Abstract
Standard histology trimming practices for toxicity studies in dogs include preparation of a section of the papillary muscle associated with the left ventricular free wall—the anteriolateral or anterior muscle. In contrast, the posteromedial or posterior papillary muscle, associated with the interventricular septum, is not commonly evaluated. In humans, the posterior papillary muscle is more often affected in ischemic myocardial disease, in large part due to the absence of collateral circulation, in contrast to the anterior muscle. Due to the differential vascular supply to the papillary muscles, we sought to determine whether there is a higher incidence of spontaneous coronary vascular changes in the posterior papillary muscle versus the anterior muscle in dogs. The hearts of 30 vehicle-treated or untreated beagle dogs that were euthanized for other purposes were collected and sectioned in a consistent manner to include both papillary muscles for histologic evaluation. The posterior muscle consistently had higher incidences of intramural coronary arteries affected by vascular medial hypertrophy, medial arteriosclerosis, intimal hyperplasia, and/or disruption or loss of the internal elastic lamina. This observation may have significant implications for the evaluation and characterization of spontaneous and xenobiotic-induced cardiovascular lesions in dogs.
Introduction
The papillary muscle (PM) is a common site of xenobiotic-induced cardiovascular lesions in laboratory species. 1,2 The PM, attached to valve cusps by chordae tendineae, is responsible for the opening of atrioventricular valves during diasystole. In people, the PMs are subject to injury, such as myocardial infarction, as the sequelae of coronary artery disease. 3,4 The latter illustrates the importance of the coronary circulation and perfusion of the PMs, which differ between the 2 PMs (in the left ventricle). The anterior PM is located on the left ventricular free wall and is supplied by the left anterior descending artery as well as the left circumflex artery. The posterior PM located on the interventricular septum, by contrast, is only supplied by the right coronary artery. 5,6 This disparity contributes to a higher incidence of posterior PM rupture in the context of myocardial infarction in people. 4,7,8 This rupture can lead to mitral valve regurgitation, which is a clinically important prognostic indicator. 3,9 Furthermore, the PMs are the last portion of the heart to be perfused, and thus show evidence of inadequate myocardial oxygenation, are more sensitive to hypoxia, and are more prone to ischemic injury. 8 In dogs, the posterior PM is variably supplied by the septal branch of the left coronary artery and/or left circumflex artery, while the anterior PM is supplied by several branches of the left and right coronary arteries. 10,11
Trimming practices for nonclinical toxicity studies as documented in the literature are vague and include methodology such as “left ventricle (including apex and papillary muscle)” 12 or “papillary muscle.” 13 Importantly, due to potential variations in necropsy and trimming practices, it is possible that different PMs are evaluated by a pathologist for any given animal within a study. Based on private communications, some companies pursue a more extensive trimming practice to include both PMs with emphasis on the insertion site of chordae tendineae as a standard protocol, or follow an extended sampling of both PMs for known cardioactive compounds.
Due to the potential for differential response to stressors between the left ventricular PMs, based on coronary circulation and human functional data, and the general lack of distinction for which PM is evaluated in toxicity studies, we sought to evaluate both PMs in young purpose-bred beagle dogs not exposed to any xenobiotic compound. With this approach, the characterization of spontaneous findings in both PMs in dogs could be performed both by light microscopic evaluation of hematoxylin–eosin (HE) slides and staining with Verhoeff-van Gieson (VVG) for the internal elastic lamina (IEL), and Masson’s trichrome (MT) for collagen deposition. This would inform a historical control database for both PMs in dogs, provide a thorough description of the normal variation of intramural coronary vascular morphology (currently limited in the literature), as well as illustrate a potential for disparate responses to stressors between the PMs. Based on our findings that there is a greater incidence of remodeled intramural coronary vessels associated with the posterior PM compared to the anterior PM, we propose that it is indeed critical to consistently sample one or the other PM within a given study. We also propose that consideration be given to evaluating both PMs on a case-by-case basis, such as for toxicologic evaluation of potentially cardioactive compounds.
