Abstract
Liver resection is a suitable option for the treatment of certain hepatic conditions, particularly hepatocarcinomas, in patients with cirrhosis. However, this disease impairs liver regeneration, which increases the risk of liver failure and postoperative death. Supportive treatments for regeneration of the remaining liver may be useful for the recovery of these patients. We demonstrated that nutritional hepatotrophic factors (NHF) is an effective regenerative stimulus for cirrhotic livers in rats subjected to partial hepatectomy (PH). The rats with thioacetamide-induced cirrhosis were subjected to PH, and they were divided into 2 groups. One group received intraperitoneal administration of NHF, and the other group received saline solution. After 12 days, biometric data, collagen content, hepatocyte regeneration (proliferation cell nuclear antigen immunochemistry), and profibrotic gene expression (Collagen-α1, matrix metalloproteinase 2, tissue inhibitor of metalloproteinase 1, and transforming growth factor beta 1) were assessed. The results indicated that the rats treated with NHF after PH had an increased liver size, a reduced amount of collagen, and a higher hepatocyte proliferation index compared with the rats that underwent PH alone. In addition, collagen-α1 gene expression was decreased in the NHF-treated rats. Thus, postoperative improvement in the liver morphology following NHF treatment may cause a significant decrease in the risk of liver failure and mortality after hepatic resection.
Keywords
Introduction
Intraperitoneal administration of a parenteral solution of nutritional hepatotrophic factors (NHF), consisting of a mixture of amino acids, vitamins, trace elements, and specific hormones (e.g., insulin, glucagon, and triiodothyronine [T3]), has been shown to simulate the production of hepatotrophic substances of splanchnic origin that reach the liver through the portal system (Parra et al. 1994). In fact, it has been demonstrated that the in vivo administration of NHF has a strong ability to accelerate and augment cellular proliferation as well as the growth of normal (Parra et al. 1994, 1995), fibrotic (Cogliati et al. 2010), and cirrhotic (Guerra et al. 2009) liver tissues. Moreover, in fibrotic or cirrhotic livers, NHF treatment causes a reduction in the amount of tissue collagen and an improvement in the liver morphology and function (Cogliati et al. 2010; Guerra et al. 2009; Parra et al. 1996). Furthermore, hepatocyte regeneration often results in the regression of liver cirrhosis (Kawada 2011).
However, cirrhotic livers demonstrate a decreased regenerative ability, and hence, the regenerated liver tissue after hepatic resection has high levels of fibrosis (Andiran et al. 2000; Moser, Kneteman, and Minuk 2000; Redai, Emond, and Brentjens 2004; Suárez-Cuenca et al. 2008). Additionally, the removal of liver tissue may result in acute liver failure. Therefore, hepatectomy is a risky procedure in patients with liver cirrhosis (Moser, Kneteman, and Minuk 2000; Min et al. 2011; Zimmermann and Reichen 1998; Wilasrusmee et al. 2004). However, in cases of liver trauma, liver infection, or liver neoplasms, liver resection may be necessary in patients with cirrhosis (Merli et al. 2002; Redai, Emond, and Brentjens 2004). Surgical resection is considered the treatment option of choice for patients with hepatocarcinoma (HCC) as well as for those with concomitant cirrhosis (Moser, Kneteman, and Minuk 2000; Poon and Fan 2004; Mittler, McGillicuddy, and Chavin, 2011; Zarrinpar, Kaldas, and Busuttil 2011). Patients with compensated cirrhosis are the best candidates for surgical resection (França et al. 2004; Llop et al. 2011; Bruix and Sherman 2011).
Hence, the objective of this study was to evaluate whether the administration of NHF in rats following partial hepatectomy (PH) can accelerate the regeneration of cirrhotic liver tissue and restore the functional liver mass, thereby promoting a more rapid morphofunctional recovery. To this end, we evaluated the following aspects of liver status: (1) absolute and relative liver size; (2) the amount of collagen tissue (measured using morphometry); (3) the rate of cellular proliferation (measured using the proliferating cell nuclear antigen [PCNA] labeling index); and (4) the relative expression of genes involved in extracellular matrix production, including collagen alpha 1 (Col-α1), matrix metalloproteinase 2 (MMP-2), tissue inhibitor of metalloproteinase 1 (TIMP-1), and transforming growth factor beta 1 (TGF-β1; measured using RT-PCR).
Materials and Method
Animals and Experimental Design
Forty female Wistar rats weighing between 195 g and 210 g were used in this study. The animals were maintained in accordance with the Guide for the Care and Use of Laboratory Animals. The rats received a standard rat diet with ad libitum access to water and were maintained under a controlled photoperiod, humidity, and temperature. All experimental procedures were approved by the Bioethics Commission of the School of Veterinary Medicine and Animal Science of the University of São Paulo (protocol #1641/2009).
All rats received intraperitoneal injections of 4% thioacetamide (TAA) (Sigma-Aldrich, St. Louis, MO; 150 mg/kg dissolved in 0.9% NaCl) 3 times per week for 15 weeks. The animals were weighed before each injection, and the doses were adjusted accordingly. After the resting period, the rats that underwent a 40% PH were randomly divided into the following two groups: a group that was treated with intraperitoneal injections of NHFs (PH + NHF group, N = 20) daily for 12 days and a group that received saline injections (0.9% NaCl) daily instead of NHF for 12 days (PH + S group, N = 20). The detailed experimental design is shown in Figure 1.
