Abstract
Acute respiratory distress syndrome (ARDS) is a severe pulmonary reaction requiring hospitalization, which is incited by many causes, including bacterial and viral pneumonia as well as near drowning, aspiration of gastric contents, pancreatitis, intravenous drug use, and abdominal trauma. In humans, ARDS is very well defined by a list of clinical parameters. However, until recently no consensus was available regarding the criteria of ARDS that should be evident in an experimental animal model. This lack was rectified by a 2011 workshop report by the American Thoracic Society, which defined the main features proposed to delineate the presence of ARDS in laboratory animals. These should include histological changes in parenchymal tissue, altered integrity of the alveolar capillary barrier, inflammation, and abnormal pulmonary function. Murine ARDS models typically are defined by such features as pulmonary edema and leukocyte infiltration in cytological preparations of bronchoalveolar lavage fluid and/or lung sections. Common pathophysiological indicators of ARDS in mice include impaired pulmonary gas exchange and histological evidence of inflammatory infiltrates into the lung. Thus, morphological endpoints remain a vital component of data sets assembled from animal ARDS models.
Keywords
Introduction
Acute respiratory distress syndrome (ARDS) was first described in humans nearly 50 years ago during the Vietnam War, when it was referred to as “shock lung” or “DaNang lung” (Ashbaugh et al. 1967). This severe clinical condition requires hospitalization and can be incited by a plethora of causes. Major inciting causes of ARDS can be grouped into 2 pathophysiological categories: (1) direct pulmonary pathogenesis, including bacterial and viral pneumonia, inhalation injury, aspiration pneumonitis, or thoracic trauma that includes trauma to lung parenchyma, and (2) indirect extrapulmonary pathogenesis, such as extrathoracic sepsis, trauma, shock, burn injury, transfusion, and others.
ARDS is very well defined in humans by a list of clinical parameters. The typical signature includes an acute onset, radiological evidence of bilateral cellular infiltration into pulmonary tissue, a ratio of arterial partial pressure of oxygen (PaO2) to the fraction of inspired oxygen (FiO2) of less than 300 (i.e., pronounced hypoxemia), and no clinical evidence for elevated pulmonary arterial pressure (Bernard et al. 1994). This definition has been challenged and subsequently redefined several times (Ferguson et al. 2005; Raghavendran and Napolitano 2011; Ranieri et al. 2012). Most recently, a panel of clinical experts endorsed by the American Thoracic Society (ATS) established specific cutoffs for PaO2/FiO2 ratios that classify patients as having mild, moderate, or marked ARDS (Ranieri et al. 2012). In addition, this so-called Berlin definition recommended the abolishment of the term “acute lung injury” and removed ancillary variables besides PaO2/FiO2 ratios from the definition of ARDS on the grounds that they are not predictive of patient mortality (Ranieri et al. 2012). Even these revised and improved criteria of ARDS pose practical diagnostic challenges in humans. The main confounders in clinical diagnosis are dynamic and rapid changes in blood oxygenation levels, which are influenced substantially by positive end-expiratory pressure (PEEP), as well as the subjectivity of evaluation of chest radiographs (Villar et al. 2007; Rubenfeld et al. 1999; Meade et al. 2000).
Recent retrospective studies have documented that the incidence of ARDS has been roughly halved (from 81 to 38 cases per 100,000 people per year) in the timespan from 2001 to 2008 (Li et al. 2011). The study’s authors concluded that this trend was due to improved treatment protocols for critical care patients. These include institution of low tidal volume ventilation, a restrictive transfusion policy, and increased staffing of pulmonary intensive care wards (Li et al. 2011). However, a controversy continues to exist in the literature regarding trends of ARDS mortality rates, with some studies reporting decreased mortality rates over the last few decades, whereas others find no changes (reviewed in Blank and Napolitano 2011). This is largely due to the challenges experienced in attempting to diagnose this condition and the poor correlation between clinical and pathologic diagnoses, as well as due to variation in inclusion/exclusion criteria of patient selection in each study (reviewed in Blank and Napolitano 2011).
The current review has 2 main objectives. The first is to introduce the principal features that are used to define ARDS in experimental animal models. The second is to provide an overview of mouse models used to investigate mechanisms driving various aspects of the ARDS response and treatment options designed to counter 1 or more of these factors. Consideration of these interrelated aims will permit researchers to devise more appropriate studies, apply better experimental and analytical methods, and increase the pace at which potential ARDS breakthroughs may be explored.
Principal Features Used to Define ARDS in Animal Models
Consensus has always been difficult to obtain regarding the characteristics of ARDS in experimental animal models. While the clinical criteria established in humans may be translated to laboratory animals (Aeffner et al. 2012; Traylor, Aeffner, and Davis 2013), some of the experimental procedures are technically challenging, and the specialized equipment for lung function testing in animals is only available in select laboratories. Furthermore, certain measurements can be incompatible with other desirable endpoints, which make it difficult to achieve an experimental design that measures the entire spectrum of potential diagnostic parameters in a single animal.
In 2011, a committee assembled by the ATS published a workshop report proposing a list of potential indices that should comprise the main features of experimentally induced ARDS in animals (Table 1). Important traits include (1) histological evidence of injury to pulmonary cell populations and tissues, (2) altered integrity of the alveolar capillary barrier, (3) the presence of an inflammatory response as indicated by infiltrating leukocytes, and (4) physiological changes consistent with pulmonary dysfunction (Matute-Bello et al. 2011). To diagnose ARDS in an animal model, 3 of these 4 criteria must be present. In addition, the ATS committee subclassified findings as “very relevant” or “somewhat relevant” for each criterion. For example, when assessing the histological evidence of tissue injury, “accumulation of neutrophils in the alveolar or interstitial space” and “thickening of alveolar walls” are both considered to be “very relevant,” whereas “atelectasis” and “hemorrhage” are categorized as “somewhat relevant” (Matute-Bello et al. 2011). The ATS committee proposed that at least 1 “very relevant” point must be present in 3 of the 4 overarching criteria to confirm a diagnosis of ARDS in research animals. Possible tools available to address the different criteria proposed by the ATS will be addressed later in this review.
A list of ARDS features in animal models according to the ATS workshop report.

Source: Matute-Bello et al. (2011).
Note. ARDS = acute respiratory distress syndrome; ATS = American Thoracic Society; BALF = bronchoalveolar lavage fluid; FiO2 = fraction of inspired oxygen; MPO = myeloperoxidase; PaO2 = arterial partial pressure of oxygen.
aFeature cannot be applied to murine models of ARDS.
A number of small and large animal species have been utilized to study ARDS. These include, but are not limited to, mice (Gong et al. 2015; Letsiou et al. 2015), rats (Xin et al. 2015; Smith et al. 2015), rabbits (Walther et al. 2014; Uzawa et al. 2015), guinea pigs (Kabir et al. 2009; C. Q. Yang et al. 2009), dogs (Y. Yang et al. 2014), sheep (Engel et al. 2015; Asmussen et al. 2014), pigs (Emr et al. 2015; Ziebart et al. 2014), horses (Fard et al. 2014), and nonhuman primates (Hukkanen et al. 2009; Fremont et al. 2008). Ideally, animal models of ARDS should reproduce the pathophysiological changes that occur during the development of ARDS in humans, as well as follow a similar progression over time (Matute-Bello, Frevert, and Martin 2008). However, no single model has been developed that can fully recapitulate all aspects of human ARDS (Matute-Bello, Frevert, and Martin 2008; Martin and Matute-Bello 2011; Bastarache and Blackwell 2009). For example, the presence of comorbidities and the effects of concurrent therapeutic interventions such as fluid therapy, parenteral nutrition, and prolonged ventilation are rarely modeled experimentally (Matute-Bello et al. 2011; Bastarache and Blackwell 2009). However, animal models incorporating several inciting stimuli are more likely to reflect human ARDS, which often is associated with 2 or more simultaneous insults (two-hit hypothesis; Rotstein 2003). It is therefore crucial to be aware of the limitations of each model and to have in-depth knowledge of the particular aspects of ARDS, which are being modeled with each different approach. This ensures that the selected test is the most appropriate choice for answering the research question being asked. While each animal species possesses advantages and disadvantages as an experimental platform for investigating ARDS, this review will focus on the mouse, which is the most commonly used animal model for ARDS research. The preeminence of mice in this regard is shown by the number of ARDS publications in which mice are used as an experimental model (>290 publications listed on PubMed since 1991).
A number of differences in gross and microscopic anatomy exist between the respiratory tract structures of mice and humans (Teuting and Dintzis 2011). These differences are listed in Table 2. The structural divergence between the 2 species can affect a number of analytical endpoints and thus needs to be considered, especially in evaluating histological features of ARDS. The implications of variations in lung structure thus require careful consideration when seeking to extrapolate data acquired in mice to predict potential responses in humans.
Comparative gross and microscopic anatomy of human and mouse respiratory tracts.
