Abstract
Administration of pediatric anesthetics with N-methyl D-aspartate (NMDA)-receptor antagonist and/or γ-aminobutyric acid (GABA) agonist activities may result in neuronal degeneration and/or neuronal cell death in neonatal rats. Evaluating pediatric drug candidates for this potential neurotoxicity is often part of overall preclinical new drug development strategy. This specialized assessment may require dosing neonatal rats at postnatal day 7 at the peak of the brain growth spurt and evaluating brain tissue 24 to 48 hours following dosing. The need to identify methods to aid in the accurate and reproducible detection of lesions associated with this type of neurotoxic profile is paramount for meeting the changing needs of neuropathology assessment and addressing emerging challenges in the neuroscience field. We document the use of Fluoro-Jade B (FJB) staining, to be used in conjunction with standard hematoxylin and eosin staining, to detect acute neurodegeneration and neuronal cell death that can be caused by some NMDA-receptor antagonists and/or GABA agonists in the neonatal rat brain. The FJB staining is simple, specific, and sensitive and can be performed on brain specimens from the same cohort of animals utilized for standard neurotoxicity assessment, thus satisfying animal welfare recommendations with no effect on achievement of scientific and regulatory goals.
Keywords
Introduction
Administration of pediatric anesthetics with N-methyl D-aspartate (NMDA)-receptor antagonist and/or γ-aminobutyric acid (GABA) agonist activities has been known to result in accelerated neuronal degeneration and neuronal cell death in the brain of neonatal rats that may result in long-term memory and cognition deficits into adulthood. 1 –4 Although anesthetics are a well-known group of agents that can alter NMDA and GABA receptor activity, other neurotropic agents for the treatment of seizure disorders or anxiety also can alter NMDA and GABA receptor functions, including phenytoin, vigabatrin, and valproate. 5 The US Food and Drug Administration (FDA) may request that a prospective potentially neurotropic pharmaceutical product be evaluated for its potential to cause perinatal neuronal degeneration and/or cell death, especially if that product is to be indicated for the pediatric population. The authority for FDA to require these studies is provided by the Pediatric Research Equity Act of 2003 and the Best Pharmaceuticals for Children Act of 2002, both of which impact toxicologic assessments in juvenile animal studies. One accepted method to document this effect is to perform a short-term study in postnatal day (PND) 7 to 9 neonatal rats. This specialized study requires dosing neonatal rats at PND 7 and evaluating brain tissue within 1 to 2 days of this dosing regimen (typically at PND 8-9).
The use of silver stains, such as amino cupric silver stain, to aid standard hematoxylin and eosin (HE) staining in the detection of degenerating or necrotic neurons has been used to fulfill regulatory needs to assess the potential effects of this class of drugs. 6,7 However, procedural challenges of these types of staining procedures, such as the need for gelatin embedding and thick (approximately 40-80 µm) frozen sectioning, warrant the investigation of alternative methodologies that can be performed on standard 5 µm sections that are serially microtomed from the same paraffin-embedded blocks of brain tissue used for HE staining.
Fluoro-Jade B (FJB) staining can substantially aid the evaluation of neuronal degeneration and cell death when used in conjunction with HE staining in routine preclinical safety assessment studies conducted in accordance with good laboratory practices (GLP). Degenerative and/or necrotic neurons were detected consistently in a dose-dependent manner in those specific subanatomic sites of the brain recommended for evaluation as per the Society of Toxicologic Pathology (STP) guidance for assessment of neurotoxicants. 8
Materials and Methods
Fifty male and female neonatal Sprague Dawley Cr1: CD(SD) rat pups (each weighing approximately 12-19 g) at PND 7 were utilized in the study. These pups were chosen from litters of time-mated female rats that were allowed to deliver naturally. All animals (dams and pups) were housed in solid bottom cages in a facility fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International.
On PND 7, 10 pups per sex were administered 1 mg/kg MK-801 hydrogen maleate (CAS Number 77086-22-7; dizocilpine hydrogen maleate; Research Biochemicals, Inc) in 0.9% sodium chloride by intraperitoneal injection. This regimen has been shown to induce neurotoxicity in neonatal rats. 9 On PND 7, a GABA agonist test article (hereinafter “GABA agonist”) known to modulate GABA type A receptors in the brain was administered via oral gavage to 10 pups/sex/dose as a suspension in a vehicle diluent. The GABA agonist was given at a low dose, mid dose, and high dose divided in 2 doses/day at dose volume 5 mL/kg. The dose range of GABA agonist was chosen to cover a range extending from an anticipated no observed adverse effect level to a dose which should lead to some toxicity findings and was determined by prior pharmacokinetic studies. Also included was a group of 10 pups/sex that received vehicle only.
