Abstract
Vascular endothelial growth factor (VEGF) receptor tyrosine kinase (RTK) inhibitors are reported to cause reversible mucosal hyperplasia (adenosis) in the duodenum of rats; however, the pathogenesis is not fully elucidated. Using lenvatinib, a VEGF RTK inhibitor, we characterized the histologic time course of this duodenal change in rats. At 4 weeks, there was degeneration and necrosis of Brunner’s gland epithelium accompanied by neutrophil infiltration around the affected glands. At 13 weeks, the inflammation was more extensive, and Brunner’s gland epithelium was attenuated and flattened and was accompanied by reactive hyperplasia of duodenal epithelium. At 26 weeks, the changes became more severe and chronic and characterized by marked cystic dilation, which extended to the external muscular layer. These dilated glands exhibited morphological characteristics of duodenal crypt epithelium, suggestive of replacement of disappeared Brunner’s glands by regenerative duodenal crypt epithelial cells. Similar changes were not present in similar time course studies in dog and monkey studies, suggesting that this is a rodent- or species-specific change. Based on the temporal progression of Brunner’s gland lesion, we identify degeneration and necrosis of the Brunner’s glands as the primary change leading to inflammation, cystic dilatation, and regeneration with cells that are morphologically suggestive of duodenal crypt epithelium.
Keywords
Introduction
Many anticancer therapies are now directed against tumor-related angiogenesis. Strategies in development include small-molecule receptor tyrosine kinase (RTK) inhibitors as well as monoclonal antibodies directed against vascular endothelial growth factor (VEGF) or VEGF receptors (Ellis and Hicklin 2008; Tugues et al. 2011). Lenvatinib methanesulfonate (lenvatinib: formerly known as E7080) is a potent inhibitor of VEGF RTK that targets the split-kinase family of RTKs including fms-like tyrosine kinase 1 (FLT1) and kinase insert domain receptor (KDR). Lenvatinib also inhibits fibroblast growth factor receptor 1 and platelet-derived growth factor receptor β RTK.
Toxicity with multitargeted RTK signaling inhibitors in various organ systems have been reported in nonclinical species, and includes reversible thickening of the growth plate (physeal dysplasia), impaired formation of corpora lutea, incisor dental caries, bone marrow hypocellularity, and adrenocortical hemorrhagic necrosis (Ryan et al. 1999; Gerber et al. 1999; Patyna et al. 2008). In the small intestine, glandular hyperplasia, sometimes associated with inflammation in the intestinal wall, has been reported in nonclinical safety evaluation of a multitargeted RTK inhibitor, sunitinib (Patyna et al. 2008). Recently, duodenal epithelial hyperplasia and “infiltration” of mucosal glands into the lamina muscularis and partly into the peritoneum and gastric wall were reported in a 26-week rat toxicity study of a VEGF receptor inhibitor (Ettlin et al. 2010). The lesion was first diagnosed as malignant and clinical studies were put on hold. However, due in part to partial regression of the lesions after dosing termination, the lesion was ultimately assessed as adenosis with reversible hyperplasia and clinical trials were allowed to resume (Thomas et al. 2005; Ettlin et al. 2010).
We have experienced similar duodenal changes in rat nonclinical toxicity studies with lenvatinib that were not present in dog and monkey preclinical trials. In the present study, we examined duodenal tissue from a series of rat toxicity studies (4-, 13-, and 26-week repeated dose oral toxicity studies) to assess time course progression and pathogenesis of the duodenal lesion. Our longitudinal histologic examination supports the notion that the duodenal lesion at 26 weeks is reactive hyperplasia and has also identified degeneration, necrosis, and inflammation of the Brunner’s gland as the earliest primary inciting cause.
Materials and Methods
Animals and Husbandry
Eight-week-old male and female Sprague-Dawley rats were supplied by Charles River Laboratories Japan Inc. (Shiga, Japan)—the males weighed 238 to 285 g and the females weighed 164 to 197 g at the start of treatment for the 4-, 13-, and 26-week studies, respectively. They were housed in a clean air room maintained at 20° to 23°C and a relative humidity of 51 to 63% with a 12-hr light cycle, and provided approximately 21 g/day in males and approximately 14 g/day in females of CRF-1 commercial rodent chow (Oriental Yeast Co., Ltd., Tokyo, Japan). Sterilized tap water was provided to animals in all studies ad libitum.
The study protocols were approved by the Laboratory Animal Care and Use Committee and were conducted by Eisai Co., Ltd. in accordance with the principles of Good Laboratory Practice.
Drug
Lenvatinib was supplied by Eisai Co., Ltd. (Ibaraki, Japan) for all 3 studies. All dose levels are based on the free base equivalent mg/kg dose. Lenvatinib was formulated in an aqueous vehicle.
Study Design
Lenvatinib was administered orally by gavage once a day for 4-, 13-, and 26 weeks to male and female rats (10 animals/group/sex for the 4- and 13-week studies; 15 animals/group/sex for the 26-week study). On the basis of the results of a 7-day dose range toxicity study, dose levels were 0, 10, 30, and 100 mg/kg for the 4-week study, and 0, 0.4, 2, and 10 mg/kg for the 13- and 26-week studies.
The study designs are summarized in Table 1. The first day of administration was defined as day 1. In the 4-week study, 1 male and 1 female at 100 mg/kg died and were necropsied on day 22 and day 29, respectively. Three males and 1 female at 100 mg/kg became moribund and were euthanized and necropsied between days 24 to 27. One female at 30 mg/kg died of gavage error and was necropsied. There were no dead or moribund animals in the 13-week study. In the 26-week study, 2 males at 10 mg/kg died on day 84 and day 140, respectively. Six males were euthanized due to poor condition between days 94 to 147 and were necropsied. The remaining 7 males were euthanized and necropsied on day 150 (week 22). Three females at 10 mg/kg were euthanized due to poor condition between days 135 to 147 and were necropsied. All surviving animals were euthanized by exsanguination under isoflurane anesthesia following completion of respective dosing period and were necropsied.
Study design.
