Abstract
The effects of particulate matter (PM) on cardiopulmonary health have been studied extensively over the past three decades. Particulate matter is the primary criteria air pollutant most commonly associated with adverse health effects on the cardiovascular and respiratory systems. The mechanisms by which PM exerts its effects are thought to be due to a variety of factors which may include, but are not limited to, concentration, duration of exposure, and age of exposed persons. Adverse effects of PM are strongly driven by their physicochemical properties, sites of deposition, and interactions with cells of the respiratory and cardiovascular systems. The direct translocation of particles, as well as neural and local inflammatory events, are primary drivers for the observed cardiopulmonary health effects. In this review, toxicological studies in animals, and clinical and epidemiological studies in humans are examined to demonstrate the importance of using all three approaches to better define potential mechanisms driving health outcomes upon exposure to airborne PM of diverse physicochemical compositions.
Introduction
The health impacts of exposure to particulate matter (PM) are well established. According to the World Health Organization data (2019), 9 of 10 people breathe polluted air—this exposure kills 7 million people every year. 1 Even at concentrations below current United States and European Union regulatory standards, community death rates rise and fall with pollution levels. 2 Exposure to PM manifests in a diverse number of ways. Complications can be as simple as eye, nose, and throat irritation, coughing, and wheezing, or as severe as irregularities in cardiac function. Of greatest concern are those effects on the heart and the lungs. Within federal pollution limits, approximately 60,000 cases of respiratory problems/heart attacks can be attributed to air pollution exposure in the United States. 1
There are three general study types for evaluating health effects of environmental exposure to PM. These include (1) toxicological, (2) human clinical, and (3) epidemiological methodologies, each of which have advantages and disadvantages.
Toxicological studies are controlled experiments using animals or cells. The advantages of toxicological studies are controlled exposure conditions, readily measured outcomes, and the ability to explore mechanisms by observing cellular and molecular changes. Compared to epidemiology studies, toxicological studies are limited in animal sample size, as well as a general inability to study susceptible subgroups reflective of the human population. In addition, interspecies differences in anatomy and physiology must be taken into consideration when extrapolating to humans. For example, mice and rats do not have respiratory bronchioles as do dogs, monkeys, and humans. Mice and rats also take 100 to 125 breaths/min, while humans only take 12 to 14 breaths/min. These anatomical and physiological factors affect the dose and thus the damage to exposed lungs.
Human clinical studies involve experiments in environmentally controlled situations or, as is often the case for inhalation studies, in isolated chambers. These studies enable the measurement of effects in humans exposed to known PM concentrations. The disadvantages of human clinical studies are typically related to the greater investments (eg, time, money) required relative to animal studies. Such costs impose limitations in (1) the number of subjects for any given study, (2) the duration of exposure that in general only allows for the measurement of acute effects, and (3) the reproducibility of environmental PM compositions and exposure conditions.
Epidemiological studies involve large cohorts of humans in real-world situations. The inclusion of a wide range of exposures to mixed pollutants presents an advantage and a disadvantage. Confounding factors such as differing populations or geographical areas could affect the study outcome, but also includes susceptible subgroups (eg, the elderly individuals and children), which offer the ability to capture a more complete range of human response patterns. Variability between subgroups may present challenges, particularly with respect to detecting statistically significant relationships. Fortunately, there are sophisticated statistical techniques to control for confounding factors to enhance more accurate exposure assessments.
Our intent in this review is to demonstrate the importance of using all three approaches of toxicological, human clinical, and epidemiological studies to better define and elucidate the potential causes for cardiopulmonary disease, based on airborne PM of diverse chemical and physical composition. Such differences can affect both the interpretation of the pathobiology and the toxicology seen in human and animal studies.
Although not comprehensive, this review provides examples of a series of studies, which demonstrate how epidemiological studies in humans can be interfaced with toxicological studies in animals to provide greater insights for potential biological mechanisms of effect.
Particulate Matter and Heart Rate Variability
Epidemiological studies have shown that there can be an increase in cardiovascular morbidity and mortality following short-term exposure to PM associated with air pollution. 3,4 Long-term exposure to particulate air pollution has also been associated with cardiovascular events leading to reduced life expectancy. 5 Although there appears to be little doubt that PM exposure poses a significant cardiovascular health risk, the underlying causes are poorly understood. The decreased heart rate variability (HRV) associated with PM exposure has gained considerable attention as a potential cause. 4 Heart rate variability is the variation among time intervals between successive heartbeats. When measured on an electrocardiogram (ECG), this between-beat interval on the QRS wave is called the RR interval. Decreased HRV has been shown to be an index of decreased cardiac autonomic function and is associated with increased susceptibility to ventricular arrhythmias and risk for cardiovascular-related sudden death. 6 –13
Epidemiological studies show significant associations between exposure to PM with particles of aerodynamic diameter of ≤2.5 µm (PM2.5) and cardiovascular-related morbidity. 14 –17 These epidemiological studies have been validated in human clinical studies. In a study by Vallejo et al, 18 40 young healthy adults from the Mexico City were assessed to determine the association between PM2.5 and HRV. All individuals underwent 13 hours of Holter electrocardiographic and PM2.5 personal exposure monitoring. The standard deviation of normal RR intervals and the percent difference between adjacent normal RR intervals larger than 50 milliseconds (pNN50) were used for the evaluation of HRV. Results showed a statistically significant negative association between pNN50 and cumulative exposure to PM2.5, such that the former decreased by 0.08% (P = .01) when there was a 30 µg/m3 increase in PM2.5 in the 2 hours prior to the measurement. This study demonstrated acute HRV effects of environmental exposure to PM2.5 in young healthy adults. However, due to a number of limitations in human studies, those mechanisms mediating reduced HRV by PM can be better addressed in animal studies.
As previously stated, an advantage of animal toxicological studies is the ability to explore mechanisms through the observation of cellular and molecular changes. Pham et al 19 hypothesized that PM2.5 exposure decreases the excitability of cardiac vagal neurons in the nucleus ambiguus to decrease HRV. Mice were exposed for 3 days to laboratory-generated PM2.5 (soot only or iron-soot). Heart rate variability was measured over a 24-hour period as ECG telemetry recordings. Cardiac vagal neurons were retrogradely labeled with fluorescent dye to determine the intrinsic properties of these neurons in the nucleus ambiguus. Following PM2.5 exposure, a reduction in neuronal excitability in cardiac vagal neurons was observed. Additional studies have also shown similar changes in HRV due to exposure to PM. 20 –23
Cardiovascular effects associated with PM exposure need to be explored further. The findings of these toxicological studies show that decreased excitability of cardiac vagal neurons might be a potential cause for the cardiovascular effects observed in humans.