On PND 8 and at least 24 hours after the injection of MK-801 or at least 12 hours after the second injection of GABA agonist or vehicle alone, all animals were euthanized by intraperitoneal injection of sodium pentobarbital. This is the ideal time to detect neuronal death based on previous institutional validation studies to obtain maximal detection of neuronal lesions (data not shown) and is the age of rat that coincides with the peak of the postnatal brain growth spurt. 10 Upon opening the calvarium, the brain was fixed in situ in 10% neutral buffered formalin. This study was approved by an Institutional Care and Use Committee and performed in accordance with the US Department of Health and Human Services, FDA, US Code of Federal Regulations, Title 21, Part 58: GLP for Nonclinical Laboratory Studies; and complied with all applicable sections of the Final Rules of the Animal Welfare Act (CFR, Title 9), the Public Health Service Policy on Humane Care and Use of Laboratory Animals, 11 and with Guide for the Care and Use of Laboratory Animals. 12
After fixation, the brains were trimmed according to the best practices of the STP into 7 blocks of tissue, embedded in paraffin, and sectioned at 5 microns. 8 Serial sections of each block of tissue were stained with either HE or FJB. The FJB-stained slides from all animals were scanned by a Hamamatsu fluorescence slide scanner, and digital images were prepared.
Microscopic evaluation of brain tissue from all animals was conducted by a board certified anatomic veterinary pathologist, and the severity and subanatomic location of neuronal death were recorded using Paxinos and Watson. 13 A 5-point severity score of 1 to 5 (minimal, mild, moderate, marked, severe) was used to document the findings. Findings were confirmed by a second board certified anatomic veterinary pathologist during a GLP peer review.
Fluoro-Jade B Staining Method
The 5-micron formalin-fixed, paraffin-embedded slides for FJB were placed in 2 changes of xylene for 2½ minutes each and then through 4 changes of 100% ethanol for 2 minutes each. Slides were rinsed in distilled water for 1 minute and placed in a 0.06% solution of potassium permanganate (Sigma Aldrich) for 15 minutes. The FJB stock solution was made of 500 mL deionized water and 0.05 g of FJB (Histo-Chem Inc); 50 mL of this FJB stock solution was mixed with 450 mL of 0.1% acetic acid to make a 0.001% FJB working solution. Slides were stained in FJB working solution for 30 minutes in a dark room to protect slides from light. Following this incubation, the slides were rinsed in 3 changes of deionized water for 1 minute each, then dried in a 37 °C oven until fully dry, then dipped in xylene and mounted with DPX (Sigma Aldrich) mounting media. The slides were kept in a closed flat to protect from light. The FJB sections were imaged with an FITC filter cube with an excitation spectra of 485/20-25 and an emission spectra of 525/30-32.
Results
All animals survived until scheduled sacrifice on PND 8. In all animals, including untreated controls and all treated groups, evidence of physiologic neuronal cell death associated with neonatal brain development was present and characterized by scattered individual neurons with a shrunken pyknotic nucleus and condensed eosinophilic cytoplasm. These cells were visible by HE but also were fluorescent positive on the FJB stain. Approximately 1 to 2 individualized fluorescent cells in each 20× field of view were considered to represent this normal physiologic process.
Evidence of neuronal death beyond expected physiological cell loss was present in the GABA agonist and MK-801-treated groups of animals, and the data are summarized in Table 1.
Incidence and Severity of Neuronal Cell Death in the Brain.
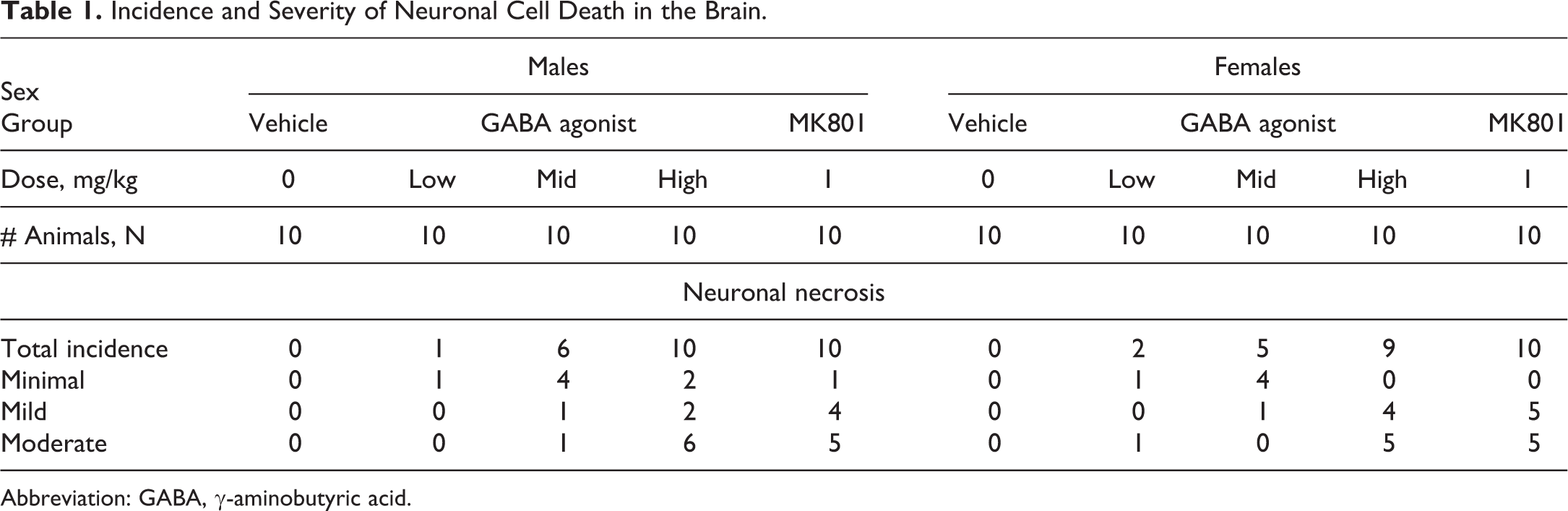
Abbreviation: GABA, γ-aminobutyric acid.
