Abstract
NOD/Shi-scid/IL-2Rγnull (NOG) mice are humanized with CD34+ hematopoietic cells (huNOG mice) and are commonly utilized for biological or medical research on human therapeutics. In the present study, nine 26-week-old, female huNOG mice were utilized for testing proprietary immune–modulating drugs over a 3-week period at the University of Washington. Two of the 9 mice developed unilateral swelling of a tibiotarsal joint with associated paresis of the affected limb. Full necropsies were performed after euthanasia at experimental end point, and routine tissues and affected tibiotarsal joints were evaluated. An expansile, multilobular mass composed primarily of chondroid cells associated with the calcaneal tendon was present within 1 tibiotarsal joint of both mice. A small focus of well-differentiated bone was present within the mass of 1 mouse. In addition, the calcaneal periosteum was expanded by a chondroid mass in 1 mouse. The cartilaginous masses associated with the calcaneal tendon were interpreted as a hyperplastic or low-grade neoplastic process accompanied by endochondral ossification, and the mass associated with the calcaneal periosteum was interpreted as chondroid metaplasia. Although the etiology of these lesions is unclear, their prevalence in 2/9 (22%) huNOG mice is interesting and may have biological significance for future studies involving the huNOG mouse model.
Keywords
The NOD/Shi-scid/IL-2Rγnull (NOG) mouse is a newer generation of severely immunodeficient mice that is commonly utilized in animal models of human disease (M. Ito et al. 2002). Their lack of lymphocytes and natural killer (NK) cells as well as impaired macrophage function render these mice particularly efficient recipients of heterologous cells, making the NOG mouse an ideal model for engraftment studies (R. Ito et al. 2012). HuNOG mice are humanized with CD34+ hematopoietic cells, with the goal of mimicking a human immune system for biological and medical research on human therapeutics, making huNOG mice ideal candidates for research involving infectious disease, cancer, allergies, and regenerative medicine (Walsh et al. 2016; Honeycutt et al. 2015). In the present study, 2 of the 9 (22%) huNOG mice utilized in a proprietary immune–modulating drug study developed unilateral tibiotarsal swelling and hind limb paresis. The purpose of this evaluation was to characterize the lesions that were present in the tibiotarsal joints of these mice which appear to be previously unreported. Because the huNOG mouse is a relatively new animal model of human disease that has the potential for widespread use, it is important to report lesions that may be a spontaneous background lesion.
Material and Method
Nine 26-week-old, female huNOG mice (Taconic Biosciences, Hudson, New York) were obtained by the University of Washington for a proprietary immune–modulating drug study. Animals were group housed in an Association for Assessment and Accreditation of Laboratory Animal Care International accredited facility under approved Animal Care and Use Committee guidelines. The study was conducted in compliance with the Animal Welfare Act, the Guide for the Care and Use of Laboratory Animals, and the Office of Laboratory Animal Welfare. Animals were fed irradiated Picolab Rodent Diet 20 #5053 (Lab Diet, St. Louis, MO).
The proprietary drugs were administered by intraperitoneal injection 5 times per week, over the course of 3 weeks. During the second week of the study, 2 mice housed in separate cages were noted to have developed unilateral swelling of a tibiotarsal joint (mouse A in left hind, mouse B in right hind) accompanied by paresis of the affected limb that did not progress in size of the swelling or severity of paresis over the remaining week of the study, from the time the lesions were first noted until euthanasia. All mice, including those affected, were euthanized at the experimental end point and full necropsy examinations were performed. Routine tissues, including samples from skull, lung, kidney, heart, liver, mesentery, spleen, ovaries, colon, and spinal cord, in addition to the affected tibiotarsal joints, were processed for routine histopathologic analysis at the University of Washington Veterinary Diagnostic Laboratory. Samples were fixed in 10% neutral buffered formalin, processed, sectioned, and stained (hematoxylin and eosin) for histopathologic evaluation. Tibiotarsal joints were decalcified prior to embedding. Initial tissue evaluations were conducted by the University of Washington Veterinary Diagnostic Laboratory, with further consultation on the affected tibiotarsal joints by a specialist in orthopedic pathology.
