Abstract
Epidemiological studies report associations between air pollution (AP) exposures and several neurodevelopmental disorders including autism, attention deficit disorder, and cognitive delays. Our studies in mice of postnatal (human third trimester brain equivalent) exposures to concentrated ambient ultrafine particles (CAPs) provide biological plausibility for these associations, producing numerous neuropathological and behavioral features of these disorders, including male-biased vulnerability. These findings raise questions about the specific components of AP that underlie its neurotoxicity, which our studies suggest could involve trace elements as candidate neurotoxicants. X-ray fluorescence analyses of CAP chamber filters confirm contamination of AP exposures by multiple elements, including iron (Fe) and sulfur (S). Correspondingly, laser ablation inductively coupled plasma mass spectrometry of brains of male mice indicates marked postexposure elevations of Fe and S and other elements. Elevations of brain Fe and S in particular are consistent with potential ferroptotic, oxidative stress, and altered antioxidant capacity-based mechanisms of CAPs-induced neurotoxicity, supported by observations of increased serum oxidized glutathione and increased neuronal cell death in nucleus accumbens with no corresponding significant increase in caspase-3, in male brains following postnatal CAP exposures. Understanding the role of trace element contaminants of particulate matter AP as a source of neurotoxicity is critical for public health protection.
Introduction
Numerous epidemiological studies report associations of various metrics of air pollution (AP) exposures with characteristics of neurodevelopmental disorders and neurodegenerative diseases. These have included reports of increased risk for autism spectrum disorder (ASD), attention-deficit/hyperactivity disorder (ADHD), slower cognitive development in children, schizophrenia, and a more rapid cognitive decline with age in adults, as summarized in a series of recent reviews. 1 -10 Several studies have also reported neuropathological changes in response to AP exposures, which to date, include reductions in brain white matter (myelin) and corresponding brain disconnectivity, reductions in gray matter volume, and altered basal ganglia structure. 11 -16 Air pollution as a risk factor for neurodevelopmental disorders and neurodegenerative diseases would carry heavy social and economic burdens. Annual US costs just for ASD are US$11 to US$60 billion according to the Centers for Disease Control, while estimated costs for Alzheimer’s are US$290 billion (Alzheimer’s Association).
Biological plausibility for the neurotoxic effects of AP has also begun to emerge from studies using animal models. 6,17 -19 Based on the fact that for different particle sizes of the same chemistry, ultrafine particulate (UFP) matter is considered the most reactive component of AP per unit mass because of its greater surface area/mass ratio available for adsorption of organic and inorganic contaminants, 20 we have examined the impact of developmental exposures to concentrated ambient ultrafine particles (CAPs) on brain and behavior in mice since 2013 as reviewed here. Exposures were carried out from postnatal days (PND) 4 to 7 and 10 to 13 for 4 h/d. This period is considered analogous to human third trimester brain development 21,22 and is characterized by significant neuro- and gliogenesis. 23 Mass CAPs concentrations across these studies to date have ranged from 22 to 121 µg/m3; levels at the lower-mid part of this range are similar to PM2.5 levels in global cities. 24 -26
These postnatal (PN) CAPs exposures in mice resulted in marked neuropathological effects, as well as behavioral dysfunctions, with these effects occurring predominantly in males, as previously reported. 27 -33 One dramatic neuropathological effect was a male-specific persistent ventriculomegaly (ie, enlarged lateral ventricles) occurring at 96 µg/m3 and observed even at PND 270, that is, the last time point examined. Subsequent exposure cohorts revealed that postnatal CAPs produced male-specific ventriculomegaly even at 44 μg/m3, the lowest levels yet examined with sufficient sample sizes to date. In addition, marked reductions in size and myelination of the corpus callosum (CC), the largest white matter tract of the brain and a structure critical to interhemispheric connectivity, were found. Male biased microglial activation (inflammation) was found in CC (340% of control) as late as PND 270, the last time point examined to date. A profile of male-specific elevated brain glutamate, with consequent excitatory–inhibitory imbalance was also observed. Behavioral impairments, as assessed to date, reveal increased impulsivity/cognitive inflexibility and altered social interactions, including reduced social preference for a novel mouse versus a cagemate. 28,31,31,34
Collectively, these findings following postnatal CAPs exposures suggest that AP can reproduce numerous characteristics of a number of neurodevelopmental disorders, including the enhanced vulnerability of males to such conditions. While ASD has unique features, it also shares features with several other neurodevelopmental disorders. 35 -40 Autism spectrum disorder and early childhood-onset schizophrenia 41 -44 share abnormalities of glutamatergic systems, 45 -47 inflammatory mechanisms, 48 and neuropathological features, including ventriculomegaly and disconnectivity. 49 -51 Attention-deficit/hyperactivity disorder occurs at higher rates in children with ASD. 52 Both involve increases in impulsive behavior and are considered disorders of hemispheric disconnectivity. 36 Like ASD, ADHD is male-biased and its rates have increased significantly from 7.8% in 2003 to 9.5% in 2007 and 11.0% in 2011 to 2012 according to the Centers for Disease Control (CDC). Attention-deficit/hyperactivity disorder can also include ventriculomegaly 53 and alterations in glutamatergic function, 54 brain white matter, 55 brain connectivity, 56 and inflammation. 57 Thus, the neurotoxic consequences of AP seen in these experimental models may contribute risk not only for ASD but for other neurodevelopmental disorders as well. Differences in timing of AP exposure during pregnancy could contribute to the heterogeneity and subgroups of ASD 58 or other neurodevelopmental disorders.
A collaborative study carried out at NYU (Tuxedo, New York) 59 -61 obtained brains of mice following gestational (GE, days 0.5-16.5) fine/UFP CAPs exposures, that is, a 1st + 2nd trimester human brain equivalent time point. The GE exposures produced ventriculomegaly in both sexes (see 31,59,60 ) at PND 11 to 15. In contrast to postnatal CAPs, GE exposures increased rather than decreased CC size and myelination, effects that were still present at PND 57 to 61, the last time point available for examination. Increased CC size and myelination are actually seen early in the ASD brain, 62 followed only later by reductions in size and myelination, as seen with our CAPs exposures. 63,64 The findings produced by GE CAPs exposures to date confirm that AP-induced neurotoxicity is not unique to exposures carried out in Rochester, and despite the heterogeneity of AP compositions, similarities exist in their potential to disrupt white matter development in the central nervous system (CNS). In addition, they reconfirm sex differences in outcome and also underscore the importance of timing of AP exposures during brain development.
The observations of neuropathological effects from particulate AP exposures at 2 different sites also suggested that similar AP components could be involved in this neurotoxicity. Consequently, we carried out studies to determine whether the carbon-based nanoparticle itself, that is, the base of UFPs, or perhaps a known inflammatory component of AP, for example, endotoxin, 65 could produce such effects by generating atmospheres of each and exposing mice to them alone or in combination. However, neither exposure alone nor the 2 combined were able to reproduce the developmental neurotoxicity of postnatal exposure to ambient CAPs. 65 Trace element contaminants were subsequently considered as another potential source of postnatal CAPs neurotoxicity. Subsequent analyses, elaborated here, reveal that trace element contaminants of AP may be a likely source of its neurotoxic consequences, based on the presence of known elemental neurotoxicants in the exposures and in excess in brain following such exposures. The increases in these elements, which can be taken up into brain from bloodstream but also via the olfactory and trigeminal nerves, 66 -68 moreover, suggest specific mechanisms of CAP-induced neurotoxicity via ferroptosis, as schematized in Figure 1, and their actions in brain would be likely to influence pathways consistent with reported biomarkers of neurodevelopmental disorders. 69 -75 In addition, data across different exposures were utilized to determine preliminary concentration–effect functions for various outcomes where available.