Impaired Neonatal Lung Growth
Many epidemiological, clinical, and toxicological studies have demonstrated exposure to PM during development has a detrimental effect on children’s respiratory health. 24 –32 Symptoms range from increased incidence of respiratory infection, decreased lung function, asthma exacerbation, and, in some cases, impaired lung development. One of the most cited studies on the relationship between air pollutants and respiratory health is the Southern California Children’s Health Study (CHS). 33 This cohort was established in 1992 and has since involved more than 12,000 school-aged children. 29,34,35 It is well studied and understood that human and animal lungs continue to develop postnatally. In the human, the lung will not be fully developed until approximately 21 years of age. This fact opens the hypothesis that there is a developmental window in which a child may be more susceptible to the detrimental effects of PM than an adult.
In a report focusing on 10- to 18 year-old youth from the CHS cohort, Gauderman et al 36,37 measured the lung function of over 1,700 school-aged children on an annual basis for 8 years. This age range was designed to capture a critical period of lung development. In children exposed to several pollutants, including, but not limited to, PM2.5 and elemental carbon (EC) statistically significant deficits, were observed in the forced expiratory volume in 1 second (FEV1; the maximum amount of air that can be forcefully exhaled in 1 second). These results were consistent across both males and females with and without preexisting respiratory conditions and worsened slightly for those who spent significantly more time outdoors. Forced vital capacity (the amount of air that can be forcibly exhaled from the lungs after taking the deepest breath possible), FEV1, and maximal mid-expiratory flow (the peak expiratory flow taken from the flow–volume curve mid-spirometry) are measures that can reflect lung volume. The youth in this study were in a specific developmental time window when alveoli are still growing, suggesting impaired lung function in children could be related to changes in lung growth. 38
Effects of PM on the developing lung have been studied and debated in the literature. The precise mechanism for these effects is complex and still being elucidated. In a study by Pinkerton et al, 38 the respiratory effects of acute exposure to combustion-generated particles were examined in neonatal rats. Neonatal rats undergo a rapid growth stage in their first 3 weeks of life, creating a perfect model to study this window of postnatal development. Ten-day old rats were exposed by whole-body inhalation 6 h/d for 3 consecutive days to laboratory-generated particles <0.1 µm in diameter, at a concentration of 250 µg/m3. Two hours prior to necropsy, they were injected with bromodeoxyuridine (BrdU), a nucleotide analog used to identify cells undergoing DNA synthesis, to enable the analysis of lung development and cell proliferation.