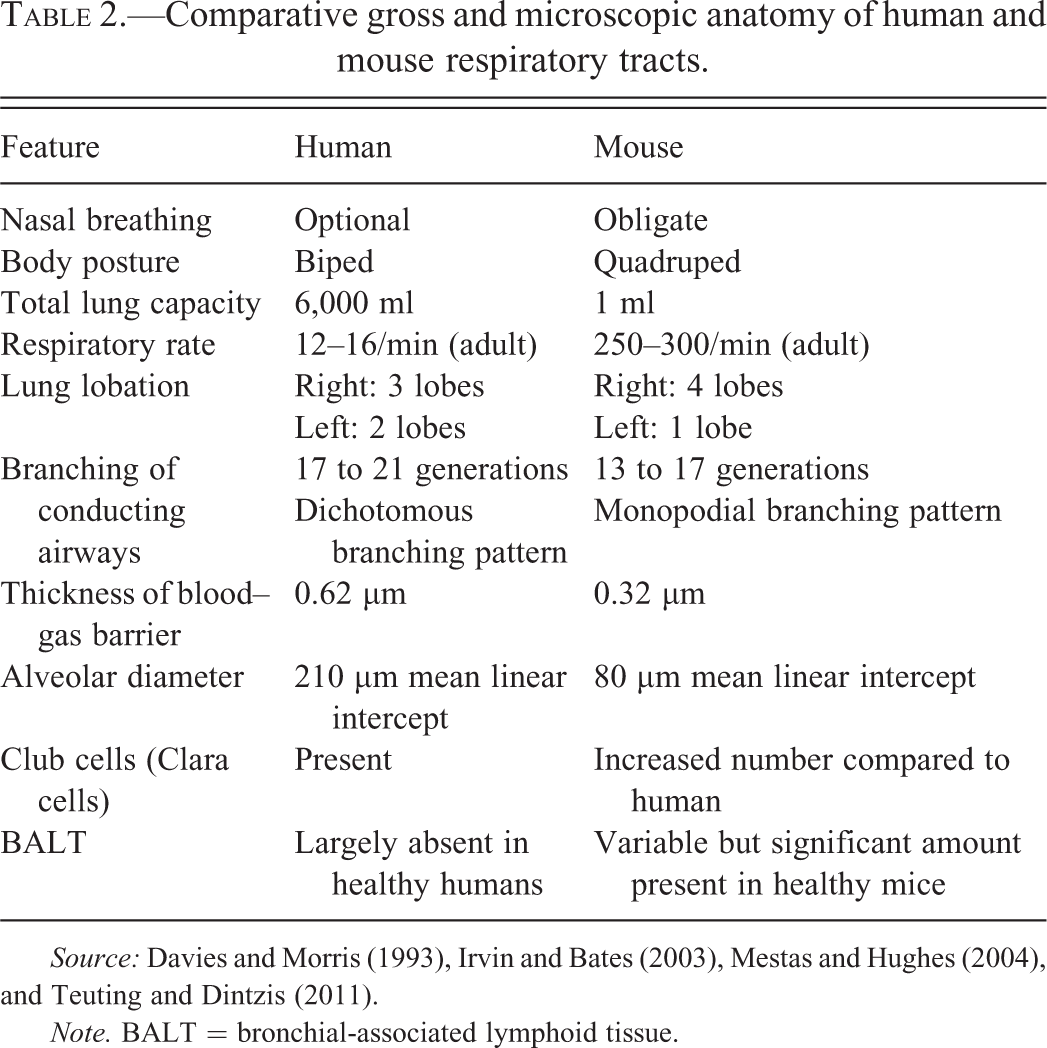
Source: Davies and Morris (1993), Irvin and Bates (2003), Mestas and Hughes (2004), and Teuting and Dintzis (2011).
Note. BALT = bronchial-associated lymphoid tissue.
Another important extrapulmonary difference between animals and humans that needs to be considered is the character of the innate immune response (Legrand, Weijer, and Spits 2006). This arm of the immune system is responsible for the rapid initiation and fast progression of acute diseases like ARDS. Specific factors that must be addressed when translating animal data to estimate human innate immune responses include differences in production of nitric oxide (Schneemann et al. 1993; Schromm et al. 2000; Thoma-Uszynski et al. 2001) and chemokines (Zlotnik and Yoshie 2000; Tsai et al. 2000) as well as expression of chemokine receptors (Fan et al. 2007) and Toll-like receptors (Rehli 2002). Large differences exist among different mouse strains and genetically engineered mice with regard to the immune system (reviewed in Sellers et al. 2012; Seymour, Sundberg, and Hogenesch 2006), and these differences can impact the pattern of lesions when modeling ARDS (Matute-Bello, Frevert, and Martin 2008; Goldman et al. 2014).
The majority of mouse models for ARDS focus on reproducing known risk factors that have been identified in human patients and therefore aim to injure the alveolar epithelium, the capillary endothelium, or both. Intratracheal administration of acid or bleomycin, prolonged hyperoxia, and prolonged mechanical ventilation at high tidal volume all induce ARDS by causing direct damage to the bronchoalveolar epithelium. Similarly, intravenous injection of oleic acid or endotoxin results in direct damage to the pulmonary vascular endothelium. Experimental methods of targeting both endothelium as well as alveolar epithelium are ischemia/reperfusion models, sepsis models, and models secondary to peritonitis (cecal ligation and puncture; reviewed in Matute-Bello, Frevert, and Martin 2008). These latter strategies rely on the production of multiple endogenous signaling molecules that control the innate immune response. The 3 most commonly used mouse models of ARDS over recent years are mechanical ventilation with high tidal volumes or high peak inspiratory pressures, pulmonary or systemic administration of endotoxin (lipopolysaccharide), and airway instillation of live bacteria (Matute-Bello et al. 2011). While not included in this list, induction of acute lung injury using influenza virus instillation also has been well established (Dong et al. 2012; T. Xu et al. 2006; Traylor, Aeffner, and Davis 2013; Zhang et al. 2012; Aeffner, Woods, and Davis 2014; Dai et al. 2014). These models may be used in tandem.
When utilizing genetically engineered mice, it is crucial to select the appropriate controls to differentiate true findings related to ARDS induction from incidental effects related to the strain-specific genetic background (Sigmund 2000). The current gold standard is to use wild-type offspring of heterozygous breeding pairs to create an age- and sex-matched, wild-type inbred strain by backcrossing for at least 20 generations. When this is not possible, congenic mice for which 10 generations of backcrossing has yielded a background homogeneous at nearly all (99.8%) gene loci should be used. Finally, it is crucial that mice of both strains are housed under the same environmental conditions and are free of all murine pathogens (Matute-Bello, Frevert, and Martin 2008).
Features, Measurements, and Practical Aspects of ARDS Research in Mice
Table 1 lists all major features and sub-features of ARDS animal models, including those described as “very relevant” and “somewhat relevant” by the ATS (Matute-Bello et al. 2011). For a platform to be considered as a useful ARDS model, 3 of these major criteria need to be fulfilled by demonstrating at least 1 very relevant subfeature. The remainder of this review describes the application of these criteria in mouse ARDS models.
Main Feature: Measurement of Histological Evidence of Tissue Injury
To collect mouse lungs for histological evaluation, animals should be euthanized and the specimen harvested as soon as possible thereafter. Whenever possible, a euthanasia route should be chosen that does not target the lungs, such as intravenous injection of an overdose of anesthetic, as opposed to, for example, CO2 narcosis. Analysis of the pulmonary parenchyma may be facilitated by partial exsanguination of the animal during euthanasia to decrease the amount of blood in lung sections. This can be achieved by an incision in the axillary region or by opening the abdominal cavity to cut the aorta while the heart is still beating. Exsanguination is especially helpful when immunohistochemical staining of sections is planned. The cytoplasm of intact erythrocytes often exhibits substantial background labeling when immunoperoxidase-based procedures are employed. Moreover, clotted blood regularly results in nonspecific binding of primary antibodies.
Processing of lung for histopathologic examination requires specialized but simple refinements to optimize preservation of fine pulmonary architecture. Intratracheal instillation of fixative results in better preservation of lung morphology, compared to whole body perfusion (Braber et al. 2010). To optimally prepare formalin-fixed, paraffin-embedded (FFPE) samples, the lungs need to be inflated via the trachea with neutral buffered 10% formalin or 4% paraformaldehyde instilled at room temperature under a constant pressure of 15 to 25 cm H2O. Inflation at a constant pressure can be achieved using a pressure column (Renne et al. 2001; Lum and Mitzner 1985; Braber et al. 2010). Inflation is easiest when the animal is in supine position (dorsal recumbency) via ventral tracheotomy with the exposed lungs remaining in situ within the open thoracic cavity (see Figure 1). After fixative instillation, the trachea should be tied off (usually about midway between the larynx and carina) to preserve inflated architecture. The lungs and heart should then be removed en bloc and submerged in fresh formalin for approximately 24 to 48 hr. Properly inflated specimens should still float at or just beneath the surface of the solution, even if pulmonary lesions are severe. This fixation technique ensures optimal preservation of parenchymal architecture for qualitative histological and immunohistochemical evaluation of the above-listed criteria. However, such routine processing results in significant organ shrinkage and, therefore, will have an impact on quantitative measurements such as lung volume (Lum and Mitzner 1985; Braber et al. 2010). If tissue needs to be stored for a longer period prior to processing, it should be switched to phosphate-buffered saline (PBS) and stored at 4°C to prevent overfixation, which will interfere with certain downstream procedures, such as the terminal deoxynucleotidyl transferase deoxyuridine triphosphate nick end labeling (TUNEL) apoptosis assay. Hence, to ensure intra- and inter-study comparability of data, fixation times should be consistent, and defined and documented in study protocols.
