Abstract
Uterine leiomyomas in miniature pet pigs occur similarly to those in women with regard to frequency, age, parity, and cycling. Clinical signs, gross, and histologic features of the porcine tumors closely resemble uterine leiomyomas (fibroids) in women. Although fibroids are hormonally responsive in women, the roles of estrogen and progesterone have not been fully elucidated. In this study, immunohistochemistry was used to assess the expression of the steroid hormone receptors, estrogen receptor alpha (ER-α), estrogen receptor beta (ER-β) and progesterone receptor (PR), and cell proliferation markers, proliferating cell nuclear antigen (PCNA) and Ki-67 in tumor and matched myometrial tissues sampled from miniature pigs. A “quickscore” method was used to determine receptor expression and labeling indices were calculated for the markers. ER-α/β and PR were localized to the nuclei of smooth muscle cells in both tissues. PR expression was intense and diffuse throughout all tissues, with correlation between tumors and matched myometria. Conversely, ER-α expression was variable between the myometrial and tumor tissues, as well as between animals. ER-β expression was low. PCNA and Ki-67 were localized to the nucleus and expression varied among tumors; however, normal tissues were overall negative. These findings support further investigation into the use of the miniature pig as a model of fibroids in women.
Keywords
Introduction
Fibroids are an extremely common benign neoplasm affecting the female reproductive tract, with an estimated cumulative incidence of 70 to 85% in American women and leading to clinical problems such as pain, abnormal bleeding, and infertility (Baird et al. 2003; Buttram and Reiter 1981; Cardozo et al. 2012; Cramer and Patel 1990; Ellenson and Pirog 2010; Segars et al. 2014). Despite an abundance of research that has well-characterized these tumors, determined risk factors, and allowed advances in treatment and prognosis, the pathogenesis of fibroids is not fully understood and efforts to prevent development or cure existing disease are elusive. Although the Eker rat is an established animal model with a high spontaneous occurrence of uterine leiomyomas (∼65%; Walker, Hunter, and Everitt 2003
Miniature, or potbellied, pigs attained popular pet status in the mid-1980s and an aging pet population has enabled veterinarians to identify the uterine leiomyoma as the most common tumor in intact females (Ilha et al. 2010). These tumors occur spontaneously at a similar frequency to that observed in humans, affecting approximately 65% of pigs aged 5 years or older. Both gross and histologic features of the porcine tumor are similar to those seen in women (Mozzachio, Linder, and Dixon 2004), and the pig has a 21-day, year-round estrous cycle (Swindle 2007) that more closely approximates that of women as compared to current animal models of this tumor. Additionally, porcine uterine leiomyomas are more common in nulliparous animals as parity appears to be protective, just as it is in women. Although the complex signaling pathways involved in fibroid pathogenesis remain the focus of ongoing research, there is clear evidence that estrogen and progesterone play a significant role in the growth of these tumors in women, through interaction with their respective receptors (Borahay et al. 2015; Kim and Sefton 2012). However, the receptors for these critical steroid hormones have not yet been evaluated in the minipig. Furthermore, cell proliferation is an important indicator of growth of these tumors. Increased cell proliferation has been reported to be a significant contributor to growth and is autonomous for each tumor in a given woman (Dixon et al. 2002).
In order to gain a better understanding of the pathology of the tumors in women, animal models are necessary. Earlier we reported the potbellied, or miniature, pig (Sus scrofa) as a potential animal model because their uterine smooth muscle tumors grossly and histomorphologically resemble human fibroids (Mozzachio, Linder, and Dixon 2004). The goal of this study was to assess the immunoexpression of the steroid hormone receptors, estrogen receptor alpha (ER-α), estrogen receptor beta (ER-β) and progesterone receptor (PR) as well as the endogenous cell proliferation markers, proliferating cell nuclear antigen (PCNA) and Ki-67, in miniature pig leiomyomas and matched normal myometrial tissues.
Materials and Methods
Tissue Collection, Fixation, and Histological Stains
Tissue samples of all gross masses and matched normal myometrium, as well as a myometrial sample from an ovariectomized female, were obtained at surgery or necropsy from client-owned or rescue minipigs presenting to the North Carolina State University College of Veterinary Medicine; pigs ranged from approximately 6 to 12 years of age. Tissues were placed in 10% neutral-buffered formalin at room temperature for 24 to 72 hr, then processed, and paraffin-embedded using routine methods. Samples were sectioned at 4 to 5 µm and stained with hematoxylin and eosin (H&E) and Masson’s Trichrome. Unstained tissue sections were cut at 4 to 5 µm and used for immunohistochemical studies.
Immunohistochemical Stains
In the initial retrospective study, immunohistochemical staining for α-smooth muscle actin was performed to confirm the smooth muscle origin of the tumors; results have been previously published (Mozzachio, Linder, and Dixon 2004). In the current study, immunohistochemical staining for the steroid hormone receptors, ER-α, ER-β, and PR, as well as the cell proliferation markers, PCNA and Ki-67, was performed on leiomyoma and animal-matched normal myometrial tissues from a subset of minipigs from the original study. In addition, the atrophied uterus of a single ovariectomized minipig was included to determine the extent of steroid hormone receptor expression in the myometrium in the absence of ovarian hormones. All tissues were evaluated by routine light microscopy and a total of 22 tumors and animal-matched normal myometrium from 7 minipigs were assessed for the sex steroid hormone receptors. A total of 10 tumors and animal-matched normal myometrium from 6 minipigs were evaluated for the proliferation markers.
