Abstract
Tanezumab, an anti-nerve growth factor (NGF) antibody, is in development for management of chronic pain. During clinical trials of anti-NGF antibodies, some patients reported unexpected adverse events requiring total joint replacements, resulting in a partial clinical hold on all NGF inhibitors. Three nonclinical toxicology studies were conducted to evaluate the effects of tanezumab or the murine precursor muMab911 on selected bone and joint endpoints and biomarkers in cynomolgus monkeys, Sprague-Dawley rats, and C57BL/6 mice. Joint and bone endpoints included histology, immunohistochemistry, microcomputed tomography (mCT) imaging, and serum biomarkers of bone physiology. Responses of bone endpoints to tanezumab were evaluated in monkeys at 4 to 30 mg/kg/week for 26 weeks and in rats at 0.2 to 10 mg/kg twice weekly for 28 days. The effects of muMab911 at 10 mg/kg/week for 12 weeks on selected bone endpoints were determined in mice. Tanezumab and muMab911 had no adverse effects on any bone or joint parameter. There were no test article–related effects on bone or joint histology, immunohistochemistry, or structure. Reversible, higher osteocalcin concentrations occurred only in the rat study. No deleterious effects were observed in joints or bones in monkeys, rats, or mice administered high doses of tanezumab or muMab911.
Nerve growth factor (NGF) is essential for neuronal survival during development and is a key modulator of nociception and pain signaling in adults (Mantyh et al. 2011). NGF administration induces increased mechanical and thermal hyperalgesia, allodynia, hypersensitivity, and generalized muscle pain, whereas blockade of NGF function decreases pain behaviors (Apfel 2002; Apfel et al. 2000; Dyck et al. 1997; Koewler et al. 2007; Sabsovich et al. 2008; Sevcik et al. 2005; Shelton et al. 2005).
Tanezumab, a humanized monoclonal antibody, binds NGF and prevents its interaction with the high-affinity transmembrane tyrosine kinase receptor (TrkA) and the low-affinity NGF receptor (p75). Our understanding of signaling through these two receptors has steadily grown in complexity, with many diverging and converging intracellular pathways, and it has become clear that the specifics of this signaling depend heavily on the cellular context in which the receptors are expressed (Denk, Bennett, and McMahon 2017). Tanezumab does not, however, directly affect either receptor (Abdiche, Malashock, and Pons 2008). In clinical trials of patients with chronic pain, tanezumab reduced pain and improved function (Balanescu et al. 2014; Brown et al. 2012; Brown et al. 2013; Ekman et al. 2014; Katz et al. 2011; Kivitz et al. 2013; Lane et al. 2010; Schnitzer et al. 2015; Spierings et al. 2013). However, during the conduct of tanezumab clinical trials, adverse events initially described as osteonecrosis that required total joint replacement led the U.S. Food and Drug Administration (FDA) to place all NGF inhibitors on partial clinical hold (FDA 2012, 2016; Pfizer 2012). This increased frequency of joint-related adverse events was unexpected, as no issues with bone or joints were seen in previous nonclinical studies (including a 6-month toxicity study in adolescent/young adult nonhuman primates) evaluating the effects of anti-NGF agents using multiples of the clinical dose (Zorbas et al. 2011). A blinded independent adjudication committee reviewed all reported cases of osteonecrosis, as well as cases of total joint replacements for which radiographs obtained within 9 months of surgery were available, and determined that rapid progression of osteoarthritis (RPOA), rather than osteonecrosis, was the underlying issue (Hochberg et al. 2016; Pfizer 2012). In 2012, the arthritis advisory committee voted overwhelmingly to continue clinical trials with added risk mitigation procedures (https://wayback.archive-it.org/7993/20170404145629/https://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/ArthritisAdvisoryCommittee/UCM307879.pdf) and the FDA lifted the partial clinical hold on tanezumab, allowing the resumption of clinical studies for chronic pain.
As part of a continuing investigation of tanezumab safety and in an attempt to further elucidate clinical bone and joint findings, three nonclinical studies investigated the effects of tanezumab or its murine precursor (muMab911) in three species commonly used in nonclinical toxicology studies (cynomolgus monkeys, Sprague-Dawley [SD] rats, and C57BL/6 mice). There are no validated animal models of RPOA; all animals used in the studies reported here were normal. The data produced are still relevant because anti-NGF agents are being used in clinical trials for indications other than osteoarthritis in patient populations with normal joints such as cancer pain and lower back pain. We used histologic and radiologic techniques and measured serum biomarkers of bone metabolism to assess whether tanezumab affects intrinsic properties of bone.
Materials and Method
Animal care conformed to the Guide for the Care and Use of Laboratory Animals (National Research Council 1996) with oversight by local Institutional Animal Care and Use committees. Monkeys were individually housed. The targeted range of temperature and relative humidity were between 18 and 29°C and 30 and 70%, respectively, with a 12-hr diurnal light cycle. Fresh drinking water was provided ad libitum and monkeys were fed twice daily with Purina Mills, Inc. (PMI) LabDiet® Laboratory Fiber-Plus® biscuits along with produce treats and access to enrichment toys. Rats were housed individually in polycarbonate cages with Sani Chip bedding and offered Certified Rodent Diet #2016C (Harlan Laboratories, Inc.) ad libitum. Water was offered ad libitum. Environmental controls for the animal room were set to maintain 18 to 26°C, a relative humidity of 30 to 70%, a minimum of 10 room air changes/hour, and a 12-hr light/12-hrdark cycle. Mice were housed 5/cage in polycarbonate cages with Anderson’s Bed O’Cob bedding and offered Purina Lab Diet 5053 ad libitum. Water was offered ad libitum.
Study 1 (Monkey Study)
Purpose-bred, treatment-naive, sexually mature male (Covance Research Products Inc., Alice, TX, USA) and female (Shin Nippon Biomedical Labs [SNBL, USA] Scientific Resource Center, Alice, TX, USA) cynomolgus monkeys were administered tanezumab 0 mg/kg/week subcutaneous (sc) (control), 4 mg/kg/week intravenous (iv), 30 mg/kg/week iv, or 30 mg/kg/week sc injections once weekly for 26 weeks (dosing period; Online Supplemental Table S1). Previous toxicity studies with tanezumab have been conducted using either sc or iv routes of administration, and the vehicles used were identical to those in the present study; therefore, it was decided that one control group was sufficient. This also had the benefit of reducing the number of primates used in this study. The sc route has a similar toxicity profile compared with the iv route, and a head-to-head comparison of these two routes at 30 mg/kg/week was of interest. Intravenous injections were administered into the cephalic or saphenous vein; sc injections were administered in the lower back. Reversibility of any changes observed during dosing was evaluated in an 8-week recovery period. At study initiation, male monkeys were aged 4.5 to 9.8 years and weighed 4.4 to 10.0 kg; females were aged 5.3 to 6.3 years and weighed 2.8 to 4.2 kg.
At necropsy, whole-body perfusion with 4% paraformaldehyde was conducted, gross observations and organ weights were recorded, and selected tissues were collected. Statistical analysis is found in Online Supplemental Text S1.
Study 2 (Rat Study)
Male and female SD rats (Charles River Laboratories, Portage, MI, USA) were aged at least 6 weeks but less than 8 weeks and weighed 100 to 350 g at dosing initiation. Rats were administered tanezumab 0 mg/kg/dose (control), 0.2 mg/kg/dose (low dose), 1 mg/kg/dose (medium dose), or 10 mg/kg/dose (high dose) twice weekly via iv injection into a tail vein for at least 28 days (total of nine doses). Reversibility, persistence, and delayed occurrence of any effects were evaluated in up to 10 animals following an 8-week recovery period (Online Supplemental Table S1). Tissue was collected at necropsy following whole-body perfusion (standard and neural tissues) with 4% methanol-free formaldehyde or immersion fixation in 10% neutral buffered formalin (bone and standard tissues). Statistical analysis is found in Online Supplemental Text S2.
