Abstract
Oral gavage studies with β-myrcene in male F344 rats showed a complex renal pathology comprising both alpha2u-globulin (α2u-g) nephropathy, an unusual nephrosis involving the outer stripe of outer medulla (OSOM), and an increased incidence of renal tubule tumors by 2 years. In the 90-day and 2-year studies, respectively, α2u-g nephropathy and linear papillary mineralization were observed in males at the two lower doses but were absent from the high dose. Nephrosis was characterized by dilation of the S3 tubules, nuclear enlargement (including karyomegaly), and luminal pyknotic cells, all in the outermost OSOM. Nephrosis was minimal at the higher doses in the 90-day study, but progressed to a severe grade in males dosed with 1,000 mg/kg for 2 years. Renal tubule tumors developed in treated groups with incidences up to 30% in the 250 and 500 mg/kg male dose groups. Tumors at the lower doses in males may have been associated with α2u-g nephropathy, while those at higher doses in both sexes may have been due to the nephrosis. Because β-myrcene induced a complex spectrum of renal pathology, the α2u-g nephropathy mechanism cannot be the sole mechanism of carcinogenesis in these rats.
Introduction
β-Myrcene is an acyclic unsubstituted monoterpene occurring naturally in a large variety of botanical species, but as a major constituent in hop and bay oils and in lemon grass oil (Madyastha and Srivatsan 1987; Guenther, Zimmerman, and Wildermuth 1994). It is also produced commercially in high volume for use in essential oils as intermediates in the production of terpene alcohols. β-Myrcene is therefore widely used as a flavoring additive in the production of food and soft beverages, in alcoholic beverages, and in the manufacture of cosmetics, soaps, and detergents (Madyastha and Srivatsan 1987; Lorente et al. 1989; Delgado et al. 1993; Kuney 1994). Lemon grass, also known as citronella, is a popular ingredient in Asian cooking and is used in some insect repellants.
Because of its high volume of commercial production and high level of human exposure through oral, inhalation, and dermal routes, the National Toxicology Program (NTP), National Institutes of Environmental Health Sciences, National Institutes of Health conducted a series of toxicity tests with β-myrcene in rats and mice, including 2-year carcinogenicity studies (NTP 2010). An additional prompt for NTP selection was the close structural relationship of β-myrcene with d-limonene, a chemical believed to produce renal tubule tumors through an alpha2u-globulin (α2u-g) mode of action (NTP 1990). Based on increased incidences of renal tubule adenomas in the 2-year carcinogenicity bioassay, the NTP concluded that there was clear evidence of carcinogenic activity of β-myrcene in male F344/N rats, and equivocal evidence of carcinogenic activity in female rats. The work presented here describes the unusual, dose-dependent nature of the renal pathology induced in rats by this chemical, and attempts to trace the pathogenesis of the lesions through time, and their relationship to the resultant renal tubule tumors.
Material and Method
Chemical
β-Myrcene was obtained from Millennium Specialty Chemicals (Jacksonville, FL) in 2 lots, one each for the 90-day and 2-year studies. Identity and purity determinations were conducted at Battelle Columbus Operations (Columbus, OH), and Galbraith Laboratories, Inc (Knoxville, TN). The purity of each lot was determined by elemental analyses, gas chromatography with flame ionization detection (GC-FID), and high performance liquid chromatography. The overall purity of the lot used for the 90-day study was determined to be greater than 90%, and that for the 2-year study was greater than 93%. The bulk chemical was stored in amber glass bottles sealed with Teflon®-lined lids at −20°C or less. Stability was monitored during the 90-day and 2-year studies using GC-FID with no detection of any degradation of the chemical.
Experimental Animals and Housing Conditions
The studies were conducted by Battelle Columbus Operations (Columbus, OH) in facilities accredited by the American Association for the Accreditation of Laboratory Animal Care (Rockville, MD) and in accordance with U.S. Public Health Service Policy on Humane Care and Use of Laboratory Animals, the National Academies of Sciences Guide for the Care and Use of Laboratory Animals, and the Battelle Institutional Animal Care and Use Committee. Male and female F344/N rats were obtained at 5 to 6 weeks of age from Taconic Farms Inc. (Germantown, NY) and were quarantined on arrival at Battelle in environmentally controlled rooms for 11 (males) or 12 (females) days. The environment consisted of a temperature of 72° ± 3°F, relative humidity of 50% ± 15%, and a 12-hr light/dark cycle. Feed (irradiated NTP-2000 wafer feed, Zeigler Brothers, Inc., Gardners, PA) and water were available ad libitum. All rats were housed 5 per cage.