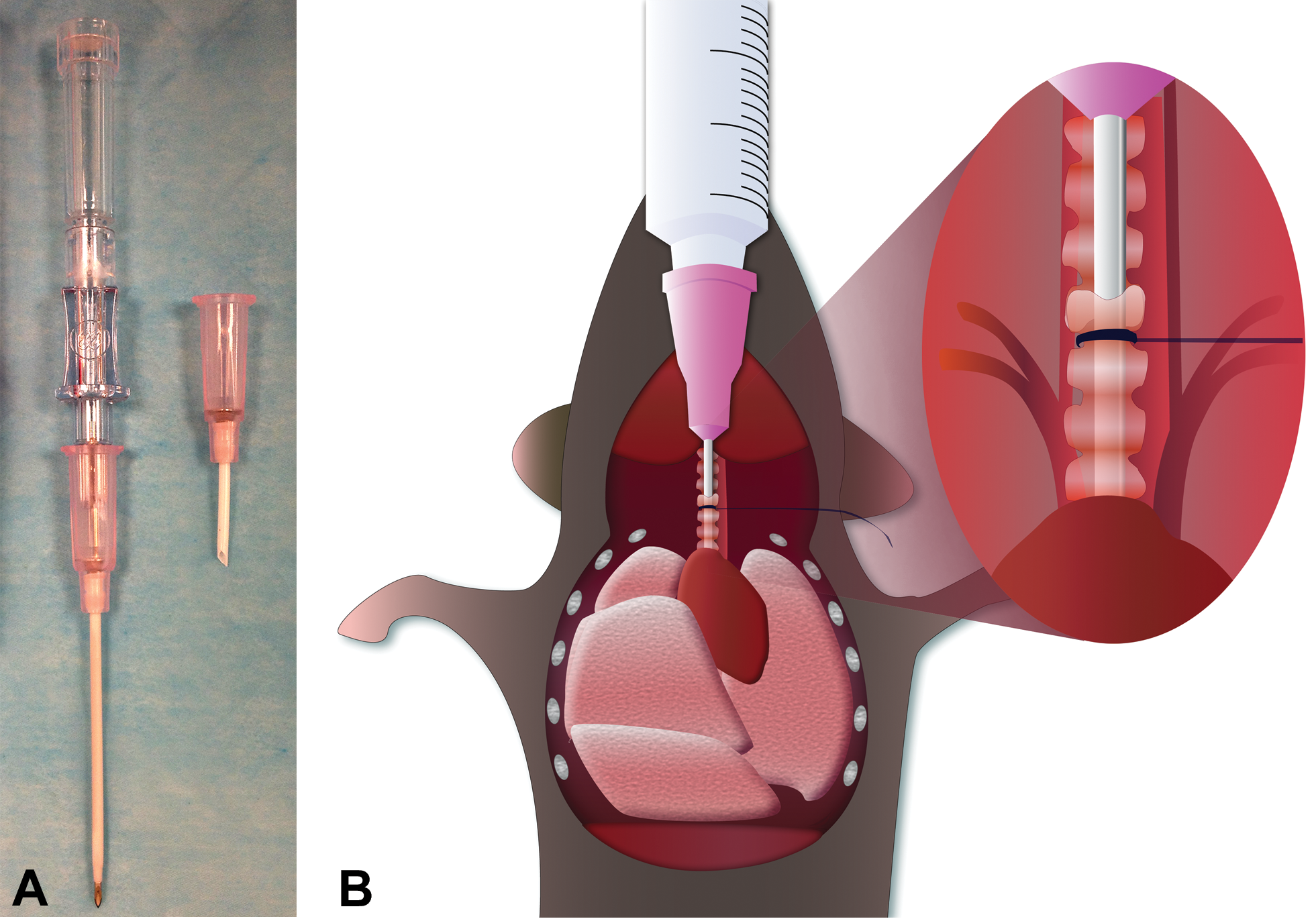
Method for routine tracheotomy and bronchoalveolar lavage (BAL) in the mouse. (A) Tracheotomy canulas can be custom-made by trimming an 18-gauge Teflon® catheter (left side) to an appropriate length (about 1–1.5 cm) and beveling the tip to allow for easy insertion into the trachea (right side) via an incision between 2 cartilage rings. (B) To harvest bronchoalveolar lavage fluid (BALF), the trimmed 18-gauge catheter is inserted into the trachea about midway between the larynx and the carina (i.e., the junction site of the trachea and main bronchi) and secured in place by a ligature. A syringe is connected to the catheter, and 1 ml sterile saline (pH 7.4, room temperature) is infused into the airway. (The image displays the lung with the lobes completely inflated by infusion of fluid.) To complete the procedure, the instilled fluid is aspirated again with the connected syringe. This process is immediately repeated 2 to 3 times with the same fluid to maximize the number of cells that are harvested.
If the entire respiratory tree/tract is available for histological evaluation, comprehensive examination is facilitated by placing the entire organ into a histological cassette with the ventral lobar surfaces oriented “down” in the cassette. This positioning will permit the histology laboratory staff to take sections that include longitudinal cuts showing lumina of multiple major conducting airways (Figure 2A). However, other orientations and sectioning techniques have been used, including the separation of all lung lobes followed by cutting longitudinal step sections parallel to the axis of the main lobar bronchus (Renne et al. 2001). This approach allows weighted scoring of histopathology findings in individual lung lobes as well as assessment of airways, but requires specialized training and is more time consuming.
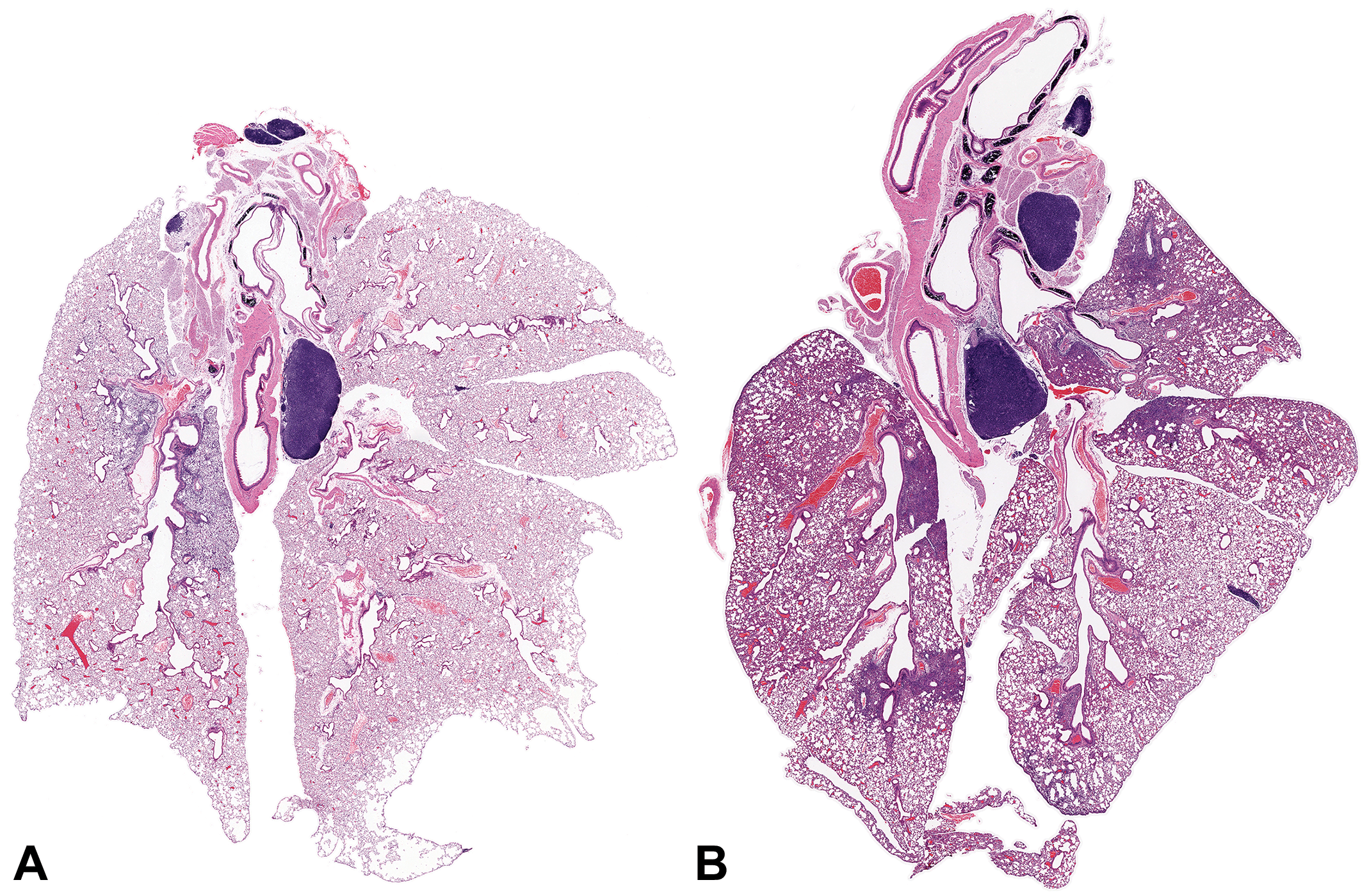
Representation of mouse lungs in the optimal orientation for histopathologic analysis. Both images display subgross images of hematoxylin and eosin (H&E)-stained sections of entire mouse lungs, with the left lobe oriented to the left of the images. The lungs were fixed by inflation in situ with neutral buffered 10% formalin, removed intact, and then immersed in fresh fixative for 48 hr. Fixed lungs were placed in a cassette with the ventral surface oriented down so that histologic sections would include longitudinal sections of major airways. (A) BALB/c mouse lung, 2 days after intranasal inoculation with the A/H1N1/WSN influenza virus. Mild emphysema-like artifacts (i.e., variably-sized clear spaces) introduced by inflating the lungs with formalin are apparent at the lobar edges, especially of the right lung lobes. (B) BALB/c mouse lung, 6 days after intranasal inoculation with A/H1N1/WSN influenza virus. Regional enhancement of lung inflammation is indicated by increased basophilia and reduced airway spaces in many regions, indicating an influx of inflammatory cells and fluid. The multifocal nature of the lesion indicates that this change may be missed if only parts of the lungs are available for histologic examination. The affected regions are more prominent in the left lung lobe because the animal was placed on its left side after viral inoculation to recover from anesthesia. Both panels: hematoxylin and eosin (H&E) stain; original objective magnification used: 0.5×.
