Abstract
Lung cancer is the most common cause of cancer-related deaths in humans worldwide. There is strong evidence that the tobacco-specific nitrosamine 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK) and its metabolite 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol (NNAL) play an important role in carcinogenesis caused by tobacco products. NNK and racemic NNAL are reported to induce lung and pancreatic tumors in rats. The carcinogenicity in Fischer 344 rats of NNK, NNAL, and its enantiomers (R)-NNAL and (S)-NNAL has been studied recently, and all test compounds induced significant numbers of lung tumors. We report here the detailed histopathological and immunohistochemical characterization of these tumors and their aggressive nature as shown by their metastasis locally and to the pancreas. The spectrum of treatment-related histopathological findings comprised pulmonary alveolar/bronchiolar (A/B) epithelial hyperplasia, A/B adenomas, and A/B carcinomas. A/B carcinomas frequently exhibited local invasion/metastasis within the mediastinum and thoracic cavity and distant metastasis to the pancreas that was confirmed by immunohistochemistry using the lung-specific markers prosurfactant protein-C and club (Clara) cell-10. Our observation regarding metastasis to the pancreas was an important, and unexpected, finding in this study both for the experimental animal model and potential human relevance.
Lung cancer is the most common cause of cancer-related deaths in humans worldwide, with average survival times measured in months. In the United States, lung cancer mortality is comparable to the combined cancer-related deaths due to breast, pancreas, prostate, and colorectal cancers (U. S. Cancer Statistics Working Group 2013). Tobacco smoking is responsible for about 70% of lung cancer and 30% of pancreatic cancer-related deaths worldwide (Stewart and Wild 2014). Therefore, it is important to understand the underlying mechanisms of carcinogenesis, cells of origin, and types of tumors caused by tobacco carcinogens in order to develop effective approaches to cancer prevention in smokers and smokeless tobacco users.
4-(Methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK) is present in all tobacco products—both combusted and smokeless—and is one of the key components of tobacco products, which induces tumors of the lung in rats, mice, hamsters, and ferrets (Balbo et al. 2014; Hecht 1998, 2002; Akopyan and Bonavida 2006). NNK and the related tobacco-specific nitrosamine N′-nitrosonornicotine are considered “carcinogenic to humans (Group 1)” by the International Agency for Research on Cancer (IARC 2012). NNK is metabolically converted to its metabolite 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol (NNAL) in all biological systems, both in vitro and in vivo, and is present in the urine of virtually all tobacco users as well as in people exposed to secondhand smoke (Hecht 1998, Hecht, Stepanov, and Carmella 2016; Roethig et al. 2009). NNAL exists in enantiomeric forms, (S)-NNAL and (R)-NNAL, due to the existence of a chiral center at its 1-position. (S)-NNAL is the major enantiomer formed in lung and liver microsomal fractions (Hecht, Spratt, and Trushin 1997), but studies in vivo demonstrate that both isomers are formed and are present in urine, including human urine, depending on the system investigated (summarized in Balbo et al. 2014). NNK has been reported to induce exocrine pancreatic tumors in rats and Syrian golden hamsters (Rivenson et al. 1988; Schuller et al. 1994). In a previous study of the carcinogenicity in rats of NNK and NNAL administered in the drinking water, the pancreatic tumors were characterized as exocrine ductular carcinomas based on histomorphology (Rivenson et al. 1988). More recently, a carcinogenicity study of NNK and the enantiomers of NNAL administered in the drinking water to Fischer 344 (F344) rats has been performed (Balbo et al. 2014). In addition, carcinogenicity of tobacco-specific N-nitrosamines and histopathological features of lung, liver, and pancreatic tumors have been reported previously (Rivenson, Hecht, and Hoffmann 1991; Belinsky et al. 1990).
This study describes the detailed histopathology and immunohistochemical characterization of NNK- and NNAL enantiomer–induced pulmonary neoplasms and their isolated metastasis to the pancreas in male F344 rats.
Materials and Methods
Test Compounds
Racemic NNAL, (S)-NNAL, and (R)-NNAL were prepared as described previously and characterized by 1H-NMR (nuclear magnetic resonance) and MS (mass spectrometry; Hecht, Spratt, and Trushin 1997; Hecht, Young, and Chen 1980). NNK was purchased from Toronto Research Chemicals (Toronto, Canada). Chemical purities, metabolism, and DNA adduct formation of these compounds were reported elsewhere (Balbo et al. 2014; Hecht, Young, and Chen 1980).