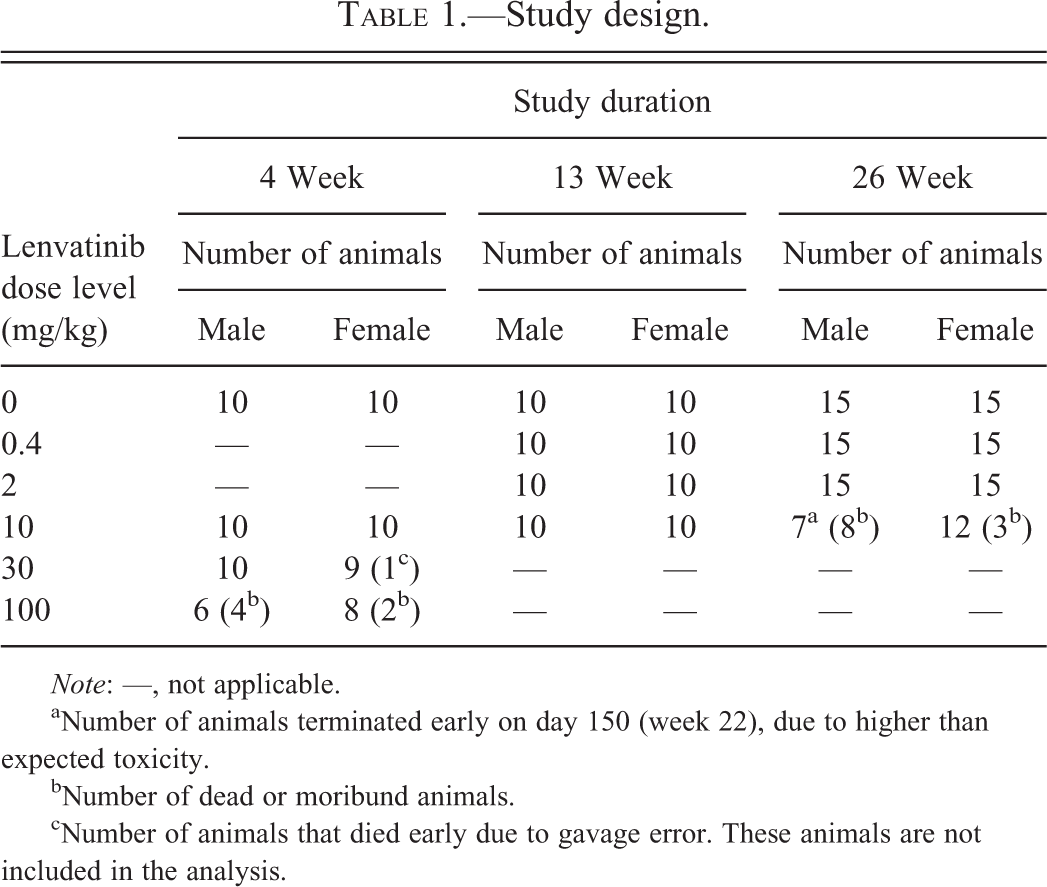
Note: —, not applicable.
aNumber of animals terminated early on day 150 (week 22), due to higher than expected toxicity.
bNumber of dead or moribund animals.
cNumber of animals that died early due to gavage error. These animals are not included in the analysis.
Microscopic Examination
The following tissue samples were collected from all animals in each group: liver, kidneys, brain, spinal cord, sciatic nerve, trachea, lung, heart, aorta, spleen, thymus, adrenals, thyroids, parathyroids, pituitary, testes, epididymides, ovaries, uterus, vagina, salivary glands, eyes, optic nerves, Harderian glands, tongue, submaxillary/mesenteric lymph nodes, pancreas, urinary bladder, prostate, seminal vesicles, coagulating glands, esophagus, stomach, small and large intestine, bone marrow, sternum, femur, thigh muscle, mammary gland, skin, incisors, molars, and gross lesions. In this article, we focus on the lesions in duodenum/Brunner’s gland. At necropsy, the stomach was opened along the greater curvature from esophagus to duodenum, leaving a section of opened distal esophagus and proximal duodenum attached to the stomach. These were fixed in neutral buffered 10% formalin and embedded in paraffin, and sections of 2 to 3 µm were prepared and stained with hematoxylin and eosin (H&E). The section from the pylorus to proximal duodenum (including Brunner’s glands) was used for examination. The results from the 4-, 13-, and 26-week studies are presented in this article. For further investigation, several sections with representative lesions from each study were additionally stained with Watanabe silver method for the histologic structure or periodic acid–Schiff reaction (PAS) for goblet cells in duodenal mucosa.