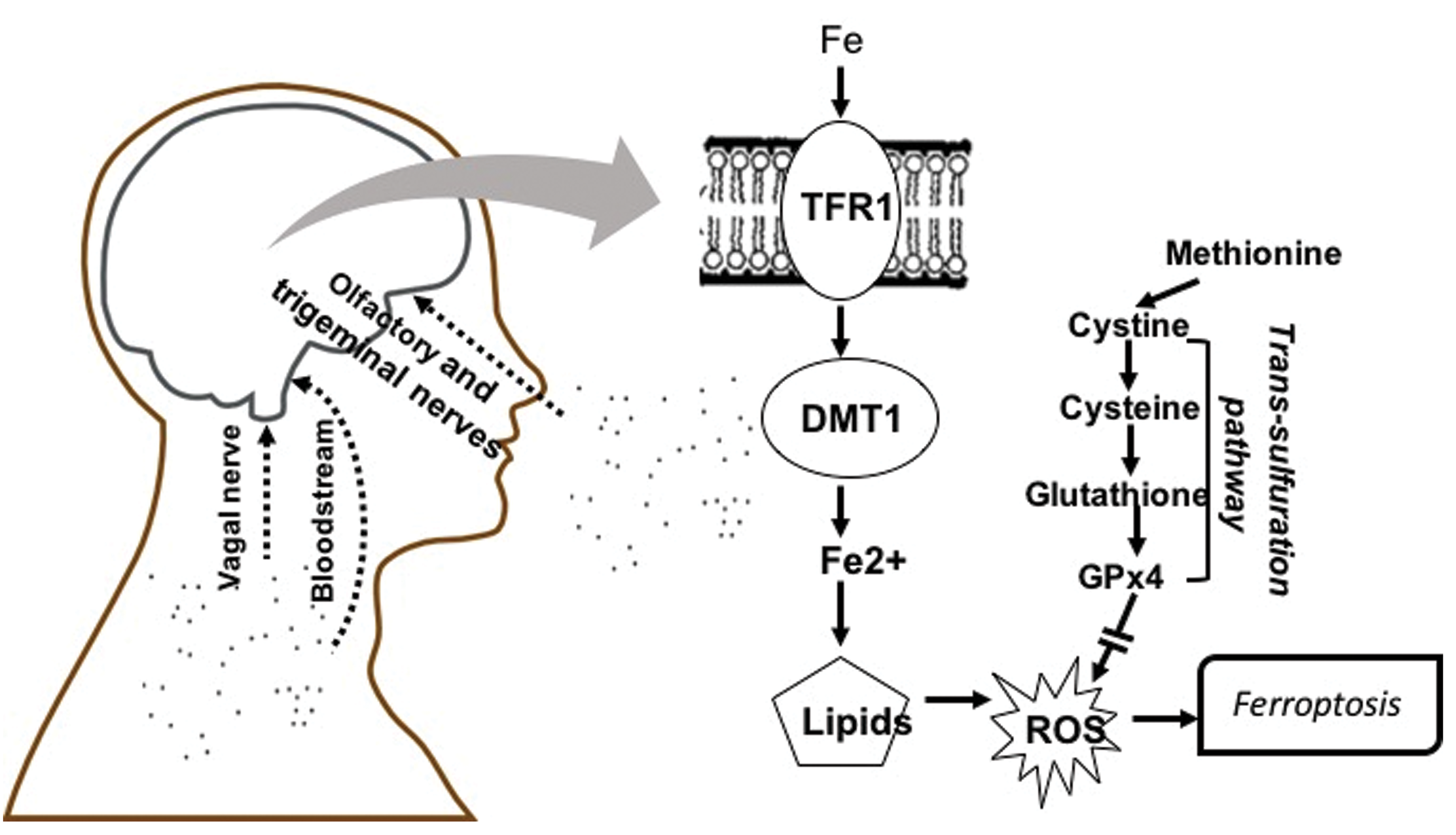
Schematic diagram showing entry routes of ultrafine particles to brain via olfactory and trigeminal nerves, vagal nerve, and bloodstream. The consequent elevation of brain metals as contaminants of ultrafine particles, for example, Fe, raise the potential for ferroptotic mechanisms of brain neuropathology associated with air pollution. Both transferrin-bound and nontransferrin (labile) Fe can be released into cells with Fe3+ (ferric iron) reduced to Fe2+ (ferrous iron) followed by movement into cytosol via the divalent metal transporter 1. Potential increases in the redox active Fe2+ can lead to reactive oxygen species via the Fenton reaction and lipid peroxidation. Ferroptosis is negatively regulated by glutathione and glutathione peroxidase 4, derived from the trans-sulfuration pathway, which if reduced can facilitate ferroptosis, an iron-and lipid peroxidation-dependent form of cell death. DMT1 indicates divalent metal transporter 1; GPx4, glutathione peroxidase 4; ROS, reactive oxygen species; TRF1, transferrin.
Methods and Materials
Animals and Postnatal CAP Exposures
Male and female C57BL6/J mice 8 weeks of age from Jackson Laboratories (Bar Harbor, Maine) were acclimated to a housing room for 1 week and then bred monogamously for 3 days. Pregnant dams were singly housed with litters until weaning on PND 21. To preclude litter-specific effects, only single pups/sex/litter were used in all of these studies. All mice used in this study were treated humanely and with regard for alleviation of suffering and approved by the University of Rochester Institutional Animal Care and Use Committee.
Pups were placed singly in compartmentalized whole-body exposure chambers. They were exposed to filtered air (Air) or CAPs using the Harvard University Concentrated Ambient Particle System (HUCAPS) fitted with a size-selective inlet and a high-volume UFP (≤100 nm) concentrator (10-20x) that takes in outdoor air at 5000 L/min and concentrates ambient UFP, as previously described. 27 -30 Exposures lasted for 4 hours per day from 0700 to 1100 for 4 days per week from PND 4 to 7 and PND 10 to 13, with exposure timing corresponding to peak vehicular traffic outside the intake valve of the HUCAPS instrumentation. A condensation particle counter (TSI 3022A) provided particle counts. Mass concentration was calculated using idealized particle density (1.5 g/cm3). A scanning mobility particle sizer was used to determine particle size distribution and median particle diameter + geometric standard deviation. Flow of CAPs-enriched and filtered air was maintained at 35% to 40% relative humidity and 77°F to 79°F. Average exposure mass concentrations from these exposures were 22, 44, 53, 96, and 121 μg/m3.Teflon filter samples from the breathing zone inside the exposure chamber were taken for chemical analysis of the constituents. Teflon filter samples were analyzed by the Desert Research Institute (Reno, Nevada) in a temperature-controlled package at below 4° C for elemental analyses by an energy dispersive X-ray fluorescence analyzer (Epsilon 5; PANalytical Company, Almelo, the Netherlands) as previously described. 76
Brain Pathology
To preclude any effect of anesthetics on measured end points, brains were extracted following decapitation without sedation and placed into 4% paraformaldehyde for 24 hours and then cryoprotected in 30% sucrose until they sank. Brains were stored at 4°C until they were sectioned and then stored in sucrose- and ethylene glycol containing cryoprotectant and kept at −20°C.
Corpus callosum area determination
The size of the CC (approximate adult-equivalent Bregma range 0.74-0.2 mm) was determined by tracing the area of interest in at least 3 adjacent coronal sections of slide-mounted brain using Neurolucida (MBF, Williston, Vermont). Software enumerated the area of interest in square micrometers.
Myelin basic protein immunostaining and image analysis
To determine the extent of myelination, every fourth section (PND 14) or every sixth section (PND 270) was stained for myelin basic protein (MBP). Briefly, brain sections were washed of cryoprotectant and placed into primary antibody for MBP (EMD Millipore; MAB386, Burlington, MA). Tissue was then placed into biotinylated secondary antibody (BA-9401; 1:200 dilution; Vector Labs, Burlingame, California) for 1 hour and the stain was visualized using 3-3′-diaminobenzidine (DAB). Expression of MBP was analyzed using Image Pro Plus 7.0 (Media Cybernetics, Rockville, MD) using segmentation and thresholding to estimate a percentage area of the region of interest that was positive for MBP expression.
Laser Ablation Inductively Coupled Plasma Mass Spectrometry
Brain thin sections of 40 µm were prepared by cryosectioning and mounted onto a microscope slide for elemental imaging by laser ablation inductively coupled plasma mass spectrometry (LA-ICP-MS). Slides were not subbed (coated with chromium potassium sulfate to prevent metal contamination). The LA-ICP-MS system used was an ESI Lasers NWR 213 laser ablation unit coupled to an Agilent 8900 ICP-MS operated in O2 reaction mode to enhance detection of S by mass shifting to SO at 48 m/z. The laser “spot” size was 50 µm2 and the laser scan speed was 250 µm/s and the laser shot frequency was 20 Hz. The sample was analyzed in a series of line scans with a spacing of 50 µm. The ICP-MS collected data for Al, S, Ca, Cr, Mn, Fe, Ni, Cu, Zn, Se, Cd, and Pb with a total acquisition time of 0.2 seconds. The raw ICP-MS data were concatenated into an Excel sheet, and relative X and Y coordinates for each data point were added. The resulting file was then imported into SMAK elemental imaging software to generate 2D elemental images of each section. 77 The images show the relative concentration differences in each element across the thin section as no quantitative calibration was performed during this analysis.
Atomic Absorption Spectrophotometry
Atomic absorption spectrophotometry was used to measure levels of metals in olfactory bulb. After wet weights were recorded, tissue were digested with a 1:1 mixture of nitric and perchloric acids, samples brought to a constant volume, and determination of metal levels performed by graphite AAS (Hitachi 170-70, Tokyo, Japan). For each analysis, controls (blanks) and standards were carried out in 0.7% nitric acid and calibration curves determined at the beginning and end of a sample run.
Serum Oxidized Glutathione
Blood was collected during sacrifice into prechilled tubes and spun at 1500g for 20 minutes and serum was removed. Serum oxidized glutathione (GSH) was measured using a commercially available colorimetric assay kit (Arbor Assays, Ann Arbor, Michigan) according to the manufacturer’s instructions. Quality controls fell within appropriate ranges with intra-assay sample variability below 15% variance.
Immunohistochemistry
Caspase-3
Briefly, brain sections incubated in primary antibody solution (1:2000; 9662; Cell Signaling Technology, Danvers, Massachusetts) at 4°C for 24 hours. Sections were then washed and incubated with biotinylated secondary antibody (1:200, anti-rabbit, BA1000; Vector Labs) for 1 hour; the stain was visualized using DAB and quantified using ImageJ with established thresholding methods. 78
Silver staining
The method was carried out on brain sections using the FD NeuroSilver Kit II (FD NeuroTechnologies, Inc, Columbia, Maryland), according to the manufacturer’s instructions.
Statistical Analyses
Statistical analysis was carried out using JMP11 (Cary, North Carolina) with initial between-group analyses of variance that included all experimental factors (sex and treatment) in full factorial designs. Main effects and interactions were then pursued in post hoc t tests as appropriate. P values ≤.05 were considered statistically significant.