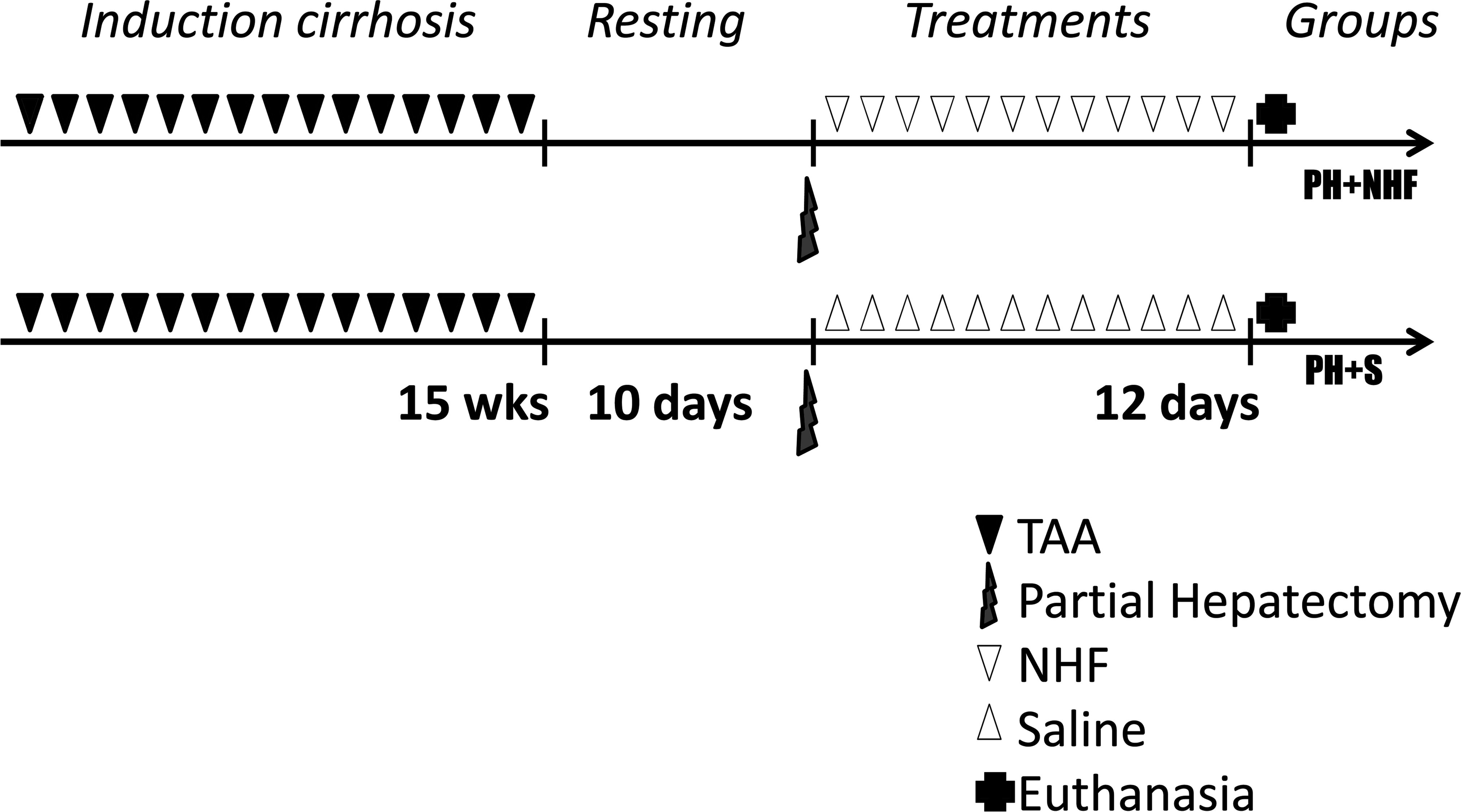
Thioacetamide-induced cirrhotic rats were subjected to different experimental procedures. Partial hepatectomy (PH) was performed in both groups. The PH + NHF group received intraperitoneal injections of nutritional hepatotrophic factors (NHF), and the PH + S group received saline injections (S; 0.9% NaCl) under the same conditions.
Surgical Procedures
All procedures were performed under continuous isoflurane/O2 inhalation. Rats in both the PH + NHF and PH + S groups were subjected to a 40% PH. The surgical procedure was performed according to the method described by Higgins and Anderson (1931), with the exception that only the median lobe was resected for removing approximately 40% of the hepatic mass (Martins and Neuhaus 2007).
Composition and Administration of Nutritional Hepatotrophic Factors
The parenteral solution of NHFs consisted of 104 g of glucose, 2 mg of pyridoxine, 2 mg of calcium pantothenate, 30 mg of thiamine, 4 mg of riboflavin phosphate, 1.43 g of potassium chloride, 1.50 g of sodium bicarbonate, 50 mg of nicotinamide, 750 mg of potassium monophosphate, 500 mg of magnesium sulphate, 500 mg of vitamin C, 62.5 IU of insulin, 0.625 mg of glucagon, 2.5 mg of folic acid, 31.25 µg of vitamin B12, and 3.125 mg of zinc sulphate. All these hepatotrophic substances were added to a 10% amino acid solution (200 ml) containing 180 mg of
The NHF solution was administered intraperitoneally to the rats in the PH + NHF group twice a day for 12 days (40 ml/kg/day), whereas the rats in the PH + S group received physiological saline injections under the same conditions. One day after the end of treatment, all of the rats in both groups were euthanized by excessive exposure to anaesthesia, according to the methods of the American Veterinary Medical Association (AVMA), and liver samples were obtained.
Biometric Data
Immediately after euthanasia, the body weight (BW) and liver weight (LW) were measured and recorded. The LW relative to BW values, also known as hepatosomatic index, were obtained using the following formula:
Blood Collection and Biochemical Analysis
Blood samples were obtained by cardiac puncture while the animals were under anaesthesia (immediately prior to euthanasia) and were processed immediately to obtain the plasma concentrations of alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), and total bilirubin (TBil) using the VetTest 8080 Blood Chemistry Analyser (Iddex, New York, NY).
Quantification of Collagen and Immunohistochemistry
Liver samples collected at the time of PH and at the time of euthanasia were fixed in Bouin and methacarn fixatives and embedded in paraplast. Tissue sections (5 µm in thickness) fixed in Bouin were stained using the picrosirius red method, and they were then subjected to collagen quantification and fibrosis scoring. Sections from the material fixed in methacarn were used for PCNA immunohistochemistry.
The staging of fibrosis and architectural changes was performed according to the methods of Ishak, where the presence of occasional nodules of collagen is considered incomplete cirrhosis, or score of 5, and the presence of regular and usual nodules is considered complete cirrhosis, or score of 6 (Ishak et al. 1995). The staging was performed to ensure that only those rat livers with a score of 6 that were diagnosed based on the hepatectomy samples prior to the NHF or saline treatment were used and analyzed in the experiments.