Tumor Study and Histopathology
The tissues used in this study were from our recent 2-year carcinogenicity bioassay of NNK and the (R) and (S) enantiomers of its metabolite, NNAL in F344 rats described in detail by Balbo et al. (2014). Groups of 15 to 24 male F344 rats were administered the test compound in drinking water as follows: NNK (24 rats, 5 ppm), (S)-NNAL (22 rats, 5 ppm), (R)-NNAL (24, 5 ppm), racemic NNAL (15 rats, 10 ppm), and negative control with no treatment. The method of preparation, purification, storage, and dilution of the aqueous stock solutions of these test compounds were described previously (Balbo et al. 2014). Husbandry and experimental procedures were in compliance with requirements set forth by the University of Minnesota Institutional Animal Care and Use Committee. After complete gross pathological examination, tissue sections from target organs including lungs, pancreas, nasal cavity, and common sites for metastasis in the rat such as liver and kidney were reviewed by histopathological examination. The target organs were selected based on previous carcinogenicity studies (Rivenson et al. 1988). The National Toxicology Program (NTP) classification scheme for pulmonary epithelial neoplasms and International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) guidelines for proliferative lesions in the respiratory system of rat were used to classify pulmonary neoplasms in this study (Boorman and Eustis 1990; Renne et al. 2009).
Immunohistochemistry
Five micrometer sections were prepared from paraffin-embedded blocks of the target organs; lungs and pancreas, and organs of potential metastasis; liver and kidney, for staining with club (Clara) cell 10 (CC-10), prosurfactant protein-C (SP-C), or cytokeratin-19 (CK-19) antibodies. The tissue sections were deparaffinized in xylene and rehydrated through decreasing grades of alcohol followed by either Tris-buffered saline or citrate buffer automation wash buffer (Biocare Medical, Pacheco, CA). Antigen retrieval was done by trypsin digestion. Endogenous peroxidase was quenched by incubating the tissue sections with 3% hydrogen peroxide. Subsequently, the sections were incubated with the primary antibody either CC-10 (1:500, goat anti-CC10 polyclonal antibody, clone T-18, Santa Cruz Biotechnology, Inc., Dallas, TX), SP-C (1:500, rabbit polyclonal SP-C antibody, Millipore, MA), or CK-19 antibody (Santa Cruz Biotechnology, Inc.). For the negative controls, tissue sections were incubated with normal serum from the species in which the primary antibody was raised. The concentration of immunoglobulin in the negative control serum was normalized to that of