All positive control MK-801-treated males (10/10) and females (10/10) had neuronal cell death. For these, severity was mild to moderate in 19 animals and minimal in 1 female. Many of the GABA agonist-treated males and females had minimal to moderate neuronal death with a dose–response profile for incidence and severity. The nature and pattern of neuronal death for both MK-801 and GABA agonist-treated groups were similar. In HE stained sections, neuronal death was characterized by clusters of neurons with pyknotic to karyorrhectic nuclei and dense eosinophilic cytoplasm and the presence of scattered nuclear debris. In FJB-stained slides, the death appeared as clusters of fluorescent-positive granular material of variable size. By comparing serial sections of HE- and FJB-stained slides, it was readily apparent that the granular material represented fluorescent-positive dead contracted neurons often admixed with fluorescent-positive cellular debris.
The severity score for neuronal death in any subanatomic region of the brain was a qualitative determination utilizing the FJB stain and the following guidelines in Table 2. The overall brain tissue for each animal was then assigned the highest severity score present in any 1 subanatomic region after evaluating all subanatomic sites in the 7 sections of brain.
Severity Score for Neuronal Cell Death Based on Fluoro Jade B Stain in Any 1 Subanatomic Region.a

a The overall severity score for the entire brain was the highest score for any single subanatomic regions of the brain.
Neuronal death was more readily detectable with the FJB stain when compared to the HE stained tissue. With HE stained tissue, the visualization of neuronal death, whether minimal, mild, or moderate, in any subanatomic region of the brain required the use of at least a 20× objective lens. With FJB-stained tissue, areas of neuronal death were visible with a 4× objective lens. The FJB had an additional advantage over the HE stain because it helped reveal the clustering pattern of a low number of dead cells and was therefore better able to distinguish small pathologic clusters (graded minimal to mild) from widely scattered, normal physiologic cell loss.
