Abstract
As a result of biotribocorrosion, the surface of a titanium (Ti) biomedical device can be a potential source of systemic contamination with Ti nanoparticles (NPs). Although NPs can be chemically similar, differences in particle size may lead to different biological responses. The aim of this experimental study was to determine Ti trace levels in skin appendages and plasma and explore the influence of NP size on trace levels using a murine model. Results showed the presence of Ti traces in the nails, hair, and plasma. The concentration of the smallest NPs (5 Nm) was higher than that of 10 Nm NPs in all the studied samples. Irrespective of NP size, Ti levels were always lower in plasma than in skin appendages. Ti levels were higher in nails than in hair. Ti NPs size influenced trace concentration levels in hair/nails, suggesting that 5 Nm Ti particles are more easily eliminated through these skin appendages. Given that the nails showed the highest levels of Ti, and that these skin appendages are not exposed to agents that can leach out Ti, as occurs with hair, we propose the nails as the most suitable and reliable bioindicator for monitoring systemic contamination with Ti.
Humans are exposed to different types of particles, which can enter the body mainly through inhalation, ingestion, and/or skin contact (Oberdörster, Oberdörster, and Oberdörster 2005). As a result of the increasing use of biomaterials in medical applications, the surface of metallic biomedical devices can be a potential source of systemic contamination with microparticles (MPs, 0.1–100 µm) and nanoparticles (NPs, 1–100 nm; Jacobs, Gilbert, and Urban 1998; Revell 2006). Titanium (Ti) is widely used in the manufacture of dental and orthopedic implants due to its excellent biocompatibility (Anderson et al. 2004). It is a highly reactive metal, and on exposure to air or fluids, it rapidly develops a passivating layer of titanium dioxide (TiO2). However, as a result of electrochemical corrosion processes, frictional wear (tribology), or a synergistic combination of the two, ions/particles may be released from metal implants (Jacobs, Gilbert, and Urban 1998; Olmedo, Tasat, et al. 2012). The combined effect of electrochemical, mechanical, and biochemical factors is known as tribocorrosion (Mathew et al. 2014).The chemically active metal ions/particles may bind to the surrounding tissues, but may also bind to proteins and be disseminated via the vascular and lymphatic systems to distant organs (Woodman, Black, and Jimenez 1984; Jacobs et al. 1991). A number of researchers in the field of orthopedics have found metal ions/particles in body organs and fluids. It is therefore well-documented that Ti ions/particles pass into the surrounding tissues, reach the plasma, and are excreted in urine (Jacobs et al. 1991, 1998; Urban et al. 2000). Moreover, previous works by our research group showed systemic distribution of the Ti particles transported by blood cells of the phagocytic-mononuclear lineage (Olmedo et al. 2005); the Ti was found to deposit in organs that have macrophagic activity, as is the case of the liver, spleen, and lungs (Olmedo, Guglielmotti, and Cabrini 2002; Olmedo et al. 2005, 2011; Olmedo, Tasat, Evelson, et al. 2008, Bruno et al. 2014), and cause an increase in oxidative stress in lung macrophages (Olmedo et al. 2005, 2011; Olmedo, Tasat, Guglielmotti, et al. 2008). At the oral tissue level, we observed the presence of Ti in peri-implant tissue of failed human dental implants (Olmedo, Fernández, et al. 2003), in oral mucosa in contact with cover screws (Olmedo, Paparella, et al. 2012), in cells exfoliated from oral mucosa in contact with Ti dental implants (Olmedo, Nalli, et al. 2013), and in reactive lesions in peri-implant mucosa (Olmedo et al. 2010).
Because skin appendages (hair and nails) can be considered excretory products (Abdulrahman et al. 2012), the determination of trace elements in skin appendages has been a subject of continuous interest in environmental and biomedical sciences (Arnold and Sachs 1994). Hair and nails are valuable biological materials since they can provide information about the concentration of some trace elements, which may be more concentrated in these skin appendages than in other biological materials. Thus, the hair and nails have remarkable potential as biomarkers (Arnold and Sachs 1994; Benko 1995; Kasai, Lida, and Uchida 2003; Abdulrahman et al. 2012). Moreover, skin appendages can be easily sampled, transported, handled, and stored, which make them suitable material for monitoring trace element concentration (Zhunk and Kist 1995). In this regard, there are studies reporting the presence of traces of copper (Cu), zinc (Zn), chromium (Cr), cobalt (Co), manganese (Mn), iron (Fe), cadmium (Cd), arsenic (As), molybdenum (Mo), nickel (Ni), and lead (Pb) in hair and nails (Abdulrahman et al. 2012; Rodriguez de la Flor, Hernández-Vaquero, and Fernández-Carreira 2013). Particularly in the case of Ti, the presence of metallic traces in skin appendages has been reported in patients with Ti alloy spinal implants (Kasai, Lida, and Uchida 2003) and Ti miniplates used in orthognathic surgery (Bozkus, Germec-Cakan, and Arun 2011).
