Abstract
Gastric mucosal injury is frequently observed in nonclinical studies of nonhuman primates. Because microscopic evaluation of stomach is generally a terminal procedure, our objective was to determine whether serum pepsinogen I (PG I) could serve as a noninvasive biomarker for detection of gastric mucosal injury in monkey. Serum PG I was measured using a commercial human immunoassay in cynomolgus monkeys (n = 166) prior to dosing and/or terminally in 11 studies of up to 1 month duration. Mean (SD) PG I values (ug/L) for monkeys with (n = 59) and without (n = 100) gastric mucosal degeneration were 101 (215) and 28 (12.6), respectively. For monkeys with baseline and terminal PG I data, mean (SD) fold change (ratio of terminal to baseline PG I) for monkeys with (n = 57) and without (n = 76) glandular degeneration were 4.1 (11.3) and 1 (0.28). Receiver operating characteristic area under the curve (AUC) data demonstrated moderate diagnostic accuracy for serum PG I for glandular degeneration, AUC (SE) 0.789 (0.04), with improved diagnostic accuracy as a fold change of baseline, AUC (SE) 0.816 (0.04), consistent with the large interindividual but low intraindividual variability of serum PG I values in control monkeys. These data demonstrate that serum PG I is a useful biomarker of drug-induced gastric mucosal injury in the cynomolgus monkey.
Although gastric mucosal injury is not uncommon in toxicology studies, there are currently no noninvasive biomarkers that reflect gastric mucosal status in preclinical species. Pepsinogens are aspartic proteinases involved in peptide proteolysis, which play a prominent role in gastric digestion in vertebrates. Produced as inactive proenzymes that are converted into pepsin in the acid environment of the gastric lumen, several isoforms of pepsinogens exist within and across species. In humans, pepsinogens I and II (PG I and PG II) are the 2 major pepsinogens (Gritti, Banfi, and Roi 2000; Plebani 1993). In cynomolgus monkey, PG I predominates, constituting nearly 90% of the total pepsinogen in gastric extracts, with the remainder as PG II (Kageyama 1994).
In humans, PG I is exclusively produced by the chief and mucous neck cells of the fundus and body of the stomach, while PG II is produced by these cells as well as by the epithelium of the cardiac and pyloric glands, Brunner’s glands of the proximal duodenum (Samloff and Liebman 1973), prostate gland (Gritti, Banfi, and Roi 2000), and lung (Foster et al. 2004). In cynomolgus monkey, PG I has been localized to chief cells of the mucosa of the fundus, body, and to a lesser extent pyloric antrum (Vidal, Mirabile, and Thomas 2008); localization of PG II in the stomach has not been evaluated, but PG II has been identified in the lung and prostate of Japanese monkey (Moriyama, Kageyama, and Takahashi 1983; Moriyama et al. 1985).
Pepsinogens are primarily secreted into the gastric lumen, but a small percentage diffuses into the blood stream (Mukoubayashi et al. 2007; Gritti, Banfi, and Roi 2000). Serum levels of pepsinogens in humans are considered to reflect the morphologic and functional state of the gastric mucosa (Gritti, Banfi, and Roi 2000; di Mario and Cavallaro 2008). For example, although PG II is more widely distributed in human stomach, gastric mucosal atrophy is associated with progressive decreases in serum PG I levels while serum PG II levels generally remain stable. Thus, as chronic atrophic gastritis progresses from the antrum toward the body of the stomach, decreases in the PG I/PG II ratio are used to monitor the development and progression of atrophic gastritis, an important risk factor in the development of gastric cancer. Increases in PG II are more sensitive to chronic gastritis caused by Helicobacter pylori than changes in PG I or PG I/II ratio (di Mario et al. 2006). Since H. pylori is another risk factor for gastric carcinogenesis, serology is increasingly combined with measurement of pepsinogens in at-risk populations (Ikeda et al. 2016).
