Abstract
Dexamethasone was given in 2 oral dosing regimens with repeat dose oral administration of the gamma secretase inhibitor (GSI), PF-03084014, in Sprague-Dawley (SD) rats in order to evaluate the effects of coadministration of dexamethasone on GSI-induced goblet cell hyperplasia (GCH) in the intestinal tract. Safety end points were evaluated in 1 week and 1 month studies. The dosing regimens tested in the 1-month studies included a 1-week pretreatment with 1.0 mg/kg dexamethasone followed by a 3-week repeat dose treatment with 100 mg/kg GSI or concurrent intermittent treatment with 1.0 mg/kg dexamethasone on weeks 1 and 3 and repeat dose treatment with 100 mg/kg GSI for 4 weeks. Pretreatment with dexamethasone for 1 week transiently mitigated the severity of intestinal GCH for up to 1 week. Intermittent coadministration of dexamethasone on weeks 1 and 3 with GSI repeat dosing for 4 weeks mitigated intestinal GCH for up to 4 weeks post treatment. Treatment-related morbidity and mortality occurred on day 7 with 150 mg/kg GSI and 5 mg/kg dexamethasone coadministration, and on days 13, 14, and 23 with 100 mg/kg GSI and 1 mg/kg dexamethasone coadministration.
Introduction
The Notch1 receptor is a class I membrane protein complex that acts as a transcriptional activator upon Jagged or Delta-like molecule ligand binding and directly transduces information from extracellular signals to changes in nuclear gene expression (Paganin and Ferrando 2011). Notch1 pathway signaling activation involves 2 successive proteolytic cleavage steps. The first proteolytic cleavage step occurs in the extracellular domain and involves ADAM/TACE (A disintegrin and metalloprotease/tumor-necrosis-factor converting enzyme) family and the second proteolytic cleavage step occurs in the transmembrane domain and involves the gamma (γ) secretase multisubunit protease complex (Nefedova and Gabrilovich 2008). Upon the second cleavage of Notch receptor by the γ secretase protein complex, the Notch intracellular domain (NICD) translocates to the nucleus and initiates gene transcription (Nefedova and Gabrilovich 2008).
The Notch1 signaling pathway controls the development and differentiation of T cell progenitors (Ferrando 2009). Notch1 signaling pathway activation mutations are present in a majority of patients with T-cell acute lymphoblastic leukemia (T-ALL) and γ secretase inhibitors (GSIs) are a therapeutic modality of interest for patients with T-ALL (Ferrando 2009). Further, GSIs enhance the anti-leukemic effect in glucocorticoid-resistant T-ALL cell lines and this anti-leukemic effect translates in vivo by coadministration of the glucocorticoid, dexamethasone (Real et al. 2009; Samon et al. 2012). Unfortunately, inhibition of γ secretase in the gut of T-ALL patients causes dose limiting gastrointestinal goblet cell hyperplasia (GCH) because the intestinal tract epithelium also expresses Notch1 and Notch2 receptors (Ferrando 2009). Fortunately, concurrent intraperitoneal (ip) injection of the glucocorticoid dexamethasone in in vivo GCH models ameliorates the gastrointestinal GCH (Stewart 1990; Real et al. 2009; Samon et al. 2012). Thus, the ideas emerge for GSI and dexamethasone combination therapy for patients with T-ALL because of the synergistic anti-leukemic effects and mitigation of the dose limiting intestinal GCH (Stewart 1990; Real et al. 2009; Samon et al. 2012).
PF-03084014 is a selective reversible, noncompetitive GSI that potently inhibits Notch signaling by reducing NICD and Notch target genes Hairy and Enhancer-of-split (Hes-1) and cellular homolog myelomatosis proto-oncogene (cMyc) in both cells and tumors (Wei et al. 2010). Antitumor efficacy for PF-03084014 in Notch-driven human xenograft models occurs as a result of NICD modulation (Wei et al. 2010), and the antitumor activities likely occur through its direct effects on the cell cycle and apoptosis (Wei et al. 2010). Intermittent i.p. dosing of dexamethasone in a 2-week repeat oral dose xenograft study with PF-03084014 in athymic mice ameliorates GCH without loss of antitumor efficacy (Wei et al. 2010). In a recent report, PF-03084014 given orally to mice for 5 days diminishes the Ki67+ proliferative compartment of the ileum while cotreatment with i.p. dexamethasone after 5 days reverses GSI-induced GCH (Samon et al. 2012). Thus, PF-03084014 is currently in clinical development for the treatment of patients with T-ALL and advanced solid tumors (Paganin and Ferrando 2011) because it appears promising that cotreatment with dexamethasone mitigates the potential GCH in those patients (Wei et al. 2010). However, a preclinical safety study design mimicking the clinical route of PF-03084014 administration with oral dexamethasone administration would be essential to confirm mitigation of GCH.
The current preclinical study aimed to determine whether a relevant clinical route and dose of dexamethasone administration ameliorates GSI-induced GCH. This article reports a study in Sprague-Dawley (SD) rats after repeat oral dosing with a combination of dexamethasone and PF-03084014. The objectives of this work include characterization of systemic exposure of dexamethasone and PF-03084014 either alone or in combination therapy, characterization of safety end points following the oral administration of PF-03084014 alone, PF-03084014 and dexamethasone pretreatment or PF-03084014, and intermittent concurrent dexamethasone treatment.
Materials and Methods
A 7-day repeat dose toxicokinetic (TK) study and two 1-month combination repeat dose oral safety studies in rats are reported. The 7-day TK study was conducted to determine the maximum tolerated oral dose of dexamethasone to lessen test article–related GCH. The dose of PF-03084014 (150 mg/kg/day) selected in this study was based on the predicted therapeutic free plasma concentration in humans and a 4-fold higher dose tolerated in rats for 7 days. The two 1-month combination repeat dose oral safety studies were conducted to determine the optimal oral dexamethasone-dosing regimen to ameliorate PF-03084014-related GCH. The dose of PF-03084014 (100 mg/kg/day) selected in the 1-month studies was lowered in order to improve the clinical tolerability for 30 days of repeat oral dosing and ensure systemic exposure adequately covered the predicted therapeutic free plasma concentration in humans (∼37 nM).
Animals and Husbandry
In all studies, standard procedure and conditions were applied in a facility accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. All procedures involving laboratory animals were reviewed and approved by the Pfizer Inc. Institutional Animal Care and Use Committee associated with the facility. The 7-day TK study and two 1-month oral safety studies were conducted in 6- to 8-week-old male SD (Crl:CD®[SD]) rats that were supplied by Charles River Laboratories, Inc. (Portage, MI).
Drug
PF-03084014, [(S)-2-((S)-5,7-difluoro-1,2,3,4-tetrahydronaphthalen-3-ylamino)-N-(1-(2-methyl-1-(neopentylamino) propan-2-yl)-1H-imidazol-4-yl) pentanamide] (Wei et al. 2010), was supplied by Pfizer Global Research and Development, La Jolla Laboratories (San Diego, CA). All dose levels are represented as the free base equivalent mg/kg dose. PF-03084014 was formulated in 0.5% methylcellulose and given to study animals by oral gavage.
PF-03084014 was assayed using a validated LC-MS/MS method, and toxicokinetic calculations were performed using the noncompartmental approach with the aid of Watson Bioanalytical LIMS (v7.2.0.03, Thermo Electron Corp.)
Study Design
Three SD rat study designs are listed in Figure 1. In the 7-day repeat dose TK study, 3 male rats per group were orally dosed with either 0.5% methylcellulose vehicle, PF-03084014 at 150 mg/kg/day, PF-03084014 at 150 mg/kg/day coadministered with 1 of the oral dexamethasone doses (0.25, 1.0, 2.5, or 5.0 mg/kg/day) or dexamethasone at 5 mg/kg/day and euthanized at 24 hr after the last dosing. The blood collection time points for determining PF-03084014 or dexamethasone mean systemic plasma concentrations were 1, 2, 4, 7, and 24 hr post dosing. Test article–related findings were determined by assessing changes in clinical signs, pre- and post-dose body weights. Blood samples for assessing systemic exposure of PF- 03084014 and dexamethasone were collected from all treatment groups at various times on days 1 and 7 of the study. Blood samples for hematology evaluation were also collected from all the animals at 24 hr after the last dosing. The mean group changes in hematology parameters for treated rats were expressed as a percentage change using this formula [(mean day 8 treated − mean day 8 vehicle control)]/(mean day 8 vehicle control) × 100%] when the magnitude of the change was less than 100% or expressed as fold change using this formula (mean value day 8 treated)/(mean value day 8 vehicle control) when the magnitude of the change was greater than 100%. Necropsy was performed 24 hr after the last dose, body weights were recorded, and tissues were collected and submitted for histopathologic examinations.