According to reports in the dermatologic literature, metallic particles can also deposit in the skin, causing metallic discoloration (Peterson et al. 2006; Aasly 2007; Jonas et al. 2007). Metals such as copper (Cu), silver (Ag), and thallium (TI) have been found to deposit in the skin. Metallic discoloration is caused mostly via the blood stream, by exposure to an external source of contamination, or more rarely, by metal traces released into the tissue in contact with a prosthetic implant (metallosis; Park et al. 2013). Specifically regarding Ti, there are only a few reports of Ti pigmentation of the skin caused by metal particles released into the peri-implant tissues (Seong et al. 1996; Akimoto, Hara, and Suzuki 2003; Park et al. 2013). To our knowledge, there are no reports in the literature of Ti ions/particles originating from the surface of metallic implants and migrating systemically to deposit in the skin, at distant sites from the implant.
One of the weaknesses of the human body is that it has poor mechanisms for the elimination of most metals, especially for those known as nonessential metals (Abdulla, Parr, and Iyengar 1993). Ti is a nonessential element in that no enzymatic pathway has been elucidated requiring it as a cofactor, that there is no homeostatic control of Ti, and that Ti is not invariably detected in the newborn (Woodman et al. 1984). Hence, the presence of Ti in the body and the biokinetics and potential biologic effects of Ti are of great interest.
NPs have a greater surface-to-volume ratio as compared to MPs. They are therefore biologically more reactive and potentially more harmful to body tissues (Oberdörster et al. 2005). The toxic effects of different particles have been associated with their physicochemical properties, which include size, shape, crystalline structure, chemical composition, area and surface load, porosity, distribution, and aggregation state, among others (Oberdörster et al. 2005; Bruno et al. 2014). Thus, although NPs can be chemically similar, differences in particle size may result in different biological responses (Revell 2006). According to our search of the literature, there are no studies reporting the possible differential effect of Ti particle size on the concentration of Ti in the skin and skin appendages.
The aims of the present study were (1) to quantitate Ti traces in the hair and nails of animals injected with different sized TiO2 particles, (2) to explore the influence of NP size on the concentration of Ti in skin appendages, (3) to evaluate whether there is a relation between Ti levels in plasma and in hair and nails, and (4) to determine the presence of agglomerates of Ti dioxide particles in the skin, using a murine model.
Materials and Methods
Animal Treatment Protocol
Following our experimental model (Olmedo, Guglielmotti, and Cabrini 2002), male Wistar rats (n = 30) weighing ∼100 g were randomly injected intraperitoneally (IP) with a suspension of 99% purity anatase TiO2 NPs (Nanostructured and Amorphous Materials, Los Alamos, NM, USA) in 5 ml 0.9% sodium chloride (NaCl; Baxter Argentina S.A.), at a dose of 1.6 / 100 g body weight. In order to ensure a homogenous distribution of the particles in all the suspensions, the latter were sonicated for 15 min prior to injection. Experimental animals were injected with one of the following: TiO2-NPs10 (n = 10) with an average particle size (APS) of 10 Nm (specific-surface area: 120 m2/g) or TiO2-NPs5 (n = 10) with an APS of 5 Nm (specific surface area: 200–220 m2/g). Control animals (n = 10) were injected with an equivalent volume of vehicle.
All procedures were performed in compliance with the National Institutes of Health (NIH) guidelines for the care and use of laboratory animals (NIH Publication—Guide for the Care and Use of Laboratory Animals: Eighth Edition, 2011) and the guidelines of the School of Dentistry of the University of Buenos Aires (Res. [CD] 352/02 and Res. [CD] 694/02). Adequate measures were taken to minimize animal pain and discomfort. The protocol was approved by the institutional experimentation committee (School of Dentistry of the University of Buenos Aires, Resolution Number 28/11/2012-37).
Body Weight, Behavior, and General Health
In order to detect abnormal signs and conditions associated with toxicity throughout the experimental time, the following endpoints were evaluated: Body weight changes: At the onset of the experiment and prior to euthanasia, the animals were weighed using an APX-1502 balance (Apex Series Balances, Denver Instruments Co.) in order to detect significant changes in body weight. Body weight of experimental animals was compared to that of controls. All the animals were fed ad libitum, and food and water consumption was continuously monitored throughout the study. Behavioral signs: Sleeping and grooming patterns, excitability in response to external stimuli, and activity levels were closely monitored to detect abnormal behavior. General health: The animals’ appearance, including coat texture, hair color, and amount, as well as posture, and amount, color, and consistency of feces were monitored daily to detect signs indicative of pain, distress, and/or discomfort.
Characterization of TiO2-NPs
Prior to injection, APS and particle morphology were confirmed using a scanning electron microscope (SEM) equipped with a field-emission filament (Carl Zeiss Supra 40 microscope, Oberkochen, Germany). For this purpose, the NPs were placed on a conductive carbon tape and analyzed without being coated. Images were obtained using an in-lens detector and 4-kV acceleration voltage. The particles were chemically identified by energy dispersive X-ray spectroscopy (EDS; Oxford Instruments, Bucks, UK).
