP full membership criteria be tightened and that new categories of membership for less experienced individuals be established. Final decisions were presented to the entire SOPEP membership in a 1980 ballot. The outcome of the election was that SOPEP members chose to (1) require letters of recommendation from experienced toxicologic pathologists for SOPEP admission as either a Full Member or a newly created Associate or Affiliate Member and (2) maintain the Society’s focus on education, leaving the accreditation function to other societies with established means for certifying pathologists and/or toxicologists.
The SOPEP experienced a time of transition during the last years of the decade. In 1978, the EC agreed to change the objective of the Society’s flagship publication from a member-focused periodical to a more science-oriented journal. This shift was undertaken to enhance the stature of the Society’s subject matter expertise and to broaden the appeal and relevance of toxicologic pathology research to toxicologists, health authorities, and other scientists.
1
The transition occurred through a name change for the SOPEP journal from

A, Evolution of the society emblems and logos. In 1973, the original crest (left) of the Society of Pharmacological and Environmental Pathologists (SOPEP) was designed by Dr J.R.M. Innes. Upon change of the organization’s name to the Society of Toxicologic Pathologists in 1980, a new logo (center) was designed and later revamped at the beginning of the 90s (right). B, In 2000, the Society logo was updated (left) to reflect the name change to Society of Toxicologic Pathology, and this version is still in use today. Special logos were created to commemorate the Society’s landmark 25th (center) and 50th (right) anniversaries.
Growing Up—1981 to 1990
Despite the transition, business of the Society continued as usual. Periodic “Discussion Groups” organized around specific topics were held regularly, with most meetings taking place in the Northeast US and hosted by local pharmaceutical firms. Attendance at these regional meetings was steady, typically involving 30 to 50 members. Such sessions were designed to integrate basic information with practical applications. For example, one highly successful 2-day workshop in 1980 on “Necropsy Techniques for Technicians Involving Laboratory Animals” included wet labs. Despite the dedication to “staying the course,” tectonic shifts in the regulatory environment with respect to product discovery and development catapulted the STP to a more vital position in ensuring the efficacy and safety of new chemicals and pharmaceuticals.
The importance of the annual scientific meeting as a prime STP function was obvious in the early 1980s. The First International Symposium was convened on May 10-12, 1982, in Reston, VA, thereby establishing the precedent that STP meetings would be held near Washington, DC on a regular basis to facilitate interactions with regulators and scientists from federal government agencies. Indeed, the first 4 STP Symposia were held in Northern Virginia or Washington, DC, after which symposia were rotated among hotels in large urban centers in the eastern United States for the remainder of the decade (Supplemental Table 1). Nonetheless, the Symposium was touted as “international” due to the growing numbers of STP members based in several European countries (primarily England, France, Germany, and Switzerland). Manuscripts based on talks given at this Symposium were assembled into the first meeting-focused journal issue to provide a permanent educational resource to benefit Society members and other stakeholders (especially health authorities tasked with regulating chemical and pharmaceutical products).
The themes for International STP Symposia in the early 1980s covered broad topics (Supplemental Table 1). The theme of the First Symposium in 1982—“Rodent Liver Nodules: Significance to Human Risk”—was controversial, and topics for the next 2 Symposia were equally momentous: “Design of Carcinogenicity Studies: Considerations in Pathology Interpretation” in 1983 and “Estimating Human Safety from Animal Tumor Data” in 1984. Thereafter, Symposium themes for the remainder of the decade focused on toxicologic pathology methods and findings in various target systems (Supplemental Table 1). Regional “Discussion Groups” continued in both the United States and Europe as a supplemental means for professional engagement, but attendance at local meetings waned to some degree with the rising attendance at the annual symposia (approximately 200-250 per year by the mid-1980s).
Entering its second decade, the STP exerted substantial effort to raise the domestic and international profiles of the Society and its members as preeminent specialists in this scientific field. A Professional Relations Coordinator role was created in 1981 “[t]o establish a working basis on which the Society will be recognized by the scientific, regulatory, and business communities as a credible organization of specialized experts.” Additional initiatives during this period included further exploration of a certification mechanism for toxicologic pathologists and a proposal to consider preparing repositories of common experimentally induced findings in laboratory animal species to provide more consistency in microscopic diagnoses within toxicologic pathology reports across product classes and sponsor companies. The certification quest remained at the discussion stage for the remainder of the decade. The repository proposal culminated with EC decisions to establish STP subcommittees with international membership to design and execute 2 interrelated projects. The first effort, launched in 1981, was to begin long-range planning of a Registry of Toxicologic Pathology for Animals (RTPA). The second venture, started in 1988, was to begin the assembly of species- and organ-specific Standard Nomenclature and Diagnostic Criteria (SSNDC) guides describing and illustrating nonproliferative and proliferative lesions in mice and rats; posters presenting initial terminology proposals were first shown at the 1988 International Symposium. These multiyear projects were instrumental in providing a common set of diagnostic training materials for STP members and health authorities.
Growing recognition of STP as a reliable source for toxicologic pathology knowledge allowed the Society to increase outreach efforts to influence public policy and promote public safety. Several opportunities were pursued by the EC. For example, STP President Harris Mosher met in 1981 with members of the ACVP Council to discuss closer ACVP/STP interaction on shared endeavors. This outreach reflected the acknowledged reliance of employers in North America for ACVP-certified individuals as the bulk of toxicologic pathologists. A principal outcome of this close ACVP/STP collaboration was a shared commitment to shaping best professional practices in toxicologic pathology. In 1985, the newly constituted External Affairs Committee of STP and a parallel committee of ACVP were charged with reviewing an American Statistical Association proposal stating that coded (“blinded”) histopathologic evaluation should be the preferred approach when examining tissue sections for rodent lifetime carcinogenicity bioassays. Parallel responses in 1986 by the STP and ACVP advocated instead for informed (nonblinded) evaluation as the best practice for generating initial diagnoses with “blinded” review of findings in potential target organs (when warranted) to confirm the dose response and/or establish cutoff values (eg, no-observed-adverse-effect-level). 4,5 Importantly, the STP position paper offered detailed reasons for this stance in alignment with the fundamental goal of such bioassays. The relevance of this STP analysis and expert opinion is evident in the continued acceptance of nonblinded histopathologic evaluation followed if needed by blinded review of target organs as the default approach for use in nonclinical toxicity and carcinogenicity studies today. 6
The expanding annual STP budget reached $116 000 annually by 1987. Expenditures were concentrated on supporting the annual symposium and other educational events, but other initiatives were begun to fulfill additional STP goals. For example, the STP in collaboration with similar organizations in other regions of the world provided initial funding in 1985 to found the International Federation of Societies of Toxicologic Pathologists (IFSTP); STP member Michael Iatropoulos, MD, PhD, served as the first IFSTP Secretary–General. In 1987, a Junior Investigator Award of $400 was established to promote interest in the field. The growing success of STP resulted in a continued expansion of membership (reaching about 360 in 1986), and growing fiscal commitments led to a gradual increase in Society dues for individual members (from $25 at the start of the decade to $100 by the end) as well as increased emphasis on Sustaining membership (corporate sponsorship).