primary antibody. Subsequently, the sections were incubated with a secondary antibody followed by incubation with EnVision+ system-HRP polymer–based detection solution (Dako North America, Inc., Carpinteria, CA) for 30 min. Antigen–antibody complexes were visualized using 3,3′-diaminobenzidine chromogen. Finally, the slides were counterstained with hematoxylin, dehydrated through increasing grades of alcohol, cleared in xylene, and coverslipped. For CC-10 and SP-C staining, sections of normal rat lungs were used as positive control tissue. For CK-19, normal rat pancreas was used as a positive control. For all of these antibodies, intracytoplasmic immunoreactivity was analyzed in comparison to controls.
Results
In our recently reported carcinogenesis study (Balbo et al. 2014), all of the rats in various treatment groups had grossly visible neoplastic lesions in the lung lobes with no grossly discernible tumors in control rats. Representative gross pathology images of lung tumors are shown in Figure 1A. Within the NNK- and racemic NNAL-treated groups, there were several rats with intrathoracic invasion or mediastinal metastasis of pulmonary carcinomas (Figure 1B).
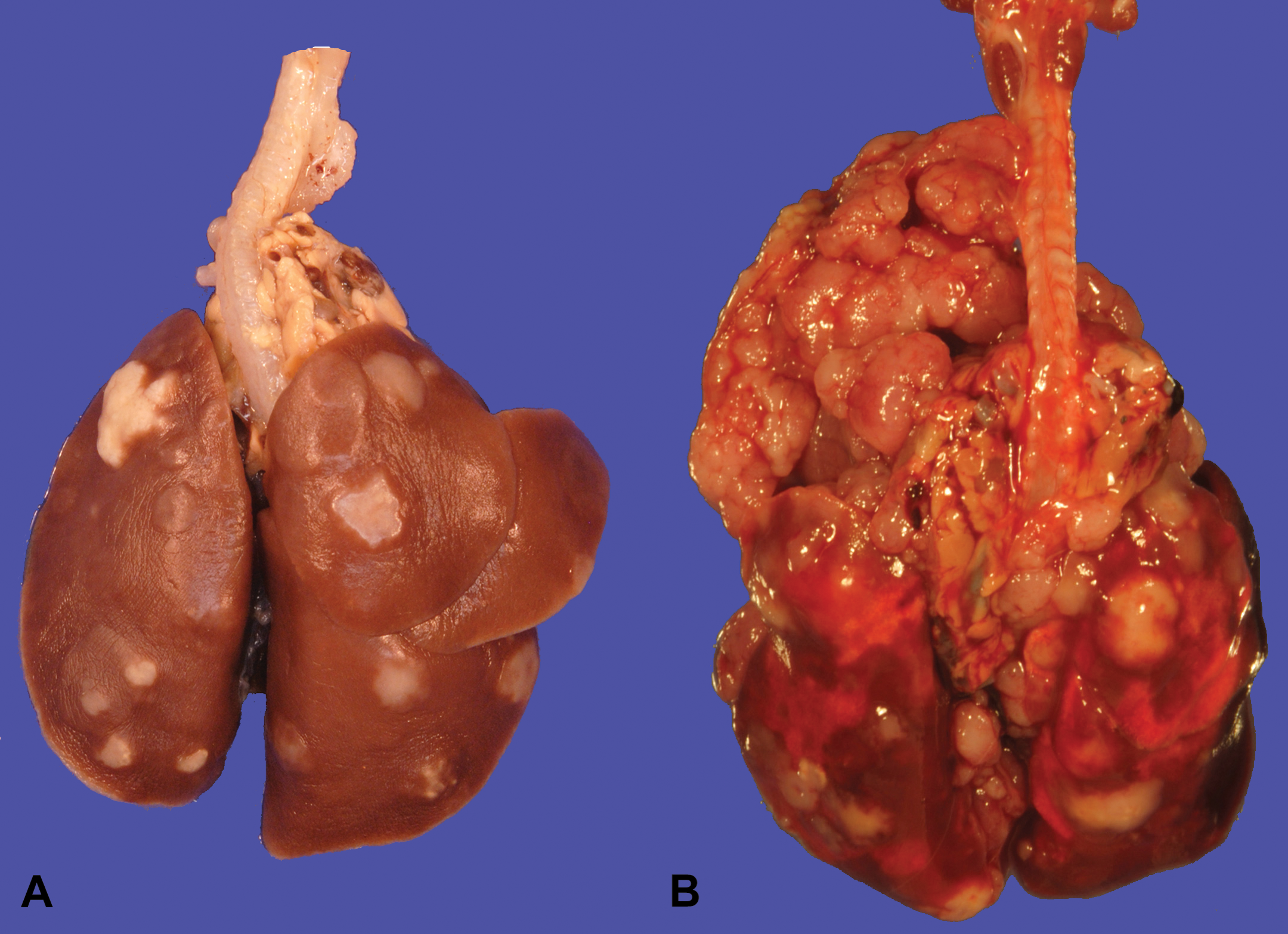
Gross images of lungs (A): (R)-NNAL with pulmonary tumors and (B): (S)-NNAL with intrathoracic invasion and mediastinal metastasis. NNAL = 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol.
Histopathology
Treatment-related proliferative lesions were observed only in lungs and pancreas in all treated groups with no incidence of pulmonary tumors in control rats. Based on the morphological features, the pulmonary lesions were categorized as alveolar/bronchiolar (A/B) epithelial hyperplasia, A/B adenoma, or A/B carcinoma as per the NTP scheme for primary pulmonary proliferative lesions of epithelial origin (Dixon and Maronpot 1991) and INHAND guidelines on proliferative lesions of the respiratory tract of rat and mouse (Renne et al. 2009).
A/B Hyperplasia
The A/B hyperplastic lesions were focal or multifocal areas of hypercellularity, with indistinct peripheral margins and composed of round to oval or cuboidal to columnar often/mostly hypertrophic alveolar type II cells with abundant eosinophilic cytoplasm prominently outlining alveolar walls and CCs with prominent apical protrusions; mitotic figures were not evident in any of the cell populations (Figures 2A and 3A). Areas of hyperplasia were often centered on terminal bronchioles and associated with influx of alveolar macrophages. Lung sections from all of the rats treated with NNK, racemic NNAL, (S)-NNAL and (R)-NNAL had focal or multifocal areas of A/B hyperplasia.
