Abstract
As a eusocial species, Apis mellifera, the European honey bee, is effectively a superorganism—a group of genetically related individuals functioning as a collective unit. Because the unit of selection is the colony and not the individual, standard methods for assessing toxicologic pathology can miss colony-level responses to stress. For over a decade, US populations of honeybees have experienced severe annual losses attributed to a variety of environmental stressors varying temporally and geographically; differentiating among those stressors is accordingly a high priority. Social interactions among individuals in this social species, however, mean that the “footprint” of stressors such as pesticides, phytochemicals, pathogens, and parasites may be most discernible in individuals that did not themselves directly encounter the stressor. For example, neurotoxic effects of pesticides on nurse bees may impair their behavioral responses to queen-destined larvae, which may then emerge as adults with altered anatomy or physiology. Similarly, pesticide-induced size alterations in nurse hypopharyngeal glands, which produce royal jelly, the exclusive food of larval and adult queens, may disproportionately affect the queen’s (and thus colony) health. Thus, evaluating toxicologic pathology in the honeybee requires a new perspective and development of assays that preserve the social context that ultimately determines colony health.
Introduction
Model species are important research subjects across many scientific disciplines, including toxicologic pathology. Drosophila melanogaster has been the premier model organism for genetic studies for more than a century and, in 2000, its genome was the first insect genome to be sequenced. While other insect genomes were sequenced due to their medical or economic importance, by 2006, the genome was sequenced of an insect that is both economically important and an important model for research—the European honey bee Apis mellifera. Its genome was the first to be sequenced from a nonhuman eusocial species. 1 Its economic importance derives primarily from the status of the honeybee as the world’s premier managed pollinators; its contributions to US agriculture through pollination have been estimated to exceed 18 billion dollars annually. 2 The utility of the honeybee as a managed pollinator for crop pollination, however, reflects in large part (as do so many of its other distinctive features) its biology as a eusocial species.
Although advanced eusociality has evolved independently across all animals only about 18 times, collectively these groups include some of the planet’s most successful species. Three attributes define eusociality 3 : cooperative brood care, including defense of the nest; reproductive division of labor, with nonreproductive individuals working on behalf of fertile individuals; and overlap of generations such that offspring assist their parents during some part of their lives. Thus, social insects and other eusocial animals are effectively superorganisms—a group of genetically related individuals functioning as a collective unit. In such groups, the unit of selection is the colony or hive, and no individual can survive for any length of time independently. A honeybee colony, for example, comprises a single reproductive female (the queen), several hundred males (drones) whose sole function is to fertilize virgin queens, and, depending on conditions, from 20 000 to 50 000 sterile females (workers), all of who are sisters (ie, daughters of the same queen) who forego reproduction to take on all remaining tasks necessary for maintenance of the colony. Social species present a unique challenge for toxicologic pathology studies in that conventional approaches are designed to detect direct impacts on individuals exclusively and not indirect effects on the colony due to disruption of social interactions.
Beyond being uniquely challenging, designing conceptual approaches and developing suitable tools specifically for toxicologic pathology of honeybees is uniquely important. Insect pathology differs qualitatively from other subdisciplines of pathology in that in most cases the insect pathologist is working toward the goal of identifying disease-causing organisms for the express purpose of controlling noxious or harmful insects. The goals of insect pathologists converge with their colleagues in allied pathology subdisciplines when the subjects of study are beneficial insects—predators or parasites used for biological control, species reared in laboratories for research, insects that are raised for commercial products (including lac and silk), and pollinators. The honeybee is the rare species that is beneficial as both a pollinator and a source of useful products (including but not limited to honey and beeswax). Thus, recognizing signs of toxin or pathogen exposure and determining mechanisms by which these stress agents cause mortality are of significant economic and academic importance.