Following deparaffinization in xylene and rehydration through graded alcohols, endogenous peroxidase was blocked using 3% H2O2. Heat-induced antigen retrieval was performed using either a microwave or the Decloaker (Biocare Medical, Concord, CA). Nonspecific sites were blocked using normal horse or goat serum at room temperature for 20 min, respective to the secondary antibody used. Tissues were then incubated with each of the primary antibodies listed in Table 1 for 1 hr at room temperature, followed by incubation with the appropriate secondary antibody for 30 min at room temperature. Negative controls consisted of normal mouse IgG at the same concentration as the primary antibody. Immunoreactive complexes were detected by an avidin–biotin affinity system (Vectastain Elite ABC kit; Vector Laboratories, Burlingame, CA) and visualized with 3,3′-diaminobenzidine chromogen (DakoCytomation, Carpenteria, CA). Finally, the sections were counterstained with hematoxylin, dehydrated through graded ethanol, cleared in xylene, and coverslipped.
Immunohistochemical Stains.

aImmunotech, Inc., Marseille, France.
bVector Laboratories, Burlingame, CA.
cAbD Serotec, Raleigh, NC.
dMillipore, Billerica, MA.
eJackson Immunoresearch Laboratories, West Grove, PA.
fDakocytomation Corporation, Carpinteria CA.
Microscopic Evaluation
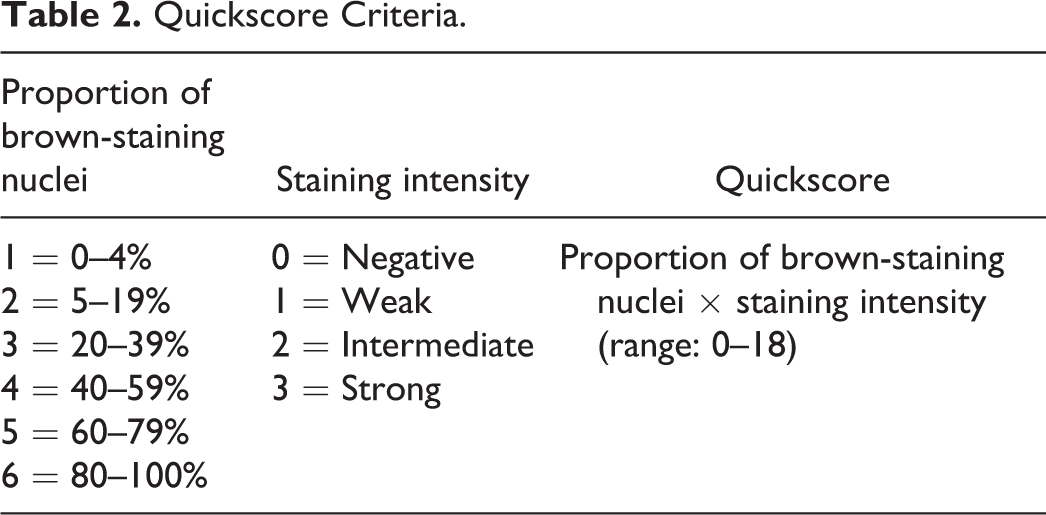
All tissues were evaluated via routine light microscopy. For the sex steroid hormone receptors, the semiquantitative method described by Detre, Jotti, and Dowsett (Detre, Jotti, and Dowsett 1995) was utilized to calculate a “quickscore” based on immunostain average intensity and the overall percentage of positive staining (Table 2). The tissues that were stained for the steroid hormones were independently scored by two coauthors. For the proliferation markers, labeling indices were calculated by dividing the number of positive (brown-staining) nuclei by a total count of 1,000 cells multiplied by 100.
Quickscore Criteria.
Statistical Analysis
Because normal tissue was sampled near tumor tissue, the statistical analysis accounted for this pairing of the data. Furthermore, quickscores were not normally distributed, so a nonparametric test, the Wilcoxon signed ranks test, was used to compare tumor to normal tissues. P values are two sided and considered statistically significant if less than .05 (Conover 1971).
Results
Tissue samples of all gross masses and matched normal myometrium from minipigs ranging in age from approximately 6 to 12 years, as well as a myometrial sample from an ovariectomized minipig, were evaluated for sex hormone receptors and proliferation markers using immunohistochemistry. Immunoexpression of the sex hormone receptors was localized to the nuclei of normal and neoplastic smooth muscle cells and positive staining was indicated by a variable brown color. ER-α expression (Figure 1A and B) was highly variable, even among different tumors in the same animal. Quickscores ranging from 1 to 18 were assigned to the tumors evaluated, and matched normal myometrium was higher, lower or equivalent to that of the tumor tissue, with a quickscore range of 5 to 18. The ER-α quickscore in the ovariectomized female was 11. ER-β was weakly expressed (Figure 1C and D), averaging a quickscore <6 in the majority of tumor tissues and in all normal myometrial tissues. The ER-β quickscore for the ovariectomized female was 1. In contrast, PR expression was typically intense and diffuse throughout both leiomyoma (Figure 1E) and normal myometrial (Figure 1F) tissue, often attaining the maximum quickscore value of 18 (tumor range 10–18, normal myometrial tissue range 12–18). PR expression was high but slightly less intense in the ovariectomized female, with a resultant quickscore of 12.
