Abstract
Periodic acid-Schiff (PAS)-positive granular deposits in the hippocampus have been reported previously in certain inbred mouse strains such as C57BL/6 and the senescent-accelerated mouse prone-8. Here, we report for the first time that similar PAS-positive granules age dependently occur in SJL/J mice, a mouse strain, for instance, used for central nervous system disease research. Moreover, similar granules stained intensely positive with a polyclonal antibody directed against p75 neurotrophin receptor (p75NTR). Granular deposits were absent in young mice and developed with aging in CA1 and CA2 regions of the hippocampus. Interestingly, granules significantly diminished in SJL/J mice previously treated with cuprizone, a copper chelator, which is a useful model for toxic demyelination. The presented data support the idea that granules might be the result of an imbalance of redox-active metals and/or a dysregulation of complementary mechanisms that regulate their homeostasis in astrocyte–neuron coupling, respectively. It remains to be determined whether the unsuspected immunoreactivity for p75NTR represents a false-positive reaction or whether p75NTR is crucially involved in the pathogenesis of age-related hippocampal granular deposits in mice.
Introduction
Periodic acid-Schiff (PAS)-positive granular deposits have been reported as an incidental finding in the geriatric mouse brain of certain strains such as C57BL/6, senescent-accelerated mouse prone-8 (SAMP8), and others (Jucker and Ingram 1994; Jucker, Walker, Kuo, et al. 1994a; Jucker, Walker, Schwarb, et al. 1994b; Krass et al. 2003; Manich et al. 2013). However, in the majority of strains, these granules are rarely reported and many inbred strains such as BALB/c, A/J, CBA/J, and DBA/2J lack such granules (Jucker and Ingram 1994; Jucker, Walker, Kuo, et al. 1994a).
Extracellular PAS-positive granular deposits are primarily located in the hippocampus but may also occur in other regions of the brain such as in piriform cortex and cerebellum (Jucker, Walker, Schwarb, et al. 1994b). They have shown to gradually increase in size, number, and distribution during aging (Jucker, Walker, Kuo, et al. 1994a). Although certain strains of mice such as C57BL/6 exhibit age-related cognitive impairments, linked to functional alterations of the hippocampus (Fouquet et al. 2011), it remains unclear whether the formation of granules is causally associated with functional abnormalities. SAMP8 mice exhibit an earlier onset of granule formation, starting as early as 2 to 4 months (Akiyama et al. 1986). The center of individual granules consists of aggregated crystalline-like fibrillar and granular material and contains no resemblance to any cell organelle. Granules are surrounded by a discontinuous membrane and the space in between contains some abnormal mitochondria and other membrane-like structures, which are not completely degenerated (Kuo et al. 1996). The majority of granules are arranged in clusters, anatomically in close association with astrocytic processes in C57BL/6 mice, while some are found scattered in the neuropil (Jucker, Walker, Kuo, et al. 1994a; Kuo et al. 1996). Moreover, it has been proposed that granule formation might be linked to neurons, for instance, in terms of enlarged synaptic terminals (Manich et al. 2011; Mitsuno et al. 1999). PAS reactivity of granules has been proposed to indicate a predominant carbohydrate composition such as glycogen, glycoproteins, and proteoglycans (Jucker, Walker, Kuo, et al. 1994a; Manich et al. 2013). Hippocampal granules in the rodent brain share antigenic properties with Lafora bodies and similarities with corpora amylacea, indicating polyglycosan composition; however, PAS-positive granules in mice differ in size, distribution, and organization (Mitsuno et al. 1999). In addition to argentophilia in certain silver staining procedures, granules were demonstrated to react positive with antibodies against heparan sulfate proteoglycan (HSPG; Jucker, Walker, Kuo, et al. 1994a; Kuo et al. 1996). Since HSPG is involved in the pathogenesis of Alzheimer’s disease, it has been suggested that these granules might share some early aspects with Alzheimer’s disease pathogenesis. Moreover, immunoreactivity for amyloid β (Aβ) and hyperphosphorylated tau protein in granules of SAMP8 mice highlighted this strain as an attractive model for neurodegenerative diseases such as Alzheimer’s disease and tauopathies (Manich et al. 2013; Manich et al., 2011). However, as a major drawback in this conclusion, pseudoimmunoreactivity with many polyclonal and monoclonal antibodies including antibodies against Aβ and tau protein has been reported (Jucker and Ingram 1994; Manich et al. 2013). In particular, the presence of a neoepitope of glycosidic nature was recently demonstrated in granular structures of SAMP8 mice, which is recognized by a contaminant immunoglobulin (Ig)M present in several antibodies obtained from mouse ascites and both mouse and rabbit sera (Manich et al. 2013).
