Abstract
Re-endothelialization of vascular lumen after endovascular procedures is a critical healing milestone and is subjected to routine pathological evaluation during preclinical safety assessment of new cardiovascular devices. Gross evaluation, microscopic evaluation, and scanning electron microscopy (SEM) are the methods of choice for evaluation of vascular surfaces. In this article, we present a new digital imaging approach of surface topography herein referred to as topographical digital microscopy (TDM) that is able to meet the objectives of endovascular healing assessment in a single instrumental platform combined with the same sample preparation techniques as for histology or SEM. This platform is taking advantage of digitally managed illumination, X-Y stitching, and Z-stacking to enable direct optical imaging of tissue surfaces at levels of details ranging from the macroscopic to the cellular level. This technique is enabled by advances in digital optical microscopy and provides images in color and 3 dimensions that can help in the analysis, especially in distinguishing biologically meaningful observations from technical preparation artifacts and in visualizing surface topography.
Keywords
Introduction
Scanning electron microscopy (SEM) is the gold standard for surface evaluation of biological and biomaterial surfaces 1,2 and figures prominently in regulatory guidelines throughout the world. 3 –5 It is used for confirmatory observation of endothelial cell coverage of surfaces of interest in conjunction and in complement to detailed macroscopic observations and microscopic evaluation of histological sections. Scanning electron microscopy is the de facto go-to imaging platform to assess endothelial coverage of vascular surfaces and implanted vascular grafts. 6 –15 It offers the advantage of a very visual 3-dimensional like topographical perspective where the typical cobblestone appearance of confluent endothelial cells can be demonstrated. Alternatively, surface abnormalities indicative of incomplete healing such as presence of thrombus, adherent leukocytes, or uncovered biomaterial can also be evidenced. While the evaluation of SEM images is relatively intuitive, the technology is complex and expensive. 1,2,16
Recent advances in digital topographical optical microscopy (TDM) such as extended depth of field, rapid and integrated Z-stacking (composite digital compiling of multiple images capturing different depth of field intervals on the vertical Z axis), X-Y stitching (digital combining of multiple overlapping image fields in the X-Y plane), as well as high-dynamic range (digital enhancement of the luminosity range to enhance contrast and color) are now affording optical alternatives that rival the SEM platform for assessment of vascular and biomaterial surfaces. Direct optical imaging offers some advantages over SEM such as ease of deployment and color as well as observation of specimens in ambient conditions at magnifications ranging from the macroscopic to the microscopic level. The system also has the advantage of functioning as a digital scanning platform for microscopic preparations.
Background
Medical implants and interventional devices designed to interface with the cardiovascular system will typically affect the delicate endothelial cell layer covering vascular surfaces. Disruption of the endothelium is one of the chief causes of thrombo-embolisms. Therefore, establishing the course of endothelium healing is an essential aspect of safety assessment for these devices. 13 The time course of complete vascular healing must be well understood in preclinical animal models to predict healing in the patient population and establish the risk period during which preventative antithrombotic medication should be prescribed.
Vascular healing is evaluated in animal studies through detailed macroscopic observation at necropsy and histologically as well as through SEM analysis of the vascular surface. 13,14 Macroscopy is an indispensable step to study vascular integrity and remodeling and assess for the presence of grossly visible changes that could signal poor healing such as hemorrhage, thrombus formation, stenosis, excessive dilation, and local or downstream embolization and ischemia. Histopathology provides detailed views into the state of healing at the tissue and cellular levels from which key insights on the pathophysiology of adverse events can be derived. Scanning electron microscopy was developed in the 1960s and 1970s and the spectacular nature of the 3-dimensional images it generates has made a very big impact in the public at large and has supported tremendous advances in materials sciences and biology. 2 Specimen preparation for conventional SEM requires complete dehydration achieved chemically or through critical point drying followed by surface coating with an electro conductive layer of metal or carbon (sputter coating). 2,16 Environmental SEM operating under subatmospheric pressures and wet conditions enables observation of hydrated specimens 17 within vacuum and hence hydration limits, resulting in some resolution loss. 2,16
Scanning electron microscopy plays a specific role in safety assessment in that it is generally used as a confirmatory visual to ascertain that endothelialization has occurred. 13,14 The advantage of SEM assessment of vascular healing is that unlike histology it scans the entire surface of the experimental vascular site and can hence be used to quantify endothelial surface coverage of vessel or implant surfaces and detect problem areas. The main limitations are that being purely a surface visualization technique it provides no insight into the subjacent tissue response. Vessels must be cut open and stretched or affixed to a support for observation which can disrupts the structural integrity of the implant site unless accomplished extremely carefully. In addition, in its conventional form (high vacuum SEM or HVSEM) the terminal dehydration through chemical or critical point drying and metal sputter coating renders this preparation irreversible and therefore destructive with respect of availability of the tissue specimen for other endeavors such as histology. Osmium tetroxide postfixation of the tissue is also sometimes used to enhance surface contrast which further impedes tissue recovery for histology. Variable pressure SEM or environmental SEM (ESEM) partially overcomes some of these limitations as it does not require high levels of depressurization in the specimen chamber which preserves some of the specimen hydration and can be accomplished without sputter coating. The imaging resolution under ESEM is however appreciably lower 2 and in our experience not always diagnostic for endothelial coverage of endovascular surfaces. In addition, variable degrees of dehydration do occur while the specimen is standing in the depressurized ESEM chamber which can lead to surface artifacts such as cracks or surface exfoliations.