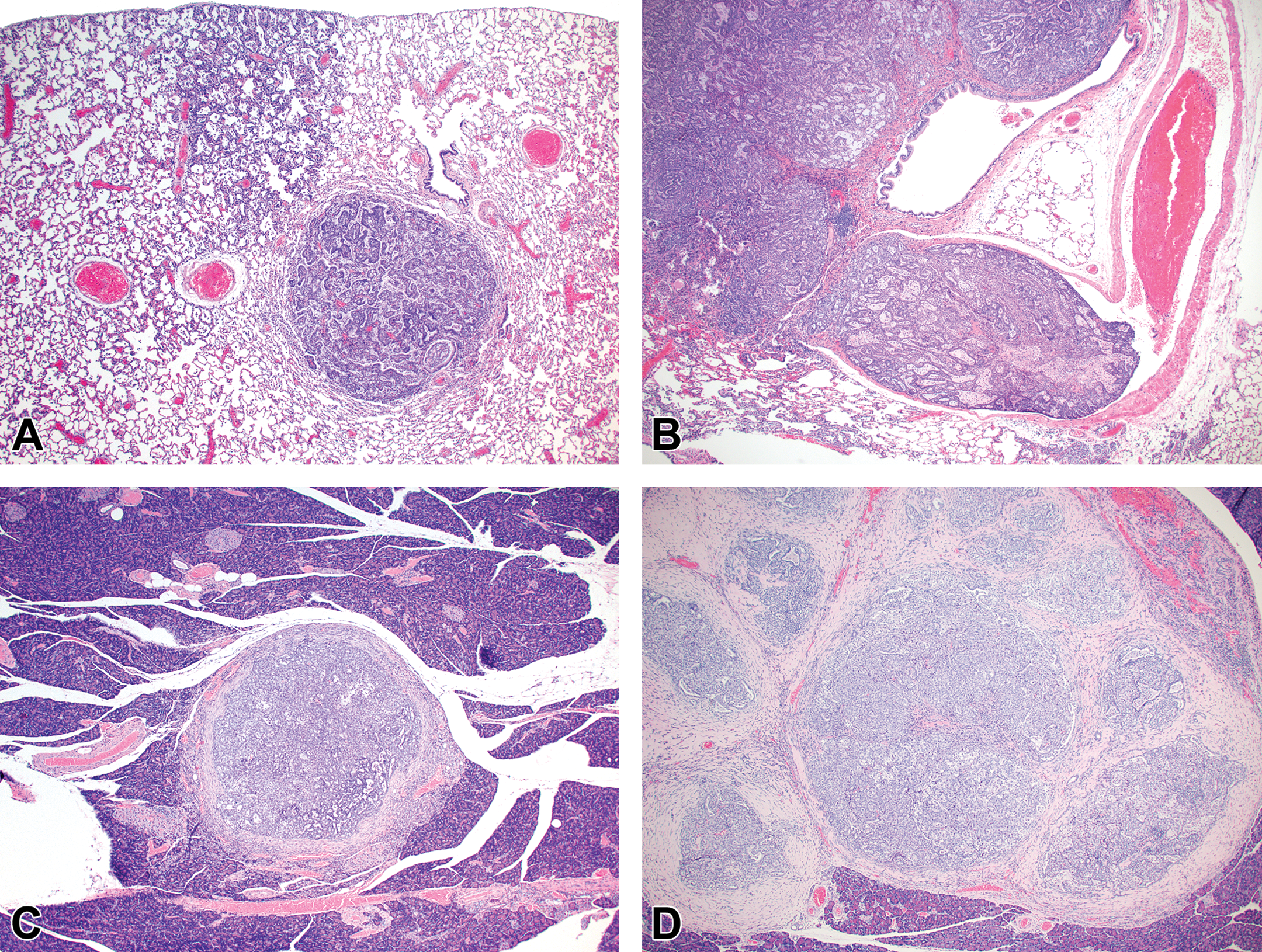
Photomicrographs of proliferative lesions in lung and pancreas. Lung: rac-NNAL-treated rat, alveolar/bronchiolar (A/B) hyperplasia and A/B adenoma (A) and carcinoma with a tumor embolus within a medium caliber blood vessel (B). Pancreas: NNK-treated rat, a focal carcinoma (C) and carcinoma with marked scirrhous reaction (D). NNAL = 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol; NNK = 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone.
A/B Adenoma
A/B adenomas were generally well demarcated, slightly compressed the adjacent nonneoplastic normal parenchyma with strongly convex border and had solid, tubulopapillary, alveolar, or mixed histologic growth patterns. Adenomas of A/B origin were composed of monomorphic populations of well-differentiated epithelial cells with variably distinct cell borders, moderate to abundant amounts of eosinophilic cytoplasm, basally located nuclei with finely stippled chromatin and 0 to 1 prominent nucleolus. Mitotic figures were rare. There were low numbers of A/B adenomas with atypia characterized by a higher degree of pleomorphism, increased mitotic figures, and occasional extension into adjacent bronchioles. All the rats treated with NNK, (R)-NNAL, (S)-NNAL, or racemic NNAL in this study had adenomas except 1 rat treated with (R)-NNAL (Figure 2A).
A/B Carcinoma
A/B carcinomas were generally large, poorly demarcated, irregular nodular growths that effaced the normal pulmonary architecture or often occupied an entire lobe. The neoplastic cells were predominantly arranged as tubulopapillary or alveolar or mixed growth patterns (Figures 2B and 3D). Neoplastic cells were cuboidal to columnar with variably distinct borders, moderate amounts of eosinophilic to foamy cytoplasm, and centrally or basally located nuclei with finely stippled chromatin. Mitotic index was high, ranging from 3 to 6 per 40× high power field (HPF) with moderate to marked anisocytosis and anisokaryosis. There were multifocal areas of squamous metaplasia and marked necrosis within the larger A/B carcinomas. There was evidence of vascular invasion (Figure 2B) or complete occlusion of the bronchial lumen in some rats. As previously reported from this carcinogenicity study (Balbo et al. 2014), all the rats treated with racemic NNAL, and 17/24, 10/22, and 6/24 rats treated with NNK, (S)-NNAL, (R)-NNAL, respectively, had A/B carcinomas. In 6/24 of the NNK-treated rats, 9/15 of the rac-NNAL-treated rats, 2/22 of the (S)-NNAL-treated rats, and 2/24 of the (R)-NNAL-treated rats, there were multifocally extensive to diffuse mediastinal metastasis with intrathoracic invasion (Figure 1B). The histomorphology of these tumors was similar to A/B carcinomas within the pulmonary parenchyma. Proliferative lesions in lung and pancreas in different treatment groups in this study are tabulated in Table 1.
