Abstract
Chromated copper arsenate (CCA) is used as a wood preservative worldwide. Exposure to it may adversely affect human health. Some events have increased human exposure to CCA, including the Great East Japan Earthquake, which generated a large amount of lumber debris from CCA-treated woods. We elucidated the toxicity due to daily exposure to CCA over a 4-week period at doses of 0, 8, 40, and 80 mg/kg/day in Wistar Hannover rats. Chromium (Cr) and arsenic (As), but not copper, were detected in the plasma samples of rats treated with various doses of CCA. Males and females showed sedation, and males had poor body weight gain. The clinical pathologies observed in both sexes included hypochromic and microcytic anemia, hepatic and renal dysfunction, and changes in lipid and glucose levels. Histopathologically, males and females showed forestomach hyperkeratosis, mucosal epithelial hyperplasia in the small intestine, rectal goblet cell hypertrophy, and lipofuscin deposition in the proximal renal tubule. Females showed diffuse hepatocellular hypertrophy with increased 8-hydroxydeoxyguanosine levels. These results indicated that oral administration of CCA mainly affected hematopoietic, gastrointestinal, hepatic, and renal systems owing to the toxic effects of As and/or Cr. Major toxic effects were observed in both sexes receiving 40 and 80 mg/kg/day.
Most wood preservatives contain toxic or hazardous chemicals that can adversely affect human health. Wood preservatives have many applications, such as in construction of buildings, public playground equipment, flower beds, and outdoor residential structures, and they can be released into the environment, that is, soil, groundwater, and air (Dickey 2003; Kim et al. 2007; Rahman et al. 2004; Shibata et al. 2007). Amid a rising tide of concern about preservatives that contain arsenic (As), especially chromated copper arsenate (CCA), CCA-treated new lumber has not been manufactured for residential purposes in the United States since 2004 (US Environmental Protection Agency 2003). However, many CCA-treated structures still exist, and this chemical can persist in treated wood for up to 10 to 40 years (McQueen and Stevens 1998). Therefore, the possibility of current and future human exposure to toxic metals from CCA-treated wood remains.
The metals in CCA are usually present in the form of oxides, and wood is industrially treated using a vacuum-pressure impregnation process (Hingston et al. 2001). Although large amounts of preservative remain in wood after prolonged exposure, even small percent losses may be environmentally significant, due to the high toxicity of the components. Toxic components include soluble copper (Cu) hydroxide ions, CuSO4, chromium (Cr) trioxide, HCrO4-, or CrAsO4. Hexavalent Cr (Cr[VI]), present in the CCA treatment solution, leaches from wood much more readily than trivalent Cr (Cr[III]), the predominant form after fixation occurs (Dickey 2003). Cr(III) can be oxidized to Cr(VI) in water or soil under certain conditions after leaching from wood. Similarly, arsenate (As[V]) found in CCA leaches less readily than arsenite (As[III]). Both As and Cu are more likely to leach from wood than Cr. Interestingly, As(III) concentrations in CCA are inversely proportional to Cr(VI), indicating that Cr(VI) may oxidize As(III) in the CCA mixture. The presence of Cr(VI) and the absence of As(III) in commercial supplies suggest that fixation may not be completed in impregnated timber available in the market (Hingston et al. 2001).
The toxicity due to environmental exposure to CCA in human is controversial at present because the levels to which humans are exposed to CCA are extremely low under common environmental circumstances; however, several data suggested the accidental exposure to CCA might affect human health. Peters et al. (1984) reported that family members experienced recurring neurological and medical illness over three years, especially during the winter months due to burning CCA-treated wood with a small stove to heat their house. The same authors also reported that woodworkers developed hematological, dermal, and neuropsychological diseases from burning and sawing CCA-treated wood (Peters et al. 1986). Natural disasters, such as the Great East Japan Earthquake, might increase exposure of CCA to human. The earthquake and the following tsunami on March 11, 2011, generated a huge amount of lumber debris, including CCA-treated woods, which had the potential to spread elements in CCA, such as As, Cr, and Cu, into the environment (Ohgami et al. 2015; Shibata, Solo-Gabriele, and Hata 2012). In the fieldwork study after the Great East Japan Earthquake, measurement of elements in 233 pieces of lumber debris collected revealed that 5 pieces (2.1%) were CCA-treated wood containing 10,000 ± 8869 mg/kg of Cr, 2064 ± 1319 mg/kg of Cu, and 3380 ± 2328 mg/kg of As (Ohgami et al. 2015). The environmental monitoring survey of soil in the disaster area of the earthquake was conducted by the Ministry of the Environment. As a result of investigation at 70 sites in Miyagi prefecture, measured values (0.011 to 0.15 mg/L) exceeding the soil elution standard (0.01 mg/L) were detected at 25 sites (35.7%) for As (Ministry of the Environment 2012), although it is not clear whether these values were derived from CCA-treated wood. Similar findings on the measurement of As in lumber debris generated by Hurricane Katrina were reported in New Orleans (Dubey, Townsend, and Solo-Gabriele 2007). These high As values exceeding the reference levels do not directly lead to human hazard, but it is important to obtain information for risk assessment against accidental high concentration exposure to CCA.
Although each of the three chemicals in CCA is individually toxic to the digestive, respiratory, urinary, nervous, circulatory, dermatologic, and immune systems (Abernathy et al. 1999; Frantzen 1998; Matos et al. 2013; Ryan, Huet, and MacIntosh 2000; Schäfer et al. 1999), the overall toxicity of CCA remains unknown, even basic information. Previously, we reported the overall toxicity of CCA to the immune system, especially the allergenicity, by showing its sensitizing activity via a local lymph node assay (Fukuyama et al. 2008). In order to provide basic toxicity information useful for risk assessment of CCA, we conducted a 4-week, daily dose, oral toxicity study of CCA at a maximum tolerated dose (MTD) in rats, considering accidental high-dose CCA exposure. We found that CCA had a broad toxic potential in several organs, especially the hematopoietic, gastrointestinal, hepatic, and renal systems.
Materials and Methods
Chemicals
Three commercial CCA formulations exist and are referred to as types A, B, and C (Hingston et al. 2001). The CCA type B mixture we used contained arsenic (V) oxide (45.1% as As2O5, w/w), chromium (VI) oxide (35.3% as CrO3, w/w), and copper (II) oxide (19.6% as CuO2, w/w) (American Wood Preservers Association 2005). Arsenic oxide was purchased from Kishida Chemical Co. (Osaka, Japan), and chromium oxide and copper oxide were purchased from Kanto Chemical Co. (Tokyo, Japan).
Animals and Treatment
Six-week-old specific pathogen-free (SPF) Wistar Hannover (BrlHan: WIST@Jcl[GALAS]) rats of both sexes were purchased from Japan Clea, Inc. (Shizuoka, Japan). Two animals were housed in each suspended stainless steel cage in an air-conditioned animal room with a temperature, 22 ± 3°C; humidity, 50 ± 20%; ventilation for the circulation of fresh air, 10 times or more per hr; and illumination, 12 hr/day (lights on at 7:00 and lights off at 19:00). Animals received a standard commercial diet of MF Mash (Oriental Yeast Co., Ltd., Tokyo, Japan), and tap water was provided ad libitum. During the experimental period, all animals were handled in accordance with the Guidelines for Animal Experimentation issued by the Japanese Association for Laboratory Animal Science (JALAS 1987) and with the Code of Ethics for Animal Experimentation of the Institute of Environmental Toxicology.
Arsenic oxide and chromium oxide were dissolved in saline, and copper oxide was then suspended into this mixture. Four groups of ten rats/sex/group received daily gavage administration of CCA at doses of 0, 8, 40, and 80 mg/kg/day for 28 days, based on a preliminary dose range–finding study. Administration of the test substance to healthy animals was initiated at the age of seven weeks.
Analysis of Total Cr, Cu, and As in Blood
Blood samples from one control rat of each sex and three treated rats of each sex, administered different doses of CCA, were collected after 4 weeks of treatment and stored at −70°C until analysis. Using plasma samples, total Cr, Cu, and AS levels were measured by inductively coupled plasma mass spectrometry (Agilent 7500ce ICP-MS, Agilent Technologies, Santa Clara, CA) and atomic absorption photometry with hydride generation (Spectr AA220, Varian, Palo Alto, CA). For analysis, yttrium was used as an internal standard.
Clinical Signs, Body Weight, and Food Consumption
All animals were clinically observed every day during the study period. Body weight and food consumption of each animal were measured weekly.
