Abstract
The purpose of this study was to establish a 2-stage model of urinary bladder carcinogenesis in male Sprague-Dawley rats to identify tumor promoters. In phase 1 of the study, rats (
Keywords
Rodent lifetime bioassays are routinely employed in the pharmaceutical and chemical industries to assess the carcinogenic potential of compounds in development. These studies are designed to detect both direct and indirect tumor initiators, but they are also able to detect tumor promotion and progression activity if a compound-related increase in the incidence, or a shortening in the latency to onset, of spontaneous background tumors is observed. In addition to these rodent lifetime bioassays where a single compound is assessed, there has been interest by regulatory agencies (Food and Drug Administration, European Medicines Agency, etc.) in conducting nonclinical studies specifically designed to assess the tumor-promoting activity of drugs following administration of a tumor-initiating compound (Buckley, Silva-Lima, and Tirmenstein 2015). Recently, a health authority requested that we conduct a 2-stage tumor promotion study in rats to assess the capacity of our development drug to promote urinary bladder tumors and compare it to a known urinary bladder tumor promoter that served as a positive control.
The 2-stage tumor promotion model has been used to examine tumor promotion in several different tissues including skin, liver, stomach, urinary bladder, and pancreas (Cohen 1998). Although there are numerous manuscripts in the literature describing the 2-stage tumor promotion assay in academic settings, it is not widely used in drug development. In these models, rodents are first dosed with a tumor initiator, to induce mutations in the DNA of the target cell population, followed by dosing of the putative tumor promoter or a vehicle control. In most cases, the initiating agent is a genotoxic agent, while the tumor promoter is usually nongenotoxic and acts epigenetically by increasing cellular proliferation and/or blocking apoptosis. Cell proliferation can occur in response to tissue injury and inflammation or as a more specific response to a mitogenic stimulus such as interaction of the chemical with nuclear receptors such as those of the peroxisome proliferator-activated receptor (PPAR)α or constitutive androstane receptor (CAR) types. The promotional phase of the model usually involves months of repeated dosing following a relatively short initiation phase. After several months of promoter treatment, the incidence and severity of tumors are scored and compared between rodents receiving the putative tumor promoter and those receiving the vehicle control. Compounds that significantly increase the incidence and severity of tumors over those treated with the vehicle control are classified as tumor promoters (Takahashi et al. 1992; Hagiwara et al. 1993).
Conceptually, the theory behind the 2-stage tumor promotion model is straightforward. However, the successful design of these studies is often challenging. There is considerable variation in how the 2-stage tumor promotion models are conducted with regard to types of initiating agents used; the dose of initiator and promoter administered; the timing of administration of initiators and putative promoters; time for the development of tumors (necropsy times); and differences in species, sex, and rodent strains used. These differences make standardization of the model for routine tumor promoter identification difficult.
The objective of the current study was to establish a 2-stage model of urinary bladder carcinogenesis in male Sprague-Dawley rats to screen for potential urinary bladder tumor promoters. We selected the compound N-butyl-N-(4-hydroxybutyl)-nitrosamine (BBN) as the urinary bladder tumor initiator, as it has been shown to specifically induce urinary bladder tumors in rodents. Studies have shown that BBN is metabolized to reactive metabolites that are subsequently transported in the urine to the urinary bladder where they covalently bind to DNA in the urothelium and initiate carcinogenesis (Vasconcelos-Nóbrega et al. 2012). In addition, studies have demonstrated that BBN can induce mitochondrial alterations leading to the opening of the mitochondrial permeability transition pore and subsequent oxidative stress in the liver of mice (Oliveira et al. 2013). Reactive oxygen species can damage DNA and thereby provide another mechanism for generating DNA mutations (Nakae et al. 1997), although it is not known if BBN also induces oxidative stress in the urinary bladder of rats.
To establish the 2-stage model, we initially conducted a dose-ranging study in male Sprague-Dawley rats to select the correct dose of BBN to induce tumor initiation. In this study, we also conducted immunohistochemistry (IHC) analysis to demonstrate that BBN increases markers of oxidative DNA damage (8-hydroxyguanosine) in rat urinary bladders. We then selected 3 compounds that have been demonstrated in the literature to act as urinary bladder tumor promoters via different mechanisms in rodents to assess in our model: rosiglitazone, sodium ascorbate, and uracil. Lubet et al. (2008) demonstrated that the PPAR gamma agonist, rosiglitazone, can function as a urinary bladder tumor promoter in rats by an unknown mechanism. Studies have also shown that dietary sodium
Material and Method
Animals
Studies were conducted at Charles River Laboratories (CRL), Senneville, Quebec, Canada. All studies were conducted in an Association for Assessment and Accreditation of Laboratory Animal Care International-accredited facility and in accordance with the guidelines of the USA National Research Council and the Canadian Council on Animal Care. All animal care and study procedures were approved by the CRL Institutional Animal Care and Use Committee prior to study conduct. Male Harlan Sprague-Dawley rats were obtained from Harlan Laboratories Inc., Indianapolis, IN, USA. At the start of dosing, male rats were 6 to 7 weeks old and weighed between 166 and 229 g. Rats were housed in groups of up to 3 rats per cage in stainless steel wire mesh floor cages. Animal room temperatures were maintained at temperatures of 19°C to 25°C and humidity at 30 to 70%. Animal rooms were on a 12-hr light/dark cycle. All rats had free access to water during the studies and, unless otherwise indicated, were fed Harlan Diet #2018C: Certified 18% Protein Diet ad libitum.
Chemicals
BBN (98.5% purity) was purchased from Tokyo Chemistry Industry Co., Ltd., Tokyo, Japan. The (+)-Sodium
Study Designs
BBN dose-ranging study
An initial BBN dose-ranging study was conducted in rats to determine the optimal dose of BBN for inducing urinary bladder tumors in the subsequent 2-stage tumor promotion study. Ninety-five male Sprague-Dawley rats were divided into 5 groups and dosed twice weekly by oral gavage with 0 (vehicle control;
BBN Dose-ranging Study: Incidence of BBN-related Microscopic Findings in Urinary Bladder during Week 24.