Lung and Pancreatic Proliferative Lesions in Male F-344 Rats Given (S)-NNAL, (R)-NNAL, NNK, or Racemic NNAL in Drinking Water.a
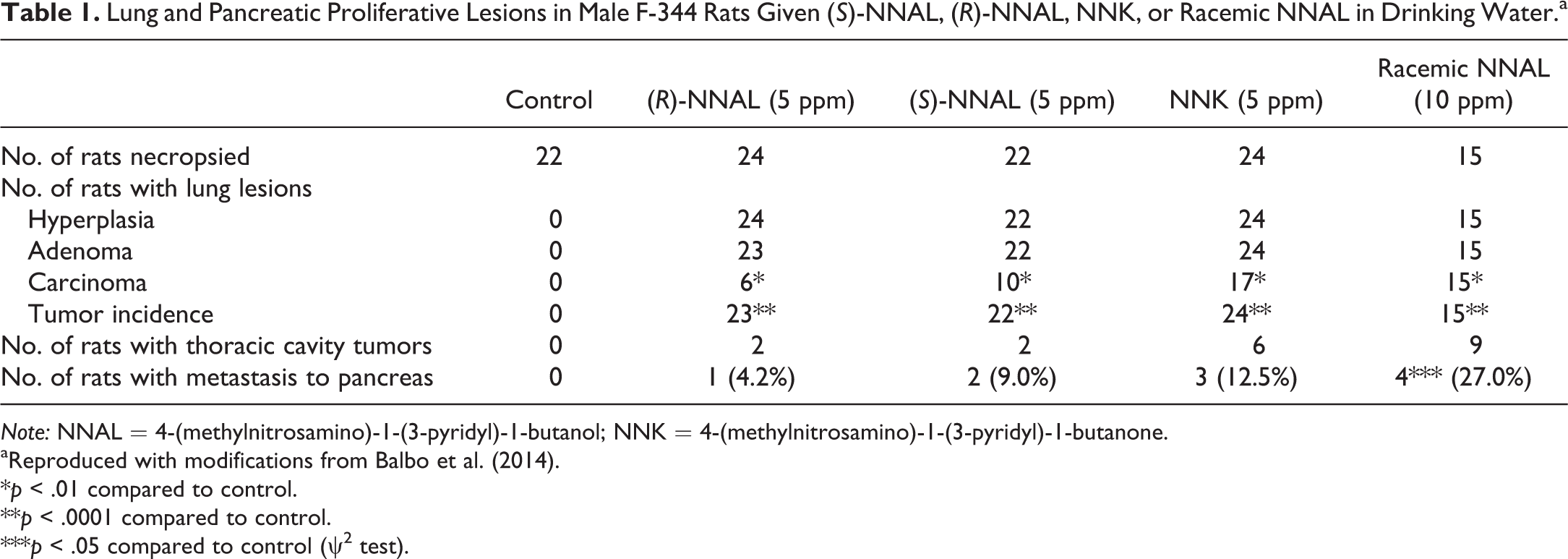
Note: NNAL = 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol; NNK = 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone.
aReproduced with modifications from Balbo et al. (2014).
*p < .01 compared to control.
**p < .0001 compared to control.
***p < .05 compared to control (ψ2 test).
Malignant Tumors in the Pancreas and Kidney
Tumors in the pancreas were only detected microscopically and ranged from 1 mm to 8 mm in diameter. Multifocally, expanding, compressing, and replacing the normal pancreatic tissue, there were nodular masses composed of cuboidal epithelium arranged in tubulopapillary growth patterns with moderate to marked scirrhous reaction (Figure 2C and D). Three of 24 NNK-treated, 4/15 of the racemic NNAL-treated, 2/22 of the (S)-NNAL-treated rats, and 1/24 of the (R)-NNAL-treated rats had 1 or more (up to 6) discrete malignant tumors within the pancreas. Some of these tumors were close to the pancreatic islets. As previously reported (Balbo et al. 2014), a total of 22 pancreatic tumors were observed in treatment groups with no tumors in control rats. One rat treated with racemic NNAL had a single, large, malignant tumor within the kidney with tubulopapillary growth pattern (Figure 3J). Immunohistochemistry with lung-specific markers confirmed that all of these tumors were metastatic pulmonary carcinomas (see below).