Ophthalmology, Urinalysis, Hematology, and Blood Biochemistry
Ophthalmological examination and urinalysis were conducted at 4 weeks of treatment as previously reported (Kojima et al. 2009). For the collection of fresh urine samples, test animals were placed in individual metabolism cages for about two hours in the morning. Cumulative urine samples were also collected by individually placing animals in the above metabolism cages for 24 hr for determining urinary appearance and volume. Overnight fasting was not conducted before urinalysis, and rats took food and water freely during urine sampling. Hematological and blood biochemical examinations were carried out after 4 weeks of treatment as previously reported (Kojima et al. 2009). Blood samples were collected from the posterior vena cava of each animal under ether anesthesia following overnight fasting. Although the use of ether as an anesthetic in experimental animals is now strongly discouraged, the experiment in this report was performed in 2004, when ether was more widely used.
Organ Weight Measurement and Pathological Examination
At necropsy, after 4 weeks of treatment, the weights of organs including the brain, pituitary, lungs, thymus, liver, kidneys, spleen, prostate, seminal vesicles/coagulating glands, and testes were measured. Relative organ weights were expressed as percentages of body weight. Systemic organs were fixed in 10% buffered formalin solution, sectioned, and stained with hematoxylin and eosin for histopathological examination. Schmorl’s reagent and Prussian blue were also applied to identify brown pigments in the kidneys.
Oxidative Stress Measurement
After 4 weeks of treatment, 8-hydroxydeoxyguanosine (8-OHdG) levels were measured using liver samples obtained from six animals in order of ascending animal numbers in each group. Measurement of lipid peroxide contents was conducted using liver samples from all surviving animals. Both parameters were measured as previously reported (Harada et al. 2003).
Statistical Analysis
All data are expressed as mean ± standard deviation. The significance of differences between control and treated groups regarding body weight, food consumption, urinalysis, hematological and blood biochemical parameters, organ weights, and oxidative stress was determined with Dunnett’s multiple comparison test (two-sided analysis). Fisher’s exact test (one-sided analysis) was used for the evaluation of the incidence of clinical signs, ophthalmologic and pathological findings. A p value ≤ .05 or .01 indicated statistical significance.
Results
Chemical Analysis in Blood
Total Cr and As levels were detected in plasma samples from rats treated with CCA at all dose levels but not in rats treated with the vehicle control (Figure 1). The levels of plasma Cu in treated rats were comparable to the levels in control rats (data not shown).
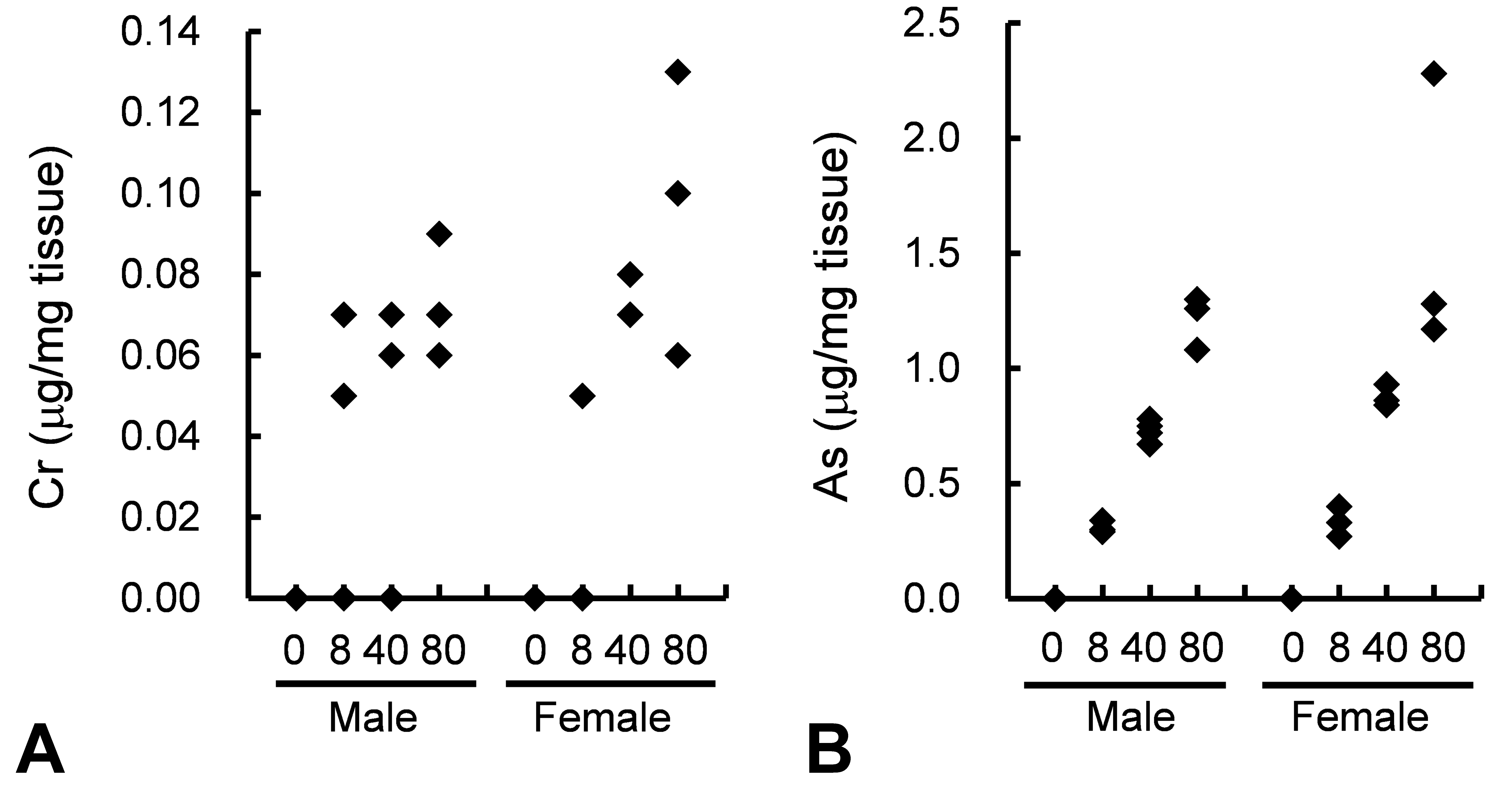
Plasma concentration of total Cr and As in rats treated with CCA at 0, 8, 40, and 80 mg/kg/day for 4 weeks. Blood samples were collected to determine the concentration of total Cr (A) and As (B). N = 1 in the control group, and N = 3 in each treated group for each sex. Cr = chromium, As = arsenic, CCA = chromated copper arsenate.
Clinical Signs, Body Weight, and Food Consumption
Male rats receiving 80 mg/kg/day and female rats receiving 40 and 80 mg/kg/day of CCA showed sedation, increased salivation, and soiled fur in the abdominal or external genital region during the treatment period (Online Supplemental Table 1). One male receiving 80 mg/kg/day died in the first week, but the cause of death was unknown. Males receiving 80 mg/kg/day gained lesser weight than control subjects throughout the treatment period, and males receiving 40 mg/kg/day showed low body weight gain at week 4 (Figure 2). Consistent with poor body weight gain, these males showed a remarkable reduction in food consumption during the study period (Online Supplemental Figure 1A). Females receiving 40 and 80 mg/kg/day also showed reduced food consumption, especially in the early phase of the study (Online Supplemental Figure 1B), but they gained weight at the same rate as the control subjects did (Figure 2).