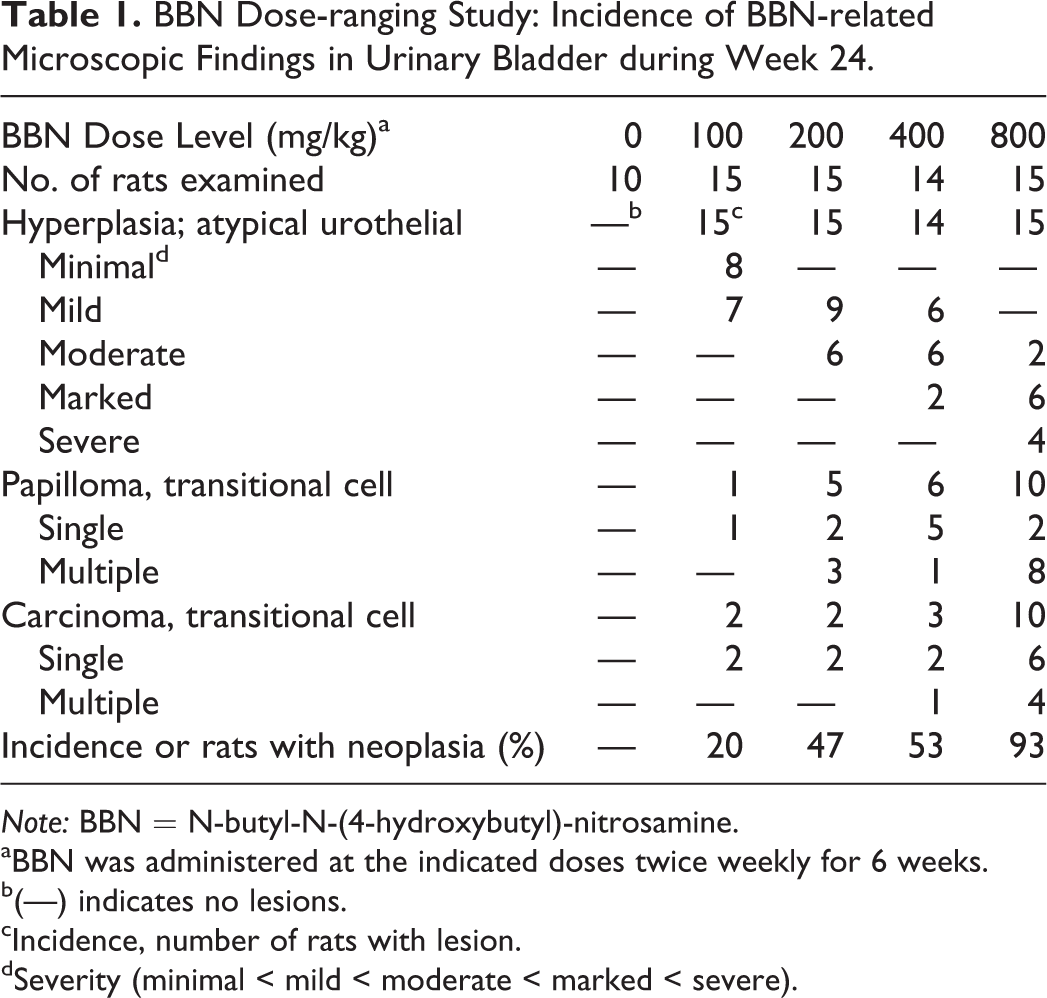
aBBN was administered at the indicated doses twice weekly for 6 weeks.
b(—) indicates no lesions.
cIncidence, number of rats with lesion.
dSeverity (minimal < mild < moderate < marked < severe).
Two-stage BBN tumor promotion study
In the first phase of the experiment, 170 male Sprague-Dawley rats were dosed twice weekly by oral gavage with 100 mg/kg BBN (dose selected from BBN dose-ranging study where a 20% incidence of neoplasms was observed and provided sufficient opportunity to assess a promoter-induced effect) in vehicle (20% ethanol in water) for a total of 6 weeks. A urinalysis was conducted during week 7 to assess the condition of the urinary bladder, and any rats with hematuria (diagnosed by a positive dipstick test or by microscopic evaluation of urine sediment) were removed from the study to eliminate rats with potential renal/urinary bladder injuries and/or preexisting urinary bladder tumors. These conditions would be expected to complicate the identification of urinary bladder tumor promoters. No gross or microscopic pathology evaluations were performed on rats that were eliminated from the study due to hematuria.
In the second phase of the experiment, beginning 8 weeks from the start of BBN dosing, 160 of the BBN-treated rats were divided into 4 groups of 40 rats/group receiving one of the following treatments for 26 weeks: (1) daily oral gavage with the rosiglitazone vehicle (90% PEG-400) at 5 ml/kg/day, (2) daily oral gavage with 25 mg/kg/day rosiglitazone (Lubet et al. 2008) in vehicle, (3) dietary 5% sodium ascorbate (Fukushima et al. 1988) formulated in Harlan Diet #2018C: Certified 18% Protein Diet, and (4) dietary 3% uracil (Shirai et al. 1986) formulated in Harlan Diet #2018C: Certified 18% Protein Diet. Rats administered rosiglitazone or vehicle received Harlan Diet #2018C: Certified 18% Protein Diet throughout the entire experiment with no additions. All surviving rats were necropsied 34 weeks after the initial dosing of BBN. The experimental design followed in the BBN 2-stage tumor promotion study is summarized in Online Supplemental Table 2.
BBN Dose-ranging Study: Immunohistochemistry of Urinary Bladders Collected at Week 14.
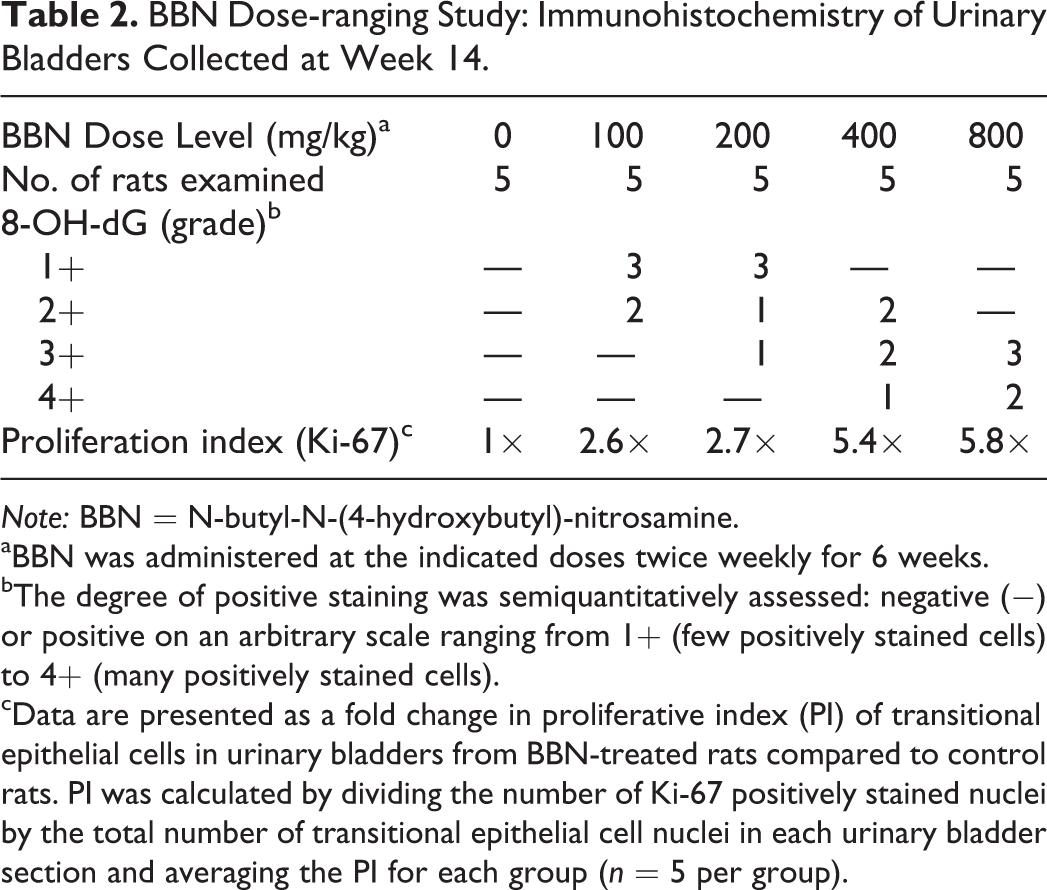
aBBN was administered at the indicated doses twice weekly for 6 weeks.
bThe degree of positive staining was semiquantitatively assessed: negative (−) or positive on an arbitrary scale ranging from 1+ (few positively stained cells) to 4+ (many positively stained cells).
cData are presented as a fold change in proliferative index (PI) of transitional epithelial cells in urinary bladders from BBN-treated rats compared to control rats. PI was calculated by dividing the number of Ki-67 positively stained nuclei by the total number of transitional epithelial cell nuclei in each urinary bladder section and averaging the PI for each group (
Mortality and Clinical Observations
Mortality/moribundity checks were performed twice daily. Clinical observations were recorded at least once weekly. Veterinary examinations were performed once pretreatment and as needed throughout the study.
