Abstract
Chronic progressive nephropathy (CPN) occurs commonly in rats, more frequently and severely in males than females. High-grade CPN is characterized by increased layers of the renal papilla lining, designated as urothelial hyperplasia in the International Harmonization of Nomenclature and Diagnostic Criteria classification. However, urothelium lining the pelvis is not equivalent to the epithelium lining the papilla. To evaluate whether the epithelium lining the renal papilla is actually urothelial in nature and whether CPN-associated multicellularity represents proliferation, kidney tissues from aged rats with CPN, from rats with multicellularity of the renal papilla epithelium of either low-grade or marked severity, and from young rats with normal kidneys were analyzed and compared. Immunohistochemical staining for uroplakins (urothelial specific proteins) was negative in the papilla epithelium in all rats with multicellularity or not, indicating these cells are not urothelial. Mitotic figures were rarely observed in this epithelium, even with multicellularity. Immunohistochemical staining for Ki-67 was negative. Papilla lining cells and true urothelium differed by scanning electron microscopy. Based on these findings, we recommend that the epithelium lining the papilla not be classified as urothelial, and the CPN-associated lesion be designated as vesicular alteration of renal papilla instead of hyperplasia and distinguished in diagnostic systems from kidney pelvis urothelial hyperplasia.
Based on the general view that the mucosal surfaces of the collecting ducts, calyces, pelvis, ureter, bladder, and urethra all have the same embryologic origin, it is widely held that the term urothelium applies to the lining epithelium of all of these parts of the kidney and lower urinary tract (Miyazaki and Nishiyama 2017), including the lining of the renal papilla. However, at a more detailed level, the collecting system (collecting ducts, calyces, pelvis, and ureter) develops by successive branching of the distal portion of the ureteric bud. In contrast, the urothelial lining of the bladder and urethra derive from the cloacal endoderm via the urogenital sinus (Song and Yosypiv 2011). The epithelium lining the outer surface of the renal papilla is an extension of the cuboidal epithelium of the distal collecting ducts, which under normal conditions, is a single-cell layer. The true kidney pelvis begins at the fornices of the kidney papilla and transitions rapidly from a single-cell layer to a multilayered, stratified epithelium with the typical appearance of urothelium (Korshid and Moffat 1974; Silverblatt 1974). The urothelium is highly specialized, with a single basal layer, one to a few layers of intermediate cells, and superficially, characteristic umbrella-like cells that are large, thin, and polygonal (Hicks, Ketterer, and Warren 1974; Pauli, Alroy, and Weinstein 1983). Tight junctions hold the superficial cell layer together as a major barrier to the urine. The luminal membrane of the urothelial superficial cells is composed of a series of plaques and ridges. Ultrastructurally, the luminal membrane is typified by an asymmetric unit membrane and, internally, by fusiform vesicles (Hicks, Ketterer, and Warren 1974; Pauli, Alroy, and Weinstein 1983). The luminal membrane of the urothelium consists of highly specialized, protective plaques, composed of uroplakins, which are membranous glycoproteins unique to urothelium (Jacobs et al. 1976; Wu et al. 2009; Lee 2011). Although the urothelium by light microscopy and the luminal surface ultrastructurally is similar from kidney pelvis to urethra, the uroplakin content differs along with its expandability, reflecting the different embryonic origins of the different parts of the urinary tract (Wu et al. 2009).