Hitting Full Stride—1991 to 2000
Possibilities for STP to impact human, animal, and environmental health enjoyed a renaissance as the millennium drew to a close. Increasingly sophisticated product classes (eg, biologics, cell and gene therapies, medical devices) and research models (eg, in vitro and ex vivo preparations, genetically modified organisms) expanded the prospects for toxicologic pathologists to participate in both discovery and development research. Simultaneously, important issues facing the profession—such as toxicologic pathology certification, fluctuating supply and demand of certified toxicologic pathologists, nomenclature harmonization, pathology best practices for histopathologic evaluation, and peer review—continued as primary considerations for the STP and its members. Dr Carl Alden’s Presidential Message in 1990 predicted that several major areas would capture the focus of the profession in the ensuing decade: “The individual who limits his/her interests and interactions to descriptive pathology service will be the technician rather than the technologist of the future.…Our challenge in the future is to help the public understand and deal rationally with issues posed by potential exposure to drugs, chemicals, and products or by consumption of foodstuffs. We may need to broaden our involvement in risk management and take a leadership role in communicating to the public.” 7 Society efforts during the 1990s ensured steady progress toward addressing such issues and enhancing intellectual opportunities for STP members. The Society’s expanding activities outgrew the ability of an all-volunteer force to fulfill essential organizational functions, so in 1993, the EC selected an experienced association management firm, Talley Management, to undertake all Society administrative needs including membership records, financial oversight, EC meetings, communications, and annual symposia support. Linda McGillicuddy served as the first STP Executive Director, and in due course, she was succeeded by Stephanie Dickinson. The Society remained with Talley until 2002.
Steady growth in member numbers during the 1990s testified to the perceived value of STP programming and service projects. Approximately 500 members were on the roll in 1991. By 1994, numbers had increased by nearly 20% to 598, most of whom were Full or Associate members; the remainder were Sustaining (corporate), Emeritus, or Honorary members (Figure 4). The change in membership from 1994 to 1995 revealed several important trends. Numbers were increasing (by 7.5%, to 643 total members with 466 Full members) even while the number of Emeritus members was also rising. Members were based primarily in North America (48% from the East Coast, 20% from the Upper Midwest, 5% from the West Coast), with just over a quarter of members located on other continents. By 1999, total STP membership stood at 754 of which 70% were employed by the pharmaceutical industry, 20% by CROs, and the remaining 10% by academic and government research laboratories, despite a due increase in 1998 (to $185 annually). Of these 754 individuals in STP, 68% were Full members, 13% were Associate members, 8% were Emeritus or Honorary members, and 3% were Sustaining members (Figure 4). An important innovation in 1999 is that the STP roster now included 8 Student members.
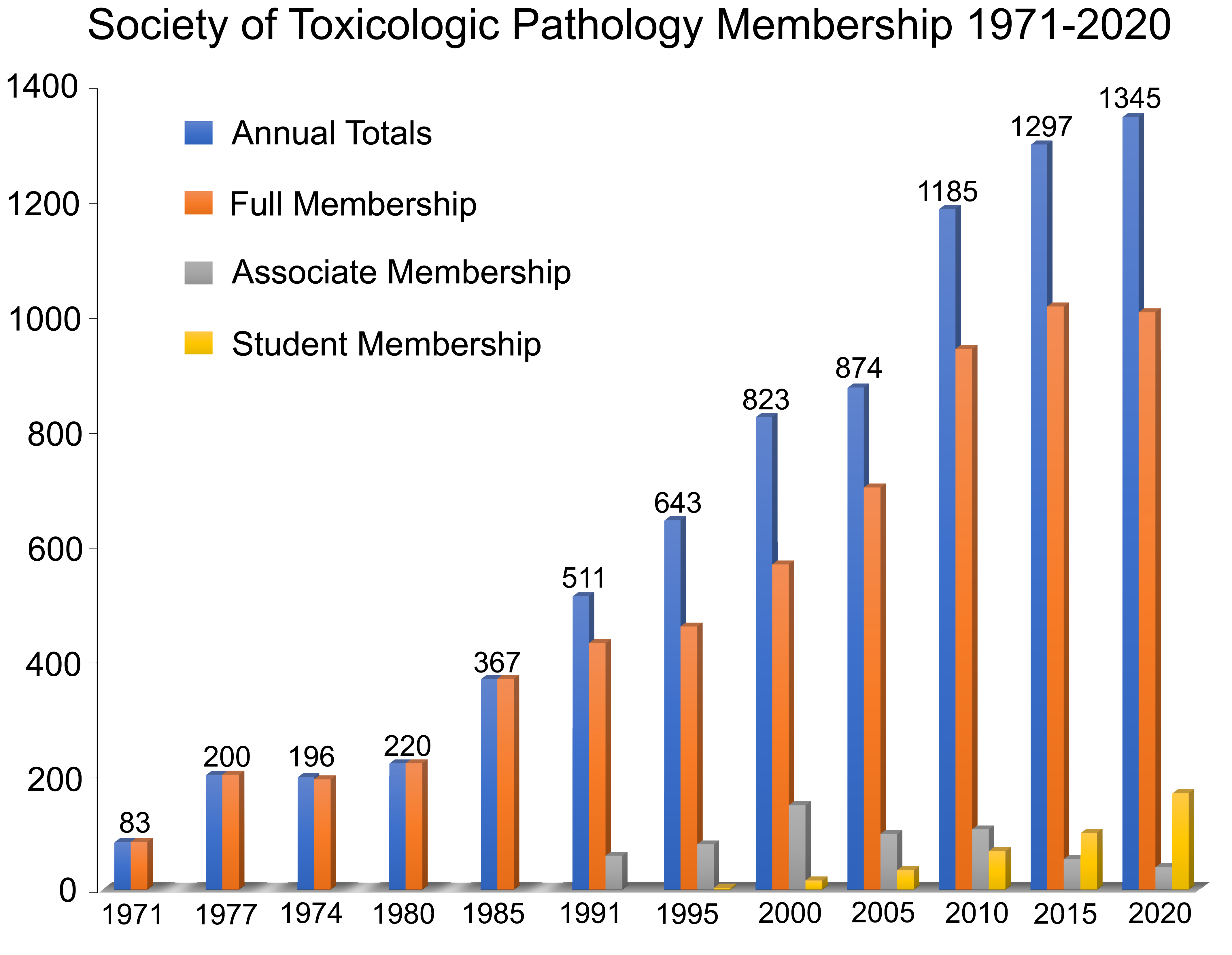
Membership trends for the society over the first 50 years (1971-2020).
