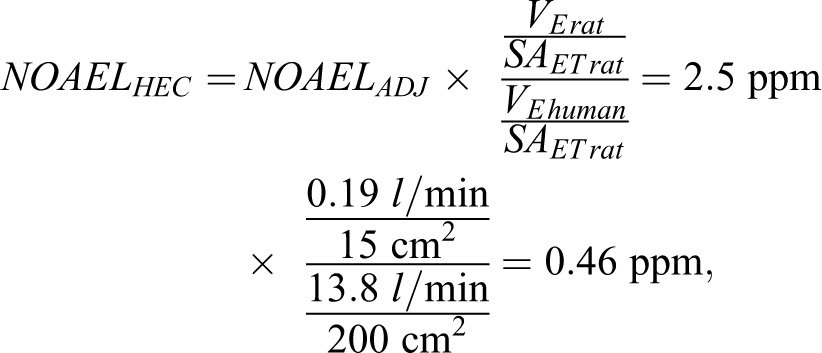Abstract
Nasal pathology can play an important role in the risk assessment process. For example, olfactory neuron loss (ONL) is one of the most sensitive end points seen in subchronic rodent hydrogen sulfide (H2S) studies and has been used by several agencies to derive health-protective toxicity values. Alternative methods that rely on computational fluid dynamics (CFD) models to account for the influence of airflow on H2S-induced ONL have been proposed. The use of CFD models result in toxicity values that are less conservative than those obtained using more traditional methods. These alternative approaches rely on anatomy-based CFD models. Model predictions of H2S delivery (flux) to the olfactory mucosal wall are highly correlated with ONL in rodents. Three major areas of focus for this review include a brief description of nasal anatomy, H2S-induced ONL in rodents, derivation of a chronic inhalation reference concentration for H2S, and the use of CFD models to derive alternative toxicity values for this gas.
Introduction
Chemical risk assessments performed by most regulatory agencies incorporate 4 steps: hazard identification, dose–response assessment, exposure assessment, and risk characterization. Completed hazard assessments can lead to a dose–response assessment and subsequent derivation of a toxicity value. Examples of toxicity values can include reference doses, reference concentrations (RfCs), or unit risks produced by the Integrated Risk Information System of the US Environmental Protection Agency (EPA); minimal risk levels for hazardous substances generated by the Agency for Toxic Substances and Disease Registry; and reference exposure levels developed by the State of California Office of Environmental Health Hazard Assessment, among others. Other agencies develop toxicity values for occupational exposures. These include threshold limit values developed by the American Conference of Governmental Industrial Hygienists and occupational exposure limits developed by a number of other agencies. An important step in the derivation of a toxicity value is identifying the critical effect and the appropriate point of departure (POD) associated with that effect. The POD is the dose–response point used in subsequent low-dose extrapolations. For noncancer effects, the POD can represent a benchmark dose, a no-observed-adverse-effect level (NOAEL), or a lowest observed-adverse-effect level (LOAEL). Derivation of a toxicity value often involves application of one or more uncertainty factors or modifying factors to the POD to derive a final value. 1 Commonly used uncertainty factors include default values of 10 for: (1) animal to human (ie, interspecies extrapolation), (2) variations in susceptibility within the human population (interhuman variability), (3) studies where only an LOAEL is reported, and (4) subchronic exposure. In cases where the scientific literature may be limited, a modifying factor can also be used (often = 3) to account for database uncertainties.
