Abstract
A substantial quantity of data on Sprague-Dawley (SD) and Hannover Wistar rats strains have been published concerning their source, diet, and housing conditions, as well as the incidences of nonneoplastic lesions and neoplasms observed in different laboratories. Differences between the commonly used rat strains provided by different breeders (i.e., CD (SD) vs. Harlan Sprague-Dawley strain or Crl: WI(Han) vs. Wistar Hannover (Han)–derived strain, continued breeding by RCC Ltd., Switzerland, thereafter continued breeding by Harlan) may include, but are not limited to, body weight, incidence, and onset of major nonneoplastic lesions and neoplasms, and these can impact the development of a nonclinical safety program. Fisher 344 (F344) and SD rat strains generally have the highest tumor incidences, exceeding that in Wistar rats. Certain tumors are more commonly observed in one strain, and for some, the difference in incidence may be so significant that the tumor may even be considered characteristic for a specific strain (e.g., thymoma in Wistar and amphophilic renal adenoma in SD).
The advantages of using rats in toxicity testing were summarized by Kacew and Festing (1996) as (a) metabolic pathway similarities to humans, (b) numerous similar anatomical and physiological characteristics, (c) a large database, which is extremely important for comparative purposes, and (d) the ease of breeding and maintenance of animals at relatively low cost. The authors pointed out that the choice of the rat strain may be complicated, considering the existence of more than 200 different strains. An overview of differences in the most commonly used rat models was presented by Weber, Razinger, et al. (2011).
A number of publications deal with the spontaneous incidences of nonneoplastic lesions and neoplasms of different rat strains (Bode et al. 1985; Bomhard 1992; Bomhard, Karbe, and Loeser 1986; Bruner et al. 2009; M. Chandra, Riley, and Johnson 1992; Haseman, Hailey, and Morris 1998; Kaspareit-Rittinghausen and Deerberg 1991; Kaspareit-Rittinghausen, Deerberg, and Rapp 1987; Kaspareit-Rittinghausen et al. 1990; Nakazawa et al. 2001; National Toxicology Program [NTP] 2010, 2011; Nogueira de Sousa Trotte, Menezes, and Tortelly 2008; Poteracki and Walsh 1998; Prejean et al. 1973; Walsh and Poteracki 1994). Breeders also made their data available to the public (Charles River 2004, 2011; Envigo 2016; Kaspareit-Rittinghausen et al. 1990).
Publications on specific spontaneous neoplasms in some organs or organ systems include hibernomas in Sprague-Dawley (SD) rats (Bruner et al. 2009) or Wistar rats (S. Chandra et al. 2008), renal tumors (Zwicker et al. 1992), amphophilic-vacuolar renal tubule tumors in SD rats (Crabbs et al. 2013), and spontaneous histiocytic sarcomas in Wistar inbred strain crossed with an inbred yellow, pink-eyed strain (after World War II; BDII/Han) rats (Kaspareit-Rittinghausen and Deerberg 1988).
Less often, other conditions, for example, source, diet, housing conditions, mortality rates, and so on, were published (Charles River 2004, 2011; Envigo 2016; Weber 2012).
All these published data represent cohorts of control data, collected by different institutions throughout the years. It was assumed that these control data might not be stable over time, perhaps due to genetic drift, or simply by changing nomenclatures. Tennekes et al. (2004) examined control data from several institutions over 20 years and found changes in the incidences of spontaneous neoplasms in skin (fibroma and keratoacanthoma), thyroid (C-cell and follicular cell adenomas/carcinomas), uterus (stromal polyp), testes (Leydig cell tumor), and hemolymphoreticular system (mesenteric lymph node hemangioma and malignant granular lymphocytic leukemia) in Wistar, SD, and F344 rats; some increased, others decreased. Changes were not common but occurred more often in outbred Wistar and SD rats than in the inbred rat strain F344 rat.
This article is an update of Weber, Razinger, et al. (2011). The data refer mainly to compiled and published data from breeders and users and include the following strains: F344 (NTP 2010), Harlan Sprague-Dawley strain (Hsd: SD; Weber, Kaiser, and Klein 2012), Charles River, CD rat, Sprague-Dawley-derived (Crl: CD(SD); Charles River 2004), Crl: WI(Han) (Charles River 2011), and Wistar Hannover (Han)–derived strain, continued breeding by RCC Ltd., Switzerland, thereafter continued breeding by Harlan (RccHan™: WIST; Weber, Razinger, et al. 2011; Envigo 2016). The amount of published data for Hsd: SD rats is limited. There was a comparison of selected data published by Dinse et al. (2010) where the control data from 9 oncogenicity studies in Hsd: SD rats were compared to data from 9 oncogenicity studies in F344 rats. Another report dealt with selected data in Hsd: SD females from oncogenicity studies (Brix et al. 2005). There are published data from the NTP (2011), albeit restricted to data from 50 males and 52 females. These data are comparable with those of Hsd: SD rats presented by Weber, Kaiser, and Klein (2012), who used 120 animals per sex, and hence only these data were tabulated for this publication. When neoplastic lesions of Hsd: SD rats were compared with data of RccHan™: WIST rats, the main differences were higher incidences of mammary gland tumors, tumors of the parathyroid gland, pheochromocytomas, and tumors of the exocrine pancreas (Weber, Kaiser, and Klein 2012). In the following section, only tumor entities that were reported in any of the aforementioned strains at incidences of >0.5% in any gender are discussed.
Tumor Bearers
The number of tumors is lowest in Wistar rats. This is related mainly to myeloid leukemia and Leydig cell tumors in F344 rats, and the higher incidences of pituitary gland adenoma and mammary gland neoplasms in SD rats (Table 1).
Rate of Tumor Bearers Comparing F344 (National Toxicology Program 2010), Hsd: SD (Weber, Kaiser, and Klein 2012), and RccHan™: WIST Rats from Studies Performed at Harlan Laboratories Ltd., Switzerland, during 1981 to 2006 (Weber, Razinger, et al. 2011)—Total Incidence (%).

Note. F344 = Fisher 344; Hsd: SD = Harlan Sprague-Dawley strain; RccHan™: WIST = Wistar Hannover (Han)–derived strain, continued breeding by RCC Ltd., Switzerland, thereafter continued breeding by Harlan.
Liver
The liver is a major target organ due to its involvement in metabolism, and therefore might be considered one of the most sensitive organs in carcinogenicity studies. Nevertheless, the rate of spontaneous tumors including hepatocellular adenoma and carcinoma is not high in any of the strains discussed (Table 2) despite significant differences between different rat strains in metabolism, cytochrome P450 isoenzyme induction, and sensitivity to naturally occurring substances (Kacew and Festing 1996).
Comparison of Selected Liver Neoplasms from Different Rat Strains Including F344 (National Toxicology Program 2010), Crl: WI(Han) (Charles River 2011), Crl: CD(SD) (Charles River 2004), Hsd: SD (Weber, Kaiser, and Klein 2012), and RccHan™: WIST Rats from Studies Performed at Harlan Laboratories Ltd., Switzerland, during 1981 to 2012 (Weber, Razinger, et al. 2011; Envigo 2016)—Total Incidence (%).
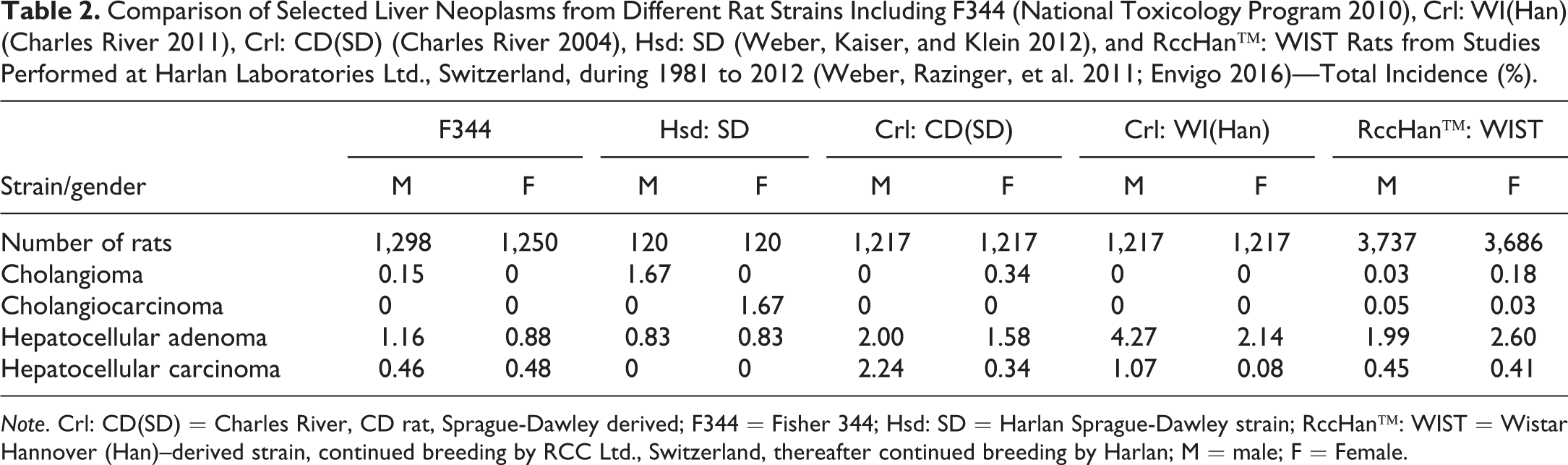
Note. Crl: CD(SD) = Charles River, CD rat, Sprague-Dawley derived; F344 = Fisher 344; Hsd: SD = Harlan Sprague-Dawley strain; RccHan™: WIST = Wistar Hannover (Han)–derived strain, continued breeding by RCC Ltd., Switzerland, thereafter continued breeding by Harlan; M = male; F = Female.