Body weight and food consumption
Individual rat body weights were measured once weekly in the BBN dose-ranging study and twice weekly in the BBN 2-stage tumor promotion study. Food consumption was measured weekly in both studies.
Urinalysis
In the BBN dose-ranging study, urine was collected at room temperature during weeks 6 (at completion of the BBN dosing period), 10, 14, and 24 from individual rats placed in metabolism cages overnight. Rats were deprived of food but not water during the urine collection procedure. Urine was evaluated for color, appearance, urinary sediment, blood, and total volume.
During the BBN 2-stage tumor promotion study, urine was collected at 7 (prior to phase 2) and 10 weeks (during phase 2) from the start of the experiment. Urine samples were collected overnight in a container cooled on wet ice and stored refrigerated until analysis. Urine was evaluated for color, appearance, specific gravity, urinary sediment, blood, bilirubin, ketones, pH, and total volume. Urine chemistry measurements included creatinine, N-acetylglucosamine, protein, glucose, sodium, potassium, and chloride.
Pathology
In both the BBN dose-ranging and the 2-stage BBN tumor promotion studies, all rats that survived to their scheduled necropsies were fasted overnight prior to necropsies and euthanized by isoflurane anesthesia and subsequent exsanguination via the abdominal aorta. For rats that died on study, a necropsy was conducted and specified tissues were retained. Isoflurane anesthesia, and subsequent exsanguination, was also used in cases where rats were euthanized for humane reasons. All necropsies included gross examinations of the carcass, musculoskeletal system, all external surfaces and orifices, cranial cavity and external surfaces of the brain, and thoracic, abdominal, and pelvic cavities with their associated organs and tissues.
Organ weights
In the BBN 2-stage tumor promotion study, urinary bladders from all rats were weighed at necropsy. Prior to removal, the ventral serosal surface of the urinary bladder from the fundus to the neck was marked with permanent ink. Urinary bladders were then voided of urine by gentle compression, ligated at the neck near the origin of the urethra, and removed by cutting distal to the ligature. After removal, ligated urinary bladders were weighed as quickly as possible to avoid drying.
Microscopic Pathology
In both the BBN dose-ranging and the 2-stage BBN tumor promotion studies, representative samples of urinary bladders and all gross lesions (when possible) were collected. Ligated urinary bladders were removed, weighed, and gently infused with 10% neutral buffered formalin by injecting from a syringe through a narrow gauge needle at the cranial aspect of the fundus. Entire urinary bladders were then immersed in 10% formalin with ligature in place to maintain optimal inflation during fixation. The tissues were fixed for 1 to 2 weeks before processing. Prior to embedding, urinary bladders were bisected transversely into 2 approximately equal halves. Urinary bladders from all rats were embedded in paraffin, sectioned at approximately 4 to 6 μm thickness, mounted on glass slides, and stained with hematoxylin and eosin (H&E). Both halves were embedded in 1 block, so that initial sections were obtained from the cut surface. Urinary bladder blocks were then step-sectioned at approximately 0.5-mm intervals through the entire block for H&E staining, so that 6 to 10 slides (each with 2 sections of urinary bladder) were obtained. Histopathological evaluations were performed by a board-certified veterinary pathologist, and findings were subject to peer review. Criteria for grading of urothelial hyperplasia was based on section area affected as follows: minimal—less than 10%, mild—10 to 20%, moderate—20 to 40%, marked—40 to 70%, and severe—greater than 70% of section area affected.
IHC
In the BBN dose-ranging study, IHC to demonstrate oxidative DNA damage and cell proliferation was performed. Rat urinary bladders (
Analysis of Data
For quantitative numerical data where
Results
BBN Dose-ranging Study
Mortality and clinical observations
BBN administration was tolerated at all doses in the dose-ranging study. The only clinical observations observed in this study were dose-related increases in salivation (days 4 to 39) at ≥400 mg/kg BBN.
Food consumption and body weights
BBN had no effect on food consumption at any dose, but there was a transient decrease in body weights (3.4% and 6.3% decrease relative to controls at 400 and 800 mg/kg, respectively) during the dosing phase of the study (first 6 weeks of the study). Mean body weights had returned to control levels in all BBN dose groups by week 14.
Urinalysis
BBN had no effect on urinalysis parameters measured in the dose ranging study (data not shown).
Histopathology
At week 14, urothelial hyperplasia was observed in the urinary bladders of all rats treated with BBN (Figure 1A and B). TCC was only observed in 3 rats at this time point (2 of the 5 rats at 400 mg/kg [40%] and 1 of the 5 rats at 800 mg/kg [20%]). By 24 weeks, there was a dose-related increase in the incidence of transitional cell papilloma and TCC in urinary bladders across all BBN groups (Table 1). The incidence of neoplasms at 24 weeks was 0%, 20%, 47%, 53%, and 93% at 0, 100, 200, 400, and 800 mg/kg, respectively. Proliferative urothelial lesions were not observed in the urinary bladders of control rats in this study.