In some cases, the need to preserve delicate molecules from the disruptive effects of typical fixative agents requires preparation of frozen lung sections. Unfortunately, preserving the ability to detect antigen structure and/or function usually reduces the degree to which cellular detail is maintained, although tissue shrinkage is generally minimal in the absence of fixation. In our experience, airway inflation should be performed with a 30% (by volume) sucrose solution mixed with 70% (by volume) Tissue-Tek® Optimal Cutting Temperature (OCT)™ medium (Sakura Finetek USA, Torrance, CA). Other dilution/diluent combinations, for example 1:1 or 1:2 mixtures of OCT with PBS, have been successfully used as well (Maeno et al. 2007; Fehrenbach et al. 1998). The chosen mixture should ensure that the rather viscous embedding material reaches the distal alveoli without the need to apply high inflation pressures, which will disrupt the delicate tissues of the distal lung (Mercer et al. 2008). In our laboratory, inflated samples are placed in pre-labeled plastic molds and covered with more OCT. They are then placed on dry ice or floated in liquid nitrogen on an aluminum foil weighing boat to accomplish rapid freezing. An alternative approach is to freeze cryo-preserved lung tissue in isopentane (2-methylbutane) that has been precooled in liquid nitrogen (Erickson et al. 2011; Bratthauer 2010). Frozen specimens are wrapped in clean aluminum foil and placed inside sealable plastic bags for storage at −80°C until sectioning (Mercer et al. 2008).
Histological sections of FFPE tissues are typically stained with hematoxylin and eosin (H&E) and evaluated using conventional histopathologic methods with respect to very relevant and somewhat relevant features of ARDS (Table 1). When diagnosing ARDS-like lesions in mice via histology, one has to remember that hyaline membranes, a common feature in human ARDS patients (Thille et al. 2013; Katzenstein, Bloor, and Leibow 1976), are rarely present in mice with ARDS despite clear evidence of increased alveolocapillary permeability in both species (Matute-Bello et al. 2011; Matute-Bello, Frevert, and Martin 2008). The cause of this discrepancy is currently unknown but may be related to the fact that human patients generally undergo mechanical ventilation for several days prior to death, which may result in development of more severe lesions such as hyaline membranes. In contrast, mice with ARDS are often euthanized in the early stage of disease for ethical reasons and are almost never subjected to prolonged mechanical ventilation analogous to that used in human intensive care units. The only study documenting hyaline membrane formation in mice describes them in animals that have succumbed naturally several days after influenza infection rather than being euthanized at a defined time point (Fukushi et al. 2011). This finding suggests that hyaline membrane formation may occur in mice under certain circumstances. However, the absence of detail in this latter report regarding the length of time between death and lung fixation also suggests that the hyaline membranes in these mice might in part represent the consequence of postmortem autolysis. Further work will be needed to confirm the presence and extent of hyaline membranes as a possible feature of mouse ARDS models.
Lesion patterns in mouse ARDS models may also be impacted by the choice of experimental methods. For example, intranasal and intratracheal treatment with liquid substances may produce patchy pulmonary lesions in mice. The effects may be manifested as multifocal lesions within all lung lobes or minimal changes on one side relative to extensive lesions on the other. This latter tendency is especially obvious if small volumes are instilled and/or if mice are placed on one side to recover from anesthesia (Figure 2B). This limitation showcases the need for histological evaluation to include the entire lung (as described earlier). When seeking to verify that the instilled agents have indeed reached the lower respiratory tract, colloidal carbon (1:100 dilution) can be added to the instillate and subsequently identified within alveolar macrophages by microscopic evaluation of histological sections (Mizgerd et al. 1996). This cross-check provides important verification that the experimental treatment has been successful, which is especially critical when quantitative morphological assessments are to be undertaken.
Accumulation of neutrophils in the alveolar or the interstitial space
The role of neutrophils in the pathogenesis of ARDS in human patients remains controversial. While many researchers view neutrophilic infiltrates and subsequent tissue injury as one of the main inciting features of ARDS (X. Zhou, Dai, and Huang 2012; Williams and Chambers 2014), the syndrome also develops in neutropenic patients or those who have recently undergone full-body bone marrow irradiation (Mokart et al. 2012; Azoulay and Darmon 2010; Ognibene et al. 1986; Laufe et al. 1986). This divergence is most likely due to the vast variety of stimuli that can incite the development of ARDS.
Most mouse models of ARDS are characterized by a prominent neutrophilic infiltrate into the alveolar space and interstitial septa, but the extent can be variable both within and across lobes. When low numbers of neutrophils are seen within alveolar spaces in histological sections, this observation should be verified by acquiring total and differential cell counts of bronchoalveolar lavage fluid (BALF). This cytological method is far more sensitive relative to routine histological examination because the entire lung volume is sampled and quantitative data are available for statistical analysis. Increased numbers of alveolar neutrophils are not visible upon histology if the total BALF neutrophil count is less than 106 (Matute-Bello et al. 2011). At the same time, it is important to point out that BALF neutrophil counts reflect the state of the intraalveolar population only and do not allow interpretation of the quantity of neutrophils in the interstitial compartment.
To assess cell infiltrates via histology, neutrophils within alveolar spaces, alveolar septa, and the interstitium are reviewed. Typical features evaluated using this qualitative analysis are the distribution and quantity of cells. Scores may be rendered for the lung as a whole or less commonly for specific compartments (e.g., alveolar spaces vs. interstitium). Alveolar septa can contain neutrophils within the interstitium as well as adhering to the endothelium. If the tiered grading scheme outlined subsequently is utilized, 1 to 5 neutrophils per high-power field (10× ocular with 40× objective magnification) result in a score of 1, more than 5 in a score of 2, and a complete absence of neutrophils is considered a score of 0 (Matute-Bello et al. 2011).
Formation of proteinaceous debris in the alveolar space
ARDS affects the air–blood barrier, leading to progressive breakdown of capillary wall and epithelial integrity, which permits leakage of protein-rich edema fluid (interstitial fluid and plasma proteins) into the alveoli (Sakka 2013). This proteinaceous fluid presents itself as a lightly eosinophilic material that ranges from homogenous to fibrillar (i.e., fibrin strands) in appearance in H&E-stained sections. While this evidence of pulmonary edema is readily visible and easy to interpret in humans and larger animals, the method of inflating mouse lungs can dilute protein-rich fluid or even wash it out of airways. When present, it should be interpreted as pulmonary edema, but its absence cannot be taken as proof that this change did not occur. Other methods, such as assessing protein content in BALF or measuring wet/dry weight ratios of the lung, are more meaningful methods of quantifying pulmonary edema and vascular leakage. Accordingly, additional methods should be used to confirm the presence or absence of histological findings in this context (see Measurement of Alterations of the Alveolar Capillary Barrier subsection).
Thickening of the alveolar wall
While inflation of murine lung samples is crucial to allow any credible histological examination, the assessment of alveolar wall thickness is especially dependent upon uniform inflation of airways across the entire specimen as well as among animals and groups. To minimize overinterpretation, only septa that have at least doubled in thickness versus unaffected septa in untreated control animals should be considered relevant for this criterion. In addition, all alveolar septa adjacent to airways or blood vessels should be omitted from the evaluation, as these high-pressure passages are naturally thickened by additional bands of collagen (Matute-Bello et al. 2011).
Enhanced injury as measured by a standardized histology score
The ATS workshop paper includes a semiquantitative and weighted histological grading scheme that can easily be utilized by a well-trained comparative pathologist for scoring mouse ARDS lesions. Furthermore, this system may be employed readily to perform objective coded (blinded) scoring of the extent of lung injury in multiple animals, which will enable statistical analysis of histological findings (Matute-Bello et al. 2011). For this purpose, the cited grading scheme mandates that at least 20 fields should be evaluated in each animal using a 40× (high power) objective. Only fields that are at least half-filled with alveoli and their lumina should be evaluated for scoring. To avoid interobserver variability, all sections within a study should be interpreted by the same experienced pathologist, and ideally over a short period of time. Because mice with ARDS do not develop hyaline membranes, this feature is omitted from the grading scheme, as a score of “zero” for this feature will result in the same overall composite score. The modified grading scheme is shown in Table 3. Examples of representative lesions for common scores are displayed in Figure 3.
Lung injury scoring system adapted for mouse models of ARDS.
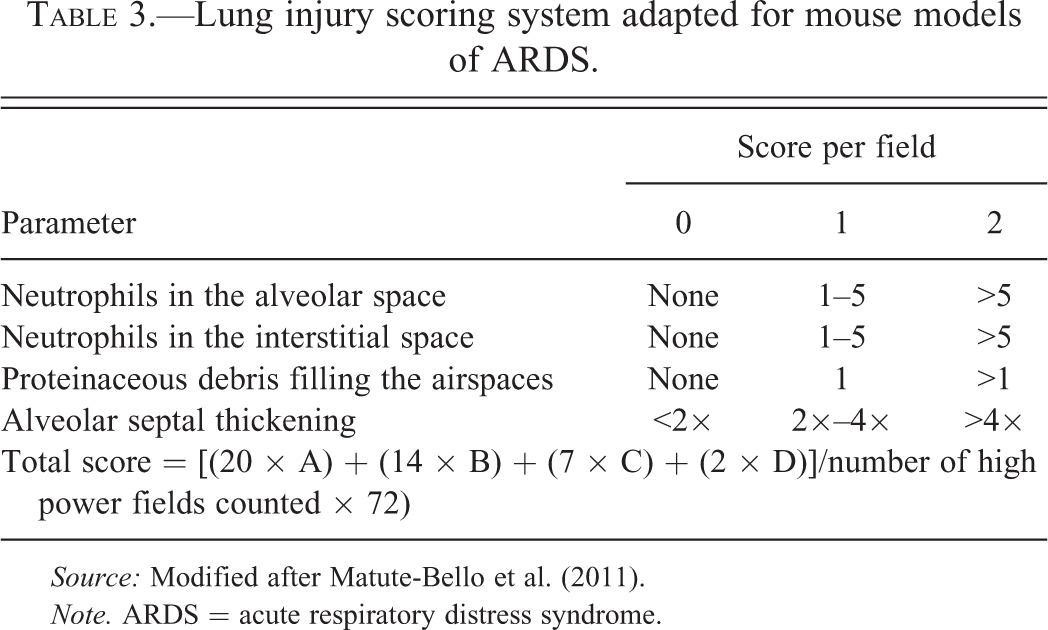
Source: Modified after Matute-Bello et al. (2011).
