Abstract
In this opinion piece, we discuss some proposed principles for designating adversity and nonadversity of histopathological changes. The suggested approach categorizes the classes of findings noted in toxicity studies with illustrations and examples and suggests adversity or nonadversity for each class, in the authors’ opinions, with rationales. Although the suggestions and examples offered in this opinion piece are generally in agreement with Society of Toxicologic Pathology best practices guideline on adversity, the authors suggest and highlight occasional divergences and differences of opinion. This is because making an adversity call is a complex and challenging topic that is difficult to simplify. Some of the challenges in deciding on adversity are discussed, especially those related to making an adversity call on a histopathological finding in isolation, based on the nature and extent of severity. The authors demonstrate some of these situations with examples. Finally, the authors suggest, in contrast to the guidelines, occasional use of a separate category for findings that are less easily classified.
*This is an opinion article submitted to the Toxicologic Pathology Forum. It represents the views of the author(s). It does not constitute an official position of the Society of Toxicologic Pathology, British Society of Toxicological Pathology, or European Society of Toxicologic Pathology, and the views expressed might not reflect the best practices recommended by these Societies. This article should not be construed to represent the policies, positions, or opinions of their respective organizations, employers, or regulatory agencies.
Keywords
Introduction
Regulatory toxicity studies provide data on the toxic effects of test items and identify their adverse and nonadverse effects, as well as the doses at which these adverse effects occur. From these studies, one can conclude the lowest dose at which adversity is observed or the highest dose at which adversity does not occur. However, these determinations should be based on collective toxicity studies data as a whole (eg, clinical observations, clinical pathology) and not on anatomic pathology data alone. A collective weight of evidence approach is recommended by Society of Toxicologic Pathology best practices guidelines. These nonclinical adversity determinations are subsequently used to assess potential risks to humans.
Study pathologists are often requested to designate test item–related findings as adverse or nonadverse to help with overall adversity calls. Making an adversity call is a complex and challenging topic that is difficult to simplify. However, this opinion piece generally discusses histopathology findings alone and attempts to define adversity and nonadversity in isolation, as this situation is not uncommon. The reader is invited to refer to the 2 best practice publications listed below for a review of the literature on the history of the definitions of adversity in nonclinical safety and risk assessment, as well as the currently endorsed definition.
Pandiri et al 3 in their publication “Proceedings of the STP 35th Annual Symposium: Continuing Education Course: Is It Adverse, Nonadverse, Adaptive, or Artifact?” provided illustrations for some examples on adversity and nonadversity. In this opinion piece, we have similarly attempted to illustrate some principles of designating adversity and nonadversity of histopathological findings, based on our perspective and experience, with suitable photomicrographs of histopathology lesions from toxicity studies.
Discussion
Histopathological findings encountered in a study can be spontaneous/incidental or test item related. The incidental findings encountered in toxicity studies encompass a wide variety of findings, including physiological phenomena such as female cyclicity, pregnancy, immaturity, age-related pathology, and on occasion, lesions produced by coexisting disease processes.
The toxicological pathologist’s first challenge is to differentiate test item–related lesions from background pathology. 2 Many background lesions, particularly in lifetime studies where spontaneous neoplastic and degenerative lesions are common, may be harmful to the animal. However, adversity is not determined for these incidental findings in the anatomic pathology report, as they are not test item related and therefore not relevant to safety assessment. The authors are fully in agreement with this principle. The anatomic toxicologic pathologist’s concern is solely with test item–related findings and their adversity, in relation to harm done to animals within that toxicity study/test system.
When test item–induced findings are present, they may be primary or secondary, progressive or nonprogressive, and reversible or irreversible. Induced changes can also vary greatly in their severity, ranging from subtle minor effects, adaptive changes, and hormonally mediated effects, to extensive histopathological changes that are incompatible with life, either in isolation or in combination with other findings. Thus, induced lesions have to be interpreted carefully by toxicologic pathologists, and their adversity attributed within the context and objectives of the study. In view of the variable nature of these changes and their morphologic presentation(s), a clear understanding of each lesion or combination of lesions, and their impact, is essential before one can attempt to categorize any findings as adverse or nonadverse. This is because pathogenic mechanisms and functional impact, among other factors, have a bearing on whether the lesions should be considered adverse or not. In addition, potential progression, although not listed as a criterion in the STP best practices guidelines, might have to be taken into consideration on certain occasions. Severity grading may relate to determination of adversity, and it is sometimes necessary to describe the rationale for the specific grading scheme for a given study within the anatomic pathology report. 1,4
Factors such as intended therapeutic use, serious unmet medical need, or lack of human relevance are irrelevant to the determination of adversity within a nonclinical safety assessment. These aspects are relevant to the overall risk assessment and should be addressed elsewhere, such as in regulatory submission documents.
It should be considered that, using some of the principles suggested in this opinion paper, there might be histopathological findings which do not clearly fall into one category or another, for example, in the case of novel lesions with little or no published literature information. In these circumstances, the toxicologic pathologist may possibly need to indicate that these findings are unclassifiable. In such cases, scientific documents prepared using data from multiple studies may offer additional information or provide context to better evaluate these findings. This could be accomplished using an appropriate rationale for submission to the regulatory authorities within the overall context of a drug or chemical development program, such as the case of vehicle-related vacuolation in the choroid plexus. However, in the vast majority of cases, it should be possible for the pathologist to provide guidance on adversity and nonadversity of induced findings.
Adverse Findings
The definition of adversity as published by the Scientific Regulatory Policy Committee Working Group of the STP
1
is “A term indicating harm to the test animal.” Adverse histopathological findings can thus be construed as those associated with harm or damage to the test animal. The authors suggest the following as categories of histopathological findings in which harm to the animal occurs and in which a classification of “adverse” should be considered: Changes which are clearly adverse. Changes which are adverse only at higher severities. Changes which are adverse only when seen in combination with deleterious findings. Test item–related exacerbations of spontaneous findings.
1. Changes Which Are Clearly Adverse
Neoplasia
Test item–related tumors, whether benign or malignant, should be considered adverse in short-term studies regardless of whether the mechanism of induction is direct or indirect. For example, malignant lymphoma induced by immunomodulators, mammary tumors derived in rodents through hormonal pathways, and thyroid tumors secondary to hepatic microsomal induction are generally considered adverse as development of a tumor indicates perturbation of normal function sufficient to result in uncontrolled expansion of a cell population and is indicative of harm. In carcinogenicity studies, it is not usually necessary to designate adversity to neoplastic or non-neoplastic findings.
Hyperplasia, focal, or atypical
Atypical hyperplasia, when induced by a test item (eg, atypical mammary gland hyperplasia; Figure 1) in shorter term toxicity studies, might be considered adverse, as such lesions have the potential to progress to neoplasia. Similarly, focal and nodular hyperplasia could also be considered adverse, in our opinion, due to their potential for progression (Figure 2). These suggestions by the authors diverge from the papers by Kerlin et al 1 and Palazzi et al 2 which limit the adversity call to the confines of the duration of the study regardless of anticipation of effects in longer duration toxicity studies. The authors suggest using potential theoretical progression of a lesion on occasion, by virtue of their specific morphological features, to designate adversity rather than using actual toxicity study data and/or relying on longer term toxicity studies to further investigate progression of the lesion.
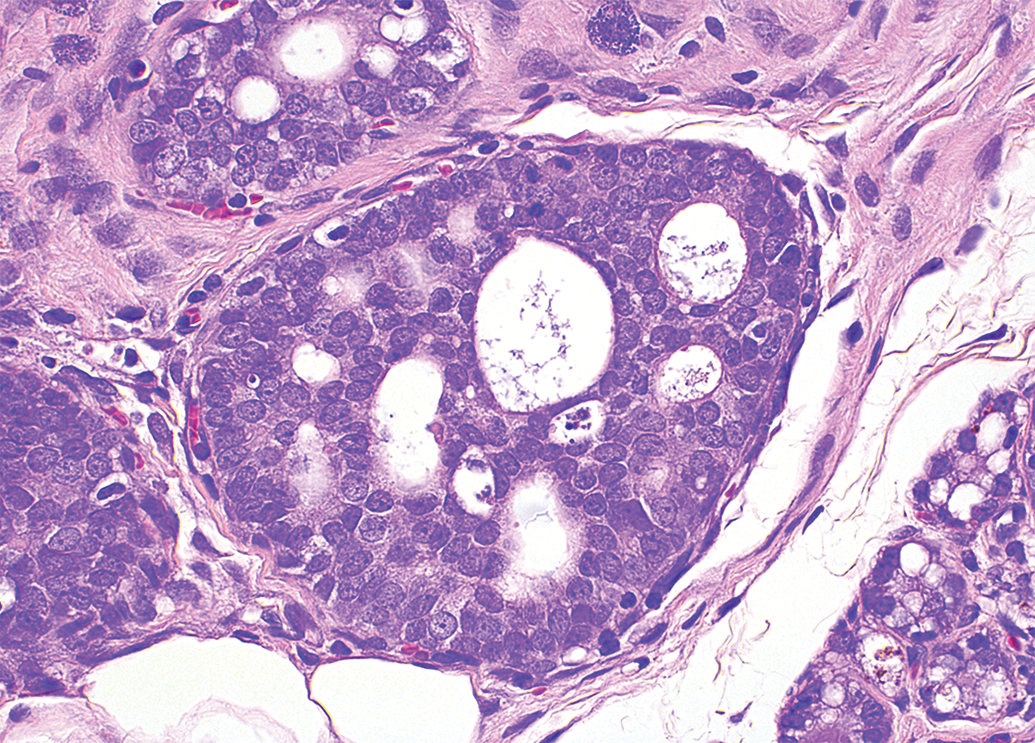
Atypical hyperplasia, mammary gland, Sprague-Dawley rat. H&E.