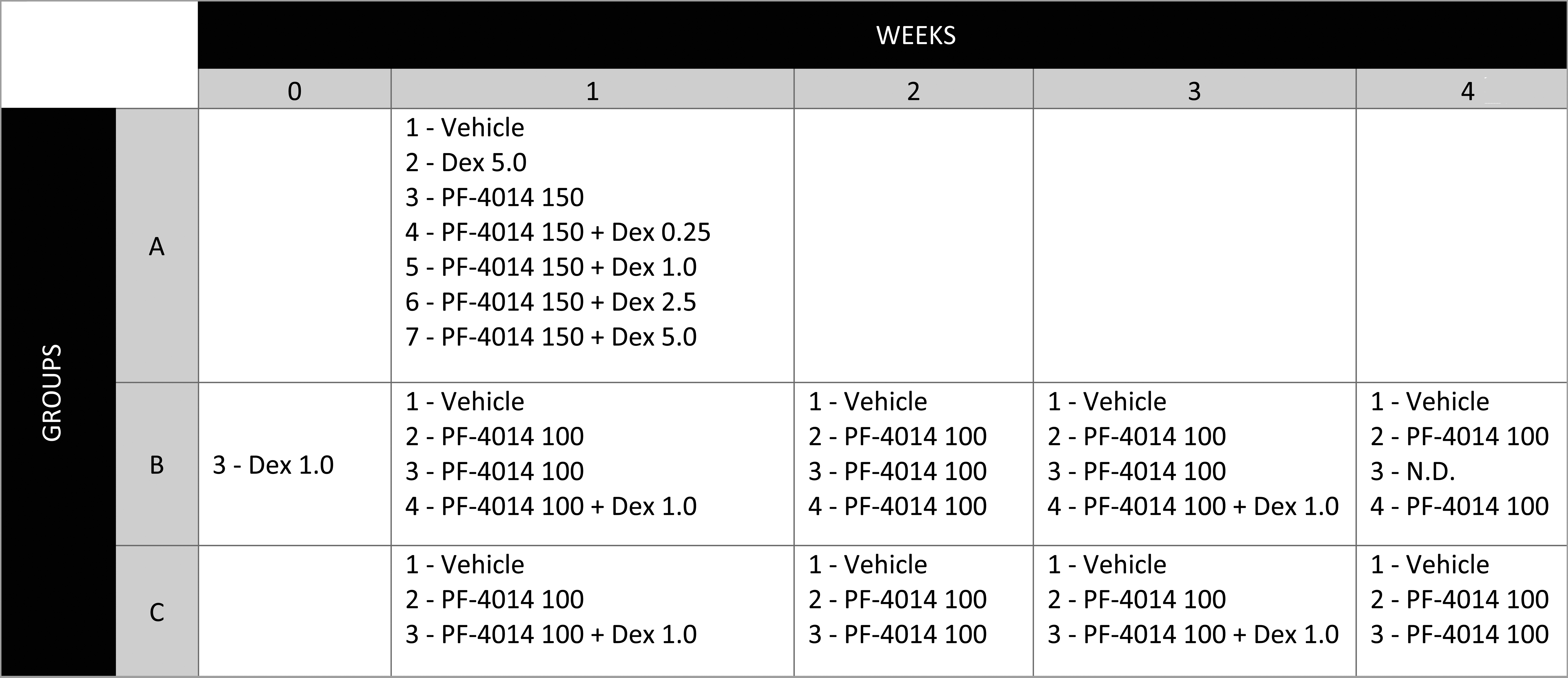
Study designs for the 7-day repeat dose toxicokinetic study and two 1-month repeat dose studies. (A) TK Study: Rats were placed into 7 groups of 3 and orally dosed for 7 days with either vehicle, 5.0 mg/kg/day dexamethasone, 150 mg/kg/day PF-03084014, or 150 mg/kg/day PF-03084014 in combination with one of the following doses of dexamethasone (0.25, 1.0, 2.5, or 5.0 mg/kg/day). (B) First 1-month study: Rats were placed into 4 groups of 3 and orally dosed with vehicle or 100 mg/kg/day PF-03084014 for 4 weeks or pretreated (week 0) with 1.0 mg/kg/day dexamethasone for 1 week followed by treatment with 100 mg/kg/day PF-03084014 on weeks 1, 2, and 3 or 100 mg/kg/day PF-03084014 in combination with 1.0 mg/kg/day dexamethasone on weeks 1 and 3 with a dexamethasone dosing holiday on weeks 2 and 4. (C) Second 1-month study: Rats were placed into 4 groups of 5 and orally dosed with vehicle or 100 mg/kg/day PF-03084014 for 4 weeks or 100 mg/kg/day PF-03084014 in combination with 1.0 mg/kg/day dexamethasone on weeks 1 and 3 with a dexamethasone-dosing holiday on weeks 2 and 4.
In the first 1-month combination repeat dose study, 12 rats per group were orally dosed with vehicle (0.5% methylcellulose), PF-03084014 at 100 mg/kg/day or a combination of PF-03084014 at 100 mg/kg/day and dexamethasone at 1 mg/kg/day. Rats treated with a combination of PF-03084014 and dexamethasone were pretreated with dexamethasone for 7 days and then given PF-03084014 at 100 mg/kg/day for 3 weeks or treated with PF-03084014 at 100 mg/kg/day for 4 weeks and dexamethasone was concurrently administered at 1 mg/kg/day during the first and third week of study. Three rats per group were euthanized at weekly intervals 24 hr after the last dose on days 8, 15, 22, and 28 of the study. Test article–related findings were determined by assessing changes in clinical signs, pre- and post-dose body weights. Blood samples for assessing systemic exposure of PF-03084014 and dexamethasone were collected from all treatment groups on days 8, 15, 22, and 29 at termination. Necropsy was performed at study termination, body weights were recorded, and tissues were collected and submitted for histopathologic examinations.
In the second 1-month combination repeat dose study, a larger sample size (20 rats per group) was orally dosed with vehicle (0.5% methylcellulose), PF-03084014 at 100 mg/kg/day or a combination of PF-03084014 at 100 mg/kg/day and dexamethasone at 1 mg/kg/day. Rats were given PF-03084014 for 4 weeks and dexamethasone was concurrently administered during the first and third week of study. Five rats per group were euthanized at weekly intervals at 24 hr after the last dose on days 8, 15, 22, and 29. Test article–related findings were determined by assessing changes in clinical signs, pre- and post-dose body weights. Blood samples for the assessment of systemic exposure to PF-03084014 and dexamethasone were collected from all treatment groups at 1, 2, 4, 7, and 24 hr on days 1, 7, 14, 21, and 28 at termination. Necropsy was conducted at study termination, body weights were recorded, and histopathologic examinations were performed.
The grading scheme for the histopathogical changes used in all studies ranged from minimal to severe and was based on the percentage of the tissue affected. A grade of minimal corresponded to ≤10%, mild corresponded to >10% to <25%, moderate corresponded with >25% to <50%, and severe corresponded to >50% of the tissue affected.
Statistical Analysis
Hematology data were analyzed using a one-way analysis of variance (ANOVA) model with Dunnett’s follow-up tests of each group versus the vehicle group. The analysis was performed either on the raw data (parametric) or on the rank-transformed data (nonparametric), depending on the historical distribution of each parameter. All tests were two-sided except for mean platelet volume (MPV) and large unstained cells (LUC), for which testing was for increases only. A significance threshold of 0.05 level was used. All data are reported as M ± SD.
For the 7-day TK study, GCH incidences were analyzed using the Cochran-Armitage test (Armitage 1955) to study the trend of incidence change with the increasing dose of dexamethasone when coadministrated with PF-03084014 (150 mg/kg/day). The control group was the group treated with PF-03084014 only. Due to the sparseness of the data, a dichotomous categorization of the GCH severity was adopted—“Abnormal” for any non-zero severity and “Normal” for zero severity.
For the 1-month studies (experiments 1 and 2), differences from the positive control group (PF-03084014 only group) were examined using Fisher’s exact test (Fisher 1922). The aforementioned dichotomous categorization of severity was adopted, except for the colon in the first week of Experiment 1 where the PF-03084014 only and dexamethasone-pretreated group exclusively had severity scores of 1 and 2, respectively. In this case, merging severity 1 and 2 would not allow one to distinguish the 2 treatment groups, and hence severity 1 and 2 were separated as were on the original scale.
All the categorical data statistical tests were 1-sided, and the significance level was set to 5% (i.e., p < .05). All the statistical analyses were carried out using SAS version 9.2 (Cary, NC).