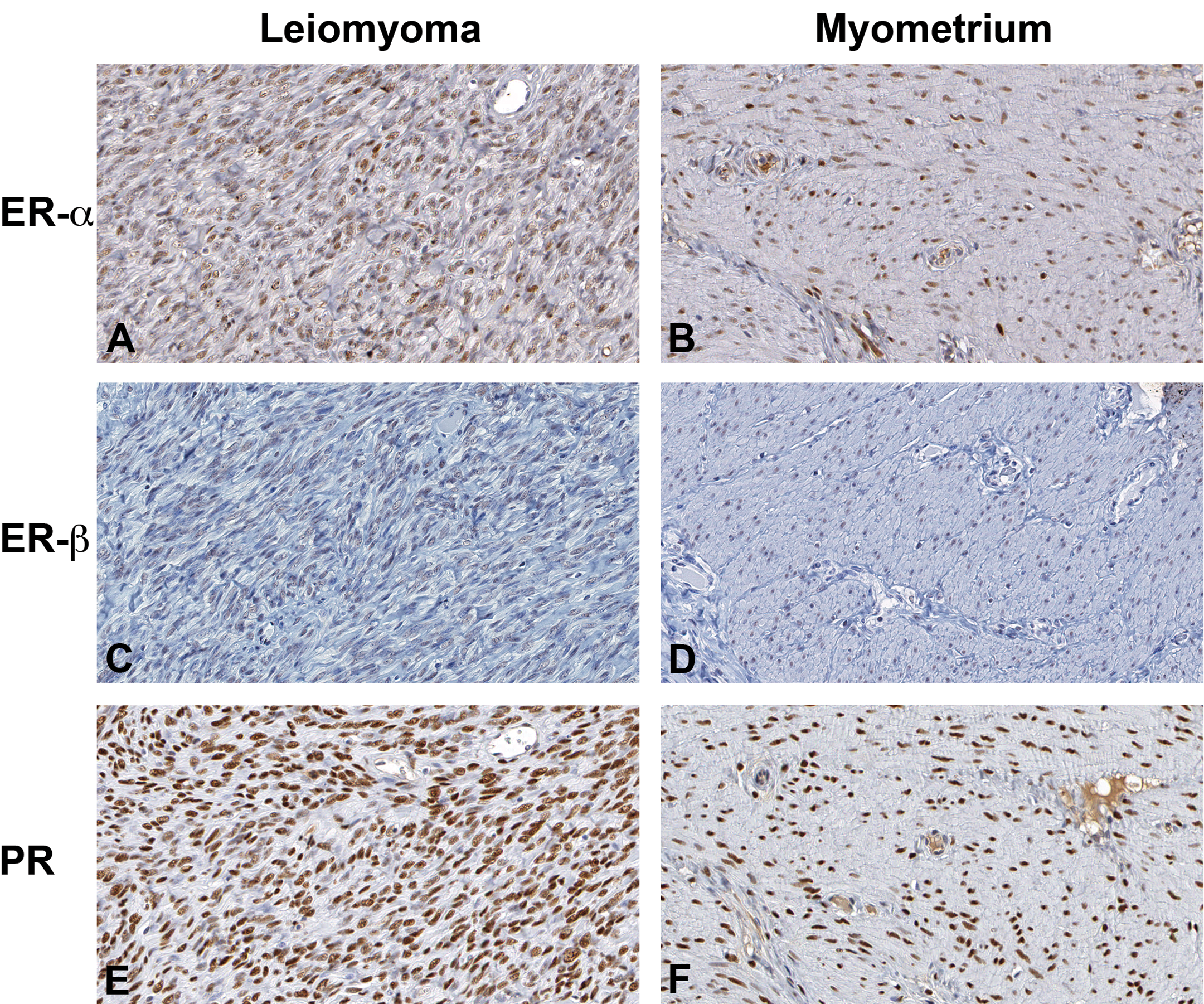
Immunohistochemical stains for sex steroid hormone receptors in porcine leiomyoma and matched normal myometrium. (A) and (B), ER-α immunoreactivity in tumor (A) versus normal myometrium (B). (C) and (D), ER-β immunoreactivity in tumor (C) versus normal myometrium (D). (E) and (F), PR immunoreactivity in tumor (E) versus normal myometrium (F). The intensity of positive brown nuclear staining ranges from weak (1C, D) to intermediate (1A, B) to strong (1E, F). ER-α = estrogen receptor alpha; ER-β = estrogen receptor beta; PR = progesterone receptor.
There was no significant difference in sex steroid hormone receptor expression between normal and neoplastic tissues for all receptors (Figure 2). However, PR expression levels were overall significantly higher (P < .05) in both leiomyoma and matched myometrial samples compared to expression of ER-α and ER-β in these tissues. Expression of ER-α was significantly (P < .05) increased compared to ER-β in both tumors and normal myometrial samples. A summary of quickscore values as well as individual quickscores for the sex steroid hormone receptors in normal and tumor tissues is presented in Figure 2 and Table 3, respectively.