Developmental changes in the lung parenchyma, the terminal bronchioles, and the proximal alveolar region were examined (Figure 1). These changes were quantified by BrdU expression, as well as the appearance of new alveoli, observed through the presence of secondary alveolar septa (Figure 2). No significant changes in the terminal bronchioles or lung parenchyma were observed between the filtered air (FA) and PM-exposed animals. In contrast, within the proximal alveolar region, there were significantly less BrdU-labeled cells in animals exposed to PM versus FA. These findings suggested a significant impairment of cellular growth in a critical region, which could plausibly produce a dramatic decrease in the formation of new alveoli during postnatal lung development.
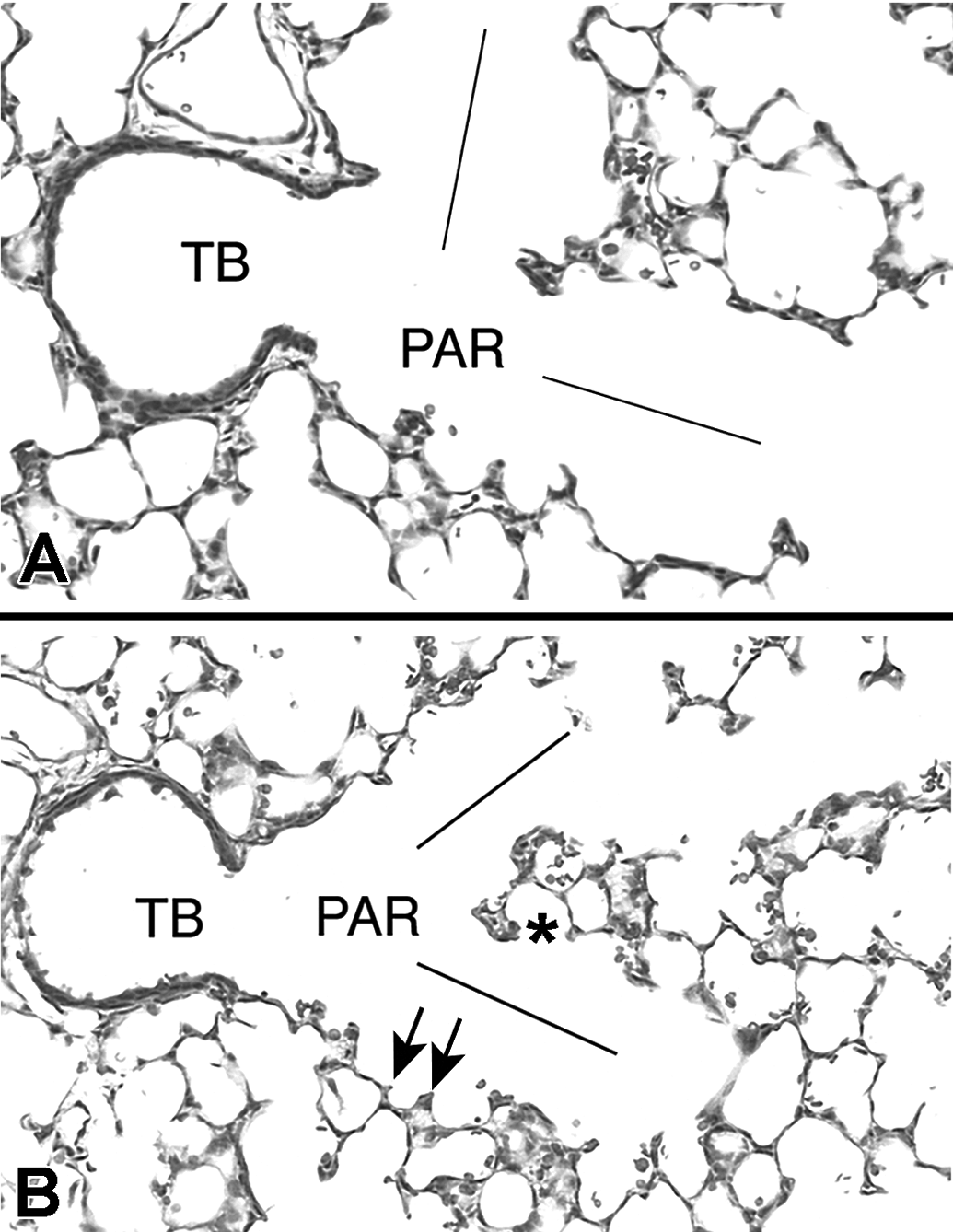
Light micrographs of the terminal bronchiole (TB) and proximal alveolar region (PAR) in the neonatal rat lung exposed to filtered air (A) or to soot and iron particles (B). The lines delineate the alveolar ducts arising from the terminal bronchiole comprising the PAR. Secondary alveolar septa (arrows) are typical in rapidly developing lungs. Alveolar outpocketings (*) show newly formed alveoli. Reproduced with permission from Pinkerton et al. 38
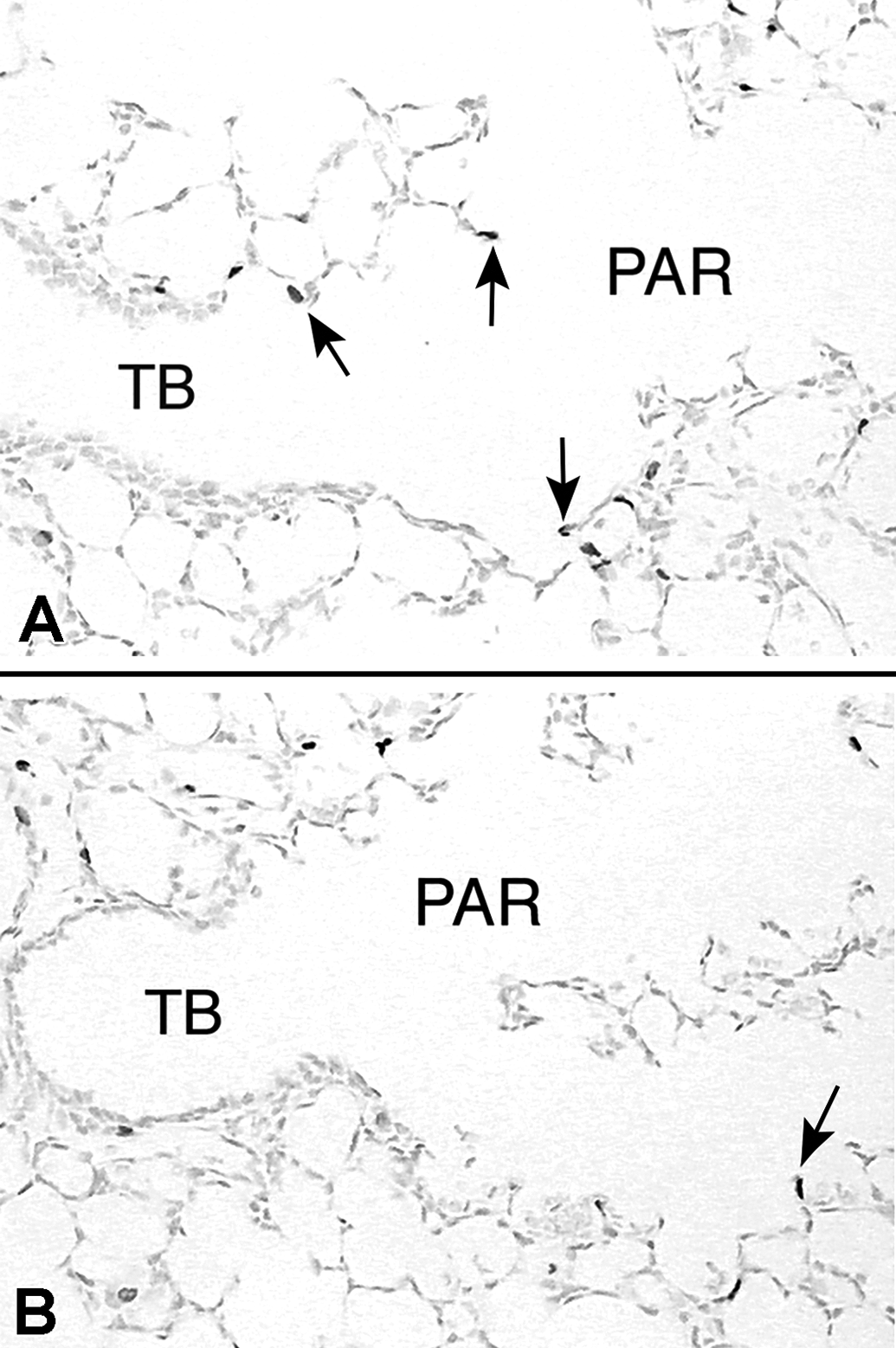
Lung-tissue sections stained immunohistochemically for the detection of bromodeoxyuridine (BrdU) from an animal exposed to filtered air (A) and an animal exposed to soot and iron particles (B). Cell nuclei incorporating BrdU are darkly stained (arrows). The nucleus of cells not undergoing DNA synthesis has a lighter appearance in these micrographs. Reproduced with permission from Pinkerton et al. 38
Regional PM Composition and Health Outcomes—Fresno, California
Recent epidemiologic studies have suggested a strong correlation between particle-induced health effects and particle size for the fine (<2.5 μm) and ultrafine (<0.1 μm) size fractions of PM. 39,40 These size fractions can be readily deposited deep in the lung to irritate the lung parenchyma and translocate into the blood stream. 41 Particle mass, composition, size, and number have all been implicated in PM toxicity in animal and human studies.
California’s Central Valley has some of the highest ambient PM concentrations found in the United States. The valley encompasses rich farming area and extensive urban development creating a unique setting for exposure to both geogenic and anthropogenic PM. 42 Agriculture, fires, wind-blown dust, diesel and gasoline engine exhaust, power plant emissions, and home heating all contribute to atmospheric PM in the Central Valley. 43 Since ambient particles come from different sources, they comprise a large number of compounds including organic carbon (OC), elemental carbon (EC), crustal components and metals. 44 The specific constituents or properties responsible for biological effects on the respiratory system have not been fully determined.