Materials and Methods
Animals
Animals (N = 30) included in this study were approximately 1-year-old purpose-bred male beagle dogs weighing 8.1 to 10.7 kg. The majority (N = 28) of dogs were briefly exposed to anesthesia and invasive cardiac monitoring prior to euthanasia. These dogs were in the control group and were not exposed to any experimental pharmaceutical agent. Animals were fasted overnight the day prior to anesthesia and were sedated by pentobarbital bolus (35 mg, intravenous) and maintained by pentobarbital infusion (6 mg/kg/h) for between 3 and 5 hours. Dogs were intubated and invasive monitoring included a Swan Ganz catheter (5.5F) advanced into the pulmonary artery via the right jugular vein, and a dual-tip micromanometer catheter advanced into the left ventricle via the right carotid artery. At the end of the study, animals were euthanized with 10 mL potassium chloride (2 mEq/mL). N = 2 dogs were not exposed to any experimental compounds and were euthanized with pentobarbital (4 mL). All procedures were approved by Abbvie’s Institutional Animal Care and Use Committee and carried out in American Association for Accreditation of Laboratory Animal Care-accredited facilities.
Tissue Collection and Trimming
Prospective collection of whole hearts was performed by immersion fixation in 10% neutral buffered formalin immediately after death. After a minimum of 48 hours fixation, the hearts were grossly examined, and all 4 chambers were opened. The heart was cut into 8 tissue sections for histologic evaluation (Supplemental Figure 1). These sections included: Right ventricle, tricuspid valve cusp, right atrium, right atrial appendage Left ventricle (basilar aspect), posterior mitral valve cusp, left atrium, extramural coronary branch Posterior PM (M. Papillaris dorsalis) of the left ventricle Left ventricle (basilar aspect), anterior mitral valve cusp, left atrial appendage Anterior PM (M. Papillaris ventralis) of the left ventricle Interventricular septum, septal mitral valve, tricuspid valve cusps Apex to include left wall, right wall, septum Heart base to include cross-sections of ascending aorta, pulmonary trunk, intertruncal adipose tissue
Histologic Evaluation and Scoring
All tissue sections were processed routinely, stained with HE, and examined histologically by a board-certified veterinary pathologist (R.K.). The incidence and character of spontaneous vascular findings was documented and compared between the anterior and posterior PM sections. Papillary muscle sections were stained with VVG to optimize the evaluation of the IEL, and MT to highlight medial collagen deposition. Additionally, step sections of representative tissue sections were restained with VVG to evaluate for branching architecture of the coronary vessels.
Establishing a grading scheme for evaluation of intramural coronary vascular findings, each HE section was assigned scores based on the incidence of remodeled intramural coronary vessels. Remodeling was defined as medial hypertrophy, medial cell nuclear hypertrophy, medial sclerosis/collagen, and/or medial vacuolar degeneration. Semiquantitative incidence of hypertrophic vessels was recorded as a score of 1 (rare vessels affected in examined sections of PM), 2 (few), 3 (several), and 4 (many). This score was added to a score for character of the lesion, if present, which included a score of 1 (medial degeneration/vacuolation present in affected vessels) and/or 2 (medial collagen present in affected vessels). Subsequently, this score was added to a score for the IEL on VVG-stained section, which included a score of 1 for either disruption of the IEL or intimal hyperplasia, or 2 for absence of the IEL. These scores (for HE and VVG) were summed for a final composite score per animal for the posterior PM and anterior PM (Supplemental Table 1).
Statistical Methods
Comparison between mean scores was accomplished by a Student t test. P values below .05 were considered statistically significant.
Results
Animals
All dogs were clinically healthy at the time of euthanasia. There were no gross observations in the hearts or vasculature of any dogs in this study.
Higher Incidence of Coronary Vascular Hypertrophy in the Posterior PM
A nonhypertrophied/ nonremodeled vessel is shown in Figure 1. Vascular medial hypertrophy in this study was defined as irregular thickening of the vascular media with enlargement of nuclei, open or clumped chromatin, and increased amount of smooth muscle cell cytoplasm. This finding was common and was present in the posterior PM of 29 (97%) of 30 dogs, and in the anterior PM of 27 (90%) of 30 of dogs in the study. An example of the extent and incidence of this finding in the posterior PM is provided in Supplemental Figure 2. This finding was often accompanied by narrowing of the vascular lumen (Figures 2A and 3A). Occasionally, smooth muscle cells may be degenerate, characterized by vacuolation of the cytoplasm, fragmentation of smooth muscle cells, and/or pyknosis of the nucleus. Additionally, segmental collagen deposition in the vascular media can be observed as focal absence of smooth muscle nuclei and replacement by pale eosinophilic homogenous material. This material was highlighted as collagen using MT stain (Figures 2B and 3B). In VVG-stained sections, hypertrophied vessels were often affected by either disruption or absence of the IEL (Figures 2C and 3C). Step sections confirmed that these changes in IEL were not due to the evaluated location being near vascular branch points. Other common findings included intimal hyperplasia, with proliferation of cells and accumulation of extracellular matrix luminal to the IEL (Figure 3C).