Pancreas
Endocrine tumors are among the most commonly recorded neoplastic lesions in oncogenicity studies (Bode et al. 1985; Bomhard 1992; Nakazawa et al. 2001).
Islet cell neoplasms are common in oncogenicity studies (Table 3). In some strains, the incidences of pancreatic neoplasms may reach high mean incidence values. These tumors are more often encountered in males than in females and published incidences in males are approximately 5% to 7.5%. The incidences are similar in all rat strains, except Crl: WI(Han) rats (Table 3), where the incidence was less than half of that in other strains. Many fewer neoplasms are recorded for the endocrine pancreas compared with islet cell tumor incidences (Table 4).
Comparison of Selected Pancreatic Neoplasms from Different Rat Strains Including F344 (National Toxicology Program 2010), Crl: WI(Han) (Charles River 2011), Crl: CD(SD) (Charles River 2004), Hsd: SD (Weber, Kaiser, and Klein 2012), and RccHan™: WIST Rats from Studies Performed at Harlan Laboratories Ltd., Switzerland, during 1981 to 2012 (Weber, Razinger, et al. 2011; Envigo 2016)—Total Incidence (%).
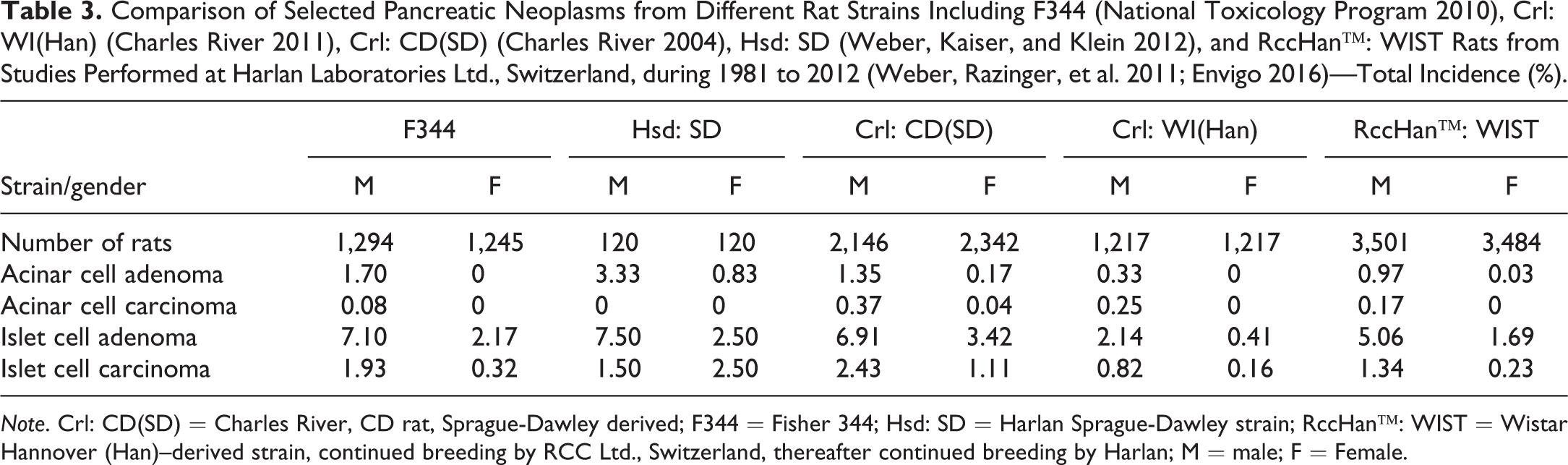
Note. Crl: CD(SD) = Charles River, CD rat, Sprague-Dawley derived; F344 = Fisher 344; Hsd: SD = Harlan Sprague-Dawley strain; RccHan™: WIST = Wistar Hannover (Han)–derived strain, continued breeding by RCC Ltd., Switzerland, thereafter continued breeding by Harlan; M = male; F = Female.
Comparison of Selected Skin Neoplasms (Mean Incidences >0.5% in One Sex, in Any Strain) from Different Rat Strains Including F344 (National Toxicology Program 2010), Crl: WI(Han) (Charles River 2011), Crl: CD(SD) (Charles River 2004), Hsd: SD (Weber, Kaiser, and Klein 2012), and RccHan™: WIST Rats from Studies Performed at Harlan Laboratories Ltd., Switzerland, during 1981 to 2012 (Weber, Razinger, et al. 2011; Envigo 2016)—Total Incidence (%).
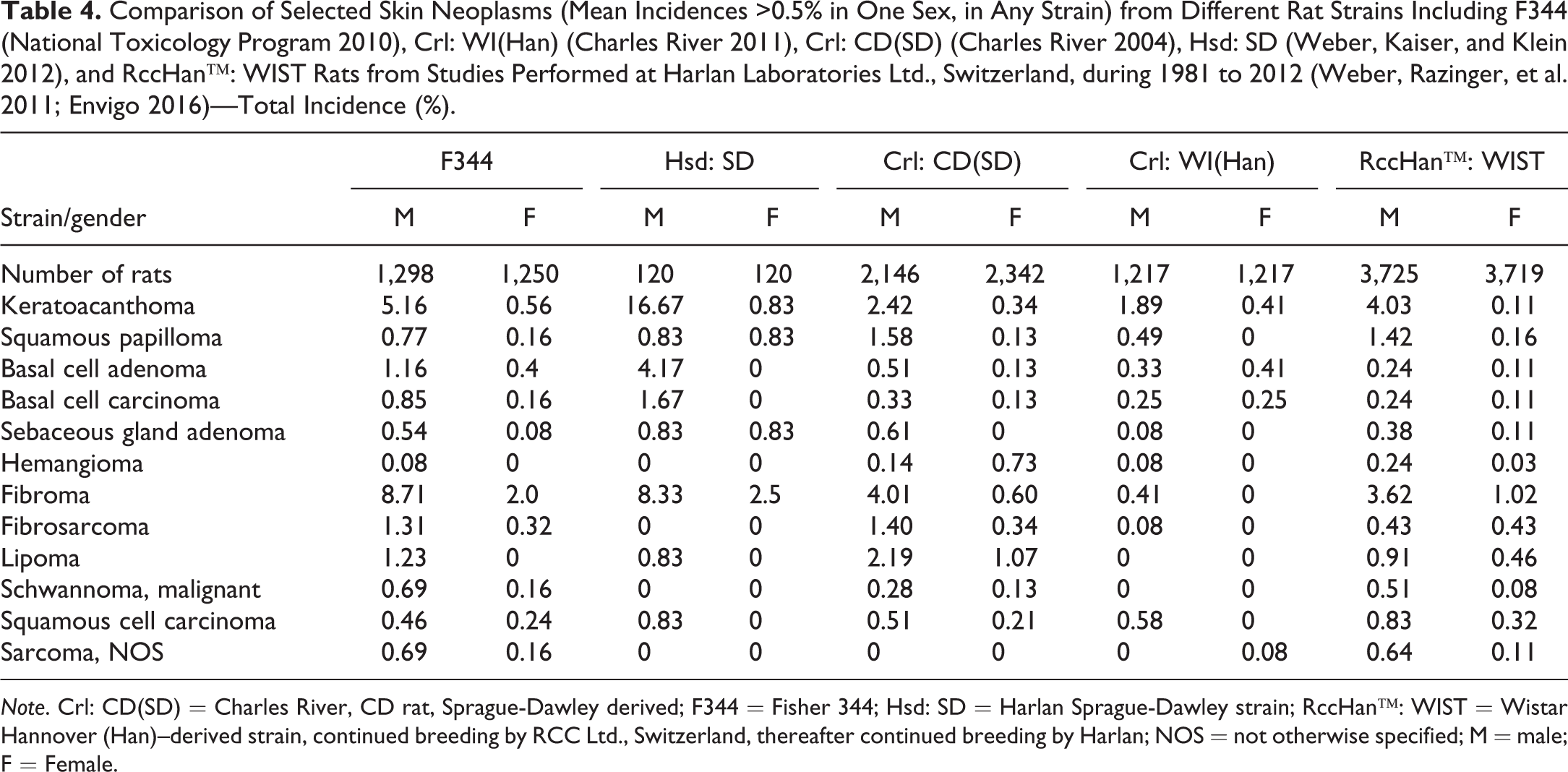
Note. Crl: CD(SD) = Charles River, CD rat, Sprague-Dawley derived; F344 = Fisher 344; Hsd: SD = Harlan Sprague-Dawley strain; RccHan™: WIST = Wistar Hannover (Han)–derived strain, continued breeding by RCC Ltd., Switzerland, thereafter continued breeding by Harlan; NOS = not otherwise specified; M = male; F = Female.
Skin
Skin neoplasms are often diagnosed in oncogenicity studies, although certain types of neoplasms are rarely encountered. Fibromas were the most commonly reported tumors of the skin/subcutis in control animals, with males significantly more affected than females. The highest rates were published for F344 and Hsd: SD rats. Wistar rats are in general less affected. Keratoacanthoma is another very common skin tumor in rats that also has a higher incidence in males of all strains. The data presented in Table 5 are consistent with the high incidences mentioned for keratoacanthomas by various authors for SD (M. Chandra, Riley, and Johnson 1992; Nakazawa et al. 2001) and Wistar (Poteracki and Walsh 1998) rats. Neoplasms that were reported at incidences >0.5% in any strain consist of squamous papilloma, basal cell adenoma and carcinoma, sebaceous gland adenoma, hemangioma, fibrosarcoma, lipoma, malignant schwannoma, squamous cell carcinoma, and sarcoma not otherwise specified (NOS).