Immunoassays for PG I and PG II have shown diagnostic utility for monitoring of gastric injury in patients; however, in nonclinical studies and in veterinary clinical practice, pepsinogen activity assays are more commonly used as a surrogate for peptic activity due to the lack of species-specific immunoassays. As these catalytic assays lack specificity because they measure the activities of other serum proteases in addition to pepsin, there is a need for development of specific assays to evaluate gastric mucosal status. Since chief cells are widely distributed throughout the stomach in monkeys, serum PG I is an excellent candidate marker of gastric mucosal injury. Our objective was to evaluate the diagnostic performance of serum PG I as a marker of drug-induced mucosal injury in a retrospective analysis of toxicology studies in the cynomolgus monkey.
Materials and Methods
Animals
Captive bred cynomolgus monkeys of Mauritian, Indonesian, or Chinese origin were obtained from Covance Research Products, Inc. (Denver, Pennsylvania), Primate Products (Miami, Florida), and Charles River Laboratories (Houston, Texas). Monkeys were aged 2.5 to 6 years and were singly housed (at 64°F to 84°F, with a relative humidity of 30% to 70%) on a 12 hr-light/12-hr dark cycle. Monkeys were fed a commercially available diet supplemented with 6 to 8 biscuits/day and a daily allotment of fresh fruit. Water was provided ad libitum. All studies were conducted in accordance with the GlaxoSmithKline (GSK) Policy on the Care, Welfare and Treatment of Laboratory Animals and were reviewed by the Institutional Animal Care and Use Committee either at GSK or by the ethical review process at the institution where the work was performed.
Study Samples
Sera for PG I analysis were collected from cynomolgus monkeys (n = 166) on nonclinical investigative, dose range, or definitive toxicity studies of up to 1 month duration conducted by GSK. Test articles were given once daily at 3 dose levels to male or female monkeys (n = 1–3 per dose) for 4 to 28 days. Monkeys were dosed by the oral route except for one inhalation study, and two studies included a recovery phase for evaluation of reversibility of gastric mucosal injury. Blood samples for standard clinical pathology parameters and serum PG I measurement were collected from the femoral vessel of unanesthetized fasted monkeys once or twice prestudy and at study termination for all studies, with the exception of two studies where only a terminal sample was collected for PG I analysis. Monkeys were sacrificed by exsanguination under pentobarbital anesthesia following an intravenous or intramuscular injection of ketamine hydrochloride. Protocol tissues were collected and fixed in 10% neutral buffered formalin, embedded in paraffin, and stained with hematoxylin and eosin for histopathologic evaluation. Sections of stomach from control and drug-treated monkeys were evaluated retrospectively by a single primary (D.E.) and peer review (S.R.) pathologist; principal histopathologic changes and consistency of grading were agreed by the study pathologists. Histopathology was assessed with knowledge of treatment group but without knowledge of PG I or other clinical pathology data. Morphologic diagnoses were determined for major pathologic processes based on comprehensive evaluation of stomach sections that included cardia, fundus, body, and antrum. Histopathology severity scores were assessed on a scale from 0 (no change) to 4 (severe pathology). For the biomarker performance analysis, lesion severity and localization or subtopography were not considered, and animals with histopathology score = 0 were defined as “negative” and animals with histopathology score > 0 were defined as “positive.”
PGI Assay
Monkey serum PG I was measured with the Biohit Diagnostics Human PGI ELISA Kit (Helsinki, Finland). Prior to assessment of study samples, method validation procedures were conducted to confirm this kit provided acceptable performance standards for measurement of PG I in monkey serum (Lynch 2016). Intra- and interassay precision, linearity, and recovery evaluations were performed using monkey sera. Intraassay precision was evaluated in monkey serum samples run across 20 wells of a single 96-well plate. For interassay precision, samples were run in quadruplicate across 5 of the 96-well plates, and the Biohit quality control sample was run in duplicate across 10 of the 96-well plates. Stability of PG I in monkey serum was evaluated by measuring concentrations in freshly collected samples and after frozen storage (−20°C) for 6 months. Biologic variation of PG I was evaluated in serum samples from study controls from 5 male and 4 female primate studies (n = 18 male and 10 female monkeys), in which the number of animals per control group ranged from 1 to 8. Control monkeys were fasted overnight and serially sampled between 2 and 6 times at intervals from 5 days to 63 days, and gastric histopathology was examined at study termination. Serum PG I concentrations were also measured for 8 stock monkeys (n = 4 males and 4 females) over 4 weekly sample collections, with collection of a final nonterminal sample when not fasted to assess food effects on serum PG I concentrations.