Two signature STP accomplishments in the 1990s were (1) the production of a compilation of nearly 3 dozen SSNDC guides providing standardized diagnostic terminology for mouse and rat lesions in all major organs and systems, and (2) the launch of a repository of images (photographs) and data to permit reliable pathology evaluation and peer review. These initiatives were released in the early-1990s. The SSNDC guides (https://www.toxpath.org/ssndc.asp), completed in 1996, were designed to be more inclusive than contemporaneous monographs released by the International Agency for Research on Cancer (IARC) 8,9 as the STP publications also incorporated nonproliferative lesions and information on background findings while the IARC fascicles were limited to proliferative changes. Accordingly, the SSNDC documents rapidly gained in stature as essential references for toxicologic pathology practitioners. The RTPA, a core STP goal established in the Society’s constitution, was inaugurated in 1993 as a collaborative undertaking by the STP and the American Registry of Pathology. 10 The STP transferred responsibility for publishing the SSNDC guides to the RTPA in the mid-1990s. These Society efforts were supplemented by the individual initiative of several STP members to edit reference texts for mice, 11 -13 rats, 13,14 and nonrodents 15 that combined basic anatomic and physiologic information with detailed descriptions of many spontaneous and toxicant-induced lesions.
The STP was a principal contributor during the 1990s to multidisciplinary efforts to improve safety assessment and regulatory science. For example, STP members were key leaders in evaluating the biological effects of dietary restriction on animal viability, as well as the incidences and severities of chronic diseases and neoplasms during lifetime rodent carcinogenicity bioassays. 16 -19 Members of STP were also influential discussants in defining appropriate practice standards for such routine toxicologic pathology tasks as clinical pathology interpretation, 20 -22 pathology peer review, 23 -26 quantitative analysis, 27 and collection and trimming of unusual organs (eg, inner ear, 28 larynx 29 ). The Society’s dedication to collaborative science in the field was affirmed by the EC decision in 1999 to support the creation of 2 toxicologic pathology groups. The first involved a petition to the Society of Toxicology (SOT) Council for permission to institute a Toxicologic and Exploratory Pathology Specialty Section, which was granted; this long-standing section was recently redesignated as the Comparative Toxicology, Pathology, and Veterinary Specialty Section. The second endeavor, supported by the STP EC, was the creation of the International Academy of Toxicologic Pathology (IATP, https://www.iatpfellow.org/), a global organization for accreditation of experts (Fellows) by credential review. The IATP has a multinational member base that offers a worldwide resource for scientific expertise and education in toxicologic pathology.
The emphasis on ensuring a steady stream of qualified toxicologic pathologists as well as many new prospects open to the toxicologic pathology profession led the EC to launch a strategic planning initiative to better define the Society’s core roles and areas of future emphasis. The inaugural STP strategic planning session, held in the mid-1990s, emphasized that the Society’s main opportunities for serving its members, the scientific community as a whole, and the general public were to provide education in toxicologic pathology and to help influence regulatory perspectives. The educational function was essential since academic training programs in pathology are not designed to provide in-depth instruction in toxicologic pathology. The Society’s commitment to facilitate good regulatory decisions led to the formation of the STP Regulatory Affairs Committee, which later became the Scientific and Regulatory Policy Committee (SRPC). This committee communicates the viewpoint of toxicologic pathologists on technical topics (eg, appropriate collection, processing, and analysis of pathology samples 30 -35 ) or regulatory positions (eg, performance of core pathology tasks such as histopathology data collection 36,37 and peer review 38,39 ) to health authorities and international consortia (eg, the Organisation for Economic Co-operation and Development [OECD]). These communications may be initiated by the SRPC to address a perceived need or may be undertaken specifically to answer a call for public comment by the health authority. Working Groups for SRPC initiatives are drawn mainly from the global membership of STP, supplemented as needed by colleagues from other global societies of toxicologic pathology and sometimes subject matter experts from allied fields (eg, toxicologists, quality assurance professionals, regulatory reviewers).
An additional outcome of this STP strategic planning session was the recognition that a mechanism for confirming the qualifications of toxicologic pathologists was desirable. Indeed, the discussion of certification for toxicologic pathologists accelerated during the 1990s. Sustained interest in the issue was driven by the absence of formal training programs in toxicologic pathology in North America and the existence of credentialing programs in toxicologic pathology administered in other geographic regions by regional societies of toxicologic pathology. In 1990, the STP International Educational Standards Committee presented recommendations for standardizing education in toxicologic pathology across national borders to the EC. In 1992, the STP Educational Standards Committee devised a “Training Curriculum for Toxicologic Pathologists.” This proposal was shared with both the EC and the ACVP Council, which led to the formation of a joint ACVP/STP committee to investigate formalized education, training, and possible certification of toxicologic pathologists in North America. This effort was intended to produce a steady stream of well-trained, entry-level toxicologic pathologists who possessed a common base of basic and applied knowledge that would allow them to quickly assume greater responsibilities in all aspects of toxicologic pathology practice. The outcome of this joint initiative was an open forum at the 1995 ACVP annual meeting to explore the establishment of a formal subspecialty in toxicologic pathology. In 1997, the STP EC charged the Educational Standards Committee to seek ACVP Council approval for ACVP board certification candidates in anatomic or clinical pathology to tailor their examination by substituting a toxicologic pathology module (offered between 1998 and 2004) in place of an existing special species pathology module.