Comparison of Selected Female Mammary Gland Neoplasms from Different Rat Strains Including F344 (National Toxicology Program 2010), Crl: WI(Han) (Charles River 2011), Crl: CD(SD) (Charles River 2004), Hsd: SD (Weber, Kaiser, and Klein 2012), and RccHan™: WIST Rats from Studies Performed at Harlan Laboratories Ltd., Switzerland, during 1981 to 2012 (Weber, Razinger, et al. 2011; Envigo 2016)—Total Incidence (%).

Note. Crl: CD(SD) = Charles River, CD rat, Sprague-Dawley derived; F344 = Fisher 344; Hsd: SD = Harlan Sprague-Dawley strain; RccHan™: WIST = Wistar Hannover (Han)–derived strain, continued breeding by RCC Ltd., Switzerland, thereafter continued breeding by Harlan.
Mammary Gland
Mammary gland tumors were recorded at week 23 in one Hsd: SD rat (Weber, Kaiser, and Klein 2012), and single cases were also recorded in 28-day (female, adenoma) and 13-week (female, fibroadenoma) studies in RccHan™: WIST rats (Weber 2012). These neoplasms were present as palpable masses. The size of growing tumors, even when benign, may cause an early sacrifice of the affected animals. In inhalation studies with SD rats that usually have higher body weights than Wistar strains, palpable masses may even be a limiting factor to placing the animals into the tubes. Mammary gland neoplasms are considered a major cause of morbidity/death (Weber et al. 2011; Table 6).
Comparison of Leydig Cell Tumor Incidences from Different Rat Strains Including F344 (National Toxicology Program 2010), Crl: WI(Han) (Charles River 2011), Crl: CD(SD) (Charles River 2004), Hsd: SD (Weber, Kaiser, and Klein 2012), and RccHan™: WIST Rats from Studies Performed at Harlan Laboratories Ltd., Switzerland, during 1981 to 2012 (Weber, Razinger, et al. 2011; Envigo 2016)—Total Incidence (%).
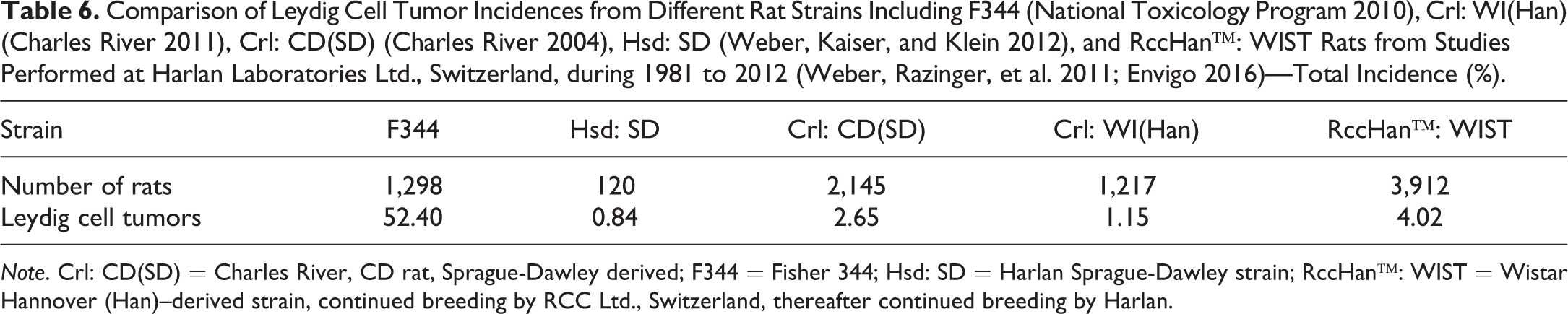
Note. Crl: CD(SD) = Charles River, CD rat, Sprague-Dawley derived; F344 = Fisher 344; Hsd: SD = Harlan Sprague-Dawley strain; RccHan™: WIST = Wistar Hannover (Han)–derived strain, continued breeding by RCC Ltd., Switzerland, thereafter continued breeding by Harlan.
In general, SD strains are by far more affected by mammary gland neoplasms (up to 72.5%, Weber 2012) than Wistar strains. Fibroadenoma constitutes the largest number of neoplasms. The data that have been presented for Wistar strains show a lower incidence of fibroadenoma in Crl: WI(Han) rats when compared to RccHan™: WIST rats. The incidence of adenocarcinoma, while higher in SD strains, varies dramatically between different SD strains/suppliers, with approximately 22% reported in Crl: CD(SD) rats (Charles River 2004) compared with 13% in Hsd: SD rats (Weber, Kaiser, and Klein 2012).
Other neoplasms are rarely encountered, with the exception of adenoma that is reported for Crl: CD(SD) rats at an incidence of approximately 7% (Charles River 2004).
Testes
The most common tumor in male F344 rats is the interstitial (Leydig) cell tumor at a mean incidence of 81% (Mitsumori and Elwell 1998; NTP 2010) but is also reported to reach almost 100% (Nolte et al. 2010; Table 7). It is deemed that compounds which cause interstitial cell proliferating lesions will not be recognized in carcinogenicity studies in F344 rats due to the high spontaneous incidences (Davis 1995). It was assumed that stress may affect both the incidence of Leydig cell and pituitary gland lesions. This was related mainly to individual caging that directly impairs testosterone synthesis and produces Leydig cell atrophy with a subsequent feedback increase in the synthesis of luteinizing hormone by the anterior pituitary. Leydig cells have specific receptors for glucocorticoids. The 11-beta-hydroxysteroid dehydrogenase inactivates glucocorticoids. However, this enzyme is depleted under chronic stress conditions. Therefore, glucocorticoids affect the steroid synthesis. The negative feedback may cause effects in the pituitary gland (Nyska et al. 1998).
Comparison of Selected Ovarian Neoplasms (Mean Incidences >0.5% in One Sex, in Any Strain) from Different Rat Strains Including F344 (National Toxicology Program 2010), Crl: WI(Han) (Charles River 2011), Crl: CD(SD) (Charles River 2004), Hsd: SD (Weber, Kaiser, and Klein 2012), and RccHan™: WIST Rats from Studies Performed at Harlan Laboratories Ltd., Switzerland, during 1981 to 2012 (Weber, Razinger, et al. 2011; Envigo 2016)—Total Incidence (%).
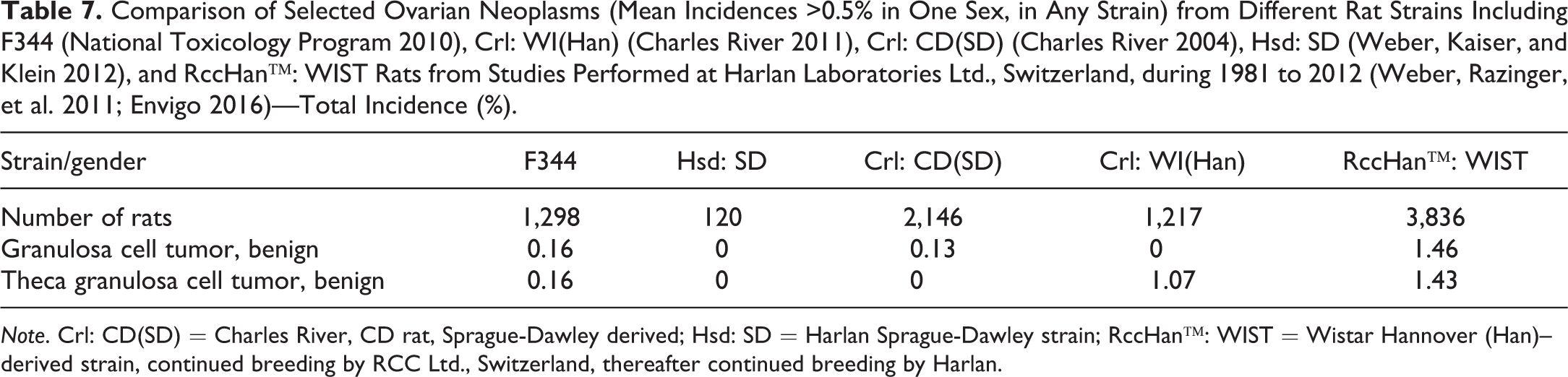
Note. Crl: CD(SD) = Charles River, CD rat, Sprague-Dawley derived; Hsd: SD = Harlan Sprague-Dawley strain; RccHan™: WIST = Wistar Hannover (Han)–derived strain, continued breeding by RCC Ltd., Switzerland, thereafter continued breeding by Harlan.
In other strains, much lower incidences have been reported from contract research organizations, that is, approximately 0.8% to 4% in SD and Wistar rats (Charles River 2004, 2011; Envigo 2016). Higher rates extracted from the Registry of Industrial Toxicology (RITA) database were reported by Nolte et al. (2010) as 13.7% in Wistar-derived rat, continued breeding by Unilever (Nolte et al. 2010). Most Leydig cell tumors are of a benign nature but may cause early sacrifice of the tumor bearers.
Female Reproductive Organs
There is also a large variety of tumor entities in female reproductive organs. No neoplastic lesion is common in ovaries. Incidences at >0.5% are only reported for granulosa cell tumors or theca granulosa cell tumors, and the highest incidences are noted in Wistar strains (Table 8).
