Abstract
Dura mater defects are a common problem following neurosurgery. Dural grafts are used to repair these defects; among them are biodegradable polymeric synthetic grafts. ArtiFascia is a novel synthetic and fibrous Dural graft, composed of poly(
Introduction
Defects in the dura mater are a common problem in clinical practice. They can result from accidental or iatrogenic trauma, inflammation, tumors, or congenital malformations. 1 –4 In such cases, it is essential to create a water-tight closure of the dura in order to avoid cerebrospinal fluid (CSF) leaks, brain herniation, infections, adhesions, and cortical scarring. 5 –7 In cases where the dura mater cannot be repaired by primary closure, a dural graft can be used.
Several types of dural grafts have been used in clinical or experimental settings, including autologous tissue grafts (eg, temporal fascia, fascia lata femoris, and periosteal flaps), allografts (eg, lyophilized cadaveric dural grafts), and xenografts (eg, bovine pericardium and porcine small intestinal submucosa). 8 However, these grafts are associated with a number of drawbacks, such as limited availability, risk of infection (eg, Creutzfeldt-Jakob disease), and immunorejection. 4,9
Natural materials, such as collagen or alginate, have emerged as promising dural grafts. 10,11 Collagen grafts are biocompatible and support cell ingrowth and tissue integration. Indeed, collagen-based grafts are widely used nowadays, with several commercial products available. 12,13 Despite this, commercially available dural grafts still possess the following limitations: Up to 32% CSF leak rate and poor mechanical strength. 4,14 –17
Dural grafts should possess several important characteristics. They should support tissue regeneration and resorb while allowing endogenous neodura to grow; should not induce immunologic or inflammatory reactions; should be strong enough to provide a water-tight closure without rupturing; should be easy to handle and suture; and should be cost-effective. 4,6,16 Biodegradable synthetic materials have many of these required features, and indeed, they are gaining popularity in many different medical applications. 18 –21 Several such materials were tested as dura mater grafts. 1,4,22 –25 Nevertheless, their use is still limited, and their effects on neural tissue is still a matter of research. Therefore, there is a need for proper preclinical assessment of biodegradable materials for use as dura grafts, using appropriate animal models and by performing a detailed histopathological evaluation of the implanted tissue.
In this paper, we assessed biodegradability and local tolerance of ArtiFascia (Nurami Medical), a novel synthetic dural graft composed of poly(
Animals, Materials, and Methods
Test Item
The test item was ArtiFascia, a novel synthetic fibrous scaffold designed for use as a dura mater graft that has a porous structure. ArtiFascia matrix is composed of PLCL and PDLCL.
Control Item
Commercially available dural repair matrix made of processed bovine collagen (originating from bovine deep flexor tendon). The matrix is porous.
Animal Model
Eighteen HsdOkd:NZW male rabbits, approximately 3 to 5 months old, were obtained from Envigo RMS. The rabbits were housed individually and provided with approximately 100 g/rabbit/day Teklad 7078S rabbit diet and free access to drinking water, supplied to each cage via polyethylene bottles with stainless steel sipper tubes. Automatically controlled environmental conditions were set to maintain the temperature at 17 °C to 23 °C with a relative humidity of about 30% to 70%, a 12-hour light/12-hour dark cycle and 15–30 air changes/hour.
Experimental Design
Prior to the surgical procedure, the animals were administered an opioid analgesic (Buprenorphine at a dose level of ∼0.05 mg/kg) and Enrofloxacin (Baytril, Bayer) at a dose of ∼5 mg/kg by subcutaneous injection. Afterward, the animals were anesthetized by an intramuscular injection of a mixture of Ketamine and Xylazine (35 and 5 mg/kg, respectively). Following sedation, the animal’s tail was clipped to enable the placement of an O2 saturation probe, and a catheter was introduced to its ear vein. Throughout the surgical procedure, animals were monitored for heart rate and blood O2 saturation level and inhaled with 100% O2 at a rate of 1 to 1.5 L/min via face mask. Additionally, Lactated Ringer’s solution was administered intravenously (IV) throughout the operation. Anesthesia was enhanced by Isoflurane 0.5% to 2% delivered via the oxygen mask. Following sedation, the fur was clipped from the bridge of the snout between the eyes to the caudal end of the skull/calvarium using electric clipper. The clipped area was then scrubbed using 4% wt/vol Chlorhexidine Gluconate (SEPTAL SCRUB, Teva Medical) and wiped with ethanol 70%.
