Abstract
We observed spontaneous dwarfism among Wistar Hannover GALAS rats, caused by primary hypothyroidism. The dwarf (D) rats showed markedly high serum thyroid-stimulating hormone (TSH) values. In this study, we investigated the incidence and histopathological characteristics of the proliferative lesions of the thyroid follicular cells in our D rats. Gross enlargement of thyroid and histological vacuolar change of the thyroid follicular cells with age-related progression suggesting diffuse hypertrophy/hyperplasia were seen in all the D rats. In addition, focal proliferative lesions—namely hyperplasias, activated hyperplasias, adenomas, and adenocarcinomas—were seen in D rats aged over 50 weeks. Taken together, these results suggested that the sustained stimulation of elevated levels of serum TSH caused and enhanced diffuse hypertrophy of thyroid follicular cells and subsequent development of focal proliferative lesions.
Introduction
Long-term perturbation of the pituitary–thyroid axis by various xenobiotic or physiologic alterations, such as iodine deficiency, partial thyroidectomy, and natural goitrogens in food, causes high incidence of proliferative lesions, including hyperplasia and adenomas of follicular cells in rat thyroid (Capen, DeLellis, and Yarrington 2010). Furthermore, a number of cases of thyroid cancer developing in dyshormonogenetic goiters have been reported in patients with long-standing uncontrolled hypothyroidism (Medeiros-Neto et al. 1998).
We previously observed spontaneous dwarfism caused by hypothyroidism in the offspring of Wistar Hannover GALAS rats. Serum thyroid hormone (TH) values in these animals were markedly low, while serum thyroid-stimulating hormone (TSH) values were markedly high, suggesting that the dwarfism was caused by primary hypothyroidism (Doi et al. 2004; Kokoshima et al. 2007). A previous study revealed that the dwarfism found in a litter of Wistar Hannover GALAS rats was an autosomal recessive inheritance and suggested that the single nucleotide polymorphism of thyroglobulin genomic DNA causes this abnormal phenotype (Sato et al. 2006, Accession No. AB678721).
To our knowledge, no detailed reports have yet discussed proliferative lesions in the thyroid follicular cells of spontaneously hypothyroid animals. Here, we report the incidence and histopathological characteristics of the proliferative lesions derived from thyroid follicular cells in dwarf (D) offspring of Wistar Hannover GALAS rats.
Materials and Methods
We obtained a total of 36 D rats from our colony that originated from 2 D male rats (Doi et al. 2004). A total of 8 rats were sacrificed at ages 8 and 45 weeks (2 males and 2 females at each point). The remaining 28 rats were kept alive until death or euthanasia due to deteriorated health at ages 18 to 108 weeks (12 males and 16 females). As normal controls (N), 20 Wistar Hannover GALAS rats of both sexes aged 8 and 45 weeks (5 males and 5 females of each age) purchased from CLEA Japan Inc. (Shizuoka, Japan) were used in this study (Table 1).
Animals used in the study.

Necropsy was performed after euthanasia by exsanguination under anesthesia with an intraperitoneal injection of sodium thiopental or as soon as the animal died. Thyroids were removed and fixed in 10% neutral phosphate-buffered formalin. The thyroids from 2 male and 2 female specimens of 8- and 45-week-old D rats and 45-week-old N rats as well as 10 (7 males and 3 females) D rats euthanized due to deteriorated health condition were weighed before fixation. Furthermore, the relative organ weight to body weight was calculated. The thyroids of all animals were embedded in paraffin, sectioned, and stained with hematoxylin and eosin (H&E) for microscopic examination. For immunohistochemical examination, sections of the thyroid were stained for proliferating cell nuclear antigen (Rat PCNA, mouse monoclonal antibody; Dako JAPAN Inc., Tokyo, Japan) by the labeled streptavidin biotin method (DAKO LSAB kit; Dako JAPAN Inc.). As the negative control, the PCNA antibody was replaced with Antibody Diluent (DAKO Antibody Diluent; DAKO North America, Inc., Carpinteria, CA). As the positive control, a section of ovary in 108-week-old female D rat was used. PCNA-positive cells were counted in 12 or all 400× field of the focal proliferative lesions and PCNA-positive cells per 400× field were calculated.
All animals were cared for in accordance with the principles outlined in the guide for the care and use of laboratory animals prepared by the Japanese Association for Laboratory Animal Science and our institution.
Results
Necropsy Findings in 8- and 45-week-old N rats and D rats
Thyroids in 8- and 45-week-old D rats were enlarged compared to same-aged N rats and the difference in thyroid size between N and D rats was greater in 45-week-old rats than in 8-week-old rats. Absolute and relative organ weights were markedly high for thyroids of 45-week-old D rats compared to those of same-aged N rats (Table 2). Relative organ weights were over 2 times higher for thyroids of 8-week-old D rats compared to those of same-aged normal Wistar Hannover GALAS rats (n = 13, our laboratory background data). In addition, absolute and relative thyroid weights in D rats were higher in females than in males.
Average thyroid weights.