Comparison of Selected Uterine Neoplasms (Mean Incidences >0.5% in One Sex, in Any Strain) from Different Rat Strains Including F344 (National Toxicology Program 2010), Crl: WI(Han) (Charles River 2011), Crl: CD(SD) (Charles River 2004), Hsd: SD (Weber, Kaiser, and Klein 2012), and RccHan™: WIST Rats from Studies Performed at Harlan Laboratories Ltd., Switzerland, during 1981 to 2012 (Weber, Razinger, et al. 2011; Envigo 2016)—Total Incidence (%).
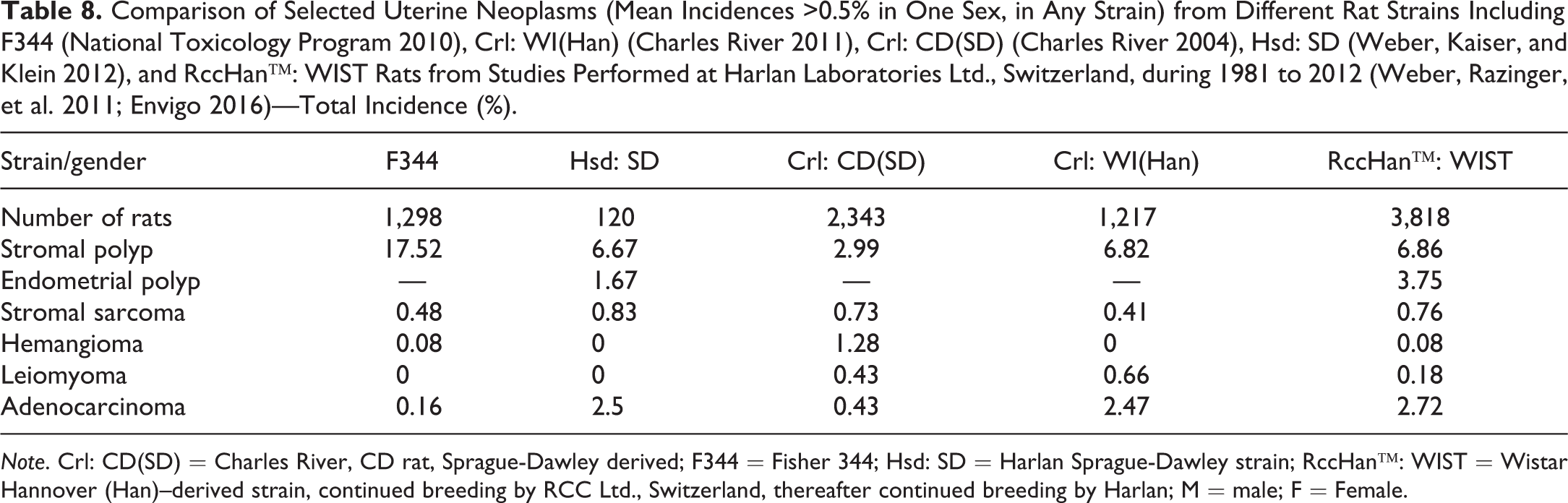
Note. Crl: CD(SD) = Charles River, CD rat, Sprague-Dawley derived; F344 = Fisher 344; Hsd: SD = Harlan Sprague-Dawley strain; RccHan™: WIST = Wistar Hannover (Han)–derived strain, continued breeding by RCC Ltd., Switzerland, thereafter continued breeding by Harlan; M = male; F = Female.
The most common tumor in the uterus is the stromal polyp, with the highest incidence in F344 rats (approximately 17%), followed by endometrial polyps reported for Hsd: SD and RccHan™: WIST rats only. Adenocarcinoma was noted at approximate incidences of 2.5% in Wistar and Hsd: SD rats. Hemangioma was reported at incidences above 1% in Crl: CD(SD) rats. Stromal sarcoma and leiomyoma were noted also at incidences >0.5% (Table 9). All other neoplasms are rarely encountered in any strain.
Comparison of Selected Neoplasms of the Pituitary Gland from Different Rat Strains Including F344 (National Toxicology Program 2010), Crl: WI(Han) (Charles River 2011), Crl: CD(SD) (Charles River 2004), Hsd: SD (Weber, Kaiser, and Klein 2012), and RccHan™: WIST Rats from Studies Performed at Harlan Laboratories Ltd., Switzerland, during 1981 to 2012 (Weber, Razinger, et al. 2011; Envigo 2016)—Total Incidence (%).
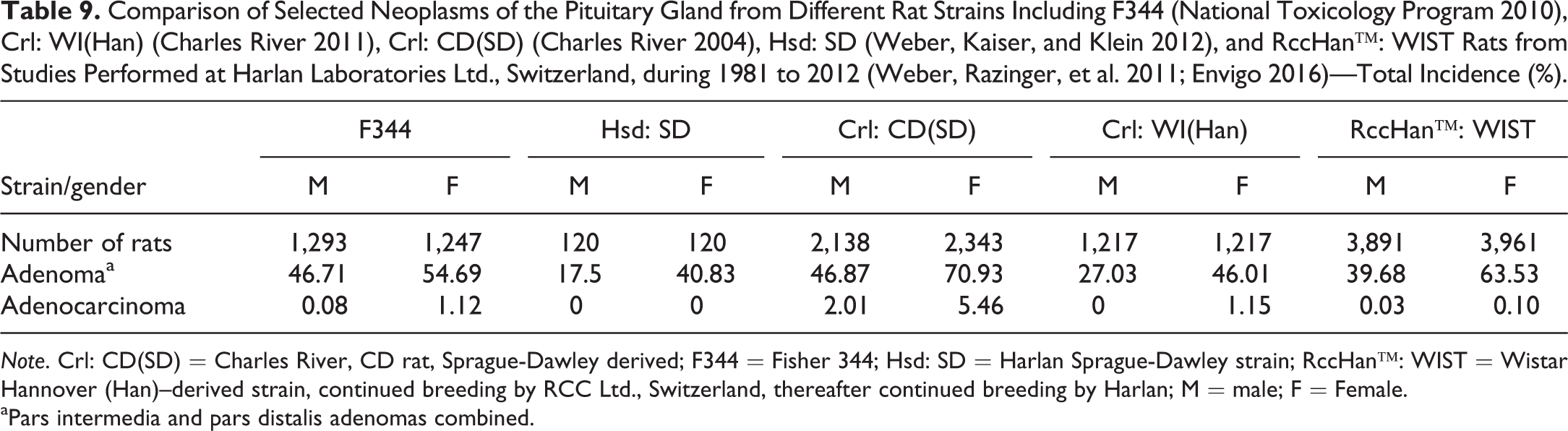
Note. Crl: CD(SD) = Charles River, CD rat, Sprague-Dawley derived; F344 = Fisher 344; Hsd: SD = Harlan Sprague-Dawley strain; RccHan™: WIST = Wistar Hannover (Han)–derived strain, continued breeding by RCC Ltd., Switzerland, thereafter continued breeding by Harlan; M = male; F = Female.
aPars intermedia and pars distalis adenomas combined.
Pituitary
In Table 4, adenomas of the pars intermedia and anterior are combined due to the fact that several pathologists did not differentiate between these tumor types. They diagnosed “adenoma” without modifier. However, the number of pars intermedia adenomas is extremely low. The data obtained from most commonly used rat strains reveal the highest incidences of pituitary gland adenomas in Crl: CD(SD) females (approximately 71%), followed by female RccHan™: WIST (approximately 64%). In contrast, in Hsd: SD rats, the incidence of pituitary adenomas were lowest. Here, the smaller sample number may be a possible cause for such a difference between SD strains. Except in F344 rats, there is a huge difference between sexes, with females affected almost twice as often as males. Adenocarcinomas are rarely diagnosed and that may be due to the difficulty of differentiating between metastases and compression of larger tumors. In one RccHan™: WIST rat male, the diagnosis of adenocarcinoma of the pars distalis was made (Envigo 2016).
Thyroid Glands
The thyroid gland is an important target organ in carcinogenicity studies due to the inducibility of follicular tumors secondary to liver changes (Alison, Capen, and Prentice 1994).
Thyroid gland follicular tumors were recorded at increasing differences in males of the Alderley Park strain (a Wistar-derived strain) over decades (Tucker 1997). Therefore, it is not unusual that the highest incidences of follicular tumors are recorded in male Wistar rats. Maximal incidences of adenomas in control animals were 12% for RccHan™: WIST and approximately 12% for Crl: WI(Han) rats. Follicular carcinomas were reported at maximal control incidences of approximately 9% for RccHan™: WIST rats and 6% for Crl: WI(Han) rats.
For C-cell tumors, there were no significant differences between Wistar strains and SD strains. F344 rats have a higher incidence of this tumor especially in males (Table 10).
Comparison of Selected Thyroid Gland Neoplasms from Different Rat Strains Including F344 (National Toxicology Program 2010), Crl: WI(Han) (Charles River 2011), Crl: CD(SD) (Charles River 2004), Hsd: SD (Weber, Kaiser, and Klein 2012), and RccHan™: WIST Rats from Studies Performed at Harlan Laboratories Ltd., Switzerland, during 1981 to 2012 (Weber, Razinger, et al. 2011; Envigo 2016)—Total Incidence (%).