SJL is a Swiss mouse inbred strain from Swiss-Webster stock from 3 different sources, developed by Lambert in 1955. It has become widely used in many fields of research including central nervous system disease. No data for SJL mice regarding age-related granular deposits exist so far.
Materials and Methods
In order to screen for the potential occurrence of PAS-positive granules in SJL/J mice, the brains of 17 neurologically healthy SJL/J (SJL/JOlaHsd) mice were used (referred to as screening study in the following text). The subjects consisted of animals with an age of 33 to 391 days. The mice were kept according to the applicable German regulations of use of animals in research (file reference/local permission number: 42500/1H). Sacrifice was done by general anesthesia with medetomidine (2.5 mg/kg) and ketamine (500 mg/kg). The collected brains were fixed in 10% formalin for 24 hr, embedded in paraffin wax, and cut at 4-µm thickness in coronal sections with a microtome.
In addition, the occurrence of PAS-positive granules was evaluated on archived brain tissue of SJL/JCrl mice, used in an earlier and unrelated animal experiment of a combined infection with Theiler’s murine encephalomyelitis virus (TMEV) and cuprizone treatment, respectively (permission number: 33.9.42502-04/07/1331), referred to as cuprizone study in the following text (Herder et al. 2012). In order to assess the effect of cuprizone treatment on the formation of granules, TMEV-infected mice were excluded from the present study. The control group consisted of 11 age-matched animals, which received a mock injection, whereas the cuprizone-treated group gained cuprizone feeding (n = 9) in addition to mock infection. The latter group was fed ad libitum with 0.2% (w/w) cuprizone (bis(cyclohexanone) oxaldihydrazone, C9012; Sigma-Aldrich, Germany) mixed into a powdered standard rodent chow (ssniff® R/M-H; Soest, Germany) with cuprizone addition between the age of day 70 and 105 (Herder et al. 2012). Brains were screened for the presence of hippocampal granules at an age of 231 days, 126 days after cessation of cuprizone feeding (control: n = 6; cuprizone treated: n = 5), and 280 days, 175 days after cessation of cuprizone feeding (control: n = 5; cuprizone treated: n = 4), respectively.
All samples were stained with hematoxylin and eosin (H&E) and PAS. Moreover, immunohistochemistry using a polyclonal rabbit anti-p75 nerve growth factor receptor (p75 neurotrophin receptor (p75NTR)) antibody (AB1554 Millipore; primary dilution 1:800) was performed according to standard procedures. Briefly, sections were deparaffinized and rehydrated through a graded series of alcohols. Endogenous peroxidase activity was blocked by treatment of slides with 30% H2O2 and 85% ethanol. Blocking was done using goat normal serum diluted in phosphate-buffered saline (PBS) 1:5 at room temperature for 20 min. Pretreatment for antigen retrieval consisted of 15 min with Triton-X (0.3%). Sections were incubated with the primary antibody overnight at 4°C. A biotinylated goat antirabbit secondary antibody was used, followed by detection with avidin–biotin–peroxidase complex (ABC; Vector Laboratories, Burlingame). 3,3′-Diaminobenzidine tetrahydrochloride (DAB) with H2O2 (0.03%, pH 7.2) served as a chromogen.
Multiple microphotographs were taken from all slides using an Olympus bx51 camera system. Total hippocampal area was measured using analySIS® 3.1 software (SOFT Imaging System, Münster, Germany). Quantification of granules was done on PAS-stained slides as well as on sections that underwent p75NTR immunohistochemistry, by counting the number of granule clusters. Here, a cluster was defined as exhibiting more than 3 individual granules. For each slide, the number of clusters per square millimeter of the hippocampus area was calculated and used for statistical analyses. Additionally, the mean number of granules per cluster was evaluated. For all statistical analyses, SPSS version 20 (IBM Corporation, Armonk, NY) was used, and p values lower than .05 were considered significant. Spearman rank correlation coefficient was calculated to evaluate a possible correlation between PAS-positive clusters and age as well as PAS positivity and p75NTR immunoreactivity, respectively. Nonparametric Mann-Whitney U test was applied to compare the number of clusters between control animals and cuprizone-fed mice. Box plots were created using GraphPad Prism, version 6.01 (GraphPad Software, La Jolla, CA).
Results
Clusters of PAS-positive Granules Age Dependently Increase in the Hippocampus of SJL/J Mice
No granules were seen in H&E-stained slides in any of the SJL/J mice of the screening study; however, numerous granules, which reacted intensely positive with PAS staining, were noted in all animals with an age over 230 days, while they were entirely absent in young animals below 54 days of age. The individual granules exhibited a round homogeneous morphology with a size of approximately 2 to 5 µm and were located predominantly in the CA1 and CA2 region of the hippocampus, while they were not observed in the CA3 region. Granules were found in all sublayers of the hippocampus, excluding granular, and polymorph layer of the dentate gyrus. The granules were commonly haphazardly arranged in round to oval clusters, consisting of a mean of 12.75 individual granules (Figure 1). As assessed in PAS staining, the number of hippocampal clusters showed a highly significant correlation with the age of the animals (Spearman’s r = .856; p < .0001; Figure 2).