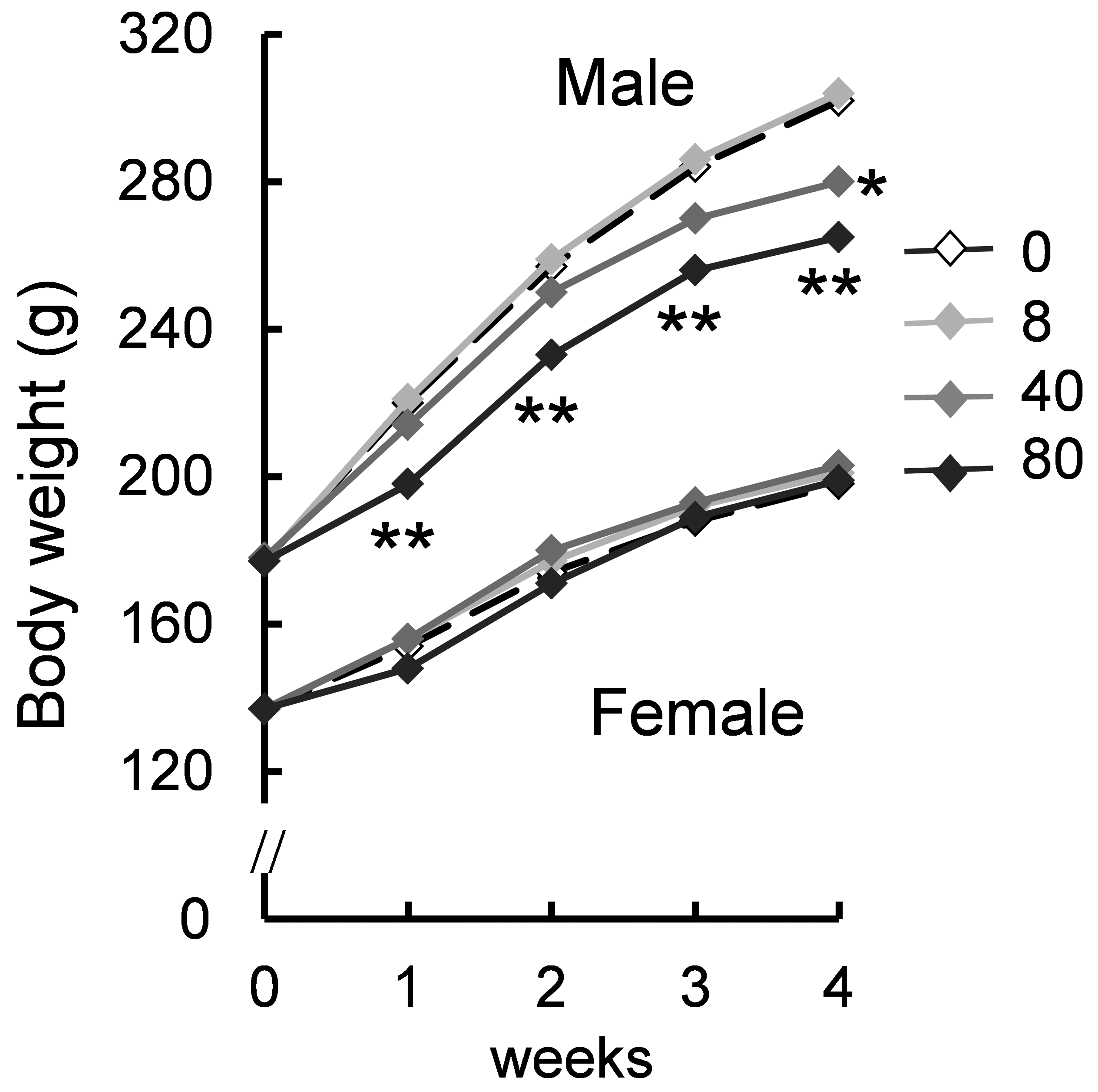
Time course of body weight changes in rats treated with CCA at 0, 8, 40, and 80 mg/kg/day for 4 weeks. During the treatment, body weight was measured weekly. *p ≤ .05 or **p ≤ .01 versus control group by Dunnett’s multiple comparison test. CCA = chromated copper arsenate.
Ophthalmology and Urinalysis
No treatment-related ophthalmological changes were detected in any of the treated groups (data not shown). Males and females receiving 80 mg/kg/day of CCA showed lesser urine-specific gravity than control subjects (male control, 1.033 ± 0.011 vs. male high dose, 1.018 ± 0.012, p ≤ .05; female control, 1.038 ± 0.019 vs. female high dose, 1.016 ± 0.009, p ≤ .01). This change was associated with a reduction in protein and ketones (data not shown) and/or an increase in urine volume (male control, 13.4 ± 3.1 mL/day vs. male high dose, 23.0 ± 13.7 mL/day; female control, 10.4 ± 5.9 mL/day vs. female high dose, 25.1 ± 15.2 mL/day, p ≤ .01). A similar change in protein levels was observed in females receiving 8 and 40 mg/kg/day of CCA.
Hematology
Males receiving 80 mg/kg/day and females receiving 40 and 80 mg/kg/day of CCA had significantly lesser hemoglobin concentration (Hb), mean corpuscular hemoglobin, and mean corpuscular hemoglobin concentration than the control subjects did. Lower Hb levels in individual total and mature erythrocytes were confirmed by assessing cellular hemoglobin concentration mean of total red cells and mature erythrocytes (CHCMm) in rats receiving 80 mg/kg/day of CCA (Figure 3 and Online Supplemental Table 2). Significant decreases and decreasing trends in both mean corpuscular volume of total erythrocytes and mature erythrocytes were observed in females administered 40 and 80 mg/kg/day and in males administered 80 mg/kg/day. A significant decrease in hematocrit (Ht) levels was observed in females administered 80 mg/kg/day. Females receiving 40 and 80 mg/kg/day showed significantly higher reticulocyte counts than control subjects, and rats of both sexes receiving 80 mg/kg/day had significantly lower mean corpuscular volume of reticulocytes and content of hemoglobin per reticulocyte (Chr; Figure 4). Consistent with these findings, characteristic alterations in distribution width were observed in rats receiving 40 and/or 80 mg/kg/day for cell volume (red cell distribution width, anisocytosis) and/or for cell hemoglobin concentration (hemoglobin distribution width, anisochromia). Although there were fluctuations in some parameters in males receiving 40 mg/kg/day and females receiving 8 mg/kg/day of CCA, anemia (i.e., reductions in Ht, Hb, and/or erythrocyte count) was not clearly detected. Males and females receiving 40 and 80 mg/kg/day showed significant increases in total leukocyte and lymphocyte counts, and animals receiving 80 mg/kg/day also showed significant increases in neutrophil count in males and monocyte count in both sexes. A significant decrease in eosinophil count was noted in females treated with 80 mg/kg/day.
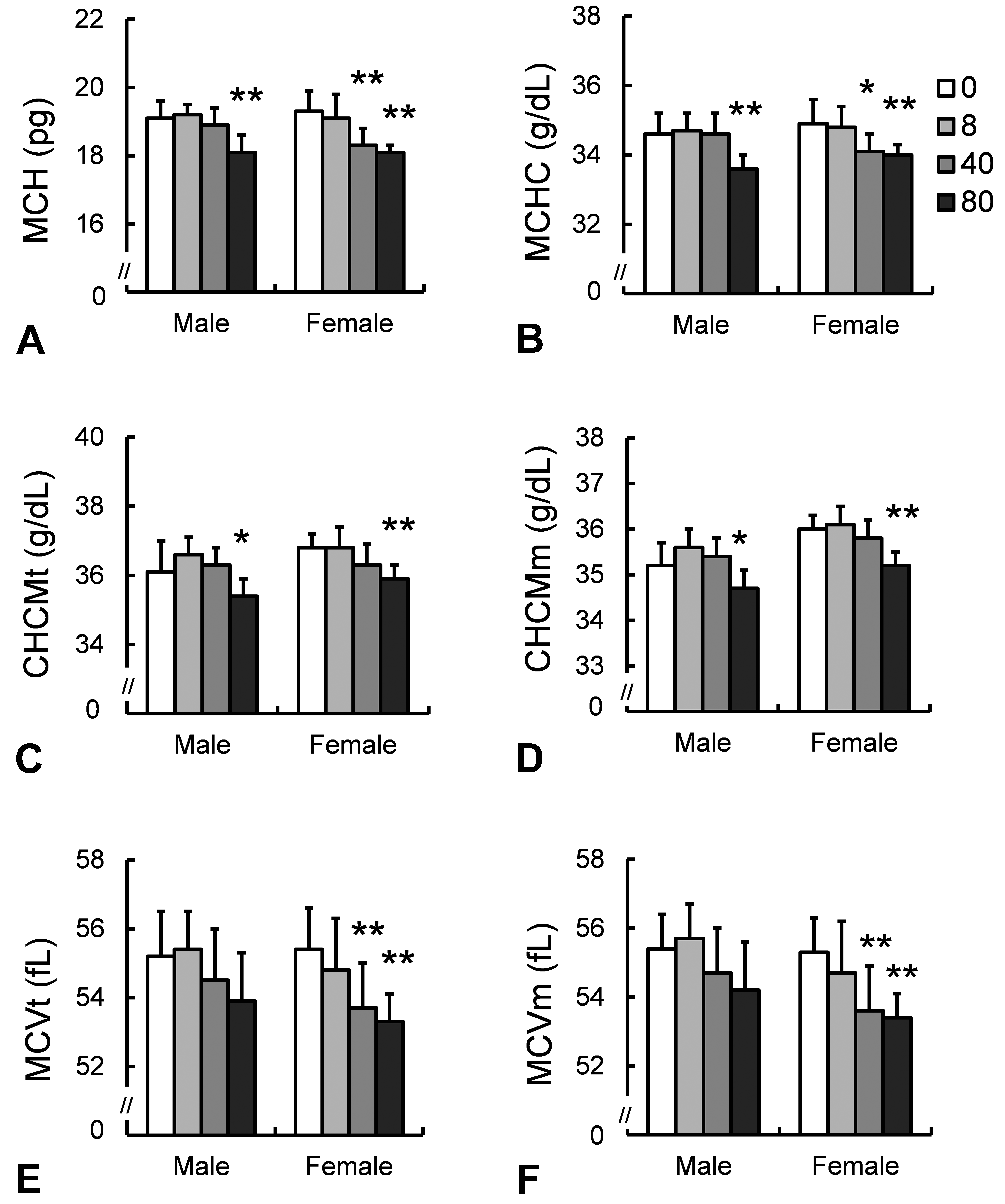
The levels of MCH, MCHC, CHCM, and MCV in rats treated with CCA at 0, 8, 40, and 80 mg/kg/day for 4 weeks. Blood samples were collected and subjected to hematology. (A) MCH; (B) MCHC; (C, E) CHCM and MCV in total erythrocyte (CHCMt and MCVt, respectively); and (D, F) CHCM and MCV in mature erythrocyte (CHCMm and MCVm, respectively). *p ≤ .05 or **p ≤ .01 versus control group by Dunnett’s multiple comparison test. MCH = mean corpuscular hemoglobin, MCHC = mean corpuscular hemoglobin concentration, CHCM = cellular hemoglobin concentration mean, MCV = mean corpuscular volume, CCA = chromated copper arsenate.