For dural defect induction and implantation procedures, the animals were placed on a heated surface in ventral recumbency and ∼0.3 mL of 1% lidocaine was injected subcutaneously along the sagittal midline of the skull. A midline skin incision was made over the scalp from the nasal bone to just caudal to the middle sagittal crest to obtain visualization of the calvarium. The periosteum covering the calvarium was sharply divided down the sagittal midline and pushed laterally while elevated from the underlying skull. The soft tissue was retracted to expose the underlying parietal bones and drilling area was irrigated with physiological saline. An 8-mm hole was drilled on each side of the parietal bone penetrating through the calvarial bone using a trephine burr with constant saline irrigation to the level of the dura. To enable repositioning of the bone piece in its original orientation, a small hole (approximately 2.0 mm in diameter) was drilled through the outer surface of the bone at the area closest to the nose (ie, at “12 o′clock”). A round piece measuring 4 mm in diameter of the underlying dura mater was removed from each of the formed bone defects using a biopsy punch and scissors. A 6-mm piece of either ArtiFascia or Control was placed without suturing on top of each of the formed dural defects to obtain complete coverage. Control graft was moistened with physiological saline according to its instructions of use. Bone flaps were then repositioned into the bone defects. The sites of drilling were photographed to enable identification of the implantation areas at termination. The periosteum and subcutaneous tissues were sutured with Vicryl 3-0 sutures and the skin was closed with surgical clips. Upon incision closure, the operated area was rinsed with physiological saline to remove remaining blood.
Postoperatively, animals were administered subcutaneously with buprenorphine at a dose of ∼0.05 mg/kg twice daily and Enrofloxacin (Baytril) at a dose of ∼5 mg/kg once daily for up to 3 days post-surgery. An Elizabethan collar was placed for 7 days on the animals’ head to prevent self-mutilation.
Animals were monitored clinically for the duration of 1, 6, and 12 months following implantation (consisted of 4, 6, and 8 animals in total for each termination time point, respectively, see Table 1). The study was performed after receiving approval from the National Council for Animal Experimentation (No. IL-16-03-72).
Experimental Design.
Viability, Clinical Signs, and Body Weight
Examination of any systemic adverse effects or abnormal clinical signs (eg, changes in gait, posture, and neurological abnormalities such as the presence of bizarre behavior, tremors, convulsions, sleep and coma, etc) was carried out on the day of surgical procedure following recovery from anesthesia, daily for the first-week postimplantation and once weekly thereafter until the respective termination point.
Additionally, examination of local reaction (eg, swelling and discoloration) at the incision area as well as cage side clinical observations for viability or overt clinical signs are recorded once each day on regular working days throughout the entire observation period.
Determination of individual body weights of animals was made on arrival, on the day of surgical procedure, once weekly thereafter, and on the respective sacrifice day.
Necropsy and Tissue Handling
At each termination time point, the rabbits were euthanized by an IV overdose of Na Pentobarbitone. Following termination, the skin and soft tissue at the top of the skull were removed to enable visualization of the calvarial bone. The drilling sites were identified and evaluated macroscopically for alterations in the tissue (Figure 1). Additionally, both drilling areas were marked using India Ink and photographed.