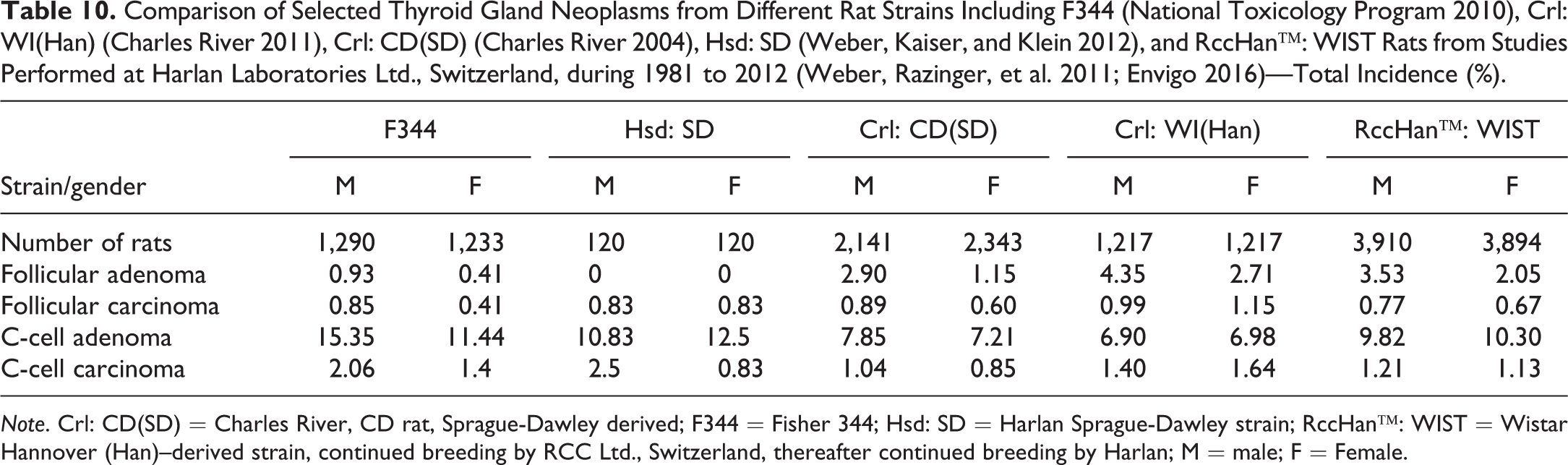
Note. Crl: CD(SD) = Charles River, CD rat, Sprague-Dawley derived; F344 = Fisher 344; Hsd: SD = Harlan Sprague-Dawley strain; RccHan™: WIST = Wistar Hannover (Han)–derived strain, continued breeding by RCC Ltd., Switzerland, thereafter continued breeding by Harlan; M = male; F = Female.
Adrenal Glands
The most common tumor of the adrenal gland is the benign pheochromocytoma. The incidence is much higher in SD and F344 rats than in Wistar strains. Males are more affected than females. This is considered related to a higher incidence and severity of chronic progressive nephropathy (Nyska et al. 1999). Regarding cortical tumors (adenomas and carcinomas combined), SD rats are more affected than other rat strains (Table 11). In general, malignant tumors are not often encountered. Very rarely, complex pheochromocytomas or ganglioneuromas are reported.
Comparison of Selected Adrenal Gland Neoplasms from Different Rat Strains Including F344 (National Toxicology Program 2010), Crl: WI(Han) (Charles River 2011), Crl: CD(SD) (Charles River 2004), Hsd: SD (Weber, Kaiser, and Klein 2012), and RccHan™: WIST Rats from Studies Performed at Harlan Laboratories Ltd., Switzerland, during 1981 to 2012 (Weber, Razinger, et al. 2011; Envigo 2016)—Total Incidence (%).
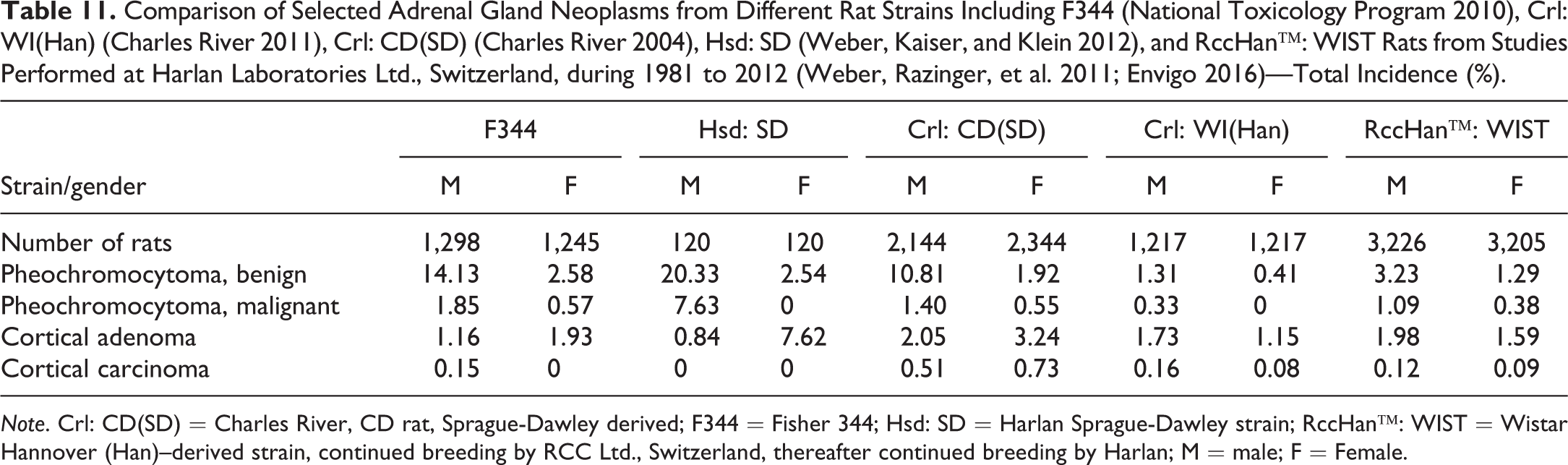
Note. Crl: CD(SD) = Charles River, CD rat, Sprague-Dawley derived; F344 = Fisher 344; Hsd: SD = Harlan Sprague-Dawley strain; RccHan™: WIST = Wistar Hannover (Han)–derived strain, continued breeding by RCC Ltd., Switzerland, thereafter continued breeding by Harlan; M = male; F = Female.
Mesenteric Lymph Nodes
In general, the spontaneous rate of mesenteric lymph node angiomas (benign vascular neoplasms including lymphangioma and hemangioma) ranges from rare to common depending on the strain used, and higher incidences are recorded in males (Radi and Morton 2012). In the presented data, the strain with the highest incidence of hemangioma and hemangiosarcomas is the RccHan™: WIST rat (Table 12). In lymph nodes other than the mesenteric lymph nodes, the tumor incidences were below 1%. In SD strains, F344 rats, and also for Crl: WI(Han), the lesions in mesenteric lymph nodes are not reported separately, as findings in all lymph nodes were combined, likely due to the lower incidences in these strains (Charles River 2004, 2010; Envigo 2016; Zwicker et al. 1992).
Comparison of Vascular Tumor Incidences of the Mesenteric Lymph Nodes from Different Rat Strains Including F344 (National Toxicology Program 2010), Crl: WI(Han) (Charles River 2011), Crl: CD(SD) (Charles River 2004), Hsd: SD (Weber, Kaiser, and Klein 2012), and RccHan™: WIST Rats from Studies Performed at Harlan Laboratories Ltd., Switzerland, during 1981 to 2012 (Weber, Razinger, et al. 2011; Envigo 2016)—Total Incidence (%).
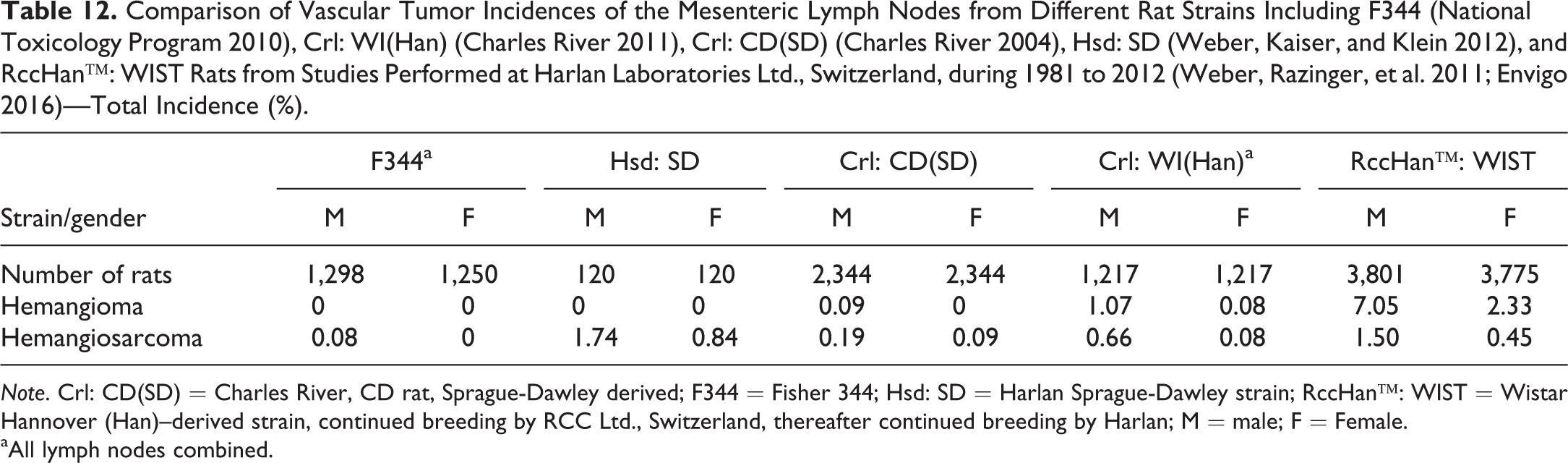
Note. Crl: CD(SD) = Charles River, CD rat, Sprague-Dawley derived; F344 = Fisher 344; Hsd: SD = Harlan Sprague-Dawley strain; RccHan™: WIST = Wistar Hannover (Han)–derived strain, continued breeding by RCC Ltd., Switzerland, thereafter continued breeding by Harlan; M = male; F = Female.
aAll lymph nodes combined.