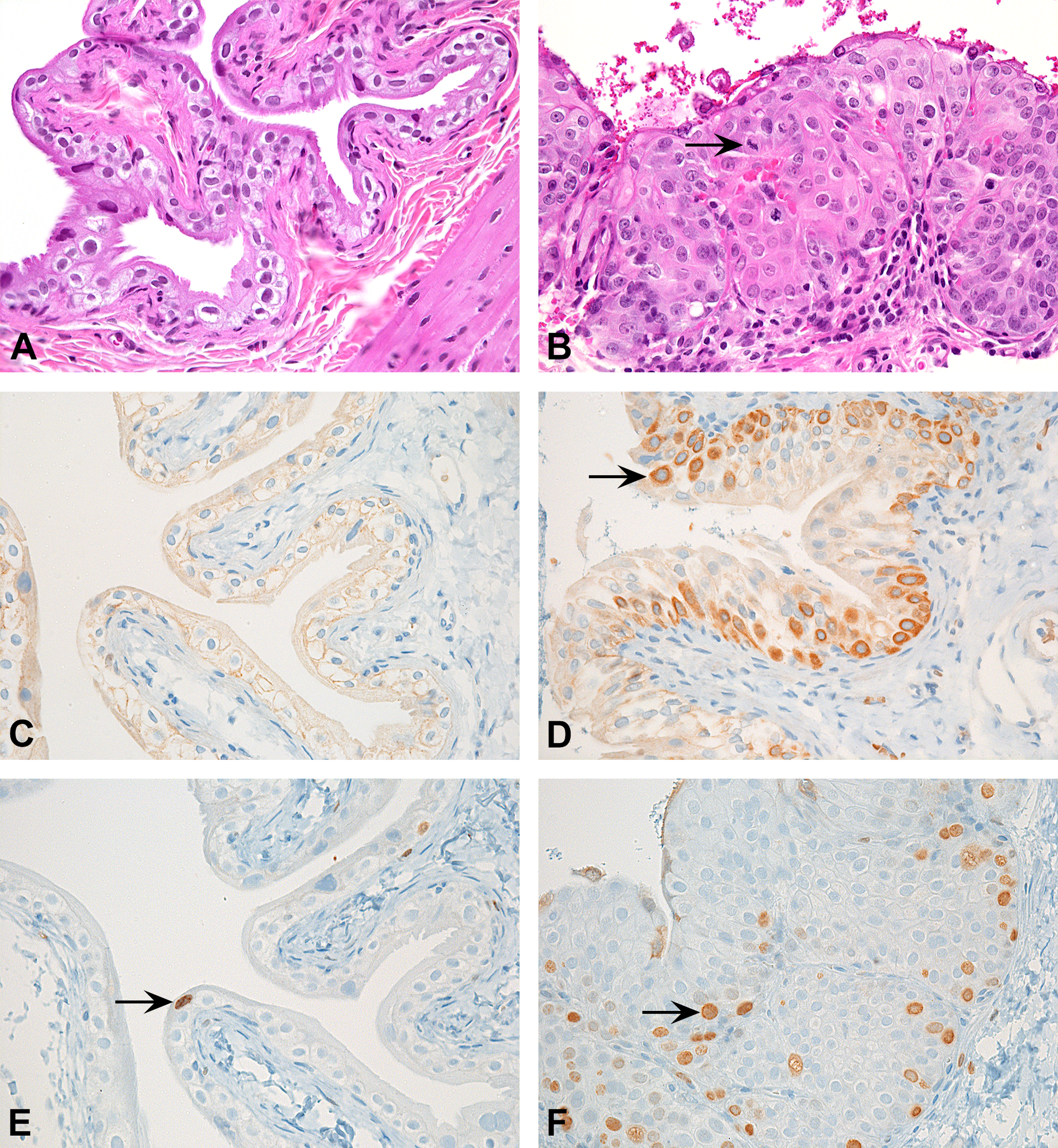
Urinary bladder histopathology and immunohistochemistry (IHC) for oxidative DNA damage (anti-8-OH-dG) and cell proliferation (anti-Ki67) of urinary bladders from control and BBN-treated rats on Week 14. (A) Normal urothelium, control, H&E stain. (B) Urothelial hyperplasia, 400 mg/kg BBN, note mitotic figure (arrow), H&E stain. (C) Normal urothelium, control, anti-8-OH-dG IHC, note lack of 8-OH-dG expression. (D) Urothelial hyperplasia, 400 mg/kg BBN, anti-8-OH-dG IHC, note urothelial cell showing perinuclear 8-OH-dG expression (arrow). (E) Normal urothelium, control, anti-Ki67 IHC, note low number of urothelial cells showing nuclear Ki67 expression (arrow). (F) Urothelial hyperplasia, 400 mg/kg BBN, anti-Ki67 IHC, note increased numbers of urothelial cell showing nuclear Ki67 expression (arrow). BBN = N-butyl-N-(4-hydroxybutyl)-nitrosamine; H&E = hematoxylin and eosin.
IHC
IHC evaluations of urinary bladders from BBN-treated and control rats at week 14 demonstrated a dose-dependent increase in oxidative DNA damage (8-OH-dG) and cell proliferation (Ki-67; Figure 1C, D, E, and F and Table 2). No 8-OH-dG was detected in vehicle control rats. Ki-67 PI values generally increased with increasing doses of BBN and ranged from 1 to 2.8, 2.3 to 6.8, 3.1 to 6.0, 4.7 to 16.3, and 5.5 to 18.1 with doses of 0, 100, 200, 400, and 800 mg/kg BBN, respectively.
BBN 2-stage Tumor Promotion Study
Mortality and clinical observations
One rat was euthanized during dosing with BBN (week 5) due to decreased activity and overall poor condition. Mortality was also observed in 3 rats administered BBN followed by uracil. One rat was euthanized during week 11 due to decreased activity and overall poor condition, and 2 rats were found dead during week 13. No pathological findings that were considered contributory to death in any of the rats that died on the study were observed. There was an increased incidence of liquid and soft feces in rats dosed with rosiglitazone and its vehicle (90% PEG-400). In addition, some of the rats fed uracil in the diet exhibited signs of suspected dehydration throughout the study beginning on week 11.
Food consumption and body weight
Food consumption was increased in rats dosed with rosiglitazone and decreased in rats fed uracil (Figure 2b). Decrease in mean food consumption in rats given uracil in the diet occurred during the first half of the exposure period with the largest effects observed at the start of dosing and progressively returning to control levels by the end of the study. These effects most likely reflect the palatability of the uracil diet. Body weights reflected these food consumption trends. At the end of the study, rats dosed with BBN/rosiglitazone had a statistically significant, 11% higher, mean body weight compared to controls at the end of the study, while BBN/uracil-treated rats had a significant decrease of 13% lower mean body weight compared to controls (Figure 2a).