Study 3 (Mouse Study)
Female C57BL/6 mice (Jackson Laboratories, Bar Harbor, ME, USA) were aged approximately 12 weeks and weighed approximately 19 g at dose initiation (Online Supplemental Table S1). Animals were administered 10 mg/kg muMab911 or mouse immunoglobulin G1 (control; 1 mg/mL) weekly via sc injection (in the nape of the neck) for 12 weeks. Both hind limbs were collected at necropsy following whole-body perfusion and were stored in 4% paraformaldehyde at 4°C prior to dissection of hip and stifle joints prior to decalcification.
Analyses
Bone biomarker determination (studies 1 and 2 only)
Serum samples were collected twice prior to first dose in study 1, on days 85 and 169 of the dosing phase, and during recovery on days 223 and 218 (males and females, respectively). Study 2 samples were collected on day 30 of the dosing phase and day 57 of the recovery phase. Samples were analyzed for bone biomarkers osteocalcin, carboxy-terminal telopeptide fragment of type 1 collagen (CTX1), and type 1 collagen N-terminal propeptide (P1NP; Online Supplemental Text S3).
Toxicokinetics (TK) and antidrug antibodies (ADAs; studies 1 and 2 only)
Plasma concentrations of tanezumab and serum concentrations of anti-tanezumab ADAs were determined from samples collected at predetermined times and measured using a validated enzyme linked immunosorbent assay (ELISA) at ICON Development Solutions (Whitesboro, NY, USA; Online Supplemental Text S4).
Bone histopathology
Bone histopathology examination was performed on decalcified sections of the hip joint (including femoral head, intertrochanteric bone, and acetabulum) and the stifle joint (distal femur and proximal tibia) stained with hematoxylin and eosin stain, toluidine blue, Masson’s trichrome, and immunohistochemistry for Factor VIII and proliferating cell nuclear antigen (PCNA; studies 1, 2, and 3) and on undecalcified sections of the contralateral femoral head and proximal tibia using modified Goldner’s trichrome (transmitted and polarized light) and toluidine blue (for studies 1 and 2). Ground sections of mandible were evaluated for study 1.
Toxicology
Toxicology evaluations for study 1 included clinical observations, food consumption, body weight, electrocardiogram, echocardiography, ophthalmology, blood pressure, rectal temperature, hematology, coagulation, serum chemistry, peripheral blood lymphocyte immunophenotyping, gross pathology, organ weights, and histopathology of bone and nonneural tissues/organs or reproductive parameters. Assessment of toxicity in study 2 was based on mortality, clinical signs, body weight, food consumption, ophthalmic findings, bone biomarker analysis, bone tissue histopathology, mCT, and clinical and anatomic pathology. Toxicology evaluations for study 3 included mortality, clinical signs, body weight, and bone tissue histopathology.
Bone imaging
Radiography and mCT were conducted on mandible, stifle, and hip joints (study 1; Online Supplemental Text S5); the distal femoral metaphysis for cancellous bone; and the femoral mid-diaphysis for cortical bone (study 2; Online Supplemental Text S6). Survey radiographs, including pelvis, hip, and stifle joints, were taken prior to dosing and prior to scheduled necropsy (study 1).
Results
Study 1 (Monkey Study)
Bone biomarkers
There were no tanezumab-related effects on serum bone biomarker parameters in the dosing or recovery phases (Online Supplemental Table S2).
TKs and ADA
TK parameters were similar for male and female monkeys within a dose group. There were no quantifiable concentrations of tanezumab in plasma or cerebrospinal fluid samples from control animals. Incidence of ADA by tanezumab dose was as follows: 4 mg/kg iv: 0% (n = 0/11); 30 mg/kg iv: 8% (n = 1/12); 30 mg/kg sc: 5% (n = 1/20); and control (sc): 65% (n = 13/20). Although plasma tanezumab concentrations were affected by ADA, group mean values for maximum drug concentration observed in plasma (C max), time at which C max was first observed (T max), and area under the plasma drug concentration–time curve (AUC) were not significantly altered, as there was only one ADA-positive animal each in the 30-mg/kg iv and sc groups. Systemic exposure on days 1, 22, 85, and 169 increased with tanezumab dose (Online Supplemental Table S3). After termination of dosing, quantifiable plasma tanezumab concentrations were observed in animals from the 30-mg/kg sc group through to on day 232 (last sampling day) except for the one ADA-positive animal whose last measurable tanezumab concentration was on day 85—48 hr postdose. Mean tanezumab concentration in this group (excluding the one ADA-positive animal) on day 232 (146 μg/mL) was approximately 14% of the concentration on day 169 (last treatment day) at 24-hr postdose (1030 μg/mL).
Bone histopathology
There were no tanezumab-related histopathologic findings in the hip joint, stifle joint, or mandible. Histopathologic evaluation revealed no evidence of osteonecrosis at any bone site examined (Figure 1). Findings of minimal-to-mild osteoarthritis were present in at least one joint in all monkeys regardless of treatment. These included articular cartilage fibrillation with occasional osteophytes and increased subchondral bone thickness, which were typical for the species, age, sex, and joint examined (Figure 2 and Table 1). There were no findings indicative of rapidly progressive osteoarthritis, nor were osteolysis or bone atrophy present in any of the examined tissues. Immunohistochemistry revealed no differences among treatment groups in vascularity (Factor VIII) or cell turnover (PCNA). Because there were no tanezumab-related bone histopathology findings in dosing-phase animals, bones from recovery-phase animals were not evaluated.
