Abstract
The European Society of Toxicologic Pathology organized an expert workshop in May 2018 to address adversity considerations related to thyroid follicular cell hypertrophy and/or hyperplasia (FCHH), which is a common finding in nonclinical toxicity studies that can have important implications for risk assessment of pharmaceuticals, food additives, and environmental chemicals. The broad goal of the workshop was to facilitate better alignment in toxicologic pathology and regulatory sciences on how to determine adversity of FCHH. Key objectives were to describe common mechanisms leading to thyroid FCHH and potential functional consequences; provide working criteria to assess adversity of FCHH in context of associated findings; and describe additional methods and experimental data that may influence adversity determinations. The workshop panel was comprised of representatives from the European Union, Japan, and the United States. Participants shared case examples illustrating issues related to adversity assessments of thyroid changes. Provided here are summary discussions, key case presentations, and panel recommendations. This information should increase consistency in the interpretation of adverse changes in the thyroid based on pathology findings in nonclinical toxicity studies, help integrate new types of biomarker data into the review process, and facilitate a more systematic approach to communicating adversity determinations in toxicology reports.
Introduction
Thyroid hormones (THs) thyroxine (T4) and 3,5,3′-triiodothyronine (T3) regulate diverse biological processes, from early growth and development to key metabolic and bioenergetic functions. Potential disruption of thyroid gland function thus represents an important human health concern in the safety evaluation of drugs, chemicals, and other regulated substances. Accordingly, health regulatory agencies require assessment of thyroid gland morphology and TH concentrations in the blood as part of various nonclinical testing guidelines. Studies with thyroid endpoints range from standard repeat-dose toxicity studies in adult animals to developmental bioassays. One of the most common thyroid effects observed in these studies is follicular cell hypertrophy and/or hyperplasia (FCHH). Although this finding generally indicates activation of the hypothalamic-pituitary-thyroid (HPT) axis, it may result from a number of distinct mechanistic pathways. These pathways represent both thyroidal and extrathyroidal mechanisms, operate in different life stages, result in different health outcomes, and exhibit important qualitative and quantitative differences across species. This complexity has led to inconsistent interpretations of FCHH, with potentially important regulatory implications. In one context, FCHH may be considered an indicator of altered thyroid gland function or capacity, while in another it may be viewed as a normal physiologic compensatory response to maintain HPT homeostasis.
To help address this issue, the European Society of Toxicologic Pathology (ESTP) organized an international expert workshop in Berlin, Germany, in May 2018. This event built on the foundation of prior adversity working groups coordinated by the ESTP 1 and the US Society of Toxicologic Pathology (STP), 2 which focused on defining and communicating adversity in nonclinical toxicity studies. The 2018 ESTP thyroid workshop included a 10-month preparatory phase consisting of presentations and discussions by the expert group. Participants included pathologists, toxicologists, and research and regulatory scientists from Europe, Japan, and the United States. Experts represented several different sectors, including pharmaceutical and agrochemical industries, contract research organizations, academic research laboratories, and regulatory affairs for drug, food, and chemical safety. These panelists were invited by the workshop chairs and the ESTP board based on their expertise in thyroid gland biology, pathology, and toxicology.
The purpose of the workshop was to increase alignment among toxicologic pathologists and regulatory scientists on how to characterize and determine adversity of thyroid FCHH. Specific aims were to: identify common pathways/mechanisms leading to thyroid FCHH; provide criteria and consensus recommendations (where possible) for characterizing adversity of thyroid FCHH; and describe mechanistic data/assays that may provide key context for adversity determinations related to thyroid FCHH.
The workshop was not intended to provide comprehensive coverage of thyroid gland biology, but rather to focus specifically on information relevant to interpreting FCHH in nonclinical toxicity studies. For example, thyroid cancer may influence the interpretation of FCHH, as discussed herein, but our goal was not to provide an in-depth review of thyroid carcinogenesis and its assessment. Similarly, the issue of HPT disruption was discussed with respect to thyroid FCHH, but the broader issues surrounding designation of chemicals as endocrine disruptors (ED), effects of THs on peripheral organs, as well as the topic of neurodevelopmental toxicity as a result of HPT disruption were considered out of scope for this publication.
The following summary document captures major discussion points, interpretive issues, panel recommendations, and areas of disagreement among workshop participants. The ultimate aims of this information are to increase consistency in the interpretation of thyroid pathology findings in nonclinical toxicity studies, identify key types of biomarker data that may add value to the decision-making process, and facilitate a more systematic approach to communicating thyroid adversity calls in regulatory reports. This manuscript has been reviewed and endorsed by major toxicologic pathology organizations, including the ESTP, the STP, the British STP, the Dutch STP, the French STP, the Japanese STP, the Latin American Society of Toxicologic and Experimental Pathology, and the International Academy of Toxicologic Pathology. See Supplemental Table 1 for a list of abbreviations.
Background Considerations: Thyroid Pathophysiology and Comparative Biology
Thyroid Hormone Biosynthesis
The thyroid gland is the largest organ in the body with exclusive endocrine function. The basic structure of the thyroid is unique, consisting of variably sized follicles with intraluminal colloid produced by follicular cells (FCs). The biosynthesis of TH is also unique in that the final assembly occurs extracellularly within the colloid. Thyroid FC trap iodide (and other essential raw materials) from the plasma and transport it to the lumen, where it is oxidized to iodine (I2) by thyroid peroxidase (TPO). Thyroglobulin, a large homodimeric glycoprotein, is synthesized in FCs and released into the colloid. 3 Thyroid peroxidase also catalyzes the linking of iodine to tyrosyl residues in thyroglobulin as well as their combination to yield the monoiodotyrosine (MIT) and diiodotyrosine (DIT) intermediates, which combine to form the 2 biologically active iodothyronines, T4 and T3. Thyroid stimulating hormone (TSH) from the pituitary gland stimulates multiple processes to increase TH synthesis and secretion including endocytosis of colloid proteins by FCs. The colloid droplets fuse with lysosomes, and enzymes cleave T3 and T4 from thyroglobulin. Cytoplasmic THs are then exported by the transmembrane transporter, monocarboxylate transporter 8, across the basolateral FC membrane and enter the systemic circulation. A key regulator of TH synthesis is the sodium-iodide symporter (NIS), an integral protein in the basolateral membrane of thyroid FCs that actively imports iodide against a concentration gradient. Chemicals such as perchlorate and thiocyanate can selectively inhibit the NIS and active iodide transport, effectively blocking the ability of the gland to synthesize TH. Iodotyrosine dehalogenase also regulates TH synthesis by removing iodide from MIT and DIT residues in FCs. The iodide is then recycled to the follicle lumen to iodinate new tyrosyl residues or released into circulation.
Thyroid Hormone Function
The majority of TH effects are mediated by T3, which is the active endogenous metabolite of T4 and is the result of enzymatic monodeiodination. The bioactivation of T3 is a peripheral activity depending on the amount and functionality of the converting enzyme in specific peripheral tissues.
Thyroid hormone can act on many different target cells. General effects include (1) increased basal metabolic rate; (2) increased glycolysis, gluconeogenesis, and glucose absorption from the intestine; (3) stimulation of new protein synthesis and turnover; (4) increased lipid metabolism and conversion of cholesterol into bile acids and other substances, activation of lipoprotein lipase, and increased sensitivity of adipose tissue to lipolysis by other hormones; (5) stimulation of heart rate, cardiac output, and blood flow; and (6) increased neural transmission, cerebration, and neuronal development in young animals. Under certain conditions (eg, carbohydrate starvation, in neonatal animals, liver and kidney disease, severe illness), T4 is preferentially monodeiodinated to 3,3′,5′-triiodothyronine (reverse T3) instead of T3 and reverse T3 concentration in the blood increases. This is due to preferential expression of deiodinase 3 and decreased activity of deiodinase 1 (DIO1). Since reverse T3 is biologically inactive, it provides a mechanism to modulate the metabolic effects of TH.
Thyroid Hormone Regulation and Clearance
Negative feedback control of TH secretion is coordinated by the anterior pituitary and certain hypothalamic nuclei, which respond to circulating T3 and T4 concentrations. Thyroxine must be deiodinated to T3 by intracellular deiodinases prior to TH receptor (THR) binding. Secretory neurons in the hypothalamus respond to decreases in plasma TH concentration by synthesizing and releasing thyrotropin-releasing hormone (TRH). Thyrotropin-releasing hormone binds to receptors on pituitary thyrotropes (basophils), leading to the release of TSH, stored in TSH-containing secretory granules of the anterior pituitary, into systemic circulation. Binding of TSH to FCs increases the rate of T4 and T3 synthesis and secretion. If the secretion of TSH is sustained (hours or days), thyroid FCs increase in size and become more columnar (hypertrophic) and follicular lumens reduce in size, due to the increased processing of colloid. Conversely, increased circulating levels of T3 and T4 result in lower circulating TSH. The expression of both the TRH gene and the 2 separate genes for the TSH α and β subunits are repressed by binding of T3 to THRs. Decreased TSH leads to enlarged thyroid follicles due to an accumulation of colloid. Follicular cells lining these follicles are flattened or low cuboidal in shape.