Results
Routine tissues did not contain significant gross or microscopic lesions. In mouse A, sections of mid-tibia through tarsus revealed an expansile, multilobular mass composed of a chondroid population of cells that exhibited moderate anisokaryosis, anisocytosis, and numerous binucleate cells with no mitoses observed (Figures 1 –3). The periosteum of the calcaneus was moderately thickened, elevated, and bilaterally, symmetrically expanded by a mass composed of hyaline cartilage, in which the component cells progressed from flattened cells that were contiguous with the periosteum, to spindloid cells, to large, hypertrophic chondrocytes (Figure 3). These cells exhibited moderate anisokaryosis and anisocytosis and included numerous binucleate cells with no mitoses observed. The overlying epidermis exhibited marked acanthosis (Figure 4). In mouse B, similar lesions were present, including an extra-articular mass associated with the calcaneal tendon and a locally extensive chondromatous proliferation that elevated and was contiguous with the periosteum of the calcaneus but involved less tissue and was smaller in size than that in mouse A (Figures 5 –7). The mass associated with the calcaneal tendon in mouse B contained a small focus of well-differentiated bone containing marrow spaces at the dorsoplantar margin (Figure 8). In both mice, minimal associated inflammatory cells were present. Other structures of the tibiotarsal joints contained no significant lesions.
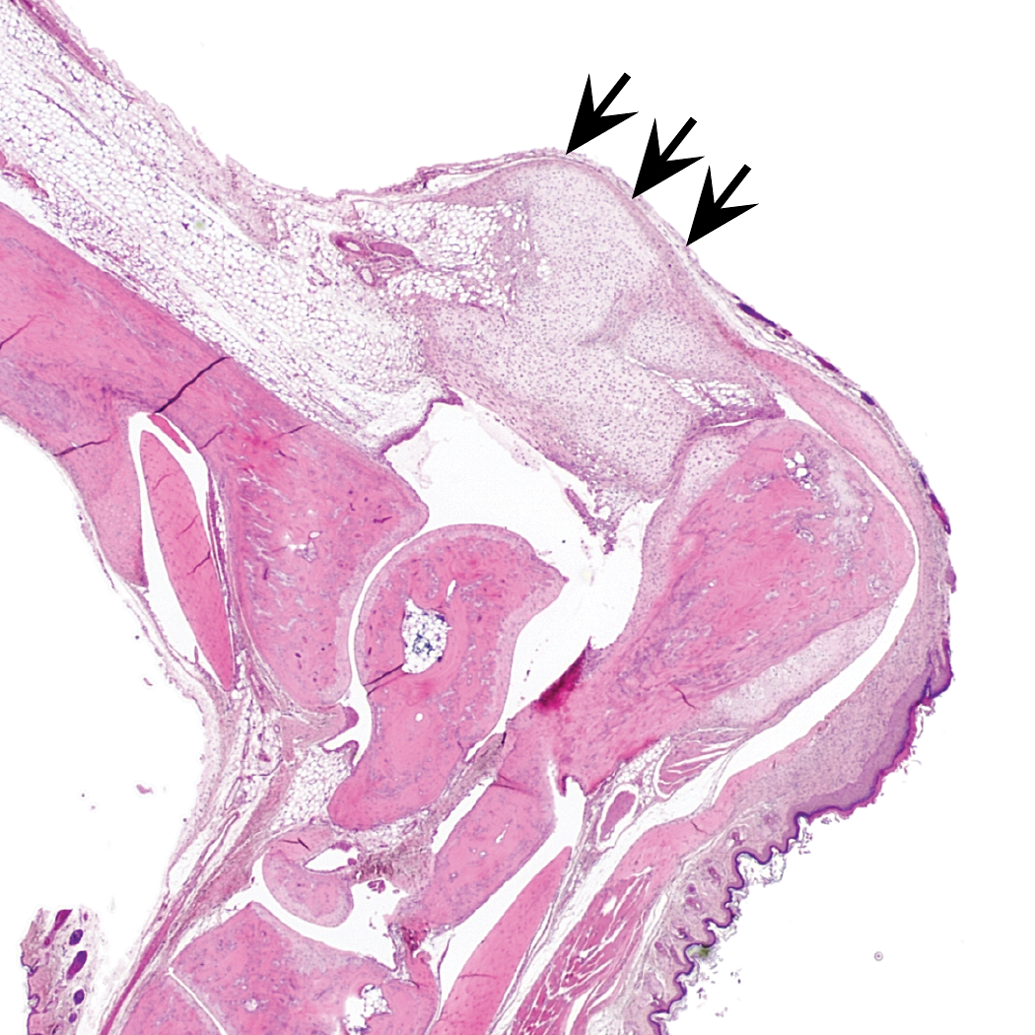
Mouse A: low magnification of chondroid mass (arrows) associated with the calcaneal tendon and periosteal chondroid metaplasia of the calcaneus.