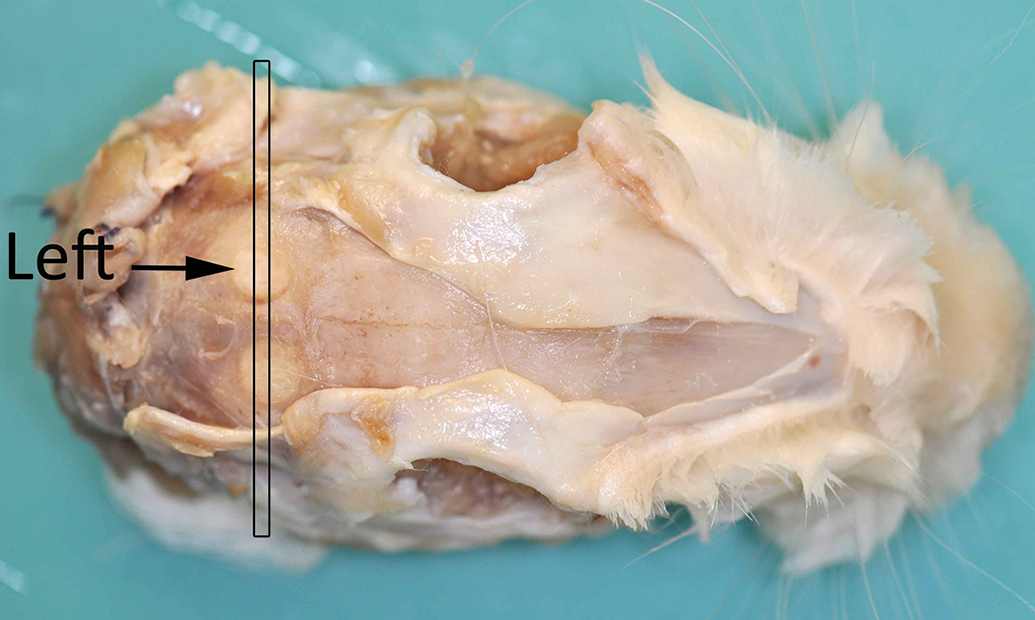
Gross appearance of the defect sites at necropsy.
Animals were subjected to perfusion performed with physiological saline followed by 10% neutral-buffered formalin administered to the carotid artery using a high-pressure infusion bag with the concurrent severing of the jugular vein. The entire head with the brain and both right and left deep cervical lymph nodes were collected following removal of the skin and mandibles.
The skull and brain were trimmed in the coronal plane on either side of the defects, with the implanted devices in situ, using an Exakt diamond band saw. Following decalcification, the skull was trimmed as shown, to include both implantation sites and adjacent brain in one section (Figure 2). Samples were processed xylene-free and embedded in paraffin, sectioned approximately at 5-µm thickness and stained xylene-free with hematoxylin and eosin to produce one slide per implantation site (ie, left and right).
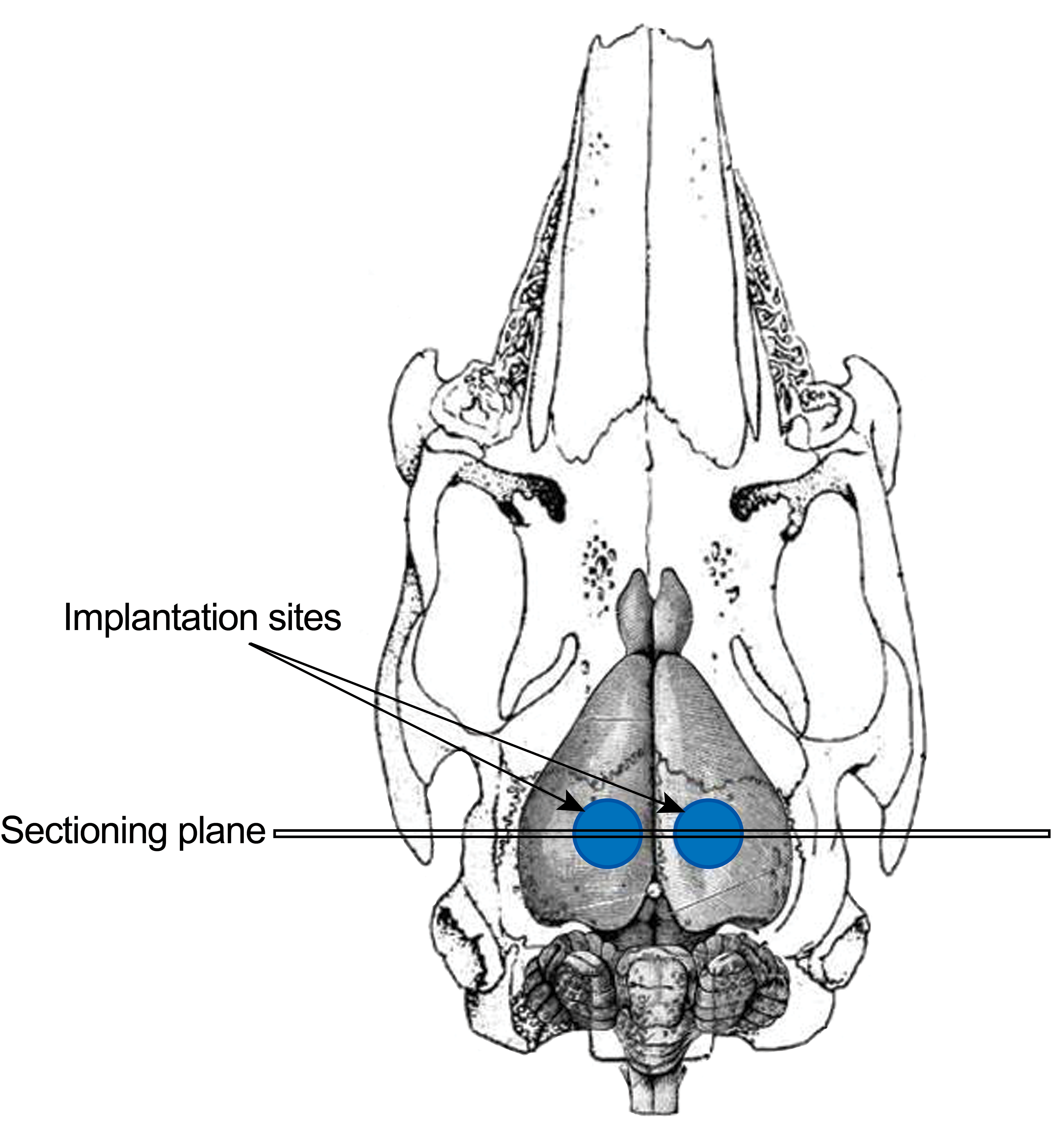
A schematic drawing of the histology sectioning plane.
Histopathological Evaluation
The biological response was assessed and recorded per the following parameters and grading scales:
Number and distribution as a function of distance from the material/tissue interface of the inflammatory cell types, namely, polymorphonuclear neutrophilic leucocytes, lymphocytes, plasma cells, eosinophils, macrophages, and multinucleated cells (Table 2)
Presence, extent, and type of necrosis, scored according to the following grading scale: 0 = none, 1 = minimal, 2 = mild, 3 = moderate, 4 = severe.
Extent of neovascularization, fibrosis/fibrous capsule, and fatty infiltrate (Table 3).
Other tissue alterations (eg, fibroplasia, thrombosis, and foreign body reaction).
Quality and quantity of tissue ingrowth.
Histological Evaluation System for the Cell Type/Response.
a phf, per high powered (400×) field.
Histological Evaluation System for the Tissue Response.