Implications of Honeybee Eusociality on Toxicologic Pathology
The first publication on the honeybee genome provided suggestive evidence of the impact of eusociality on honeybee toxicology and pathology. 1 Among the highlighted findings was the fact that the honeybee genome contains fewer immunity-related genes and detoxification genes than do genomes of solitary insect species; these reductions are thought to be due in part to replacement of genome-encoded responses to diseases and toxins with cooperative behaviors. “Social immunity” in honeybees, for example, involves allogrooming, hygienic removal of dead or dying individuals, use of antimicrobial plant resins in propolis (the mixture of resinous plant secretions, saliva, and beeswax that makes up the “bee glue” used to seal cracks and line cells throughout the hive), and collective elevation in hive temperatures to kill microbes 4 ; the evolution of such behaviors may have reduced dependence on genes encoding immunity-related proteins or peptides. The possibility exists as well that the evolution of collective behaviors relating to food gathering and processing contributed to losses of genes encoding detoxification-related enzymes. In most organisms, detoxification involves 3 phases: phase 1—metabolic alteration of the toxin; phase 2—conjugation for increased solubility; and phase 3—transporter-mediated removal of conjugated products from the body. Relative to other genomes, the honeybee genome displays reductions in genes associated with all 3 phases of detoxification. 5 Honeybees and possibly other eusocial species may rely on an additional phase—“phase 0”—during which collective social behaviors are used to reduce exposure to toxins. In honeybees, phase 0 may include selective consumption (avoidance of toxic food) 6 as well as food preparation (eg, in-comb heating during conversion of nectar to honey and fungal fermentation during conversion of pollen into beebread). 5
Most of the reduction in phase 1 metabolism in the honeybee genome involves the cytochrome P450 monooxygenase (P450) enzymes. These multifunctional enzymes are encoded by a large gene superfamily; in insects, P450s are involved in the biosynthesis of endogenous substrates (eg, hormones and pheromones) and in detoxification of exogenous substrates (eg, xenobiotics and volatile signaling compounds). The honeybee genome has 46 P450 genes, approximately half the inventory found in D. melanogaster. Reductions are most pronounced in 2 clades: the CYP3 clade, which encodes detoxification genes, and the CYP4 clade, which encodes pheromone-synthesizing and -degrading enzymes as well as enzymes involved in synthesis of hydrocarbons and other lipids. 5 These reductions appear to leave honeybees particularly vulnerable to synergistic interactions among substrates competing for access to P450 catalytic sites; toxins that interfere with P450 functions thus have the potential to compromise both toxin clearance and chemical communication.
Much as cells within a body depend on chemical signals to coordinate their activities, social insects depend more heavily on chemical senses for intraspecific communication than do solitary species. Like most other social insects, honeybees are well-endowed with glands (with approximately 15 exocrine glands located throughout the body), and the majority of these glands produce multicomponent pheromone blends. 7 Disruptions in gland function caused by pathogens or toxins, although not lethal to an individual, can profoundly influence colony health by interfering with nest homeostasis and social regulation. The queen, for example, produces both multicomponent primer pheromones (mandibular gland pheromone or queen signal) that direct physiological, behavioral, and developmental changes in workers and release pheromones that elicit immediate behavioral responses in workers and drones. A sex pheromone blend, for example, attracts numerous drones during the virgin queen’s nuptial flight. Workers produce pheromones associated with alarm, defense, and foraging recruitment; nestmate recognition; and orientation and even larvae produce pheromones that regulate brood development and induce nursing behavior in workers.
For more than a decade, US populations of honeybees have experienced atypically high levels of annual losses that have been attributed to a number of stressors that vary temporally and geographically; differentiating among those stressors is accordingly a high priority. 8,9 The effect of stressors on individual bees may not reflect effects of those stressors on colony health, and the “footprint” of stressors such as pesticides, pathogens, and parasites may be most clearly discernible in individuals that did not directly encounter the stressor. Toxins ingested by nurse bees, for example, may, by compromising nursing behavior, affect the morphology and physiology of the emerging queen yet have no substantial impacts on the morphology or physiology of the nurse bees that consumed the toxins.
A variety of sublethal effects of pesticides have been documented in honeybee toxicology studies, but identifying their causative mechanisms has proved difficult. Many sublethal impacts are associated with changes in P450 gene expression. CYP4G11, 1 of the 4 CYP4 P450s in the honeybee CYPome, appears to have 2 functions—(1) cuticular hydrocarbon and comb wax production and (2) odorant clearance. 10 Multiple stressors have been associated with differential expression of CYP4G11—expression is downregulated in workers experiencing colony collapse disorder 11 or exposed to glyphosate or to varroa mite infestation and deformed wing virus 12 as well as in larvae exposed to the neonicotinoid insecticide imidacloprid. 13 The consequences of CYP4G11 downregulation on pesticide resistance, pathogen responses, or larval development and subsequent worker behavior, however, are unclear. Differential expression of the CYP3 clade detoxification genes (particularly CYP9Q genes) 14 can be similarly difficult to interpret.