Results
Concentration Effect Functions for Postnatal UFPs
Postnatal CAPs exposure concentrations across our studies to date have ranged from mass concentrations of 22 to 121 μg/m3. In order to ascertain the impact of concentration, we plotted magnitude of effect against exposure concentrations for outcomes in males that were measured at 2 or more of the exposure concentrations. The results are depicted in Figure 2. As shown in the left panel, ventriculomegaly was not observed in preliminary assessments following a 22 μg/m3 exposure, but trending increases in size of the lateral ventricle were observed at 44 μg/m3,F(1,17) = 3.98, P = .062, in males and significantly further increased at 96 μg/m3 as previously reported and shown in Figure 3. 28,31 No changes in myelination were evident at 22 or 44 μg/m3, while, as previously reported, marked reductions occurred as concentrations increased to 96 μg/m3. 28,31
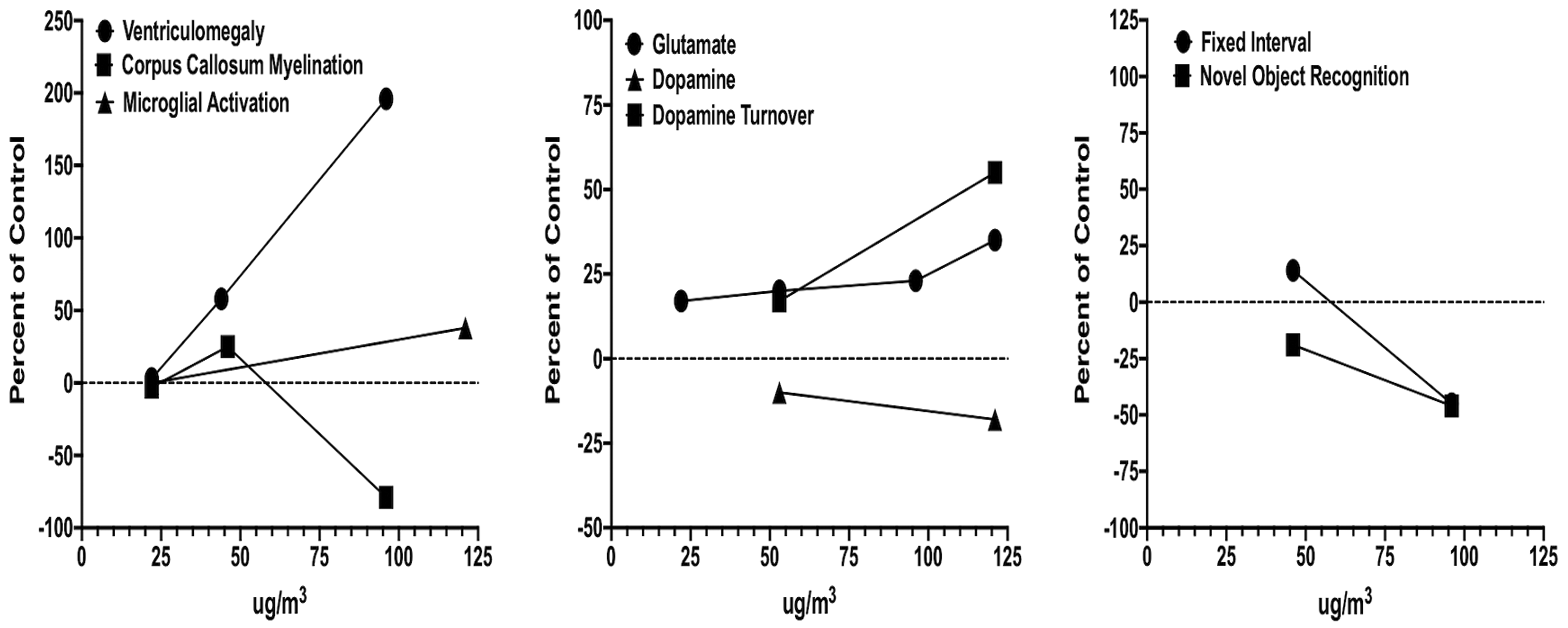
Concentration–effect functions for neuropathological (left), neurochemical (middle), and behavioral (right) consequences for concentrated ambient ultrafine particle (CAP) exposure. Data are from males only and presented as percentage of the corresponding filtered air control data and the CAP concentration (x-axis). Unpublished and previously reported data. 27 –32,82
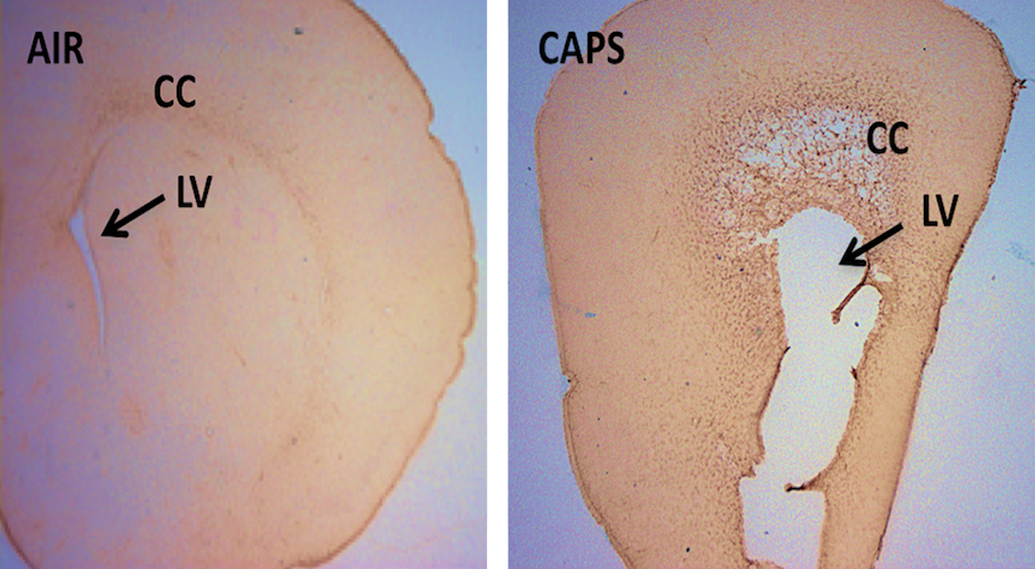
Coronal unstained brain sections from a male brain exposed to filtered air (left) or to 96 µg/m3 concentrated ambient ultrafine particles (CAPs; right) during the postnatal period representing the extreme of ventriculomegaly observed. The enlarged lateral ventricle (LV) below the corpus callosum (CC) of the CAPs-exposed brain on the right panel is indicated by an arrow. 28
The middle panel depicts alterations in dopamine and glutamate levels and function. Even at the lowest concentrations generated in our studies to date (22 μg/m3), increases in levels of glutamate have been observed, with slight increases in the magnitude of elevation at the highest concentration generated to date (121 μg/m3). 28 -30,33 Findings also suggest increases in levels of dopamine turnover in brain, again increasing with increasing CAPs exposure concentration. 29,30 With increases in dopamine turnover, levels of dopamine appear to decline with increasing CAPs exposure concentration. 29,30
The right panel depicts 2 behavioral baselines that have been measured in 2 different exposure cohorts, that is, behavior on a fixed interval (FI) schedule of food reward, as a measure of temporal learning, and novel object recognition, an index of short-term memory, as previously described. 30,17 Slight increases in rates of response on the FI schedule (delayed learning) at 44 μg/m3 were observed, while at 96 μg/m3, marked reductions in response rates (delayed learning and delayed reward) were found that persisted across sessions. Novel object recognition was already significantly impaired at 44 μg/m3, with even further impairments in performance observed at 96 μg/m3.
Assessment of Trace Elements in CAPs Exposures
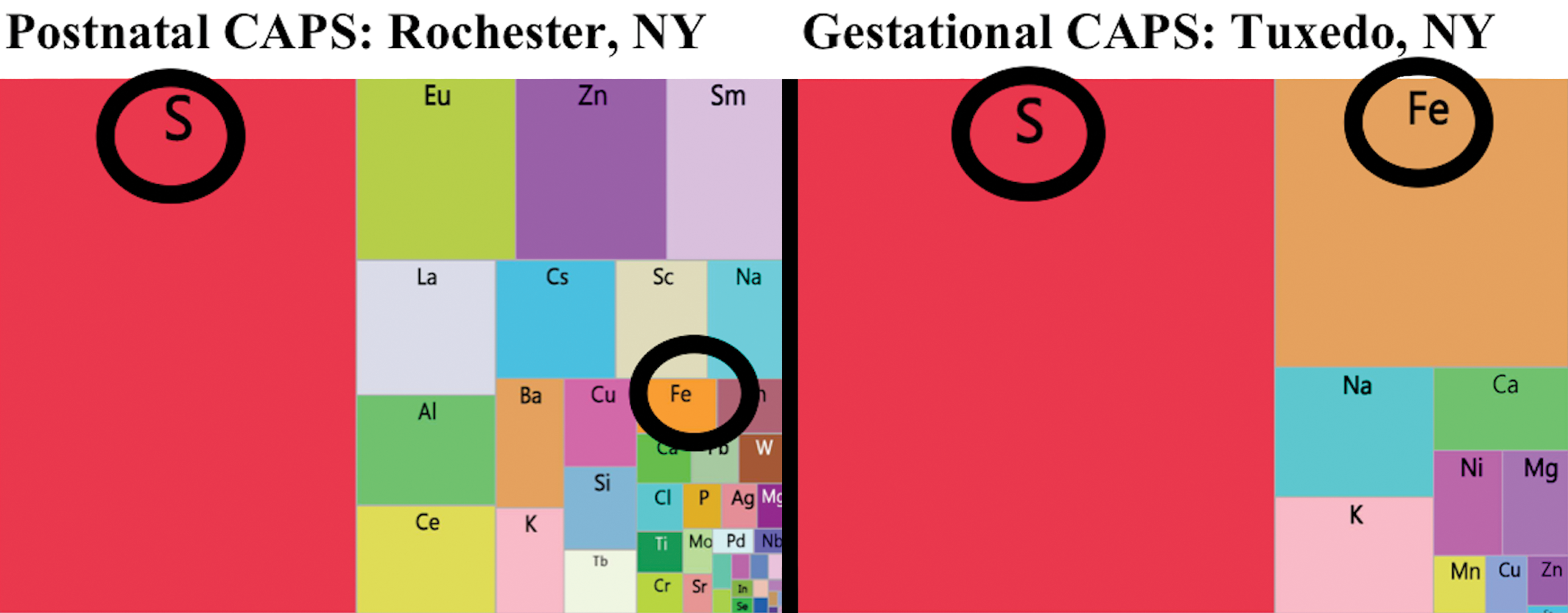
X-ray fluorescence analyses were utilized to examine the elemental exposures associated with various CAPs exposure studies. As shown in the tree maps for one such exposure from Rochester, New York and another from Tuxedo, New York in Figure 4, all such analyses revealed multiple trace elements were contaminants of CAPs, regardless of the site of the exposure. In addition, as expected, the profile of elevation of trace elements in CAPs differed somewhat by exposure site, with S being the highest in Tuxedo with a relatively high Fe levels as well; S was also highest in Rochester, but Fe was not as high as in Tuxedo.
Elemental contaminants of the AP exposures are further characterized in Figure 5 where the top row depicts fold changes in various elements relative to the filtered air chamber exposure levels for 3 different exposures. As it shows, levels of sulfur (S) showed dramatic increases in virtually all these exposures. In addition, increases in iron (Fe) levels were also consistently evident. Potassium levels likewise showed increases. Increases in numerous other elements were also observed, but fold-changes were less systematic across such exposures.