Immunohistochemistry
PG I localization in monkey stomach was assessed in formalin-fixed paraffin embedded sections of monkey stomach stained on the Ventana Discovery system. All reagents used were from the Discovery DABMap kit (Ventana Medical Systems, Tuscon, AZ) unless specified. Briefly, paraffin sections were deparaffinized and rehydrated. Sections were subjected to heat-induced antigen retrieval using Ventana Cell Conditioning 1 buffer and were blocked with 3% hydrogen peroxide, avidin/biotin blocking system, and a DAKO protein block (Agilent, Santa Clara, CA). A mouse monoclonal antihuman PG I antibody (U.S. Biologicals, Swampscott, Massachusetts) was used at a concentration of 5 ug/ml, and an isotype-matched mouse IgG (Southern Biotechnology, Birmingham, AL) was used as a negative control. PG I was labeled using an antimouse biotinylated secondary antibody (Vector Laboratories, Burlingame, CA), followed by streptavidin peroxidase, and detected with 3,3 diaminobenzidine.
Data Analysis
Absolute and fold change values of PG I (expressed as the ratio of terminal to baseline value) were determined for control (vehicle) and drug-treated monkeys. Sources of biological variation were quantified and partitioned using Proc MIXED [MIXED Procedure of SAS/STAT, 2008] in SAS (Cary, NC). Diagnostic accuracy of PG I for gastric mucosal degeneration was evaluated by receiver operating characteristic (ROC) analysis (MedCalc®, Version 12.3, Ostend, Belgium) for both the absolute PG I value and the fold change of PG I from baseline.
Results
PG I Assay Validation and Biological Variation
The Biohit ELISA assay demonstrated good precision (intra assay CV% < 6, inter assay precision CV% < 15), dilutional linearity (r 2 = .999), and recovery (95–109%) in monkey serum (Supplementary Tables 1 and 2; Supplementary Figures 1 and 2). Although the original lot of Biohit reagent was not available for measuring PG I in serum samples reanalyzed after storage at −20°C for 6 months, PG I concentrations (Mean ± SD) were comparable to respective values for fresh samples, thus serum PG I is stable for up to 6 months at −20°C (Supplementary Table 3).
There were no sex differences in PG I values: the average baseline serum PG I concentration was 31.7 ug/L for males and 28.2 ug/L for female monkeys (n = 76 and 55, respectively). While all study samples were collected from fasted animals, there did not appear to be a food effect on serum PG I values based on data from a small cohort of monkeys sampled at weekly intervals over 1 month (Supplementary Figure 3). As this was a retrospective study, the data used in this performance analysis were not designed for estimation of biological variation; however, serial PG I measurements in vehicle control monkeys from several studies indicate greater variation in PG I values between monkeys than within individual monkeys (Figure 1). This was confirmed by a variance components analysis of control data, which demonstrated that between animal variability (CVB) was the largest contributor to biological variation for PG I, approximating 75% of the total variance, estimated CVB of 46.4 and 64.7 for males and females, respectively, while within animal or residual variance and the variability between studies (CVw) comprised a minor percentage of the total variance (Table 1).