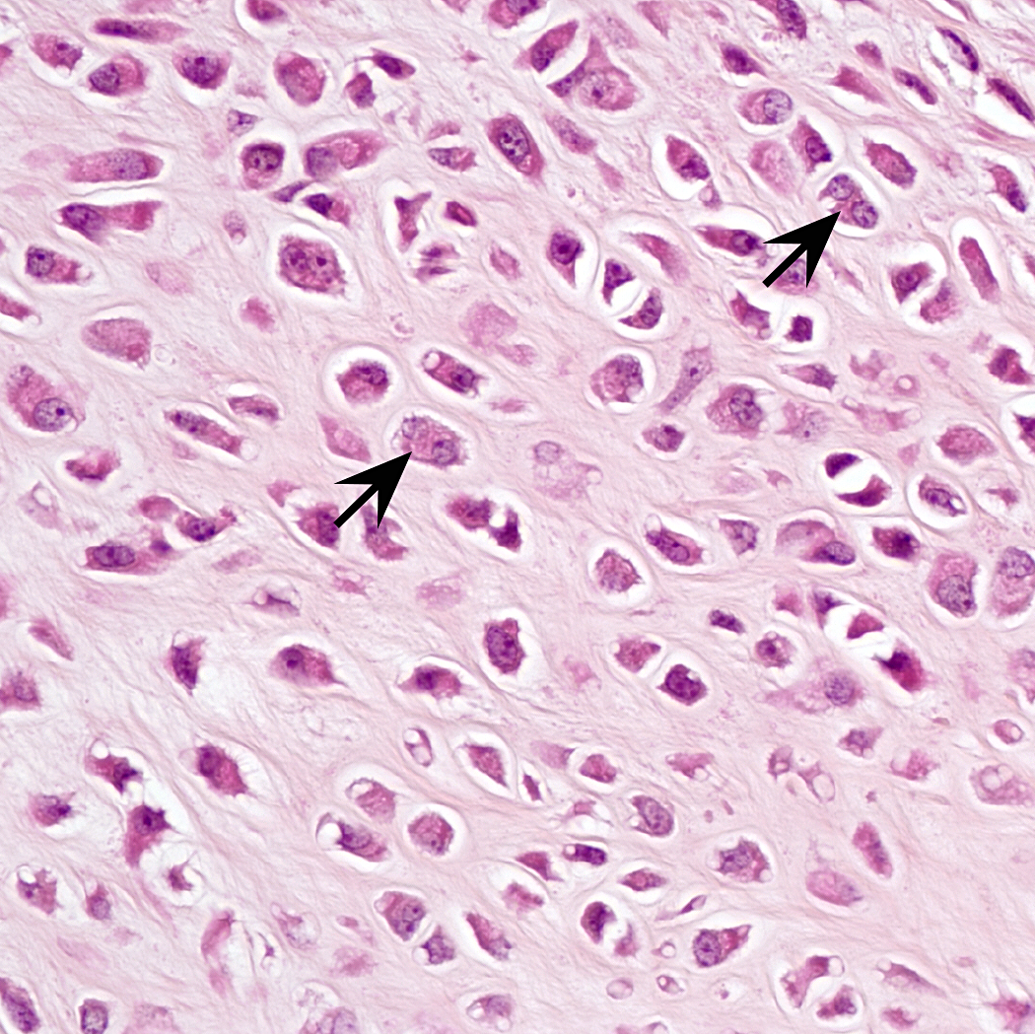
Mouse A: high magnification of chondroid mass, demonstrating binucleate cells (arrows), anisocytosis, and anisokaryosis.
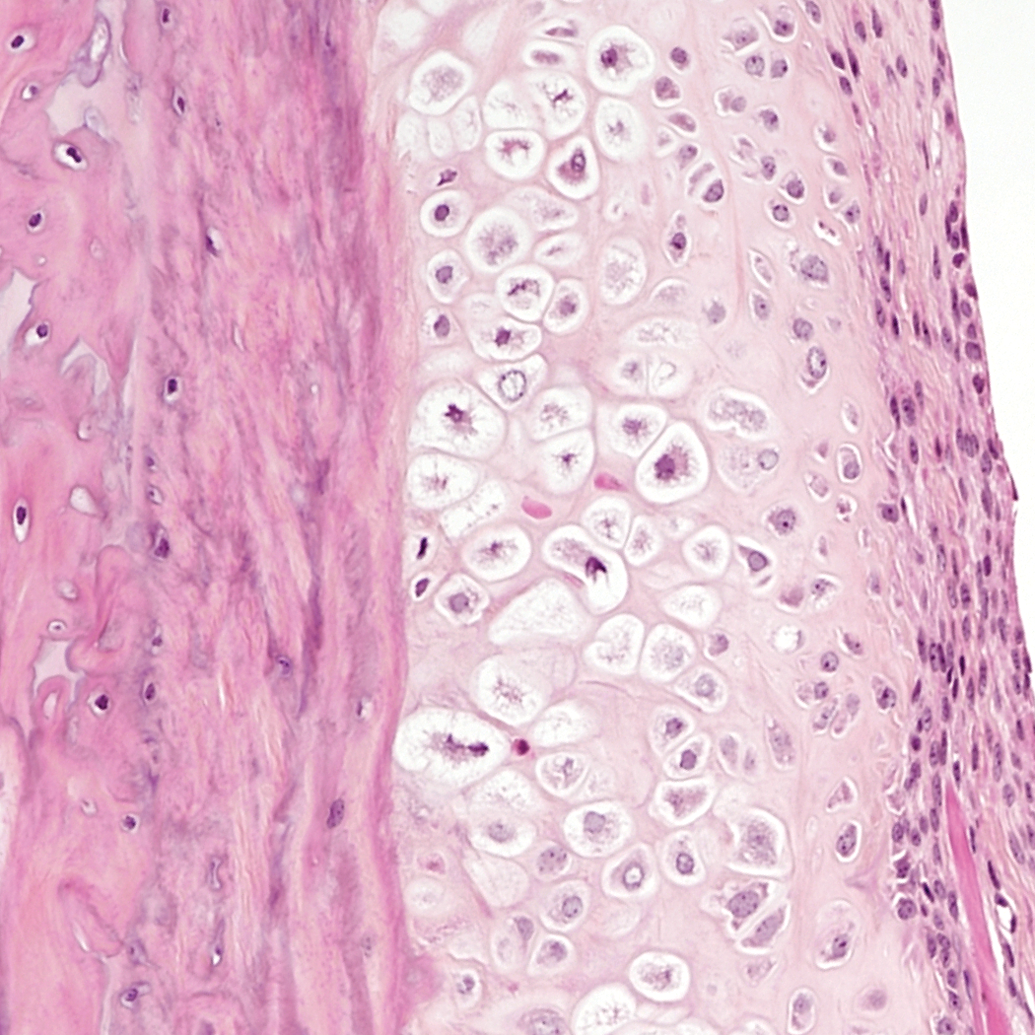
