Abstract
Most uterine cancers, the most common gynecological malignancies in women in developed countries, are hormone-dependent endometrial adenocarcinomas (EACs) that express estrogen and progesterone receptors. Although rat strains exist with a high spontaneous incidence of EAC, the Fischer 344 (F344) strain, previously one of the most commonly used strains in carcinogenicity testing, is not a high-incidence strain. To better understand the biology of this neoplasm, we assessed estrogen receptor α (ER), progesterone receptor (PR), and Ki-67 expression using immunohistochemistry in spontaneous EAC in 18 F344 rats used as control animals in 2-year National Toxicology Program bioassays. Of the 18 tumors, 9 were well-differentiated and 9 were poorly differentiated. Most tumors, 7/18, were ER+PR+, as observed in women. Of the remainder, 6/18 were ER+PR−, 2/18 were ER−PR+, and 3/18 were ER−PR−. Well-differentiated tumors were ER+ (8/9) more often than poorly differentiated tumors (5/9). The percentage of ER+ tumors (72%) in rats was similar to that seen in women, but rats less frequently had PR+ (50%) tumors than women. The heterogeneous estrogen and progesterone receptor immunophenotypes observed in F344 rats in this study highlight the importance of evaluating hormone receptor expression in animal models used for chemical evaluations.
Introduction
Endometrial cancer is the most common gynecologic malignancy in developed countries and the fourth most common malignancy overall in women in the United States and Europe (Burke et al. 2014; Ferlay et al. 2013). The most common type of endometrial cancer is type I endometrial adenocarcinoma (EAC), typically with endometrioid morphology. These tumors are generally low grade, arise in a background of hyperplasia, express estrogen receptor α (ER) and progesterone receptor (PR), and have a good prognosis (Amant et al. 2005; Sorosky 2012). Moreover, ER and PR immunoreactivities in human endometrial cancer have a significant inverse relationship with immunoreactivity of Ki-67, a marker of proliferation (Obeidat et al. 2013). Given the importance of animal models in the study of carcinogenesis or assessment of risk for humans, our objective was to characterize hormone receptor and Ki-67 expression in Fischer 344 (F344) rats with spontaneous endometrial cancer.
Endometrial cancers are generally considered estrogen-dependent neoplasms because they typically express sex steroid receptors and because unopposed or elevated estrogens have been identified as risk factors (Amant et al. 2005; Kounelis et al. 2000; Sorosky 2012). Unopposed estrogen replacement therapy in menopause is associated with most type I endometrial cancers, leading to a 2- to 20-fold increased risk, however, concurrent progestogen use significantly reduces this risk (Burke et al. 2014). Risk factors for EAC that also result in estrogenic stimulation unopposed by progesterone include obesity, anovulation, polycystic ovarian syndrome, nulliparity, early menarche, late menopause, and tamoxifen use (Amant et al. 2005; Burke et al. 2014; Sorosky 2012).
Dependence of endometrial carcinomas on estrogen exposure has been presumed but not experimentally tested in F344 rats, an important strain used in carcinogenicity testing. The F344 rat has until recently been one of the most commonly used strains in carcinogenicity bioassays in the United States and is also commonly used in Japan. This strain was adopted as the strain of choice for carcinogenicity testing by the National Cancer Institute (NCI) in 1970 and, until recently, it was the strain of choice for the National Toxicology Program (NTP). The F344 rat strain has also been used extensively by the chemical industry for safety evaluation. This neoplasm in F344 rats has been associated with advanced age (Tang and Tang 1981) but has not been well characterized in this strain. Characterization of endometrial neoplasms in F344 rats in the context of putative risk for human beings requires identifying the presence or absence of steroid receptors, as EAC is generally a steroid-responsive neoplasm in women.
Materials and Methods
This retrospective study used formalin-fixed, paraffin-embedded uterine tissues from control animals from previously conducted 2-year bioassays from the NTP archives. Animal care and use were in accordance with the United States Public Health Service Policy on humane care and use of laboratory animals and the Guide for the Care and Use of Laboratory Animals. Eighteen endometrial cancers were identified in control F344 rats from 13 studies. Unstained sections of each uterus were used for immunohistochemistry or stained with hematoxylin and eosin for histopathology.
