Abstract
Testicular effects of chemical mixtures may differ from those of the individual chemical constituents. This study assessed the co-exposure effects of the model germ cell- and Sertoli cell-specific toxicants, X-irradiation (x-ray), and 2,5-hexanedione (HD), respectively. In high-dose studies, HD has been shown to attenuate x-ray-induced germ cell apoptosis. Adult rats were exposed to different levels of x-ray (0.5 Gy, 1 Gy, and 2 Gy) or HD (0.33%), either alone or in combination. To assess cell type-specific attenuation of x-ray effects with HD co-exposure, we used laser capture microdissection (LCM) to enrich the targeted cell population and examine a panel of apoptosis-related transcripts using PCR arrays. The apoptosis PCR arrays identified significant dose-dependent treatment effects on several genes, with downregulation of death receptor 5 (DR5), Naip2, Sphk2, Casp7, Aven, Birc3, and upregulation of Fas. The greatest difference in transcript response to exposure was seen with 0.5 Gy x-ray exposure, and the attenuation effect seen with the combined high-dose x-ray and HD did not persist into the low-dose range. Examination of protein levels in staged tubules revealed a significant upregulation in DR5, following high-dose co-exposure. These results provide insight into the testis cell-specific apoptotic response to low-dose co-exposures of model testicular toxicants.
Introduction
There is a need for improved methods to study toxicant exposures in the context of complex mixtures or co-exposures, as exposure to single toxicants is a rare occurrence. Many factors influence the complexity of mixture studies, including the number of cellular and molecular targets, the amount and dose levels of the toxicant mixture components, and the interactions that can occur between these components. To add to this, the study of low-dose effects of toxicant mixtures becomes complicated because many of the common tools for assessing toxicological effects are no longer applicable. Most often, the doses studied produce low-level effects that are difficult to measure analytically as they fall below the limits of detection of lower-precision platforms, such as gene microarrays. These low-level effects induced by low-dose mixtures of toxicants can still have biological significance, when they are spread across an entire pathway, such as apoptosis.
The use of model toxicants with specific target organs has allowed for the investigation into the underlying mechanisms of mixtures exposures. Recent studies with well-characterized model germ cell- and Sertoli cell–specific toxicants, X-irradiation (x-ray), and 2,5-hexanedione (HD), respectively, have been useful in the investigation of co-exposure effects in the testis. HD is a metabolite of n-hexane and methyl-n-butyl ketone, two commonly used solvents, and HD exposure inhibits Sertoli cell function through enhanced microtubule assembly and stability (Boekelheide et al. 2003). X-ray induces apoptosis in the actively dividing spermatogonia cell population through DNA damage (van der Meer et al. 1992). Also, certain stages of spermatogenesis are more susceptible to x-ray exposure (Hasegawa et al. 1997). High-dose studies performed with these toxicants have demonstrated that x-ray-induced germ cell apoptosis is attenuated following a priming exposure to HD (Campion et al. 2010a; Campion et al. 2010b; Yamasaki, Sandrof, and Boekelheide 2010).
Although the above studies begin to reveal mechanistic insights into mixtures behavior, these have largely been with whole testis tissues. Whole testis studies provide some insight into the response to toxicant exposure, but they are limited in that the actual effect of the sensitive cell population is diluted when combined with other testicular cell types. The testis is a particularly complex tissue, with several interacting cell types and germ cells in varying stages of development. To overcome this issue, we used laser capture microdissection (LCM), which has become a particularly useful tool in the study of toxicant exposure in the testis. LCM has many applications, including the molecular profiling of diseases (i.e., tumor cells from an organ; Lili et al. 2013; Murphy et al. 2013) and the examination of cell-type-specific toxicant responses (Campion et al. 2010b; Sluka et al. 2008). In the exploration of stage-specific testicular sensitivity to x-ray exposure, it was found that stages I through VI are the most susceptible and that the greatest increase in germ cell apoptosis is seen in stages II and III (Yamasaki, Sandrof, and Boekelheide 2010). When applied to the examination of the stage-specific effects of high doses of HD and x-ray in an LCM selected sensitive cell population, LCM revealed that Fas induction by 5 Gy x-ray is significantly attenuated by HD co-exposure (Campion et al. 2010b). Attenuation of Fas with HD and x-ray co-exposure within the sensitive cell population only begins to uncover what occurs following exposure in the complex apoptotic pathway.
The apoptosis pathway is useful in the investigation of toxicant mixtures in the testis, as germ cell apoptosis has been identified as the ultimate adverse effect of both HD and x-ray exposure. Apoptosis is a complex system of cell death that can be activated either through intracellular driven “stress sensors” such as p53 or through extracellular signals such as the cell surface receptors Fas and DR5. The activation of the apoptotic pathway through either mechanism results in p53-mediated activation of the caspase cascade through upregulation of Bcl2-associated X protein (Bax), Fas, and DR5, with simultaneous repression of Bcl2. This cascade ultimately results in the apoptosis of the cell. (For an in-depth review of male germ cell apoptosis, see (Shaha, Tripathi, and Mishra 2010).
Low dose exposures remain largely unstudied and many risk assessment decisions for low doses have been based on high-dose extrapolations, which do not necessarily reflect actual low-dose exposures (Amundson, Do, and Fornace 1999). To enhance our ability to detect small gene changes in low-dose exposures that would otherwise have been lost within the noise inherent in whole genome array studies, LCM-derived material was used in conjunction with an apoptosis pathway–specific real-time polymerase chain reaction (qRT-PCR) array platform. We hypothesized that low-dose exposure to HD and x-ray and examination of an LCM enriched cell population would allow for the identification of a transition point in the time line of adaptive and adverse effects within the apoptosis pathway. With the combination of an improved LCM method for qRT-PCR array application, a potential adaptive response of 0.5 Gy x-ray is shown with an overall decrease in expression of both pro- and antiapoptotic genes. Seminiferous tubules were also staged through transillumination-assisted dissection for examination of the protein levels of significantly altered transcripts, which illustrated significant upregulation of a key apoptosis mediator. This study sheds light on the complex stage-specific apoptotic response of the testis to co-exposures of model toxicants.
Materials and Methods
Animals
Adult male Fischer 344 rats (200–250 g) were purchased from Charles River Laboratories (Wilmington, MA) and allowed to acclimate for 1 week after arrival. All rats were housed in community cages in a temperature- and humidity-controlled environment with 12 hr light–dark periods and given Purina Rodent Chow 5001 (Farmer’s Exchange, Framingham, MA) and water ad libitum. All experimental animal protocols were approved in compliance with the National Institute of Health guidelines by the Brown University Institutional Animal Care and Use Committee.