Thyroid hormones are inactivated either by deiodination or hepatic conjugation by phenol sulfotransferase and UDP-glucuronosyltransferase (UGT) and excreted in the bile. Side chain deamination and decarboxylation and cleavage of an ether linkage are relatively minor pathways of degradation (species differences will be outlined below). A wide variety of drugs and chemicals can influence TH metabolism by inducing one or more classes of hepatic microsomal enzymes that increase degradation of T3 and T4. The stepwise monodeiodination of T4 in the liver, kidney, and elsewhere is also important in the metabolism of TH. 4
Species Differences in Thyroid Responses
Impairment of TH economy by various xenobiotics or physiologic alterations (eg, iodine deficiency) typically results in increased blood concentrations of TSH. In the rats, increased TSH causes diffuse FCHH, and, with sufficient time, an increased incidence of adenomas and carcinomas (discussed further below). Male rats are particularly susceptible to these changes. For example, in a review of rodent carcinogenicity studies previously submitted to the US Environmental Protection Agency (US EPA), 11% (35/307) of chemicals resulted in thyroid tumors in male rats compared to 4% (13/307) in females. 5 In rats, a sexual difference is also observed with respect to TSH levels, which are greater in males compared to females (historical control data BASF, Crl: WI (Han) rats: 3.3-14.4 µg/L in males and 3.1-8.0 µg/L in females [all studies were performed in compliance with animal welfare regulations]). Mice can exhibit a similar range of FC responses but are generally less susceptible to disruption of TH economy compared to rats, 6,7 although in some cases mice do appear to be more sensitive than rats. 7
Although the general function of TH is well-conserved in mammals, there are important species differences in the metabolism and response of the thyroid to chronic variations in TSH, which limits the value of a direct comparison of thyroid gland parameters between laboratory animals and humans. One important difference is the plasma half-life of TH that varies widely in different mammalian species. For example, the half-life of T4 is much shorter in rats (12-24 hours) compared to humans (5-9 days) due to rapid metabolism and excretion. In humans and nonhuman primates, circulating T4 is bound primarily to thyroxine-binding globulin (TBG), but this high-affinity binding protein is only present in low levels in rats 8 and has only 9% of the serum binding capacity as that in humans. 9 Additionally, the binding affinity of human TBG for T4 is approximately 1000 times greater than that of transthyretin, which is an abundant, high capacity, low affinity T4-binding protein in mammals but highly conserved across all species. As expected, the percentage of unbound active T4 is lower in species with high TBG concentrations compared to those where T4 binds predominantly to albumin, transthyretin, and some lipoproteins. In general, T3 binds with less affinity to transport proteins compared to T4, which results in a faster turnover and shorter plasma half-life in most species. Another potential species difference relates to the metabolism and clearance of TH. In humans, inactivation by deiodination is considered the predominant route of metabolism of T4 and T3, 10 whereas in rats, hepatic glucuronidation has been historically assumed to be the major pathway of T4 clearance. 11 However, other studies have demonstrated that deiodination is likely the predominant pathway in this species as well. 12 Thus, a more systematic evaluation of T4 metabolism and clearance in rats is still needed. Induction of UGTs in the rat liver by xenobiotics often increases the rate of T4 and T3 metabolism and elimination and results in decreased serum T4 and increased serum TSH concentrations.
Thyroid Gland Carcinogenesis
There are inherent differences in the incidence, tumor types, and clinical history of thyroid cancer in mammals. 13 Thyroid follicular tumors are particularly common in rats, depending on the strain, which suggests that rats may have a genetic predisposition for thyroid tumors. In rats, thyroid tumors are more common in males compared to females, and follicular adenoma is the most common subtype. As described above, the rat also has important differences in TH production, metabolism, and excretion (TH economy) compared to other mammalian species and humans. Rats, particularly males, have high secretory capacity, rapid metabolism, and excretion of TH. Perturbation of TH economy results in rapid (as little as 1 week) morphologic changes in the rat thyroid gland that includes FCHH. 14 Adult rats (particularly males) have been shown to have greater serum free T4 concentrations and T4 turnover compared to humans. 11,15 However, such direct comparison has raised methodological concerns during the workshop, based on the need to thoroughly validate T4 kits for rat use, the different matrices of rat samples, and human derived standard curve materials.
Persistently increased TSH is considered an important key event for FCHH and eventual thyroid tumor development in the rat. 16 The TSH action on FCHH also may depend on iodide and selenium status of the thyroid gland. 17 –19 Generally, rats are fed diets with adequate to high levels of iodide. Dietary requirements for rats are 150 to 200 µg of iodide per kg diet; 20 µg per kg diet will produce deficiency. Supplementation of rats with iodide does not change serum hormone concentrations but does increase thyroid and tissue concentrations of iodine. 20
Thyroid cancer is common in humans and is increasing in incidence. However, death from thyroid cancer is low, because most cancers are low grade and removed early. Thyroid tumors are more frequent in women compared to men and most tumors are papillary carcinomas. 21 –23
Although humans develop goiter (grossly visible thyroid follicular hyperplasia) in response to chronic increases in endogenous TSH concentrations, such as occurs with iodide deficiency or exposure to goitrogens, TSH suppression does not decrease thyroid cancer recurrence in low and intermediate risk patients. However, endemic goiter is still an important clinical condition depending on the geographic location. What is not clear is whether chronically increased serum TSH and goiter represent a preneoplastic condition in humans. There are no or minimal increases in cancer incidence in humans with goiter, although some epidemiologic studies have shown an association between higher thyroid cancer risk and iodine deficiency. 24,25 There may be an increase in thyroid cancer in patients with immune-mediated Hashimoto thyroiditis.
Women have an increased incidence of papillary carcinomas, which rises at the beginning of the reproductive years. However, in postmenopausal women, incidences in males and females approximate and equalize by 85 years of age. 26
Follicular cell adenomas and carcinomas of the thyroid gland in rats are usually preceded by FCHH, which can be induced by chemicals associated with disruption of TH economy. The FC tumors in rats can also be promoted by mitogenic stimulation of focal thyroid hyperplasia. 6 Human thyroid cancer typically does not show this type of progression (ie, from hyperplasia to adenomas and carcinomas). 22 The different diagnostic criteria and terminology between rat and human FC tumors make direct comparisons impractical. According to the World Health Organization classification system, 27 human thyroid tumors are predominantly papillary carcinomas (85%). 21,22 Thyroid tumors in rodent nonclinical toxicity studies exhibit different histological patterns, as described in the International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) nomenclature 6 but are not typically stratified by subtype.
Workshop members discussed whether subclassifying rodent thyroid tumors could add value to the translatability to humans.
Thyroid Gland Parameters in Nonclinical Toxicity Studies
Thyroid Gland Weights
Standard endpoints in nonclinical toxicity studies include thyroid gland weights, histopathology, and serum TH concentrations. Developmental studies include brain morphology as an indicator of thyroid function. Although thyroid gland weights can provide a sensitive indicator of HPT disruption, 30 they can also be difficult to accurately measure in rodents. A previous STP working group recommended collection of thyroid gland weights in all species except mice. 31 For rats, thyroid glands should be dissected in a randomized sequence to eliminate bias and minimize the impact of technical errors (eg, collection of adjacent muscle). Handling artifacts can also be reduced by dissecting and weighing after fixation. 32 In mice and rat pups, thyroid weights often have high standard deviations due to technical errors, and artifacts associated with dissecting and weighing thyroid glands can complicate the microscopic assessment. Because of these factors, it is recommended that thyroid glands in mice and rat pups be collected with surrounding tissue to obtain high quality sections for histopathologic evaluation, precluding measurement of thyroid weights. The relationship between thyroid gland weights in adult rats and histopathology can vary by study. Weight changes may result from alterations in follicular size and colloid content in the absence of FCHH, or when higher weight from FCHH is offset by smaller follicles containing less colloid.
Thyroid Gland Histopathology
Morphologic features of FCHH
Changes in thyroid gland morphology are often the most reliable indicator for HPT disruption. Rats are generally more susceptible to FCHH compared to mice, 6,30 although in some cases the mouse is more sensitive. 7 Follicular cell hypertrophy in rats presents as a diffuse lesion characterized by increased cellular size and height. Follicle lumina may be reduced in size and filled with normal to pale eosinophilic colloid. Follicular cell hyperplasia, in contrast, can occur as either a diffuse or focal lesion. The follicles in diffuse hyperplasia have an increased number of cuboidal to low columnar FCs and are often smaller in size with reduced colloid. Follicular cells can pile up and form small papillary projections that project into the colloid lumens. 6
Follicular cell hypertrophy in rats may progress to and often occurs concurrently with diffuse hyperplasia in the same thyroid section, making it difficult to discern between these two changes. In the latter case, a combined diagnosis of hypertrophy/hyperplasia might be considered. As in other endocrine organs, increased functional demand is reflected by thyroid enlargement due to cellular hypertrophy and hyperplasia. 33
A severe diffuse FCHH is shown in Figures 1 and 2. Both lesions are also expected to return to normal in rats if the stimulus is withdrawn. Focal hyperplasia, in contrast, is regarded as a preneoplastic lesion in rats 6 that may or may not be reversible. Diffuse hyperplasia typically occurs due to chronic stimulation by increased serum TSH concentrations, whereas focal hyperplasia is thought to result from an induced or stochastic DNA mutation followed by increased cell turnover (stimulated or autonomous). According to the INHAND nomenclature, a combined diagnosis of hypertrophy/hyperplasia can be used if appropriate. 6 The combined term in these cases implies the diffuse type of hyperplasia. Similarly, the Standard for Exchange of Nonclinical Data format allows for the combined diagnoses.