Note. ARDS = acute respiratory distress syndrome.
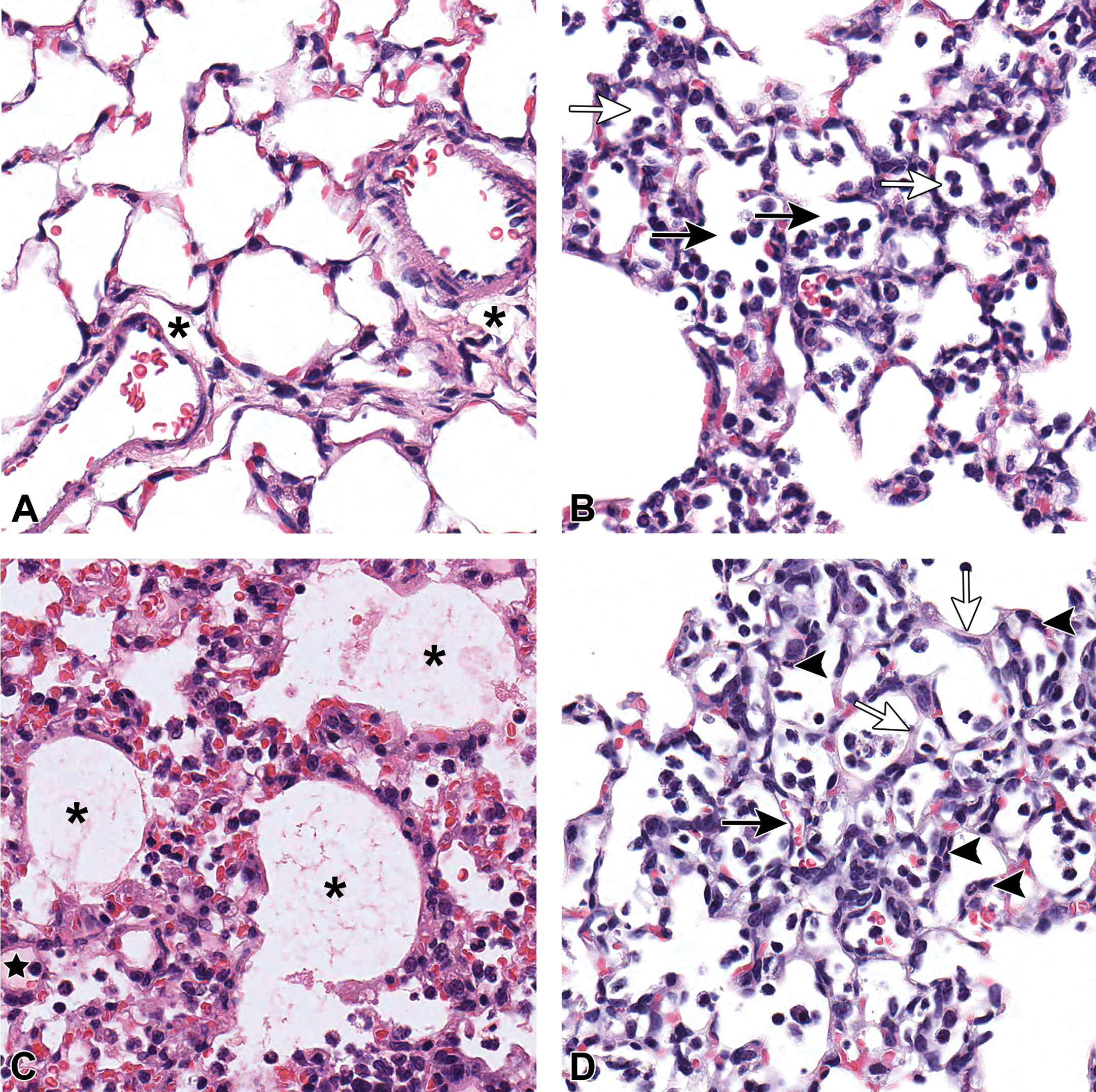
Representative histological features for common lesion scores observed in mouse models of acute respiratory distress syndrome (ARDS). (A) Normal lung (score = 0) displaying thin alveolar septa and largely cell-free airspaces. Individual alveolar macrophages can be observed in a few alveoli (not shown). Asterisk denotes the expansion of the perivascular space (also commonly seen in peribronchial tissue); this change should be interpreted as artifact. (B) Neutrophils in the alveolar spaces of mice with ARDS. Open arrows denote alveoli with 1 to 5 neutrophils, score = 1. Solid arrows denote alveoli with >5 neutrophils, score = 2. (C) Proteinaceous debris filling the alveolar spaces. Asterisks denote fibrillar to granular eosinophilic protein strands. Black star denotes homogenous eosinophilic material, representing protein-rich fluid. (D) Alveolar septal thickening and interstitial neutrophils. Solid arrow denotes septum of normal thickness (also displayed in panel A), score = 0. Open arrows denote septa >4× greater in thickness, score = 2. Solid arrow tips are pointing at interstitial neutrophils. The ARDS lesions in panels B, C, and D were induced by intranasal inoculation with A/H1N1/WSN influenza virus. All images: hematoxylin and eosin (H&E) stain; C57BL/6 mice; original objective magnification used: 20×.
Due to the fact that many mouse models of ARDS result in a patchy distribution of histological findings and pathological processes, entire lungs should be analyzed whenever possible to document and grade histological lesions. If this broad-based assessment is not possible, a variety of ways have been developed to help perform an unbiased evaluation of tissue in histological sections of limited area. Examples of such objective practices include successive random displacements or randomized generation of (x, y) coordinates as well as stratified sampling and Poisson-disk distribution (Matute-Bello et al. 2011).
Somewhat relevant histological evidence of tissue injury
The morphologic features listed as somewhat relevant to a diagnosis of ARDS have to be evaluated with caution in the mouse. Peracute pulmonary hemorrhage can be a euthanasia artifact and is indistinguishable from acute intravitam bleeding that occurs in diseased lungs prior to euthanasia. However, both can be distinguished from chronic hemorrhage by the presence of hemosiderin deposition in the latter lesion. Grossly observed changes in lung color, surface texture, and consistency can be indicators of pulmonary disease and should be documented. However, these changes can only be assessed prior to lung inflation with fixative solution. Moreover, atelectasis observed microscopically in the inflated mouse lung generally should be interpreted as a failure to fully inflate the lung and maintain this expanded state during tissue processing (i.e., a handling-induced artifact) as opposed to true atelectasis due to pulmonary disease.
Expanded peribronchial spaces are commonly seen in inflated murine lung sections. These clear zones typically are attributed to pressure-driven extravasation of fixative solution during processing and should not be misinterpreted as peribronchial or interstitial edema (Matute-Bello et al. 2011). In contrast, genuine alveolar edema can be recognized by its homogeneous and often fibrillar eosinophilic appearance within alveolar spaces.
Main Feature: Measurement of Alterations of the Alveolar Capillary Barrier
The formation of pulmonary edema begins to be morphologically visible with the increased prominence of interstitial space (i.e., accumulation of fluid) in the loose connective tissue immediately around the vasculature and bronchioles, followed by an increase in fluid in the interstitium (Parker and Townsley 2004). Alveolar flooding occurs only after the extravascular lung water in interstitial tissues increases by more than 35% (Wu et al. 1995). The development of edema does not occur at a constant rate or follow an even distribution throughout the lung (Wiener-Kronish and Broaddus 1993).
Translocation of a tracer from the blood into the pulmonary air spaces
A very accurate and widely used way to quantify vascular leakage in murine ARDS models has been the measurement of efflux of vascular markers into the airways. Common markers include radiolabeled or fluorochrome-tagged albumin, fluorescent-labeled high molecular weight dextran, or dyes (Looney et al. 2006; Wolk et al. 2008; Sutton et al. 2003; Y. Yang, Yang, and Schmidt 2013; Gotts, Abbott, and Matthay 2014). Any of these tracers can be injected intravenously in the mouse and then measured quantitatively either by homogenizing the entire lung and subtracting the tracer amount in the blood volume or by harvesting BALF to perform the measurement directly. With all these tracers, it is important to verify that the label has not dissociated from the protein (Modelska et al. 1999). Evans blue dye is commonly used as an intravenously injected tracer to define regions of vascular leakage (Gonzales et al. 2014; Gong et al. 2015). The amount of the translocated dye can be measured in formamide-treated, homogenized lungs at an excitation wave length of 620 nm and emission wave length of 680 nm. Mathematical determination of the Evans blue dye concentration should include a correction for heme pigment contamination (Tremblay et al. 1997). The advantage of using fluorescently labeled dextran over Evans blue dye is that the former requires less sample processing and no heme pigment correction (S. E. Sinclair et al. 2004). A major disadvantage of using radiolabeled tracers is that mouse tissues must subsequently be safely stored until the labeling isotope is no longer radioactive.