Nasal Anatomy
Broad reviews of nasal toxicology are available. 2,3 In some cases, nasal lesions are identified as the critical effect by risk assessors. The importance of performing nasal evaluations in support of chemical hazard assessments is embedded in some test guidelines. For example, some inhalation testing requirements of the Toxic Substances Control Act (eg, CFR 799.9135) require that multiple transverse sections of the nasal cavity be evaluated histologically. Likewise, Organization for Economic Co-operation and Development guidelines for inhalation toxicity studies also include recommendations for the evaluation of multiple transverse nasal sections. 4 These guidelines are a response to the 3-dimensional complexity of most mammalian nasal passages and the need to evaluate all nasal epithelial subtypes. There are 4 main epithelial subtypes found in the mammalian nasal cavity: squamous, respiratory, transitional, and olfactory. The distribution of these epithelial subtypes varies by anatomical location in the nose. 3 In most mammals, the olfactory epithelium is located in the dorsal or dorsoposterior aspect of the nasal cavity (Figure 1). The olfactory mucosa contains both the olfactory epithelium and the underlying lamina propria. The olfactory mucosa is characterized histologically by the presence of a pseudostratified columnar olfactory epithelium, mucin-secreting Bowman glands, and olfactory axons that traverse the lamina propria. The olfactory epithelium contains olfactory sensory neurons, glial-like sustentacular cells, and basal cells. Olfactory neurons comprise the major cell type found in the olfactory epithelium. Expression patterns of olfactory receptors help identify several subzones of olfactory neurons. Horizontal and globose basal cells in the olfactory epithelium give rise to new olfactory neurons and sustentacular cells.
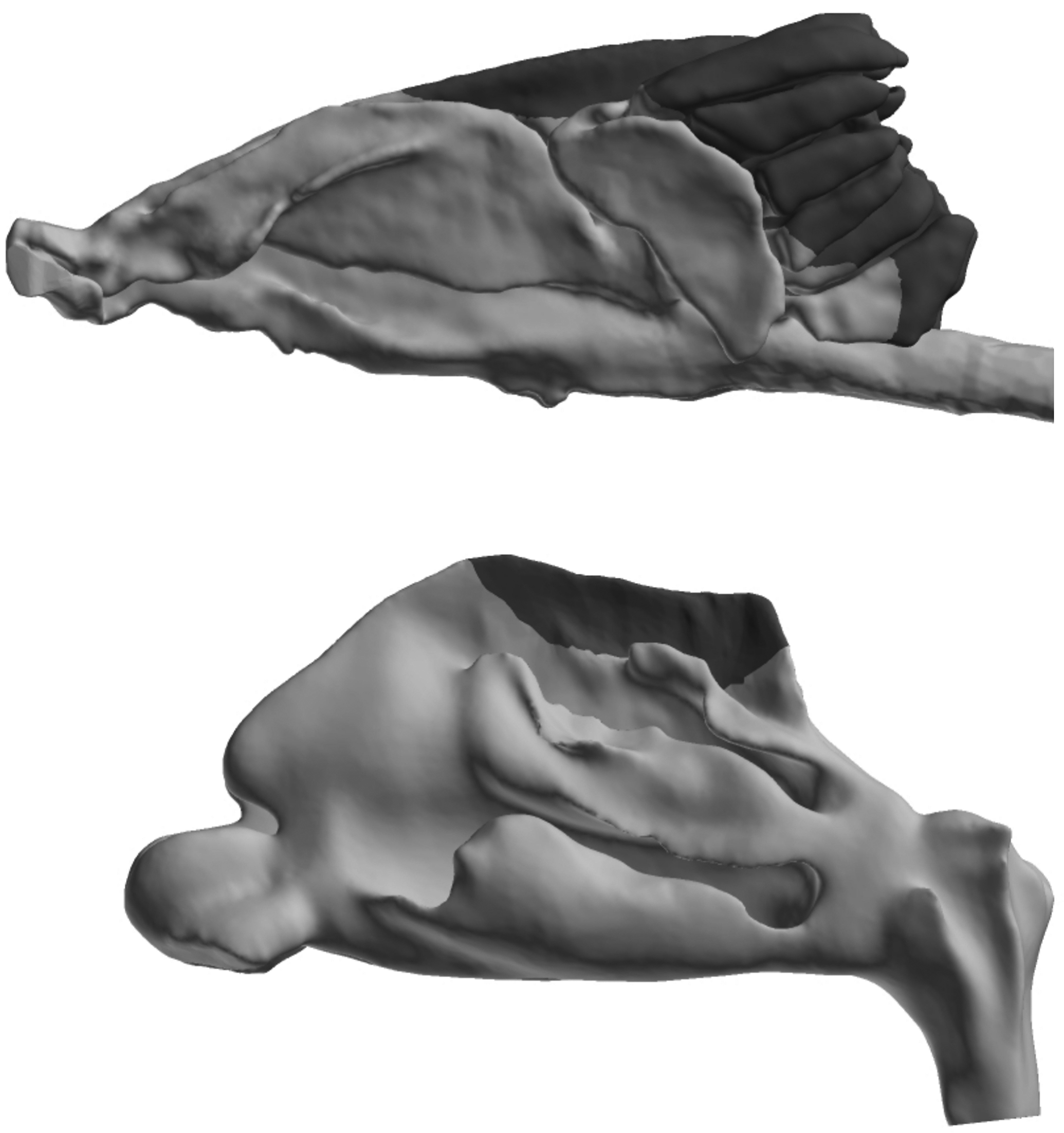
Lateral view of rat (top) and human (bottom) nasal airflow models (gray) with their corresponding olfactory regions (black). The nostrils are to the left, and the nasopharynxes are to the right. Figure compliments of Dr. Jeffry D. Schroeter.
