Abstract
Lysosomes have a central role in cellular catabolism, trafficking, and processing of foreign particles. Accumulation of endogenous and exogenous materials in lysosomes represents a common finding in nonclinical toxicity studies. Histologically, these accumulations often lack distinctive features indicative of lysosomal or cellular dysfunction, making it difficult to consistently interpret and assign adverse dose levels. To help address this issue, the European Society of Toxicologic Pathology organized a workshop where representative types of lysosomal accumulation induced by pharmaceuticals and environmental chemicals were presented and discussed. The expert working group agreed that the diversity of lysosomal accumulations requires a case-by-case weight-of-evidence approach and outlined several factors to consider in the adversity assessment, including location and type of cell affected, lysosomal contents, severity of the accumulation, and related pathological effects as evidence of cellular or organ dysfunction. Lysosomal accumulations associated with cytotoxicity, inflammation, or fibrosis were generally considered to be adverse, while those found in isolation (without morphologic or functional consequences) were not. Workshop examples highlighted the importance of thoroughly characterizing the biological context of lysosomal effects, including mechanistic data and functional in vitro readouts if available. The information provided here should facilitate greater consistency and transparency in the interpretation of lysosomal effects.
Keywords
Introduction
The determination of adversity based on morphologic changes in nonclinical studies is a critical but often challenging part of risk assessment. In order to better define and describe characteristics that aid in the determination of adverse pathological effects, the European Society of Toxicologic Pathology (ESTP) organized an international expert workshop in 2015 to elaborate on, and further refine, efforts on the topic by the U.S. Society of Toxicologic Pathology (STP; Kerlin et al. 2016). The tiered approach for the evaluation of adversity recommended by Palazzi et al. (2016) was also intended to serve as the basis for further organ- or lesion-specific workshops planned by the ESTP. To build on this foundation, and apply many of the concepts outlined in these recent publications, a subsequent workshop was organized by the ESTP to address adversity issues related specifically to lysosomal accumulation in nonclinical studies.
The topic of lysosomal accumulation was chosen for several reasons: (1) this change is frequently encountered in nonclinical toxicity studies (Logan et al. 2012) due to the central role of lysosomes in cellular uptake, storage, intracellular trafficking, and digestion of endogenous and exogenous material; (2) it is caused by a wide variety of pharmaceutical and chemical agents, and in many cases linked to specific physicochemical properties; and (3) it results in histological features that are often quite nondescript or similar across different types of compounds, independent of their chemical structure and the mechanism of lysosomal uptake. The latter features may be characterized in pathology reports by various general terms such as “cytoplasmic vacuolation,” “intracytoplasmic granules,” “hyaline droplets,” or “pigmentation,” which may not adequately convey the nature of the accumulation or the biological consequences. Many of these relatively quiescent diagnoses can lead to difficult (and perhaps inconsistent) decisions regarding adversity, especially when there is limited basis for evaluating the potential for impaired cellular or organ function, or capacity to respond to additional stress. Such considerations can greatly influence the development and safety evaluation of new pharmaceutical and chemical entities.
Lysosomal accumulations can result from a large number of diverse substances, both endogenous and exogenous in origin, which may have very different biological contexts. On the one hand, lysosomes may be considered organelles that enable detoxification and cell survival by storage and degradation of a foreign material. From this perspective, a given lysosomal accumulation may be considered simply as a benign indicator of physiological clearance (i.e., the lysosome doing its job). On the other hand, the same accumulation may be interpreted as an indicator of lysosomal dysfunction, with consequences for major cell functions. From this standpoint, lysosome pathology may indicate cytotoxicity possibly leading to cell death and therefore be considered adverse (i.e., based on cellular injury and dysfunction). This dichotomy highlights the interpretive challenge of lysosomal accumulations and, in some cases, the importance of communicating information beyond a simple generic histopathological diagnosis. Mechanistic investigations may include evaluation of cell- or tissue-specific clearance pathways and mitochondrial toxicity, given that impaired lysosome function may impact autophagy and lead to increases in oxidative stress, mitochondrial dysfunction, inflammation, and cell death (J. Lee, Giordano, and Zhang 2012; Boya et al. 2005).
There was general agreement by the working group that adversity considerations and criteria would vary depending on the mechanism of accumulation and pathophysiological context. Since the full spectrum of test items inducing lysosomal accumulation could not be feasibly covered during the workshop, representative compound classes and types of accumulation were selected. These case examples included polyethylene glycol (PEG)-modified (PEGylated) compounds, therapeutic oligonucleotides, phospholipidosis (PLD), iron, lipofuscin, and alpha-2u (a2u) globulin accumulation. Other chemical classes such as nanoparticles, particulate matter such as diesel exhaust and dyes, and fibers were not included in the scope of this workshop.
A face-to-face workshop was held in Barcelona, Spain, on September 23–24, 2016, after a 7-month preparatory phase consisting of teleconferences with expert contributions and discussions. The expert group was comprised of 23 pathologists, toxicologists, and research scientists from Europe, the United States, and Japan, working in a diverse range of settings, including industry (including Contract Research Organizations), research, and regulatory affairs for industrial chemicals, environmental and food chemicals, and pharmaceuticals. As in the previous workshop, panelists, including a chemist and an investigative toxicologist, were invited by the workshop chairs and the ESTP board based on their expertise in the field of lysosomal accumulation. The primary goals of this workshop were to (1) develop a better understanding of the pathways/modes of action leading to lysosomal accumulation and potential functional consequences, (2) provide criteria for toxicologic pathologists to assess adversity of histopathologic findings associated with lysosomal accumulation, and (3) describe methods and experimental considerations to further inform adversity decisions related to lysosomal accumulation.
This resulting final workshop document has been reviewed and endorsed by major toxicologic pathology organizations including the ESTP, the British Society of Toxicologic Pathology, the Dutch Society of Toxicologic Pathology, the French Society of Toxicologic Pathology, the Society of Toxicologic Pathology, the Japanese Society of Toxicologic Pathology, the Latin American Society of Toxicologic and Experimental Pathology, the Society of Toxicologic Pathology—India, the Chinese Pharmaceutical Association—Specialty Group of Toxicology Pathology, the Chinese Society of Toxicology—Toxicologic Pathology Specialty Section, the International Federation of Societies of Toxicologic Pathologists, and the International Academy of Toxicologic Pathology.
The Lysosome
Lysosomes were first discovered in the early 1950s using biochemical methods as part of an attempt to localize glucose-6-phosphatase in liver cells (Bainton 1981). Subsequent studies showed lysosomes to be membrane-bound organelles present in virtually all eukaryotic cells. While initially considered to be simple cellular “waste bags,” lysosomes are now considered to be major metabolic coordinators of the cell (Saftig and Klumperman 2009). They facilitate degradation of extracellular material as well as intracellular components and contribute to many other key cellular processes such as plasma membrane repair, cholesterol homeostasis, antigen presentation, and cell death (Xu and Ren 2015). The fundamental pathophysiological relevance of lysosomal function is highlighted by the fact that the Nobel Prize for Physiology or Medicine has been granted twice for lysosomal research to Christian de Duve for their initial discovery as organelles (Bainton 1981) and, more recently, to Yoshinori Oshumi for elucidation of the process of autophagy (Harnett et al. 2017).
Primary lysosomes are membrane-bound organelles, ranging in size from ∼0.2 to 0.8 µm, that originate from the Golgi complex by budding via the trans-Golgi network. Their size, distribution, and number within a cell may vary depending on nutrient availability. It has been estimated that nutrient-rich mammalian cells may have several hundred lysosomes; upon starvation, this number drastically decreases and lysosome size increases (Xu and Ren 2015). Their luminal and membrane components are initially synthesized by the rough endoplasmic reticulum (ER) and then transferred to the Golgi apparatus via transport vesicles. The relationship between primary lysosomes, the Golgi apparatus, and the ER was identified in the late 1950s by Novikoff and coworkers (Novikoff 1976). The acidic luminal contents of lysosomes (pH ∼5) and the presence of more than 60 acid hydrolases allow degradation of many types of macromolecules (Xu and Ren 2015). The lysosomal membrane is a single phospholipid bilayer with a high content of glycosylated membrane proteins such as lysosomal associated membrane protein (LAMP)-1 and LAMP-2, which represent about 80% of lysosomal membrane proteins, lysosomal integral membrane protein 2, and cluster of differentiation 63 (CD63). These heavily glycosylated membrane proteins form a luminal glycocalyx protecting the lysosome from autodigestion (Saftig and Klumperman 2009).
Lysosomes degrade and recycle macromolecules taken up from both extracellular and intracellular compartments (Figure 1). Extracellular materials can be internalized either by the uptake of extracellular submicroscopic constituents in an endosome (endocytosis) or by the uptake of extracellular pathogens and cellular debris in phagosomes (phagocytosis). Intracellular materials are acquired via the uptake of cellular components in autophagosomes (autophagy; Turk and Turk 2009; Boya et al. 2005; Appelqvist et al. 2013; Stern, Adiseshaiah, and Crist 2012). Endosomes result from invagination of the plasma membrane around extracellular material, forming a vesicle. Vesicles then fuse with a lysosome to form an endolysosome (secondary lysosome), where the material is degraded by lysosomal hydrolases. After phagocytosis, the phagosome containing the ingested macromolecules fuses with the lysosome to form the phagolysosome, in which the foreign material is enzymatically degraded (Stern, Adiseshaiah, and Crist 2012).
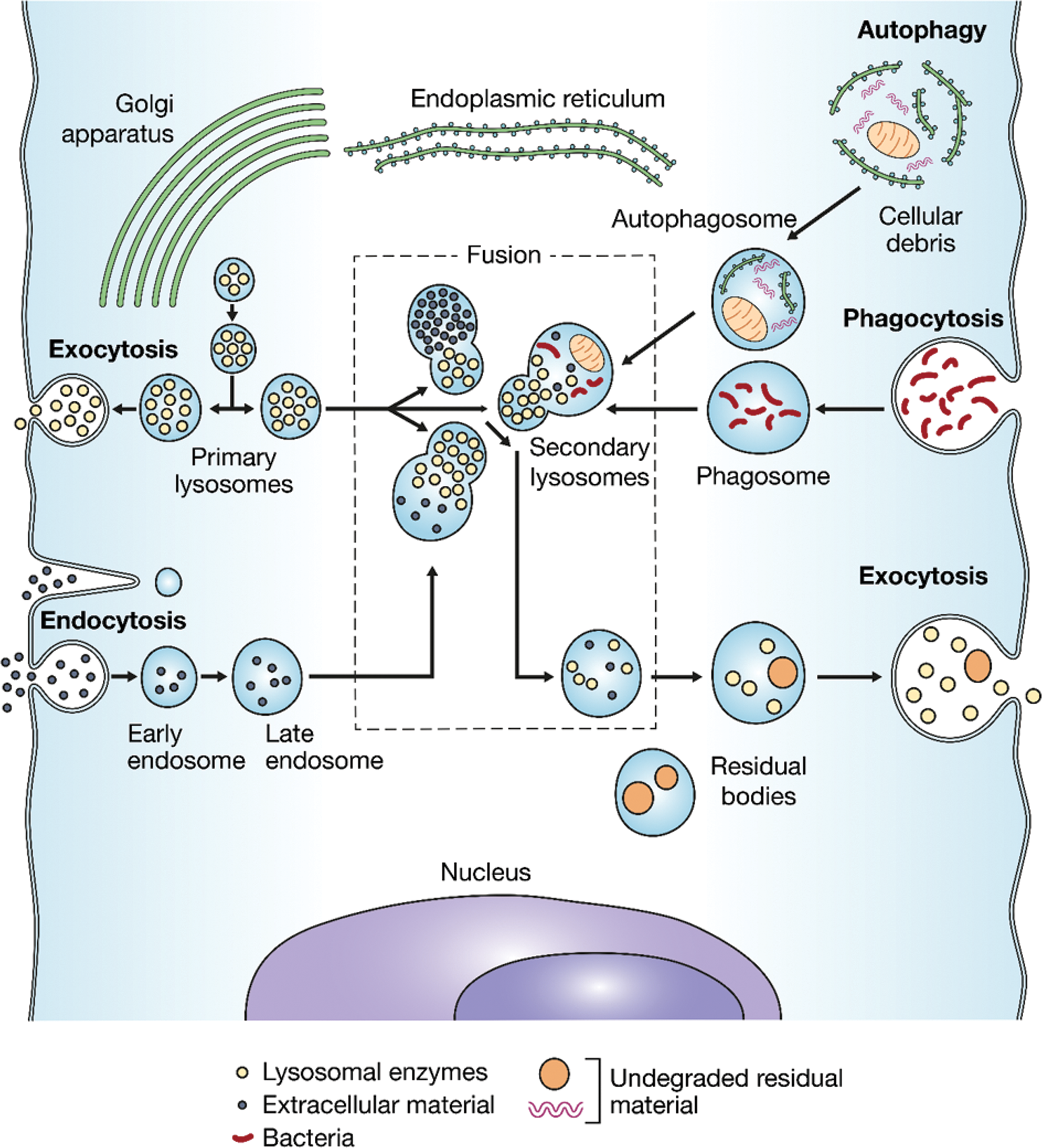
The endosomal–lysosomal apparatus, responsible for the intracellular digestion of macromolecules via the processes of autophagy (uptake of cytoplasmic constituents), phagocytosis (uptake of extracellular pathogens and cellular debris), and endocytosis (uptake of extracellular submicroscopic constituents). The resulting phagosomes and endosomes fuse with lysosomes where material is degraded and recycled. Residual material is removed from the cell via exocytosis (active transport of molecules out of the cell). Adapted from Desnick and Schuchman (2002).
Three types of autophagy can be distinguished: (1) chaperone-mediated autophagy, (2) microautophagy by direct engulfment, and (3) macroautophagy via sequestration of cytoplasmic material within an autophagosome. Autophagy controls important physiological functions in which cellular components need to be degraded and recycled, and a basal rate of autophagy is required to maintain cellular homeostasis (Appelqvist et al. 2013). Autophagy is also a means to provide energy to the cell during starvation and other types of stress; to eliminate intracellular pathogens, as well as damaged proteins and organelles (such as dysfunctional mitochondria); and as a quality control mechanism to counteract the negative consequences of aging. Disrupted autophagy has been linked to several age-related diseases including neurologic conditions such as Parkinson’s disease and Alzheimer’s disease (J. H. Lee et al. 2010; Bellettato and Scarpa 2010) and cancer (Kirkegaard and Jäättelä 2009). There is current research focused on developing drugs that can target autophagy in various diseases.