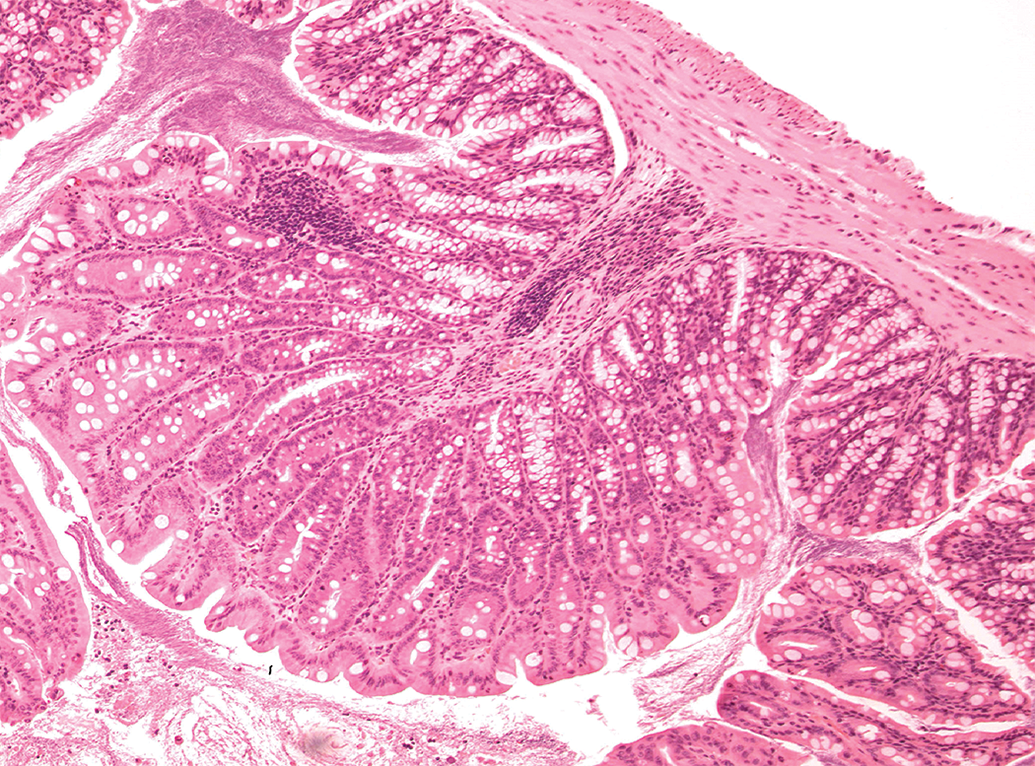
Focal colonic hyperplasia, Sprague-Dawley rat. H&E.
Necrosis and inflammation
Focal necrotic changes in parenchymatous organs are sometimes seen in toxicity studies as background, and differentiating test item–induced lesions from spontaneous changes can be challenging. If necrotic lesions are established as test item related, whether due to local or systemic effects, the likelihood of adversity should be considered as there is permanent damage to and/or loss of cells, even if low grade (eg, hepatocellular necrosis, zonal, focal, or diffuse; Figure 3). Primary test item–related necrosis of the myocardium, renal tubules, mucous membranes, skin, adrenal, pituitary, testes, lymphoid organs, and central nervous system might also be considered adverse, regardless of severity grading, in many cases. For example, multifocal degeneration in the heart of a rat (Figure 4) might be considered adverse. Another example is that of necrosis caused by test items administered by intramuscular or subcutaneous injection. Apart from a minimal degree of procedure-related local tissue trauma, hemorrhage, and inflammation (Figure 5), vehicle- or test item–related necrosis at the site of injection and in adjacent connective tissue (Figure 6), whether due to local or systemic effects of the test item, indicates tissue damage and might be considered adverse.
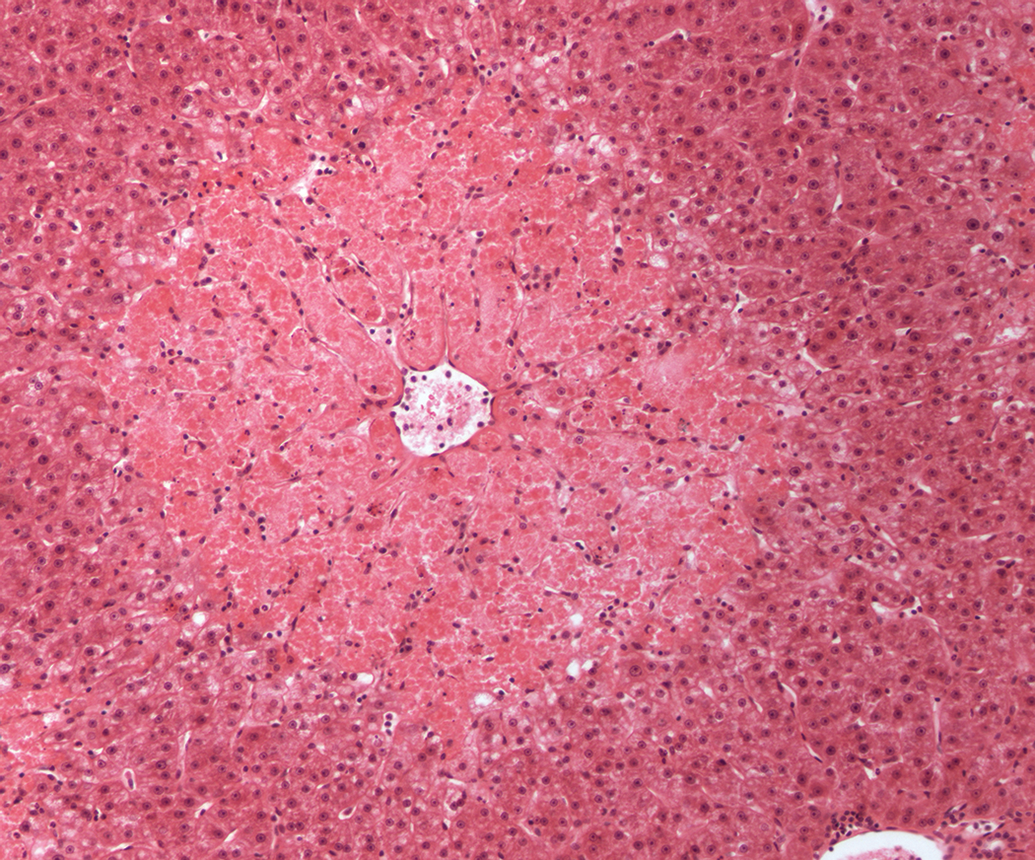
Centrilobular hepatocellular necrosis, cynomolgus monkey. H&E.
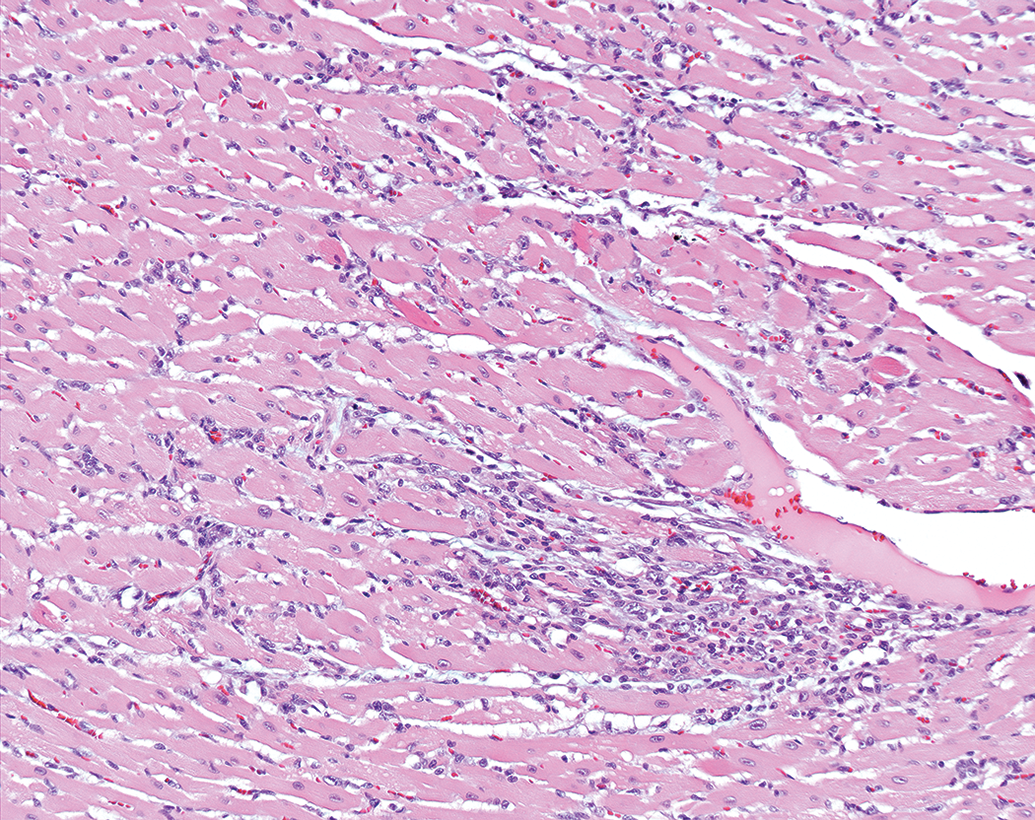
Multifocal myocyte degeneration with inflammatory infiltrate, heart, Sprague-Dawley rat. H&E.
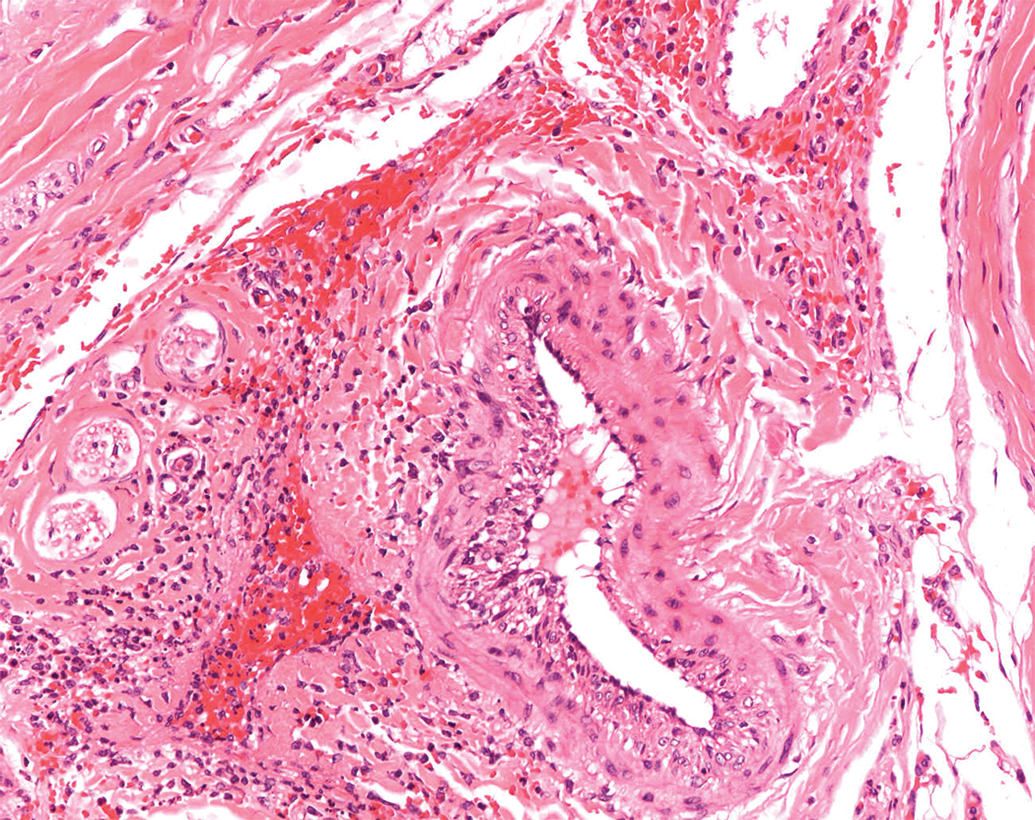
Minimal perivascular inflammation and hemorrhage, intravenous injection site, tail, Sprague-Dawley rat. H&E.