The STP enjoyed several significant milestones during the 1990s that confirmed the success and ongoing relevance of the organization. The Society established an online presence in 1996 with the construction of a website; the domain “toxpath.org” was chosen for STP since the name “stp.org” was already in use. In 1997, the President’s Reception at the Annual Symposium celebrated “25 Years of the STP.” In 1998, the first Continuing Education (CE) short courses—“Genetically Engineered Mice for Use in Medical Research and Safety Assessment” and “Molecular Pathology For Toxicologic Pathologists”—were held the Sunday afternoon prior to the Annual Symposium. In 1999, the website was enhanced by launching new pages on the website for career management, quarterly newsletters, and a TOXPATH listserv. Finally, the name of the organization was revised in 2000 to the STP, thus affirming that membership in the Society was open to all scientists (eg, toxicologists, regulatory reviewers, quality assurance experts) with an interest in the field. An updated list of membership criteria was devised to attract individuals with a wider array of toxicologic pathology interests (eg, toxicologists, regulatory reviewers).
The Habit of Success—2001 to 2010
The STP experienced a dramatic transformation during the 2000s, with an updated constitution and a fundamental shift in focus from primarily addressing the needs and interests of STP members to an expanded role as a global leader in toxicologic pathology education and technical standardization. In addition, a revised strategic plan (completed in 2004) was implemented, adding new goals of advocacy, collaboration, globalization, and recruitment to the traditional objectives of providing CE to Society members and providing pathology expertise to positively influence regulatory affairs; the largest change in the STP vision statement in the 2000s was the need to act globally. These new goals were approached using a number of vigorous new STP committees and societal initiatives, the outcomes of which are still in effect today.
The success of these STP endeavors was aided by the transition to a new professional management group (Association Innovation & Management [AIM]) headquartered near Washington, DC and by a substantial expansion of the Society’s website and information technology (IT) infrastructure to provide greater convenience and access of STP members to Society benefits and services. An additional driver of the greater IT capabilities during this time was a pilot project to build an STP member-only online Toxicologic Pathology Wiki, but this effort waned quickly as a result of limited Wiki input (due to heavy member workloads and corporate constraints on posting intellectual property).
Membership in STP boomed in the 2000s, rising to 874 members by the middle of the decade and 1185 by the end (Figure 4). The bulk of this increase was in the Full member category; the number of Associate members remained steady at approximately 100 throughout the 2000s. Interest among trainees was robust as indicated by an increase in Student members from fewer than 20 in 2000 to 67 in 2010. The cadre of Emeritus members rose modestly but steadily over the entire time span, reaching 69 at the end of the decade.
Perhaps the most influential STP activity during the 2000s was the regular publication of papers designed to facilitate global harmonization of toxicologic pathology nomenclature and practices with respect to regulatory-type (ie, GLP-compliant and GLP-like) nonclinical toxicity studies. Growing out of the success of the SSNDC guides, the STP played a leading role in expanding involvement in the formation of standardized nomenclature both within the Society and globally, to conform to the new strategic plan. To ensure worldwide acceptance and endorsement, the STP SRPC established a draft proposal in 2005 with the involvement of European and Japanese colleagues to revise the standard nomenclature for lesions in the rat and mouse. In collaboration with the Registry of Industrial Toxicology Animal data, the “International Harmonization of Nomenclature and Diagnostic Criteria for Lesions in Rats and Mice” (INHAND) was established in partnership with 3 other allied organizations: the British Society of Toxicological Pathology (BSTP), the European STP (ESTP), and the Japanese STP (JSTP). This effort permitted the negotiation of globally recognized terminology for nonproliferative and proliferative lesions in major organs and systems of these 2 species. The final lexicons were approved by the governing bodies of these societies, published in either the
Another influential STP activity during the 2000s was a collaboration with other toxicologic pathology organizations (eg, primarily but not solely among the STP and the BSTP, ESTP, JSTP, and/or IFSTP). For example, an international meeting in Kobe, Japan in 2004 included representatives from the JSTP (as host society), ESTP, STP, IATP, and IFSTP; this event reaffirmed the commitment of the STP to act as a global partner to promote the toxicologic pathology profession. A particular emphasis in the late 2000s was interaction with the IFSTP to explore a global pathway for the recognition of qualified toxicologic pathologists. Multilateral discussions led to an IFSTP proposal, sent to all 10 then-existing societies of toxicologic pathology around the world, that credential review might offer a suitable means for global certification of toxicologic pathology proficiency. 41,42 A vigorous debate within the STP ended in the rejection of the IFSTP credential review proposal by the STP EC, 43 but further negotiation among the 10 societies (moderated by the IFSTP) did result in a globally applicable curriculum for training toxicologic pathologists who were endorsed by all global societies. 44 Several STP members were instrumental in framing the international discussion and bringing the universally recognized curriculum to fruition.
A third essential STP activity during the 2000s was to improve the recruitment and training of future toxicologic pathologists. Specific STP efforts to address the growing need for toxicologic pathologists included expanded educational programming (funded by regular budget allocations and a 2003 capital campaign); creation of new trainee-focused awards, grants, and internships; formation of an STP Career Development and Outreach Committee (CDOC) to explain the advantages and mechanisms for beginning a career in the field; and partnership with the ACVP to offer educational opportunities in toxicologic pathology to veterinary students and pathology residents. A signature initiative to achieve this aim was the formation in 2004 of the ACVP-STP Coalition for Veterinary Pathology Fellows, whereby pathology graduate students or residents were provided with 3 years of funding for their academic program and also training experiences working on-site in a CRO or industrial toxicologic pathology laboratory. During this period, the STP also began sending complimentary copies of
Interactions between STP and various users of toxicologic pathology data were another major activity in the 2000s. Partnerships between STP and allied life science groups (eg, International Life Sciences Institute [ILSI], PhRMA–Drug Safety Subsection [DruSafe], SOT) or associated sciences (eg, Society of Quality Assurance) as well as various regulatory agencies (eg, EPA, FDA, Health Canada) were undertaken strategically to promote wider understanding by nonpathologists and regulators of unique toxicologic pathology concerns. For example, STP organizers and speakers collaborated with the American College of Toxicology (ACT) to hold the first “Pathology for Nonpathologists” course in 2004. This course was designed to introduce toxicologists to core pathology concepts including comparative anatomy and physiology, spontaneous and test article-induced lesions, anatomic and clinical pathology methods, and case studies in various organ systems. Regular publications and seminars by STP provided introductions to the pathologist’s perspective on such core concepts as the difference between interim notes and pathology raw data as well as appropriate practices for pathology data generation, reporting, and pathology peer review.