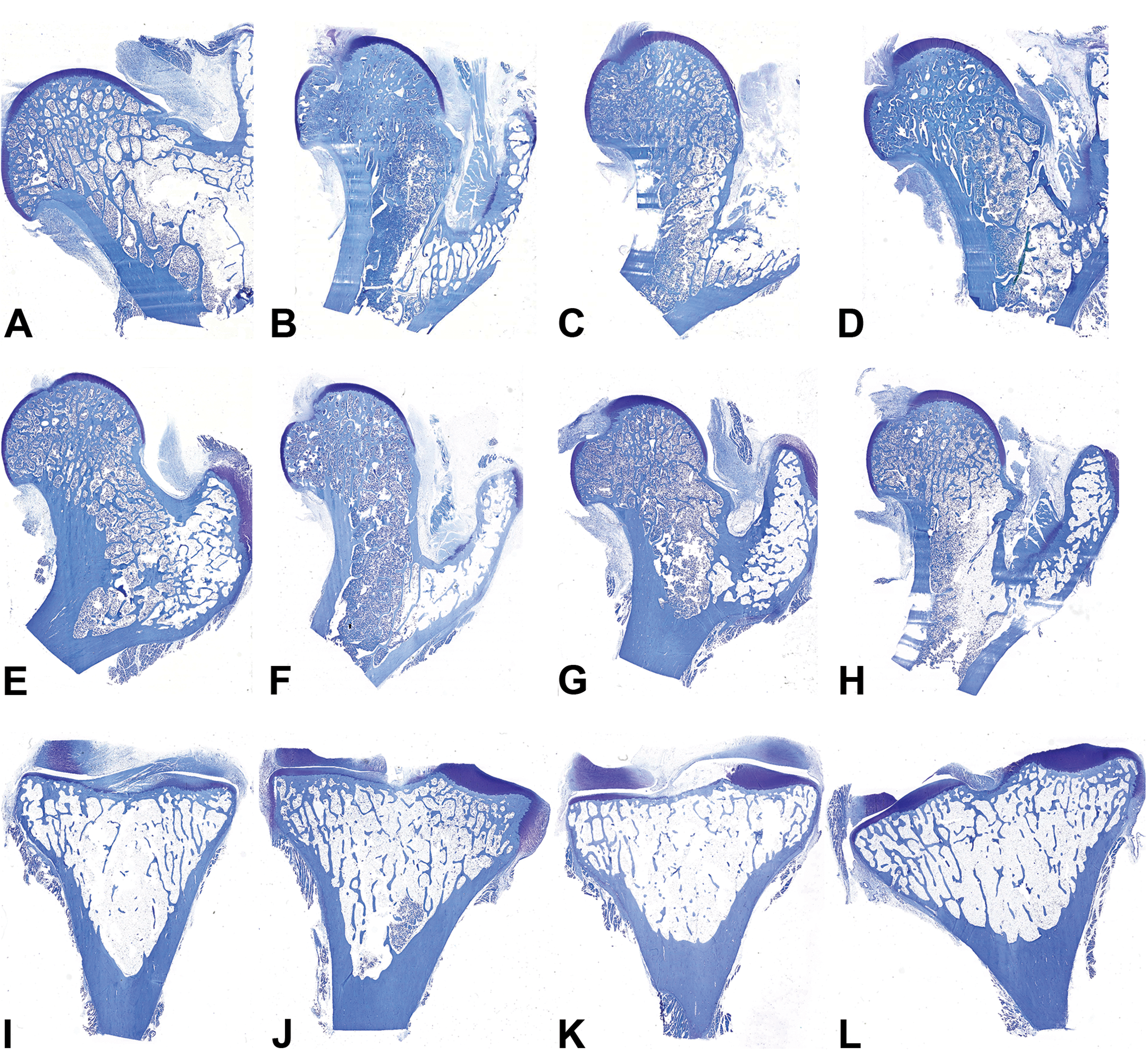
Representative proximal femur and proximal tibias from male and female monkeys. From left, control (0 mg/kg, sc); 4 mg/kg iv; 30 mg/kg iv; 30 mg/kg sc. Top row: male proximal femurs; middle row: female proximal femurs; bottom row: female proximal tibias. No evidence of osteonecrosis or joint destruction was observed in any sample. Decalcified bone, Toluidine blue stain.
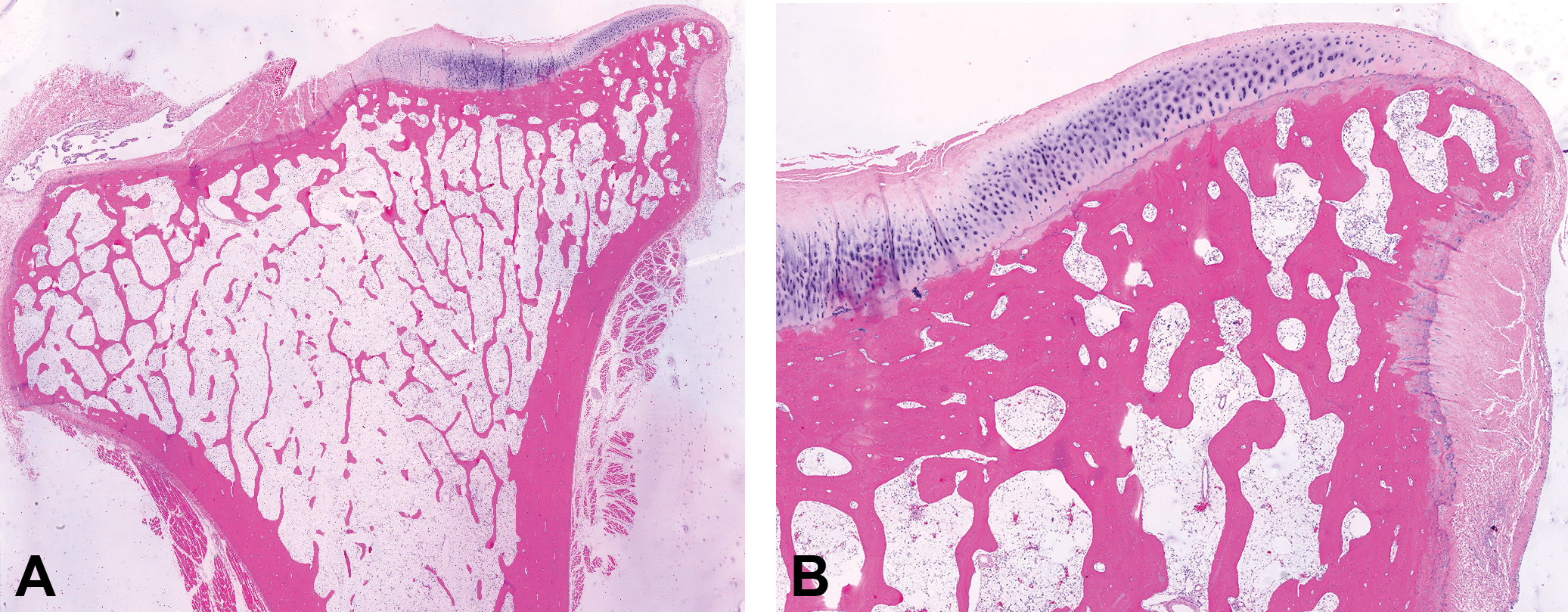
Early osteoarthritis in the proximal tibia from the oldest monkey in study 1 (aged 9.8 years at the initiation of dosing), which was dosed with 30 mg/kg/week of tanezumab subcutaneously. (A) Overview of entire proximal tibia with medial tibial plateau. (B) Medial tibial plateau demonstrating mild articular fibrillation, increased subchondral bone thickness, and abaxial osteophyte. All joint changes noted for this animal were typical for this species, sex, and age and were not related to administration of tanezumab. Decalcified bone, hematoxylin and eosin stain.
Incidence of Histopathology Findings for Right Proximal Tibia (Decalcified) from Study 1.