The quantification of fibrillar collagen content was performed by histomorphometric analysis, wherein 3 random fields from 5 random, nonserial slides from each rat were examined under an Olympus BX-60 (Olympus, Tokyo, Japan) optical microscope using a 20× objective, and the images were captured with an AxioCam HRc (Carl Zeiss, Thornwood, NY). The collagen content was measured using an image analysis system, AxioVision 4.8.2.0 (Carl Zeiss, Thornwood, NY), which separates the collagen fibres (stained red) from the background (stained light pink) by thresholding and calculates the percentage of collagen per microscopic field. The collagen content is typically reported as the percentage of red-stained fibers (Dias et al. 2008). The mean percentage of the fibrous area from the 5 histologic sections obtained from 1 rat is equivalent to the volumetric proportion (VP) of collagen fibres in the liver of this rat (Cogliati et al. 2010). The following 2 types of comparisons for VP were performed: intergroup comparison (between samples from the rats of the PH + NHF group and those from the rats of the PH + S groups after euthanasia) and intragroup comparison (between samples from the same rat in each respective group after hepatectomy and after euthanasia).
Methacarn-fixed sections were immunostained for PCNA (monoclonal mouse PC10; 1:100 in PBS; Dako, Glostrup, Denmark) using the LSAB + System HRP kit (DakoCytomation, Carpinteria, CA), according to the manufacturer’s instructions. The positive cells became evident following the application of DAB (3′3-diaminobenzidine tetrahydrochloride) and the hematoxylin counterstain. Fragments of the small intestine were used as internal positive controls for verifying the antibody specificity of the immunohistochemical reaction. The PCNA labeling index value was defined as the percentage of labeled cells per 1,000 liver cells (Andiran et al. 2000) when viewed using the 40× objective.
RNA Purification and Real-time PCR
Total RNA was extracted from 40 mg of frozen liver tissue using an RNAspin mini kit (GE, Fairfield, CT), and real-time PCR analysis was performed using an ABI Prism® 7000 Sequence Detection System (Applied Biosystems, Carlsbad, CA). TaqMan Universal Master Mix (Applied Biosystems, Carlsbad, CA) was used for each PCR reaction. The β-actin forward primer (ACCATGAAGATCAAGATCAT), the β-actin reverse primer (AGATTACTGCCCTGGCTCCTA), and the internal β-actin TaqMan probe (FAM-CCACCAATCCACACAGAGTACTT) were used to detect the expression of the housekeeping gene β-actin (Park et al. 2010). The primers for the detection of Col-α1 (Rn00801649), TGF-β1 (Rn00695755m1), TIMP-1 (Rn00587558m1), and MMP-2 (Rn01538167m1) were purchased from Applied Biosystems (Applied Biosystems, Carlsbad, CA). The relative gene expression analysis was performed using the 2–ΔΔCt method (Livak and Schmittgen 2001).
Statistical Analysis
The samples were analyzed for normality using the Kolmogorov–Smirnov test. The parametric data were compared using Student’s t-test for the intergroup means and Student’s paired t-test or analysis of variance (ANOVA; two-way ANOVA with the Bonferroni posttest) for the intragroup means, and the nonparametric data were compared using the Mann–Whitney test. All values are expressed as the M ± SD. Differences in the mean values were considered statistically significant when the p value was <.05. The data analysis was performed using Minitab 16 (Minitab, State College, PA)
Results
Rat Cirrhosis Model
No mortality was observed during the cirrhosis induction period, and not all of the rats developed complete cirrhosis (Ishak score 6) following the administration of TAA; this result was verified by the histopathologic examination of the liver samples taken at the time of PH. Among the rats with an Ishak score of 6 (22 rats or 55% of the initial sample size), 11 rats were randomly assigned to the PH + NHF group, and 11 rats were randomly assigned to the PH + S group. No ascites, jaundice, or neurological alterations were found in the rats over the course of the entire experimental period, similarly that occurs in compensated cirrhosis (Regimbeau et al. 2008). Cirrhotic rats demonstrated a high degree of fibrosis, the presence of regenerative nodules, disruption of the histological architecture, and bile duct proliferation; however, neoplastic lesions were not observed.
Biometric Data
In the rats of the PH + NHF group, the liver weight and its relative to body weight values increased by 8.4% and 5.6%, respectively (p < .05; Table 1), thereby indicating an absolute and relative increase in the liver mass compared with the rats of the PH + S group.
Comparison of biometric parameters between the groups following the administration of saline or nutritional hepatotrophic factor solution.
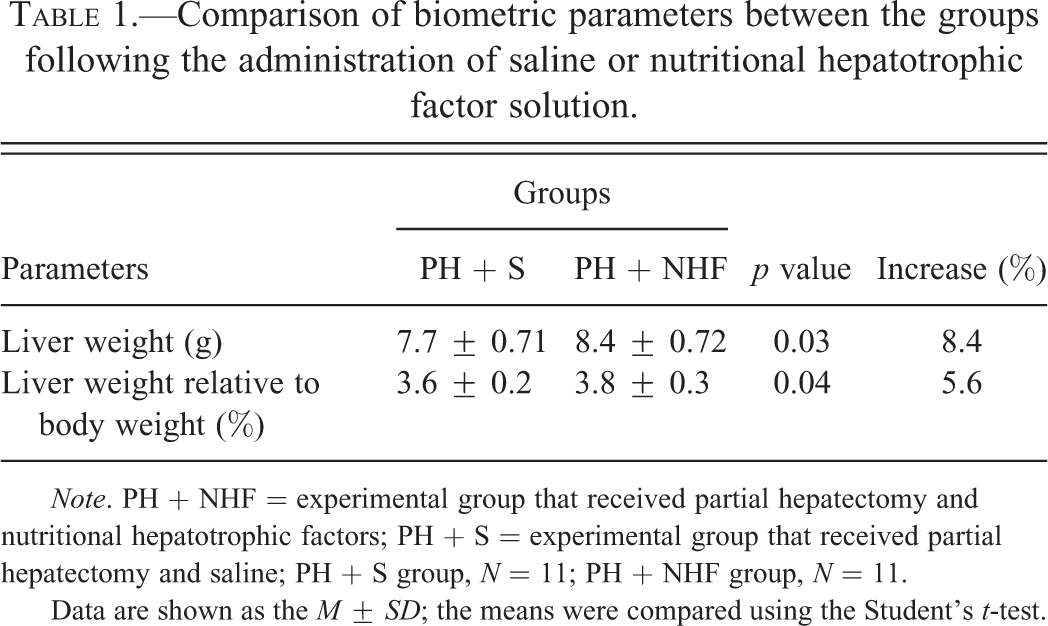
Note. PH + NHF = experimental group that received partial hepatectomy and nutritional hepatotrophic factors; PH + S = experimental group that received partial hepatectomy and saline; PH + S group, N = 11; PH + NHF group, N = 11.