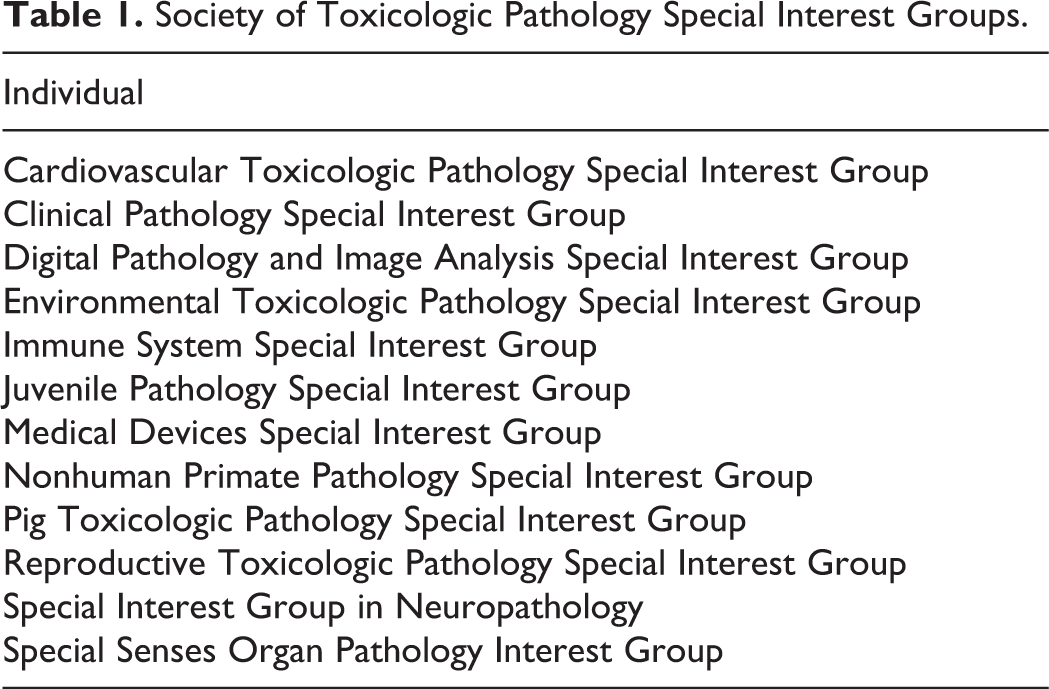
In addition to the education of other stakeholders, STP sought to enhance CE and consensus-building opportunities for STP members. The STP annual symposium served as a focal point for these efforts. In 2000, the STP symposium became an officially accredited CE activity through the American Association of Veterinary State Boards, thus allowing attendees to obtain both relevant state-of-the-art technical information and CE credits necessary to maintain state licenses. In 2002, the first satellite meeting was given as a free presymposium CE session sponsored by the US National Toxicology Program; this tradition has continued to this date. An innovation begun in the mid-2000s was the institution of the first STP Special Interest Groups (SIGs), which were designed to provide an additional forum for members with a common interest to interact (Table 1). The EC decided that SIG status would be limited to common scientific interests (eg, toxicologic pathology of an organ or species) and not demographic attributes (eg, gender, race, or geographic region). The first “Town Hall” meeting (on Peto odds ratio meta-analysis for rodent carcinogenicity studies) was held at the 2001 STP symposium. The Town Halls were designed to first introduce attendees to a topic of current interest and then permit real-time exchanges among STP members. These open forums proved to be powerful vehicles for providing member feedback to the EC and shaping Society policies and actions. Major topics discussed during the first Town Halls included best toxicologic pathology practices (eg, determining a cause of death and the use of historical control data), the approach to defining internationally harmonized diagnostic nomenclature, optimizing interactions between IFSTP and the global societies of toxicologic pathology, and the need for and means to provide a globally relevant certification of toxicologic pathologist quality. 43 In addition to the increasingly popular annual symposium, the STP implemented 1-day regional meetings in 2008 to offer even more CE options and improve professional networking; the first such event was held in Summit, NJ and focused on neuropathology. Regional meetings occurred intermittently thereafter and typically were confined to East Coast and West Coast cities with large numbers of members from pharmaceutical companies.
Society of Toxicologic Pathology Special Interest Groups.