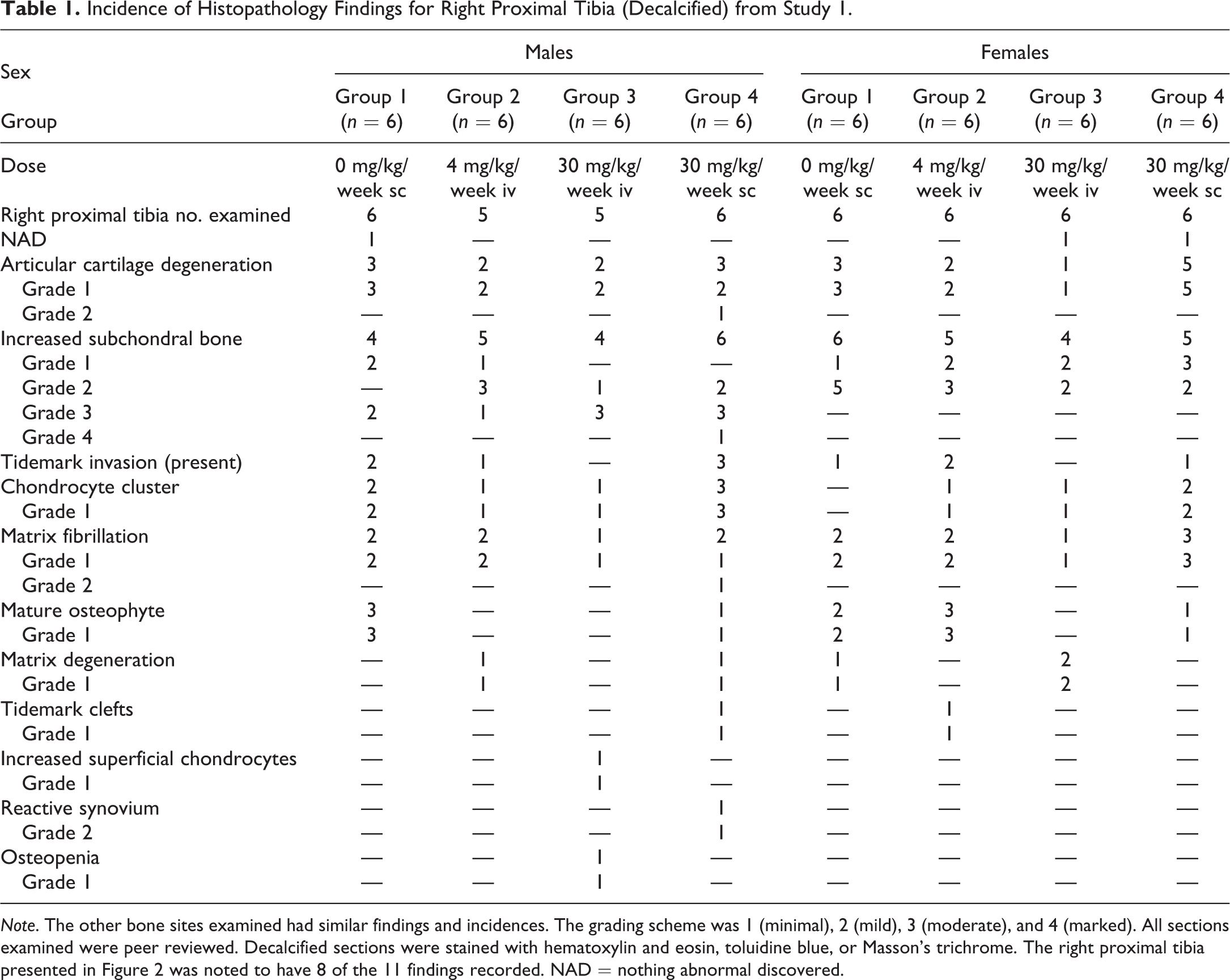
Note. The other bone sites examined had similar findings and incidences. The grading scheme was 1 (minimal), 2 (mild), 3 (moderate), and 4 (marked). All sections examined were peer reviewed. Decalcified sections were stained with hematoxylin and eosin, toluidine blue, or Masson’s trichrome. The right proximal tibia presented in Figure 2 was noted to have 8 of the 11 findings recorded. NAD = nothing abnormal discovered.
Toxicology
No adverse effects of tanezumab treatment were observed in clinical observations, food consumption, body weight, electrocardiogram, echocardiography, ophthalmology, blood pressure, rectal temperature, hematology, coagulation, serum chemistry, peripheral blood lymphocyte immunophenotyping, gross pathology, organ weights, histopathology of nonneural tissues/organs, or reproductive parameters (Palazzi et al. 2016). Three animals were euthanized early due to clinical signs unrelated to tanezumab administration (Online Supplemental Text S7).
Imaging, mCT
Radiography revealed that 11 of 22 male monkeys exhibited completely or partially open growth plates at the proximal tibial epiphyses (Online Supplemental Figure S1). Incomplete growth plate closure was observed in three of five male monkeys in the 4-mg/kg iv group, one of five in the 30-mg/kg iv group, four of six in the 30-mg/kg sc group, and three of six controls. All female monkeys had closed growth plates at the proximal tibial epiphysis and femoral head. Radiography revealed no bone deformities or fractures and there were no tanezumab-related findings noted on in vivo survey radiographs or ex vivo radiography.
mCT revealed that the amount of cancellous bone (bone volume) at the femoral head and neck was similar in all male and female monkeys regardless of treatment (Figure 3). Average trabecular number and trabecular thickness at the femoral head and neck were similar in all monkeys, and variations were within physiological limits and did not reach statistical significance. Also, the average trabecular number and trabecular thickness of the mandible below the second molar were similar in all monkeys and variations were within physiological limits (Online Supplemental Figure S2).
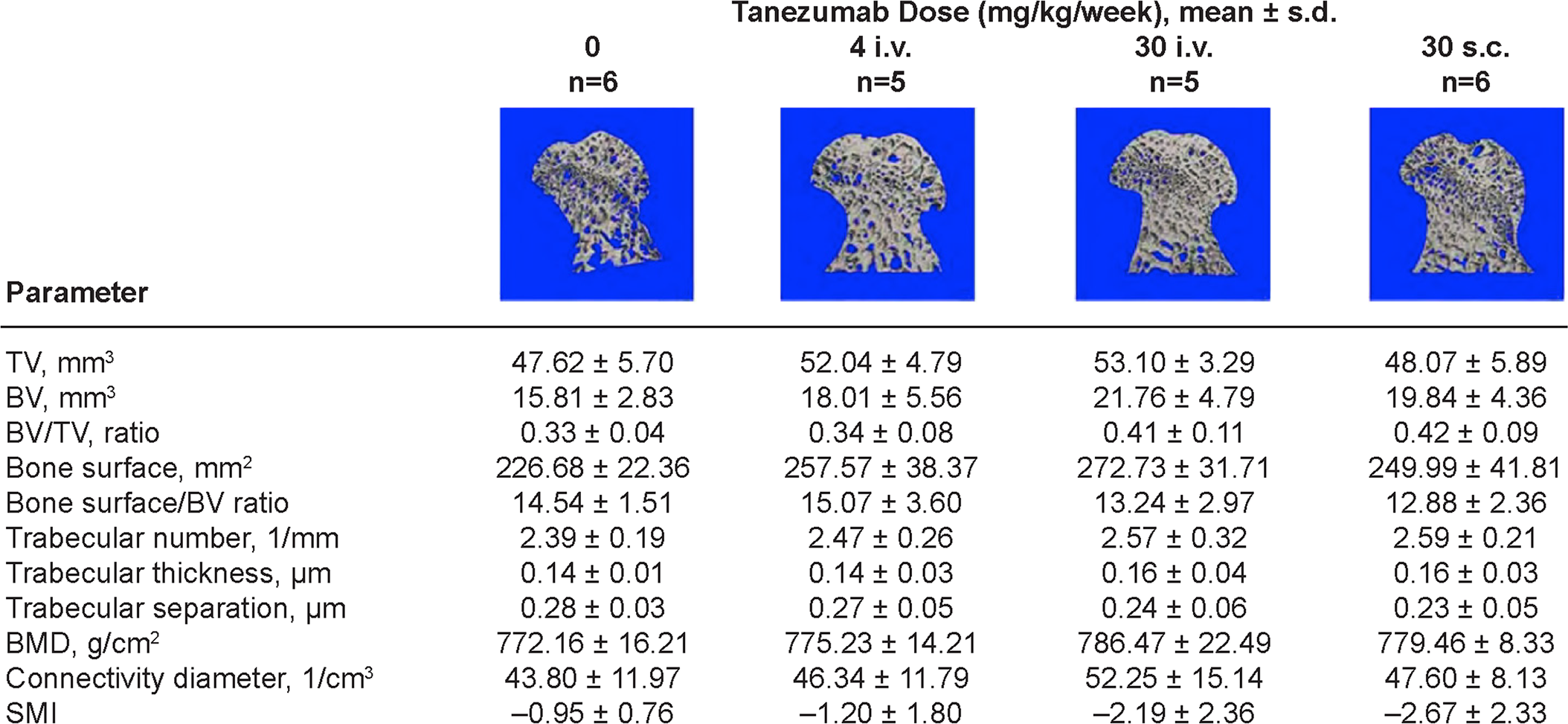
Three-dimensional mCT results and representative images of the femoral head and neck assessing cancellous bone mass and structure in male monkeys. BMD, bone mineral density; BV, bone volume; mCT, microcomputed tomography; s.d., standard deviation; SMI, structural model index; TV, tissue volume.