Results
Tolerability
In the 7-day repeat dose TK study, treatment-related mortality occurred on day 7 in 1 rat receiving 150 mg/kg/day PF-03084014 in combination with 5 mg/kg/day dexamethasone. In the first 1-month combination repeat dose study, treatment-related morbidity occurred on day 14 in a rat that was given 100 mg/kg/day PF-03084014 concurrently with 1.0 mg/kg/day dexamethasone during the first week of study. Treatment-related morbidity included development of a swollen hind limb, and this clinical finding resulted in early termination of this animal on day 14 of study. Because there was a treatment-related mortality in 1 rat on day 23, the remaining animals in that treatment group were terminated on day 23 of study. In the second 1-month combination repeat dose study, 1 rat given vehicle or 100 mg/kg PF-03084014 was sacrificed on the first and second week, respectively, due to gavage error. An unscheduled sacrifice of 1 rat given 100 mg/kg PF-03084014 and 1.0 mg/kg dexamethasone occurred on day 13 due to moribund condition.
Hematology
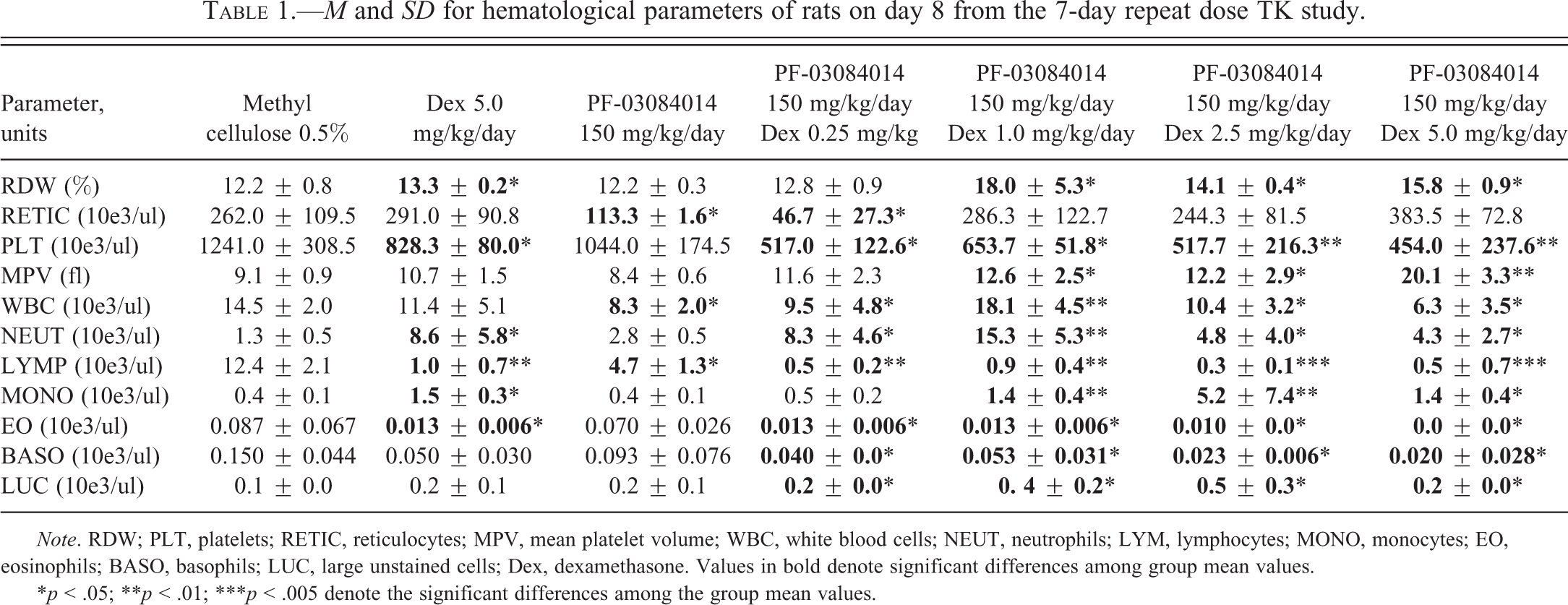
The M and SD of hematology parameters for the 7-day repeat dose TK study are shown in Table 1. In rats given 5 mg/kg dexamethasone alone, the red cell distribution width was increased by 8.5%, the total mean white blood cell (WBC) count was decreased (−21.4% control mean), whereas the mean total neutrophil and mean total monocyte counts were increased 6.7-fold and 4.1-fold control means, respectively. The mean total large unstained cell count was increased (35.7% control mean). Mean total lymphocyte, eosinophil, and basophil counts were decreased (−91.9%, −84.6%, and −66.7% control means, respectively). Thus, rats given 5 mg/kg/day dexamethasone alone resulted in red cell anisocytosis and in a steroid-induced leukogram that was characterized by neutrophilia, lymphopenia, monocytosis, and eosinopenia. In rats given 150 mg/kg/day PF-03084014 alone, the mean total WBC count, lymphocyte, and basophil counts were decreased (−42.6%, −61.6%, and −37.8% control means, respectively) and neutrophil count was increased (2.3× control mean). The mean total reticulocyte count was decreased (−56.7% control mean). Thus, PF-03084014 treatment appeared to have a significant effect on the total WBC count and lymphocyte count. In rats given the combination treatment of PF-03084014 and ≥0.25 mg/kg/day dexamethasone, the mean red blood cell distribution width was slightly increased (15.5–47.4% control mean) in rats given ≥1.0 mg/kg dexamethasone with PF-03084014, but a dose response was not apparent. Total platelet counts were decreased (−47.3% to −63.4% control mean) in rats given ≥0.25 mg/kg dexamethasone with PF-03084014, but a dose response was not apparent. MPV was increased (33.3% to 2.2-fold control mean) in rats given ≥1.0 mg/kg dexamethasone with PF-03084014. Mean WBC counts were decreased −28.3% control mean in rats given 0.25, 2.5, or 5.0 mg/kg dexamethasone with PF-03084014, but increased 25.4% in rats given 1.0 mg/kg dexamethasone with PF-03084014. Mean neutrophil counts were increased 3.3-fold to 12-fold; mean monocyte counts were increased 1.3-fold to 3.8-fold; and mean large unstained cell counts were increased 1.5-fold to 3.0-fold respective control means in rats given 0.25 to 5.0 mg/kg dexamethasone with PF-03084014. Total mean lymphocyte counts were decreased −92.7% to −97.9%; mean eosinophil counts were decreased −84.6% to −100%; and mean basophil counts were decreased −36.7% to −88.9% of respective control means in rats given 0.25 to 5.0 mg/kg dexamethasone with PF-03084014. Overall, combination treatment of PF-03084014 and dexamethasone mimicked a steroid-induced leukogram.
M and SD for hematological parameters of rats on day 8 from the 7-day repeat dose TK study.
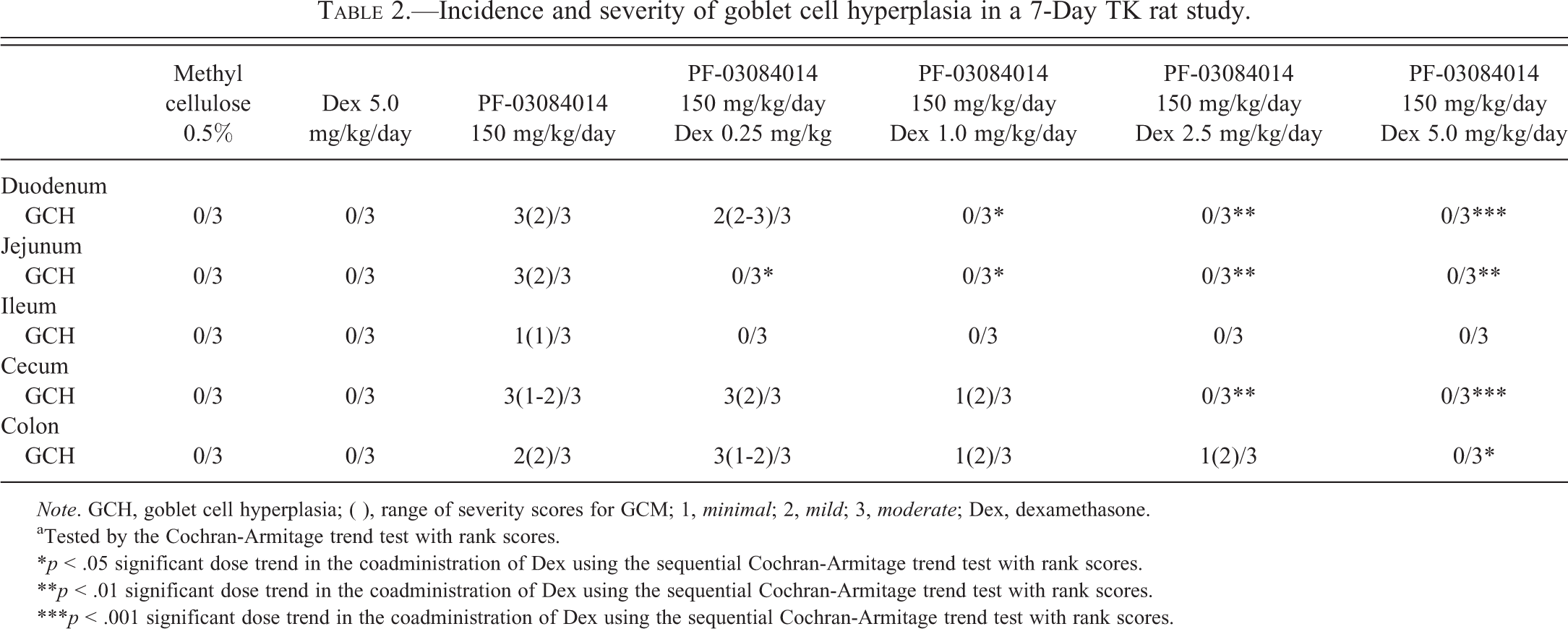
Note. RDW; PLT, platelets; RETIC, reticulocytes; MPV, mean platelet volume; WBC, white blood cells; NEUT, neutrophils; LYM, lymphocytes; MONO, monocytes; EO, eosinophils; BASO, basophils; LUC, large unstained cells; Dex, dexamethasone. Values in bold denote significant differences among group mean values.