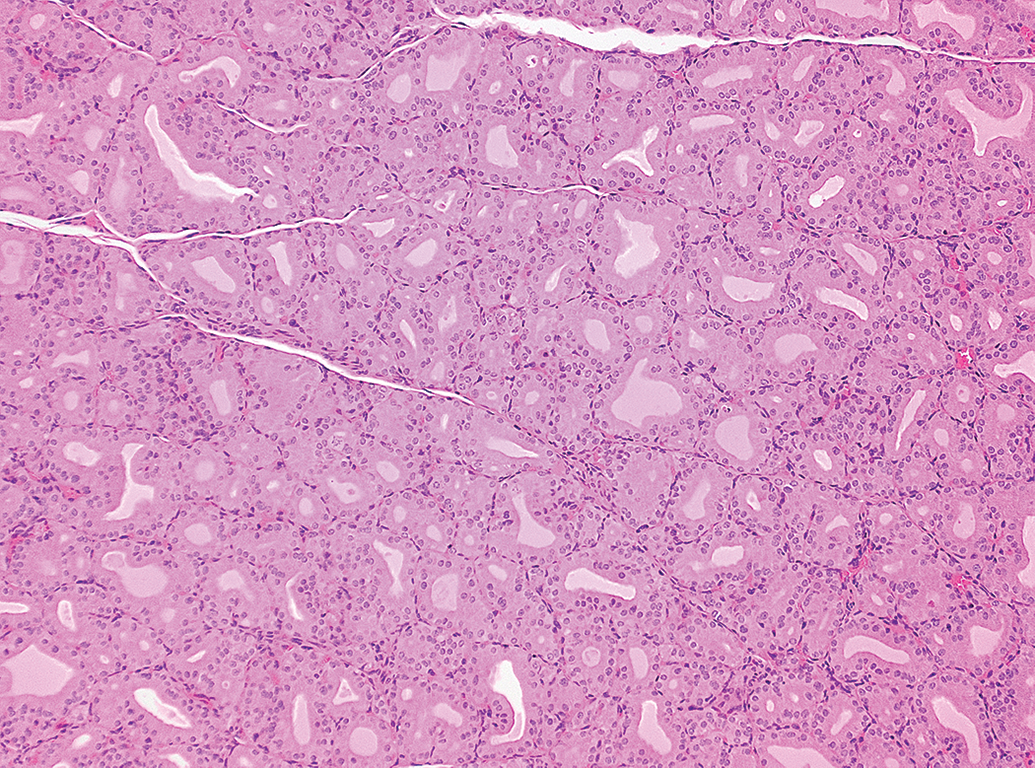
Thyroid gland, male Sprague Dawley rat, positive control study with PTU (2.5 mg/kg body weight/d, 42 days treatment). Severe diffuse follicular hypertrophy and hyperplasia. Hematoxylin and eosin (H&E) stain. Original objective 10×.
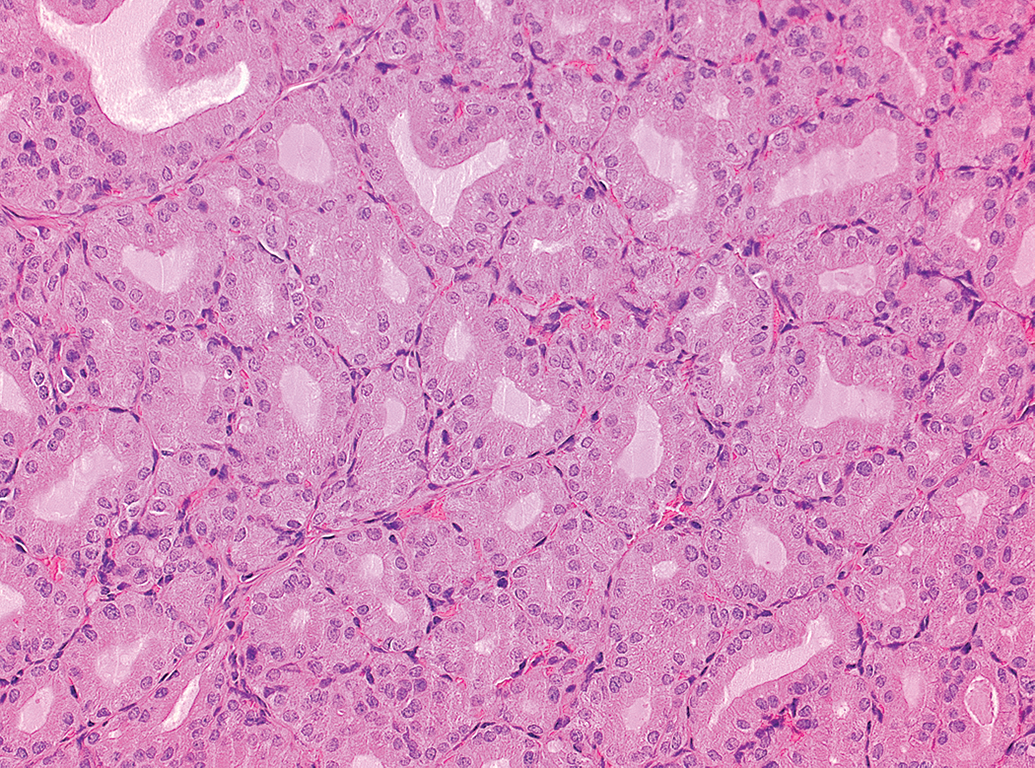
Thyroid gland, male Sprague Dawley rat, positive control study with PTU (2.5 mg/kg body weight/d, 42 days treatment). Higher magnification of Figure 1. Severe diffuse follicular hypertrophy and hyperplasia. H&E stain. Original objective 20×. H&E indicates hematoxylin and eosin.
Severity grading
Severity grades for FCHH can help determine whether an observed finding is test article-related and, if so, where the no/lowest observed adverse effect level should be set. Although previous guidances have proposed various systematic scoring criteria for FCHH, 34 workshop members did not support a uniform set of scoring criteria and indicated that when a grading scheme for FCHH is utilized, it should be well described. General grading recommendations have been described. 39 Participants noted that the inherent variation in thyroid gland morphology across different species, sexes, strains, test facilities, and studies (eg, based on different metabolic states, housing conditions, diets, and age of animals) makes it impractical to apply a common set of grading criteria. When lesion severity is important for study interpretation, then detailed criteria should be outlined in the narrative of the pathology report. Criteria must be definable, reproducible, and meaningful 40 and include the degree of change (eg, estimate of effect on cell number and size and follicle size), the distribution (eg, extent of gland affected), and the lesion characteristics (eg, presence of atypical cytologic or architectural features).
Reporting in control animals
Diffuse FCHH and colloid alteration are routinely observed in control animals, especially in male rats, and the incidence may vary widely across studies and testing facilities. These changes may also be qualitatively similar to those resulting from a test article. Historical control data (along with severity grades) may be important in such cases to determine whether an effect is test article-related or potentially adverse.
Altered colloid
Active thyroid glands in rats generally present with basophilic colloid, whereas inactive glands have more eosinophilic colloid. 41,42 The basophilic colloid may appear granular or clumped and contain foci of mineralization. Colloid from active thyroid glands may also have an increased number of clear vacuoles at the apical border of thyroid FCs, 43,44 which are thought to represent a fixation artifact even though the extent of the vacuoles may still correlate with follicular activity. The current INHAND term for these colloidal changes in rats is “colloid alteration.” 6 Workshop members noted that altered colloid also occurs in mice and dogs. This finding in rats increases with age as a spontaneous change. 45 When this finding is test article-related, then the colloid alteration is often accompanied by diffuse FCHH. Notably, it is often not reported as a distinct diagnosis from FCHH unless pronounced. 6
Workshop participants discussed the relationship between altered colloid and thyroid function. Exposure of rats to polychlorinated biphenyls (PCBs) have resulted in dose-dependent FCHH with abnormal accumulation of large colloid droplets and irregular lysosomes on ultrastructural evaluation.
46
It was hypothesized that PCBs were limiting colloid droplet–lysosome interaction, thereby inhibiting proteolysis of thyroglobulin necessary for TH release. However, it is not clear whether this specific type of colloidal change relates to colloidal alteration observed routinely in both control and treated rats. In addition, long-term treatment of rats with tembotrione, a 4-hydroxy-phenylpyruvate dioxygenase (HPPD) inhibitor, resulted in colloid alteration with FCHH. These changes were ultimately considered to be caused by tyrosinemia secondary to HPPD inhibition in rats and not relevant to human health risk assessment.
47
Thyroid Hormones
Measurement of serum hormones of the HPT axis is a required part of several different types of nonclinical toxicity studies. Diurnal variation of serum levels of T3, T4, and TSH 48 require blood sampling in a study to be limited to a defined time frame (eg, 2 hours) each morning. 49,50 Stress is discussed as an important factor among several factors influencing serum hormone levels of the HPT axis. However, various kinds of stress seem to alter THs differently, therefore a common change cannot be elucidated. 48,51,52 Nevertheless, it is advisable to minimize stress due to handling of rats just prior to blood sampling. In humans, the measurement of free TH in the blood is preferred. 53 The total serum TH levels are measured in rats because of different protein-binding characteristics of TH in rodents and due to limited blood volume. It remains to be demonstrated whether in the case of rat toxicity studies, free TH has a higher sensitivity or is more accurate than total TH. 54 –57 Tissue concentrations of TH have been used as a method to measure hormones at important target sites, such as brain. 58 –61 However, measuring tissue hormone concentrations is technically challenging and not part of standard requirements currently. Standardization and interlaboratory validation of hormone extraction protocols for tissue hormone measurements are difficult, and the sensitivity of tissue versus serum TH concentrations is not well established.