Bone mineral density (BMD) was also similar among animals, and there were no tanezumab-related effects. Further, BMD of the cortical bone, bone volume, and cortical bone thickness were similar among females and similar among males. There were no differences in polar moment of inertia among males or among females (data not shown). There were also no differences between dose groups in any cancellous bone parameters at the proximal tibial metaphysis (Online Supplemental Figure S1). Similarly, cancellous bone parameters at the proximal tibial epiphysis showed similar values in all dose groups in both sexes (data not shown). There were no significant differences between groups in any of the structural cancellous bone parameters. There were also no statistically significant differences in structural parameters or BMD values between the medial and lateral subchondral regions within the same dose group or between control and treated monkeys (Online Supplemental Figure S1).
Study 2 (Rat Study)
Bone biomarkers
There were tanezumab-related higher mean serum osteocalcin concentrations in the rats. Males (37–46% from mean control) and females (10–25% from mean control) had higher osteocalcin at ≥0.2 mg/kg, which reversed by the end of the 8-week recovery phase. No dose–response relationship was found (Table 2). There were no other tanezumab-related differences in any other bone biomarker measured (P1NP, CTx) or in total alkaline phosphatase (ALP).
Rat Biomarker Data.
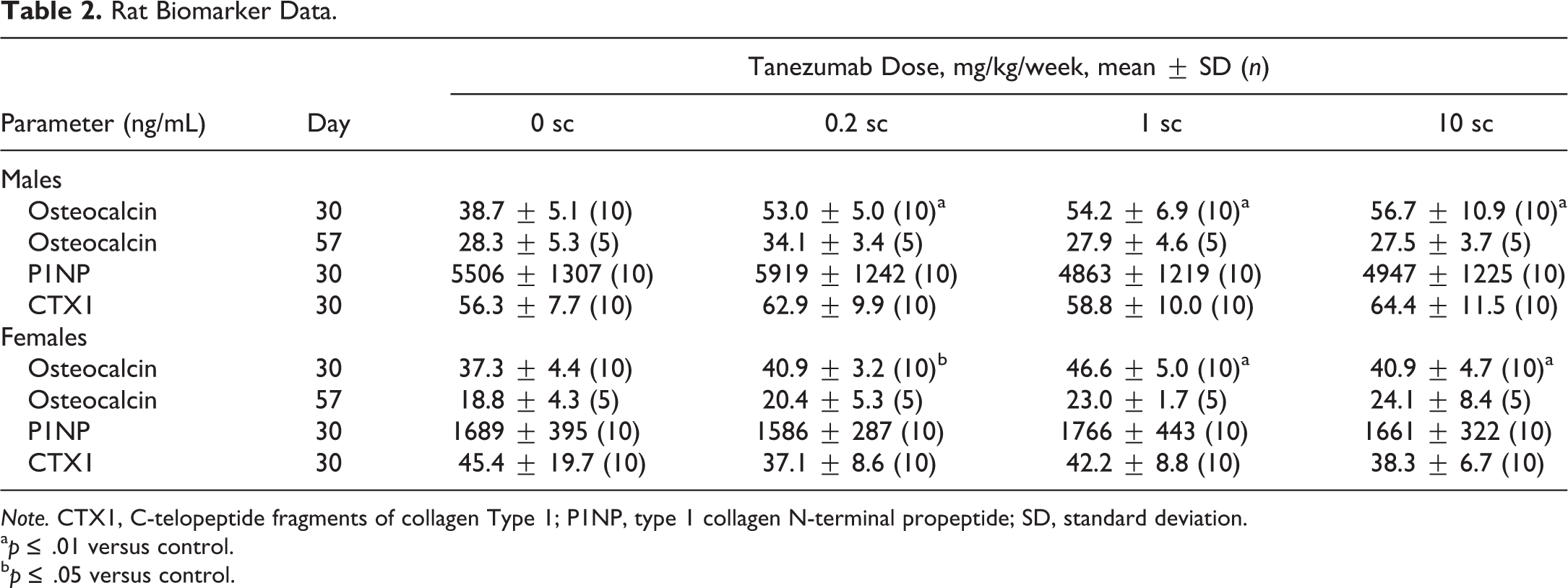
Note. CTX1, C-telopeptide fragments of collagen Type 1; P1NP, type 1 collagen N-terminal propeptide; SD, standard deviation.
a p ≤ .01 versus control.
b p ≤ .05 versus control.
TKs and ADA
Tanezumab exposure was similar for male and female rats within a dose group. Mean T max was between 1.0- and 6.8-hr postdose for all groups in the satellite TK animals (Online Supplemental Table S4; Online Supplemental Text S8). The ADA results are also presented in Online Supplemental Text S8.
Bone histopathology
There were no tanezumab-related histopathologic findings and no evidence of osteonecrosis. The physes were normal, and production of primary and secondary spongiosa between control and high-dose rats was similar (Figure 4A and B), indicating no effect on endochondral ossification or cortical shell formation. All bone specimens had normal lamellar collagen orientation when examined using transmitted and polarized light. Factor VIII and PCNA immunohistochemistry showed no differences in bone between control and tanezumab-treated animals. Because there were no findings in high-dose rats, neither bone samples from rats at the two lower doses nor bones from recovery-phase groups were evaluated.