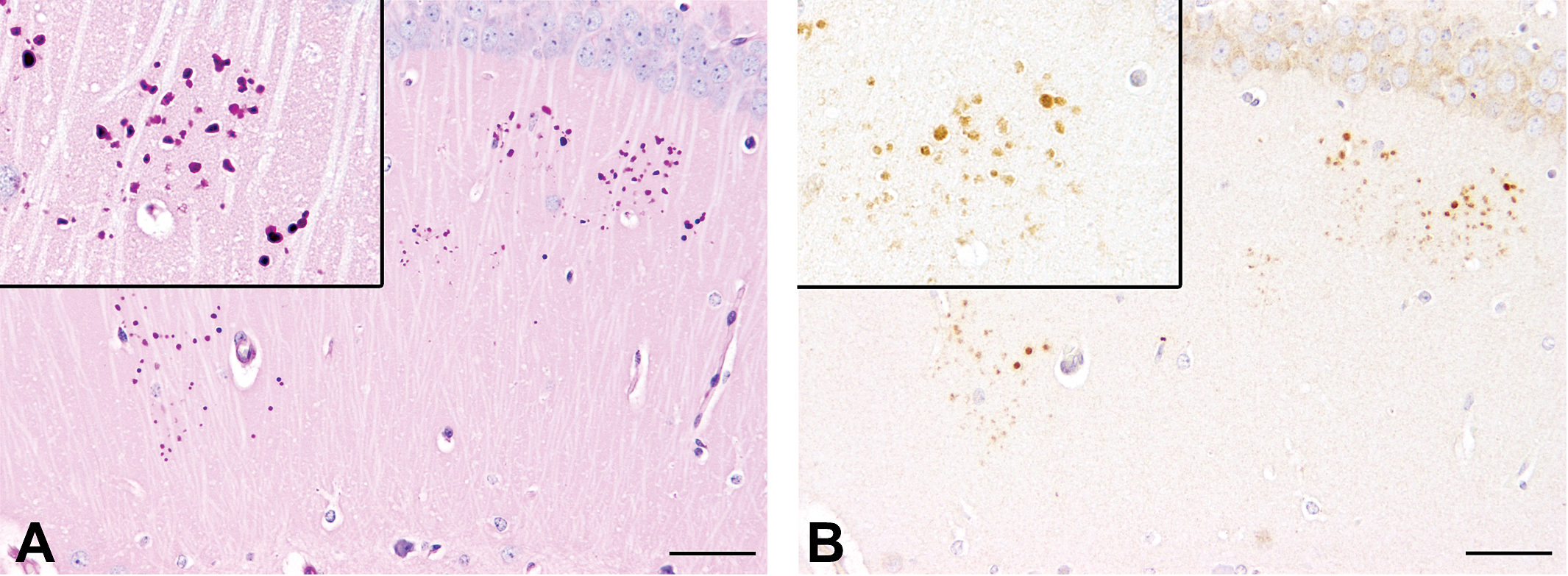
Periodic acid-Schiff (PAS) staining (A) and p75 neurotrophin receptor (p75NTR) immunohistochemistry (B) for granules in the hippocampus of a 336-day-old SJL/J mouse. (A) PAS staining reveals numerous intensely positive clusters of granules, which predominantly appear in the CA1 and CA2 region of the hippocampus. Inset: magnification of a cluster, consisting of more than 30 individual granules, which itself measure approximately 1 to 3 µm in diameter. (B) Serial section of (A). Granules stain positive for p75NTR. Inset: magnification of p75NTR-immunoreactive granules. Scale bars = 50 µm.
