Abstract
Microchip (passive radio-frequency identification device) implantation is a common and widely employed means of animal identification in laboratory animal facilities. However, these devices have been associated with tumors of the skin and subcutis in rodents. While microchip-associated tumors are rare, they pose a challenge for accurate diagnosis and documentation in preclinical toxicity studies. Documentation of these tumors should differentiate microchip-associated lesions with spontaneously occurring or test article–induced tumors. Standardizing criteria for microchip-associated lesions will aid the diagnostic process and allow for preclinical regulatory standardization. To this end, the Registry of Industrial Toxicology Animal-data have developed clear recommendations for diagnosis and documentation of microchip-associated lesions.
Keywords
Summary of Major Recommendations
Recording and histologic examination of all macroscopic abnormalities, including those suspected to be microchip-associated; recording of macroscopic observations under the organs “skin” or “subcutis,” with a descriptive entry (e.g., “with microchip”) either as a free text entry or modifier; recording of histologic observations under the organ “implantation site” if histopathology characterizes mesenchymal histogenesis for the tumor macroscopically marked with microchip; macroscopic–microscopic correlation across organs; and utilization of tumor nomenclature as published by the International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) working groups for tumors in rats and mice.
Aim
Registry of Industrial Toxicology Animal-data (RITA) is a pathology database for the collection of validated histopathological data on spontaneous tumors and preneoplastic lesions of control animals. Data generated for this database thus serve to enhance interlaboratory harmonization of applied terminology (Bahnemann et al. 1995; Kittel et al. 2004; Morawietz et al. 2004; Ruehl-Fehlert et al. 2003) for use in preclinical toxicity studies. This RITA publication comprises a literature review on microchip (passive radio-frequency identification device) implantation devices and microchip-associated tumors, as well as a statement on the current, often inconsistent, practice of sampling and recording of microchip-associated tumors. Recommendations for improved sampling and recording are provided subsequently. With these recommendations, RITA aims to enhance interlaboratory consistency and comparability of histopathological results of (regulatory) preclinical toxicity studies. These recommendations may explicitly serve as guidance to avoid confounding the incidence of test article–induced or incidental background lesions with device-related subcutaneous soft tissue proliferations. Moreover, with the aid of these recommendations, regulators’ insight into histopathological results of preclinical toxicity studies will benefit from the straightforward decision and recording process. Further, consistent recording of microchip-associated tumors, although not of particular interest in preclinical toxicity studies, will ensure that the scientific community keeps track of the lesions, which may be subject to drift in particular strains of rodents or particular test article–related situations.
Resources of Information
The
With the objective of providing information about the current practice of sampling and recording of microchip-associated tumors, an
Background
To the best of the authors’ knowledge, no published recommendations for microchip implantation sites exist for laboratory rodents. The World Small Animal Veterinary Association (WSAVA) has published recommendations for companion, zoo, and wildlife animals. Their guidelines state that if a species is not explicitly mentioned and its adult length is <17 cm from the spinal cord to the shoulder blades (i.e., rats and mice), microchips should be placed subcutaneously between the shoulder blades (WSAVA). In our experience, the most commonly used sites in laboratory rodents include the dorsal or lateral neck (primarily left side), shoulder, and thoracic regions.
When microchips are implanted into the subcutis of the aforementioned tissues, they elicit a foreign body response. Several long-term biocompatibility studies in rats and mice have shown that tissue reactions consist of a fibrous capsule that envelops the microchip with an associated minimal inflammatory response (Ball et al. 1991; Rao and Edmondson 1990; Sacco 1992). However, these biocompatibility studies did not characterize neoplastic changes associated with the transponder. Since the 1980s, microchip transponder identification has increasingly been used in preclinical toxicity studies, which has corresponded with increased observation of mesenchymal tumor development at implantation sites (Albrecht 2010).
Microchip-associated Tumors: Historical Data from Primary Literature
Several studies as well as retrospective analyses were published characterizing spontaneous microchip-associated tumors in rats and mice (Table 1). However, some terminology used in the studies listed in Table 1 is no longer recommended by current INHAND nomenclature of soft tissue tumors in rats and mice (see Current Nomenclature of Soft Tissue Tumors section). All
Frequency and Type of Microchip-associated Tumors in Various Strains of Laboratory Rodents—Literature Data.