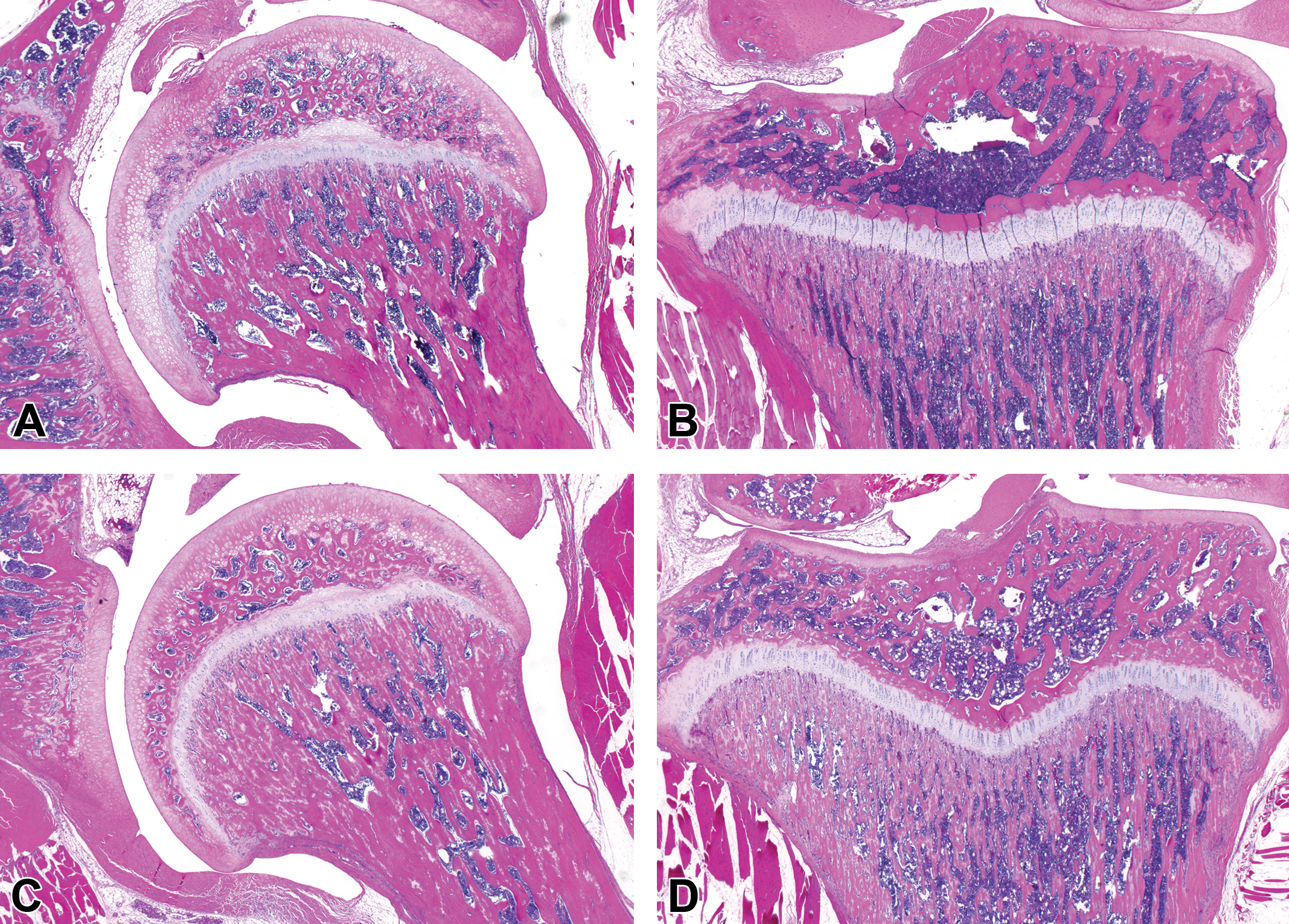
(A-D) Representative female rat hips and proximal tibias. From left, control rat hip joint (A), control rat proximal tibia (B), 10 mg/kg/dose hip joint (C), 10 mg/kg/dose proximal tibia (D). For hip, note intact joint, no evidence of osteonecrosis or joint destruction, and similar physis width. For tibia, note similar physis width. No evidence of impairment of endochondral ossification, and no evidence of osteonecrosis or joint destruction. Decalcified bone, Hematoxylin & Eosin (H&E) stain. (E-H) Female proximal tibia physis. From left, control (E), control (F), 10 mg/kg/dose (G), 10 mg/kg/dose (H). Note similar physis width, normal physis morphology, and normal vascular penetration (Factor VIII) at physis/bone interface. There is no evidence of impairment of endochondral ossification. Decalcified bone, H&E stain for (E) and (G); Factor VIII immunohistochemistry for (F) and (H).
Toxicology
Tanezumab-related clinical signs included sores/scabs at numerous locations (neck, shoulders, ears, head) starting by day 8 of the dosing phase in both sexes. This occurred at all doses although there was some evidence of dose-dependency, particularly between the 0.2 and 1 mg/kg/dose. Although the sores/scabs resolved in some animals in the dosing and/or recovery phase, sores/scabs remained in the majority of the animals throughout the remainder of the study (Online Supplemental Table S5). In addition, one female given the 0.2 mg/kg/dose, one male and one female each given the 1 mg/kg/dose, and two males given the 10 mg/kg/dose developed the first evidence of tanezumab-related sores/scabs in the recovery phase. The skin findings correlated with a minor inflammatory response in clinical pathology parameters. No toxicologically significant changes in body weight or other clinical pathology parameters were noted (Online Supplemental Text S9).
Imaging, mCT
Caliper measurements of femoral length and width in male and female rats did not reveal any treatment-related differences on longitudinal bone growth or femoral diaphyseal diameter (Figure 5). In addition, X-ray radiography revealed no bone deformities or fractures. There were no statistically significant differences in any of the measured cortical bone parameters among treatment groups; animal-to-animal variation was within expected physiological variations (Figure 6). All cancellous bone parameters at the distal femoral metaphysis were similar among dose groups and sexes, although female rats generally exhibited more cancellous bone at the distal femoral metaphysis compared with males (Figure 5), as would be expected (David et al. 2006).
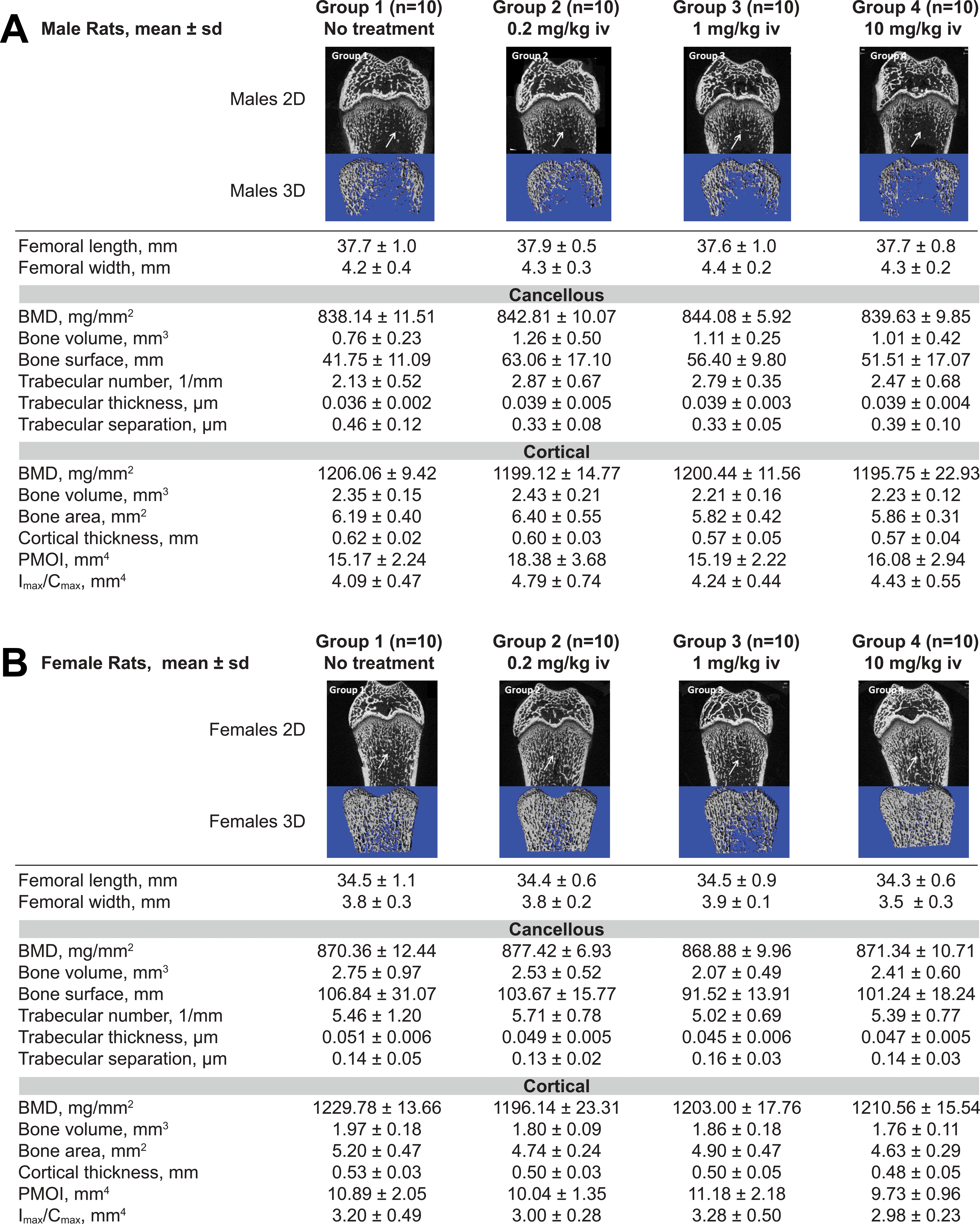
Representative two-dimensional mCT images of the entire distal femur and three-dimensional images of the cancellous bone from distal femoral metaphysis (arrows) in (A) male and (B) female rats. Arrows indicate secondary spongiosa where 3-D structural analysis of cancellous bone was performed. Note more cancellous bone in secondary spongiosa in female rats relative to males. 2/3-D, two/three-dimensional; BMD, bone mineral density; C max, maximum plasma concentration; I max, maximum inertia volume; PMOI, polar moment of inertia; s.d., standard deviation.