Thymoma
Thymoma is a typical Wistar-related tumor, with females more affected than males (Table 13). There are different morphological expressions, with variants ranging from mainly lymphocytic to epithelioid cell compositions. Most common expressions are benign thymomas with mainly lymphocytic cell composition.
Comparison of Incidences on Thymoma from Different Rat Strains Including F344 (National Toxicology Program 2010), Crl: WI(Han) (Charles River 2011), Crl: CD(SD) (Charles River 2004), Hsd: SD (Weber, Kaiser, and Klein 2012), and RccHan™: WIST Rats from Studies Performed at Harlan Laboratories Ltd., Switzerland, during 1981 to 2012 (Weber, Razinger, et al. 2011; Envigo 2016)—Total Incidence (%).
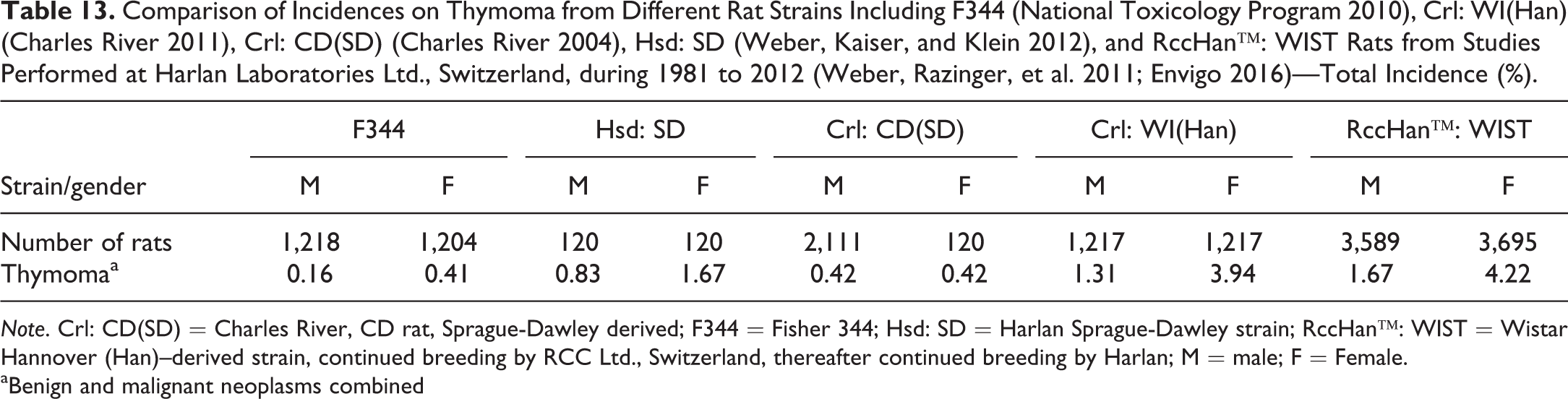
Note. Crl: CD(SD) = Charles River, CD rat, Sprague-Dawley derived; F344 = Fisher 344; Hsd: SD = Harlan Sprague-Dawley strain; RccHan™: WIST = Wistar Hannover (Han)–derived strain, continued breeding by RCC Ltd., Switzerland, thereafter continued breeding by Harlan; M = male; F = Female.
aBenign and malignant neoplasms combined
Systemic Neoplasia
Among myeloid leukemia, malignant lymphoma, and histiocytic sarcoma, the most common systemic neoplasia in rats is chronic myeloid leukemia (LGL leukemia) in F344 rats, which has been recorded at mean incidences of over 38% in male and over 21% in female F344 rats. The remaining systemic neoplastic changes are recorded below a mean incidence of 4% in any rat strain (Table 14). Systemic neoplasia involves many, if not all, organs of an animal. An uncommonly high incidence of histiocytic sarcomas was reported from BDII/Han inbred rats (12.5%) by Kaspareit-Rittinghausen and Deerberg (1988). The highest incidence of this tumor was found in the subcutis. Other organs frequently involved were lungs and liver.
Comparison of Incidences on Systemic Neoplasia from Different Rat Strains Including F344 (National Toxicology Program 2010), Crl: WI(Han) (Charles River 2011), Crl: CD(SD) (Charles River 2004), Hsd: SD (Weber, Kaiser, and Klein 2012), and RccHan™: WIST Rats from Studies Performed at Harlan Laboratories Ltd., Switzerland, during 1981 to 2012 (Weber, Razinger, et al. 2011; Envigo 2016)—Total Incidence (%).
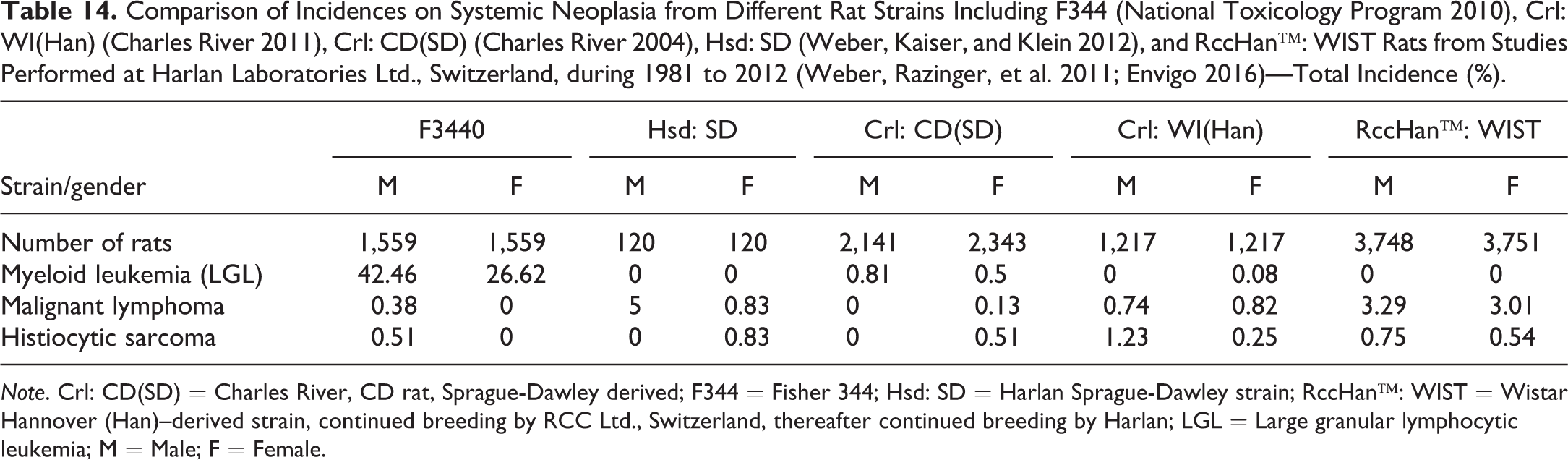
Note. Crl: CD(SD) = Charles River, CD rat, Sprague-Dawley derived; F344 = Fisher 344; Hsd: SD = Harlan Sprague-Dawley strain; RccHan™: WIST = Wistar Hannover (Han)–derived strain, continued breeding by RCC Ltd., Switzerland, thereafter continued breeding by Harlan; LGL = Large granular lymphocytic leukemia; M = Male; F = Female.
Brain
The incidence of neural neoplasms in rats varies with strains and sex but is also dependent on the number of sections made per organ (Weber, Garman et al. 2011). The granular cell tumor is the most common neoplasm of the central nervous system. The highest incidences are recorded in RccHan™: WIST rats. Males of all strains are more affected than females. In contrast, astrocytoma was reported as the brain tumor with the highest incidence in Crl: CD(SD) (Table 15).
Comparison of Selected Brain Neoplasms (Mean Incidences >0.5% in One Sex, in any Strain) from Different Rat Strains Including F344 (National Toxicology Program 2010), Crl: WI(Han) (Charles River 2011), Crl: CD(SD) (Charles River 2004), Hsd: SD (Weber, Kaiser, and Klein 2012), and RccHan™: WIST Rats from Studies Performed at Harlan Laboratories Ltd., Switzerland, during 1981 to 2012 (Weber, Razinger, et al. 2011; Envigo 2016)—Total Incidence (%).