Increase in total BALF protein concentrations or high molecular weight proteins
The protein concentration in harvested BALF primarily reflects vascular leakage into the alveolar space. However, increased protein can also be derived from dead and dying inflammatory cells and pneumocytes. In addition, the airway protein concentration can increase as a result of the diminished ability of injured lungs to clear alveolar fluid (Matute-Bello et al. 2011; Sartori and Matthay 2002).
To harvest BALF, mice are anesthetized using high doses of injectable anesthetics (e.g., ketamine/xylazine at 87 mg/kg and 13 mg/kg, respectively, delivered intraperitoneally), and a tracheotomy is performed to insert a 20-gauge stainless steel cannula or trimmed 18-gauge Teflon catheter (Figure 1). Next, a fixed volume (usually 1 ml) of sterile normal saline (pH 7.4, room temperature) is instilled into the lungs via the cannula and then aspirated; this cycle is repeated 2 to 3 times using the same fluid, after which the fluid is collected (Figure 3B). Lavage samples should be centrifuged at 500 × g for 10 min at 4°C to separate out cells, after which supernatants can be stored at −80°C for further analysis (Davis et al. 2006; Naito et al. 2013; Wolk et al. 2008).
Since it is difficult to standardize any BALF measurements, appropriate control specimens should be acquired from untreated animals studied under the same circumstances and processed by the same methods, as protein concentrations can increase significantly in non-perfused lungs (Tremblay et al. 1997). Since hydrostatic pressure can force intravascular protein out of blood vessels and into alveoli and the interstitium, an additional control sample can be generated in animals exhibiting increased left atrial pressure (to model higher hydrostatic pressure; Matute-Bello et al. 2011).
A variety of assays are available to measure protein concentration in BALF. The usual options include the Bradford method, the Biuret method, or the bicinchoninic acid (BCA) assay kit (e.g., by Thermo Fisher Scientific; Waltham, MA; Davis et al. 2004; Letsiou et al. 2015; H. Wang et al. 2014). Instead of measuring total protein concentration, it is also possible to quantify increased vascular leakage into the lung by measuring the concentration of very high molecular weight proteins (e.g., IgM) that normally are only present in the blood stream using an enzyme-linked immunosorbent assay (ELISA; Chun et al. 2010). In addition, species-specific albumin ELISA kits are available to measure murine albumin (Wolk et al. 2008).
It is important to note that harvesting BALF renders the lungs unsuitable for histology, as it induces artifacts such as ruptured alveoli due to the large volume instilled. However, by tying off the left (single) lung lobe before the BAL procedure and then using half the saline volume (i.e., 0.5 ml), lavage fluid can be harvested from just the right lung lobes. This approach makes the left side available for measurement of lung wet/dry ratios. Alternatively, the left lung can be homogenized for measurement of viral titers, or extraction of proteins for Western blotting and cytokine ELISAs. This approach permits the correlation of structural and chemical changes in the same experimental subject.
Somewhat relevant features of altered alveolar capillary barrier integrity
A very easy and commonly used method to assess total lung water content as a surrogate for assessing pulmonary edema is the determination of wet/dry lung weight ratios. For this procedure, lungs are extracted from the mouse, weighed immediately, and then dried in an oven at or below 56°C (Parker and Townsley 2004; Davis et al. 2004; Liu et al. 2012; He et al. 2015). Some investigators prefer to homogenize lungs before drying them (Looney et al. 2006). Organ weights are taken daily during the drying process until the weight is stable. Depending on the temperature of the oven, this period can be as soon as 2 to 3 days after sample harvest. This method, however, has several caveats. First, identical sample preparation among animals is important. Features that need to be controlled to maintain consistency include whether or not the animal is exsanguinated during euthanasia, whether the harvested lung is blotted on tissue paper to remove external blood prior to drying, and whether the lung is adequately dissected free of nearby organs like the trachea, esophagus, and thymus (if present). Second, it is of uttermost importance to use a very fine analytical scale (3 digits after the decimal point) with such light samples (especially after drying), as small errors may have a large effect on data quality. Finally, this method assesses total water content only and does not distinguish between fluid leakage into the alveolar spaces versus water accumulation in the interstitium (Parker and Townsley 2004; Davis et al. 2004).
Similar to measuring leakage of a protein from the blood stream into the alveolar space, the translocation of instilled substances from the air space into the plasma can be assessed. For this method, an exogenous (foreign) or labeled protein or dextran can be used (Wolk et al. 2008). However, the use of an albumin-based method can lead to overinterpretation of translocation due to potential transcytosis of this protein through endothelial cells (Predescu, Vogel, and Malik 2004).
Magnetic resonance imaging (MRI) and assessment of alveolar fluid clearance were not included in the ATS workshop paper. However, the authors believe that these assays are important to include as powerful analytical tools for examining pulmonary edema in mouse models of ARDS. While expensive, MRI is an elegant tool for serial in vivo visualization of edema progression in the lung. While it does not give detailed information about the compartmental location of the water signal (alveolar vs. interstitial), the imaging can be done under survival anesthesia in live, ventilated mice (Aeffner et al. 2012), which helps to reduce interindividual variation in experimental animals. Because, unlike measurement of wet/dry weights, MRI is not a terminal procedure, animals can be allowed to recover, enabling the researcher to either track disease evolution or make the animals available for other kinds of measurements. This option provides the potential for reducing overall animal numbers in studies while providing multifaceted data sets. Rendering of stacked 2-dimensional MRI scans can be used to reconstruct the whole lung in 3 dimensions. This permits calculation of the total lung volume as well as the total volume containing fluid (Aeffner et al. 2012). A net increase in total lung volume is commonly observed in mice with ARDS over the course of the disease due to progressive edema development and increased respiratory effort, which together result in recruitment of more alveoli into the total lung volume (Aeffner et al. 2012; Karmrodt et al. 2006).
While MRI requires expensive equipment, measurement of alveolar fluid clearance rate as a means of assessing damage to the alveolar capillary barrier is far less costly. In essence, alveolar fluid clearance rate reflects the electron transport capability of bronchoalveolar epithelial cells. This tightly regulated process maintains a thin but essential film of fluid lining the air space that directly reflects the ion transport capacity of alveolar type II epithelial cells. Briefly, sodium (Na+) ions are actively transported from the alveolar lining fluid into the interstitial alveolar space via Na+ channels in the apical membranes of the epithelial cells (Davis and Matalon 2007). This process is facilitated by the basolateral sodium–potassium adenosine triphosphatase (ATPase), which exchanges 3 intercellular Na+ ions for 2 extracellular potassium (K+) ions through the energy generated by adenosine triphosphate (ATP) hydrolysis. The resulting electrochemical gradient results in movement of chloride (Cl−) ions along with Na+ into the subepithelial interstitial space. The net movement of NaCl from the airspace-lining fluid into interstitial tissue creates an osmotic gradient across the alveolar epithelium, which causes water to translocate to the interstitium. When normal processes that regulate ionic equilibrium are impaired, pulmonary edema develops (Ware and Matthay 2001; Schuster and Marklin 1986; Fronius, Clauss, and Althaus 2012; Ware et al. 1999; Matthay, Folkesson, and Clerici 2002). It has been shown that the degree of impaired alveolar fluid clearance in human ARDS patients is a predictor of hospital mortality (Ware and Matthay 2001; Sartori and Matthay 2002). Clearance measurements in mice are undertaken by instilling a known concentration of bovine serum albumin in iso-osmolar saline into the dependent lung of an anesthetized, mechanically ventilated, live mouse placed in left lateral recumbency. In this position, instillation (via tracheotomy) of 200 μl will completely fill the single dependent left lung lobe in an 8- to 12-week-old mouse. The animal is ventilated for 30 min, after which residual intra-alveolar fluid is aspirated via the tracheotomy needle. The recovered volume in normal mice is very low (˜40 μl) with a high protein concentration (as water is transported into the interstitium while bovine serum albumin remains in the air spaces). Aspiration of larger volumes of fluid with a lower protein concentration is indicative of diminished alveolar fluid clearance (Davis et al. 2004; Aeffner et al. 2011; Hardiman, Lindsey, and Matalon 2001). For this procedure, it is of utmost importance that ventilators are calibrated for ventilation pressure and volume to ensure that all animals are exposed to the same experimental parameters. It has been shown that variations in ventilation pressure alone can affect clearance rates in healthy mice (Yu et al. 2009). It is also essential that mice remain alive throughout the procedure as a buildup of bronchoalveolar interstitial pressure following hemostasis tends to oppose fluid clearance.
The protein concentration of aspirated fluid can be measured with the same assays as noted earlier for total BALF protein concentration. It is important to remember to measure a pre-instillation sample in additional to experimental samples.
Main Feature: Measurements of the Inflammatory Response
Increase in the absolute number of neutrophils in BALF
As previously mentioned, pulmonary inflammation is a prominent component of ARDS. In human patients, it has been shown that neutrophil numbers in the BALF correlate with disease severity (D. G. Sinclair et al. 1994). Assessment of inflammation may be performed by subjective and/or semiquantitative histological assessment of inflammation and specifically neutrophil accumulation as described earlier. More quantitative analysis may be undertaken by cytological evaluation of BALF. Measurements should include total cell counts and differential counts for all major leukocyte classes (McGarry, Protherone, and Lee 2009); with these values, the total number of neutrophils within the alveolar compartment can be calculated (Guo et al. 2012). It is important to evaluate both the total number and relative quantities (percentage) of the cells, as the influx of other inflammatory cell populations can greatly influence the overall percentage of neutrophils without producing a substantial change in the total cell number, and vice versa. Since differential cell counts also quantify the numbers of lymphocytes, macrophages, and eosinophils present in the sample, this method has the potential to yield meaningful data related to other aspects of lung inflammation in ARDS.