Inflammation with necrosis, subcutis, dog H&E.
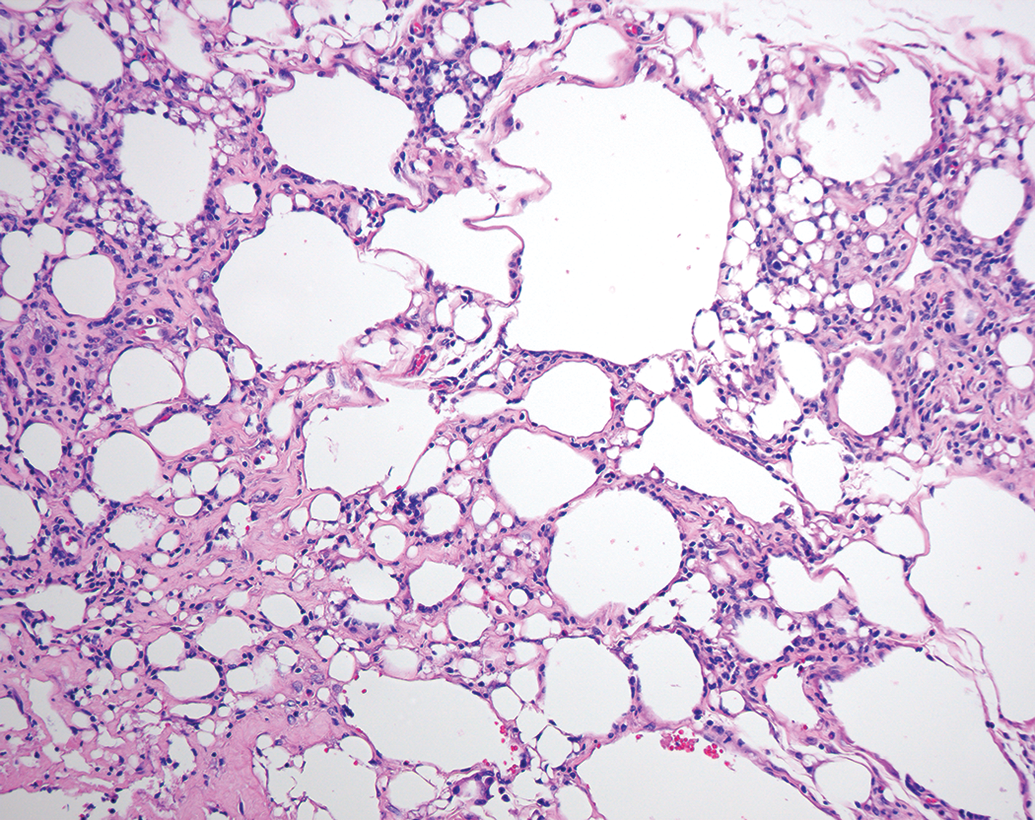
Primary test item–related inflammatory responses usually reflect tissue damage. Test item–related intestinal inflammation, vasculitis (Figure 7), glomerulonephritis, and interstitial nephritis are among some of the examples that could generally be considered adverse, in the authors’ opinion, regardless of severity grade. However, test item–related inflammation is not uncommon, and there may be examples of test article–related inflammation that are not reflective of tissue damage.
Mural inflammation, arteriole, cynomolgus monkey. H&E.
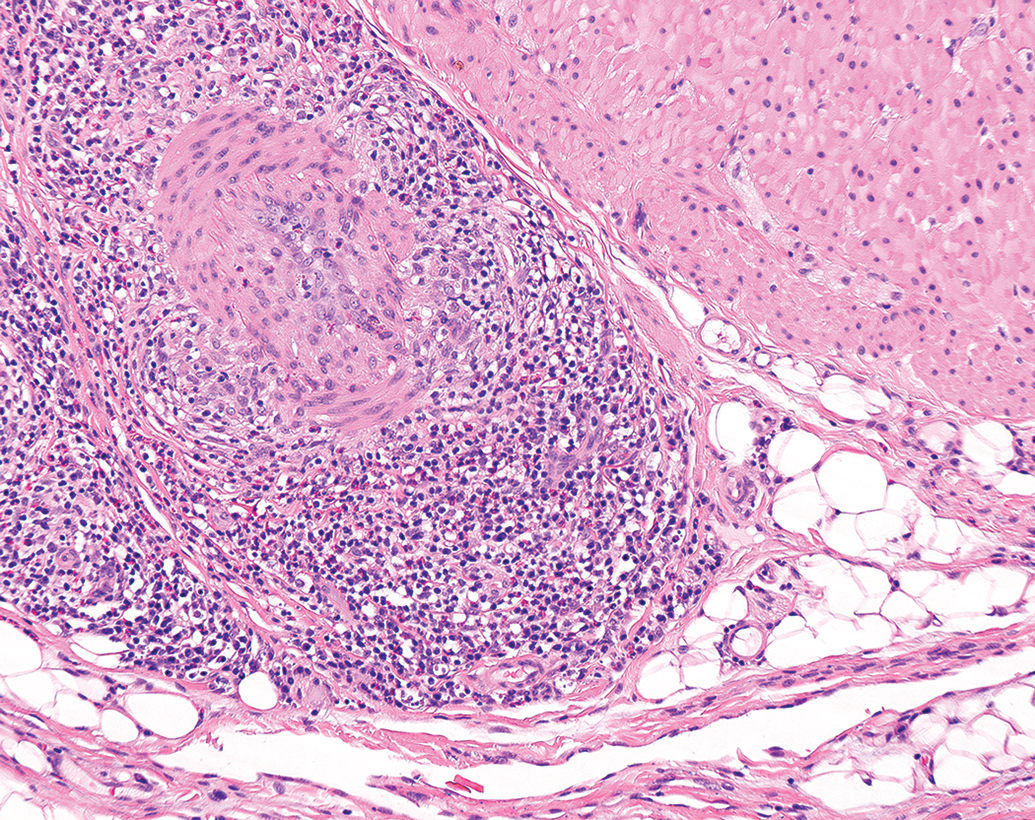
Chronic inflammation of the lungs in inhalation studies, when associated with tissue damage, might be considered adverse. Other examples of clearly adverse findings include test item–related mucosal erosions and ulcerations, fibrosis in parenchymal organs such as the lungs (Figure 8), liver and kidneys, cardiac valvular inflammation, and thrombosis as they all indicate tissue damage and harm. Peripheral nerve degeneration, seen sometimes as a primary and direct effect and also secondary to hypoglycemia with several antidiabetic compound classes, is considered adverse, in the authors’ opinion and in the opinion of others regardless of mechanism. 3
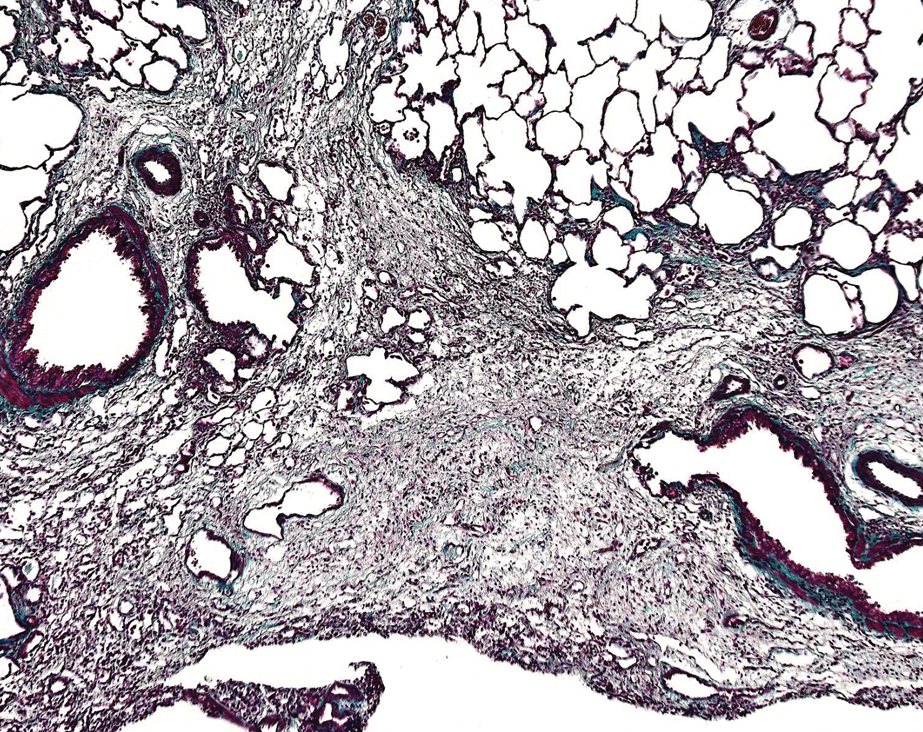
Pulmonary fibrosis, lung, mouse; Masson’s trichrome stain.