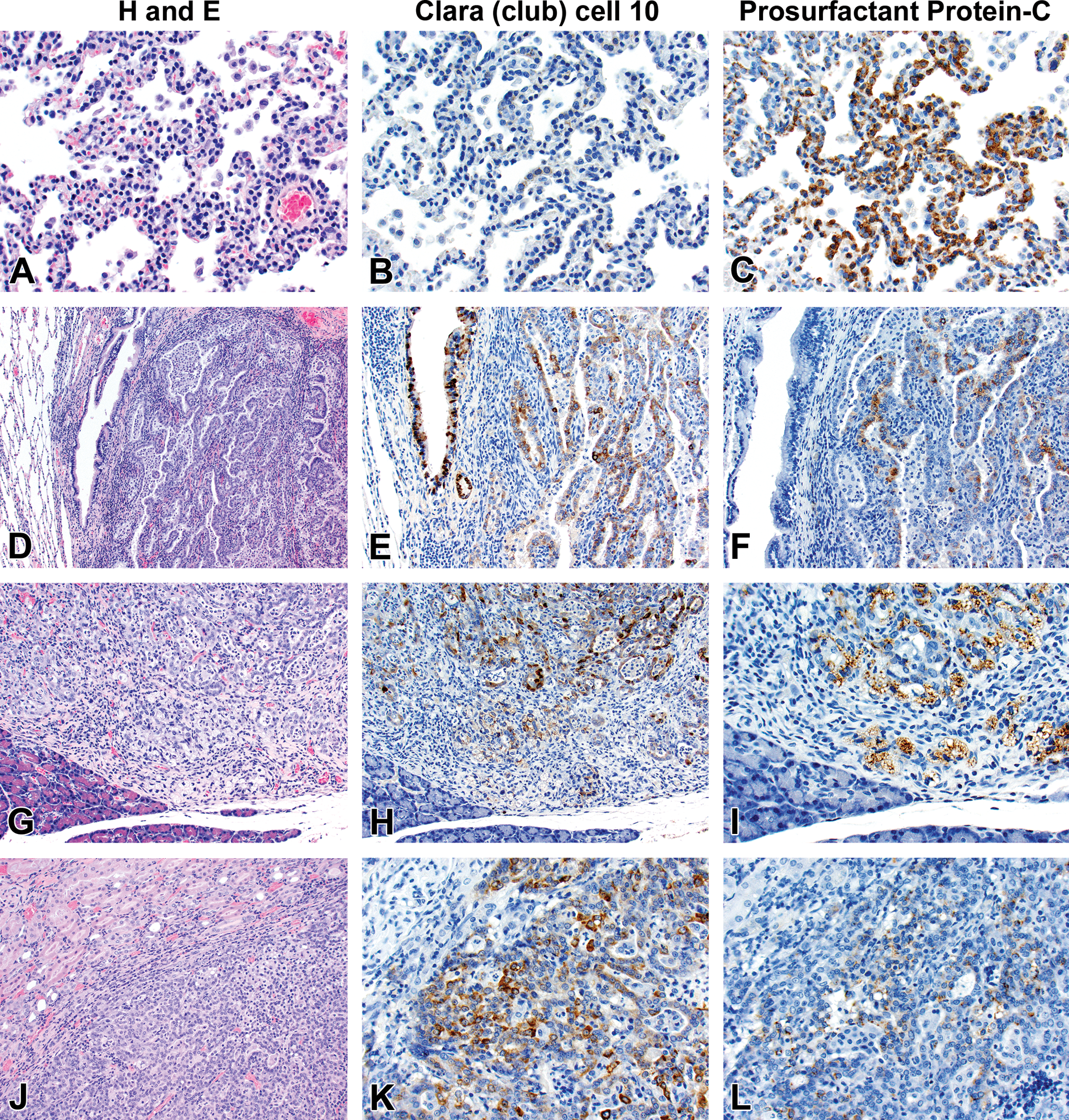
Photomicrographs of histology and immunohistochemistry of lung tumors, metastatic tumors in the pancreas and kidney. Panel A–C: Lung: alveolar/bronchiolar hyperplasia (A); multifocally, a few hyperplastic pneumocytes exhibit weak intracytoplasmic immunoreactivity for CC-10 (B); and diffusely, the hyperplastic cells exhibit strong intracytoplasmic immunoreactivity for SP-C (C). Panel D–F: Pulmonary carcinoma: the tumor cells exhibit strong immunoreactivity for CC-10 and SP-C. Panels G–I and J–L: pancreas and kidney, respectively. The tumor cells exhibit strong immunoreactivity for CC-10 and moderate immunoreactivity for SP-C. CC-10 = club (Clara) cell-10; SP-C = prosurfactant protein-C. Reprinted by permission from Elmore et al. (2016), figure 3D–3I.