When assessing inflammatory cell populations, BALF is collected as described earlier. Some investigators add 1% ethylenediaminetetraacetic acid (EDTA) in saline, pH 7.4, to the sampling fluid to prevent cell clumping (Matute-Bello et al. 2011; Bhargava et al. 2013). Newly harvested BALF specimens should be kept on ice and processed as soon as possible to prevent cell death and lysis, which can impact the number of cells available to be counted (McGarry, Protherone, and Lee 2009; Naito et al. 2013; Aeffner and Davis 2012). To separate the cells for counting, BALF should be centrifuged at 500 × g for 10 min at 4°C (E. Zhou et al. 2014). The supernatants can be stored at −80°C for further analysis, including measurement of cytokine levels (Zhang et al. 2015). The remaining cell pellet is resuspended in saline so that the total live cell counts may be obtained via trypan blue dye exclusion using a hemocytometer. Later, the remaining cells or a portion of them can be processed as cytospin preparations (by centrifugation at 1,000 rpm for 5 min on a Cytospin™ 4 unit; Thermo Fisher Scientific, Waltham, MA) and dyed with Wright–Giemsa stain or a similar method. Differential cell counts typically are performed at 10 (ocular) × 100 (objective) magnification via oil-immersion microscopy by counting a minimum of 200 cells (optimal: 300 or more cells; Naito et al. 2013; McGarry, Protherone, and Lee 2009; Aeffner, Woods, and Davis 2014).
If flow cytometry equipment is available, neutrophils can be quantified rapidly in automated fashion. Neutrophils are recognized using specific antibodies for cell type–specific markers such as Ly6G, their characteristic scatter profile due to the cytoplasmic granularity, or typically both features in combination (Aggarwal et al. 2013; McAuley et al. 2007; Reutershan et al. 2006). In addition to BALF analysis, flow cytometry can also be performed on whole lung homogenates to determine total lung leukocyte numbers (Deroost et al. 2013).
Increase in lung myeloperoxidase (MPO) activity or protein concentration
While the assessment of the total neutrophil numbers does not give information about the activation state of counted cells, measurements of MPO activity may reveal the functional state of this cell population. MPO is present in all cells of the myeloid lineage, but it is most abundant in the azurophilic granules of neutrophils (Nauseef, Olsson, and Arnljots 1988). Enzyme activity can be measured in both BALF cell pellets and whole lung homogenates. For the latter, lungs are harvested as described earlier and stored at −80°C until enough samples are collected. Lungs are then homogenized by hand or mechanically (e.g., FastPrep-24 instrument; MP Biomedical, Santa Ana, CA), after which enzyme activity is measured using a commercially available MPO kit or a colorimetric assay according to the manufacturer’s instructions (e.g., Mouse Myeloperoxidase ELISA Kit, Cell Sciences, Canton, MA; Lomas et al. 2003; Hirsh et al. 2004; Neff et al. 2005; Hoke et al. 2007; Aeffner, Woods, and Davis 2014; E. Zhou et al. 2014). If whole lung MPO is to be measured, it is important to homogenize the lungs in 1 ml of cold hexodecyltrimethylammonium bromide buffer (50 mM KPO4 and 0.5% hexodecyltrimethylammonium bromide [pH 6.0]) to preserve MPO activity (Hoke et al. 2007).
Increased concentrations of pro-inflammatory cytokines
Pulmonary inflammation in ARDS is in part mediated by pro-inflammatory cytokines (Barnett and Ware 2011). Pro-inflammatory cytokines and chemokines can readily be measured in BALF. A variety of ELISA kits and multiplex assays for murine cytokines are commercially available. Common pro-inflammatory cytokines that may contribute to development of ARDS include tumor necrosis factor-alpha (TNF-α; Y. Xu et al. 2014; L. Wang et al. 2015), interleukin-1-beta (IL-1β; Maron-Gutierrez et al. 2013), interleukin-6 (IL-6; L. Wang et al. 2015), macrophage inhibitory factor (MIF; also known as macrophage migratory inhibitory factor [MMIF] or glycosylation-inhibiting factor [GIF]; Lai et al. 2003), transforming growth factor-beta (TGF-β; Peters et al. 2014), and platelet-activating factor (PAF; Matute-Bello et al. 2011; Nagase et al. 1999). An increase in BALF neutrophils in mice is often associated with elevated BALF levels of keratinocyte chemoattractant (KC, also known as CXCL1) and macrophage inflammatory protein 2-alpha (MIP-2α, also known as CXCL2), which are the murine analogs of the neutrophil-recruiting chemokine interleukin-8 (IL-8; Bhargava et al. 2013; Y. Xu et al. 2014).
When handling and storing BALF samples, it is important to pay attention to the susceptibility of each cytokine to degradation. Some molecules need added buffers and proteinase inhibitors, and others (such as TNF-α) are particularly sensitive to repeated freezing and thawing. Therefore, BALF samples should be stored in small aliquots so that each cytokine can be measured from samples that have been thawed only once. Aliquot size, buffer composition, and storage requirements are dependent on the requirements of the specific cytokine detection kit to be used.
Several alternatives exist for evaluating pro-inflammatory mediators in the lung. Instead of BALF, whole lung homogenates can be used for these measurements (Tumpey et al. 2005; Bhargava et al. 2013). Rather than assessing cytokine protein concentrations, Northern blot or (increasingly) quantitative real-time polymerase chain reaction (PCR) can be used to measure cytokine gene expression (Avasarala et al. 2013; Downs et al. 2013). However, gene expression levels do not necessarily reflect protein expression levels, and therefore mRNA expression data should be verified by measuring the concentration of the actual protein where feasible. In addition, real-time PCR analysis of mRNA in lung samples can be challenging. Since RNA is easily degraded, RNAse inhibitors (e.g., RNAlater, Thermo Fisher, Carlsbad, CA) should be added to specimens upon collection. In addition, investigators should pay close attention to keeping samples chilled during homogenization.
Another important consideration is the choice of reference genes for standardization of cytokine gene expression levels between samples. So-called housekeeping genes such as glyceraldehyde 3-phosphate dehydrogenase (GAPDH) and actin are widely used for this purpose, but recent reports view them more and more critically as not having sufficiently stable expression levels. These variations have led many investigators to favor hypoxanthine-guanine phosphoribosyltransferase (HPRT) or 18S rRNA as reference RNAs for quantitative real-time polymerase chain reaction (PCR); (Glare et al. 2002; Bas et al. 2004; Schmittgen and Zakrajsek 2000). However, at present no widely accepted consensus exists regarding the most appropriate reference genes for use in animal models of ARDS. Probably the most important issue is to consistently use the same reference RNA for all quantitative real-time PCR studies.
Somewhat relevant features of the inflammatory response
With regard to assessing the inflammatory environment in the lung and the status of the entire animal, the main somewhat relevant feature is increased pro-coagulation activity in peripheral blood samples. The procedure needs to be validated by the laboratory conducting the study, in particular to build a historical database of normal reference values in the test species and strain. Commercial kits are available to measure parameters such as thrombin–antithrombin complexes in BALF and whole lung homogenates (Naito et al. 2013). Similar kits are available to assess complement factors or matrix metalloproteinases (Naito et al. 2013; Hirsh et al. 2004).
An alternative feature of some relevance is to define the degree to which the leukocyte profile in lung samples shift from an acute to a more chronic phenotype. Chronic stages of ARDS, which are less commonly assessed in mouse models, are accompanied by a conversion from a more neutrophilic process (typical or acute ARDS) to a predominantly monocytic inflammatory environment. This shift can be observed and assessed via histology as well as BALF cell counts and flow cytometry, as described earlier. Conversion to chronic disease in mouse ARDS models occurs at variable time points, depending on the specific model used. In general, little information regarding the extent or timing of this change is present in the literature.
Main Feature: Measurements of Physiological Dysfunction
Hypoxemia
While hypoxemia is one of the most important readouts for functional impairment of the lung, it is important to point out that hypoxemia is only the result of ARDS, not a direct measurement of injury severity per se. Hypoxemia can also result from various physiological mechanisms that are independent of ARDS. In addition, this readout obviously does not apply to ARDS models in which the injury is the result of prolonged ventilation with 100% oxygen (Matute-Bello et al. 2011).
The easiest and most reliable assessment of peripheral blood oxygenation (SpO2) in mice is by pulse oximetry. This procedure is done by applying the probe to the neck or hind limb (with the choice of location depending on the type of probe available for the study). It is of utmost importance to use pulse oximetry equipment that is specifically designed for use in mice (e.g., MouseOx instrument, Starr Life Sciences, Oakmont, PA). Before such instruments were available, pediatric pulse oximetry devices were commonly used. However, the sensors in these analyzers were unable to detect the high-frequency pulsatile nature of arterial blood flow in mice and therefore gave an artificially low reading reflecting the average of arterial and venous blood oxygenation levels (Davis et al. 2006). Generally, SpO2 values of less than 90% are viewed as hypoxemic; however, rigid cutoff values do not exist for either animals or humans, so decisions regarding the relevance of decreased readings are subject to interpretation. The advantage of pulse oximetry over direct measurements of arterial blood gases is that the latter method is a terminal procedure, while the former can be performed on conscious mice on a repeated basis with minimal stress to the animals (e.g., by using collar clips while letting the mouse range with relative freedom throughout the cage). This transcutaneous measurement works in mice with white or light-colored coats without having to remove the hair, while in animals with black or dark-colored hair, the fur needs to be removed first by shaving or another method. For repeated measurements, we recommend depilation using mild chemical products for hair removal developed for humans, such as Veet® or Nair™, applied under light anesthesia and rinsed off well with abundant water to minimize skin irritation. This strategy will result in delayed regrowth of hair and allows for reliable pulse oximetry for up to 7 days after hair removal.