Histopathology
The percentage of solid differentiation was evaluated based on the International Federation of Gynecology and Obstetrics (FIGO) system for human endometrioid or serous carcinomas of grade 1 (≤5% nonsquamous solid growth pattern), 2 (6–50% nonsquamous solid growth pattern), or 3 (>50% nonsquamous solid growth pattern; Creasman 1989). Each tumor was also graded by consensus of two pathologists as well (low-grade) or poorly differentiated (high-grade), based on the lowest degree of differentiation present. Nuclear atypia was scored as mild (+), moderate (++), or marked (+++). For neoplastic glandular areas, the average number of mitotic figures per high power (40×) field (hpf) was determined after counting 10 hpfs. Mitotic index was calculated separately for glandular and solid areas for tumors with neoplastic solid growth. Solid areas in some of these tumors with both glandular and solid differentiation consisted of fewer than 10 hpfs. In such cases, the solid area mitotic index was counted as the average number of mitotic figures per hpf.
Immunohistochemistry
Uterine sections were stained for nuclear expression of ER, PR, and Ki-67. The primary antibodies used were mouse monoclonal ERα clone 6F11 (NCL-L-ER-6F11; Leica Biosystems, Newcastle upon Tyne, UK) at a 1:100 dilution, mouse monoclonal PR clone 10A9 (MAB4298; Abnova, Taipei City, Taiwan) at a 1:50 dilution, and rabbit monoclonal Ki-67 (RM-9106-S; Thermo Scientific, Fremont, CA) at a 1:100 dilution. ERα was examined rather than ERβ because ERα is the predominant receptor in the uterus of rats and women, and it has been proposed that estrogen exerts its effects primarily through ERα in endometrial carcinomas in women (Shughrue et al. 1998; Utsunomiya et al. 2000). The staining procedure used a streptavidin–biotin–alkaline phosphatase method and antigen retrieval using heat and citrate buffer pH 6.0 to 6.1 as previously described (Wood, Hester, and Cline 2007). Sections were incubated with the primary antibodies overnight at 4°C. The chromogen used was Vector Red (Vector Laboratories, Burlingame, CA) and immunostained sections were counterstained with Mayer’s hematoxylin. Positive controls were normal rat uterus for ER and PR and rat gastrointestinal tract for Ki-67, and negative controls for each immunostain used nonimmune serum from the same species as the primary antibody in place of primary antibodies.
Immunohistochemical staining was evaluated using a semiquantitative score based on nuclear staining intensity and percentage of cells staining in neoplastic epithelial cells (Fuqua et al. 2003). The proportion of positively stained tumor cells was estimated as 0 (none), 1 (<1/100), 2 (1/100 to 1/10), 3 (1/10 to 1/3), 4 (1/3 to 2/3), or 5 (>2/3). An intensity score representing average intensity was assigned as 0 (none), 1 (weak), 2 (moderate), or 3 (strong). The sum of the proportion and intensity scores was the total score, which ranged from 0 to 8 (0–2 = negative; 3–8 = positive).
Results
Histopathology
EACs in rats generally displayed complex interdigitating cords or glandular structures and acini lined by a single layer of neoplastic cuboidal to tall columnar epithelial cells (Figures 1A, 2A, and 4A). Neoplastic epithelial cells had moderate amounts of eosinophilic cytoplasm and large vesicular nuclei with generally one prominent nucleolus. Neoplastic glandular structures were commonly dilated by variable amounts of necrotic cellular debris and viable and degenerate neutrophils (Figure 3A). Desmoplasia, with neoplastic acini surrounded by abundant connective tissue, was prominent in most tumors (Figure 3A). Intra-tumoral necrosis was common, with two cases having abundant necrosis. Three cases displayed either myometrial or vascular invasion. Invasion in many of the cases was considered likely but could not be reliably assessed owing to a lack of normal tissues present.