Significant species differences exist with respect to the amount of the nasal cavity that is lined by the olfactory mucosa (Figure 1). Approximately 50% of the rodent nasal cavity is lined by olfactory mucosa, 5 whereas less than 5% of the macaque or human nasal cavity is lined with this mucosa. 6,7 The higher relative size of the rodent olfactory mucosa can result in increased delivery of inhaled chemicals to this epithelial subtype thereby predisposing rodents to olfactory lesions when compared to primates. Another important anatomical feature found in species with a well-developed sense of smell (eg, carnivores and rodents) is the presence of a dorsal meatus and a caudally located olfactory recess. 8 In carnivores, the dorsal meatus helps split the inspired air into olfactory and respiratory air streams. 8 The olfactory airstream is directed toward the sensory neuron-rich olfactory recess, while the respiratory stream largely bypasses the carnivore olfactory epithelium. 8 Airflow predictions in rodent airways also demonstrate these airflow patterns. The less complex human nasal cavity lacks both the dorsal meatus and the olfactory recess, and airflow in the human nose has different airstream patterns when compared to rodents and other species with a robust sense of smell (ie, microsmatic). Another important species difference between humans and rodents commonly used in toxicology studies is that rodents are obligate nasal breathers. This anatomical limitation may further predispose rodents to nasal (portal of entry) effects following chemical inhalation. Chemical exposure to the nasal epithelium during exhalation is also an important consideration as is nasal pathology from systemic exposure. 2
Collectively, these anatomical and physiologic features evolved to enhance the delivery of odorants to the sensory olfactory epithelium. 8–9 Other factors that influence nasal odorant delivery are the partition coefficient, aerodynamic diameter, and other physicochemical properties of the odorant. For example, highly soluble odorants deposit primarily along the canine dorsal meatus, whereas more volatile odorants are preferentially delivered to the olfactory recess. 9 Odorant deposition patterns in the canine nasal cavity are also influenced by inspiratory airflow rate—with higher inspiratory flow rates favoring olfactory deposition. 9 Uptake of other chemicals is likewise influenced by both vapor solubility (as measured by blood: air partition coefficient) and inspiratory flow rate. In addition, nasal delivery of particles is dependent upon inspiratory flow rate and particle size.