Macro- and Microscopic Study of the Skin
All the animals were euthanized by IP overdose of Ketamine Chlorhydrate (Holliday-Scott S.A., Buenos Aires, Argentina) and xylazine chlorhydrate 2% (Kensol, König, Argentina) 6 months postinjection. Animal death was confirmed by opening of the chest cavity to ensure loss of respiratory function. Scalp skin samples (1 × 1 cm) were obtained, examined macroscopically using a stereo magnifier (Zeiss Stemi 2000-C, Jena, Germany), fixed in 10% buffered formalin, and embedded in paraffin to obtain histological sections transverse to the direction of the hair flow. Section thickness was 6 microns. In all cases, the sections were treated with saturated solutions of picric acid to remove formalin pigments (acid hematin). The sections were stained with hematoxylin and eosin (H&E) or Grenacher’s carmine and examined using conventional and polarized light microscopy. Grenacher’s carmine is a red dye that stains nuclei and cytoplasm pink. This stain facilitates identification of brownish Ti deposits in tissues and/or inside cells. The histological sections stained with H&E showing particle deposits were evaluated by SEM (Carl Zeiss Supra 40 microscope, Oberkochen, Germany), and the chemical composition of the particles was determined by EDS (Oxford Instruments, Bucks, UK). Briefly, the areas showing deposits of particles were outlined on the back of the glass slide using a diamond pencil. This procedure facilitates identifying the location of the deposits when observing the sections by SEM. The samples were then placed in xylol for 24 hr in order to remove the coverslips, after which they were coated with a thin 20 Nm layer of gold in a vacuum evaporator. The coated samples were then placed on a conductive carbon tape. Images were obtained using an in-lens detector and 4-kV acceleration voltage.
Ti Concentration in Skin Appendages (Hair and Nails)
In order to assess Ti levels in skin appendages, hair samples (0.1 g) were obtained from the back, and nail samples (0.1 g) were obtained from the front paws of experimental and control rats. The skin appendages were cut into small pieces (approximately 1 mm long) to ensure feasible and fast digestion of the samples. They were disintegrated for 24 hr in ultrapure nitric acid (65%, Merck, Darmstadt, Germany). Ti quantitation was performed using inductively coupled plasma-mass spectrophotometry (ICP-MS; Elan DRC II, PerkinElmer Life and Analytical Sciences, Shelton, CT).Ti concentration was quantified in microgram per liter.
Concentration of Ti Levels in Plasma
Prior to euthanasia, 2 ml of blood were obtained by tail vein puncture for quantitation of plasma levels of Ti. Determinations were performed using ICP-MS.
Statistical Analysis
The results were compared employing one-way analysis of variance or Student’s t test, accordingly. Values are expressed as mean and standard deviation. Statistical significance was set at p < .01.
Results
None of the experimental animals showed alterations in body weight, behavior, or general health throughout the experiment, as compared to controls.
Characterization of TiO2-NPs
Physicochemical analysis of TiO2-NPs showed the same characteristics as those previously reported by our research group (Bruno et al. 2014). SEM confirmed APS of the TiO2-NPs employed in the experiment to be 10 and 5 Nm (Figure 1A and B); the particles were lentil-shaped and formed agglomerates. Chemical analysis using EDS confirmed the presence of Ti in all the particles (Figure 1C).

SEM images of TiO2-NPs. Note the lentil shape and average particle size (APS) of the particles: (A) 5 Nm APS and (B) 10 Nm APS. Original magnification: ×500,000. (C) Energy-dispersive X-ray spectroscopy analysis of the particles; the spectrum corresponding to titanium is shown. SEM = scanning electron microscope; NP = nanoparticle.
Macro and Microscopic Study of the Skin
Macroscopic examination using a stereo magnifier revealed no changes in texture and/or color of the skin in any of the studied groups. Similarly, microscopic evaluation showed no differences in tissue structure between experimental and control animals. However, skin samples from both the TiO2-NPs experimental groups (5 and 10 Nm) had agglomerates of fine brownish NPs in different zones of the connective tissue (Figure 2A–D). Particles were mostly found accumulated around the hair follicles (Figure 2A, B, and D) and/or phagocytosed by macrophages (Figure 2D). No inflammatory infiltrate, giant-cell reaction, or vascular alterations were observed. TiO2-NP deposits were found to be birefringent by polarized light microscopy. A case corresponding to the TiO2-NPs5 group is shown as an example (Figure 2E and F). SEM demonstrated the presence of agglomerates of the different sized- and shaped-particles in the connective tissue (Figure 3A). Elemental analysis by EDS confirmed that the observed deposits contained Ti (Figure 3B). Skin samples corresponding to the control group showed no particle deposits.