Immunohistochemistry
Immunohistochemistry was performed on the tissue sections of all tumors in the pancreas, kidney, and lung sections from selected paraffin blocks of corresponding hematoxylin and eosin–stained tissue sections. Appropriate negative controls were used as detailed in Materials and Methods section. There was no nonspecific immunoreactivity in any of the species-specific antibodies in negative control sections.
SP-C
The SP-C antibody specifically labeled type II pneumocytes in sections of the lung used as a positive control and unaffected pulmonary parenchyma within the treated groups. All sections of the lungs from treated groups with A/B hyperplasia, A/B adenoma, and A/B carcinoma showed strong positive immunoreactivity for SP-C (Figure 3C and F). The immunoreactivity was intracytoplasmic and punctate or finely granular. There was a weak and patchy immunoreactivity for SP-C in some of the pulmonary A/B adenomas and carcinomas. All the thoracic cavity and mediastinal tumors exhibited strong immunoreactivity for SP-C. Twenty-two pancreatic metastases and 1 renal metastasis, all exhibited strong intracytoplasmic immunoreactivity for SP-C (Figure 3I and L). Within the kidney and pancreas, there was no immunoreactivity for SP-C in any of the constituent cell types.
CC-10
In sections of the lung used as positive control tissue, the antibody specifically labeled the CCs within the bronchioles. All A/B hyperplasia exhibited weak intracytoplasmic immunoreactivity (Figure 3B). A/B adenomas and A/B carcinomas exhibited strong immunoreactivity for CC-10 (Figure 3E). The thoracic cavity and mediastinal tumors exhibited strong immunoreactivity similar to lung tumors. Metastatic tumors in the pancreas (22/22) and kidney (1/1) were strongly positive for CC-10 (Figure 3H and K). There was no positive immunoreactivity in any of the cell types within the pancreas and kidney.
CK-19
In the sections of the pancreas used as positive control, the antibody weakly labeled the pancreatic ductular epithelium. Within the lung, which was used as a negative control, the antibody labeled type I pneumocytes, type II pneumocytes, and bronchial epithelial cells. All the lung tumors, thoracic cavity tumors, and metastatic tumors in the pancreas and kidney showed weak intracytoplasmic immunoreactivity (data not shown).
Discussion
A/B adenomas and carcinomas are the most common spontaneous and chemically induced pulmonary neoplasms in B6C3F1 mice and F344 rats (Dixon and Maronpot 1991). Based on experimental animal and human epidemiological studies, tobacco is causally linked with cancer of the lung, oral cavity, larynx, esophagus, pancreas, renal pelvis, nasal cavity, cervix, and urinary bladder (IARC 2012). Tobacco-specific carcinogen NNK’s metabolite NNAL and its enantiomers are found in human urine samples of tobacco users and many people exposed to secondhand smoke (Hecht et al. 2002; Roethig et al. 2009; Xia et al. 2011). Most risk factors for pancreatic cancer, such as genetic disposition, metabolic diseases or chronic pancreatitis, cannot be easily influenced. By contrast, tobacco usage is one of the strongest risk factors for exocrine pancreatic cancer, and it is a completely avoidable hazard. In rodents, several tobacco-derived carcinogens including NNK and NNAL in rats (Rivenson et al. 1988) and N-nitroso-2,6-dimethylmorpholine (cis-NNDM) in rats and hamsters (Kokkinakis and Scarpelli 1989; Lijinsky, Reuber, and Reznik-Schuller 1982) have been reported to induce pancreatic ductal tumors. There are reports describing morphological features of lung and pancreatic tumors induced by tobacco-specific carcinogens (Belinsky et al. 1990; Rivenson et al. 1988; Rivenson, Hecht, and Hoffmann 1991); however, in this study, we report further immunohistochemical characterization of the lung tumors and their specific metastatic lesions to the pancreas.
The number and size of tumors and extent of involvement of pulmonary parenchyma vary with different tobacco-specific carcinogens. Based on our recent study (Balbo et al. 2014), NNK is the most potent carcinogen when compared to its enantiomers, (S)-NNAL and (R)-NNAL in the male F344 rat. Treatment-related microscopic lesions in the lung from this study are in substantial agreement with the previous chronic bioassay of NNK and NNAL in rats (Rivenson et al. 1988); however, using immunohistochemistry, we were able to demonstrate that the pancreatic tumors observed were metastatic tumors rather than primary pancreatic exocrine ductular carcinomas as initially diagnosed based on histomorphology (Rivenson et al. 1988).