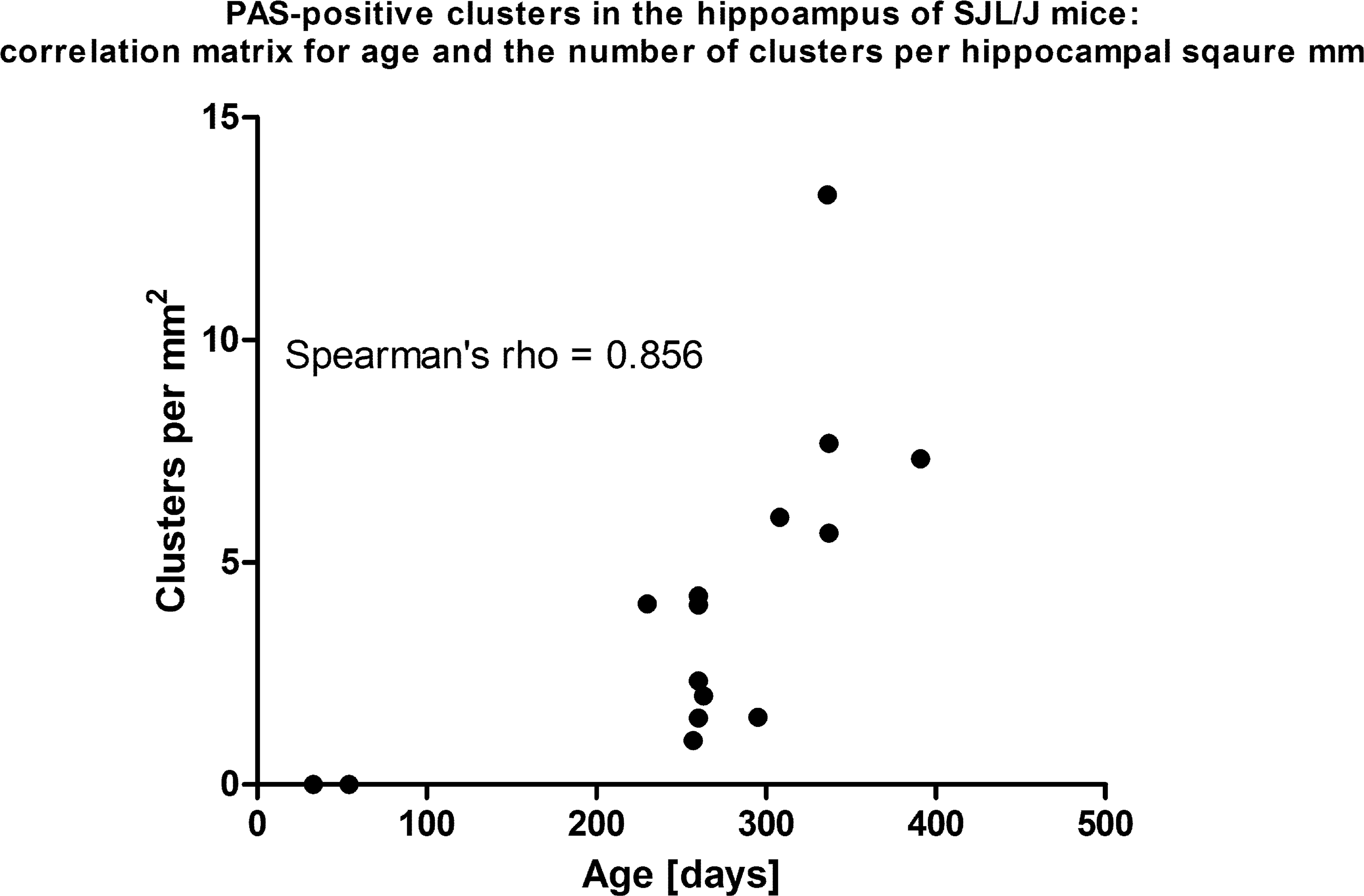
Correlation matrix displaying the interdependence of age and the number of periodic acid-Schiff (PAS)-positive clusters in the hippocampus of SJL/J mice. There is strong correlation between both variables as estimated by Spearman’s rank correlation coefficient (.856).
Cuprizone Treatment Diminishes the Number of PAS-Positive Granular Clusters
Control mice of the cuprizone study exhibited some clusters in the hippocampus (Figure 3) comparable to the SJL/J mice from the screening study. Generally, mice, with an age of 231 days old showed less granules than mice with an age of 280 days (Figure 4); however, the number did not reach such high amounts as in mice with an age of 300 days and older derived from the screening study. Interestingly, in mice previously fed with cuprizone, remarkably less PAS-positive clusters were observed than in age-matched controls (Figure 3). Specifically, at an age of 231 days, the number of PAS-positive clusters was significantly lower in mice previously treated with cuprizone as compared to age-matched controls (p = .009; Figure 4). However, the mean number of granules per cluster did not show significant differences in this group (p = .329; data not shown). At an age of 280 days, the number of PAS-positive granular clusters was still appreciated to be lower in previously cuprizone-fed mice compared to age-matched controls, although this difference did not reach the level of significance (p = .063; Figure 4). However, the mean number of granules was significantly lower in animals previously treated with cuprizone compared to controls (p = .032; data not shown).