In contrast to this typical multicellular layering of the urothelium, the lining epithelium of the papilla is a single layer of regularly shaped cuboidal cells lacking an asymmetric unit membrane and fusiform vesicles (Korshid and Moffat 1974). By scanning electron microscopy (SEM), the cells lining the renal papilla have a luminal surface showing microvilli and microplicae, not the plaques and peaked ridges of urothelium (Carroll et al. 1974; Andrews and Porter 1974; Bulger, Siegel, and Pendergrass 1974). Also, there is no evidence of single cilia typically seen on the luminal surface of collecting duct cells (Carroll et al. 1974; Andrews and Porter 1974; Bulger et al. 1974). This particular mucosal lining is relevant to an emerging controversy that has regulatory implications for the risk assessment of chemicals exacerbating chronic progressive nephropathy (CPN), a common renal disease in rats (Hard, Johnson, and Cohen 2009; Hard et al. 2011, 2013). CPN is a recognized confounder of renal pathology interpretation in subchronic and chronic toxicity bioassays (Wolf and Mann 2005), particularly in male rats, which are more severely affected than females. One of the many morphologic changes that typify advanced CPN in chronic bioassays is the appearance of multicellularity of the renal papilla lining, which has traditionally been diagnosed as urothelial hyperplasia (Frazier et al. 2012) or transitional epithelial (cell) hyperplasia (National Toxicology Program 2007). In this communication, we question the classification of this lesion regarding both urothelial and hyperplastic, based on light microscopic, scanning electron microscopic, and immunohistochemical investigations.
Materials and Method
Animals for Light Microscopic Evaluation of Serial Sections of End-stage CPN
Hematoxylin and eosin (H&E)-stained serial sections of left and right kidneys (sectioned at 5 µm intervals for a distance of 0.25 mm) were reexamined from four F344 male rats with end-stage CPN from a previous 2-year bioassay (Hard and Seely 2005) to trace the nature and extent of renal papillary lesions associated with CPN of advanced severity. For each of the 4 rats, one kidney was sectioned transversely, and the other was sectioned sagittally. This assessment was supported by more than 20 years of experience evaluating rat kidneys with advanced CPN.
Animals for Immunohistochemistry and SEM
Eight untreated male F344 rats from a previous study (Cohen et al. 1995) and seven untreated male Sprague-Dawley rats used as breeders in a previous protocol (unpublished) were used in the present study. The respective protocols were approved by the University of Nebraska Medical Center Institutional Animal Care and Use Committee. At sacrifice, F344 rats were 133 to 135 weeks old (aged rat group), and Sprague-Dawley rats were 10 to 15 weeks old (young rat group). All eight F344 rats and five of the seven Sprague-Dawley rats were used for immunohistochemistry; the other two Sprague-Dawley rats were used for SEM evaluation.
Study Design for Evaluation of Lining of Kidney Papilla
Kidney tissue was fixed in 10% phosphate-buffered formalin, dehydrated, and embedded in paraffin by standard procedures. Kidney tissue from all of the aged F344 rats had a diagnosis of CPN, of which four had a severe grade (grade 4) CPN, accompanied by the typical CPN-related papilla lining alteration diagnosed previously as “urothelial hyperplasia.” The remaining four aged F344 rats had CPN of low-grade severity, usually grade 2, without the renal papilla lining alteration. All kidneys from the five young Sprague-Dawley rats were graded as normal, that is, CPN grade 0. The differentiation of the papilla lining epithelium in both young and aged male rats was assessed by immunohistochemical staining for uroplakins, proteins that are specific for urothelium (Wu et al. 2009). The proliferation rate of the renal papilla lining was assessed by Ki-67 immunohistochemical staining (Wood et al. 2015) and by evaluation of H&E-stained kidney tissue for the presence of mitotic figures.
Uroplakins and Ki-67 Immunohistochemistry
Kidney sections from young and aged rats were used for immunohistochemical detection of uroplakins or Ki-67. Urinary bladder and intestinal tissue were used as positive controls for uroplakins and Ki-67, respectively. The stratified, nonpapillary renal pelvis lining served as an internal control for uroplakins, and the tubules in the renal parenchyma served as an internal control for Ki-67 labeling. Briefly, after deparaffinization of the sections and exposure of the slides to 3% H2O2 for 20 min to quench endogenous peroxidase activity, heat-induced antigen retrieval was performed in 0.01 M citrate buffer, pH 6.0, using a pressure cooker for 5 min. One percent nonfat milk was used to block nonspecific staining. Sections were immunostained using polyclonal rabbit total bovine uroplakin antibody (gift from Dr. Tung-Tien Sun, NYU School of Medicine, New York, NY), diluted 1:200, or monoclonal mouse anti-rat Ki-67 antibody (Dako, Carpenteria, CA), diluted 1:25, overnight at 4°C. Secondary antibody incubation was performed either with anti-rabbit IgG (for uroplakin) or anti-mouse IgG (for Ki-67) diluted 1:200 for 1 hr followed by incubation using the avidin–biotin–peroxidase complex (ABC) for 45 min (Vectastain Elite ABC kit, Vector Laboratories, Burlingame, CA). Positive reactions resulted in brown cytoplasmic staining for uroplakins or brown nuclear staining for Ki-67 using DAB (3,3′-diaminobenzidine) as the substrate (DAB substrate kit, Vector Laboratories). Uroplakin and Ki-67 staining were assessed as negative or positive.
