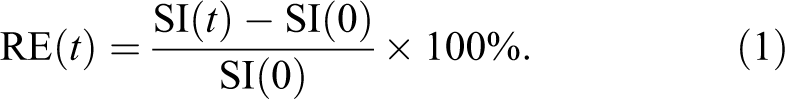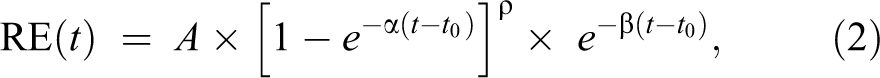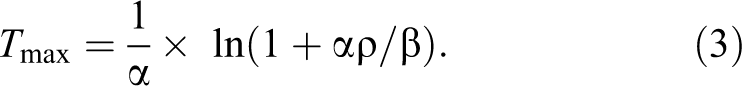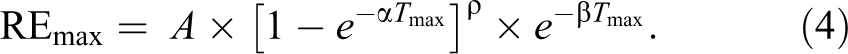Abstract
To determine if amiodarone induces hepatic phospholipidosis (PLD) sufficient to detect changes in hepatobiliary transporter function as assessed by gadoxetate dynamic contrast–enhanced magnetic resonance imaging (DCE-MRI), rats were orally dosed with vehicle (1% methyl cellulose) or amiodarone (300 mg/kg/day) for 7 consecutive days. Gadoxetate DCE-MRI occurred at baseline, day 7, and following a 2-week washout of amiodarone. At day 7, the gadoxetate washout rate was significantly decreased compared to the vehicle group. Blood chemistry analysis revealed no significant changes in liver enzymes (alanine aminotransferase [ALT]/aspartate aminotransferase [AST]/alkaline phosphatase [ALP]), bilirubin, or bile acids between vehicle or amiodarone groups. Hepatic PLD was confirmed in all rats treated with amiodarone at day 7 by transmission electron microscopy. Following the 2-week washout, there was no ultrastructural evidence of hepatic PLD in rats and the gadoxetate washout rate returned to baseline levels. This is the first study to show the application of gadoxetate DCE-MRI to detect hepatobiliary functional changes associated with PLD and offer a potential new technique with clinical utility in patients suspected of having PLD. These results also suggest PLD itself has functional consequences on hepatobiliary function in the absence of biomarkers of toxicity, given the cause/effect relationship between PLD and function has not been fully established.
Keywords
Introduction
Cationic amphiphilic drug (CAD)-induced phospholipidosis (PLD) is a phospholipid storage disorder resulting in excessive accumulation of phospholipids in tissues. Tissue PLD, a lysosomal phospholipid storage disorder, is a recognized side effect of CADs with more than 200 drug candidates having induced PLD in preclinical studies (Tengstrand, Miwa, and Hsieh 2010). Interaction with negatively charged phospholipids results in a massive accumulation of both phospholipids and the inducing drug in affected tissues, noted as electron-dense lamellar inclusions or whorls in the cytoplasm when observed with transmission electron microscopy (TEM; Hein, Lullmann-Rauch, and Mohr 1990). PLD can occur in virtually any tissue, and although the induction mechanism is well understood and generally accepted, the prevalence and toxicological implications in humans are still unclear (Reasor and Kacew 2001) particularly in those areas where no obvious evidence of cell injury is observed. However, after cessation of drug administration, PLD is reversible with drugs effluxing from cells. Therefore, the presence of PLD is currently not considered a toxicologic effect (Baronas et al. 2007). CADs appear to make “good” drugs because their amphiphilic characteristics allow them to cross biological membranes with relative ease, in particular the blood–brain barrier (Reasor, Hastings, and Ulrich 2006). The physiochemical properties most commonly shared by CADs are a hydrophobic ring structure on the molecule and a hydrophilic side chain with a charged cationic amine group. These 2 structural entities provide the amphiphilicity that is common to CADs (Halliwell 1997). The hydrophobicity of CAD molecules favors their interaction with membrane receptors. The hydrophobic structure enhances the CAD molecule’s ability to pass through plasma membranes when they are not ionized. The ionized form of the molecule tends to remain within the membrane and contribute to membranous changes (Joshi et al. 1988). The positively charged nature of CADs can also influence the movement of sodium or calcium ions across cell membranes, thus affecting receptor-mediated reactions (Mortuza et al. 2003). For these reasons, a number of CADs are marketed as drugs.
Amiodarone, marketed in the United States as Cordarone, is an antiarrhythmic drug which comes with black box warnings both for severe hepatotoxicity and for inducing PLD. Amiodarone induces PLD, and the target organs for PLD include the lung, liver, and spleen (Goldman et al. 1985; Lewis et al. 1988; Kannan et al. 1991; Reasor et al. 1996), justifying its widespread use as the reference drug for the general phenomenon of CAD-induced PLD. Amiodarone initiates a specific and nearly complete inhibition of phospholipid degradation by inhibiting lysosomal phospholipases A1 and A2 (Halliwell 1997). Two theories show how inhibition of lysosomal phospholipase activity becomes the primary mechanism by which CADs induce PLD. The first involves CADs binding to hydrophobic and hydrophilic moieties of phospholipids, resulting in complexes that are indigestible by lysosomal phospholipases (Reasor and Kacew 2001). The second theory involves direct inhibition of lysosomal phospholipases by the CADs (Kubo and Hostetler 1985; Martin et al. 1989).
Gadolinium ethoxybenzyl dimeglumine (gadoxetate), marketed as Eovist® in the United States and Primovist® in Europe, is a liver-specific magnetic resonance imaging (MRI) contrast agent approved for the focal detection of hepatocellular carcinoma. After intravenous injection, gadoxetate distributes into the vascular and extravascular spaces during the arterial, portal venous, and late dynamic phases and ultimately into the hepatocytes and bile ducts during the hepatobiliary phase. The hepatocyte uptake of gadoxetate occurs mainly via the organic anion transporter polypeptides (oatp1b1 and b3) located on the sinusoidal membrane of the hepatocyte. The biliary excretion of gadoxetate occurs primarily via the multidrug resistance–associated protein (mrp2) in the canalicular membrane of the hepatocyte. Gadoxetate transport and efflux in rat liver have been previously characterized to confirm hepatic uptake, and efflux occurs in rat hepatocytes (Jia et al. 2014; Ogawa et al. 2014). Drugs that are known inhibitors of the uptake transporters for gadoxetate will inhibit the increase in liver signal intensity (SI, uptake) after gadoxetate injection. Drugs that are inhibitors of mrp2 will prolong the efflux/washout rate of SI (Ulloa et al. 2013).