Chemicals
HD (CAS# 110-13-4, ≤99% purity), and all other chemicals were purchased from Sigma Aldrich (St. Louis, MO) unless otherwise noted.
Toxicant Exposure
An established treatment protocol (Markelewicz, Hall, and Boekelheide 2004; Yamasaki, Sandrof, and Boekelheide 2010) was used to expose the rats to 2,5-hexanedione (HD) in the drinking water for 17 days as a 0.33% solution. For the co-exposure groups, animals were given a subsequent exposure of either 0.5 Gy, 1 Gy, or 2 Gy caudal half-body radiation on the 17th day of exposure, at a dose rate of 0.31 Gy/min using an RT 250 Philips kVp x-irradiation machine (Philips, Hamburg, Germany). The time line of the dosing paradigm for HD and x-ray co-exposure, in addition to the 3-hr and 12-hr time point for tissue collection, is illustrated in Figure 1. This resulted in a total of seven treatment groups: control, 0.33% HD, 0.5 Gy x-ray, 1 Gy x-ray, 2 Gy x-ray, 0.33% HD + 0.5 Gy x-ray, 0.33% HD + 1 Gy x-ray, and 0.33% HD + 2 Gy x-ray. At 3 hr following irradiation, a group of rats was euthanized by CO2 asphyxiation and testes were collected (n = 5; for each of the seven treatment groups) and immediately embedded in optimal cutting temperature (OCT) compound (Sakura Finetek, Torrance, CA), as described previously (Yamasaki, Sandrof, and Boekelheide 2010) for LCM. Another group of rats were euthanized at 12 hr following irradiation for transillumination seminiferous tubule dissection. The testes were collected (n = 5; control, 0.33% HD, 2 Gy x-ray, 0.33% HD + 2 Gy x-ray) and either embedded in OCT compound, fixed in modified Davidson’s fluid for plastic and paraffin embedding, or detunicated, longitudinally bisected, and placed in 35°C collagenase solution (0.25% trypsin [Life Technologies, Grand Island, NY], and 0.1% Type III collagenase, in phosphate-buffered saline [PBS]) for transillumination dissection of spermatogenesis stages I through VI, to mimic the stages captured for transcriptional analysis.
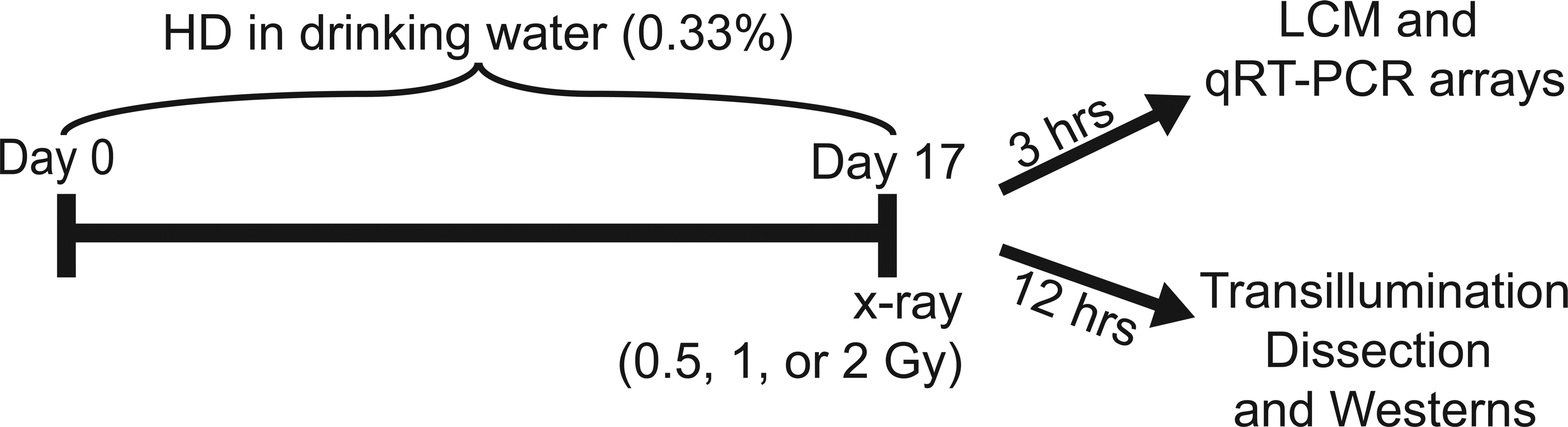
Exposure paradigm. HD was administered to Fischer 344 rats as a 0.33% solution in the drinking water ad libitum for 17 days. X-ray was given as a single dose of 0.5 Gy, 1 Gy, or 2 Gy on the 17th day. Animals were divided into two time point groups, depending on downstream endpoints. For the 3 hr time point, the animals were exposed to all doses and dose combinations (n = 5; seven groups: 0.33% HD, 0.5 Gy x-ray, 1 Gy x-ray, 2 Gy x-ray, 0.33% HD + 0.5 Gy x-ray, 0.33% HD + 1 Gy x-ray and 0.33% HD + 2 Gy x-ray) and testes were taken at 3 hr following x-ray exposure for LCM and qRT-PCR arrays. For the 12 hr time point, a group of animals was dosed only with the highest doses (n = 5; 0.33% HD, 2 Gy x-ray, and 0.33% HD + 2 Gy x-ray) and the testes were taken for transillumination dissection and Western blotting.
LCM and qRT-PCR Arrays
Given the stage-specific effects of HD and x-ray on apoptosis (Campion et al. 2010b; Yamasaki, Sandrof, and Boekelheide 2010), the seminiferous tubules were staged according to the position and shape of the elongated spermatid nuclei using the standards outlined by Leblond and Clermont (1952). LCM, RNA extraction, and RNA concentration were performed as described previously in a companion article (Catlin et al. 2014). Briefly, 10 μm sections of frozen embedded testis tissue were fixed with 75% ethanol, stained with Arcturus Histogene Staining Solution (Applied Biosystems, Carlsbad, CA, USA), dehydrated and allowed to air dry in a hood prior to LCM. LCM was performed with the PixCell IIe Laser Microdissection System (Arcturus Bioscience, Inc.) according to the manufacturer’s protocol. The cell layers of seminiferous tubules at spermatogenesis stages I through VI were captured as previously described (Catlin et al. 2014; Campion et al. 2010b), because x-ray-sensitive spermatogonia are within in these stages of spermatogenesis (Yamasaki, Sandrof, and Boekelheide 2010). The LCM samples were processed for RNA as previously described (Catlin et al. 2014), with the following exception: the number of LCM replicates for each sample was reduced to three (from seven), since a PCR-based amplification step was used to decrease the amount of starting material necessary.