SEM Evaluation
Kidney tissue from two of the seven young Sprague-Dawley rats was fixed in Trump’s fixative, dehydrated through an ascending series of ethanol concentrations (EtOH; 50–100%) followed by 1:1 100% EtOH and hexamethyldisilazane (HMDS) and 100% HMDS. The tissue was coated with gold prior to examination by SEM, and analysis was performed using a Quanta 200 Scanning Electron Microscope (FEI Company, Hillsboro, OR, USA).
Results
Morphology of Renal Papilla Alteration in End-stage Rat Kidney
Of the four F344 rats with serially sectioned end-stage CPN-affected kidney, three of the four sagittal sections had no renal papilla in the section. One sagittal section had a detached stump representing the lower quarter of the papilla including the papilla tip. The four transverse sections all included some papilla, but in two cases, this comprised only the proximal third arising from the medulla. Two rats had a reasonable representation of the length of the papilla along with the distal third of papilla from a sagittal section; thus, three papillary specimens provided information on CPN-related papillary lesions.
The CPN-associated papilla lining lesions tended to involve the lower half of the papilla and not the upper half. If the papilla section included only the proximal third of the papilla (which is often the case in practice), no CPN-related papillary lesions were observed.
From the serial sections of the three papilla specimens, there were approximately 15 individual lesions that were tracked at 5-µm intervals in one direction from the point of sectioning. In contrast to the normal smooth epithelial lining of the papilla and the smooth multilayered urothelium of the kidney pelvis (Figure 1A), the papilla lining lesions were small, blister-like outpouchings or vesicular hollow protrusions from the lining into the pelvis lumen (Figures 1B). The lining epithelium of many of these vesicles retained the appearance of a single-cell layer (Figures 1C–E), but in some, multicellularity appeared to be associated with collapse of the vesicle, or was a result of tangential sectioning of the vesicle wall (Figure 1E and F). Tracking along the serial sections, many lesions became complex and multilocular, but in all cases, the lesion eventually disappeared or remained a simple vesicle. There was no progression beyond this vesicular appearance and no evidence of proliferative hypercellularity. As the edge of a disappearing vesicle was approached, the vesicle wall assumed a multicellular, solid appearance, but this was due to tangential sectioning of a curved surface. The vesicles were devoid of histologically identifiable contents, except for very pale wisps of structureless material, a scattering of small dense nuclei, and a rare macrophage or fibroblast. In one vesicular lesion, there was hemorrhage, which persisted through many serial sections. An occasional mitotic figure was observed in the papilla lining, but rarely in the vesicle walls. Also, there was no evidence of inflammation in any papillary lining lesion.