Results
Normal Brunner’s Glands in the Duodenal Mucosa of Adult Rat
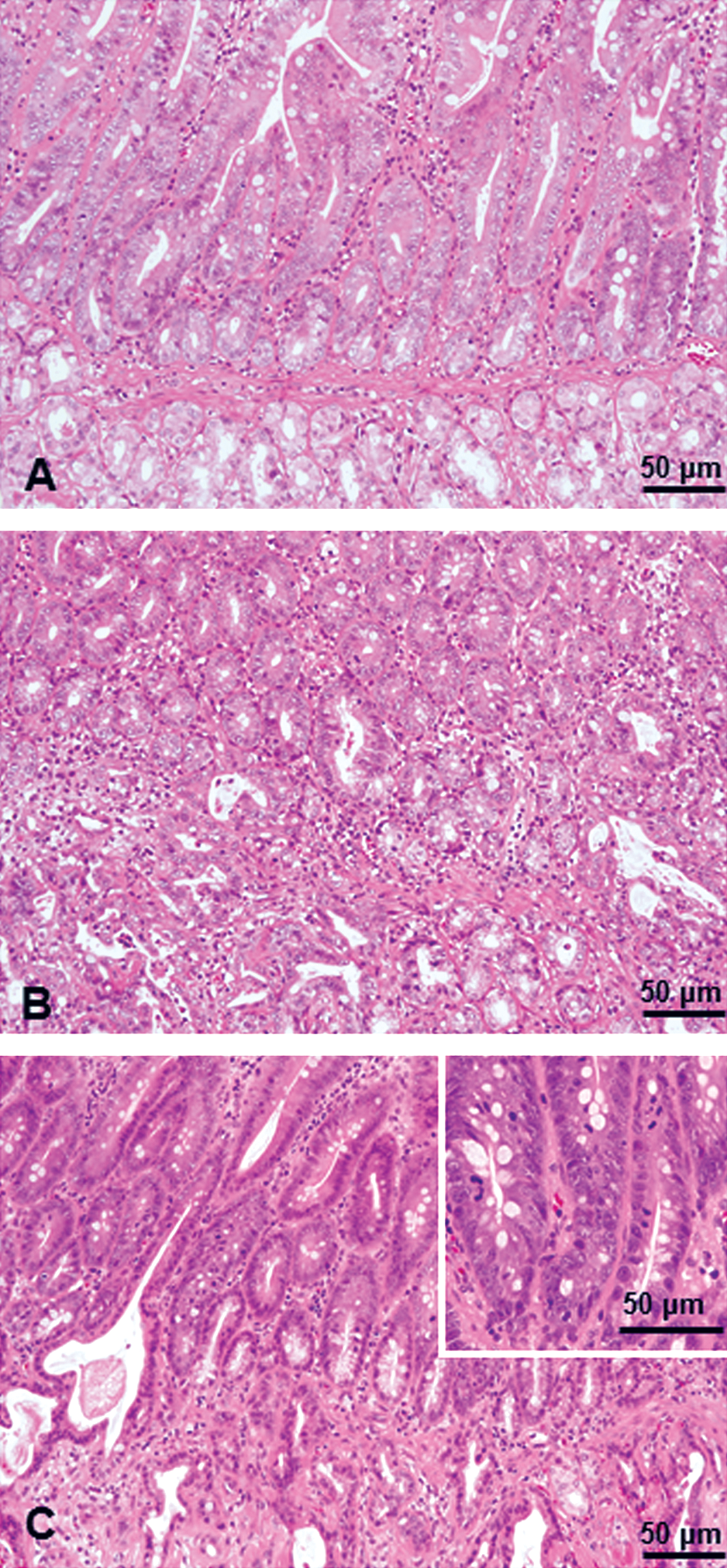
In the longitudinal tissue section from the gastric pylorus to proximal duodenum, Brunner’s glands are found in the submucosa and composed of acini that open into branched tubules (Figure 1A and C). The ducts from Brunner’s glands open into the bases of the intestinal glands immediately after passing through the muscularis mucosae (Figure 1B). No age-related histological differences were noted in the Brunner’s gland of rats used in the present study.
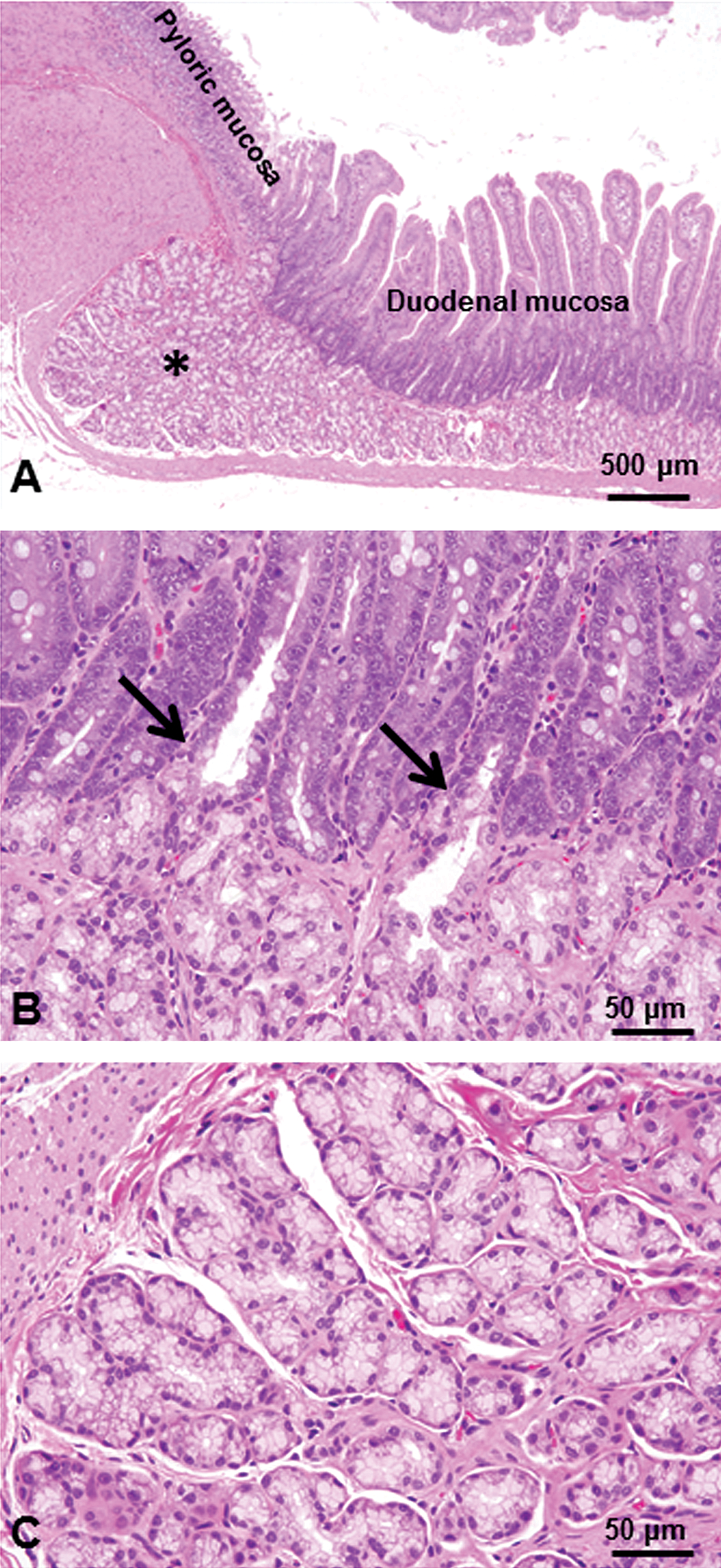
Brunner’s glands in the normal duodenal mucosa of adult rat. Brunner’s glands (*) are found in the submucosa and are composed of acini that open into branched tubules (A, C). The ducts from Brunner’s glands open into the bases of the intestinal glands immediately after passing through the muscularis mucosae (arrows, B). H&E staining.