a Terms no longer recommended by the current nomenclature. NOS = not otherwise specified.
Table 1 clearly depicts a gender independent higher incidence in genetically engineered p53+/− mice, compared to conventional rodent strains. Blanchard and colleagues (1999) argue that, although component materials of the microchip transponders are assumed to be free of genotoxic potential, p53+/− mice have a higher risk of tumor development at microchip implantation sites. This may be due to leachates or energy from the signal transmitted by the transponder (Blanchard et al. 1999).
It has been tried to characterize microchip-associated tumors with a variety of immunohistochemical (IHC) markers for mesenchymal-origin cells. IHC studies can use antibodies against α-smooth muscle actin (α-SMA), α-sarcomeric actin, desmin, and myogenin to confirm that microchip-induced sarcomas are not of myogenic origin (Elcock et al. 2001; Le Calvez, Perron-Lepage, and Burnett 2006). Le Calvez, Perron-Lepage, and Burnett (2006) observed desmin and α-SMA staining of a large proportion of tumor cells not considered to be of myogenic origin by routine histology (Le Calvez, Perron-Lepage, and Burnett 2006). Elcock and colleagues (2001), on the other hand, observed neither desmin nor α-SMA staining but did observe consistent vimentin staining in microchip-induced sarcomas (Elcock et al. 2001). Vimentin staining was also seen in two studies of microchip-induced sarcomas in domestic animals (Carminato et al. 2011; Vascellari, Melchiotti, and Mutinelli 2006). Only one IHC study of a microchip-induced sarcoma in a house musk shrew (
Current Nomenclature of Soft Tissue Tumors
Table 2 lists tumors of soft tissue, skeletal muscle, and mesothelium in rats and mice that are recognized by INHAND nomenclature (Greaves et al. 2013). Detailed descriptions of all neoplasms listed in Table 2 are beyond the scope of this article. For this purpose, study pathologists should refer to INHAND nomenclature or the goRENI website (goRENI; Greaves et al. 2013).
INHAND Nomenclature of Tumors of Soft Tissue, Skeletal Muscle, and Mesothelium in Rats and Mice.

aNo fully substantiated report on rhabdomyomas in laboratory rodents available. NOS = not otherwise specified.
Some diagnostic terms used in the studies listed in Table 1 are no longer valid when applying INHAND nomenclature including malignant fibrous histiocytoma, benign fibrous histiocytoma, and hemangiopericytoma. Historically, malignant fibrous histiocytomas were considered to be a group of largely undifferentiated or primitive sarcomas. Today, neither INHAND nor World Health Organization (WHO) classification of human tumors recommends use of this term. While WHO nomenclature subdivides these tumors according to their IHC and electron microscopic characteristics, INHAND criteria recommend the term “fibrosarcoma, pleomorphic” (Fletcher 2006; Fletcher et al. 2006; Greaves et al. 2013). Pleomorphic fibrosarcoma is the most common tumor type to develop in rats and mice in response to subcutaneous implants (Brand, Johnson, and Buoen 1976; Kirkpatrick et al. 2000), including microchip implants (Nolte et al. 2006; Palmer, Palazzolo, and Ryan 1998; Tillmann et al. 1997). According to INHAND recommendations, the term “fibrosarcoma, pleomorphic” should be used for tumors that exhibit a variable histologic pattern—ranging from a storiform pattern with uniform plump spindle cells or small rounded cells to a highly pleomorphic pattern of bizarre spindle cells and tumor giant cells—but still demonstrate fibrous tissue features such as interwoven bundles of collagen (goRENI; Greaves et al. 2013). When composed of sheets of undifferentiated round, spindle, or pleomorphic cells that cannot be attributed to specific tissue histogenesis, the term “sarcoma, not otherwise specified” should be used.
The term “benign fibrous histiocytoma” should no longer be employed; instead, these masses should be defined with “fibroma” (goRENI; Greaves et al. 2013). In addition, hemangiopericytomas are no longer included in current INHAND nomenclature. These lesions are now categorized with the terms listed in Table 2. For human tumors, it is now generally accepted and recommended to use “solitary fibrous tumor” for extrameningeal fibrous proliferations resembling the tumors formerly termed hemangiopericytomas (Fletcher et al. 2006; Fletcher 2006).