*p < .05; **p < .01; ***p < .005 denote the significant differences among the group mean values.
Body Weights
In the 7-day repeat dose TK and first 1-month combination repeat dose studies, treatment with dexamethasone alone and dexamethasone with PF-03084014 caused moderate to marked body weight loss (−10% to −27%) after 7 days treatment. In the second 1-month combination repeat dose study, a similar magnitude of body weight loss (−10% to 22%) occurred with repeat dosing on the first week or third week of treatment with 100 mg/kg PF-03084014 and 1 mg/kg dexamethasone. When dexamethasone was not coadministered with PF-03084014 on the second week of study, increases (4%) in body weight were noted, suggesting that the body weight loss was reversible.
Gross and Histopathologic Findings
7-Day Repeat Dose TK and Range-finding Study with Dexamethasone
Gross findings in rats from the 7-day repeat dose TK study are listed in Supplementary Table 1. Spleens and thymuses were small in rats given 150 mg/kg PF-03084014, 5.0 mg/kg dexamethasone, or 150 mg/kg PF-03084014 in combination with ≥0.25 mg/kg dexamethasone. Abnormal liver discoloration was observed in rats given PF-03084014 in combination with ≥0.25 mg/kg dexamethasone. Abnormal stomach discoloration was observed in rats given PF-03084014 in combination with ≥1.0 mg/kg dexamethasone. Abnormal kidney discoloration was observed in rats given PF-03084014 in combination with 0.25 mg/kg dexamethasone or ≥2.5 mg/kg dexamethasone. A jejunal perforation was observed in 1 rat given 150 mg/kg PF-03084014 in combination with 5.0 mg/kg dexamethasone.
Histopathological findings in rats from the 7-day repeat dose TK study correlated with a majority of the gross findings and are in Supplementary Table 2. The small spleens and small thymuses in rats given PF-03084014, dexamethasone, or combination treatment correlated with a mild to marked decreased lymphocyte cellularity of those organs. In addition, there was a significantly decreased cellularity of gut-associated lymphoid tissues (GALT) in rats given PF-03084014 in combination with ≥0.25 mg/kg dexamethasone. The stomach discoloration in rats given PF-03084014 in combination with ≥0.25 mg/kg dexamethasone correlated with gastric erosions/ulceration, vascular thrombosis, and the presence of mucosal bacterial colonies. Mucosal glandular atrophy was also observed in the stomachs of rats given PF-03084014 in combination with ≥1.0 mg/kg dexamethasone. Minimal inflammation characterized by neutrophil infiltrates within the gastric serosal layer was observed in 1 rat given 150 mg/kg PF-03084014. Duodenal erosions/ulcerations, inflammation, vascular thrombosis, and glandular dilatation were observed in a rat given PF-03084014 in combination with 0.25 mg/kg. Bacterial colonies and glandular dilatation were observed in rats given either PF-03084014 or PF-03084014 in combination with 0.25 mg/kg or 5.0 mg/kg dexamethasone. Liver discoloration observed in rats given PF-03084014 in combination with ≥0.25 mg/kg dexamethasone correlated with hepatocyte vacuolation and areas of hepatic necrosis often accompanied by bacterial colonies. The abnormal kidney discoloration in rats given PF-03084014 and ≥2.5 mg/kg dexamethasone correlated with renal tubular necrosis and the presence of bacterial colonies.
The incidence and severity of GCH in rats from 7-day repeat dose TK study are listed in Table 2. GCH was observed in both the small and large intestines of rats given 150 mg/kg PF-03084014 and was most consistently observed in the duodenum and jejunum. GCH in the duodenum was significantly ameliorated in rats given PF-03084014 in combination with ≥1.0 mg/kg dexamethasone. In the jejunum, GCH was significantly ameliorated in rats given PF-03084014 in combination with ≥0.25 mg/kg dexamethasone. In the cecum and colon, GCH was ameliorated in rats given PF-03084014 in combination with ≥2.5 mg/kg dexamethasone or 5.0 mg/kg dexamethasone, respectively. Because 1.0 mg/kg dexamethasone mitigated the severity of GCH in the intestines when coadministered with PF-03084014, the oral dose of 1.0 mg/kg/day dexamethasone was selected for evaluating the dosing regimen in the 1-month rat studies.
Incidence and severity of goblet cell hyperplasia in a 7-Day TK rat study.
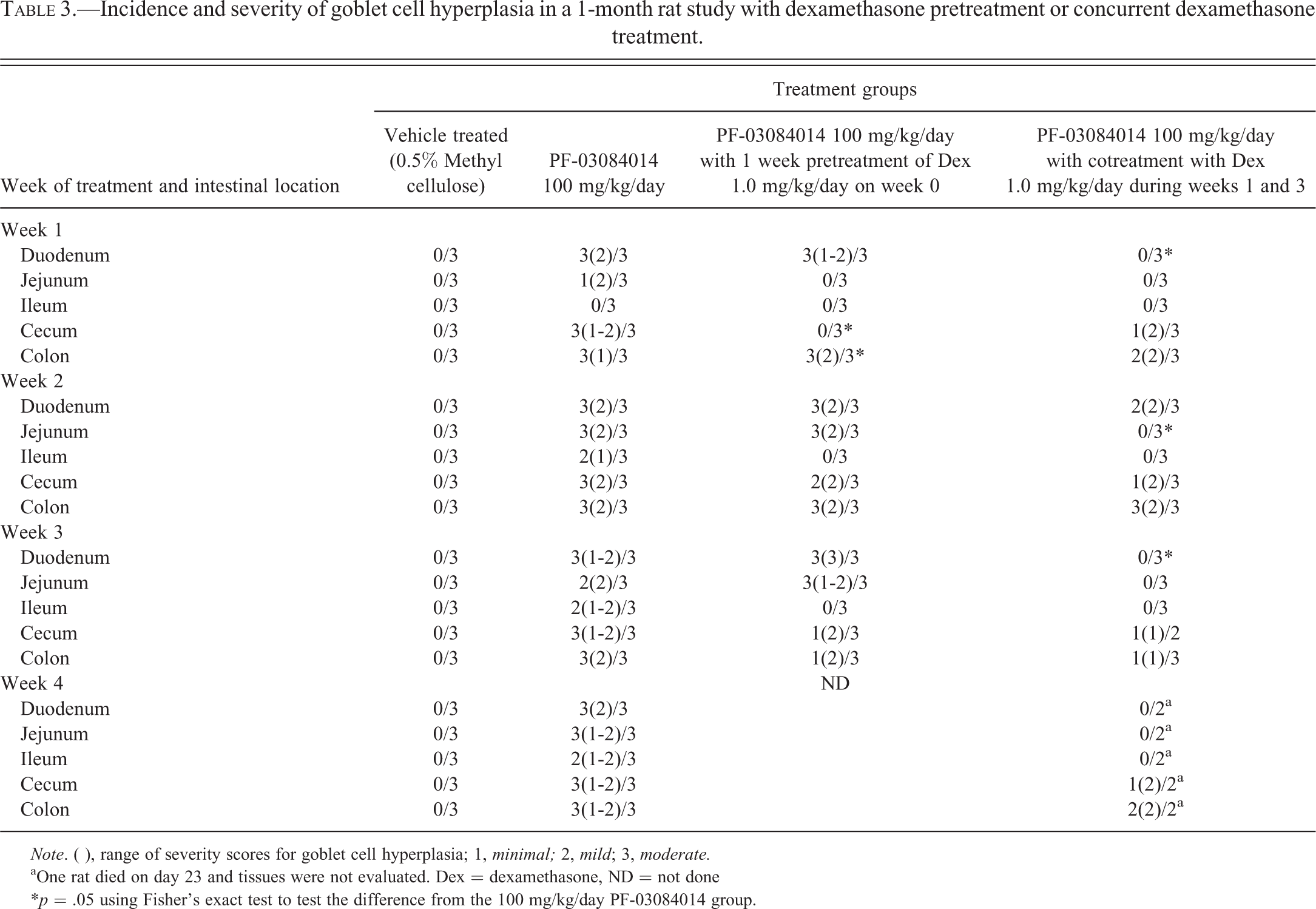
Note. GCH, goblet cell hyperplasia; ( ), range of severity scores for GCM; 1, minimal; 2, mild; 3, moderate; Dex, dexamethasone.
aTested by the Cochran-Armitage trend test with rank scores.