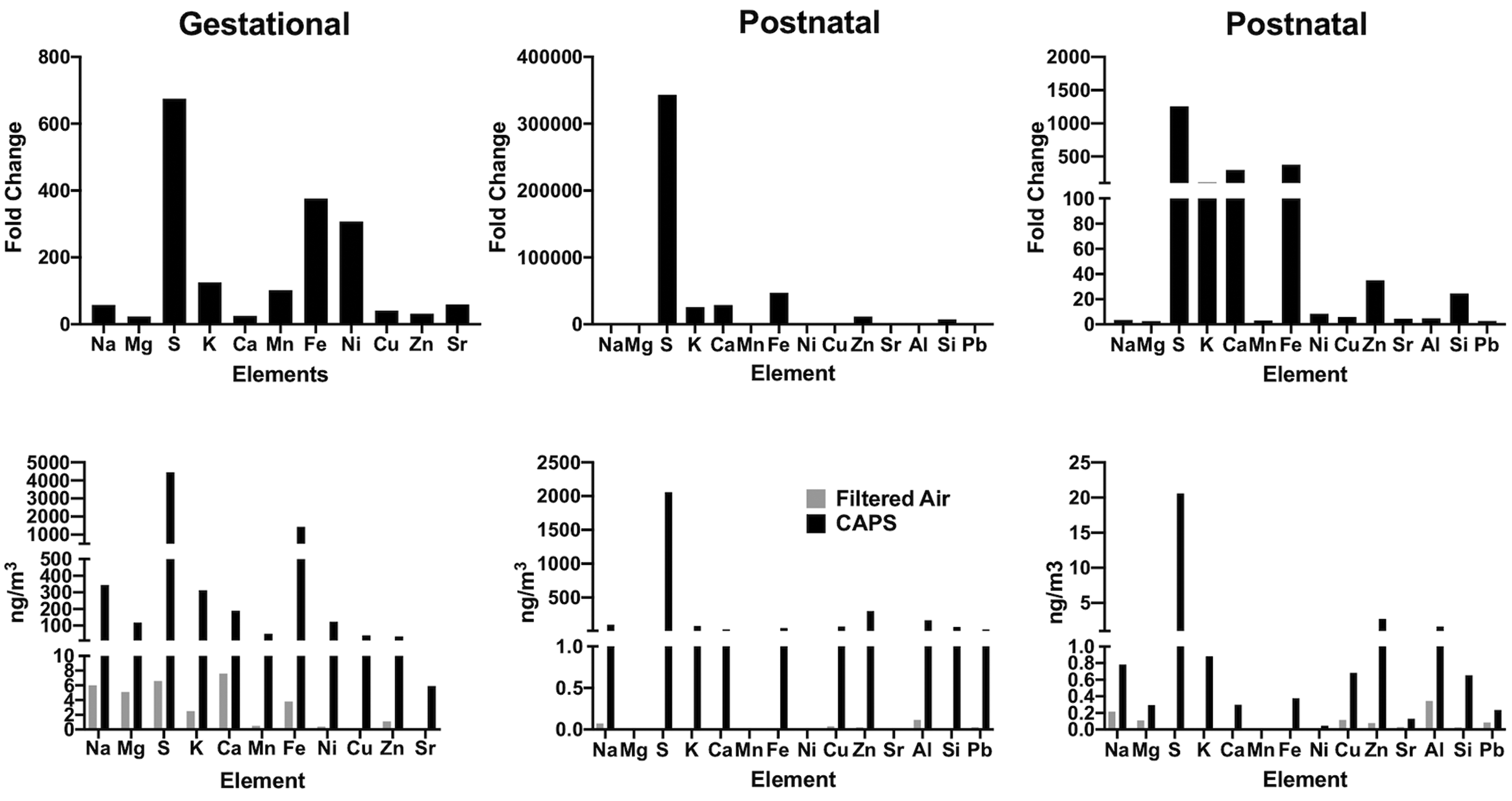
Top row shows fold changes in various elements relative to levels in filtered air control chambers for gestational (left column) and for 2 postnatal exposures (middle and right columns) following X-ray fluorescence analyses. Corresponding data in nanograms per cubic meters are depicted along the bottom row.
Total concentrations in nanograms per cubic meters are depicted in the bottom row of Figure 5 relative to filtered air controls for these same exposure cohorts. As these plots show, S levels ranged from 2000 to 4000 ng/m3 and Fe levels from approximately 500 to 1000 ng/m3. Other metals of note in these exposures included zinc (Zn: range from 2.7 to 297 ng/m3), copper (Cu: range from 0.68 to 69 ng/m3), aluminum (Al: range from 1.65 to 164 ng/m3), potassium (K: range from 0.88 to 313 ng/m3), and silica (Si: range from 0.65 to 64 ng/m3).
Assessment of Trace Elements in Brain
Subsequently, LA-ICP-MS was carried out on 40-µm sagittal sections after postnatal CAPs exposure from a randomly chosen filtered air control male and a CAPs-exposed male brain. Figure 6 contrasts the levels of Fe and S in these brains at PND 14, that is, 24 hours after completion of exposures. As it shows, postnatal CAPs markedly elevated the levels of both elements in brain relative to levels in filtered air control brain. These elevations, moreover, were seen diffusely across the brain rather than regionally, as would be consistent with the broad functions of Fe and S in brain. As would be suggested by the analyses of filters from exposure chambers, other metals were likewise elevated in CAPs brain post exposure (Figure 7), in particular levels of Cu and to some degree calcium (Ca). Notably, increases in Al were also seen that were somewhat diffuse across regions but showed a selectively high accumulation in hippocampus. In contrast, levels of 2 essential metals in brain appeared to be of a lower concentration in brain, specifically manganese (Mn) and Zn. Collectively, these findings suggest brain metal dyshomeostasis.
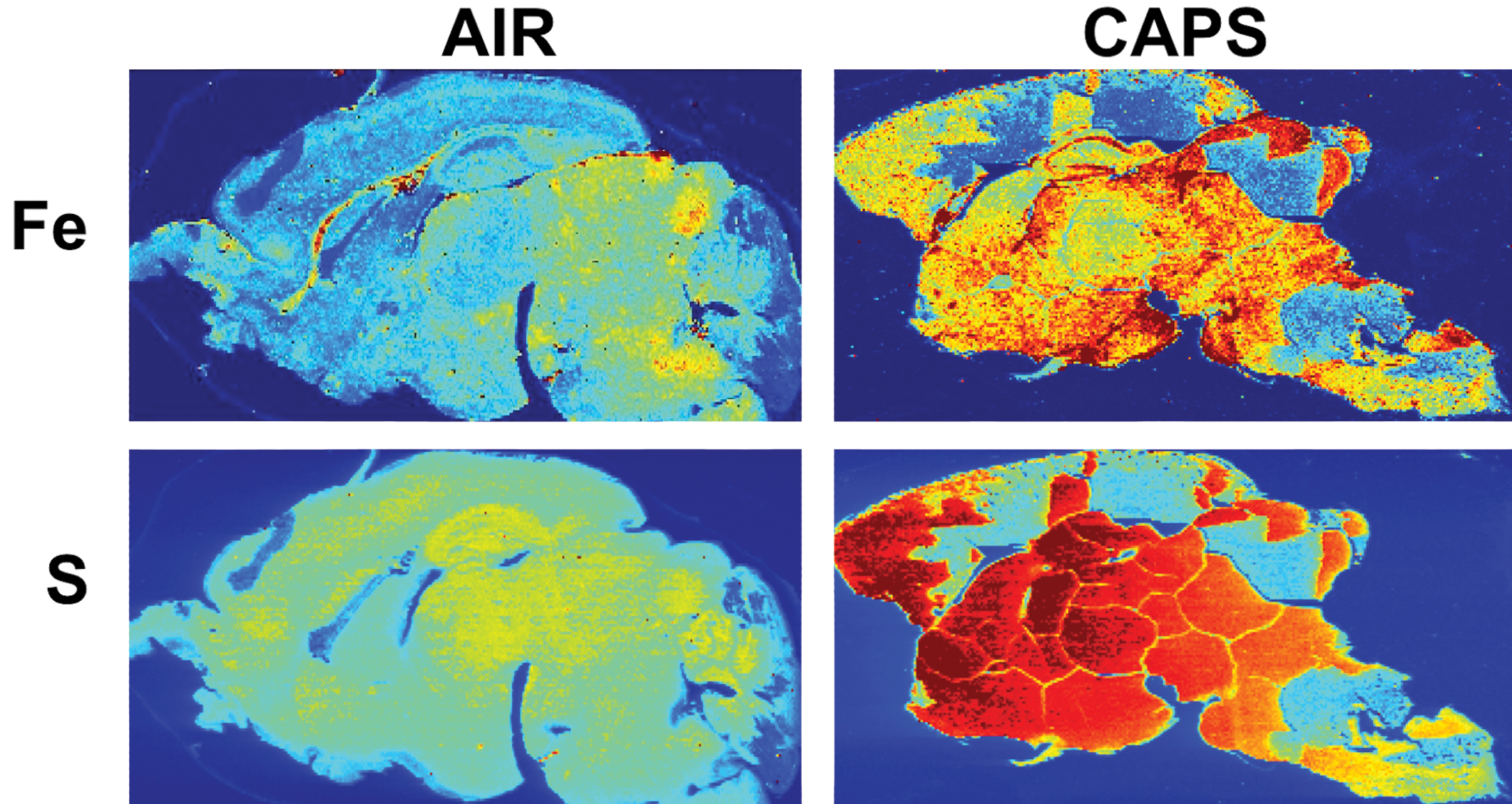
Brain iron (Fe) and sulfur (S) levels are increased in postnatal concentrated ambient ultrafine particles-exposed males after analysis by inductively coupled plasma mass spectrometry in one randomly chosen filtered air control male and 1 male exposed to concentrated ambient ultrafine particles. These images show a relatively diffuse pattern of increases across brain regions. Brains from females not yet examined. Exposure concentrations averaged 45 µg/m3.