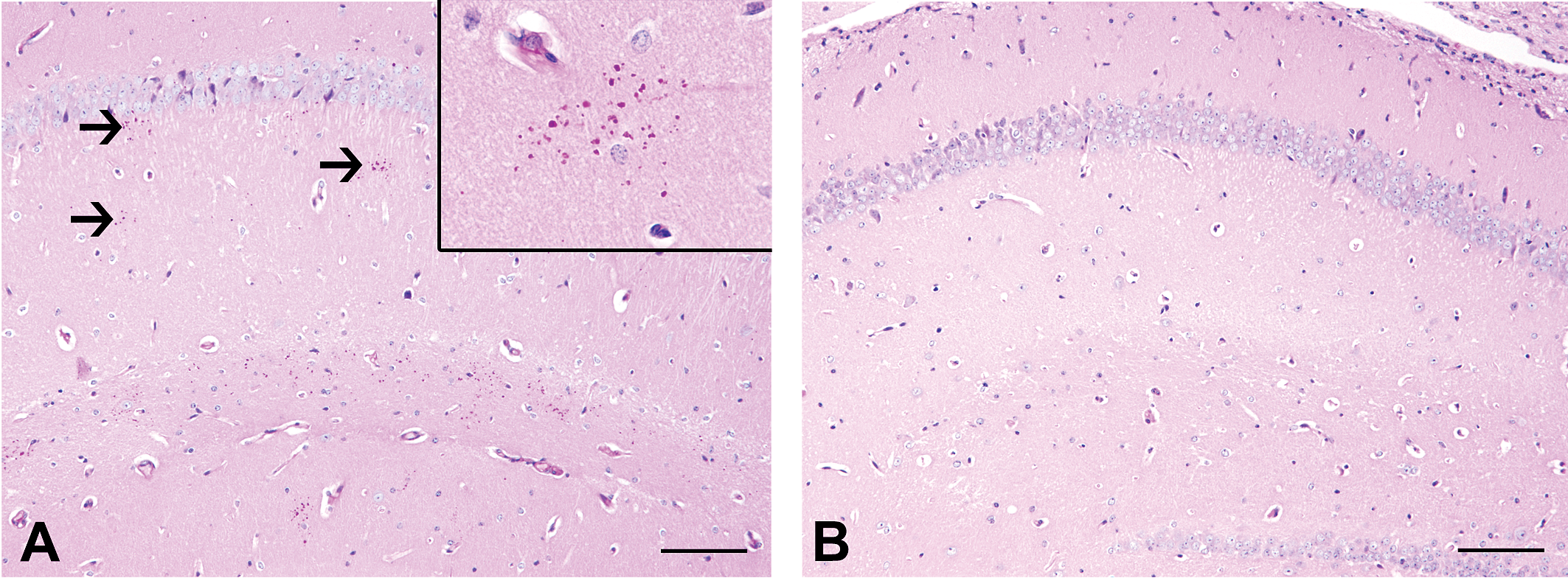
Periodic acid-Schiff (PAS) staining for granules in the hippocampus of a control mouse (A) and an age-matched SJL/J mouse, which received treatment with the copper chelator cuprizone. (A) PAS staining reveals some positive clusters of granules in the CA1 region of the hippocampus (arrows). Inset: higher magnification of a cluster of PAS-positive granules of the same mouse. (B) In contrast, the age-matched mouse, previously treated with cuprizone, lacks clusters. Scale bars = 100 µm.