Exocytosis is a secretory pathway involving lysosomes and occurs in many mammalian cell types. Material is released into the extracellular space by fusion of the lysosome with the cell membrane, and membrane components of the lysosome are integrated into the external plasma membrane. This mechanism plays a major role not only in excretory functions but also in immune responses, bone resorption, cell signaling, and plasma membrane repair (Andrews 2000).
Altered lysosomal function may have other important effects on cellular health beyond cellular catabolism. Lysosomal membrane permeabilization and other types of damage to the lysosomal membrane can leak cathepsins and other hydrolases into the cytosol, where they degrade various cell components and trigger apoptosis (Boya and Kroemer 2008). These lysosome-mediated cell death pathways can function in normal physiology as well as disease, for example, in mammary gland involution after lactation (Kreuzaler et al. 2011) and embryonic remodeling (Tsukamoto et al. 2008). In addition to hydrolases, lysosomal release of iron and calcium, cytosolic acidification, and formation of reactive oxygen species may lead to cell death.
The study of lysosomal storage diseases (LSDs) has led to further insights into lysosomal function. These are inborn pathologies characterized by incomplete degradation and accumulation of undigested material in lysosomes, often due to hereditary absence or reduced activity of lysosomal enzymes or transport proteins. Over 50 types of human LSDs have been described to date (Boya 2012). It is recognized today that clinical disease in the context of LSDs is not simply a consequence of accumulation but rather of perturbation of complex cell-signaling mechanisms. The fact that most LSDs have prominent central nervous system (CNS) manifestations, despite more pronounced lysosomal accumulation in other organs, indicates the high vulnerability of postmitotic cells to lysosomal dysfunction due to their limited regenerative potential and lack of compensatory cellular metabolic pathways (Bellettato and Scarpa 2010).
Different in vitro assays are currently available to assess mechanisms of induced lysosomal dysfunction. For example, autophagy can be monitored in vitro by evaluating autophagosomes, which increase in number and size following inhibition of lysosome–autophagosome fusion (Klionsky et al. 2012). This process can be demonstrated with antibodies against microtubule-associated protein 1 light chain 3, a marker for autophagosomes. Likewise, histomorphologically, size and quantity are considered indicative of lysosomal dysfunction and can be identified using lysosome-specific stains (e.g., Lysotracker® dyes) in combination with quantitative image analysis software (Khoh-Reiter et al. 2015). The functional capacity of macrophages can be assessed by different in vitro and in vivo/ex vivo assays (Lebrec and Burleson 1994; Burleson and Burleson 2008; Drevets, Canono, and Campbell 2015). In addition, different in silico, in vitro, and in vivo tools are available to screen for the potential of a compound to induce PLD (a common type of lysosomal accumulation) or to monitor it. Selected examples are provided in Table 1.
Selected Assays/Tests Used to Screen for the Potential to Induce PLD or Other Types of Lysosomal Accumulation and to Assess Lysosomal Function.
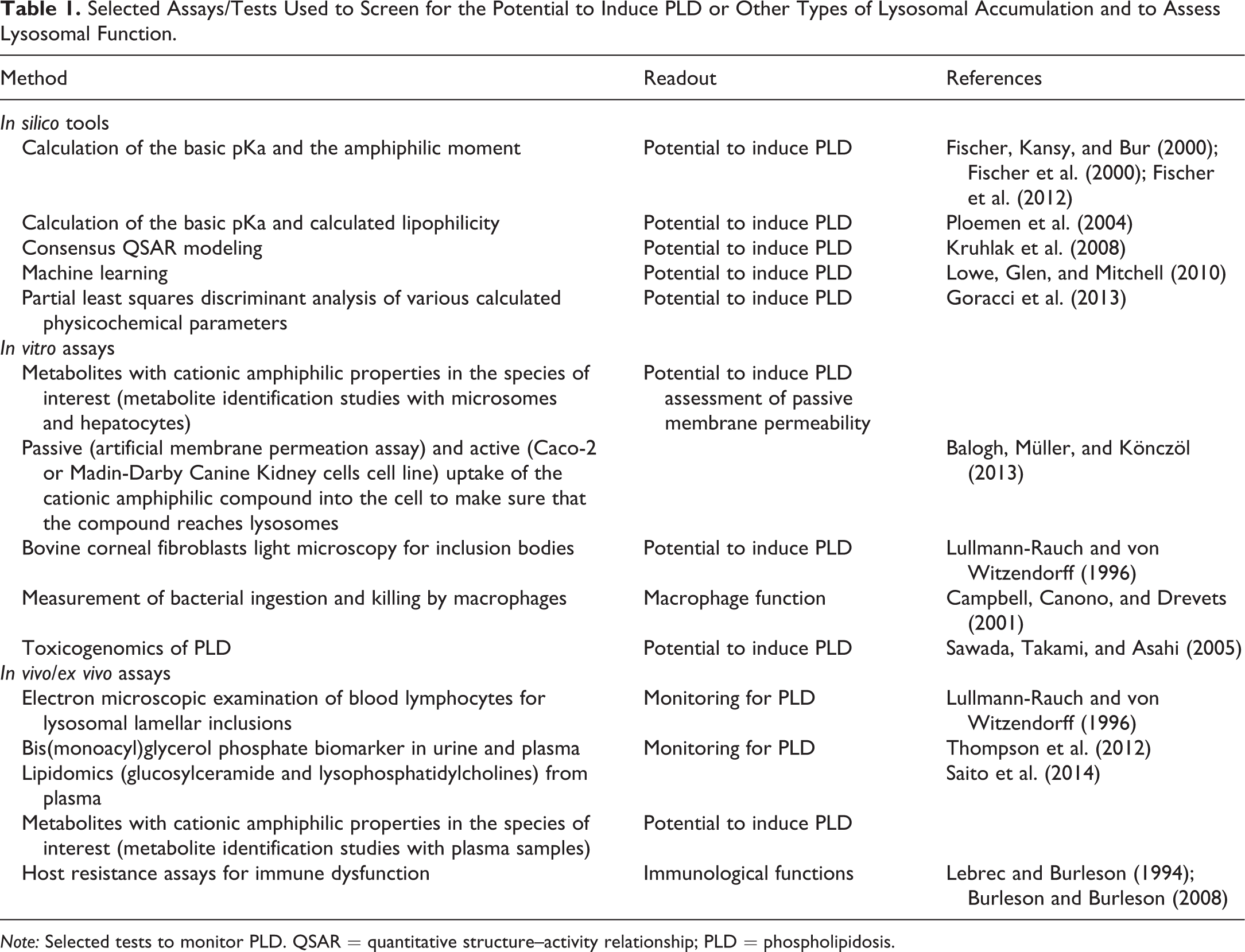
Note: Selected tests to monitor PLD. QSAR = quantitative structure–activity relationship; PLD = phospholipidosis.
Lysosomal Accumulation and Entities Discussed
A variety of endogenous and exogenous substances are known to enter lysosomes. Xenobiotics administered in nonclinical studies are often taken up by lysosomes as part of their role in removing foreign material. This general mechanism is relevant for PEGylated compounds and antisense oligonucleotides (ASOs). However, the specific physicochemical properties of exogenous compounds can also lead to their accumulation in lysosomes and eventually to a disturbance of lysosomal function. This is almost exclusively observed with cationic amphiphilic drugs (CADs). In their nonionized form, these compounds can easily traverse lipid bilayers. However, once within the lysosome, they are ionized by the acidic pH and become trapped, leading to the morphologic features of PLD.
Endogenous substances can also accumulate in lysosomes (Höhn and Grune 2013). A common example is lipofuscin, which is composed of aggregates of damaged proteins and lipid resulting from the degradation of cell components. Other examples include nutrients such as iron, which is recycled within lysosomes (Kurz, Eaton, and Brunk 2011), and proteins such as a2u, which is reabsorbed and degraded in proximal tubular epithelial cells of the kidney (Hard et al. 1993). If lysosomal digestion is inhibited or incomplete, accumulation occurs, resulting in lysosomal engorgement or increased numbers of lysosomes per cell. As with inborn LSDs, these changes can manifest microscopically as cytoplasmic granules, vacuoles, or droplets. Characteristic features of different compound classes/types of accumulation and methods for detection are shown in Table 2 and are discussed in detail below.
Characteristic Features and Methods for Detection of the Different Compound Classes/Types of Accumulation.
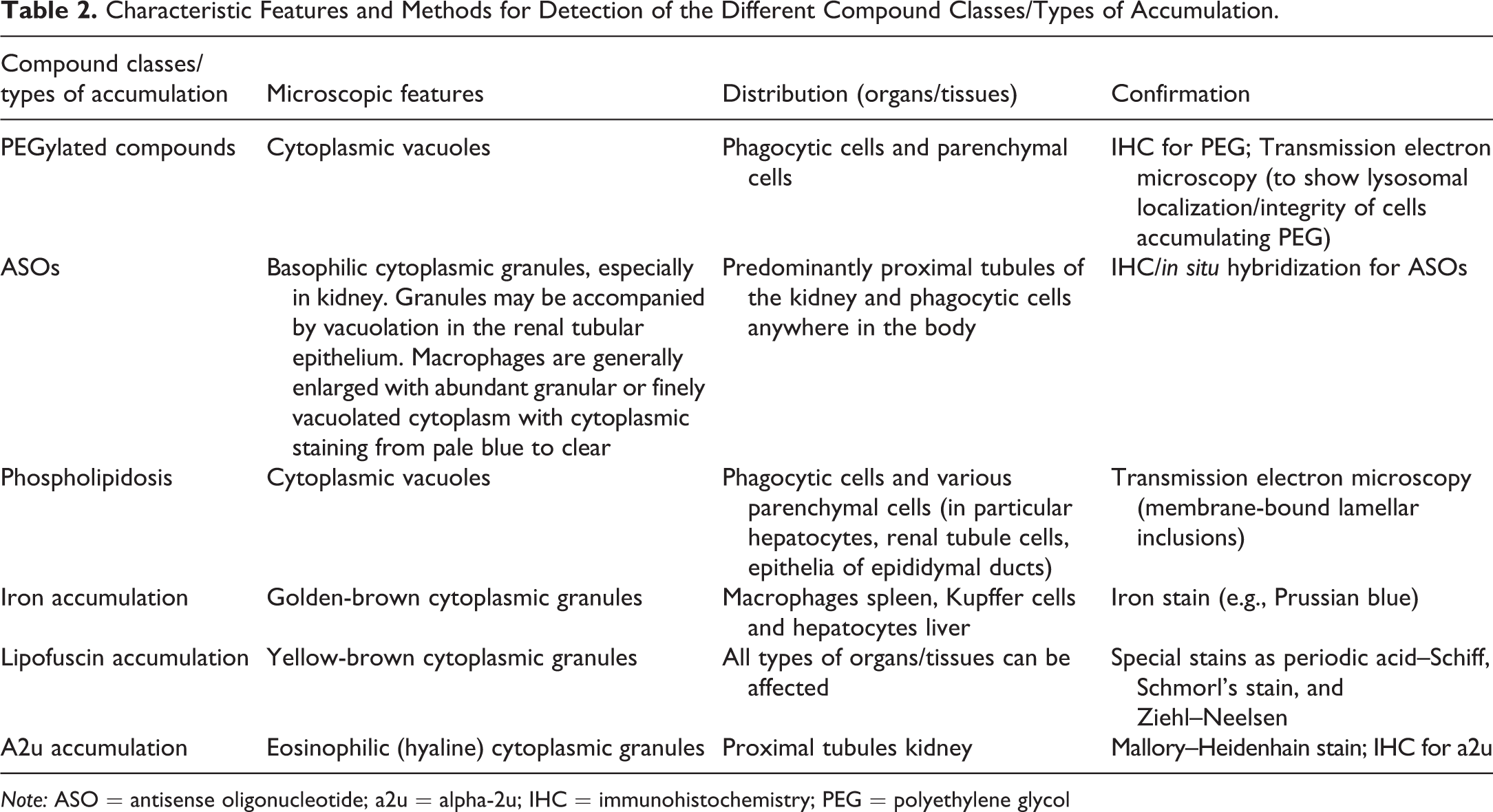
Note: ASO = antisense oligonucleotide; a2u = alpha-2u; IHC = immunohistochemistry; PEG = polyethylene glycol
PEGylated Compounds
PEG is a medical and industrial compound with a diverse array of commercial applications. Drug molecules (commonly peptides and proteins) can be PEGylated in order to improve pharmacokinetics and reduce immunogenicity. Currently, there are 12 PEGylated drugs on the market (Turecek et al. 2016). It has been postulated that PEGylated proteins are removed from circulation through the existing protein-specific mechanism, target-mediated uptake (including receptor-mediated uptake), and endocytosis (Baumann et al. 2014; Markovsky et al. 2012; Yu et al. 2004). The protein part of the complex is degraded by lysosomal enzymes within endolysosomes, while the PEG part is probably not metabolized, as there is a limited repertoire of enzymes available in lysosomes to perform this function. PEG, metabolic products, and unmetabolized PEGylated proteins are released by exocytosis or following cell turnover (Baumann et al. 2014). PEG then returns to the lymphatic system and the blood and is eliminated primarily by renal filtration with or without reabsorption by tubular epithelial cells.
It is hypothesized that PEG acts by attracting and binding water molecules. This hygroscopic action causes gradual fluid accumulation in lysosomes, leading to distension that is visible histologically as clear cytoplasmic vacuoles (Bendele et al. 1998). Baumann et al. (2014) suggested that several factors collectively influence the sites and extent of cell vacuolation, including the molecular weight and characteristics of PEG, the balance of nonspecific uptake versus receptor-mediated uptake, dose and dosing frequency, the uptake rate for specific cell types, and the rate of exocytosis and cell turnover of tissues accumulating PEG. In addition to phagocytic cells, PEG-associated vacuoles can be seen in a wide variety of parenchymal or interstitial cells across various tissues/organs including hepatocytes, renal tubular cells, thyroid gland follicular cells, endocrine cells of the pancreas and adrenal cortex, epithelial cells of the urinary bladder, epididymis, prostate, and mammary gland, endothelial cells of multiple organs, ciliary body cells of the eye, epithelial cells of the choroid plexus (CP), and neurons.