A study by Smith et al 45 found concentrated ambient particles (CAPs), composed of PM2.5 from the Central Valley, produced respiratory changes in healthy adult rats. In their study, Sprague-Dawley rats were exposed in three separate experiments to FA or PM2.5 during the fall of 2000 in Fresno, California. Exposures were for 4 h/d for 3 consecutive days. The 3-day mean mass concentration of particles ranged from just under 200 to over 800 μg/m3. Particle mass and elemental composition were determined over the course of each experiment. Following exposure, bronchoalveolar lavage (BAL) was collected to determine cell viability, total cell number, and cell differentials.
Physicochemical analysis of CAP samples from fall 2000 demonstrated PM enriched with ammonium nitrate, OC, EC and metals. Nitrate and OC were the predominant PM constituents, accounting for 80% of the total CAP mass (Figure 3). Bronchoalveolar lavage cell permeability is an indicator of decreased membrane integrity. Permeable (nonviable) BAL cells from rats exposed to FA ranged from 6% to almost 11%. In contrast, CAP exposure significantly increased the proportion of nonviable BAL cells over control animals (Figure 4). In addition, significant increases were also noted in the number of macrophages and neutrophils following PM exposure (P < .05). 45 These observations strongly suggest exposure to elevated concentrations of PM2.5 in Fresno is associated with mild, but statistically significant, cellular effects in the lungs of healthy adult rats.
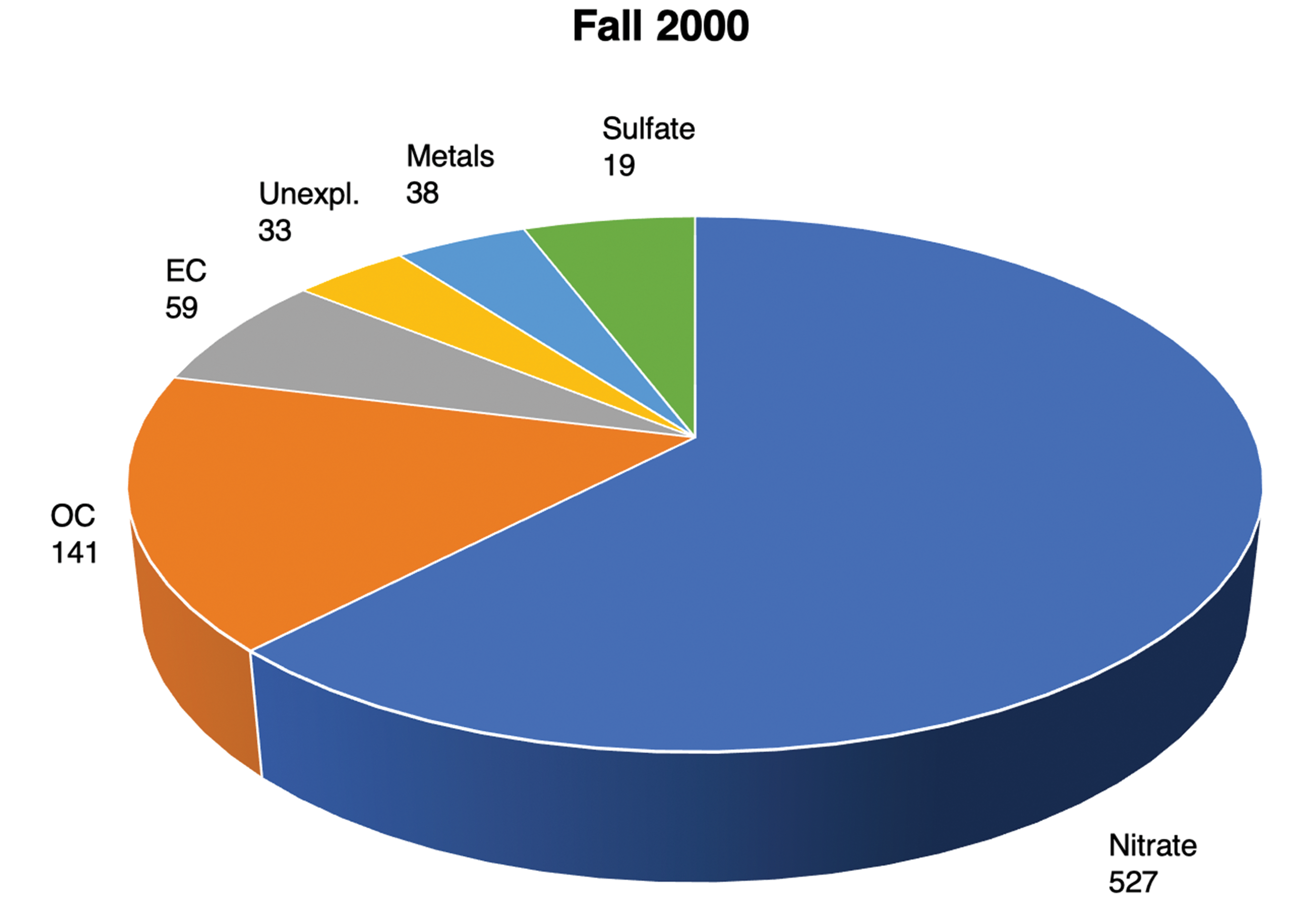
Chemical composition of concentrated ambient particles during fall 2000 exposures. The mass concentration (μg/m3) is shown for each species measured over the 3-day period of exposure. EC is elemental carbon, OC is organic carbon and the unexplained fraction (Unexpl) represents that portion of the total particle mass not accounted for by chemical analysis. Modified from Smith et al. 45