Mouse A: high magnification of chondroid metaplasia of the periosteum.
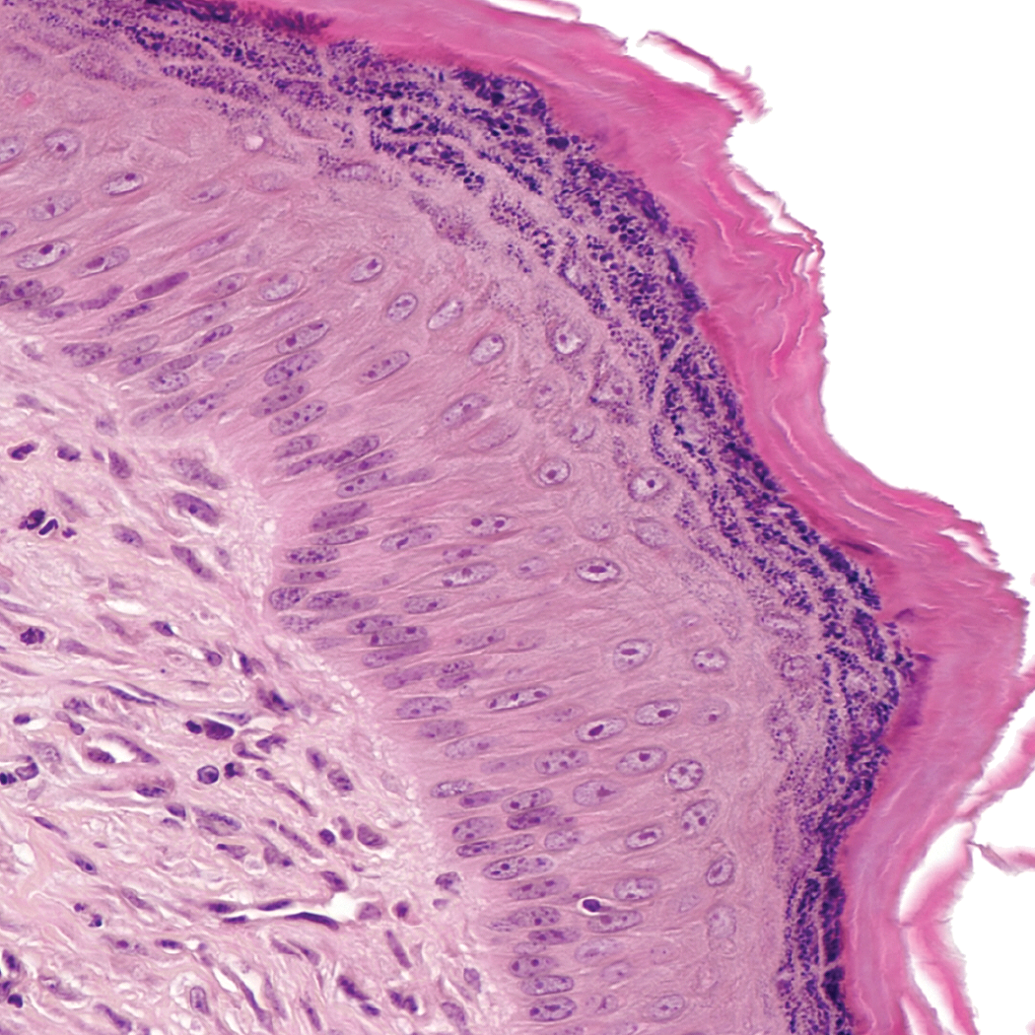
Mouse A: markedly acanthotic epidermis overlying mass.