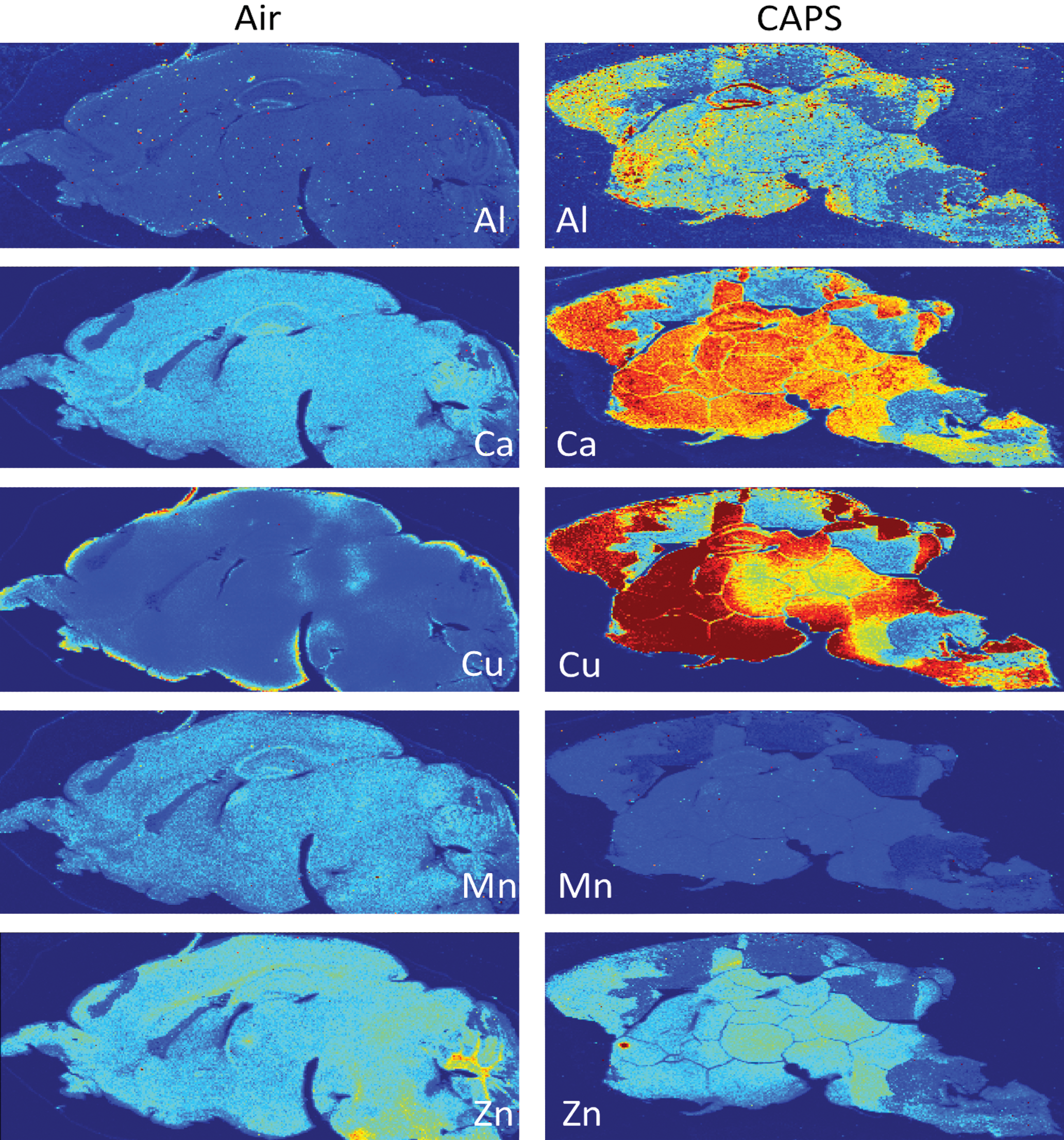
Brain levels of aluminum (Al), calcium (Ca), copper (Cu), manganese (Mn), and zinc (Zn) from a randomly chosen male (air) and concentrated ambient ultrafine particles–exposed male following postnatal exposures. Exposure concentrations averaged 45 µg/m3.
Given the demonstrated olfactory and trigeminal nerve uptake of elemental contaminants of UFPs, and since the LA-ICP-MS analysis is not quantitative, olfactory bulb tissue from PND 14 CAPs exposed and filtered air-exposed brains of males and females were analyzed by atomic absorption spectrophotometry for levels of Al, Fe, and Zn. As can be seen from Figure 8, in support of the ICP-MS images, significantly increased levels of both Fe, 65%, F(1,11) = 47.58, P < .0001, and Zn, 43%, F(1,11) = 20.09, P = .0008, were found in males, with a similar trend for Al increases, F(1,11) = 3.495, P = .086, relative to filtered air controls after exposures to 44 µg/m3. Females showed a significant increase only in levels of Al by 120%, F(1,11) = 6.56, P = .0265, but not in Fe or Zn.
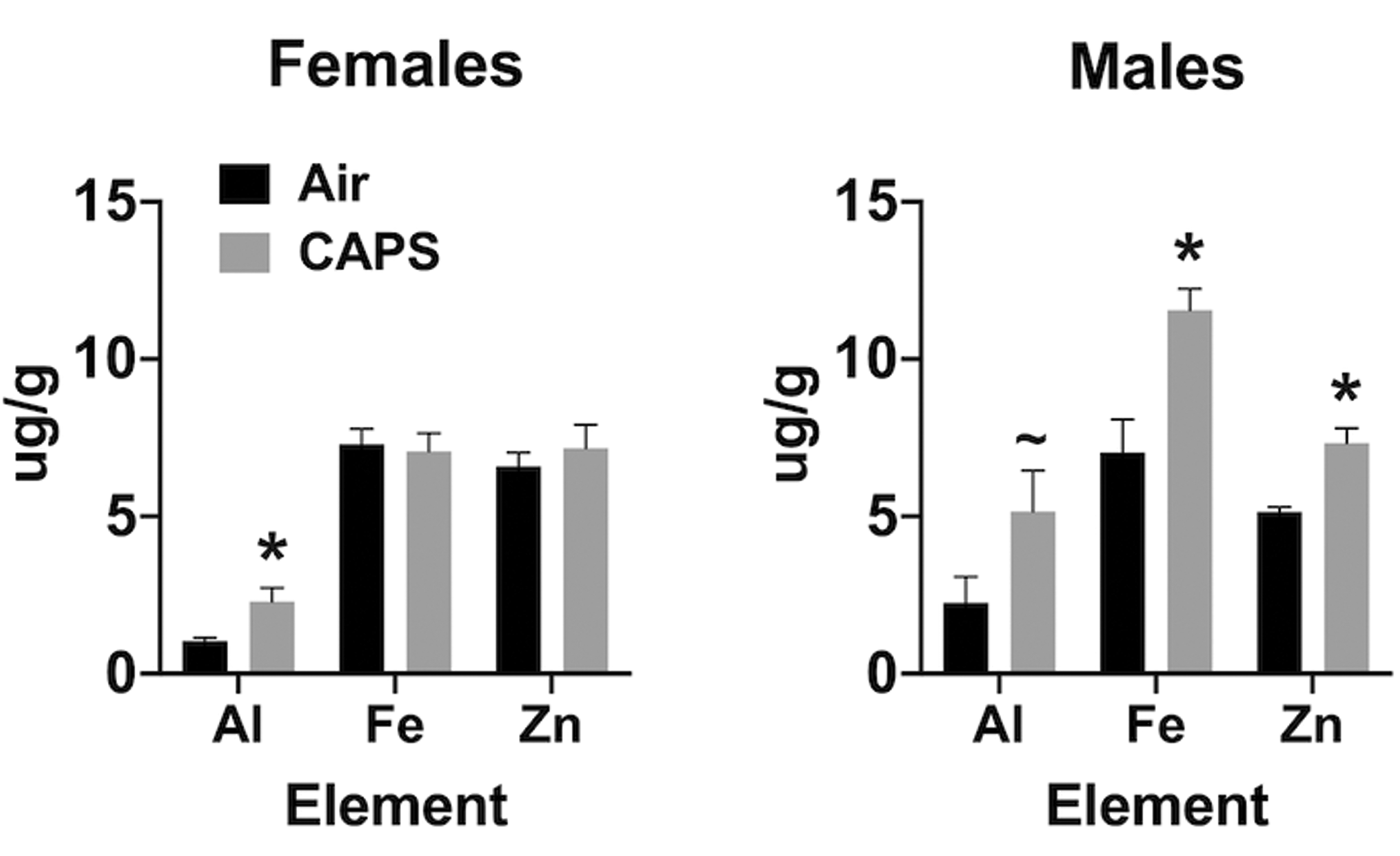
Mean ± standard error levels (µg/g) of aluminum (Al), iron (Fe), and zinc (Zn) in olfactory bulb following postnatal concentrated ambient ultrafine particles exposure concentrations of 45 µg/m3 in females (left) and males (right). * indicates statistical significance; ∼ indicates marginal significance.
Potential Ferroptotic Mechanisms
Increases in Fe and S in brain suggested the potential for ferroptotic mechanisms could underlie the neuropathological changes observed. 79 -82 Ferroptosis is a nonapoptotic fatal combination of Fe toxicity, antioxidant depletion due to disruption of glutathione peroxidase-4 (GPx4), that is, trans-sulfuration, and lipid peroxidation-induced membrane damage. 83 Ferroptosis is negatively regulated by GSH, a product of the trans-sulfuration pathway, making S levels critical. An inverse relationship between Fe overload and GSH, hydrogen peroxide, and reactive oxygen species (ROS) are well-documented. 84 -86 Thus, high brain Fe and low GSH or GPx4 would facilitate ferroptosis.
Oxidized GSH
To determine the potential involvement of ferroptotic mechanisms in these effects, levels of serum-oxidized GSH were measured and revealed male-specific increases as shown in Figure 9, F(1,8) = 10.9, P = .0108. These effects were seen following postnatal CAPs exposures where the exposure concentrations averaged only 22 µg/m3.