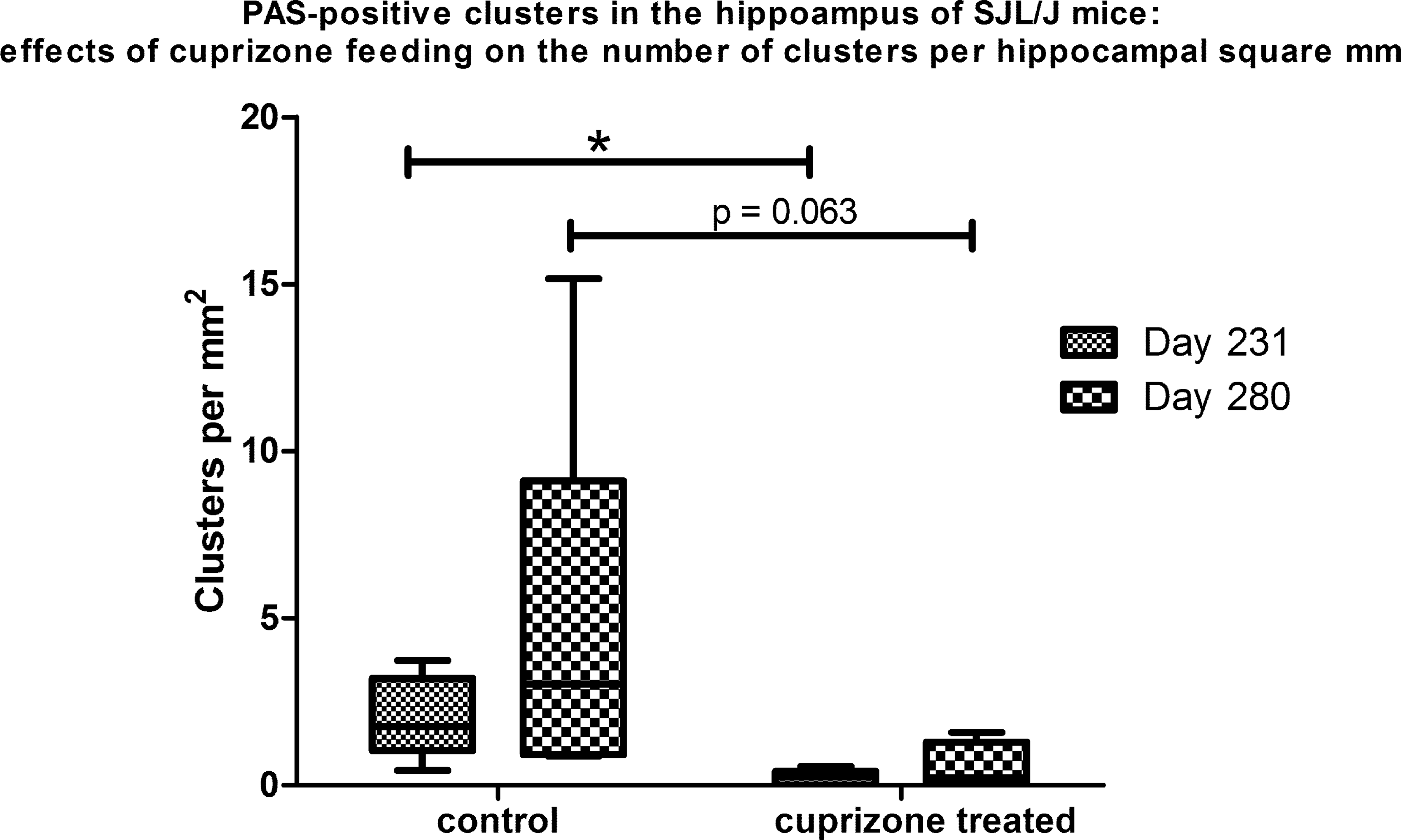
Box and Whisker plots displaying the effect of cuprizone treatment on the number of periodic acid-Schiff (PAS)-positive clusters in the hippocampus of SJL/J mice. Mice were fed with cuprizone, a demyelinating agent, from the age of 70 days up to 105 days; 126 days after cessation of treatment, at an age of 231 days cuprizone-fed mice have significantly less PAS-positive hippocampal clusters as compared to age-matched controls (*p = .009); 175 days after cessation of treatment, at an age of 280 days, the mean number of hippocampal granules is still appreciated to be higher in controls than in cuprizone-fed mice, however, not reaching the level of significance (p = .063). Mann-Whitney U test.
Similar Clusters of Granules React Immunopositive for p75NTR
Using a polyclonal antibody directed against p75NTR, the granules stained intensely immunopositive (Figure 1). p75NTR immunoreactivity strongly correlated with PAS staining (Spearman’s r = .968; p < .0001) in SJL/J mice of the screening study, suggesting that both stainings reacted with similar structures. Similarly, the number of clusters per hippocampal area showed a strong correlation in mice derived from the cuprizone study (Spearman’s r = .733; p = .010 at day 231 and Spearman’s r = .812; p = .008 at day 280, respectively). In order to substantiate the positivity of granules for both PAS and p75NTR, a double-staining procedure was performed on representative slides. Subsequent immunohistochemistry for p75NTR of sections, which were previously stained with PAS, revealed a colocalization of both staining methods in granules, although it appeared relatively blurred due to strong interference of both stainings in the same structures (Figure 5).