Due to the current lack of biomarkers, PLD induction and progression cannot be followed in healthy volunteers or patients and can therefore restrain drug development or drug approval (Reasor, Hasting, and Ulrich 2006). Pharmaceutical companies attempt to implement early in silico and in vitro strategies to screen out potential PLD-inducing drugs in the selection of drug candidates (Morelli et al. 2006; Tomizawa et al. 2006; Nioi et al. 2007; Chatman et al. 2009; Mesens et al. 2010). In 2010, the Food and Drug Administration formed a special panel specifically to address the challenges associated with drug-induced PLD. In this study, we asked whether amiodarone can induce hepatic PLD sufficiently to alter hepatobiliary transporter function as assessed by changes in gadoxetate dynamic contrast–enhanced (DCE)-MRI kinetics. Additionally, does gadoxetate DCE-MRI offer potential value in assessing functional liability of compounds known to induce PLD in drug development? Lastly, does gadoxetate DCE-MRI offer potential clinical translational utility in assessing liver function in response to PLD-inducing drugs?
Materials and Methods
Animal Model
Male control (Sprague Dawley) rats ∼9 weeks of age (body weight [BW] 250–300 g) were purchased from Charles River (Wilmington, MA) for the MRI study. The study consisted of a vehicle group (n = 4) and the amiodarone-treated group (n = 8). Following the 7 days of amiodarone treatment, 4 rats were sacrificed for ultrastructural confirmation of PLD, while the remaining 4 amiodarone-treated rats were allowed to recover to undergo gadoxetate DCE-MRI after 2 weeks following the cessation of amiodarone treatment. Rats were allowed free access to food and water and were acclimated for a minimum of 1 week prior to starting the MRI study. Rats were not fasted prior to MRI procedure. For all MRI time points, rats were placed in a supine position inside the magnet and anesthetized using 1.5–2.0% isoflurane anesthesia with a flow rate of ∼1 L/min of medical grade air. All animal procedures complied with the guidelines of the Institutional Animal Care and Use Committee at GlaxoSmithKline.
Amiodarone Formulation
Amiodarone hydrochloride (batch no. 32456; Sigma-Aldrich, St. Louis, MO) was formulated as a suspension in 1% methyl cellulose in reverse osmosis–treated water and administered orally to rats at a dose volume of 10 ml/kg. Groups of male rats were given amiodarone at doses of 0 (vehicle) or 300 mg/kg/day once daily in the morning for 7 days following the initial baseline gadoxetate MRI. The amiodarone dose level was selected from a previous GlaxoSmithKline (GSK) safety assessment study where the 300 mg/kg/day oral dose for 7 days was sufficient to cause hepatic PLD, while showing no increases in liver enzymes, bilirubin, or bile acids. Amiodarone at 300 mg/kg/day in a 7-day rat toxicity study was sufficient to cause hepatic PLD without accompanying increases in liver enzymes and that desirable profile was the rationale for selecting the 300 mg/kg dose.
Gadoxetate DCE-MRI
Gadoxetate DCE-MRI was performed at 4.7 T (Bruker Biospec, Billerica, MA) using a 72 mm volume coil. A tripilot scout sequence (repetition time [TR]/echo time [TE]/flip angle [FA] = 88.3 ms/1.2 ms/15°, field of view [FOV] = 10 cm × 10 cm, slice thickness = 2 mm, spatial resolution = 781 μm, matrix 128 × 128, and total acquisition time [TA] = 1 min 2 sec) was used to determine optimal anatomical coronal slice of liver. For the gadoxetate DCE-MRI, an intragate fast low angle snapshot sequence was used with the following MRI sequence parameters (TR/TE/FA = 7.12/1.8 ms/30°, FOV = 6 cm × 6 cm, slice thickness = 2 mm, spatial resolution = 234 μm, matrix 256 × 256, TA = 1 hr 5 min). Baseline (precontrast) images were acquired after 5 min, immediately followed by a tail vein injection of a clinical dose of 25 μmol/kg of gadoxetate (Eovist™—Bayer HealthCare, Whippany, NJ) to acquire postcontrast images for 60 min. A retrospective reconstruction of the DCE-MRI data was performed for 1 min temporal resolution. Data analysis consisted of a manual tracing of the liver to obtain mean SI. DCE-MRI was performed at baseline, day 7, and 2-week drug washout time points (Figure 1).
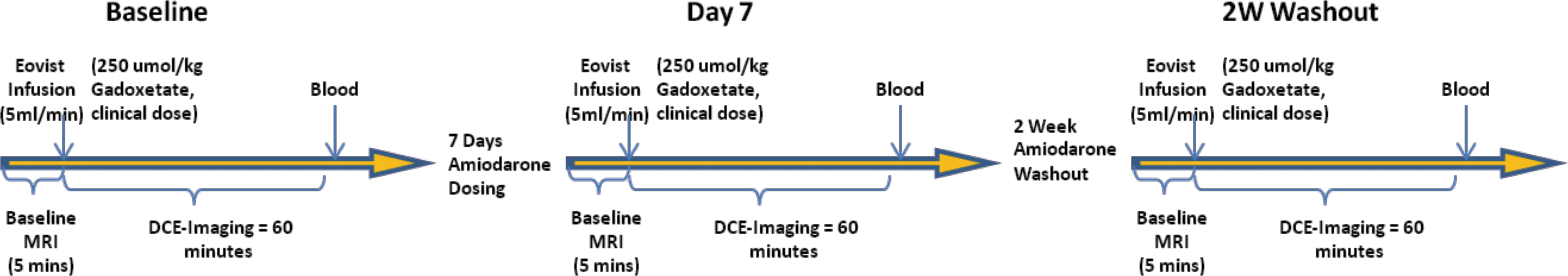
Study time line—gadoxetate dynamic contrast–enhanced magnetic resonance imaging time points and oral dosing of amiodarone over 1 week followed by the 2-week washout of amiodarone.
Mathematical Modeling of DCE Data
An empirical mathematical model (EMM) was used to calculate the gadoxetate uptake and washout rate kinetics (Saito et al. 2012). When analyzing the DCE-MRI data in the liver using a pharmacokinetic model, traditional pharmacokinetic modeling is complicated by a dual vascular blood supply from the hepatic artery and portal vein. Thus, a simple and reliable method is desired for analyzing the DCE-MRI data without vascular input functions. The SI at time t, SI(t) was obtained by manually tracing a region of interest around the liver parenchyma. The contrast agent concentration as a function of time was estimated by comparing SI(t) to the SI before contrast agent injection, SI(0). SI(0) was obtained by averaging the SI of the 5 precontrast frames of the liver. Relative enhancement at time t, RE(t) was defined as follows:
The EMM with 5 parameters was used to fit the time SI curves obtained from the DCE-MRI images following gadoxetate administration:
where A = the upper limit of the SI, α = the rate of contrast uptake (min−1), β = the rate of contrast washout (min−1), ρ = the parameter related to the slope of early uptake, and t 0 = the rise time point (min). The parameters were calculated using the nonlinear least squares method. It is important to note that the initial approximation of the parameters was critical as poor initial guesses resulted in the failure to converge on a good fit. The initial guesses based on previous experience were defined as follows: A = the maximum value of RE(t), α = 0.2 min−1, β = 0.03 min−1, ρ = 0.3, and t 0 = 5 min. The time to maximum relative enhancement (T max) was calculated by setting the derivative of equation (2) equal to zero:
The maximum relative enhancement (REmax) was obtained by substituting equation (3) into equation (2) as follows:
Blood Chemistry
Blood was collected at baseline (predose) and immediately following each MRI time point. Blood was collected in 1.5 ml heparin–lithium-coated tubes and centrifuged (3,000 rpm) to collect plasma for analysis of blood chemistry indices. A Beckman Coulter AU640 Chemistry Analyzer (Beckman Coulter Inc., Brea, CA) was used for all blood chemistry analysis. The assays used were as follows: alkaline phosphatase (ALP, Beckman Coulter reagent): p-nitrophenyl phosphate method; alanine aminotransferase (ALT, Beckman Coulter reagent): ultra-violet/nicotinamide adenine dinucleotide (UV/NADH) method; aspartate aminotransferase (AST, Beckman Coulter reagent): UV/NADH method; total bile acids (TBA, Diazyme Laboratories reagent): Enzyme cycling method; and bilirubin, total (Beckman Coulter reagent): Jendrassik-Grof method.