90-day Toxicity Study
Groups of 10 male and 10 female F344/N rats were administered β-myrcene in corn oil by gavage at dose levels of 0, 250, 500, 1,000, 2,000, and 4,000 mg/kg body weight, 5 days per week for 14 weeks. These dose levels were selected based on published lethal dose data. All rats in the 4,000 mg/kg group died or were sacrificed moribund during the first week except one male that died on day 11. The 90-day toxicity study included a special segment (as a scheduled sacrifice required by the study protocol) in which 10 male and 10 female rats were administered each of the above doses for 23 days. As in the core study, all rats in the 4,000 mg/kg group died by the end of the first week.
Complete necropsies and microscopic examinations were performed on all rats. All major tissues were fixed and preserved in 10% neutral buffered formalin, processed and trimmed, embedded in paraffin, sectioned to a thickness of 4 to 6 µm, and stained with hematoxylin and eosin (H&E) and/or Mallory–Heidenhain (MH) for microscopic examination. Additionally, H&E-stained kidney sections were examined under ultraviolet illumination at a wavelength of 45 to 49 nm in order to observe autofluorescence of the lysosome pattern of proximal tubules by one of the authors (G.C.H.).
Immunohistochemical staining of α2u-g was performed on male and female rat kidney sections using a monoclonal anti-rat α2u-globulin antibody (R&D Systems Inc., Minneapolis, MN). The secondary antibody was anti-mouse IgG and avidin–biotin peroxidase was used as the method of detection of the antigen–antibody complex (Vectastain Elite ABC Kit, Vector Laboratories, Burlingame, CA).
2-year Carcinogenicity Study
Groups of 50 male and 50 female F344/N rats were administered β-myrcene in corn oil by gavage at doses of 0, 250, 500, and 1,000 mg/kg body weight, 5 days per week for 105 weeks (2 years). Survival of male rats in the 250 and 500 mg/kg dose groups, and all dosed groups of females, was similar to the respective vehicle controls; but all males at the 1,000 mg/kg dose had died by week 89 (20.5 months), with an average survival of 467 days (15.4 months) for the group. Nephrosis was considered to be the cause of death in all cases.
As for the 90-day study material, all major tissues (including grossly observable lesions and right and left kidneys) were fixed in 10% neutral buffered formalin and embedded in paraffin, and sections were stained with H&E. For standard or routine histological examination of kidneys, one section was prepared from each of the right and left organs. For extended evaluation of proliferative renal lesions, additional sections of both kidneys from the residual formalin-fixed wet tissues were embedded in separate paraffin blocks and step sectioned at 1-mm intervals to produce up to 8 additional sections of each male and female kidney.
Grade of Lesion Severity
Each lesion entity was graded on a scale of 0 to 4, with 0 representing absence of the lesion; 1, minimal; 2, mild; 3, moderate; and 4, marked grades of severity. For most lesions, the severity grades represented an increasing percentage of the kidney or kidney zone involved. For hyaline droplet nephropathy, which was confined to the cortex, the severity grade was based on a combination of increasing staining intensity and disruption of the normal pattern of droplets, as has been described previously (Hard 2008). Immunohistochemical staining for α2u-g was not graded but evaluated by observation of the intensity and specific staining pattern.