Where applicable, the severity of histopathological findings was graded using semiquantitative grading scale of 5 grades as follows: 0 = no lesion, 1 = minimal change, 2 = mild change, 3 = moderate change, and 4 = marked change.
The amount of residual implanted material was evaluated and scored according to the following grading scale 26 : 0 = No apparent degradation; 1 = Minimal degradation of implant, some minor dissolution on edges, cracks in implant and/or small fragment present; 2 = Moderate degradation of implant with cracks in implant and/or some fragments; 3 = Marked degradation of implant with the presence of several fragments; and 4 = Abundant degradation of implant with (almost) complete fragmentation.
Neodura is defined by a well-delineated layer of densely collagenous connective tissue situated between newly formed bone and the brain surface or deep within the surgical fibrous scar that is parallel to the leptomeninges and of a thickness of the order of the normal dura. 27 A fibrous scar is typically thicker, not tangentially polarized and follows the surgical planes rather than the brain surface.
Results
Survival, Clinical Observations, and Body Weight Gain
Animals were separated into Control and Test subgroups according to Table 1. No treatment-related mortality or abnormal clinical signs occurred in any of the animals throughout the 12-month observation period.
One animal from the control 12-month group was sacrificed on humane grounds at 11 months post-op due to lack of appetite and significant weight loss. At necropsy, trichobezoar (ie, hair ball) in the stomach was found, which was most likely associated with liver disease. This incident was considered to be nontreatment-related. Three animals were replaced due to nontreatment-related surgical complications or animal’s death during recovery from anesthesia.
Intraoperative observations consisted mainly of slight to mild bleeding detected either following removal of the bone or during defect induction in the dura with similar incidence in both groups.