Histology/TEM/Pathology
For light microscopic evaluation, sections of liver that were in the imaging FOV were collected at necropsy from treated (n = 4, sacrificed at day 7), recovery (n = 4), and vehicle control (n = 4) rats and fixed in 10% neutral-buffered formalin. Tissue was trimmed, processed to paraffin wax, sectioned at 5 μm in thickness, and H&E stained. For TEM, formalin-fixed liver samples from selected treated, control, and vehicle control rats were postfixed in 2.5% glutaraldehyde with 0.1 M cacodylate buffer, osmicated, processed to Spurr’s resin (Electron Microscopy Sciences Inc., Hatfield, PA), sectioned on an ultramicrotome (Leica Microsystems Inc., Buffalo Grove, IL), collected on grids, stained with uranyl acetate and lead citrate, and visualized on a JEM-1230 TEM (JEOL, Boston, MA). Digital images were captured using an Orius digital camera (GATAN Inc., Warrendale, PA). Stained H&E sections and electron microscope digital images were evaluated morphologically by a board-certified veterinary pathologist.
Bioanalytical Procedures for Amiodarone Concentration
Amiodarone was extracted from 10 μl rat plasma by protein precipitation using 100 μl of acetonitrile. After vortex mixing and centrifugation, 50 μl of the supernatant was diluted with 50 μl of water. Concentrations of amiodarone were determined against a standard curve over the range of 10–10,000 ng/ml by ultra high-performance liquid chromatography–tandem mass spectrometry (UHPLC-MS/MS).
The LC-MS/MS system consisted of an ACQUITY™ UPLC integrated system from Waters (Milford, MA) and a triple quadrupole mass spectrometer API4000 (Applied Biosystems/MDS-Sciex, Concord, Ontario, Canada). The analytical column was BEH C18, 2.1 mm × 50 mm with 1.7 μm particle size from Waters. The column was held at room temperature, and the sample compartment was at 10°C. Mobile phase A consisted of 10 mM ammonium formate adjusted to pH 3.0 with formic acid, and mobile phase B was 90/10 acetonitrile/methanol (v/v). A linear gradient ran from 55% B to 85% B for 0.8 min and held at 85% until 1.2 min, after which the system returned to initial condition. The total run time was 1.5 min, and the flow rate was maintained at 0.75 ml/min. An API4000 with a turboionspray was operated in the positive ionization mode, with the multiple reaction monitoring (MRM) transition of m/z 648–58 for amiodarone analysis.
Mrp2 IC50 Assay
Amiodarone, benzbromarone (an mpr2 inhibitor used as a positive control inhibitor), and estradiol glucuronide (EG, used as a probe substrate) were supplied by Sigma. [3H] estradiol 17-β-
Membrane vesicles prepared from recombinant baculovirus-infected Sf9 cells expressing the rat mrp2 transporter, and reagent kits were obtained commercially (Life Technologies, Carlsbad, CA). Stock solutions of amiodarone and benzbromarone were prepared in dimethyl sulfoxide (DMSO) and stored at 20°C. Working solutions of amiodarone were prepared at 10× concentrations of 1, 3, 10, 30, 100, 300, and 1,000 μM by diluting the appropriate DMSO stock solution in reaction buffer (50 mM MOPS-Tris, 70 mM KCl, and 7.5 mM MgCl2) such that the final amount of organic solvent in each test system was constant and ≤1% (v/v). Working solutions of benzbromarone were prepared at a 10× concentration of 1,500 μM.
Mrp2 inhibition assays were performed using the assay conditions recommended by the vesicle manufacturer. Briefly, mrp2 membrane vesicles (50 μg protein) were preincubated for 10–15 min at 37°C in reaction buffer in the presence or absence of amiodarone or positive control inhibitor benzbromarone. Triplicate incubations were used for each concentration of test compound and positive control inhibitor. Following preincubation, reactions were initiated by the addition of 10 mM magnesium adenosine triphosphate) solution containing 50 μM [3H]EG. Additional incubations were performed in the absence of inhibitor and in the presence of 10 mM magnesium adenosine monophosphate solution containing 50 μM [3H]EG for passive transport. After a 5-min incubation, reactions were terminated by the addition of chilled stopping buffer (40 mM MOPS-Tris and 70 mM KCl). Incubations were transferred to a 96-well glass fiber filter plate and vacuum suctioned. The filter plate was washed 3 times with stopping buffer and filtered by vacuum suction, and 100 μL of microscint scintillation cocktail was added to it. An aliquot of the 50 μM [3H]EG substrate solution was removed and the radioactivity measured using a liquid scintillation counter.
Additionally, we did not collect any absolute or relative liver weight data, so we cannot confirm there were any increases in absolute liver weights during the study.
Statistical Analysis
Prism software 5.0 (Graphpad, San Diego, CA) was used for all statistical analysis. A one-way analysis of variance was used with a Tukey post hoc test for all multigroup (group# > 2) comparisons with significance of *p < .05, **p < .01, or ***p < .001. In case of comparisons between 2 groups, an unpaired two-tailed t-test was used with significance of *p < .05, **p < .01, or ***p < .001. Spatial mapping of gadoxetate washout rate was performed using MATLAB R2013b (Mathworks, Natick, MA).
Results
Amiodarone induced a significant decrease in BW by day 4 which was maintained through day 21 (**p < .01). Mean BW increased 14% in vehicle-treated rats, whereas the mean BW in amiodarone-treated rats decreased 8% during the 7-day period (Figure 2). During the 2-week amiodarone washout, the amiodarone group showed a larger absolute increase in BW (115 g) compared to the vehicle group (95 g), but these differences were not significant.