Proper validation of TH assays with rodent samples is critical for accurate and reproducible measurements. The validation procedure can be performed according to the European Medicines Agency (EMA) Guideline on bioanalytical method validation 62 and industry white papers. 63 –65 Intra-assay precision and sensitivity are key parameters for measuring TH in toxicity studies. Hormone assays contribute to one-quarter to one-third of the total variation among control groups of rat studies (BASF, unpublished). According to the Organization for Economic Co-operation and Development (OECD) test guidelines, the maximum acceptable total coefficient of variation (CV) among controls is 25% for T3 and T4 and 35% for TSH. 66,67 The CV is not dependent on the age of the rats. 50 The difference between the control group mean and the lowest quantifiable value of the assay is crucial for detecting potential decreases in T4 or T3 concentrations. This is particularly important in fetuses and postnatal day 4 pups with low TH levels where sensitive assays are necessary. 50 Following assay validation, a positive control study with a reference compound is recommended. The T4 kits which passed the validation procedure may fail in some cases to detect a significant decrease in a reference study because of different matrices of rat samples and human-derived standard curve materials. 50
Mechanisms Leading to FCHH
Mechanistic information can provide important perspective on findings in a toxicity study and aid in the risk assessment. If a compound is associated with effects on the follicular epithelium of the thyroid, then understanding the mechanism of the change is relevant to risk assessment. The US EPA has identified 15 potential mechanisms or modes of action (MOAs) that may alter thyroid function, which are organized around the concept of one or more molecular initiating events (MIEs). Each of these events is linked to an important component of TH signaling or function. However, some of the MOAs/MIEs are not commonly affected by exposure to environmental chemicals. For example, very few thyroid-active compounds interact directly with the TH receptor,
68,69
making this mechanism a lesser cause of concern. The EPA has thus prioritized mechanisms for thyroid perturbations based on currently understood relevance.
68
Briefly, the most commonly encountered mechanisms to consider during interpretation are: enhanced hepatic clearance, inhibition of TPO, inhibition of NIS, and inhibition or activation of iodothyronine deiodinases.
Activation of hepatic nuclear receptors and subsequent UGT enzyme induction to increase T4 sulfation and/or glucuronidation is most common in rodent toxicity studies. 70 This type of information can be part of a weight of evidence (WOE) review. For example, low-grade FCHH in the rat due to a liver-mediated MOA may be less of a concern compared to FCHH due to a different MOA. It is generally accepted that rodent thyroid tumors induced by chronic induction of UGT have little or no relevance to humans with respect to carcinogenesis, 71 although regulatory acceptance would still require demonstration of evidence specific to the test article. Thyroid carcinogenesis in rats may be an indicator of disruption of the HPT axis. 33
Toxicants that affect TH economy can disturb the thyroid gland itself or affect the HPT axis or metabolism even though they may have similar morphologic and biochemical changes in thyroid function, including decreased T4 (and/or T3), increased TSH, increased thyroid gland weight, and thyroid FCHH. Many thyroid toxicants produce a similar pattern of morphologic effects and it can be difficult to determine the precise mechanism for altered thyroid function. Additional mechanistic data collected in the nonclinical toxicity studies will aid in identification of the most probable mechanism.
The spectrum of effects in repeat-dose toxicity studies often provides a pattern suggestive of test-article effects on the HPT axis that may include decreased T4 and/or T3, increased serum TSH, increased thyroid weight, hypertrophy and vacuolation of pituitary thyrotropes, and thyroid histopathology (FCHH and decreased amount of colloid). Changes in TH levels which are due to hepatic enzyme induction with enhanced T4 metabolism and clearance generally accompany increased liver weights and hepatocellular hypertrophy. An increase in thyroid FC tumors may be seen in chronic 2-year studies. Effects on T4 and/or TSH blood levels in repeat-dose toxicity studies are sufficient to trigger regulatory concern, even in the absence of morphologic findings.
Pathologists organizing the study data in a way to evaluate thyroid-associated tissues often use a WOE-based approach. 72 –77 Another approach is to use adverse outcome pathways (AOPs) to organize data along increasing levels of biological complexity (thyroid-related AOPs appear as Supplementary Information 1).
There is ongoing interest in using the AOP framework as an organizing concept for presenting and analyzing toxicity data and identifying knowledge gaps. This framework is intended to help build context and regulatory confidence around shorter-term effects, including FCHH or changes in serum T4, and help determine when they may (or may not) result in adverse health effects. Additionally, this approach should enable integration of new alternative methods and data to improve interpretation of morphologic data and streamline current testing paradigms. Multiple pathways are available to further characterize the MOA/MIE of a xenobiotic, 68,78 but are beyond the scope of this article.
The MOA information provides useful context for evaluating pathologic effects and their human relevance. Ultimately, adversity should be based on observed effects, not target pathways. Thorough characterization of the tissue alterations accompanied by supportive mechanistic study data should allow the pathologist to determine whether the thyroid effect is adverse or not and also provide insight into the relevance of the finding(s) for patients receiving a drug or to individuals exposed to a xenobiotic in the environment.
Assessing Thyroid Gland Effects
Relationship to Test Article
Adversity in the context of nonclinical toxicity studies is only relevant to effects that are test article-related. 1 It is important for this initial determination to review experimental and technical factors that can affect thyroid-related end points as confounding factors. These include age, sex, pregnancy status, estrous stage, changes in body weight, and feed intake, all of which can affect the HPT axis. 48,79 –81 Caloric restriction (either 36 hours of complete fasting or 21 days of 50% food restriction) was reported to decrease serum T4 concentrations in male Wistar rats. 82 Reproductive studies have shown that parental diet, birth weight, and litter size can all influence TH concentrations in the offspring. 83 –85 Another important confounder of serum TH level is stress, including acute events such as transport to a new room or handling prior to blood sampling. 48,86 Systemic illness and starvation can lead to a condition known as nonthyroidal illness syndrome, which is associated with decreased T3 and T4, and TSH within reference range or decreased. 86 –88 These studies highlight the need for considering study context when interpreting thyroid endpoints.
Consistent assessment of FCHH depends on the proper understanding of normal or background variability. Although incidence and severity versus concurrent controls are the primary determinants of whether a finding is regarded as test article-related, thyroid FCs can show a considerable degree of variation, especially in male rats (Figures 3 and 4). Both figures represent either end of the spectrum of normality (especially in short-term studies). For comparison, Figures 1 and 2 show a severe FCHH in a rat, treated with PTU.
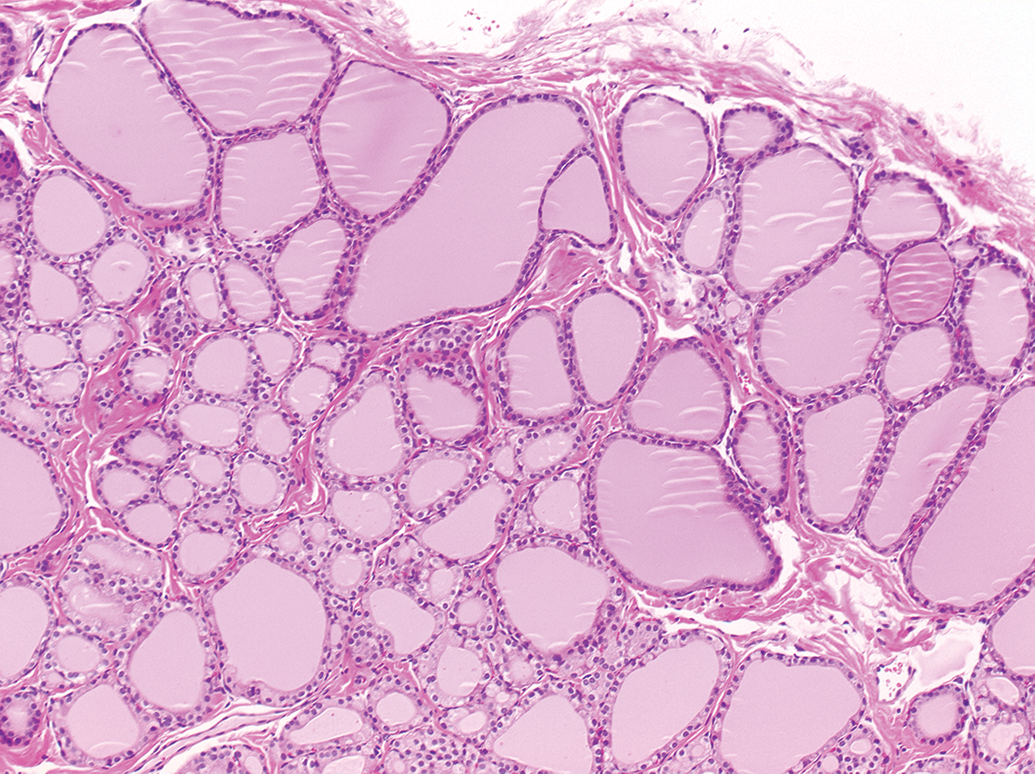
Thyroid gland, male control Sprague Dawley rat, 90-day study. Hematoxylin and eosin (H&E) stain. Original objective 10×.