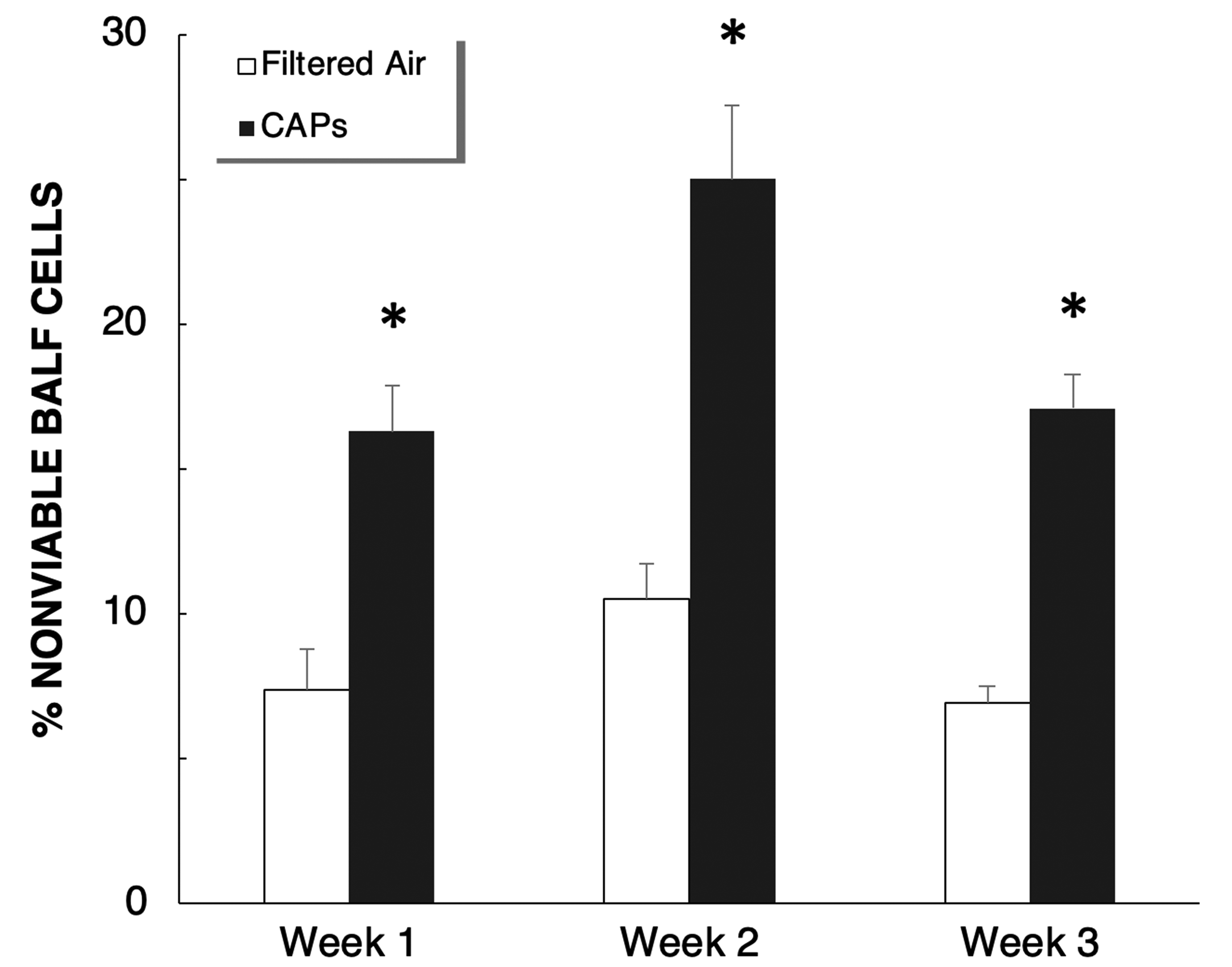
Percentage of nonviable (membrane permeable) cells in bronchoalveolar lavage fluid during the 3 weeks of fall 2000 exposures in Fresno. Error bars indicate standard error. * P <.05, compared with filtered air. Modified from Smith et al. 45
An inquiry arising in both toxicological and epidemiological studies is the specific chemical components leading to observed health effects. As noted in Figure 3, the dominant chemical components of the Fresno PM were nitrates and carbon. Therefore, we asked the question whether aerosolized nitrate and carbon particles alone are sufficient to cause enhanced cellular permeability and cell death. Under laboratory conditions, ammonium nitrate (150 µg/m3) and EC (100 µg/m3) were aerosolized to a respirable size fraction of PM to observe whether an acute 3-day exposure (6 h/d) similar to that conducted in Fresno would lead to injury in the lungs of healthy adult rats.
To measure the effect of cell viability and injury, our laboratory used a 3-dimensional imaging technique described by Postlethwait and colleagues to examine airway cell permeability. 46 One day following the end of this 3-day exposure, animals were deeply anesthetized and the lungs were perfused via the airways with ethidium homodimer-1 (EthD-1) solution that has a high binding affinity to DNA. Microdissection of the airways was done, followed by incubation with a second nuclear dye, YOPRO-1. The airways were subsequently examined by confocal microscopy to determine the presence of EthD-1-positive (injured) cells (Figure 5). In contrast to FA controls, numerous cells at airway bifurcations demonstrated increased permeability (EthD-1-positive cells), suggesting significant toxicity of ammonium nitrate and carbon black PM exposure.