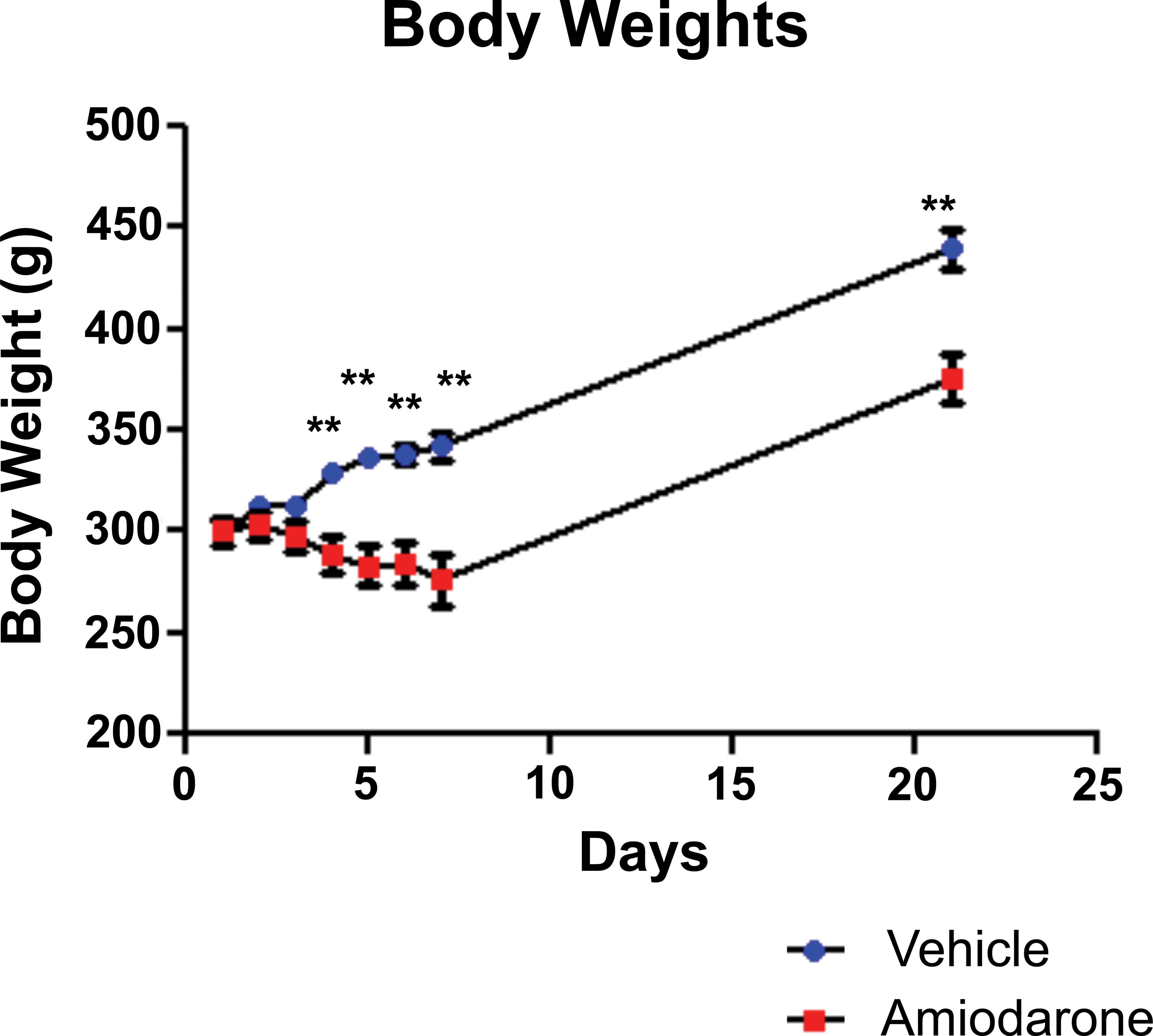
Rat body weights (BWs)—amiodarone induced a significant decrease in rat BW by day 4 which was maintained through day 21. The rate of BW increases upon the cessation of dosing until 2 W washout was similar between the vehicle and amiodarone groups. Data presented as mean ± SEM (**p < .01).
The concentration of amiodarone in blood after 7 days was 9.1 μmol. After the 2-week washout, the concentration of amiodarone was 0.09 μmol, indicating 2 weeks were adequate time to produce a washout effect of the drug. Blood was collected after each imaging session (baseline, day 7, and 2-week washout), and amiodarone did not induce any significant increases in plasma liver enzymes (AST/ALT/ALP), total bilirubin, or TBAs over the 7 days of oral dosing. After the 2-week washout of amiodarone, no significant differences were observed in plasma liver enzymes, total bilirubin, or TBAs (Table 1).
Blood Chemistry Parameters.
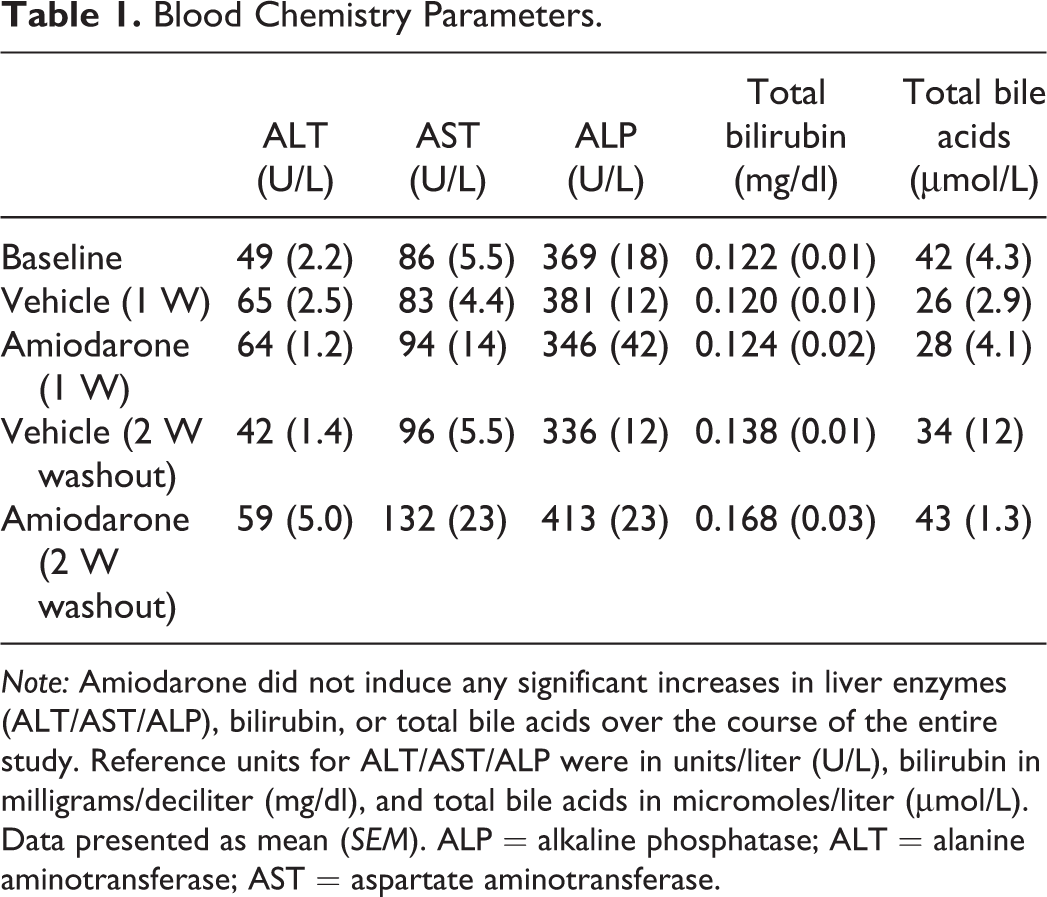
Note: Amiodarone did not induce any significant increases in liver enzymes (ALT/AST/ALP), bilirubin, or total bile acids over the course of the entire study. Reference units for ALT/AST/ALP were in units/liter (U/L), bilirubin in milligrams/deciliter (mg/dl), and total bile acids in micromoles/liter (μmol/L). Data presented as mean (SEM). ALP = alkaline phosphatase; ALT = alanine aminotransferase; AST = aspartate aminotransferase.