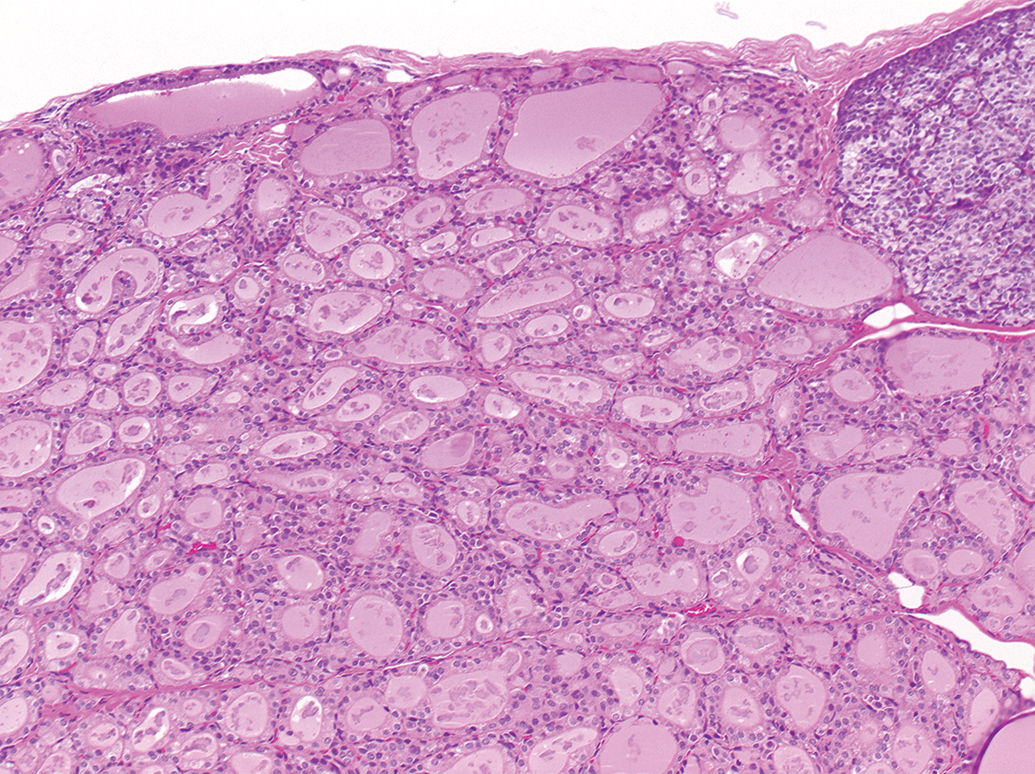
Thyroid gland, male control Sprague Dawley rat, 90-day study. Follicular hypertrophy and colloid alteration. H&E stain. Original objective 10×. H&E indicates hematoxylin and eosin.
The variability in control animals makes it challenging to distinguish low-grade effects from background variation. Other factors such as seasonal variations of iodine in the feed may also influence this morphologic variation.
Characterizing Adversity in Nonclinical Toxicity Studies
A prior ESTP working group presented the following working definition of adversity: “In the context of a nonclinical toxicity study, an adverse effect is a test article-related change in the morphology, physiology, growth, development, reproduction or life span of the animal model that likely results in an impairment of functional capacity to maintain homeostasis and/or impairment of the capacity to respond to an additional challenge.” 1(p812) The assumption based on this definition is that the thyroid glands have a physiological capacity to compensate for changes in hormonal and metabolic conditions as reflected by transient increases or decreases in activity. The workshop participants proposed that diffuse FCHH in the absence of other morphological changes such as focal hyperplasia or neoplasia should not be considered intrinsically adverse. Additional parameters indicating impairment of cell/tissue/organ function or reserve capacity should also be considered. These may include lesion severity, associated effects, including hormonal changes, MOA, and experimental factors such as life stage.
Severity of FCHH
Currently, there are no harmonized grading criteria or severity thresholds for calling FCHH adverse or nonadverse. However, minimal to mild diffuse FCHH occurs routinely in healthy control adult rats, and alterations within this range may thus represent an adaptive or compensatory nonadverse response to metabolic and environmental conditions (ie, FCs properly responding to physiologic cues). Thus a low severity grade, in the absence of other data indicating harm to the animal, may be sufficient to justify a nonadverse interpretation in the context of a given study. Moderate to severe FCHH is less frequently observed as a spontaneous finding and thus more likely to represent altered thyroid function or capacity. A case study illustrating how severity can influence the adversity decision appears as Supplementary Information 2.
The key essentials for adversity considerations of FCHH are visualized in Figure 5.
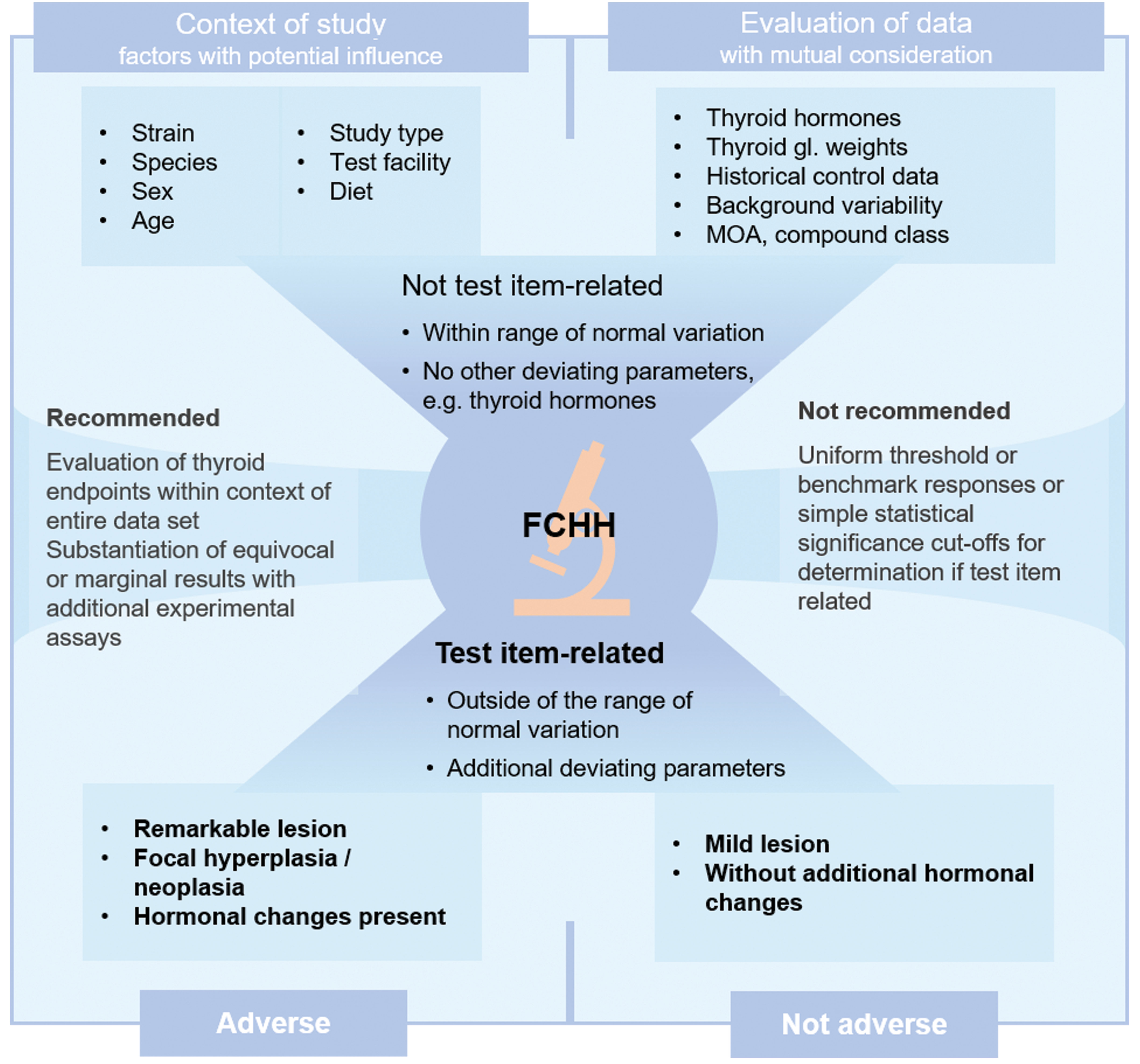
Key essentials for adversity considerations of follicular hypertrophy and hyperplasia in the thyroid gland.
Effect constellation of FCHH
The overall spectrum of findings provides important context for adversity decisions.
1
In the case of thyroid effects, other histopathological changes, TH levels, and thyroid weights can provide important supporting information regarding thyroid/HPT disruption and interpretation of FCHH.
When changes in serum TH concentrations are observed, low serum T4 is often given the most weight due to its direct association with adverse developmental effects.
92
–96
Increased TSH provides support for antithyroid MOAs and induces thyroid FCHH but it is not a causative key event in developmental outcomes and in some cases may be treated with less concern compared to low serum T4. Moreover, detecting effects on TSH are difficult due to high interanimal variability and variability in the timing and/or duration of changes in serum concentrations. Thus, the absence of a TSH effect, especially at a single time point, does not necessarily rule out a TSH-mediated effect. Serum total T3 concentrations are often given the least weight, since they do not reflect active T3 concentrations in target organs. A related issue is the use of TH thresholds or BMRs to guide adversity decisions based on correlations with adverse developmental outcomes.