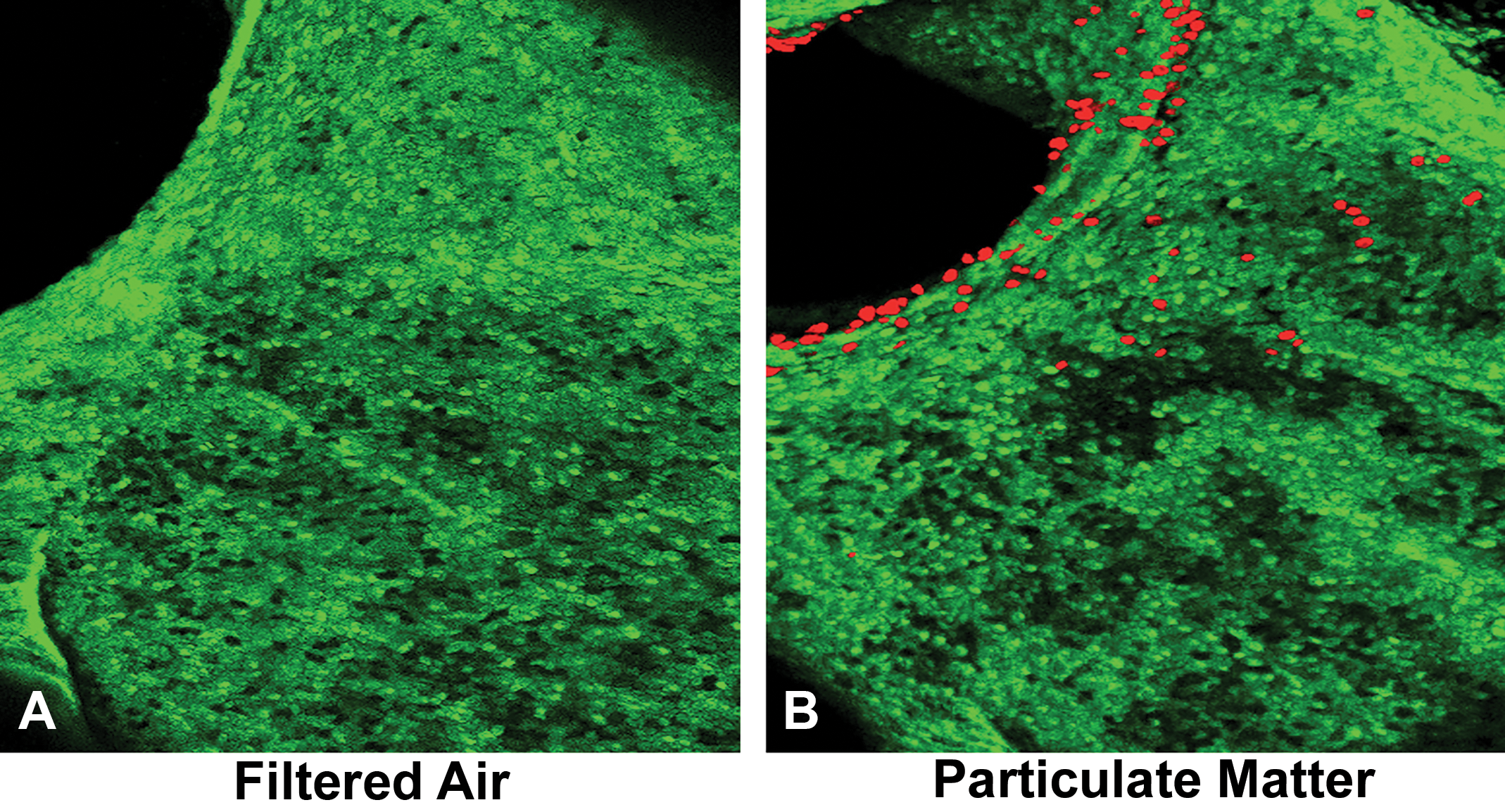
Three-dimensional image of airway bifurcations following exposure to filtered air (A) and particles (B). Epithelial cell permeability is indicated by ethidium homodimer-1 positive cells (in red).
Lung Pathology of Fresno Coroner Case Studies
Fresno has among the highest inhalable particle concentrations in the United States. 47 Individuals residing in this region are at increased risk of consistently high exposure to ambient PM. 45 Pinkerton et al 48 examined the lungs of 40 deceased Hispanic males, with a median age of 33 years who had lived an average of 16 years in Fresno County. Most of the subjects were farmworkers or in other blue-collar occupations. These individuals had been healthy and died of nonrespiratory causes.
Lung autopsy specimens were collected from the Fresno County Coroner’s Office. Two distinct airway paths into the apicoposterior and apicoanterior portions of the left upper lung lobe (Figure 6) were followed to examine the relationship between the retained carbonaceous and mineral dust in the lungs and remodeling of the airways. Parenchymal tissues and associated terminal and respiratory bronchioles of each airway path were analyzed. Pathological changes in the human lung were assessed by semiquantitative evaluation.
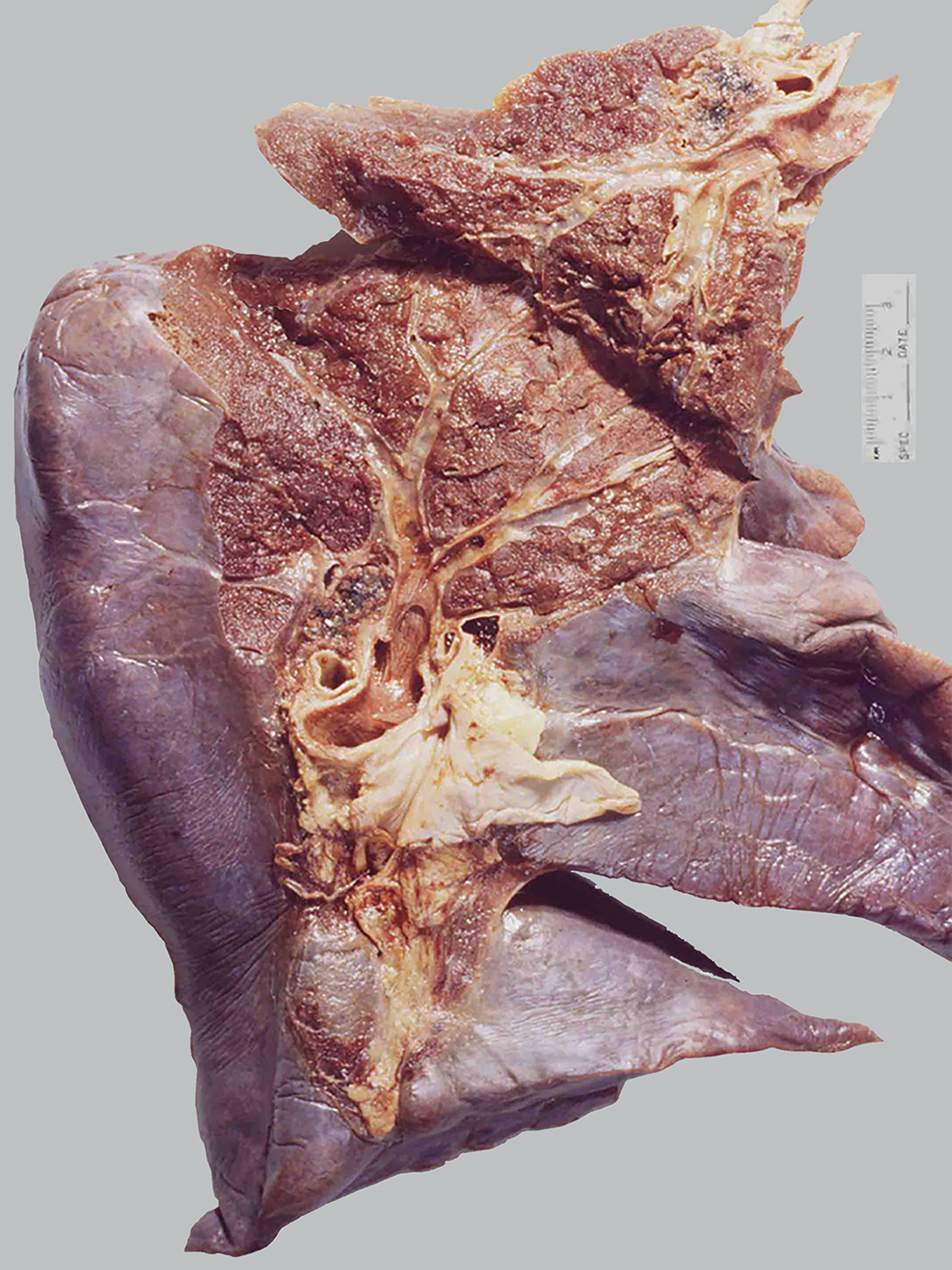
Airway dissection of the left human lung. Anatomically distinct airway paths beginning at the left mainstem bronchus are followed to the apical regions of the left upper lobe. Modified from Pinkerton et al. 48
Results demonstrated little evidence of visible particle accumulation in the larger conducting airways with the exception of bronchus-associated lymphoid tissues and peribronchial connective tissues. The relative absence of particles in the larger conducting airways likely reflected more rapid clearance of particles deposited in these regions (Figure 6).