Light microscopic changes in amiodarone-treated rats included multifocal hepatocellular and Kupffer cell cytoplasmic vacuolation characterized by poorly defined vacuoles which were generally clear but occasionally contained a flocculant material. This is consistent with the microscopic changes associated with drug-induced PLD, and electron microscopy was used to confirm the presence of PLD.
Electron microscopic evaluation of livers of vehicle control rats showed no evidence of multifocal lysosomal lamellar bodies at day 7 (Figure 3A) as compared to the amiodarone-treated group where lysosomal multilamellar bodies were prominent in hepatocytes and Kupffer cells (Figure 3B), confirming the ultrastructural presence of PLD. Following the 2-week washout of amiodarone, the ultrastructural presence of multilamellar bodies was not found confirming the known reversible effects of PLD after the 2-week washout (Figure 3C).
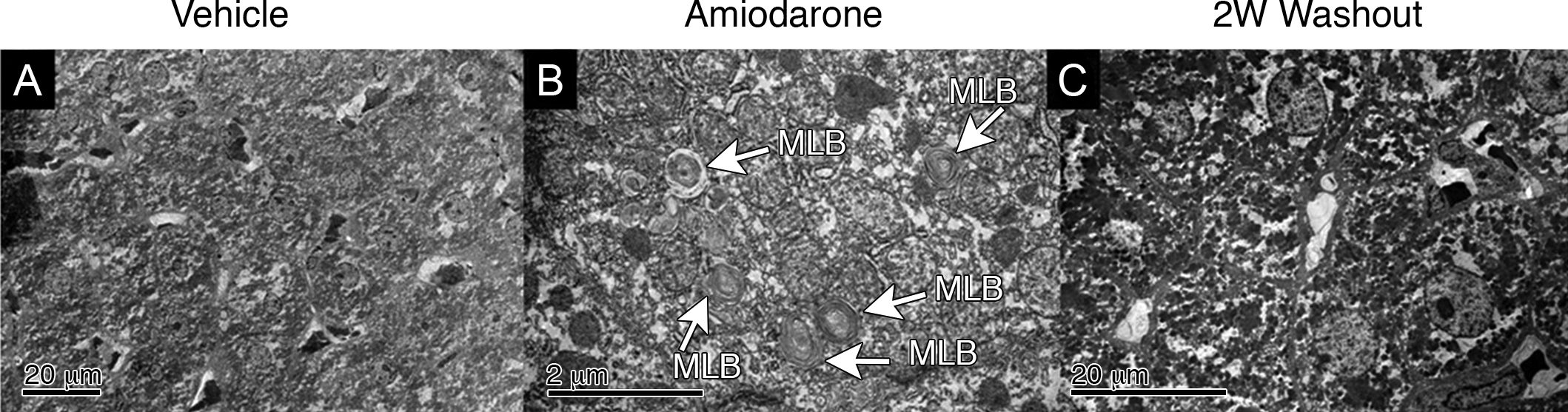
Electron microscopy of rat liver. (A) Representative vehicle liver showing no abnormalities detected upon the electron microscopy evaluation. (B) Representative amiodarone liver showing histological evidence of phospholipidosis with multifocal lysosomal multilamellar bodies after 7 days of oral dosing of amiodarone at 300 mg/kg/day. (C) Representative 2 W washout liver showing no evidence of PLD.
A representative series of coronal images at baseline and multiple time points postgadoxetate injection shows early peak SI in liver, occurring within 2–3 min postinjection followed by a steady decrease in mean signal over the remaining 60 min of the MRI acquisition (Figure 4). The gadoxetate uptake rate did not significantly change over the course of the study in either the vehicle or amiodarone group (Figure 5A). However, the gadoxetate washout rate was significantly decreased (*p < .05) in the amiodarone group after 7 days which increased back to baseline levels after the 2-week washout of amiodarone (Figure 5B). In the amiodarone-treated group, all rats (n = 8) showed a decrease in their gadoxetate washout rate after 7 days of dosing after which all rats allowed to recover during the 2-week washout showed an increase in their washout rate back to baseline levels (Figure 5C). The REmax was slightly decreased in the amiodarone group (ns; Figure 5D), and the T max was slightly longer in amiodarone group (ns; Figure 5E). The RE(t) profile at baseline (Figure 5F) is similar between groups as would be expected, given that at this baseline time point rats have yet to be dosed with vehicle or amiodarone. The RE(t) profile after 7 days of dosing with amiodarone (Figure 5G) shows the amiodarone group with a decreased peak RE(t) in conjunction with a more gradual washout profile as compared to the vehicle group. Both the uptake rate and REmax were not different between groups after 7 days of amiodarone treatment, suggesting similar rates, and the amounts of gadoxetate were entering hepatocytes, suggesting altered uptake kinetics by inhibition of uptake transporters is not contributing to the altered washout kinetics observed in this study. After the 2-week washout of amiodarone, the profiles between the vehicle and amiodarone groups appear similar (Figure 5H).