In the authors’ opinion, test item–related biliary plugs and porphyrin pigment in hepatocytes, supported by consistent changes in clinical chemistry parameters, indicate a functional perturbation (Figure 9). These findings are not seen as background changes and are indicative of functional disturbances to the animal. They might be considered adverse according to the authors’ experience and the best practice guidelines. 1
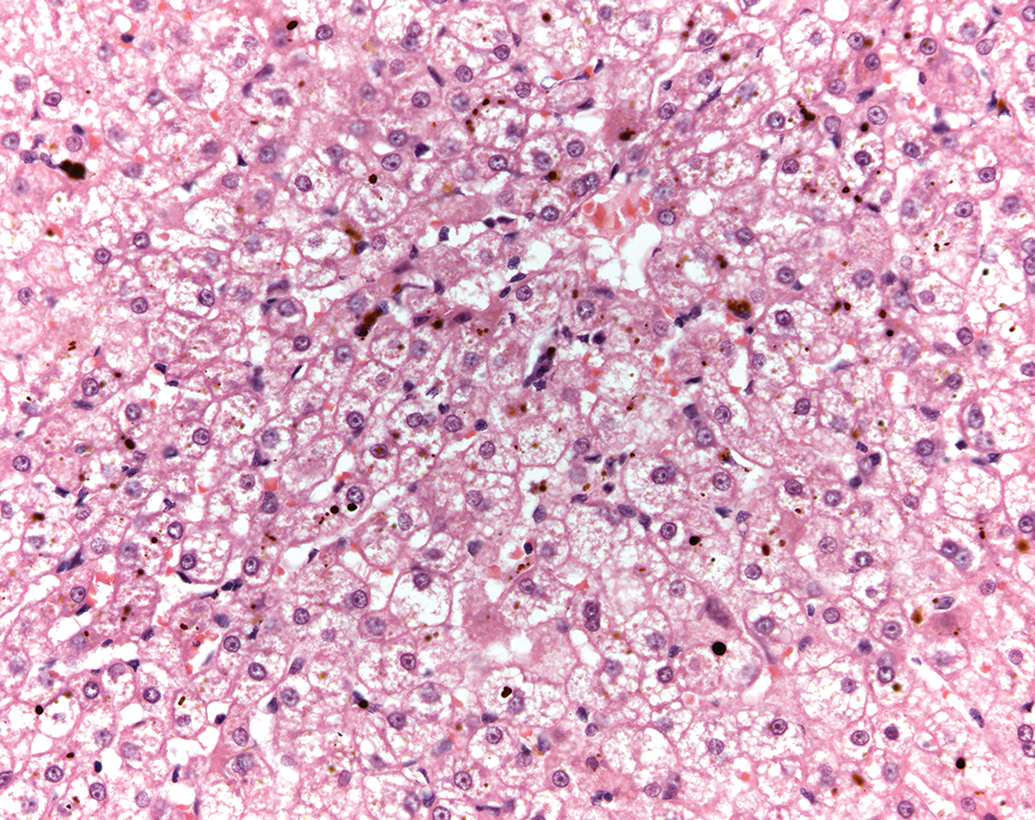
Cholestasis and porphyria, liver, beagle dog. H&E.
2. Adverse Only at Higher Severities
The authors consider that a number of histopathological changes may be adverse only at higher severity levels, 5 as these higher severities are likely, in our experience and opinion, to be associated with changes in clinical signs or chemistry parameters that reflect functional disturbances. Examples of such lesions include alveolar macrophage aggregations in the lung, which are generally considered adverse when they are of a severity that could potentially impair airflow and gaseous exchange. Other degenerative lesions, such as fat deposition in the hepatocytes 6 and renal tubular basophilia, are also generally considered adverse at higher severities alone. Similarly, test item–related apoptosis in lymphoid and other organs at high severities is adverse, as at these levels they are likely to be associated with abnormalities in clinical pathology and other parameters, indicating functional impairment and can thus might be considered “critical.” 4 Some immunomodulators can cause prominent increases or decreases in cellularity in lymphoid organs which could be classified as adverse, as at these levels immune function is likely to be altered. However, such test item–related changes in lymphoid cellularity, when occurring at lower severity grades, can manifest without evidence of immune system function compromise (eg, secondary infections). Loss of hematopoietic cells from the bone marrow, when at higher severity grades, might be considered an adverse finding, particularly when associated with disturbances of hematological parameters.
3. Adverse When Lesions Occur in Conjunction With Deleterious Findings
Some induced lesions are nonadverse when seen in isolation, but when present in conjunction with other changes, such as inflammation, ulceration, hyperplasia, metaplasia, necrosis, or fibrosis, indicate a more complex process involving tissue damage and thus may be considered adverse. 4 For example, pulmonary alveolar macrophages at a low severity, when seen together with inflammation, fibrosis, and/or epithelial hyperplasia of the bronchioalveolar epithelium, are part of an adverse process 7 (Figure 10).
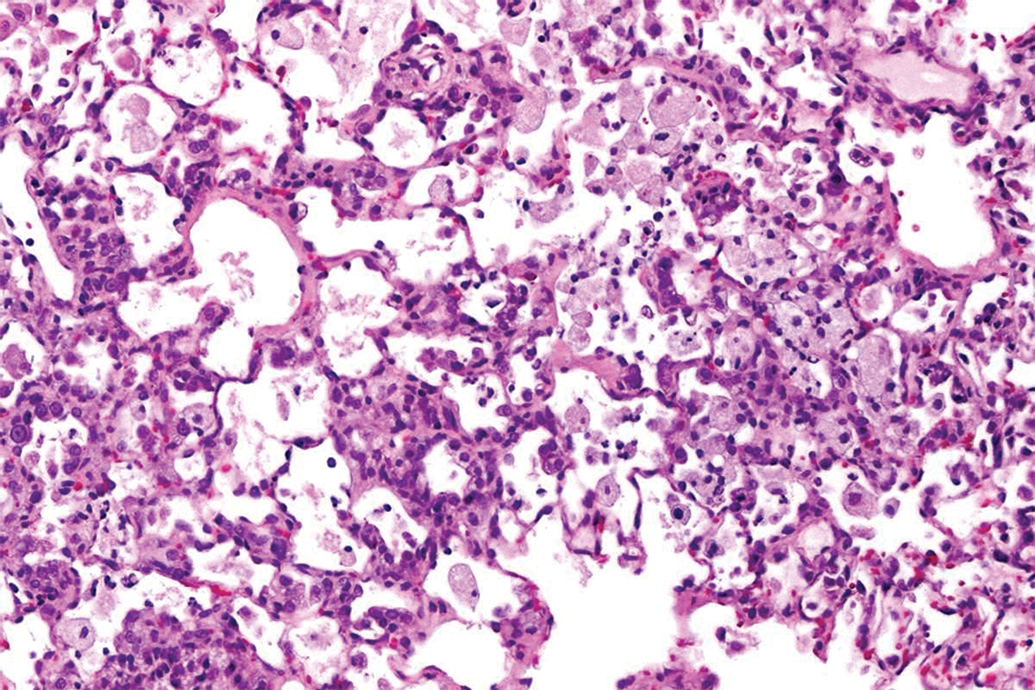
Alveolar macrophages with inflammatory cell infiltration and alveolar epithelial hyperplasia, lungs, Han Wistar rat. H&E.
Accumulations of exogenous or endogenous substances, often at higher severities, when associated with histopathological findings indicative of damage such as degeneration, inflammation, or extracellular spillage, should be considered adverse (Figure 11).
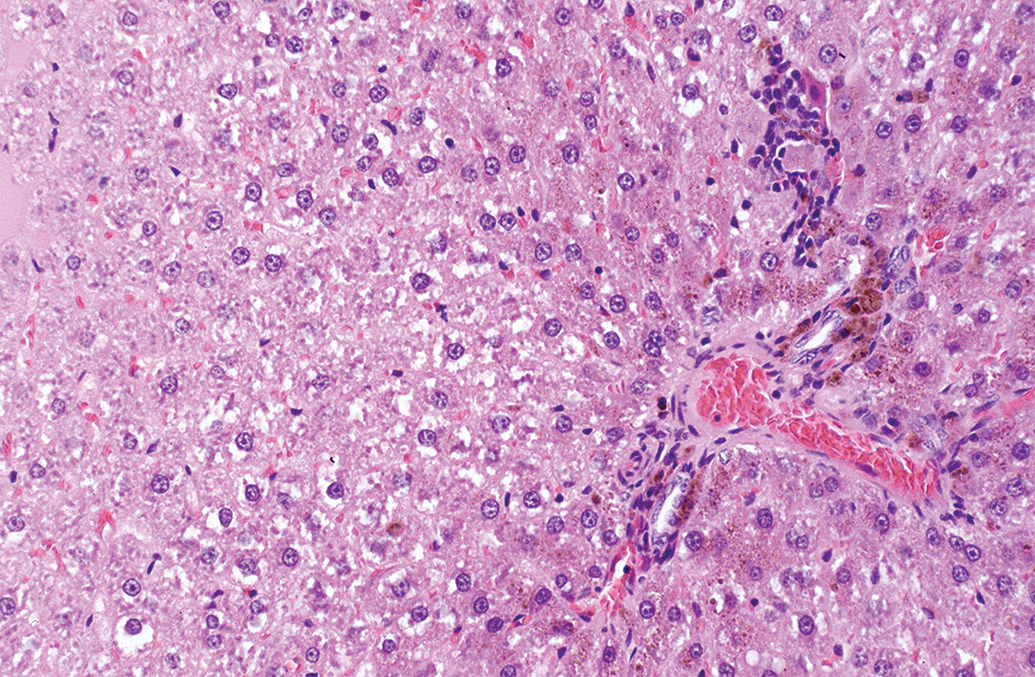
Hepatocyte pigment with inflammatory infiltrate, liver, Han Wistar rat. H&E.
Similarly, laryngeal squamous metaplasia, even when seen at low severities but in conjunction with inflammation or ulceration, might be adverse (Figure 12). 8 Hepatocyte hypertrophy when associated with degeneration and necrosis might be considered adverse, 6 and in these instances, there is often evidence of tissue damage as shown by leakage of liver specific enzymes. 9
Squamous metaplasia and inflammation, larynx, Han Wistar rat. H&E.