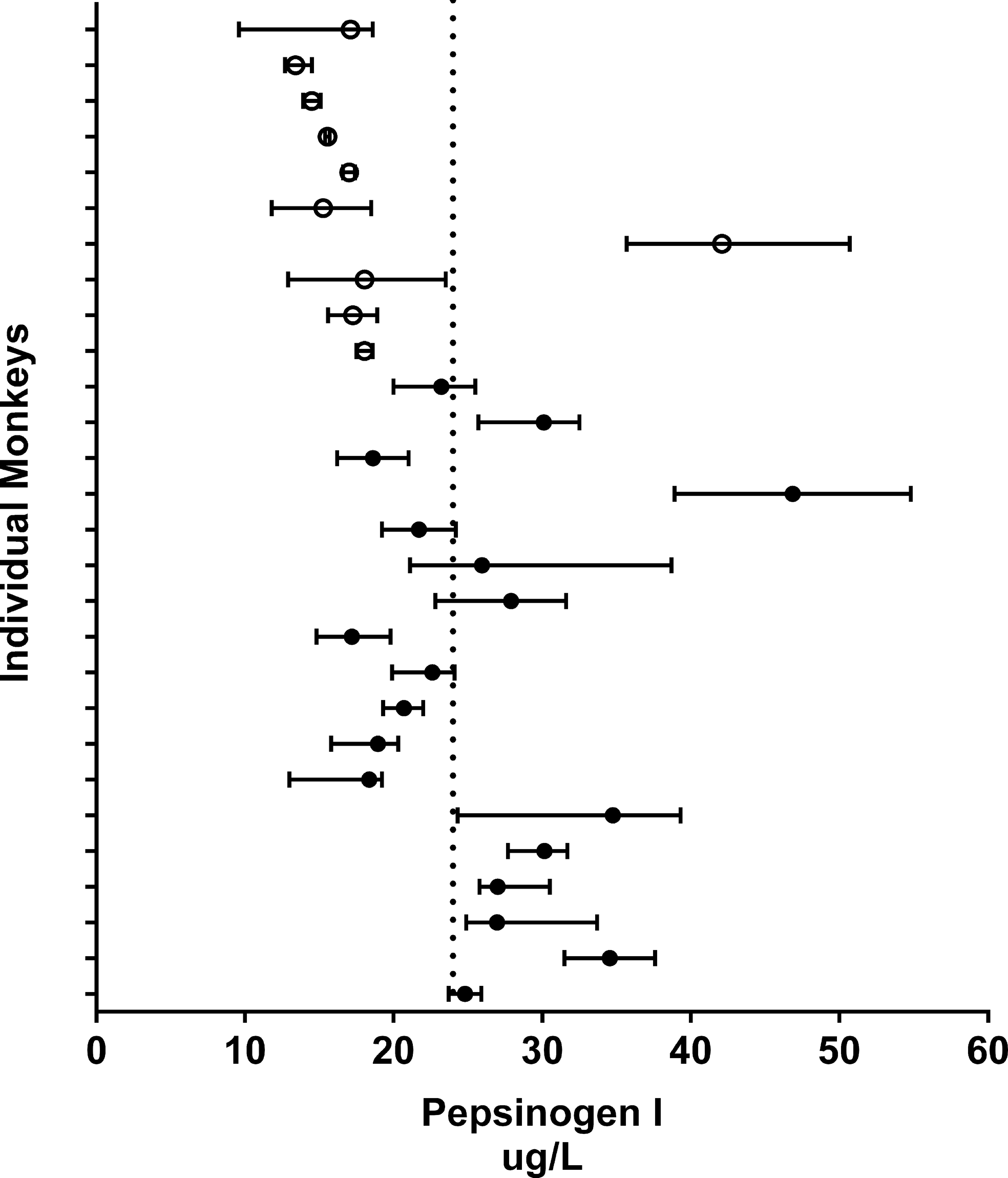
Biologic variation of pepsinogen I (PG I) in control monkeys. For each monkey, the horizontal bars indicate the median and range of PG I collected for 18 males (closed circles) and 10 females (open circles) between 2 and 6 times at intervals between 5 days and 63 days. The dashed vertical line represents the population mean (24 ug/L).
Sources of Variance of pepsinogen I (PG I) in Control Monkeys.