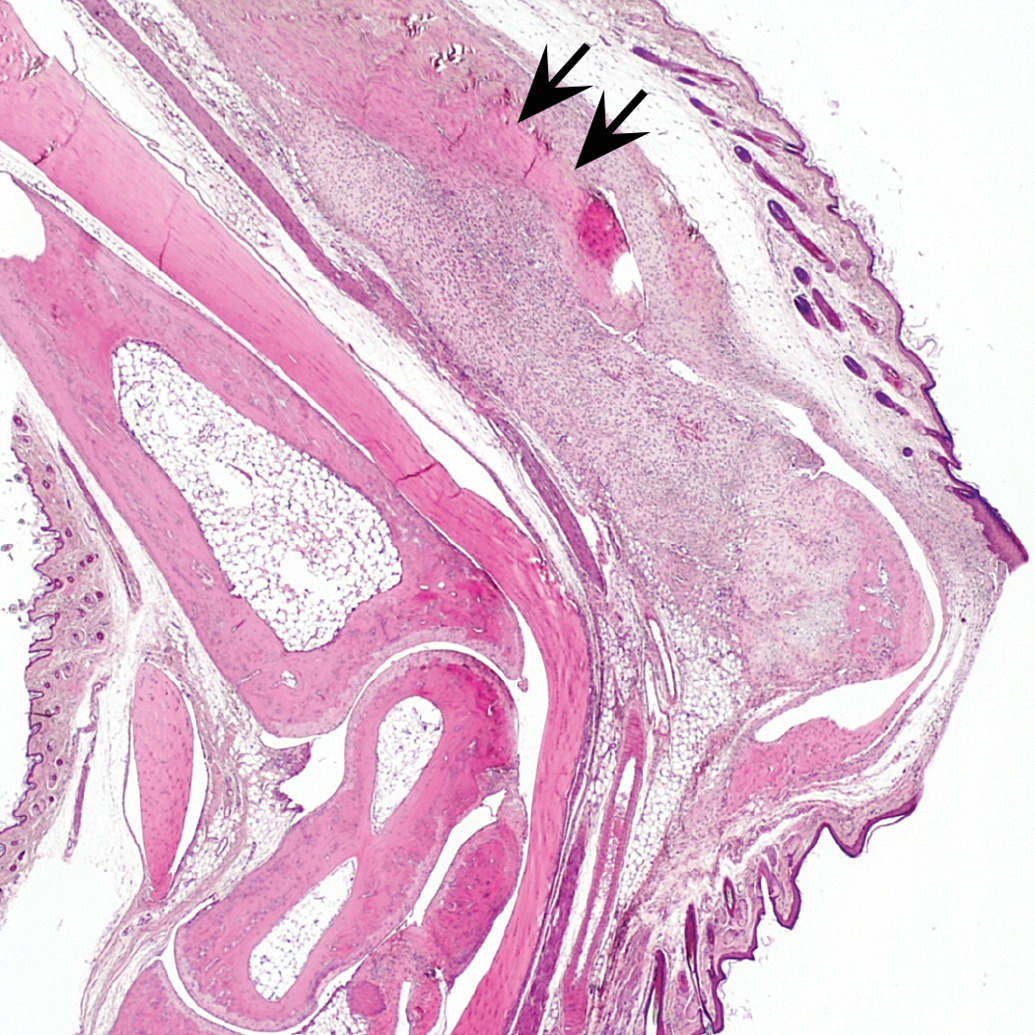
Mouse B: low magnification of extra-articular mass associated with calcaneal tendon (arrows).