Our recent chronic bioassay (Balbo et al. 2014) resulted in a number of important observations including evidence for chemical induced multistep carcinogenesis (Barrett 1993) with progression of lesions from A/B epithelial hyperplasia to atypical hyperplasia to A/B adenoma to A/B carcinoma in most of the lung sections from treatment groups. There was evidence of aggressive malignant behavior of A/B carcinomas including extensive necrosis, squamous metaplasia, marked anisocytosis and anisokaryosis, high mitotic index, intravascular invasion, and local invasion of the mediastinum and thoracic cavity. Our observation of the presence of extrapulmonary intrathoracic cavity and mediastinal carcinomas in several animals has not been previously reported from tumors induced by tobacco carcinogens. However, alveolar bronchiolar tumors that are purely or largely mediastinal with alveolar bronchiolar histological features are reported in F344 rats from several NTP chronic studies (Howroyd et al. 2009).
Pulmonary neoplasms can be diagnosed by their location and typical histomorphology. However, diagnosing isolated metastatic carcinomas in organs like kidney, pancreas, or other parenchymatous organs is often difficult, as the neoplastic cells may have similar morphology to primary neoplasm of these organs. In this study, besides a detailed histomorphological examination, immunohistochemical characterization of A/B neoplasms by using pulmonary epithelial–specific markers including SP-C (type II pneumocytes marker) and CC-10 and a previously reported pancreatic ductular epithelial marker, CK-19 (Kasper, von Dorsche, and Stosiek 1991; Real et al. 1993) was performed. Based on the histomorphology and immunohistochemical characteristics, the intrathoracic and mediastinal tumors were interpreted to have originated from A/B carcinomas either by direct extension or by seeding the thoracic cavity, mediastinum, and pleural surfaces. All of the malignant tumors in the pancreas and 1 tumor in the kidney exhibit strong immunoreactivity for SP-C and CC-10, indicating that all the malignant tumors in the pancreas and kidney are indeed metastatic pulmonary alveolar/bronchiolar adenocarcinomas. The morphology of the metastatic lesions in the pancreas and kidney closely resembles the primary lung A/B carcinomas. Another observation indicating that the pancreatic tumors are not primary pancreatic ductular neoplasms is the absence of a developmental progression of the lesions from adenoma to carcinoma within the pancreas, while in the lung, progression of lesions was observed from A/B hyperplasia to A/B adenoma to A/B carcinomas. Moreover, all of the animals with pancreatic neoplasms also had A/B carcinomas, further reinforcing that the pancreatic tumors are indeed metastatic A/B carcinomas. The kidney is one of the common sites of metastasis of malignant pulmonary tumors in rodents (Boorman and Eustis 1990). However, in this study, only 1 rat treated with rac-NNAL had focal metastasis in 1 kidney. Immunoreactivity to CK-19 was equivocal as pancreatic ductular epithelium, alveolar epithelium, and bronchial epithelium exhibit moderate immunoreactivity to CK-19, and the expression of CK-19 in rat lungs has been previously reported (Schlage et al. 1998). Therefore, CK-19 may not be a specific pancreatic ductular epithelial marker in rats.
The most common sites of metastatic spread from primary lung cancer include the brain, bone marrow, liver, adrenal glands, and skin in humans and the liver and kidney in rats. Isolated metastasis to the pancreas is a rare event. In humans, about 2% of pancreatic cancers are metastatic from another primary site (Roland and van Heerden 1989; Stankard and Karl 1992; Z’Graggen et al. 1998). In humans, isolated pancreatic metastases are most often from renal cell carcinoma, lung cancer, breast, and colon cancer (Bonapasta et al. 2010; Niess et al. 2013; Showalter, Hager, and Yeo 2008). There are no reports of isolated metastases to the pancreas in rats. The underlying mechanism of specific isolated metastasis of A/B carcinomas to pancreas in humans in rodents is not clear.
In conclusion, tobacco-specific carcinogens NNK, NNAL and its enantiomers, induced a spectrum of histopathological lesions including A/B hyperplasia, A/B adenoma, and A/B carcinomas in the lungs of male F344 rats and isolated metastasis of A/B carcinomas to the pancreas. Immunohistochemical characterization of tumors in this study has demonstrated that pancreatic tumors in NNK- and NNAL-treated rats are metastatic lesions arising from primary lung tumors rather than primary pancreatic tumors as had been originally reported on the basis of histomorphology alone.
Footnotes
Acknowledgments
We sincerely thank Drs. Jerry Hardisty and colleagues at Experimental Pathology Laboratories, Inc., and at the National Toxicology Program (NTP), Research Triangle Park, for reviewing lung and pancreatic tumors from this study; Paula Overn for preparation of all histology and immunohistochemistry slides, Josh Parker for gross pathology image preparation, and Dr. Janardhan Kyathanahalli, Integrated Laboratory Systems and NTP for sharing CC-10 and SP-C Immunohistochemistry protocols.
Author Contribution
All authors (RK, CJ, SB, SH, MO) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by U. S. National Cancer Institute grant (CA-81301).