*p < .05 significant dose trend in the coadministration of Dex using the sequential Cochran-Armitage trend test with rank scores.
**p < .01 significant dose trend in the coadministration of Dex using the sequential Cochran-Armitage trend test with rank scores.
***p < .001 significant dose trend in the coadministration of Dex using the sequential Cochran-Armitage trend test with rank scores.
1-Month Combination Repeat Dose Safety Study with Dexamethasone Pretreatment or Dexamethasone Coadm-inistration
Gross and histopathologic findings from the 1-month repeat dose study with PF-03084014 and dexamethasone pretreatment or dexamethasone concurrent treatment are listed in Supplementary Table 3. Thymuses were small in most rats given 100 mg/kg PF-03084014 and pretreatment or concurrent treatment with 1.0 mg/kg dexamethasone, and this finding was significantly different from vehicle and PF-03084014 alone treatment groups. Spleens were small in 3 rats given dexamethasone concurrently and in a single rat given dexamethasone pretreatment. Tan foci in the liver and kidneys were observed in a rat given dexamethasone concurrently. Pale foci in the liver were observed in rats given PF-03084014 and ≤2.5 mg/kg dexamethasone. Histopathological findings correlated with a majority of the gross findings. The small thymuses and small spleens correlated with a mild to severe overall decreased lymphocyte cellularity in those organs. Areas of decreased cellularity included the thymic cortex and/or medulla, mantle layer, and follicular areas of the spleen and were significantly different in combination treatment groups for the thymus only. In addition, there was a significant mild decreased cellularity of GALT in most rats given PF-03084014 in combination with dexamethasone pretreatment or dexamethasone concurrent treatment. Tan foci in the liver and kidneys and the swollen hind limb of a rat given concurrent dexamethasone treatment correlated with the presence of abscesses in those tissues. Gastric glandular necrosis was observed in 1 to 4 rats given dexamethasone pretreatment or concurrent treatment, respectively, and was characterized by minimal to moderate single cell necrosis of mucosal cells. A significant mild jejunal villous atrophy characterized by blunting of the villi was observed in 4 rats given dexamethasone concurrently.
The incidence and severity of GCH in the intestinal tracts are listed in Table 3. After the first week of GSI treatment, coadministration of dexamethasone with PF-03084014 significantly mitigated GCH in the duodenum, and there was a trend of reduced the incidence of GCH in the jejunum, cecum, and colon when compared to GSI treatment alone. When rats were pretreated for 1 week with daily administration of dexamethasone at 1 mg/kg/day prior to treatment with PF-03084014, the GCH was significantly mitigated in the cecum and colon but not in the duodenum, jejunum, or ileum. Hence, pretreatment with dexamethasone had less of a protective effect on PF-03084014 mediated duodenal GCH than concurrent dexamethasone administration. Duodenal GCH was observed in the cotreatment group during week 2 when dexamethasone treatment was withheld and was significantly mitigated in the jejunum. However, coadministration of dexamethasone with PF-0308414 during week 3 significantly mitigated GSI-induced GCH in the duodenum only and produced a trend that suggested reduced GSI-induced GCH in the jejunum, ileum, and large intestines compared to rats given PF-03084014 alone. After the fourth week of treatment with PF-03084014 when dexamethasone treatment was withheld, GSI-induced GCH was mitigated in the small intestines only. GSI-induced GCH increased with time with PF-03084014 repeat dosing and when dexamethasone treatment was withheld. Therefore, the intermittent coadministration-dosing regimen was further assessed in a larger number of animals in the next 1-month rat study.
Incidence and severity of goblet cell hyperplasia in a 1-month rat study with dexamethasone pretreatment or concurrent dexamethasone treatment.
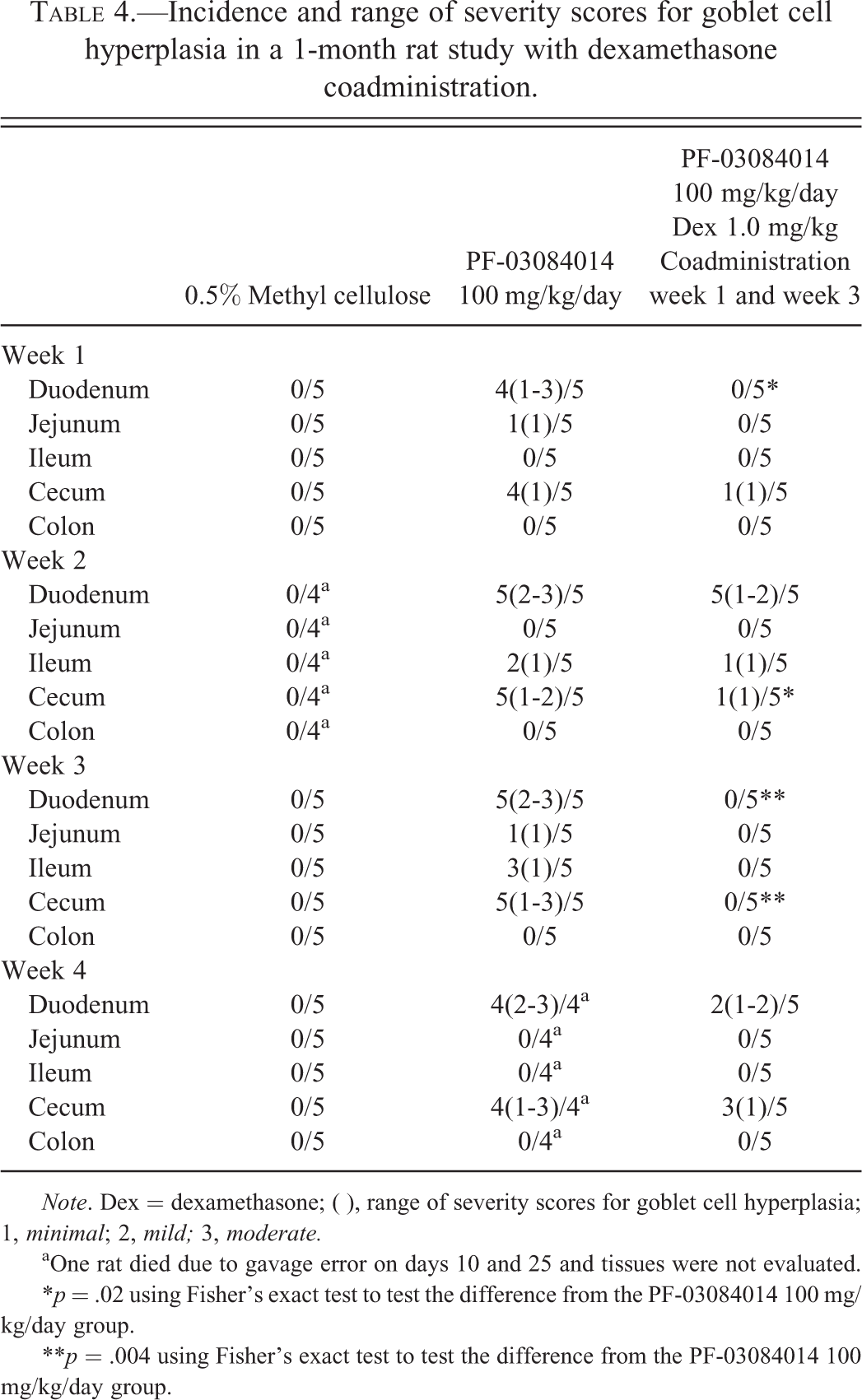
Note. ( ), range of severity scores for goblet cell hyperplasia; 1, minimal; 2, mild; 3, moderate.
aOne rat died on day 23 and tissues were not evaluated. Dex = dexamethasone, ND = not done
*p = .05 using Fisher’s exact test to test the difference from the 100 mg/kg/day PF-03084014 group.