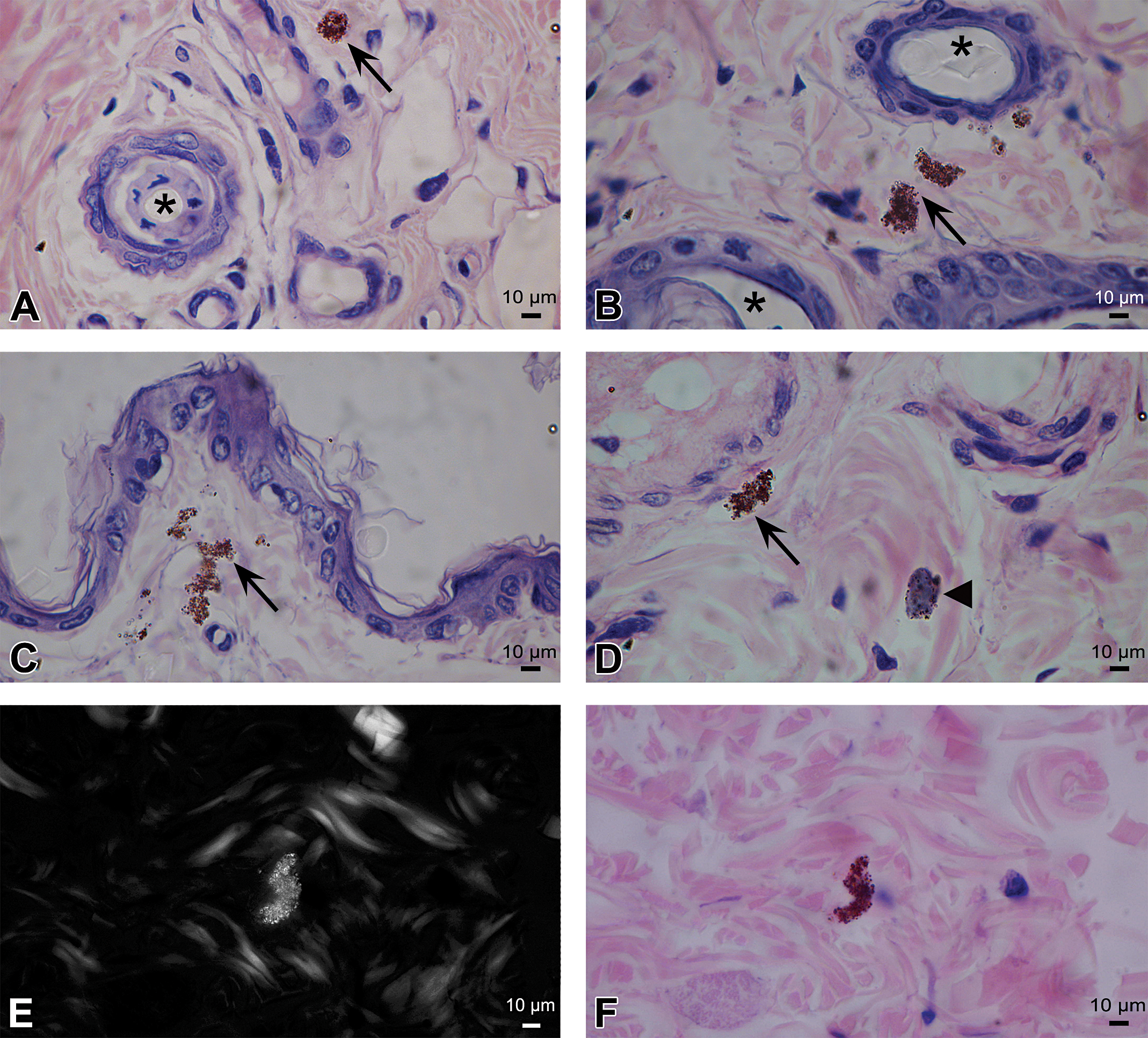
Histological analysis of the skin (TiO2-NPs5). (A–D) Particle agglomerates (→) with no associated inflammatory response can be observed in different areas of the tissues; (▴) macrophage; (*) hair follicle. H&E Stain. (E) Ti deposit showed birefringence under polarized light microscopy. (F) The same section shown in (E) as seen by light microscopy. H&E Stain. H&E = hematoxylin and eosin; NP = nanoparticle.
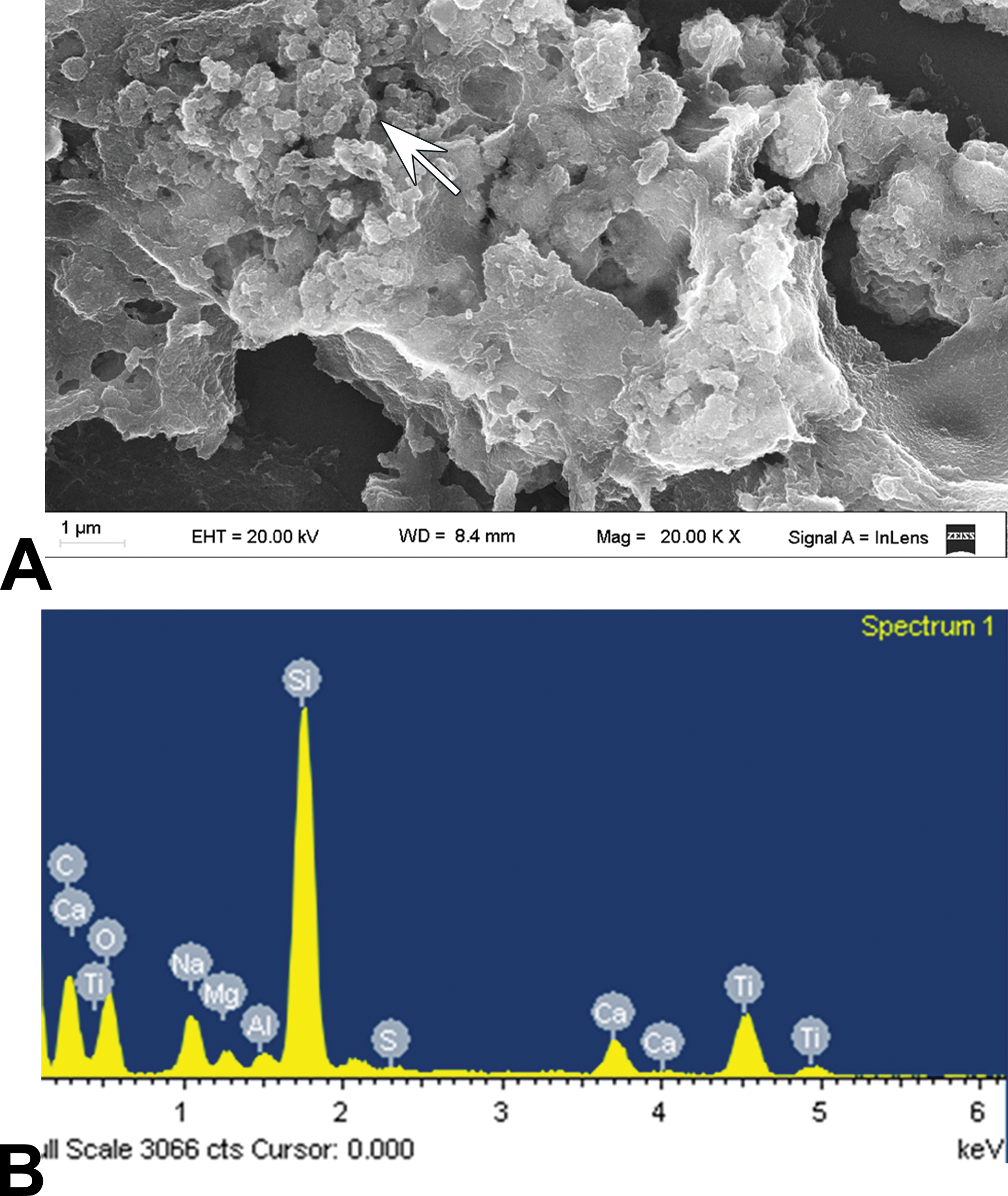
Ti agglomerates in the skin. (A) Scanning electron microscopy examination of TiO2-NPs5 agglomerates (→). (B) Energy-dispersive X-ray spectroscopy of the deposits. The spectrum corresponding to titanium is shown. NP = nanoparticle.
Concentration of Ti in Skin Appendages and Plasma