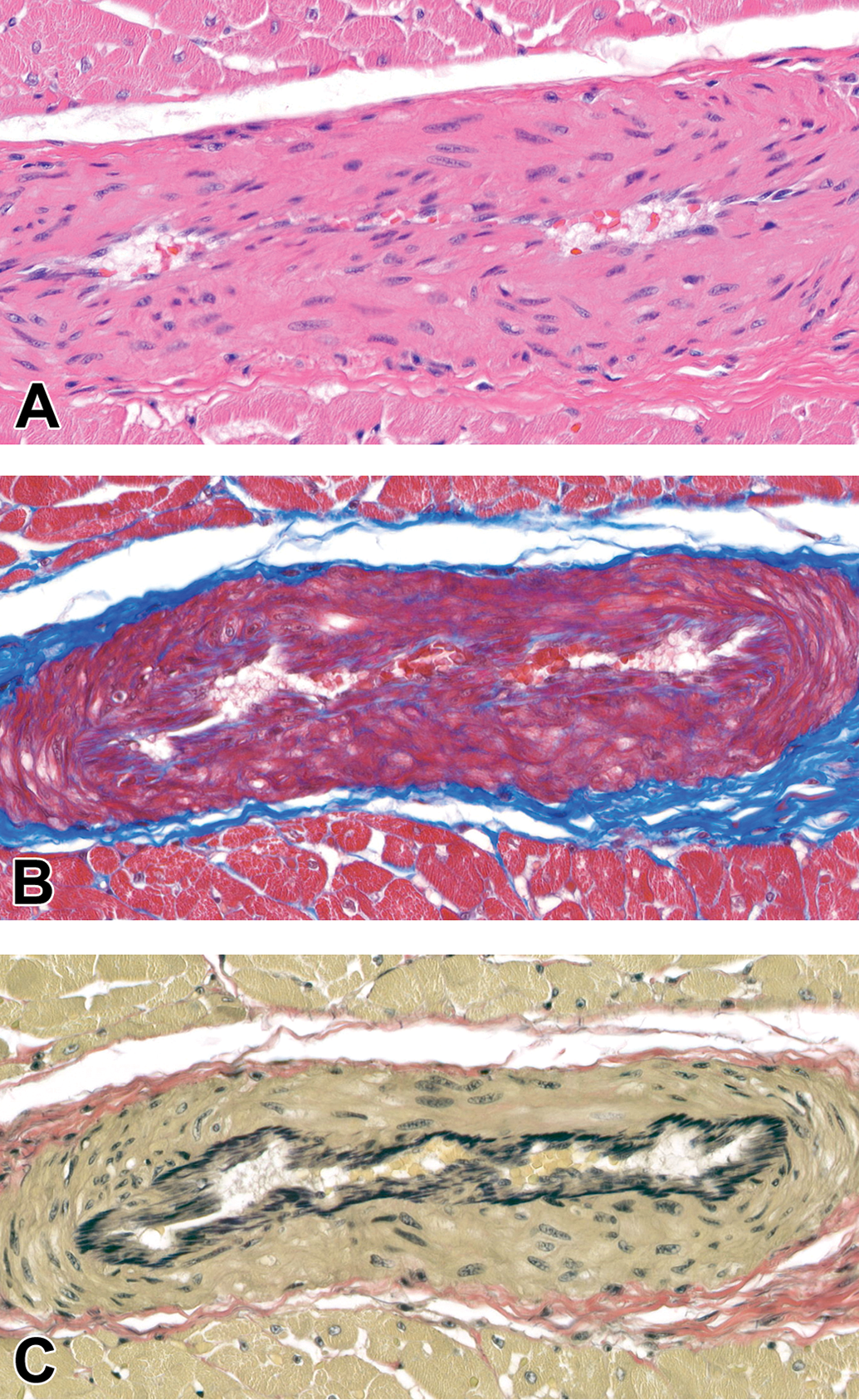
Nonremodeled intramural coronary vessel, posterior papillary muscle, dog. (A) Hematoxylin and eosin (HE), (B) Masson’s trichrome (MT), (C) Verhoeff-van Gieson (VVG). Original Objective ×40.