Additionally, in 2 implantation sites of each group, the drilled bone flap was broken in half at its depth (ie, to upper and lower parts), and both halves were repositioned on top of each other. In 1 implantation site subsequently implanted with ArtiFascia, similar bone fracture occurred yet the lower part was removed by pieces thus only the upper part of the bone was repositioned. Mean body weight was comparable between the 2 groups throughout the 12-month follow-up period.
Macroscopic Observations
No abnormalities were detected during macroscopic examination of the drilling areas in the skull and the deep cervical lymph nodes in any of the animals.
Microscopic Observations
One-month termination point
In the Control-implanted group, there was a minimal presence of macrophages and occasional multinucleated giant cells, lying at the interface with the collagenous implanted material, which characteristically was contiguous with the native dura mater (Figure 3A and B and Supplemental Tables 1 and 4). A minimal fibrotic reaction was present within the implanted material. Few (minimal) polymorphonuclear cells were dispersed within the implanted material. No other inflammatory reaction was present in the adjacent brain tissue. No abnormalities were detected in the lymph nodes.
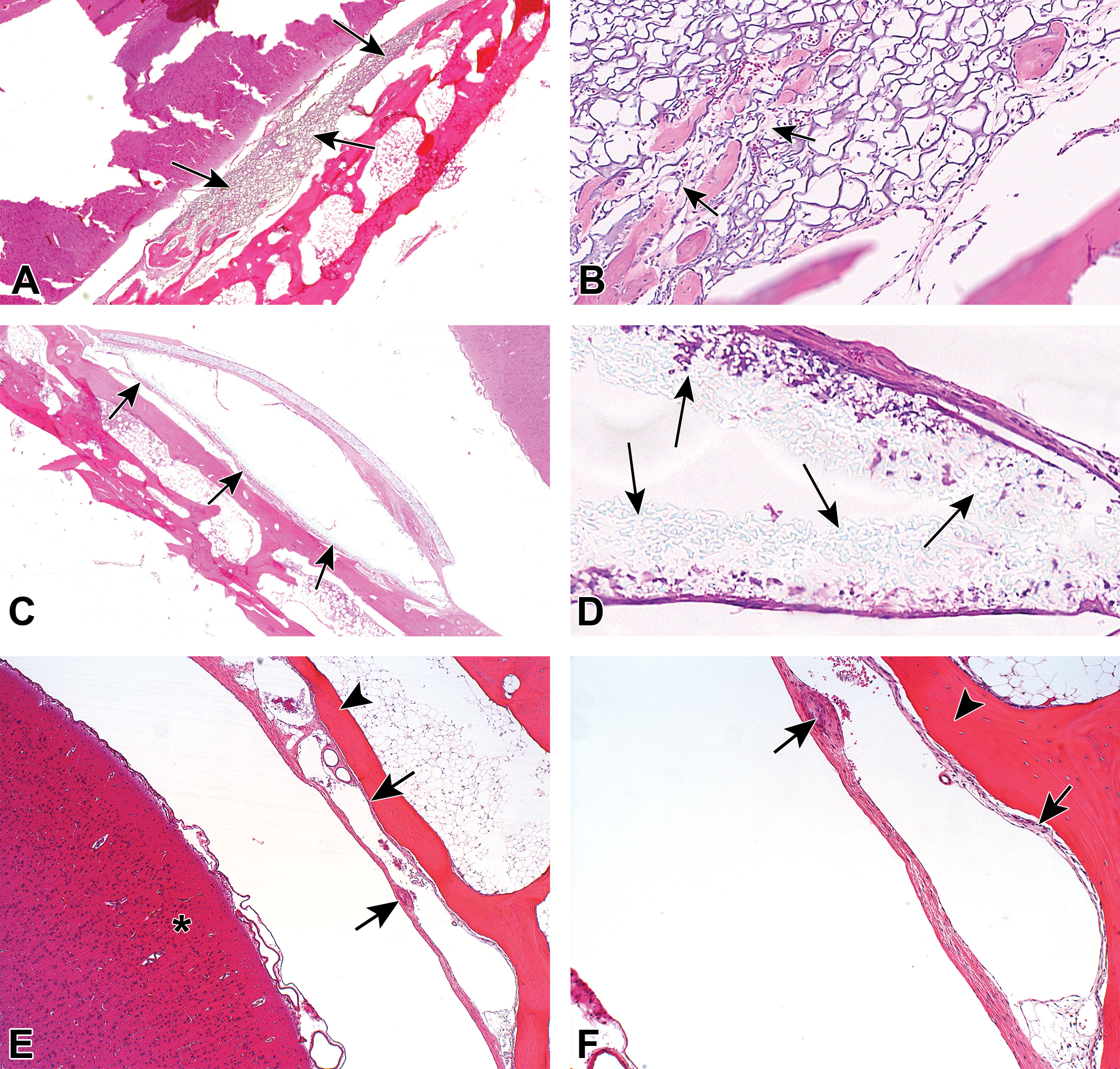
A-B, Animal implanted with Control 1-month postimplantation. Low (A) and high (B) magnification view of the implantation site. There is only a minimal (grade 1) presence of macrophages lying at the interface with the implanted material. A minimal fibrotic reaction is present within the implanted material (arrows). Few (minimal) polymorphonuclear cells are dispersed within the implanted material. No other inflammatory reaction is present in the adjacent brain tissue. At the periphery of the 8-mm hole drilled at the parietal bone (ie, penetrating through the calvarial bone), newly reactive bone can be seen. C-D, Animal implanted with ArtiFascia, 1-month postimplantation. Low (C) and high (D) magnification views of the implantation site. The arrows indicate the capsule surrounding the implanted device (C) and the implanted device (D) located at the dura mater. There is only a minimal presence of macrophages and giant cells, surrounded by a capsule. No other inflammatory reaction is present within the capsule. E-F, Animal implanted with Control, 6-month postimplantation. Low (E) and high (F) magnification views of the implantation site. The arrows indicate the presence of minimal fibrotic capsule surrounding the implanted device (ie, neodura), located at the dura mater level. The implanted device is completely degraded, leaving an empty space surrounded by a thin capsule. No inflammatory reaction is present within the capsule or in the adjacent brain tissue. Externally, the site of implantation is completely covered with newly formed mature bone (arrowhead). Asterisk indicates the brain. G-H, Animal implanted with ArtiFascia, 