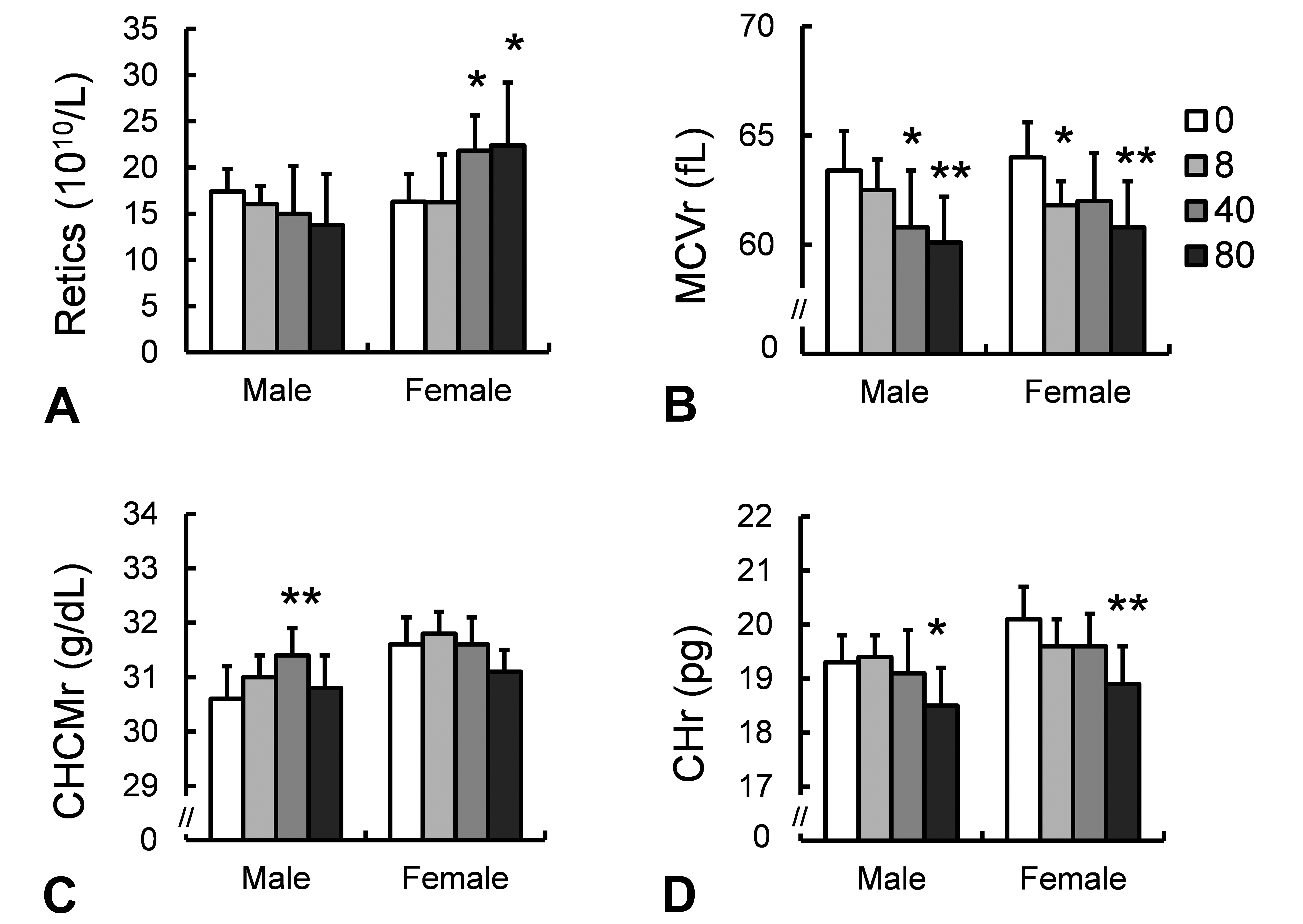
Reticulocyte analysis in rats treated with CCA at 0, 8, 40, and 80 mg/kg/day for 4 weeks. Blood samples were collected and subjected to hematological studies. (A) Reticulocyte count (Retics); (B) MCV in reticulocyte (MCVr); (C) CHCM in reticulocyte (CHCMr); and (D) Content of hemoglobin per reticulocyte (Chr). *p ≤ .05 or **p ≤ .01 versus control group by Dunnett’s multiple comparison test. CCA = chromated copper arsenate, MCV = mean corpuscular volume, CHCM = cellular hemoglobin concentration mean.
Blood Biochemistry
γ-Glutamyl transpeptidase (GGTP), total cholesterol, blood urea nitrogen (BUN), triglyceride (TG), and total bilirubin (T.Bil) levels in males and/or females treated at 80 mg/kg/day of CCA were significantly higher than those in control subjects; GGTP and T.Bil were higher in females than in males; high BUN and TG were observed in males and females, respectively (Online Supplemental Table 3). Although there was no statistically significant difference, relatively high aspartate aminotransferase (AST) and alanine aminotransferase (ALT) were noted in males and females treated at 80 mg/kg/day. These increases in AST and ALT depended on one male and one female that showed extremely high values (AST 267 and 400U/L and ALT 203 and 87U/L in male and female, respectively) and corresponding histopathological changes (focal hepatocellular necrosis and bile duct hyperplasia) in the liver. Total protein, albumin, and globulin levels in both sexes of the high-dose group were significantly lower than those in the control subjects. These were accompanied by an increase in albumin/globulin ratio. Similar findings were also observed in females receiving 40 mg/kg/day. Glucose (Gluc) was not consistently altered in rats of either sex by CCA administration: A decrease was observed in males administered 40 and 80 mg/kg/day, and an increase was observed in females administered 80 mg/kg/day. Significant increases in inorganic phosphorus (P) and potassium and a significant decrease in chloride in females receiving 40 and 80 mg/kg/day were found; a significant increase in P was also noted in males received the highest dose. Other significant changes were incidental or of little toxicological relevance.