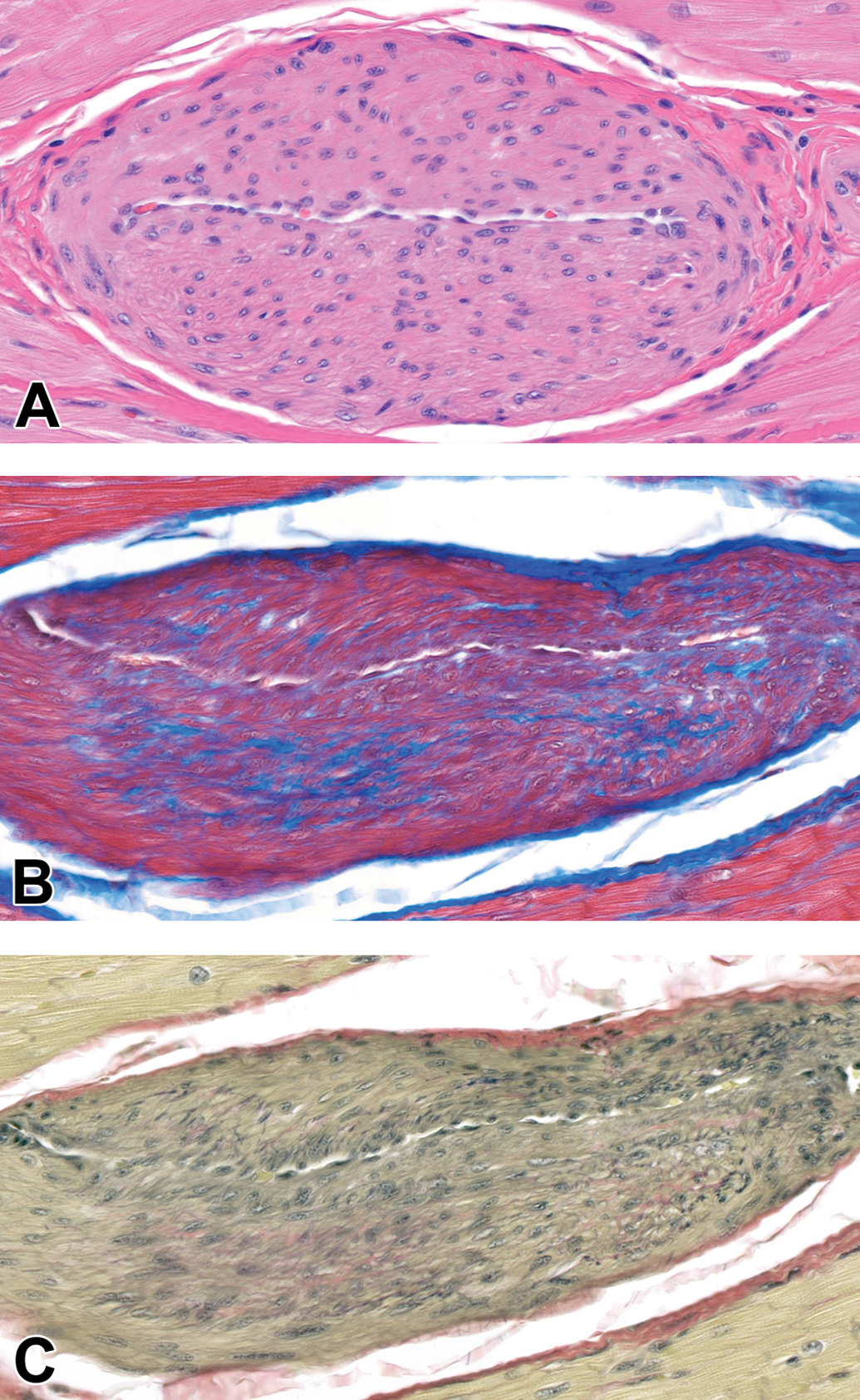
Remodeled intramural coronary vessel, posterior papillary muscle, dog (Figure 2 from same papillary muscle section as for Figure 1). A, Note irregularly thickened tunica media with enlarged smooth muscle cell nuclei characterized by open chromatin. The lumen is narrowed, hematoxylin and eosin (HE). B, Increase in deposition of collagen within the tunica media. Masson’s trichrome (MT). C, Circumferential loss of internal elastic lamina (IEL). Verhoeff-van Gieson (VVG). Original Objective ×40.