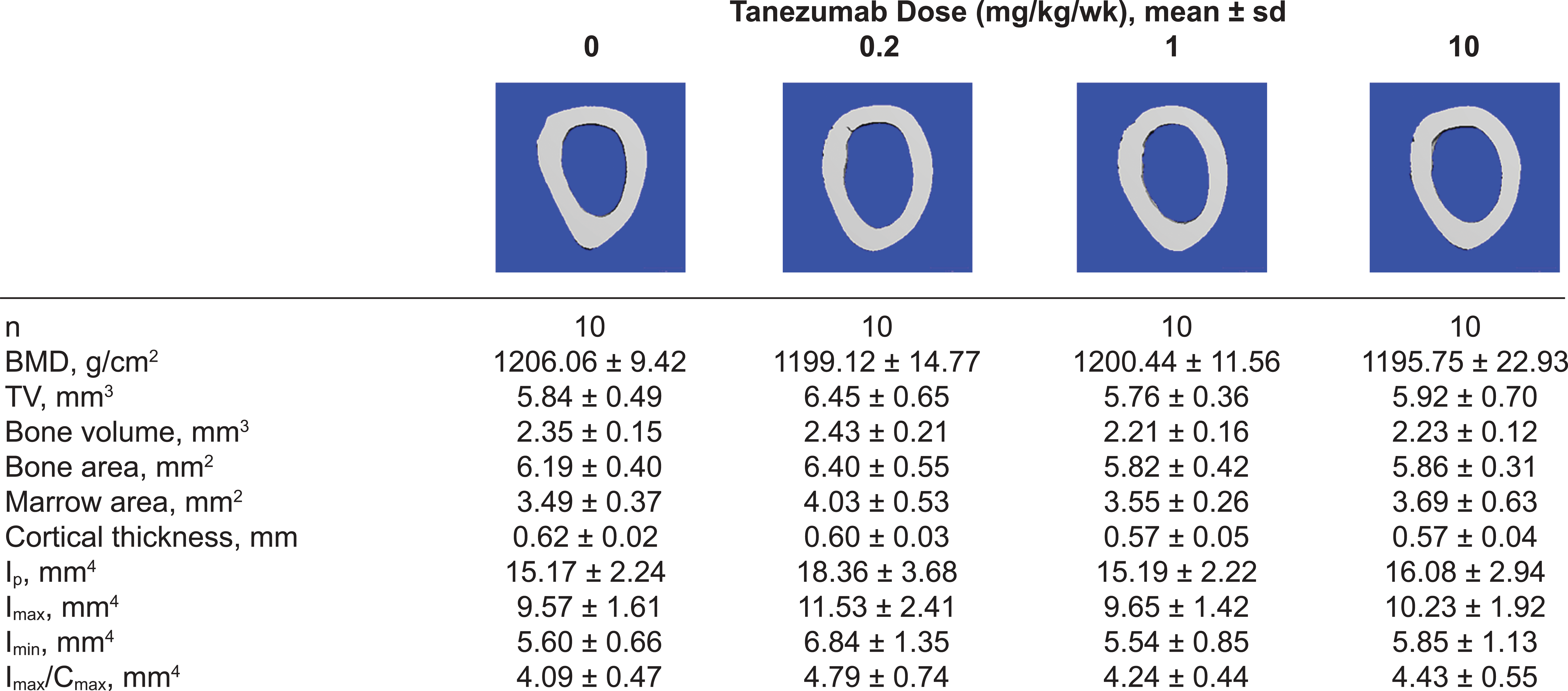
Representative images and numerical results of two-dimensional analysis of cortical bone parameters obtained at the femoral mid-diaphysis in male rats using mCT imaging. BMD, bone mineral density; C max, highest drug concentration observed in plasma; I max, maximum inertia value; I min, minimum inertia value; I p, polar moment of inertia; s.d., standard deviation; TV, tissue volume.
Study 3 (Mouse Study)
Toxicology
No abnormal clinical signs or significant differences in body weight were observed between muMab911- and immunoglobulin G1 (control)-treated groups; all mice survived to scheduled termination.
Bone endpoints
There were no muMab911-related histopathologic findings in joints or bones. There was no evidence of osteonecrosis or other bone pathology, and long bone physes were normal. All bone specimens had normal lamellar collagen orientation under transmitted and polarized light. Immunohistochemical staining was as expected for Factor VIII and PCNA, which showed no differences between control bone and animals administered muMab911.
Discussion
Despite differences in bone mechanics, gross and microanatomic differences compared with the human skeleton, monkeys are considered the most suitable laboratory animal to model the physiology and pathology of the human skeleton (Bagi, Berryman, and Moalli 2011; Brommage 2001; Jerome and Peterson 2001). In addition, radiologic and histologic evidence shows that monkeys of both sexes naturally develop early lesions of osteoarthritis by 10 years of age and are a natural model for osteoarthritis in humans (Carlson et al. 1996; Carlson et al. 1994; Radin et al. 1991). Therefore, these animals are good models for recreating clinical bone and joint findings from clinical tanezumab studies.
In these studies, tanezumab was administered to cynomolgus monkeys at doses that were large multiples of the clinically effective exposure (Brown et al. 2012; Brown et al. 2013). Using the clinically relevant dose in humans is a valid comparison since mature NGF sequences from humans and these monkeys are identical (https://www.ncbi.nlm.nih.gov/protein/XP_005542281.1?report=genpept). The high dose of tanezumab used in the rat studies is also much larger than that needed to maximally reduce pain behaviors in a rat model of deep somatic pain (Shelton et al. 2005). The murine precursor 911 was administered to mice at 10 mg/kg, a dose shown to be effective in multiple pain models in mice (Koewler et al. 2007; Sevcik et al. 2005). Thus, all doses used in these studies are relevant for understanding any potential adverse events resulting from clinical use.