The toxicant-induced gene response of the LCM-derived sensitive cell population was examined through the use of the RT2 Profiler PCR Array System (SABiosciences, Valencia, CA) for apoptosis pathway–related genes identified in the rat. Due to the time intensity of LCM and the number of toxicant exposure groups, a preamplification step was utilized to amplify the transcripts to be measured with the qRT-PCR array. The LCM-derived samples were amplified using the RT2 PreAmp complementary DNA (cDNA) Synthesis Primer Mix (Qiagen) with 14 ng of input RNA according to the manufacturer’s protocol (2012 version) to perform a PCR based preamplification of the genes that are present on the qRT-PCR array. The input RNA amount (14 ng) was selected based on the findings of our companion article, where 14 ng was the minimum amount of RNA that produced reproducible results and could be reasonably obtained through LCM (Catlin et al. 2014). Each sample was run in 10 μl reactions, which were loaded onto the qRT-PCR array (4 × 96-well format) using the epMotion 5075 robot automated pipetter (Eppendorf, Hamburg, Germany). An ABI-7900HT PCR machine (Applied Biosystems, Grand Island, NY) was used to run the arrays with the following cycling program: 95°C for 10 min, 40 cycles of 95°C for 15 sec and 60°C for 1 min, followed by a dissociation step.
Raw Ct values were normalized to the geometric mean of five housekeeping genes (Actb, B2m, Hprt1, Ldha, and Rplp1) to generate relative mRNA levels of each target gene, and the ▵▵Ct method (Schmittgen and Livak 2008) was used to analyze expression for each transcript. A one-way analysis of variance (ANOVA) was performed to assess the significance of the transcripts and corrected for multiple comparisons using the Tukey’s test (Prism 5; Graphpad Software, Inc., La Jolla, CA). The fold changes determined by qRT-PCR analysis for the seven significantly altered transcripts were uploaded into Multi-experiment Viewer (MeV version 10.2) and hierarchical clustering was performed on the transcripts using average linkage clustering for Euclidean Distance.
Transillumination Seminiferous Tubule Dissection
To examine protein levels at the same stages that were isolated through LCM, stage-specific sections of the seminiferous tubules were dissected from rat testes using transillumination methods adapted from those detailed previously (Chapin et al. 2001). Following removal from the animal, the collagenase solution immersed testis was immediately placed horizontally in a 35°C incubator (Thermo Hybaid, Franklin, MA) and shaken vigorously for 8 min. After digestion, the collagenase solution was decanted and the seminiferous tubules were washed three times with ice cold PBS. The loosely associated seminiferous tubules were then poured into a 100 cm3 glass petri dish and kept on ice during tubule dissection. Using forceps, the seminiferous tubules were transferred onto a petri dish on a Nikon SMZ-U Stereoscopic Microscope (Nikon, Melville, NY) and staged according to the methods and images previously described (Chen et al. 2003; Danner, Kirchhoff, and Ivell 2009). Stages I through VI were isolated for protein extraction to represent the same stages that were laser captured for the qRT-PCR experiments. The staged sections of seminiferous tubules were cut away using microscissors and placed in a 1.5-ml tube with 1 ml of PBS on ice, until allstage I through VI tubule segments had been isolated. Following the completion of tubule segment isolation, the seminiferous tubules were spun down at maximum speed (14,000 rpm) for 1 min and the PBS was removed. The tubule segments were then flash frozen and stored at −80°C until protein extraction.
Total Protein Preparation and Western Blot Analysis
Total protein lysates were prepared through homogenization of both the transillumination dissected seminiferous tubules and whole testis tissue in radioimmunoprecipitation assay (RIPA) buffer (150 mM NaCl, 1% NP40, 0.5% sodium desoxycholate, 0.1% sodium dodecyl sulfate (SDS), 50 mM Tris-Hcl pH 8.0). Lysates were homogenized on ice every 15 min for 1 hr and then cleared of cells and tissue debris through centrifugation at maximum speed (14,000 rpm) for 20 min. Protein concentrations were measured using the DCTM protein assay (Bio-Rad, Hercules, CA), with bovine serum albumin (BSA) as a standard.
For the Western blots, 15 μg of protein was denatured by boiling for 10 min in Laemmli SDS sample buffer and separated on a 12% SDS PAGE gel. Proteins were transferred onto a polyvinylidene fluoride (PVDF) membrane (Thermo Scientific, Waltham, MA) and blocked for 1 hr with 5% milk in tris-buffered saline (TBSt; 50 mM Tris pH 8.0, 150 mM NaCl, 0.1% Tween 20). Total cellular proteins were detected using primary antibodies against DR5 (1:250; Abcam, Cambridge, MA) and Caspase-7 (Casp7; 1:1,000; Cell Signaling, Danvers, MA) coupled with horseradish peroxidase-conjugated secondary antibody (1:750 and 1:2,000, respectively). The emitter-coupled logic (ECL) chemiluminescent substrate (Pierce, Rockford, IL) was used as the detection reagent and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) as the internal control for gel loading. HeLa cell lysate (StressGen Biotechnologies Corp., Victoria, British Columbia) was used as a positive control for both DR5 and Casp7; MCF7 cells treated for 24 hr with cisplatin were used as a positive control for cleaved Casp7. Total protein from rat brain was prepared as a negative control for DR5.
Protein levels for DR5, Pro-Casp7, and cleaved Casp7 were quantified by scanning the films. The band intensities were analyzed using the ImageJ software and the methods detailed at http://www.lukemiller.org/journal/2007/08/quantifying-western-blots-without.html (Accessed August 22, 2013). The percentage of cleaved Casp7 was calculated by dividing the GAPDH normalized values for cleaved Casp7 by the normalized values for total Casp7.
Statistical Analysis
For Western blots, the data were graphed in Prism 5 (Graphpad Software, Inc., La Jolla, CA) and are shown as the individual means ± standard error of the mean (SEM). Statistical differences (p ≤ .05) were calculated with Prism 5 software and using a Student’s unpaired two-tailed t-test. Pearson correlations between different exposures were performed using the cor function in the R statistical environment.
The incremental effect of HD co-exposure to each x-ray exposure used both paired t-tests and paired Wilcoxon rank sum tests were calculated in R (functions t.test and wilcox.test).
