Abstract
The equine hoof capsule, composed of modified epidermis and dermis, is vital for protecting the third phalanx from forces of locomotion. There are descriptions of laminitis, defined as inflammation of sensitive hoof tissues but recognized as pathologic changes with or without inflammatory mediators, in the earliest records of domesticated horses. Laminitis can range from mild to serious, and signs can be acute, chronic, or transition from acute, severe inflammation to permanently abnormal tissue. Damage within the intricate dermal and epidermal connections of the primary and secondary lamellae is often associated with lifelong changes in hoof growth, repair, and conformation. Decades of research contribute to contemporary standards of care that include systemic and local therapies as well as mechanical hoof support. Despite this, consistent mechanisms to restore healthy tissue formation following a laminitic insult are lacking. Endogenous and exogenous progenitor cell contributions to healthy tissue formation is established for most tissues. There is comparably little information about equine hoof progenitor cells. Equine hoof anatomy, laminitis, and progenitor cells are covered in this review. The potential of progenitor cells to advance in vitro equine hoof tissue models and translate to clinical therapies may significantly improve prevention and treatment of a devastating condition that has afflicted equine companions throughout history.
Introduction
Aristotle described the relationship between equine laminitis, commonly called “founder,” and overfeeding of barley around 350 BC. 1 Laminitis is a multifactorial condition in which there is disruption of the distal phalangeal suspensory apparatus at the dermal–epidermal junction secondary to pathologic inflammatory or endocrine cascades or vascular compromise. 2 -4 Based on a systematic review of published evidence, the incidence of laminitis varies widely, 1.5% to 34%, depending on the specific equine population evaluated. 5 There has been relatively little progress to prevent or reverse this devastating disorder, despite decades of research that have contributed to helpful therapeutic interventions. 6 Efforts are complicated by the numerous conditions associated with laminitis which range from intestinal obstruction and severe unilateral lameness to dietary insults and both endocrine and metabolic diseases. 7,8 Regardless of the inciting cause, lamellar tissue within a hoof affected by laminitis often does not return to normal, and the affected hoof may require continuous management to maintain comfort and some degree of function. 9,10 Contemporary treatments for laminitis typically consist of one or more systemic and local therapies as well as mechanical support of the laminitic tissue. 6,10 Increased awareness and application of novel approaches to study, prevent, and treat laminitis, including use of progenitor cells to create ex vivo models, will significantly augment the current armamentarium against this devastating condition.
Adult multipotent stromal cells (MSCs) are found in most mammalian tissues including the equine hoof. 11 The immature cells can differentiate into diverse cell lineages and exhibit variable immune privilege and self-renewal capabilities. 12 Lamellar tissue formed after severe laminitis contains dyskeratinized epidermal cells, suggesting abnormal formation and/or maturation of progenitor cells. 13 Much of the adult equine MSC research is focused on those from bone marrow, umbilical cord blood, and adipose tissue, with the intent to promote healing in bone, tendon, or cartilage. 14 -16 However, equine hoof progenitor cells are largely unexplored, and they may provide a novel target for study, prevention, and treatment of laminitis.
Equine Hoof Structure
The equine hoof capsule is a cornified, modified epidermis consisting of 5 regions: periople, wall, white line, sole, and frog. The wall has 3 layers: from the outside, they are the stratum externum (tectorium), stratum medium, and stratum internum (lamellatum; Figure 1). The stratum externum is a few millimeters of cornified tissue formed by germinal cells in the perioplic corium located superficial to the coronary corium that produces the stratum medium. 17 The thick stratum medium is composed of pigmented tubular and intertubular horn. 18 As new cells form in the coronary corium, existing cells keratinize and eventually cornify as they move distally. 19
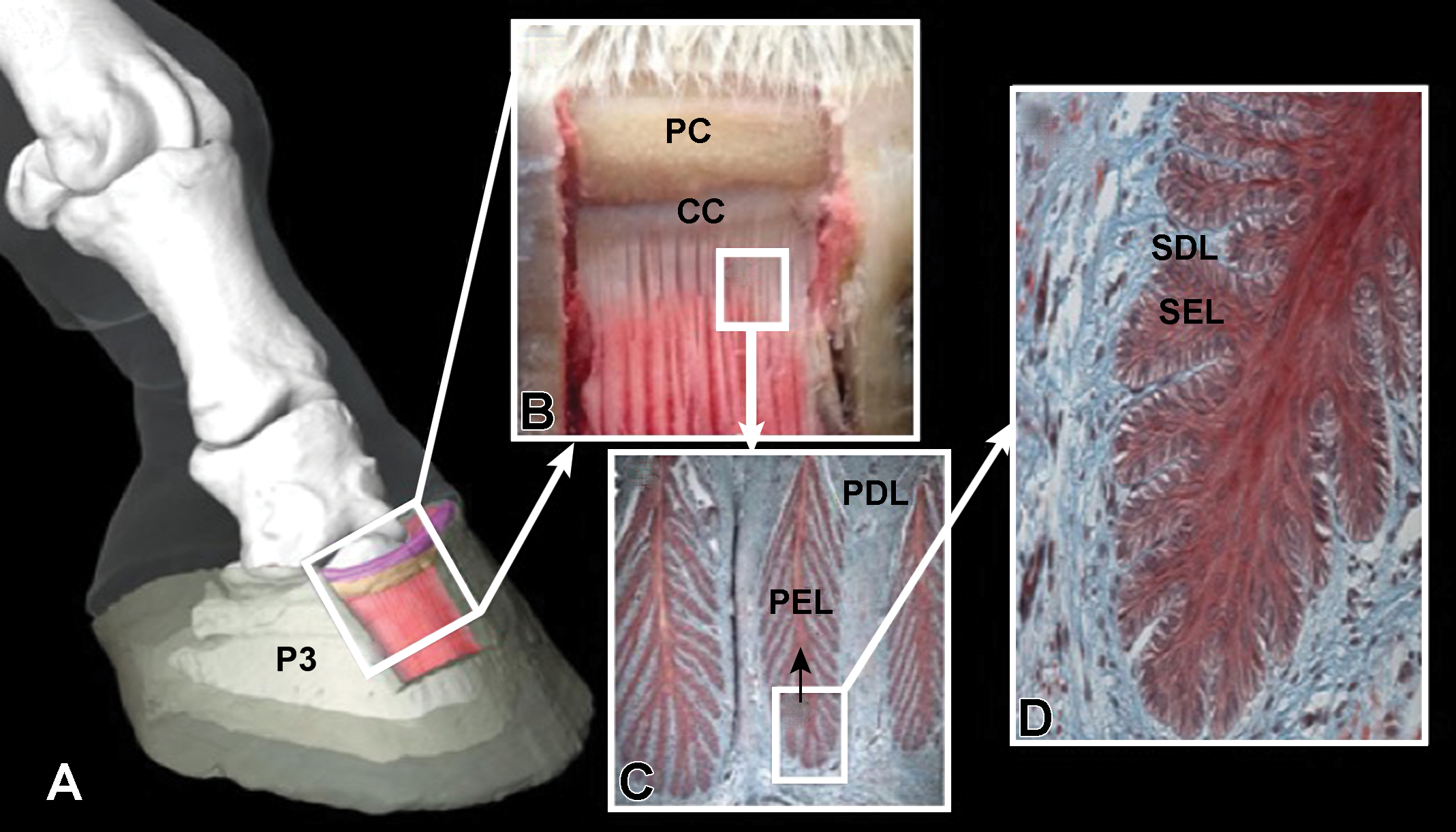
Illustration of the internal components of the equine hoof (A) in images with no (B) to progressively higher magnification (C, original objective 10×; D, original objective 20×). The perioplic corium (PC; B) and coronary corium (CC; B) are shown in addition to the primary epidermal lamellae (PEL; C, black arrow) and primary dermal lamellae (PDL; C) and the secondary epidermal lamellae (SEL; D) and secondary dermal lamellae (SDL; D). P3 = third phalanx.