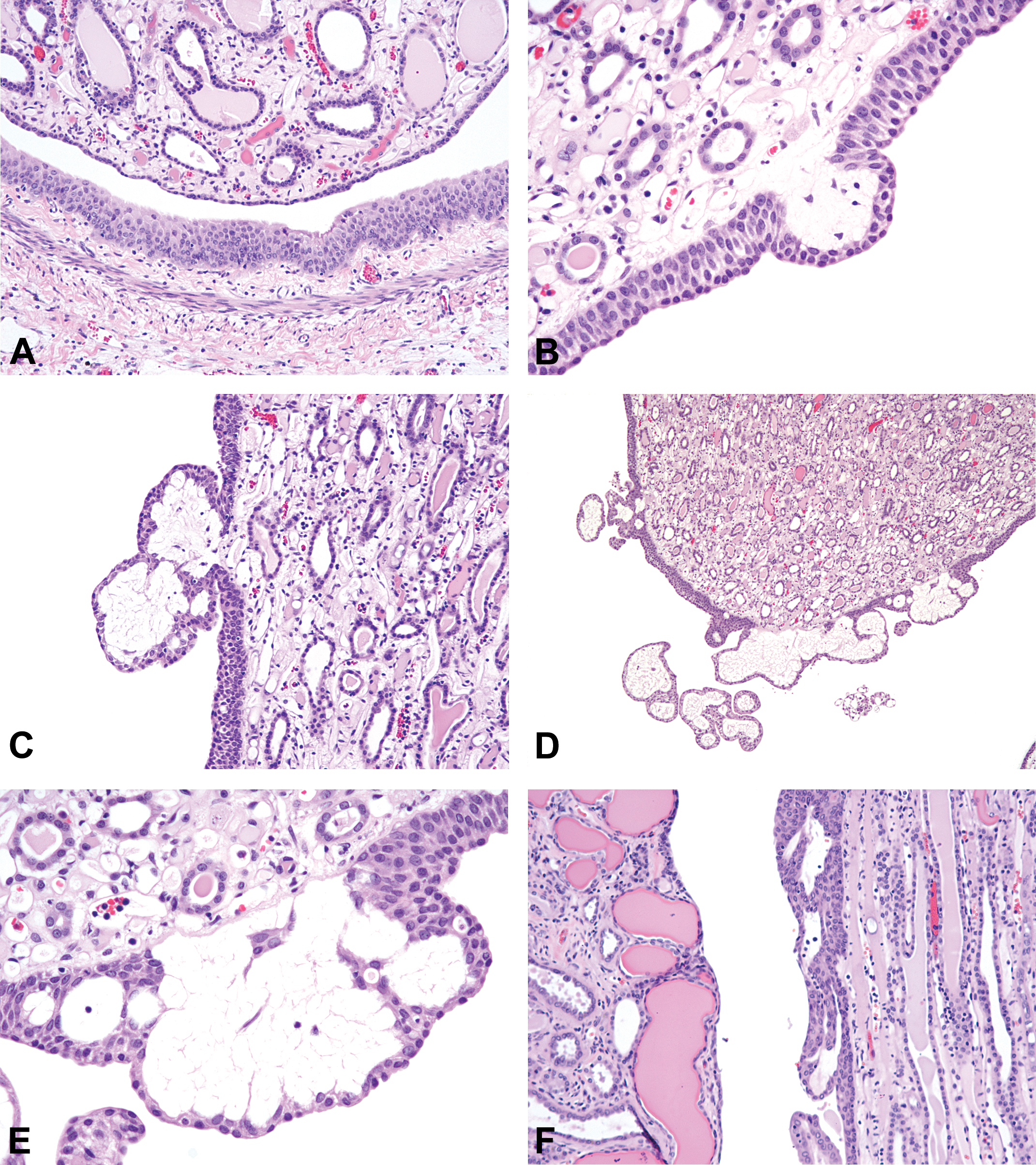
(A). Renal papilla (top) with single-cell layer lining epithelium contrasted to the multilayered true kidney pelvis urothelium (bottom). (B) At their edge, chronic progressive nephropathy (CPN)-associated lesions commence and end as a blister-like or vesicular, outpouching of the papilla lining. The apparently thickened papilla lining is likely accentuated by tangential sectioning. (C) A papillary lesion becoming complex, with two apparent vesicles. The loose interior of each vesicle is structureless, containing a reticulum of wispy, poorly staining material. The epithelial lining is a single layer, with multiple layers extending to the adjoining papilla. (D). Typical complex vesicular lesion at the papilla tip consisting of several vesicular profiles, most lined by a single epithelial layer. (E) Higher magnification of part of the lesion in (D). The outer lining of the vesicular outpouching retains the single-cell layer, but the basal epithelium appears thickened, probably due to tangential sectioning. The vesicle interior is a loose network of structureless wispy material or collapse of vesicles. (F) Papilla lining lesion where the vesicle outpouching has collapsed to give an appearance of multicellularity. Cortex of kidney showing CPN is on the left.