Designing toxicity assays for honeybees is fundamentally different from designing toxicity assays for solitary insects in that the colony is the unit of replication (not the individual bee); colonies change in size, composition, and physiological attributes seasonally, and the genetic composition of a colony changes over time depending on how the queen allocates stored sperm from multiple drones. 15 Moreover, due to food processing and trophallaxis (social food exchange), individuals who collect contaminated raw materials (eg, nectar, which is converted to honey, and pollen, which is converted to beebread) are not the individuals who ultimately ingest whatever toxins remain in food after processing. The lethal concentration 50 (LC50) assays for assessing risks to nontarget species are not designed for eusocial species with a complex behavioral repertoire; pesticides present a unique threat to honeybees in that sublethal effects on behavior and physiology can jeopardize the health of nestmates and the colony overall.
To some degree, honeybees have evolved in a relatively benign chemical environment. While other types of insect herbivores can consume a diversity of plant tissues, bees are restricted to consuming floral products, including nectar and pollen. As a tissue produced primarily as a reward for mutualistic insect visitors, nectar typically, is less well chemically defended by toxic phytochemicals than are other plant tissues. Pollen, too, is defended by a limited range of phytochemicals. Both nectar and pollen, however, contain high levels of flavonoids that can present a toxicological challenge to herbivores. 16 Bees have had a long evolutionary association with floral nectar and pollen and display multiple adaptations for utilizing these resources. Honey and pollen, important foods for workers and worker-destined larvae, are rich in biologically active phenolic acids, such as p-coumaric acid, and flavonols, particularly the polyphenol quercetin; these phytochemicals upregulate CYP9Q3 involved in pesticide detoxification in the midgut. 17 p-Coumaric acid also upregulates genes encoding P450s in 2 other Clan 3 subfamilies, subsets of phase II and phase III detoxification genes, and at least 1 immunity gene. Upregulation of CYP9Q genes has functional significance in that adding p-coumaric acid to a diet of sucrose increases midgut metabolism of coumaphos, a widely used in-hive acaricide, by ∼60%. 18
In larval bees, however, exposure to p-coumaric acid has different consequences. While worker-destined female larvae are fed protein-rich secretions of the hypopharyngeal, mandibular, 19 and postcerebral 20,21 glands (called worker jelly) for 3 days and then are fed a mix of beebread, honey, and jelly, 19 queen-destined female larvae are fed royal jelly from the same glands exclusively, with only trace contaminants of plant foods. 19,22 RNA-Seq analysis of larvae revealed that, as in adults, detoxification and immunity genes are upregulated in response to p-coumaric acid, but, in addition, expression of a suite of genes involved in caste determination is affected by p-coumaric acid, with the phytochemical acting as a partial chemical castrating agent that prevents worker-destined larvae from emerging as reproductive adults with functional ovaries. 23
Like p-coumaric acid, quercetin is a ubiquitous component of the diet; multiple members of the CYP6AS subfamily metabolize quercetin 24 and are upregulated by p-coumaric acid as do members of the much smaller CYP9Q subfamily. Despite its universal presence in the honeybee diet, quercetin can have adverse sublethal effects on bees. Consumption of nectar with high concentrations of quercetin interferes with worker responsiveness to queen mandibular pheromone, which, among other functions, inhibits worker ovary development and elicits queen care behavior; reduced responsiveness results in socially disruptive aggressive behavior toward the queen. 25 Moreover, feeding 3-day-old bees, which normally consume only worker jelly, quercetin results in downregulation of 15 genes in the oxidative phosphorylation pathway, potentially slowing growth and reducing energy available for withstanding stress. 26
Because bees depend on P450-mediated metabolism for both chemical communication and xenobiotic metabolism, disruption of this enzyme system by environmental stressors has tremendous potential to influence colony health. Although honeybee detoxification systems evolved in a relatively benign chemical environment, contemporary agricultural practices are exposing bees to escalating numbers of novel xenobiotics. Mullin et al 27 reported detecting “…121 different pesticides and metabolites within…wax, pollen, bee and associated hive samples.” In the presence of novel xenobiotic challenges, bee P450s may be especially prone to synergistic interactions among substrates because there are relatively few of them, particularly in subfamilies, that detoxify xenobiotics (ie, CYP9Q). Docking these 121 pesticides and metabolites in silico into the active pocket of a molecular model of CYP9Q1 identified 68 compounds that fit into the pocket, including multiple triazole fungicides; all of these compounds have the potential to synergize phytochemicals and other pesticides. To cite just one of many examples, when adult worker bees are fed quercetin in the presence of the triazole fungicide myclobutanil, concentrations of (unmetabolized) quercetin in the hemolymph increase significantly, and the amount of adenosine triphosphate (ATP) present in thoracic muscles declines, which may compromise the ability of workers to forage. 26
Impacts of stressors such as xenobiotics on workers can have ramifications on colony health if sufficient numbers of workers are affected. A honeybee colony cannot function without a healthy queen, and workers are largely responsible for maintaining the health of the queen and for rearing new queens. Nutrients from pollen and honey are processed by nurse workers to produce royal jelly, the sole queen larval food, via the hypopharyngeal, mandibular, and postcerebral glands, with the major royal jelly proteins synthesized in and secreted from acini of hypopharyngeal glands. 28,29 Thus, the anatomy, morphology, and consequent functionality of the hypopharyngeal gland are more important in the social context of the colony than they are to the life of the individual nurse bee and as such provides an exemplar of the specific challenges of evaluating toxicologic pathology in the honeybee.