4. Test Item–Related Exacerbation of Spontaneous Findings as Adverse
Severity and incidence of spontaneous, often age-related, histopathological findings may be increased as a result of test item administration. Increased incidence and severity of chronic progressive nephropathy (CPN) and cardiomyopathy (CM) in rodents are good examples, as nephrotoxicity can often manifest as increased incidence and severity of CPN, especially in chronic toxicity studies. Similarly, cardiotoxicity can exacerbate CM in rodent chronic toxicity studies (Figure 13). Although these specific syndromes (CPN and CM) are not seen in man, test item–related exacerbations resulting in functional impairment might be considered adverse in the context of the associated study,
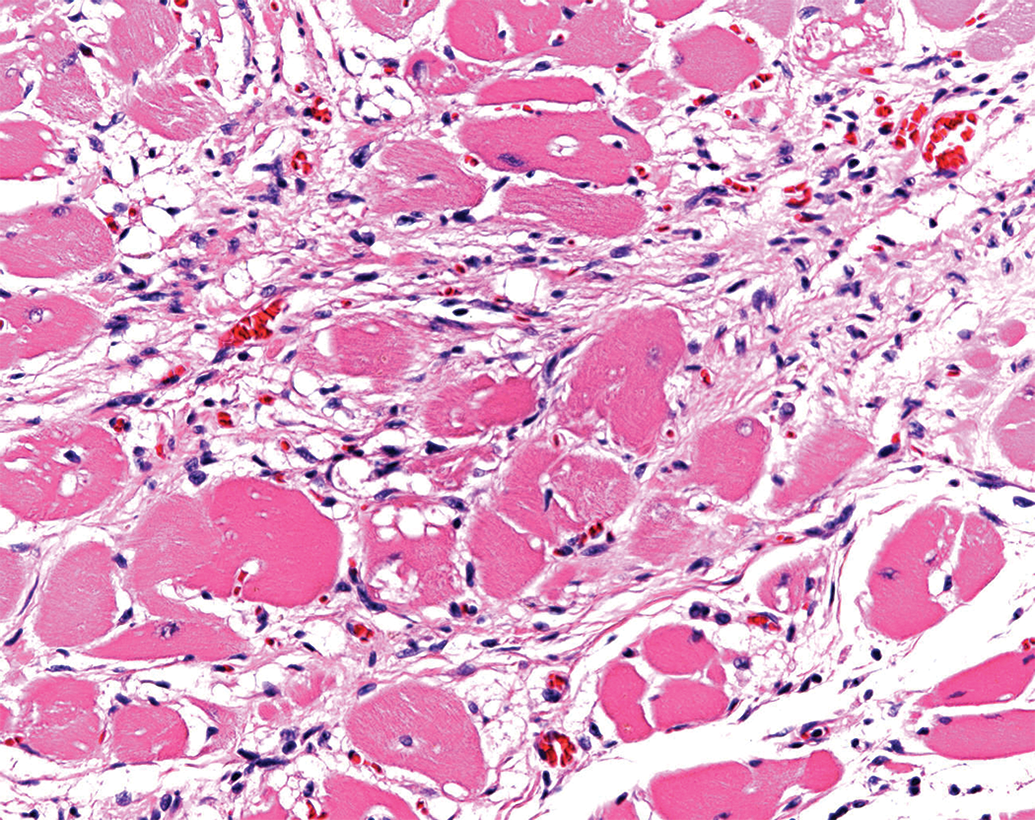
Cardiomyopathy, heart, Sprague-Dawley rat. H&E.
Nonadverse Findings
A number of low-grade test item–related changes which occur in various tissues and organs are generally considered nonadverse. Many of these findings have been extensively studied and well described in the literature, and based on their mechanisms of induction, lack of progression over a long period, reversibility, and other similar criteria can be classified as nonadverse when at a severity which doesn’t harm the animal within the context of the study. These include: Adaptive changes. Secondary changes and physiological responses due to minor perturbations of homeostasis. Minor changes due to pharmacological effects. Test substance–related changes of low severity, with no functional disturbance.
1. Adaptive Changes
Many induced lesions are adaptive changes that are reversible and show little negative functional impact. For example, centrilobular hepatocyte hypertrophy in the liver, seen following administration of agents like phenobarbital, is due to microsomal proliferation and enzyme induction and results in liver enlargement coupled with an increase in liver weight. When seen in isolation in the absence of degenerative changes, this finding might be considered nonadverse.
Cecal enlargement with mucosal hypertrophy occurs as an adaptive response to changes in the diet or alterations in variables such as cecal pH or flora and can manifest histopathologically as diffuse mucosal hypertrophy, sometimes with minimal diffuse hyperplasia (Figure 14). These changes might be considered nonadverse when seen without evidence of inflammatory or degenerative changes or of clinical or functional disturbance.
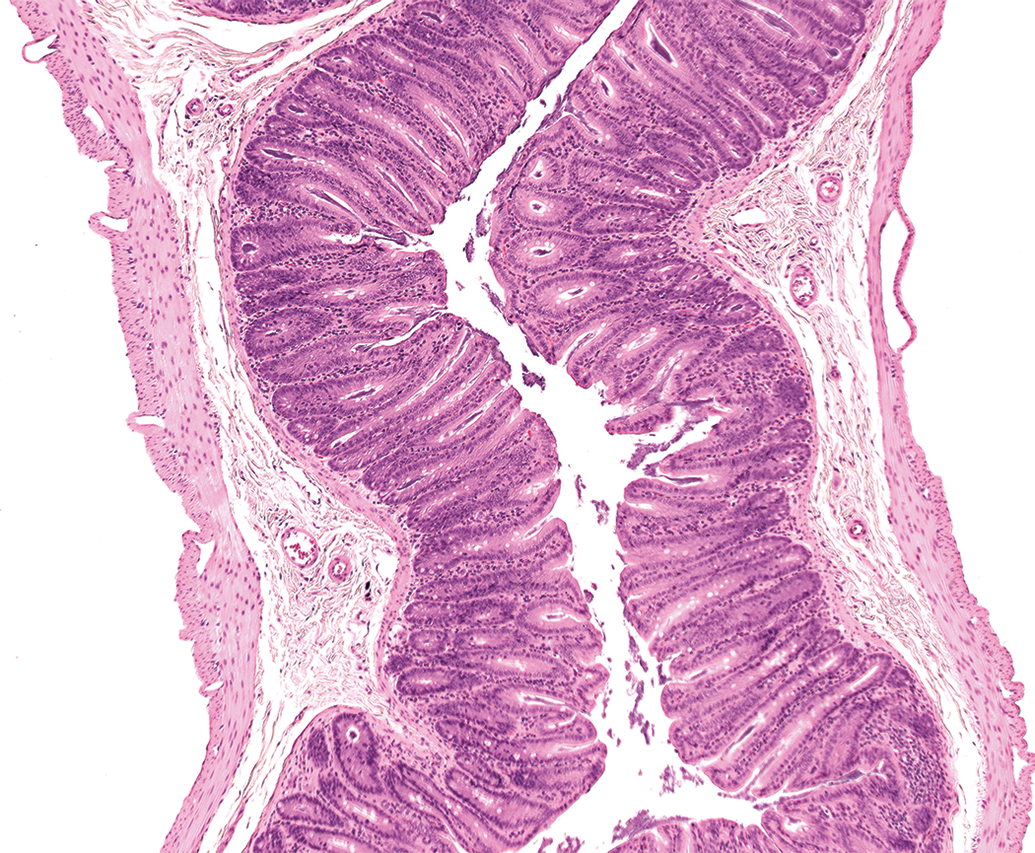
Diffuse mucosal hypertrophy, caecum, Sprague-Dawley rat. H&E.
Minimal to mild squamous metaplasia of the laryngeal mucosa in rodents, when seen in isolation, might be considered adaptive and nonadverse. 3,8,10 Likewise, minimal to slight decreases or increases in extramedullary hematopoiesis in the rodent spleen and other organs can be regarded as nonadverse if seen in isolation (ie, no changes in peripheral hematology or bone marrow histopathology).
2. Secondary Changes and Physiological Responses Due to Minor Perturbations of Homeostasis
Changes in one organ are often secondary to induced primary changes in another organ or to physiological changes in the host. Thyroid follicular hypertrophy, occurring secondary to hepatic enzyme induction, might be considered adaptive and nonadverse when it occurs as a diffuse change at low degrees of severity (Figure 15). Secondary adaptive hypertrophy can occur in other endocrine organs as well. Induced zona glomerulosa hypertrophy of the adrenal glands develops secondary to urinary electrolyte imbalances, such as increased sodium excretion or increased urinary volume. Morphologically, this appears as bilaterally diffuse, even thickening of the zona glomerulosa, often with vacuolation, and shows no progression with length of treatment, even in carcinogenicity studies (personal observation, Figures 16A, B).
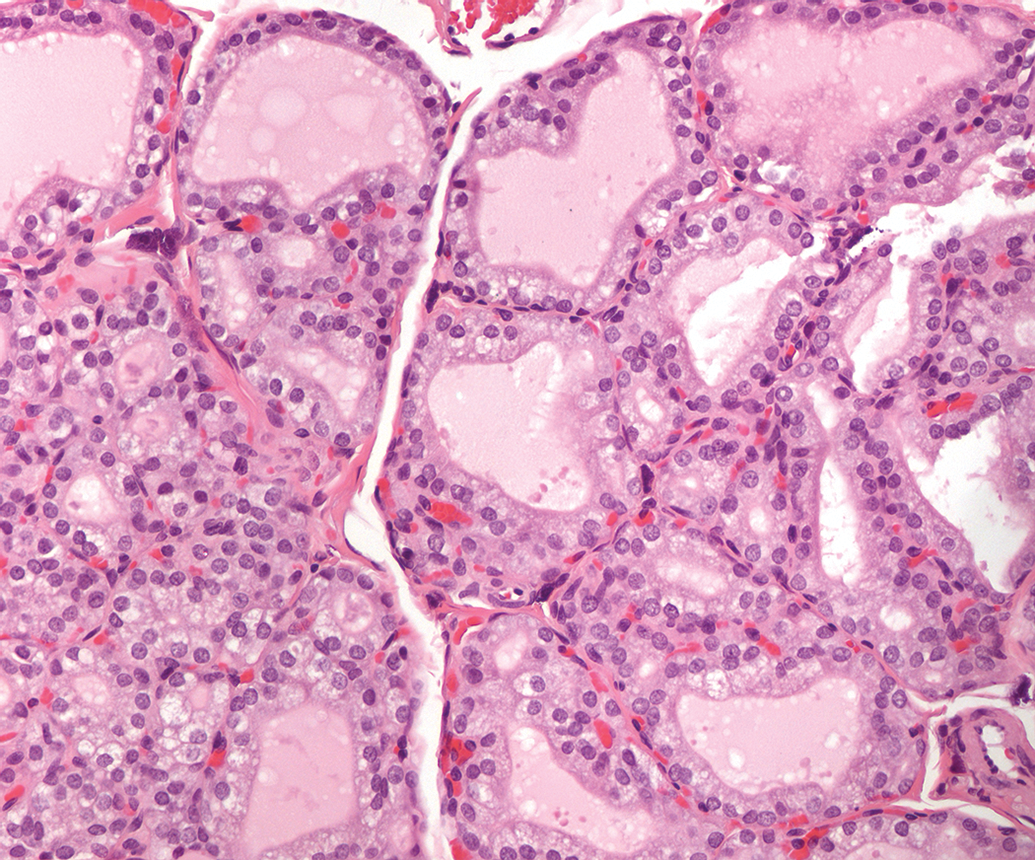
Follicular cell hypertrophy, thyroid, Sprague-Dawley rat. H&E.