Data are shown as the M ± SD; the means were compared using the Student’s t-test.
Histopathology and Quantification of Collagen
The VP of collagen in the biopsy samples taken at the time of PH was similar in all the rats, and this result indicated similar levels of cirrhosis among the selected rats (those with an Ishak score of 6). The morphometric measurements showed that the VP of collagen was decreased in both the groups (intragroup comparison), regardless of whether the rats had received NHF. However, the reduction in the VP of collagen was greater in the group that had received NHF (PH + NHF group, 49.1%, p < .05) than in the group that had received only saline solution following PH (PH + S group, 28.7%, p < .05; Figure 2).
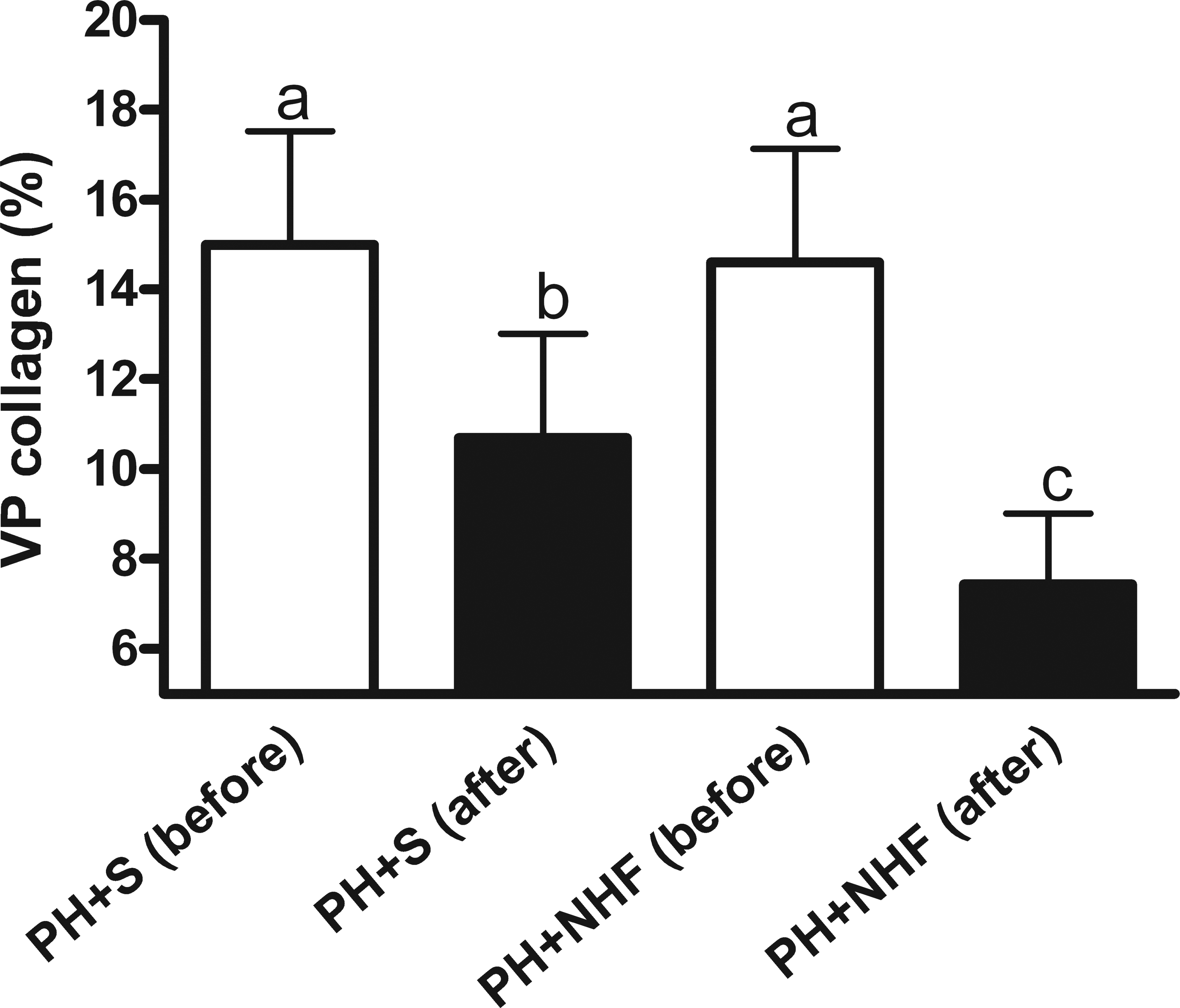
The volumetric proportion (VP) of collagen in livers of rats submitted to PH before and after the indicated treatments (M ± SD, intragroup comparison). The white bars represent the 2 groups prior to the administration of saline (PH + S) or nutritional hepatotrophic factors (PH + NHF), whereas the black bars represent the same 2 groups after the treatment administration period. Two-way ANOVA and the Bonferroni posttest were performed. Different letters indicate statistically significant differences (p < .05).
The qualitative assessment of collagen content and the distribution of collagen fibers revealed a clear decrease in the extent of the fibrotic area, a thinner septa and fewer regenerative nodules in both the experimental groups compared with the same cirrhotic livers prior to the indicated treatment (intragroup comparison). However, according to the measurements shown previously, these findings were the most evident in the PH + NHF group (Figure 3).