The TiO2-NPs5 group showed the highest concentration of Ti in both skin appendages (hair and nails; p < .01). No significant differences in either nail or hair concentrations of Ti were observed between the TiO2-NPs10 and control groups (Figure 4).
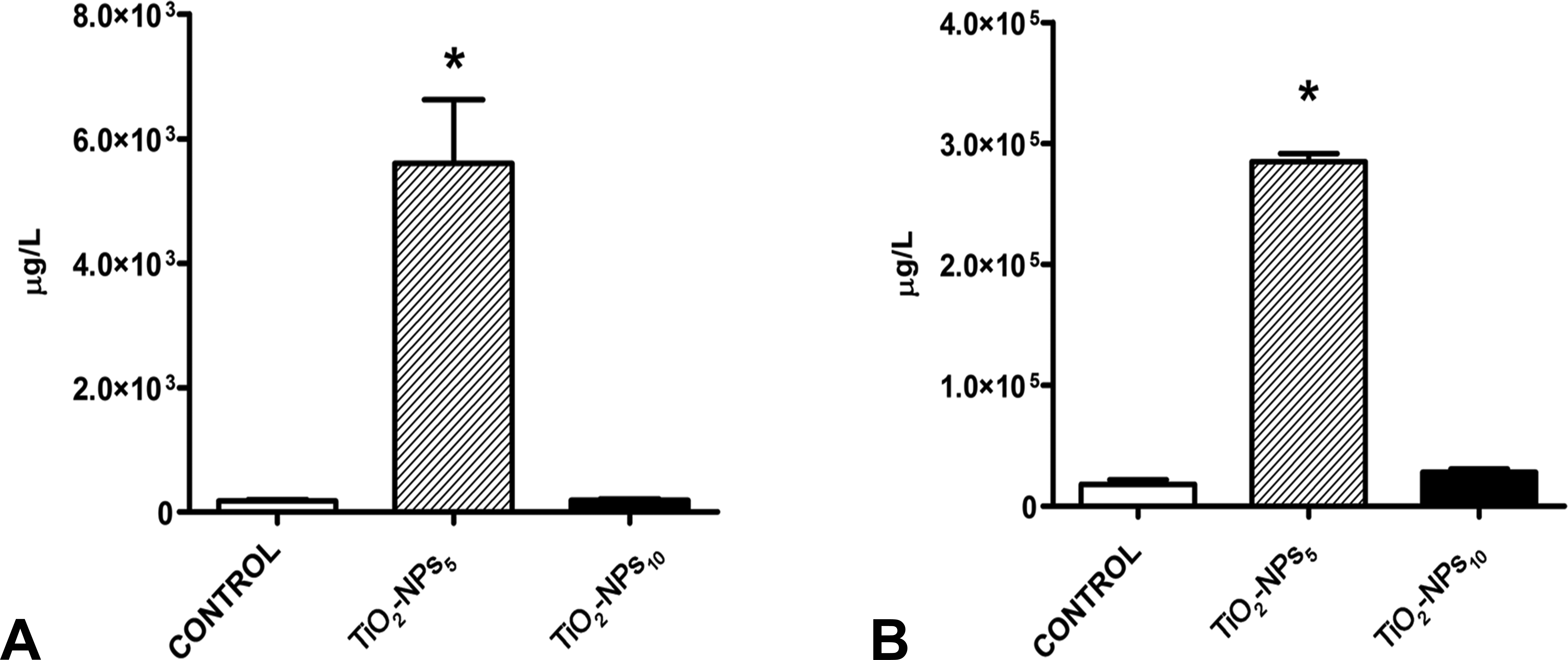
Ti concentration in skin appendages using ICP-MS. Ti concentration in hair (A) and nails (B) was determined 6 months postinjection. The histograms show the mean ± SD; levels were significantly higher in TiO2-NPs5, *p < .01. TiO2-NPs5 group concentration was 50-fold higher in nail than in hair samples. Values corresponding to hair are expressed as 103 µg/L, whereas nail values are expressed as 105 µg/L. ICP-MS = inductively coupled plasma-mass spectrophotometry; NP = nanoparticle; SD = standard deviation.