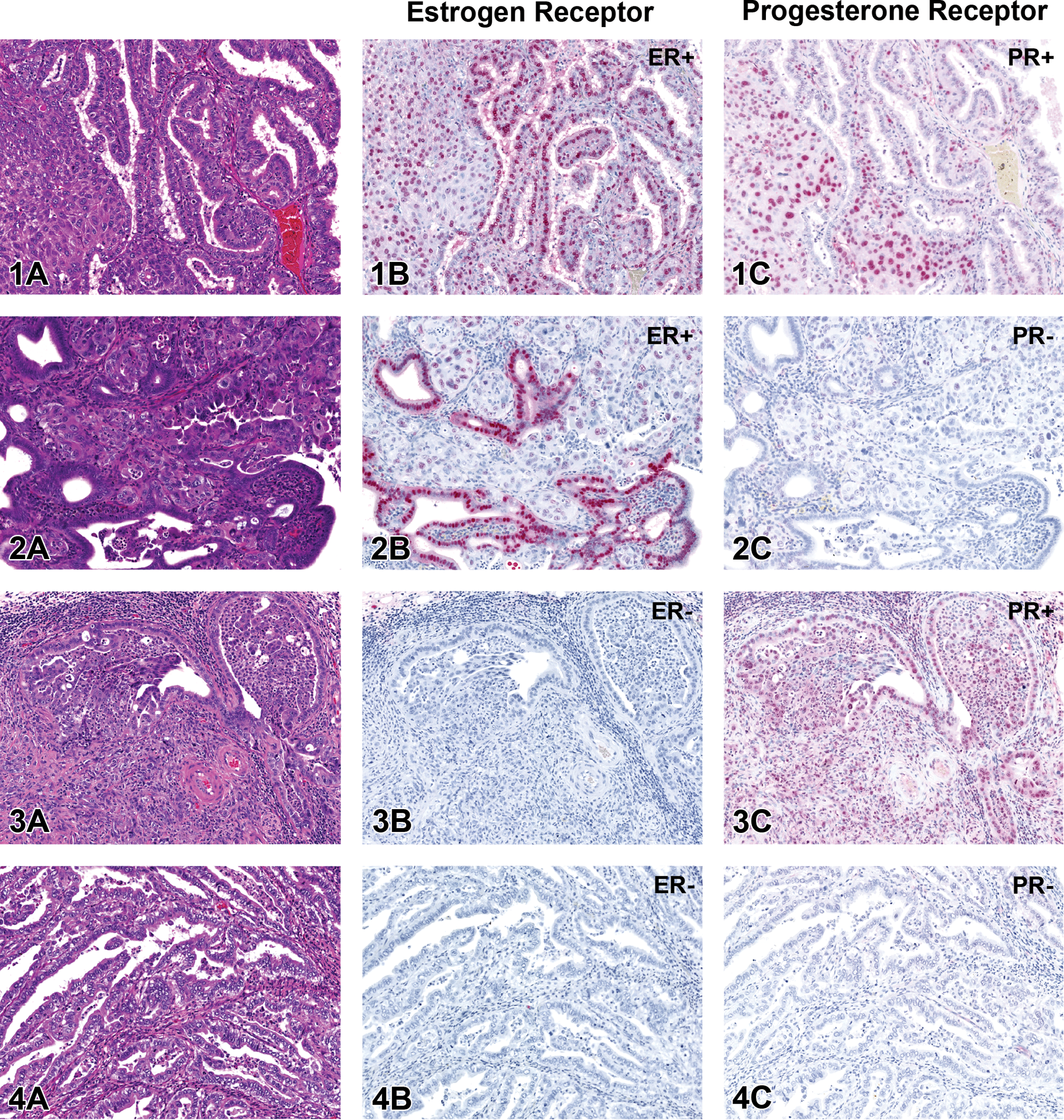
Four different estrogen receptor alpha (ER) and progesterone receptor (PR) immunophenotype combinations in endometrial adenocarcinomas in control F344 rats. In Figures 1–4, serial sections of four tumors are stained with hematoxylin and eosin (panel A) or demonstrate ER (panel B) or PR (panel C) immunoreactivity (Vector Red chromogen and hematoxylin counterstain). FIGURE 1. This tumor is ER+PR+. FIGURE 2. In this ER+PR− tumor, neoplastic areas with glandular differentiation are ER+ whereas neoplastic areas with solid differentiation are ER−. FIGURE 3. This tumor is ER−PR+. FIGURE 4. This tumor is ER−PR−.