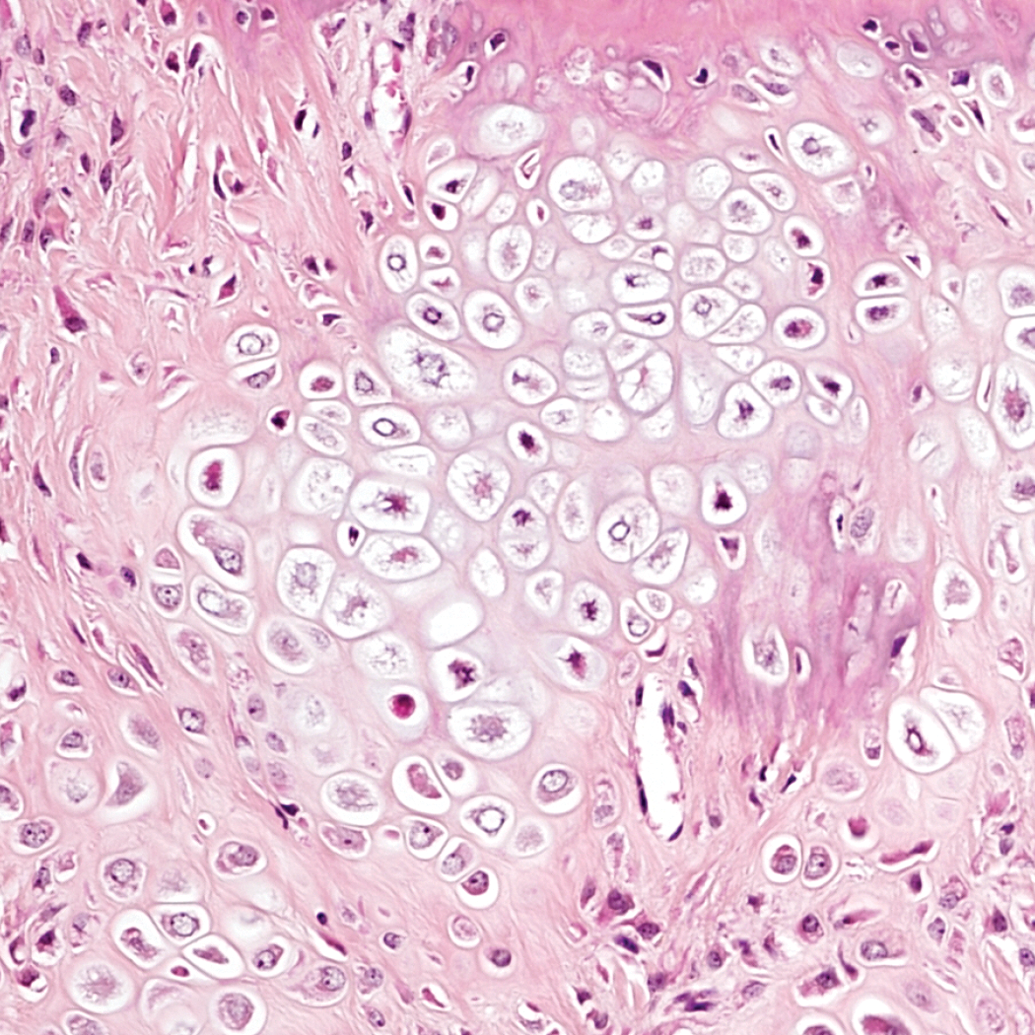
Mouse B: high magnification of chondroid mass, demonstrating chondroid cells that progress from flattened, to spindloid, to large, hypertrophic chondrocytes.