In the current study, no differences were found in radiologic evaluation, BMD, or structural parameters in cortical or trabecular bone evaluated by mCT in tanezumab-treated monkeys versus controls, nor were there any tanezumab-related findings in bone and joint histopathology or serum bone biomarkers in sexually mature monkeys following 6 months of treatment at weekly doses of 4 or 30 mg/kg. Instead, findings were consistent with early osteoarthritis, as is typical for the species, age, sex, and joints examined, and were not tanezumab-related (Carlson et al. 1994; Carlson et al. 1996; Ham et al. 2002). Thus, although the monkeys in this study were too young to exhibit marked osteoarthritic changes in subchondral bone at the proximal femur and tibia, there was no evidence that prolonged tanezumab treatment effects normal bone physiology or exacerbates early osteoarthritis.
Although 65% of control monkeys showed ADAs to tanezumab, tanezumab was not detected in any control animals. The reason for positive ADA samples in control animals could not be determined, but it was not attributable to dosing error (i.e., no control animals were exposed to tanezumab).
In SD rats, the long bone metaphyses are sites of high bone cellular activity (Frost and Jee 1992; Wronski, Cintron, and Dann 1988). Cortical bone physiology in rats is well characterized, and changes observed in periosteal and endocortical bone envelopes following surgical or chemical manipulation are often predictive of cortical bone physiology in humans (Miller et al. 1991; Stenstrom, Hansson, and Thorngren 1977). The 4-week dosing used in this study allowed for comprehensive evaluation of bone metabolism and evaluation of tanezumab effects on bone mass and structure. As with the monkey study, tanezumab treatment in rats had no effects on structural parameters of cancellous bone, cortical or cancellous BMD, cortical bone thickness, bone geometry, or structural integrity. There were no tanezumab-related effects on rat long bone physes and metaphyses. Abnormalities in osteoblast or osteoclast function are readily recognizable in these areas, which are undergoing the processes of endochondral ossification and cortical bone formation in rats of the age used in this study.
The observed skin changes in study 2 were characteristic of self-trauma (excoriation) likely due to pruritus (itchy skin) or altered skin sensation, but no clear cause could be identified. A potential role for involvement of mast cells was evaluated using toluidine blue staining (which stains the granules of mast cells) on histologic sections of some skin lesions in animals administered the 10 mg/kg/dose from the dosing-phase necropsy. This revealed no increase in mast cell numbers or degranulation over what would be expected in inflamed skin. No other tanezumab-related macroscopic or microscopic findings were noted in other nonneural tissues.
Recent nonclinical observations have suggested a clear importance of NGF-TrkA signaling for promoting sensory innervation and development of long bones in mice (Tomlinson et al. 2016; Tomlinson et al. 2017). It is interesting to note the potential causes of the different results obtained in this work by Tomlinson et al. with that described herein, where we saw no effect of administration of anti-NGF antibodies. First, our results were obtained in mice (and rats and monkeys) that were freely moving, but did not have any extra load imposed on their bones. Second, our results were obtained in animals that had been administered an anti-NGF antibody, whereas the results of Tomlinson et al. were obtained by direct inhibition of TrkA signaling. We observed that in mice, there were no muMab911-related clinical signs or histopathologic findings in joints and bone, and furthermore, no osteonecrosis was observed. All bone specimens had normal lamellar collagen orientation; immunohistochemistry results for Factor VIII and PCNA were similar among treatment groups. Exactly how inhibition of TrkA signaling influences bone homeostasis during mechanical loading remains to be determined. However, our results provide evidence for long-term tolerability of a mouse NGF antibody in rodents and tanezumab in nonhuman primates since no deleterious effects of tanezumab on bone and joint tissues were observed.
The only tanezumab-related finding in biomarkers across studies was higher mean osteocalcin concentration at doses ≥0.2 mg/kg in rats. Osteocalcin, a matrix protein produced by osteoblasts during bone synthesis, is mainly a marker of bone formation, although increases can also be seen with bone resorption (Ivaska et al. 2004). The higher osteocalcin levels reversed following an 8-week recovery and were not accompanied by concomitantly higher P1NP, considered a more sensitive indicator of increased bone formation than osteocalcin in rats (Hale et al. 2007), and there were no tanezumab-related changes in ALP. There were also no changes in serum CTX1, a bone resorption marker (Rosen et al. 2000). Often, all three parameters are altered in states of increased bone turnover, such as occurs in ovariectomized rats (Rissanen et al. 2008). Also, the presence of permanently open growth plates and bone modeling can cause natural variation in osteocalcin secretion in rats (Bagi et al. 2012). Based on the low magnitude of the osteocalcin values, reversibility, no concurrent change in P1NP or CTX1, lack of complementary histopathology findings, and the presence of open growth plates in all rats in all groups, the described change was not adverse and there was no evidence of an effect on bone turnover.
Conclusion
No animal model entirely mimics human conditions because no animal species has skeletal or biomechanical properties identical to humans. Also, disease conditions, comorbidities, and co-treatments can influence the efficacy and safety of tested compounds. Therefore, availability and translatability of biomarkers to the clinical environment becomes an important issue. Normal mice, rats, and monkeys are routinely used in nonclinical safety studies, and serum biomarkers of bone metabolism are well established and validated. By applying well-designed protocols using three species, we assessed tanezumab safety using multiple techniques.
Our results show that the administration of tanezumab or muMab911 did not alter bone metabolism in normal, healthy animals, a finding also confirmed in a rat model of osteoarthritis (LaBranche et al. 2017). Although safety studies are traditionally performed in normal animals, our experience in the development of tanezumab suggests that the preemptive use of available disease models that mimic the general arthritis patient population should also be considered to further assess the efficacy and safety of novel compounds since the human adverse events were not predicted in normal standard laboratory animal species.
Supplemental Material
Supplemental Material, DS1_TPX_10.1177_0192623318772501 - Effects of Monoclonal Antibodies against Nerve Growth Factor on Healthy Bone and Joint Tissues in Mice, Rats, and Monkeys: Histopathologic, Biomarker, and Microcomputed Tomographic Assessments
Supplemental Material, DS1_TPX_10.1177_0192623318772501 for Effects of Monoclonal Antibodies against Nerve Growth Factor on Healthy Bone and Joint Tissues in Mice, Rats, and Monkeys: Histopathologic, Biomarker, and Microcomputed Tomographic Assessments by Kathryn E. Gropp, Cathy S. Carlson, Mark G. Evans, Cedo M. Bagi, William J. Reagan, Susan I. Hurst, David L. Shelton, and Mark A. Zorbas in Toxicologic Pathology
Footnotes
Acknowledgments
The authors would like to thank Shana Dalton (Covance study director), Sato Oneda (SNBL [Everett, WA] study director), Thomas Cummings (Pfizer), Edwin Berryman (Pfizer), and Colleen Huddleston (Pfizer).
Author Contributions
Authors contributed to conception or design (KG, ME, CB, WR, SH, DS, MZ); data acquisition, analysis, or interpretation (KG, CC, ME, CB, WR, SH, DS, MZ); drafting the manuscript (KG, CB, WR, SH); and critically revising the manuscript (KG, CC, ME, CB, WR, SH, DS, MZ). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Ethics Approval
Oversight by local Institutional Animal Care and Use Committees.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kathryn E. Gropp, Cedo M. Bagi, William J. Reagan, Susan I. Hurst, and Mark A. Zorbas are employees of Pfizer and own stock and/or stock options in Pfizer. Mark G. Evans and David L. Shelton were employees of Pfizer during the development of the article. Cathy S. Carlson has no conflicts of interest to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Medical writing support was provided by Joseph Oleynek at Engage Scientific Solutions and was funded by Eli Lilly & Co. and Pfizer. These studies were funded by Pfizer.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