In this study, the regions of the brain affected by neuronal death were generally similar between GABA agonist and the MK-801 treatment, with few exceptions. For both MK-801 and GABA agonist-treated animals, the most commonly and most severely affected areas included the retrosplenial granular and dysgranular regions of the cerebral cortex (Figure 1), the dorsal subiculum of the hippocampus, the dorsolateral and ventromedial regions of the thalamus (Figure 2), and the frontal/motor cortex (Figure 3). The inferior colliculi and medial geniculate were commonly affected with the GABA agonist, but not with MK-801. The full list of subanatomic sites affected is presented in Table 3 and depicted in a panel of schematics of coronal sections of the brain taken from Paxinos and Watson (Figure 4). 13 When the motor/frontal, somatosensory, or cingulate cortex was affected, the superficial most layers (layers I and II) were involved.
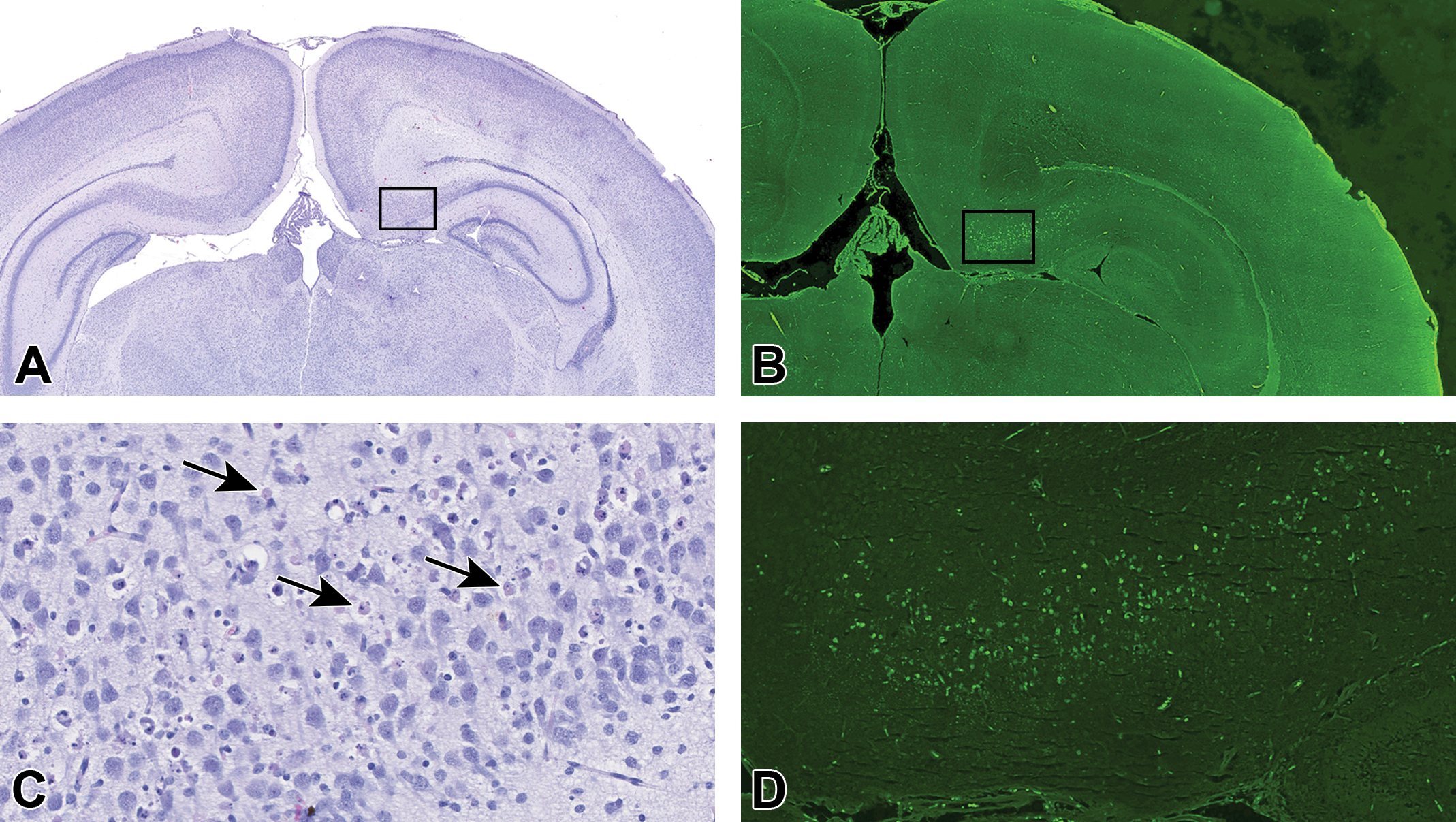
Retrosplenial granular cortex of rat treated with MK-801. A, HE (low magnification): The retrosplenial granular cortex of this animal (outlined zone) has moderate neuronal necrosis, which is not visible at low magnification (1.0×). B, FJB (low magnification): The retrosplenial granular cortex region of this animal (outlined zone) has moderate neuronal necrosis visible at low magnification (1.5×). C, HE (high magnification): The outlined zone reveals moderate neuronal necrosis/apoptosis (arrows) at higher magnification (27×). D, FJB (middle magnification): This outlined zone reveals moderate neuronal necrosis/apoptosis visible at 7.5× magnification.
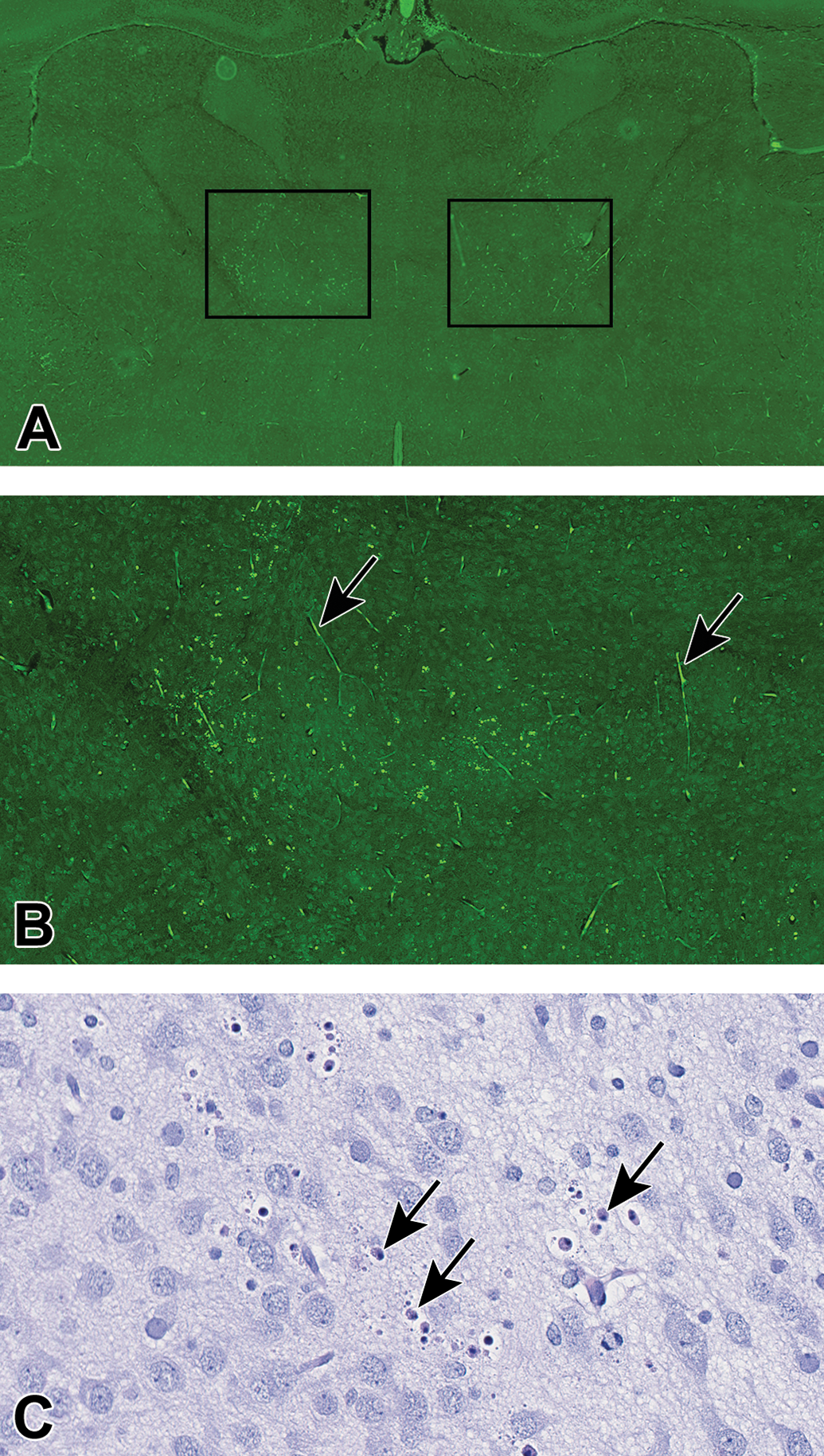