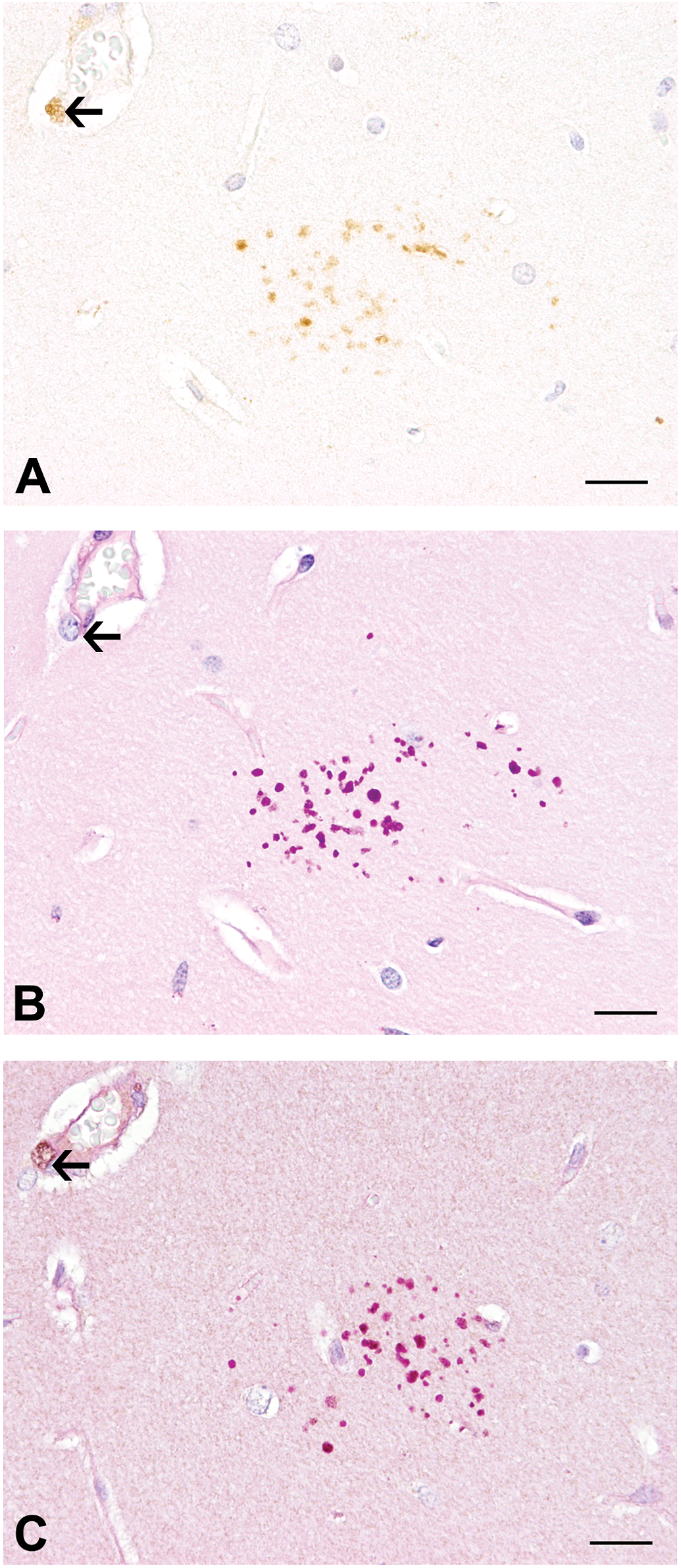
p75 neurotrophin receptor (p75NTR) immunohistochemistry (A), periodic acid-Schiff (PAS) staining (B), and double staining with PAS, followed by immunohistochemistry for p75NTR (C) of granules in the hippocampus of an aged SJL/J mouse in serial sections. The double-staining procedure reveals simultaneous positivity of granules for both PAS and p75NTR in (C), although the signal appears blurred due to strong staining interference. Note the perivascular cell (arrows), which reacts positive for p75NTR in (A) and (C) but not with PAS (B). Scale bars = 20 µm.
Discussion
The present results demonstrate that PAS-positive granular structures occur in the hippocampus of SJL/J mice, a mouse strain not reported to be prone to this phenomenon before. The described lesion is consistent with similar granules, reported to age dependently emerge in other inbred mouse strains such as C57BL/6 and SAMP8 mice, thus emphasizing that this unusual lesion may be less strain specific than previously thought.
Interestingly, we unraveled that cuprizone feeding of mice resulted in diminished numbers of hippocampal PAS-positive clusters long term after cessation of treatment, indicating a preventive impact of cuprizone on the formation of hippocampal granules. Cuprizone is a copper chelator, used as an efficient demyelinating agent, and thus represents an important model for demyelinating diseases such as multiple sclerosis (Herder et al. 2011, 2012; Skripuletz et al. 2013).
Modulatory effects of certain drugs on the formation of such granules have been reported previously. Here, treatment of SAMP8 mice with resveratrol, a caloric restriction mimetic with antidiabetogenic properties, and feeding of apolipoprotein E-deficient mice with an antioxidant diet have shown to similarly result in a decrease in granules in the hippocampus (Porquet et al. 2013; Veurink et al. 2003). In fact, several lines of evidence indicate that age-dependent hippocampal granules may be the result of oxidative stress (Veurink et al. 2003; Porquet et al. 2013).