The focus of this review, the stratum internum, is composed of epidermal and dermal lamellae that are continuous with the tubular horns of the stratum medium and the periosteum of the distal phalanx, respectively. The suspensory apparatus of the distal phalanx is a network of collagen bundles which connects the distal phalanx to the stratum internum and permits the hoof wall to act as a principal weight-bearing structure. 20 Collagen bundles extend from the distal phalanx and insert on the lamellar basement membrane between the secondary dermal and epidermal lamellae. 21 While the epidermis is nonsensitive and avascular, the dermis has arteries, veins, nerves, and capillaries in a dense matrix of tough connective tissue. 22,23 Both epidermal and dermal lamellae have frond-like shapes, primary lamellae, that have smaller frond-like structures, secondary lamellae, on their surface. The junction between the epidermal and dermal components of the lamellae is a complex basement membrane to which epithelial basal cells are attached via hemidesmosomes (Figure 2) or electron-dense adhesion plaques, and the dermis is connected via insertion of anchoring fibrils composed of proteins such as collagen and laminin. 24,25 The highly complex morphology of the primary and secondary lamellae can vary regionally, likely due to different functions and load-bearing stresses. 26
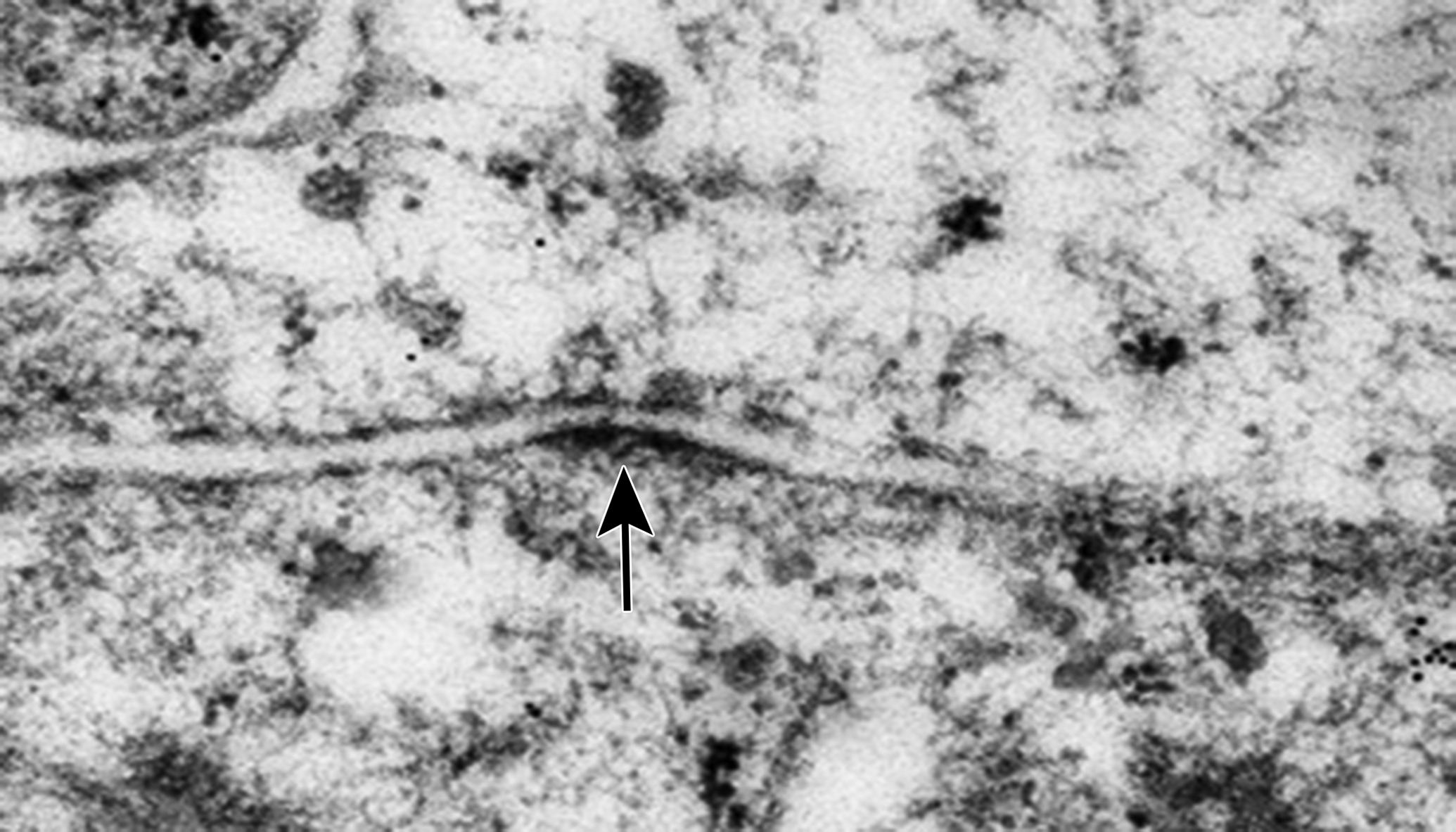
Transmission electrophotomicrograph of an electron-dense, desmosome-like structure (arrow) between cultured cells from equine hoof stratum lamellatum.
Keratins
Keratins are the major structural proteins, largest subgroup of intermediate filament proteins, and vital cell signaling participants in epithelial cells. They are nonsoluble proteins with distinct physicochemical properties, intermediate structures, and mechanical behaviors that broadly divide them into soft or hard keratins and contribute to the variable properties of different hoof regions. 27 -29 Disulfide bonds within keratin filaments convey strength and hardness proportionate to the number of bonds present, and hard keratins tend to have more than soft. 30 Amino acid sequences determine the secondary, tertiary, and quaternary structures of keratins. 31 They are often combined with other proteins such as keratin filament-associated proteins and mineral binding enzymes and can vary from fibrous to amorphous. 32,33 In the epidermis, keratins produced by keratinocytes exist as keratohyaline granules within the cells. 34 Keratin 14 is a cytokeratin typically found in the cytoskeleton of epidermal cells, and it is expressed by basal cells between the secondary epidermal and dermal lamellae (Figure 3). As the names imply, soft keratins are typically the largest component of relatively soft cornified epithelia, while hard keratins are the major population in hard cornified tissue.
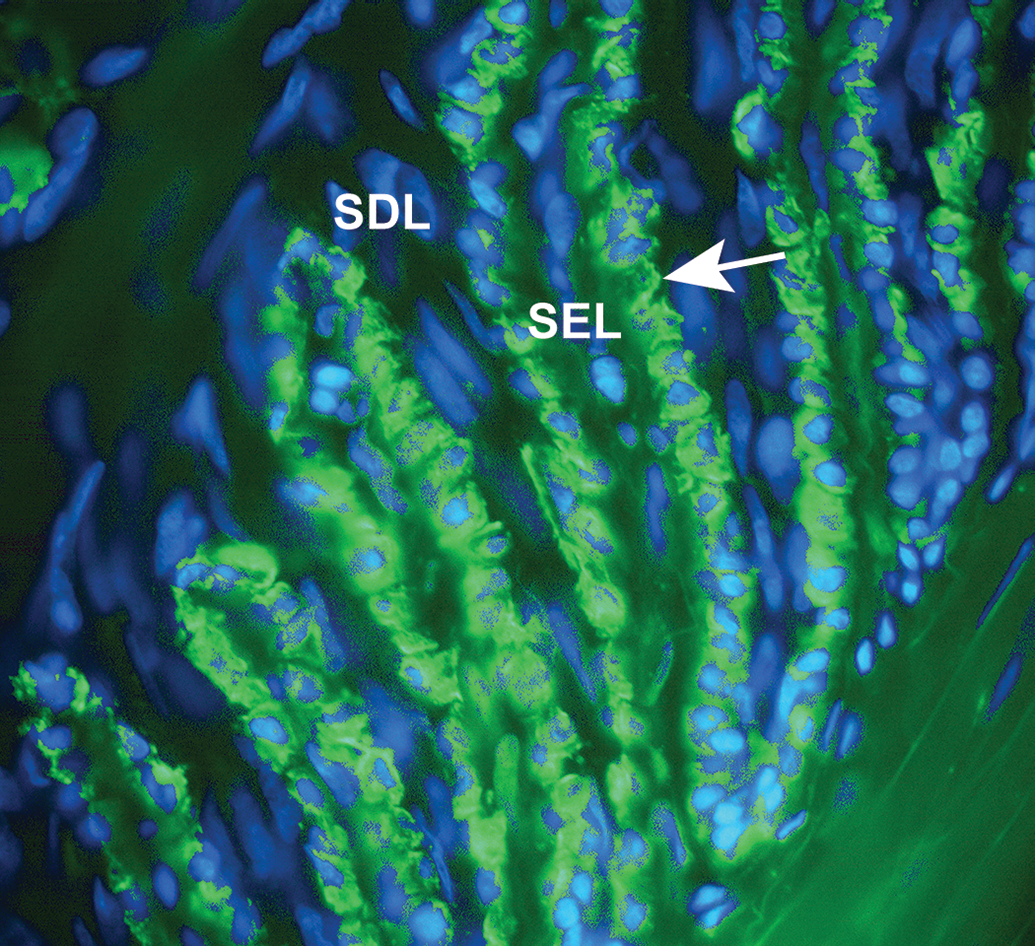
Photomicrograph showing fluorescent labeling of keratin 14 (arrow, green), in basal cells between secondary epidermal lamellae (SEL) and secondary dermal lamellae (SDL) within the stratum internum of equine hoof tissue. Nuclei are stained blue.