In contrast, terminal and respiratory bronchioles arising from each pathway revealed varying degrees of wall thickening and remodeling (Figure 7A). Walls with marked thickening contained moderate-to-heavy amounts of carbonaceous and mineral dusts (Figure 7B). Wall thickening was also associated with increases in collagen and interstitial inflammatory cells, including dust-laden macrophages. These changes were analyzed and scored in order of respiratory bronchiole generation (Figure 8). A highly significant (P < .001) difference was noted in the degree of histologic changes for all features, including fibrosis, interstitial wall inflammation, amount of carbonaceous pigment, and amount of birefringent dust pigment found by respiratory bronchiole generation. First-generation respiratory bronchioles demonstrated the greatest changes compared to second-generation respiratory bronchioles, while second-generation respiratory bronchioles demonstrated greater changes compared to third-generation respiratory bronchioles (P < .001).
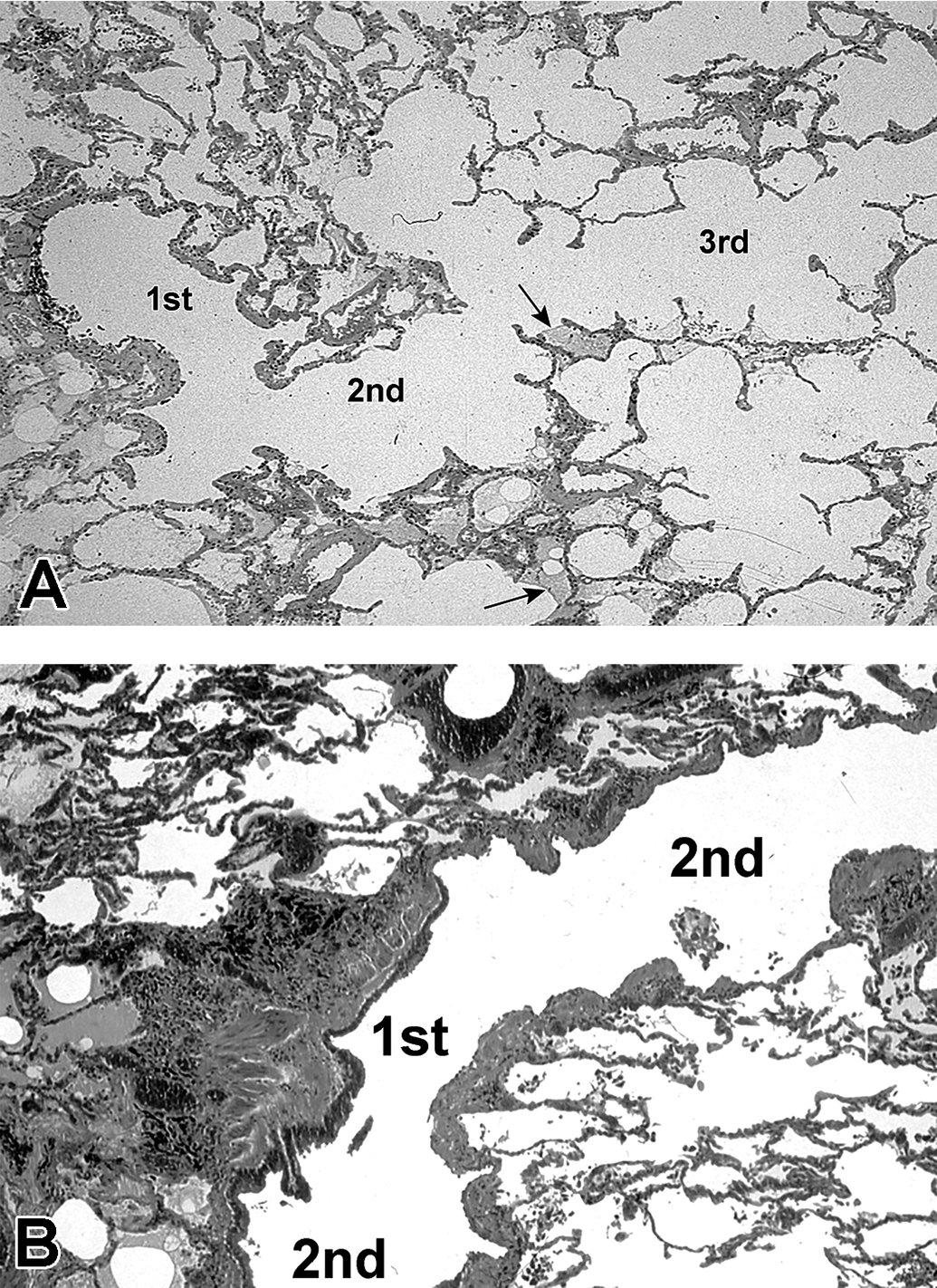
A, Contiguous first-, second-, and third-generation respiratory bronchioles in longitudinal profile from the lung of a Fresno County resident. Minor postmortem filling of some alveoli with edema fluid is noted (arrows) with no other pathological changes. B, First- and second-generation respiratory bronchioles from a Fresno County resident showing severe grades of pathologic change in the respiratory bronchioles. Specifically, there are increases in collagen, smooth muscle, and visible pigment. Also note that the changes are maximal in the first-generation respiratory bronchiole, with a progressive decrease in these tissue responses in more distal generations. Modified from Pinkerton et al. 48
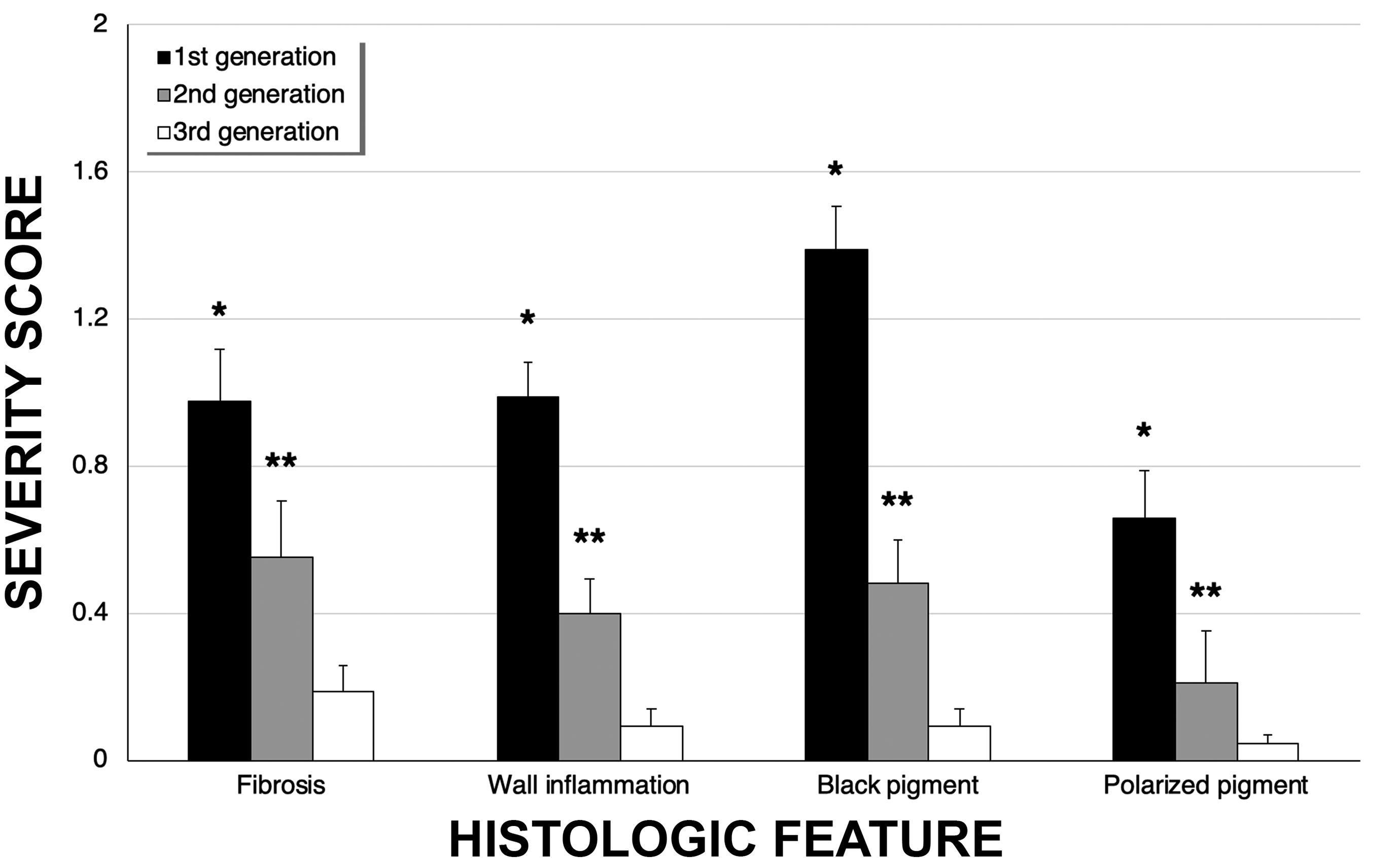
Bar graph showing the relationship between the severity scores for first-, second-, and third-generation respiratory bronchioles. *For all features, severity scores were significantly greater (P < .001) in first-generation respiratory bronchioles compared to the second and third generations. **All scores for second-generation respiratory bronchioles were significantly greater (P < .001) than that for the third-generation respiratory bronchioles. Adapted from Pinkerton et al. 48
