Abstract
The microminipig has considerable potential as an animal model to evaluate general toxicity; however, there are few studies on the male reproductive system, particularly regarding spermatogenesis. The objectives of the present study were to clarify the stages of the seminiferous epithelium cycle on the basis of spermiogenesis and to determine the duration of spermatogenesis in the microminipig. Eleven microminipigs from 6 to 9 months of age were used for histological analyses. Spermiogenesis and stages of the seminiferous epithelium cycle were classified according to the degree of acrosomal development as shown by the periodic acid-Schiff reaction. Three of the animals were intravenously injected with 5-bromo-2-deoxyuridine to determine the duration of spermatogenesis by immunohistochemistry. Spermiogenesis was classified into 15 steps according to the morphological development of the acrosome, nucleus, and flagellum. The seminiferous epithelium cycle was classified into 11 stages based on the steps of spermatid development and germ cell associations. The length of the seminiferous epithelium cycle and the overall spermatogenesis process in the microminipig were estimated to be approximately 9.1 and 40.9 days, respectively. The results indicate the potential application of the microminipig in the evaluation of testicular toxicity, such as spermatogenesis disruption, in general toxicity studies.
Introduction
The microminipig (Fuji Micra Inc., Shizuoka, Japan) has been used as an animal model for various experimental applications, such as understanding the pathogenesis of disease (Miyoshi et al. 2010; Kawaguchi et al. 2011; Sugiyama et al. 2011; Yamaguchi et al. 2014; Kawaguchi et al. 2014); in pharmacologic (Ohno et al. 2015), toxicologic (Yoshikawa et al. 2013), and pharmacokinetic studies (Murayama et al. 2009; Mogi et al. 2012; Higashi et al. 2014; Guruge et al. 2015); and in regeneration medicine (Nakagawa et al. 2015; Murata et al. 2015). Their body weight is less than 10 kg at 6 months of age, whereas minipigs weigh from 15 to 20 kg at the same age. Therefore, microminipigs are suitable for studies with limited amounts of test substances and housing spaces. Furthermore, based on animal welfare, the microminipig is one of the promising candidates for nonrodent animal models particularly for drug discovery research and preclinical toxicity studies (Kaneko et al. 2011). Despite its considerable potential as a laboratory animal, there are limited studies in the literature regarding the male reproductive organs of the microminipig, particularly those aspects related to spermatogenesis, which are one of the most relevant chemical targets.
In pharmaceutical studies, almost regardless of the cellular target within the reproductive system, the most common morphological consequence of toxicity is spermatogenic disturbance. Spermatogenesis is a complex biological process involving cellular transformations that produce male haploid germ cells from diploid spermatogonial stem cells (Hess and Renato de Franca 2008). It is, therefore, important to understand the process and dynamics of spermatogenesis in the studied species (Lanning et al. 2002). In the minipigs in general, however, the different spermatogenesis stages are not fully characterized, in contrast to rats, dogs, or primates (Bode et al. 2010).
Two techniques have been well established for the study of spermatogenesis. The first technique relies on the morphologic changes of the acrosome in the developing spermatids using the periodic acid-Schiff (PAS) reaction. Using this approach, a number of steps during spermiogenesis, namely, spermatid development from round spermatids to spermatozoa, could be defined, which provides the basis for the identification of the stages of the seminiferous epithelium cycle (Clermont and Leblond 1955; Amann and Schanbacher 1983). The second technique involves radiolabeling or immunodetection of DNA during its synthesis in immature primary spermatocytes using tritiated thymidine or bromodeoxyuridine (BrdU) labeling, which enables the time course of spermatogenesis and the pattern of spermatogonial divisions to be ascertained (Amann and Schanbacher 1983; Nakai, Van Cleeff, and Bahr 2004; Choudhuri et al. 2005).
The objectives of the present study were to identify the stages of the seminiferous epithelium cycle and to determine the duration of the spermatogenesis in the microminipig by a thorough histological and immunohistochemical evaluation of the testis.
Materials and Methods
Animals
Eleven microminipigs from 6 to 9 months of age, considered as young adult (Kaneko et al. 2011), were used for this study. The animals were born and raised in a closed breeding colony at the Shizuoka Prefectural Research Institute of Animal Industry, Swine and Poultry Research Center (Shizuoka, Japan), which Fuji Micra Inc. had provided with microminipigs for the purpose of research. All animals were housed individually or in pairs in pens of at least 0.54 m2/animal under conventional condition with a 12-hr light–dark cycle. The animals were weaned at 4 weeks of age and then fed a commercial diet (Marubeni Nisshin Feed Co., Ltd., Tokyo, Japan), as instructed by Fuji Micra Inc., at 2 to 3% of body weight/animal/day, with ad libitum access to tap drinking water. Before testis sample collection at necropsy, all animals received intramascular injections of 0.1 ml/kg of Domitor (1.0 mg/ml medetomidine hydrochloride, Nippon Zenyaku Kogyo Co., Ltd., Fukushima, Japan) or Dorbene (1.0 mg/ml medetomidine hydrochloride, Kyoritsu Pharmaceutical Industry Co., Ltd., Nara, Japan) with additional 0.1 ml/kg of Dormicum (5 mg/ml midazolam, Astellas Pharma Inc., Tokyo, Japan) as anesthesia. After a veterinarian confirmed the unconscious state of the animals, they were euthanized by exsanguination (Close et al. 1996). Body weight was measured at necropsy. The testes and epididymides were removed, and subsequently, the testes were separated from the epididymis and weighed. Collected testes were cut transversely in the middle of the major axis, as this provides uniform cross sections through all the seminiferous tubules (Lanning et al. 2002). All experiments and procedures were approved by the Animal Care and Use Committee of Shizuoka Prefectural Research Institute of Animal Industry, Swine and Poultry Research Center, which complies with the Guidelines for Proper Conduct of Animal Experiments by the Science Council of Japan: the representative organization of the Japanese scientist community (2006).
Histological Preparations
Testicular tissues were fixed in formalin–sucrose–acetic acid solution (24% v/v of a 37% solution of formaldehyde, 72% v/v of a 5% sucrose solution, and 4%v/v acetic acid) for 2 weeks or in modified Davidson’s solution (30% v/v of a 37% solution of formaldehyde, 15% v/v ethanol, 5% v/v glacial acetic acid, and 50% v/v distilled H2O) for 1 week, then dehydrated in graded series of ethanol, and embedded in paraffin (Latendresse et al. 2002; Maita et al. 1999). Paraffin sections (3 μm) were stained with PAS-hematoxylin (PAS-H) alone or were used for immunohistochemistry.