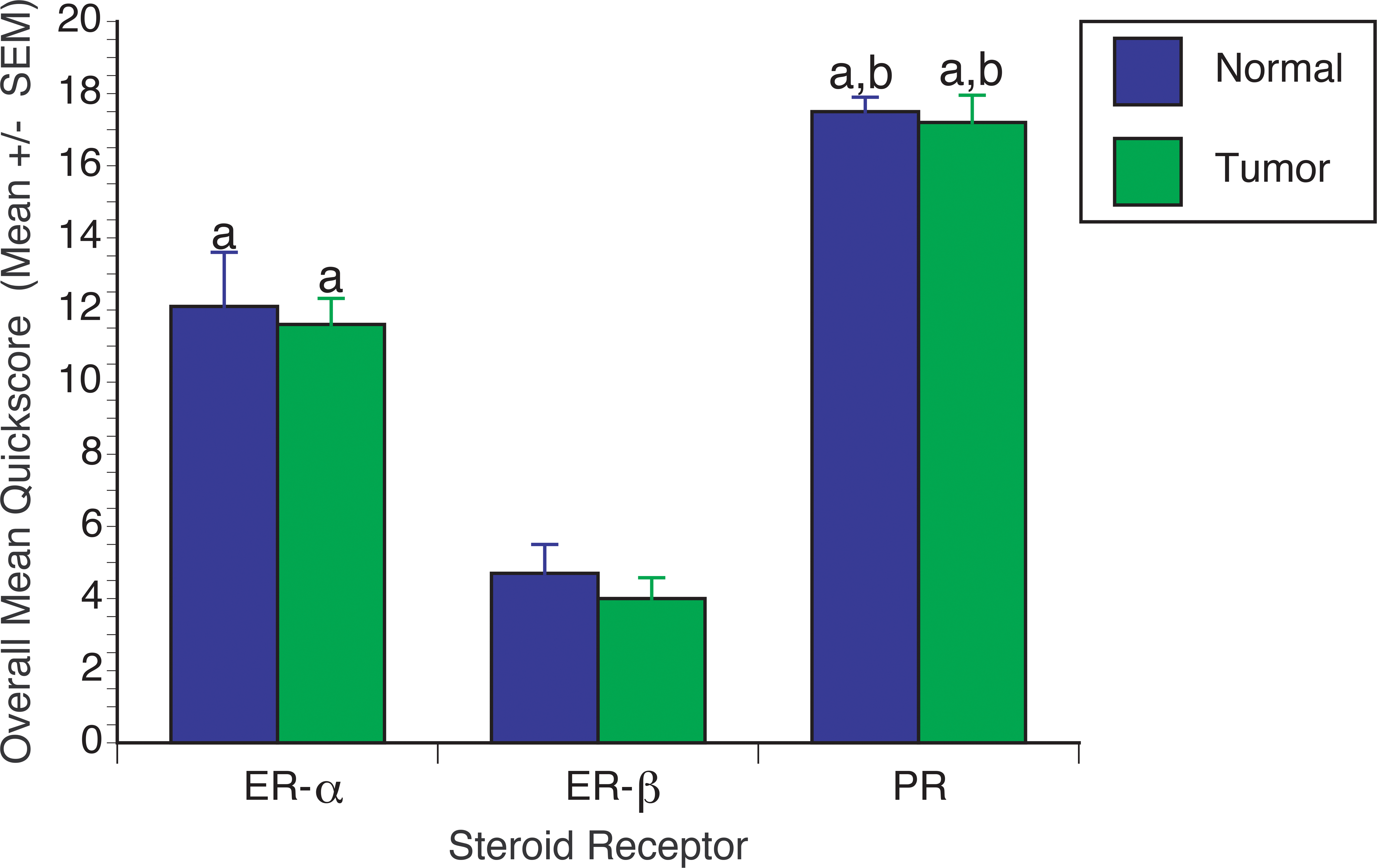
Graph shows overall average (± standard error of mean) quickscores for steroid receptors in uterine leiomyomas and matched myometrial tissues from miniature pigs. ER-α and PR expression levels were significantly higher in both leiomyoma and matched myometrial samples compared to expression of ER-β. Expression of PR was significantly higher than that of ER-α in both tumors and normal myometrial samples. There were no significant differences between tumor and normal tissues for each steroid receptor evaluated. a, Statistically different from ER-β (P < .05); b, statistically different from ER-α (P < .05).
Individual Steroid Receptor Quickscores in Miniature Pig Tissues.