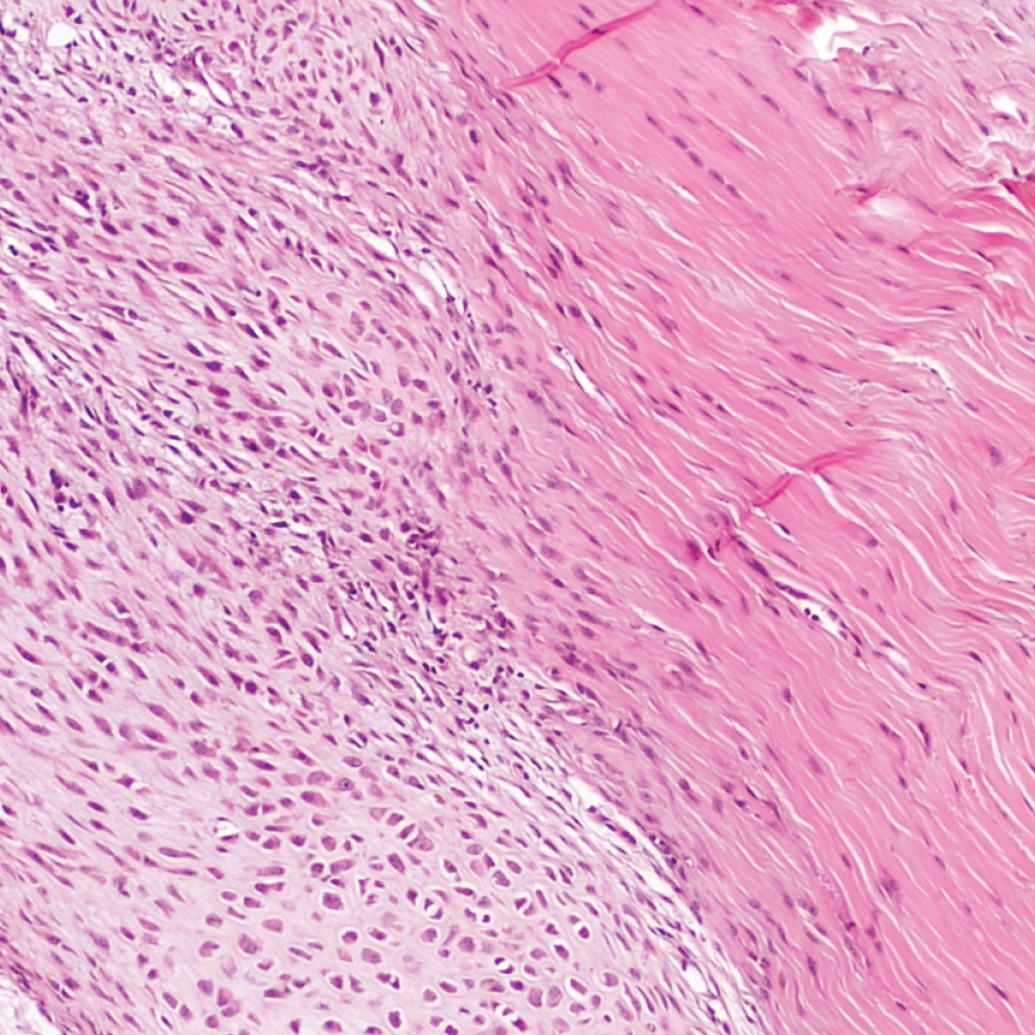
Mouse B: transition of calcaneal tendon to chondromatous mass.
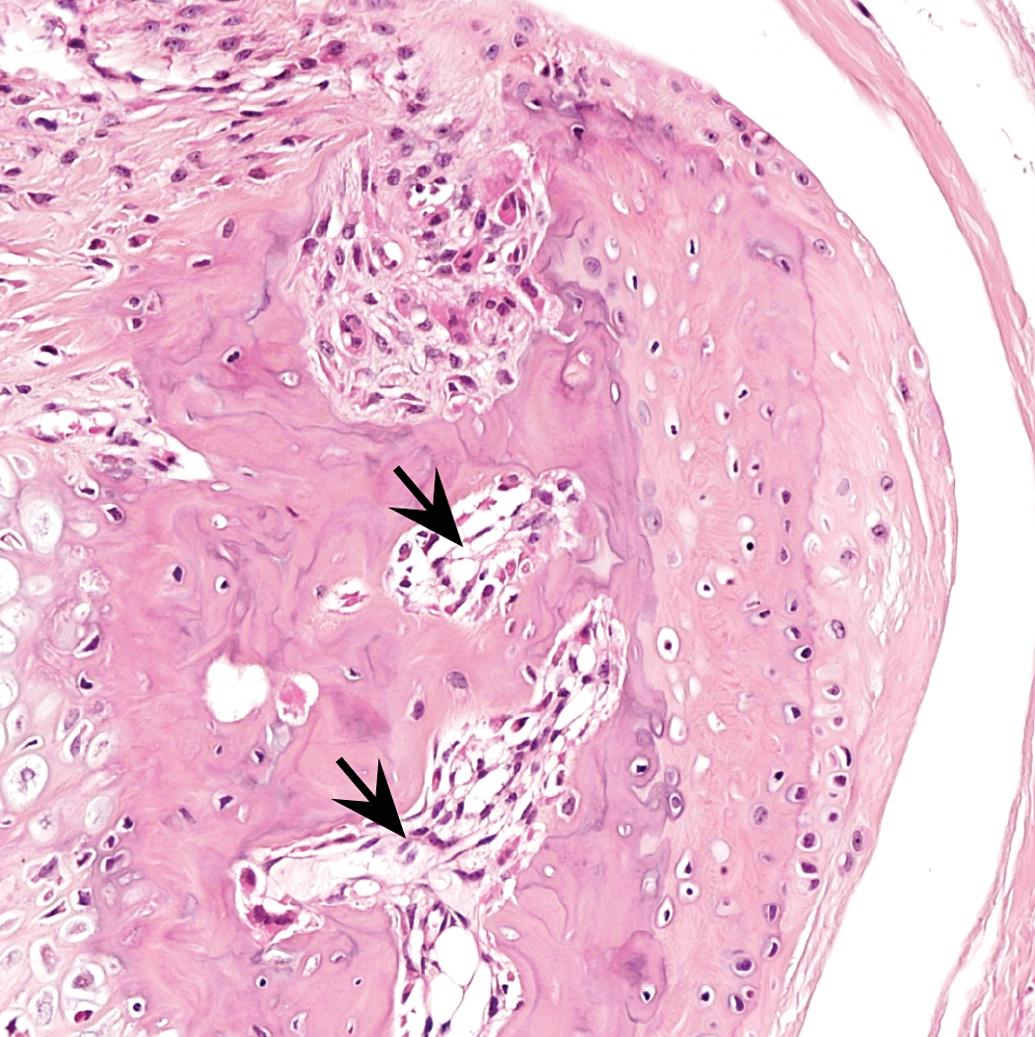
Mouse B: focus of well-differentiated bone with marrow spaces (arrows).