Time Course of Brunner’s Gland Lesions
Histopathological findings in the Brunner’s gland and duodenum are summarized in Table 2.
Histopathological findings in the Brunner’s gland and duodenum.
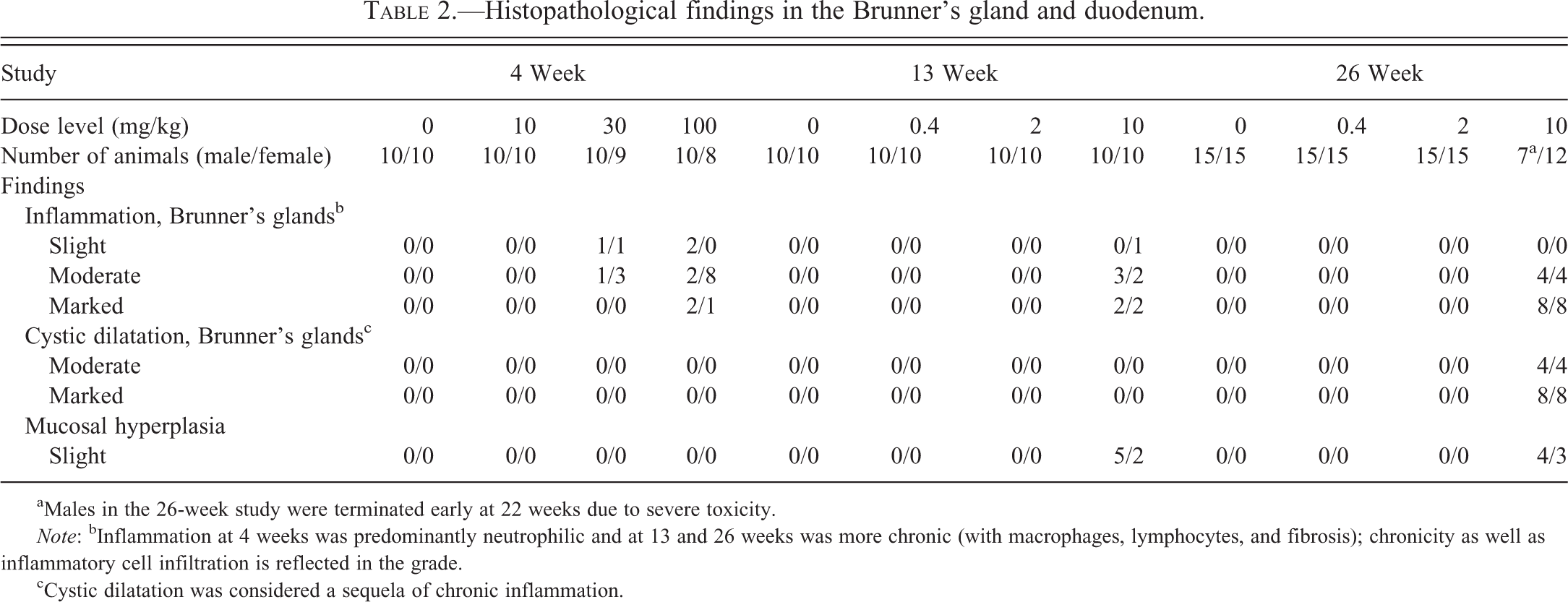
aMales in the 26-week study were terminated early at 22 weeks due to severe toxicity.
Note: bInflammation at 4 weeks was predominantly neutrophilic and at 13 and 26 weeks was more chronic (with macrophages, lymphocytes, and fibrosis); chronicity as well as inflammatory cell infiltration is reflected in the grade.
cCystic dilatation was considered a sequela of chronic inflammation.
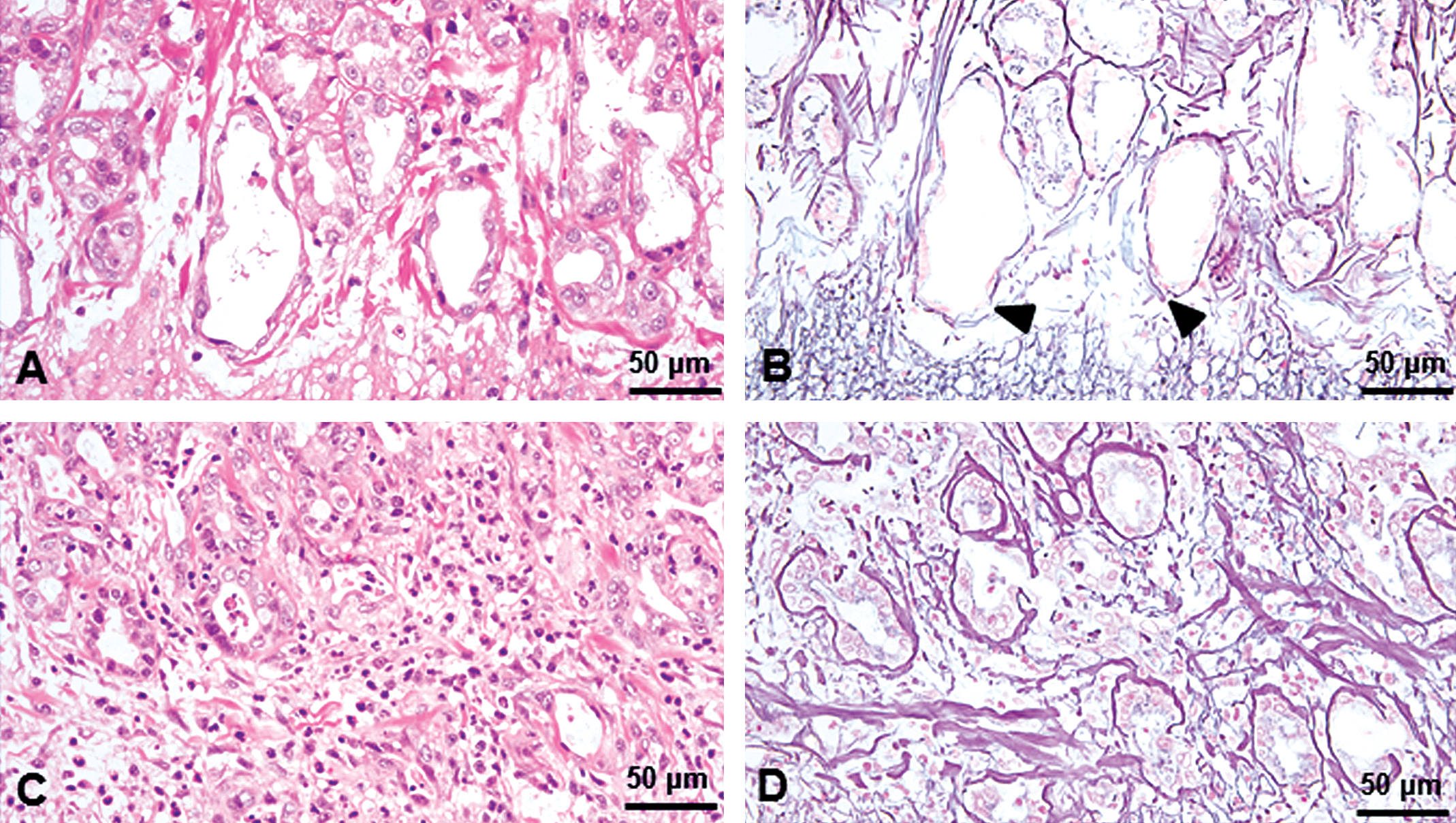
In the 4-week study, at 30 mg/kg, inflammation of Brunner’s gland characterized by degeneration/necrosis of glandular epithelial cells with slight dilation of Brunner’s glands accompanied by neutrophil infiltration of the interstitium surrounding the Brunner’s glands was observed (Figure 2A and B). In some areas, the observed epithelial degeneration/necrosis was accompanied by neutrophil infiltrates around the affected Brunner’s glands (Figure 3A) and disruption of the basement membrane as an early lesion in silver-stained sections (Figure 3B), suggesting that the Brunner’s gland epithelial degeneration/necrosis resulted in leakage of glandular secretions into the interstitium and induction of the inflammatory response. In the lesions with greater severity, the inflammation also extended into the surrounding mucosa, muscular layers, or both. Duodenal mucosa appeared within normal limits (Figure 4A).