Results
In the 90-day study, three sets of histopathologic change were observed in the kidneys of treated animals depending on dose group: hyaline droplet accumulation in the cortex, an unusual form of nephrosis in the outer stripe of outer medulla (OSOM), and the exacerbation of chronic progressive nephropathy (CPN), a spontaneous disease entity (data summarized in Table 1). In the 2-year carcinogenicity study, 3 sets of dose-dependent histopathologic change in the kidney included linear mineralization in the papilla, nephrosis in the OSOM, and CPN exacerbation (data summarized in Table 2). In addition, renal tubule proliferative lesions, either hyperplasia, adenoma, and/or carcinoma were present in relatively low incidences (albeit greater than controls) at all dose levels in the 2-year study (data summarized in Table 3). In addition to the renal lesions, β-myrcene-induced lesions in other organs included olfactory epithelial degeneration and chronic inflammation in the nose and increased porphyrin pigment in the Harderian gland in the 90-day study, and chronic inflammation of the nose in the 2-year study. Tests for genetic toxicity were negative.
Incidences of non-neoplastic lesions in F344 rats in the 90-day gavage study of β-myrcene.
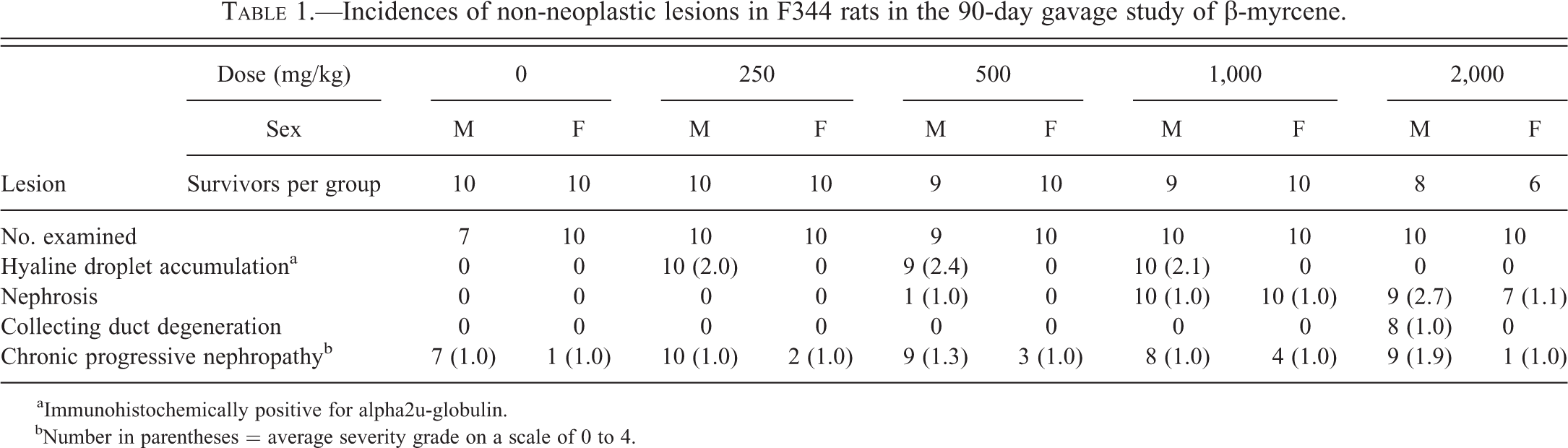
aImmunohistochemically positive for alpha2u-globulin. bNumber in parentheses = average severity grade on a scale of 0 to 4.
Incidences of non-neoplastic lesions in F344 rats in the 2-year gavage study of β-myrcene.