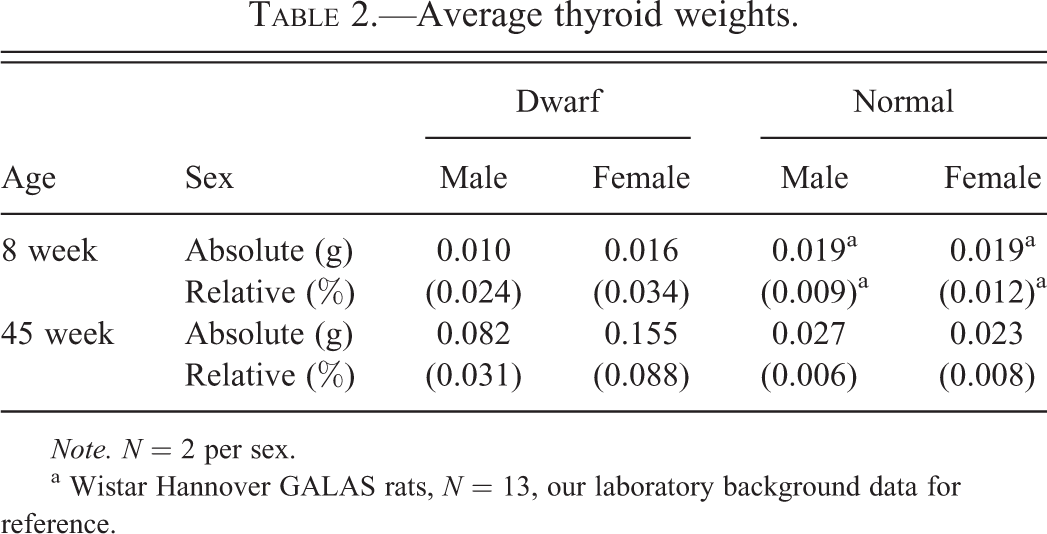
Note. N = 2 per sex.
a Wistar Hannover GALAS rats, N = 13, our laboratory background data for reference.
Necropsy Findings in Dead or Moribund Sacrificed D Rats
Animal number, age, sex, thyroid weights, body weights, and gross and histopathological findings are shown in Table 3. Thyroid enlargement was observed in all animals. In addition, thyroids were so large that they compressed the trachea and subsequently suffocated 1 female (No. 22). Enlarged thyroids showed a nodular appearance in 3 females (Nos. 20, 25, and 27) and were accompanied with a single nodule and a whitish patch in 1 female each (Nos. 16 and 26, respectively; Figure 1), suggesting tumor presence. Except for 3 males (Nos. 3, 4, and 8), absolute and relative weights of thyroids were high compared to those of same-sex 45-week-old D rats. Furthermore, in those 3 males, both absolute and relative weights of thyroids were high compared to those of 45-week-old male N