Olfactory Neuron Loss Following Hydrogen Sulfide Inhalation
Evolutionary adaptations in nasal anatomy and physiology can also lead to deleterious effects at the olfactory epithelium following xenobiotic exposure. One such example involves inhalation of hydrogen sulfide (H2S) gas. Inhalation of H2S is an important environmental, occupational, and public health concern. Toxic effects from H2S inhalation are characteristically dose related and most notably involve the nervous, cardiovascular, and respiratory systems. 10 In humans, inhalation of ≥100 ppm H2S for only a few minutes can result in incoordination, memory and motor dysfunction, and anosmia (so-called “olfactory paralysis”). Other olfactory effects including hyposmia, dysosmia, and phantom smells (phantosmia) have been reported in individuals recovering from H2S exposure. 10 In animals, a single 3-hour exposure to either 200 or 400 ppm H2S or 5 consecutive daily 3-hour exposure to ≥80 ppm H2S resulted in olfactory mucosal necrosis in the rat dorsal medial meatus. 11 Respiratory epithelial metaplasia was also observed in this study along the lateral wall of the ventral meatus following a single 3-hour exposure to ≥80 ppm H2S. 11 This metaplastic lesion was no longer present in animals that were exposed repeatedly to H2S. 4 Brenneman et al 11 also evaluated recovery of the olfactory mucosa at 2 and 6 weeks following the 5-day exposure. They found the olfactory epithelium was restored to its normal thickness following 6 weeks of recovery, although some abnormalities including small cysts lined by regenerating olfactory epithelial cells were noted in the olfactory epithelium. Replacement of olfactory neurons is dependent upon a functional basal layer in the olfactory epithelium. The rat olfactory epithelium will regenerate from the basal cell layer within 30 days of full-thickness injury. The H2S-induced effects on the olfactory mucosa have been replicated in other short-term rodent inhalation studies. 12,13 Inamura and coworkers 12 reported that rat olfactory neurons affected by H2S inhalation express NAD(P)H quinone dehydrogenase 1 (NQO1). The NQO1 reduces quinones and may lower cellular production of free radical and toxic oxygen metabolite. 12 The toxicologic significance of this finding with respect to nasal toxicity of H2S remains unknown. Short-term inhalation studies in rats using microarray analysis of tissues collected from affected sites show that H2S alters expression of genes associated with a variety of biological processes including cell cycle regulation, protein kinase regulation, and cytoskeletal organization and biogenesis. 14 Olfactory mucosal necrosis is also seen in subchronic H2S inhalation studies performed in B6C3F1 mice 15 and several strains of rats (Figure 2). 15,16 An NOAEL for lesions in the olfactory mucosa of 10 ppm has been identified in these subchronic studies. 16
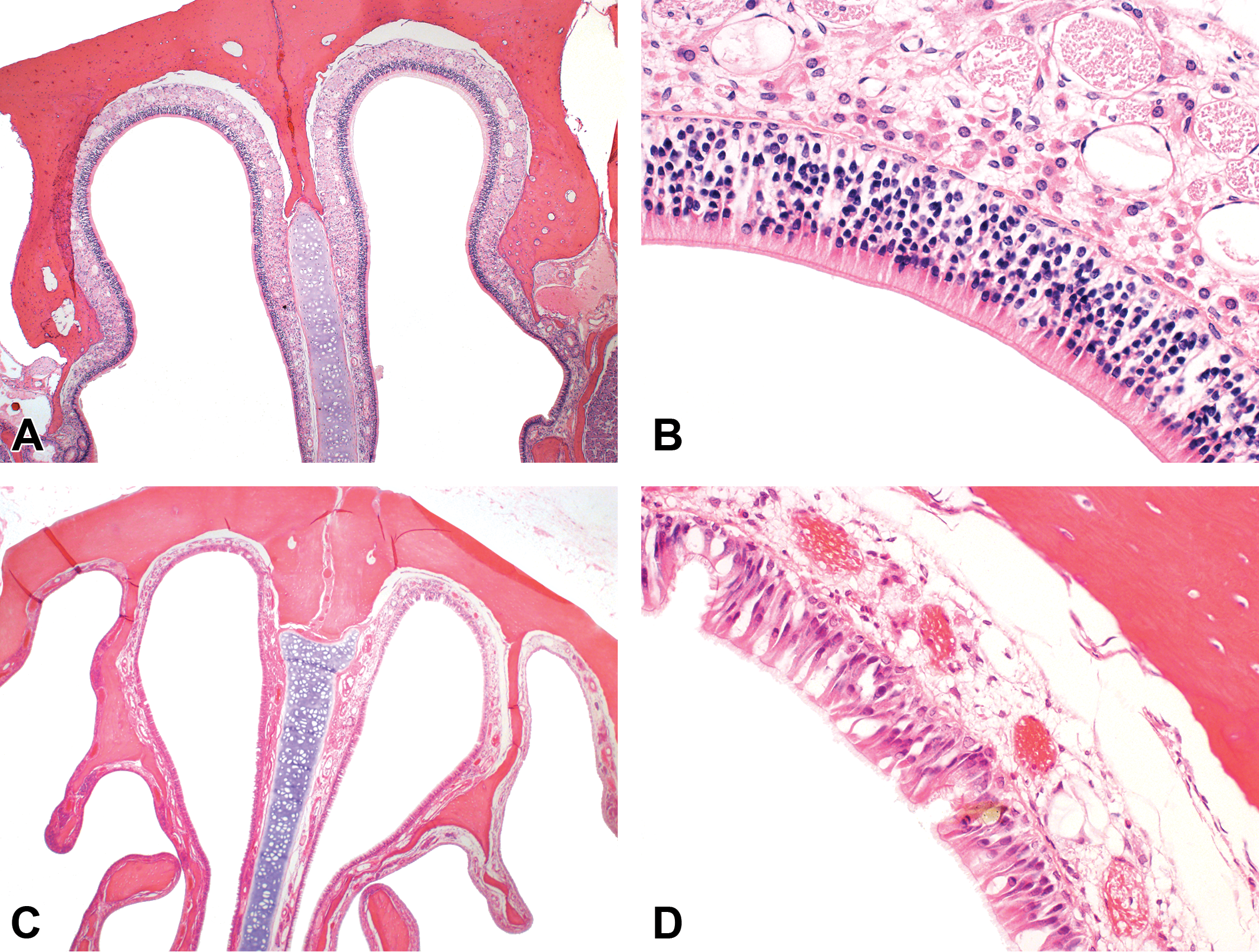
Olfactory neuronal loss in the rat dorsal medial meatus following subchronic (6 hr/d, 5 d/wk, 13 weeks) exposure to either air (A and B) or 80 ppm hydrogen sulfide (H2S; C and D). Original objective either ×4 (A and C) or ×40 (B and D).
Use of Nasal lesions in the Derivation of an RfC for H2S