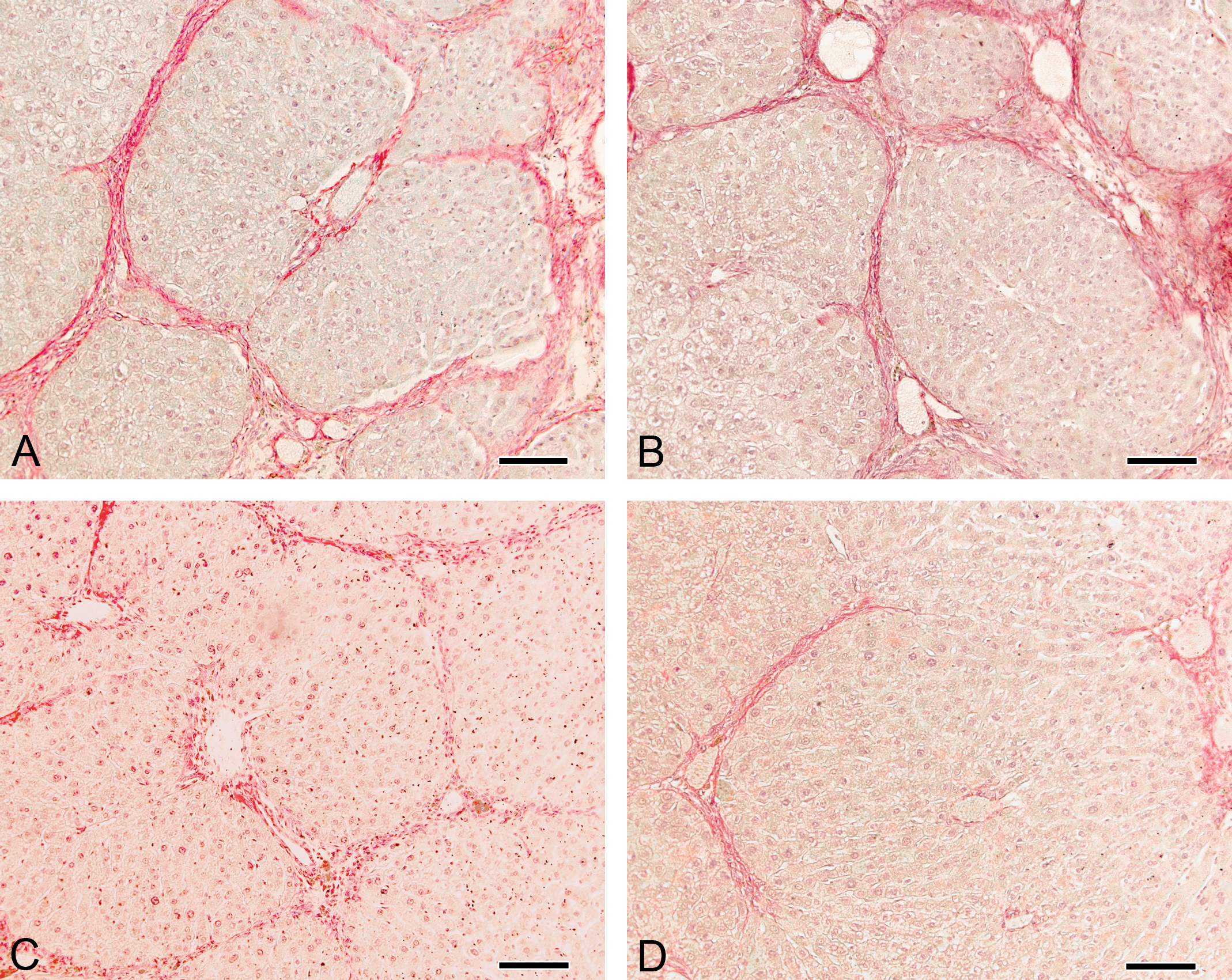
Liver sections stained using the picrosirius red method, wherein the collagen is highlighted in dark red. A and B: livers of 2 rats at the time of partial hepatectomy (PH) prior to treatment; C: liver sample from the same rat (A) after receiving saline (S) for 12 days following PH; D: liver sample from the same rat (B) after receiving nutritional hepatotrophic factors (NHF) for 12 days following PH. Magnification: 200×.
PCNA Labeling Index
The PCNA proliferation index values were higher in the rats of the PH + NHF group than those in the rats of the PH + S group (1.29 ± 0.23% and 0.89 ± 0.41%, respectively; p = .01; Figure 4), thereby indicating a difference of 44.9% between the 2 groups.
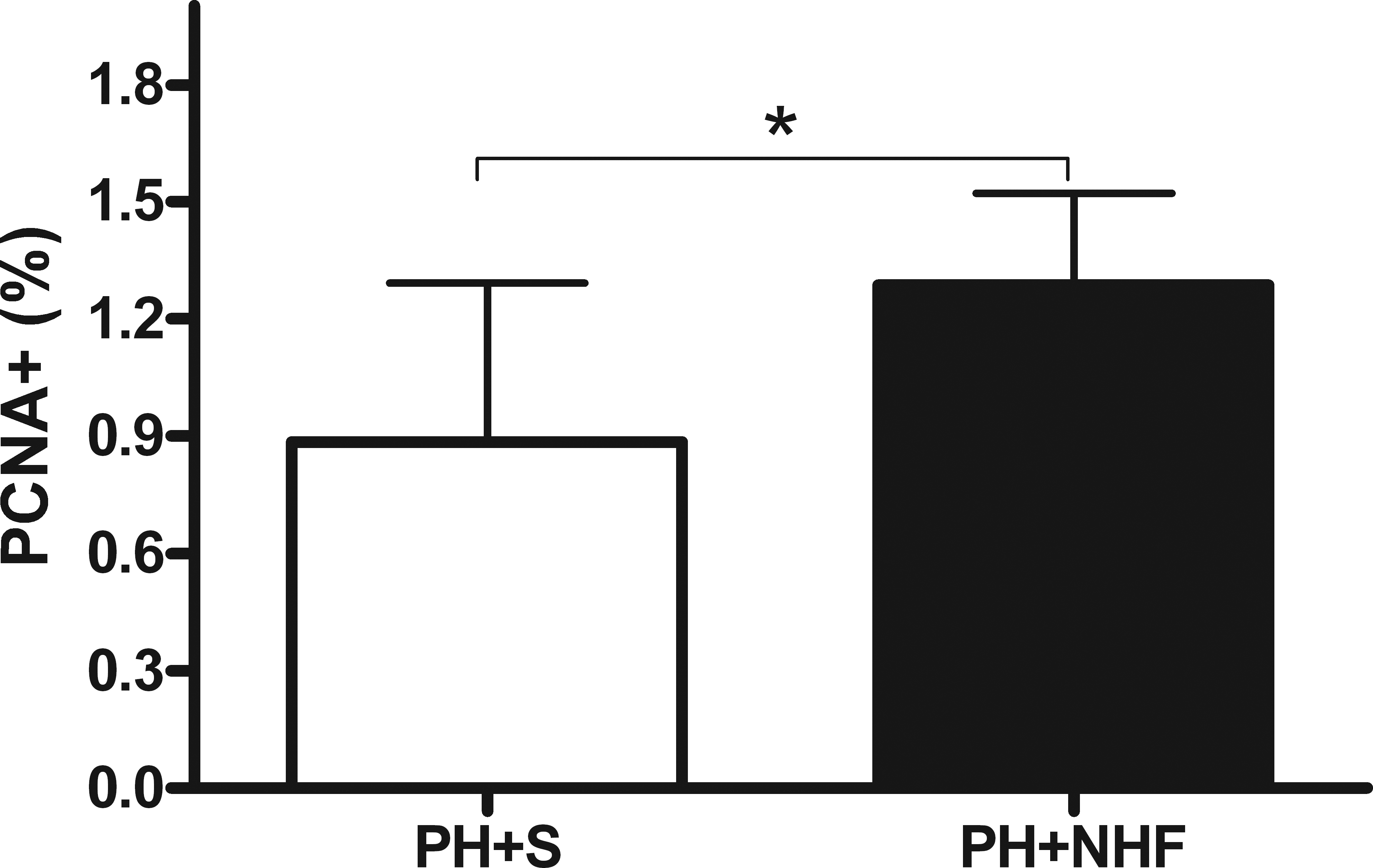
PCNA labeling index values displayed as a percentage (M ± SD). The white bar represents the group that received saline (PH + S), whereas the black bar represents the group that received nutritional hepatotrophic factors (PH + NHF). Student’s t-test was performed (*p < .05).