rats. Nodules of the pituitary, suggesting tumor development, were seen in 4 males and 10 females, and were considered to have been the cause of death or deterioration in all cases, due to compression of the adjacent brain.
Animal information, thyroid and body weights, gross and histopathological findings in thyroids of dwarf (D) rats.
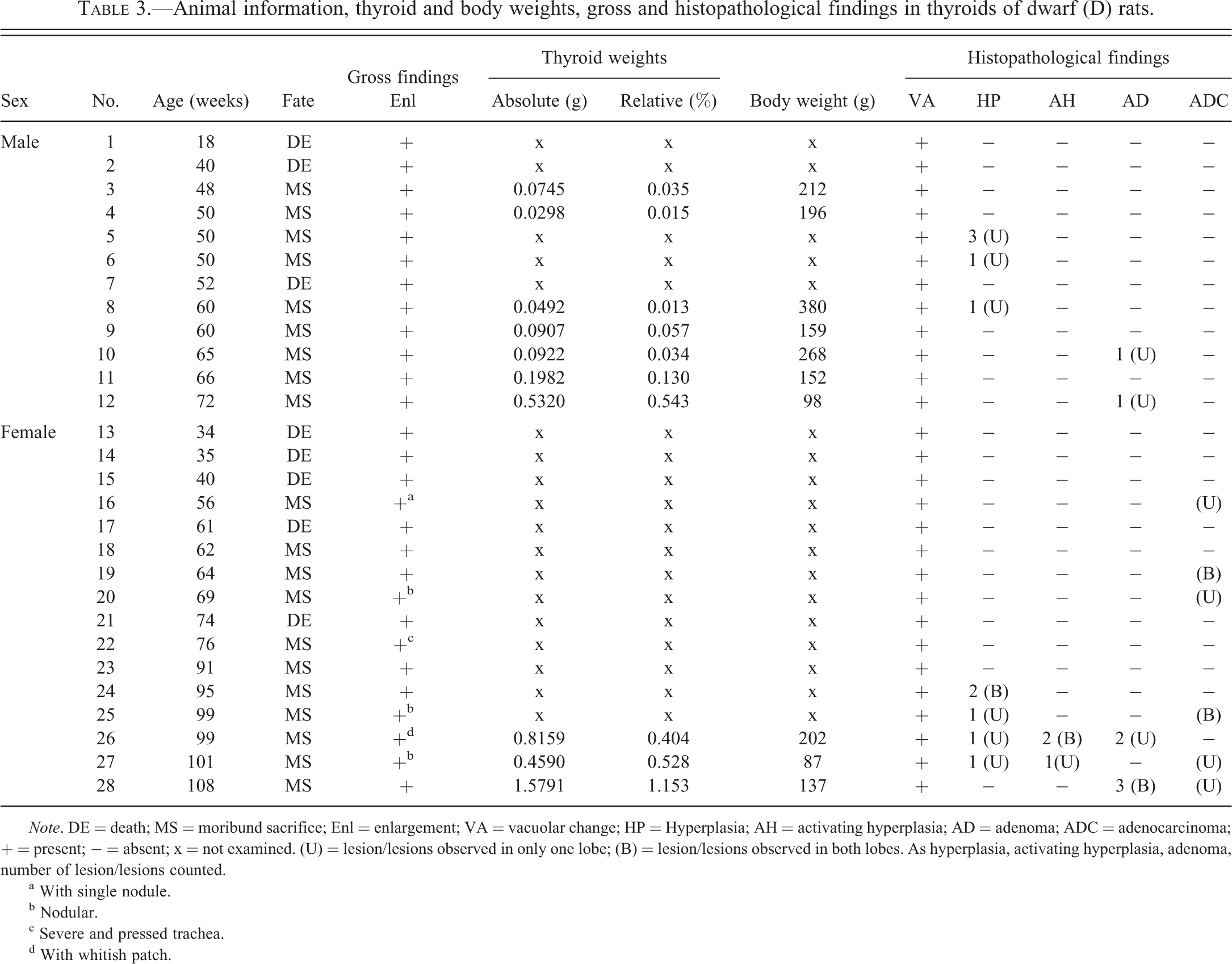
Note. DE = death; MS = moribund sacrifice; Enl = enlargement; VA = vacuolar change; HP = Hyperplasia; AH = activating hyperplasia; AD = adenoma; ADC = adenocarcinoma; + = present; − = absent; x = not examined. (U) = lesion/lesions observed in only one lobe; (B) = lesion/lesions observed in both lobes. As hyperplasia, activating hyperplasia, adenoma, number of lesion/lesions counted.
a With single nodule.
b Nodular.
c Severe and pressed trachea.
d With whitish patch.
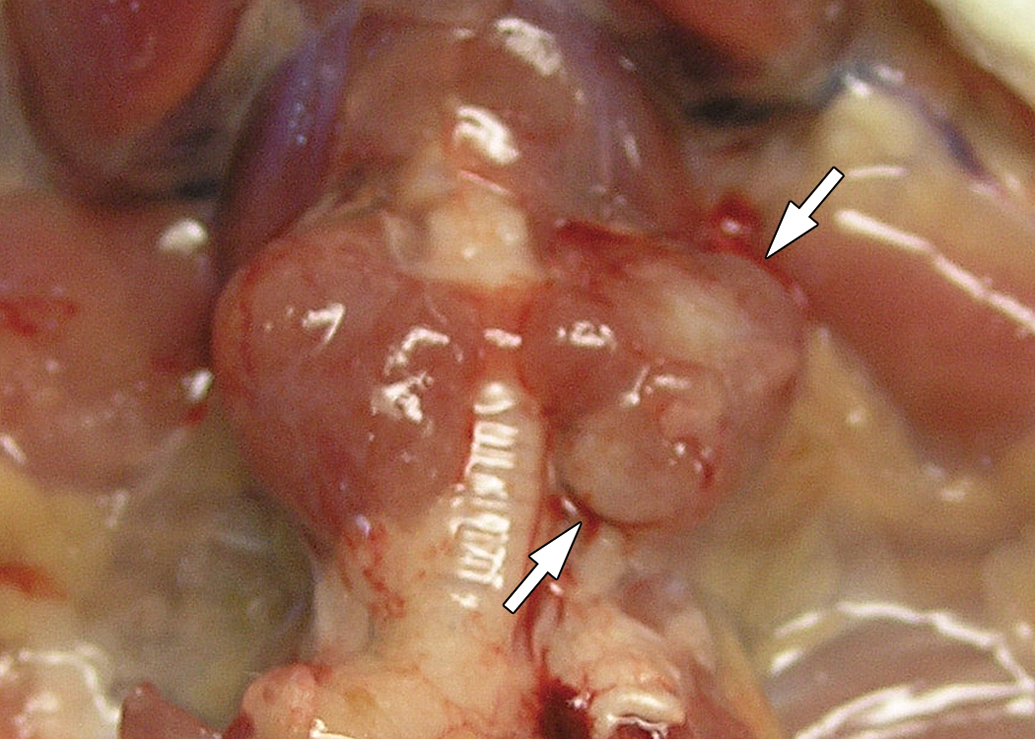
Thyroids of a 99-week-old female dwarf rat (No. 26). Enlargement and whitish patch (white arrow) are seen.