1-Month Combination Repeat Dose Safety Study with Intermittent Coadministration of Dexamethasone
Gross and histopathologic findings from the 1-month repeat dose study with 100 mg/kg PF-03084014 or 100 mg/kg PF-03084014 coadministered with 1.0 mg/kg oral dexamethasone are listed in Supplementary Table 4. Small thymuses were observed in all rats given PF-03084014 with dexamethasone and in 6 rats given PF-03084014 alone. Small spleens were observed in 7 rats given combination treatment. The occurrence of the small thymuses in the combination treatment group was significantly different from rats given PF-03084014 treatment alone. The small thymuses and spleens correlated the histopathologic finding of mild to marked decreased lymphocyte cellularity in those organs. Tan foci in the liver and kidneys were observed in 2 rats given the combination treatment and correlated with the histopathologic findings of tissue abscess. The gross findings in 1 rat terminated on day 13 due to poor clinical condition included tan foci in the stomach, liver, kidneys, and mesenteric lymph node, and the tan foci correlated the histopathologic findings of abscesses/inflammation in those tissues. Mild decreased cellularity of GALT was observed in 16 rats given PF-03084014 alone and mild decreased cellularity occurred in 9 rats given combination treatment. The difference in severity for GALT hypocellularity was significant for the combination treatment group. The pale discoloration of the liver in 2 rats given combination treatment correlated with the histopathologic findings of marked hepatocyte vacuolation. Four rats given 100 mg/kg PF-03084014 had minimal to mild jejunal eosinophilic inflammation after second or third week but was absent by week 4.
The incidence and severity of GCH in the intestinal tracts of rats given 100 mg/kg PF-03084014 or 100 mg/kg PF-03084014 coadministered with 1.0 mg/kg oral dexamethasone are listed in Table 4. A significant amelioration of GCH was observed in the duodenum during the first week, in the cecum during the second week, and in the duodenum and cecum during third week of Study in rats given concurrent dexamethasone treatment on weeks 1 and 3. There was only a trend suggestive of decreased incidence of GCH in the duodenum and cecum during week 4 because of the small sample size due to an unscheduled death of 1 animal during week 4 given 100 mg/kg PF-03084014 alone. In summary, intermittent concurrent administration of dexamethasone with repeat dosing of PF-03084024 reduced the incidence and/or severity of intestinal GCH for up to 4 weeks of treatment.
Incidence and range of severity scores for goblet cell hyperplasia in a 1-month rat study with dexamethasone coadministration.
Note. Dex = dexamethasone; ( ), range of severity scores for goblet cell hyperplasia; 1, minimal; 2, mild; 3, moderate.
aOne rat died due to gavage error on days 10 and 25 and tissues were not evaluated.
*p = .02 using Fisher’s exact test to test the difference from the PF-03084014 100 mg/kg/day group.
**p = .004 using Fisher’s exact test to test the difference from the PF-03084014 100 mg/kg/day group.
Representative histopathologic features of duodenal GCH are illustrated in Figure 2. After week 1 of treatment with PF-03084014, the numbers of goblet cells in the duodenum were moderately increased (Figure 2C) while numbers of goblet cells were decreased to background or vehicle control levels (Figure 2A and B) with concurrent administration of dexamethasone and PF-03084014 (Figure 2D). At the end of the second week, the numbers of goblet cells in the duodenum were moderately increased with PF-03084014 treatment alone (Figure 2E) compared to the mildly increased numbers of goblet cells observed in rats at the end of the second week following the 1-week dexamethasone dosing holiday (Figure 2F). After 3 weeks of treatment with PF-03084014 alone, the numbers of goblet cells in the duodenum remained moderately increased (Figure 2G) while the numbers of goblet cells in rats were decreased to background levels at the end of the third week with coadministration of dexamethasone and PF-03084014 (Figure 2H). After the fourth week of treatment, the numbers of goblet cells in the duodenum were remained moderately increased in rats with PF-03084014 treatment alone (Figure 2I) compared to the mildly increased numbers of goblet cells observed in rats at the end of the fourth week with PF-03084014 following the 1-week dexamethasone dosing holiday during the third week of study (Figure 2J).
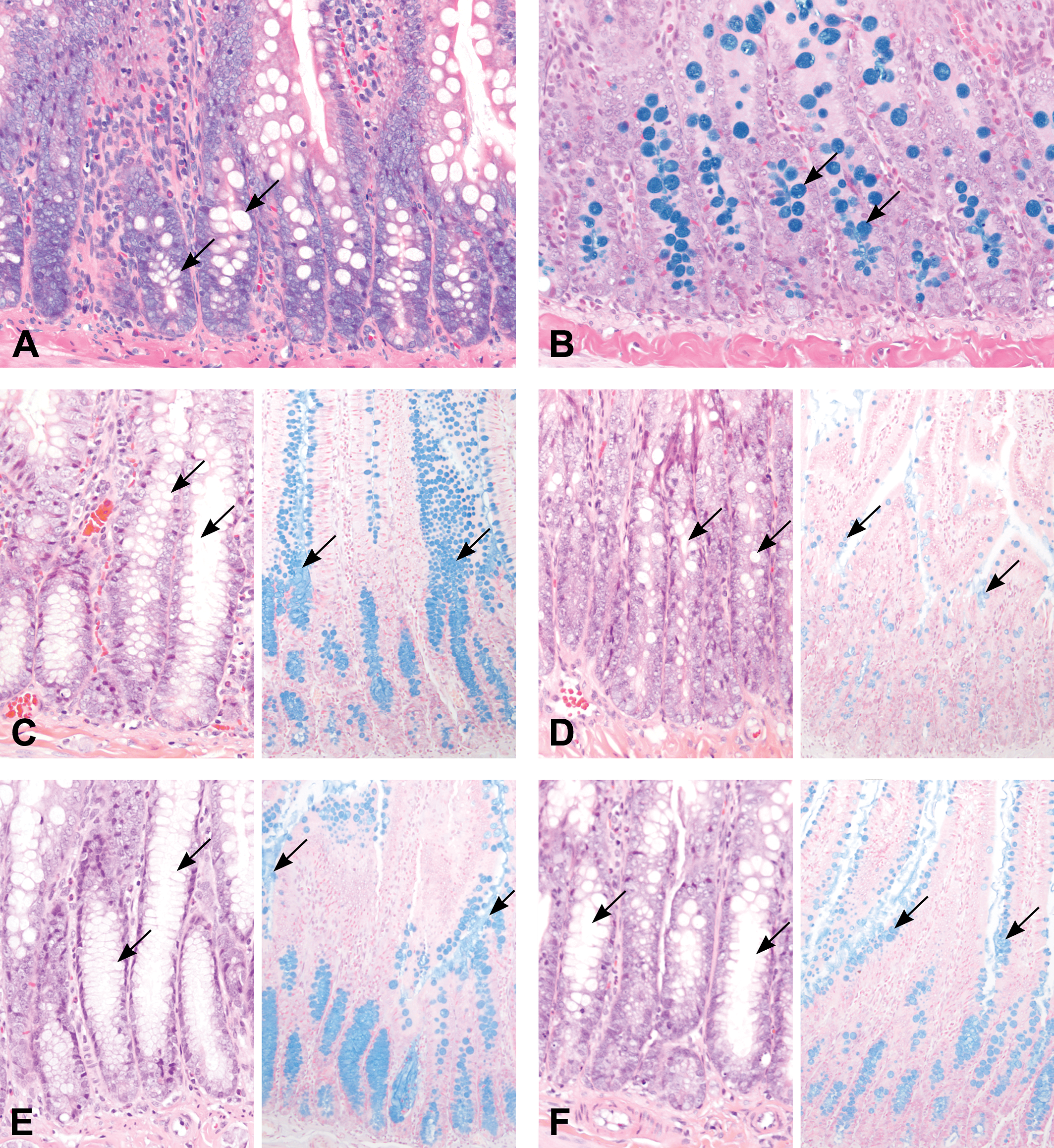
Microscopic findings of Hematoxylin and Eosin (H&E)- and Alcian blue (AB)-stained sections of rat duodenal tissue. Duodenum from a rat 4 weeks after treatment with the vehicle stained with H&E (A) or AB (B). (C) Duodenum from a rat 1 week after treatment with 100 mg/kg/day PF-03084014. Note the moderately increased numbers of goblet cells in the crypts or mild goblet hyperplasia (arrows) on the H&E and AB panels. (D) Duodenum from a rat 1 week after concurrent administration of 100 mg/kg/day PF-0308414 and 1 mg/kg/day dexamethasone. Note the number of goblet cells (arrows) was decreased and similar to vehicle treated controls (A and B). (E) Duodenum from a rat 2 weeks after treatment with 100 mg/kg/day PF-03084014. Note the moderate goblet hyperplasia (arrows). (F) Duodenum from a rat 2 weeks after treatment with 100 mg/kg/day PF-03084014 and a 1-week dexamethasone-dosing holiday. Note the mildly decreased numbers of goblet cells (arrows) compared to (E). (G) Duodenum from a rat 3 weeks after treatment with 100 mg/kg/day PF-03084014. Note the moderate goblet hyperplasia (arrows). (H) Duodenum from a rat 3 weeks after treatment with 100 mg/kg/day PF-03084014 and concurrent administration of Dexamethasone during the third week of Study. Note the numbers of goblet cells (arrows) are similar to numbers of goblet cells in the control (A and B). (I) Duodenum from a rat 4 weeks after treatment with 100 mg/kg/day PF-03084014. Note the moderate goblet hyperplasia (arrows). (J) Duodenum from a rat 4 weeks after treatment with 100mg/kg/day PF-03084014 and a 1-week dexamethasone-dosing holiday. Note the numbers of goblet cells (arrows) are similar to the numbers of goblet cells in (F).