Scanning electron microscopy assessment of vascular surfaces is a high magnification topographic technique and for practical purposes useful magnifications range from ×10 for general topographical overview to ×500 for detailed view of surface cellular elements. Recent advances in digital optical topographic microscopy are enabling levels of magnification and resolution that rival magnifications used for biological and materials surface evaluation using SEM. In this article, we compare these technical platforms (SEM and TDM) for endovascular surface evaluation to ascertain the performance and usability of direct and digitally enabled optical technique for topographic imaging, specifically addressing vascular endothelial coverage assessment in preclinical animal studies.
Materials and Methods
A total of 4 male Yukatan minipigs (weight range from 30-45 kgs) were used in accordance with an Institutional Animal Care and Use Committee approved protocol. All procedures were performed under general anesthesia and using strict aseptic technique. Two side-wall aneurysms (one per carotid artery) were created in each animal. Approximately, a 5 cm segment of the external jugular vein was ligated and harvested to form the dome of the aneurysm. Both common carotid arteries (CCA) were then exposed and a venous pouch was attached in an end-to-side fashion. The arteries were then monitored to ensure there was no leaking at the suture site. Finally, the incision was closed.
Two weeks post aneurysm creation, each CCA was treated with a flow diverter through femoral artery access. Either a test (Test) or one of two control flow diverter (Control 1 or Control 2) was placed in the CCA.
Upon scheduled termination 30 days post diverter implantation, a limited necropsy was performed. The carotid arteries were exposed, excised, and flushed with saline, fixed in 10% neutral buffered formalin, and shipped to the histopathology laboratory for SEM evaluation. Upon receipt of the fixed specimens, the arteries were bisected parallel to the long axis, cutting through the vessel wall and diverter. The bisected vessels were processed for SEM by rinsing in 0.1 M phosphate buffer (pH 7.2), post fixation in 1% osmium tetroxide for 1 hour, dehydration in a graded series of ethanol, critical point dried, and sputter coated with a molecular layer of gold. The vessels were mounted on an aluminum stub and placed in a scanning electron microscope (JEOL JSM-IT500 HV) and examined under high vacuum.
For TDM evaluation, the vessels previously prepared for SEM (critical point dried and sputter coated) were placed on the stage of a Keyence VHX7000 digital microscope and examined directly in ambient conditions. For direct comparison, fields chosen for SEM were matched and imaged at similar magnification levels using the Keyence optical system (at a minimum, one field per vessel).
Scanning electron microscopy and TDM images were scored for endothelial cell coverage (Table 1) and presence of surface fibrin, thrombus, or adherent leukocytes (Table 2) by a board-certified veterinary pathologist. Surface artifacts were also recorded. Observed morphologies were scored in a semiquantitative fashion described below.
Scoring Definitions for Endothelialization.

Scoring Definitions for All Other Morphologies.

The tabulated data are presented with supplemental materials available online.
Study Limitations
The specimens evaluated were representative of chronic postimplantation observation periods when the endothelial surface is expected to be healed and completely endothelialized. There were no vessels lacking endothelial coverage other than caused by postmortem erosion (handling artifact) and there were no pathological features on the vascular surfaces examined such as thrombus, postmortem clot, or plaque. While it will be critical to assess TDM on a variety of conditions, we do not consider these limitations to be critical to the objectives of this publication because where endothelial cells were artifactually eroded, this was visible with both imaging platforms. Regarding visualization of pathology such as thrombosis or plaque, the TDM platform offers the advantage of supporting a 3D evaluation of the wet fresh or fixed vascular surface at high magnification and in color which can be an advantage over the SEM platform alone which is in grayscale. The lag in fine cellular detail discussed below for TDM should be compensated by the benefits derived from color interpretation.