Histological and Immunohistochemical Findings in 8- and 45-week-old N Rats and D Rats
Vacuolar change and enlargement of the follicular cell characterized by huge intracytoplasmic vacuoles in the follicular cells, little colloid formation, and a decrease in follicular size were observed in all D rats, corresponding to observations in our previous studies (Doi et al. 2004; Kokoshima et al. 2007). Vacuoles were localized at the base of the follicular cells and were weakly eosinophilic (Figure 2A, B). In 8-week-old D rats, follicular cells were uniformly columnar and clearly showed single layer arrangements and clear lumina (Figure 2A). In 45-week-old D rats, the shape of follicular cells became irregular due to cells pressing against each other, and layer arrangements and lumina became unclear, showing an acinar-like appearance (Figure 2B). PCNA-positive cells were rarely seen, and no focal proliferative lesions were observed in any 8- or 45-week-old D rats.
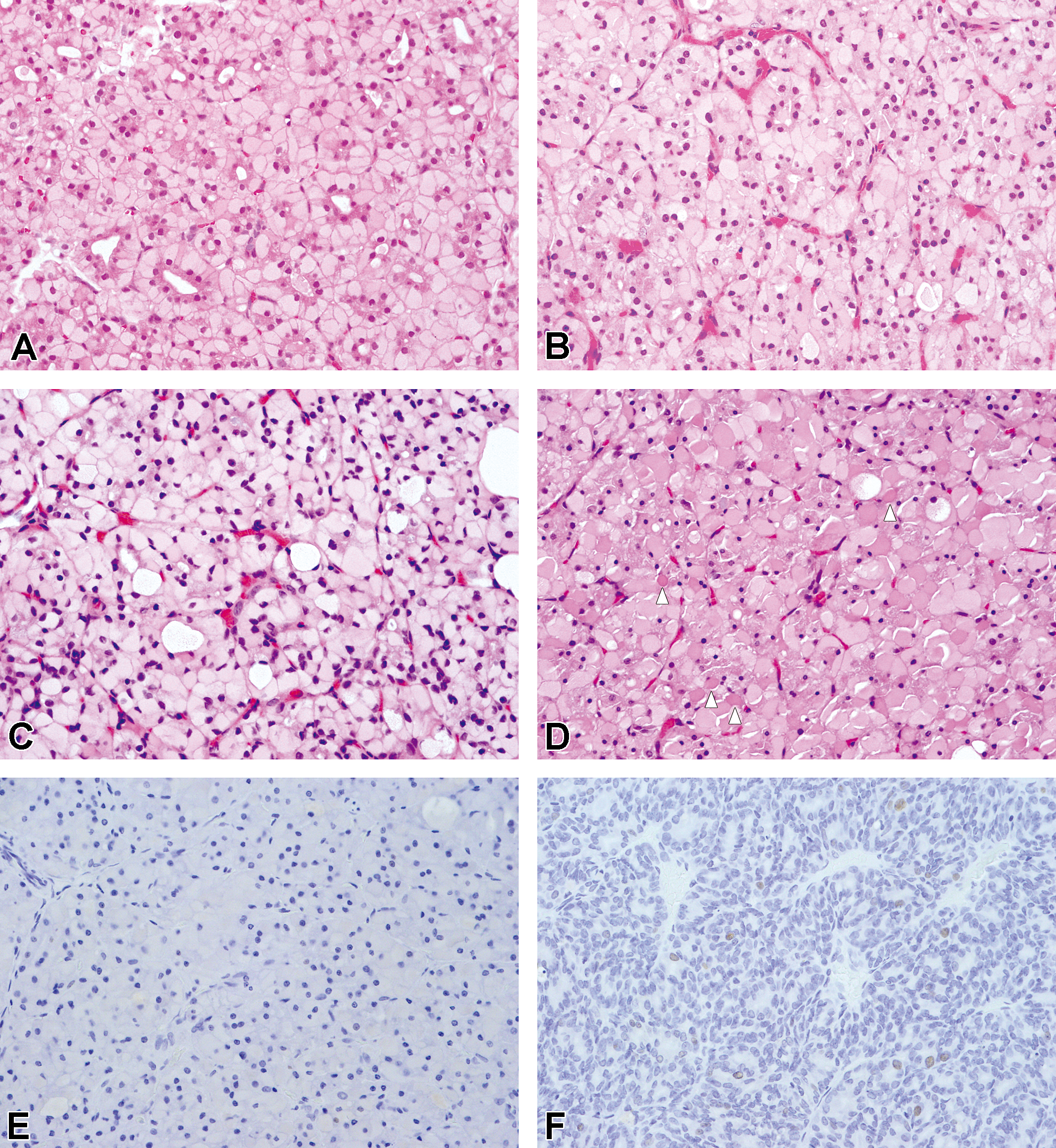
Vacuolar change of follicular cells with age-related progression. (A) 8-week-old dwarf rat. Single layer arrangements of columnar follicular cells are present. (B) 45-week-old dwarf rat. The shape of follicular cells is irregular and follicular structure is unclear showing acinus-like appearance. (C) 56-week-old dwarf rat. Follicular structure is more unclear showing lobular-like appearance (No. 16). (D) 108-week-old dwarf rat. Lobular-like appearance and degenerating cells are seen (white arrow head, No. 28). (A–D: H&E. Original objective lens magnification: 40×). (E) 69-week-old rat (No. 20). No PCNA-positive cell is seen. (F) 108-week-old rat (No. 28). Rare PCNA-positive cells are seen. (E, F: PCNA immunohistochemical stain. original objective lens magnification: 40×). H&E = hematoxylin and eosin; PCNA = proliferating cell nuclear antigen.