Uroplakin Staining
The epithelial cells lining the nonpapillary parts of the renal pelvis were positive for cytoplasmic uroplakin staining in all kidney samples. No cytoplasmic uroplakin staining was observed in the epithelium lining the length of the papilla in any of the rats, whether young or old, or whether affected by advanced CPN papilla lining lesions or not (Figure 2A–C).
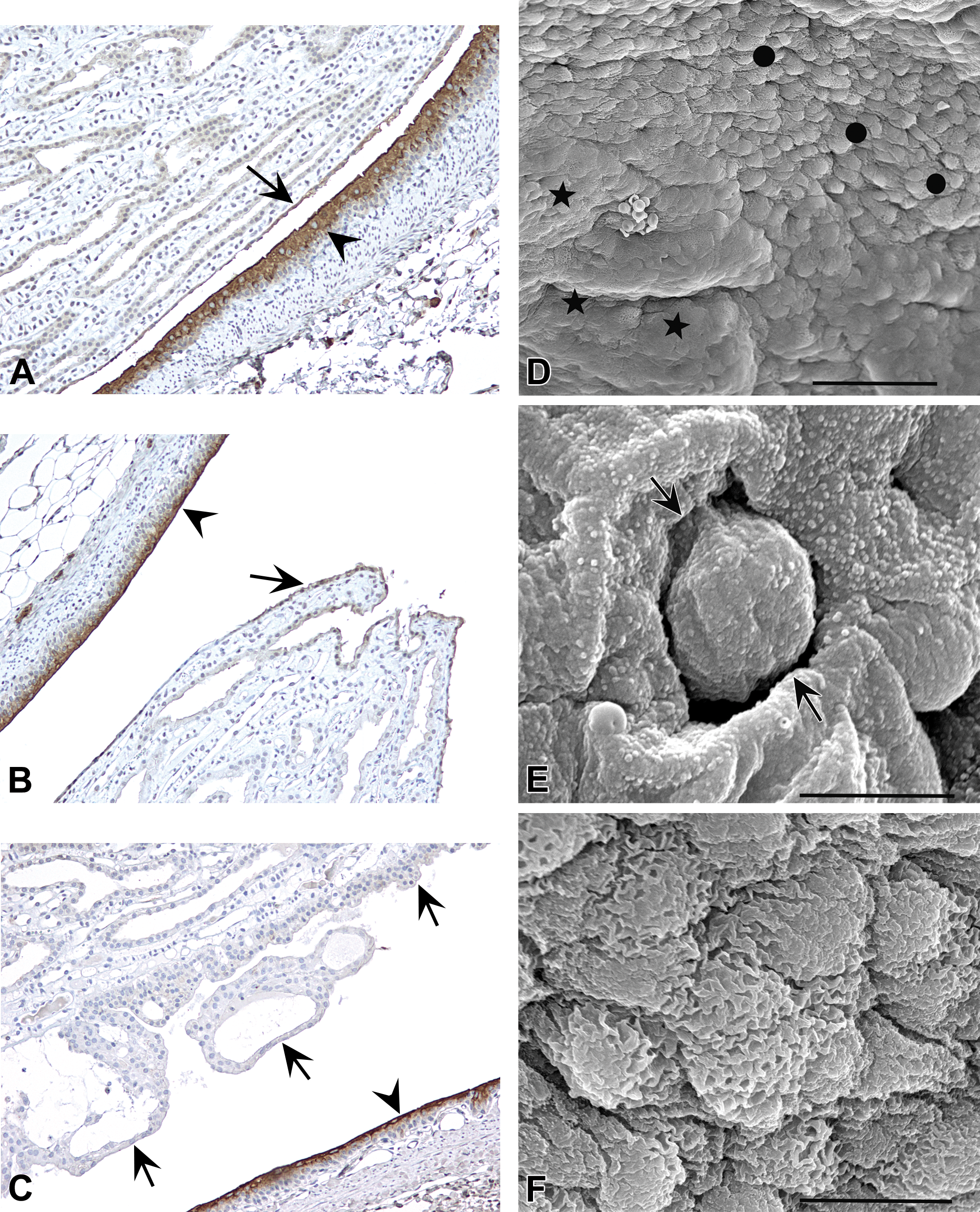
(A) Immunohistochemical staining of a kidney of a young rat without chronic progressive nephropathy (CPN) for uroplakins, showing strong staining of true kidney pelvis urothelium (arrowhead) but lack of staining for the epithelial lining of the renal papilla (arrow) and collecting duct epithelium. Slight edge effect staining was noted within the apical aspect of the renal papilla epithelium and occasionally in the collecting ducts, but no cytoplasmic staining was observed. (B) Immunohistochemical staining of a kidney of an old rat with CPN but without vesicular lesion of renal papilla, showing strong staining of true kidney pelvis urothelium (arrowhead) but lack of staining for the epithelial lining of the renal papilla (arrow) and collecting duct epithelium. (C) Immunohistochemical staining of a kidney of an old rat with CPN but with vesicular lesion of renal papilla, showing strong staining of true kidney pelvis urothelium (arrowhead) but lack of staining for the epithelial lining of the renal papilla (arrows) and collecting duct epithelium. Epithelial lining of vesicular lesions does not stain. (D) Scanning electron microscopy (SEM) of luminal surface at fornix showing large polygonal cells (stars) of the true urothelium irregularly merging with smaller cells of the papilla lining epithelium (circles). (E) SEM of papilla with opening of collection duct (arrows). Cells lining the papilla and collecting duct have numerous short, uniform microvilli, with no evidence of cilia. (F) SEM of cells lining the papilla showing microplicae on the surface.
Ki-67 Staining and Mitotic Figures