92,96
Life stage considerations
The thyroid gland and HPT axis differ biologically based on developmental stage, including a limited capacity for compensation and higher susceptibility to permanent effects in early life. Epidemiologic studies have shown an association between maternal hypothyroidism and cognitive effects in offspring, 97,98 raising health concerns about developmental neurotoxicity when thyroid effects are observed. According to previous ESTP recommendations “adversity should be defined only within the context of the animal model…[,] within the temporal restrictions imposed on the study design, and with the amount of information available at the time of the integrated toxicology report.” 1(p 816) Accordingly, thyroid effects in an adult model should be distinguished to the extent possible from putative or hypothetical effects in a developmental setting. This separation can be challenging in some cases, particularly for environmental chemicals with potential for maternal or early life exposure. Thus, FCHH or lower serum T4 in standard nonclinical toxicity (adult-only) studies, whether considered adverse or not, may often trigger requests for developmental thyroid studies (eg, European Union Guidance for the identification of endocrine disruptors 2018). 49 Ideally, thyroid findings in dams (prenatal toxicity studies) or female animals after delivery (reproduction toxicity studies) should be assessed in combination with findings in the offspring.
Other Types of Supporting Data
Workshop participants noted that for many case examples there simply was not enough information available to make an informed interpretation of FCHH. Greater biological context is typically needed. This section outlines several alternative assays, markers, and data that can help provide this context. These ancillary biomarkers should generally be rare in a toxicology study but well suited for use in special investigative studies of impact on thyroid function.
Molecular Biomarkers
Various molecular markers can provide additional information for characterizing FCHH in nonclinical toxicity studies. For example, immunohistochemical markers such as Ki-67 and BrdU may assist in quantifying FC proliferation, establishing a thyroid MOA, and establishing reference dose levels. 99 Immunohistochemical detection of increased TSH or thyrotropes in the pituitary gland can also be used to support a TSH-mediated pathogenesis for FCHH, 100,101 although caution should be used in quantifying TSH, given variability in its release from thyrotrophs and in sectioning of the pituitary (in many cases of FCHH, thyrotroph hypertrophy/hyperplasia may not be evident). Recent studies have used in situ hybridization 102 and quantitative polymerase chain reaction 7 analysis to measure TSH-β subunit messenger RNA in the pituitary as an indicator of TSH induction. These methods may be particularly useful when data on serum TH are either not available or difficult to interpret (eg, because of timing of the samples).
Where available, analysis of the plasma metabolome in rats may also help to confirm a test article-related thyroid effect interfering with the thyroid gland structure and/or function and to differentiate between different mechanisms. 103,104
Structural Alerts
Efforts to interpret structural information regarding antithyroid activity date back to the 1940s, 105,106 initially without or with only limited mechanistic understanding. Increasing information on the HPT axis and the underlying biochemistry has led to a system for sorting and classifying available information. Knowledge regarding antithyroid mechanisms and corresponding chemical structures has now been assembled for many chemical classes (Table 1, which only includes direct mechanisms). In the future, high-throughput screening will considerably expand our knowledge and understanding, while the development of in silico tools for predicting potential antithyroid activity is expected in the longer term. Initial steps in that direction have already been taken. 107 Chemical alerts, however, may not always be helpful for reliable prediction of in vivo responses. There is currently a lack of understanding how in vitro/ex vivo inhibition translates into in vivo effects and to what extent other factors such as iodine deficiency in the diet or differences in the type of TPO inhibition play a role.
Chemical Alerts Suggesting Interactions With the HPT Axis.
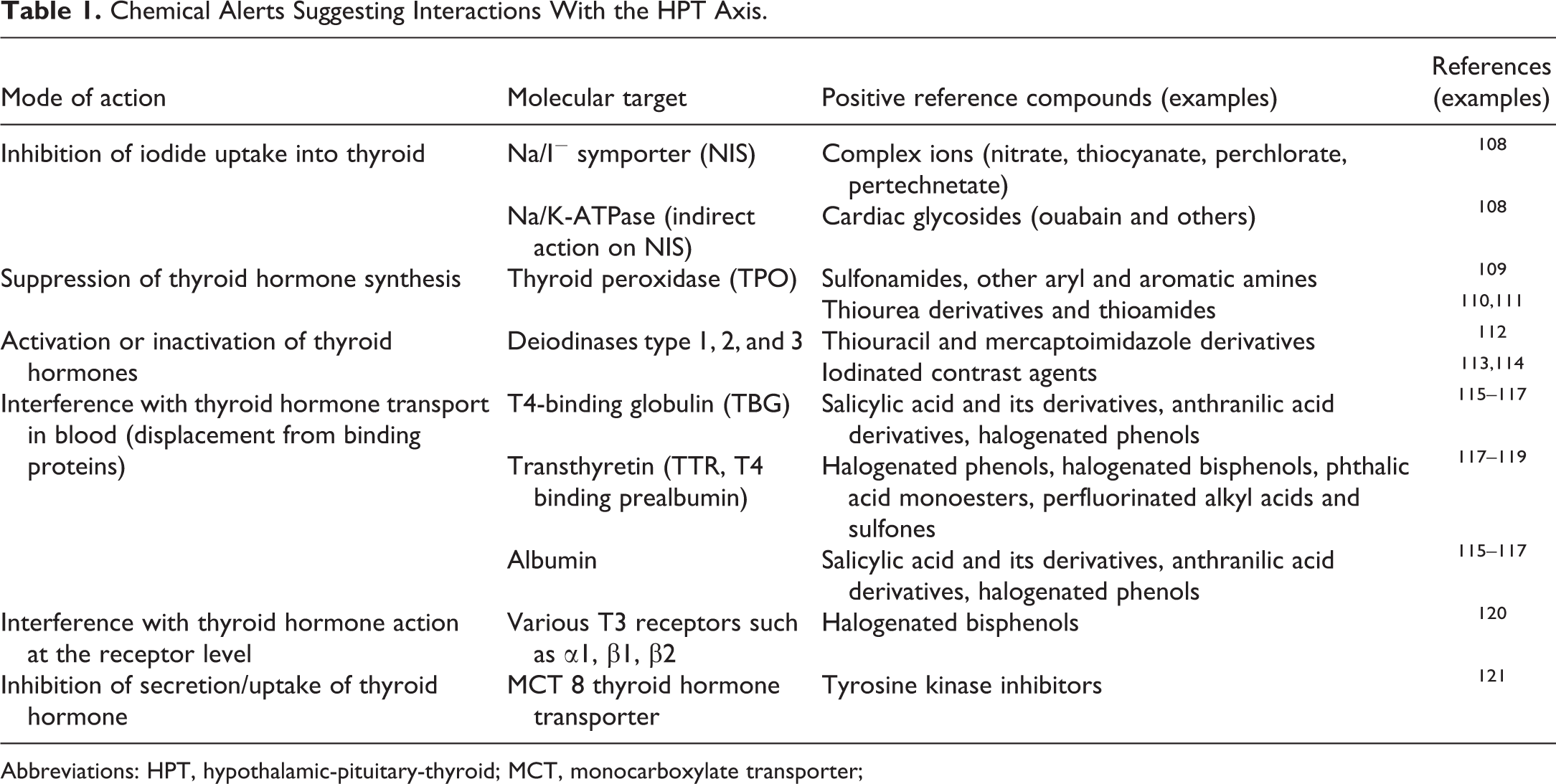
Abbreviations: HPT, hypothalamic-pituitary-thyroid; MCT, monocarboxylate transporter;
Perchlorate Discharge Test
This test measures the accumulation of 125 I in the thyroid gland (phase I), followed by measurement of thyroid: blood 125 I ratio using discharge with perchlorate (phase II). 122 –125 In phase I, compounds that inhibit 125 I uptake into the thyroid or organification of 125 I in the gland (eg, NIS and TPO inhibitors) will have lower accumulation of radiolabel in the thyroid gland, whereas compounds that alter TH levels at extrathyroidal sites (eg, by enzyme induction and enhanced T4 metabolism) generally increase the uptake of 125 I into the thyroid gland. Phase II animals are treated with perchlorate which releases unbound 125 I from the thyroid. Bound 125 I (eg, that which has been incorporated into TH) is retained in the gland. Thus, compounds that prevent TH synthesis such as TPO inhibitors will result in a greater release of 125 I into blood with perchlorate treatment and a decrease in the thyroid:blood 125 I ratio. This ratio will also decrease with compounds that inhibit 125 I uptake. In contrast, rats with an extrathyroidal mechanisms of thyroid toxicity will continue to incorporate 125 I into TH synthesis and release little 125 I into the blood, resulting in an increased thyroid:blood 125 I ratio relative to toxicants that target the thyroid gland itself (thyroid:blood 125 I ratio may be similar to euthyroid rats). Thus, the perchlorate discharge test differentiates toxicant effects occurring in the thyroid gland from effects occurring at extrathyroidal sites of action. Agents acting at extrathyroidal sites include FD&C Red No.3, SK&F 93479, phenobarbital, arochlor 1254, and β-napthoflavone. An agent directly acting at the thyroid gland, such as PTU, inhibits TPO and DIO1. Noxythiolin, which alters iodide organification in vitro, has no effect in an in vivo perchlorate discharge test. 122 The perchlorate discharge test has also been used clinically to diagnose thyroid diseases.
In Vitro Assays
The Tox21 and US EPA Toxicity Forecaster (ToxCast) databases include a variety of high-throughput in vitro assays that can be used to examine potential of a xenobiotic to cause thyroid toxicity (Table 2). These data may be useful to eliminate or focus on specific MIEs when examining existing chemistries or using “read across” to related compounds that have undergone high-throughput testing. Additional high-throughput assays have been developed in US EPA laboratories but are not yet included in the ToxCast database. These include assays on the NIS, TPO, and the DIO1. 126 –129
ToxCast and Tox21 Assays (Version 7) That May Provide Thyroid MIE Information.a

Abbreviations: ELISA, enzyme-linked immmunosorbent assay; MIE, molecular initiating event; mRNA, messenger RNA; THR, thyroid hormone receptor; TPO, thyroid peroxidase; TSH, thyroid stimulating hormone; UGT, uridine diphosphate glucuronosyltransferase; SULT, sulfotransferase.
a Many assays also include cytotoxicity assays to verify specificity.