Thalamus of rat treated with MK-801. A, FJB (low magnification): The thalamus of this animal treated with MK-801 has clusters of fluorescent (necrotic) neurons in the dorsolateral and ventromedial (outlined zones) thalamus regions that can be detected at low magnification (4×). B, FJB (middle magnification): There is moderate clustering of fluorescent-positive necrotic neurons and debris apparent at 12×. Note the autofluorescent red cells within capillaries (arrows). The linear pattern of distribution of these autofluorescent red cells within capillaries of the brain parenchyma can be distinguished from the clustered pattern of necrotic neurons that result from the toxic effect. C, HE (high magnification): In the ventromedial region of the thalamus, high magnification (34×) is necessary to readily detect the necrotic neurons (arrows). FJB indicates Fluoro-Jade B.
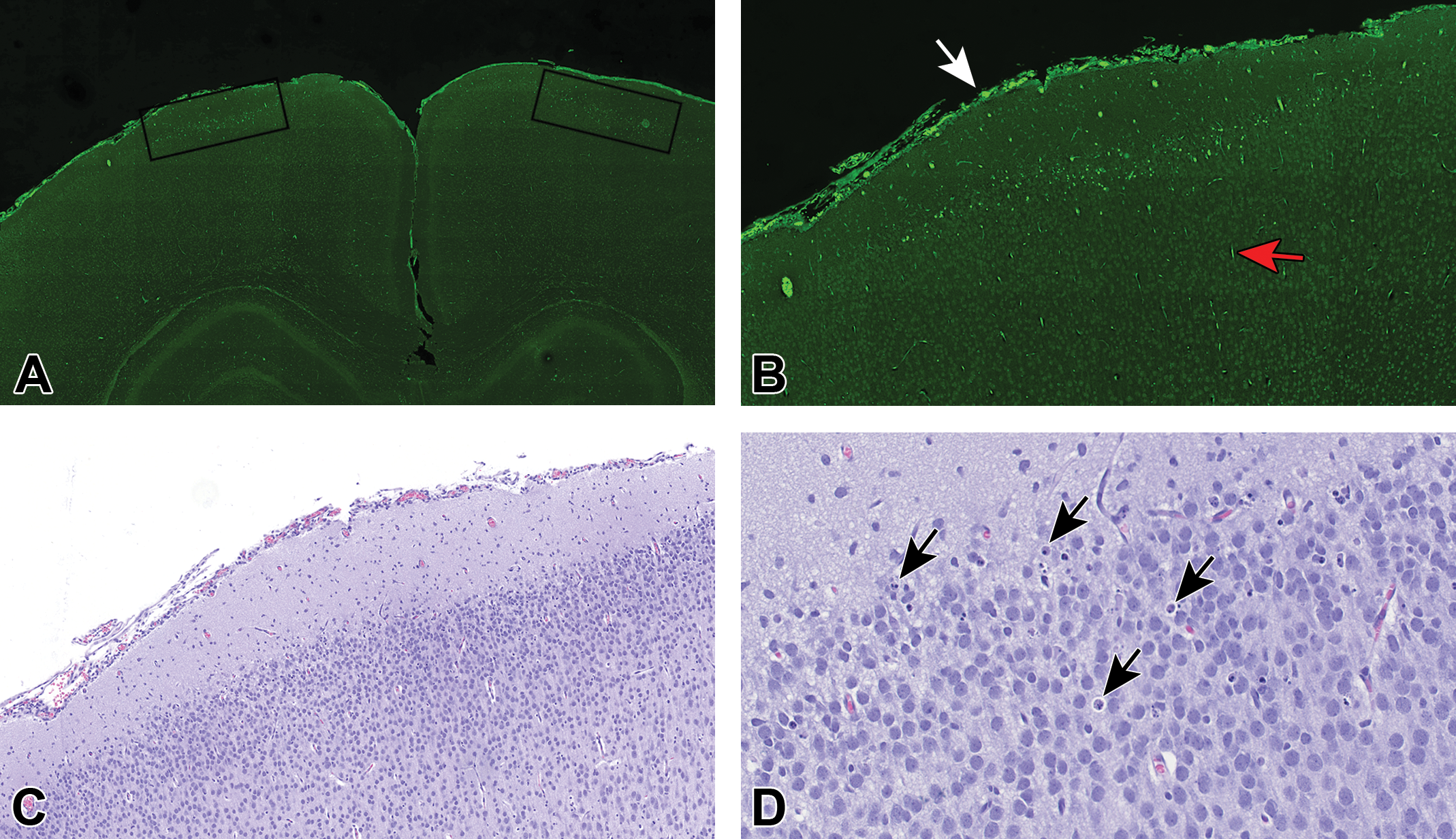
Frontal cortex of rat treated with MK-801. A, FJB (low magnification): The frontal (motor cortex) of this rat has a cluster of fluorescent neurons in the superficial levels (levels I and II) of the cerebral cortex (outlined zones). At this low magnification (3×), the clustering pattern of the necrosis can be detected. B, FJB (middle magnification): At mid magnification (9×) of the outlined zone, the cluster of necrotic neurons and cellular debris is obvious. Necrotic neurons can be readily differentiated from the autofluorescent red cells in the meninges (white arrow) and within capillaries of the parenchyma (red arrow). C, HE (middle magnification): At mid magnification (9×) of the outlined zone in panel A, the necrotic neurons and cellular debris are difficult to detect. D, HE (high magnification): High magnification of the frontal cortex (as outlined in panel A) at 30× is generally necessary to detect necrotic neurons (arrows).
Subanatomic Regions of Brain Affected by MK801 and GABA Agonist in This Study.a

Abbreviation: GABA, γ-aminobutyric acid.
a Subanatomic regions reported were limited to those subanatomic regions examined in the 7 sections of brain.
b Levels I and II affected.
c The thalamus was examined at approximately Bregma level −3.24.
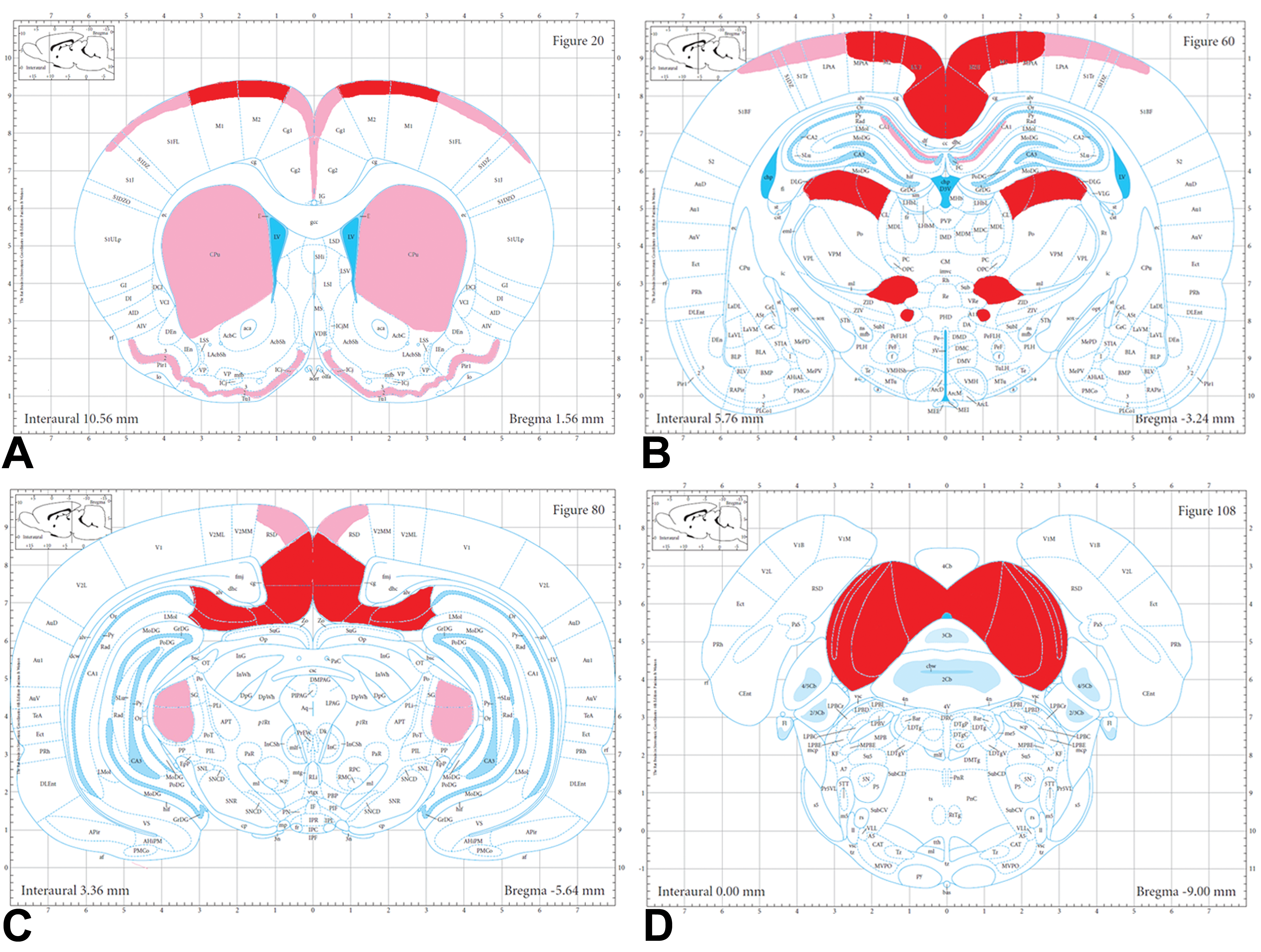