Histological and Immunohistochemical Findings in Dead or Moribund Sacrificed D Rats
All dead or moribund sacrificed D rats showed vacuolar change with age-related progression. That is, in rats aged over 50 weeks, follicular appearance was less clear than in 45-week-old D rats showing lobular-like appearance (Figures 2C, D). In addition, female rats aged over 64 weeks exhibited degeneration of follicular cells with condensed nucleus (4 females: Nos. 22, 26, 27, and 28; Figure 2D), thickening of capsules, and parenchymal divisions by connective tissue (6 females: Nos. 19, 20, 22, 25, 26, and 28).
Although thyroid sizes were markedly increased, few PCNA-positive cells were observed in follicular cells (Figure 2E), except for only 1 female (No. 28). In follicular cells of this oldest animal, only a few PCNA-positive cells were observed (Figure 2F).
We also observed focal proliferative lesions of follicular cells in the 13 dead or moribund sacrificed D rats (Table 3). We diagnosed these lesions according to World Health Organization (WHO) classification (Botts et al. 1994), International Harmonization of Nomenclature and Diagnostic Criteria for Lesions in Rats and Mice (http://www.goreni.org/), and from textbooks (Capen et al. 2010; Hardisty and Boorman 1990), and classified these focal proliferative lesions as hyperplasia, adenoma, and adenocarcinoma based on the histological and PCNA-immunoreactive features.
Animals laden with these focal proliferative lesions were all aged over 50 weeks, and 4 had more than 2 types of lesions.
Hyperplasia was defined as a noncapsulated and uncompressed focal lesion and showing an alteration in the follicular structure and/or follicular cell shape (Figure 3A–D). They could be divided into 2 types mainly by PCNA-positive rates. One was a small round lesion and showed no PCNA-positive cells (Figure 3E) except for 1 lesion showing 1 PCNA cell in whole lesion (0.3 PCNA-positive cells per 400× field). They showed 3 histological patterns: cystic dilatation of the follicle (Figure 3A), crowded appearance with relatively small-sized follicular cells (Figure 3B), or focus of expanded follicular cells with a huge intracytoplasmic vacuole (Figure 3C). The follicular cells of this hyperplasia had similar or larger sized intracytoplasmic vacuoles compared to the surrounding follicular cells (Figure 3A–C, inset). The other one was a poorly