Development of the hypopharyngeal glands begins soon after workers emerge as adults after pupation and achieve peak weight and size within 6 to 12 days 30 ; at this point, nurse bees tend queen cells, monitoring the queen larvae and provisioning them with royal jelly. Consuming pesticide-contaminated pollen may interfere with the quality of care, such as frequency and duration of visits provided to the larval queen cell by nurse bees. For example, many almond orchards in California are currently treated during bloom with a tank mix of the insecticide chlorantraniliprole and the fungicide propiconazole. With high LC50 values, these 2 pesticides have been considered “bee-safe” and in fact are not acutely toxic to workers under field conditions. 31 The potential exists, however, for sublethal effects of ingesting this pesticide combination that can influence colony health indirectly via socially mediated interactions, specifically nurse care of brood that can be measured only within the context of the colony.
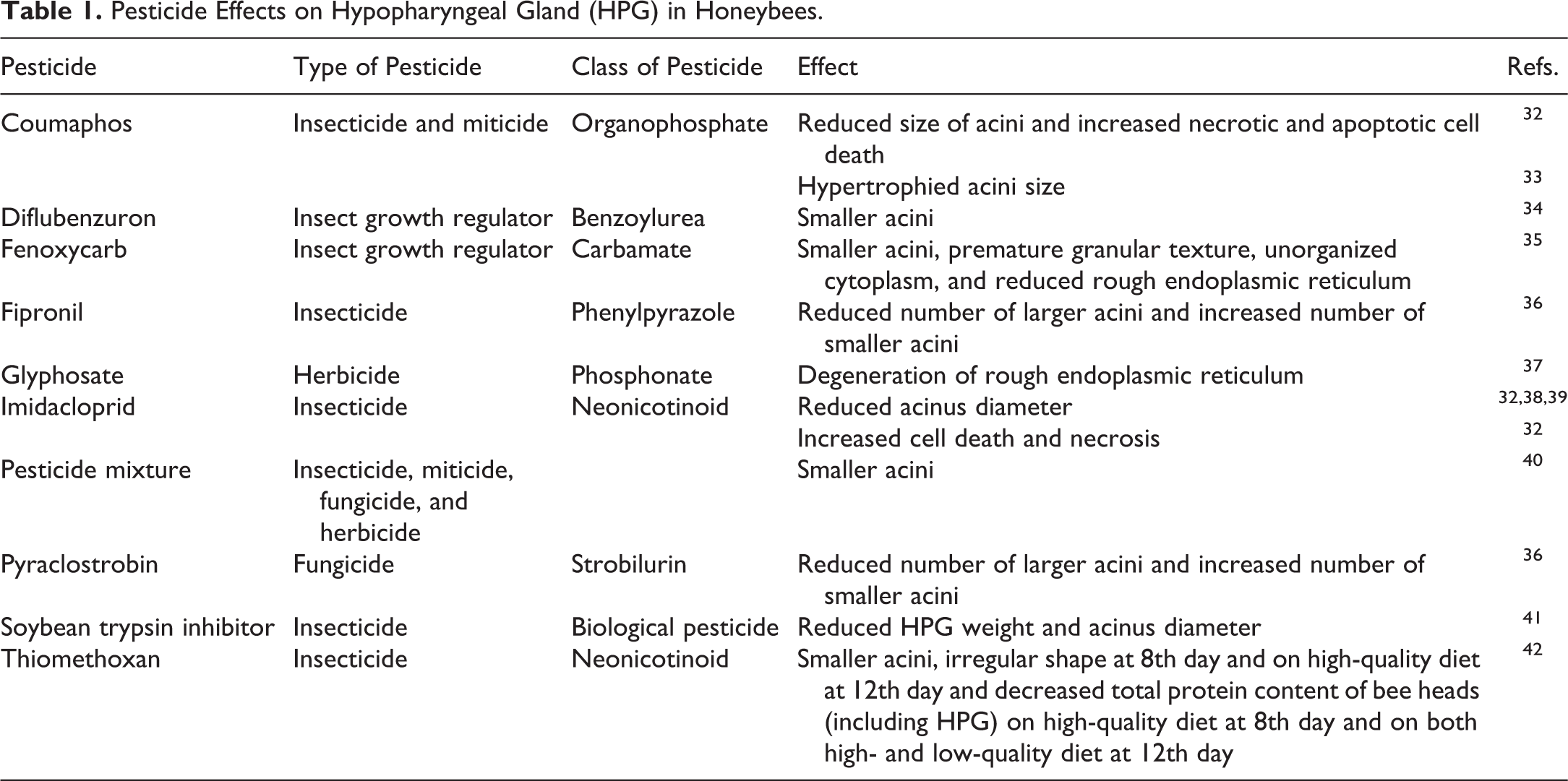
Pesticides can affect the quality of nursing in 2 ways—by altering the function of the glands producing components of royal jelly, thereby reducing the amount or quality of queen food available, and by altering the frequency, duration, and quality of nursing behavior. Hypopharyngeal gland size, morphology, and activity are known to be affected by a variety of pesticides, including herbicides, fungicides, and insecticides (Table 1). Reductions in gland size and function can lead to decreased royal jelly secretion 43 and consequent colony failure through diminished brood production, premature transition from nursing to foraging behaviors, shortened worker life span, and reduced colony size. 38 Moreover, pesticides, including fungicides, herbicides, 6 and insecticides, 44 can alter nurse food consumption patterns and increase the risk of transmission of unmetabolized pesticides to larvae via royal or worker jelly. For example, although not known to affect hypopharyngeal gland size or function, chlorothalonil may, like several other pesticides, act as a feeding stimulant. 6 Nurses exposed to pesticide-contaminated pollen may thus respond to its presence by consuming greater quantities of pollen and hence pesticide, possibly leading to production of glandular secretions with higher levels of pesticide contaminants passed on to the developing queen-destined larva. Finally, queens tended by nurse bees adversely affected by pesticide consumption may experience developmental delays and, upon emergence, may exhibit performance deficits, such as reduced flight capacity (which in turn could have lifelong impacts on fertility and egg production if she fails to mate with sufficient number of males).