Hepatic Biochemistry
There was a decrease of 23.7% in the ALP level in the rats of the PH + NHF group compared with that in the rats of the PH + S group (p < .01). However, no differences were found in the ALT, AST, GGT, or TBil levels between the 2 groups (Table 2).
Comparison of the hepatic biochemistry parameters between the 2 groups after the administration of saline or nutritional hepatotrophic factor solution.
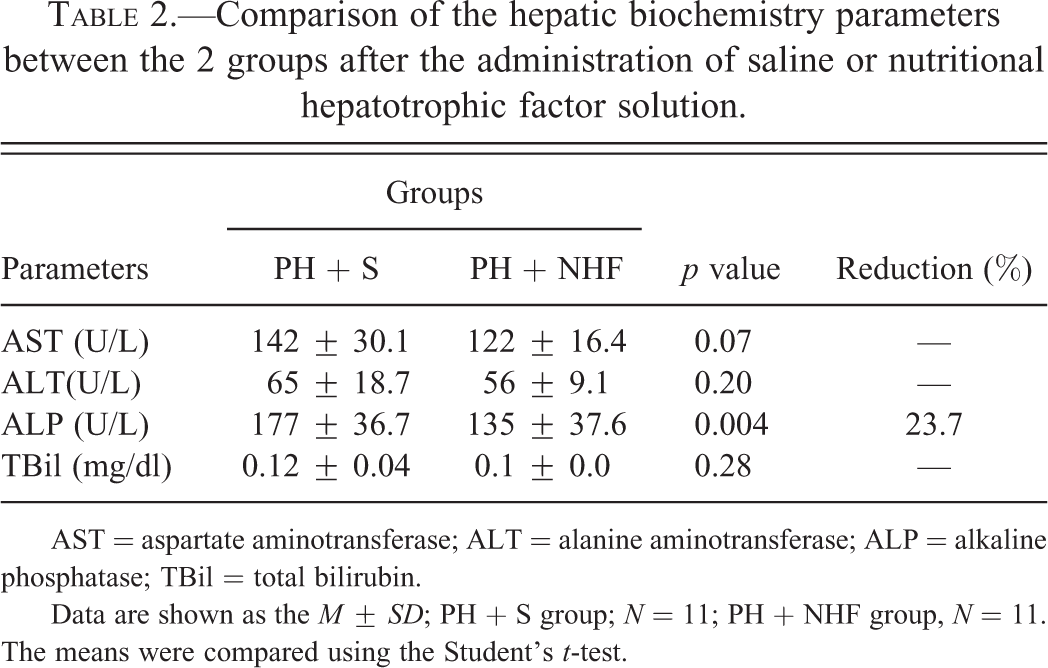
AST = aspartate aminotransferase; ALT = alanine aminotransferase; ALP = alkaline phosphatase; TBil = total bilirubin.
Data are shown as the M ± SD; PH + S group; N = 11; PH + NHF group, N = 11. The means were compared using the Student’s t-test.
Gene Expression
There was a reduction of 54% in the relative expression of Col-α1 in the PH + NHF group compared to the PH + S group after the respective treatments (0.46 ± 0.29 and 1.00 ± 0.34, respectively; p = .048; Figure 5). However, no differences were observed in the expression levels of MMP-2, TIMP-1, or TGF-β1 between the two groups.