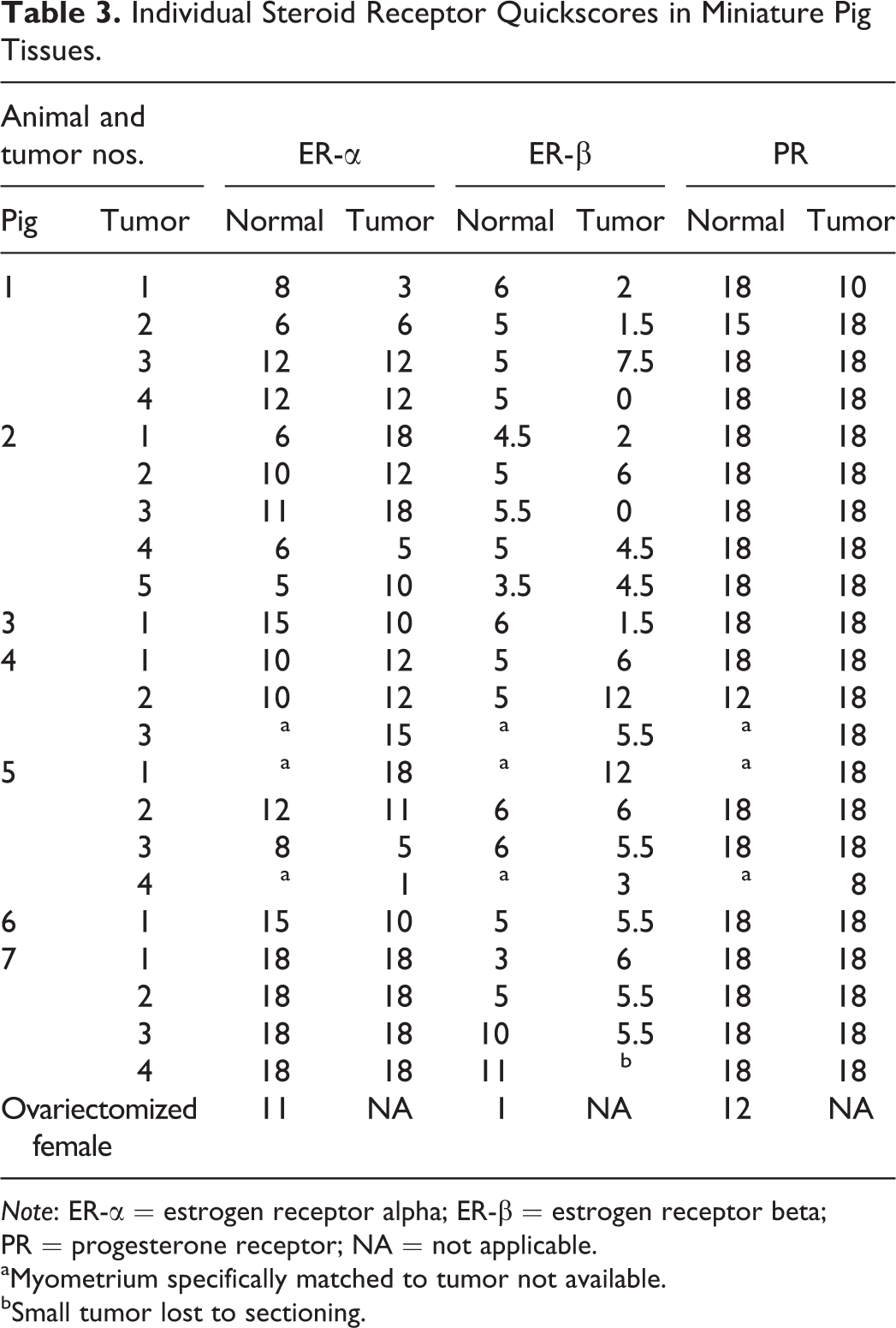
Note: ER-α = estrogen receptor alpha; ER-β = estrogen receptor beta; PR = progesterone receptor; NA = not applicable.
aMyometrium specifically matched to tumor not available.
bSmall tumor lost to sectioning.
PCNA expression (Figure 3A) was greater than that of Ki-67 (Figure 3B), and both markers were present within the majority of tumors but absent or minimally expressed in normal myometrium. Labeling indices in neoplastic tissue ranged from 0% to 52.2% for PCNA and from 0% to 6.7% for Ki-67. Expression was autonomous for individual tumors in a given animal, although large tumors (>30 cm) typically demonstrated greater expression than small tumors (<2 cm).