Affected areas of the rat brain by GABA agonist or MK-801 (NMDA receptor antagonist). This panel of schematics from Paxinos and Watson 13 of the adult rat brain shows those areas of the rat brain commonly affected by GABA agonists or NMDA-receptor antagonists in the context of this study. A, In the cerebral cortex from Bregma 1.56 mm, the areas affected include the superficial layers (layers I and II) of the cingulate, motor, somatosensory cortices, as well as the piriform cortex and the basal ganglia. B, At the level of the thalamus at Bregma −3.24 mm, the areas affected include the retrosplenial granular cortex; the superficial layers (levels I and II) of the motor and somatosensory cortex; the retrosplenial dysgranular cortex; and the dorsolateral and medial zones of the thalamus. The CA1 area of the hippocampus was affected by GABA agonist, but not MK-801, in this study. C, At the level of the rostral midbrain at Bregma −5.64 mm, the areas affected included the retrosplenial granular cortex as it merges with the dorsal subiculum of the hippocampus, the retrosplenial dysgranular cortex (RSD), and the medial geniculate. The medial geniculate was a target area primarily for GABA agonist. D, At the level of the caudal midbrain at Bregma −9.00 mm, the area affected included the inferior colliculi, which was more commonly and severely affected with the GABA agonist. Red—most commonly and severely affected. Pink—less commonly and less severely affected. GABA indicates γ-aminobutyric acid; NMDA, N-methyl D-aspartate.
Discussion
The ability of MK-801, an NMDA-receptor antagonist, to cause acute neurodegeneration has been known since 1987. 1 In adult rats, single doses of MK-801 produce dose-dependent vacuolation and necrosis of neurons in the posterior cingulate/retrosplenial cortex. 14,15 This vacuolar lesion, often referred to as the Olney lesion, is characterized by cytoplasmic vacuoles in the retrosplenial and posterior cingulate cortices within 4 hours of treatment. After 1 to 2 days, the vacuolation is no longer observed and these affected neurons become necrotic. 14