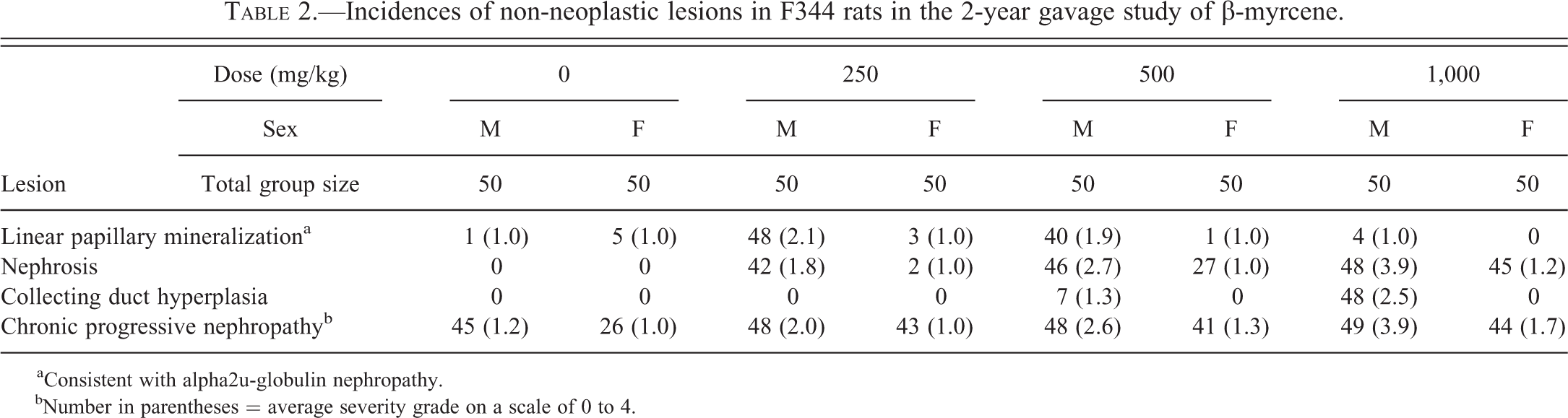
aConsistent with alpha2u-globulin nephropathy. bNumber in parentheses = average severity grade on a scale of 0 to 4.
Incidences of renal tubule proliferative lesions in F344 rats in the 2-year gavage study of β-myrcene.
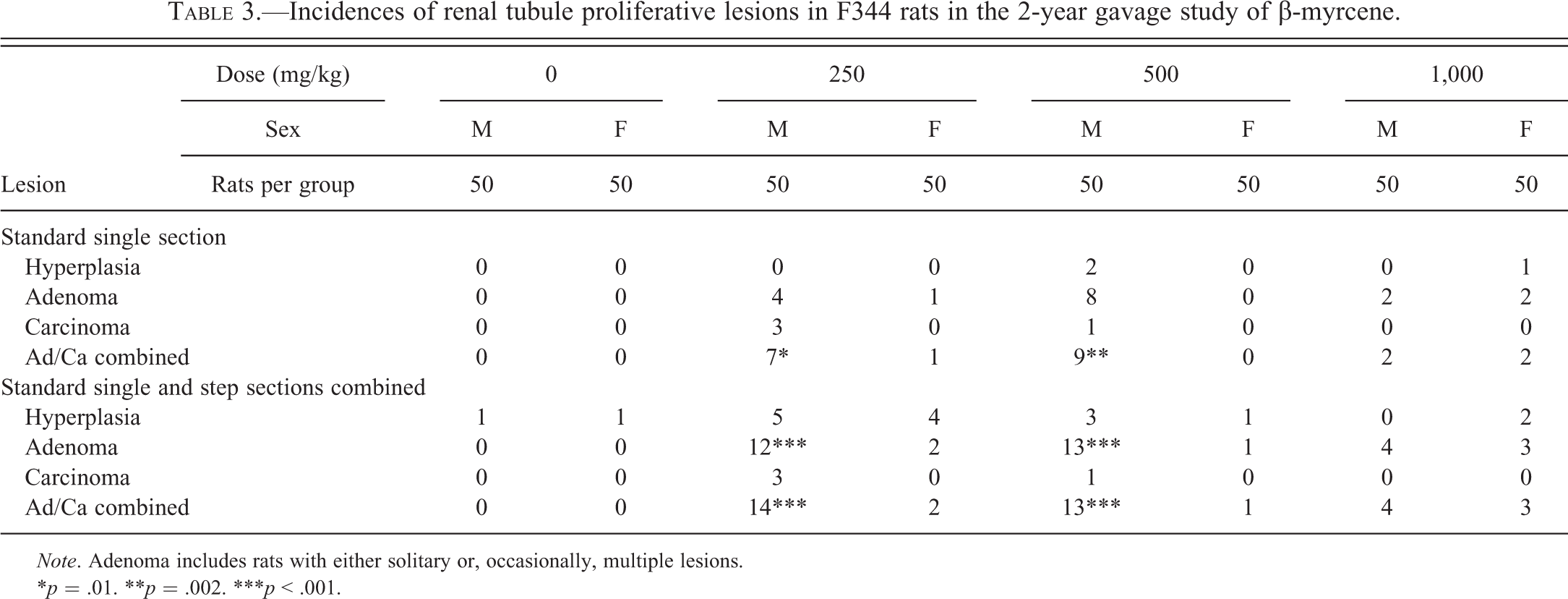
Note. Adenoma includes rats with either solitary or, occasionally, multiple lesions.