6-month postimplantation. Low (G) and high (H) magnification views of the implantation site. The implanted device is overlying the dura mater, composed of 2 layers (black arrows), surrounded by fat tissue (dash arrows), with the presence of small islands of trabecular bone (white arrows). Each of the layers of the implanted device is composed of a dense mesh of convoluted and tightly intertwined clear fibers, which is consistent with the polymer nature, associated with a minimal presence of macrophages and multinucleated giant cells. No other inflammatory reaction is present within the cavities, capsule, or in the adjacent brain tissue. The site of implantation is completely covered with newly formed mature bone. The presence of neodura formation at the site of operation is suggesting progressive healing of the previously induced dural defect. I-J, Low (I) and high (J) magnification views of the Control implantation site 12 months postimplantation. The implanted device was completely degraded. No inflammatory reaction is present at the site of implantation or in the adjacent brain tissue. The site of implantation is completely covered with newly formed mature bone with leptomeninges over the brain. K-L, Animal implanted with ArtiFascia, 12-month postimplantation. Low (K) and high (L) magnification views of the implantation site. Within the dura mater, the implanted device is identified, and its cavities filled with macrophages and giant cells (arrows), surrounded by a thin mature fibrotic capsule. There is increased trabecular bone formation (arrowheads) when compared to the previous time points, indicative of time-related increased bone maturation. The implanted device is filled with relatively abundant macrophages and multinucleated giant cells (grade 2, mild), with no evidence for the dense mesh of convoluted and tightly intertwined clear fibers. This characteristic is suggesting complete absorption of the polymeric component of the device. No other inflammatory reaction is present within the cavities, capsule, or in the adjacent brain tissue, indicating good tolerability. The site of implantation is completely covered with newly formed mature bone. There is evidence for neodura formation at the site of operation, suggesting complete healing of the previously induced dural defect.
In the ArtiFascia-implanted group, there was minimal fibrotic capsule surrounding the entire implanted device, located at the dura mater level (Figure 3C and D and Supplemental Tables 1 and 4). There was minimal presence of macrophages and multinucleated giant cells, lying over the capsule. The remnants of the implants could be easily identified as a dense mesh of convoluted and tightly intertwined clear fibers, consistent with the implant structure, intermixed with the minimal macrophage reaction. No other inflammatory reactions were present within the capsule or in the adjacent brain tissue. No abnormalities were detected in the lymph nodes.