demarcated, irregular-shaped lesion with markedly high cell density (Figure 3D). These hyperplastic cells were small in size, with vacuoles smaller than that of the surrounding follicular cell (Figure 3D, inset), and showed relatively high PCNA-positive rates (average 1.3 PCNA positive cells per 400× field [range: 0.9–2.1], Figure 3F). The former hyperplasias were seen in males aged 50 weeks or older and females aged 95 weeks or older with 1 male and 1 female having 2 or more hyperplasias (Nos. 5 and 24; Table 3). The latter activated hyperplasia was noted in just 2 females at ages 99 and 101 weeks (Table 3).
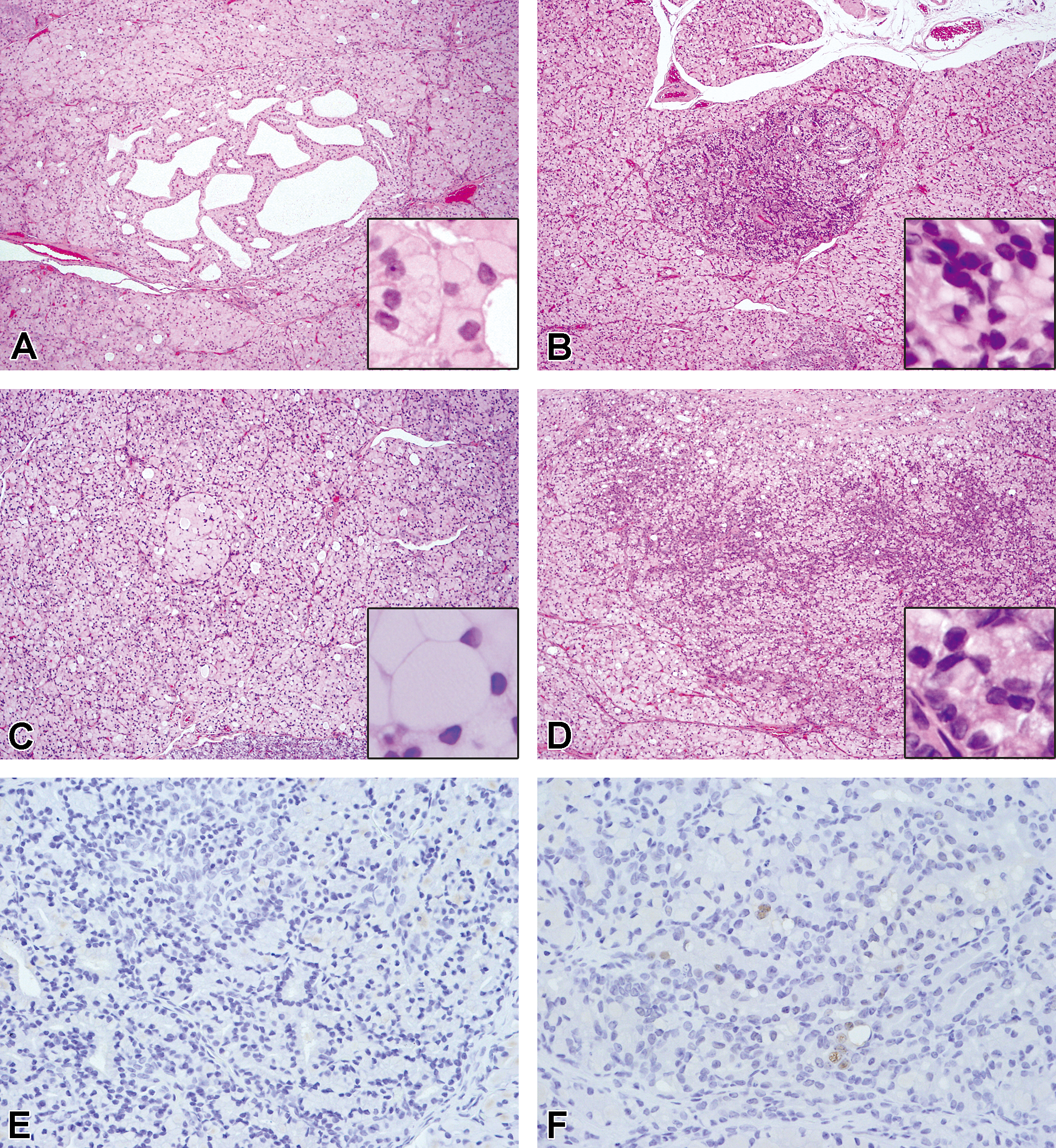
Hyperplasia and activated hyperplasia of follicular cells. (A) Hyperplasia showing cystic dilatation of follicle, 95-week-old rat (No. 24). (B) Hyperplasia showing a crowded appearance with small-sized follicular cells, 95-week-old rat (No. 24). (C) Hyperplasia showing huge intracytoplasmic vacuoles, 50-week-old rat (No. 5); in hyperplasia, intracytoplasmic vacuole is larger or almost the same size compared to surrounding hypertrophic follicular cells (A–C, inset). (D) Activated hyperplasia showing irregular shape and high cell density, 99-week-old rat (No. 26). Their intracytoplasmic vacuole is distinctly smaller than that of surrounding hypertrophic follicular cells (inset). (A–D: H&E. Original objective lens magnification: 10×, inset: 40×). (E) Hyperplasia, crowded type, in a 95-week-old rat (No. 24). No PCNA-positive cell. (F) Hyperplasia in a 99-week-old rat (No. 26). 2.1 PCNA cells per 400× field. (E, F: PCNA immunohistochemical stain. Original objective lens magnification: 40×). H&E = hematoxylin and eosin; PCNA = proliferating cell nuclear antigen.
Adenoma was defined as a demarcated round lesion with minimal circumference compression. Some adenomas were partially or completely surrounded by a fibrous capsule. Follicular patterns (Figure 4A) and solid patterns (Figure 4B) were noted. Neoplastic follicular cells had enlarged and hyperchromatic nuclei and very small-sized vacuoles or no vacuoles (Figure 4A, B, inset), and showed high PCNA-positive rates (average 1.9 PCNA positive cells per 400× field [range: 0.0–7.5], Figure 4E). Adenomas were seen in 2 males at ages 65 and 72 weeks and 2 females at ages 99 and 108 weeks. Two males had 1 adenoma each and 1 female had 2 adenomas, the other 1 had 3 adenomas (Table 3).