The type and number of keratins within a tissue tend to change with age and pathology. 35,36 Accordingly, there are distinct differences in keratins within the stratum lamellatum of unaffected versus laminitic hooves. In endocrinopathic laminitis, lamellar tissue is reported to be partially replaced with aberrant keratin and keratinization of abaxial aspects of primary epidermal lamellae is increased compared to normal. 37 In addition, keratinocyte hyperproliferation is documented in hooves affected by insulin-induced laminitis. 37,38 Abundant keratohyaline in the normal hoof stratum externum and cytokeratins in epidermal basal cells are not evident in laminitic hoof tissue. 39,40 Cytokeratin 14 clusters around the nucleus in hooves with carbohydrate overload (CHO) induced laminitis compared to the normal cytoplasmic distribution. 40 Based on this information, changes in cytokeratin and keratin content and location provide a unique mechanism to determine cell phenotype, maturity level, and pathology within equine hoof tissue.
Growth
The hoof wall of a normal adult horse grows at a rate of approximately 0.23 to 0.35 inches (6-9 mm) per month. 41 Stratum medium growth occurs by division of epidermal basal cells on papillae within the coronary corium. 20 As the basal cells divide, they drive older cells toward the ground surface to create the tubular and intertubular horns of the hoof wall. 17 There are several reports describing surrounding tissue growth in the stratum internum which is thought to occur, in part, by proliferation of basal cells in the proximal lamellae. Theories exist about the process by which the strong attachment with the stratum medium is maintained while it grows downward. 19 It is generally agreed that tissue homeostasis includes balanced synthesis and enzymatic degradation of extracellular matrix by resident cells. Proteins such as tissue inhibitors of metalloproteinase and metalloproteinases (MMPs) appear to be important to the process of controlled tissue turnover required for hoof growth and repair. 42 The enzymes can also cause excessive degradation of key proteins that stabilize the epidermal–dermal interface like laminin when tissue homeostasis is disrupted by increased neutrophil MMP (eg, MMP2 and MMP9) production in the presence of pro-inflammatory cytokines (eg, interleukin [IL] 1β and 8). 43 -46 While it is clear that coordination of tissue production and degradation is disrupted by inflammatory mediators, the exact contributions of and consequences to resident progenitor cells have yet to be determined.
Mediators of Tissue Damage
There are 4 recognized stages of laminitis: developmental, acute, subacute, and chronic. 47 The developmental phase occurs between the first insult and the appearance of initial clinical signs. The acute phase extends from first clinical signs to displacement of the third phalanx, thought to result from a combination of tension from the deep digital flexor tendon, weight-bearing forces, and breakdown of the dermal–epidermal junction. If the third phalanx is not displaced within 72 hours, the condition is classified as subacute. Subsequent lamellar tissue failure with progressive third phalanx displacement is considered chronic laminitis (Figure 4). 47,48
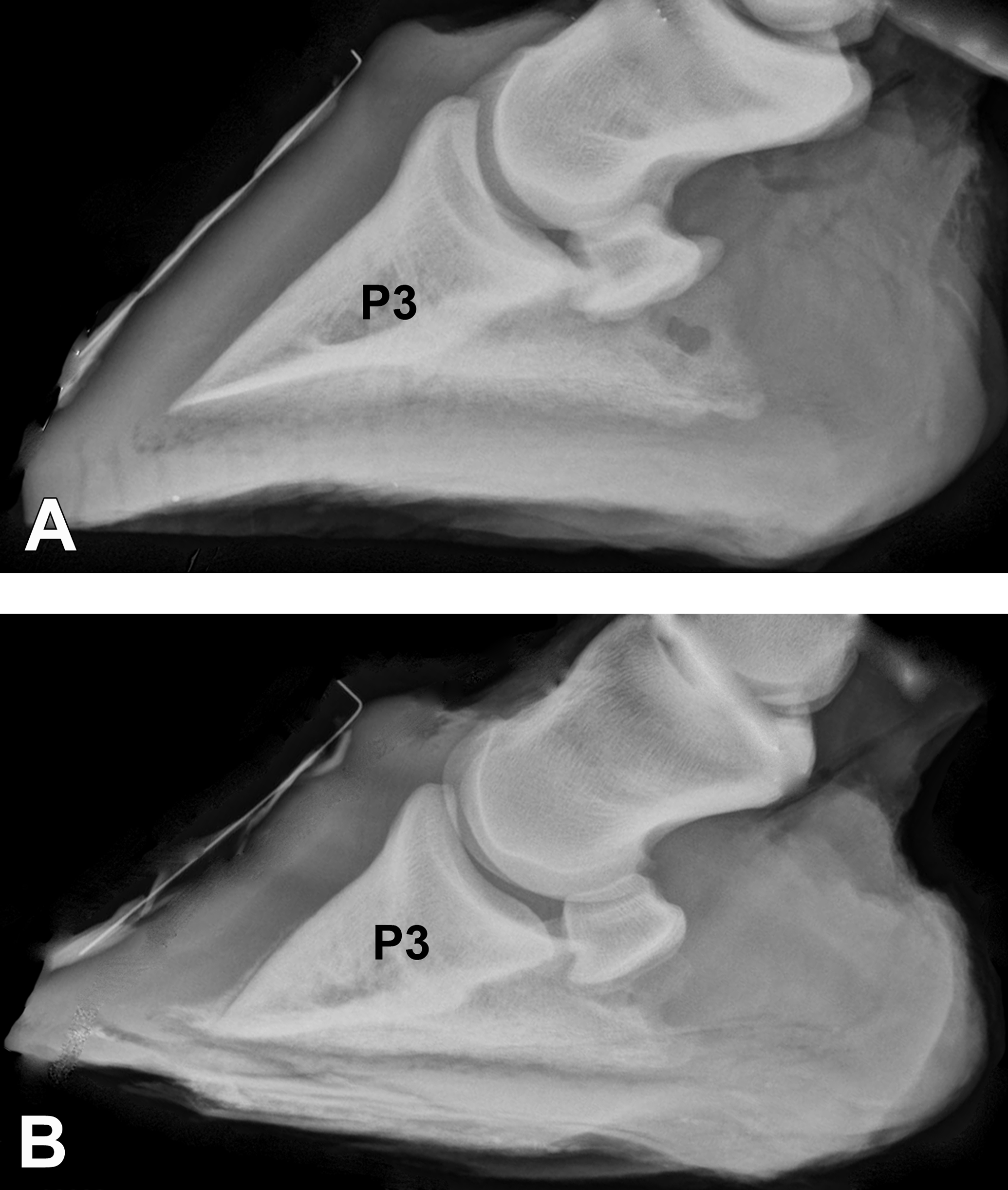
Radiographs of an unaffected (A) and laminitic hoof (B). The third phalanx (P3) is rotated and displaced distally in the radiograph of the hoof with laminitis.
Laminitis can occur secondary to any number of systemic or local insults. Inflammatory or endocrine cascades as well as circulatory compromise may all contribute to tissue damage. 49,50 Predisposing conditions for laminitis are numerous, ranging from equine metabolic syndrome to pituitary pars intermedia dysfunction, and most are associated with some level of systemic inflammation and/or hormonal imbalance. 51 Obesity may increase predisposition to laminitis by disrupting hoof homeostasis, in part, by adipose tissue production of inflammatory cytokines such as tumor necrosis factor α, IL-1β, and IL-6, all of which decrease systemic insulin receptor signaling and insulin sensitivity. 52 Additionally, obesity contributes to adipose tissue dysregulation and altered adipokine production including increased leptin and decreased adiponectin, an insulin-sensitizing hormone. 53 Obesity is also associated with elevated cortisol levels from overexpression of 11β-hydroxysteroid dehydrogenase 1 in human patients with insulin resistance, type 2 diabetes, and hyperlipidemia. 54 Regardless of the inciting cause, laminitis can seriously damage the crucial epidermal–dermal bond that maintains structural integrity of the hoof soft tissues. Similarly, due to differences in the root cause of laminitis as well as individual characteristics, disease severity and progression vary widely in equine individuals.