Six-month termination point
In the Control-implanted group, the site of implantation was identified within the dura mater level surrounded by a minimal fibrotic capsule (Figure 3E and F and Supplemental Tables 2 and 4). The implanted device was completely degraded, leaving an empty cavity surrounded by a thin capsule. There was evidence for progressive neodura formation at the site of operation, suggesting healing of the previously induced dural defect. No inflammatory reaction was present within the capsule or in the adjacent brain tissue. The site of implantation was completely covered externally with newly formed mature bone. No abnormalities were detected in the lymph nodes.
In the ArtiFascia-implanted group, the implanted device was still identified (ie, degradation score 2) within the dura mater level (Figure 3G and H and Supplemental Tables 2 and 4). Each layer of ArtiFascia was composed of a dense mesh of convoluted and tightly intertwined clear fibers, which was consistent with the implant structure, associated with minimal presence of macrophages and multinucleated giant cells. No other inflammatory reactions were present within the cavities, capsule, or in the adjacent brain tissue. There was evidence for neodura formation at the site of operation, suggesting healing of the previously induced dural defect, without any evidence of on-going active inflammatory reaction. The site of implantation was covered externally with the calvarium. Higher magnification indicated that new bony trabecula, fibrotic, or fat tissue septa were noticed within the implanted device and/or underneath the implant (Figure 3H and Supplemental Tables 2 and 4). The new bony trabecula growth was strictly localized to the site of the implant, without evidence for extension to, and/or compression of the underlying brain. No abnormalities were detected in the cervical lymph nodes.
Twelve-month termination point
In the Control-implanted group, the implanted device was completely degraded. No inflammatory reaction was present at the site of implantation or in the adjacent brain tissue. Externally, the site of implantation was completely covered with newly formed mature bone. Toward the brain, there was evidence for neodura formation at the site of operation, suggesting healing of the previously induced dural defect (Figure 3I and J and Supplemental Tables 3 and 5).
In the ArtiFascia-implanted group, the implanted device was identified, composed of layers of cavities filled with monomorphic population of macrophages (which are consistent with an anti-inflammatory M2-like phenotype), and giant cells, surrounded by a thin mature fibrotic capsule, the neodura, formed at the site of the implantation. The lack of remnants of the implanted graft suggests complete absorption of the polymeric component of the device. No other inflammatory reactions were present within the implantation site, capsule, or in the adjacent brain tissue, indicative of good tolerability. The site of implantation toward the brain was completely covered with neodura (ie, fibrotic capsule) suggesting complete healing of the previously induced dural defect. Additionally, trabecular bone formation was also observed within and around the implanted device, comparable to the previous time points, indicative of time-related integration (Figure 3K and L and Supplemental Tables 3 and 5).
Discussion
This study shows that ArtiFascia, a novel dura mater graft composed of PLCL and PDLCL, is safe at 1, 6, and 12 months after implantation as a dural graft. No evidence for local irritation (inflammation) was noted, suggesting excellent local tolerability of ArtiFascia. The presence of histiocytic reaction is the expected phagocytic and foreign body reaction to biodegradable materials. 18 –21,28 The macrophage population observed at the absorption sites of the ArtiFascia-implanted sites were monomorphic, which is consistent with an anti-inflammatory M2-like phenotype: abundant cytoplasm indicative of phagocytic activity. The M2 macrophages are instrumental in tissue repair through the promotion of neovascularization, phagocytosis, and fibrosis. 29,30 There is no literature on M2 morphometry in the rabbit or other animal species; therefore, this distinction was based on indirect literature describing the M2 phenotype in humans. 31,32
Such tolerability for the materials that compose ArtiFascia, PLCL and PDLCL, has been shown previously in other tissues, and they are Food and Drug Administration–approved for orthopedic applications and wound closures. 33,34 Although for these applications, there is a relative abundance of data to show their local tolerance in the tissue, their effect on dura mater has been reported only sparsely in the literature.