Spermiogenesis and Stages of the Seminiferous Epithelium Cycle
In the beginning, the terminologies used in this study are defined as follows: spermatogenesis, the entire process of division and differentiation of spermatogonial stem cells into spermatozoa (sperm); spermiogenesis, a part of spermatogenesis when immature round spermatids are transformed into elongated spermatids; cycle, a complete and ordered series of cell associations occurring in a particular segment of seminiferous epithelium over time; and stage, a defined group of germ cell types at certain phases of development within cross-sectioned tubules (Setchell 1978; Russell 1990; Haschek, Wallig, and Rousseaux 2010). To characterize the spermiogenesis process, spermatid development was assessed. This process was evaluated by using the sections stained with PAS-H, which was separated into steps according to the morphological development of the acrosome, nucleus, and flagellum, as previously reported (Clermont and Leblond 1955). Stages of the seminiferous epithelial cycle were identified on the basis of the morphological changes of the spermatids during spermiogenesis, presence of meiotic divisions, and overall associations of the germ cells (Berndtson 1977). Spermatogonia and spermatocytes were differentiated from each other based on the characteristics of the nuclear chromatin (Setchell 1978). Both testes from all 11 animals were examined to ensure consistency in the subdivision of steps and stages. Relative stage frequencies were determined from the analysis of 200 seminiferous tubule cross sections per testis by light microscopy. As with pigs in general, a tubular cross section on microminipig was composed of one cell association and was recognized as single stage. Both testes from each animal were analyzed.
BrdU Injections
To estimate the duration of the spermatogenesis process, 3 microminipigs (6 months of age) were intravenously injected with BrdU (5-bromo-2-deoxyuridine, Wako, Tokyo, Japan), at a dose of 10 mg/kg, approximately 21 days and 1 hr before the collection of the testes. BrdU is an analog of thymidine, which is a specific marker of cells that are in the process of synthesizing DNA at the time of injection. The period of injection was selected based on previous studies (Costa et al. 2010).
Immunohistochemistry
An antibody against BrdU (M0744) was purchased from Dako (Glostrup, Denmark). The immunohistochemical analysis was performed as follows. The sections were deparaffinized and heated 3 times at 98°C for 5 min each in a microwave oven using Target Retrieval Solution (pH 6.0, Dako) for antigen retrieval. The slides were then incubated with 2N HCl at room temperature for 30 min to partially denature the cellular DNA, followed by neutralization in 2 washes of 0.1-M borate buffer at pH 8.5 for 5 min each. The sections were washed twice in phosphate buffered saline (PBS) and then were immersed in absolute methanol and 0.3% H2O2 for 30 min to quench endogenous peroxidase activity. After blocking nonspecific binding with Protein Block, Serum-Free (Dako) for 10 min, the slides were incubated with the primary antibody at 1:100 in Antibody Diluent, Dako REAL (Dako) for 1 hr at room temperature. After being thoroughly rinsed twice in PBS, the slides were incubated with EnVision+ Dual Link System-HRP (Dako) for 30 min at room temperature and then with DAB+ Substrate Chromogen System (Dako) to detect the immunoreactivity. The sections were then stained with PAS-H. The analyses of these sections were performed by light microscopy to detect the germ cell type immunostained and to identify the stages of the seminiferous epithelium cycle at which these cells were observed.