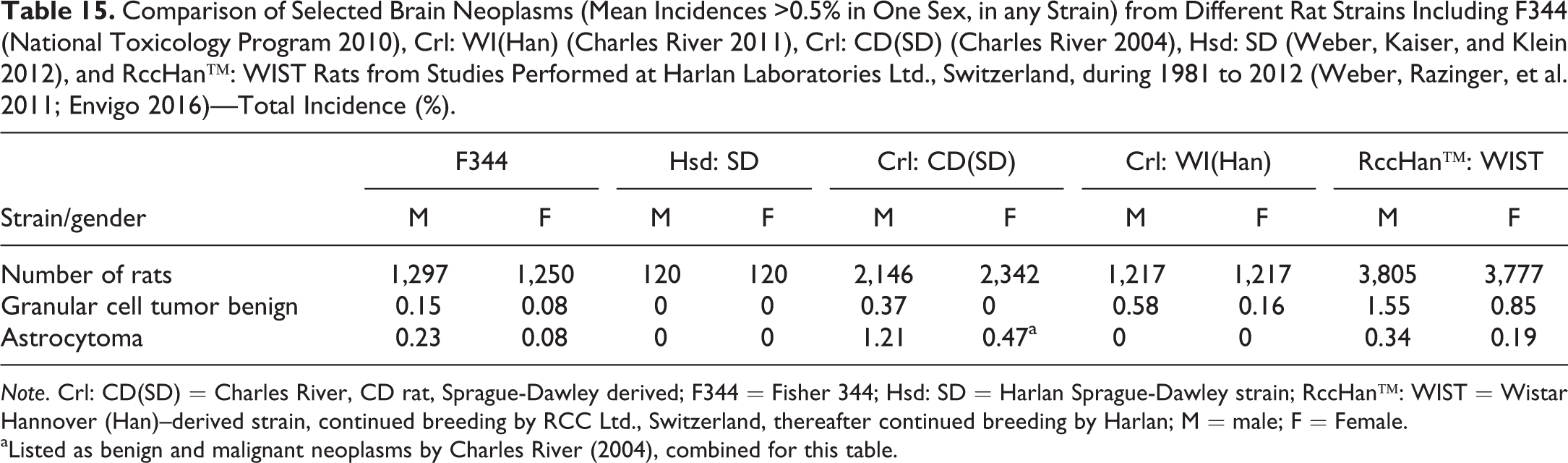
Note. Crl: CD(SD) = Charles River, CD rat, Sprague-Dawley derived; F344 = Fisher 344; Hsd: SD = Harlan Sprague-Dawley strain; RccHan™: WIST = Wistar Hannover (Han)–derived strain, continued breeding by RCC Ltd., Switzerland, thereafter continued breeding by Harlan; M = male; F = Female.
aListed as benign and malignant neoplasms by Charles River (2004), combined for this table.
Neoplasms in Other Organs
The following neoplasms were published at incidences >0.5% but below 1% in any strain (F344, Hsd: SD, Crl: CD(SD), Crl: WI(Han), and RccHan−: WIST) or sex (Table 16). In all strains, Zymbal gland carcinoma has been recorded, except in female Wistar rats. Oral cavity papillomas or squamous cell carcinomas were noted in all rat strains except Hsd: SD females, Crl: WI(Han) males, or RccHan™: WIST females. Adenocarcinoma of the parotid gland was observed in RccHan™: WIST rats only. Vaginal polyps were noted in F344 and Wistar strains. A number of neoplasms at incidences >0.5% were reported in the body cavities, including lipomas at high incidences in RccHan™: WIST rats. Mesothelioma was noted in all rat strains (except Crl: WI(Han) rats) with a highest incidence in RccHan™: WIST rats. Other neoplasms that are not unusual in the body cavities are malignant schwannomas, hemangiosarcomas, and sarcoma NOS.
Comparison of Selected Neoplasms of Other Organs (Mean Incidences >0.5% in One Sex, in any Strain) from Different Rat Strains Including F344 (National Toxicology Program 2010), Crl: WI(Han) (Charles River 2011), Crl: CD(SD) (Charles River 2004), Hsd: SD (Weber, Kaiser, and Klein 2012), and RccHan™: WIST Rats from Studies Performed at Harlan Laboratories Ltd., Switzerland, during 1981 to 2012 (Weber, Razinger, et al. 2011; Envigo 2016)— Total Incidence (%).
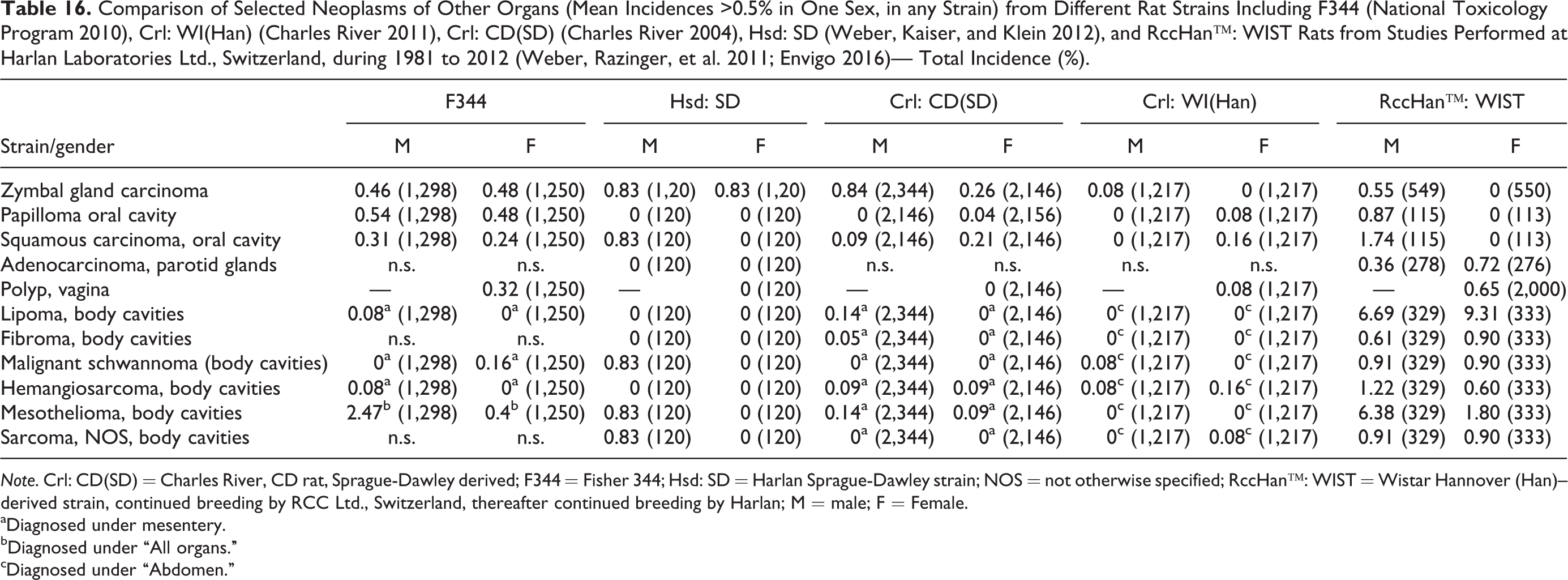
Note. Crl: CD(SD) = Charles River, CD rat, Sprague-Dawley derived; F344 = Fisher 344; Hsd: SD = Harlan Sprague-Dawley strain; NOS = not otherwise specified; RccHan™: WIST = Wistar Hannover (Han)–derived strain, continued breeding by RCC Ltd., Switzerland, thereafter continued breeding by Harlan; M = male; F = Female.
aDiagnosed under mesentery.
bDiagnosed under “All organs.”
cDiagnosed under “Abdomen.”
Neoplasms in Young Adult Rats
Neoplasms are rare in young adult animals but in toxicity studies they may cause discussions on possible early inductions. Not many data have been published, but most are available on SD rats, for example, splenic hemangiosarcomas in a 9-week-old SD rat (Shiraki et al. 2012), malignant lymphoma in a 19-week-old Crl: CD(SD) rat (Matsushima et al. 2010), rhabdomyosarcoma in an 8-week-old SD rat (Chang et al. 2008), carcinoma of the parotid gland in a 6-week-old SD rat (Tsunenari, Yamate, and Sakuma 1997), or malignant Sertoli cell tumor in the ovary of a 19-week-old female SD rat (Kinoshita et al. 2016). A survey on the occurrence of amphophilic-vacuolar phenotypic variant in young SD rats was provided by Crabbs et al. (2013). This tumor variant was reported more commonly than the conventional renal tubule tumors. They were recorded in rats at ages of 7 to 10 weeks. Females were perhaps more affected than males (approximately 1% vs. 0.4%).
A compilation of data on young SD rats was provided by Ikezaki, Takagi, and Tamura (2011). They included data from 4- (782 males and 770 females), 13- (857 males and 878 females), and 26-week toxicity studies (842 males and 874 females) conducted at Bozo Research Center Inc., from 2000 to 2009. The first neoplasms were observed in 19-week-old animals and included anterior adenoma of the pituitary, follicular cell adenocarcinoma and C cell adenoma of the thyroids, nephroblastoma of the kidneys, basal cell tumor of the skin, and malignant lymphoma. In 32-week-old animals, hemangiosarcoma of the tongue, adenocarcinoma of the submandibular glands, histiocytic sarcoma of the spleen, oligodendroglioma of the brain, and adenocarcinoma and fibroadenoma of the mammary glands were recorded. The highest incidences were found at an age of 32 weeks for mammary adenocarcinoma (0.7%) and pituitary anterior adenoma (0.3%). At this age, there a few oligodendrogliomas as well as mammary gland fibroadenomas. All other cases were single incidences.
Less information was published on neoplasms in young adult Wistar rats, for example, intracranial malignant teratoma in a 91-day-old male Wistar rat (Reindel et al. 1996). An attempt was made by Son et al. (2010) to describe the early occurrence of neoplasms using data from early decedents out of 10 two-week, 34 four-week, 36 thirteen-week, and 8 twenty-six-week studies, as well as from 29 carcinogenicity studies performed between 1995 and 2009 at Huntingdon Life Sciences, UK. Between weeks 16 and 26, there were 3 cases of malignant lymphoma. During this period, there was in addition one case of fibrosarcoma (skin/skeletal muscle) and one renal tubule carcinoma.