As with other findings, the interpretation of PEG-induced vacuolation is often based on biological context. Workshop participants noted that vacuolated phagocytic, parenchymal, or interstitial cells, in the absence of associated findings such as architectural distortion, necrosis, or inflammation, should not by default be considered adverse. Instead, location, severity, and reversibility (or partial reversibility) should be taken into account (Ivens et al. 2015). Also, in vitro tests for cellular function and capacity are viewed as valuable tools to inform potential adversity. For example, data from macrophage viability assays have been presented in submission packages of marketed compounds (Cimzia European Medicines Agency European public assessment reports). Vacuolation of neurons in the brain has been considered adverse, even in the absence of neuronal damage, because of the lack of recovery at this site and because it could not be completely ruled out that neuronal function was compromised (Example 1). The adversity decision might be more challenging if findings in the brain are limited to cytoplasmic vacuolation of the CP epithelium, as shown by a related example in cynomolgus monkeys treated for up to 52 weeks in which there was no evidence of cellular degeneration, necrosis, or architectural distortion, and clinical observations revealed no indications of neurologic dysfunction. However, no recovery was observed after 3 months, and severity was slightly increased in the female group at this time point. No additional adversity determinants could be defined based on the criteria defined by Palazzi et al. (2016), making the CP vacuolation in this case a difficult adversity call, especially when severity is low.
One issue raised by this latter example is whether a recovery period of 3 months is sufficient to definitively evaluate reversibility of effects at some sites, such as CP epithelium, where the cell turnover rate may be very low. A survey of 11 PEGylated pharmaceuticals, 7 of which demonstrated CP epithelium and/or pituitary gland vacuolation, indicated that standard recovery periods used in nonclinical toxicology studies are often too short to demonstrate reversibility but suggested reversibility of vacuoles when given sufficient recovery time. Workshop participants reported unpublished evidence of partial recovery of CP epithelium and/or pituitary gland vacuolation after recovery phases of 52 weeks in nonhuman primates (NHPs). In the case of CP epithelium, there is further uncertainty as to the turnover rate, functional impact on the production of cerebrospinal fluid, and local metabolism (Ivens et al. 2015). Therefore, data on viability and function of CP epithelial cells (e.g., ex vivo or in vitro studies) or pharmacodynamics may be supportive in the adversity assessment of nonreversible PEG-related vacuolation of CP epithelial cells, in particular for a prolonged administration in a pediatric population.
Example 1: PEG-associated lysosomal accumulation
Case Introduction
Several repeat-dose toxicity studies with a PEGylated peptide were performed up to 52 weeks in the rat and cynomolgus monkey. All studies described in this and subsequent examples were performed in compliance with animal welfare regulations.
Findings and Additional Investigations
The PEGylated compound was well tolerated in the rat and monkey with no clinical signs of adverse response or mortality. There were no changes in hematology, clinical chemistry, urinalysis, and urine chemistry. Vacuolation was observed in macrophages at the injection site (Figure 2A–C) and various organs in parenchymal cells of numerous tissues/organs including hepatocytes, endocrine, and exocrine glandular epithelium at many sites, ciliary body cells of the eyes, and neurons (Table 3, Figure 2D and E). The vacuolation was not associated with distortion of the organ, inflammation, and necrosis, and there were no changes in clinical pathology. The vacuolation was partially reversible or not reversible in the affected organs and not reversible in neurons. Neuronal vacuolation was not associated with neuronal damage as demonstrated with Fluoro-Jade staining. Immunohistochemistry (IHC) demonstrated the presence of PEG in neurons with or without vacuolation. Electron microscopy of the injection site revealed the presence of macrophages with storage vacuoles.
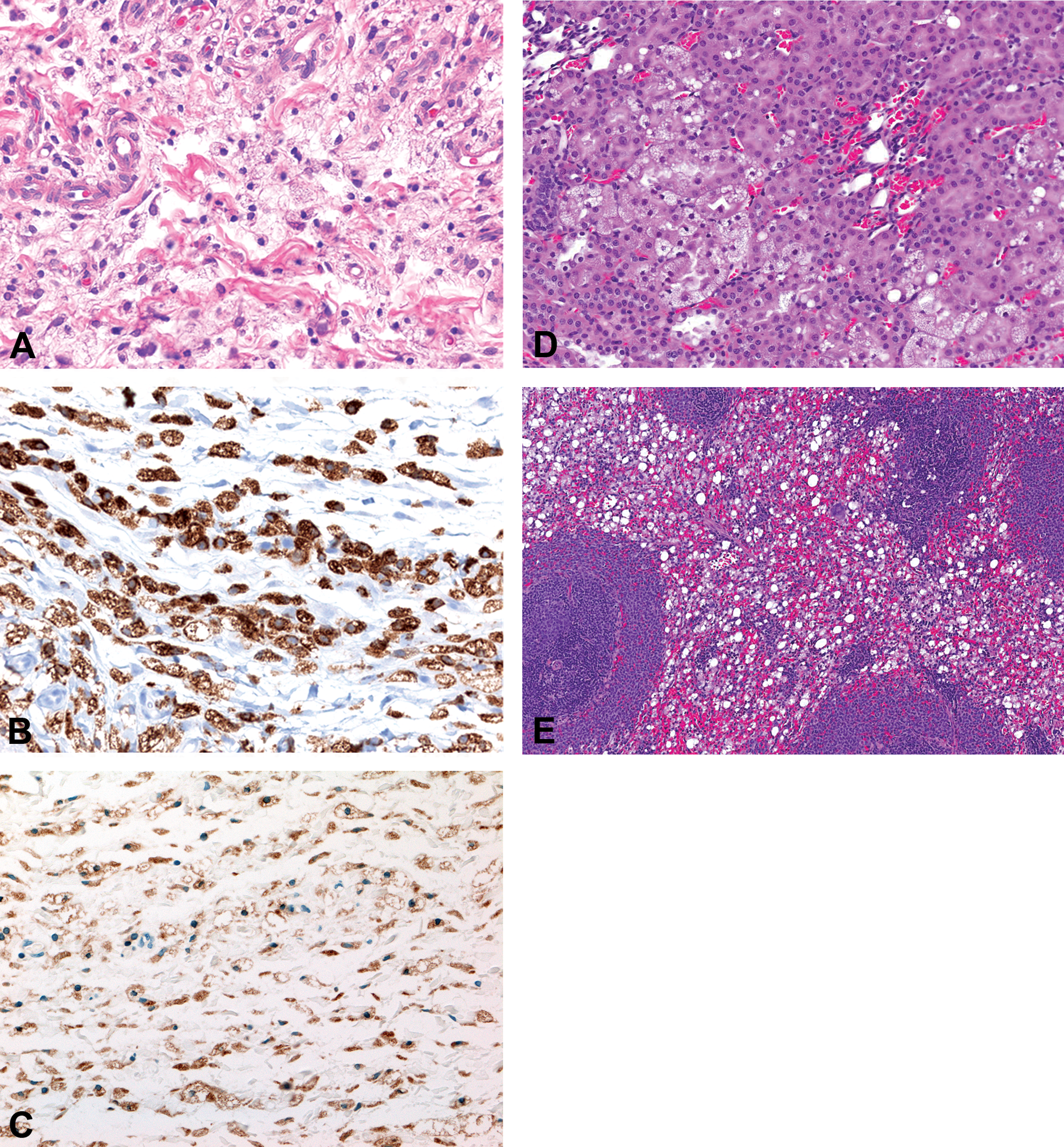
Wistar rats treated with a high dose of a polyethylene glycolylated peptide for 4 weeks (A–C) or 26 weeks (D, E). (A) Vacuolated macrophages at the injection site. Hematoxylin and eosin (H&E) stain and original objective 40×. (B) Vacuolated cells are confirmed to be macrophages based on CD68 labeling by immunohistochemistry. Original objective 40×. (C) Lysosomal associated membrane protein-2 immunohistochemical staining demonstrates lysosomal involvement. Original objective 40×. (D) Vacuolated renal tubular epithelium. H&E stain. Original objective 10×. (E) Vacuolated macrophages in the spleen. H&E stain. Original objective 10×. CD = cluster of differentiation.
Vacuolated Parenchymal Cells in Rat and Monkey Treated with a Polyethylene Glycolylated Compound.
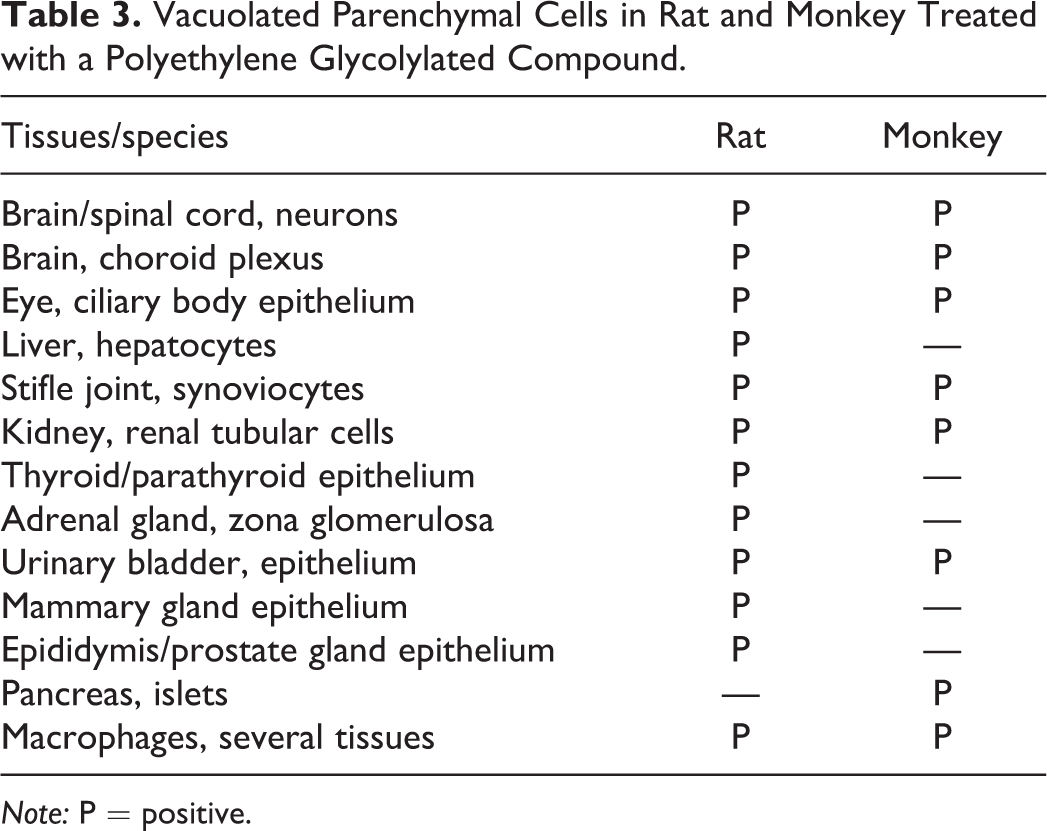
Note: P = positive.
Presenter’s Interpretation
The vacuolation of macrophages, parenchymal, and interstitial cells in various organs and tissues was not considered inherently adverse because it was not accompanied by inflammation, degeneration, or necrosis on histopathology or other indicators of cell injury or organ dysfunction, as suggested by Ivens et al. (2015). While the simple presence of PEG in neurons was considered target related and therefore not adverse, the presence of vacuolated neurons (without other evidence of cell injury) was considered adverse since chronic administration could compromise neuronal function in this poorly regenerative or nonrenewable cell type.
Antisense oligonucleotides
Histopathology findings related to accumulation of single-stranded ASOs are similar across animal species in preclinical studies. Changes are dependent on dose and tissue concentrations and reversible with tissue clearance of the drug. Intracellular uptake of ASOs is independent of the nucleotide sequence and greatest in cells with either prominent pinophagocytic properties or a well-developed endocytic-lysosomal apparatus. Targets include epithelial cells lining the proximal tubules of the kidney and phagocytic cells throughout the body, especially Kupffer cells in the liver and macrophages or histiocytes in lymphoid tissues.
First- and second-generation phosphorothioate-based (PS) ASOs, which generally have a negative charge, can interact with endogenous proteins in the plasma and on the cell surface and enter cells through nonspecific adsorptive endocytosis. In addition, PS ASOs can bind alpha-2 macroglobulin, which can then bind to specific cell membrane receptors on some cells. Tissue uptake of morpholino or peptide nucleic acid oligonucleotides, which have no or a low charge, and unmodified small interfering RNA is less effective relative to PS ASOs. It is considered that oligonucleotide can enter cells through different pathways and mechanisms of endocytosis, such as clathrin- or caveolin-mediated endocytosis as well as clathrin- and caveolin-independent pathways, as reviewed by Juliano and Carver (2015).
In the kidney, ASOs filtered through the glomeruli are readily reabsorbed by the proximal tubular epithelium via the brush border and processed into endolysosomes, as confirmed by ultrastructural analysis (Rappaport et al. 1995). In light microscopic evaluation, these endolysosomes appear as variably sized cytoplasmic granules exhibiting basophilia in hematoxylin and eosin (H&E)-stained sections because ASOs are similar to native nucleic acids, such as RNA and DNA, in that they stain with hematoxylin. However, in NHPs, ASO-related cytoplasmic granules may also exhibit variable degrees of eosinophilia. Common histologic features in the kidneys of ASO-treated animals include the presence of basophilic granules in normal or minimally enlarged proximal tubular epithelial cells, without evidence of cellular degeneration, at cortical ASO concentrations up to 3,000 µg/g (typically seen following once weekly doses of 20 mg/kg for ≥13 weeks). With some ASO chemistries, however, such as third-generation phosphorodiamidate morpholino–modified sequences, basophilic cytoplasmic granules in the epithelium of renal tubules and histiocytes are not a typical histological feature (Carver et al. 2016).
Oil Red O-negative cytoplasmic vacuolation or presence of a perinuclear unstained halo sometimes accompanies the basophilic granules at higher kidney concentrations (>1,500 µg/g) in NHPs and rats. These are considered secondary to extraction of the ASO from endolysosomes during tissue fixation/processing and/or water accumulation within ASO-containing endolysosomes due to the osmotic properties of ASOs. This vacuolation generally does not occur in mice. ASOs are highly water soluble and extractable, leading to this change in formalin-fixed tissues, whereas the vacuolation is less prevalent in tissue perfused with glutaraldehyde. It was demonstrated in the examples shared at the workshop that ASO accumulation, in and of itself, was not associated with renal tubular dysfunction. There were no associated increases in serum blood urea nitrogen or creatinine, kidney resorptive functions were maintained, and urinalysis end points were typically within normal limits (Engelhardt 2016).