The exact reason for the decreased number of PAS-positive clusters in mice treated with cuprizone remains unclear. It should be emphasized that the number of investigated individuals is rather low and prevents conclusive interpretations on this finding. However, despite the low number of individuals, our observations indicate that there might be an inverse effect of cuprizone-induced alteration in the levels and bioavailability of redox-active metals and their dependent enzymes on the formation of granules. The hippocampus has unique regulatory demands for metals, such as copper, iron and zinc, as each contributes to synaptic plasticity, and copper and zinc are pivotal neuromodulators in synapses (Jones, Beard, and Jones 2008). The exact mechanisms of general cuprizone toxicity are not fully understood so far. However, the neurotoxic effect of cuprizone is believed to be in part attributed to its capability of chelating copper, ultimately leading to chronic copper deficiency with subsequent inhibition of copper-dependent enzymes (Benetti et al. 2010). Moreover, activity of monoamino oxidase B, an iron-dependent enzyme, has been detected in PAS-positive granules of SAMP8 mice (Nakamura et al. 1995). Interestingly, cuprizone has been shown to inhibit monoamino oxidase B activity in C57BL/6 mice (Xu et al. 2009). The preventive effect on the formation of PAS-positive granules might thus be explained by the persistence of altered redox-active metals homeostasis in the brain for a prolonged time after cessation of cuprizone feeding.
Interestingly, excess of redox-active metals such as copper and iron can lead to the generation of toxic reactive oxygen species. Astrocytes are considered to be the main regulators of metal homeostasis in the brain (Scheiber, Mercer, and Dringen 2014). Histochemically, the highest cellular copper levels are found in astrocytes (Scheiber, Mercer, and Dringen 2014). Copper-induced stimulation of glycolytic flux in astrocytes could have far-ranging consequences for the glucose metabolism of the brain in conditions of copper overload (Scheiber, Mercer, and Dringen 2014). In fact, the composition of PAS-positive granules points to some abnormalities in glucose metabolism (Mitsuno et al. 1999). Interestingly, clusters of PAS-positive granules were observed in CA1 and CA2 but were absent in the CA3 region of the hippocampus. This might be explained by the fact that neurons in the hippocampal CA1 region are shown to be particularly sensitive to oxidative stress, whereas those in CA3 are comparatively resistant (Wang et al. 2007), potentially suggesting compromised astrocyte protection.
Similar granules exhibited intense immunoreactivity for p75NTR, which has not been reported so far. Reported immunopositivity of granules includes Aβ, phosphorylated tau, HSPG, laminin, and others (Manich et al. 2011, 2013). A recent report however significantly contributed to the elucidation of partly diverging and contrary results regarding the immunoreactivity of hippocampal granules, especially in SAMP8 mice. The authors demonstrated that false-positive immunoreactivity of granules is attributed to contaminant IgM in many commercially available antibodies, thus challenging previously reported immunopositivity for antigens implicated to play a role in Alzheimer’s disease (Manich et al. 2013). These authors characterized a neoepitope of glycosidic nature in the granules, which was the causative factor for the production of false-positive results (Manich et al. 2013).
In our study, immunoreactivity of similar granules was demonstrated using a polyclonal antibody against p75NTR. In light of recent reports on pseudoimmunoreactivity of granules, a false-positive reaction has to be taken into account. However, the presented findings warrant further investigations on this topic. In fact, p75NTR is a pivotal molecule in many neurodegenerative diseases, such as Alzheimer’s disease, and plays a crucial role in many demyelinating diseases (Knowles et al. 2013; Lempp et al. 2014 ).
Taken together, the presented results demonstrate that (a) SJL/J mice represent another so far not described mouse strain, which is prone to the formation of hippocampal granules, (b) cuprizone feeding diminishes the number of clusters of PAS-positive clusters, indicating that cuprizone due to its ion chelator capability may influence oxidative stress in the hippocampus, and (c) hippocampal granules in SJL/J mice appear to be immunoreactive for p75NTR, warranting further investigations on the potential role of p75NTR in the pathogenesis of this unusual age-dependent lesion in the hippocampus of mice.
Footnotes
Acknowledgments
The authors gratefully acknowledge the excellent technical assistance of B. Buck, P. Grünig, C. Namneck, C. Herrmann, C. Schütz, and K. Schöne.
Author Contribution
Authors contributed to conception or design (A.C., W.B., and I.S.); data acquisition, analysis, or interpretation (A.C., V.H., F.H., A.B., W.B., and I.S.); drafting the manuscript (A.C., W.B., and I.S.); and critically revising the manuscript (V.H., F.H., and A.B.). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A. Cana was supported by a scholarship provided by the European Union.