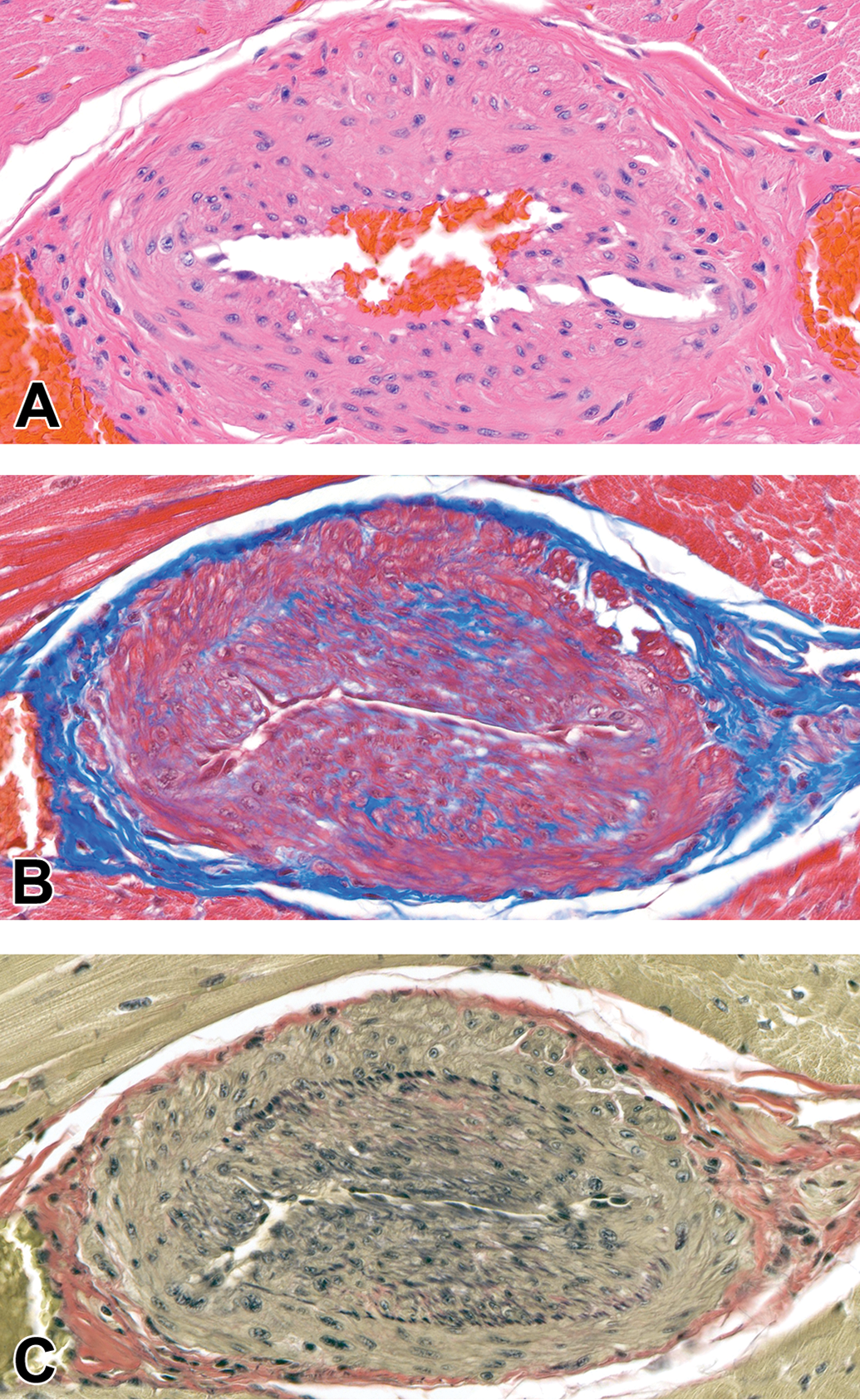
Remodeled intramural coronary vessel, posterior papillary muscle, dog. A, Note irregularly thickened tunica media with enlarged smooth muscle cell nuclei characterized by open chromatin (HE). B, Increase in deposition of collagen within the tunica media. Masson’s trichrome (MT). C, Segmental loss of internal elastic lamina (IEL) with intimal hyperplasia. Verhoeff-van Gieson (VVG). Original Objective ×40.
The sum of semiquantitative scores was compared between sections of posterior PM, which includes the posterior section of the left ventricle (interventricular septum), and the anterior muscle, which includes the anterior section of left ventricle (free wall). The composite scores ranged from 1 to a highest possible score of 9. The average HE-only score for the posterior PM was 3.46 (±1.38) and for the anterior PM was 2.16 (±1.26; P = .0003; Figure 4A). The average composite score for the posterior muscle was 5.16 (±2.15), while the average for the anterior muscle was 3.40 (±1.90; P = .0013; Figure 4B).