No nuclear staining was observed for Ki-67 in the cells lining the renal papilla in any of the rats including those with vesicle-like outpouchings of the papilla lining and collapsed, multicellular forms. Acting as a positive control, a few Ki-67 positive cells were observed in the proximal tubules of the cortex and in the pelvic urothelium and rarely in the collecting ducts. No mitotic figures were observed in the renal papilla lining of any of the rat kidneys used for immunohistochemical investigation, nor were any observed in the papilla lining cells in the corresponding H&E-stained sections.
SEM Evaluation
SEM of the kidney pelvis showed the typical structure of the luminal surface of the large urothelial superficial cells with plaques and peaked microridges (Jacobs et al. 1976). At the fornix, there was an abrupt, albeit irregular transition to the type of cells lining the renal papilla (Figure 2D), which were considerably smaller and cuboidal to polygonal in shape with microvilli and/or microplicae (Figures 2E-F; Bulger et al. 1974). We did not observe cilia on any cells lining the papilla nor did we detect cilia on the lining cells of the collecting ducts as they opened onto the pelvic space (Figure 2E).
Discussion
There is an absence of immunohistochemical staining for uroplakins of the epithelium lining the renal papilla, and the luminal surface cells lining the papilla do not share the appearance of superficial urothelial cells, as observed by SEM. Thus, the lining epithelium of the rat kidney papilla is clearly not urothelium. The appearance of the cells lining the renal papilla is cuboidal by light microscopy and has a luminal surface distinct from the true urothelium as viewed by SEM. Based on these observations, the lining epithelium of the rat kidney papilla needs to be categorized separately from the true urothelium of the kidney pelvis lining in classification schemes and in histopathology evaluations of kidney. We recommend that the nomenclature in International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) for the single-cell lining of the renal papilla be specified as cuboidal lining of renal papilla, as has been previously described in Cohen (2013) and Greaves (2012). The present INHAND site clearly distinguishes between papilla and pelvis but is not clear as to the lining epithelium of the papilla being urothelial or not. Furthermore, in practice, the lesion on the papilla associated with CPN is commonly referred to as renal pelvic urothelial (transitional cell) hyperplasia.