Results
Under both imaging platforms (SEM and TDM) and based on the images and as recorded in the tabulated scores (score 4) for endothelialization for both the SEM and TDM analyses (given in the Supplemental Materials), the carotid arteries showed full coverage of the neck and diverter stent at 30 days by a thin layer of tissue that was diffusely endothelialized (score 4 endothelialization). Endothelial coverage was evident by SEM from the typical cobble stone appearance of the surface, denoting the regular arrangement of confluent endothelial cell nuclei (Figures 1A, C, E and 2A, C). Topographical digitalmicroscopy images displayed very similar features: the regular distribution of bulging nuclei as well as cobble stone appearance was evident (Figures 1B, 1D, 1F and 2B, D). There were occasional red blood cells, and platelets (Figure 3A) found on SEM images which were also visible albeit not as evident on TDM images (Figure 3B). Fibrin or thrombus were not observed on SEM and TDM images.

Side-by-side illustration of representative SEM and TDM images. The SEM montage (A) and low magnification (×20) TDM image (B) are very similar except for the gold sputter coating color in the TDM image; the black rectangles are the areas detailed in C-F. The thin tissue coverage of the stent diverter is visible in both imaging modes (C, SEM ×100) and (D, TDM ×300) endothelial coverage is apparent and confirmed at high magnification (E, SEM ×500) and (F, TDM ×1000) as evidenced by the regular cobblestone pattern of confluent endothelial cells. There are small surface cracks (arrowheads, black and green in SEM and TDM images, respectively) visible with both techniques as well as cellular debris that is more evident with SEM (E, black arrow) compared to TDM (F, green arrow) where the sloughed cells are visible in a golden-brown hue slightly darker against the gold endothelial background. SEM indicates scanning electron microscopy; TDM, topographical optical microscopy.

Comparative illustration of surface artifacts as shown with SEM (gray images A montage and C, ×250) and with TDM (golden images B, ×500; D, ×400 and E, ×400). Terminal endothelial erosions are seen on SEM (black arrows in A); the same erosions are clearly visible under TDM (black arrows in B). The SEM image A shows a particle at the lower left corner (black arrowhead in A) that appears black in the TDM view (B) and can be discounted as particulate that deposited after sputter coating. A white particle (green arrowhead in B) is also visible on the TDM image and not present in SEM image A indicating that it appeared post sputter coating (no gold coating) and represents dust that sedimented on the specimen between SEM and TDM steps. C and D illustrate erosions that occurred terminally prior to sputter coating (green ovals) and cracks (green arrows) that show the grayish appearance of the uncoated tissue on SEM indicating that they opened in the tissue after coating and are therefore preparation or handling artifacts. Image E illustrate the ability to manipulate the viewing perspective through 3D software on the TDM viewing platform. SEM indicates scanning electron microscopy; TDM, topographical digital microscopy

Surface and cellular details with SEM (A, ×500) and TDM (B, ×1000). The SEM field in A shows partial endothelial cell coverage in the left of the image (white arrow) whereas the area at right is largely devoid of cellular coverage and shows the underlying exposed collagen matrix of the intima (black asterisk in SEM image A and white asterisk in TOM image B). Some cellular details, namely adherent red blood cells (green oval) are somewhat less evident under TDM (B) than SEM (A), due to reflection or glaring artifacts from the gold layer. Smaller particles possibly consistent with adherent platelets (green arrows) are also visible with both techniques although more evident under SEM (A) compared to TDM (B). SEM indicates scanning electron microscopy; TDM, topographical digital microscopy.
There were a number of routine preparation artifacts recorded in SEM images such as localized foci and areas lacking endothelial coverage (Figure 2A, 2C, 3A). Based on the torn appearance of the edges and the absence of fibrin or platelet deposition on the exposed subintimal collagen, these features were interpreted as postmortem erosions associated with tissue handling (erosions) or were due to processing namely dehydration cracks in the surface (Figure 2C). These artifacts were also visible on TDM images (Figures 2B, 2D, 2E, 3B).