A device based on PLCL electrospun fibers was found to be safe in a rat neurosurgical spine procedure model and was effective in reducing scar formation when followed for 30 days by reducing epidural fibrosis in the damaged dura. 35 Yamada et al tested a copolymer composed of PLCL and poly(glycolic acid) in 31 rabbits with dural defects. 23 The rabbits were followed for 26 months, demonstrating the formation of a dura-like tissue in the area of implantation, without the presence of inflammation. The same material was also tested in 20 patients during neurosurgical operation, and although the transient subcutaneous collection of CSF was found in 3 patients, a computed tomography scan performed more than 6 months following implantation did not show any abnormal findings. 36 Our study further confirms the local safety and tolerability of a PLCL/PDLCL biodegradable material when implanted as a substitute for the dura mater in rabbits. Furthermore, no evidence for abnormalities in the local lymph nodes were noted both macroscopically and histologically in any of the operated animals.
ArtiFascia and the Control device are indicated to repair human dura, a 0.5- to 1-mm-thick soft connective tissue. The rabbit’s dura is significantly thinner (99 µm in rabbit vs 564 in human), 27 and we estimated that this structural difference caused both implants to occupy the space where once the drilled bone piece was located. Consistent with this, Figures 3E and F show a relatively large empty cavity where the Control graft was implanted. The formation of new bone trabecula within and around the implantation sites suggests that this space is refilled with new bone as part of a normal wound healing process. Owing to the relatively slower degradation time of ArtiFascia vs Control observed in this study (6-12 months vs <6 months, respectively), reactive bone that was observed after 1 month at the periphery of the Control (Figure 3A and B), increased over time in the ArtiFascia (Figure 3G and H). These observations suggested that both Control and ArtiFascia did not interfere with normal bone growth at the site of implantation in the rabbit model. The promotion of well-ordered local trabecular bone induction at the implantation site contributes to the healing and enhances the integrity of the induced bone defect without interfering with the growth of a new Dura.
Though the current study did not assess or compare the performance of the grafts in terms of CSF leakage, neodura regeneration and bone growth, the generation of fibrotic capsule surrounding the entire implanted device (ie, neodura) observed at 1-month follow-up (Figure 3C and D), suggests that ArtiFascia has the potential to rapidly generate a water-tight seal at the implantation site and help reduce CSF leakage rates. This proposal is supported by the observation that no CSF leakage was observed in a clinical study of ArtiFascia that included 6 patients with a follow-up of 6 months (data not published). Further studies are required to assess the performance of ArtiFascia in animal and clinical settings. Ideally, degradation dynamics should be similar between test and Control grafts in a comparison study. Degradation time of ArtiFascia was longer than the Collagen grafts used here. Longer degradation time can have several advantages, among them, longer period of leakage prevention and longer period where the graft may serve as a scaffold for tissue regeneration. This can be particularly important when healing is impaired following chemotherapy or radiotherapy. Occasional ruptures that were observed in ArtiFascia at 6 months point (see Figure 3H) suggest that ArtiFascia performance is longer than the tested Collagen graft but is still limited to several months. This is a typical degradation profile of the polymers that comprise the synthetic graft. In summary, the current study supports the use of PLCL/PDLCL biodegradable materials, and specifically ArtiFascia, as substitutes for dura mater after dural damage, based on its favorable local tolerability and biodegradability.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_0192623320947075 - Local Tolerance and Biodegradability of a Novel Artificial Dura Mater Graft Following Implantation Onto a Dural Defect in Rabbits
Supplemental Material, sj-docx-1-tpx-10.1177_0192623320947075 for Local Tolerance and Biodegradability of a Novel Artificial Dura Mater Graft Following Implantation Onto a Dural Defect in Rabbits by Yuval Ramot, Sagi Harnof, Ido Klein, Netanel Amouyal, Michal Steiner, Nora Nseir Manassa, Amir Bahar, Serge Rousselle and Abraham Nyska in Toxicologic Pathology
Footnotes
Acknowledgments
The authors thank Armando Tellez for critical review of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Nora Nseir Manassa and Amir Bahar are employees of Nurami Medical Nanofiber Technology. Yuval Ramot and Serge Rousselle have received consultation fees from Nurami Medical Nanofiber Technology. Sagi Harnof is a member of the Scientific Advisory Board of Nurami Medical Nanofiber Technology.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Nurami Medical Nanofiber Technology.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