Pesticide Effects on Hypopharyngeal Gland (HPG) in Honeybees.
Conclusion
In summary, evaluating toxicologic pathology in the honeybee requires a different perspective to be applied in developing assays that take into consideration the social context that ultimately determines colony health. In a way, doing so is a dramatic departure from the standard practices in the field; in fact, histopathological findings can be identified as “adverse” according to Kerlin et al 45 when they are associated with harm to the test animal. With eusocial species such as A. mellifera, even expanding the definition of “harm to the test animal” to include any histopathological change that interferes with the ability of the test animal to carry out its social function may not suffice. In the case of the honeybee hypopharyngeal gland, even if pesticides reduce the size of the gland acini and interfere with royal jelly production in workers, queens as both larvae and adults are tended not only by a single individual worker but by a retinue of workers; to a great extent, honeybees can compensate for reduced efficiency or even losses of workers carrying out a specific social function by recruiting other workers as replacements. 46
Despite the challenges of interpreting findings, toxicologic pathology is of enormous value as an approach to identifying impacts of stressors and differentiating among them at the cellular, organ, and tissue level. Those researchers most concerned about the long-term future of honeybees and beekeeping in the United States can benefit from reaching out to their counterparts who work with vertebrates (including, or maybe even especially, humans) to figure out how best to determine how the complexities of complex social living influence impacts of toxicologically inimical environments.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This manuscript was prepared with grant support from the USDA Agriculture and Food Research Initiative (AG 2017-67013-26533), the Almond Board of California, and the North American Pollinator Protection Campaign to May R. Berenbaum.