Length of the Seminiferous Epithelium Cycle
The length of the seminiferous epithelium cycle was estimated based on the stage frequencies and the most advanced immunostained germ cell types corresponding to the different time points after BrdU injections (Almeida, Leal, and Franca 2006; Costa et al. 2010). The formula used was as follows:
The lengths of each stage of the cycle were determined by taking into account the cycle length and the percentage of each stage. The total duration of spermatogenesis was estimated based on the length of 1 cycle and the number of cycles necessary for this process to be completed, from type A spermatogonia to spermiation. In mammals, the number of cycles is known to be approximately 4.5 (Amann and Schanbacher 1983).
Results
Biometric Data
Mean body and testis weights of the microminipigs were 11.6 ± 1.9 kg and 44.8 ± 7.8 g (right testis, 22.2 ± 4.6 g; left testis, 22.5 ± 3.8 g), respectively, providing a gonadosomatic index (relative testis weight = absolute testis weight/body weight) of 0.39 ± 0.05%.
Spermiogenesis and Stages of the Seminiferous Epithelium Cycle
Spermiogenesis was classified into 15 steps according to the morphological development of the acrosome, nucleus, and flagellum. The morphological characteristics of each step are described in Figure 1 and Table 1. Steps 1 to 3 correspond to the Golgi phase of spermatid development, steps 4 to 8 the cap phase, steps 9 to 13 the acrosomal phase, and steps 14 to 15 the maturation phase.
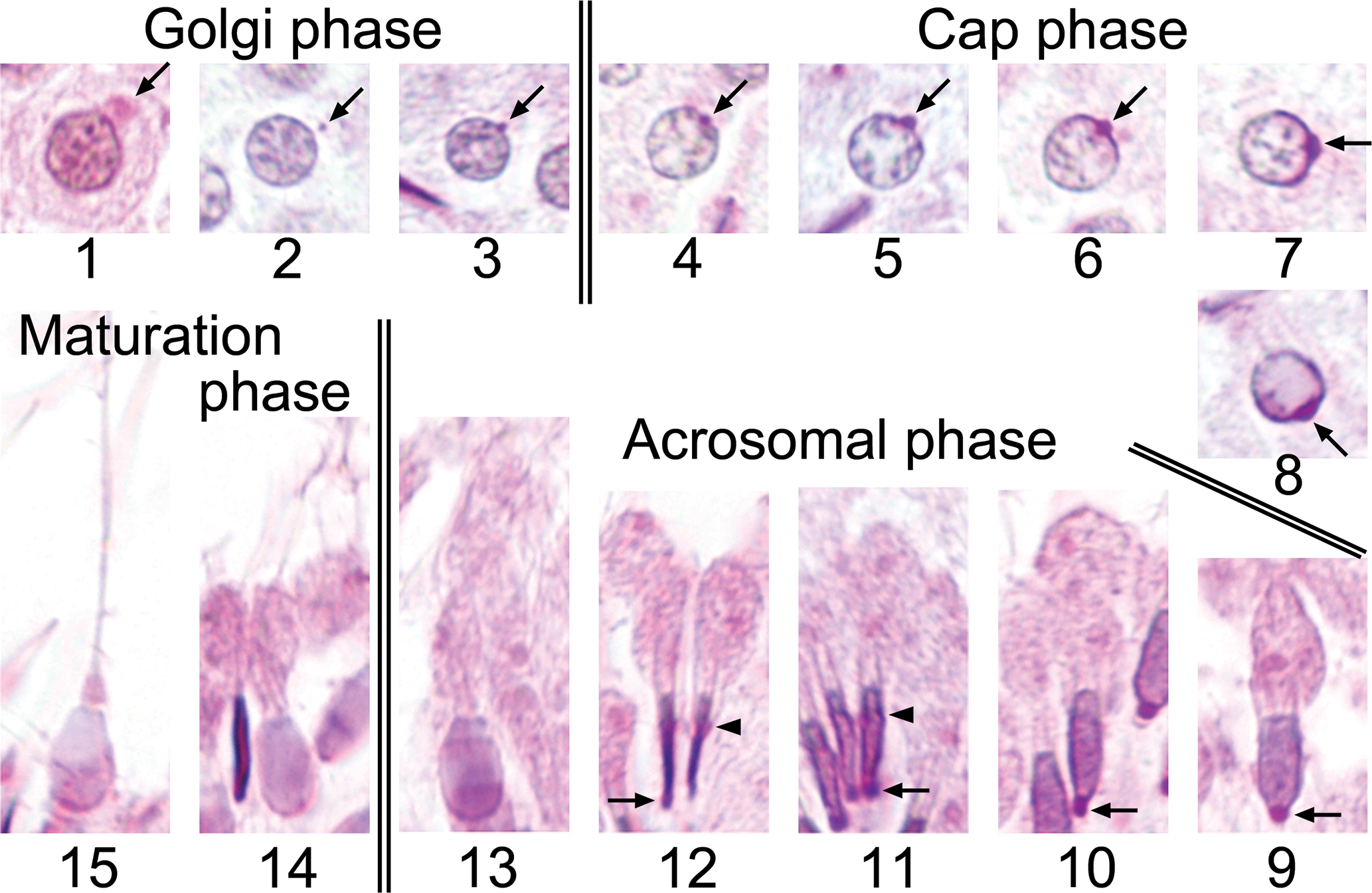
Spermiogenesis in the microminipig with periodic acid-Schiff (PAS)-hematoxylin staining. Steps 1 to 3 Golgi phase. Steps 4 to 8 cap phase. Steps 9 to 13 acrosomal phase. Steps 14 to 15 maturation phase. The arrows indicate the Golgi apparatus (step 1) and the acrosomic granule (steps 2 to 12). The arrowheads indicate the caudal edge of the nuclei with prominent PAS reactivity (steps 11 and 12).
Summary of the morphological characteristics of the spermiogenesis in the microminipig.

Stages of the seminiferous epithelium cycle were classified into 11 stages based on the steps of the spermatid development, meiotic divisions, and germ cell associations. The morphological characteristics of each stage are described in Figure 2 and Table 2. The mean percentages of the relative stage frequencies were as follows: stage I, 8.4 ± 1.2%; stage II, 4.3 ± 1.0%; stage III, 6.2 ± 0.9%; stage IV, 6.6 ± 1.8%; stage V, 7.2 ± 2.2%; stage VI, 10.4 ± 2.5%; stage VII, 14.5 ± 2.1%; stage VIII, 18.9 ± 2.8%; stage IX, 6.9 ± 1.6%; stage X, 5.1 ± 1.1%; and stage XI, 11.5 ± 1.8%.
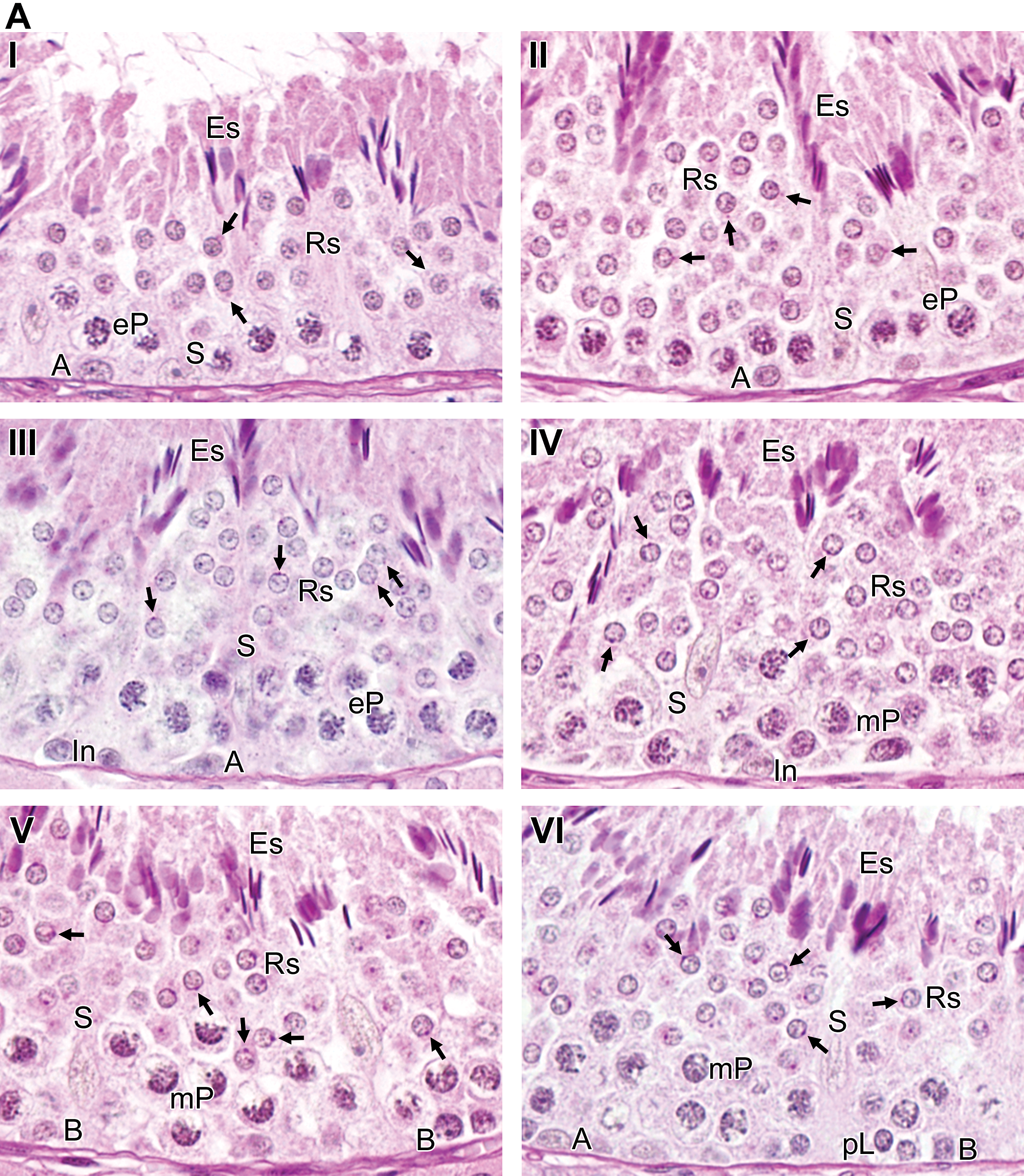
Stages of the seminiferous epithelium cycle in the microminipig classified according to the development of the acrosomic system. Eleven stages, indicated by Roman numerals, were characterized. (A) Stages I to VI represent the early stages. (B) Stages VII to XI represent the late stages. The arrows indicate the Golgi apparatus (stage I), the acrosomic vesicle and granule (stages II–VIII). A = type A spermatogonia, In = intermediate spermatogonia. B = type B spermatogonia, pL = preleptotene spermatocytes; L = leptotene spermatocytes; Z = zygotene spermatocytes; eP = early pachytene spermatocytes; mP = mid pachytene spermatocytes; lP = late pachytene spermatocytes; D = diplotene spermatocytes; M = meiotic figures; Rs = round spermatids; Es = elongated spermatids; S = Sertoli cells; Rb = residual bodies. Periodic acid-Schiff-hematoxylin staining.