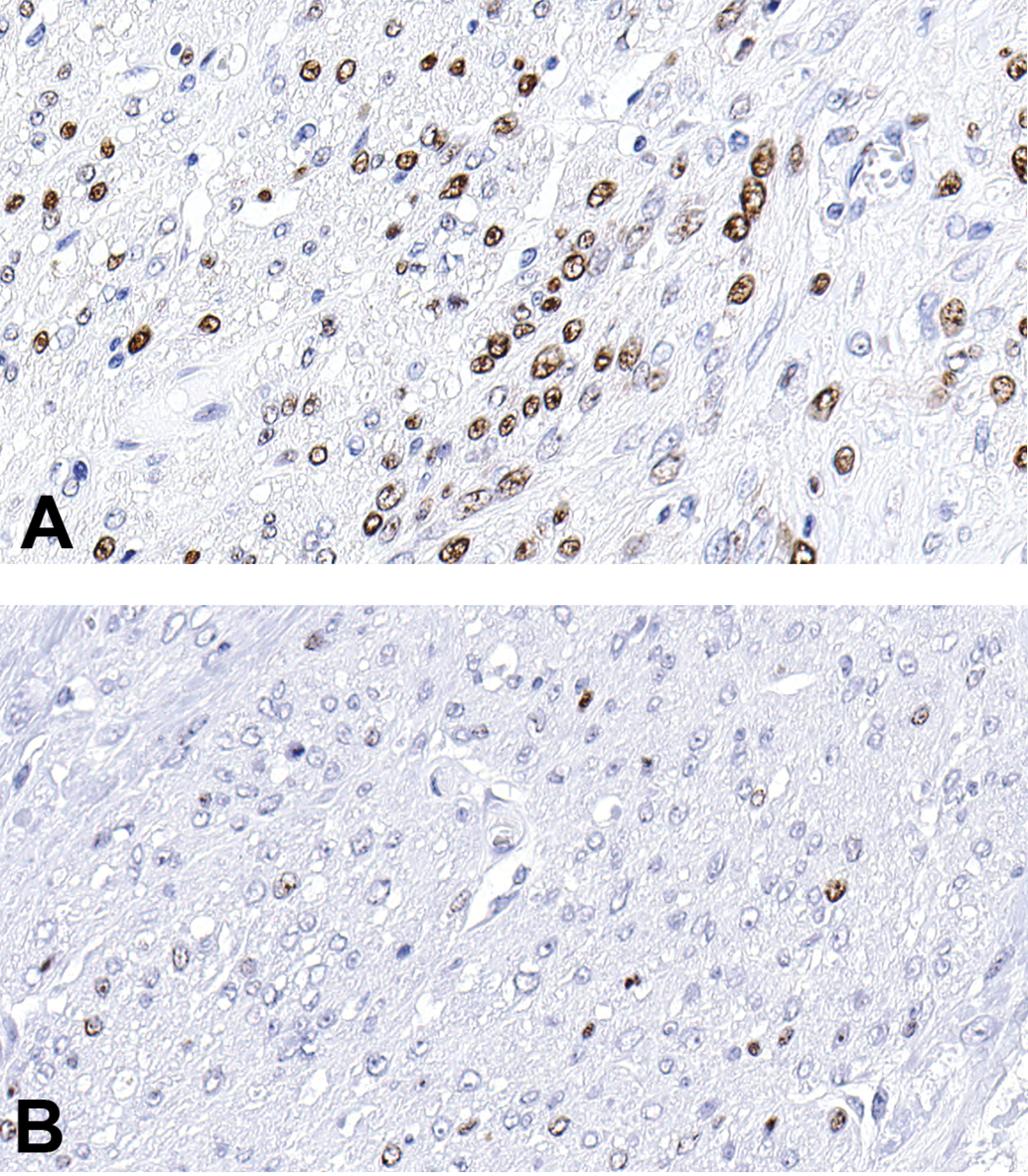
Proliferation marker immunohistochemical staining in a porcine leiomyoma, PCNA (A) and Ki-67 (B). Positive staining is indicated by brown coloration of tumor nuclei, and PCNA is expressed to a greater degree than Ki-67. PCNA = proliferating cell nuclear antigen.
Despite the absence of historical information on clinical signs of estrus, ovarian and uterine evaluation suggested that all pigs were actively cycling. However, specific estrous cycle stage data could not be ascertained.
Conclusions
The miniature pig has been identified as a promising animal model of fibroids in women (Mozzachio, Linder, and Dixon 2004). High spontaneous occurrence, clinical presentation, effects of parity, and gross (Figure 4) and histologic (Figure 5) features are remarkably similar to observations in humans (Parazzini et al. 1996; Segars et al. 2014). Although the roles of estrogen and progesterone in uterine leiomyoma development and growth are complex, the effects are elicited through their respective receptors, and the expression of these receptors has been described as a key feature of any potential animal model (Segars et al. 2014).
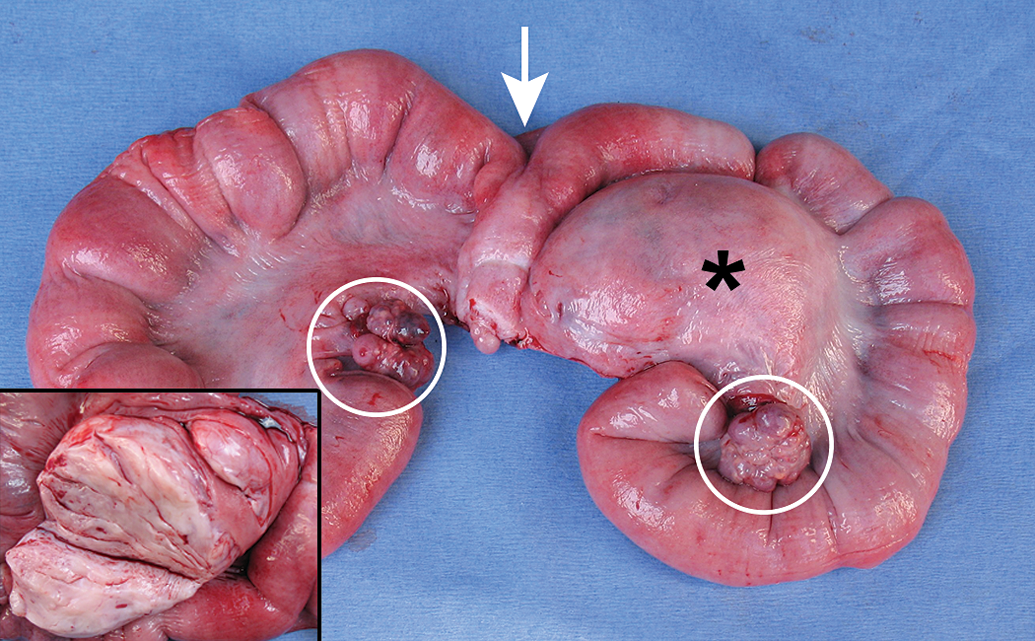
Minipig uterine leiomyoma: A firm, round, well-circumscribed mass (*) extends from the serosa of the uterine horn into the broad ligament. On cut surface (inset), the mass is white to tan and whorled. Arrow = uterine bifurcation; Circle = ovary.