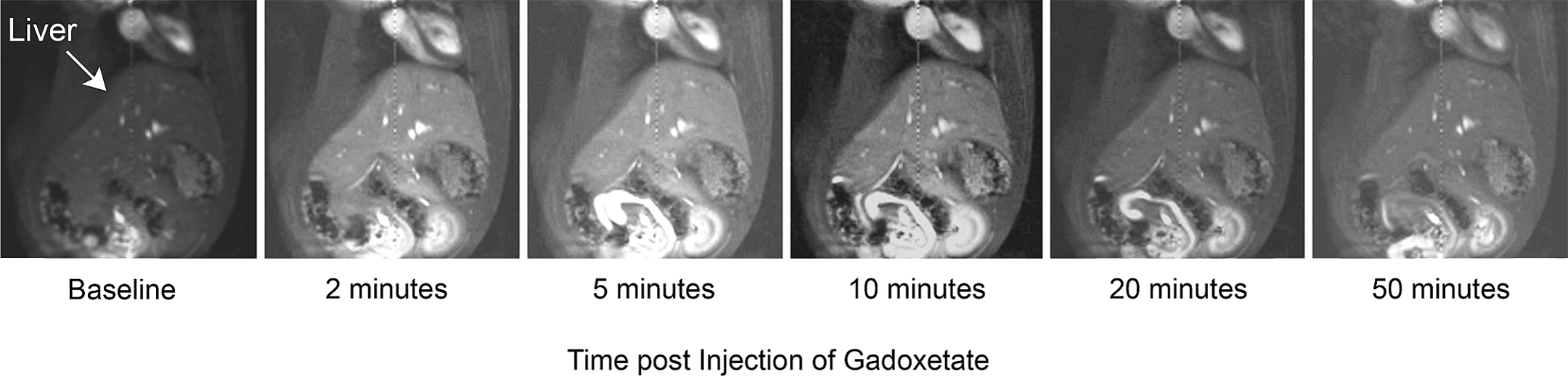
Representative MRI images illustrating gadoxetate signal intensity change in liver. A representative series of coronal images at baseline and specific times postgadoxetate injection showing signal increase and eventual decrease over the course of the dynamic contrast–enhanced magnetic resonance imaging experiment.
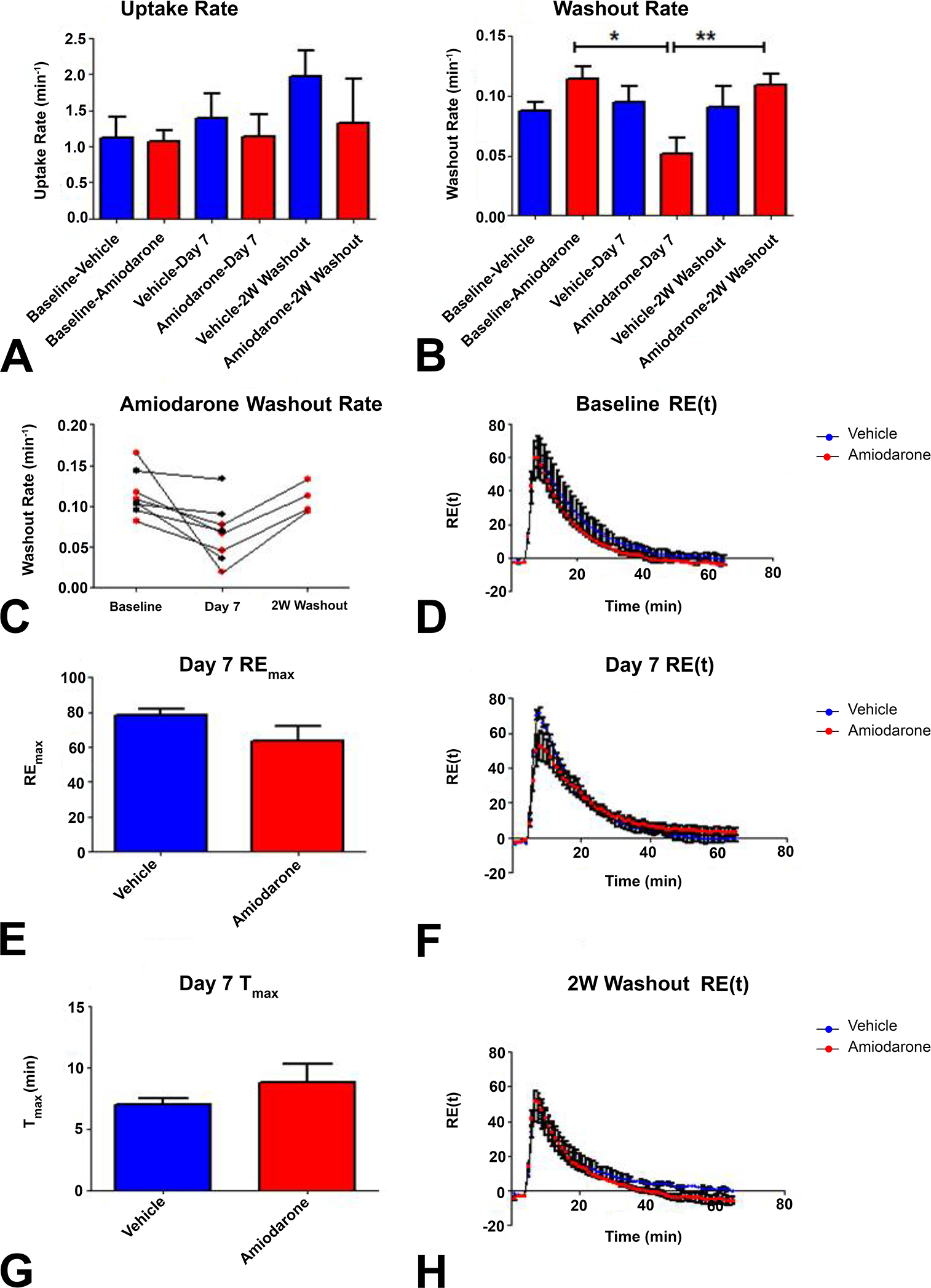
Gadoxetate kinetics. (A) Amiodarone induced no significant changes in gadoxetate uptake rate during the study. (B) Gadoxetate washout rate was significantly decreased in amiodarone group at day 7 and showed significant increases to baseline level after the 2 W washout of amiodarone. (C) Amiodarone group individual profiles for gadoxetate washout rate showing consistent decrease from baseline and subsequent increase after washout (red dots). Black dot animals were sacrificed for ultrastructural evaluation of PLD. (D) REmax was not different at day 7 between vehicle and amiodarone groups. (E) T max was not different between groups. (F–H) Baseline, day 7, and 2 W washout RE(t) profiles over the 65 min gadoxetate experiment. REmax = maximal relative enhancement. T max = time to reach maximal relative enhancement. Data presented as mean ± SEM (*p < .05 and **p < .01).
Color spatial maps of the gadoxetate washout rate of a representative vehicle- and amiodarone-treated rat show the decreased washout in amiodarone-treated liver (i.e., green/blue color overlay indicative of a decreased washout rate) after 7 days of oral dosing compared to the vehicle-treated animal (i.e., orange/red color overlay indicative of a normal washout rate; Figure 6A). In the same representative animals, the individual gadoxetate washout rates show the decrease at day 7 and the return to baseline levels after the 2-week washout of amiodarone (Figure 6B).