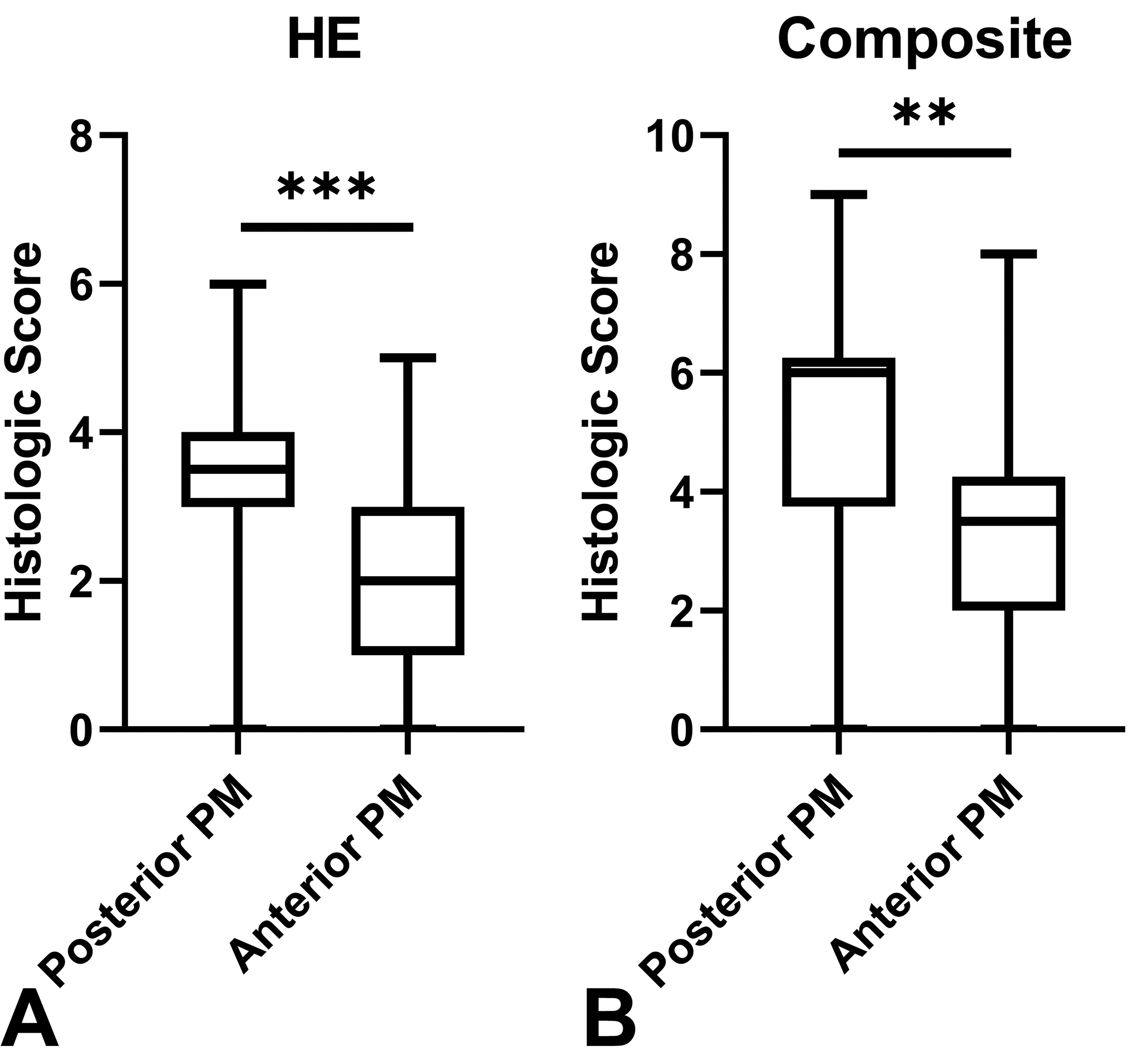
A, Semiquantitative scores of incidence of intramural coronary vascular hypertrophy in hematoxylin and eosin–stained sections. ***P = .0003. B, Composite semiquantitative scores of incidence of intramural coronary vascular hypertrophy in hematoxylin and eosin–stained and Verhoeff-van Gieson-stained sections. **P = .0013. PM = papillary muscle.