Pathology
All rats treated at 40 and 80 mg/kg/day of CCA showed luminal dilatation of the duodenum and distention of the cecum at necropsy (data not shown). In the gastrointestinal tracts, hyperkeratosis in the forestomach, mucosal epithelial cell hyperplasia in the small intestine, and hypertrophy of goblet cells in the rectum were observed histopathologically (Table 1, Figure 5). Hyperkeratosis was mild and was found mainly at the limiting ridge between the forestomach and the glandular stomach. Mucosal epithelial cell hyperplasia corresponded to an increase in villous length, and the changes were almost the same in the duodenum, jejunum, and ileum. There was no obvious increase in crypt depth. Hypertrophy of goblet cells refers to increased size of mucous-containing vacuoles in mucosal epithelial cells. No erosion, ulcer, or inflammation was detected in the gastrointestinal tract. Concurrent with the gastrointestinal lesions, sinus erythrocytosis was observed in the mesenteric lymph nodes, suggesting that bleeding also occurred in the gastrointestinal tract. In organ weight measurement (Online Supplemental Table 4), females receiving 40 and 80 mg/kg/day of CCA showed significant increases in absolute and relative weights of the liver and kidneys, in accordance with histopathological observations of diffuse hepatocellular hypertrophy and brown pigment deposition in the proximal renal tubules, respectively (Figure 6). Males receiving 80 mg/kg/day also showed significant increases in relative weights of the liver and kidneys, which were associated with the same proximal lesions in the kidney. The brown pigments were identified as lipofuscin because they were positive for the Schmorl reaction (Figure 6) and negative for Prussian blue staining (data not shown). Although there was no statistically significant difference in the incidences of the findings, moderate focal hepatocellular necrosis and bile duct hyperplasia in the liver were observed in one male and one female with high AST and ALT values as described above in the CCA 80 mg/kg/day group. Hepatocellular necrosis was found multifocally, and there is no localization in the centrilobular or periportal region. Both findings are sometimes observed as spontaneous lesions, but their severities were clearly exceeding the degree normally seen in rats of this age. Thus focal hepatocellular necrosis and bile duct hyperplasia noted in one male and one female treated at 80 mg/kg/day were judged to be treatment-related. The absolute and relative weights of the thymus in males and females receiving 40 and 80 mg/kg/day of CCA were significantly lower than those of the control subjects; a similar change was observed in females receiving only 8 mg/kg/day of CCA. Cortical atrophy in the thymus was observed in a small number of males. Other organ weight changes were minor, and no corresponding histopathological findings were evident.
Selected Histopathological Lesions from Rats Treated with CCA.
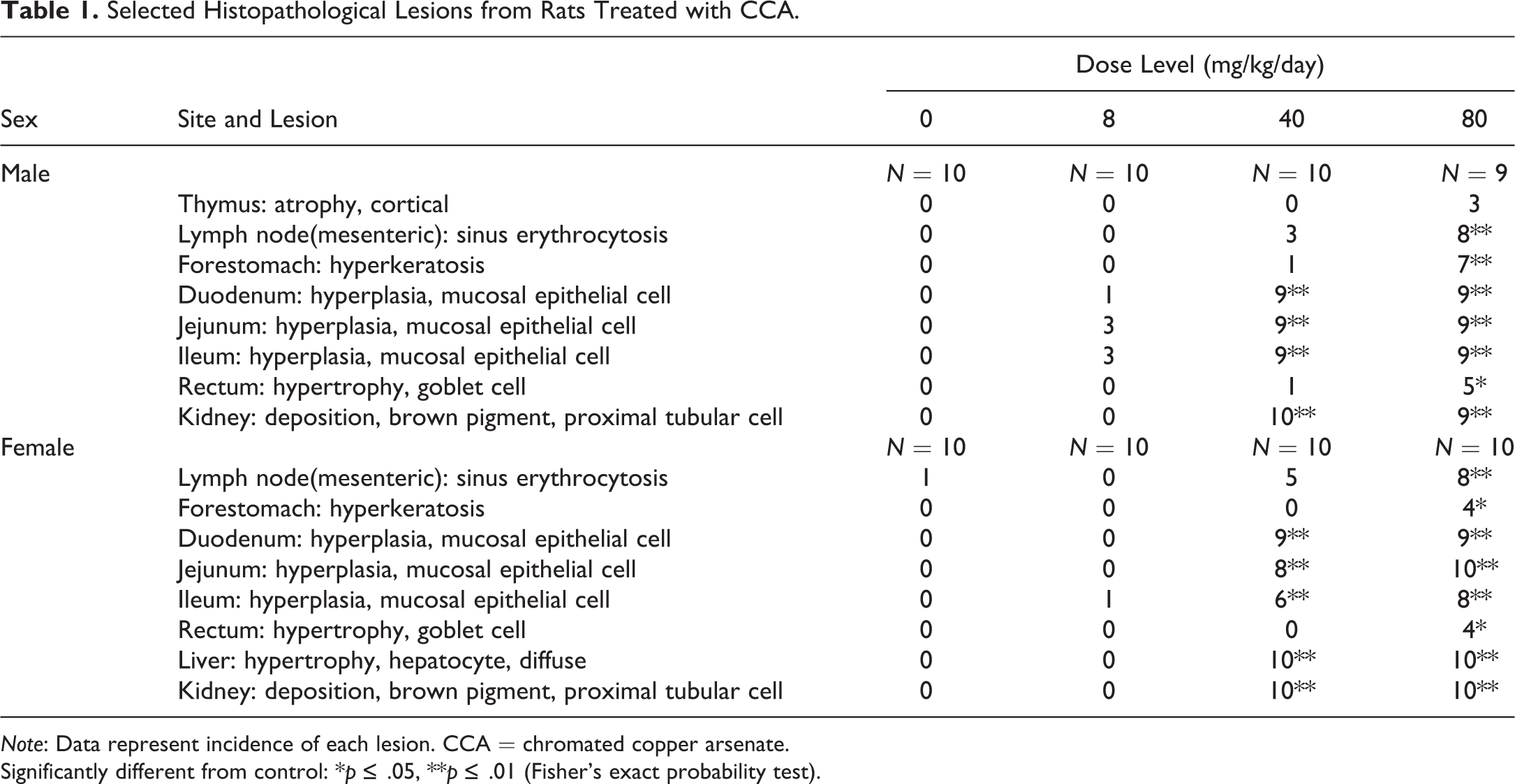
Note: Data represent incidence of each lesion. CCA = chromated copper arsenate.
Significantly different from control: *p ≤ .05, **p ≤ .01 (Fisher's exact probability test).
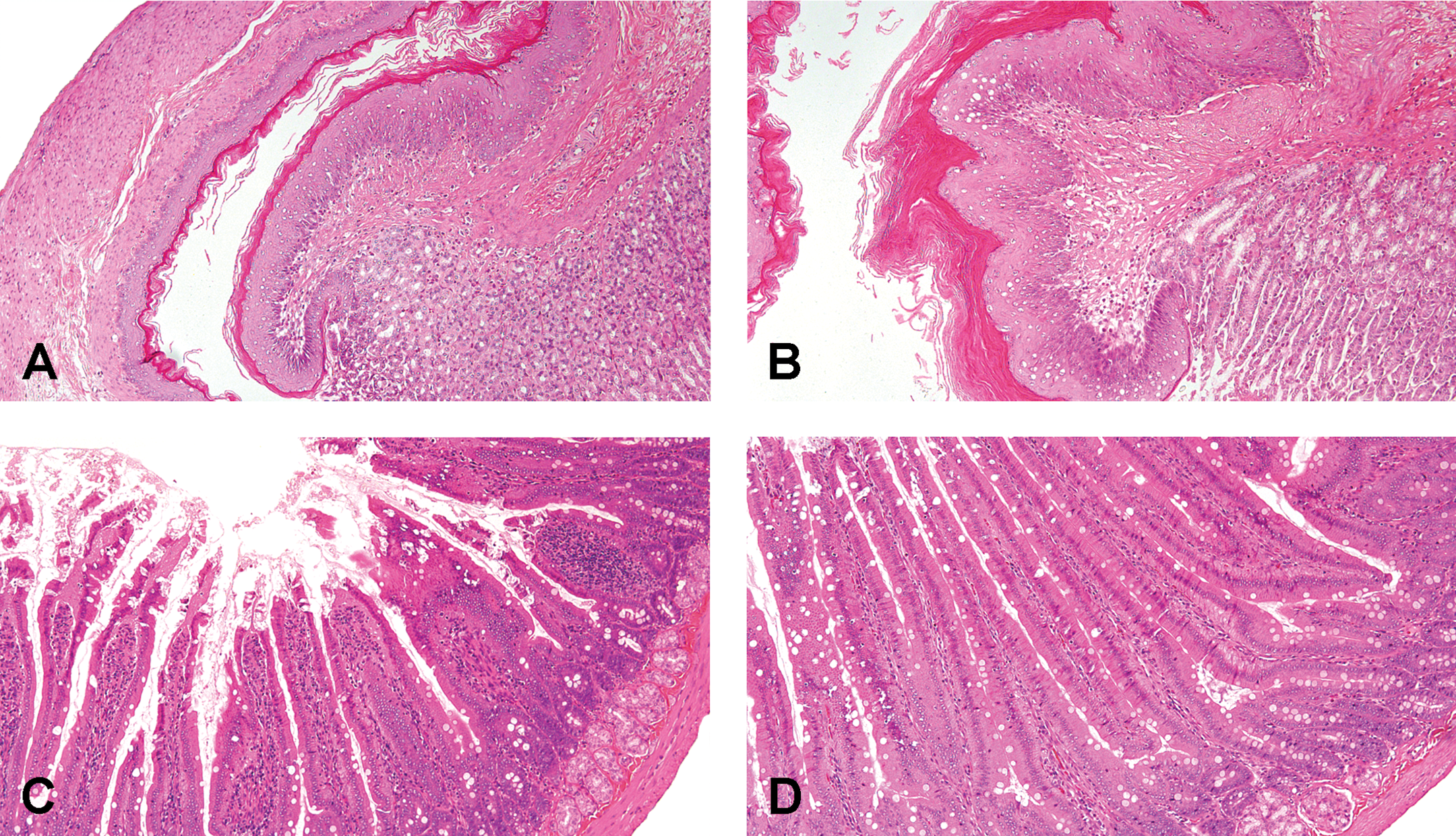
Representative images of histopathological lesions in rats treated with CCA. Micrographs of the forestomach (A and B) and duodenum (C and D) stained with H&E were obtained from rats orally administered CCA at 0 (A and C; normal) and 80 mg/kg/day (B and D) for 4 weeks. (B) Hyperkeratosis of squamous epithelial cell; (D) Mucosal epithelial cell hyperplasia. CCA = chromated copper arsenate, H&E = hematoxylin and eosin.