Inducible Models of Laminitis
To study the chronology and pathophysiology of laminitis, toxicants and high doses of carbohydrates and insulin have been used historically to repeatably induce the condition in horses for sequential evaluations as disease progresses. 55,56 Pathologic changes vary with toxin and dose. 57 -59 Use of endotoxin to cause laminitis is controversial since lipopolysaccharide administration to normal horses tends to create signs of clinical sepsis and decrease digital perfusion, but it does not appear to cause laminitis in otherwise normal horses. 46,60 -62 Carbohydrate overload with corn starch and wood cellulose flour or black walnut extract (BWE) cause microstructural changes in the hoof which resemble those of naturally occurring laminitis (Figure 5). 57,63 Oligofructose derived from chicory root and hyperinsulinemia are also used to create laminitis. 58,64 Years of research have resulted in a diverse set of mechanisms to induce laminitis for purposes of study.
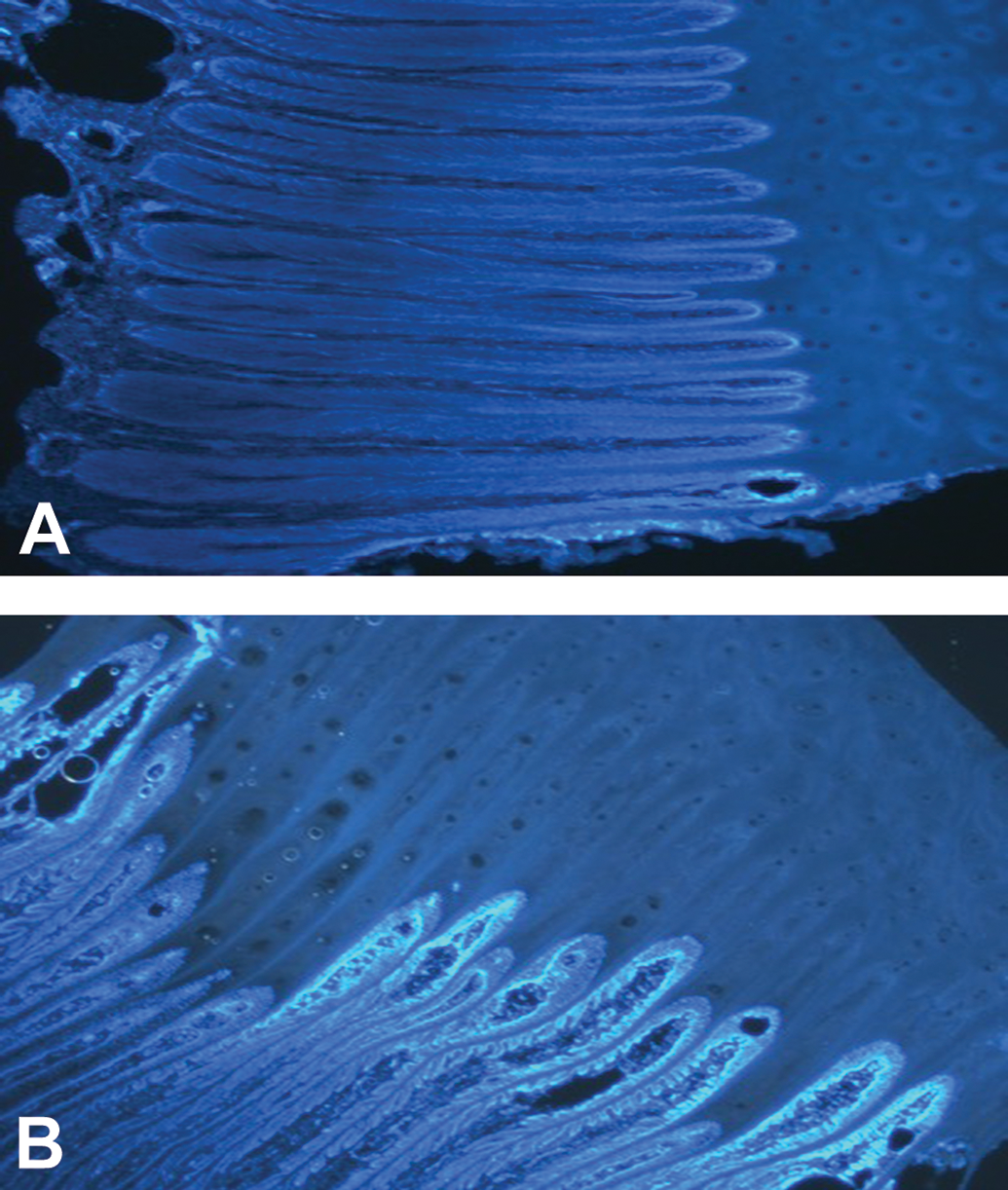
Fluorescent photomicrographs of the stratum internum from an unaffected (A) and laminitic (B) hoof with nuclei stained blue with 4′, 6-diamidino-2-phenylindole. The lamellae are misshapen in the laminitic hoof.
Given the role of resident cells in tissue homeostasis and repair, careful consideration of changes in both cells and tissue is necessary to compare the models of acute laminitis to the naturally occurring condition (Table 1). In models and naturally occurring laminitis, innate and adaptive immune cells accumulate around microvasculature and the stratum lamellatum epidermal–dermal interface (Table 1). 65,66 Calprotectin, an inflammatory protein, is expressed by extravasated white blood cells on the epidermal side of the basement membrane in natural and induced laminitis. 67,68 Microstructural damage and cell loss on and around the basement membrane is comparable among models and laminitis; however, changes in naturally occurring laminitis can be more subtle than experimentally induced laminitis. Basal cell cytoskeleton collapse is associated with glucose deprivation and MMP activation in the oligofructose model. 44 Hemidesmosomes between basal cells are lost in the oligofructose, CHO, and insulin models. 69 -71 In the CHO model, there is basement membrane necrosis and loss of epithelial cells between the secondary epidermal and dermal lamellae. 72 With high-dose insulin, lamellar epithelial cells are abnormally elongated and subject to dyskeratosis and apoptosis. 13,38
Comparison of Microstructural Tissue Changes from Induced and Naturally-occurring Laminitis.

Abbreviations: BWE, black walnut extract; CHO, carbohydrate overload; OF, oligofructose.
Gene and protein expression between naturally occurring and experimentally induced laminitis are somewhat comparable. In natural laminitis, the transcription factor, P63 (a determinant of stratified epithelial stem cell proliferation), decreases, while proteins involved with vascular inflammatory reactions such as gamma-histone H2AX and endothelin-1 expression increase. 37,73,74 Inflammatory ILs such as IL-8, IL-1β, and IL-6 are typically increased and anti-inflammatory ILs such as IL10 decrease in the BWE and oligofructose models. 75 Cell signaling is disrupted in the oligofructose and CHO models. Of particular note is that suppression of the Wnt signaling mechanism necessary for mesenchymal to epithelial transition of basal epithelial cells in the CHO model potentially interferes with commitment to the epithelial lineage necessary for organized epithelial tissue formation. 76 Basal cell gene expression of factors associated with both intercellular cell connections and communication, like β-catenin and integrins in the CHO model and insulin-like growth factor-1 receptor expression in the insulin model, are reduced. 77 -80 Induction of laminitis in otherwise normal hooves is one mechanism to study the acute pathogenic mechanisms of the condition for potential therapeutic targeting. Unfortunately, the models do not exactly replicate the natural course of the disease that often includes persistent abnormalities in hoof tissue production and maturation. Focus on viable, immature cells that contribute to tissue formation and repair is likely to reveal a new avenue of discovery in the quest to counteract the life-changing effects of laminitis.