Results
To first examine the effect that low doses have on overall gene expression of the toxicant sensitive cell population, a qRT-PCR array with genes representing the apoptosis pathway was used. Analysis of the qRT-PCR data found that 7 of the 84 apoptosis-related transcripts were significantly altered at one or more of the doses, with fold changes ranging from −6.37 to 3.05 (Figure 2 and Supplemental Table 1). The altered transcripts encompassed both pro- and antiapoptotic genes and included Fas, Casp7, Birc3, DR5, Naip2, Sphk2, and Aven. The greatest transcript response was seen following exposure to the lowest dose of 0.5 Gy, where all of the seven transcripts were significantly altered. The number of altered transcripts decreased as the x-ray dose increased (n = 3 and 1; for 1 Gy and 2 Gy x-ray, respectively). The influence of HD on the x-ray-induced alterations was examined next. The addition of HD to the x-ray exposure resulted in significantly enhanced transcript alterations at 1 Gy and 2 Gy x-ray plus HD (n = 6 and 3, respectively). No significant response was seen with HD exposure alone. Fas was the only transcript that was significantly upregulated across all of the exposures (with the exception of the 0.33% HD exposure), while the rest of the significantly altered transcripts were downregulated. Hierarchical clustering grouped the transcripts according to direction of fold change, with no further apparent separation of transcripts into distinct groups.
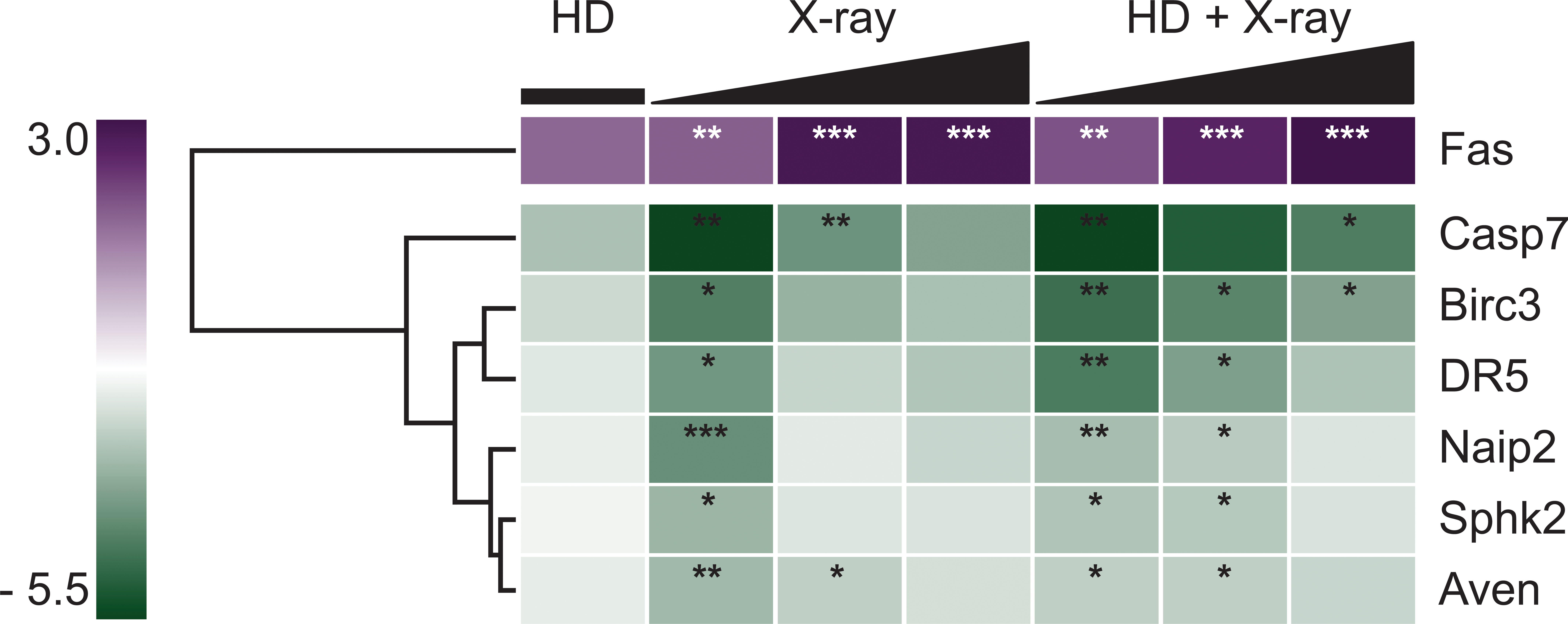
Heatmap displaying hierarchical clustering of qRT-PCR data. Analysis of qRT-PCR data identified seven significantly altered transcripts, altered at least with one exposure. Expression of the seven transcripts is expressed as fold change (relative to control). Hierarchical clustering separated the transcripts into two groups, those that were upregulated (n = 1; purple) and those that were down regulated (n = 6; green). The intensity of color for each transcript reflects the level of transcript expression, as depicted by the bar to the left of the dendrogram. Significance of a transcript following an exposure is indicated, *p ≤ .05. **p ≤ .01. ***p ≤ .001.
In the examination of the targeted germ cell population dose response to x-ray, the greatest difference in overall effect was elicited by exposure to the lowest x-ray dose, 0.5 Gy. To show the difference in response to 0.5 Gy compared to 1 or 2 Gy, the log fold changes for 0.5 Gy were plotted against the log fold changes for both 1 and 2 Gy x-ray (Figure 3). The blue “x” signs indicate the comparison of 0.5 Gy to 1 Gy x-ray, while the red “+” signs indicate the comparison of 0.5 Gy to 2 Gy. The shift in response of 1 or 2 Gy away from the dashed line shows that fewer genes were responding to these exposures than to the 0.5-Gy exposure. The data are similar even after the addition of HD to x-ray exposure (data not shown) and illustrates the limited influence HD has on x-ray-induced alterations at low-dose exposures.