The concentration of Ti in the nails (105) was found to be two orders of magnitude higher than in hair (Figure 4A and B; 103). As shown in Figure 4A and B, Ti concentration was 50-fold higher in the nails than in hair in the TiO2-NPs5 group (284,649 ± 14,695 vs. 5,606 ± 2,041 µg/L, respectively) and was 150-fold higher in the nails than in hair in the TiO2-NPs10 group (28,063 ± 4,895 vs.188 ± 51.1 µg/L, respectively).
Plasma concentration of Ti (Figure 5) was significantly different in both the TiO2-NPs5 and TiO2-NPs10 groups compared to controls (p < .01) but did not differ between the TiO2-NPs5 and TiO2-NPs10 groups (p > .01).
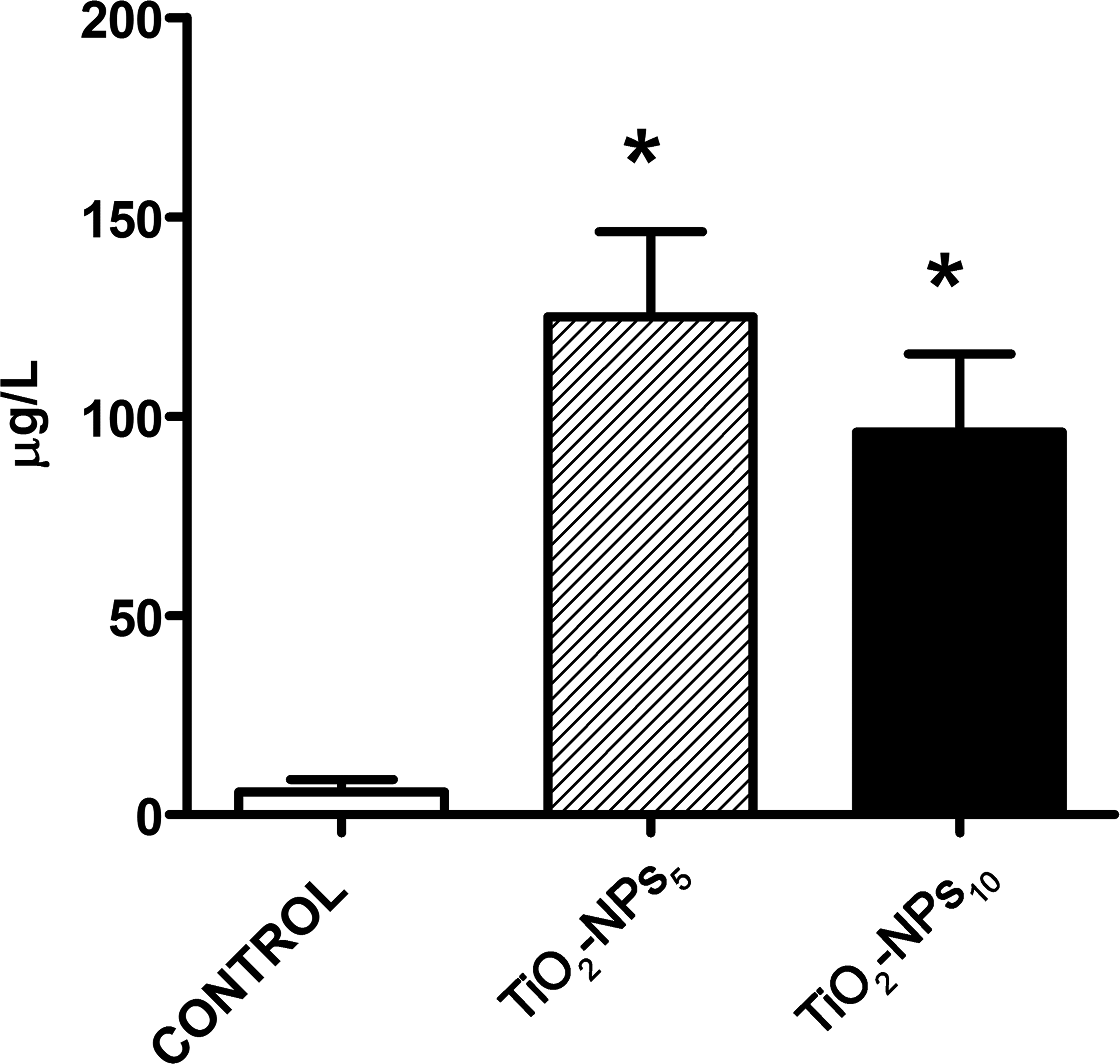
Ti concentration in plasma using ICP-MS. Ti concentration in plasma was determined 6 months postinjection. The histograms show the mean ± SD,*p < .01 compared to the control group. ICP-MS = inductively coupled plasma-mass spectrophotometry; SD = standard deviation.