Discussion
Scanning electron microscopy currently stands as the method of choice for assessment of endothelial surfaces and for evaluation of biomaterials alterations after ex-vivo or in-vivo testing. 3 - 5 Other complementary techniques have been utilized such as selective immunodetection of endothelial cells using en-face submicroscopic confocal fluorescence detection of CD31 or Von Willebrand associated antigen. 10,18 These techniques are indirect, technically challenging to execute routinely in a controlled manner and are best used as a research tool.
Direct imaging of live or fixed biological surfaces at the cellular level is challenging due to optical properties of hydrated cells, namely transparency, diffraction, or reflectivity. Live cells can be imaged directly using phase contrast. 19 Phase contrast imaging is limited to single cell layer and not suitable for whole tissue mounts. Optical imaging of surface features (topography) of wet tissues requires an opaque surface. This can be accomplished through critical point drying and sputter coating. At the time of this publication, our testing was limited to processes that can support SEM evaluation since this remains the industry standard for evaluation of biological surfaces. We have tested the TDM platform on specimen coated with gold which is often favored for SEM due to its stability and highly conductive properties. Under direct illumination gold has the inconvenience of being highly reflective which challenges 3D reconstruction algorithms at very high magnifications, even in the high dynamic range mode (HDR or “pseudo-SEM” mode). Polarization can reduce reflectivity and improve detail reconstruction but hyper reflectivity is at this point the main limitation of the technique and is likely responsible for the lag in fine detail resolution at the highest magnification levels (×300-×500) relative to HVSEM. This current limitation makes it more difficult to positively identify small objects (2-10 microns) such as platelets or red blood cells. However, we are finding that low- and mid-magnification ranges (×20-×250) produce images that rival HVSEM and are diagnostic for surface integrity and endothelial cell coverage, showing the characteristic cobblestone distribution of endothelial cell nuclei, sometimes more prominently than under HVSEM.
There are inherent advantages to a direct optical technique such as TDM compared to SEM, namely the ability of the TDM platform to faithfully record colors. With gold coated specimens this helps identify artifacts that occur after the coating process such as dehydration or tissue handling surface cracks or environmental particulate that may deposit on the specimen surface. These artifacts will characteristically not show gold coating and appear in shades of gray for the tissue and in their natural color for