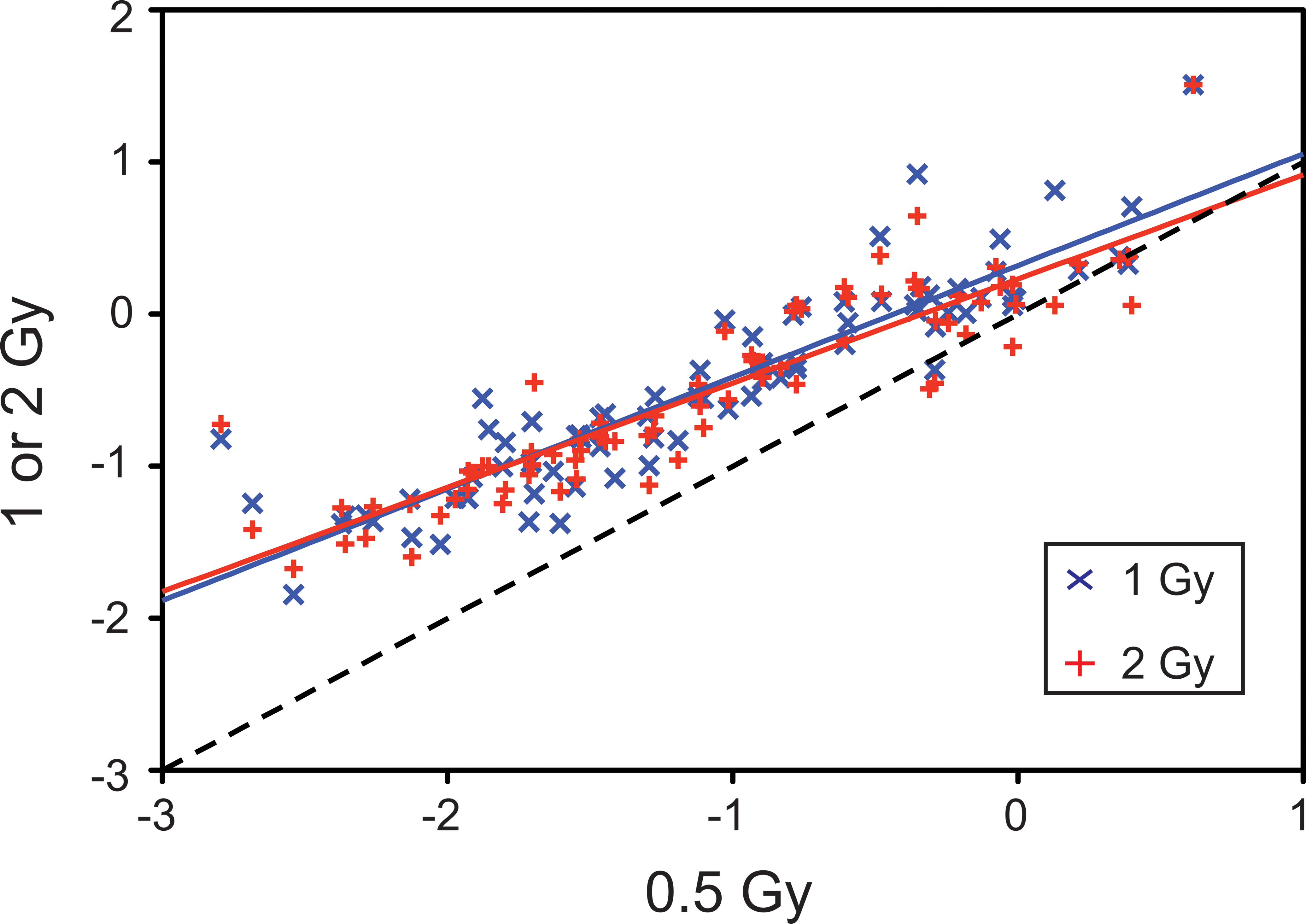
Dose response comparison of 0.5 Gy x-ray to both 1 and 2 Gy x-ray. The log fold changes for 0.5 Gy were calculated and plotted against the log fold changes for 1 Gy or 2 Gy, represented by a blue “×” or red “+”, respectively. The dotted line represents the correlation where the slope = 1.
The transcriptional analysis at 3 hr was complemented by an investigation of DR5 and Casp7 protein levels at 12 hr. The 2-Gy dose of x-ray was chosen for protein level examination, due to previous studies that showed high levels of apoptosis following 2 Gy exposure (Hasegawa et al. 1997; Yamasaki, Sandrof, and Boekelheide 2010). Both DR5 and Casp7 are important members within the apoptotic pathway that were significantly altered at the transcript level. For protein detection by Western blotting, both whole testis tissue and stage I through VI seminiferous tubules were probed. The seminiferous tubules were staged through microdissection using a transillumination technique. In stage I through VI seminiferous tubules, protein levels of DR5 increased nonsignificantly with exposure to either x-ray or HD alone (Figure 4). However, exposure to the combination of HD and x-ray resulted in a significant increase in DR5 protein at 12 hr following exposure. There was no statistically significant difference in the percentage of cleaved Casp7 following any of the exposures, although there was a slight increase with the x-ray alone and x-ray plus HD exposures (Figure 5). When examined in whole testis tissues, there were no significant alterations in either DR5 or the percentage of cleaved Casp7.
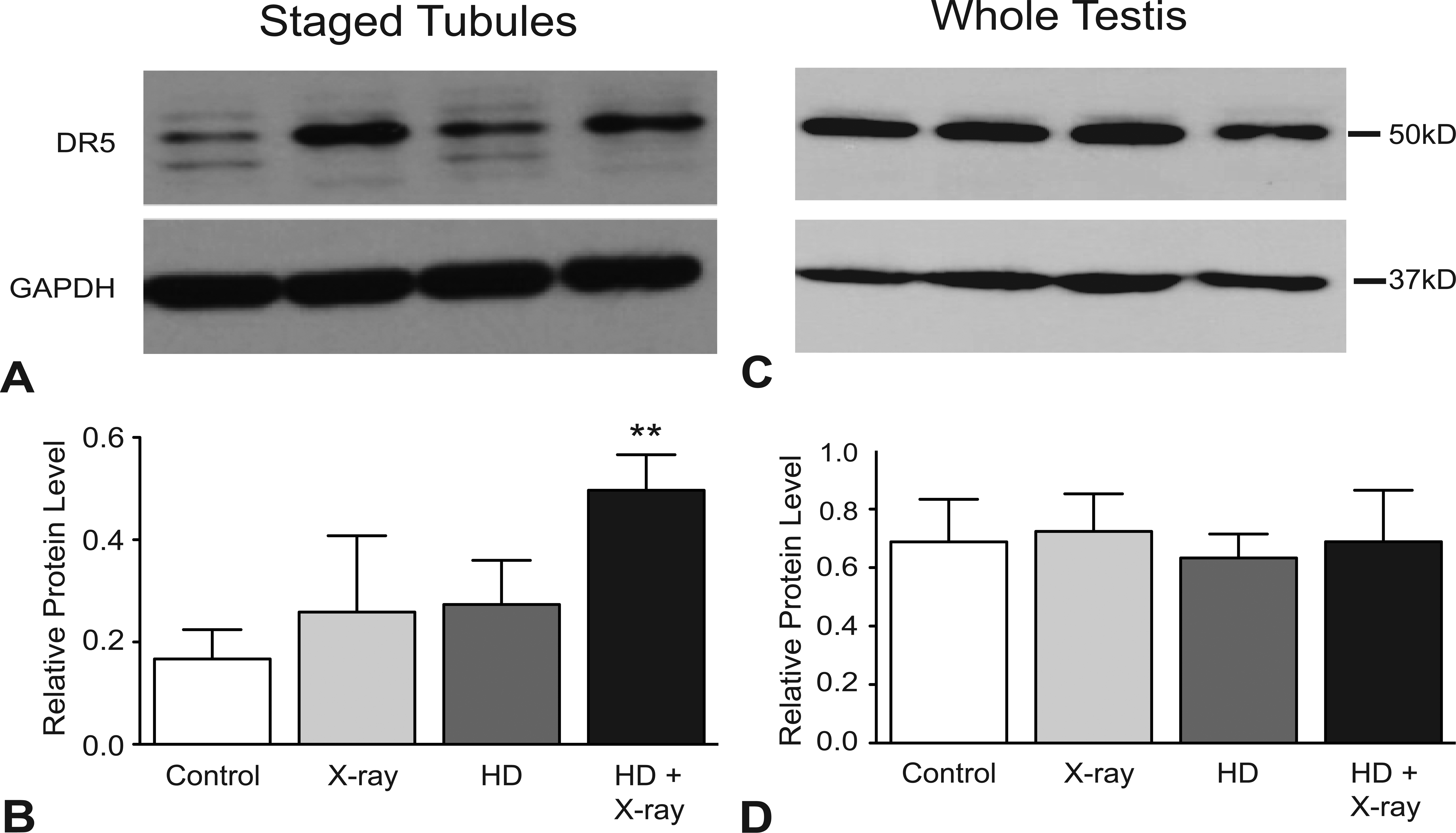
Western blot analysis of DR5 in staged seminiferous tubules and whole testis. Total cellular proteins from stages I–VI seminiferous tubules (A–B) or whole testis (C–D) were detected using primary antibody against DR5 (1:250). Representative Western blot images are shown for each exposure group (control, 2 Gy x-ray, 0.33%, and the co-exposure of 2 Gy x-ray with 0.33% HD; A and C). When quantified, DR5 expression in the staged seminiferous tubules was slightly increased with exposure to either x-ray or HD and was significantly enhanced following co-exposure to HD and x-ray (B). These differences in DR5 expression were not seen with whole testis protein (D). Significant differences between groups was achieved when p ≤ .05.