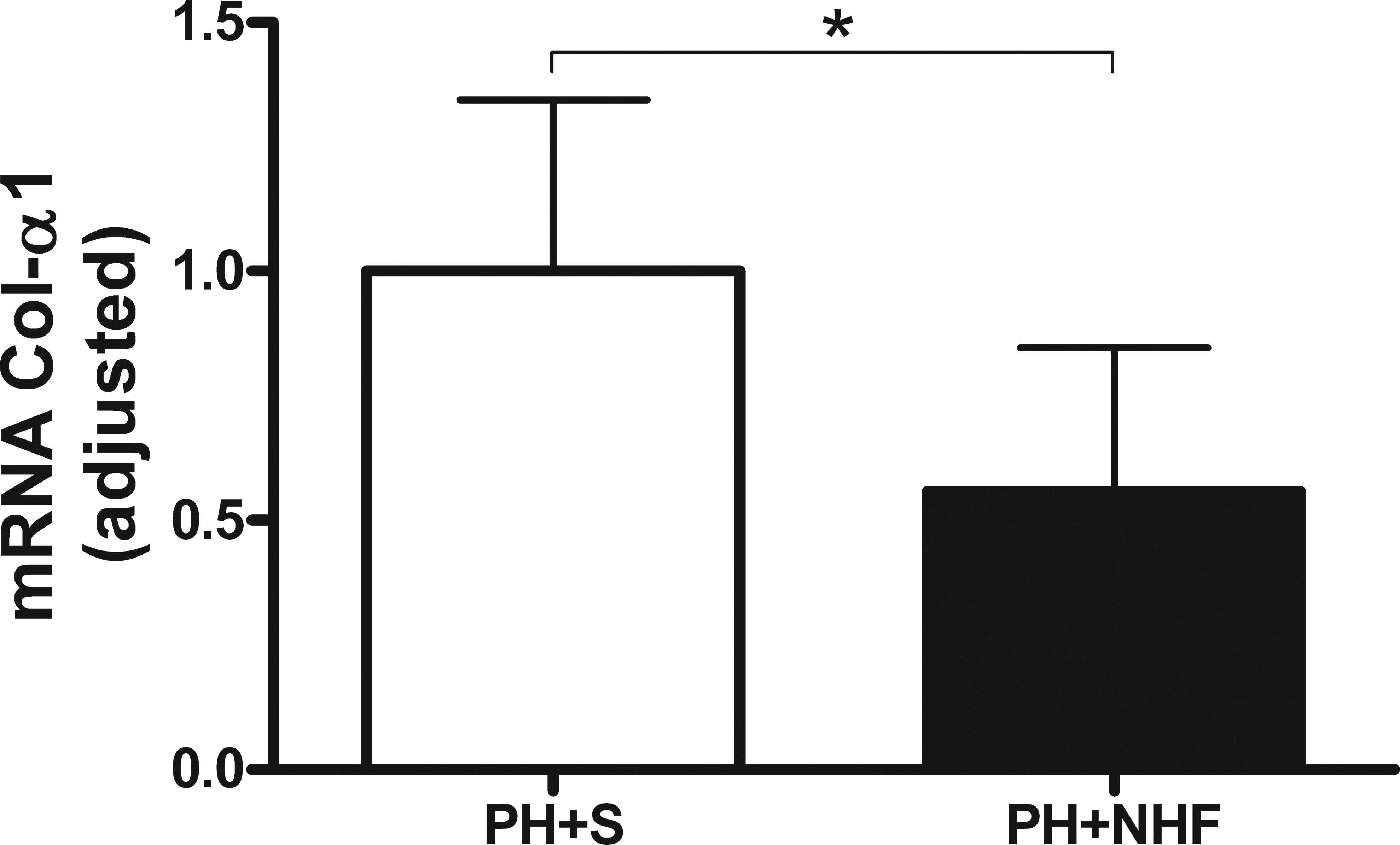
Relative expression of collagen α 1 (M ± SD). The white bar represents the group that received saline (PH + S), whereas the black bar represents the group that received the nutritional hepatotrophic factor solution (PH + NHF). Student’s t-test was performed (*p < .05).
Discussion
Liver resection may cause fatal injury to the liver tissue, and this outcome suggests that more effective methods need to be developed to minimize liver damage and promote liver regeneration (Min et al. 2011). Regeneration of the remaining liver tissue may be particularly useful in cirrhotic patients undergoing hepatic resection (Moser, Kneteman, and Minuk 2000).
In the present work, the administration of NHF was demonstrated to provide an additional effective regenerative stimulus in the rats with liver cirrhosis that underwent PH, and this finding was demonstrated by the higher PCNA proliferative index values in the rats of the NHF-treated group. In addition, the biometric parameters indicated that the absolute and relative livers sizes of the rats treated with NHF were greater than those of the rats that received saline solution. Rats in the PH + NHF group demonstrated an increase of 8.4% and 5.6% in the liver weight and liver weight relative to body weight, respectively. Minimal liver function appears to be adequate to sustain life (Moser, Kneteman, and Minuk 2000), and NHF was shown to promote a more rapid recovery and an increase in the hepatic mass. Moreover, the NHF-treated rat livers had a larger VP of parenchyma with a reduction in the extent of fibrotic area. The collagen content in the livers of rats that received NHF was significantly reduced by 49.1% after treatment (intragroup comparison), whereas the collagen content in the cirrhotic livers of rats that received saline solution was reduced by 28.7% (intragroup comparison). The reduction in the collagen content was expected because the PH procedure in itself is an important regenerative stimulus, and it has previously been studied as a potential anti-cirrhotic treatment (Hashimoto and Watanabe 1999). The intergroup comparison (between the NHF-treated group and the saline-treated group) provided an indication of the ability of the NHF treatment to accelerate the reversion of liver fibrosis, considering that the relative reduction in the liver collagen was 27.9% after NHF treatment. Morphologically, this observed decrease in the liver collagen can be explained by the reduction in the quantity of regenerative nodules and fibrous interportal septa, as was noted during the microscopic evaluation of liver histology. These findings, which indicate an improvement in the morphological aspects of the liver, suggest an improved internal blood flow and improved recovery conditions for the local tissue. A reduction in the extent of fibrotic area in a cirrhotic liver can enhance the blood flow, increase the blood supply to the residual liver parenchyma and ensure rapid tissue growth (Redai, Emond, and Brentjens 2004).
These results confirm the ability of the NHF solution to accelerate liver regeneration and increase the VP of the liver parenchyma following PH. In our previous work, we have shown that the NHF treatment can reduce the collagen content (37.1%) in cirrhotic animals (independent of PH) due to increased degradation of the extracellular matrix (Guerra et al. 2009).
Regeneration of the liver parenchyma was associated with higher PCNA labeling index values. Administration of NHF has previously been shown to increase the rate of hepatocyte proliferation in fibrotic (Cogliati et al. 2010) and cirrhotic livers (Guerra et al. 2009). In the present work, the rats that received NHF showed a significant increase of 44.9% in the PCNA labeling index compared with the rats of the PH + S group, and this result indicates a greater regenerative capacity of the liver among the rats in the NHF-treated group (Min et al., 2011). Following PH, the peak of cellular proliferation is known to occur within the first 24 hr in healthy livers and within the first 48 hr in cirrhotic livers, and the level of cellular proliferation decreases after 72 hr (Hashimoto and Watanabe 1999). In the current study, the treatment duration was 12 days, and the cells from the rats subjected to PH demonstrated a low rate of proliferation at the end of this period. However, the administration of NHF provided a regenerative stimulus during the 12 days of treatment. Thus, it is likely that prolonged treatment with NHF will further accelerate liver regeneration and reduce collagen deposition in the hepatic parenchyma.