Multipotent Stromal Cells
As indicated above, adult MSCs are routinely isolated from numerous tissues in the horse. 86,87 Tissue stroma is derived from mesoderm, and undifferentiated cells in the space retain the capacity to alter their morphology and function to address tissue repair and maintenance needs. 88 By definition, adult MSCs exhibit specific characteristics in vivo such as the ability to adhere to polystyrene and the capacity to differentiate into distinct cell types from the same embryonic layer under specific induction conditions (Figure 6). 89,90 Additional criteria required by the International Society of Cell Therapy are the expression of some or all of a panel of surface antigens, including CD105, CD44, CD29, CD73, CD90, and no expression of CD45, CD34, CD14, CD11b, or CD19, as well as lineage-specific protein and gene expression following chemically induced differentiation. 91,92 Other stem cell markers often applied to identify MSCS include markers associated with embryonic development such as octamer-binding transcription factor 4, stage-specific embryonic antigen 3 (SSEA-3), SSEA-4, and podocalyxin (TRA-1-60). 93,94 Typically, MSCs possess some capacity for self-renewal, though not immortality like totipotent cells, and they have variable immune privilege. 95
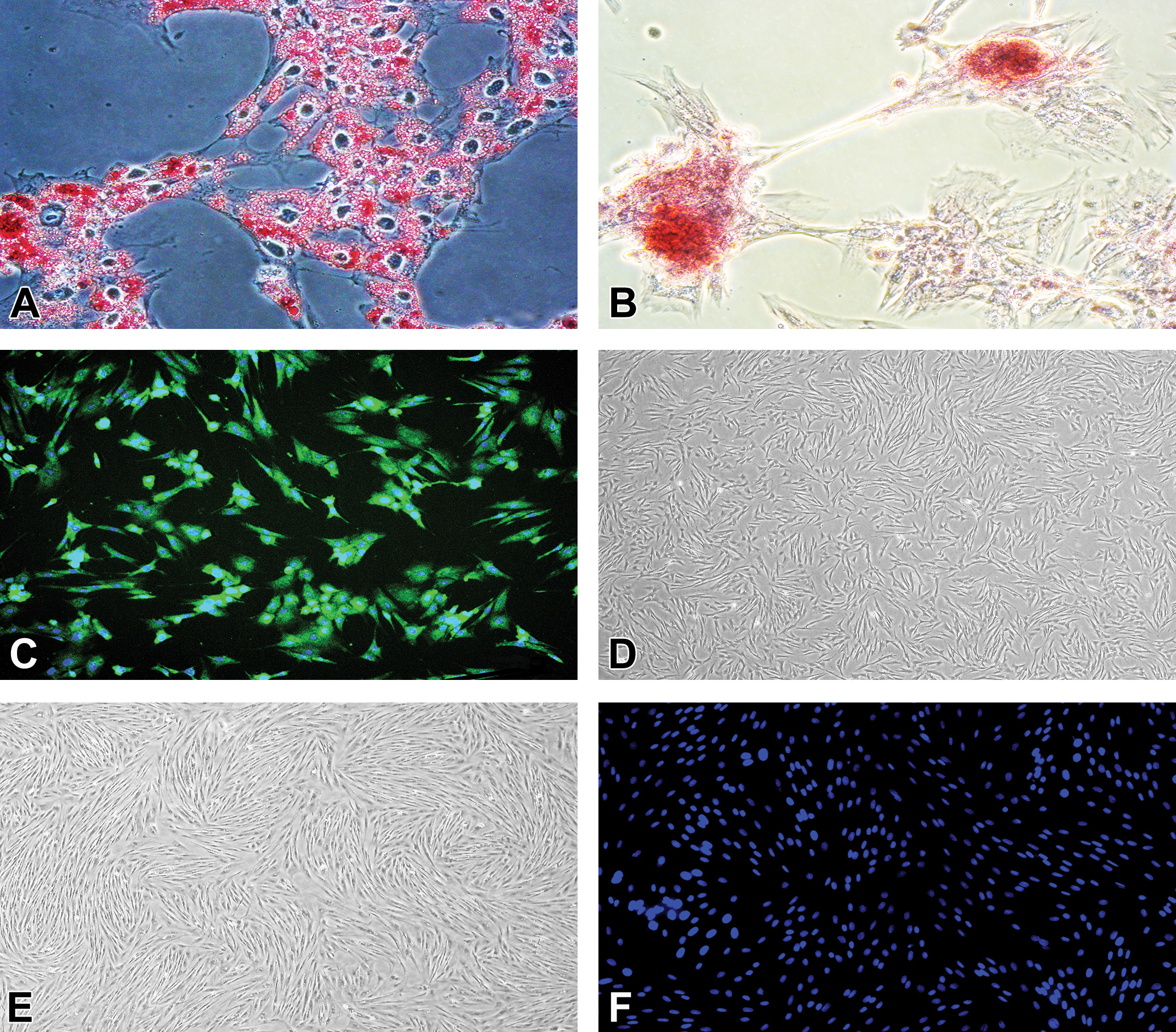
Photomicrographs of lamellar progenitor cells following culture in adipogenic (A), osteogenic (B), or neurogenic (C) induction medium as well as stromal medium (D, E, F). Adipose droplets, calcium deposits, and microtubule-associated protein 2 (Map2) protein expression were stained with Oil Red O (A, D, red), Alizarin red (B, E, red), and anti-map2 with anti-mouse IgG (C, F, green), respectively. Nuclei were stained with 4′, 6-diamidino-2-phenylindole (C, F, blue).
Primary cell isolates are usually heterogeneous collections of cells with multiple immunophenotypes that reflect different plasticity and maturity. 96 Despite this, the cells have characteristic microstructure and ultrastructure. 97 Current knowledge supports that progenitor cells can differentiate into distinct cells from similar origins as well from other embryonic layers, a process commonly referred to as transdifferentiation. 98 They also maintain the capacity to “regress” to an earlier stage of development both by natural and synthetic means. 99 Additionally, exogenous MSCs can both guide and be guided by endogenous cells. 100,101 Despite similar behaviors and morphologies, undifferentiated cells vary among species and tissues. 102 This requires individual characterization to elucidate the distinct properties and potential of cells from each parent tissue and species. 91,92
The ability to culture and manipulate progenitor cells in vitro creates a virtual plethora of opportunities to study normal and abnormal tissue regeneration to direct progressive therapeutic discovery and translation of cellular therapies. 103 Autologous (same individual) and allogeneic (same species, different individual) MSCs appear to promote tissue healing in horses (including hoof) and other species. 104 -106 Despite this, epigenetic factors are thought to influence the plasticity of progenitor cells, with cells from a given tissue having the best potential to help repair that tissue. 107 Reports are mixed surrounding the efficacy of non-tissue-specific MSC therapies in the hoof and other tissues like tendon, potentially due to the consequences of epigenetic programming that is not overcome by limited in vitro manipulation. 108 -110 This evidence strongly supports the need to isolate and characterize progenitor cells from the hoof to understand the effects of lamellar pathology on them as well as the potential of exogenous cells or neotissues to reverse or prevent the disease process.
Flow cytometry and fluorescence-activated cell sorting (FACS) are universal tools to quantify distinct cell characteristics within a heterogeneous progenitor cell isolate. Flow cytometry is used to differentiate cells based on size, volume, and intensity of fluorescence from labeled antibodies that bind to antigens of interest on or within the cells. 111 The system is typically composed of a “flow” mechanism by which cells pass through a detection apparatus composed of a laser activator and light detectors. The light scatter is used to evaluate size and fluorescence detectors quantify light intensity within specific wavelength ranges. 112 Fluorescence-activated cell sorting is a more complex form of flow cytometry that relies on most of the same concepts but has the added feature of sorting the cells based on size and fluorescence. To accomplish this, an electrostatic charge is applied to fluid droplets containing individual cells. Based on the presence or absence of a charge, the cells are collected into distinct reservoirs. 113,114 The process of cell sorting is performed with live cells to allow culture following separation. The level of complexity varies among flow cytometry and FACS systems and can include relatively few to many detection channels to permit separation of cells expressing a wide assortment of proteins. Recently, FACS was used to separate cells that expressed both K14 and CD105 (K14+, CD105+) from equine hoof primary cell isolates for evaluation and characterization of both a distinct progenitor cell subset and a heterogenous cell population. 115
Epidermal Progenitor Cells
Epidermal progenitor cells, are, as the name implies, derived from the embryonic ectoderm. 116 They inhabit the basal layer of the epidermis and participate in tissue repair and cyclic renewal under the direction of growth factors like epidermal and fibroblast growth factors. 117,118 Like MSCs, they also possess the capability to differentiate down distinct cell lineages from multiple embryonic layers. 119 Notably, MSCs are reportedly capable of assuming an epidermal phenotype. 120 Keratins expressed by epithelial cell populations of complex tissues, including the hoof, vary with cell maturation stage well as specific function(s) (Figure 7). 31 In human epithelial tissues, there is distinct keratin expression in basal cells, suprabasal cells, transit-amplifying cells, and keratinocytes with keratins 5, 14, and 15 typically expressed by immature cells and keratins 1, 9, and 10 by mature cells. 36 Human epidermal stem cells and transit-amplifying cells can also be distinguished by high integrin 1β expression and low accumulation of rhodamine (Rh123) versus medium integrin 1β expression and high accumulation of rhodamine, respectively. 121 As the progenitor cell knowledge base expands, so does awareness of distinctions between unique cell populations. Continued diligence to maintain nomenclature and standards will support consistency and repeatability in progenitor cell research.