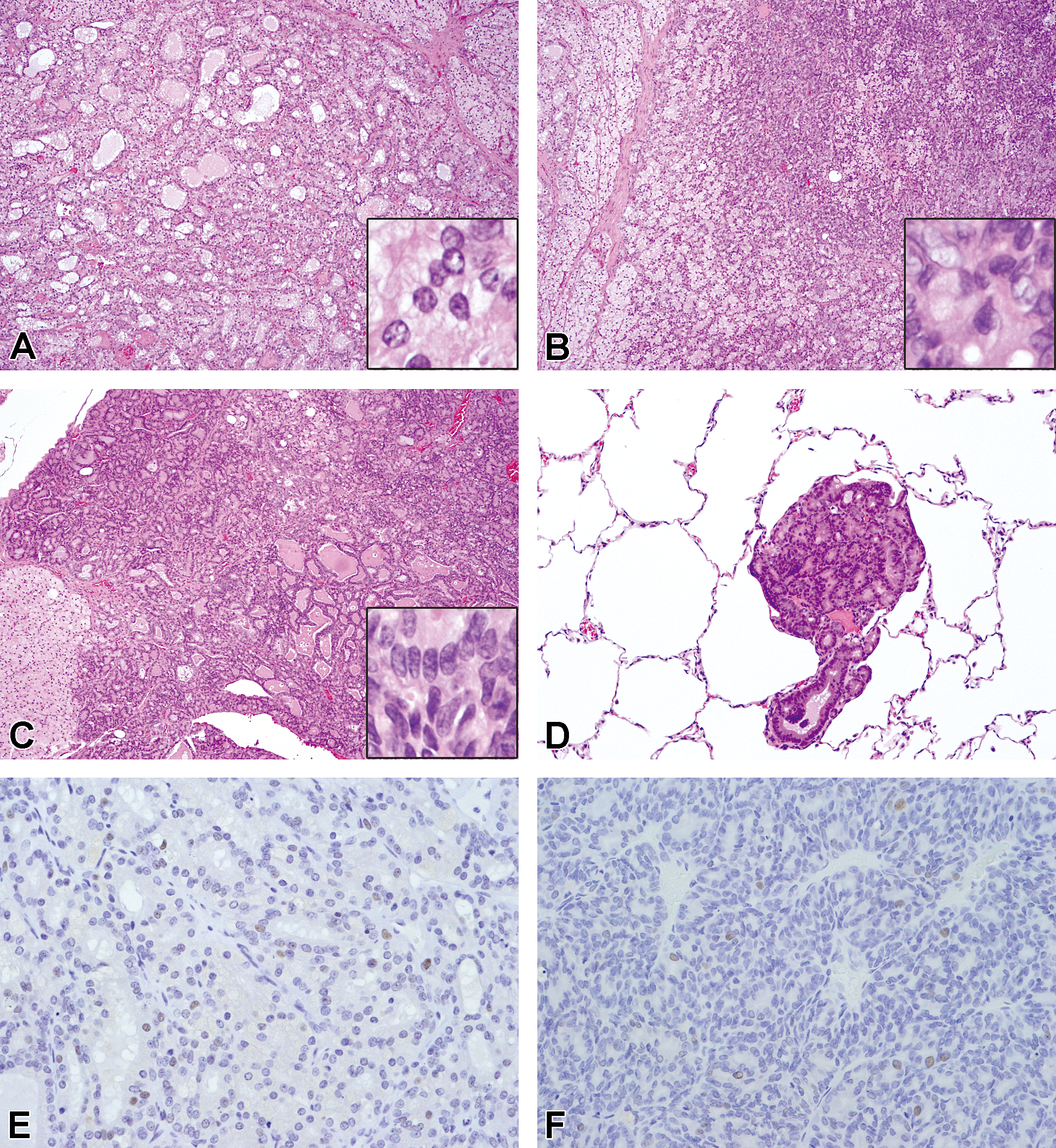
Neoplastic lesions of follicular cells. Adenoma, follicular (A) and solid (B) pattern, 99-week-old rat (No. 26). The cells have enlarged nuclei and very small or no intracytoplasmic vacuole (A and B, inset). Adenocarcinoma showing irregular follicular proliferation (C) with lung metastasis (D), 64-week-old rat (No. 19). The cells have enlarged and hyperchromatic nuclei and no or very small intracytoplasmic vacuole (C, inset). (A–D: H&E. Original objective lens magnification: A–C; 10×, inset: 40×, D; 20×). (E) Adenoma, 99-week-old rat (No. 26). 7.5 PCNA cells per 400× field were seen. (F) Adenocarcinoma, 64-week-old rat (No. 19). 10.4 PCNA cells per 400× field were seen. (E, F: PCNA immunohistochemical stain; original objective lens magnification: 40×). H&E = hematoxylin and eosin; PCNA = proliferating cell nuclear antigen.
Adenocarcinoma was defined as a large round or irregular-shaped lesion with invasion into the capsule, surrounding parenchyma, or blood vessels. In 3 cases (Nos. 16, 19, and 25), we observed metastasis to the lung (Figure 4D). Adenocarcinomas showed structure atypia such as irregular follicular, papillary, and a solid proliferative pattern (Figure 4C). Neoplastic follicular cells had enlarged and hyperchromatic nuclei with atypia and very small-sized or no vacuoles (Figure 4C, inset). Although few mitotic figures were seen, PCNA-positive rates were markedly high (average 5.4 PCNA positive cells per 400× field [range: 0.6–10.4], Figure 4F). Adenocarcinoma was noted in 6 females aged 56 weeks or older in 1 or both lobes. Nodular enlargement and nodules observed at necropsy were related to adenocarcinoma.
Discussion
Thyroids from all D rats in this study showed diffuse vacuolar change of follicular cell with age-related progression. Age-related increases in thyroid size and weight accompanied by morphological change of follicular cells and structure with thickening of capsules and parenchymal divisions by connective tissue suggests that diffuse hypertrophy/hyperplasia of follicular cells, so-called goiter, occurred in our D rats.
As prolonged TSH stimulation caused by various chemicals or drugs is well known to result in diffuse hypertrophy/hyperplasia of follicular cells (Capen, DeLellis, and Yarrington 2010), it is possible that hypertrophy/hyperplasia of thyroid follicular cells caused by persistently high serum TSH values occurred in these D rats.