A similar approach was made by Weber (2012) on the occurrence of spontaneous neoplasms in 2-, 4-, and 13-week studies (Table 17). The most commonly affected organ was the kidney (renal tubular adenoma and adenocarcinoma, nephroblastoma), and these tumors were detected in all study types but with increased incidences in 4- and 13-week studies. Malignant lymphoma was recorded in 13-week studies with higher incidences in females. At this period, there were also 2 pituitary adenomas (pars anterior) and 2 fibroadenomas in the mammary gland diagnosed in females. All other tumors were single incidences, including oligodendroglioma, C-cell adenoma, adrenal medulla ganglioneuroma, ameloblastoma (tooth), salivary gland carcinoma, tongue hemangiosarcoma, colon leiomyoma, rectum adenocarcinoma, dermal basal cell tumor mammary gland adenoma, thymic squamous carcinoma, and mesenteric lymph nodes: hemangioma, uterine stromal polyp, and malignant schwannoma in the abdominal cavity.
Comparison of Incidences on Neoplastic Lesions Related to Organ Systems Recorded in 2-, 4-, and 13-Week Studies Performed with RccHan™: WIST Rats at Harlan Laboratories Ltd., Switzerland, during 1981 to 2012 (Weber 2012)—Total Incidence (%).
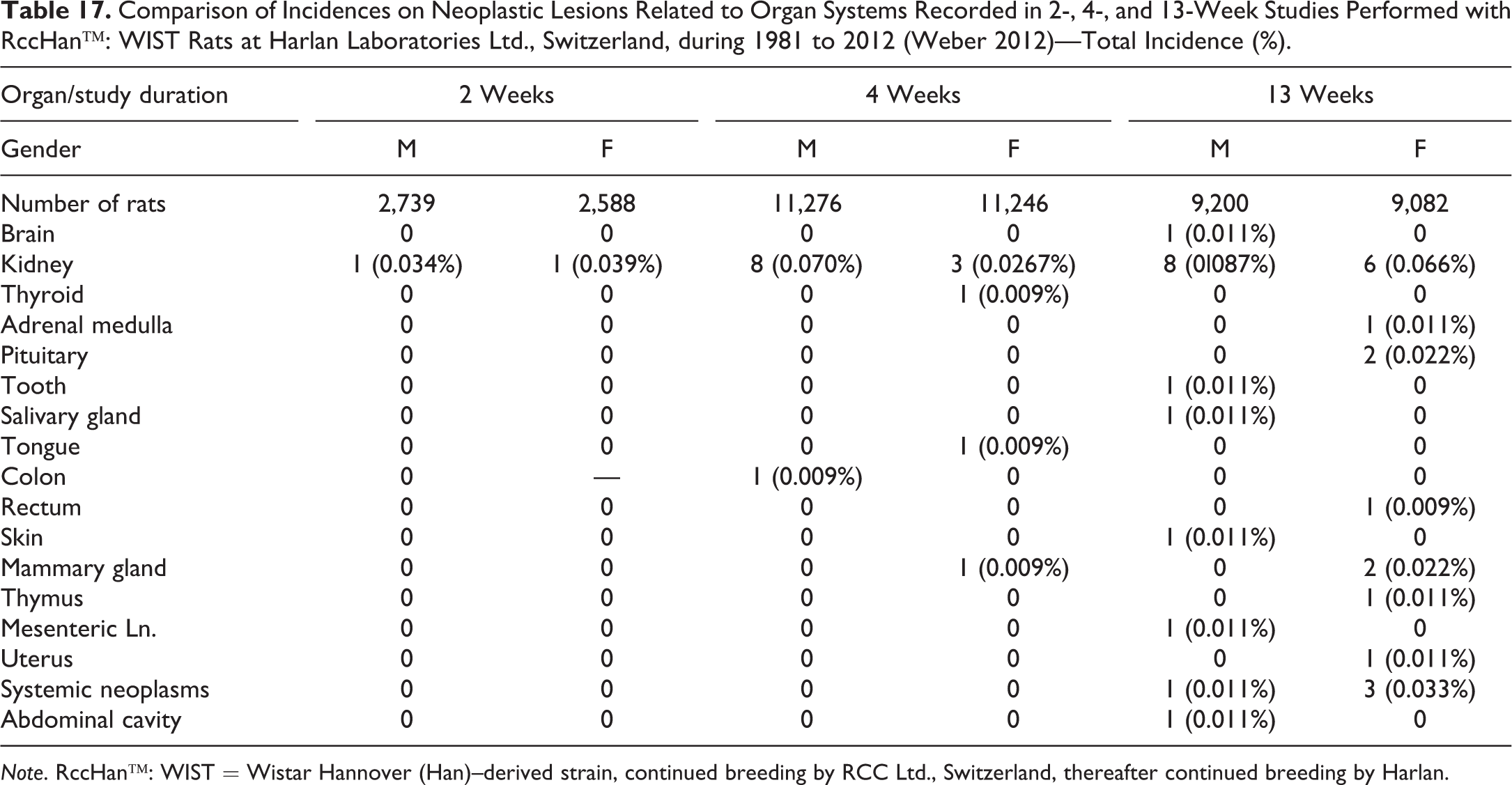
Note. RccHan™: WIST = Wistar Hannover (Han)–derived strain, continued breeding by RCC Ltd., Switzerland, thereafter continued breeding by Harlan.
Summary
A significant amount of published data, provided either by breeders or pathologists, is available on the incidence of neoplastic lesions in different rat strains. There are so many different kinds of rats, however, that the data can be difficult to generalize. The main rat models in use are SD and Wistar rats, so a summary of neoplastic incidences should concentrate in the first instance on these strains. The F344 rat is of lesser significance.
Furthermore, it should be considered that SD and Wistar rats are delivered by different breeders that are breeding these animals for many generations. In this case, differences between substrains (e.g., Hsd: SD and Crl: CD(SD)) and even breeding colonies at different locations within a substrain may cause differences in control data. “Genetic drift cannot be prevented; however, breeding strategies such as randomization of breeder selection can aid in delaying this inescapable event. Moreover, to ensure that the variable genetic profile of an outbred model is sustained, the breeding scheme of an outbred colony should be optimized to avoid inbreeding by mating unrelated animals only…” (Weber et al. 2011). However, a common trend in tumor incidences (and also body weight, mortality, breeding index, etc.) may apply to a specific strain including the substrains.
The tumor incidence in SD rats is higher than in Wistar rats. This is related mainly to the higher incidences of pituitary gland adenomas (in females, up to approximately 70% in SD rats vs. 64% in Wistar rats) and mammary gland neoplasms, especially the fibroadenoma (in females, up to approximately 72% in SD rats vs. 22.3% in Wistar rats). The highest incidences of adenocarcinomas of the mammary glands were reported in Crl: CD(SD) rats (22.2%), and adenomas were reported in this rat strain at incidences of 6.92%.
High incidences of neoplasms are expected in endocrine organs. Besides pituitary gland adenomas, islet cell tumors in males reach mean incidences of approximately 7%, and benign pheocromocytomas approximately 20%, again with higher incidences in SD rats. Also, cortical tumors (adenoma and adenocarcinoma) were recorded at higher mean incidences in SD rats but in females (up to 7.6%).
In contrast, thyroid follicular tumors are recorded at higher mean incidences in Wistar strains (up to approximately 5% vs. 4% in SD rats). For C-cell tumors, the mean incidences are very variable and are reported as 6.9% to 10.83% throughout Wistar and SD rats.
Vascular neoplasms in lymph nodes are considered to be Wistar related, for example, in RccHan™: WIST, the total mean incidence for hemangioma and hemangiosarcomas is reported at approximately 9% that is exceeding the incidences in other rat strains by far. This is also true for thymoma that is recorded in Wistar females up to 4%.
The most common dermal tumor is fibroma with highest mean incidence in SD rats (up to 8%) followed by keratoacanthoma up to 17%, again in SD rats.
In the reproductive organs, stromal polyps are common in the uterus throughout all strains (between 3% and 7%). Adenocarcinomas of the uterus are much more often recorded in Wistar rats compared to SD rats, at mean incidences up to approximately 3%. Leydig cell tumors in the strains discussed have been recorded up to 4% in Wistar rats versus 3% in SD rats, but even higher incidences have been reported for Wistar rats (Nolte et al. 2010).
All other tumors appeared to have mean incidences of 2% and lower. Generally, benign tumors are more often diagnosed. Regarding the most common tumors, the highest incidences in females are for pituitary gland adenomas, mammary gland neoplasms, adrenal cortical neoplasms, thymomas, and dermal neoplasms; and in males, the highest incidences are for islet cell tumors, pheochromocytomas, and vascular tumors.
Rare tumors are considered to be rare in all important rat strains/substrains (e.g., chordoma, craniopharyngioma, paragangliomas, etc.). Occasionally, there are reports on rarely encountered tumors that suddenly appear at high incidences, for example, hibernoma at incidences of up to 12% in control animals (Bruner et al. 2009; S. Chandra et al. 2008). These, however, are notable exceptions.
In young adult rats (studies up to 26 weeks), the data on tumor incidences are limited. However, there are several case reports or organ-related summaries published (e.g., Chang et al. 2008; Crabbs et al. 2013; Matsushima et al. 2010; Reindel et al. 1996; Shiraki et al. 2012; Tsunenari, Yamate, and Sakuma 1997), and summary data are available in the literature (e.g., Ikezaki, Takagi, and Tamura 2011; Lanzoni et al. 2007; Son et al. 2010). Tumors may be encountered in animals as young as 9 weeks. Malignant lymphomas and renal tumors (especially the amphophilic-vacuolar renal tubule tumor in SD rats and the renal tubular adenoma and nephroblastoma in Wistar rats) are not unusual in young adult animals.
Footnotes
Author Contribution
All authors (K.W.) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