Nasal effects seen in subchronic rodent studies were used to derive the inhalation RfC. An RfC is an estimate of a continuous inhalation exposure to the human population, including sensitive subgroups that is likely to be without an appreciable risk of deleterious effects during a lifetime. 17 According to the US EPA, it “can be derived from a NOAEL, LOAEL, or benchmark concentration, with uncertainty factors generally applied to reflect limitations of the data used.” 17
The RfC for H2S developed by the US EPA in 1994 relied on a subchronic study sponsored in the early 1980s by the then Chemical Industry Institute of Toxicology (CIIT). 15 An abstract describing this study 18 reported that subchronic exposure of B6C3F1 mice to 80 ppm H2S resulted in nasal mucosal inflammation and 30 ppm represented a subchronic NOAEL for this lesion in mice. Nasal lesions were not reported in rats following H2S exposure in the CIIT study. 18 A retrospective reassessment of the nasal and lung histologic specimens obtained from the subchronic CIIT inhalation study revealed the presence of olfactory neuronal loss in both species. 15
The RfC for H2S was subsequently updated by the US EPA in 2003. 19 The NOAEL of 10 ppm in rats identified by Brenneman et al 16 was used by the US EPA as the POD in the derivation of the revised RfC. 19 The US EPA adjusted the NOAEL (NOAELADJ) reported by Brenneman et al 16 to account for continuous exposure (24 hours/d vs 6 hours/d) resulting in an NOAELADJ = 2.5 ppm. A human equivalent concentration NOAEL (NOAELHEC) was subsequently calculated using US EPA standard methods for chemicals with a gas-induced respiratory effect in the nasal (ie, extrathoracic) region and the following formula:
Where VE = minute volume and SAET = surface area of the extrathoracic region. The
The RfC of 1.5 ppb (2.1 µg/m3) was subsequently determined by applying an aggregate uncertainty factor of 300 (representing a factor of 3 for interspecies extrapolation, 10 for interhuman variability, and 10 for subchronic exposure) to the NOAELHEC. 19 Some individuals 20 have criticized the US EPA derivation of the RfC questioning whether effects seen by Brenneman et al 16 at the LOAEL should be considered adverse, since the effects seen in this study were likely reversible. H2S-induced olfactory neuron loss in rodents has been considered an adverse effect by other organizations, including the US National Academies in their derivation of occupational exposure limits for submariners. 21 In addition, the National Academies has recently defined an adverse effect as “A biological change in an organism that results in an impairment of functional capacity, a decrease in the capacity to compensate for stress, or an increase in susceptibility to other influences.” 22 Chemical-induced olfactory neuron loss would be considered an adverse effect under this definition. Additional concerns regarding the appropriateness of the time and dosimetric adjustments used by the agency have also been raised by critics of the RfC. 20