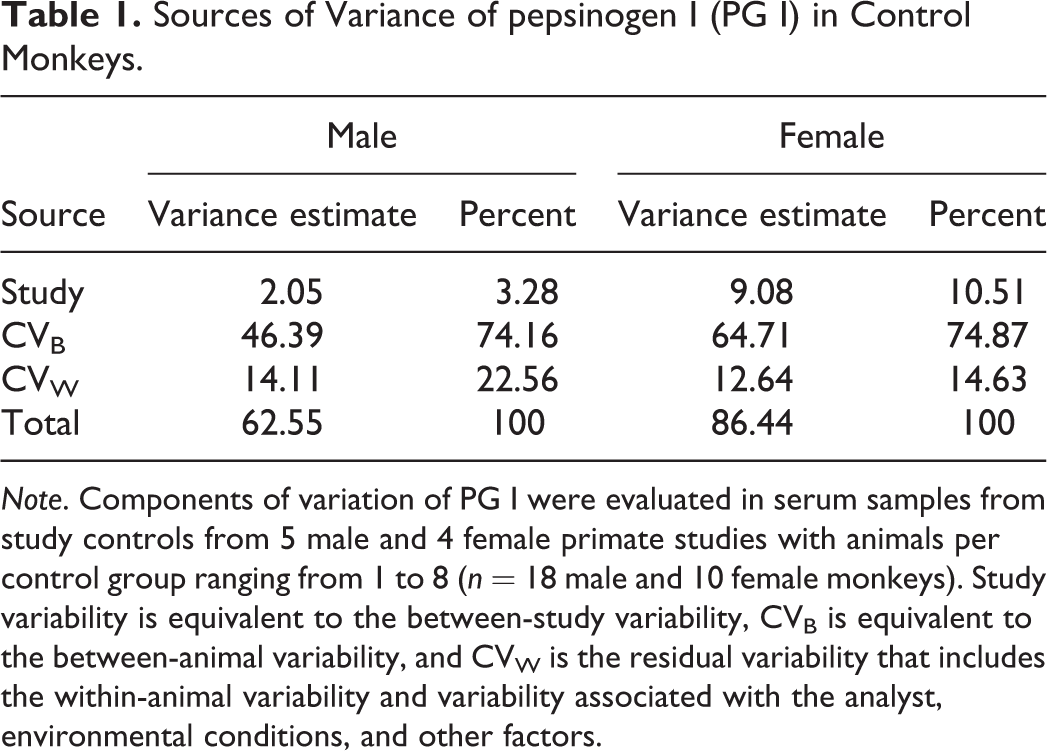
Note. Components of variation of PG I were evaluated in serum samples from study controls from 5 male and 4 female primate studies with animals per control group ranging from 1 to 8 (n = 18 male and 10 female monkeys). Study variability is equivalent to the between-study variability, CVB is equivalent to the between-animal variability, and CVW is the residual variability that includes the within-animal variability and variability associated with the analyst, environmental conditions, and other factors.
Gastric Histopathology and PG I Localization
Gastric histologic findings were grouped into six general morphologic diagnoses (Table 2). No gastric abnormalities were observed in approximately half of the monkeys, and glandular degeneration with or without regeneration was the most prevalent pathologic finding. Glandular degeneration was characterized by single cell necrosis, dropout, or loss of the oxyntic glandular epithelium, often with dilated glands lined by attenuated epithelium and luminal debris (Figure 2E and F). A subset of monkeys with glandular degeneration had evidence of regeneration manifested as cytoplasmic basophilia and increased mitotic activity of the glandular epithelium. Erosion or ulceration was of low incidence and was associated with glandular degeneration/regeneration in approximately half of the affected monkeys. Glandular degeneration with or without regeneration and mucosal erosion or ulceration were considered primary manifestations of gastric injury, and the finding of glandular degeneration was used as the reference gastric lesion for the ROC analysis. Eosinophilic or mononuclear mucosal infiltrates and interstitial fibrosis were secondary findings of relatively low incidence that were generally associated with glandular degeneration/regeneration and were not evaluated in the ROC analysis.
Incidence of Gastric Mucosal Findings and Associated Pepsinogen I (PG I) Values.