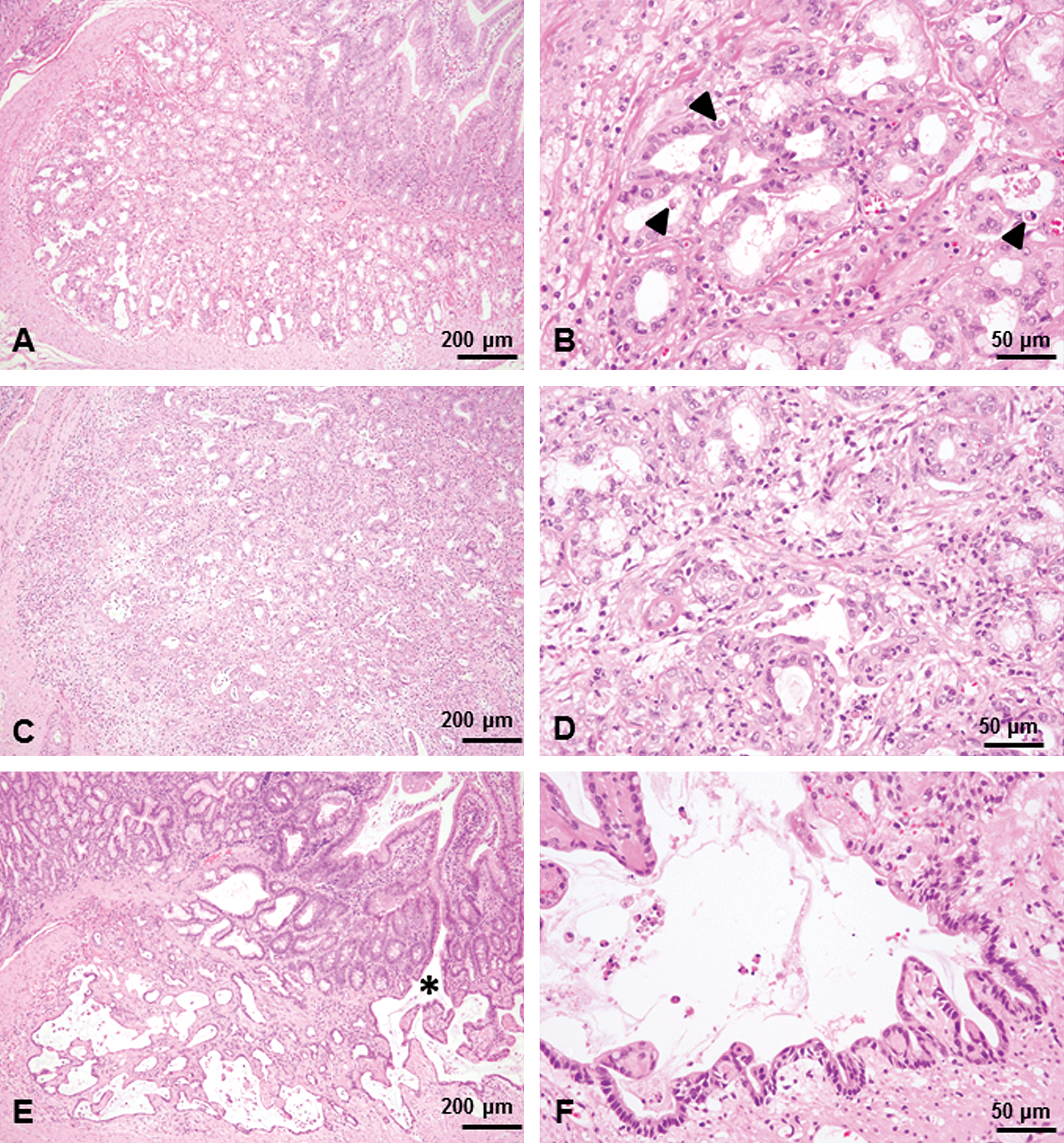
Brunner’s gland lesions under low magnification from a male from the group treated with 30 mg/kg for 4 weeks (A, B), the group treated with 10 mg/kg for 13 weeks (C, D), and the group treated with 10 mg/kg 26 weeks (E, F). Slight dilation of Brunner’s glands and cell debris in the glandular lumina, accompanied by inflammatory cell infiltration of the interstitium surrounding the Brunner’s glands were noted (A). Degeneration/necrosis of glandular epithelial cells (arrowheads) with dilation of Brunner’s glands were noted and were accompanied by neutrophil infiltration of interstitium surrounding the Brunner’s glands (B). Inflammation was marked and widespread, and inflammatory cells within the Brunner’s gland lumen and interstitial edema were also present (C). Inflammation and associated disruption of the glandular structure were extensive and severe and were accompanied by presence of degenerated exfoliated cells and inflammatory cells within the Brunner’s gland lumen (D). Brunner’s glands lined by flattened epithelial cells accompanied by inflammatory cell infiltration and interstitial fibrosis (E). Note that the dilated glands expanded into muscular layer (*) and were lined by duodenal mucosal epithelium. Dilated Brunner’s glands were lined with flattened and atrophic epithelial cells and formed large cystic cavities (F). H&E staining.