Summary of the cellular associations during the seminiferous epithelium cycle in the microminipig.

The percentages of the premeiotic (VIII–X), meiotic (XI), and postmeiotic (I–VII) stages were 30.8%, 11.5%, and 57.7%, respectively.
Immunohistochemistry
In all 3 studied animals, a BrdU-positive reaction was detected in the cells located in the basal compartment at stages II, III, V, and VII and in those located in the luminal compartment at stage X. In addition, there were a few BrdU-positive cells in the luminal compartment at stage VIII. According to the morphological characteristics, such as shape of the nucleus and accumulations of the chromatin (Setchell 1978), the BrdU-positive cells were identified as follows: cells with ovoid nuclei and dust-like chromatin at stage II were type A spermatogonia (Figure 3A); cells with ovoid nuclei and conspicuous crust-like accumulations of the chromatin within the nucleus and/or attached to the nuclear membrane at stage III were intermediate spermatogonia (Figure 3B); cells with ovoid to round nuclei and prominent crust-like accumulations of the chromatin attached to the nuclear membrane at stage V were type B spermatogonia (Figure 3C); cells with round nuclei and prominent crust-like accumulations of the chromatin throughout the nucleus at stage VII were preleptotene spermatocytes (Figure 4A); cells with small round nuclei at stage VIII were round spermatids (step 8; Figure 3D); and cells with sharpened nuclei at stage X were elongated spermatids (step 10; Figure 4B).
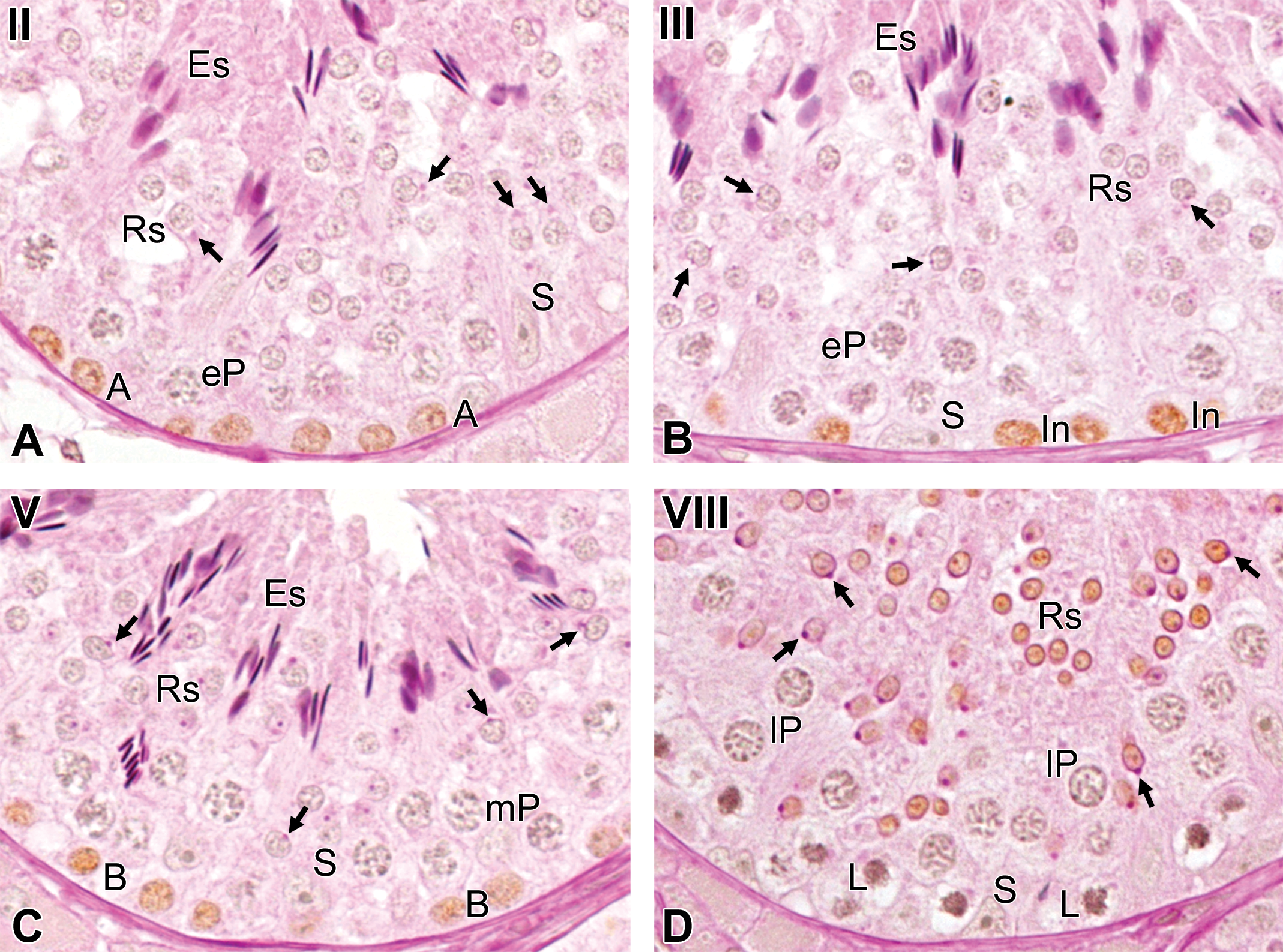
The germ cell types immunostained after different time periods following intravenous injection with bromodeoxyuridine. (A) At 1 hr after injection, type A spermatogonia: cells located in the basal compartment with ovoid nuclei and dust-like chromatin, at stage II. (B) At 1 hr after injection, intermediate spermatogonia: cells located in the basal compartment with ovoid nuclei and conspicuous crust-like accumulations of the chromatin within the nucleus and/or attached to the nuclear membrane, at stage III. (C) At 1 hr after injection, type B spermatogonia: cells located in the basal compartment with ovoid to round nuclei and prominent crust-like accumulations of the chromatin attached to the nuclear membrane, at stage V. (D) At 21 days after injection, step 8 spermatids: cells located in the luminal compartment at stage VIII. The arrows indicate the acrosomic granule. A = type A spermatogonia, In = intermediate spermatogonia. B = type B spermatogonia, L = leptotene spermatocytes; eP = early pachytene spermatocytes; mP = mid pachytene spermatocytes; lP = late pachytene spermatocytes; Rs = round spermatids; Es = elongated spermatids; S = Sertoli cells. Stained with periodic acid-Schiff-hematoxylin.