Under the head of European Union Network of Laboratories for the validation of alternative methods, the Joint Research Centre is coordinating a validation study of in vitro methods for the detection of thyroid disruptors. Based on the OECD scoping document on in vitro and ex vivo assays for the identification of modulators of TH signaling (OECD Series on Testing and Assessment No. 207, OECD, 2017), 16 in vitro methods are currently being established. 130
Regulatory Considerations
The regulatory consequences of the thyroid adversity assessment vary according to global regions and different regulated industry sectors. This regulatory diversity was not discussed in depth during this workshop but clearly influenced several topics. Below is a summary of the current regulatory framework for several global regions/sectors, which aims to highlight similarities and differences, impacted by thyroid adversity considerations including those addressed during this workshop.
Europe (European Chemicals Agency/European Food Safety Authority)
Thyroid gland carcinogenicity and TH-mediated noncarcinogenic effects are typically addressed separately under the current European Union (EU) regulatory frameworks. Both cases require the careful assessment and identification of thyroid gland adversity due to its serious regulatory impact.
Assessment of thyroid tumors
Hazard classification of a chemical or xenobiotic as carcinogenic is typically performed under the Classification, Labelling and Packaging (CLP) regulation, 131 based on the United Nations’ Globally Harmonised System. Extensive guidance on the application of the CLP criteria to support the harmonised classification and labelling for certain hazardous substances has been developed by the European Chemicals Agency (ECHA) and is updated approximately every 2 years. 132
The guidance states that substances are classified according to their potential to cause cancer in humans. There may be direct evidence on the carcinogenicity to humans from epidemiological studies. However, in most cases the available information on carcinogenicity will be primarily from animal studies. The relevance in this case of the findings in animals to humans must be considered.
This guidance includes the recognition that some mechanisms of tumor formation are considered not relevant for humans, for example, certain thyroid tumors in rodents mediated by UGT induction, supported by reference to International Agency for Research on Cancer 133 and EU specialized experts. 134 Under CLP, each conclusion is dependent on a WOE of the available MOA data.
According to the EU specialized experts as documented in 1999, 134 there is convincing scientific evidence that humans are considerably less sensitive than rodents (especially rats), regarding (1) perturbation of TH homeostasis induced by nongenotoxic xenobiotics and (2) development of epithelial thyroid tumors after long-term exposure to such agents.
Thyroid hormone mediated noncarcinogenic adversity
Coincident with the preparation of this workshop, 2 pieces of EU legislation were updated which triggered explicit consideration of thyroid toxicity, as one of four endocrine modalities of regulatory focus: Commission Regulation (EU) 2018/605
135
amended the Plant Protection Products Regulation
136
and Commission Delegated Regulation (EU) 2017/2100
71
pursuant to the Biocidal Products Regulation (EU) No 528/2012 both
49
set out scientific criteria for the determination of endocrine-disrupting properties of plant protection products and biocides, respectively. These criteria are applicable to relevant dossiers from specific dates in 2018. According to these regulations (2017/2100 and 2018/605), EDs (endocrine disruptors) are substances (with respect to humans) with the following features: Show adverse effects in an intact organism or its progeny, which is a change in the morphology, physiology, growth, development, reproduction or life span of an organism, system or (sub)population that results in an impairment of functional capacity, an impairment of the capacity to compensate for additional stress, or an increase in susceptibility to other influences. Have an endocrine MOA, that is, alter the function(s) of the endocrine systems. The adverse effect is the consequence of the endocrine MOA.
In response to this new regulatory requirement, the European Commission asked the European Food Safety Authority (EFSA) and the ECHA to develop a guidance document for the implementation of the scientific criteria for the determination of endocrine-disrupting properties pursuant to the Biocidal Products Regulation (EU) No 528/2012 and the Plant Protection Products Regulation (EC 2017). The current version of this guidance document was published in June 2018. 49 Specifically relevant to thyroid toxicity is Appendix A “Additional considerations on how to assess the potential for thyroid disruption for human health.” The aim of this appendix is to provide: “…additional guidance on which data could be provided and considered in the weight of evidence to substantiate that some specific thyroid effects are not human relevant and how to address specific thyroid related developmental neurotoxicity (DNT) concerns. This appendix is not intended to be exhaustive and covers all MoAs associated to thyroid effects for which the principles detailed in this guidance should be applied.”
Appendix A recognizes that there can be quantitative species-specific “…differences in the systemic regulation of TH levels between commonly used experimental animal models and humans”.53(pp102,135) The following concepts are recommended when interpreting data from experimental animals: Substances inducing histopathological changes (ie, FCHH, focal hyperplasia, and/or neoplasia) in the thyroid with or without changes in the circulating levels of TH would pose a hazard for human TH insufficiency in adults as well as pre- and postnatal neurological development of offspring. Substances that alter circulating levels of T3 and/or T4 without histopathological findings would still present a potential concern for neurodevelopment. Humans and rodents are considered to be equally sensitive to thyroid disruption in the absence of substance-specific data which provide proof of the contrary (including cases where liver enzyme induction is responsible for increased TH clearance).
Appendix A also provides additional ideas on how to investigate human relevance of a thyroid effect observed in rats to investigate the MOA of the thyroid disruption and on the WOE for human relevance. However, it also recognizes that there is ongoing uncertainty about how thyroid disruption can be appropriately investigated due to a lack of relevant test methods.
Like CLP, each conclusion is dependent on an assessment of the WOE of the available data. A conclusion on whether the endocrine-disrupting properties have been met or not will require consideration of adverse effects according to the IPCS definition, 137 a TH mediated MOA and a biologically plausible link between them as outlined in the criteria. 71,135
US Environmental Protection Agency
Follicular cell hypertrophy and/or hyperplasia associated with environmental chemicals in nonclinical studies generally indicates activation of the HPT feedback loop with increased TSH and decreased circulating TH. Areas of uncertainty include the relationship between morphologic (FCHH) and underlying hormonal changes (T3, T4, and TSH), between rodent and human pathophysiology, and between adult and developmental settings. As an example at the workshop, perchlorate was used to highlight these issues and the importance of neurodevelopmental considerations when evaluating thyroid changes. 138 Early life neurological impairments are often the primary human health concern for environmental chemicals that induce thyroid FCHH based on experimental and epidemiologic evidence showing a direct association between low maternal T4 during pregnancy and neurodevelopmental outcomes in offspring. 96,139 Hormonal changes in a nonclinical toxicity study (decreased T3/T4 or increased TSH) may be considered a more relevant endpoint to the human health risk assessment rather than morphologic changes (FCHH) given that low TH is the more proximate driver of neurodevelopmental effects. This “biomarker” issue adds complexity to adversity decisions regarding FCHH especially at the study level when a complete list of effects may not be available.
The panel discussion focused on the role of TH changes in interpreting adversity of FCHH. It was generally agreed that the rat thyroid FC is more sensitive to HPT disruption compared to human. However, it is not clear from current evidence whether or to what extent TH changes in the rat differ from those in the human given a similar exposure to an antithyroidal agent. The question remained what magnitude TH changes in the rat may reach the level of adversity, with or without FCHH. A 20% decrease in T4 has previously been proposed as a BMR for adversity, but to date this type of threshold approach has not been applied in regulatory assessment. An EPA Science Advisory Panel in 2011 stated, “For the Agency to be able to use precursor key events as the basis for a [point of departure] for a risk assessment, the quantitative relationships must be understood between precursor events or key events within a causal path leading to a disease or adverse outcome”. 140
Any evidence of HPT disruption, including FCHH, can serve as a trigger for developmental data requests. Agrochemicals are the main legislative driver in the Food Quality Protection Act (FQPA) of 1996 which directs EPA to use an additional 10-fold safety margin to protect infants and children in setting pesticide tolerances. 141 Early life stages are more sensitive to thyroid-disrupting chemicals, given reduced compensatory abilities, the potential for permanent alterations related to HPT function, and other biological differences in susceptibility. 142 The implication is that points of departure for thyroid disruption based on adult studies may not be adequately protective in the developmental setting. The rat DNT study was used historically to address these types of questions. However, questions about the sensitivity of the DNT to detect neurological outcomes resulting from TH disruption during development led the EPA Office of Pesticide Programs to establish a special mechanistic study called the developmental or comparative thyroid assay as a surrogate study. 34
The FC tumors in addition to neurodevelopmental effects are associated with antithyroid MOAs in longer-term studies of rodents (rats in particular). The relevance of these tumors to human health has been widely debated because carcinogenicity of the thyroid gland follows different patterns based on animal species. 15 The US EPA along with other chemical regulatory authorities consider thyroid FC tumors in rodents to be of potential human relevance and manage human health risks through the MOA framework. 143 Demonstration of an antithyroid (or other non-mutagenic) MOA under this system allows a threshold method of quantitative risk assessment to be used based on a precursor event such as increased thyroid FC proliferation or FCHH. The premise here is that a reference dose level protective of HPT disruption in a sensitive species such as rat should also protect against potential FC tumors in human populations. In an integrated assessment, FC tumors in a chronic study may thus shift the interpretive context for FCHH in a shorter-term study. This risk-based approach is based in part on the amended FQPA of 1996, which exempted pesticide residues from the Delaney Clause by permitting EPA to approve tolerances that would pose no more than a “negligible risk” to consumers. 144
Thyroid hormone signaling is an important part of the Endocrine Disruptor Screening Program (EDSP), which has a broad mandate under the FQPA to screen chemicals for potential endocrine activity. Currently, the EDSP consists of two assay batteries, which target estrogen, androgen, thyroid, and steroidogenic pathways. Thyroid endpoints are required as part of several Tier 1 EDSP assays, most notably the rat pubertal development and thyroid function assay, 145,146 and their histopathology endpoints have been previously described. 147 These endpoints are evaluated in context with the totality of the data in determining whether an effect is adverse. Some of the factors considered in this context include dose selection, variability of the assay, variation of the endpoint per se, and consistency with effects seen throughout the database. 148 While thyroid activity may trigger a test order under the EDSP, the US EPA does not have a labeling mandate or systematic formula for calling a chemical an ED.