Histologic structures of the Brunner’s gland lesions from a male from the group treated with 30 mg/kg for 4 weeks (A and B) and the group treated with 10 mg/kg for 13 weeks (C and D). In the 4-week study, epithelial degeneration/necrosis was accompanied by neutrophil infiltrates around the affected Brunner’s glands (A) and disruption of the basement membrane (arrowheads) was observed as an early lesion (B). Disruption of the basement membrane and glandular structure were prominent in the 13-week study (C and D). A and C: H&E staining; B and D: Watanabe silver method.
Duodenal mucosa from the group treated with 30 mg/kg for 4 weeks (A), the group treated with 10 mg/kg for 13 weeks (B), and the group treated with 10 mg/kg 26 weeks (C). The duodenal mucosa appeared within normal limits in the 4-week study. In the 13-week study, the inflammation became prominent and extended to the lamina propria and submucosa. In the 26-week study, the crypts are markedly elongated and lined by basophilic and hyperplastic epithelium. This change is suggestive of reactive hyperplasia to sustained inflammation in Brunner’s glands. H&E staining.
In the 13-week study, at 10-mg/kg dose, the inflammation was more extensive and severe than that observed in the 4-week study and was accompanied by presence of inflammatory cells within the Brunner’s gland lumen and interstitial edema (Figure 2C and D). The epithelium lining the Brunner’s glands was attenuated and flattened, and disruption of the basement membrane and glandular structure were clearly demonstrated in silver-stained sections (Figure 3C and D). In the duodenal epithelium, prominent inflammatory cell infiltration extended to the lamina propria and submucosa (Figure 4B).
In the 26-week study, at 10 mg/kg, the changes were more severe than those at 13 weeks and characterized by marked dilation of Brunner’s glands and chronic inflammation (Figure 2E and F). In the most severe lesions, the dilated Brunner’s glands were lined with flattened and atrophic epithelial cells and formed large cystic cavities that extended into the muscularis externa as well as the mucosal layer. Interestingly, there was continuity between the dilated Brunner’s glands and the duodenal mucosal epithelium (Figure 5C). The Brunner’s glands were often lined by a single layer of cuboidal to columnar epithelial cells with a basally located nucleus that morphologically resembled duodenal crypt epithelial cells; in addition, there were scattered goblet cells that were PAS positive (Figure 5D). The inflammation was characterized by moderate to marked neutrophilic and lymphocytic infiltration around and within the dilated Brunner’s glands, extended into the adjacent mucosa, muscularis externa, and serosa, and was accompanied by fibrosis of Brunner’s gland interstitium and reactive hyperplasia of the duodenal epithelium (Figure 4C).
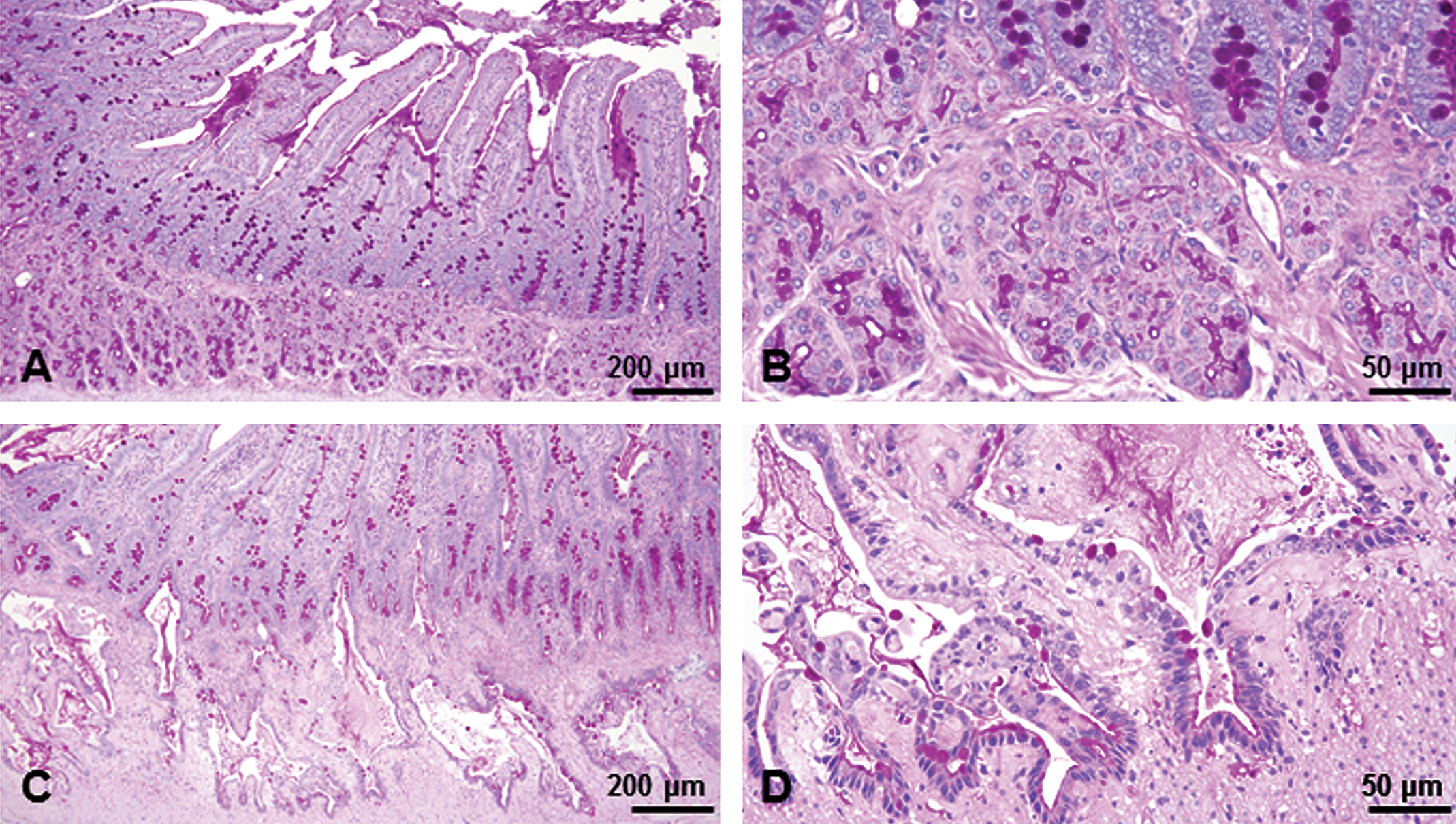
Brunner’s glands from a male from the control group (A, B) and the group treated with 10 mg/kg for 26 weeks (C, D). In the control group, the scattered goblet cells in the duodenal mucosal epithelium and secretion in Brunner’s gland were PAS positive (A, B). Dilated Brunner’s glands were lined with flattened epithelial cells and formed large cystic cavities that extended into the muscularis externa as well as mucosal layer (C). The Brunner’s glands were often lined by a single layer of cuboidal to columnar epithelial cells with a basally located nucleus that morphologically resembled duodenal crypt epithelial cells; in addition, there were scattered goblet cells that were PAS positive (D). PAS reaction. PAS = periodic acid–Schiff reaction.