Other Spontaneous Findings
Through evaluation of 7 to 8 histologic sections for each dog in the study, additional spontaneous findings of the heart and vasculature were observed. The most common of these was focal to locally extensive minimal loose mononuclear infiltrates within the epicardial adipose tissue of the right atrial appendage, occurring in 6 (20%) of 30 dogs (Figure 5). Another common finding of the atrial appendage was so-called epicardial “fronds” on the epicardial surface. These papillary proliferations are composed of loose connective tissue and lined by mesothelium and reported as incidental findings in beagle dogs. 12,14 This finding was present in the right appendage in 5 (17%) of 30 dogs and in the left atrial appendage in 2 (7%) of 30.
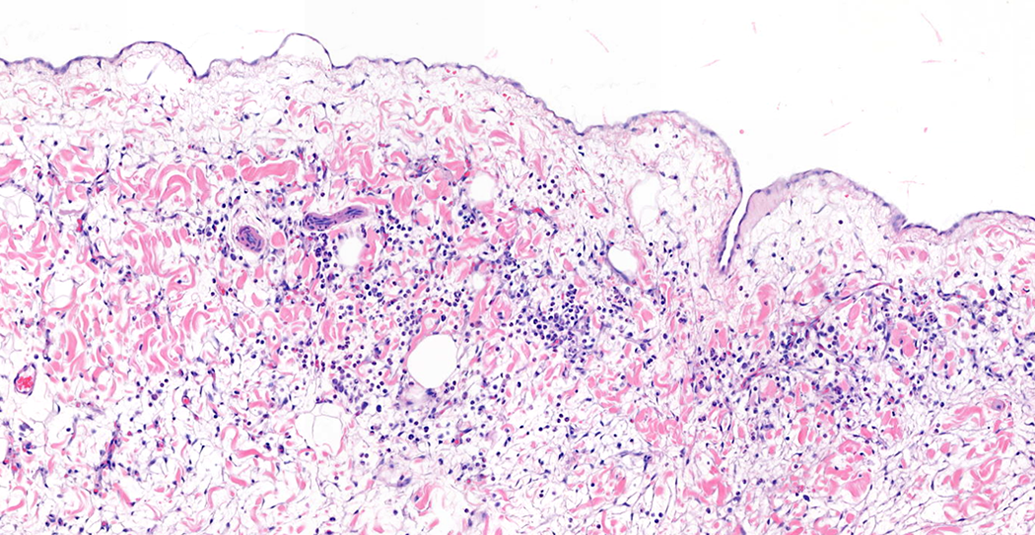
Epicardial mononuclear infiltrate, right atrial appendage, dog. Hematoxylin and eosin (HE). Original Objective ×40.
Another common finding was hemorrhage at the base of the mitral valve, which was present in 5 (17%) of 30 of dogs. Hemorrhage of the pulmonic valve was observed in one additional dog. However, this hemorrhage may be due to the passage of the Swan Ganz catheter during anesthesia. Foci of mineralization were present in the apex of the posterior PM in one dog and in the right atrial appendage in another. Finally, one dog had epicardial granulation tissue and mixed neutrophilic and histiocytic infiltrate affecting the cardiac apex. The cause of this finding was not determined, as these were naive dogs with no previous history of disease or xenobiotic exposure.
Discussion
There has been minimal description or discussion of spontaneous intramural coronary vascular hypertrophy in the PMs of dogs in the literature. Recent publications describe thickening of the tunica media of arteries as more likely to be a manifestation of postmortem hypercontraction. 12,13 This conclusion was supported in one example by lack of staining for Ki67 in the media. 12 In the current study, hypertrophied vessels were evaluated using VVG staining to examine the morphology of the IEL and the presence of intimal hyperplasia, and MT to examine the deposition of collagen within the media. Demonstration of frequent disruption or loss of the IEL in these vessels argues that this form of vascular remodeling is not artifactual, and in fact represents a physiologic response to the physical stress and microenvironmental conditions of these vessels within the PM. Step sections were performed to confirm that these changes in the IEL were not merely present at a vascular branching point. Furthermore, high incidence of intimal hyperplasia is also consistent with physiologic response to regional hypoxia and/or turbulent blood flow. 15 –17 These changes could not have arisen postmortem. Masson’s trichrome staining also demonstrated deposition of collagen within the medial tunic of many coronary vessels, also indicating a physiologic remodeling response.