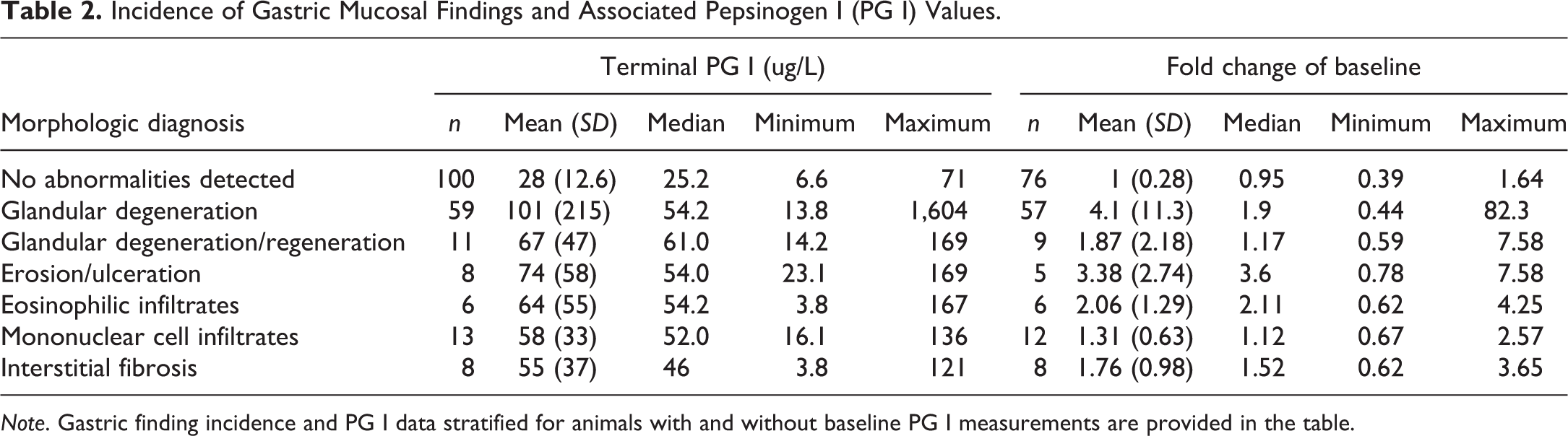
Note. Gastric finding incidence and PG I data stratified for animals with and without baseline PG I measurements are provided in the table.
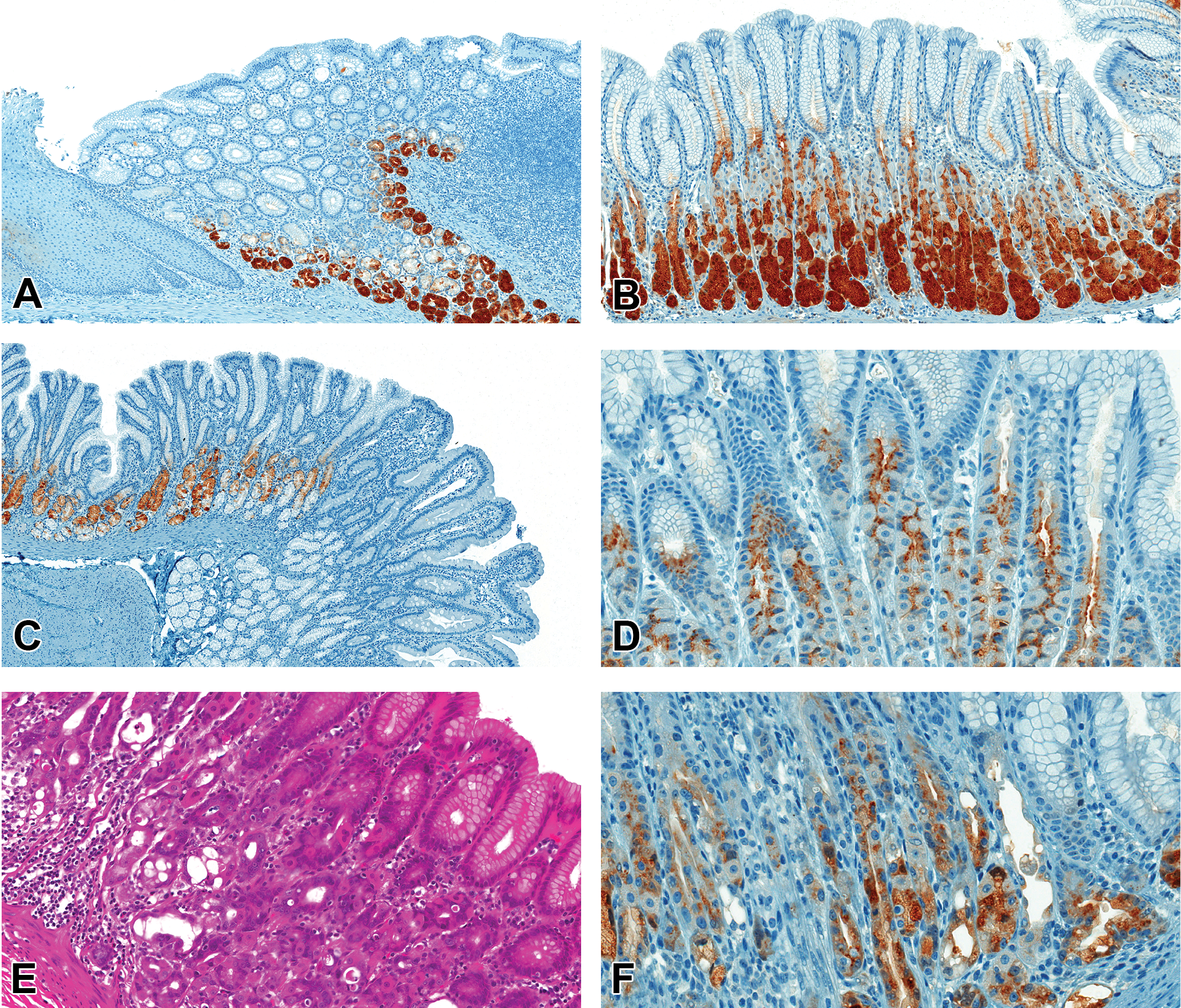
Pepsinogen I (PG I) expression in cynomolgus monkey stomach. Immunohistochemistry for PG I in control monkey stomach (A to D) and gastric mucosal injury (F, 40× original magnification). In normal monkey stomach, PG I localizes within the chief cells of the fundus (A) and body (B), particularly at the base of the gastric glands, with decreased expression in the chief cells of the mucus neck glands in the antrum/pyloroduodenal junction (C). PG I expression in the neck of gastric glands of the body of the stomach (D). Glandular degeneration (E, H&E) with unchanged PG I expression (F).
Immunohistochemistry for PG I in the gastric mucosa of cynomolgus monkeys demonstrated comparable localization in gastric chief cells of control monkeys and monkeys with drug-induced gastric mucosal degeneration. In monkey stomach, PG I–immunoreactivity localized to chief cells predominantly within the fundus and body, with limited immunoreactivity in the pylorus or antrum, and no expression in the cardia where chief cells are absent (Figure 2A–C). Immunoreactivity of PG I in the neck of the gastric glands appeared as supranuclear and apical membrane staining and bleb formation (Figure 2D), consistent with storage as proenzymes with an exosomal mode of release.