Macrophages that have engulfed ASOs generally are enlarged and have abundant granular or finely vacuolated cytoplasm that is considered to primarily reflect the lysosomal accumulation of ASO material (Henry et al. 2008; Braendli-Baiocco et al. 2017). However, accumulation of cytokines and/or other secretory substances must also be considered due to tinctorial variations in cytoplasmic staining from pale blue to clear (Engelhardt et al. 2015). Importantly, no demonstrable effect on macrophage function was observed in host defense assays in mice treated for 4 weeks with ASOs prior to infection or in in vitro phagocytosis assays with peritoneal macrophages harvested from mice treated for 4 weeks with ASOs (internal company data).
Therefore, given that (1) there was no effect on net immune capability of the host to clear infectious agents, (2) there were no effects on phagocytosis or killing capability of macrophages, and (3) ASO accumulation was not associated with renal tubular dysfunction and did not progress in severity with increased duration, the lysosomal accumulation of ASOs was not considered to represent an adverse effect. Additional details are provided in Example 2.
Example 2: Basophilic granulation of lysosomes in kidney and macrophages in NHPs given an ASO
Case Introduction
A 5-10-5 2′-methoxy-ethyl ASO was given to NHPs at 0 (control) low, mid, and high doses once weekly via subcutaneous injection for 9 months.
Findings and Additional Investigations
Basophilic granulation of the cytoplasm of proximal tubular epithelium, Kupffer cells in the liver, and sinusoidal macrophages in lymph nodes were readily evident in the high-dose group on light microscopy (Figure 3A and B). Vacuolation of tubular epithelial cells was common in mid- and high-dose animals and associated with higher tissue concentrations of drug. Aside from the vacuolation, there were no cytological features of degeneration and/or necrosis. Sinusoidal macrophages had abundant blue-gray clear cytoplasm with deep to faint basophilic granules and occasional clear vacuoles. In addition to the hypertrophy of macrophages, there was a perceptible increase in numbers of cells within the sinusoids. Basophilic granulation and cytoplasmic vacuolation generally resolved within 3 months when drug treatment was discontinued.
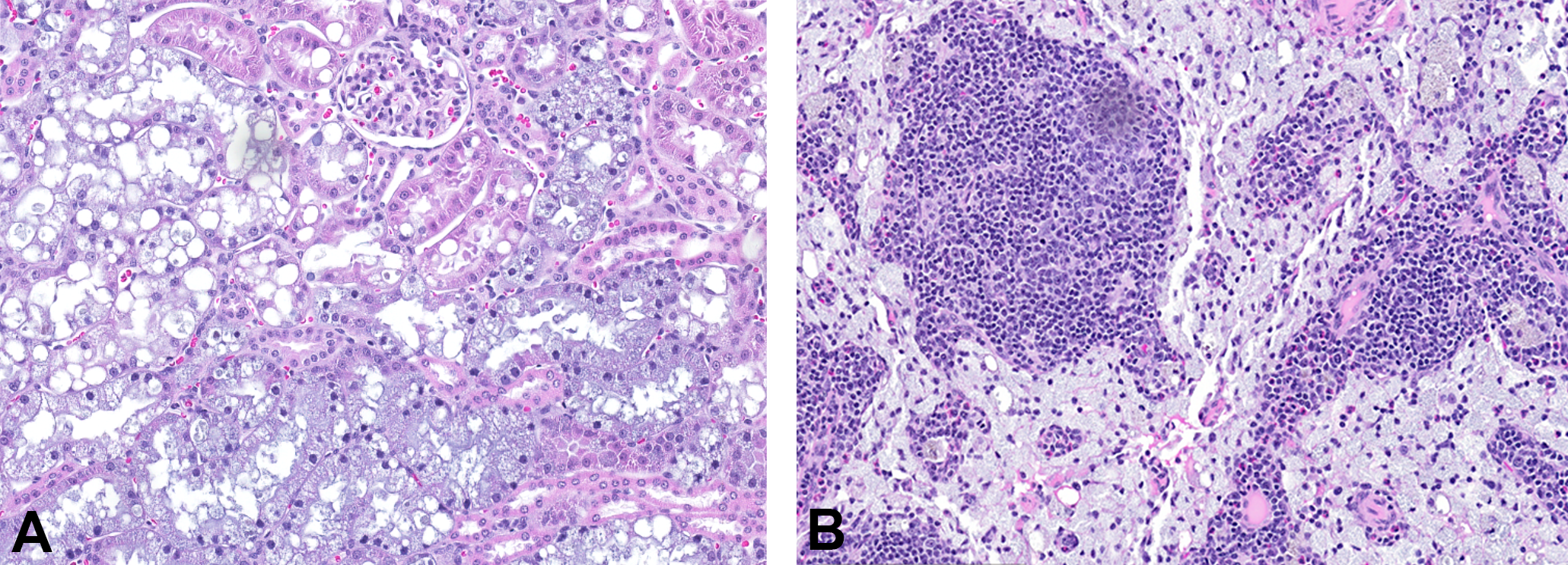
Nonhuman primates treated with a high dose of an antisense oligonucleotide once weekly for 9 months. (A) Kidney; note the enlarged epithelial cells with abundant blue-gray cytoplasm, basal displacement of nuclei, variably sized cytoplasmic vacuoles, and presence of basophilic granules. Original objective 20×. (B) Lymph node; note the hypertrophied sinusoidal histiocytes showing abundant blue-gray finely granular cytoplasm. Original objective 20×.
Presenter’s Interpretation
The accumulation of basophilic granules in these cells at doses up to the high dose was not considered to be adverse, as it was not associated with concurrent inflammation, degeneration, or necrosis, and there were no indications of altered renal function. It was interpreted that the presence of basophilic granules represents the uptake of ASOs into lysosomes, an adaptive change that does not represent an adverse effect on cell structure or function.
Phospholipidosis
PLD is characterized by lysosomal accumulation of phospholipids. It is often caused by CADs but also by some cationic hydrophilic drugs such as gentamycin. Several mechanisms are discussed, which may lead to PLD, including (1) indirect inhibition of the phospholipase activity due to massive accumulation of compound in the lysosomes, and particularly in the inner leaflet of the membrane, which changes local conditions critical for lysosomal activity (Lüllmann, Lüllmann-Rauch, and Wassermann 1975); (2) direct enzyme binding and inhibition of phospholipase activity; (3) enhanced phospholipid synthesis; and (4) disturbance of intracellular trafficking and compartmentalization of phospholipids (Sawada, Takami, and Asahi 2005; Ikeda et al. 2008). The first mechanism appears to be the most prevalent since the vast majority of small molecules associated with PLD are membrane-permeable CADs. Recent studies have demonstrated that for a well-known PLD-inducing compound, amiodarone, a massive compound accumulation could be detected by correlative nanosecondary ion mass spectrometry and electron microscopy in lysosomes with multilamellar bodies (Jiang et al. 2017).
The hallmark of PLD is cellular vacuolation, which may affect multiple cell types. Macrophages, particularly alveolar macrophages in the lungs (Halliwell 1997), and epithelial cells (e.g., hepatocytes, renal tubular cells, and epithelial cells of epididymal ducts) are commonly affected. However, other cells types such as endocrine cells, striated and smooth muscle cells, vascular endothelial cells, and nerve cells in central and peripheral nervous system, or certain compartments in the eye can also be involved (Reasor, Hastings, and Ulrich 2006, Nonoyama and Fukuda 2008). Electron microscopic features include increased size and number of lysosomes containing concentric membrane-bound lamellar bodies. These features are considered to reflect an accumulation of the inducing drug or its metabolite together with accumulated phospholipids (Reasor, Hastings, and Ulrich 2006; Jiang et al. 2017).
Effects on lysosomes can be deleterious on the entire cellular system, and CADs may act beyond the lysosome. For example, CADs are excellent at crossing the cell membrane, allowing them to exert effects inside the cell. They are also able to cross the blood-brain barrier, making them important structures for drugs requiring CNS activity. A number of CADs have been reported to be associated with findings other than PLD. These findings include QT prolongation via inhibition of the human Ether-a-go-go Related Gene potassium ion channel (L. Doessegger, E. A. Atzpodien, M. Csato, M. Cukierski, P. Day-Lollini, H. Fischer, M. Irwin, et al., pers. commun.; Willard 2010; Sun et al. 2013), mitochondrial toxicity (Begriche et al. 2011), P-glycoprotein mediated efflux, and effects on ion channel activity (Lundbaek, Koeppe, and Andersen 2010). Generally, organs presenting with PLD also have higher rates of associated injury, indicating potential damage beyond the lysosomes. There are currently no conclusive data as to whether PLD is a cause of cellular or tissue dysfunction or whether they are a separate effect of the drug itself (Reasor, Hastings, and Ulrich 2006). Several investigators have examined the effects of PLD on cell/tissue function, in particular pulmonary function, but the reported results have not been consistent (Reasor, Hastings, and Ulrich 2006).
An adversity decision concerning cases of PLD requires careful assessment of all data available, as shown by the examples shared at the workshop, and there is currently no consistency in the assessments between agrochemical and pharmaceutical industries. In pharmaceutical development, PLD is commonly considered not adverse as long as changes are not accompanied by additional/secondary pathological changes (cellular damage and/or tissue reaction such as inflammation), do not involve critical organs/tissues (e.g., the CNS, the heart, and sensory organs such as the eye), and do not occur in a generalized fashion (e.g., with numerous parenchymal cells being affected). See Examples 3–5 for additional details.
Careful consideration is given by regulatory agencies when presented with PLD submissions (see Example 6). At present, PLD findings in toxicology studies of pharmaceuticals are evaluated on a case-by-case basis. The U.S. Food and Drug Administration (FDA), for example, instituted the Phospholipidosis Working Group (PLWG) in 2004 to better understand PLD and to develop a more consistent approach for the evaluation of drug-related PLD. The relationship between PLD, signs of histopathological injury, and histopathological adversity in the PLD-exhibiting organ has not always been well understood. In pursuit of these goals, the FDA PLWG began a database of PLD-causing compounds, organs involved, and histopathological assessments. In addition, the FDA PLWG collaborated with the quantitative structure–activity relationship (QSAR) group at the FDA on structural methods of predicting PLD, and with the laboratory research group examining nonelectron microscopic methods for determining PLD, as well as looking at potential biomarkers (e.g., bis(monoacylglycerol)phosphate). This database has revealed that the number of PLD-inducing drugs is much larger than previously thought, with over 350 compounds identified to date. This scope highlights the importance of methods for assessing adversity of lysosomal accumulation and appropriate regulation of these compounds.
Although more commonly reported for pharmaceuticals, PLD has also been reported for selected classes of environmental chemicals. A presented example of a cationic amphiphilic agrochemical, developed as an insecticide, showed findings consistent with PLD in dogs, rats, and mice (Yano et al. 2002). Effects were characterized by cytoplasmic vacuolation or aggregation of foam cells in various organs and tissues, including lung, heart, liver, pancreas, kidney, reproductive organs, pituitary, adrenal, thyroid, parathyroid, and lymphoid, although the target sites differed slightly between species. PLD was confirmed by electron microscopy. For food-use pesticides, the Food Safety Commission of Japan and others use the acceptable daily intake (ADI), which is a guidance value for long-term exposure to pesticides based on the lowest no observed adverse effect level (NOAEL) for the most sensitive end point(s) in various toxicity studies. If PLD induced by a compound is the most sensitive end point for setting ADI, then PLD would generally be considered an adverse effect unless there was clear and adequate evidence to indicate that the PLD is not relevant to humans. Therefore, in 1 agrochemical example presented at the workshop but not shown as example in this article, the presence of PLD in any tissue was interpreted as an adverse effect, and the NOAEL was set at the highest dose not revealing PLD.
Example 3: PLD induced by a small molecule pharmaceutical compound in rats and monkeys
Case Introduction
A small pharmaceutical molecule was intermittently given to rats and monkeys in 90-day subchronic studies. This molecule was not a typical CAD, was not metabolized, had a low volume of distribution, and was almost entirely excreted through the kidney.
Findings and Additional Investigations
PLD presented as cytoplasmic vacuolation, which was confirmed by electron microscopy. It was identified in both rats and monkeys predominantly in the epithelium of the urinary tract via exposure to the molecule during excretion and perivascular interstitial macrophages in the majority of organs examined (Figure 4A–D). This finding was not evident in the eye or brain. At the highest evaluated dose in rats, there were 2 unexplained deaths and microscopic thymic depletion that suggested potential phospholipid overload. Partial reversibility at 4 weeks after the completion of the dosing period was seen at all doses. Increased serum urea and creatinine in rats and urinary protein excretion in rats and monkeys provided appropriate markers for evaluation in clinical trials.