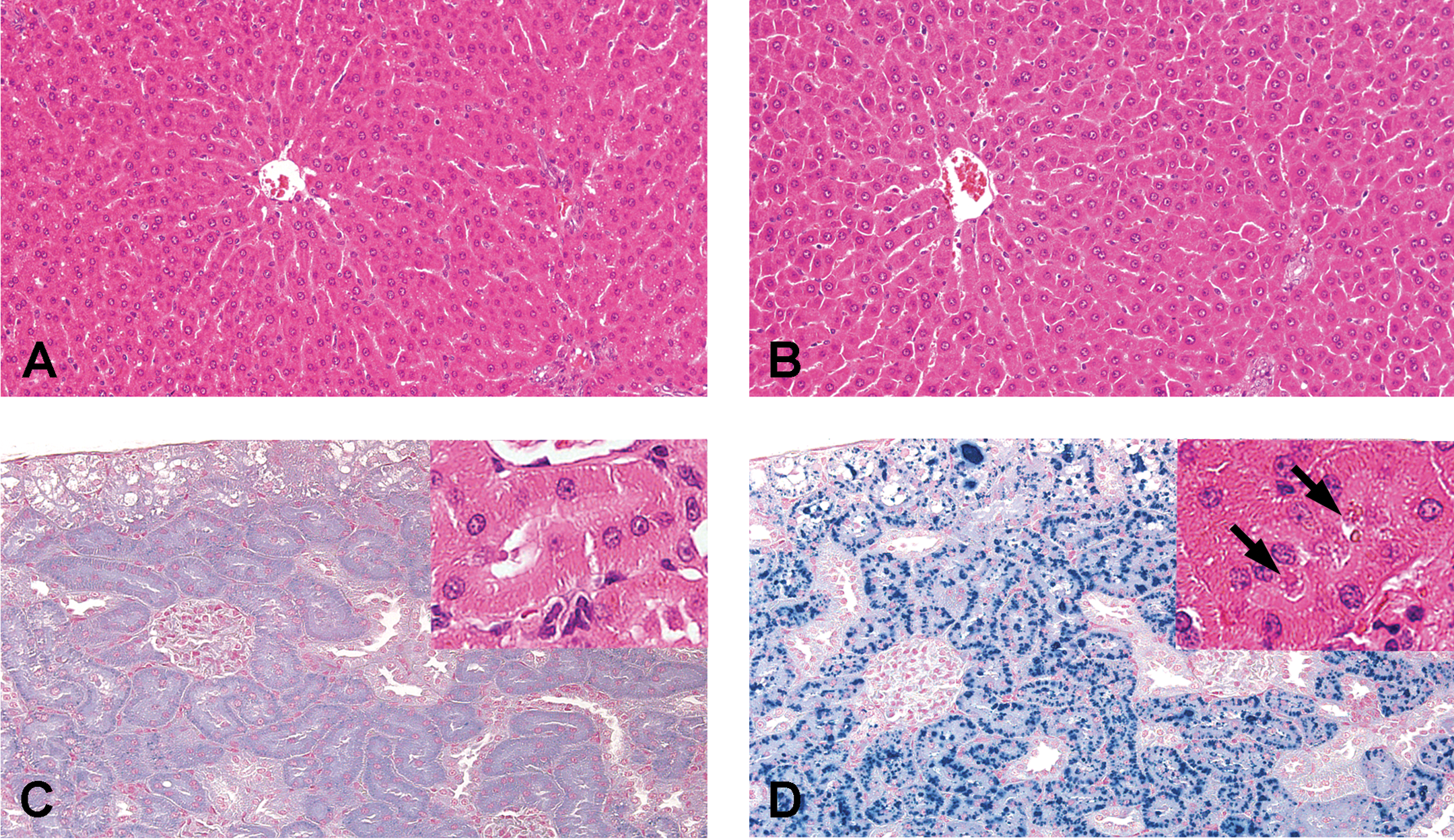
Representative images of histopathological lesions in rats treated with CCA. Micrographs of the liver (A and B) stained with H&E and of the kidneys (C and D) stained with Schmorl’s reagent were obtained from rats orally administered CCA at 0 (A and C; normal) and 80 mg/kg/day (B and D) for 4 weeks. (B) Diffuse hepatocellular hypertrophy; (D) Deposition of brown pigments stained positively in the proximal tubules. Inset (C and D): Representative images of the proximal tubule stained with H&E from CCA 0 (C) and 80 mg/kg/day (D). Arrows (D) show brown pigments deposited in proximal tubule. CCA = chromated copper arsenate, H&E = hematoxylin and eosin.
Oxidative Stress Measurement
In the liver, 8-OHdG levels in females receiving 80 mg/kg/day of CCA were significantly higher than those in the control subjects, whereas the levels in males receiving 40 and 80 mg/kg/day were significantly lower than those in the control subjects (Figure 7A). Lipid peroxide contents in males receiving 80 mg/kg/day were lower than those in the control subjects, but this change was not statistically significant (Figure 7B).
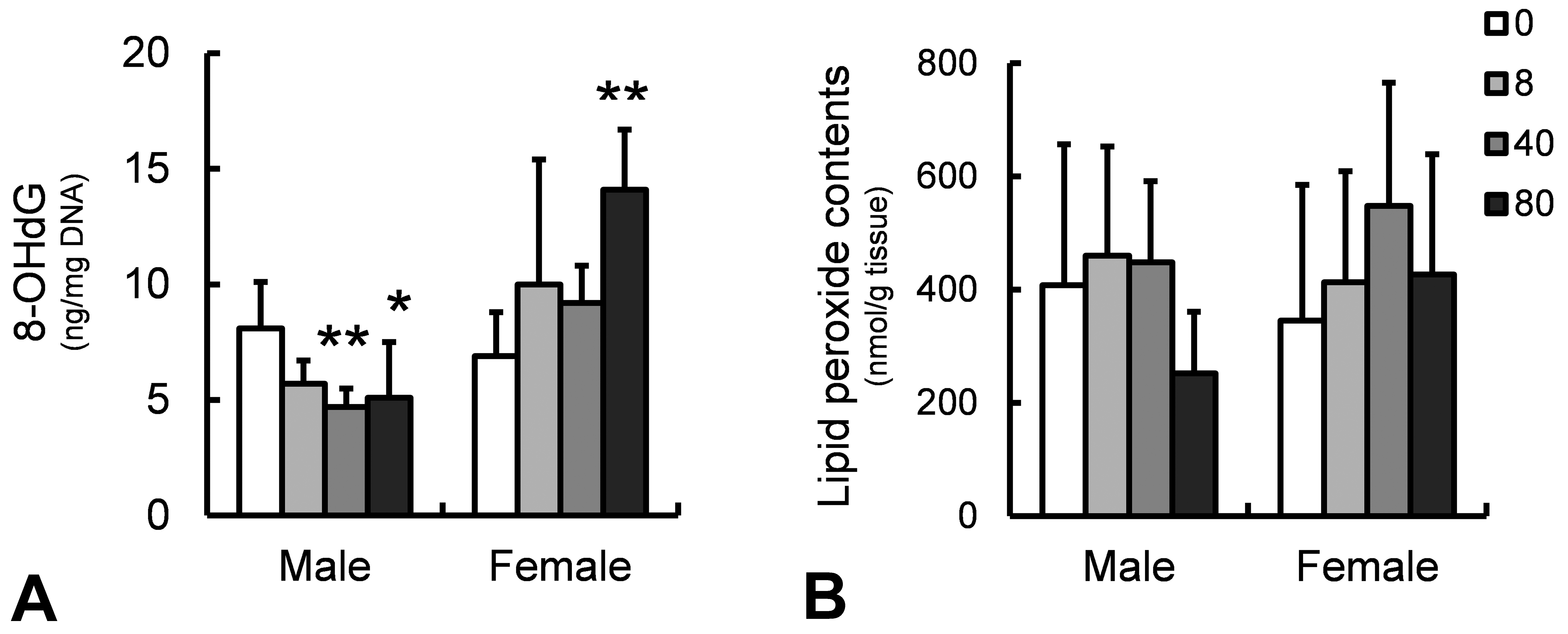
Determination of oxidative stress markers in rats treated with CCA at 0, 8, 40, and 80 mg/kg/day for 4 weeks. 8-OHdG (A) and lipid peroxide contents (B) in liver samples. *p ≤ .05 or **p ≤ .01 versus control group by Dunnett’s multiple comparison test. CCA = chromated copper arsenate, 8-OhdG = 8-hydroxydeoxyguanosine.