Since that first report by Olney et al in 1987, the acute neurodegenerative potential of specific GABA agonists and NMDA-receptor antagonists has been recognized as a possible toxic effect of anesthetic agents used on the pediatric population. 3,16 γ-Aminobutyric acid agonists include drugs such as benzodiazepines, barbiturates, propofol, etomidate, isoflurane, halothane, and alcohol. 17 Those with NMDA-receptor antagonist activity include ketamine, nitrous oxide, xenon, alcohol, and phencyclidine. 3 These anesthetics can work by 2 principal mechanisms: they increase inhibition by stimulating the GABA-A receptors (which is an inhibitory neurotransmitter early in life) or they decrease excitation by antagonizing the NMDA receptors. The pathogenesis for ketamine-induced neuronal death in young animals has been studied and is most likely as follows: blocking of the NMDA receptor (by ketamine) results in a significant reduction in extracellular concentrations of glutamate, leading to reduced physiological activation of neuronal populations, which results in decreased synaptogenesis, and eventual apoptotic degeneration of those inactive poorly synapsed neurons. 16,18 –20 A similar pathogenesis may apply to GABA agonist-induced neuronal death. That is, inhibition of a neurotransmitter like GABA during early brain development reduces physiologic activation and synaptogenesis of certain neurons, which can lead to neuronal loss of those poorly synapsed neurons. 10,21 –23
Immature brain neurons are most vulnerable to alteration in NMDA or GABA receptor function during the brain growth spurt. 10,24 In humans, the brain growth spurt starts at the beginning of the third trimester and ends several years after birth, 24 with the peak occurring between the last month of gestation and first 6 months of life. 3 In rats, the brain growth spurt runs from gestational day 20 to 21 through PND 14, with a peak occurring from PND 4 to 10. 10 Therefore, in safety assessment studies, PND 7 rats represent the middle of the peak of the brain growth spurt and are a relevant choice age for detecting toxic effects associated with test substances that alter NMDA and/or GABA receptor activity.