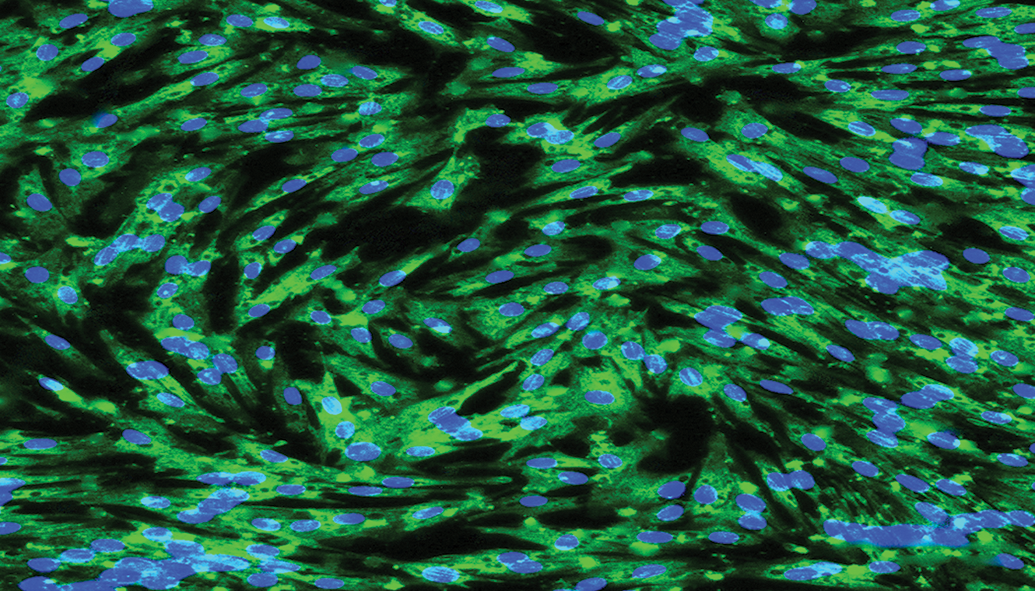
Fluorescent photomicrograph of cultured equine hoof progenitor cells stained with anti-K14 and anti-mouse IgG-488 (green). Nuclei are stained with 4′, 6-diamidino-2-phenylindole (blue).
Skin progenitor cells from most species have comparable plasticity to progenitor cells from other tissues and can mature into multiple lineages including bone, adipose tissue, nerve, and liver. 117,122 Several immunophenotypes have been identified among species like K15+, CD34+ immature keratinocytes from canine hair follicle tissue, and CD29+, K19+ cells from human, murine, and porcine skin. 118,123,124 The markers CD34 and K6 are also used to identify and isolate keratinocytes. 125 Coexpression of keratins 15 and 19 by basal keratinocytes decreases with age and indicates cell self-renewal and tissue production capabilities in human skin. 121,126 Fibroblast-like stem cells from human foreskin express CD90, CD105, CD166, and CD73, which are also found on human bone marrow–derived MSCs. 124,127 This information confirms a wide variety of epidermal progenitor cells within heterogeneous primary cell isolates, establishes plasticity of the cells, and highlights common surface antigens with MSCs.
Failure of basal cell proliferation or keratinocyte maturation is associated with mild to serious and potentially life-threatening skin pathologies. 128 A study about human scalp hair follicle progenitor cells in bald versus hairy scalp showed that there was no difference in the number of K15+ progenitor cells but that the number of CD200+, CD34+ cells was lower in bald scalp, leading to the conclusion that a failure or difference in maturation was the primary distinction between the two conditions. 129 On the other end of the spectrum is epidermolysis bullosa, an inherited skin disorder in humans and horses associated with depleted progenitor cells and disrupted homeostasis in the skin that results in blistering or sloughing epidermis. 130 The underlying pathology of the disease is failure of anchoring proteins like laminin and collagen at the dermal–epidermal junction due to variable genetic mutations. 25,131 Injection of mesodermal or epidermal progenitor cells into lesions has reportedly promoted local healing. 132 This lends credibility to the potential for exogenous progenitor cells or cell factors to restore healing potential to damaged or compromised tissue.
Hoof Progenitor Cells
The epidermal–dermal interface in the stratum lamellatum is unique to the hoof in horses and the periodontal membrane and fingernail bed in humans. 133,134 Isolation and culture of hoof progenitor cells has only recently been described in contrast to isolation of progenitor cells from the other interface tissues (Figure 8). Current knowledge supports that primary progenitor cell isolates from normal and abnormal equine hoof stratum lamellatum express ectodermal (K14, K15, K19) and mesodermal (CD29, CD44, CD105) genes and proteins, and they have both ectodermal (neurogenic) and mesodermal (osteogenic, adipogenic) differentiation abilities. 135 Further, equine hoof progenitor cells share some ultrastructural features of both epidermal and mesodermal equine progenitor cells like intracellular vacuoles and well-developed dilated rough endoplasmic reticulum cisternae (mesodermal), as well as cell surface microvilli (ectodermal). 136,137 The cells also produce extracellular matrix on scaffold carriers composed of either polyester or mineral and on decellularized lamellar matrix in vivo, confirming the ability to form neotissue on custom templates. 115,135 This information suggests that in vitro cell models of the equine stratum internum may be an alternative to in vivo induction of laminitis in otherwise normal hooves.
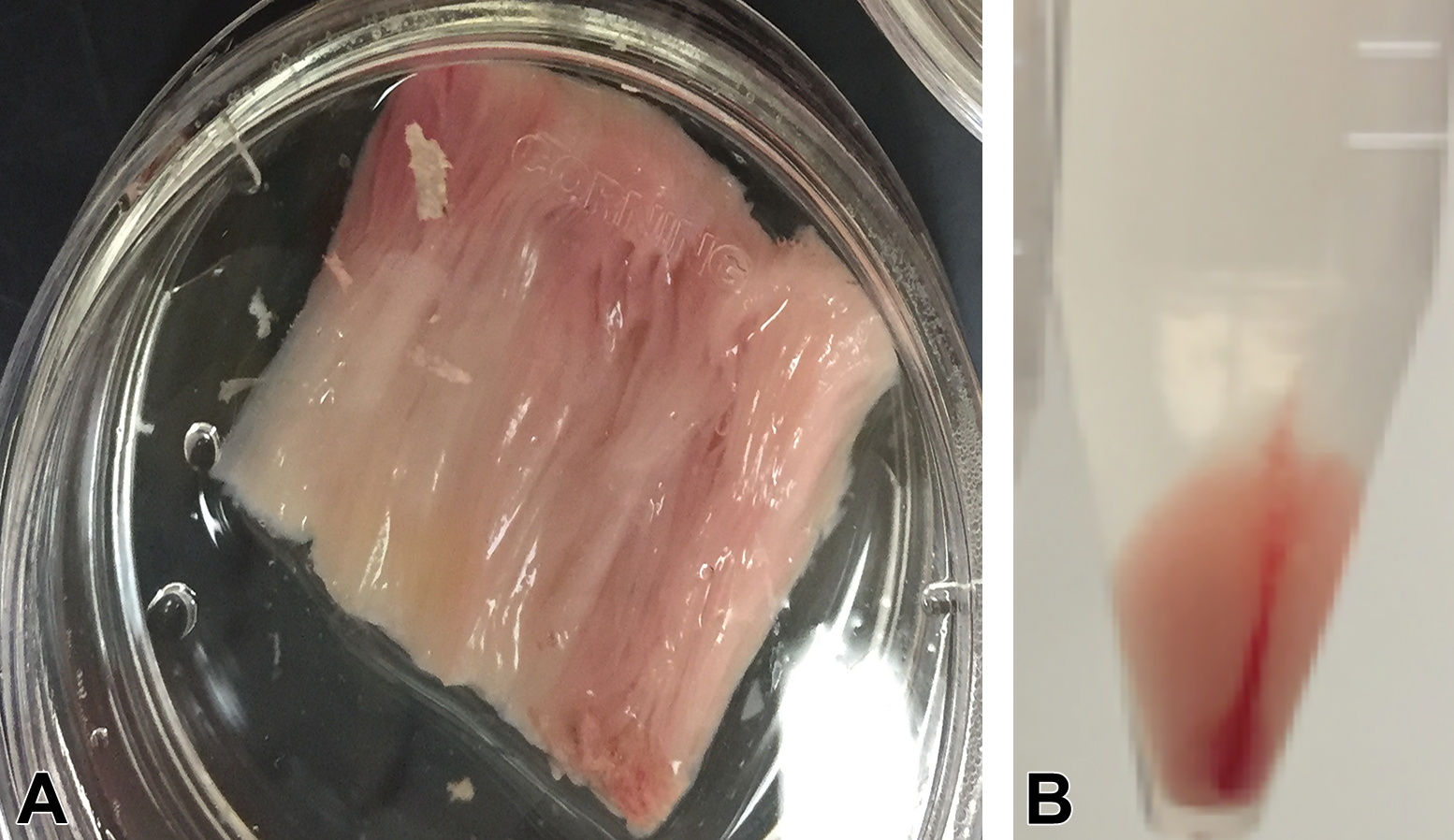
Stratum lamellatum harvested from an equine hoof (A) and the resulting cell pellet (B) after digestion, filtering, and centrifugation.