In summary, cases from the Fresno Medical Examiner’s Office demonstrated terminal bronchioles and first-generation respiratory bronchioles to be the principal sites of deposition for ambient particles and associated tissue remodeling. This transitional respiratory zone of the lungs has unique anatomical and physiological features that can lead to enhanced particle deposition and possible retention. All medical examiner cases exhibited tissue remodeling associated with the presence of carbon black and birefringent particles. The association between fibrotic lesions and particle deposition/retention also strongly suggested that inhalation of ambient PM can lead to long-term structural changes in the lungs.
Conclusion
Multiple studies have shown exposure to PM can exert adverse effects on the heart and lungs in both animals and humans. This review demonstrates that the combined use of toxicological and epidemiological studies can serve to better elucidate those mechanisms by which PM causes injury. However, there continues to exist important data gaps that need to be addressed to more fully identify and understand the cardiopulmonary health effects of PM. For example, the identification of specific sources of PM by chemical composition derived from traffic, agriculture, wood smoke, or other unique sources could allow us to better understand those components that are most toxic. A greater implementation of personal monitoring devices could also aid in the elucidation of specific sources that exacerbate disease. New toxicological and human clinical studies could be subsequently designed based on the acquisition of data from such studies.
Definitive evidence of PM toxicity enables intervention, regulation, and policy-making that could minimize exposure, especially for susceptible individuals. Studies that investigate susceptible populations, such as children and the elderly could be further developed. While epidemiological studies can identify susceptible populations, toxicological studies could aid in the development of therapeutics. Together, these approaches could assist in establishing regulatory interventions to better protect the health of susceptible populations.
The current knowledge gaps of PM toxicity requires studies in toxicological, epidemiological, as well as human clinical studies. The power of their combined use in research provides a greater ability to define biological plausibility and to seek out solutions to protect public health.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The concepts and studies described in this review have been facilitated through the following: the National Institute for Occupational Safety and Health (NIOSH) grants U01 OH010969 and U54 OH07550 and the National Institute of Environmental Health Sciences (NIEHS) grants R01 ES025229, U01 ES027288, P30 ES023513, and P51 OD011107. S.M.M. is supported by NIEHS T32 ES007059. Authors appreciate the editorial assistant of Dr. Rona M. Silva in the preparation of this review.