particulate deposits (Figure 2). Other coatings would presumably afford the same benefit. Additionally, imaging of the fresh or fixed wet tissue specimen prior to processing and sputter coating provides highly detailed and 3D images of the vascular surface with characteristic color and texture features that help a trained pathologist recognize abnormalities such as thrombi, postmortem clots or tissue healing with a much greater level of detail and confidence, even revealing to the trained eye the characteristic cobblestone appearance of endothelial cell coverage (Figure 4). The digital platform is also fitted with a very sophisticated measurement toolbox that can generate valuable quantitative data such as thrombus sizes (surfaces, volumes) or surface profiles in 3D (Figure 5) which can help document fine surface details important to rheology (characterization of blood flow pattern having direct implications for thrombogenesis, vascular remodeling, and more generally for vascular pathology).
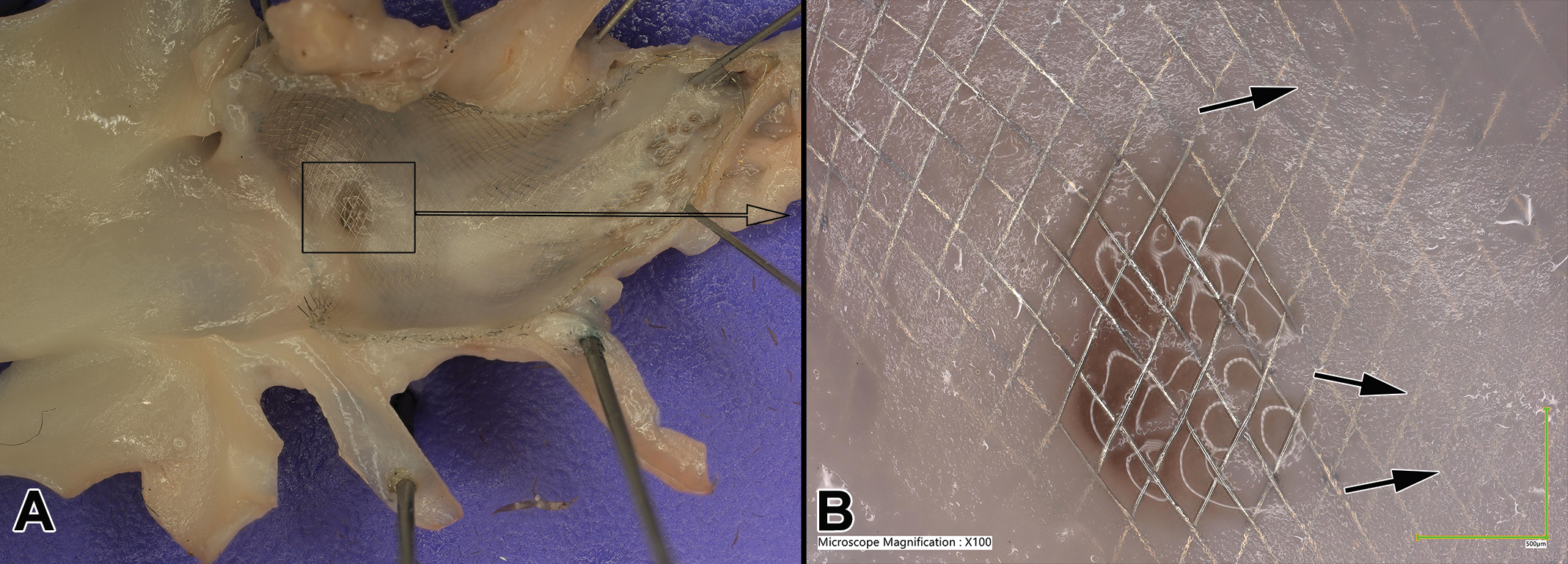
Highly detailed optical (TDM) imaging of a complex vascular surface containing a stent diverter (A: ×20). At high power (B: ×100) the area within the black box can be viewed at a level of resolution that enables the trained eye to detect endothelial cell coverage (arrows show the characteristic cobblestone appearance of endothelial cell nuclei at the surface of the neointima covering the stent). TDM indicates topographical optical digital microscopy.
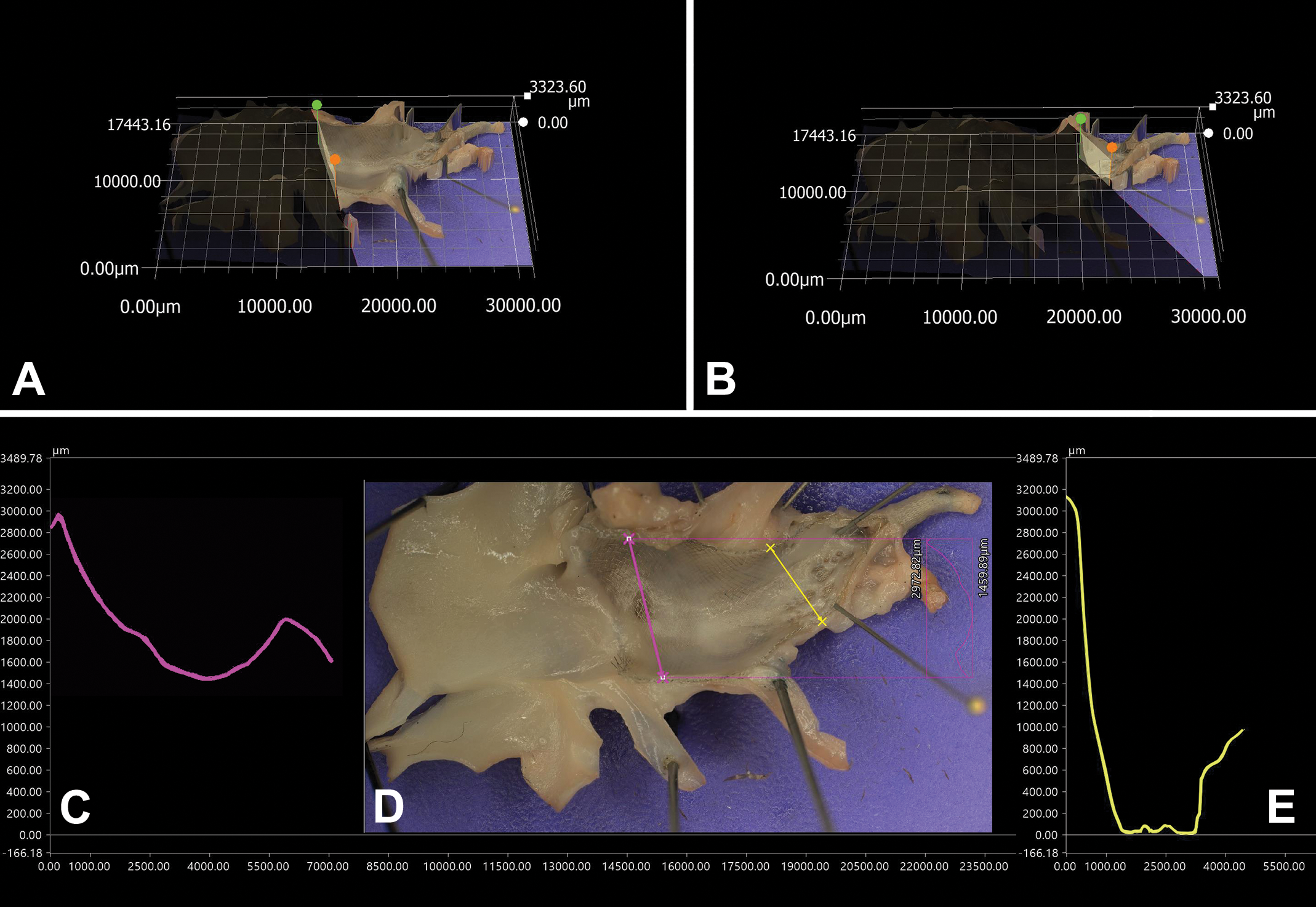
Three-dimensional macroscopy and topography. The digital microscope constructs a 3D map of the specimen surface enabling virtual sectioning of the surface (magenta and yellow lines (D), shown in 3D (A and B) with calibrated profile generation (D and E).
Relative to HVSEM, TDM constitutes a practical solution that is less capital intensive and does not require specific environmental conditions. In addition, the topographic microscope is very versatile and supports extremely well high-power 3D microscopic evaluation of biomaterials given that it was originally designed for inspection of small manufactured components such as microchips. We have also found that it can be used very effectively to produce high-quality images and digital scans of microscopic slides in bright field. The magnifications that are practical for vascular TDM evaluation (up to ×500-×1000) do not stretch the microscope capabilities as it is available with lenses that deliver in excess of ×2000 magnification, the limiting factor being the optical characteristic of the surfaces examined (reflectivity, transparency, etc). Because it has an open specimen stage and operates in ambient condition, it can be used to image live specimen or specimen placed in a clear fluid which can sometimes solve some issues with surface glare and reflectivity or to image live cells or tissues. The digitally controlled motorized staged is also fully capable of scanning microscopic slides under brightfield microscopy and the rapid Z-stacking tool makes it one of the most capable platforms to digitally scan thick ground and micropolished plastic sections which are standard for many medical device preparations.