The description of equine hoof progenitor cell isolation is typical of collagenase tissue digestion for progenitor cell isolation from adipose tissue and skin with various collagenases (eg, type I or type III), followed by separation of the cells from tissue remnants with combinations of filtration, gravity separation, and centrifugation. 138,139 Notably, progenitor cells from cryopreserved, intact equine hoof stratum lamellatum have also been isolated by collagenase digestion. 115 Unlike cells isolated from cryopreserved periodontal ligament which were indistinguishable from fresh cell isolates, the cells from cryopreserved hoof tissue show signs of senescence including an irregular cell membrane, electron-dense cytoplasm, poorly organized rough endoplasmic reticulum, and aggregated nuclear chromatin (Figure 9). 140 Cryopreservation is reported to “age” fresh cell isolates, and in vivo behavior, plasticity, growth rate, and expansion potential are impacted accordingly. 141 As such, most reports caution against consideration of continuously cultured and revitalized cells as the same. 140,142
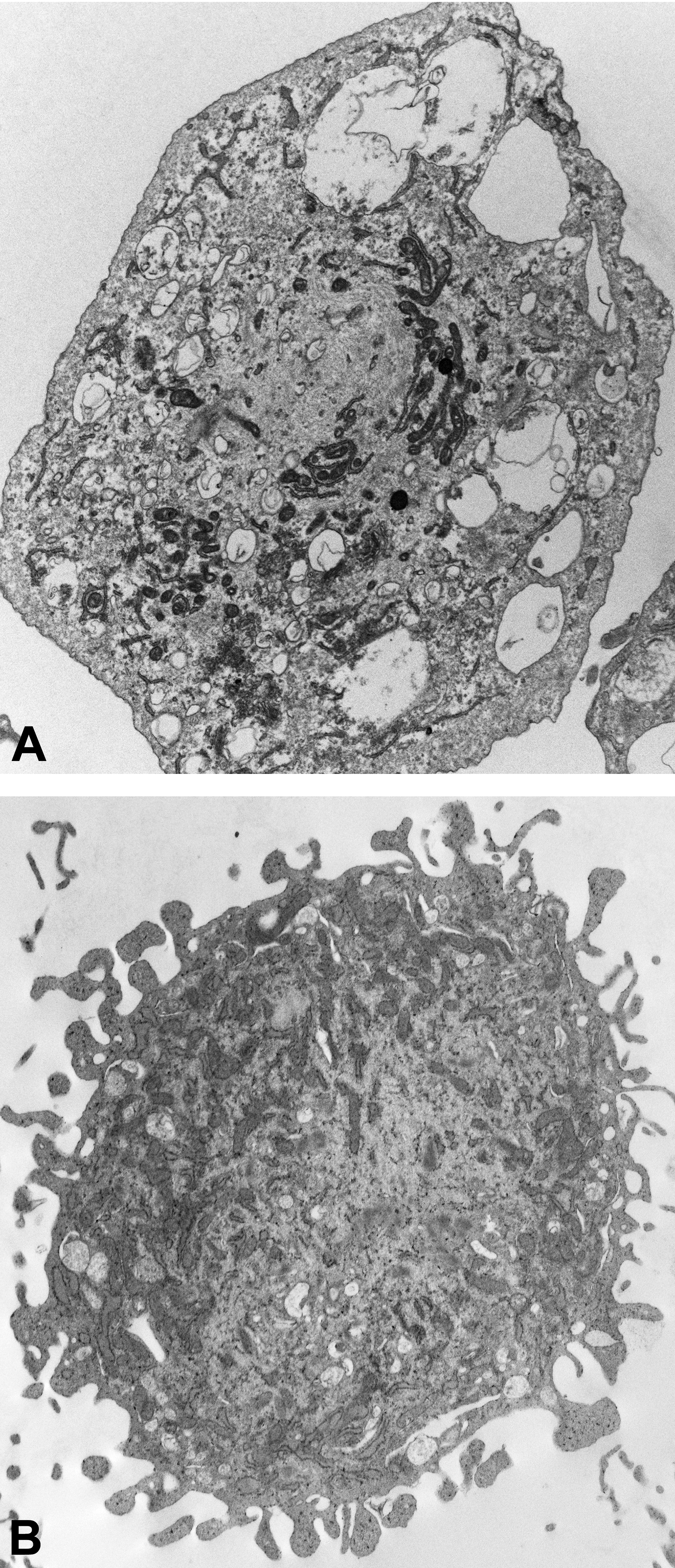
Transmission electron photomicrographs of cultured progenitor cells from fresh (A) and cryopreserved (B) equine stratum lamellatum. The cells from the cryopreserved tissue show signs of senescence.
Transdifferentiation
Transdifferentiation is a complex process by which immature cells assume a new lineage that is distinct from their embryonic origin, parent tissue, or current phenotype. 143 Numerous distinct and overlapping cell signaling pathways are associated with transdifferentiation. 144,145 Both mesodermal and epidermal progenitor cells possess transdifferentiation abilities as alluded to above. In mice and humans, bone marrow progenitor cells can assume skin and neural phenotypes. 146,147 Similarly, mesodermal progenitor cells from equine bone marrow show neural crest-like cell morphology and expression of neural proteins after neural differentiation. 148 A well-known transdifferentiation process is epidermal-to-mesenchymal transition (EMT). Controlled EMT is a key process in normal tissue healing, while uncontrolled EMT is characteristic of neoplasia. 149,150 One caveat raised by researchers is that transitional cells, or cells expressing proteins from multiple lineages, must be thoroughly characterized to confirm their identity and avoid confusion with cells that result from fusion of progenitor cells with somatic cells. 151 -154 Elucidation of the EMT process by hoof progenitor cells may provide novel insight to the pathology of laminitis.
A natural interface between the epidermis and dermis like that in human skin as well as equine hoof stratum lamellatum would reasonably contain transitional progenitor cells that can function in repair and maintenance of the complex tissue connection. 155 Recently, cells that express epidermal (K15) or mesodermal (CD105) proteins were identified in the stratum lamellatum. 156 Progenitor cell isolates that expressed both proteins were subsequently isolated from heterogenous stratum lamellatum primary cell isolates and cultured through multiple passages in vitro (Figure 10). 135 The cells showed ectodermal and mesodermal plasticity and transdifferentiation capabilities. The potential for EMT to contribute to laminitis is suggested by the abundant mesodermal wedge of fibrous tissue typical of the condition. This is especially notable given the presentation of transitional cells in the stratum lamellatum. Inflammation has been shown to favor mesodermal tissue formation. 157 This lends credibility to the potential for inflammatory cytokine-driven EMT in laminitic hooves. Given the potential for diverse neotissue formation, transitional progenitor cells may be a meaningful therapeutic target to help avoid permanent tissue damage and restore normal formation after an inflammatory insult in the equine hoof.