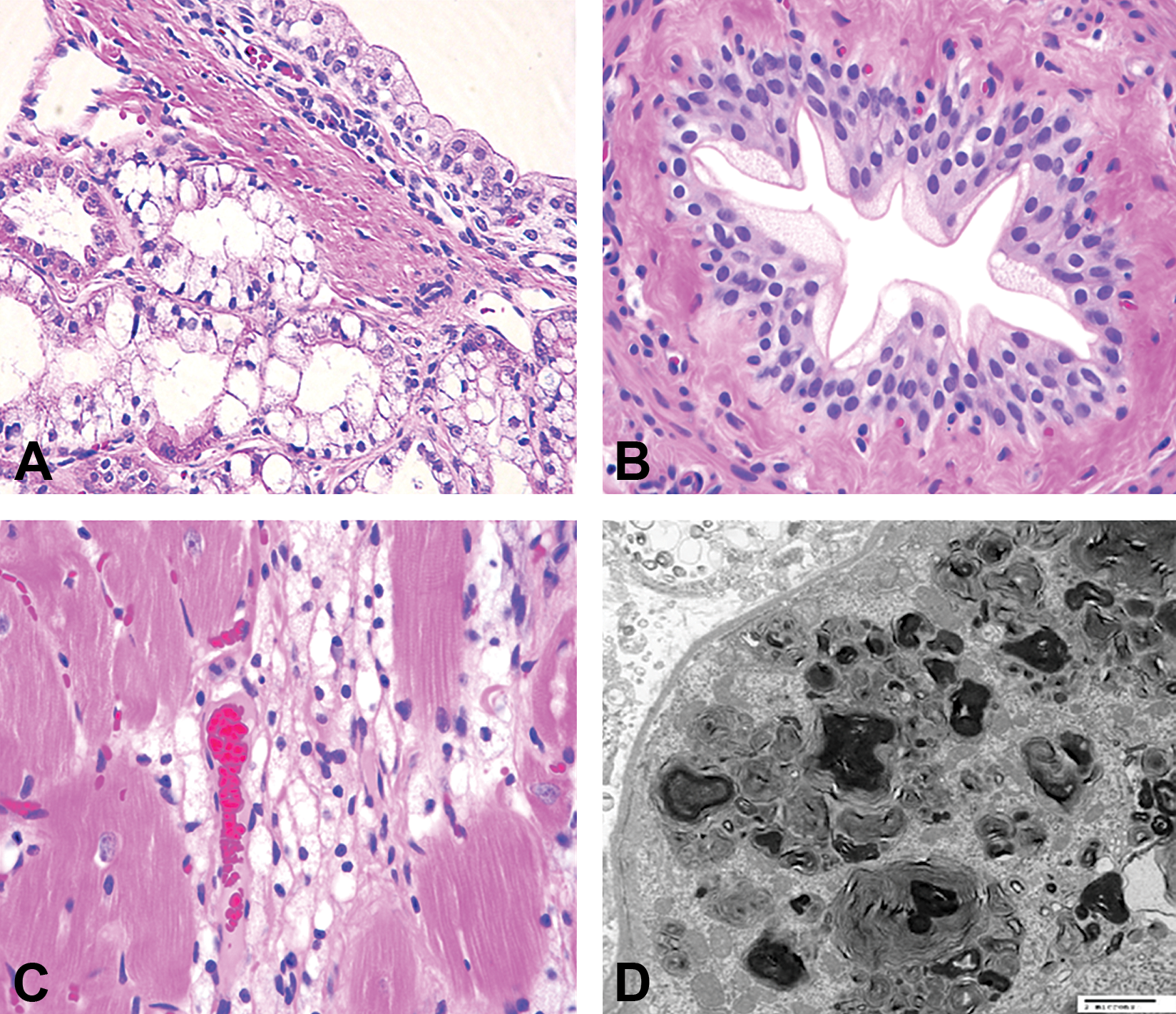
Sprague-Dawley rats treated with a high dose of a small pharmaceutical molecule for 90 days. (A) Kidney indicating tubular and pelvic epithelial vacuolation; hematoxylin and eosin (H&E) stain and original objective 10×. (B) Ureter indicating epithelial vacuolation; H&E stain and original objective 20×. (C) Heart indicating interstitial macrophages with vacuolation; H&E stain and original objective 20×. (D) Electron micrograph of kidney with intracellular membrane-bound lamellated structures.
Presenter’s Interpretation
The accumulation of phospholipid was not considered adverse because it was not associated with tissue inflammation or degeneration. However, due to the potential for phospholipid overload associated with 2 deaths at the high dose in rats, that dose in that species was considered adverse. Regulators supported moving forward in the clinic with monitoring of renal biomarkers.
Example 4: PLD induced by a small molecule pharmaceutical compound in rats
Case Introduction
A series of endopeptidase isoenzyme inhibitors were given once daily to rats in 1-week exploratory toxicity studies. The compounds were not typical CADs.
Findings and Additional Investigations
PLD was noted in the liver and confirmed by special stains including negative Oil Red O and positive Sudan Black staining and the typical lamellar cytoplasmic bodies by electron microscopy (Figure 5A and B). Different chemical series were screened in a 24-hr hepatocyte culture with test material and fluorescently labeled nitrobenzoxadiazol-phosphoethanolamine. Chemical compounds that resulted in the incorporation of the fluorescently labeled phospholipid (Figure 5C and D) all contained a piperidine ring featuring a nitrogen. This assay was used to determine the structure–activity relationship for PLD in the program. The phospholipid accumulation was not associated with tissue degeneration or altered tissue function.
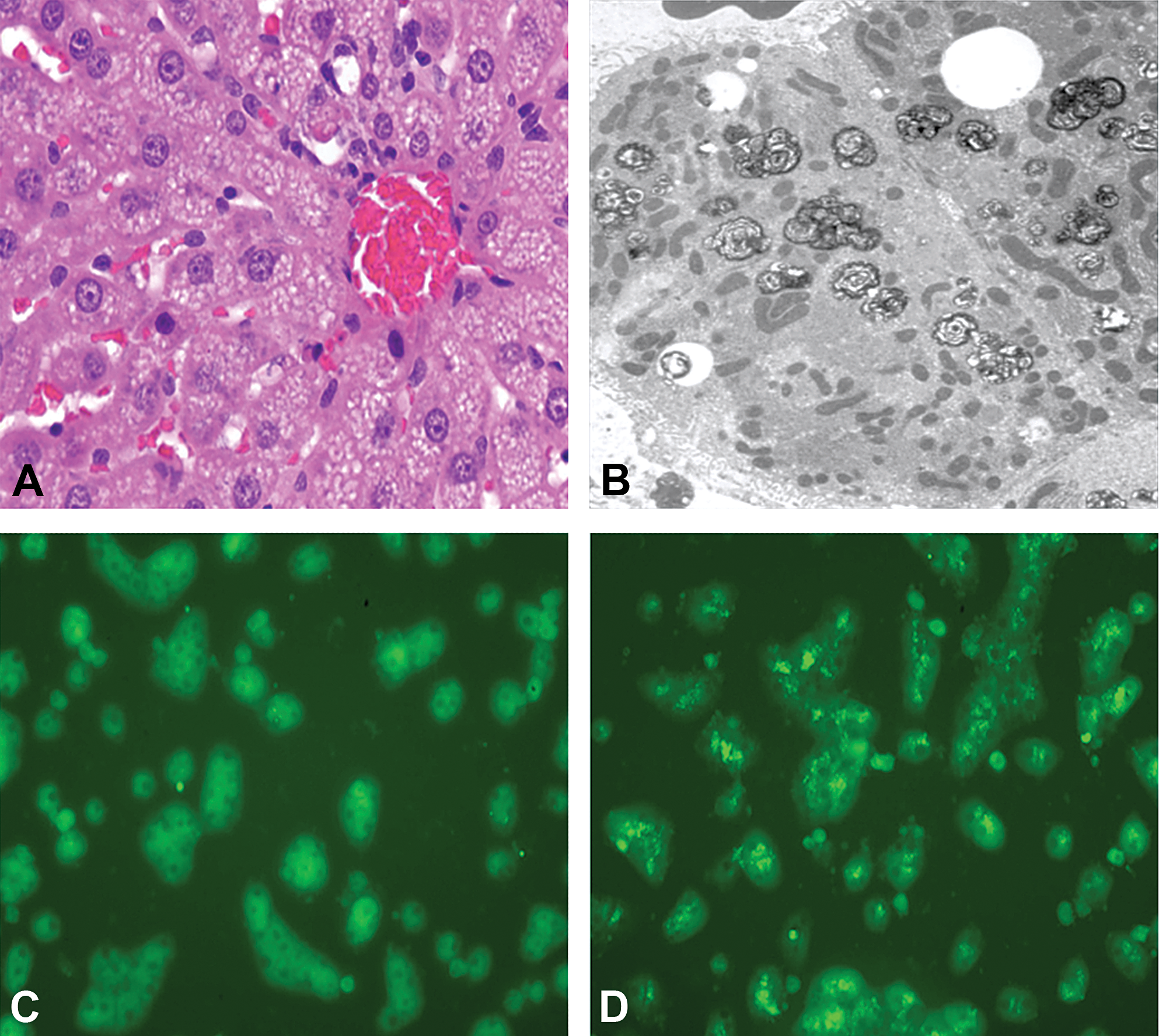
Sprague-Dawley rats treated with a high dose of a small pharmaceutical molecule for 1 week. (A) Liver; centrilobular hepatocytes with phospholipid accumulation. Hematoxylin and eosin stain and original objective 20×. (B) Electron micrograph of hepatocyte with membrane-bound lamellated structures. (C, D) 24-hr hepatocyte culture with test material and fluorescently labeled nitrobenzoxadiazol-phosphoethanolamine identified the key structure relationship with chemical series containing a piperidine ring with negative (C) and positive (D) results.
Presenter’s Interpretation
Due to the lack of tissue degeneration or other evidence of altered tissue function, PLD was not considered adverse. However, due to a low projected therapeutic margin and evaluation for a nonlife-threatening indication requiring chronic use, the hepatocyte assay was used to deprioritize compounds causing PLD.
Example 5: Drug-induced PLD in bile duct epithelium in rat
Case Introduction
A compound with high affinity for liposomes was administered orally to Sprague-Dawley rats for 2 weeks at 0 (control), low, mid, and high doses. Alerts were given by in vitro toxicogenomics–based PLD and cytotoxicity assays. Inhibitory effects on phospholipase A1 and A2 were confirmed in the additional promiscuity panel.
Findings and Additional Investigations
In the high-dose group, decreases in body weight gain and food consumption, an increase of alanine aminotransferase (ALT), and enlargement of mesenteric lymph node were observed. Histologically, cytoplasmic vacuolation in intra- and extrahepatic biliary epithelium from the mid dose onward, inflammation of intra- and extrahepatic biliary ducts, and foamy macrophages in the mesenteric lymph node at the high dose were observed. There were no changes suggesting PLD in other organs and tissues.
Presenter’s Interpretation
PLD occurred in a nongeneralized fashion in this case. Affected cell types were limited to biliary epithelial cells and macrophages in the mesenteric lymph node. The bile duct inflammation was considered to be a consequence of the necrosis of the bile duct epithelium due to severe lysosomal accumulation of phospholipid and test compound with cytotoxic profile (Figure 6A and B, Figure 7A–H). This secondary change was the basis of the adversity assessment of PLD at the high dose. The NOAEL was set at the mid dose. PLD of the biliary epithelium at this dose was judged as not adverse because of lower severity and the lack of histopathological findings other than vacuolation.
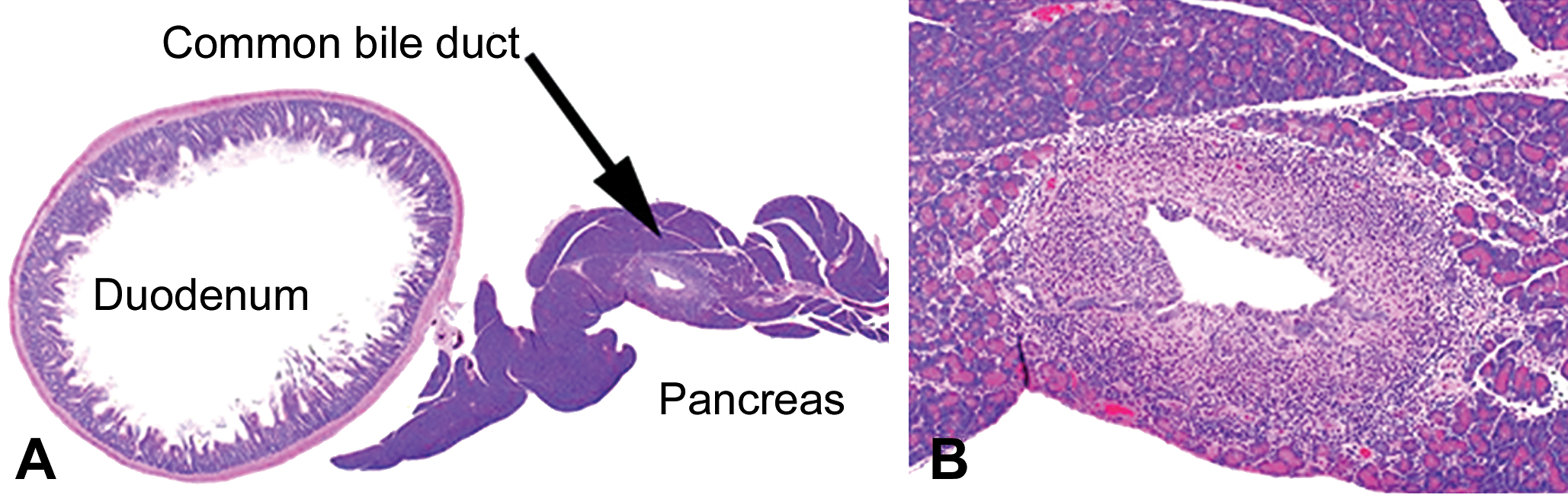
Sprague-Dawley rats treated for 2 weeks with a compound with high affinity for liposomes. Phospholipidosis accompanied by inflammation in the extrahepatic (common) bile duct in a rat treated at the high dose for 2 weeks. (A) Common bile duct running through the pancreas. Original objective 1.25×. (B) Inflammatory cells infiltrating the ductal wall. Hematoxylin and eosin (H&E) stain. Original objective 4× (B).