According to the FIGO grading system used for human endometrioid carcinomas (Creasman 1989), 12/18 tumors in this study were grade 1 (≤5% solid growth), 6/18 were grade 2 (6–50% solid growth), and 0/18 were grade 3 (>50% solid growth; Table 1). According to a grading system of low versus high grade, 9/18 tumors in this study were classified as low grade (well-differentiated), and 9/18 were classified as high grade (poorly differentiated; Table 1). Of the 9 high-grade tumors, 6 had a greater degree (6–50%) of solid, nonsquamous growth pattern, corresponding to grade 2 in the FIGO system (Table 1; Figure 2A). In 3 of these 6 high-grade tumors with solid growth, nuclear atypia was greater within the areas of solid growth pattern than in neoplastic glands (Table 1; Figure 2A). Nuclear atypia was generally mild to moderate in all 9 of the low-grade tumors, whereas 5 of the 9 high-grade tumors displayed marked nuclear atypia (Table 1). In contrast to the degree of nuclear atypia, which was frequently greater in high-grade neoplasms, the mitotic index did not show any pattern with tumor grade or nuclear atypia (Table 1). The mitotic index was generally low to moderate in the 18 tumors, ranging from an average of 0.2 to 3.1 per hpf in either the low-grade or grade 1 tumors and from 0.2 to 4.2 per hpf in either the high-grade or grade 2 tumors (Table 1).
Histopathological and immunohistochemical characteristics of the 18 spontaneous EACs in control F344 rats.
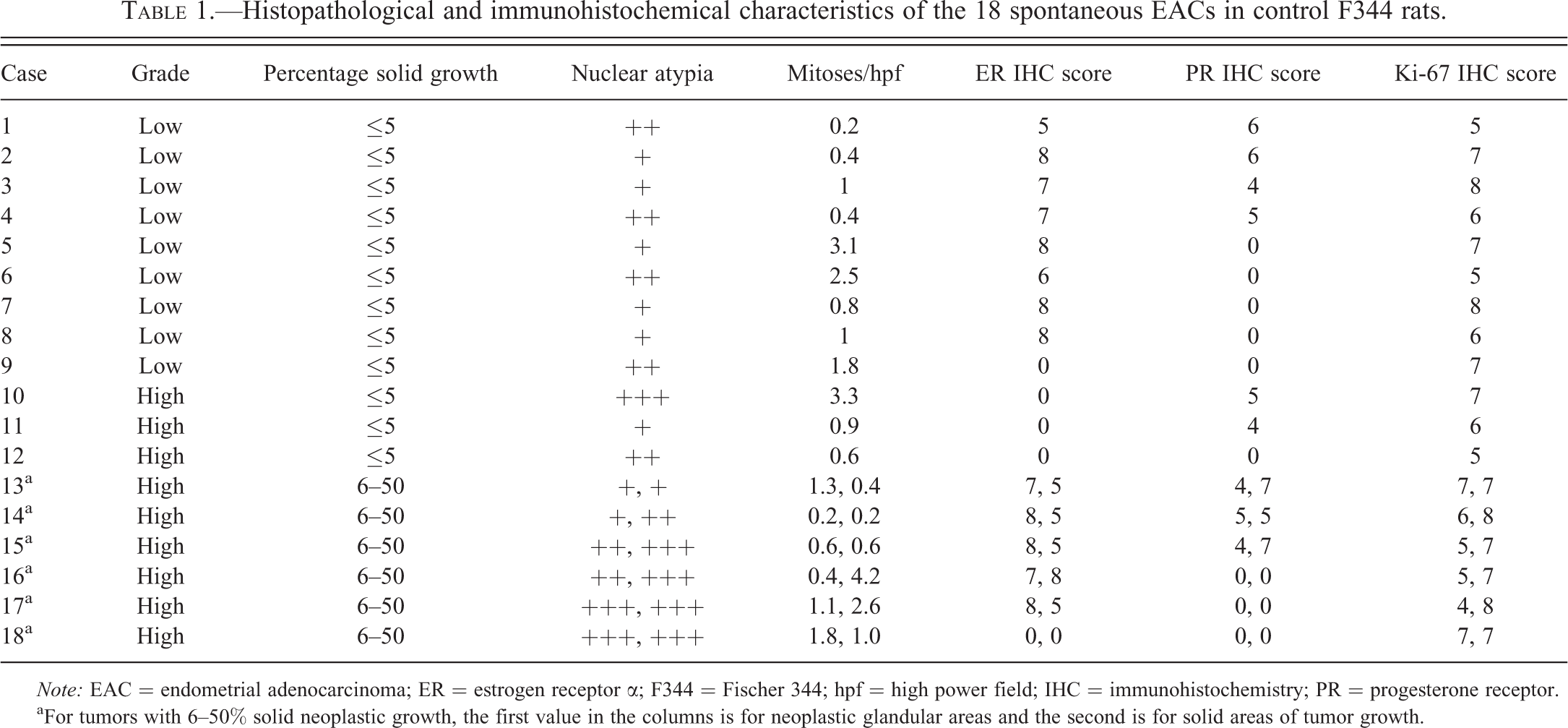
Note: EAC = endometrial adenocarcinoma; ER = estrogen receptor α; F344 = Fischer 344; hpf = high power field; IHC = immunohistochemistry; PR = progesterone receptor.