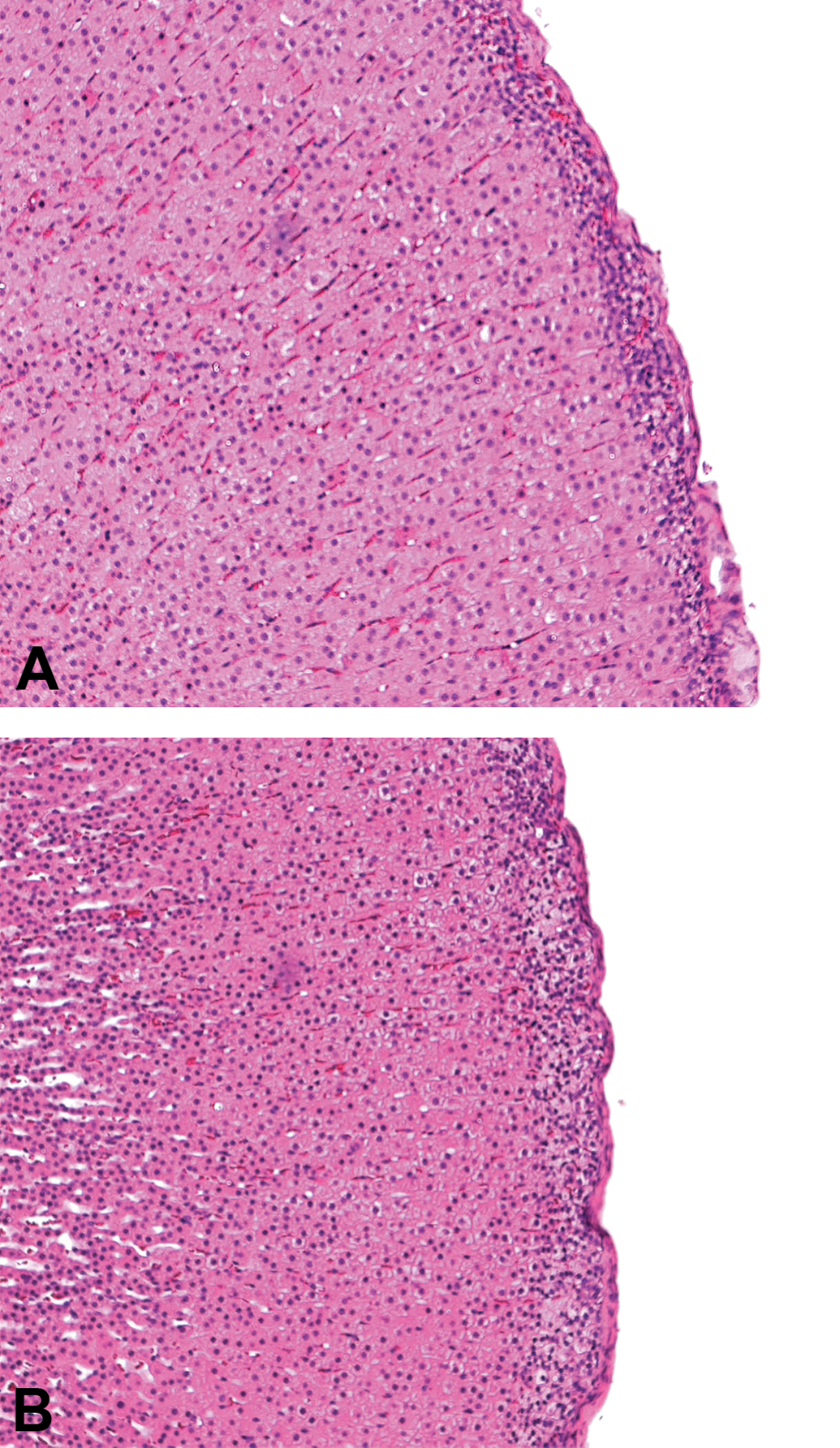
A, Control B, Zona glomerulosa hypertrophy, adrenals, Han Wistar rat. H&E.
Mild hormonally mediated changes in the female reproductive tract, accompanied by changes in the estrous cycle, might be considered nonadverse in standard toxicity studies as these effects are generally reversible, do not reflect harm to the animal in the context of the study, and fall into the category of physiological responses. Keratinization or mucification of the vaginal epithelium, dilation of the uterine lumen, and a decrease in corpora lutea are among some of the nonadverse findings that can occur due to hormonal alterations in general toxicity studies. However, these might be considered adverse in studies designed to assess reproductive performance, 2 highlighting the subjectivity of making adversity calls. Physiological responses such as increased or decreased glycogen in hepatocytes and decreased zymogen granules in pancreatic acinar cells, when on their own, are not considered indicative of harm.
3. Minor Changes Due to Pharmacological Effects
Expected or pharmacologically mediated responses may often be considered nonadverse as long as they cause no harm. However, when they are accompanied by functional impairment or tissue damage, even as expected pharmacologic effects, they could be considered adverse. 1 For example, increased or decreased cellularity of one or more compartments of various lymphoid organs (Figure 17), due to immune stimulation or immunosuppression, respectively, may be considered nonadverse at low severities, as these are not usually associated with clinical perturbations and often fall under the umbrella of expected pharmacological responses without evidence of functional impairment.
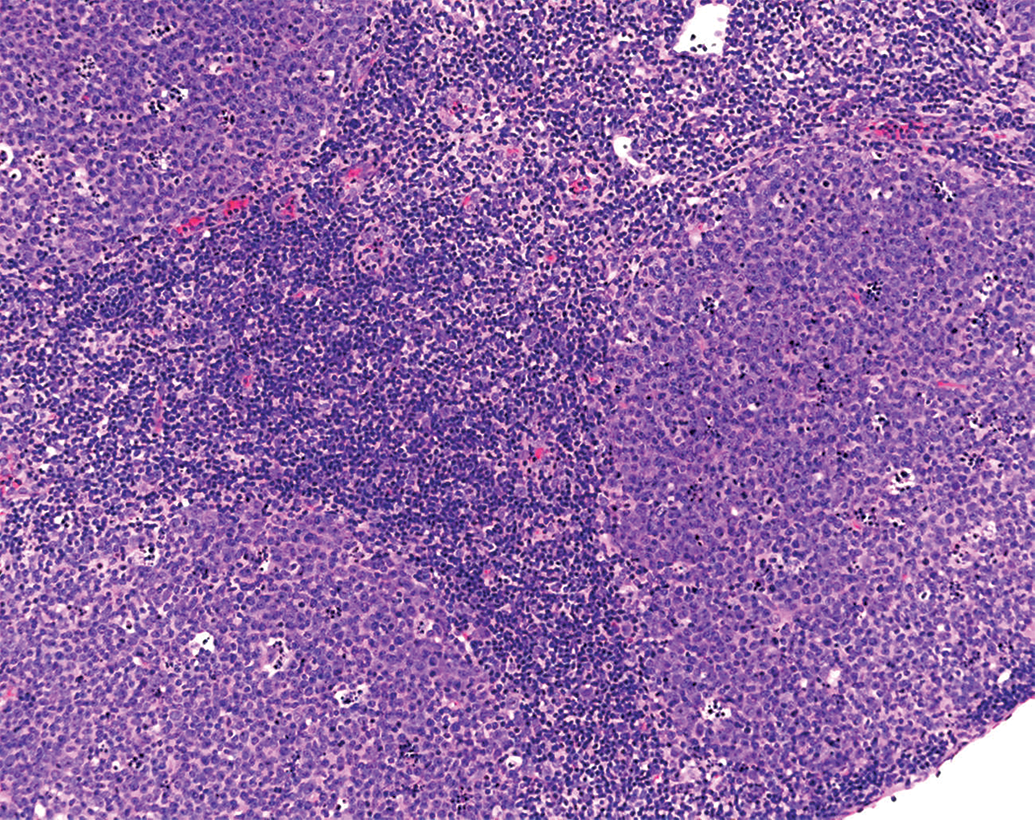
Increased cellularity, germinal centers, CD-1 mouse. H&E.
In our opinion, low-grade test item–related increases or decreases in cellularity of the bone marrow might be considered nonadverse (Figure 18), as long as hematological parameters are within normal ranges and no evidence of cellular damage or inflammation is noted. Test substance–induced inflammatory cell infiltrate, independent of tissue damage, sometimes observed with immunomodulators (eg, Toll-like receptor agonists), could be considered nonadverse.
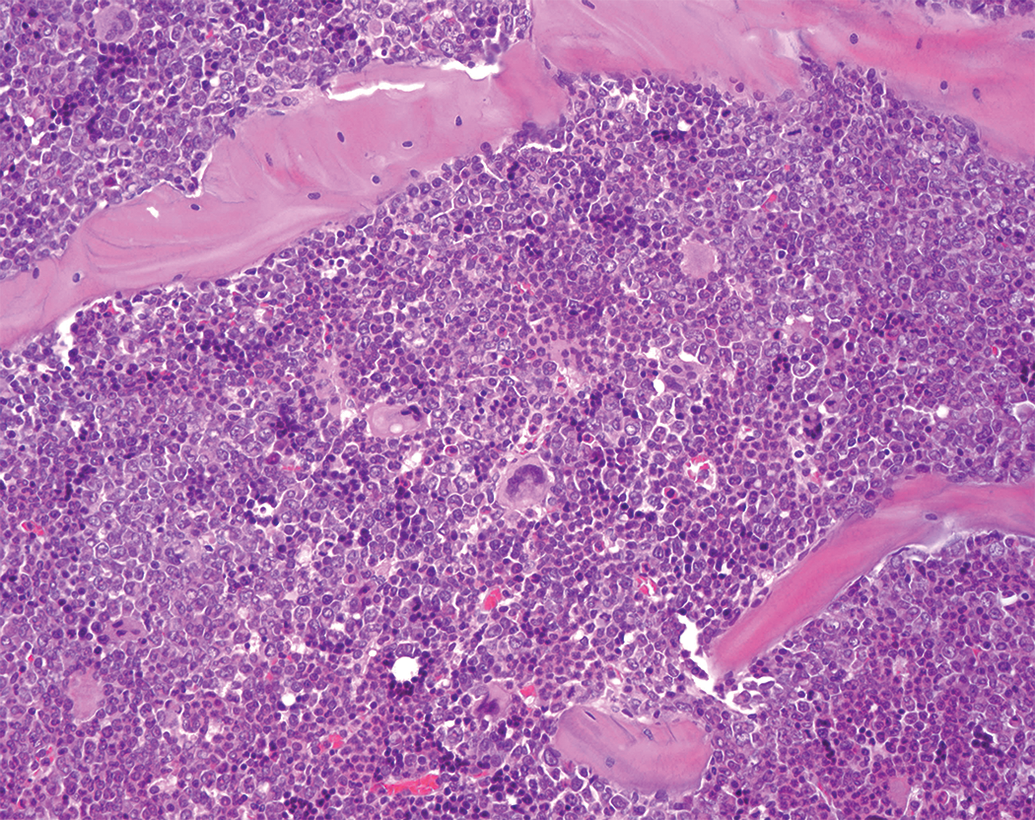
Increased granulopoiesis, bone marrow, CD-1 mouse. H&E.