Comparison of the RfC value with measurements of airborne H2S in different environments yield interesting results. Ambient H2S concentrations from natural sources have been estimated to be between 0.11 and 0.33 ppb. 10 Measurements of H2S in ambient air in some locations, however, can greatly exceed the current inhalation RfC. One of the best studied locations is the Rotorua geothermal field area in New Zealand. Residential properties in this location have had a wide range of indoor H2S air concentrations that often exceed the RfC by several orders of magnitude. 10 Measurement of H2S in exhaled breath at concentrations that exceed the RfC has been reported in studies that evaluated exhaled H2S as a biomarker for asthma, 23 chronic obstructive pulmonary disease, 24 inflammatory bowel diseases, 25 and sepsis 26 among others.
Alternative Approaches to Deriving an RfC
An alternative NOAELHEC value has been proposed that relies on the use of anatomically accurate computational fluid dynamic (CFD) models developed for human and rat noses. 27 Histologic cross-sections and computed tomography or magnetic resonance images of the nasal cavity are often used to create nasal CFS models and are used to predict nasal airflow. 28 Chemical-specific nasal CFD models consider solubility, vapor pressure, air concentration, and air flow rates to predict chemical delivery to different parts of the nasal cavity. Development of these CFD models for H2S was accomplished in several steps. An initial effort 29 showed that the distribution of olfactory epithelial lesions seen in rats following subchronic exposure correlated with regional delivery of H2 S to the olfactory epithelium. An important limitation of the Moulin et al study, 29 however, was that a range of hypothetical nasal extraction values for H2 S (20%, 40%, and 80% of the inspired H2S would be removed by the nasal cavity) were used in lieu of actual nasal extraction values. A later refinement of the H2S CFD model 27 used measured nasal extraction values that were obtained in anesthetized rats exposed to 10, 80, or 200 ppm H2S under constant unidirectional inspiratory flow rates of 75, 150, and 300 mL/min. Schroeter and colleagues 27 reported that nasal extraction of H2S was dependent on both inspired H2S concentration and air flow rate with increased nasal extraction occurring when air H2S concentrations or air flow rates were low. The revised CFD model was then used to estimate H2S flux (ie, delivery in pmol/cm2·s) to the rat airway walls. A human equivalent flux to the olfactory epithelium was then predicted using a human CFD model. Flux values in the human model were used to back calculate an NOAELHEC for H2S. These modeling efforts assume similar sensitivity between the human and the rodent olfactory epithelium. The calculated NOAELHEC derived for H2 S based on the CFD models was estimated to be 5.4 ppm—or approximately 1 order of magnitude higher that than calculated by the US EPA on the basis of default dosimetry models. 27 This higher CFD-derived NOAELHEC would also lead to a less restrictive RfC. It is unknown whether the US EPA will consider this alternative modeling approach in future revisions of the H2S RfC. A subsequent modeling effort 30 considered anatomical variation among adult (n = 5) and children (n = 2) noses. This study showed that despite predicted differences in olfactory airflow, H2S dosimetry in the olfactory region was generally similar among individuals. 30 These results may provide a basis for decreasing the size of the uncertainty factor needed to account for sensitive subpopulations.