aFor tumors with 6–50% solid neoplastic growth, the first value in the columns is for neoplastic glandular areas and the second is for solid areas of tumor growth.
Immunohistochemistry
The 18 tumors were heterogeneous in hormone receptor expression, with the 4 possible ER/PR immunophenotype combinations demonstrated in Figures 1 to 4. For those samples that also contained some normal endometrium, both the luminal and glandular epithelia generally displayed mild to moderate nuclear immunoreactivity for ER and PR. Most tumors (13/18; 72.2%) were immunoreactive for ER, whereas only 50% (9/18) were immunoreactive for PR (Tables 1 and 2). Of the tumors positive for one receptor and negative for the other, it was more common to have an absence of PR expression than an absence of ER expression (Table 2). The most common immunophenotype was dual estrogen and progesterone receptor expression, 38.9% (Table 2; Figure 1B, 1C). The second most common immunophenotype was ER+PR−, 33.3% (Table 2; Figure 2B, 2C). Less common were tumors with undetectable expression of both hormone receptors, ER−PR−, 16.7% (Table 2; Figure 4B, 4C). The least common immunophenotype was ER−PR+, 11.1% (Table 2; Figure 3B, 3C).
Percentages of four hormone receptor immunophenotype combinations (ERα and PR) in rat EACs compared to 3 published studies in human EAC.
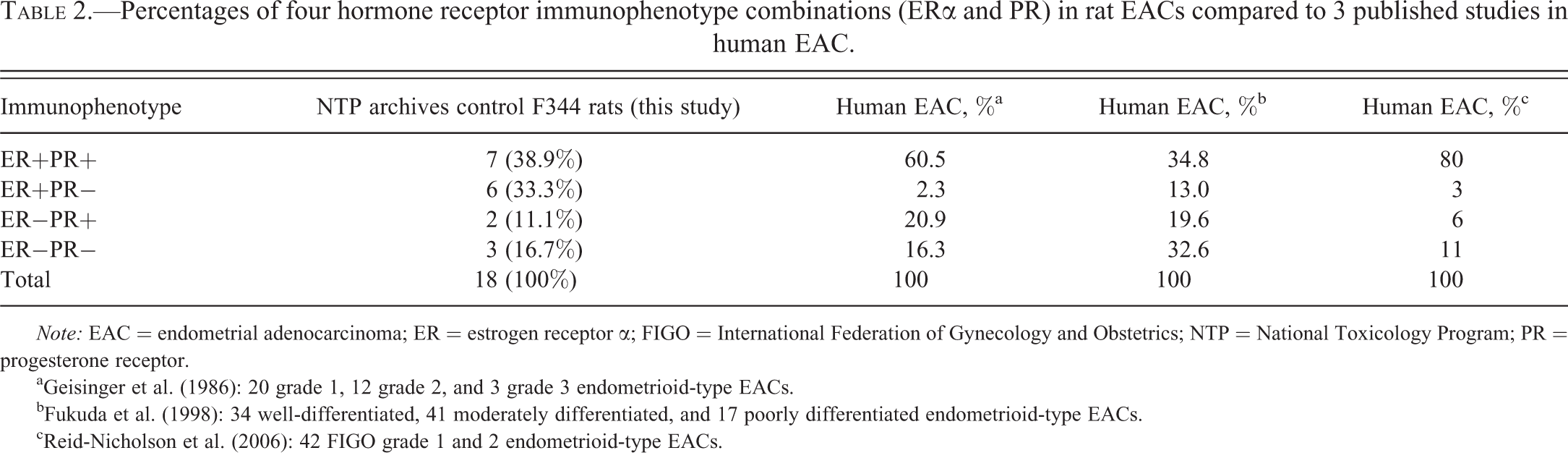
Note: EAC = endometrial adenocarcinoma; ER = estrogen receptor α; FIGO = International Federation of Gynecology and Obstetrics; NTP = National Toxicology Program; PR = progesterone receptor.