As regards the relation between Ti levels in plasma and skin appendages, Ti concentration in the TiO2-NPs5 group was 600- and 12-fold lower in plasma (125 ± 42.6 µg/L) than in the nails and hair, respectively.
Discussion
Humans can be exposed to metallic traces originating from external sources, such as foods, environmental contamination, and/or occupational exposure, or from internal sources, as is the case of subjects bearing a metallic prosthetic device (Koppang et al. 2007; Skocaj et al. 2011; Rodriguez de la Flor, Hernández-Vaquero, and Fernández-Carreira 2013). Unlike exposure to traces from external sources, few reports have analyzed exposure to metallic traces originating from a metallic prosthesis.
Although many of the eighty known metals are vital to normal physiological processes in humans (e.g., Fe, Mg, and Zn), other metals, such as Pb, Hg, and Cd, are among the oldest toxicants known to humans. Different mechanisms contribute to the elimination of metals, such as loss through breast milk, body fluids, hair, nails, and exfoliating skin, among others. Hair and nails are used as indicators for screening population groups exposed to environmental pollution, occupational pollution, and/or to biomedical devices. In this regard, the presence of Cu, Zn, Cr, Co, Mn, Fe, Cd, As, Mo, Ni, Pb, and Ti in hair and nails has been reported (Kasai, Lida, and Uchida 2003; Merha and Juneja 2005; Bozkus, Germec-Cakan, and Arun 2011; Abdulrahman et al. 2012; Rodriguez de la Flor, Hernández-Vaquero, and Fernández-Carreira 2013).
Specifically regarding Ti, there is limited information on Ti toxicity, absorption, and distribution throughout the body, despite its widespread use in the food, pharmaceutical, and medical industries. And yet there are reports of tissue and body fluid, that is, plasma and urine, contamination with Ti traces originating from internal sources, such as metallic prosthetic devices (Jacobs et al. 1991; Jacobs, Gilbert, and Urban 1998; Olmedo, Tasat, et al. 2012). The average amount of exposure of implant carrying patients to Ti from tribocorrosion remains to be determined, given the influence of a number of variables including implant type, surface area and composition of the implant, time since implant placement, presence of inflammation close to the metallic structure, the method used for quantitation (ICP-MS, ICP-Optical Emission Spectrometry analysis), and the detection limits of the measuring system, among others (Jacobs et al. 1991; Jacobs, Gilbert, and Urban 1998; Jacobs et al. 1998; Zaffe, Bertoldi, and Consolo 2003).
Hair, nails, and teeth are tissues in which trace minerals are sequestered and/or stored and can be used to monitor the highest priority toxic trace elements (Barrett 1985). Nevertheless, it should be kept in mind that the effects of dietary habits, environmental changes, and the use of medications and cosmetics may also affect trace-element concentrations in these tissues. In this regard, by feeding the animals a Ti-free diet and avoiding external sources of Ti contamination, the experimental rat model used in the present study rules out the confounding effect of the aforementioned sources of external exposure on the studied biological parameters. In humans, however, the hair and nails are subjected to external chemical agents such as those found in cosmetics. According to Wilhelm et al. (1989), metal concentrations in hair may vary with the type of shampoo and the method and frequency of shampooing, and the authors thus concluded human hair is not an appropriate indicator of systemic metal ion accumulation. As a result, estimating human Ti concentrations using hair may result in an underestimation of Ti levels. Animals are not exposed to Ti leaching agents, and animal hair is therefore not affected by this variable. Hence, the results observed in hair in the present study would be accurate and reliable.