It is unclear whether the cells lining the papilla are an extension of the collecting duct lining cells, as we did not observe cilia on the papilla lining cells. However, the general features of the cells that we could observe by SEM in the collecting ducts appeared similar to those lining the papilla. Although most epithelial cells lining the collecting ducts proximal to their connection to the papilla lining epithelium, have a single cilium, some cells do not have a cilium but have short microvilli and microplicae (Carrroll et al. 1974; Bulger et al. 1974; Andrews and Porter 1974) such as what we observed of the epithelium lining the papilla and the adjoining collecting duct epithelium.
The distinction between this lining epithelium and the true urothelium is critical in the assessment of toxicologic pathology evaluations. The types of toxicities occurring in the true urothelium of the kidney pelvis are distinct from those affecting the glomeruli, tubules, or collecting ducts but are similar to those occurring in the urothelium of the ureters and urinary bladder. However, toxic responses affecting the urothelium of the urinary bladder tend to be more frequent and more severe compared to the urothelial lining of the renal pelvis and ureters, probably due to the storage function of the bladder, resulting in extended exposure to urine (Cohen 1998; Cohen et al. 2007; Wu et al. 2009).
Serial sectioning of rat kidney illustrates the importance of including the length of the papilla in the histological specimen, as CPN-related papilla lining alterations affected mainly the distal two-thirds of the papilla. Tracking along serial sections of papilla demonstrated that the lesions were vesicular and did not transition into the solid or proliferative character typical of hyperplasia. They remained vesicular until they disappeared from the section, except in cases of lining collapse. This lesion was distinct from the hyperplastic papilla lining induced by potassium bromate, which was a multicellular thickening of the lining projecting into the pelvis lumen as papillary fronds or sessile mats (Wolf et al. 1998). In contrast, CPN-associated papilla lining alteration was consistently vesicular in nature without morphological progression to a proliferative state. By light microscopy, only rare mitotic figures were observed, and immunohistochemistry with Ki-67 antibody, a characteristic marker for DNA replication (Wood et al. 2015), revealed an absence of cell proliferation. In addition, there was no inflammatory component to this unique lesion.
The distinction between histopathologic alterations of the lining epithelium of the papilla and those of the urothelial lining of the kidney pelvis is of more than academic interest, because the papillary lesion associated with advanced CPN is currently classified by regulatory and authoritative agencies as urothelial hyperplasia or transitional cell hyperplasia. The term transitional cell hyperplasia was also used in the latest version of the INHAND classification system of histopathologic changes in rodent kidney (Frazier et al. 2012) when referring to this lesion under the heading of CPN and was also used by Hard and Khan (2004) in the latest review of CPN. Because of this inaccurate nomenclature, the CPN-related renal papilla lesion has been regarded sometimes as a distinct form of toxic response with proliferative potential, separate from CPN. It is a part of CPN, and more importantly, like CPN overall, this vesicular lesion has not been described in humans (Taal et al. 2012; Jennette et al. 2015). This lesion appears to be unique to high grades of CPN in the rat.
By definition, the term hyperplasia denotes an abnormal increase in the number of cells in a tissue or organ lining. However, it connotes a proliferative lesion. The vesicular morphology, the lack of any conspicuous proliferative activity, and the lack of progression of this renal papilla alteration to more advanced lesions, including historically an absence of neoplastic development, indicates that this is not a true hyperplastic response. Based on these differences, we recommend that the nomenclature for this renal papilla lesion be revised and referred to as vesicular alteration of renal papilla lining and that it be specified separately from kidney pelvis and separate from urothelial. In the INHAND listing for this entity (to INHAND Global Editorial and Steering Committee; Frazier et al. 2012), we recommend the following: organ: kidney, specific site: renal papilla, and descriptor: vesicular alteration of papilla lining.
Footnotes
Acknowledgment
The authors wish to acknowledge the support of the Fred and Pamela Buffett Cancer Center Tissue Sciences Facility Shared Resource, supported by the National Cancer Institute under award number P30 CA036727.
Author Contribution
All authors (NS, GH, LA, RF, KP, SC) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Fred and Pamela Buffett Cancer Center support grant (P30CA036727).