a Geisinger et al. (1986): 20 grade 1, 12 grade 2, and 3 grade 3 endometrioid-type EACs.
b Fukuda et al. (1998): 34 well-differentiated, 41 moderately differentiated, and 17 poorly differentiated endometrioid-type EACs.
c Reid-Nicholson et al. (2006): 42 FIGO grade 1 and 2 endometrioid-type EACs.
Tumor differentiation appeared to be associated with ER immunoreactivity, with low-grade tumors (8/9) tending to be ER+ more than high-grade tumors (5/9; Table 1), although the relationship was not statistically significant (data not shown). Of the low-grade tumors, most (8/9) were ER+ and less than half (4/9) were PR+ (Table 1). In contrast, fewer high-grade tumors were ER+ (5/9) than low-grade tumors, but high- and low-grade tumors had similar numbers of PR+ tumors (5/9 and 4/9, respectively; Table 1). Proliferation, represented by Ki-67 immunoreactivity, within high-grade tumors was higher in the neoplastic solid areas than glandular areas in 4 of 6 tumors with solid differentiation (Table 1). Overall, however, Ki-67 immunoreactivity was similar in both high- and low-grade tumors (Table 1).
Discussion
Spontaneously arising EACs in control F344 rats from prior NTP studies were mostly ER+ (72.2%) with varied immunoreactivity to PR. ER and PR immunoreactivities are usually correlated (Fukuda et al. 1998) as estrogen induces PR expression. Most rat tumors expressed both ERα and PR (ER+PR+), as has been observed in human tumors (Table 2; Figure 1B, 1C; Fukuda et al. 1998; Geisinger et al. 1986; Reid-Nicholson et al. 2006). However, 50% of F344 rat EACs were PR−, whereas human tumors are generally PR+ (Tables 1 and 2; Fukuda et al. 1998; Geisinger et al. 1986; Reid-Nicholson et al. 2006). After ER+PR+ tumors, ER+PR− tumors were the next most common immunophenotype in this study, but these tumors are least common in humans (Table 2; Fukuda et al. 1998; Geisinger et al. 1986; Reid-Nicholson et al. 2006). ER+PR− tumors are rare in humans, have a poor prognosis, and tend not to respond to progestin therapy (Bender, Buekers, and Leslie 2011).
The ER+PR− immunophenotype was relatively common in this study, occurring in 1/3 of the F344 rat tumors (Tables 1 and 2; Figure 2B, 2C). The ER+PR− immunophenotype has also been shown to be common in rat strains with a high incidence of spontaneous EAC. For example, in tumor cell lines derived from crosses from the high incidence BDII/Han strain, 9/10 EACs were ER+, whereas only 3/10 EACs were PR+ (Karlsson et al. 2001). The EnDA transplantable tumor model derived from the high-incidence DA/Han strain is ER+PR− but interestingly is inhibited by the progestin medroxyprogesterone acetate, despite lacking progesterone receptor expression (Horn et al. 1993). The discordant immunophenotypes ER+PR− and ER−PR+ may be explained by mutant forms of ER (Bender, Buekers, and Leslie 2011). For example, in ER+PR− tumors, there may be an interruption in the induction of PR by a mutant ER.