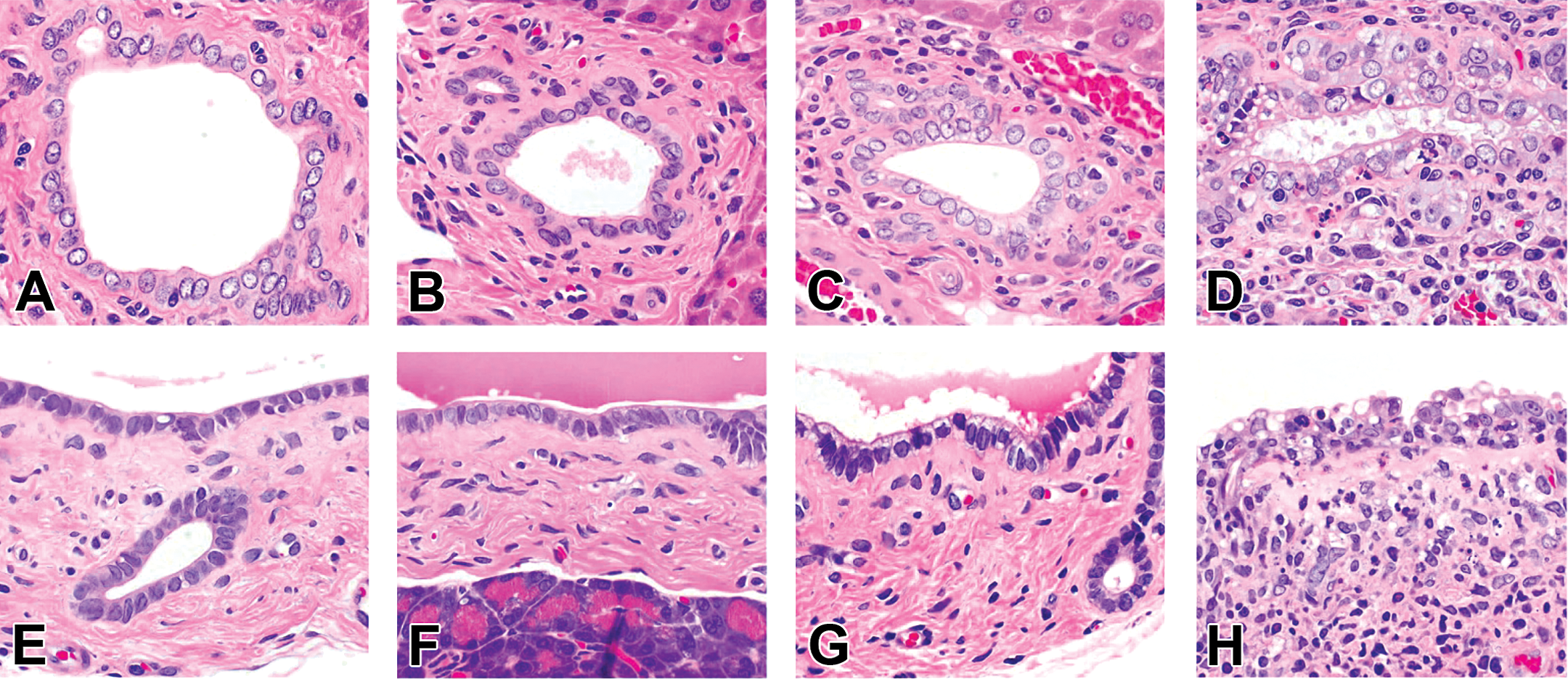
Sprague-Dawley rats treated for 2 weeks with a compound with high affinity for liposomes. Higher magnification of the intra- and extrahepatic biliary epithelium. Intrahepatic biliary epithelium from control (A), low dose (B), mid dose (C), and high dose (D). Extrahepatic biliary epithelium from the control (E), low dose (F), mid dose (G), and high dose (H). Fine vacuoles present in the apical portion of the epithelium at the mid dose (C, G). Larger vacuoles at the high dose and necrosis of biliary epithelium and inflammatory cell infiltration (D, H). H&E stain; original objective 20×.
Example 6: Two PLD case studies illustrating communication of potentially adverse pathology findings in regulatory submissions related to lysosomal accumulation
Case Introduction
CADs easily access the CNS and thus provide attractive candidates for development as antipsychotic medications. A downside to their successful drug development is that they are often accompanied by PLD. Two regulatory review case studies are presented: (1) cariprazine, antipsychotic medication indicated for bipolar disorder and schizophrenia and (2) pimavanserin, an antipsychotic indicated for Parkinson’s disease psychosis. Both cases presented with a histopathological diagnosis of PLD and a separate diagnosis of pulmonary fibrosis. Pulmonary inflammation in response to the PLD was not reported in the submissions. The cardiac drug amiodarone, which causes primary pulmonary fibrosis, is the classic example of adversity associated with PLD in the lung. Four different types of potentially fatal amiodarone–related pulmonary fibrosis are known, most of which are progressive and do not respond to discontinuation of the drug. To rule out a possible amiodarone-like primary pulmonary fibrosis, both case examples were submitted for a consult regulatory pathology review to provide an expert opinion on the findings to assist the review division in their safety and risk evaluation.
Findings and Additional Investigations
A careful reevaluation of the lung histopathology slides (cariprazine) and/or histopathological data of the submissions (cariprazine and pimavanserin) by Center for Food Safety and Applied Nutrition pathologists revealed an underlying inflammatory response at higher doses, mainly characterized by the formation of very large, multinucleated giant cells distended by eosinophilic material often containing cholesterol clefts, and accompanied by a subtle amount of mixed inflammatory cells (Figure 8A). With chronicity, fibrosis was also part of this PLD-associated inflammatory response.
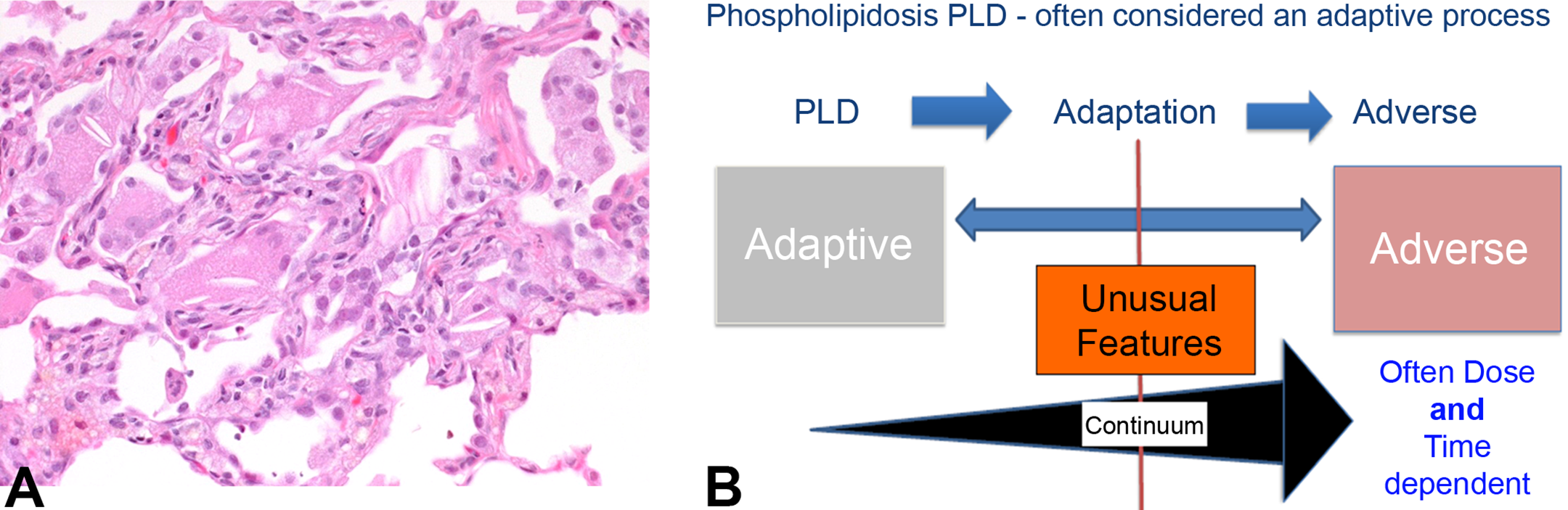
Dog treated with a high dose of cariprazine (6 mg/kg/day) for 1 year. (A) Pulmonary phospholipidosis with chronic inflammation characterized by mainly multinucleated giant cells with cholesterol clefts and occasional inflammatory cell (lymphocytes, plasma cells, or polymorphonuclear cells) and minimal interstitial fibrosis. Hematoxylin and eosin stain and original objective 20×. (B) Continuum of phospholipidosis evolving from an adaptive to an adverse response.
Presenter’s Interpretation
Although PLD is often considered an adaptive process, a shift to the adverse manifestations (e.g., inflammatory response) may become more apparent with increasing dose (Figure 8B). Unusual features, such as multinucleated giant cell with cholesterol cleft formation at higher doses, may be predictive of these changes occurring with long-term use and at lower doses. For these case examples, an early careful description of all changes associated with the PLD, such as subtle inflammation with unusual features (multinucleated giant cells with cholesterol cleft formation), would have enabled the sponsor to successfully explain minimal-to-mild chronic pulmonary inflammatory fibrosis at higher doses and/or after long-term use.
Iron accumulation
Iron is an essential nutrient that plays a key role in numerous cellular processes including oxidative respiration (e.g., by cytochromes and mitochondria) and oxygen storage and transport (e.g., hemoglobin and myoglobin). The main regulator of serum iron level is hepcidin, which acts via binding and neutralization of ferroportin, the main iron cell exporter located on enterocytes and macrophages. There is no physiological excretion pathway for iron present in the body. Iron homeostasis thus relies on the regulated balance of iron absorbed and released from the enterocytes of the small intestine and recycled iron mainly provided by macrophages of the spleen. Small amounts of iron are lost from the body physiologically by gastrointestinal blood loss, sloughing of the intestinal mucosa and skin, and via urinary and biliary excretion. Iron is a key factor for erythropoiesis, and iron deficiency typically leads to anemia. Alternatively, excess iron deposition (hemochromatosis), due to hereditary diseases, dietary conditions (e.g., wildlife species kept in captivity), and transfusions, can lead to various degenerative conditions including liver cirrhosis and cancer, diabetes (toxic iron deposition in pancreatic β cells), cardiomyopathy or congestive heart failure, arrhythmias, change in skin color, arthritis, and testicular atrophy.
Test item–induced intracellular iron accumulation occurs mainly (1) secondary to hemolysis (e.g., induction of hemolytic anemia, with Heinz bodies or Coombs-positive conditions) or (2) due to downregulation of hepcidin (with decreased hemojuvelin and bone morphogenetic protein 6). Assessment of the adversity of iron accumulation in cells generally requires an evaluation of its pathophysiological mechanism and is a case-by-case decision. Hemorrhage or hemolysis could be the primary and adverse effects of test item administration, whereas the accompanying iron-positive pigment accumulation in Kupffer cells might be assessed as nonadverse if the magnitude is low and evaluated in isolation (Example 7). In an example of test item–induced hepcidin downregulation, iron accumulation in hepatocytes was assessed nonadverse since neither degenerative nor inflammatory reactions were present, and iron accumulation was reversed (Example 8).
Example 7: Iron pigment accumulation due to hemolysis
Case Introduction
Dogs were treated with a new anticonvulsive drug for 13 weeks. A previous 4-week study showed sedation and increased liver weights but was otherwise unremarkable. At weeks 6 to 9, individual dogs from the mid- and high-dose groups showed signs of hemolytic anemia, autoagglutination, and icterus and had to be sacrificed.
Findings and Additional Investigations
The Coombs test was positive in the affected animals. The liver and spleen showed marked accumulation of hemosiderin as well as extramedullary hematopoiesis/erythropoiesis (Figure 9A–D).
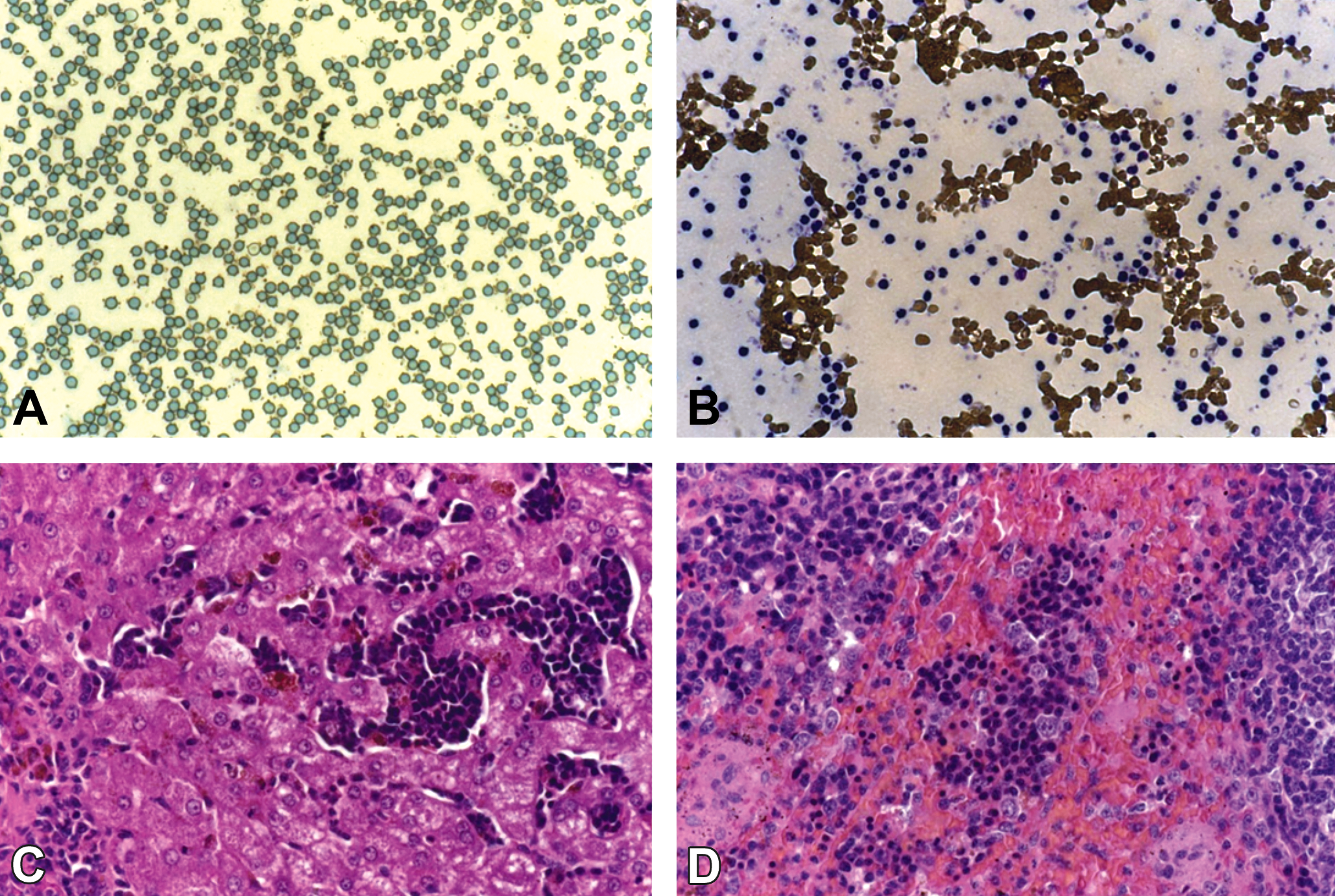
Dogs treated with a high dose of an anticonvulsive drug for 13 weeks. Blood films showing autoagglutination and varied individual responses. (A) Small aggregates of erythrocytes and platelets with paucity of white blood cells (WBCs). (B) Large aggregates with markedly increased numbers of WBCs. Original objective 63× for blood film and 20× for slides. (C, D) Hematoxylin and eosin–stained sections of liver (C) and spleen (D) showing marked extramedullary erythropoiesis and deposition of hemosiderin in Kupffer cells, macrophages, and hepatocytes. Original objective 20×.