Current Practice of Sampling and Recording
An informal survey of RITA member laboratories regarding processes for sampling and recording of microchip-associated tumors emphasized the need for consistent procedures and definite criteria that can be filtered and searched for. Laboratories use varied approaches and do not use a standardized decision tree. In general, two major diagnostic approaches are used at the time of necropsy: (1) macroscopic documentation of microchip-associated tumor with other skin tumors or (2) macroscopic documentation of tumor separately from skin tumors. Some laboratories pursue histologic characterization of microchip-associated tumors and correlate histology to macroscopic findings. Alternatively, other laboratories do not pursue further histologic assessment when a microchip is associated with the mass at the time of necropsy. Macroscopic documentation of microchip-associated tumors with other skin masses has the potential to confound incidences of spontaneous, test article–induced, and microchip-associated tumors.
Recommendations for Sampling and Recording
RITA recommends standardization for macroscopic and microscopic assessment of microchip-associated tumors in a manner that allows harmonization with future Standard for Exchange of Nonclinical Data (SEND) recommendations. Specific recommendations for sampling approaches and the rationale for these approaches are outlined below.
When observed, macroscopic records should indicate the presence of microchip-associated lesions under the organs skin or subcutis. Descriptive free text should be added to annotate with microchip if the transponder is detected within the tumor. Detection can be achieved by excising the mass from surrounding skin and soft tissue, with subsequent verification of the embedded transponder with the transponder reader. Alternatively, the tumor may be serially sectioned to identify the microchip and record the microchip number. Then, either the transponder itself or an elongated cavity must be demonstrated (Figures 1 and 2). If the microchip is identified adjacent to the mass but macroscopically not clearly embedded within or otherwise directly adjoining the mass, the free text with microchip should also be used. Not only is this important for laboratory personal to ensure exact histotechnical processing of the lesion. During histopathological examination, physical association may nevertheless be observed. Free text annotations may become obsolete with implementation of SEND; consequently, introduction of the modifier with microchip would become essential.
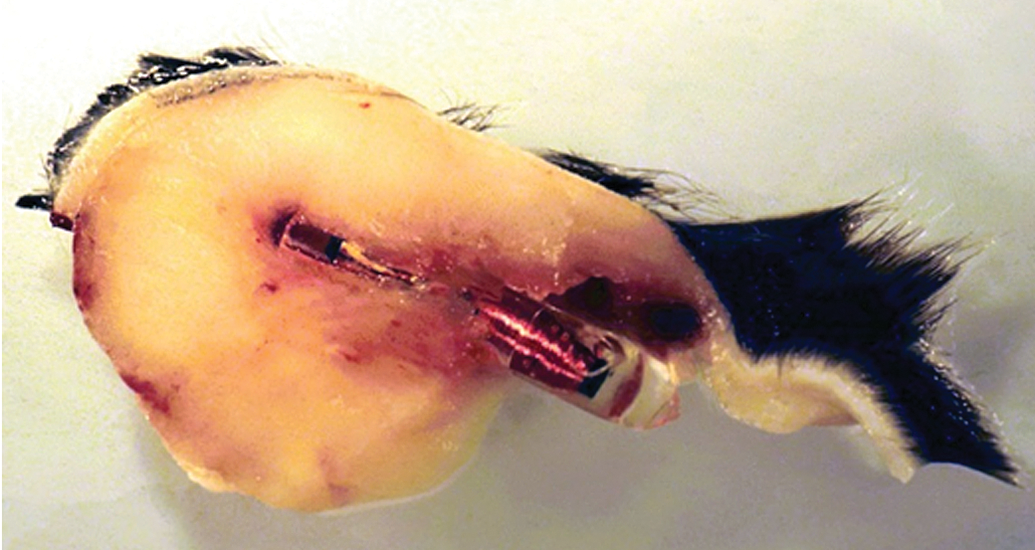
Subcutis, mouse. Microchip-associated soft tissue mass demonstrating a microchip centrally embedded within. Macroscopically, this mass should be described under “subcutis” with the free text “with microchip.”