Evaluating brain tissue for acute neuronal necrosis is more difficult in neonatal PND 7 to 8 rats because of baseline physiological cell death that occurs at this time. 3,25 This is one of the reasons pathologists turned to additional staining methods, such as amino cupric silver, to aid in the standard (HE) assessment of degenerative and necrotic neuronal lesions. 14 Fluoro-Jade B and amino cupric silver stain all parts of dead neuronal cells similarly, including cell bodies, dendrites, and axonal processes, whether it results from physiologic processes or toxic insult caused by xenobiotics such as MK-801. 26,27 The pathologist assessment must depend on the lesion pattern to differentiate between toxic and physiologic cell death. Dead neurons that are clustered and associated with cellular debris can be considered toxic, while those that are individualized and present at low levels comparable across control animals represent normal physiologic programmed cell death present in the developing vertebrate brain. 25,28
Another challenge for the pathologist is the presence of artifactual background staining observed with these types of staining procedures. Fluoro-Jade B and the amino cupric silver stain, for that matter, both stain normal red blood cells. Red cells often occur in a linear arrangement and are a consistent size which aid in the distinction. Plus with the FJB, the pathologist can readily compare the FJB- and HE-stained serial sections should any doubt arise as to the identity of the fluorescent positive material.
Fluoro-Jade B has an advantage over HE staining, not only because the necrotic neurons can be detected at lower magnification, but also because the FJB can often help differentiated artifactual “dark neurons” from peracute degenerative neurons that initially may appear dark before acquiring the deep eosinophilic cytoplasm of the dead and red neuron. 29,30 Should there be any question as to whether a neuron with abnormal tinctorial properties on HE is an artifact or is in the initial stage of degeneration, the serially sectioned FJB may help distinguish between these 2, because generally speaking only the degenerative neuron will fluoresce. In some instances, however, artifactual dark neurons have been known to fluoresce, making the distinction between peracute degenerative neurons from the artifactually dark neurons quite difficult. The authors did not experience dark neurons of any kind, whether arising from peracute degeneration or artifact in the current study. However, proper distinction between the degenerative dark neuron and the artifactual dark neuron can be made by knowing the artifactual dark neurons often have prominent corkscrew-shaped dendritic processes and deep basophilic monomorphic cytoplasm, while the peracute degenerative neurons would likely be associated with necrotic debris or other stages of degenerative/dead neurons. Needless to say, the pathologist must take into account all such features when confronted with a cluster of dark staining neurons on HE that also fluoresce on the serial sectioned FJB-stained slide.
According to the results of this study, FJB stains are able to identify neuronal death in comparable subanatomic areas as that reported for the amino cupric silver stain or TUNEL (terminal deoxynucleotidyl transferase dUTP nick end labeling) staining when rats were exposed to NMDA-receptor antagonists or GABA agonists. 3,16 Any variation between subanatomic regions between these studies is likely due to the specific test agent, differences in dosage, the trimming procedures, and/or the subanatomic regions examined.
Fluoro-Jade B staining has several advantages over the use of silver staining methodologies to detect neuronal cell death. Silver staining methodologies (in particular, amino cupric silver staining) require significant histochemical expertise and complex embedding and processing protocols in order to obtain homogenous uniform specific staining. The amino cupric silver stain takes from 5 to 8.5 hours to complete, uses up to 7 different staining solutions, requires the use of acid washed glassware, and involves disposal of heavy metals. In contrast, FJB stain run takes slightly over 1 hour and requires the preparation of 2 solutions. The amino cupric silver stain procedure involves quite commonly gelatin embedding of rodent brain sections and collecting frozen thick (usually 40-80 µm) sections of that block. 7,14,31 Both multibrain and single-brain blocks can be created. These thick gelatin embedded blocks of brain do not align well with the recommended sectioning of paraffin-embedded brain at 5 microns for GLP neurotoxicity studies. 8 Therefore, if one is to use brain tissue for the amino cupric silver stain and other brain tissue for histopathology end points recommended by the “best practice” guidelines, 2 cohorts of animals are necessary. The “best practice” guidelines recognize that routine 5 µm sections of paraffin-embedded brain are the most suitable means for evaluating changes in cytoplasmic granularity of neurons, which underscores the importance of using paraffin-embedded sections that allow for both FJB and HE stains. Other challenges associated the amino cupric silver stain pertains to the significant variability of the background stains between studies, runs, and laboratories. Together, these aspects present diagnostic and technical challenges for preclinical assessment where validated procedures that ensure consistency/reproducibility of data are paramount. The bottom line is that the amino cupric stain is best performed by experienced technicians.
Fluoro-Jade B staining has been used in neuroscience research to detect neuronal degeneration and neuronal death in general, 32,33 and specifically when caused by NMDA-receptor antagonists 34 –36 and GABA agonists. 2,26,27 We further demonstrate here that FJB can be used in concert with HE to reveal this change on standard 5 µm sections of brain tissue prepared according to the best practices of the STP in GLP safety assessment studies without the need for 2 cohorts of animals. We conclude that FJB staining is a sensitive, technically simple, unequivocal method to identify preclinical safety risk of pediatric drugs with NMDA-receptor antagonist and/or GABA agonist activities in the neonatal rat brain.
Footnotes
Acknowledgments
The authors thanks Nicole Leibold, Pam Taylor, and Kelly Richardson for their efforts in histology and special chemistry.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