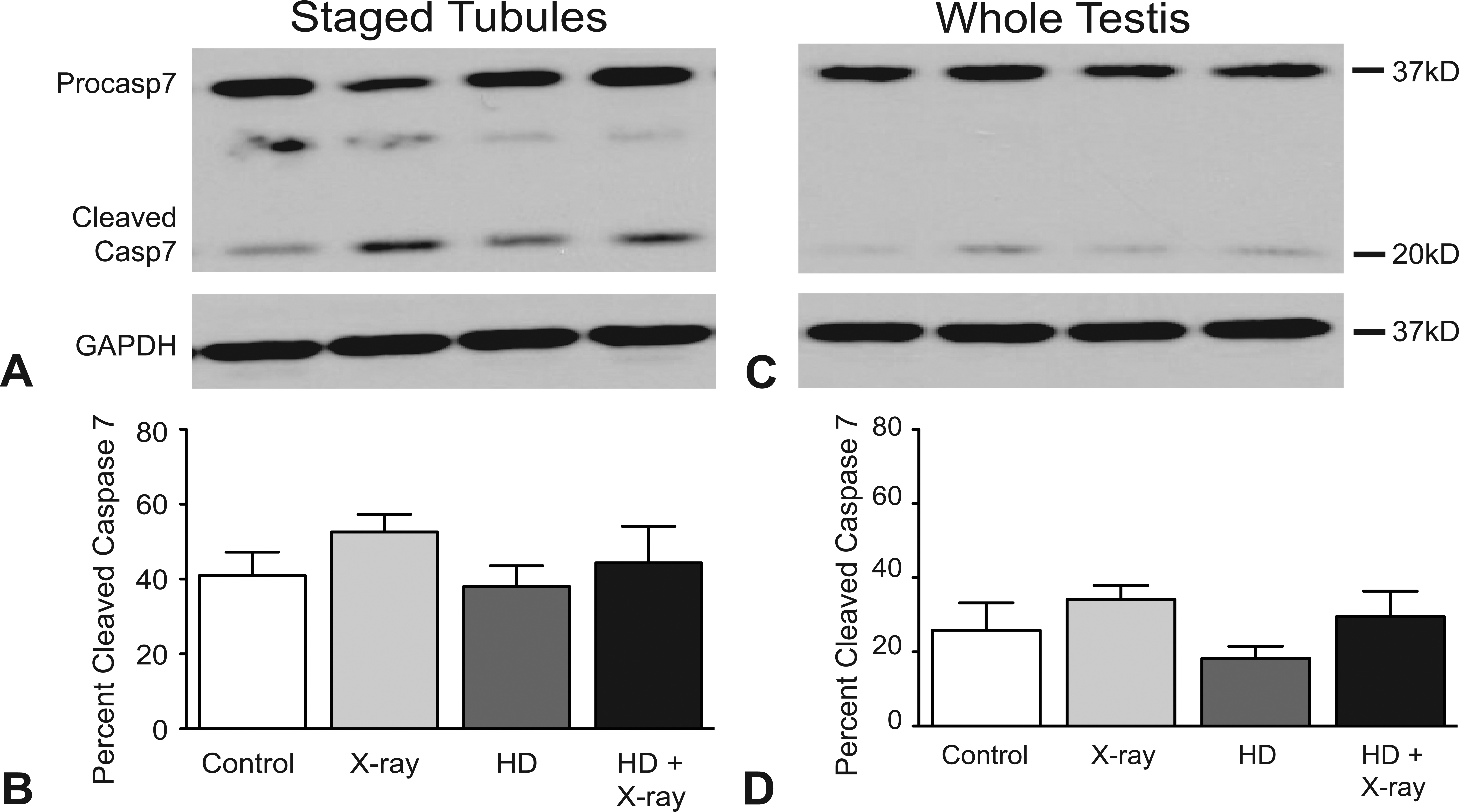
Western blot analysis of Casp7 in staged seminiferous tubules and whole testis. Total cellular proteins from stages I–VI seminiferous tubules (A–B) or whole testis (C–D) were detected using primary antibody against Casp7 (1:1,000). Representative Western blot images are shown for each exposure group (control, 2 Gy x-ray, 0.33% and the co-exposure of 2 Gy x-ray with 0.33% HD; A and C) The antibody detected both Pro-Casp7 and cleaved Casp7, and the percent of cleaved Casp7 was calculated by the division of cleaved Casp7 by total Casp7. When quantified, Casp7 expression exhibited no significant differences between exposures (B and D).