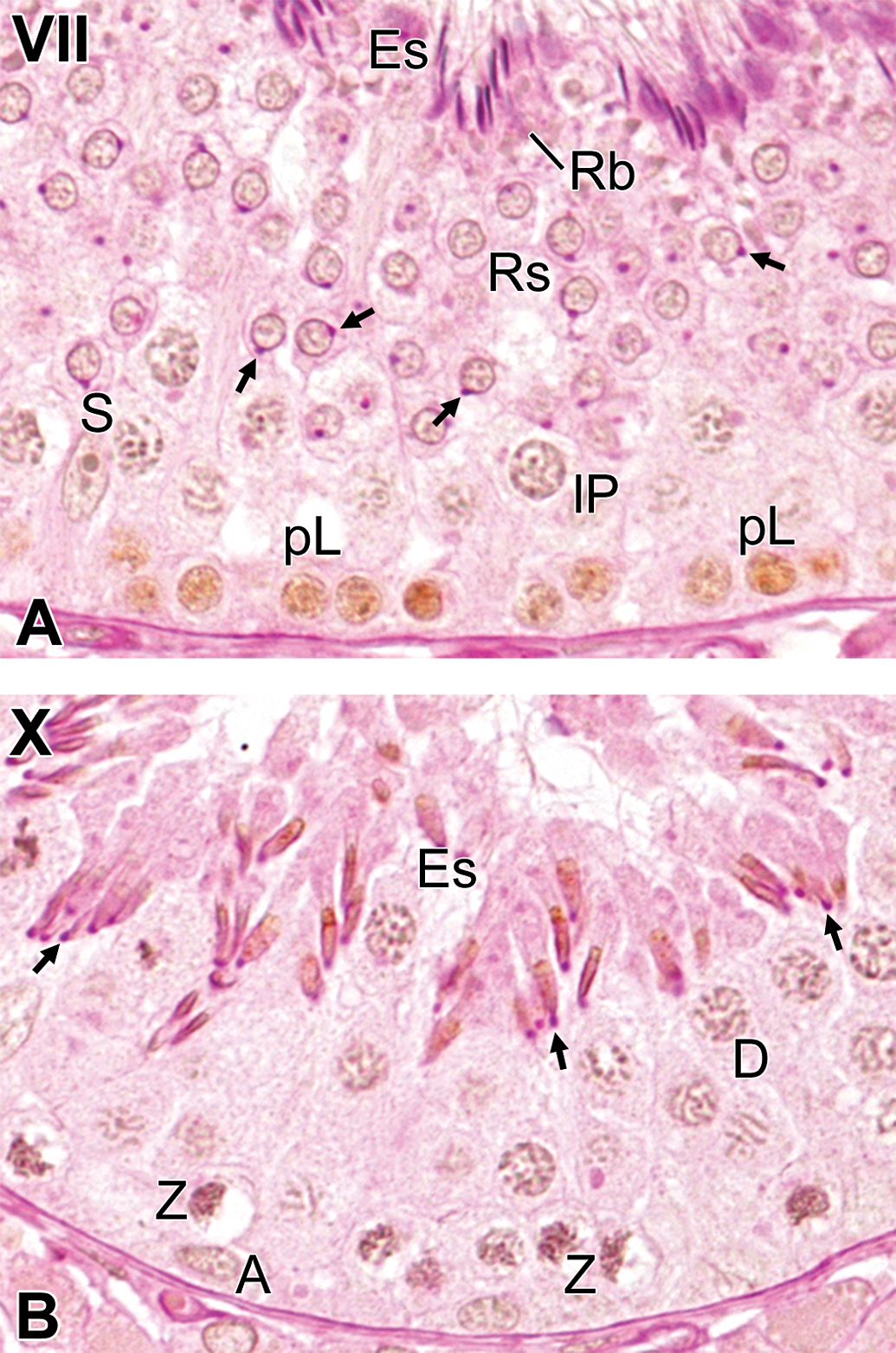
The most advanced germ cell types immunostained after different time periods following intravenous injections with bromodeoxyuridine. (A) At 1 hr after injection, preleptotene spermatocytes: cells located in the basal compartment with round nuclei and the prominent crust-like accumulations of chromatin throughout the nucleus, at stage VII. (B) At 21 days after injection, elongated spermatids (step 10): cells located in the luminal compartment with sharpened nuclei, at stage X. The arrows indicate the acrosomic granule. A = type A spermatogonia, pL = preleptotene spermatocytes; Z = zygotene spermatocytes; lP = late pachytene spermatocytes; D = diplotene spermatocytes; Rs = round spermatids; Es = elongated spermatids; S = Sertoli cells; Rb = residual bodies. Stained with periodic acid-Schiff-hematoxylin.
As BrdU is a specific marker of cells that are synthesizing DNA at the time of injection, the immunostained spermatogonia (at stages II, III, and V), and preleptotene spermatocytes (at stage VII) were the consequence of the BrdU injection 1 hr prior to the testes collection, and the immunostained round spermatids at stage VIII and elongated spermatids at stage X were the effect of the BrdU injection 21 days before.
Length of the Seminiferous Epithelium Cycle
The most advanced immunostained germ cell types, corresponding to the different time points after BrdU injections, were preleptotene spermatocytes at stage VII (1 hr after injection) and elongated spermatids at stage X (21 days after injection), as shown in Table 3 and Figure 5. The addition of stage frequencies after preleptotene spermatocytes at stage VII to elongated spermatids at stage X gave a total of 2.308 (0.423 + 1.000 + 0.885 = 2.308) cycles traversed. Based on the most advanced immunostained germ cell type and stage frequencies, the mean length of the seminiferous epithelium cycle was estimated as 9.1 days. The lengths of each stage of the cycle are shown in Figure 5. Approximately 4.5 cycles were necessary for the spermatogenic process to be completed; therefore, the total duration of spermatogenesis process in the microminipig was estimated as 40.9 days.