Food and Drug Administration
Approaches to determining adversity can not only differ widely among global regulatory authorities (eg, the United States Food and Drug Administration [US FDA] and European EMA) but also among Centers of the same regulatory agency (eg, the US FDA) depending on the regulatory context of the regulated product (eg, CDER regulating drug products vs Center for Food Safety and Applied Nutrition [CFSAN] regulating food products). 1 Specifically, adversity determinations of thyroid-related toxicity endpoints, such as hormonal imbalances (resulting in hypo/hyperthyroidism or hypo/hyperthyroxinemia), hyperplasia, or cancer, depend on the regulatory context of the compound under evaluation. Regulations and guidance are often intentionally nonprescriptive, providing the needed flexibility for a case-by-case assessment of the respective WOE approach. It is, in general, difficult for regulators to globally harmonize organ-specific adversity assessments while still providing context-specific flexibility. Differing and contradicting regulatory interpretations of similar histopathological occurrences are, therefore, not uncommon. 1
The decisions of the Centers of the US FDA are based on the best available science, incorporating new and evolving approaches and technologies (https://www.fda.gov/science-research/advancing-regulatory-science/conclusion-strategic-plan-regulatory-science).
Recently, the US FDA, alongside the US National Institutes of Health, the US EPA, and other federal agencies, made collaborative efforts to advance toxicology toward a more predictive science through programs such as Toxicology Testing in the 21st Century and the Interagency Coordinating Committee on the Validation of Alternative Methods (https://www.fda.gov/ScienceResearch/AboutScienceResearchatFDA/ucm612467.htm). The US FDA specifically advocates the increasing use of in vitro and, potentially, computer-based modeling systems for toxicology. The development of cell lines, engineered model tissues, and other cell culture approaches are deemed critical, not only to better understand underlying mechanisms in human cells following chemical exposure but also to potentially replace current approaches with more efficient and cost-effective toxicology studies (https://www.fda.gov/ScienceResearch/SpecialTopics/RegulatoryScience/ucm228212.htm).
Beyond these efforts, however, regulatory context and historical precedent (including the purpose of the evaluation and the associated laws) often determines the overall regulatory decision-making process despite acknowledged scientific findings.
For example, for food additives (under the jurisdiction of US FDA CFSAN), the argument that thyroid cancer in male rats is not relevant to humans because of an indirect MOA (increased TSH levels constituting the key event) is legally not recognized under the Delaney Clause (Federal Food, Drug, and Cosmetic Act, section 409(c)(3)). The Delaney Clause was enacted in 1958 and requires FDA not to approve the use of food additives that have been found to induce cancer in humans or animals at any dose. Resulting regulatory decisions are, therefore, a matter of law and not only based on scientific arguments. Adversity assessments of thyroid-related noncancer endpoints are, as stated above, case-by-case decisions, and the assessment approach depends on the regulatory use, data gaps, exposure, affected life stages, and inherent uncertainty of the data available. These data may trigger requests for 2-generation developmental studies, DNT studies, or extended 1-generation studies. 149
Food Safety Commission of Japan
The Food Safety Commission of Japan has no specific guidance or guideline focused on evaluation of endocrine disrupting chemicals including those affecting the thyroid. Recent toxicological evaluations of chemicals in foods which observed liver hypertrophy without indication of hepatotoxicity have been judged as adaptive but not adverse in mice, rats, and dogs, based on the guidance of expert committees on pesticide, veterinary medicines, and feed additives toxicology. Conversely, liver hypertrophy with FCHH in the thyroid has been considered adverse in many cases based on the recognition that the normal liver function/capacity is altered. A mechanistic study is needed to clarify that the thyroid effect is secondary to phase II metabolism enzyme induction including UGT activity in the liver and circulating TH levels.
Conclusions
The broad goal of the workshop was to develop criteria and consensus recommendations for characterizing the adversity of thyroid FCHH in order to facilitate a better alignment of toxicologic pathology and regulatory sciences on how to determine adversity of FCHH in the context of nonclinical toxicity studies. The workshop concluded broad agreement regarding the following recommendations: A wide variety of factors influence the response of a thyroid gland in a nonclinical toxicity study and careful comparison to concurrent controls is essential. Follicular cell hypertrophy and/or hyperplasia in adult rats without other morphological changes such as focal hyperplasia or neoplasia should not be considered intrinsically adverse at the level of an isolated animal toxicity study. Qualitative severity scores alone should not be used to determine whether a thyroid histological finding is adverse. Other hormonal and mechanistic data are necessary to fully determine the adversity of a thyroid finding at the level of an isolated animal toxicity study. Thyroid endpoints are best evaluated within the context of the entire data set for a given study, including knowledge of the compound class and MOA. Threshold or BMRs (or simple statistical significance cutoffs) should be avoided to determine whether a change is test article-related. Where possible, additional experimental assays/models/data to substantiate equivocal or marginal results are recommended.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_0192623320972009 - Adversity Considerations for Thyroid Follicular Cell Hypertrophy and Hyperplasia in Nonclinical Toxicity Studies: Results From the 6th ESTP International Expert Workshop
Supplemental Material, sj-docx-1-tpx-10.1177_0192623320972009 for Adversity Considerations for Thyroid Follicular Cell Hypertrophy and Hyperplasia in Nonclinical Toxicity Studies: Results From the 6th ESTP International Expert Workshop by Maike Huisinga, Lise Bertrand, Ronnie Chamanza, Isabelle Damiani, Jeff Engelhardt, Sabine Francke, Alexius Freyberger, Takanori Harada, Johannes Harleman, Wolfgang Kaufmann, Kevin Keane, Josef Köhrle, Barbara Lenz, M. Sue Marty, Stephanie Melching-Kollmuss, Xavier Palazzi, Gabriele Pohlmeyer-Esch, Andreas Popp, Thomas J. Rosol, Volker Strauss, Hetty Van den Brink-Knol, Charles E. Wood and Midori Yoshida in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-docx-2-tpx-10.1177_0192623320972009 - Adversity Considerations for Thyroid Follicular Cell Hypertrophy and Hyperplasia in Nonclinical Toxicity Studies: Results From the 6th ESTP International Expert Workshop
Supplemental Material, sj-docx-2-tpx-10.1177_0192623320972009 for Adversity Considerations for Thyroid Follicular Cell Hypertrophy and Hyperplasia in Nonclinical Toxicity Studies: Results From the 6th ESTP International Expert Workshop by Maike Huisinga, Lise Bertrand, Ronnie Chamanza, Isabelle Damiani, Jeff Engelhardt, Sabine Francke, Alexius Freyberger, Takanori Harada, Johannes Harleman, Wolfgang Kaufmann, Kevin Keane, Josef Köhrle, Barbara Lenz, M. Sue Marty, Stephanie Melching-Kollmuss, Xavier Palazzi, Gabriele Pohlmeyer-Esch, Andreas Popp, Thomas J. Rosol, Volker Strauss, Hetty Van den Brink-Knol, Charles E. Wood and Midori Yoshida in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-docx-3-tpx-10.1177_0192623320972009 - Adversity Considerations for Thyroid Follicular Cell Hypertrophy and Hyperplasia in Nonclinical Toxicity Studies: Results From the 6th ESTP International Expert Workshop
Supplemental Material, sj-docx-3-tpx-10.1177_0192623320972009 for Adversity Considerations for Thyroid Follicular Cell Hypertrophy and Hyperplasia in Nonclinical Toxicity Studies: Results From the 6th ESTP International Expert Workshop by Maike Huisinga, Lise Bertrand, Ronnie Chamanza, Isabelle Damiani, Jeff Engelhardt, Sabine Francke, Alexius Freyberger, Takanori Harada, Johannes Harleman, Wolfgang Kaufmann, Kevin Keane, Josef Köhrle, Barbara Lenz, M. Sue Marty, Stephanie Melching-Kollmuss, Xavier Palazzi, Gabriele Pohlmeyer-Esch, Andreas Popp, Thomas J. Rosol, Volker Strauss, Hetty Van den Brink-Knol, Charles E. Wood and Midori Yoshida in Toxicologic Pathology
Footnotes
Authors’ Note
The opinions expressed in this document are those of the authors and do not reflect views or policies of the employing institutions, including the US FDA and the FSCJ. Approval does not signify that the contents necessarily reflect the views or the policies of the Agency. Mention of trade names or commercial products does not constitute endorsement or recommendation for use.
Declaration of Conflicting Interests
The author(s) declared no potential, real or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