Conclusions
This mini review highlights the application of rodent nasal pathology to the development of a chronic inhalation RfC for H2S. Although the focus of this review was the RfC, other agencies have developed toxicity values for H2S that were also based on nasal lesions seen in rodents (Table 1). Noncancer inhalation RfCs for other chemicals have also been based on nasal effects including damage to the olfactory epithelium (Table 2). As was the case with H2S, dosimetry-based alternative approaches to deriving chronic inhalation RfCs have been developed for some of these chemicals including acetaldehyde 31 and acrolein. 32 In addition, derivation of occupational toxicity values increasingly rely on the use of CFD and other computational models to predict chemical delivery to the human nose, 33 and CFD models of the complete respiratory tract have been developed 34 to support other risk assessment methods. Although nasal pathology plays a critical role in chemical hazard assessment, it does have some important limitations. Histologic evaluation of the olfactory epithelium provides no direct information concerning the functional status of the epithelium. Changes in olfactory function (eg, odor threshold or discrimination) may go undetected in some studies, despite the presence of widespread nasal lesions. 35 Conversely, more severe functional effects are anticipated when ablation of the olfactory epithelium occurs. 36 It is currently unknown whether the rodent olfactory epithelium that recovers from H2S exposure maintains normal abilities to detect or discriminate odorants. This consideration is important since acute injury to the olfactory epithelium following acute exposure to H2S is zonal 5 as is distribution of olfactory binding proteins and receptors needed for normal olfaction.
Inhalation Toxicity Values Developed for H2S by Different Agencies.a
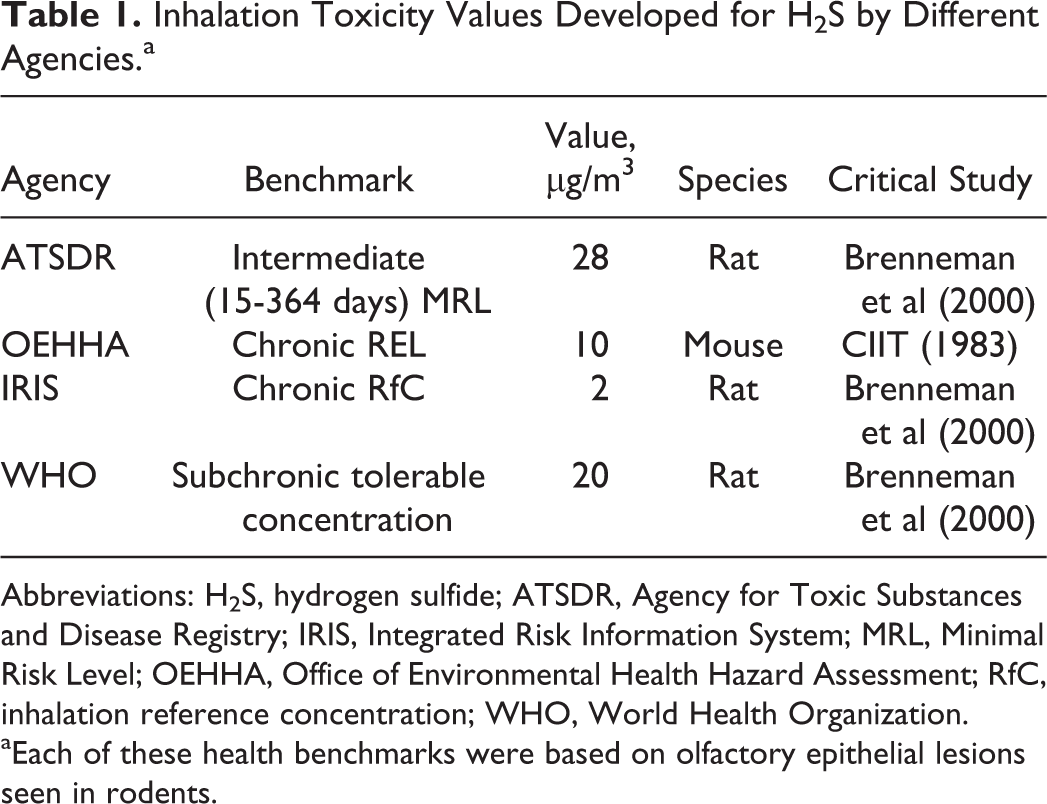
Abbreviations: H2S, hydrogen sulfide; ATSDR, Agency for Toxic Substances and Disease Registry; IRIS, Integrated Risk Information System; MRL, Minimal Risk Level; OEHHA, Office of Environmental Health Hazard Assessment; RfC, inhalation reference concentration; WHO, World Health Organization.
aEach of these health benchmarks were based on olfactory epithelial lesions seen in rodents.