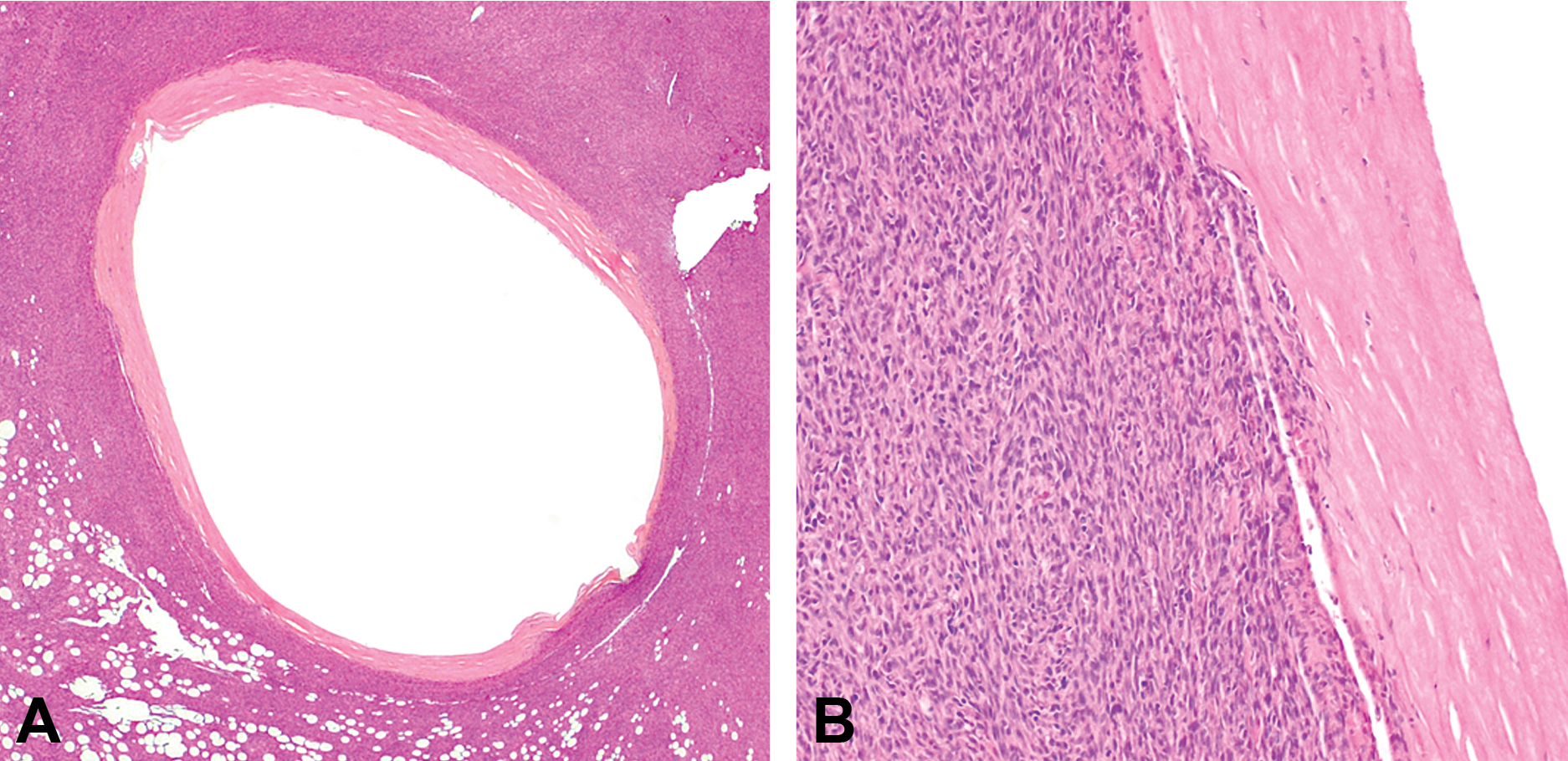
Subcutis, rat. Hematoxylin and eosin–stained tissue sections of a microchip-associated fibrosarcoma, with the oval transponder cavity completely covered by a fibrous capsule and surrounding tumor tissue. Tumor cells demonstrate typical features of a fibrosarcoma—spindle-shaped, arranged in a herringbone pattern, and invading the fibrous capsule. Microscopically, the diagnosis would be recorded under “implantation site” with macroscopic–microscopic correlation across organs. Hematoxylin and eosin stain.