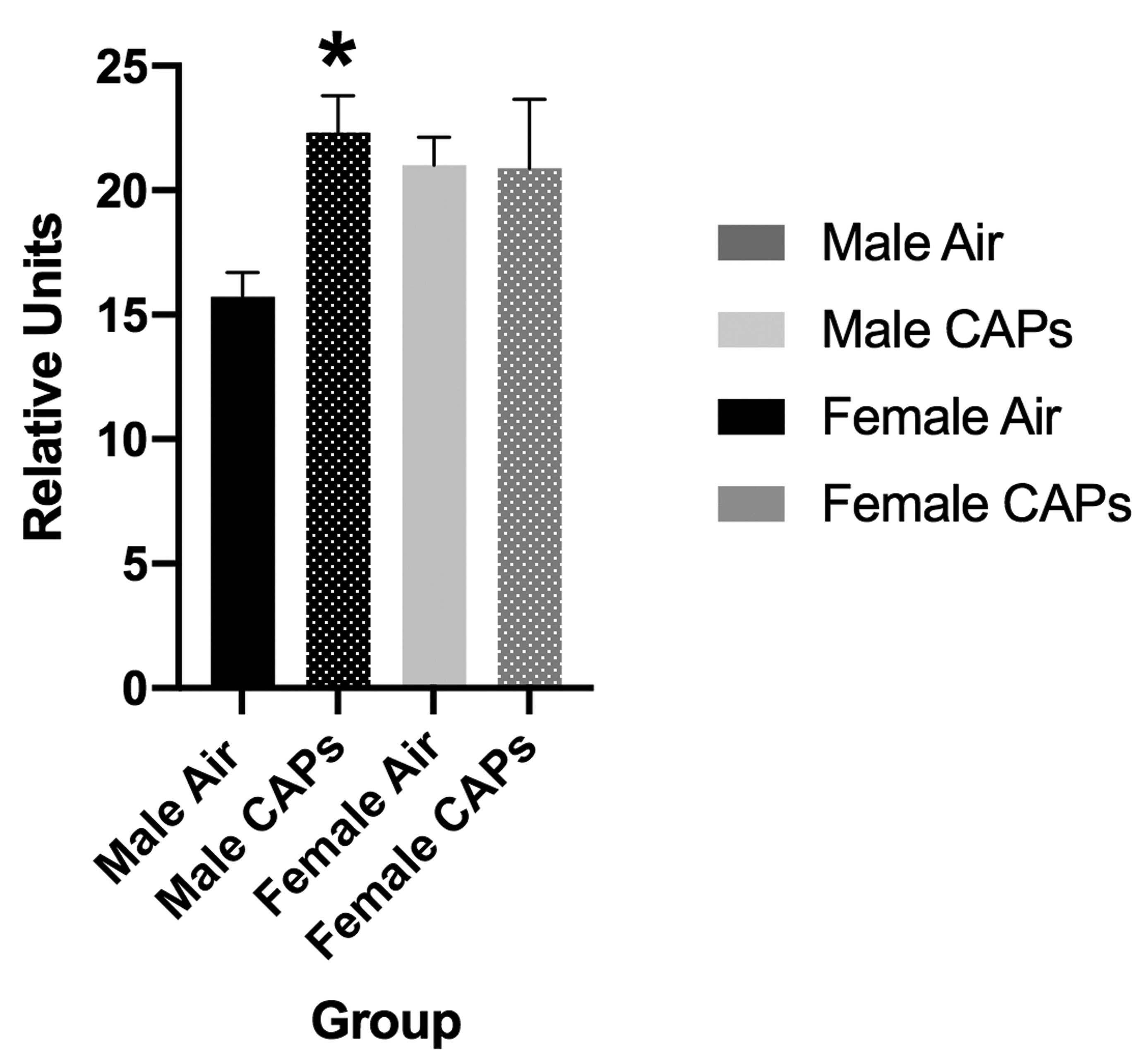
Mean ± standard error levels of oxidized glutathione in serum from males (black) and females (gray) exposed postnatally to filtered air or to 45 µg/m3 concentrated ambient ultrafine particles (CAPs). Statistically significant increases in serum oxidized glutathione were seen in males after CAPs, but not in females. * indicates statistical significance; ∼ indicates marginal significance.
Nucleus Accumbens Neuronal Cell Death
In addition, measures of neuronal death were examined at PND 14 in nucleus accumbens (NAc), a key region of the mesocorticolimbic circuit of brain critical to executive functions, after exposure concentrations averaging 44 µg/m3. For this purpose, tissue was silver stained as well as examined by caspase immunohistochemistry. Males exhibited significant increases in numbers of dead cells and reductions in numbers of live cells and total numbers of cells (Figure 10), males: dead cells, F(1,8) = 8.92, P = .0174; live cells, F(1,8) = 7.46, P = .0258; total cells, F(1,8) = 6.97, P = .0297, as measured via silver staining. While a similar trend toward an increase in dead cells was seen in females at PND 14, effects were not statistically significant. Figure 11 shows a photomicrograph depicting the increased silver staining in CAPs-exposed male NAc; importantly, these increases were not seen with caspase staining. When measured at PND 25 (Figure 12), effects were no longer evident in males, whereas significant increases in live cells (F(1,6) = 8.99, P = .024) and total cells (F(1,6) = 8.95, P = .043) were seen in females at this time point.
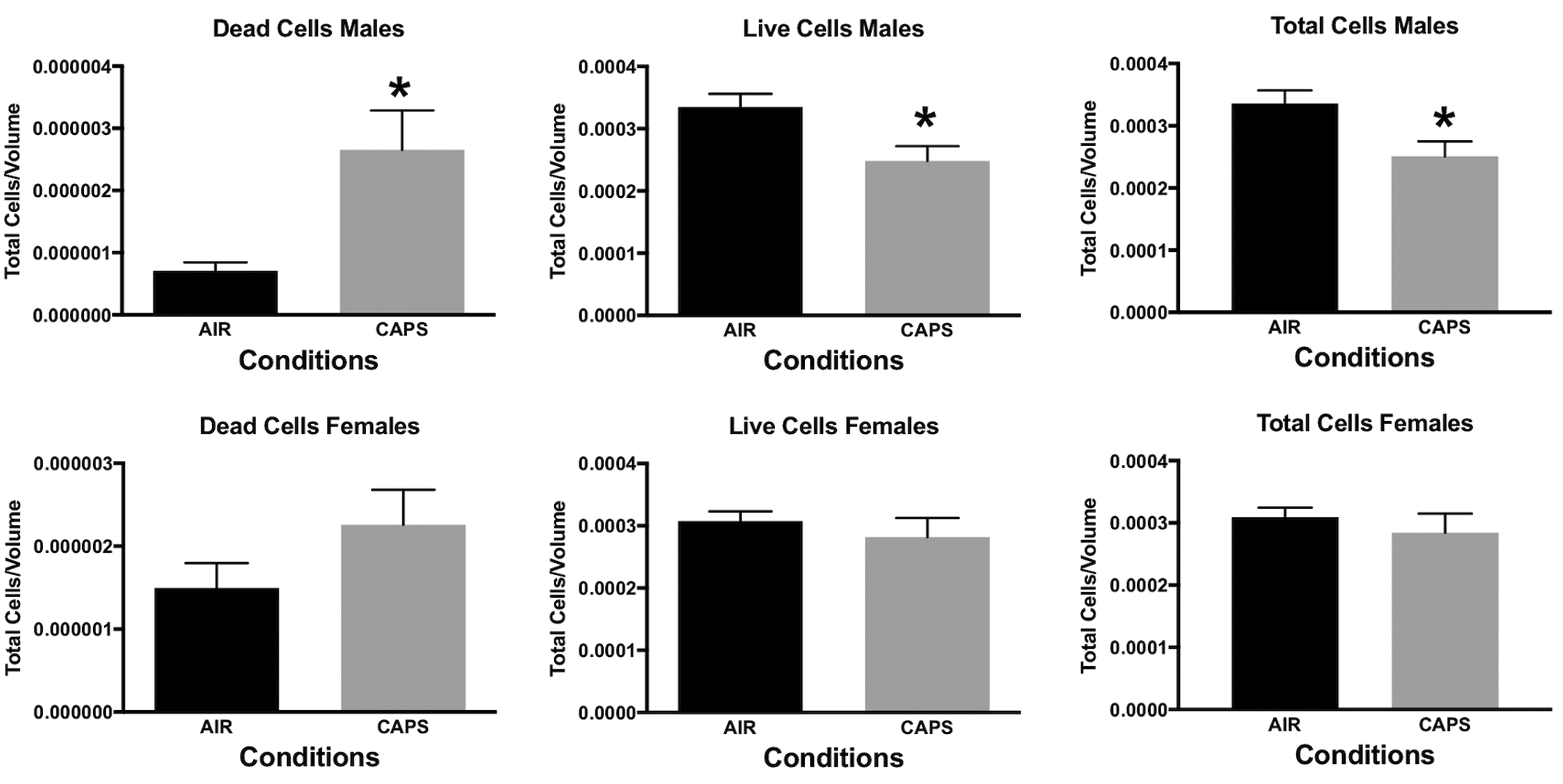
Mean ± standard error levels of dead cells (left column), live cells (middle column), and total cells (right column) in nucleus accumbens at PND 14 as evaluated by silver staining of males (top row) and females (bottom row) exposed postnatally to filtered air (air) or to 45 µg/m3 concentrated ambient ultrafine particles. Males but not females exhibited increases in dead cells and reductions in both live cells and total cells.