Example Chemicals From EPA IRIS With Chronic Inhalation RfCs Based on Nasal Lesions Seen in Rodents.a
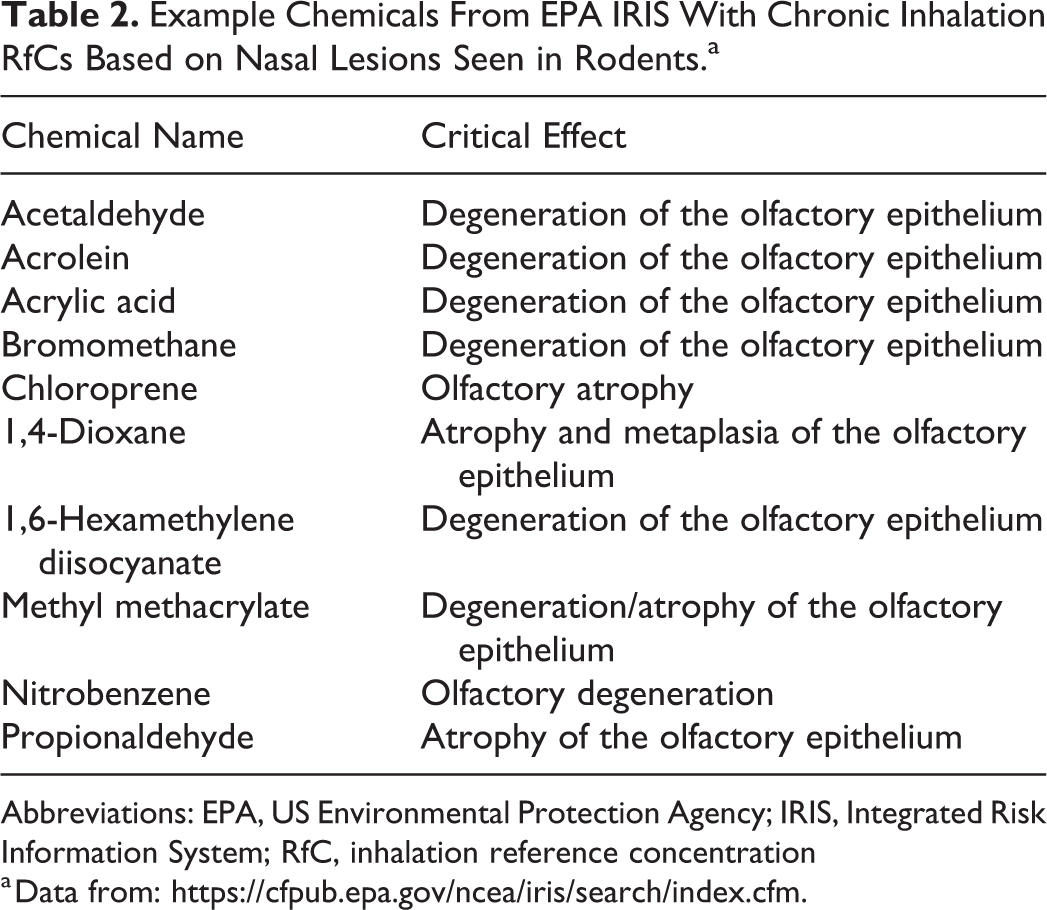
Abbreviations: EPA, US Environmental Protection Agency; IRIS, Integrated Risk Information System; RfC, inhalation reference concentration
a Data from: https://cfpub.epa.gov/ncea/iris/search/index.cfm.
Footnotes
Acknowledgments
The authors wish to thank Melanie Foster and Deepa Rao for their reading of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.