All abnormal tissues, including microchip-associated tumors, should be sampled for routine histologic examination, and the microchip should be removed. These tissues should undergo standard fixation, processing, and staining with hematoxylin and eosin. In cases of special interest, the microchip may be left in place and undergo embedding with resins (e.g., SPURR or methyl methacrylate) that require specialized sectioning. Further, abnormal tissues should be evaluated by a study pathologist. When a lesion (denoted macroscopically as with microchip) is characterized as a mesenchymal proliferation at necropsy, it should be recorded under the organ implantation site in order to avoid interference with incidences of incidental or test article–related soft tissue proliferations. The organ implantation site is usually not of main interest in studies focusing on systemic exposure to a test article. In the case of a mesenchymal proliferation that had macroscopically been observed with microchip, it may thus be necessary to add the extra organ implantation site to the organ finding table within the data capture system. Consequently, this requires this organ to be contained by or introduced into the glossary of available organs. Institutions working on the development of medical implants should define implantation sites as different organs, for example, implantation site 1 could be the site of microchip implantation, implantation sites 2, and so on, the sites of test article implantation.
RITA also urgently emphasizes the use of preferred terminology of INHAND working groups, which have published internationally established tumor nomenclature for rodents (see Current Nomenclature of Soft Tissue Tumors section).
Finally, the macroscopic–microscopic correlation must be made across organs to the macroscopic record under skin or subcutis.
The rationale for associating a mesenchymal mass denoted as with microchip at necropsy under the microscopic implantation site is based on published reports of tumor histogenesis. The macroscopic location of these tumors at necropsy (e.g., tumor located in neck and shoulder area of the animal) or degree of association with a microchip (e.g., centrally embedded, peripherally associated, adjacent but macroscopically not clearly attached) may confer diagnostic ambiguity, especially if the tumors do not undergo histologic evaluation. Currently, there is no evidence that tumors induced by transponder implants exhibit any other histogenic lineage other than cells of mesenchymal origin (Elcock et al. 2001; Le Calvez, Perron-Lepage, and Burnett 2006). Further, correlation of a macroscopic observation of a microchip-associated tumor cannot clearly be made without the knowledge of tumor cell histogenesis.
While this study focuses on rodents, the aforementioned recommendations may also be employed in unlikely cases of microchip-associated tumors in the nonrodent species used in preclinical toxicity studies.
Conclusions
Microchip transponder implantation is increasingly used in preclinical safety studies in rodents. Microchip-associated tumors have been documented in both rats and mice, although there have been modifications in accepted nomenclature for the field. Macroscopic and microscopic approaches to assess these masses vary widely among institutions. Here, RITA has proposed best practice approaches for documentation, assessment, and characterization of microchip-associated tumors in preclinical safety studies. We recommend that all masses be documented under the appropriate organ system (macroscopy: skin/subcutis; microscopy: implantation site), evaluated histologically, and appropriately characterized by the use of INHAND nomenclature. A correlation between macroscopic and microscopic findings should then be made across organs.
Footnotes
Authors’ Note
Registry of Industrial Toxicological Animal-date (RITA). Members: AbbVie, BASF SE, Bayer AG, Boehringer Ingelheim Pharma GmbH & Co. KG, F. Hoffmann-La Roche Ltd, Merck KGaA, Novartis Pharma AG, Novo Nordisk A/S, Sanofi, Syngenta Ltd, and Takeda Pharmaceutical Company Ltd.
Acknowledgments
The authors acknowledge gratefully Dr. Thomas Tillmann for the provision of the macroscopic photography of a microchip-associated lesion and Elizabeth Clark for her thorough revision and excellent conceptual suggestions.
Author Contributions
Authors contributed to conception or design (SL, FC, HE, RK, TN, MR); data acquisition, analysis, or interpretation (SL, AB, FC, HE, RK, TN, MR); drafting the manuscript (SL); and critically revising the manuscript (AB, FC, HE, RK, TN, MR). All authors gave final approval, and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
All author(s) declare no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