Discussion
The testis is comprised of several interacting cell types and germ cells at varying stages of development. This complexity proves to be a major hurdle to the study of testicular toxicant mechanisms. Due to the difficulty of separating individual cells or stages, testis gene expression studies are often performed with RNA isolated from whole testis. In this study, several different approaches were used to examine the spermatogenic stages most susceptible to two testicular toxicants, HD and x-ray. Optimized LCM methods were combined with qRT-PCR array approaches to provide mechanistic insight into individual exposures to low doses, and their combination, on target cell populations.
Here we report the application of our improved and validated LCM method, which was optimized to increase RNA yield for the study of a larger number of transcripts on a qRT-PCR array-based platform (Catlin et al. 2014). The apoptotic pathway is useful for toxicity evaluation, because a dose-dependent increase generally indicates a more “toxic” effect. Therefore, with alterations in key aspects of this pathway, conclusions can be drawn on the overall harm that is induced by toxicant exposure. The pathway approach allows for the study of low-dose mixtures, which typically do not induce large-scale changes but rather low-level changes within a pathway. Of the 84 transcripts represented on the qRT-PCR array, only 7 were significantly altered following correction for multiple comparisons (Figure 2). Of the 7 altered transcripts, there were both pro- and anti apoptotic genes represented, illustrating that for cells to undergo apoptosis, there must be simultaneous activation of proapoptotic genes with silencing of antiapoptotic genes. Surprisingly, in this study, the greatest transcript alterations were found with the lowest dose of x-ray (0.5 Gy), predominantly manifesting as downregulation of transcripts. The downregulation of both pro- and antiapoptotic genes with the low-dose of x-ray may be a survival attempt of the spermatogonial cell population to avoid the apoptosis signal triggered by downregulation of antiapoptotic genes. This effect was moderated as the x-ray dose increased, suggesting that the spermatogonial population was no longer evading the apoptosis signal.
The apoptotic pathway is highly dynamic and requires the consecutive, and sometimes simultaneous, turning on and off of pro- and antiapoptotic factors to induce apoptosis. This simultaneous upregulation and downregulation of apoptotic factors was seen with 0.5 Gy x-ray exposure, where there was simultaneous upregulation of the death receptor Fas and downregulation of both DR5 and Casp7. The smaller upregulation of Fas, in comparison to the higher doses of x-ray, combined with a strong downregulation of DR5 is quite interesting. Fas and DR5 have been shown to work in concert, interacting with the same downstream targets, and in some cases DR5 can serve as a surrogate mechanism of apoptosis activation when Fas signaling is dysfunctional (Richburg et al. 2002). At the time point measured, 3 hr, an adaptive response to 0.5 Gy exposure might be occurring to avoid apoptosis and maintain homeostasis, in part by overriding the downregulation of antiapoptotic factors that would otherwise lead to activation of apoptosis. At the higher doses, there was less overall downregulation of pro- and antiapoptotic factors and an increased expression of Fas, ultimately tipping the scales toward higher levels of apoptosis. Previous studies have found similar differences in the rate of apoptosis gene expression of several transcripts within staged seminiferous tubules. Bcl2 and p53 levels remained unaltered in stage I through VI seminiferous tubules at 12 hr with qRT-PCR, while significant increases were seen in levels of Fas and Casp3 (Campion et al. 2010b). In the same study, apoptosis enhancing nuclease (Aen) transcript expression was found to be low at 3 hr and high at 12 hr, with p53 up-regulated modulator of apoptosis (Puma) expression having the exact opposite expression profile. Fas expression peaks between 12 and 24 hr after a much lower dose of 0.075 Gy (Liu et al. 2007), a much later peak than that seen with doses of 2 and 5 Gy, where peak expression in Fas was seen between 6 to 12 hr (Embree-Ku, Venturini, and Boekelheide 2002).
Dose may be influencing the timing of apoptosis initiation, as illustrated by our study of multiple doses at a fixed time. Transcripts were altered in a much different manner with the 0.5 Gy x-ray exposure, compared to either 1 or 2 Gy exposure (Figure 3). This was shown in the dose response comparison of 0.5 Gy x-ray to 1 and 2 Gy x-ray, where there was a distinct shift in overall transcript expression. This could be a reflection of the adaptive effect occurring with the lower dose, with downregulation of proapoptotic genes to compensate for downregulation of the antiapoptotic genes. At later timepoints, the downregulation of proapoptotic transcripts could be overwhelmed by the cell damage response, as it has been shown previously with TUNEL staining that apoptosis eventually occurs with 0.5 Gy x-ray between 12 and 24 hr in stage I through VI seminiferous tubules (Hasegawa et al. 1997; Hasegawa et al. 1998). To contrast this with higher doses, peak TUNEL staining with 2 and 5 Gy x-ray was seen earlier between 6 and 12 hr following exposure in the same stages of seminiferous tubules (Yamasaki, Sandrof, and Boekelheide 2010).
The timing of the apoptosis pathway response is important as it involves the subsequent activation of many genes in a cascade. A transillumination seminiferous tubule dissection technique allowed for the separation of stages as those that were captured through LCM for examination of alterations in protein levels at 12 hr. Exposure to radiation has been shown to cause significant increases in rat testicular apoptosis at 12 hr following exposure as a result of upregulation of the Fas system (Embree-Ku, Venturini, and Boekelheide 2002; Lee et al. 1999). DR5 and Casp7 were studied further at this time point to see if the transcript level alterations were translated into protein alterations. At 12 hr following HD and x-ray co-exposure, DR5 protein was significantly upregulated in the staged seminiferous tubules (Figure 4), while no alterations were seen in DR5 protein levels when examined in whole testis. In both staged seminiferous tubules and whole testis, there were no significant differences in the amount of cleaved Casp7 (Figure 5). DR5 plays a role early on in the initiation of apoptosis, while Casp7, an executioner caspase, is activated later in the apoptosis pathway. This time difference in activation could be an alternate explanation to the significant increase in protein levels of DR5 seen with the co-exposure, while Casp7 was unchanged. Comparison of data from large-scale protein and messenger RNA (mRNA) profiling shows that protein concentrations generally do not correlate as well with their corresponding mRNAs, with a squared Pearson’s correlation of about .40 (Vogel and Marcotte 2012). The lack of correlation could be due to many underlying factors, including greater protein stability than that of mRNA, a faster protein than mRNA production rate, or simply noise from protein and mRNA measurement. However, overall these results illustrate the important role of timing in the regulation of the apoptotic pathway following toxicant exposure.