In contrast to the F344 rat strain, there are 4 rat strains with a high incidence of spontaneous EAC (Vollmer 2003). The BDII/Han strain has a >90% incidence of EAC (Deerberg and Kaspareit 1987). There is good evidence for hormone dependence in these tumors, with a 0% incidence if the rats are ovariectomized prior to onset of estrous cyclicity or fed daily progestogens for life (Deerberg and Kaspareit 1987; Deerberg et al. 1995). The DA/Han inbred strain has a 62% incidence of spontaneous, generally well-differentiated, highly metastatic EAC, and the EnDA metastasizing transplantable tumor model has been developed from this strain (Horn et al. 1993). The Donryu strain has a 35% incidence of EAC (Nagaoka et al. 1990). Tumors in the Donryu rat appear to be hormone dependent because these animals enter persistent estrus by 9 months, the tumors arise from hyperplastic lesions, and the animals have elevated serum estrogen, with a 5-fold higher serum estradiol:progesterone ratio than F344 rats at 12 months of age (Nagaoka et al. 1990, 1994). The F344 rat was used as a comparison strain by Nagaoka et al. (1990) because it is a low-incidence strain for EAC. Aside from two reports of unusually high incidences of EAC that increased over time to 12% (Ando et al. 2008) and 24% (Nyska et al. 1994), most reports of control animals from several large cohorts show an incidence of EAC in F344 rats of <1% (Chandra and Frith 1992; Haseman, Hailey, and Morris 1998; Maita et al. 1987; Solleveld, Haseman, and McConnell 1984). Not as well characterized as the BDII/Han, DA/Han, or Donryu strains, the Wistar:Han strain has a 5% incidence of uterine epithelial tumors as reported in the Registry of Industrial Toxicology Animal-data (RITA) database of carcinogenicity studies (Harleman et al. 2012), increasing to a 39% incidence of EAC in nulliparous animals if kept to the end of life (Deerberg, Rehm, and Pittermann 1981).
In addition to high-incidence rat strains, Chinese hamsters, cows, and rabbits are additional species that have a high spontaneous incidence of EAC (Brownstein and Brooks 1980; Kennedy et al. 1998). EAC in rabbits is associated with either hyperplasia or senile atrophy and can be induced by estrogen (Elsinghorst, Timmermans, and Hendriks 1984). Rabbit tumors often express both ER and PR, similar to EAC in humans (Asakawa et al. 2008).
The preponderance of PR− tumors (9/18) in this study is noteworthy in light of the fact that progestins are used as a hormonal therapy in some cases of EAC in women, and PR+ status is important for response to therapy and prognosis. Progestin therapy is used in the treatment of recurrent human cases or for fertility sparing in some young premenopausal women with well-differentiated, low-grade tumors (Bovicelli et al. 2013; Yang, Thiel, and Leslie 2011). Hyperplasia, atypical hyperplasia, and even some well-differentiated, noninvasive, low-grade EAC can be reversed by progestin therapy (Yang, Thiel, and Leslie 2011). PR status is a major predictor of the success of progestin therapy. For example, in one study using progestin therapy for recurrent cases, 37% of women with PR+ tumors responded, whereas only 8% of women with PR− tumors responded (Thigpen et al. 1999). Treatment failure can result from PR loss secondary to prolonged progestogen treatment, because progestogens inhibit PR expression (Yang, Thiel, and Leslie 2011). In addition to influencing the success of progestin therapy, positive hormone receptor status in humans is significantly associated with disease-free survival (Creasman 1993). ERα expression (Jongen et al. 2009) or PR expression (Fukuda et al. 1998), or both (Creasman 1993) are significant positive prognostic indicators for survival. Although ERβ expression is not correlated with histologic grade, stage, or survival (Shabani et al. 2007), the absence of ERα or PR expression in human EAC is an indicator of increased grade, aggressive tumor growth, poor prognosis, or decreased survival (Nyholm, Christensen, and Nielsen 1995; Shabani et al. 2007).