Mean length of the seminiferous epithelium cycle in the microminipigs.

Note. In each animal, the most advanced germ cell type immunostained 1 hr after injection was preleptotene spermatocytes in stage VII, and 21 days after injection was elongated spermatids (step 10) in stage X. The number of cycles traversed was 2.308. Mean length of the cycle is 9.1 days.
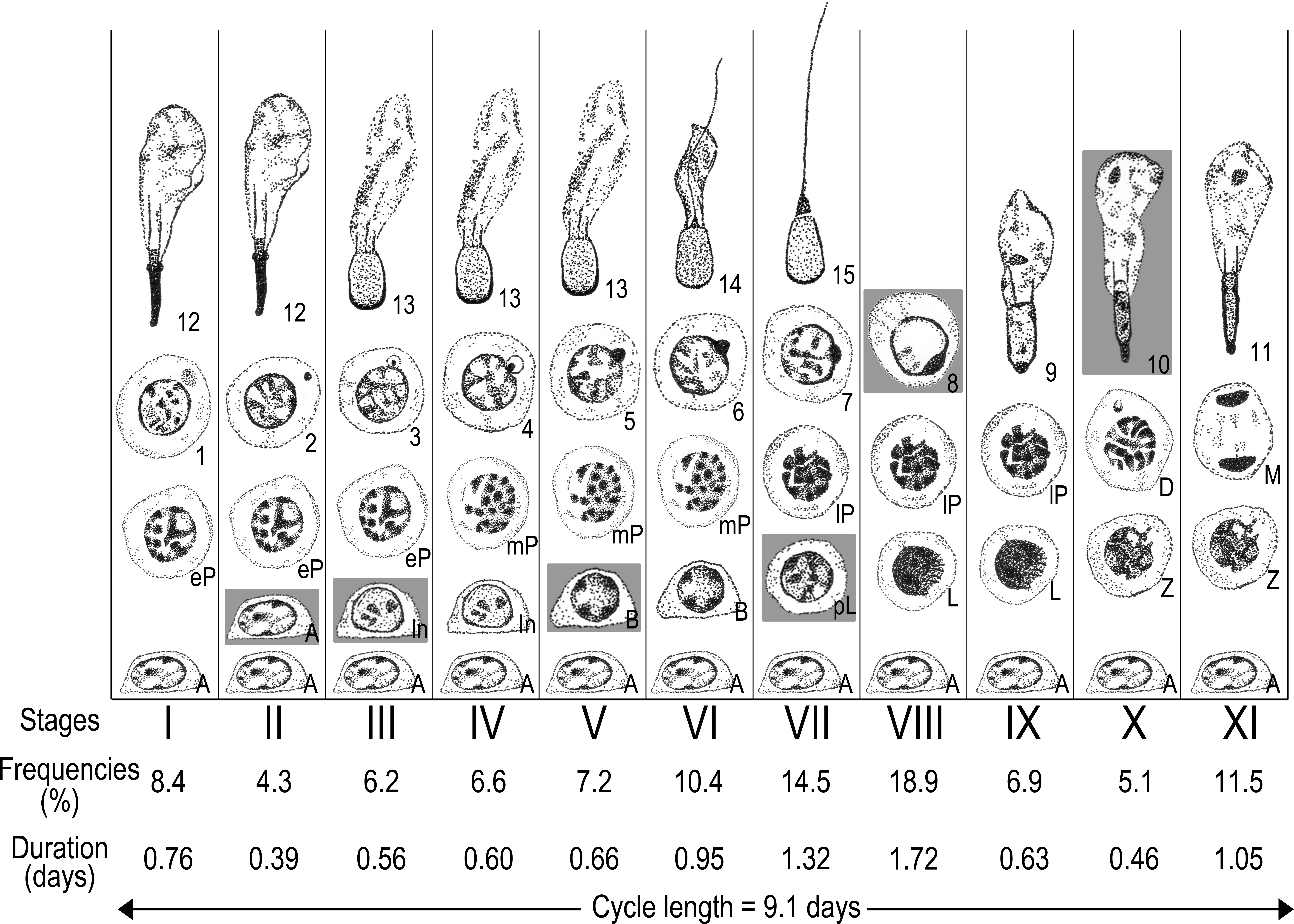
Diagram showing the cellular composition (denoted by Roman numerals I–XI), frequencies (percentage), and duration (in days) for the 11 stages of the seminiferous epithelium cycle. This diagram also illustrates the germ cell type immunodetected at different time periods (1 hr and 21 days) following bromodeoxyuridine injections (shading). A = type A spermatogonia, In = intermediate spermatogonia; B = type B spermatogonia; pL = preleptotene spermatocytes; L = leptotene spermatocytes; Z = zygotene spermatocytes; eP = early pachytene spermatocytes; mP = mid pachytene spermatocytes; lP = late pachytene spermatocytes; D = diplotene spermatocytes; M = meiotic figures; 1 to 15 = the 15 steps of the spermatid development.
Discussion
We characterized the stages of the seminiferous epithelium cycle and the duration of spermatogenesis in the microminipig using standard PAS-H staining and BrdU labeling of testicular sections. Both methods are well established for the assessment of the testis in a variety of studies. PAS-H staining is a strong tool to identify the steps of spermatid development by acrosomal analysis. BrdU labeling is a reliable method for evaluating the length of the seminiferous epithelium cycle and the total duration of spermatogenesis. To the best of our knowledge, the present study is the first to report the morphology and process of the spermatogenesis process in the microminipig.
The gonadosomatic index in the microminipig (0.39 ± 0.05%) resembled that previously reported for the domestic boar, 0.4% (França and Russell 1998). However, this index was approximately 30% higher than that of the wild boar, 0.3% (Almeida, Leal, and Franca 2006), and the Göttingen minipig at 6 months of age, 0.3% (Ellegaard-Göttingen-Minipigs 2009). Almeida, Leal, and Franca (2006) suggested that a reason for the differences in the gonadosomatic index might be the extensive reproductive selection to which most domestic pig breeds have been submitted over the centuries. Although the microminipig has not been selected based on reproductive traits, this tiny pig might have as good reproductive potential as the domestic boar.