Discussion
The present toxicity study of orally administered CCA mainly provided evidence of specific clinical signs, low body weight gain and food consumption, clinical pathological toxicities, including hypochromic and microcytic anemia, hepatic and renal dysfunction, changes in lipid and Gluc levels, and pathologies of the gastrointestinal, hepatic, renal, and immune systems in rats. The effects in males were severer than those in females because the two highest doses for the males showed decreased body weight gain greater than 10%, which makes these doses in excess of the MTD. However, the toxic profiles in both males and females were similar to each other, and these changes were plausibly induced by the toxicity of an individual heavy metal or by their combined effects. Increases in the concentration of circulating total Cr and As in plasma samples indicated that both metals contribute to systemic toxic effects in CCA-treated rats. Oral administration of As(V) or As(III) causes accumulation of those metals and their methylated metabolites in the liver, kidney, and other organs (Hughes et al. 2003; Kenyon, Del Razo, and Hughes 2005; Rodríguez et al. 2005). A major part of absorbed As(V) is rapidly reduced to As(III), which is more reactive than As(V), mainly in the blood (Vahter 2002). As(III) is oxidatively methylated by the addition of a methyl group (MMA[V]) and is reduced to trivalency MMA(III), further repeated the similar methylation reaction, and then metabolized to dimethylated arsenic (DMA[V] and DMA[III]), which is the most active form (Tapio and Grosche 2006; Yamanaka et al. 2004). These methylation process of As varies between species. Humans excrete relatively more MMA(V) into the urine than other species, suggesting that humans are slower methylators of As (Hughes 2006). Rats are very efficient in methylating As to DMA, but most of the produced DMA is retained in the erythrocytes, resulting in a slow rate of excretion in the urine (Vahter 2002). Considering the species difference in metabolic process is extremely important in extrapolating toxic effects of the test substance to humans. For Cr, reductive metabolism of Cr(VI) generates intermediate Cr(V) and Cr(IV) forms and finally yields thermodynamically stable Cr(III) (Carter 1995; Salnikow and Zhitkovich 2008).
During these metabolic processes, Cr and As generate oxidative stress, leading to DNA and protein damage, and cellular responses. To explore the extent of oxidative stress, we measured 8-OHdG levels and lipid peroxide contents in liver samples, which showed that 8-OHdG levels were increased by treatment with CCA at 80 mg/kg/day in females. Hepatic oxidative stress has been known to be strongly induced in As- or Cr-exposed rats (Bashir et al. 2006; Bhadauria and Flora 2007; Patlolla et al. 2009; Scibior and Zaporowska 2007; Wang et al. 2006). Treatment with some antioxidants prevented As-induced oxidative stress and liver injuries in mice (Santra et al. 2007) and rats (Gupta and Flora 2006). Reduced levels of 8-OHdG and lower lipid peroxide content in CCA-treated males could be associated with adaptive induction of other antioxidant genes such as superoxide dismutase and catalase, as reported in Cr(VI)-treated rats (Patlolla et al. 2009). We only observed marginal effects in the liver in the present study; however, hepatotoxicity, as suggested by the results of blood biochemistry (hypoproteinemia and increases in GGTP, T.Bil, AST, and ALT) and pathological examinations in the present study, tended to be greater in females than in males. Therefore, it is suspected that oxidative stress may be involved in hepatotoxicity induced by CCA, as observed in that by As (Abu El-Saad, Al-Kahtani, and Abdel-Moneim 2016). To elucidate the mechanism on hepatotoxicity, additional molecular pathological analyses are being conducted in our institute. The liver is also a target organ of Cu (Schafer et al. 1999), but it remains unknown how Cu may be involved in hepatotoxicity when CCA is administered in rats.
Hematological assessment revealed that males and females receiving 80 mg/kg/day and females receiving 40 mg/kg/day of CCA showed hypochromic and microcytic anemia, where small-sized mature erythrocytes and reticulocytes with a lower level of hemoglobin circulate in the peripheral blood, by assessing CHCM and Chr. The parameter CHCM is the mean Hb measured directly from individual red cells. It is not affected by error in the measurement of Ht and Hb (Saw and Tham 1988). The present study clearly showed that CHCMm and Chr levels were reduced by CCA administration, indicating that CCA impairs hemoglobin synthesis and erythrocyte maturation, which might be caused by poor iron utilization. The Chr parameter directly assesses the incorporation of iron into erythrocyte hemoglobin, free from biological variations that affect iron, unsaturated iron binding capacity, transferrin measurements, and transferrin saturation (Goodnough, Skikne, and Brugnara 2000; Thomas and Thomas 2002; Ullrich et al. 2005). Therefore, Chr is a useful early indicator of iron-deficient erythropoiesis, and therefore, we did not measure plasma iron levels in the present study. In another report, a 13-week drinking study showed that exposure to Cr(VI) induced hematological signs of microcytic anemia based on iron deficiency in rats (Suh et al. 2014). This change was closely associated with an increase in iron ion oxidation by Cr(VI) in the lumen of the small intestine, higher expression of duodenal transporters, divalent metal transporter 1 (DMT1, Slc11a2) and transferrin receptor (TFR1, Tfr1), and intracellular absorption of Cr(VI) into erythrocytes, impairing iron absorption and iron binding to heme. Recent understanding of the molecular basis of intestinal iron absorption reveals that DMT1 plays a pivotal role in iron uptake from both transferrin and nontransferrin sources, and mutant variants display hypochromic and microcytic anemia by having decreased iron uptake in intestinal cells (Canonne-Hergaux et al. 2001; Fleming et al. 1998). Our data strongly suggest that Cr might induce anemia through small intestinal dysfunction as shown by histopathological examination. Another supportive piece of evidence is that DMT1 functions as a transporter for heavy metals including iron and Cu (Wood and Han 1998). Excessive Cu exposure through oral administration of CCA might also impair iron transport in the small intestine. On the other hand, the most common cause of iron deficiency is blood loss. As described later, various histopathological findings were observed in the gastrointestinal tract in the present study, and sinus erythrocytosis indicating hemorrhage from the gastrointestinal tract was noted concurrently in the mesenteric lymph nodes. Especially, forestomach is likely to be the biggest source of bleeding because we used gavage application with CCA, an irritating substance. Therefore, hemorrhage may have been involved in iron deficiency anemia. However, in females of the 40 mg/kg/day group in which similar anemia was observed, no histopathological change was observed in the forestomach, suggesting that hemorrhage from the gastrointestinal tract is not main cause of CCA-induced anemia. Further studies are needed to clarify direct and indirect mechanisms of the anemia.