In previous studies, we explored Ti deposition in abdominal organs and observed Ti deposits in these organs both macro and microscopically (Olmedo et al. 2005, 2011; Olmedo, Tasat, Evelson, et al. 2008; Olmedo, Tasat, Guglielmotti, et al. 2008; Bruno et al. 2014). The present study, however, sought to quantitate Ti traces in the hair and nails of animals injected with different sized TiO2 particles and to determine the presence of agglomerates of TiO2 particles in the skin. Several transport mechanisms have been described for Ti, that is, systemic dissemination by the vascular and/or lymphatic systems as free particles or as phagocytosed particles within macrophages (Olmedo, Tasat, et al. 2003). The NPs observed at sites distant from the administration site (IP) may have reached such locations via one of these mechanisms. Whereas blood and other body fluids show transient concentrations of metallic traces, skin appendages provide a continuous record of elemental concentration (Abdulrahman et al. 2012). In line with this observation by Abdulrahman et al., Ti levels found in plasma at 6 months were significantly lower than those found in skin appendages, given that hair and nail samples show past exposure and accumulation of metals over a long period of time. Regarding particle size, the result of the present study showed TiO2-NPs5 concentration to be higher than that of TiO2-NPs10 in both the studied skin appendages. Given that the hair and nails can be considered excretory products, it could be stated that the smaller NPs (5 Nm) used in our study were more easily eliminated through the skin appendages.
In addition, our results showed Ti concentration to be significantly higher in the nails than in hair. Our findings are in line with a study by Bozkus et al. (2011) who reported a higher concentration of Ti in the nails than in hair in patients with Ti-6Al-4V alloy miniplates and miniscrews. Moreover, the concentration of metals other than Ti, such as Cu, Zn, Mg, Fe, Cr, Cd, As, Ni, and Pb, originating from external sources followed the same pattern (Abdulrahman et al. 2012).
The lower concentrations of metallic traces observed in hair in human studies might be attributed to the incorporation of elements into the keratin structure of hair, which takes place by binding to the sulfhydryl groups that are present in the follicular protein. In this regard, detergents such as soap and shampoos, hair pomades, lotions, hair bleaches, and dyes actually compete with the complexing ability of these reactive sites, thus leading to a significant leaching of elements from the shaft bulk (Buchancová et al. 1993). Interestingly, the results obtained using the present experimental rat model, in which the animals were not affected by the aforementioned factors, also showed that the concentrations of Ti were lower in hair than in the nails.
It could be hypothesized that Ti concentrations in the nails and hair would have different biological excretion mechanisms, which would explain our results. Taking into account particle size, it may also be true that there is a distribution problem, and the larger particles do not gain access to these skin appendages.
Several trace elements are important in biochemical processes where an excess or absence of these essential trace elements can cause serious physiological problems and even diseases (Dombovari and Papp 1998, 1999). In this regard, a pathology termed Yellow Nail Syndrome, characterized by nail changes, respiratory disorders, and lymphedema, has been reported, and Ti has been suggested to be the etiological agent (Berglund and Carlmark 2011).
There is controversy as to whether the skin, like the nails and hair, can be considered a bioindicator of Ti deposition. Regarding skin exposure, Adachi et al. (2013) found no evidence of TiO2 penetration in the viable skin areas, whereas Wu et al. (2009) demonstrated that TiO2 can penetrate into the deep layer of the viable epidermis.
With regard to internal sources, metallosis of the skin overlying a metallic Ti implant is quite rare (Park et al. 2013). Although no macroscopic changes in color or texture of the scalp skin were observed in the present study, histological examination showed the presence of agglomerates of fine brownish NPs in different regions of the connective tissue. Although it is well established that the presence of a particulate material can trigger an inflammatory response, with vascular alterations, inflammatory infiltrate, and even giant cells (Revell 2006), our results only showed the presence of macrophages with no inflammatory infiltrate or giant-cell reaction.
Biomarkers can serve as informative predictors of disease onset in asymptomatic individuals (predictive biomarkers), indicators of disease incidence and progression (diagnostic/progression biomarkers), and measures of response to treatment or surgical intervention (response biomarkers; Sumner, Ross, and Purdue 2014). The hair, nails, and skin are reliable biomarkers for monitoring and analyzing the toxicokinetic behavior of Ti. All three biological materials may be used as bioindicators of the level of systemic contamination with Ti, allowing short- and long-term clinical follow-up of an implant.
In summary, our experimental study showed the presence of Ti traces in the nails, hair, and plasma of Ti exposed rats, and that the concentration of the smallest NPs (5 Nm) was higher than that of 10 Nm NPs in all the studied samples. In addition, Ti NP size might influence trace concentration level in hair and nails, suggesting that 5 Nm Ti particles are more easily eliminated through these skin appendages.
Given that the nails showed the highest levels of Ti and that these skin appendages are not exposed to agents that can leach out Ti, as occurs with hair, we propose the nails as the most suitable and reliable bioindicator for monitoring systemic contamination with Ti.
Footnotes
Acknowledgment
We specially thank Dr. Sandra Renou for her assistance in editing the photographs.
Authors’ Contribution
Authors contributed to conception or design (DT, DO); data acquisition, analysis, or interpretation (MD, MB, BG); drafting the manuscript (DO); and critically revising the manuscript (DT, MD, MB, BG, DO). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants: UBACyT20020150100032BA and 0020130100332BA from the University of Buenos Aires and PIP 11220130100091 from the National Council for Scientific and Technological Research, Argentina.