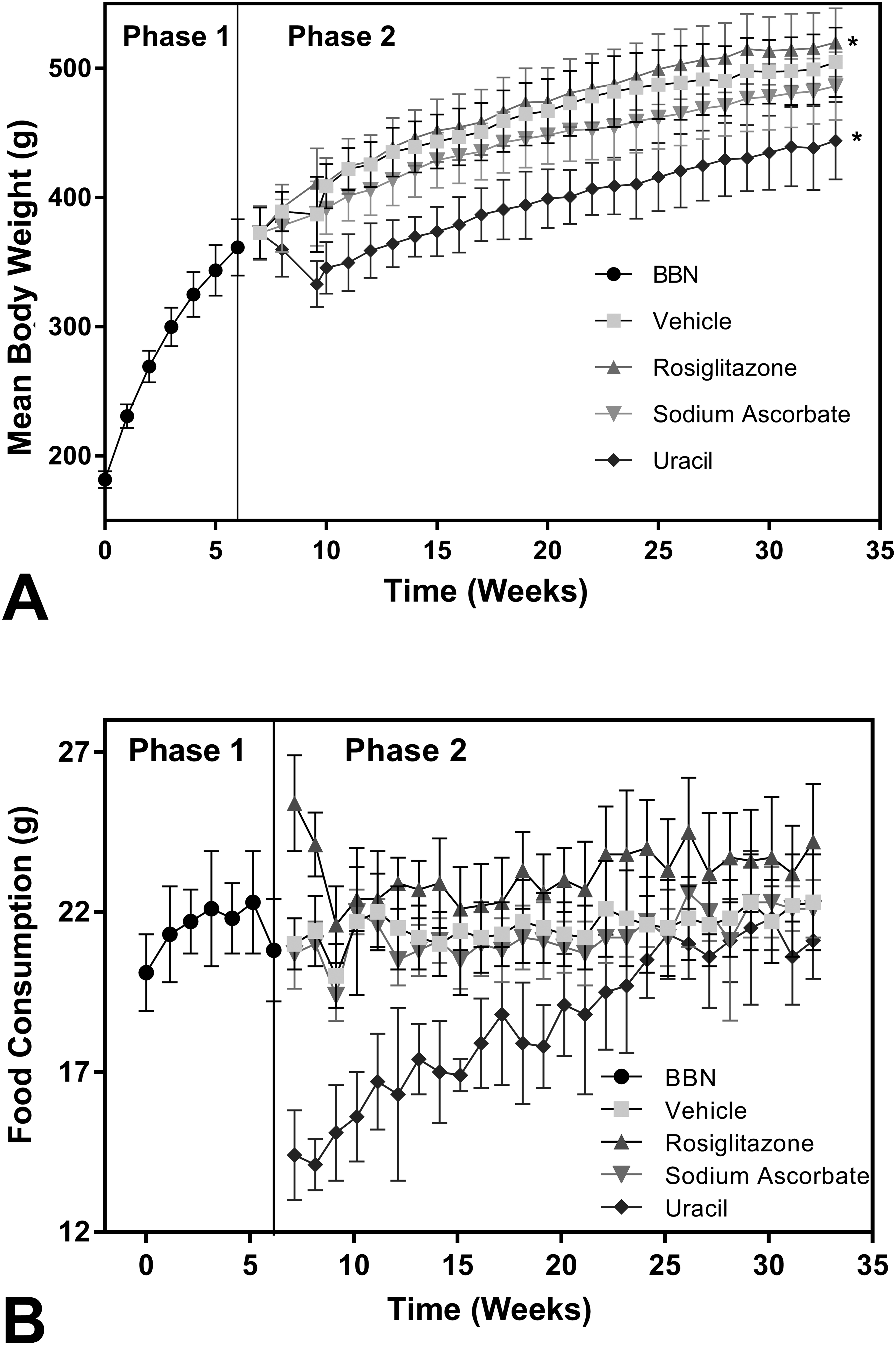
Mean body weights (a) and food consumption (b) during BBN 2-stage tumor promotion study. For food consumption and body weight determinations, each treatment group was divided into 2 groups of approximately 20 rats each. Values represent means ±
Urinalysis
BBN/rosiglitazone had no significant effect on urinalysis parameters during week 10 (Online Supplemental Table 3).
BBN 2-stage Tumor Promotion Study: Incidence of Gross Pathology Findings in Urinary Bladder during Week 34.
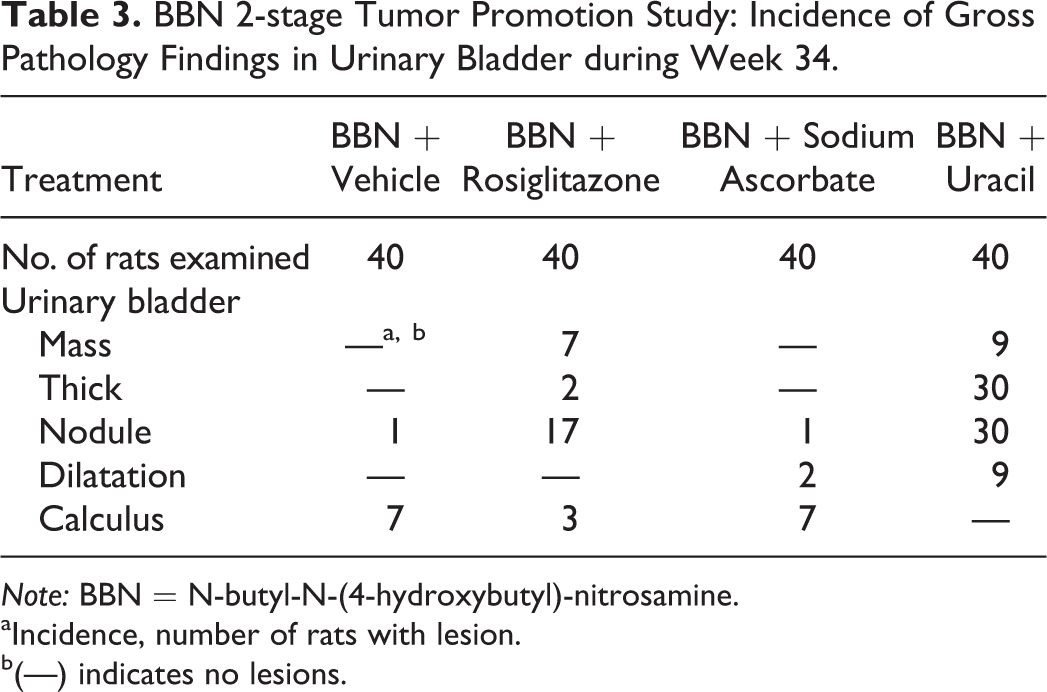
aIncidence, number of rats with lesion.
b(—) indicates no lesions.
BBN/sodium ascorbate was associated with a nonstatistically significant increase in urine volume, relative to the BBN plus vehicle controls, with a corresponding significant decrease in specific gravity. The group given BBN/sodium ascorbate also had significant increases in total sodium excretion and in the sodium/creatinine ratio. There was also a notable increase in urinary pH in rats treated with BBN/sodium ascorbate during week 10.
BBN/uracil induced significant increases in urinary volume with associated decreases in specific gravity and creatinine concentrations. BBN/uracil also significantly increased urinary total protein and total glucose.
Anatomic pathology
Gross findings in the urinary bladder of all groups included masses, thickening, nodules, and dilatation that correlated with urothelial hyperplasia, papilloma, and/or carcinomas (Table 3). Overall, the incidence of all gross findings, with the exception of calculi, was higher in the BBN plus uracil-treated group compared to all other groups. Conversely, calculi were observed in BBN plus vehicle, BBN plus rosiglitazone, and BBN plus sodium ascorbate-treated rats but not in BBN plus uracil-treated rats.
Various gross findings were also observed in the ureter and kidney of BBN plus uracil-treated rats, but these tissues were not examined microscopically as this was considered outside the scope of the study.
There was a significant increase (3.3× controls) in urinary bladder weights in rats treated with BBN plus uracil but not in rats treated with BBN plus either rosiglitazone or sodium ascorbate (Figure 3). This increase in urinary bladder weights correlated with the higher incidences of masses, thickening, and nodules in the uracil-treated rats compared to the other treatment groups.