4. Test Substance–Related Lesions of Low Severity, With No Functional Disturbance
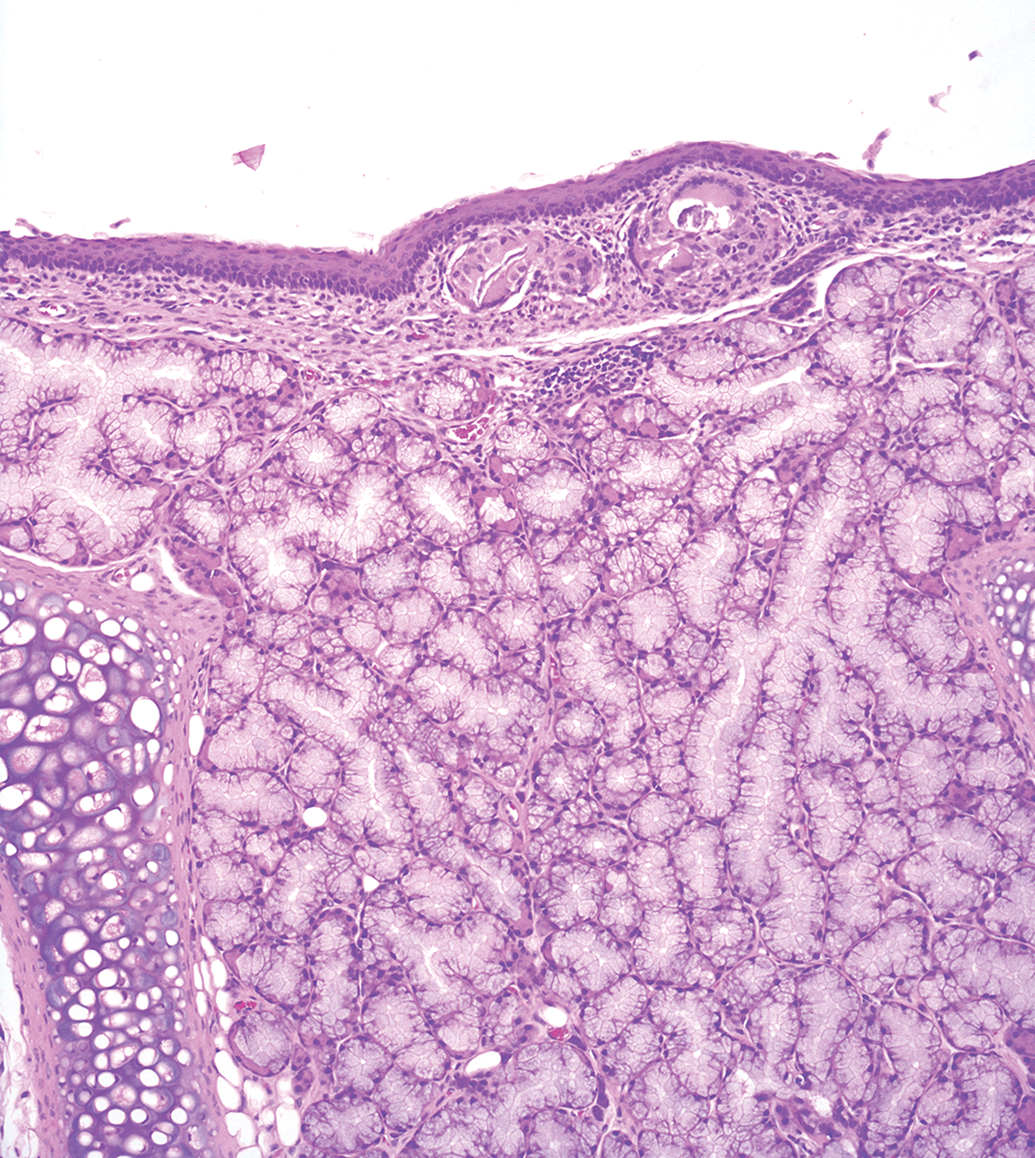
Early and low-grade degenerative changes are often difficult to classify as adverse or nonadverse. Degenerative lesions seen at low severities in many organs and tissues which show no evidence of other disturbances, such as in-life clinical parameters, might be considered nonadverse because they are not associated with functional impairment. This is distinct from cellular necrosis which is lethal, often associated with evidence of functional impairment and might be considered adverse. Examples include basophilic renal tubules, degeneration of muscle fibers, and hepatocellular fatty vacuolation. When at low severities, these findings might be considered nonadverse if they show no evidence of functional abnormalities within the specific study. For example, minimal basophilic renal tubules are not, in our experience, associated with changes in clinical chemistry parameters. In these cases, however, higher severities might indicate adversity, illustrating the subjectivity of these determinations and the overlap between adverse and nonadverse categories. Similarly, adaptive or protective changes, such as diffuse squamous epithelial hyperplasia of the rodent nonglandular stomach, bile duct hyperplasia, epidermal hyperplasia, and goblet cell hyperplasia of the respiratory tract (Figure 19), are unlikely to have associated functional impacts at low-grade severities and might therefore be considered nonadverse when they occur in isolation.
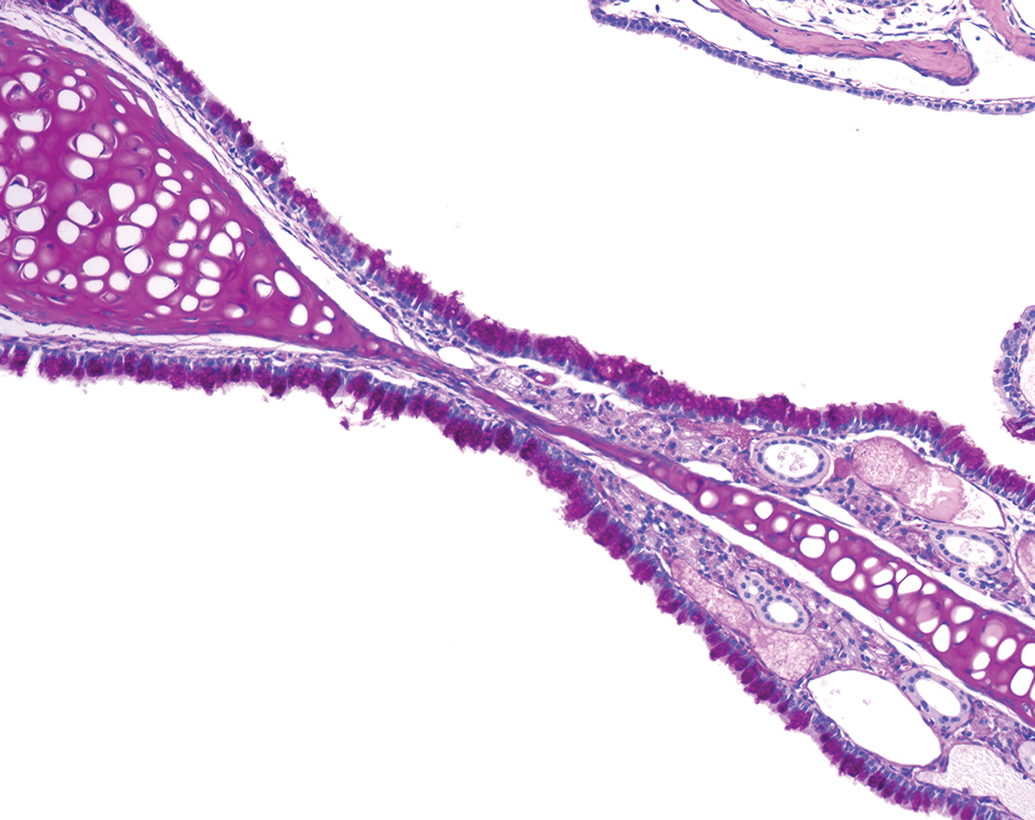
Goblet cell hypertrophy, nasal turbinates, CD-1 mouse. Periodic acid-Schiff stain.
Minimal to mild degrees of induced macrovesicular fatty change in the liver, as isolated lesions, have been considered nonadverse (Figure 20). 6 Accumulation of exogenous substances such as test items or their metabolites, when unaccompanied by changes indicative of tissue injury, might generally also be considered nonadverse.
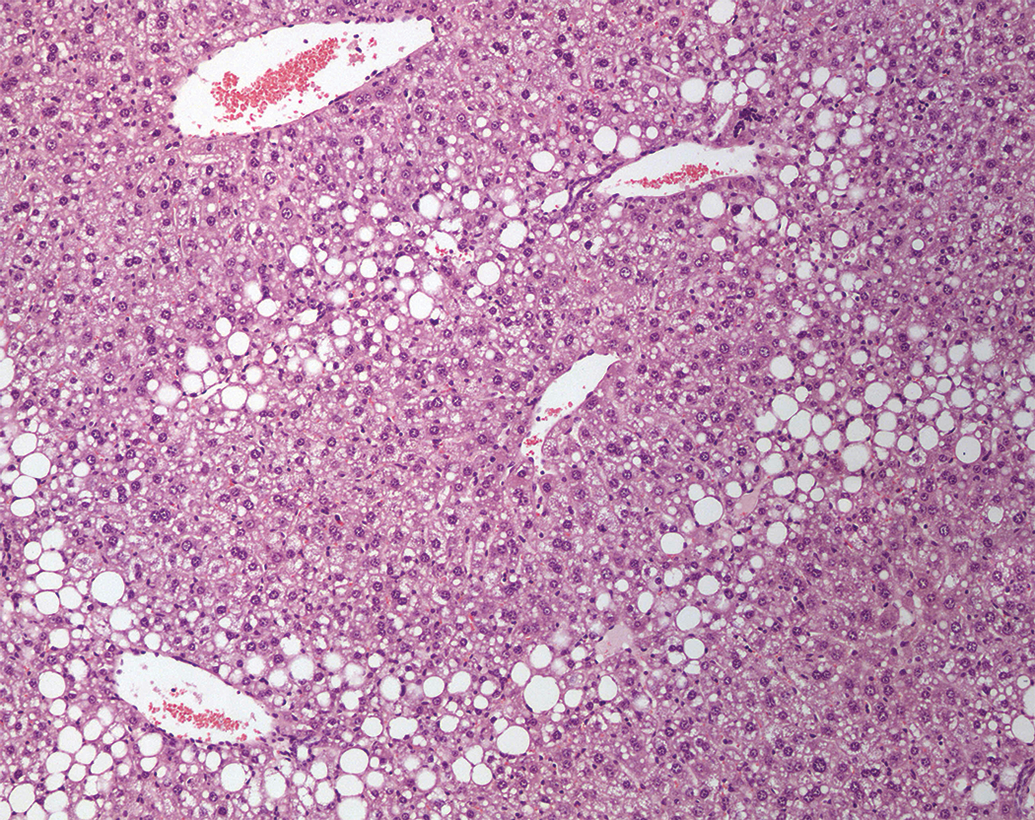
Fatty vacuolation, liver, mouse. H&E.