Spermiogenesis in the microminipig was divided into 15 steps according to the morphological development of the acrosome, nucleus, and flagellum. Steps 1 to 3, 4 to 8, 9 to 13, and 14 to 15 of the spermatids corresponded to the Golgi, cap, acrosomal, and maturation phases, respectively. During the Golgi phase, the acrosomal formation produced by the Golgi complex resembled that previously reported in other mammals (Clermont and Leblond 1955; Takahashi 1994). During the cap phase, however, acrosomal development was not subdivided as clearly as in rats and mice, probably because there are slight differences in PAS reactivity between microminipigs and rodents. During the acrosomal and maturation phases in the microminipig, there was acrosomal PAS reactivity along the caudal edge of nucleus at steps 11 and 12 and a paddle shape of the nucleus as it became mature. Although PAS-H is said to be less useful for the evaluation of testicular toxicity in nonrodent laboratory animals, such as dogs and primates, due to its restricted staining of the acrosome compared to rats and mice (Lanning et al. 2002), the present study demonstrates that PAS-H staining yields a great deal of information on spermatid development in the microminipig.
A description of the stages of the seminiferous epithelium cycle in the domestic boar was previously reported using the tubular morphology system: a method for distinguishing the seminiferous epithelium cycle based mainly on changes in the shape of the spermatid nucleus, occurrence of meiotic divisions, and arrangement of spermatids (Berndtson 1977; Cupps 1991; Swierstra 1968). This approach is widely used for reproductive studies in pigs; however, it does not allow a detailed identification of the spermiogenesis steps since the spermatid nucleus does not change in size or shape during the initial steps. Therefore, each of the 8 stages described (Cupps 1991; Swierstra 1968) may correspond to more than one of the stages identified in the present study. A comparison of the seminiferous epithelium cycle stages between the tubular morphology system and the acrosomic system in the microminipig is shown in Figure 6. It is evident that stages 5 and 6 in the tubular morphology system could be subdivided into 3 stages and 2 stages in the acrosomic system, respectively. In the present study, we adopted the acrosomic system to identify the stages of the seminiferous epithelium cycle, as this method has been used in most laboratory animals such as rats, mice, guinea pigs, Chinese hamsters, and monkeys (Leblond and Clermont 1952a; Oakberg 1956; Clermont 1960; Oud and de Rooij 1977; Dreef, Van Esch, and De Rijk 2007) and has significant advantages for the evaluation of effects of experimental treatment on the testes. One advantage of the acrosomic system is greater refinement due to the larger number of stages that could be identified. In addition, accurate identification of the stages is often possible with the acrosome system even after drastic reductions of the number of spermatids. In such cases, the tubular morphology system cannot be used without adopting new criteria, which generally cause pooling of several stages, and hence, reducing precision (Berndtson 1977).
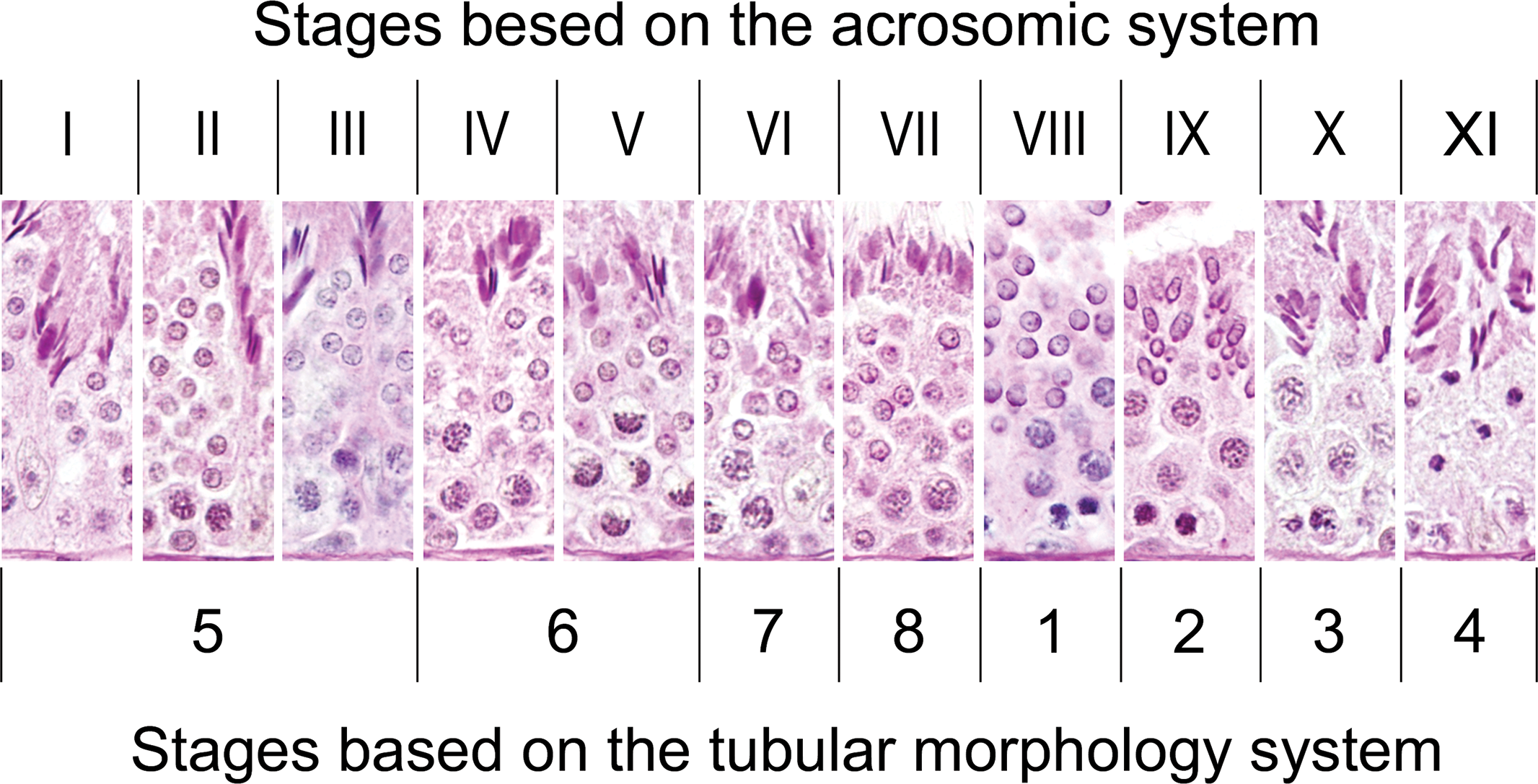
Schematic comparison between the subdivision of the seminiferous epithelium cycle in the microminipig into stages on the basis of the acrosomic system (designated by Roman numerals) and the tubular morphology system (designated by Arabic numerals). Stages 5 and 6 in the tubular morphology system can be subdivided into 3 stages (stages I, II, and III) and 2 stages (stages IV and V) in the acrosomic system, respectively.