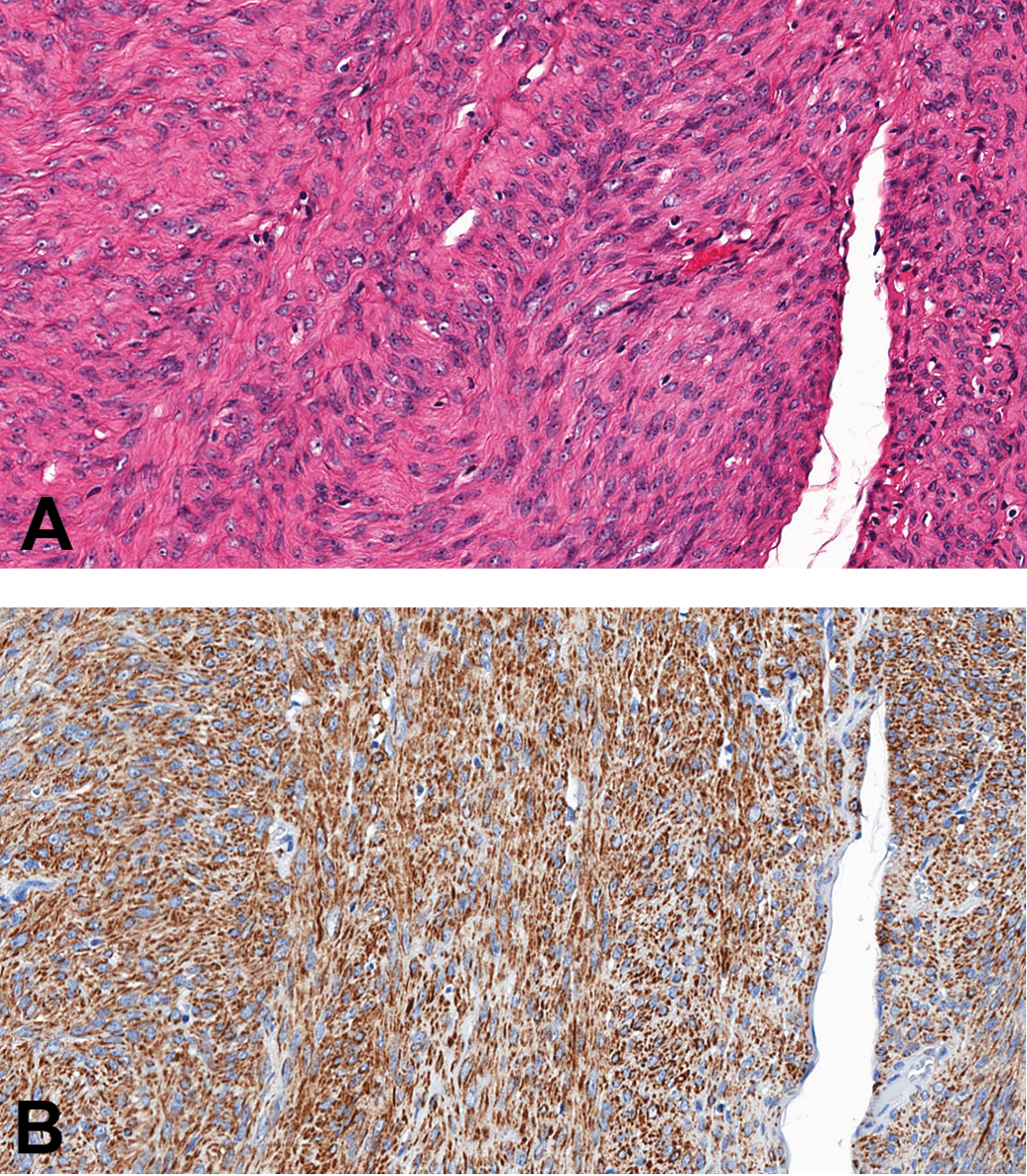
Minipig uterine leiomyoma. (A), Hematoxylin and eosin (H&E) stain showing well-differentiated smooth muscle cells arranged in interlacing fascicles. (B), Cytoplasm of tumor cells stains positively for α-smooth muscle actin.