Discussion
We have presented time course changes of histologic findings of duodenum in rats given lenvatinib, a VEGF receptor inhibitor, for 4, 13, or 26 weeks. Our data clearly demonstrate for the first time that the Brunner’s gland is a target tissue of lenvatinib in rats.
Brunner’s glands are unique to eutherian mammals, which include rodents and primates, and are confined primarily to the submucosa of the proximal duodenum. The main function of Brunner’s glands is to protect the duodenal mucosa from the acidic gastric contents and abrasion of the intestinal mucosa by the passage of rough ingesta (Florey et al. 1939; Griffith and Harkins 1956; Lang and Tansy 1983; Krause 2000). The primary effect of lenvatinib on Brunner’s gland in rats is characterized microscopically by degeneration and necrosis of Brunner’s gland epithelial cells. Because inflammation was often present in tissues surrounding the affected Brunner’s glands, it is likely that leakage of secretions from damaged Brunner’s glands into the surrounding tissues elicited focal inflammation and subsequent reactive hyperplasia of duodenal crypt epithelium (Figure 4). With continuous and prolonged damage, the lesions progressed into chronic inflammation accompanied by dilated, cystic glands lined by flattened and atrophic epithelial cells.
The toxicity of lenvatinib has been also evaluated at 3 dose levels in standard repeated dose oral toxicity studies in dogs (up to 4 weeks) and monkeys (up to 39 weeks). Lenvatinib was orally administered once a day to male and female beagle dogs (3 or 5 animals/group/sex for the 4-week study) and cynomolgus monkeys (3 or 5 animals/group/sex for the 4-week study; 3 animals/group/sex for the 13-week study; 4 animals/group/sex for the 39-week study). Although the maximum tolerated dose was established and high exposures were achieved in all the studies, there were no drug-related lesions in the duodenum of dogs or monkeys on any study. It is noteworthy that this lesion was observed only in rat but not in nonrodent species, suggesting that this is a rat-specific finding. Possible reasons for this species difference include physiologic species differences in the postnatal development of the gastrointestinal tract (Ettlin et al. 2010), species differences in expression, localization, or function of vascular endothelial growth factor receptor (VEGFR) in the Brunner’s gland, and differences in biodistribution of parent drug/metabolite/metabolites in these species. For example, species differences in localization of FLT-1/VEGFR1 was reported by Hagedorn et al. (2005) who showed a unique and mainly intranuclear immunoreactivity in Brunner’s gland epithelial cells of the mouse and rat but not dog, pig, or monkey. Lenvatinib inhibits the expression of FLT-1 in Brunner’s gland of rats, which supports the notion that lenvatinib-mediated inhibition of FLT-1 dependent intranuclear signaling processes may be important in regulating gene transcription, cell proliferation, differentiation, and secretion of Brunner’s gland epithelial cells (Figure 6).