Presenter’s Interpretation
The extensive accumulation of hemosiderin and extramedullary hematopoiesis is secondary to the induced hemolytic anemia. The latter finding is the primary adverse effect. The accumulation of hemosiderin is expected to be reversible and by itself not considered to be adverse. However, it does represent a signal for an adverse response (i.e., hemolytic anemia).
Example 8: Iron overload (hemochromatosis) due to hepcidin downregulation
Case Introduction
Rats and cynomolgus monkeys were treated with a hemojuvelin-neutralizing monoclonal antibody.
Findings and Additional Investigations
Animals showed an increase in iron level and reduced iron-binding capacity in serum. The liver showed periportal hepatocellular iron (Prussian blue positive) accumulation, and there was iron release from splenic macrophages (Figure 10A–D).
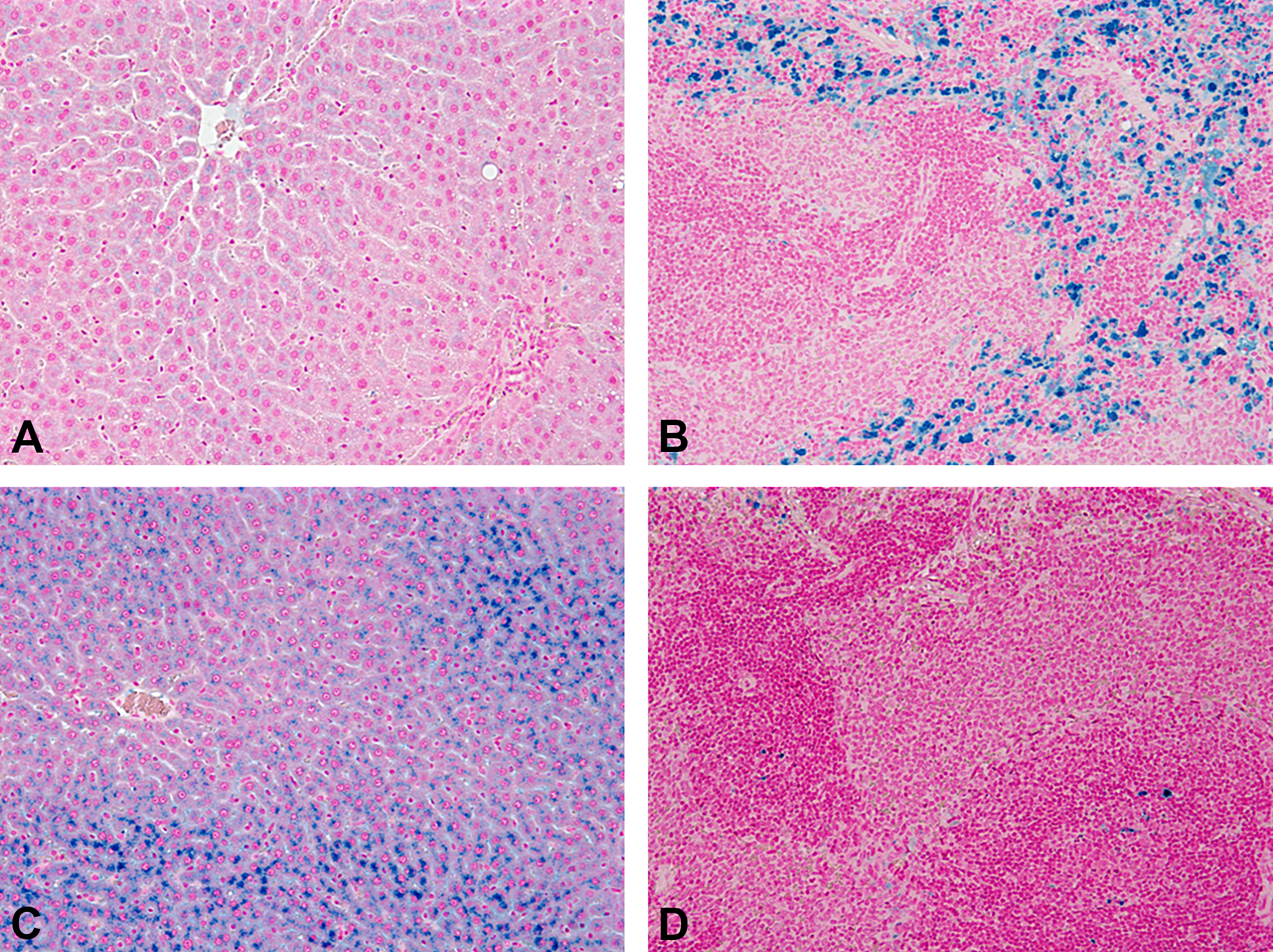
Sprague-Dawley rats treated weekly with a high dose of a hemojuvelin-neutralizing monoclonal antibody for 4 weeks. (A) Liver, control, Prussian blue. (B) Spleen, control, Prussian blue. (C) Liver, high dose, Prussian blue. (D) Spleen, high dose, Prussian blue. Original objective 20× (A and B) and 10× (C and D).
Presenter’s Interpretation
The histopathological findings were not considered adverse. The iron accumulation could be indicative of an imbalance in iron regulation via hemojuvelin—hepcidin—bone morphogenic protein 6—neogenin but is considered nonadverse unless iron overload occurs. Assessment of the drug effects should therefore include the expression of these key factors for iron regulation besides histopathological findings.
Lipofuscin accumulation
Lipofuscin and ceroid are “wear-and-tear” pigments produced through oxidation of fatty acids, often as the result of membrane turnover or damage. Accumulation of these pigments occurs with aging. The presence of lipofuscin is not directly injurious to the cell (Kumar, Abbas, and Aster 2015), but it can be an indication of prior sublethal cell injury. Lipofuscin accumulation is a common finding in the liver. Damaged organelles are removed via autophagosomes, and undigested material is retained as lipofuscin (Haschek 2010). Without obvious cell injury, it is often not possible to determine whether lipofuscin accumulation is the consequence of an increased lipid/membrane turnover or an extension of the life span of the cell. Therefore, whether lipofuscin accumulation should be considered adverse as an isolated finding has to be carefully weighed in the context of corroborating evidence. In some cases, it may be the first indicator that there is a compound-related enhancement of oxidative stress (Cattley, Popp, and Vonderfecht 2013).
A survey of prior chemical reviews by the U.S. Environmental Protection Agency (EPA) identified chemicals in which lipofuscin or ceroid was cited in the lowest observed adverse effect level (LOAEL) of a study report. The 3 primary sites were liver, kidney, and thyroid. In all cases, there were associated findings, typically indicative of direct cell injury. There was only 1 case in which lipofuscin was cited by itself as the basis for an LOAEL (a subchronic rat study), but in this specific case, there were changes noted at the same site in the corresponding subchronic mouse study, which may have influenced interpretation. In general, the working group did not consider accumulation of lipofuscin to be adverse when observed in the absence of other morphological or functional effects.
Example 9: Lipofuscin accumulation in the liver of rats
Case Introduction
A small molecule drug candidate for treatment of a CNS disorder was given to rats by daily oral gavage administration for 4 or 13 weeks. The compound had cationic amphiphilic properties and induced PLD in several organs. Its clearance was dominated by metabolism of hepatic cytochromes.
Findings and Additional Investigations
Clinical pathology revealed increases of group mean plasma ALT, aspartate aminotransferase, and glutamate dehydrogenase activity to a maximum of 4-fold compared to control. Grossly, the liver was enlarged and, after the 13-week treatment, discolored in some animals. The group mean liver weight and liver-to-body weight ratio were about 1.3-fold versus control. Histopathology revealed vacuolation of centrilobular hepatocytes and Kupffer cells after 4 weeks of treatment. Ultrastructurally, the vacuoles contained lamellar bodies, indicating PLD. The PLD was less prominent and in some animals absent after the 13-week dosing. Instead, deposits of brown pigment were present in the centrilobular region. The pigment was fine granular in hepatocytes and more compact in Kupffer cells. It was characterized as lipofuscin based on positive staining for periodic acid–Schiff and Schmorl’s stain and negative staining for Prussian blue (Figure 11A). The pigment was identified by transmission electron microscopy as tertiary lysosomes, which were loosely scattered throughout the hepatocyte cytoplasm and densely packed in Kupffer cells (Figure 11B and C). Additional histological findings in the liver were a minimal to mild increase of sinusoidal leukocytes, Kupffer cell activation, and single cell necrosis of hepatocytes.
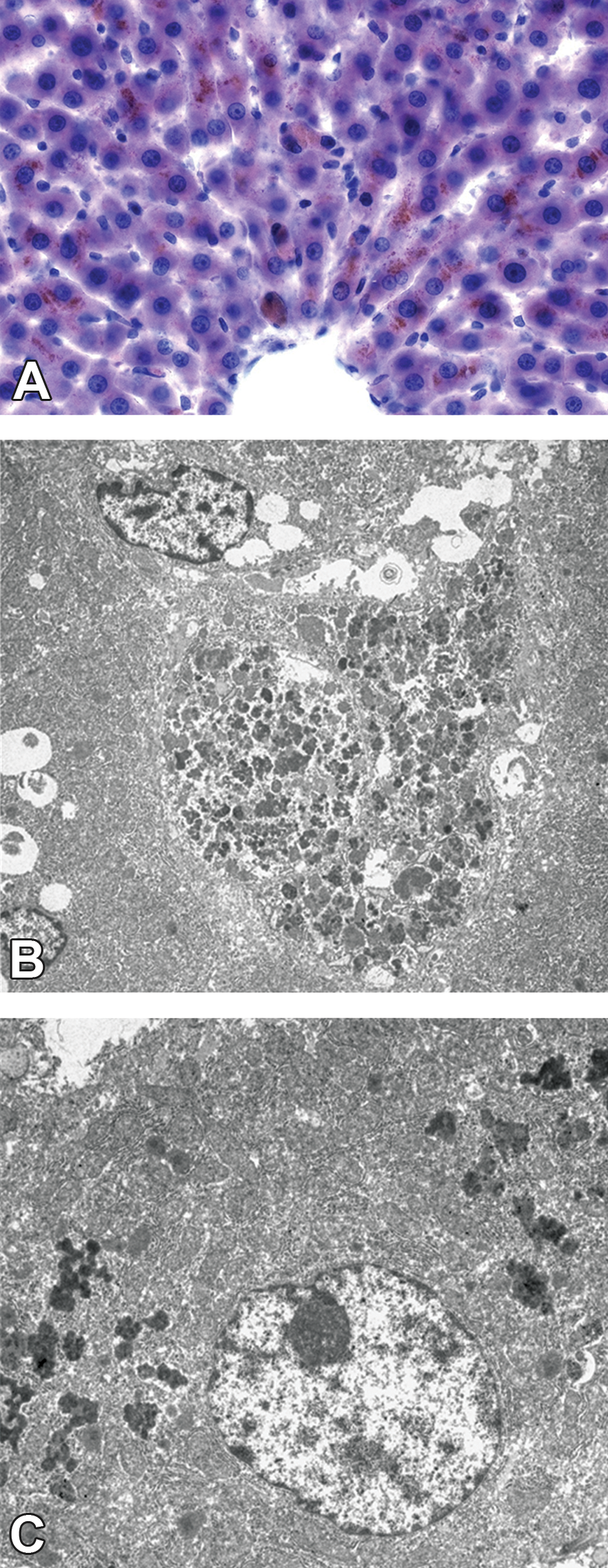
Wistar rats treated with a high dose of a small molecule for 13 weeks. (A) Frozen liver section stained with Fat Red 7 (Fat Red 7 was used, as it was easier to identify lipofuscin in frozen sections stained with Fat Red 7 than in hematoxylin and eosin– stained paraffin sections or paraffin sections stained with the periodic acid–Schiff or Schmorl’s procedure). Centrilobular region shows fine granular brown lipofuscin deposits in hepatocytes and dense accumulation in enlarged Kupffer cells. Original objective 20×. (B) Transmission electron microscopy. Hepatocyte showing tertiary lysosomes scattered throughout the cytoplasm. (C) Transmission electron microscopy. The cytoplasm of an enlarged Kupffer cell is densely packed with tertiary lysosomes.
Investigations into hepatic enzyme induction revealed an increase in centrilobular hepatocellular hypertrophy on histology and cytochrome messenger RNA but reduced enzyme activity. This latter finding was interpreted as enzyme autoinhibition. A genome-wide gene expression analysis was performed in mice, which indicated an increased expression of genes related to the proteasome as the most pertinent finding.
Presenter’s Interpretation
The autoinhibition of the drug-metabolizing cytochromes and the increased expression of proteasome-related genes were interpreted as evidence for the generation of reactive metabolites, which altered proteins. The presence of lipofuscin was interpreted as the sequela of (1) increased turnover of cellular proteins and (2) structural alteration of phospholipids, probably by oxidation. The lipofuscin accumulation was interpreted as adverse for both hepatocytes and Kupffer cells. In the case of hepatocytes, the lesion context was the most decisive criterion (increased plasma activity of hepatocellular marker enzymes, secondary to formation of reactive metabolites). Regarding the Kupffer cells, the large amount of lipofuscin was considered to potentially have impaired cellular functions by overload phenomena.
Alpha-2u globulin accumulation
A2u is a type of major urinary protein synthesized predominantly in the liver of male rats. It has a low molecular weight (<20 kDa) and is freely filtered by the glomerulus. A fraction of a2u is reabsorbed by renal proximal tubular epithelial cells. This protein appears on H&E staining as intracytoplasmic eosinophilic hyaline droplets, which are a normal background feature in the kidneys of sexually mature male rats (Frazier et al. 2012). Ultrastructurally, these droplets appear as large dense secondary lysosomes, sometimes with polyangular or irregular crystalloid shapes (Hard et al. 1993).
The amount of a2u in the proximal tubular epithelium depends on the relative rates of reabsorption and subsequent hydrolysis in secondary lysosomes. A2u in the rat has a distinctive pocket that binds certain types of small organic molecules with high affinity, causing resistance to lysosomal digestion and increased half-life of the a2u-chemical complex in proximal tubular epithelial cells. Over time, higher levels of a2u accumulation can lead to a distinct type of nephropathy characterized by proximal tubular cell death (nephrosis), granular casts in the outer medulla, linear mineralization in the papilla, and/or exacerbation of chronic progressive nephropathy. Cytotoxicity associated with a2u nephropathy can also lead to increased compensatory renal tubular cell proliferation and higher incidence of renal epithelial cell tumors with age (EPA 1991).