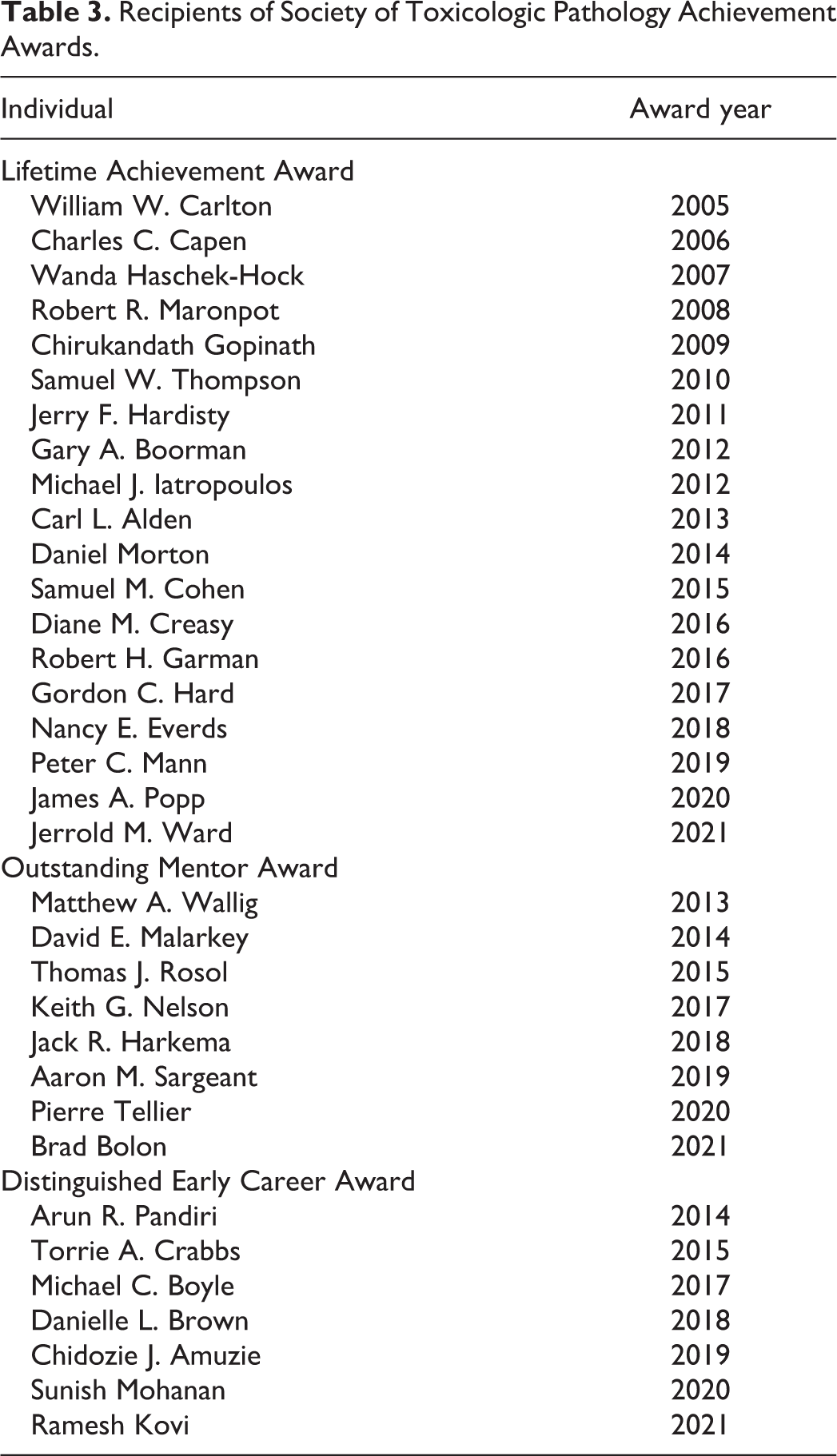
Finally, STP began to provide new opportunities for recognizing member excellence in the mid-2000s. The STP Lifetime Achievement Award was established in 2005 to celebrate an STP member who has made significant contributions to the toxicologic pathology profession over an extended (20+ years) period, engaging in such vital activities as scientific discovery, education, regulatory interactions, and/or service to the Society. The first recipient was William Carlton, DVM, PhD, a toxicologic pathologist who influenced a generation of new colleagues during a 30-year career teaching at the Purdue University School of Veterinary Medicine. The list of other STP sages who have received the Lifetime Achievement Award is shown in Table 3. Another new accolade instituted in 2005 was the STP Best Paper Award, presented to the author(s) who published the finest original research paper in
Society of Toxicologic Pathology Standing Committees.

The STP Today—2011 to 2021
The STP significantly bolstered its numbers and its educational and service activities in its fifth decade of existence. Membership in 2011 totaled 1209 individuals, including 964 Full, 76 Student, and 84 Emeritus members (Figure 4). Numbers peaked in 2016 with 1412 total members and have remained approximately constant within the range of 1300 to 1350 ever since (of which about 1000 are Full members and about 150 are Student members). The rapid increase in Student membership resulted from the investment in outreach activities by the CDOC and Membership committees during this time. Although a majority of the STP membership is based chiefly in North America, international members reside in at least 28 other countries (the top 5 of which are Canada, Japan, the United Kingdom, Switzerland, and France). The numbers of Associate members have declined each year since 2011, which coincides with the EC decision to broaden the criteria supporting an application for Full member status. The numbers of Emeritus members have risen throughout the 2010s and are now holding steady in the range of 120 to 130 individuals per year.
Member engagement occupied center stage in the STP’s fifth decade. Regular Town Hall sessions and workshops at the annual symposium offered STP members repeated opportunities to provide timely feedback on current and often controversial issues. Key topics covered in these meetings that informed subsequent STP “best practice” recommendations included global pathology peer review practices (2010) 38 ; anatomic pathology end points for recovery studies (2011) 46 ; determining, communicating, and using adverse effect data (2014) 47 ; and severity grading of histopathology findings (2017). 37 Additional topic-oriented discussions covered innovative approaches to carcinogenicity risk assessment (2012), use of INHAND terminology to fulfill the SEND (Standard for the Exchange of Nonclinical Data) requirements in FDA regulatory filings (2018), 48 and a spirited debate regarding “if, when, and how” to use coded “blinded” histopathologic evaluation in nonclinical toxicity studies (2019). 6 In addition to regular occasions for airing opinions and concerns, all STP members receive regular invitations to participate in Society activities through service on committees or as elected officers. The roster of STP SIGs expanded from 4 in 2010 to 12 in 2020 (Table 1) due to intense interest (indicated by participation of 900 members). Increased Society attention to socially responsible policies such as enhanced Animal Welfare (the “3Rs” principles [replacement, reduction, refinement]); Anti-Harassment; Diversity, Equity, and Inclusion; and Non-Discrimination were implemented to reflect cultural shifts and benefit an increasingly varied membership. An energetic online communication platform, ToxPathNet, was launched in 2010 to facilitate committee activities and afford easier access to career, learning, and networking opportunities. In 2020, the STP initiated an online presence on social networking platforms such as Facebook and LinkedIn to promote the discipline of toxicologic pathology and share ideas and common interests.