*p = .01. **p = .002. ***p < .001.
Core Segment of the 90-day Study
Hyaline Droplet Accumulation
In male rat kidney stained with MH or immunohistochemically with antibody to α2u-g, the control rat hyaline droplet pattern in the cortex was characterized by ill-defined, radial tracts of tubules (Figure 1), which contained small, well-formed, circular droplets in some tubules, and clusters of tubule cells with dense, apical aggregates or tufts of MH-positive droplets (Figure 2). This pattern, typical of young mature male F344 rats (Hard 2008), was confirmed by fluorescence microscopy. On a scale of 0 to 4, hyaline droplet accumulation in untreated male rats was assigned a grade of 0.
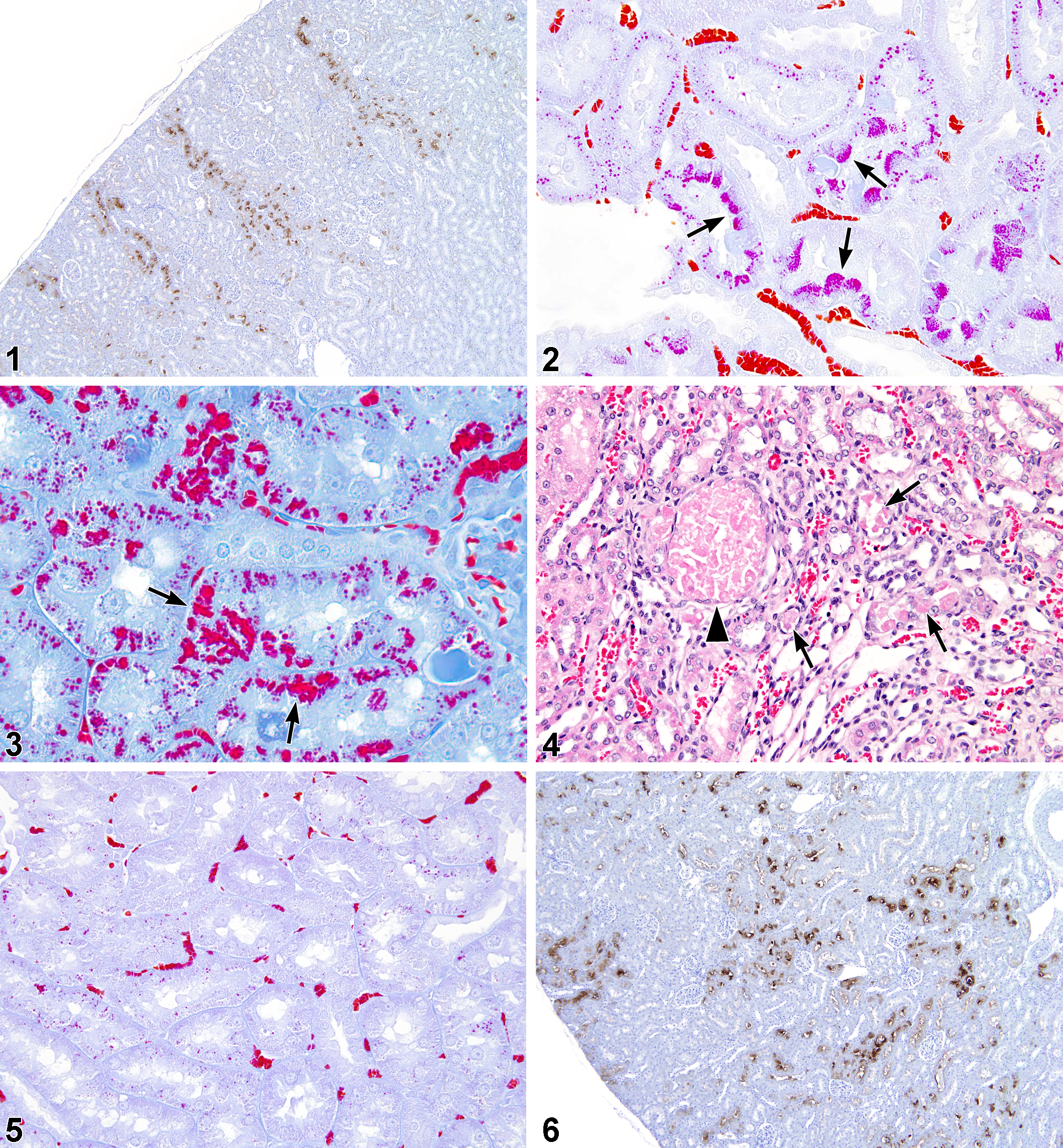
At 250 and 500 mg/kg, hyaline droplet accumulation was present in the cortex of male rats, but not easily discerned in H&E-stained tissue. However, with MH staining and under ultraviolet illumination, there was a clear increase in hyaline droplets, increased brightness of droplets, usually increased area of involvement, and disruption of the normal pattern of lysosomes, mainly in the S2 segments of the proximal tubules. The disrupted pattern contained fewer “tuft”-like cells and rounded droplets, but with increased numbers of clusters of angular or brick-shaped profiles suggestive of crystal aggregates (Figure 3). The average severity grade of hyaline droplets at 250 mg/kg was 2.0 and at 500 mg/kg was 2.4. In addition, there were a minimal number of granular casts at the junction of the OSOM and inner stripe of outer medulla (ISOM; Figure 4), which stained positively for α2u-g. Some proximal tubules at the same location contained small amounts of granular cell debris and were consistent with precursors of the larger, granular casts (Hard 2008).
At 1,000 mg/kg, hyaline droplet accumulation was of a similar mild grade as in the lower 2 dose groups (250 and 500 mg/kg), but only 1 rat had a single granular cast at the OSOM/ISOM junction indicating some reduction in the hyaline droplet response.