When comparing well- and poorly differentiated tumors, more well-differentiated (low-grade) tumors were ER+ (8/9), whereas only 5/9 of the poorly differentiated (high-grade) tumors were ER+ (Table 1). In human EAC, well-differentiated tumors are more likely to express ER and PR than poorly differentiated tumors (Geisinger et al. 1986; Kounelis et al. 2000; Reid-Nicholson et al. 2006). Also, ER expression decreases stepwise from grade 1 to grade 3 in human EAC (Jazaeri et al. 2001). Not only are ER and PR immunoreactivities significantly associated with low tumor grade in human EAC, but they have also been shown to be inversely associated with expression of the proliferation marker Ki-67 (Obeidat et al. 2013). However in the present study, Ki-67 expression was similar across all tumors, regardless of tumor grade (Table 1).
The trend toward loss of ER with high tumor grade was also observed at the level of the individual tumor within the biphasic morphology of both well-differentiated neoplastic glands and more poorly differentiated solid areas. In 4/5 ER+ tumors with solid differentiation, areas with glandular differentiation tended to have a higher ER immunoreactivity score than the less well-differentiated, solid areas (Table 1; Figure 2B). Loss of ERα immunostaining in poorly differentiated parts of a tumor has also been shown in EAC induced by tamoxifen in Wistar:Han rats (Carthew et al. 2000).
Exposure to unopposed estrogenic stimulation is a major risk factor for EAC in women (Amant et al. 2005; Burke et al. 2014). Likewise, there is evidence for excessive serum estrogen levels in some rat strains with a high incidence of EAC such as the Donryu strain (Nagaoka et al. 1990). Chemicals that cause an increased incidence of uterine carcinoma in F344 rats include 3-amino-9-ethylcarbazole hydrochloride (NCI-CG-TR-93 1978a), daminozide (NCI-CG-TR-83 1978b), 4,4′-thiodianiline (NCI-CG-TR-47 1978c), and intrauterine injection of N-ethyl-N′-nitro-N-nitrosoguanidine (Ogino et al. 1989). Chemicals causing an increased incidence of uterine carcinoma in Sprague-Dawley (SD) rats, another commonly used low-incidence strain (0.9%; Harleman et al. 2012), include pentachlorodibenzofuran (PeCDF; carcinoma; NTP TR 525 2006a) and tetrachlorodibenzodioxin (TCDD; squamous cell carcinoma; NTP TR 521 2006b). Surprisingly, the potent xenoestrogens ethinyl estradiol (SD; NTP TR 548 2010) and zearalenone (F344; NTP TR 235 1982) did not induce EAC in the commonly used SD or F344 rat strains. Thus, the influence of excessive or unopposed estrogens on tumorigenesis in F344 rats requires further characterization to understand the relevance for estrogenic risk for humans.
In summary, spontaneous EACs in F344 rats exhibited the diversity of estrogen and progesterone receptor phenotypes observed in women, with diversity sometimes exhibited within a single tumor. ER+PR+ tumors were most common in F344 rats, as has been reported for women. Well-differentiated (low-grade) tumors tended to be ER+, as reported for women. As this is a descriptive study, further work would be necessary to gain a mechanistic understanding of why PR− tumors were somewhat common in F344 rats (9/18) in this study and in rat strains with a high incidence of spontaneous EAC. Given the heterogeneity of immunophenotypes between and within tumors, this study demonstrates the importance of evaluating for presence or absence of hormone receptors in models used for chemical evaluations.
Footnotes
Acknowledgments
The authors thank Ms. Hermina Borgerink for technical assistance with immunohistochemistry, Drs. Tanasa Osborne and Darlene Dixon for their constructive comments on this manuscript, and Ms. Beth Mahler for assistance with figure preparation.
Author Contribution
Authors contributed to conception or design (MC); data acquisition, analysis, or interpretation (CW, RH, MC); drafting the manuscript (CW); and critically revising the manuscript (CW, RH, MC). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JMC has been a consultant for the Silicones Environmental, Health, and Safety Council of North America.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the Silicones Environmental, Health, and Safety Council of North America; in part by NIH training grant T32 OD010957 (CJW); and in part by the NIH, National Institute of Environmental Health Sciences.