Toxicokinetics
In the 7-day repeat dose TK study, the systemic exposure, as defined by area under the curve (AUC), and maximal concentration (Cmax) of PF-03084014 in the 7-day TK study following daily oral administration of 150 mg/kg ranged from 13 nM to 21 nM on day 1 and 46 nM to 164 nM on day 7 of treatment and the Tmax ranged from 1 to 7 hr post administration (Supplemental Table 5-1 and Figure 3A). Dexamethasone increased with increasing dose where the time of maximum plasma concentration (Tmax) ranged from 1 to 4 hr post the first day of dosing (Supplemental Table 5-2 and Figure 3B). The average free plasma concentration of dexamethasone observed 13 ± 2 nM, 65 ± 2 nM, 168 ± 13 nM, 315 ± 36 nM for 0.25, 1, 2.5, and 5 mg/kg, respectively, on day 1 and 17 ± 4 nM, 70 ± 22 nM, 127 ± 13 nM, and 130 ± 85 nM for 0.25, 1, 2.5, and 5 mg/kg, respectively, on day 7. Additionally, dexamethasone mean free systemic exposure at 5 mg/kg was similar with and without coadministration of a 150 mg/kg oral dose of PF-03084014 (315 nM compared to 257 nM on day 1 and 130 nM compared to 169 nM on day 7, respectively).
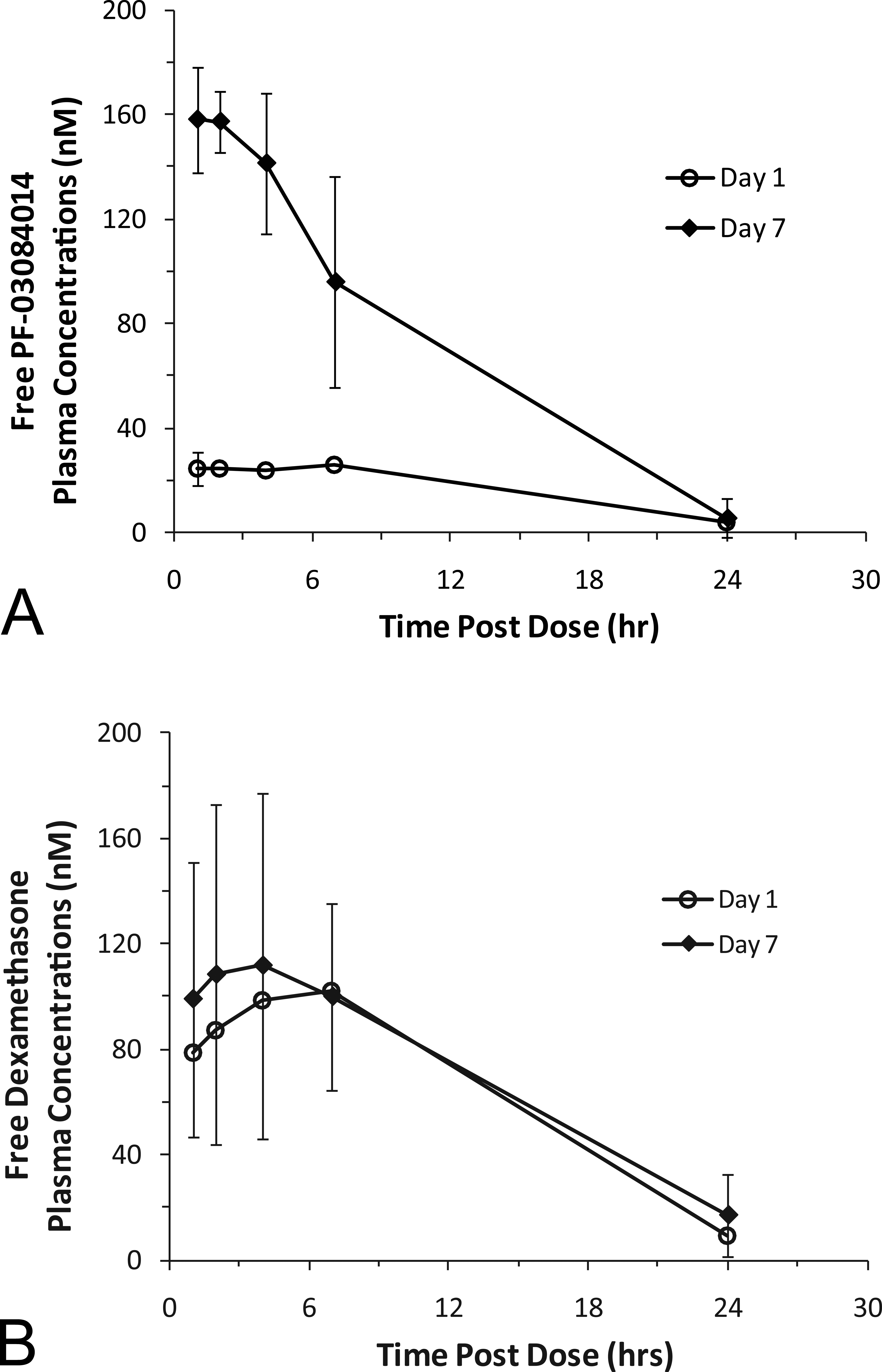
Mean free systemic plasma concentrations with SD for (A) PF-03084014 after oral daily dosing with 150 mg/kg for 1 week and (B) dexamethasone after oral daily dosing with 1 mg/kg for 1 week. The systemic free plasma concentration was determined at 1, 2, 4, 7, and 24 hr after the first dose on day 1 (
In the 1-month studies, the mean average free plasma concentration ± SD of PF-03084014 following daily oral administration of 100 mg/kg was 16 ± 2 nM on day 1, 40 ± 11 nM on day 7, 23 ± 12 nM on day 14, 69 ± 41 nM on day 21, and 17 ± 9 nM on day 28 (Supplementary Table 6-1). The Tmax for PF-03084014 in the 1-month studies ranged from 1 to 4 hr. The mean average free plasma concentration ± SD of dexamethasone following daily oral administration of 1 mg/kg when given concurrently with PF-03084014 on weeks 1 and 3 was 72 ± 8 nM on day 1, 71 ± 18 nM on day 7 and 62 ± 22 nM on day 21 (Supplementary Table 6-2). The Tmax for dexamethasone ranged from 1 to 10 hr. No detectable levels were observed during weeks when no dexamethasone was administered which was consistent with the exposure profile and low levels of dexamethasone observed at 24 hr post dose (Figure 3B).
Discussion
The morphologic diagnosis of increased numbers of goblet cells in the small intestine is divisive. The marked increase in the number of small intestinal goblet cells observed in rodents has been reported in the literature as both goblet cell metaplasia (Milano et al. 2004; Paganin and Ferrando 2011; Real and Ferrando 2009; Samon et al. 2012; Wong et al. 2004; Yang et al. 2001) and GCH (Wei et al. 2010; Zheng et al., 2009), which has led to controversy on the diagnosis for this change. The terms hyperplasia and metaplasia are descriptive terms used in pathology and are not assigned to a finding based on any known mechanistic, molecular or metabolic etiology, as different etiologies can lead to a metaplastic or hyperplastic change. Definitions of hyperplasia and metaplasia were adopted from leading medical dictionaries (Dorland 2008; Stedman 1990) and pathology texts (Jubb, Kennedy, and Palmer 2007; Slauson and Cooper 2002; Jones, Hunt, and King 1997) in assigning the morphologic diagnosis for increased numbers of goblet cells in the small intestine. Hyperplasia is defined as an increase in the number of cells in that tissue or organ. Metaplasia is defined as the abnormal transformation of an adult, fully differentiated tissue of one kind into a differentiated tissue of another kind that is not normal to that tissue or organ. Some of the most frequently noted examples of metaplastic change are the substitution of columnar or cuboidal epithelium by stratified squamous epithelium, the formation of cartilage or bone in connective tissue, and the formation of bony spicules in thickened alveolar septa of the lungs (Jubb, Kennedy, and Palmer 2007; Slauson and Cooper 2002; Jones, Hunt, and King 1997). Given these definitions for the descriptive terms and the fact that the secretory (goblet) cells and absorptive cells are normal components in the small intestinal epithelium, and both cell types belong in the normal small intestine, we used the morphologic diagnosis “goblet cell hyperplasia” to accurately label the GSI-induced finding of increased goblet cells in the small intestines of rodents.