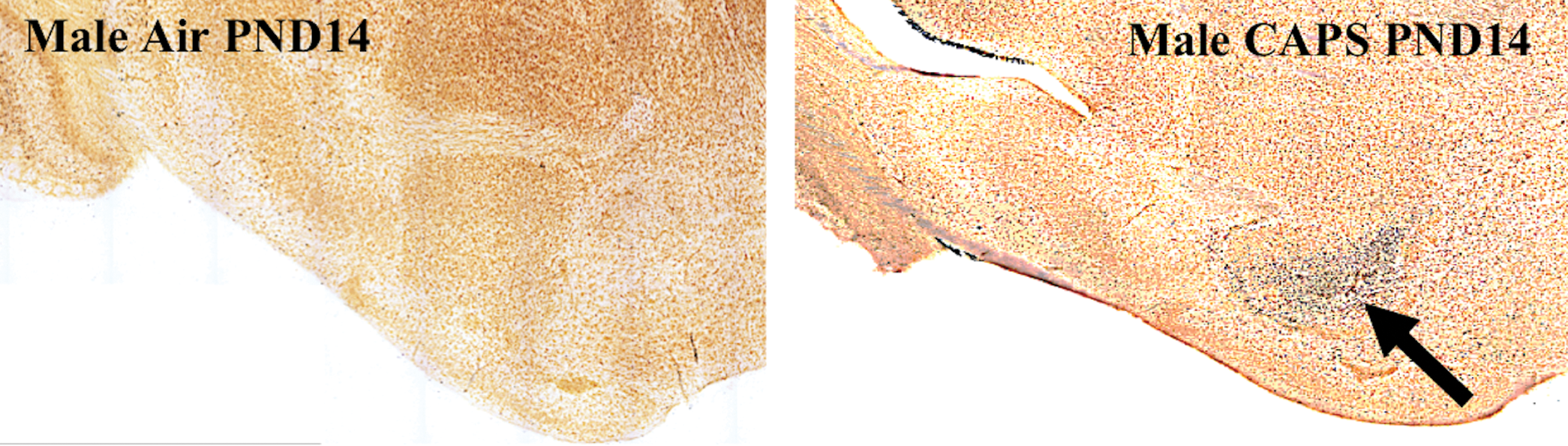
Male-specific degeneration of neurons in nucleus accumbens (arrow) as measured by silver staining as described in the Methods at postnatal day 14 following postnatal exposure to 45 µg/m3 concentrated ambient ultrafine particles. No comparable effects were seen in males exposed to filtered air, nor did caspase staining show increases.
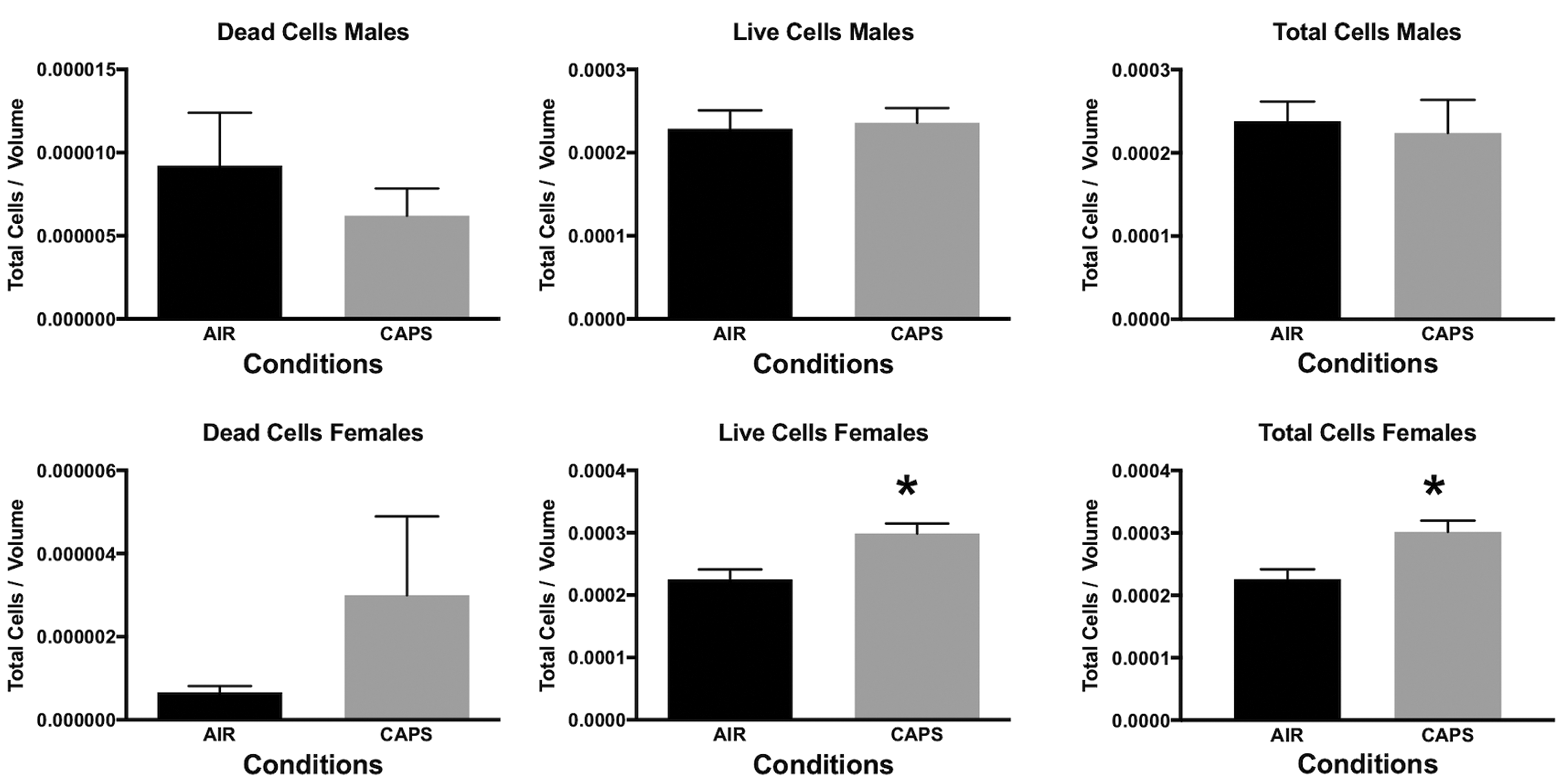
Mean ± standard error levels of dead cells (left column), live cells (middle column), and total cells (right column) in nucleus accumbens at postnatal day 25 as evaluated by silver staining of males (top row) and females (bottom row) exposed postnatally to filtered air (air) or to 45 µg/m3 concentrated ambient ultrafine particles. At this time point, changes were no longer evident in males, whereas females showed increases in both live cells and total cells.
Discussion
Collectively, our studies of postnatal and GE CAPs exposures provide biological plausibility supporting an impact of AP on brain development and behavior. 17,27 -31,59,61,60 Importantly, these studies demonstrate that the impacts of UFP matter exposures are sex dependent and differ by the specific developmental window of time in which it occurs. Further, they suggest that the adverse impacts are not geographically unique to Rochester and that similar components of AP could be responsible for the observed neuropathological consequences.
Similarities of Effects of Postnatal CAPs Exposures in Mice to Features and Hypothesized Mechanisms of Autism Spectrum Disorder and Other Neurodevelopmental Disorders in Humans
The similarity of the effects of CAPs exposures to characteristics and hypothesized mechanisms of ASD is striking as depicted for postnatal CAPs exposures in Table 1. Autism spectrum disorder is a male-biased disorder diagnosed on the basis of impaired social behaviors and restricted/repetitive behaviors. 48,87,88 Neuropathology of ASD in humans is dynamic across time and can include persistent ventriculomegaly, 64,89 -91 initial increases followed by sustained loss of white matter 63,64 and gray matter, 92 -96 loss of myelin connectivity across the hemispheres 97 -99 particularly in CC, 62 persistent brain inflammation, 100,101 and excitatory (glutamate) inhibitory imbalance 102 that correlates with IQ reductions and social and cognitive deficits. Notably, in mice, increases in white matter were seen following GE CAPs exposures, whereas postnatal exposures markedly reduced white matter. 63,64 Nucleus accumbens dysfunction in ASD has been related to social and repetitive behavior deficits and response inhibition 95,96,103,104,96 and to altered self-control in ADHD. 105,106 While ASD etiology remains unknown, inflammation is considered key, and biomarkers include altered trans-sulfuration pathways and increased oxidative stress. 69,107 -112
Comparison of ASD and PN CAPs characteristics.

Abbreviations: ASD, autism spectrum disorder; CAPs, concentrated ambient ultrafine particles; PN, postnatal.
It is important to recognize, however, that many of the phenotypic features and hypothesized mechanisms of ASD are also seen in other neurodevelopmental disorders. 35 -40 Autism spectrum disorder and early childhood-onset schizophrenia 41 -44 share abnormalities of glutamatergic systems, 45 -47 inflammatory mechanisms, 48 and neuropathological features, including ventriculomegaly and disconnectivity. 49 -51 Attention-deficit/hyperactivity disorder occurs at higher rates in children with ASD. 52 Both involve increases in impulsive behavior and are considered disorders of hemispheric disconnectivity. 36 Like ASD, ADHD is male biased and its rates have increased significantly from 7.8% in 2003 to 9.5% in 2007 and 11.0% in 2011 to 2012 according to CDC. Attention-deficit/hyperactivity disorder can also include ventriculomegaly 53 and alterations in glutamatergic function, 54 brain white matter, 55 brain connectivity, 56 and inflammation. 57 Thus, the neurotoxic consequences of AP seen in our experimental models may contribute risk not only for ASD but for other neurodevelopmental disorders as well.