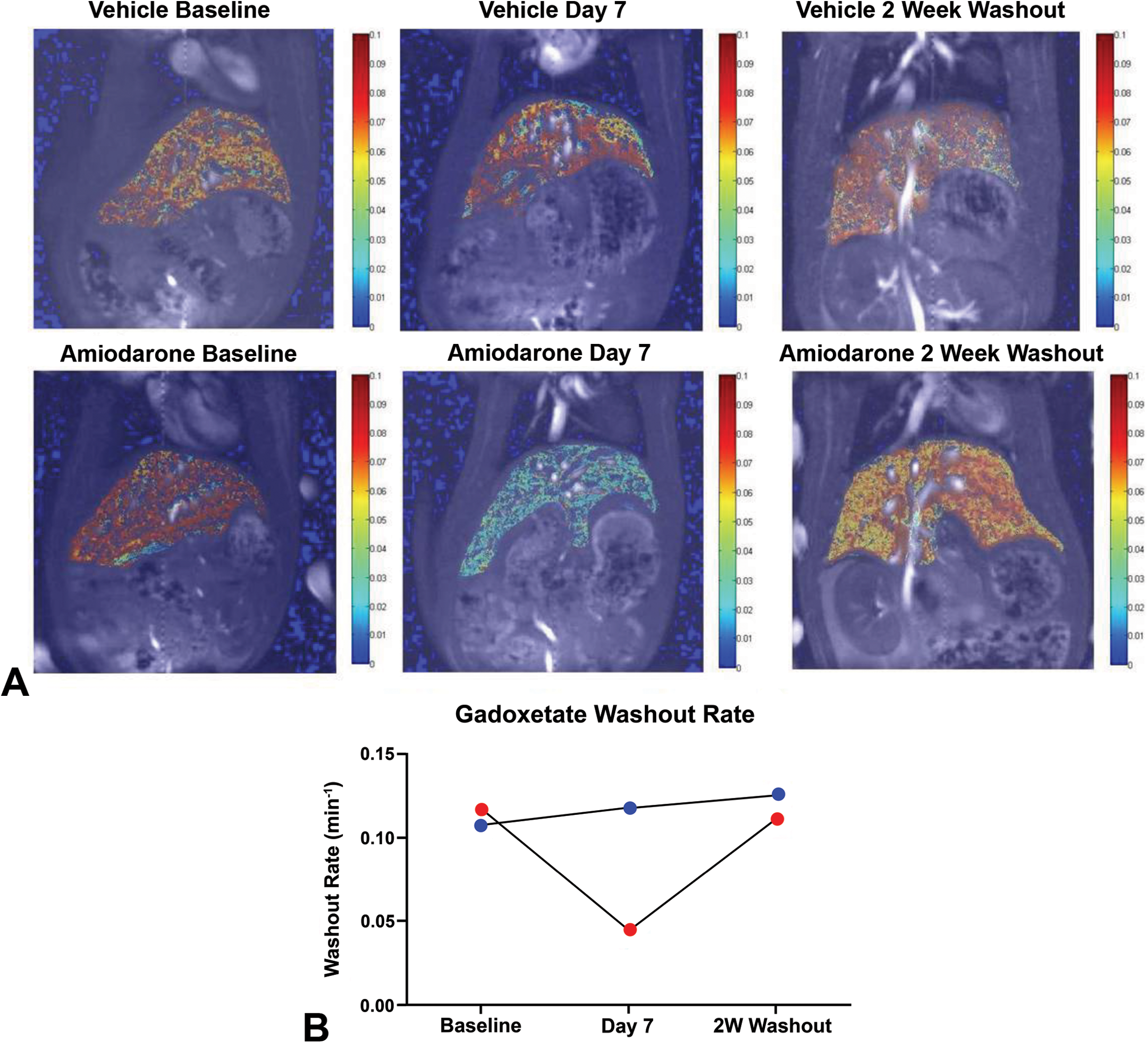
Spatial mapping of gadoxetate washout rate. (A) Color spatial maps of the gadoxetate washout rate in a representative vehicle and amiodarone rat. (B) Corresponding graph of the same animal’s washout rate highlighting the decrease at day 7, which returned to baseline level after the 2-week washout of amiodarone.
In order to confirm the effects observed on the gadoxetate washout rate are not the result of direct compound inhibition of amiodarone on mrp2, and the rat mrp2 IC50 was confirmed to be >100 μM (Table 2). Benzbromarone was used as a positive mrp2 inhibitor in this assay and was shown to inhibit [3H]EG uptake by ∼70%. This result confirms amiodarone did not directly inhibit mrp2, given the peak concentration measured after 7 days was <10 μM in the study.
Inhibition of [3H]EG Uptake by Amiodarone in Rat mrp2 Membrane Vesicles.
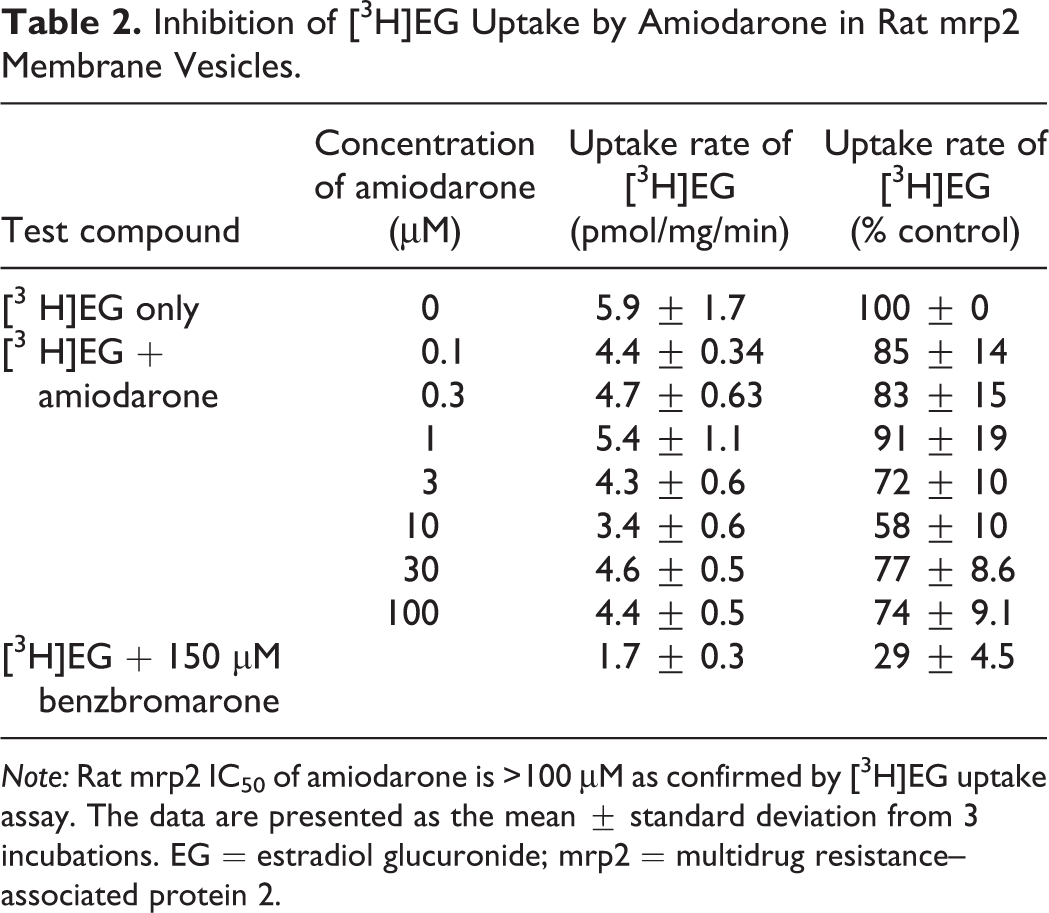
Note: Rat mrp2 IC50 of amiodarone is >100 μM as confirmed by [3H]EG uptake assay. The data are presented as the mean ± standard deviation from 3 incubations. EG = estradiol glucuronide; mrp2 = multidrug resistance–associated protein 2.