Gastrointestinal changes were clearly demonstrated by histopathological examinations. The effects may be primarily due to Cr. Cr is a carcinogen, which targets the oral cavity and the small intestines in rats and mice used in carcinogenesis studies, respectively (Stout et al. 2009). Cytotoxicity-induced regenerative hyperplasia is a key precursor event to carcinogenesis in the small intestine of mice (Haney Jr. 2015). In a 13-week study of rats, villous atrophy/blunting, single-cell necrosis, and histiocyte infiltration were observed in association with crypt epithelial hyperplasia (Cullen, Ward, and Thompson 2016). Villous necrosis and subsequent atrophy implied that the toxic effects of Cr lead to villous epithelial cell loss followed by compensatory regenerative hyperplasia in the crypt. The hypothesis was supported by the finding that Cr was distributed in villous cells but not in crypts as demonstrated by synchrotron-based X-ray fluorescence microscopy (Thompson et al. 2015). Toxicogenomic analysis also demonstrated that duodenal changes were associated with gene expression related to oxidative stress, cell death, immune response, and cell proliferation (Kopec et al. 2012). In the present case, villous necrosis and crypt epithelial hyperplasia were not clear in histopathological examination, indicating that villous damage and compensatory regenerative hyperplasia in the crypt induced by CCA treatment were slight compared to those seen in the above 13-week study with Cr. The difference might depend on study design, dose level, and treatment period. Hyperkeratosis of the forestomach and goblet cell hypertrophy in the rectum have not been reported to our knowledge. Hyperkeratosis of the forestomach was probably related to the irritant property of Cr, which affected the oral mucosal and tongue in rats (Stout et al. 2009). The toxicological significance of goblet cell hypertrophy in the rectum remains uncertain; however, it might also be related to the irritant property of Cr as it is eliminated in the distal part of the large intestine. Changes in leukocyte counts and sinus erythrocytosis in the mesenteric lymph nodes were suspected to be associated with the above gastrointestinal lesions induced by CCA. Further studies such as time-course observations in the gastrointestinal tracts are required to recognize the pathogenesis on intestinal toxicity by CCA.
Impairment of renal function by CCA administration was suggested by results of urinalysis, blood biochemistry (an increase in BUN and fluctuations in electrolytes), organ weight analysis, and histopathology. Abnormal redox homeostasis and an antioxidant response might contribute to CCA-induced kidney toxicity (Matos et al. 2013). Accumulating evidence suggests that CCA-induced renal toxicity could be caused by Cr and As. In a case report, a 46-year-old man who had worked for nine years as a cutter of stainless steel (and was thereby exposed to smoke containing Cr) exhibited chronic interstitial nephropathy with a high plasma Cr level (Petersen, Mikkelsen, and Thomsen 1994). When rats received a single injection of Cr(VI), nephrotoxicity developed, revealing altered proximal tubule function, decreased glomerular filtration, and distal segment dysfunction, part of which was prevented by α-tocophenol supplementation (Arreola-Mendoza et al. 2006). Distortion of renal antioxidant systems has been demonstrated via decreases in glutathione peroxidase, glutathione reductase, and catalase activities in kidney samples obtained from rats receiving a single injection of Cr(VI) (Pedraza-Chaverrí et al. 2005). As-induced renal toxicity has also been reported in rats (Nandi, Patra, and Swarup 2005, 2006) and mice (Kimura et al. 2005). Interestingly, NF-E2-related factor 2 (Nrf2)-mediated multidrug resistance-associated protein 1, a main transporter for As(III) efflux, is a critical mediator of renal toxicity by As(III) (Kimura et al. 2006). Although severe histopathological changes due to As (Kimura et al. 2005) or Cr (Pedraza-Chaverrí et al. 2005) exposure have been reported, our findings were restricted to lipofuscin deposition in proximal tubule cells. This discrepancy is probably dependent on study design, dose level, and treatment period.
We postulate that markedly high dose of CCA is highly toxic to multiple organ systems including the hematopoietic, gastrointestinal, hepatic, and renal systems; however, the data in dose levels in excess of the MTD might not be suitable to extrapolate to a risk assessment for human. These toxicities are plausibly mediated by each heavy metal and/or by their combined effects, as well as by oxidative stress induced by the mode of action of Cr and/or As. Further studies are required for a better understanding of the molecular toxicity profile of each organ/tissue.
Supplemental Material
Supplemental Material, DS1_TPX_10.1177_0192623318765392 - Pathological and Clinical Pathological Changes Induced by Four-week, Repeated-dose, Oral Administration of the Wood Preservative Chromated Copper Arsenate in Wistar Rats
Supplemental Material, DS1_TPX_10.1177_0192623318765392 for Pathological and Clinical Pathological Changes Induced by Four-week, Repeated-dose, Oral Administration of the Wood Preservative Chromated Copper Arsenate in Wistar Rats by Naofumi Takahashi, Toshinori Yoshida, Sayuri Kojima, Satoru Yamaguchi, Ryoichi Ohtsuka, Makio Takeda, Tadashi Kosaka, and Takanori Harada in Toxicologic Pathology
Supplemental Material
Supplemental Material, TS1_TPX_10.1177_0192623318765392 - Pathological and Clinical Pathological Changes Induced by Four-week, Repeated-dose, Oral Administration of the Wood Preservative Chromated Copper Arsenate in Wistar Rats
Supplemental Material, TS1_TPX_10.1177_0192623318765392 for Pathological and Clinical Pathological Changes Induced by Four-week, Repeated-dose, Oral Administration of the Wood Preservative Chromated Copper Arsenate in Wistar Rats by Naofumi Takahashi, Toshinori Yoshida, Sayuri Kojima, Satoru Yamaguchi, Ryoichi Ohtsuka, Makio Takeda, Tadashi Kosaka, and Takanori Harada in Toxicologic Pathology
Supplemental Material
Supplemental Material, TS2_TPX_10.1177_0192623318765392 - Pathological and Clinical Pathological Changes Induced by Four-week, Repeated-dose, Oral Administration of the Wood Preservative Chromated Copper Arsenate in Wistar Rats
Supplemental Material, TS2_TPX_10.1177_0192623318765392 for Pathological and Clinical Pathological Changes Induced by Four-week, Repeated-dose, Oral Administration of the Wood Preservative Chromated Copper Arsenate in Wistar Rats by Naofumi Takahashi, Toshinori Yoshida, Sayuri Kojima, Satoru Yamaguchi, Ryoichi Ohtsuka, Makio Takeda, Tadashi Kosaka, and Takanori Harada in Toxicologic Pathology
Supplemental Material
Supplemental Material, TS3_TPX_10.1177_0192623318765392 - Pathological and Clinical Pathological Changes Induced by Four-week, Repeated-dose, Oral Administration of the Wood Preservative Chromated Copper Arsenate in Wistar Rats
Supplemental Material, TS3_TPX_10.1177_0192623318765392 for Pathological and Clinical Pathological Changes Induced by Four-week, Repeated-dose, Oral Administration of the Wood Preservative Chromated Copper Arsenate in Wistar Rats by Naofumi Takahashi, Toshinori Yoshida, Sayuri Kojima, Satoru Yamaguchi, Ryoichi Ohtsuka, Makio Takeda, Tadashi Kosaka, and Takanori Harada in Toxicologic Pathology
Supplemental Material
Supplemental Material, TS4_TPX_10.1177_0192623318765392 - Pathological and Clinical Pathological Changes Induced by Four-week, Repeated-dose, Oral Administration of the Wood Preservative Chromated Copper Arsenate in Wistar Rats
Supplemental Material, TS4_TPX_10.1177_0192623318765392 for Pathological and Clinical Pathological Changes Induced by Four-week, Repeated-dose, Oral Administration of the Wood Preservative Chromated Copper Arsenate in Wistar Rats by Naofumi Takahashi, Toshinori Yoshida, Sayuri Kojima, Satoru Yamaguchi, Ryoichi Ohtsuka, Makio Takeda, Tadashi Kosaka, and Takanori Harada in Toxicologic Pathology
Footnotes
Acknowledgments
Authors would like to thank Ms. Yuko Chiba, Mutsumi Kumagai, Chizuko Tomiyama, Takako Kazami, Yukie Sakano, Kayoko Iijima, and Mr. Satoshi Akema for tissue preparation. Authors would also like to thank Mr. Junya Sasaki and Ms. Mariko Tomita for carrying out the clinical pathology studies. Authors specially thank Dr. Nobuaki Nakashima, Maki Kuwahara, and Yukiko Kashimoto.
Author Contribution
Authors contributed to conception or design (NT, TY, TK, TH); data acquisition, analysis, or interpretation (NT, TY, SK, SY, RO, MT, TK, TH); drafting the manuscript (NT, TY); and critically revising the manuscript (NT, TY, SK, SY, RO, MT, TK, TH). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant in aid of research on risk assessment of wood preservatives from the Ministry of Health, Labour and Welfare, Japan.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