Understanding the characteristics of spermatid development is necessary since it gives the basic concept for the staging of the seminiferous epithelium cycle (Russell 1990; Leblond and Clermont 1952b). In addition, staging is fundamental to the assessment of the testicular changes, particularly in toxicological studies (Creasy 1997). The seminiferous epithelium cycle in the microminipig was classified into 11 stages based on spermiogenesis, the steps of the spermatid development, and germ cell associations. In the domestic boar, the seminiferous epithelium cycle was classified into 10 stages (Franca et al. 2005) or 12 stages (Frankenhuis, Kramer, and de Rooij 1982) using the acrosomic system. However, the morphological development of the spermatids was not fully illustrated in both cases. The approach used in the present study to classify the seminiferous epithelium cycle was based on the detailed morphological characteristics of the spermatid development. Even though the number of stages into which the cycle is divided is arbitrary and depends on the classification criteria used by the originator of the particular scheme (Haschek, Wallig, and Rousseaux 2010), our results can precisely illustrate the changes in the seminiferous epithelium cycle and be applicable to evaluating spermatogenic disturbance.
A simplified classification of the stages of the cycle has been used in mammals, dividing the spermatogenic process into 3 phases in relation to meiosis: a premeiotic phase (after spermiation and prior to the metaphase of meiosis I), a meiotic phase (meiosis I through meiosis II), and a postmeiotic phase (after completion of meiosis until spermiation; França and Russell 1998). In the domestic boar, the frequencies of the premeiotic, meiotic, and postmeiotic phases are 28 to 32%, 11 to 13%, and 56 to 60%, respectively (França and Russell 1998; Franca et al. 2005). The frequencies of the 11 stages in the microminipig, grouped in these 3 phases of spermatogenesis, were similar to those of the domestic boar. This observation is in agreement with a number of reports (Almeida, Leal, and Franca 2006; Franca and Cardoso 1998; Neves, Chiarini-Garcia, and Franca 2002; Costa et al. 2008; Leal and Franca 2006), suggesting that phylogeny is strongly related to stage frequencies when grouped in premeiotic, meiotic, and postmeiotic phases. Although microminipigs are so small that there is always concern about their soundness and potential as a laboratory animal, the results support the fact that there are no reproductive abnormalities in them as far as spermatogenesis is concerned.
BrdU is used as a specific marker of the cells that are synthesizing DNA; the use of BrdU allows one to calculate the duration of the cycle of the seminiferous tubules in rats (Rosiepen, Chapin, and Weinbauer 1995), and it was also shown to be a useful method for evaluating cellular proliferation in somatic cells as well as the cell cycle in spermatogonia and spermatocytes (Monesi 1962). The values obtained in the microminipig for the spermatogenic cycle length (9.1 days) and the total duration of spermatogenesis (40.9 days) were similar to those found for domestic pigs; 8.6 to 9.0 and 38.7 to 40.5 days, respectively (Franca et al. 2005; Swierstra 1968). This finding does not contradict previous studies showing that the duration of spermatogenesis does not differ substantially among different breeds or strains (França and Russell 1998). The duration of spermatogenesis in the rat, dog, rabbit, and human was 51.6 days, 54.4 days, 51.8 days, and 64 days, respectively (Haschek, Wallig, and Rousseaux 2010). The shorter duration of the spermatogenesis in the microminipig, compared to other laboratory animals, is an advantage for toxicity studies where spermatogenesis is a potential target; it is also advantageous in evaluating the reversibility of these effects.
There is a general agreement that spermatogonial renewal takes place to maintain their number when type A spermatogonia divide to generate two intermediate spermatogonia (Setchell 1978). Based on the morphological characteristics of the nucleus, the spermatogonia immunostained with BrdU in stages II, III, and V in the present study corresponded to type A spermatogonia, intermediate spermatogonia, and type B spermatogonia, respectively. These results indicate that the division of type A spermatogonia into intermediate spermatogonia takes place around stage II. Therefore, stage II is presumably the starting point of the spermatogenesis. This estimation is supported by the fact that the entire process from newly produced type A spermatogonia in stage II to spermiation in stage VII spans 4.49 cycles based on the addition of stage frequencies shown in Figure 5 (0.916 + 1.000 + 1.000 + 1.000 + 0.577 = 4.493), which is consistent with the previous report that approximately 4.5 cycles are necessary for this process to be completed from type A spermatogonia to spermiation in mammals (Amann and Schanbacher 1983).
BrdU-positive elongated spermatids at stage X (descendant of preleptotene spermatocytes) and a few round spermatids at stage VIII (descendant of type B spermatogonia) were the only immunostained germ cells detected as a consequence of BrdU injection 21 days prior to sacrificing the animals. Although we expected to detect immunopositive cells that were derived from type A and intermediate spermatogonia with incorporated BrdU, it was not detected within the cycle in spite of thorough investigation. The possible reason would be that BrdU in the descendant cells of type A and intermediate spermatogonia was not easily detectable on the basis of immunohistochemical method used in the present study, because spermatogonia divided more frequently than preleptotene spermatocytes and the BrdU content within each descendant cell decreased as it underwent a series of mitotic divisions (Ramos-Vara 2005; Matos et al. 2010).
The results of the present study demonstrate that staging of the seminiferous epithelium cycle using the acrosomic system effectively describes the morphological changes in the microminipig. In addition, the duration of the spermatogenesis is almost identical to that previously reported in domestic pigs, which is relatively short compared to other laboratory animals. Therefore, we expect the information obtained by the present study to be useful as a basis for the evaluation of testicular toxicity, such as spermatogenesis disruption, in general toxicity studies. Considering that a limited number of the animals have been evaluated so far, further studies on the physiological aspects, such as hormonal regulation of the male reproductive system, and morphologic manifestations of response to injury, are required to comprehensively establish the function of the spermatogenesis in the microminipig.
Footnotes
Acknowledgments
The authors thank Professor Yoshiyasu Kobayashi (Obihiro University of Agriculture and Veterinary Medicine) for his helpful comments on earlier version of this article, Professor Motoki Sasaki (Obihiro University of Agriculture and Veterinary Medicine) for his advice during the early days of the study, and Mr. Toshiaki Fukumura (Shizuoka Prefectural Research Institute of Animal Industry, Swine and Poultry Research Center) for taking good care of the animals.
Author Contributions
Authors contributed to conception or design (AK); data acquisition, analysis, or interpretation (MO, SE, TY, YK, MS); drafting the manuscript (AK); and critically revising the manuscript (MO, SE, TY, YK, MS). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