If hypoxemia is assessed in anesthetized and/or ventilated animals, it is important to know the effect of these manipulations on blood gases. Many anesthetics are respiratory suppressants and can artificially depress oxygenation levels. Similarly, significant effects on blood gases can result from fluctuations in ventilation frequency, ventilatory pressure, tidal volume, and, most importantly, the presence or absence of PEEP (Traylor, Aeffner, and Davis 2013). In addition, inappropriate ratios between duration of inspiratory and expiratory ventilation maneuvers can result in hypoventilation and hypercapnia (increased PaCO2 levels). All measurements using anesthetized animals should be performed with animals in prone position, which will minimize the influence of body weight and chest wall stiffness on blood oxygenation.
Significant pulmonary shunting can lead to severely decreased arterial blood gases with decreased cardiac output. The resulting decrease in arterial oxygenation levels do not accurately reflect compromised pulmonary function, but rather reflect alterations in hemodynamics. Therefore, hypoxemia should never be the sole criterion upon which to base the estimated severity of lung injury.
Increased alveolar–arterial oxygen difference
While rarely performed in mice, an increase in alveolar–arterial oxygen difference (DAO2–DaO2; where A = alveolar; a = arterial) can reveal impaired gas exchange due to lung injury (Vaporidi et al. 2012). The partial pressure of oxygen in the alveoli (PAO2) is calculated via the alveolar gas equation, while arterial partial pressure (PaO2) is measured via arterial blood gas analysis. The exact formulas for these calculations can be found in the literature (Risk, Epler, and Gaensler 1984; Story 1996). The values can be significantly influenced by FiO2, so it is important to keep this value controlled and identical across different time points, groups, and individual animals. This technique is more commonly performed in rats than mice, probably due to the larger animal size and easier arterial blood sampling in the former species (Sato et al. 2010).
Somewhat relevant features of physiological dysfunction
While methods for measurement of PaO2/FiO2 ratios have recently been established in the mouse (Traylor, Aeffner, and Davis 2013; Ma et al. 2014), the procedure is technically very challenging to perform in this species. It has therefore not been included as a very relevant feature for assessment of mouse ARDS models by the ATS. Nevertheless, arterial blood gases are considered to be a more accurate measurement of hypoxemia than pulse oximetry. In addition, arterial blood gases and pulse oximetry are indicators of changes in different oxygenation parameters. While pulse oximetry measures the oxygen bound to hemoglobin (Ralston, Webb, and Runciman 1991; Webb, Ralston, and Runciman 1991; Chan, Chan, and Chan 2013), arterial blood gases reflect the amount of oxygen dissolved in the blood (Tipping, Berry, and Nesbitt 2012). While it has been shown that both readouts correlate closely in humans (Rice et al. 2007; Lobete et al. 2013), it is our experience that arterial blood gases in mice decrease significantly earlier than do pulse oximetry measurements (Traylor, Aeffner, and Davis 2013). We believe this is due to the high respiratory rate of mice, which results in a roughly 15-fold higher alveolar ventilation rate in mice than in humans. Accordingly, the mouse has a large compensatory capacity for maintaining high pulse oximetry readouts while arterial blood gases are already declining (Traylor, Aeffner, and Davis 2013). One advantage of PaO2/FiO2 over the aforementioned alveolar–arterial oxygen differences is that the ratio is corrected for FiO2, which simplifies the comparison of values acquired during different experiments.
It is essential to note that, whether they are in room air or exposed to 100% O2, arterial blood gases should only be measured in anesthetized, mechanically ventilated, live animals in the presence of appropriate PEEP. The method should be validated in each laboratory using healthy mice to confirm that PaO2/FiO2 ratios are around or close to 600 mmHg, before ARDS animals are investigated. Besides the influence of ventilation on data, this method is a surgically challenging procedure that involves isolation of the carotid artery in a ventilated, anesthetized animal and subsequent sampling of arterial blood from inside the artery while the animal’s heart continues to beat (Traylor, Aeffner, and Davis 2013). In addition, arterial blood gases are usually measured in machines developed for use in human patients, which require a minimal volume of 200 μl or more of whole blood. Depending on hydration status and disease progression, harvesting this much blood from a mouse can be very challenging. We and others have had reasonable success using the iSTAT point-of-care blood gas analyzer (Abbott, Chicago, IL; Fukushi et al. 2012; Traylor, Aeffner, and Davis 2013; Ma et al. 2014). These machines usually also assess other helpful readouts such as blood pH and PaCO2. With the protocol established in our laboratory, mice are ventilated using 100% oxygen prior to sample harvest. Since the iSTAT also measures oxygen bound to hemoglobin (SaO2; similar to the readout of pulse oximetry), this readout can be used for quality control and as grounds for data exclusion. SaO2 values below 85% on 100% O2 are considered to be indicative of faulty ventilation (Traylor, Aeffner, and Davis 2013).
Increased spontaneous changes in respiratory rate cannot be accurately assessed visually in mice due to their naturally high rates of respiration. However, subjectively, mice with ARDS are often described as breathing more heavily (i.e., a reflection of increased respiratory effort) while displaying decreased overall movement within the cage. Some pulse oximetry devices such as the MouseOx instrument have the ability to also record spontaneous respiratory rate in conscious animals, as described above (Aeffner et al. 2012). Alternatively, respiratory rates can be determined using whole body plethysmography in conscious, unrestrained mice. However, for other readouts (especially enhanced pause or Penh), this method has been shown to be inadequate (Lundblad et al. 2007; Lundblad et al. 2002; Adler, Cieslewicz, and Irvin 2004).
Increased minute volume is calculated from respiratory rate and tidal volume and is not assessed in mice as it is in humans. However, tools exist to measure tidal volume in mice during ventilation (when the respiratory rate is known), such as the Scireq flexiVent (Aeffner and Davis 2012). This computer-controlled piston-pump ventilator can be used on tracheotomized as well as intubated mice, with the latter providing for the possibility that animals can be recovered for repeated measurements. While it is a highly specialized tool and thus is not available in every laboratory, this instrument does provide additional readouts that are very meaningful but that were not specifically mentioned in the ATS workshop paper, such as airway resistance, dynamic lung compliance, and static lung compliance. The flexiVent can also be used to generate pressure–volume loops. Decreased compliance is a measurement of the decreased elasticity of the lung and is included in some definitions of ARDS (Matute-Bello, Frevert, and Martin 2008; Ferguson et al. 2005). A decrease in lung compliance has been shown to occur in some mouse models of ARDS (Aeffner et al. 2012).
Relevance of Mouse ARDS Models for Drug Discovery and Development
The greatest increase in survival rates for human ARDS patients has come from recent improvement in clinical treatment (e.g., the introduction of low-tidal volume ventilation). Comparatively little progress has been made in lowering the ARDS mortality rate via basic research to comprehend the fundamental processes that drive this disease. The multifactorial pathogenesis of ARDS limits the extent of correlation between mouse models and human patients. Nonetheless, mouse ARDS models are being employed to discover and develop new therapies for humans.
The main use of mouse ARDS models in developing new therapies for human ARDS is the identification of potential biochemical and molecular pathways responsible for the ARDS-associated inflammatory and vascular lesions. Data regarding efficacy or safety pharmacology endpoints obtained via mouse models may be used in a general sense to predict potential human responses. However, several drug candidates that appeared to have promise in animal studies have subsequently failed in human clinical trials. The authors believe that this divergence in animal and human efficacy is due chiefly to the fact that the common endpoints used for assessing responsiveness in animals—organ weights, lung lesions, and survival—provide limited information on the progression of ARDS in humans, and also do not take into consideration the advanced supportive care that human patients receive in respiratory intensive care units of hospitals. Greater familiarity by pathologists, toxicologists, and other researchers with clinically relevant ARDS endpoints should allow animal experiments to be designed that will improve the ability of mouse experiments to predict human outcomes.
To our knowledge, adult mouse ARDS models do not predict responses in infants who develop ARDS (or neonatal respiratory disease syndrome). The reason for this divergence is that respiratory distress syndrome (RDS) is due in large part to a lack of surfactant, which is not a typical part of the pathogenesis in adult ARDS patients. Exploration of RDS in mouse models usually is undertaken with animals engineered to lack surfactant genes production (Cuevas Guaman et al. 2014; Mohamed et al. 2014). However, the pig appears to be the most commonly employed animal model in this particular field of study (Preuss et al. 2014; Caminita et al. 2015).
General Comments
Veterinary pathologists will remain key investigators in evaluating ARDS in any animal models. This prominence is due to the importance of morphological assessments (e.g., histopathologic scores of lung lesions, cytological findings in cytological preparations of BALF samples) in the interpretation of ARDS-like lesions. In addition, veterinary pathologists have a unique combination of knowledge in the majority of biomedical fields relevant for ARDS research, such as pulmonary anatomy and physiology as well as clinical and anatomic pathology. On the other hand, it is important that pathologists do not rely on histological readouts alone to interpret respiratory function in an animal or specifically mouse model of ARDS, but instead incorporate a variety of the other features and especially the functional methods listed in this article into their overall interpretation.
In summary, based on the ATS workshop paper defining features of ARDS that are most relevant in animal models, here we have reviewed principal attributes of mouse ARDS models by highlighting the major features of experimental lung injury in mice. We have also provided methodologic guidance on how to approach the measurement of these features. We hope this publication will aid biomedical investigators and toxicologic pathologists who are using animal models to investigate the pathogenesis of ARDS and explore innovative means of treating this condition. Over time, we expect that new means for structural analysis of mouse ARDS models will lead to further refinements in our understanding of this poorly understood disease.
Footnotes
Acknowledgments
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