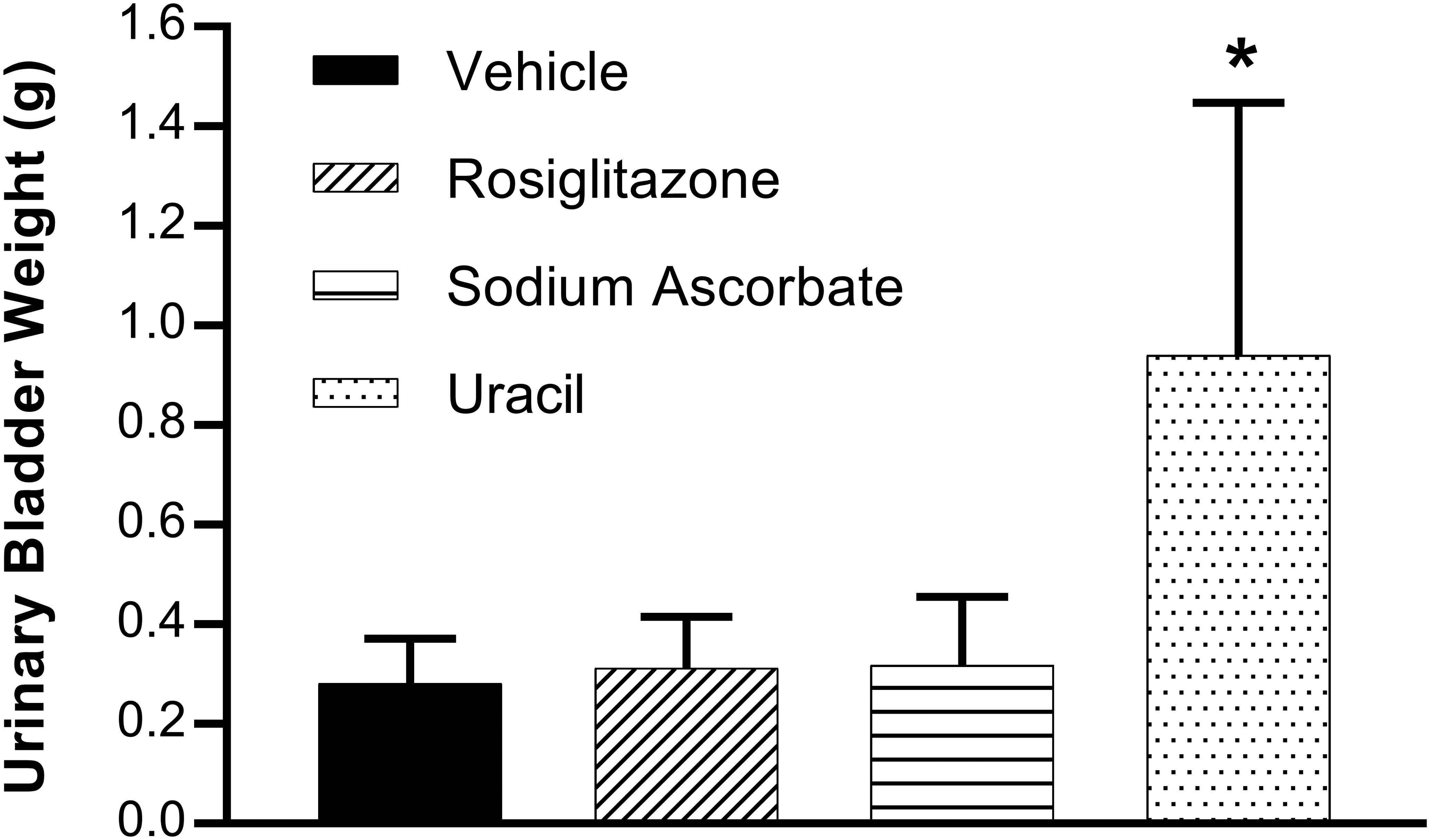
Urinary bladder organ weights during phase 2 of the BBN 2-stage tumor promotion study. Urinary bladders were voided of urine by gentle compression, ligated at the neck near the origin of the urethra, and removed by cutting distal to the ligature. After removal, the ligated urinary bladders were weighed as quickly as possible to avoid drying. Values represent means ±
Rats treated with BBN and vehicle had minimal to mild urothelial hyperplasia (83%) and papillomas (15%) in the urinary bladder. No rats had TCC in the BBN/vehicle treatment group (Table 4).
BBN 2-stage Tumor Promotion Study: Incidence of BBN-related Microscopic Findings in Urinary Bladder during Week 34.
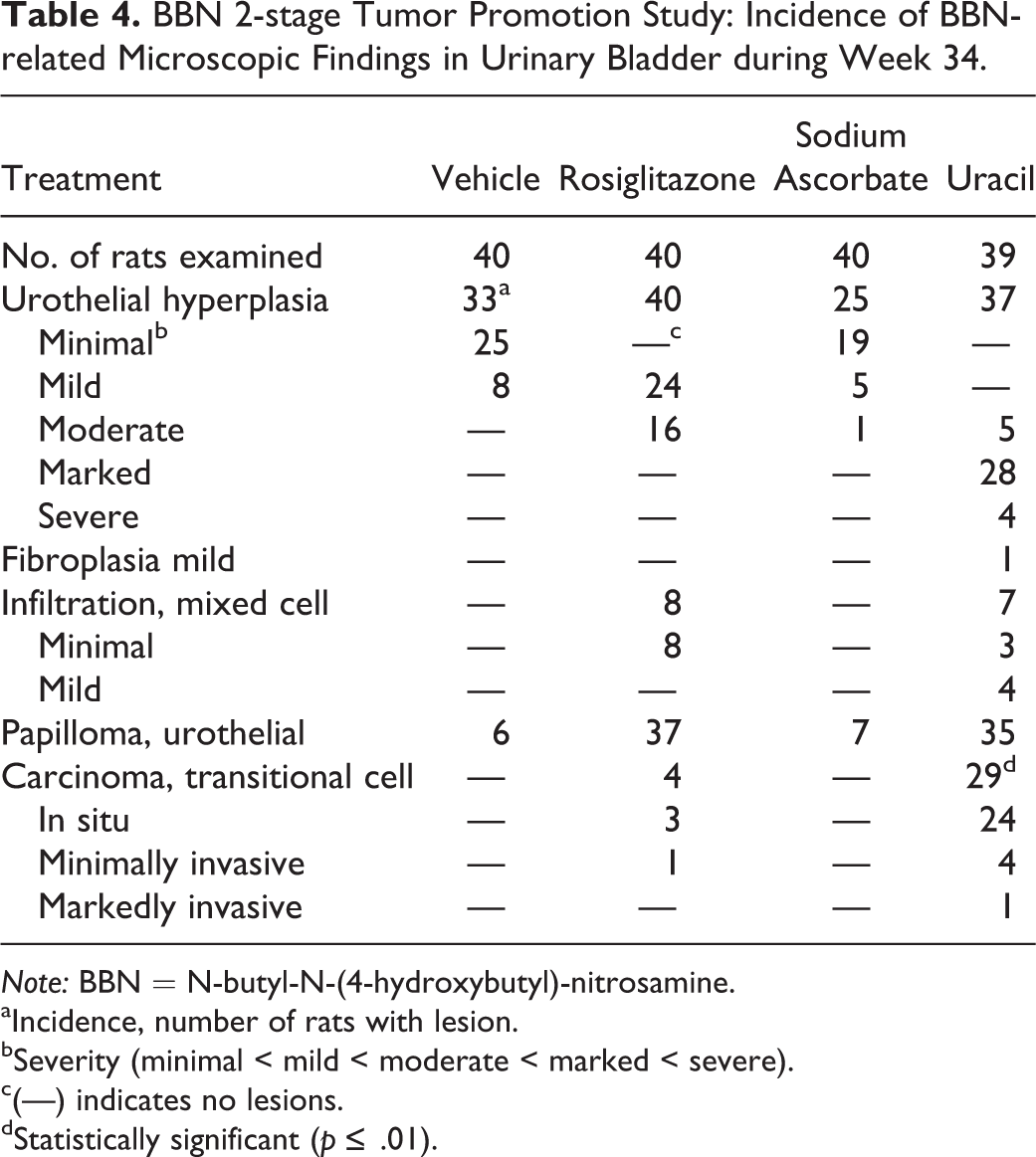
aIncidence, number of rats with lesion.
bSeverity (minimal < mild < moderate < marked < severe).
c(—) indicates no lesions.
dStatistically significant (
BBN/rosiglitazone increased the severity (mild to moderate) of urothelial hyperplasia and the incidence of urothelial papillomas (93%) over that observed with BBN/vehicle. However, there was only a 10% incidence of TCC in BBN/rosiglitazone-treated rats.