Discussion
Amiodarone induced a significant decrease in gadoxetate washout rate in liver, while inducing no changes in liver enzymes, bile acids, or total bilirubin, consistent with the lack of any toxicologically significant injury detected by light and electron microscopic evaluation. Amiodarone is not an mrp2 inhibitor in humans (Morgan et al. 2013), and the drug levels in this study did not reach a concentration required to inhibit mrp2 in rats. Because amiodarone is not a direct inhibitor of mrp2 at concentrations observed in this study, yet the liver gadoxetate washout rate was significantly decreased, the question of how PLD affects hepatobiliary transport of gadoxetate becomes paramount to understanding the functional differences induced by amiodarone in this study.
We do not have direct data from this study to address the question of intracellular accumulation of amiodarone in the hepatocyte. However, we can indirectly address this question citing Felser et al. (2013), where the authors found that in HepG2 cells exposed to amiodarone for 24 hr, amiodarone induced cytotoxicity and apoptosis at 20 μM and 50 μM concentrations, respectively. Given that we did not observe any biomarkers of cytotoxicity or evidence of apoptosis and also did not observe these changes on H&E, we can infer the drug level reached intracellularly were below 50 μM, which is well below the IC50 for mrp2 of amiodarone, so we do not believe the washout effects observed with gadoxetate are directly attributable to drug inhibition of the mrp2 transporter.
Due to the nature of PLD being reversible, we wanted to evaluate hepatobiliary function following a washout time period to determine if observed hepatobiliary functional changes would recover following the washout of amiodarone. We observed a strong association between the electron microscopy evidence showing the ultrastructural presence of multifocal lysosomal lamellar bodies associated with PLD and decreased liver gadoxetate washout rates as all animals showed a significant decrease in their gadoxetate washout rate after the 7 days of amiodarone dosing. While we did not observe any significant decreases in gadoxetate uptake rates in this study, there was a decreased peak RE(t) in the amiodarone-treated group suggesting a potential amiodarone-induced effect on oatp-mediated uptake of gadoxetate (Ferslew and Brouwer 2014). Recently published work evaluating different hepatic inducers of PLD in rat sandwich cultured hepatocytes revealed after 48 hr chronic administration of amiodarone at 10 μM, no changes in the biliary excretion index (BEI)% for taurocholate (oatp-specific probe) or rosuvastatin (mrp2-specific probe). Interestingly, as PLD-inducing drugs are known to accumulate in hepatocytes, a higher concentration (50 μm) of amiodarone was evaluated where a significant decrease in the BEI % was observed for both taurocholate and rosuvastatin, indicating chronic exposure to PLD-inducing amiodarone leads to functional changes in hepatic oatp- and mrp2-mediated uptake and excretion, respectively (Ferslew and Brouwer 2014).
The association between the ultrastructural presence of lamellar bodies and the gadoxetate washout rate was reinforced after the 2-week washout of amiodarone where we observed no ultrastructural evidence of PLD at this time point, while the gadoxetate washout rate returned to baseline levels. These results suggest the ultrastructural presence of PLD may be associated with the hepatobiliary function evidenced by the decreased washout rate of gadoxetate. However, this association between the ultrastructural presence of PLD and its subsequent effect on gadoxetate washout rate would have to be followed up with future studies using well-known inducers of hepatic PLD to determine if the functional consequences observed in this study represent a “class effect” of PLD.
The possibility exists that the altered gadoxetate kinetics observed in this study could be due to an amiodarone-induced effect on mitochondrial function. Because mrp2 function is an adenosine triphosphate–dependent process, the possibility of amiodarone transiently altering mitochondrial function/energetics would need to be investigated as a potential cause of the altered hepatobiliary function observed in this study (Beavis 1989). Amiodarone also has been shown to induce metabolic gene transcriptional changes by increasing cholesterol biosynthesis and fatty acid metabolism (Mesens et al. 2012). The question arises how metabolic gene changes might affect what is driving these functional differences we observe with gadoxetate in the presence of PLD?
In a 2006 review paper of PLD, Reasor and colleagues state, “There is no conclusive evidence to directly link the presence of CAD-induced PLD to impaired cellular or tissue function in intact organisms” (p. 577). The novel results of this study suggest PLD may be associated with hepatobiliary function as evidenced by the significant decrease in the gadoxetate washout rate which resolved back to baseline level after the 2-week washout of amiodarone. This is the first study to show the application of gadoxetate DCE-MRI to detect experimentally induced hepatobiliary functional changes associated with PLD. Gadoxetate DCE-MRI offers the potential to assess hepatobiliary function in vivo in preclinical drug development to potentially help mitigate functional liabilities associated with compounds suspected by in vitro or in silico tests to alter transporter function which may be known to induce hepatic PLD. While these results suggest PLD itself may exert transient consequences on hepatobiliary function in the absence of abnormal established biomarkers of toxicity (liver enzymes), the direct cause/effect relationship between PLD and function has not been established. The potential clinical utility of using gadoxetate DCE-MRI in patients suspected of having PLD may offer a valuable noninvasive tool to offer further insight into the hepatobiliary functional consequences of PLD.
Footnotes
Acknowledgments
The authors would like to acknowledge the excellent technical support by Debra Paul, Edward, Long, and Kathy Morasco of the Department of Laboratory Animal Sciences and Tracy Gales of Safety Assessment for their contributions to this study.
Author Contributions
Authors contributed to conception or design (SL, RP, RM, and BJ); data acquisition, analysis, or interpretation (SL, ML, LW, CG, RP, RM, and BJ); and drafting the manuscript (SL). All authors critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GSK and all authors have no personal or financial interest in any of the MRI imaging technology, imaging contrast agent, or compound (amiodarone) used in this study. The clinically available liver-specific MRI contrast agent (Eovist/Primovist) is manufactured by Bayer HealthCare. Cordarone (clinically available amiodarone HCl) is manufactured by Pfizer. All MRI instrumentation/hardware/software was developed/manufactured by Bruker Biospin (Billerica, MA). GSK and the authors did not develop any of the existing imaging technology/contrast agents used in this study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.