In this study, immunohistochemistry was used to assess the expression of the steroid hormone receptors, ER-α, ER-β, and PR, and cell proliferation markers, PCNA and Ki-67, in tumor and matched myometrial tissues sampled from miniature pigs. Reports of immunoexpression of the sex steroid hormone receptors in fibroids in women are contradictory, although the majority indicate increased expression of steroid receptors in tumor tissue compared to normal myometrium (Bakas et al. 2008; Flake, Andersen, and Dixon 2003; Jakimiuk et al. 2004; Roan et al. 2005; Tamaya, Fujimoto, and Okada 1985; Wei, Chiriboga, and Mittal 2005; Wilson, Yang, and Rees 1980). Although, in this study, there was no significant difference in steroid hormone receptor expression between normal and tumor tissues in minipigs, PR expression was greater than that of ER-α, and ER-α expression was greater than that of ER-β in both normal myometrium and leiomyomas. This has also been reported in women (Plewka et al. 2014; Zaslawski et al. 2001). For the proliferation markers, PCNA was more highly expressed than Ki-67, and leiomyomas frequently exhibited an increased proliferative rate over matched myometrium. The subset of porcine tumors evaluated for proliferation markers was small but demonstrated proliferation indices autonomous for individual tumors in a given animal. In women, the proliferative state has been reported to be autonomous for each tumor in a given individual, although independent of tumor size (Dixon et al. 2002).
It is evident that fibroids are dependent on the steroid hormones, estrogen and progesterone, which play a role in the proliferation of these tumors. Steroid hormones are also thought to be important in regulating growth factors and cytokines, which can influence uterine leiomyoma cell growth in addition to contributing to the expansion of an abundance of disorganized extracellular matrix (ECM) that consists predominantly of collagen and is believed to also play a role in tumor growth (Ciarmela et al. 2011; Leppert, Jayes, and Segars 2014). Contrarily, many of the current animal models do not share this characteristic overproduction of ECM, which is a consistent feature of many fibroids in women; however, the authors have found that, over years of characterizing minipig tumors, excessive ECM is common and appears to expand with increasing tumor size. This phenomenon of increased tumor size and enhanced collagen production has been observed in fibroids in women (Flake et al. 2013). Growth factors that appear to be important in influencing ECM production as well as tumor cell proliferation are transforming growth factor-β, basic fibroblast growth factor, and insulin-like growth factor-I, among others, which have all been reported in human leiomyomas (Flake, Andersen, and Dixon 2003; Yu et al. 2008).
Recently, there has been a shift of focus to not only include steroid hormones but explore the role of genetics and stem cells in fibroid development and growth (Moravek and Bulun 2015). Genetic abnormalities, recurrent genetic aberrations, and mutations may also contribute to the development of uterine leiomyomas (Hodge et al. 2009; Makinen et al. 2011; Parker 2007; Velagaleti et al. 2010) and are considered to play pivotal roles in tumorigenesis. Although the cellular origin of uterine fibroids remains unknown, fibroids are thought to be monoclonal tumors that originate from the transformation of a single somatic, or adult, stem cell (Canevari et al. 2005; Zhang et al. 2006), although the initiating event causing the neoplastic transformation is currently unknown. Recently, a few significant advances have been made to pave the way to understanding the pathophysiology of fibroids. Somatic stem cells have been isolated and appear to be necessary for steroid hormone–dependent growth (Mas et al. 2012; Ono et al. 2012). In particular, one study identified a somatic single-gene defect encoding mediator complex subunit 12 (MED12) gene in a majority (∼70–75%) of uterine fibroid tumors (Makinen et al. 2011). MED12 is a subunit of the mediator complex, which is thought to regulate transcription (Taatjes 2010). Another study showed that the growth of human fibroid tumors, which are dependent on estrogen and progesterone, also requires the presence of these stem cells, which make up just 1% of the tumor; moreover, the fibroid stem cells, but not the stem cells from the surrounding uterine myometrial tissue, carried MED12 mutations. Interestingly, one lab summarizes that tumors will not develop without sex hormones, especially progesterone, as well as stem cells in the environment that carry the most common mutation, MED12 (Bulun et al. 2015). Although the regulation of estrogen and progesterone on the growth of leiomyoma stem cells is unknown, it has been demonstrated that other factors such as the wingless-type/β-catenin pathway plays a critical role in uterine leiomyoma tumorigenesis in a paracrine manner (Ono et al. 2013).
Conventionally, estradiol has been considered the primary stimulus for uterine leiomyoma growth, and studies with cell cultures (Barbarisi et al. 2001) and animal models (Newbold et al. 2002) support this concept. However, there is emerging evidence that progesterone and the PR play key roles in the growth and development of uterine leiomyomas (Ishikawa et al. 2010; Kim, Kurita, and Bulun 2013). The minipig tumors express both ER and PR and could, therefore, allow study of the interaction of these steroid hormone receptor pathways, in addition to their interaction with growth factors and other cytokines in regulating fibroid cell growth and expansion of the ECM. Additionally, the minipig could serve as an excellent model for future studies to further explore the role of genetic alterations involving MED12 and/or the role of stem cells in uterine leiomyoma development and growth.
Footnotes
Acknowledgments
The authors would like to thank the NIEHS Histology and Immunohistochemistry laboratories for their technical support, and Norris Flagler, Eli Ney, and Beth Mahler for their expertise with digital imaging. The authors also thank Cynthia Swanson of WIL Research-Hillsborough, and Experimental Pathology Laboratories, Inc. for support with imaging. Lastly, the authors kindly thank Drs. Xiaohua Gao and Mark Cesta for their critical review of this manuscript. This research was supported, in part, by the Intramural Research Program of the NIH, NIEHS and DNTP.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