In normal GALAS rats, it has been reported that vacuolar change of thyroid follicular cells sometimes occurs as a spontaneous lesion (Shimoi et al. 2001). Doi et al. (2004) reported that this vacuolar change was observed in all our D rats and some of their normal littermates. Although Weber et al. (2009) reviewed these reports and termed this change thyroid dysplasia from a genetic origin only, we think it is inaccurate to indicate this morphological change as “thyroid dysplasia.” In pathology, dysplasia is defined as “alteration in its shape, size, and organization, usually affecting the epithelium, occasionally associated with neoplastic transformation” (Wallig 2010), and in common usage it is principally applied to epithelial cells that have undergone proliferation and atypical cytologic alterations considered to be closely related to hyperplasia (Cortran, Kumar, and Robbins 1989). Strictly speaking, dysplasia is used to denote deranged development; however, there is no data to prove this vacuolar change was “deranged development.” So, we are using the term vacuolar change to indicate this change in our D rats.
In addition to a high incidence of adenoma and adenocarcinoma, occurrence of characteristic nonneoplastic proliferative lesions classified as hyperplasia or activated hyperplasia was confirmed. The incidence of neoplastic lesions (16.7% in males and 43.8% in females of dead or moribund sacrificed animals) was also markedly high compared to that of BrlHan:WIST@Tac(GALAS) rat (3.9% in males, 2.1% in females, CLEA Japan, Inc. 2002) and RCCHan™:WIST, Wistar Hannover rat (4.3% in males, 2.7% in females, Harlan Laboratories Ltd. 2013). To our knowledge, this is the first report discussing proliferative lesions in the thyroid follicular cells of spontaneously hypothyroid rats.
Prolonged TSH stimulation caused by various chemicals or drugs is well known to be strongly related to the development of focal proliferative lesions, including neoplasm of thyroid follicular cells (Mitsumori et al. 1995). As the focal proliferative lesions occurred in D rats aged 50 weeks or older, induction of focal proliferative lesions may require a longer term stimulation than that which resulted in diffuse hypertrophy/hyperplasia.
Although the thyroids showing the vacuolar change with normal growth reported to be more sensitive to carcinogens than normal thyroids (Abe et al. 2012), there was no indication of increased preneoplastic and neoplastic lesions in oncogenicity studies (Weber et al. 2009). These facts also support the relationship between occurrence of these lesions in our D rats and TSH stimulation.
We were able to easily distinguish hyperplasia from surrounding parenchyma based on alteration of structures such as cystic dilatation of follicles, increases in cell density, or alteration in cell morphology such as expansion with a huge vacuole. Activated hyperplasia was thought to be the result of more active proliferation than hyperplasia, showing high cell density, high cell proliferation activity as demonstrated by the high PCNA-positive rates and irregular shapes of lesions. Hyperplasia of thyroid follicular cell by various xenobiotic or physiologic alterations such as iodine deficiency, partial thyroidectomy, and treatment regimens that induce follicular cell tumors are generally composed by cystic or irregular-shaped follicles (Capen, DeLellis, and Yarrington 2010; Mitsumori et al. 1999; Yasuhara et al. 2001). Huge vacuole or solid-type hyperplasia and activated hyperplasia observed in the present study are considered to be D rat–specific characteristic lesions by these morphological differences from previous reported hyperplasia. Our findings suggest that hyperplasia is the first proliferative lesion, as it showed the earliest occurrence and highest incidence among all focal proliferative lesions, low proliferative activity as demonstrated by small lesion size, no compression, and no or few PCNA-positive cells. No relationships were noted among any of the focal proliferative lesions due to the small number of examined animals and absence of transition between them. On the other hand, activated hyperplasia and adenoma were only seen relatively late in life (>99-week-old 2 females, >65-week-old, 2 males and females, respectively).
Incidences of activated hyperplasia, adenoma/adenocarcinoma, and multiplicity of hyperplasia were higher in females than in males. In particular, incidence of adenocarcinoma was extremely high in females (37.5%) and started to occur from as early as 56 weeks. In addition, increases in thyroid weights and the serum values of TSH were more severe in females than in males (Doi et al. 2004). This sex difference in incidence is extremely interesting, as males were believed to be more susceptible to the development of proliferative lesions in response to chronic TSH stimulation in rodents than in females (Capen, DeLellis, and Yarrington 2010). However, in humans, thyroid cancer is roughly 2.5 times more common in females than in males, with sex hormones therefore suspected to play an extremely important role in the development of thyroid cancer (Henderson et al. 1982). In the ovariectomized female rats only, a 2-stage thyroid carcinogenesis model using antithyroidal chemicals was enhanced by β-estradiol-3-benzeoate implantation (Takizawa et al. 2006). Although we failed to measure serum estrogen levels in our D rats, estrogen may play an important role in development of these proliferative lesions of thyroid follicular cells in our D rats.
In conclusion, high incidence of proliferative lesions of thyroid follicular cells was observed in our D rats and was believed to result from sustained high serum TSH levels. Furthermore, this incidence was higher in females than in males.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