PG I Changes and Gastric Mucosal Injury
PG I values measured in monkey serum across 11 toxicology studies were significantly increased, Mean (SD) = 101 (215), p < .001, for monkeys with drug-induced gastric mucosal degeneration compared to monkeys without gastric findings, 28.5 (12.6; Table 2). For studies where baseline PG I values were measured, the fold changes of PG I at study termination were increased approximately 4-fold relative to baseline values for these monkeys. Increases of similar magnitude were seen for monkeys with gastric mucosal erosion/ulceration. Although a low incidence finding, mucosal erosion/ulceration was largely associated with glandular degeneration: the 5 monkeys with mucosal ulceration and glandular degeneration with both terminal and baseline PG I values had the largest increase (3.6- to 7.6-fold respective baseline) in serum PG I values.
Diagnostic Utility of PG I for Glandular degeneration
ROC analysis was performed for PG I data using a positive classifier variable of gastric mucosal degeneration and negative classifier of no abnormalities determined. ROC analysis demonstrated that serum PG I had moderate diagnostic utility for predicting gastric mucosal degeneration (AUC [95% confidence interval] = 0.789 [0.718–0.849]). Diagnostic accuracy of serum PG I for glandular degeneration was slightly improved in an ROC analysis of the fold change in terminal PG I values compared to respective baseline PG I levels (0.816 [0.738–0.879]).