Because dexamethasone reduced PF-03084014-induced GCH when given at a high dose of 15 mg/kg/day by the i.p. route for up to 2 weeks in mice (Wei et al. 2010), we investigated whether a clinically relevant lower oral dose of 1 mg/kg of dexamethasone could reduce PF-03084014-induced GCH in rats for up to 4 weeks. Importantly, after 2 weeks of PF-03084014 treatment in mice, anti-tumor efficacy of PF-03084014 was not lost and PF-03084014-induced GCH was reduced when a high i.p. dose of dexamethasone was coadministered for 14 consecutive days, or when dexamethasone was administered for the first 7 days plus a 7 day dosing holiday or when dexamethasone was administered only on the last 7 days of a 2-week study (Wei et al. 2010). We found that intermittent oral coadministration of 1.0 mg/kg/day dexamethasone with PF-03084014 at 100 mg/kg mitigates GSI-induced GCH in the intestinal tract for up to 4 weeks post treatment. With the exception of 1 rat that was terminated on day 13 in poor clinical condition due to the presence of abscesses in multiple tissues, this dosing regimen was well tolerated in a majority of the rats for 4 weeks. The predominant side effect associated with oral intermittent dexamethasone treatment included mild to marked decreased cellularity of lymphoid tissues. The oral dexamethasone pretreatment dosing regimen was ineffective to ameliorate GSI-induced GCH for 4 weeks. Administration of 100 mg/kg PF-03084014 alone in rats was well tolerated for 4 weeks despite the minimal to mild decreased cellularity of lymphoid tissues and exceeded the predicted therapeutic free plasma concentration in humans by a 1.9-fold margin. GSI-induced GCH predominated in the small intestines compared to the large intestines and across studies reported here, GSI-induced GCH was most pronounced in the duodenum.
The success of the intermittent repeat oral dosing regimen with dexamethasone may be explained in part by the normal goblet cell turnover time in the intestines and systemic free concentrations of dexamethasone. Goblet cells differentiate from stem cells within the intestinal crypts and mature within 2 to 3 days as they migrate toward villus and then are sloughed off into the intestinal lumen. Inhibition of Notch signaling by a GSI did not affect stem cell populations in the gut but was reported to shift the balance of cells to decrease the number of absorptive enterocytes and increase the number of mucous secreting cells (Real et al. 2009; Yang et al. 2001). Reported mechanisms suggest that dexamethasone and GSI modulate intestinal goblet cells through the induction of positive and negative cell cycle regulators (Real et al. 2009). GSI treatment was shown to induce the negative cell cycle regulator, Krüppel-like zinc finger transcription factor, Klf4, within goblet cells leading to goblet cell cycle arrest and accumulation in the intestines (Real et al. 2009). In contrast, dexamethasone treatment induced the positive cell cycle regulator cyclin D2 (Ccnd2) within goblet cells and ameliorated the development of GSI-induced GCH (Real and Ferrando 2009; Real et al. 2009). These cell cycle regulatory mechanisms could explain the remarkable decrease in the incidence and severity of GCH in small and large intestines observed on weeks 1 and 3 post treatment with concurrent dexamethasone administration. Thus, after a week of a dexamethasone-dosing holiday, the protective effects of dexamethasone waned while GSI expanded the goblet cell population and resulted in the recurrence of a mild GSI-induced GCH observed on weeks 2 and 4. Because GSI-induced GCH in rats was less severe after the dexamethasone-dosing holiday compared to the GSI treatment alone, an intermittent oral dosing regimen with dexamethasone lessened the untoward GSI-induced GCH in the gut for up to 4 weeks.
GSI-induced GCH was more predominant in the small intestines compared to the large intestines. The predominant epithelial populations in the small intestines are the absorptive enterocytes followed by goblet cells and within the large intestines, goblet cells predominate over the absorptive epithelial cells (Cheng and Leblond 1974; Young and Heath 2000) and may in part explain why GCH was easily detected in the small intestines compared to the large intestines. In addition, the expression of glucocorticoid receptors within the gut mucosa varies in different regions with the highest level of dexamethasone receptor expression located within the duodenum (Pressley and Funder 1975). Thus, the greatest therapeutic effect of dexamethasone against GSI-induced GCH was readily detectable in the duodenum and may be related to the increased expression of dexamethasone receptors in the duodenum.
Combination treatment with PF-03084014 and dexamethasone had suppressive effects on the immune system. The concurrent intermittent dexamethasone-dosing regimen was more immune suppressive compared to the dexamethasone pretreatment or PF-03084014 treatment alone. In general across studies, the severity of decreased cellularity of the thymus, spleen, and GALT was greater in rats cotreated with PF-03084014 and dexamethasone. Leukogram findings from the 7-day TK confirmed the dexamethasone steroid-induced effects of neutrophilia, lymphopenia, monocytosis, eosinopenia, and basophilopenia (Stockham and Scott 2002) when dexamethasone was coadministered with PF-03084014. Treatment-related morbidity and mortality with dexamethasone and GSI coadministration resulted in unscheduled animal terminations. Moribund rats developed organized abscesses in the liver, kidneys, mesenteric lymph node, and hind limb joints. The presence of tissue abscesses in some rats was attributed to the immunosuppressive and gastric side effects associated with dexamethasone (Yokota et al. 2007; Crabtree et al. 1979). The concurrent administration of dexamethasone along with PF-03084014 treatment led to an increased susceptibility to bacterial infections. The origin of the bacterial infection was likely from the gastrointestinal tract where erosions and ulcerations were observed.
The 1-week dexamethasone pretreatment dosing regimen was insufficient to ameliorate GSI-induced GCH in the small and large intestines for up to 3 weeks when compared to rats given the intermittent dexamethasone-dosing regimen. It is considered likely that the concentration of free plasma dexamethasone at 24 hr after the last dose on day 7 during the pretreatment period (Figure 2) was insufficient to mitigate GSI-induced GCH. After ≥7 days of GSI treatment, GSI-induced GCH in the duodenum was similar to GSI-induced GCH in rats given GSI treatment alone. GSI-induced GCH was unabated on the second and third week of dosing with PF-03084014 because dexamethasone was not given and the plasma dexamethasone concentrations were below of the level of detection.
PF-03084014 treatment alone was associated with hematologic and histopathologic findings that were similar to other Notch inhibitors (Wong et al. 2004; Milano et al. 2004). GSI-induced GCH was predominant in the small intestines, and this observation can be explained by the reported shift in cell populations from absorptive epithelium to a secretory epithelium (Real et al. 2009). The inability to detect minimal GSI-induced GCH consistently in the large intestines may be explained by the predominance of goblet cell population normally present in the cecum and colon (Cheng and Leblond 1974). In general, treatment with PF-03084014 was associated with decreased numbers of lymphocytes within the marginal zone of the spleen, follicles of GALT, and the cortex of the thymus, and these histopathologic findings correlated with a leukopenia and lymphocytopenia in peripheral blood. A minimal to mild eosinophilic inflammatory response was observed in the duodenum and jejunum of rats during the first 3 weeks of treatment with 100 mg/kg/day PF-03084014. This finding correlated with the hematology findings of decreased eosinophils observed in the 7-day rat TK study. The vast majority of tissue eosinophils normally reside in the gastrointestinal tract (Bochner and Schleimer 2001). The eosinophilic accumulation within the small intestines in some PF-03084014 treated rats was above the background level of intestinal eosinophils observed in control rats. This finding was likely a direct pharmacologic effect of γ secretase inhibition because human eosinophils in vitro were reported to express Notch receptors (Radke et al. 2009). Eosinophils that had been treated with GSIs were protected from apoptosis and exhibited enhanced chemokinesis when stimulated with granulocyte monocyte colony stimulating factor (GM-CSF; Radke et al. 2009) and had enhanced maturation (Kang et al. 2005). The reasons for absence of this finding in rats after a 4-week PF-03084014 treatment and why all rats were not affected remained unclear. However, presence of this finding in some rats suggested that intestinal eosinophilic inflammation represented another potential reversible adverse effect of γ secretase inhibition.
In summary, this article described the amelioration of dose limiting gastrointestinal effects associated with repeat oral dosing of a GSI by concurrent intermittent dexamethasone administration and the other findings associated with the administration of the GSI or dexamethasone alone. Importantly, we elucidated a potential strategy for the dose and route of administration for the 2 drugs that could be translated in the clinic and allow for a continuous dosing of a chemotherapeutic agent (PF-03084014) in the event of a dose limiting GCH should develop.
Footnotes
Acknowledgments
Special thanks to individuals who directly contributed to this work: Dingzhou Li for statistical analysis, Winston Evering for helpful discussions, 32 Pathologists who responded to the morphologic diagnosis survey, Carlos Esparanza, Jessica Frey, and Anthony Wong for histology assistance, Michelle Sands and Minerva Batugo for the bioanalysis of compound levels in the samples, and Constance Benedict for graphic arts assistance.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