The qualitative and quantitative reduction in the collagen content of the liver tissue is in accordance with the gene expression levels of Col-α1, which also demonstrated a significant reduction (54%) in the rats that were administered the NHF solution compared to the rats that were administered saline. A similar reduction in the gene expression level of Col-α1 has been shown in the rats with cirrhotic livers that were treated with NHF (Guerra et al. 2009), thereby indicating that the reduction in new collagen deposition together with an increase in liver parenchyma are the primary effects of the NHF treatment. In contrast to previous studies, no differences were observed in the expression of other profibrotic genes involved in cirrhosis. In our study, an overlap of different mechanisms that are able to modulate gene expression is likely, and therefore, the analysis becomes increasingly complex because each individual may respond differently. For example, TGF-β1 is known to play an important role in liver fibrogenesis by participating in the activation of hepatic stellate cells (HSC) and inducing the expression of TIMPs and pro-collagen. However, TGF-β1 also plays an important paracrine role in the modulation of liver regeneration due to its ability to inhibit cellular proliferation and control the regeneration of liver tissue. In addition, the expression and production of TGF-β1 by non-parenchymal cells of the liver during liver regeneration is high, especially in the final stages of this process (Park et al. 2010). Moreover, TGF-β1 may play an important role in controlling the cessation of oval cell proliferation after PH (Park and Suh 1999). In the current study, both groups demonstrated a high cellular proliferative activity, and this cellular proliferative activity may have interfered with the TGF-β1 expression.
MMP-2 plays an important role in the early stages of liver injury, and it contributes to the accumulation of scar tissue (Knittel et al. 1999; Hemmann et al. 2007; Park et al. 2010). However, MMP-2 also plays an important role in the degradation of collagen I during fibrosis regression (Arthur 2000; Hemmann et al. 2007). After PH, the expression of MMP-2 is markedly elevated (Zhou et al. 2004; Kirimlioglu, Kirimlioglu, and Yilmaz 2008; Phan Van et al. 2008), and the inhibition of MMP-2 expression can prevent liver regeneration in mice (Alwayn et al. 2008). An increase in TIMP-1 expression is also one of the main aspects of hepatic fibrogenesis that serves to inhibit the activity of collagenase and decrease the degradation of the ECM. Additionally, an increase in TIMP-1 expression reduces the proliferation of hepatocytes in mice subjected to PH (Mohammed et al. 2005), and it regulates the expression of MMPs as well as that of the other cytokines that influence ECM remodeling (Bellayr, Mu, and Li 2009). Thus, resolution of fibrosis primarily depends on the degradation of collagen and the low level of TIMP-1 activity (Bruck et al. 2001). In contrast, during liver regeneration, TIMP-1 expression may be increased in response to elevated TGF-β1 levels. Thus, these findings reflect the presence of concurrent paradoxical processes that occur in regenerating cirrhotic livers. The expression of MMP-2 and TIMP-1 have also been reported to remain unchanged during the regeneration of cirrhotic livers (Hashimoto and Watanabe 1999; Lee et al. 2003).
Resection of approximately 40% of the liver tissue, which is considered a minor PH procedure, was chosen for this study because major hepatectomies pose grave risks even in patients with normal liver function (Seyama and Kokudo 2009), and the surgical risk for any type of procedure is greater in individuals with cirrhosis than in the general population (Low et al. 2004). In cirrhotic animals with impaired liver function, after PH, the remnant liver may not be able to perform the required functions, and acute hepatic failure can occur (Hashimoto and Watanabe 1999). Although regenerative responses typically occur after all types of liver resection, these responses may be more delayed after a minor PH procedure (Masson et al. 1999).
Although severe liver damage has been observed in TAA-treated rats, the levels of AST, ALT, and TBil did not differ between the groups; one possibility is that this result was observed because the period between the end of cirrhosis induction and blood collection was 23 days, and alterations in these parameters are not expected after this time period (Nakata et al. 1996; Oren et al. 1996; Ramaiah 2007). Only the ALP levels were reduced in the PH + NHF group (23.7%), thereby indicating reduced ductal injury and/or cholestasis (Ramaiah 2007), which are the common features of TAA-induced cirrhosis (Laleman et al. 2006). Furthermore, the morphological enhancement and the decrease in ductal cell proliferation in the livers of rats treated with NHF may explain the above finding.
In cirrhosis, there are global alterations in the 3 main categories of macronutrients (proteins, lipids, and carbohydrates) and in the glucoregulatory hormones, such as insulin and glucagon (Bianchi et al. 2008). In addition, several trace elements have been reported to be significantly decreased in TAA-induced cirrhotic rats (Abul et al. 2001). Thus, providing nutritional support for patients with cirrhosis may prevent progressive liver failure, reduce mortality, and improve their quality of life (Merli et al. 2002; Bianchi et al. 2008; Urata et al. 2007; Holeck 1999). In subjects undergoing major hepatic resection or surgery, the correction of malnutrition and the prevention of weight loss may reduce mortality, both prior to the surgical procedure and following the surgical procedure (Bianchi et al. 2008; Fan et al. 1994). It is important to provide metabolic support for the remnant liver during the early period after PH (Hashimoto and Watanabe 1999); the NHF solution may enrich the portal blood with nutrients that are diminished during cirrhosis, and it can also stimulate liver regeneration.
In conclusion, intraperitoneal administration of NHFs following hepatic resection in rats with cirrhosis was shown to increase cell proliferation and liver regeneration, which was accompanied by a significant reduction in the collagen content and morphological enhancement, and it provided essential nutrients that aid in postoperative recovery. Our results demonstrate that NHF is a promising candidate for an effective neoadjuvant or adjuvant therapy (Poon and Fan 2004) in patients with cirrhosis undergoing PH, to further improve the survival outcomes and patient recovery. Due to the ability of NHF to maintain high levels of hepatocyte proliferation up to as many as 12 days, prolonged treatment with NHF could potentially further improve the condition of the liver in these patients.
Footnotes
Acknowledgment
We would like to thank Formula Medicinal for preparing and kindly providing the nutritional hepatotrophic factor solution.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by FAPESP and CNPQ.