The substantial growth in STP members over the last 10 years and their generosity in serving the toxicologic pathology profession, the scientific community, and the general public have supported many new STP initiatives. The escalating range and complexity of essential functions have produced a large increase in committee numbers (Table 2). Many groups are perennial to support repetitive tasks. Key standing committees include (but are not limited to) the Annual Symposium Committee to oversee annual meeting planning and its subcommittee, the Continuing Education Subcommittee, to organize CE opportunities; the CDOC to mentor trainees, entry-level pathologists, and experienced pathologists seeking further avenues for career advancement; the Toxicologic Pathology Forum Committee to solicit expert opinions on important issues facing the profession; the SRPC to manage the STP publication efforts to communicate toxicologic pathology viewpoints to regulators and nonpathologists; and the EC to allocate the budget and integrate the activities of the other committees. In addition, ad hoc committees and task forces are assembled on a regular basis to undertake one-time tasks. Examples of recent ad hoc teams include the Scientific Program Planning Committee to assemble the symposium program and recruit speakers; various SRPC working groups, each of which is tasked with preparing a “best practice” or “points to consider” paper; the Student Outreach and Engagement Task Force; and task forces assembled to provide an STP response to a proposed regulatory position. Initially, INHAND working groups that prepared harmonized nomenclature for rodent lesions, and more recently lesions in many nonrodent species, were established as ad hoc committees, However, INHAND is now a standing committee in order to address any changes requested by members of toxicologic pathology societies or the SEND Controlled Terminology Committee under the Clinical Data Interchange Standards Consortium. The flurry of activity sustained by these committees is instrumental in STP efforts to serve as a positive influence on public health and regulatory policy. In cases where proposed regulatory guidance has international ramifications for product discovery and development with respect to toxicologic pathology work practices, the STP generally seeks to align its response with those of the other global societies of toxicologic pathology.
In 2014, the Society embarked on a regular program of direct interactions with global regulatory agencies and consortia (eg, the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use [ICH], OECD) to fulfill the Advocacy objective of the STP Strategic Plan. The goal of such interactions was to offer expertise, guidance on appropriate pathology practices, and feedback on draft regulatory guidance documents. Principal STP accomplishments in this regard include published commentary on draft guidance by FDA for biomarker qualification 49 and OECD on histopathology peer review 39 ; opinion letters (prepared by SRPC working groups and signed by the President) on FDA draft guidance on histopathology peer review, FDA data standard plans, and revisions of ICH guidelines; the STP responses were designed to attain global acceptance by the inclusion of international perspectives and coordination with parallel responses by other societies of toxicologic pathology. The STP also publicized the availability of its “best practice” and “points to consider” publications and subject matter experts to a wider audience, including regulatory agencies; prior scientific partners (ACT, ACVP, ASVCP, ILSI, SOT); and many new allied organizations (eg, American Society of Investigative Pathology, Charles Louis Davis and Samuel Wesley Thompson, DVM Foundation for the Advancement of Veterinary and Comparative Pathology, Developmental Neurotoxicology Society, Scientific Liaison Coalition [SLC], and Society for Birth Defects Research and Prevention). The continued popularity of the “Pathology for Nonpathologists” course, co-sponsored by the ACT and STP, was a signal example of successful scientific collaboration throughout this decade; the BSTP became an additional sponsor in 2015, resulting in dual delivery of the course in North America and the United Kingdom. Through these and other activities, the STP fulfills a vital function in improving technical aspects of pathology evaluations in nonclinical studies as well as the creation and application of rational regulatory policy around the globe.
Some STP collaborations were allowed to wind down in the 2010s. For example, the STP EC made the decision to withdraw from the IFSTP in 2017 to better focus educational and financial resources on activities of interest to its members. The ACVP and STP also decided to stop accepting applications for ACVP/STP Coalition for Veterinary Pathology Fellows in 2019 due to difficulties in obtaining funding for new training positions. 50 Nonetheless, 32 new pathologists were cycled through the Coalition during its existence, 51 thereby affording exposure to the profession and enriching a whole new generation of pathologists.
The STP commitment to enhanced education was showcased in the 2010s by many new achievements. In the fall of 2015, the Society participated in its first joint symposium in collaboration with the ACVP and ASVCP. This meeting was directly responsible for the peak STP membership in 2016, and the STP-sponsored scientific sessions and activities provided nearly 200 trainees with broad exposure to career opportunities in toxicologic pathology. An innovation beginning with the 2017 annual Symposium was the availability of a mobile meeting app designed to provide real-time, in-person, feedback on scientific content and additional social networking capabilities.
The menu of additional learning options was expanded to include face-to-face Modular Courses (one 2- to 3-day workshop each year on a particular scientific theme, beginning in 2013) and regional meetings (several ½- or 1-day courses per year) held at corporate or government sites or hotels located in large cities throughout North America. The Modular Courses were designed to introduce entry-level toxicologic pathologists to system-based concepts in the field, while regional meetings were organized to benefit pathologists and toxicologists at all levels of experience. Webinars (commonly 1-hour to ½-day lectures) on best pathology practices, novel product classes, or regulatory affairs began in 2012 under the care of the new Web-based Education Committee and were offered on a regular basis throughout the remainder of the decade. In 2013, an STP Speakers/Training Institution Program was initiated to link academic centers with experienced toxicologic pathologists, thereby providing a means for pathology and toxicology students and residents to obtain personalized introductions to toxicologic pathology concepts and opportunities. The STP also demonstrated an ongoing commitment to global education in toxicologic pathology by inaugurating the STP Global Scholar ToxPath Outreach Grant program in 2018. These grants provide partial support (up to USD$1500) in travel costs for STP members who are delivering educational experiences in toxicologic pathology to organizations in countries outside North America.