Discussion
In both mice, the expansile, extra-articular, chondromatous masses were interpreted as representing either a hyperplastic or a low-grade neoplastic process arising from the calcaneal tendon. Unfortunately, other joints, including the contralateral tibiotarsal joints, were not collected nor examined, and imaging was not available prior to necropsy to determine whether any additional, subclinical masses were present. The masses were fairly similar in both mice and appeared to arise from and replace areas of the calcaneal tendon. The tissue changes, however, were not reminiscent of chondrification of the tendon, which may occur as a degenerative change (Kavirayani, Sundberg, and Foreman 2012). Instead, the fibrous tissue of the tendon was replaced by cartilaginous tissue that appeared to be very well demarcated.
In mouse B, the mass appeared to be undergoing endochondral ossification with the production of a bony cap. The locally extensive chondromatous proliferation that was contiguous with the periosteum of the calcaneus was interpreted as a chondroid metaplasia of the periosteum due to its location and degree of differentiation. The marked epidermal acanthosis of overlying skin likely occurred secondary to trauma/irritation associated with the expansile nature of the underlying mass.
A disorder called heterotopic ossification (HO) is described in the human literature as the abnormal deposition of bone at extraskeletal soft tissue sites, most commonly secondary to acute tissue damage (Chakkalakal et al. 2012; Liu et al. 2013). However, HO may also occur secondary to genetic factors including fibrodysplasia ossificans progressiva (FOP), which is a rare manifestation of HO in humans in which connective tissues are gradually replaced by bone either spontaneously or after trauma due to a rare genetic mutation in Activin A receptor, Type 1 (ACVR1; Chakkalakal et al. 2012). Patients with FOP are, on average, severely debilitated by their third decade with ossification of soft tissues initially axially but that spreads throughout the appendicular regions of the body. Eventually, an affected individual will lose mobility of the joints, the rib cage, and jaw, causing issues with movement, breathing, and eating (Eisler, Tibesar, and Sidman 2011; Shore and Kaplan 2008).
Despite the presence of endochondral ossification within the chondromatous mass of mouse B, several differences exist between the lesions of the present study and HO and/or FOP. For example, there was no known history of trauma, and all other tibiotarsal joint structures and routine tissues showed no histopathological abnormalities. Secondly, HO and FOP are commonly associated with a robust inflammatory response, which was absent in the present cases (Chakkalakal et al. 2012; Medici and Olsen 2012). Furthermore, HO is primarily extracapsular and does not involve the periosteum, yet the periosteum of mouse A was markedly affected by chondromatous proliferation (Mavrogenis, Soucacos, and Papagelopoulos 2011). These factors, in combination with the absence of bony development in mouse A, render HO an unlikely diagnosis of the lesions observed in these mice. However, it is possible that, although these mice are aged 31 weeks at the time of development of these lesions, these lesions may represent the early stages of HO in both mice that may have progressed to further ossification of soft tissues.
Without further investigation of the opposite tibiotarsal joint, unaffected joints, or unaffected mice, it is difficult to ascertain the cause of these lesions at this time. Future studies are required to further investigate their cause, including whole-body radiographic studies to screen all tissues for bony lesions, in addition to histopathological analysis of grossly unaffected tibiotarsal joints. Tracking the progression of these lesions over time may also lend insights into their potential association with HO or FOP described in the human literature.
Conclusions
Although the etiology of these lesions is currently unclear, their presence in 2 of 9 (22%) huNOG mice used in this study is interesting and unusual and should be kept in mind in future studies using these animals. Future work remains to determine whether the pathogenesis of these lesions is related to genetic factors in these humanized mice and may have biological significance for future studies involving the huNOG mouse model.
Footnotes
Authors’ Note
Tissues were acquired from, processed, and initially evaluated by the University of Washington Veterinary Diagnostic Laboratory. Sery Johnson of the Carlson Laboratory aided in imaging. Timothy O’Brien of the University of Minnesota College of Veterinary Medicine and John P. Sundberg of The Jackson Laboratory aided in interpretation of lesions.
Author Contribution
Authors contributed to conception or design (AM, PT, CC); data acquisition, analysis, or interpretation (AM, PT, CC); drafting the manuscript (AM); and critically revising the manuscript (PT, CC). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