BBN/sodium ascorbate slightly decreased the incidence of urothelial hyperplasia (63%), had no effect on the occurrence of urothelial papillomas (18%), and did not result in any TCC (0%).
BBN/uracil, however, had a striking influence on the severity of hyperplasia in the bladder (moderate to severe) and on the incidence of urothelial papillomas (90%; Figure 4). There was also a marked, statistically significant, increase in the incidence of TCC (74%) observed in BBN/uracil-treated rats. In addition, there was minimal to mild mixed cell infiltration in rats treated with rosiglitazone and uracil.
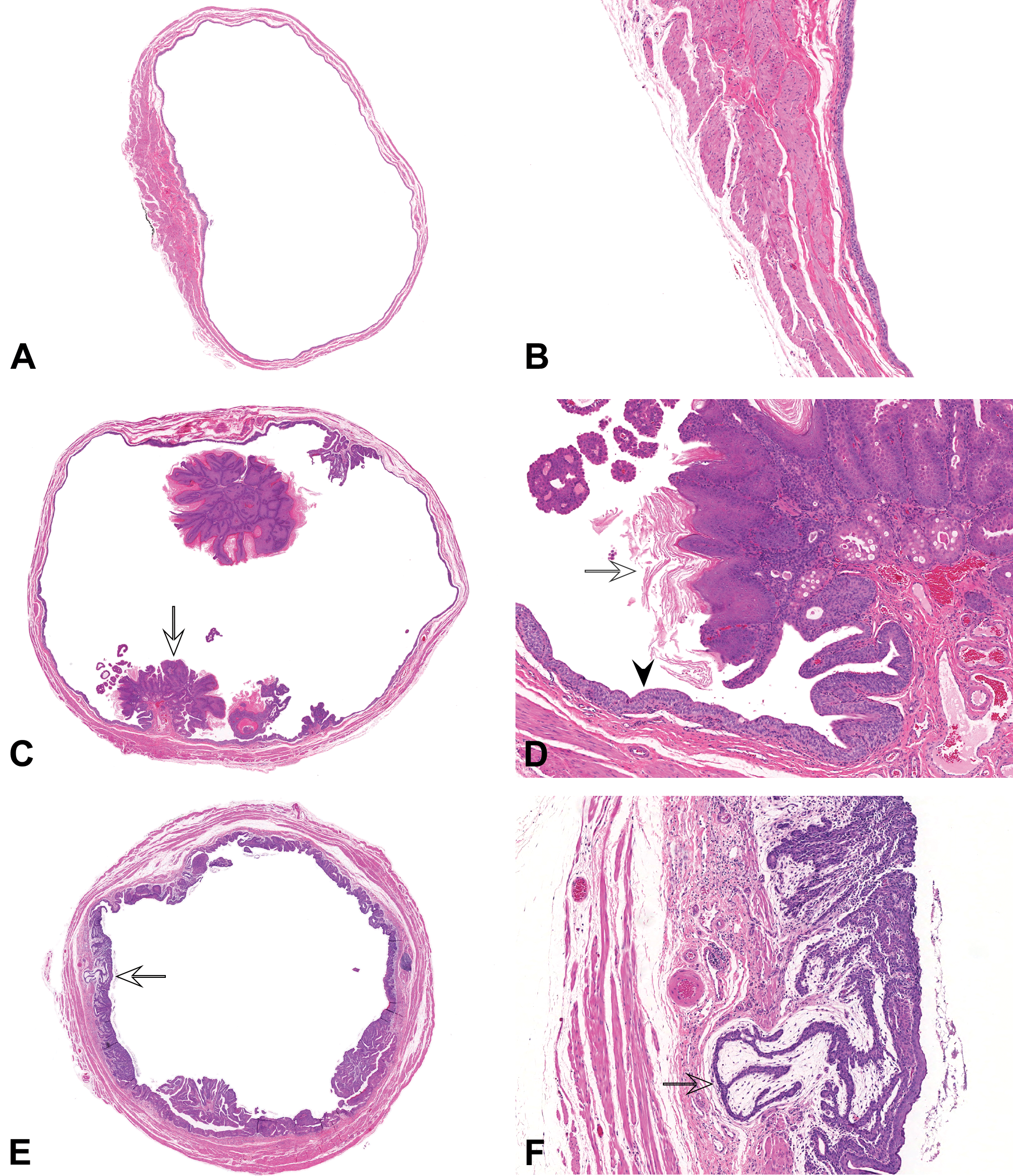
Histopathology of urinary bladder in BBN 2-stage tumor promotion study at week 34. Urinary bladders were stained with H&E and examined microscopically. A and B represent lower (Original Objective 8×) and higher (Original Objective 50×) magnification of normal urinary bladder transitional epithelium in a control rat. C to H represent images of tumors in rats treated with 100 mg/kg BBN and 3% uracil in the diet at lower (Original Objective 8×) and higher (Original Objective 50×) magnification; C and D, transitional cell papilloma with squamous differentiation. Note papillary (exophytic) growth pattern into bladder lumen of transitional epithelium on a fibrovascular stalk, with squamous differentiation and keratinization (open arrows); E and F minimally invasive transitional cell carcinoma (TCC). Note area of minimal invasion into the bladder wall (open arrows). G and H, markedly invasive TCC. Note areas of marked invasion into the bladder wall (open arrows). Note the presence of transitional epithelium hyperplasia in all images from the BBN/uracil-treated rats (arrowhead). BBN = N-butyl-N-(4-hydroxybutyl)-nitrosamine; H&E = hematoxylin and eosin.
Discussion
BBN is a potent and selective initiator of urinary bladder tumors in rodents (Vasconcelos-Nóbrega et al. 2012). For this reason, BBN was selected as the tumor initiator in our urinary bladder 2-stage tumor promotion model. A review of the literature indicated that BBN was well tolerated in rats and was not associated with significant mortality or serious adverse clinical signs in rodents. Any model resulting in the development of tumors at other organ/tissue sites could lead to decreased survival that could interfere with the accurate determination of the incidence of urinary bladder tumors in the study. In the present study, the cause of death could not be identified by macroscopic evaluation in those rats that were found dead or were euthanized in moribund condition. No neoplasms were observed in organs other than the urinary bladder.