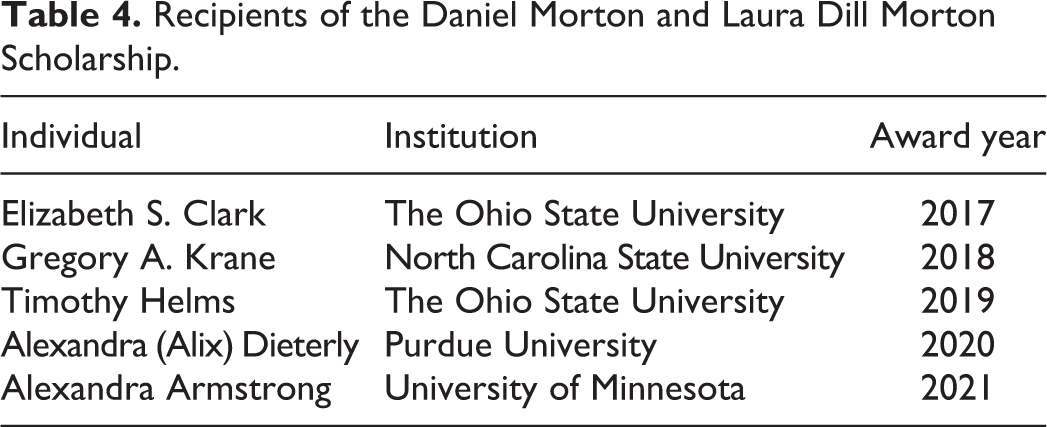
The STP proved its enduring dedication toward honoring excellence in toxicologic pathology by instituting a number of new awards during the 2010s. The Outstanding Mentor Award was inaugurated in 2013 with the intention of honoring an STP member who has significantly contributed to training toxicologic pathologists whether formally, in the academic setting, or informally as a coach during early postgraduate professional practice (Table 3). The Distinguished Early Career Award was launched in 2014 to applaud an early-career STP member with noteworthy accomplishments during the first 10 years of their toxicologic pathology career (Table 3). Multiple new merit-based awards were initiated to benefit students with an interest in the profession. The capstone prize is the Daniel Morton and Laura Dill Morton Scholarship (Table 4), an annual grant of USD$5000, begun in 2016 with funding donated by 2 longtime, devoted STP members: the late Dr Daniel Morton (an STP Past-President) and Dr Laura Dill Morton. The Scholarship is given to an outstanding student or resident with a sincere commitment and aptitude for toxicologic pathology. The IATP/STP Charles Capen Trainee Award, named in honor of the late Dr Charles Capen (an STP Past-President), has been presented since 2013 to a trainee in toxicologic pathology–related educational programs. The Capen Award comprises a USD$2000 prize plus complimentary STP annual meeting registration and STP student membership for the following year. Young Investigator Awards (first place, $750; second place, $500; third place, $250) are given to the top student poster abstracts at each annual Symposium. Other student awards that provide educational and/or travel support include the STP/SOT Comparative Toxicology, Pathology and Veterinary Specialty Section Student Travel Award (first given in 2015), the STP Environmental Toxicologic Pathology SIG Student Research Award (begun in 2019), the STP Modular Course Student Travel Grant (launched in 2016), the STP/ACVP Student Poster Award (initiated in 2013), and a large number of STP Student Travel Grants (commonly 10 or more per year).
Recipients of Society of Toxicologic Pathology Achievement Awards.
Recipients of the Daniel Morton and Laura Dill Morton Scholarship.
The close of the decade unexpectedly presented the STP—and the rest of the world—with a major challenge in the form of the global “
The History of the Society’s Journal
Shortly after its inception, the Society launched its first publications. A newsletter was distributed in February 1972, written by the first president, Dr Hans Keysser. The newsletter stated that the Publication Committee was charged with publishing a Bulletin of the SPEPs; Dr J. R.M. Innes was the first editor. The first Bulletin was published in July 1972 and contained a combination of society news, a commentary on Dr Paul Ehrlich, and a book review, with Dr Marcus Mason as the editor (Supplemental Table 2). An EC editorial on the first page of the first
The first issue of
In 1996,
At the turn of the century, the self-imposed evolution in the STP mission from a relatively “internally-focused” Society to an outward-looking organization based in education, service, and global impact led to new kinds of content in
Since 2010,
Conclusions
The STP has a rich history of scientific excellence in the discovery and development of safe products that enhance the health of humans, animals, and the world in which we live. The evolution from a small professional organization concentrated in North America to a vibrant society with a global membership and substantial international impact and relevance is validated by—among other accomplishments—(1) the many productive STP committees composed of large numbers of members who continue to voluntarily serve the Society and greater scientific community, (2) a wide and ever-expanding body of well-recognized “best practice” and “points to consider” papers on core concepts in toxicologic pathology, (3) a commitment to global education that serves the interests of both existing practitioners and the needs of the next generation, and (4) its regular and productive interactions with regulatory bodies around the globe. The resiliency of the STP in the face of COVID-19 cataclysm confirms that the Society is well positioned to continue its tradition of science in the public service for years to come. Welcome to the next 50 years.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_01926233211037934 - The Society of Toxicologic Pathology: Advances and Adventures in the First 50 Years
Supplemental Material, sj-docx-1-tpx-10.1177_01926233211037934 for The Society of Toxicologic Pathology: Advances and Adventures in the First 50 Years by Mark Hoenerhoff, Stacey Fossey, Charlotte Keenan, Agathe Bédard, Typhaine Lejeune, William Kerns, Daniel Patrick, Erin Quist and Brad Bolon in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-docx-2-tpx-10.1177_01926233211037934 - The Society of Toxicologic Pathology: Advances and Adventures in the First 50 Years
Supplemental Material, sj-docx-2-tpx-10.1177_01926233211037934 for The Society of Toxicologic Pathology: Advances and Adventures in the First 50 Years by Mark Hoenerhoff, Stacey Fossey, Charlotte Keenan, Agathe Bédard, Typhaine Lejeune, William Kerns, Daniel Patrick, Erin Quist and Brad Bolon in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-docx-3-tpx-10.1177_01926233211037934 - The Society of Toxicologic Pathology: Advances and Adventures in the First 50 Years
Supplemental Material, sj-docx-3-tpx-10.1177_01926233211037934 for The Society of Toxicologic Pathology: Advances and Adventures in the First 50 Years by Mark Hoenerhoff, Stacey Fossey, Charlotte Keenan, Agathe Bédard, Typhaine Lejeune, William Kerns, Daniel Patrick, Erin Quist and Brad Bolon in Toxicologic Pathology
Footnotes
Authors’ Note
Mark Hoenerhoff and Stacey Fossey contributed equally to organizing and completing this work. Finally, this work could not have been prepared without the significant contributions of the many Society members who served on the STP 50th Anniversary Task Force (see Supplemental Table 3 for a roster of their names).
Acknowledgments
The authors and the Society thank the Association Innovation & Management (AIM) professionals who have supported the day-to-day operations of the STP: Mr. Matt Price (Executive Director), Mr. Tierre Miller (Deputy Executive Director), Ms. Sue Pitsch (Past Executive Director), Ms. Maureen Bayley (Senior Meetings Manager), Ms. April Brewer (Program Manager), Ms. Mphatso Matenda (Senior Project Coordinator), Ms. Liz Kasabian (Exhibits Manager), Ms. Laura McCroddan (Account Manager), and Ms. Deborah O’Keefe (AIM President and Chief Executive Officer). Special thanks to Mr. William Stoeffler who has provided security services for the annual symposia and other STP-related events. The authors also extend their thanks to Ms. Stephanie Dickinson (Managing Editor) and Ms. Beth Mahler (Illustrations Editor) for their superb assistance in maintaining the excellence of the Society’s journal—including the help in optimizing the images included in this paper.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