Endogenous accumulations (with the exception of bile pigments and porphyrin) are often nonadverse (eg, pigment accumulation), but this needs to be determined on a case-by-case basis. Increased lipofuscin in hepatocytes, as an induced test article–related change, is nonadverse at low severity levels unless accompanied by inflammatory changes and/or other cellular damage. Accumulation of eosinophilic droplets in the respiratory tract mucosa is nonadverse when seen without associated inflammatory or degenerative changes and can show no progression even in long-term toxicity studies (personal observations; Figure 21). Test substance–related altered foci in the liver are often also considered nonadverse when they occur at a low incidence and/or severity within an animal or dose group or are within background levels.
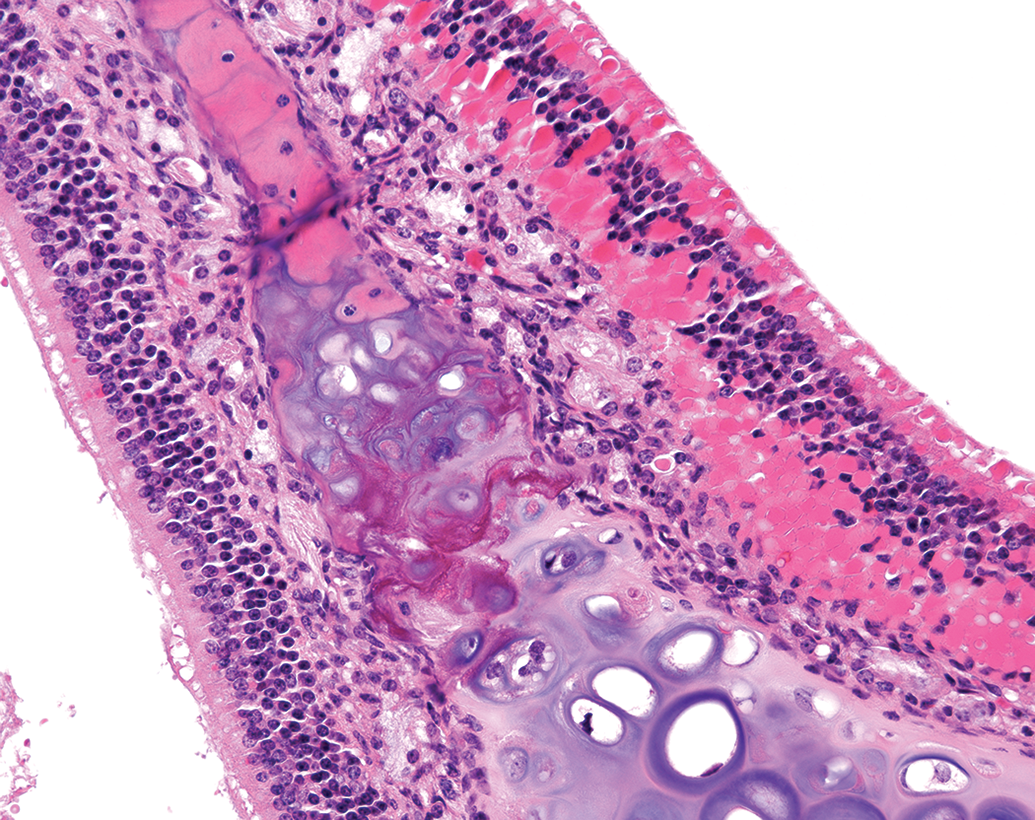
Eosinophilic droplets in olfactory epithelium, nasal turbinates, CD-1 mouse. H&E.
Recovery and Functional Impairment in Connection With Adversity Determination
In our opinion, recovery/reversibility data should not be used as the sole criterion for assigning adversity, although they may contribute to a weight-of-evidence decision within a study for a given finding or dose level. In some organ systems, test item–related lesions may no longer be apparent after the recovery period, although this might not always be indicative of recovery. For example, there may be little or no evidence of hepatocellular or neuronal necrosis after a recovery period due to regeneration (in the case of hepatocytes) and/or complete removal (in the case of neuronal necrosis) of dead cells and debris, but these changes might still be considered adverse.
Similarly, absence of detectable functional disturbance, based on in life observations/clinical pathology, is another general indicator of nonadversity, but there are several instances where this cannot be applied due to the limitations of preclinical studies. In our experience, lung lesions, even when of greater severity, may cause no apparent in-life clinical signs detectable in a general toxicology study or changes in clinical pathology parameters. Renal lesions and lesions of the central nervous system, in our opinion, often do not result in in-life functional deficiencies, or alterations in clinical pathology, until marked. This may be due to the lack of sensitivity of the tests employed, or to the high reserve capacities of organs involved.
Controversial (Difficult to Classify) Changes
In this section, we are deviating from the position papers of the STP and ESTP. There are a few examples where we cannot clearly assign adversity or nonadversity, and hence, it may be necessary to describe some lesions as “unclassifiable.”
Vehicle-related vacuolation of ependymal lining cells, noted in animals administered PEGylated compounds is, in our opinion, difficult to classify (Figure 22) as vacuolation of this nature often does not reverse even over long recovery periods, so the long-term biological implications are unclear. However, if one follows the definition of lack of evidence of harm and functional compromise within the study as suggested by STP, along with what has been described in the literature as an adaptive rather than a toxic response, 11,12 then one might make a nonadversity call for this finding. In our experience, inflammatory cell infiltration in the lungs with administration of novel compound classes by inhalation might also fall into this new category if the mechanism is not clear.
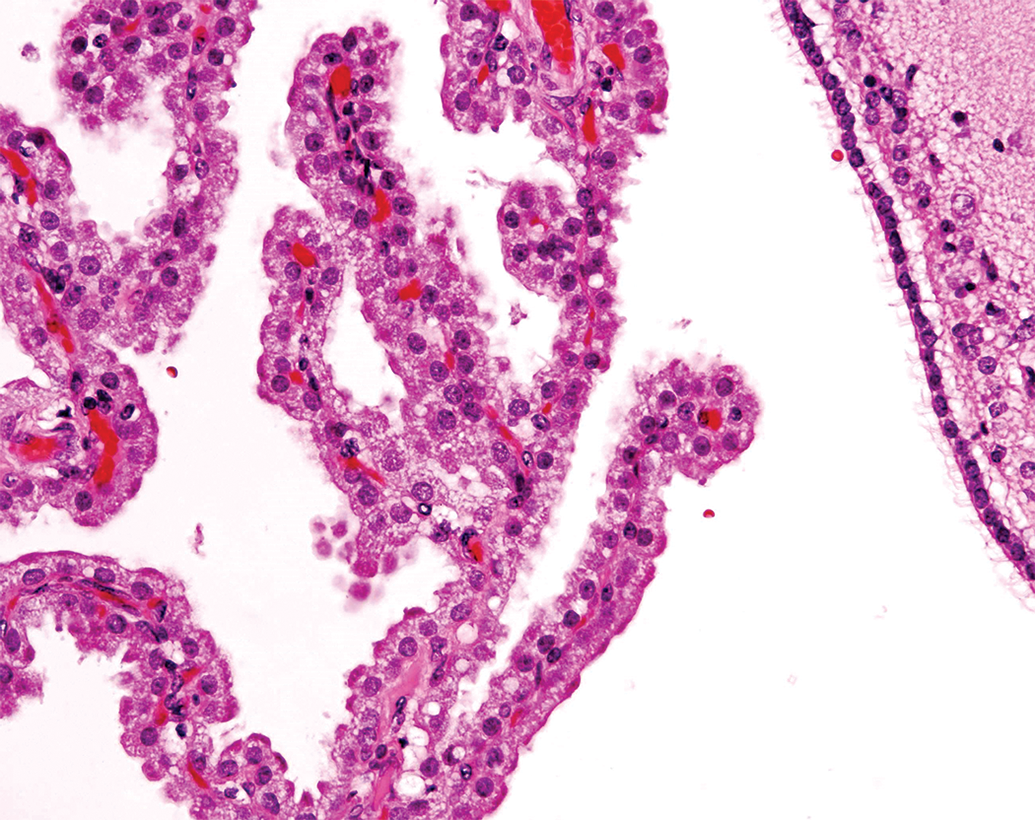
Vacuolation, choroid plexus, cynomolgus monkey. H&E.
Summary and Conclusions
In this opinion piece, we have suggested some categories and principles for designation of adversity and nonadversity of histopathological findings in acute and chronic toxicity studies, with a few divergences from the best practice guidelines, and have highlighted some of the associated challenges for the toxicologic pathologist.
In general, adversity classification of test item–related findings is often not clear-cut and depends on a number of factors. Many of these originate outside of anatomic toxicologic pathology and include in-life data, clinical pathology, and other end points from the entire toxicity study. Therefore, a weight-of-evidence approach must be utilized for adversity determination as recommended by STP best practice guidelines. While this paper expresses the authors’ opinions, every study has to be addressed on a case-by-case basis, as what is relevant to one study might not apply to another. In our opinion, occasional novel findings may be difficult to classify, and we are proposing to use “not classified” as a description for some novel lesions. In conclusion, determination of adversity in anatomic pathology subreport should be judged only in the context of the specific toxicity study involved and requires an integrated and overall weight-of-evidence approach.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