Medial hypertrophy, or thickening of the vascular smooth muscle, occurs in the heart of dogs spontaneously, and as a result of pathologic processes such as systemic arterial hypertension, mitral valve disease, or subaortic stenosis. 18 –20 In general, adaptive myogenic responses in the vessel wall occur in response to chronically increased pressure. These thickened vessels allow continued blood flow despite increased perfusion pressure; however, they are more rigid and have decreased sensitivity to vasoactive mediators, thus increasing the risk for ischemia. 20 –22 In cases of systemic arterial hypertension, medial hypertrophy can predispose to infarcts, particularly in the brain of cats. 21 Also, cats with hypertrophic cardiomyopathy frequently display medial hypertrophy of intramural coronary arteries, which may further predispose to infarction in the heart due to reduced blood flow. 23 –26 In dogs with mitral valve disease, intramural infarction occurs not uncommonly, potentially as a result of reduced lumen diameter of remodeled coronary arteries causing reduced oxygen delivery, endothelial dysfunction, and increased vascular stiffness. 18,27 –29 In the young beagle dogs described in this report, medial hypertrophy occurs not as a result of an identified pathologic condition, but likely as a physiologic response to pressure within the PM.
If we accept that coronary vascular hypertrophy and remodeling are a physiologic response to the stressors of the PM, we should subsequently consider the potential consequences of this spontaneous finding. In particular, this study demonstrates that the posterior PM is more substantially affected by these coronary vascular findings as compared to the anterior PM. Although the potential effects of this observation on the range of responses to xenobiotic-induced cardiovascular effects, for example, are not yet known, it can be hypothesized that the posterior PM may be more sensitive to toxic effects that result in hemodynamic alterations which further compromise the vascular supply to the PM. For example, minoxidil and other vasodilators result in hypotension, reduced oxygen flow and perfusion particularly in the subendocardium of the PMs, resulting in ischemia. 1,2 In people who experience coronary arterial occlusion, the posterior PM is subject to higher incidences of hypoxic damage, likely related to the differential coronary perfusion to this muscle. 5 –8 Differential perfusion pressure between the PMs may be in part responsible for the coronary vascular findings presented in this study in dogs, and therefore differential responses to injury may also occur.
Based on the observations in this prospective study, there is a semiquantitative difference in the incidence of vascular findings between the 2 left ventricular PMs in young beagle dogs. The implications of this finding for the histologic evaluation of the heart in toxicity studies are multifold. First, we demonstrate a wide range of spontaneous vascular changes in the PMs of normal dogs, which should help pathologists to ensure appropriate identification of xenobiotic-induced changes in cardiac vasculature. Second, we recommend that special consideration in tissue trimming protocol be given to consistently sectioning the PM to include the base and apex (including the insertion of the chordae) of the muscle, in order to consistently and thoroughly evaluate pathologic changes. This consistency should also extend to the identification of the specific PM evaluated within a given study. A further recommendation is made to evaluate both PMs in toxicity studies when possible, in particular for those studies that evaluate a potentially cardioactive or hemodynamically active compound. This expanded evaluation of the PMs will allow for the determination of any potential disparate effects of cardioactive compounds on one or the other PM, in particular that the posterior PM may be more sensitive to certain mechanisms of toxicity, thus reducing the risk of underappreciating a toxic effect on the heart in dogs.
Supplemental Material
Supplemental Material, sj-tif-1-tpx-10.1177_0192623320963020 - Characterization of Spontaneous Vascular Findings in the Papillary Muscles of Beagle Dogs
Supplemental Material, sj-tif-1-tpx-10.1177_0192623320963020 for Characterization of Spontaneous Vascular Findings in the Papillary Muscles of Beagle Dogs by Rebecca Kohnken and Angella Weber in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-tif-2-tpx-10.1177_0192623320963020 - Characterization of Spontaneous Vascular Findings in the Papillary Muscles of Beagle Dogs
Supplemental Material, sj-tif-2-tpx-10.1177_0192623320963020 for Characterization of Spontaneous Vascular Findings in the Papillary Muscles of Beagle Dogs by Rebecca Kohnken and Angella Weber in Toxicologic Pathology
Footnotes
Acknowledgments
The authors would like to thank Amanda Wilsey, Brandan Bird, Yevgeniya Koshman, and Scott Mittelstadt from the Safety Pharmacology group for performing the anesthetized dog studies and providing heart tissue. The authors would like to thank the necropsy technician staff for providing heart tissue. The authors also thank the Histology Laboratory staff for assistance with sectioning and special stains. Finally, the authors thank Magali Guffroy for providing feedback on the draft manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Rebecca Kohnken and Angella Weber are employees of AbbVie and may own AbbVie stock. AbbVie sponsored and funded the study; contributed to the design; participated in the collection, analysis, and interpretation of data, and in writing, reviewing, and approval of the final manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