BBN is reported to be metabolized mainly in the liver to metabolites that are excreted in the urine. In the liver, BBN is either activated to the metabolite N-nitrosobutyl(carboxypropyl)amine (BCPN) or detoxified by glucuronidation to form the BBN-glucuronide. BCPN unlike the BBN-glucuronide is a urinary bladder carcinogen and can be further metabolized to reactive metabolites that covalently bind to DNA (Vasconcelos-Nóbrega et al. 2012). Although the exact mechanism is unknown, differences in metabolism and/or DNA repair most likely explain why tumors specifically develop in the urinary bladder and not in the liver (Airoldi et al. 1994). There is also evidence that BBN can induce mitochondrial dysfunction leading to oxidative stress in mouse liver (Oliveira et al. 2013). Markers of oxidative DNA damage, 8-OH-dG, have been detected in the urinary bladders of mice treated with BBN (Lin et al. 2012), and there is evidence that BBN-induced oxidative stress contributes to the mechanism of urinary tumor formation (Parada et al. 2012).
In our dose-ranging study, there was a dose-related increase in the PI (Ki-67) and the 8-OH-dG grade observed in the urinary bladder by IHC 14 weeks following treatment with BBN. Although only 3 rats had observable TCC at the week 14 necropsy, levels of these markers correlated with the high incidence of BBN-induced urinary bladder tumors after 24 weeks. These data support the use of 8-OH-dG and Ki-67 as preneoplastic markers of BBN-induced urinary bladder carcinogenesis.
We conducted the BBN dose-ranging study to select the appropriate dose of the initiator, BBN, for use in the subsequent tumor promotion study. The dose of the tumor initiator is critical in the establishment of a 2-stage tumor promotion model. If too high a dose of an initiator is selected, the majority of rats treated with the initiator in the absence of a promoter will develop tumors, thereby lessening the sensitivity of the assay to detect tumor promotion in the initiation/promotion phase of the study. On the other hand, if too low a dose of the initiator is selected, none of the rats will develop tumors even when a potent tumor promoter is used.
In studies of this type, BBN is frequently administered to rats in the drinking water. However, we chose to administer BBN by oral gavage in order to get a more accurate measure of the BBN dose administered to each rat and to reduce some of the variability associated with drinking water administration. BBN induced a dose-dependent increase in TCC in the dose-ranging study at 24 weeks with the incidence of TCC ranging from 20% at the low dose (100 mg/kg) to 93% at the high dose (800 mg/kg). Based on these results, and the reasons above, we selected the low dose of 100 mg/kg BBN for the subsequent 2-stage BBN tumor promotion study. In the BBN dose-ranging study, 2 of the 10 rats developed TCC by the end of the study. However, no rats (0 of the 40) treated with 100 mg/kg BBN alone developed TCC at the end of the 2-stage BBN tumor promotion study. The reason for the difference in incidence is unknown but most likely reflects the low number of rats used in the BBN dose-ranging study and normal biological variation between individual rats. Regardless, the 100 mg/kg dose of the tumor initiator, BBN, was sufficient to detect tumor promotion in the subsequent 2-stage tumor promotion model.
All of the chemicals (rosiglitazone, sodium ascorbate, and uracil) tested for tumor promotion in the current 2-stage BBN tumor promotion model have previously been reported to function as urinary bladder tumor promoters in rats. Rosiglitazone was shown to be a potent urinary bladder tumor promoter in female Fischer-344 rats (Lubet et al. 2008) and in that study, daily administration of 10 mg/kg by oral gavage for 5.5 months, following BBN administration (150 mg/kg twice weekly for 8 weeks), significantly increased the incidence of urinary bladder tumors in rats at 8 months from 28% in the BBN/vehicle group to 93% in the BBN/rosiglitazone group. Lubet et al. (2008) speculated that rosiglitazone, acting as a PPAR gamma agonist, may increase the expression of angiogenic growth factors that contribute to the oncogenic process. Rosiglitazone was much less effective as a urinary bladder tumor promoter in our study possibly due to the shorter duration of administration and the lower dose of BBN that was used. The current study also used male Sprague-Dawley rats while Lubet et al. (2008) used female Fischer-344 rats. The clinical relevance of the rat rosiglitazone urinary bladder tumor promotion findings is not known and is currently a matter of debate (Turner et al. 2014).
Dietary sodium ascorbate has also been reported to be a potent urinary bladder tumor promoter in male Fischer-344 rats by a proposed mechanism involving increasing the sodium content and pH of the urine (Fukushima et al. 1988). Increases in intracellular sodium have been linked with the stimulation of DNA synthesis (Iwata et al. 1997). In contrast to these studies, sodium ascorbate did not increase the incidences of TCC in our study despite significant increases in urinary pH and total urinary sodium. In fact, rats administered sodium ascorbate had minor decreases in the levels of hyperplasia observed in the urinary bladder compared with BBN/vehicle. A study conducted by Mori et al. (1987) indicated that the capacity of dietary sodium ascorbate to function as a urinary bladder tumor promoter differed among different strains (Fischer-344 vs. Lewis) of rats tested and the type of diet (Oriental MF vs. Clea CA-1) used for administering sodium ascorbate. These factors could explain our observed lack of an effect of sodium ascorbate in promoting urinary bladder tumor formation and emphasize the need for very carefully controlled study designs in the development of these models.
In contrast to 5% sodium ascorbate, feeding 3% uracil in the diet proved to be a potent promoter of BBN-induced urinary bladder carcinogenesis in our study increasing the incidence of TCC in rats to 74% compared to 0% in the BBN/vehicle-treated rats. BBN/uracil also induced increases in the severity of urothelial hyperplasia and in the incidence of urothelial papillomas and led to significant increases in urinary bladder weights. Shirari et al. (1986, 1987) determined that 3% dietary uracil was a potent inducer of BBN-induced urinary bladder carcinogenesis in Fischer 344 rats. These researchers proposed urolithiasis as the mechanism for urinary bladder tumor promotion. Interestingly, in our study, we saw no evidence that uracil, at the same dietary concentrations administered, induced calculus formation in the urinary bladder. The reason for the lack of calculi following uracil treatments in our study is unknown. However, Shirai et al. (1986) reported that calculi formed in the bladder following uracil treatments dissolved rapidly during fixation in buffered formalin. We cannot exclude the possibility that calculi were originally present in urinary bladders of uracil-treated rats in our study but dissolved during the formalin fixation procedures.
In conclusion, we have successfully established a 2-stage model for assessing tumor promotion in rat urinary bladders with 2 known urinary bladder tumor promoting agents, rosiglitazone and uracil. The model was based on a rigorous study design that included oral gavage dosing of BBN with a large number of rats in each treatment group (
Footnotes
Authors’ Contribution
Authors contributed to conception or design (MT, EJ, TD, KC, RM, MG, BA, AB, MB, MS); data acquisition, analysis, or interpretation (MT, EJ, TD, YS, SC, KG, KC, RM, BA, SH, MC, JF, MB, MS); drafting the manuscript (MT, EJ); and critically revising the manuscript (MT, EJ, TD, YS, SC, KG, KC, RM, MG, BA, SH, MC, JF, AB, MB, MS). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Authors are employees or former employees of Bristol-Myers Squibb, AstraZeneca, Charles River Laboratories, and/or ToxPath Sciences.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