Discussion
The principal objectives of this study were to evaluate and characterize PG I as a potential biomarker of gastric mucosal injury in cynomolgus monkey. Here, we demonstrated prominent expression of PG I within chief cells of the cynomolgus fundus, body, and pylorus. As PG I is widely expressed in gastric glands in both control monkeys and monkeys with drug-induced gastric mucosal degeneration, PG I is an excellent candidate marker of gastric mucosal injury in the cynomolgus monkey.
In humans, the diagnostic use of pepsinogens has progressed from measurement of PG I or PG II individually to evaluation of a panel of markers that includes the individual pepsinogens, the ratio of PG I/II, and serology for Helicobacter (Yamaguchi et al. 2016). While in this study PG I had moderate diagnostic utility, it is possible that sensitivity could be increased by the addition of other noninvasive serum or plasma biomarkers such as ghrelin, a product of enteroendocrine cells of the oxyntic glands of the body of the stomach, or gastrin-17, a marker of antral gland function (di Mario and Cavallaro 2008).
Pepsinogen gene families and products differ among species. Where PG I is the product of multiple genes, PG II typically originates from a single gene copy in mammals (Castro et al. 2012). For example, while PG I is the product of 3 gene copies in humans, 14 different PG I cDNAs have been cloned from the stomach of orangutans (Narita et al. 2010). Similarly, 7 different pepsinogen fractions were isolated from human stomach (Samloff and Liebman 1973) and 5 pepsinogens have been isolated from macaques (Kageyama 1994). Although the specificity and cross-reactivity of the Biohit human PG I ELISA for cynomolgus monkey PG I was confirmed based on excellent precision, linearity, and recovery, it is possible that the human PG I assay did not detect the entire concentration of serum PG I in monkey sera.
The evaluation of data from a relatively large cohort of monkey studies for this meta-analysis of PG I performance is an example of the use of regulatory toxicology data in biomarker characterization. An essential component and major strength of this study was the consistency of the histopathology evaluation across the entire data set. Unlike many, often larger meta-analyses, stomachs from this study were assessed by a single study and peer review pathologist, and histopathology findings were “curated” into broad pathophysiologic categories of gastric mucosal injury in order to compensate for differences across studies due to changes in sectioning protocols over time, and to enable a more robust statistical analysis. The diagnostic utility of PG I was only estimated for the primary gastric injury manifestation of glandular degeneration. Other findings including erosion or ulceration, inflammation, and fibrosis were recorded but not evaluated against PG I due to lack of specificity, low number, and/or or presence of concomitant glandular mucosal degeneration. Further, during data curation for the purpose of the meta-analysis, lesion subtopography and severity scores were disregarded to enable a statistically robust meta-analysis across studies. Since PG I is broadly distributed throughout the glandular mucosa of the cynomolgus monkey, this had no impact on the outcome of the performance evaluation.
One of the limitations of this study was that it was retrospective and not optimally designed for the evaluation of biological variability. However, as a new biomarker, baseline PG I measurements were collected for the majority of studies to aid interpretation of PG I changes and to understand the dynamic range and idiosyncrasies of serum PG I concentrations. We capitalized upon the large number of prestudy baseline and control animal data to demonstrate that interindividual variability was the greatest contributor to biological variability of PG I, as reflected by the greater diagnostic utility of the fold change of PG I relative to baseline values. This is similar to the situation in humans, where PG levels are known to not vary significantly over a 10-yr period in the majority (90%) of adults (Yamaguchi et al. 2016). Also, as in humans, there did not appear to be food effects on PG I values in monkeys. This differs from the situation in dogs where feeding has a significant effect on PG I values (Suchodolski et al. 2003).
In conclusion, we demonstrate that measurement of serum PG I has diagnostic value as a noninvasive marker of drug-induced gastric mucosal injury in the cynomolgus monkey. Evaluation of changes in serum PG I concentration relative to baseline values optimizes diagnostic performance.
Footnotes
Authors’ Note
The current affiliations for two of the authors have changed. Carie Kimbrough is currently affiliated with PAREXEL, International, Durham, North Carolina, USA. Sabine Rehm is retired.
Acknowledgments
The authors thank Beverly Maleeff, Janice Kane, and Deatra Floyd for their expert technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