Focused ion beam electron microscopy (FIB-SEM) is a technological advance that is well suited to topographical and 3D analysis of biological structures at very high resolution (<10 nm) 20 at the subcellular level (nanostructure and intracellular architecture). The ion beam is inherently destructive (gradual milling through the material, exposing new layers for imaging). Furthermore, specimen size is limited to a few hundred microns in any one axis 21 and therefore this exciting technology is not currently applicable to evaluation of endovascular surfaces at the supracellular level.
Conclusion
Recent advances in computerized Z-stacking with high magnification optical lenses are opening new capabilities in optical topographic digital microscopy (TDM). The ability to also very quickly stitch x-y fields in combination with fast Z-stacking under various lighting conditions make it possible to very quickly generate wide fields of view in 3D and under a variety of light conditions, providing levels of details that rival what can be accomplished using SEM. We have found that specimens prepared for HVSEM can be effectively imaged under this new TDM platform supporting assessment of endovascular surface healing, namely, to demonstrate confluent coverage by endothelial cells. It will be important to continue to stress-test this platform under various conditions to demonstrate that the range of changes expected on vascular surfaces such as thrombus/fibrin deposition can be effectively detected. Additionally, we will continue to test various specimen preparation methods and lighting conditions (eg, polarization) to minimize surface reflectivity and improve detail resolution to expand upon capabilities with this platform that affords many advantages in terms of ease of use, versatility, and deployment costs.
Supplemental Material
Supplemental Material, sj-xls-1-tpx-10.1177_0192623320979908 - Digital 3D Topographic Microscopy: Bridging the Gaps Between Macroscopy, Microscopy and Scanning Electron Microscopy
Supplemental Material, sj-xls-1-tpx-10.1177_0192623320979908 for Digital 3D Topographic Microscopy: Bridging the Gaps Between Macroscopy, Microscopy and Scanning Electron Microscopy by Serge D. Rousselle in Toxicologic Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SDR is employed by a contract research organization that provides services for safety assessment of drugs and medical devices.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