Cases shared at the workshop highlighted several concepts related to the interpretation of a2u accumulation. When hyaline droplets (confirmed as a2u by IHC) were accompanied by degenerative changes such as basophilic tubules, granular casts, intratubular cellular debris, and/or nephrosis (e.g., Cesta et al. 2013; National Toxicology Program 2010), this group of effects was considered adverse in male rats. In contrast, increases in a2u droplets without evidence of tubular degeneration on histopathology or urinalysis were generally not considered adverse, given that a2u reabsorption is a normal part of proximal tubular cell function. A more challenging question is whether non-a2u accumulations (e.g., hyaline droplets) in the tubular epithelium of female rats should be considered adverse. This latter scenario would be interpreted on a case-by-case basis integrating other renal effects, distribution and severity of accumulation, reversibility, and other knowledge about the compound being studied (Palazzi et al. 2016).
Findings in the kidney associated with a2u were considered specific to the male rat and not relevant to human health, as described previously by the U.S. EPA (1991) and others. However, the pathologist should still characterize a2u effects in the study report to allow reviewers to distinguish a2u-related and non-a2u-related adversity levels. Both the STP and ESTP working groups on adversity (Kerlin et al. 2016; Palazzi et al. 2016) recommended that adversity should be applied to the model species at the study level, and human relevance should be assessed after the primary adversity call in the nonclinical studies. If it is expected that a2u-associated changes will be dismissed in the integrative assessment for human, then it is still important to know the dose levels for other potential (non-a2u-related) adverse renal effects.
A related issue involves confirmation of a2u accumulation, which is recommended using an a2u-specific marker, typically IHC (EPA 1991). Stains such as Mallory–Heidenhain or chromotrope aniline blue (CAB) may provide supportive data but are not strictly diagnostic for a2u. Given that a2u droplets are normally present in the male rat kidney, incidence alone may not be definitive, especially if a sensitive staining technique is used that shows a2u in a high proportion of concurrent male controls. In addition, the appearance of droplets may vary based on H&E protocol, influencing background rates across studies. Thus, including some type of quantitative or semiquantitative morphometric analysis of a2u (such as percentage area or intensity grade) may assist reviewers in discerning whether an actual treatment effect is present. Moreover, use of a true negative staining control such as female rat kidney is recommended to optimize staining protocols. Finally, it was noted during the workshop that current commercially available a2u antibodies for IHC may exhibit variable levels of sensitivity and specificity and should be validated prior to use. In the future, a consortium may be warranted to develop and standardize a2u antibodies.
Example 10: A2u nephropathy induced by a catalyst compound
Case Introduction
Male and female Wistar rats [Crl: WI(Han)] were given a catalyst at 0 (control), low dose, mid dose, and high doses in a repeated dosing 28-day oral feeding study (according to Organization for Economic Cooperation and Development Guideline 407). The compound is an intermediate substance used during different catalytic processes. The study was performed for Registration, Evaluation, Authorisation and Restriction of Chemicals registration in an Association for Assessment and Accreditation of Laboratory Animal Care International–accredited laboratory.
Findings and Additional Investigations
Eosinophilic hyaline droplet accumulation in the proximal tubules of the kidney was observed in all treated male but not female rats starting at the low dose, with dose-dependent increases in incidence and severity (Figure 12A–C). Droplets were confirmed by IHC staining to be a2u (Figure 12D). Associated histopathological changes, including basophilic tubules, intratubular granular casts, and intratubular cellular debris, were observed in the mid-dose and high-dose groups. Grading of the hyaline droplets was performed on Mallory–Heidenhain–stained kidney sections.
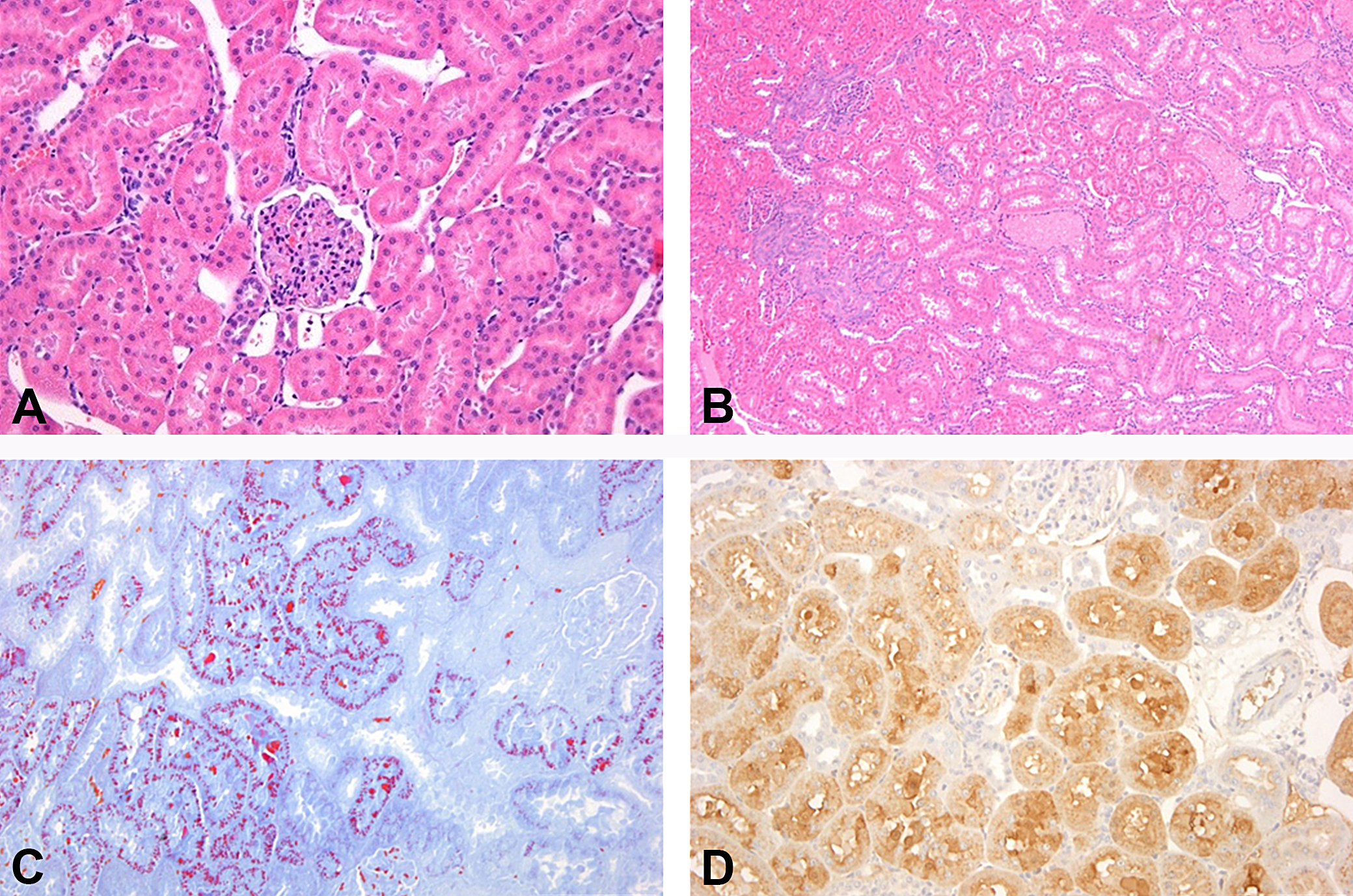
Wistar rats treated with a catalyst in a repeated dosing 28-day oral feeding study. (A) Control, male rat, no findings, hematoxylin and eosin (H&E) stain and original objective 10×. (B) Male rat treated with a high dose. Hyaline droplet nephropathy with basophilic tubules, granular casts, and intratubular cell debris. H&E stain and original objective 4×. (C) Male rat treated with a high dose. Increased eosinophilic hyaline droplets. Mallory–Heidenhain stain and original objective 10×. (D) Male rat treated with a high dose. Positively stained droplets and tubules in alpha-2u (a2u) immunohistochemistry. A2u-specific antibody and original objective 10×.
Presenter’s Interpretation
A2u hyaline droplets should always be confirmed by IHC. Additional stains such as CAB may be useful in grading of hyaline droplet severity. Renal effects associated with a2u should be clearly characterized in the study report to avoid masking of non-a2u-related effects. Here, tubular cytotoxicity associated with a2u accumulation at the mid dose would be the basis for the LOAEL. If these effects are subsequently considered not relevant to human health, other potential non-a2u-related effects may be important in determining adverse dose levels.
Discussion
Lysosomes have a central role in intracellular digestion, trafficking, and recycling of macromolecules. Disruption of these processes can occur through a diverse range of biological pathways and result in accumulation of both exogenous and endogenous materials. When interpreting xenobiotic-induced lysosomal effects, discriminating a physiologic process (simple uptake or storage) from a pathologic 1 (toxicity and impaired function) can be challenging, especially when the histopathologic diagnosis is relatively simple or nondescript. In many cases, the adversity decision will be based on contextual information, mechanistic understanding of target pathways, and functional consequences of excessive accumulation. Ancillary data may include related histopathologic effects, clinical pathology, IHC or other in situ biomarkers, and biochemical and cell culture assays to assess lysosomal function and dysfunction. Additional in vivo functional assessments (e.g., assessment of macrophage function in host defense assays, as shown for ASOs) may be informative in supporting adversity decisions. Without this context, decision-making will tend to be more conservative and potentially inaccurate.
Workshop discussions focused on general guiding principles for determining adversity and key concepts specific to lysosomal accumulation. Using the primary determinants of adversity (inherency, context, and severity) as a guide (Palazzi et al. 2016), the following factors were proposed (see also Table 4):
Factors to Be Taken into Account for Adversity Assessment.

location and affected cell population, origin of lysosomal content (e.g., endogenous vs. exogenous substance), biological consequences including cell injury and loss of function, and severity of the accumulation.
Other considerations included functional reserve and capacity for renewal of target cells/organs, reversibility, distribution, and ability to monitor function. The following list provides general recommendations, not as formulaic guidelines, but rather as points to consider when reaching or communicating a lysosomal-related adversity decision: Lysosomal accumulation in tissues without regenerative capacity (e.g., neurons or retinal cells) would generally be considered adverse, as these cellular changes may potentially affect the function of a cell that cannot be renewed (or directly assessed). In tissues with a large regenerative capacity such as liver, adversity may depend on associated changes, mechanistic information, and other factors such as reversibility, markers of injury or function, and physicochemical properties of lysosomal contents. Associated effects, such as inflammation, fibrosis, and necrosis, are common hallmarks of adversity but should still be considered as part of a weight-of-evidence evaluation. In many cases, adversity of lysosomal accumulation may depend on severity. Changes manifesting as a low severity morphological change (e.g., “minimal vacuolation” or “slight cytoplasmic granules”) may not be adverse in absence of other findings, while marked PLD or other accumulations may decrease the lysosomal reserve capacity, leading to cellular dysfunction and eventually cell death. Similarly, an extensive or unusual distribution of PLD involving multiple cell types, including parenchymal cells, would support adversity concerns. Accumulation of endogenous materials is more likely to result from physiologic processes and thus less likely to be considered adverse compared to a foreign material. However, distinction between “natural” and “non-natural” accumulations is not necessarily an informative criterion for adversity. Reversibility of an accumulation does not automatically indicate nonadversity. However, the lack of reversibility could indicate additional cause for concern and consequently increase the likelihood that a change will be considered adverse. For example, the lack of reversibility for PEG-induced vacuolation of neurons and CP epithelial cells increased uncertainty and influenced the adversity decision. Endogenous accumulations such as lipofuscin or iron/hemosiderin often indicate prior or associated cell injury that needs to be assessed for adversity. Thus, interpretation of these changes in isolation may not provide adequate context; instead, adversity should be discussed in context of other findings (e.g., oxidative injury, hemolysis). Many cases of lysosomal accumulation may be considered physiologic or adaptive under some scenarios but not others (e.g., uptake of material by alveolar macrophages, storage of glycogen in hepatocytes, and a2u reabsorption in proximal tubular epithelial cells). The pathogenesis of the accumulation and its mechanism may be important in determining threshold effects, functional consequences of excessive accumulation, and human relevance of these changes.
The various examples discussed at the workshop illustrate that lysosomal accumulation encompasses a diverse array of substances, mechanisms, cell types, and functional consequences. In addition, it was highlighted in the discussions that although adversity should initially be considered within the context of the animal model (i.e., at the study level), the intended use (e.g., pharmaceutical vs. agrochemical or industrial chemical) and assessment method may also influence the adversity decision. In pharmaceutical development, where exposed individuals will presumably derive a health benefit from the exposure (risk–benefit assessment), there may be a higher acceptance of certain findings compared to environmental chemicals where there is only a risk assessment for exposed individuals and no subsequent human trial data. Lysosomal accumulations might even be accepted without safety margin for certain indications in pharmaceutical development. For example, it was noted that there are several marketed drugs such as amiodarone that have potential for PLD development at pharmacological doses. In addition, the lack of human monitoring data for the vast majority of environmental chemicals may contribute to different levels of conservatism during review.
In summary, the expert group concluded that lysosomal accumulation should be assessed on a case-by-case basis using a weight-of-evidence approach. The examples shared during the workshop illustrate the diverse types of data that may inform these decisions and confirm the importance of thorough characterization beyond a simple histopathologic diagnosis, including mechanistic evidence and functional readouts, when available. This information will facilitate greater consistency and transparency when evaluating lysosomal accumulation and its human relevance and reduce default approaches driven by uncertainty.
Footnotes
Authors’ Note
The opinions expressed in this document are those of the authors and do not reflect views or policies of the employing institutions including the U.S. FDA. This article has been reviewed by the U.S. EPA and approved for publication. Approval does not signify that the contents necessarily reflect the views or the policies of the agency. Mention of trade names or commercial products does not constitute endorsement or recommendation for use.
Author Contribution
Authors contributed to conception or design (BL, AB JE, PF, HF, SF, RF, SG, TH, HH, WF, SK, TN, XP, GP, AP, AR, AS, LS, LT, JW, CW, MY); data acquisition, analysis, or interpretation (BL, AB JE, PF, HF, SF, RF, SG, TH, HH, WF, SK, TN, XP, GP, AP, AR, AS, LS, LT, JW, CW, MY); drafting the manuscript (BL); and critically revising the manuscript (BL, AB JE, PF, HF, SF, RF, SG, TH, HH, WF, SK, TN, XP, GP, AP, AR, AS, LS, LT, JW, CW, MY). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