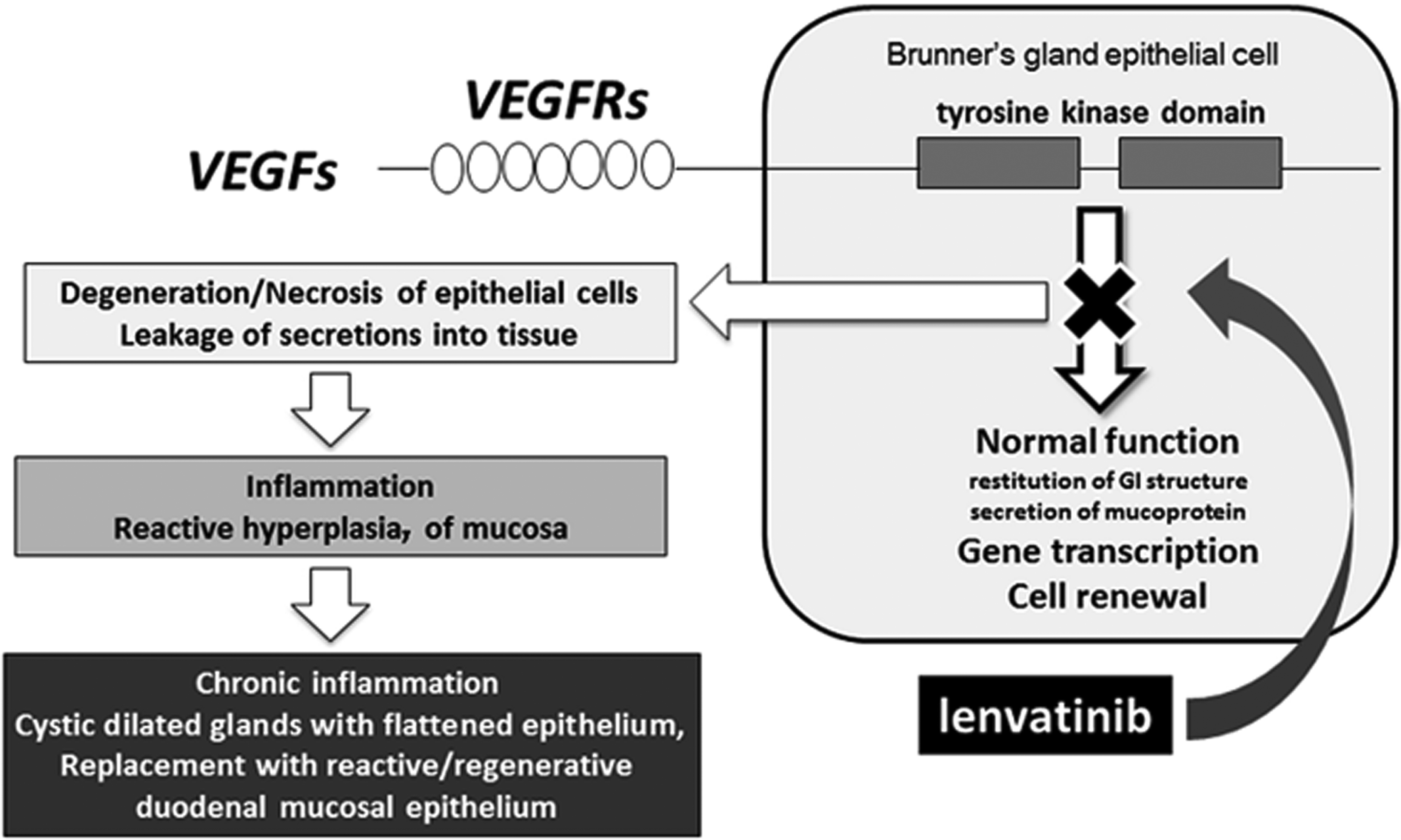
Proposed mode of toxicity in Brunner’s glands induced by lenvatinib.
In the chronic lesions at 26 weeks, the Brunner’s glands were markedly dilated, forming cystic structures that extended into the submucosa and muscular layers, and the glandular epithelium was replaced with single-layered epithelium that resembled duodenal crypt epithelium and with scattered goblet cells. This lesion was previously interpreted as the downgrowth of duodenal epithelium with adenosis and reversible hyperplasia in a 26-week rat toxicology study (Ettlin et al. 2010). However, based on our observations, our interpretation is that degeneration/necrosis of Brunner’s gland epithelium is the primary change, which then leads to inflammation and subsequent reactive hyperplasia of duodenal and pyloric mucosa, and replacement of Brunner’s gland epithelium by regenerative duodenal crypt epithelial cells, but not a “downgrowth” of the duodenal mucosa. Rather than downgrowth of the duodenal epithelium, another explanation for the change in Brunner’s gland epithelial morphology consistent with our findings may be replacement of Brunner’s gland epithelium with regenerative duodenal crypt epithelium. Comparative developmental studies of eutherian mammals have shown that Brunner’s glands derived from the duodenal mucosa, either directly or from the base of the crypts (Johnson 1910; Krause and Lesson 1967; Toofanian and Targowski 1982; Bailey et al. 1984), but in the adults, cell renewal occurs primarily from mature glandular cells within the Brunner’s glands themselves (Obuoforibo and Martin 1977). In the most severe lesions at 26 weeks, there was apparent continuity between the duodenal crypts and dilated Brunner’s glands. We think that the epithelial cells of the Brunner’s gland were replaced by duodenal crypt cells after repetitive damage to Brunner’s gland epithelial cells. We propose that while Brunner’s gland epithelium would be repopulated by epithelial regeneration of the gland itself under normal circumstances, continuous damage to the gland epithelial cells have diminished or interfered with the ability of Brunner’s gland epithelium to regenerate. While incomplete regression following a 13-week recovery period was previously reported (Thomas et al. 2005) for this lesion, we suspect that self-regeneration capacity of Brunner’s gland is partially/substantially exhausted due to continuous degeneration/necrosis (as supported by regenerative replacement of Brunner’s gland epithelium by duodenal crypt epithelial cells), and hence this lesion may require a prolonged nondosing period to show evidence of complete recovery.
Despite the presence of markedly dilated and cystic glands lined by a single layer of epithelium with scattered goblet cells in the duodenal submucosa and muscular layers in the most developed lesions, the VEGF RTK inhibitor–induced lesions morphologically differ from carcinogen-induced adenocarcinomas in rats (Sugimura, Fujimura, and Baba 1970; Tahara et al. 1982; Tamano et al. 1990; Uwagawa et al. 1991) in that features of malignancy such as individual cell invasion into the tunica muscularis and cellular atypia are not present. Even though cystic dilated Brunner’s gland extended into the tunica muscularis, this finding should be interpreted as a local extension facilitated by cystic pressure and inflammation, and lacked features of true invasion such as formation of isolated islands/cords and lymphatic/vascular invasion/metastasis. The trend of partial reversibility reported by Thomas et al. (2005) further supports that this lesion is nonneoplastic.
In conclusion, the results of our longitudinal histologic examination show that the duodenal mucosal lesion induced by VEGF RTK inhibitors starts as degeneration/necrosis of the Brunner’s gland leading to inflammation. This is the first report, to our knowledge, that presents detailed progression as well as pathogenesis of this lesion implicating Brunner’s gland epithelial degeneration/necrosis as the primary change. Our report highlights the importance of careful evaluation of Brunner’s glands for RTK inhibitors since early changes are minimal, and also underlines the importance of longitudinal, integrated evaluations in nonclinical toxicity studies in order to develop a comprehensive understanding of the pathogenesis of any lesion.
Footnotes
Abbreviations
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