Olfactory Uptake of Elemental Contaminants of AP
Inductively coupled plasma mass spectrometry analyses of brains from mice after CAP exposures suggest that AP exposures could produce brain metal dyshomeostasis, with increases in several brain metals, for example, Fe and S, and suggestive depletion of others. The observed increases in olfactory bulb Fe levels in males are consistent with the assertion that some component of this uptake occurred via nasal olfactory uptake after postnatal CAPs exposures. 113,114 Similarly, increases in brain Fe were seen in cerebellum of both sexes following GE CAPs exposures. 115 Nasal olfactory uptake of elemental AP particles has been demonstrated in both rats and concluded from studies in humans, 116 including Fe, but also Mn, Cd, Ni, Hg, Al, Co, Zn, and Cu, 113,116 -122 across olfactory epithelial barriers in the nasal passages followed by transport to olfactory bulb and movement to other brain regions. 123 In one study, intranasal instillation of either ferric or ferrous iron subsequently increased blood Fe levels in rats, with uptake relying on divalent metal transporter 1. 113 Accumulation of ferric iron in olfactory bulb was found following inhalation exposures to iron soot (40 µg/m3 Fe oxide nanoparticles) in adult female mice. 124
Inhaled exposures to SO 2 show that virtually all of it is removed from air by the nose. 125 Early studies indicate that subsequently, its derivatives enter the blood and dissociate to form the derivatives bisulfite and sulfite, 126 both of which have been found in hippocampal slices from rat brain. 127
With olfactory nerve uptake, elements bypass the blood–brain barrier such that increased levels of trace elements in brain may not be reflected in peripheral markers. In addition, sensory nerves in the upper and lower respiratory tract can translocate particles 128 that reach for example, the trigeminal ganglion or the vagal nerve. 66,68,129,130 As Fe regulatory proteins 1 and 2, and Fe transporters such as the transferrin receptor and divalent metal transporter-1, critical to tight brain regulation of Fe levels, are not fully expressed until PNDs 15 to 20 in rodents, 131,132 an extended window is open during which excess Fe or S can reach and directly influence brain development. Similarly, homeostatic regulation of Fe absorption appears in human infants at about 6 to 9 months of age. 133
Plausibility of Elemental Contaminants of AP for the Postnatal CAPs-Induced Phenotype
Notably, reported consequences of excess brain Fe or SO2 are consistent with ASD and other neurodevelopmental disorders. Normally, Fe2+ oxidation to Fe3+ releases a free radical quenching electron, with nanoparticulate Fe3+ existing in a biomineralized solid or particle. However, excess Fe2+, that is, the labile pool (like other ionic species, eg, Ce3+), has greater bioactivity than Fe3+ and produces oxidative stress and inflammation, reductions in antioxidant capacity, increased cell death markers, and altered prefrontal cortex and hippocampal glutamate function. 134 Fe(III)Cl injections into rodent brain produce ventriculomegaly. 135,136 Developing brain is particularly susceptible to excess ionic Fe2+ given its immature antioxidant defense systems, low total Fe binding capacity, and low concentrations of Fe2+ binding proteins. 137 Indeed, increased non-protein-bound Fe in umbilical cord blood is an early predictive marker of neonatal brain damage. 138 The uptake, transport, and sequestration of extrinsic Fe x O y nanoparticles may be accompanied by breakdown and in vivo processing, making it ultimately critical to distinguish extrinsic Fe x O y from intrinsic ferritin nanoparticle formation.
In what might seem contradictory to our findings, some studies have reported reduced, rather than increased, serum Fe levels in ASD, 139 -141 although this was not confirmed by a recent meta-analysis. 142 Even if such a finding was confirmed, it does not preclude a concurrent elevation of brain Fe. For example, neuroferritinopathy resulting from a mutation in the ferritin light chain gene results in elevated brain Fe and ferritin levels but reduced serum ferritin. 143 Additionally, intranasal instillation of Fe2O3 nanoparticles to rats over 7 days increased Fe brain levels, while significantly reducing serum Fe levels. 144 Such discrepancies are also seen in neurodegenerative diseases that include elevated brain Fe, for example, Parkinson disease, 145 -147 suggesting a broader metal dyshomeostasis under such conditions.
Further exacerbating the potential toxicity of elevated brain Fe, studies have demonstrated its extremely slow turnover in rodent brain (half-life of ∼9 months), with estimates on the order of decades when extrapolated to humans 148 -150 ; such slow turnover may relate to the potential role of Fe in multiple neurodegenerative diseases, including Alzheimer and Parkinson disease, 83,151 given that such exposures are cumulative across the life span.
Sulfur is critical for Fe-S cluster proteins, amino acid metabolism, and brain trans-sulfuration pathways critical to GSH production necessary to maintain cellular redox states and scavenge free radicals. Glutathione activity is very low in fetal brain, reaching adult levels only at PND 14 in rat, indicating incomplete regulation of this pathway during brain development. 137,152 Alterations in the trans-sulfuration pathway adversely influence brain and behavior. 153,154 Sulfite, taken up during SO2 inhalation, 155 can auto-oxidize, as mediated by O2 or Fe3+ (the most stable such complex), to produce sulfite radicals and ROS. 156 -158 Inhaled SO2 increases levels of brain sulfite, 159 hippocampal neuronal death, protein oxidation, lipid peroxidation, 160 brain inflammation, and oxidative damage in brain; it also alters glutamate function and reduces GSH and GPx4 levels. 161 These features correspond to the key role that chronic neuroinflammation is considered to play in ASD. 162 Of direct relevance to our findings, recent studies report that 1 ppb increases in SO2 in AP increase the odds ratios for ASD by 17%. 163
Elevated Brain Fe and S Suggest Potential Ferroptotic and Oxidative Stress-Based Mechanisms for Postnatal CAPs Effects
Elevated brain Fe and S also suggest ferroptosis and oxidative stress mechanisms of postnatal CAPs effects. Ferroptosis is a nonapoptotic fatal combination of Fe toxicity, antioxidant depletion due to disruption of GPx4, that is, trans-sulfuration, and lipid peroxidation-induced membrane damage. 83 It is negatively regulated by GSH, a product of the trans-sulfuration pathway, making S levels critical. An inverse relationship between Fe overload and GSH, hydrogen peroxide, and ROS are well-documented. 84 -86 Thus, high brain Fe, and low GSH or GPx4 would facilitate ferroptosis, as proposed in Figure 1. These mechanisms would be consistent with our observations of postnatal CAPs-induced increases in NAc neuronal cell death that were not revealed by caspase staining (Figure11), as well as the increases in levels of serum oxidized GSH (Figure 9). While ferroptosis is seen in multiple neurodegenerative diseases, 81 its role in neurodevelopmental disorders has not been investigated, although it is seen in periventricular leukomalacia of prematurity, that is, brain white matter injury arising from free radical damage to oligodendrocytes. 82,164
Male Vulnerability to Postnatal CAPs Exposures and Heterogeneity of Neurodevelopmental Disorders
Although numerous different and perhaps even multiple mechanisms could account for the enhanced vulnerability of the male brain to postnatal CAPs exposures, one attractive hypothesis relates to differences in the trajectory of colonizing activated brain microglia. Specifically, males have both a greater number of, as well as more activated (amoeboid), microglia than females in early postnatal development, 165 which could impact early neurogenesis and oligodendrogenesis and late cell toxicity. Additionally, it is interesting to consider that differences not only in the timing but also in the components of AP exposure during development could contribute to the heterogeneity and subgroups of ASD 58 or other neurodevelopmental disorders.
But Our Air Is Getting Cleaner?
One seeming inconsistency relates to the fact that US air quality has improved, whereas rates of ASD have continued to increase. Air pollution health effects are related to particulate matter (PM) size, designated as PM10 (coarse, <10 μm), PM2.5 (fine, <2.5 μm), or PM0.1 (ultrafine:UFPs; <100 nm). Ultrafine particulates, being the subject of our studies, and the same size as engineered nanoparticles, are considered the most reactive component of AP, 20 with greater surface area/mass for adsorption. Levels of PM10 and PM2.5,but not UFPs, are regulated by the U.S. Environmental Protection Agency (EPA). While it might be anticipated that UFP levels would decline with PM2.5 regulation, this has not necessarily been the case, with several studies reporting no correlations between levels of PM2.5 and UFPs. 166 -170 Ultrafine particulates are part of PM2.5 and PM10, due to the fact that UFPs agglomerate rapidly into the PM2.5 mode 171 and PM10 mode: as larger agglomerated particles, they have a high deposition efficiency on nasal mucosa, whereas de-agglomeration occurs on epithelial lining fluid so that neuronal uptake and CNS translocation takes place (olfactory and trigeminal pathways). Further understanding of how UFP and its most toxic components, alter sex-differentiated brain development in children, and alter risk for neurobehavioral disorders, is critical for public health protection.
Limitations of Animal Models for Human Risk and Research Needs
Of course rodent models have limitations in regard to characterizing human risk. Among these is the fact that rodents are altricial and thus the third trimester human equivalent of brain development occurs postnatally. Therefore, rodents are nose-breathing rather than being exposed in utero during this period. There are also differences in the anatomy of the nasal cavity between rodents and humans that can influence extent of deposition and thus transfer of particulates to brain via olfactory routes. 172 -174
To date, our studies have only begun to examine the neurotoxicity of particulate AP on brain development, but they have raised innumerable research questions that will be critical to ultimately understanding the impacts and consequences of AP. Our studies have focused primarily on white matter to date, leaving many open questions about developmental AP exposures and changes in gray matter as well as susceptibility of various brain regions. In addition, more complete characterization of effects during different windows of development will be required and would be assisted by a more complete assessment of the trajectory of effects across time; such information will also be critical to the ultimate understanding of the mechanisms by which neuropathological consequences occur. For example, with GE CAPs, we see a correlation between increased brain Fe and hypermyelination in females and a premature maturationals shift in oligodendrocytes and the myelinating cell of the CNS and suggest an impact on oligodendrocyte precursor cells. 60
Certainly, interventions would be desirable. In that regard, the elevation of brain metals always raises the suggestion by some to employ chelating agents that would bind metals and enhance their excretion. As noted by others, 175 such approaches are problematic for multiple reasons, including the complexity of brain metal homeostaisis and its differences within different cellular components and the lack of specificity of most chelating agents. Other approaches may be devised following a more complete understanding of the dynamics of particle chemistry and tissue interactions in vivo, that is, their in vivo bioprocessing. Aside from primary prevention, an understanding of the specific components of AP that contribute to risk could be used to alter regulatory policies to minimize exposures. For example, while levels of Pb in air are currently regulated by EPA, should this be extended to other metals?
Footnotes
Authors’ Note
Data will be made available to any qualified individuals within the scientific community to the extent that resources are possible to accommodate the nature of the request
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported in part by NIH Grants P30 ES001247 and R01 ES025541.