The combination of HD and x-ray exposures have some influence on gene expression at 3 hr following exposure. This was illustrated by an increase in the number of significantly altered transcripts when HD was added to x-ray exposure, suggesting an accentuated effect when HD and x-ray are combined (Figure 2). This was not expected as previous studies with higher doses of HD and x-ray have illustrated an attenuation effect with co-exposure (Campion et al. 2010b). Contrary to the DR5 3-hr gene expression data showing a non-significant downregulation in transcript levels with 2 Gy x-ray co-exposure with HD, DR5 protein was significantly upregulated at 12 hr following the same co-exposure in the staged seminiferous tubules (Figure 4). These results with HD and x-ray co-exposure indicate that the attenuation effect seen with the combined high dose of 5 Gy x-ray and 0.33% HD did not persist into the low-dose range.
The results of the current study have been summarized into a working model that illustrates the overall effect of x-ray exposure on the apoptosis pathway gene regulation and outcome (Figure 6). With 0.5 Gy x-ray exposure, death receptors DR5 and Fas have an overall decreased expression, that when combined with a significant decrease in Casp7 expression, overrides the decreased expression of antiapoptotic factors. This is a potential mechanism underlying the delayed execution of cellular apoptosis in low-dose exposed testes, as demonstrated by other studies with TUNEL staining. As the dose is increased to up to 1 or 2 Gy x-ray, the proapoptotic signal grows stronger accompanied by a decrease in antiapoptotic factors, resulting in a more rapid onset of apoptosis. It is apparent that the response to 0.5 Gy x-ray is different than that to either 1 Gy or 2 Gy x-ray, indicating an adaptive response. In addition, we employed a transillumination-assisted dissection technique to examine the protein levels of stage I through VI seminiferous tubules. There was a significant up-regulation in DR5 protein following 0.33% HD and 2 Gy x-ray combined exposure, while there was no change in Casp7 protein levels.
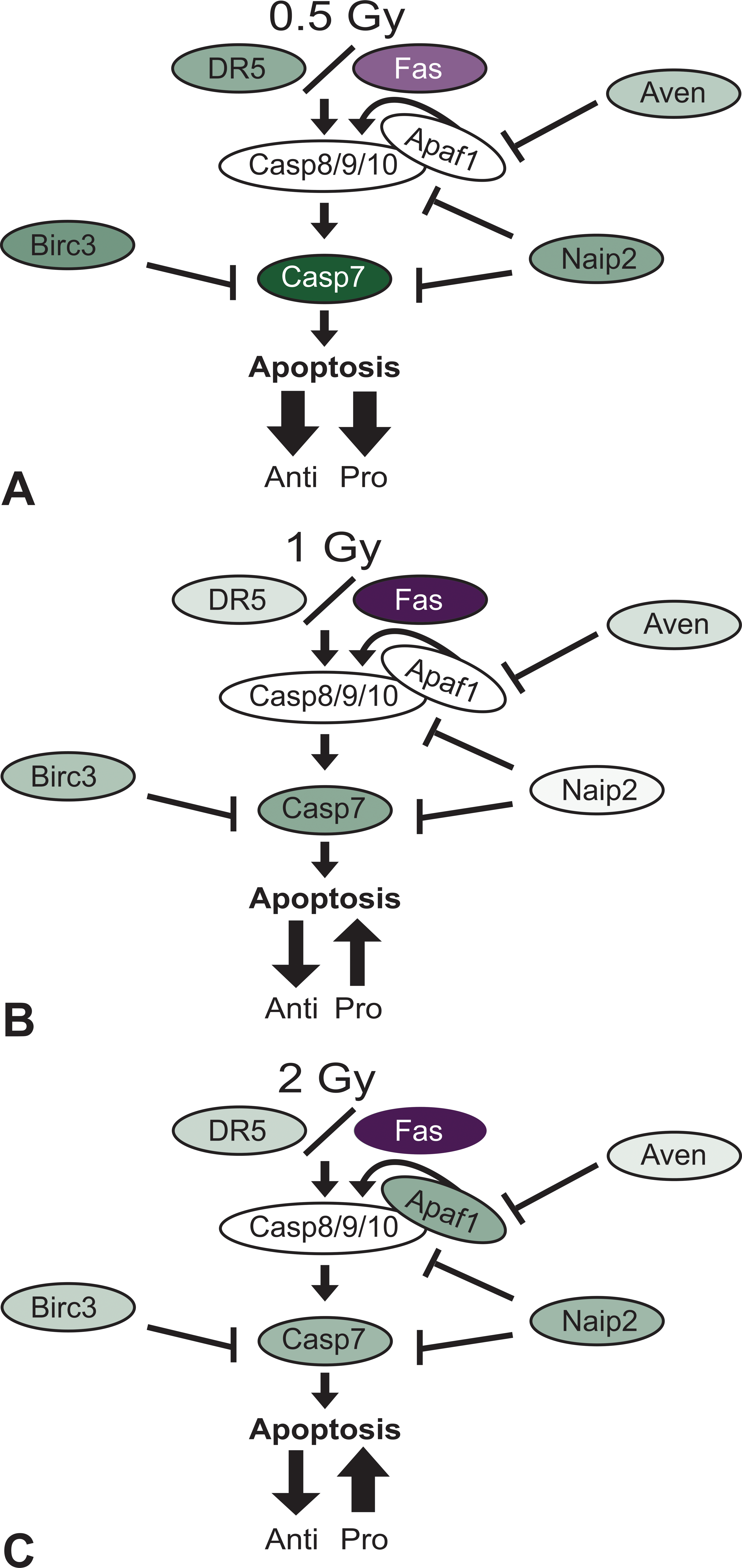
Proposed mechanism of testicular apoptotic response to x-ray exposure. The regulation of key apoptotic factors in the testis are shown following exposure to (A) 0.5 Gy x-ray, (B) 1 Gy x-ray, or (C) 2 Gy x-ray. Following 0.5 Gy exposure (A) the spermatogonial cells attempt adaptation to cell damage by simultaneously down regulating pro-and antiapoptotic factors, resulting in a delay in apoptosis activation. With 1 Gy (B) and 2 Gy (C) x-ray exposure, the proapoptotic signal is stronger, resulting in a more rapid onset of apoptosis.
This is the first study to examine a representative portion of the apoptosis pathway with either laser capture microdissected cells or staged seminiferous tubules, through the application of an improved LCM method developed in the companion study. These results provide insight into the complex mechanisms of low-dose co-exposures and the necessity of examining stage specificity of effect. More studies are needed to investigate a broader range of time points, taking advantage of cell- and stage-enrichment techniques to allow for greater insight into the mechanisms underlying apoptosis.
Footnotes
Supplemental Material
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Environmental Health Sciences at the National Institutes of Health (grant numbers P42 ES013660 and 5 T32 ES07272-17). Kim Boekelheide has funding from NIEHS, USEPA, and the American Chemistry Council. He is an occasional expert consultant for chemical and pharmaceutical companies, and owns stock in CytoSolv, an early stage biotechnology company developing a wound healing therapeutic.
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