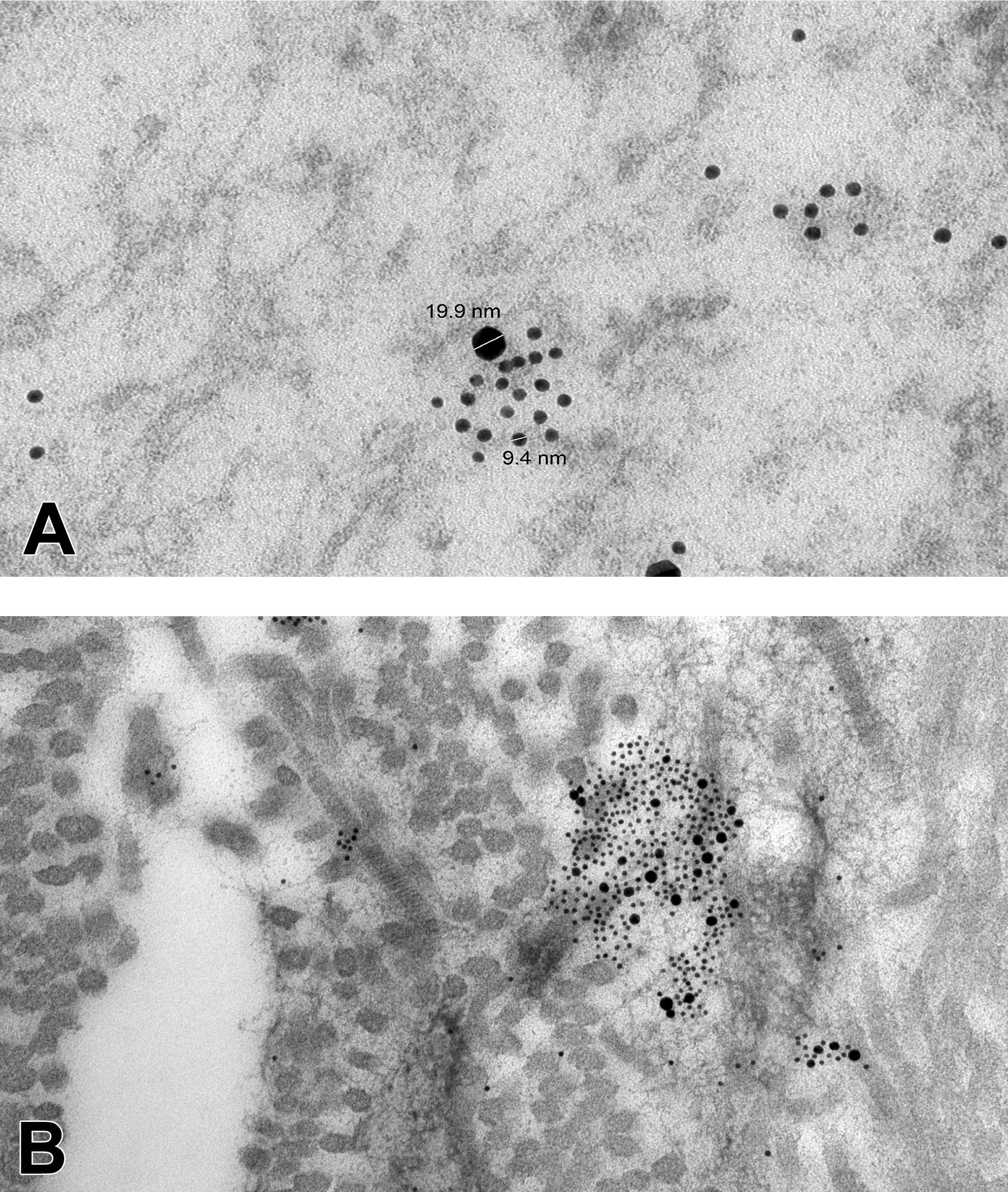
Transmission electron photomicrographs of P3 progenitor cells from equine hoof stratum lamellatum cultured on culture ware (A) and on decellularized lamellar tissue matrix (B). Gold-labeled secondary antibody labels against K15 (10 nm) and CD105 (20 nm) localized in cell cytoplasm (A) and on the fibrous construct (B).
Regenerative Medicine Scaffolds
Decellularized tissue matrix scaffolds are frequently used for in vitro and in vivo neotissue formation because they provide structural support and a favorable environment that contains various amounts of native tissue proteins to facilitate cell attachment and help guide differentiation. 158,159 The matrices can be derived from virtually any tissue and are typically decellularized and partially deproteinized via chemical and thermal means in a variety of combinations. 160,161 However, the tissue matrix does not necessarily have to come from the recipient species or tissue. Decellularized matrices have been used to support regeneration of tissues from relatively simple musculoskeletal structures to complex organs like liver. 162,163
The complicated skin structure requires a matrix framework that recapitulates or facilitates formation of the epidermal–dermal junction during healing. Maturation of keratinocytes to form the protective, outer layers of skin relies on a healthy connection between the epidermis and dermis. 164 Synthetic and natural tissue matrix alone and with progenitor cells has been used to augment skin healing, and collagen-based matrices are widely popular. 157 A report details the use of porcine small intestinal submucosa matrix to treat granulating wounds on equine distal limbs. 165 Multipotent stromal cells from equine umbilical cord blood embedded in an autologous fibrin gel are reported to improve the histologic outcomes of distal limb skin wounds. 166 Dextran-based hydrogel scaffold itself improved neovascularization and skin regeneration in porcine burn wounds by promoting angiogenic cell infiltration and accelerating healing kinetics. 167 Porcine burn wounds treated with collagen–glycosaminoglycan scaffold–MSC constructs had significantly better healing than with scaffold or cells alone. 168 While the hoof capsule has many differences from most of the epidermis, the work above establishes strong precedent for tissue matrix and tissue matrix with cells to restore normal hoof growth and repair.
Following severe inflammation, some changes in the basement membrane zone of the stratum lamellatum include altered keratinocyte proliferation, loss or reduction in vital proteins such as laminin and collagen, and decreased cell hemidesmosome connections. 2,169 Like other tissues, there appears to be abundant progenitor cells in damaged lamellar tissue. However, lack of structural and protein direction may prevent appropriate differentiation and, in fact, may misdirect differentiation via a process like EMT. It is possible that minimally invasive implantation of decellularized stratum lamellatum (Figure 11) or stratum lamellatum neotissue formed by progenitor cells under controlled laboratory conditions may provide the necessary signals for restoration of healthy hoof tissue. Continued in vitro research is necessary to advance and refine potential administration of exogenous tissue or cell–tissue constructs.

Scanning electron micrograph of decellularized stratum lamellatum showing the typical lamellar structure (A, arrow) and the protein-covered surface (B).
The Future of Progenitor Cell Therapy
Progenitor cell therapy has gained substantial popularity over the last few decades. Potential benefits of progenitor cells are abundant and include anti-inflammatory properties, direction of endogenous cells, and acceleration of tissue healing or formation, among others. 170 Current Food and Drug Administration regulations, however, place strict regulations on preparation and administration of primary cell isolates. Potential concerns about their use include lack of a predictable half-life and route of elimination, the potential for undifferentiated cells to assume pathologic phenotypes, and possible, dangerous immune reactions. 171,172 Despite this, promising results have been reported from primary cell administration. There are also other valuable options for primary cell isolates. Some include living tissue models that recapitulate normal and abnormal embryonic tissue formation as well as generation of viable tissue grafts and organs for implantation from autogenous or autologous progenitor cells. It is conceivable that neotissue models from hoof progenitor cells may provide a valuable resource for study and treatment of equine laminitis. Culture media can be modified to replicate normal, diseased, and inflamed conditions to evaluate and modify cell responses to withstand and overcome tissue abnormalities from injury or systemic illness. In vitro neotissue models may also result in viable tissue grafts and support development of custom therapeutic matrix for local administration. Inherent to these futuristic, but theoretically attainable goals, is the continued need to isolate and fully characterize native progenitor cell populations. It is only through focused, incremental advances that the power of the cell can be harnessed to restore normal function of an organ or system for the health and well-being of a whole organism.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for some of the work described in this review was provided by the Louisiana State University Equine Health Studies Program, Louisiana State University School of Veterinary Medicine, US Department of Agriculture 1433 Animal Health and Disease Research Program, and the Tynewald Foundation.