At 2,000 mg/kg, both the hyaline droplet accumulation and the normal young male rat pattern of hyaline droplets were entirely absent from the cortex (Figure 5). At high magnification, only a sporadic, small droplet was observed in cortical proximal tubules in MH-stained tissue or under ultraviolet illumination. The average severity grade of hyaline droplet accumulation in this dose group was 0; however, there were far fewer hyaline droplets than in the controls.
Immunohistochemical staining with antibody to α2u-g confirmed that the accumulating protein in the treated males at the lower doses (250 and 500 mg/kg) was α2u-g (Figure 6), but the kidney cortex of male rats in the 2,000 mg/kg dose group was negative for α2u-g staining.
In females, both the normal male rat pattern of hyaline droplets and the droplet accumulation were absent in all treatment groups (including controls), and the kidneys were immunohistochemically negative for α2u-g.
Nephrosis
There was no indication of tubule nephrosis in males or females exposed to 250 mg/kg of β-myrcene, or generally in males at the 500 mg/kg dose in the 90-day study. However, 1 male in the 500 mg/kg dose group had a minimal increase in mitotic activity involving the S3 segment of proximal tubule in the outermost part of the OSOM, which was interpreted as an early indicator of a nephrotic change. In females exposed to 500 mg/kg, there was some nuclear variability in epithelial cells of the OSOM, involving a low scatter of slightly enlarged nuclei with a prominent nucleolus, which was also consistent with an early indicator of nephrosis.
At the 1,000-mg/kg dose level, all male and female rats were judged to have slightly more advanced stages of nephrosis in the outermost region of the OSOM. These changes comprised minimal dilation of S3 tubules (which could be viewed as being within normal limits based on the width of the lumen, but contrasted with the more collapsed state of the S3 tubules in the animals without nephrosis), minimal nuclear enlargement of the epithelial lining cells of S3 tubules, and occasional degenerate (pyknotic) cells in S3 lumens. One of the male rats also had a minimal degree of single cell degeneration in the papillary collecting ducts.
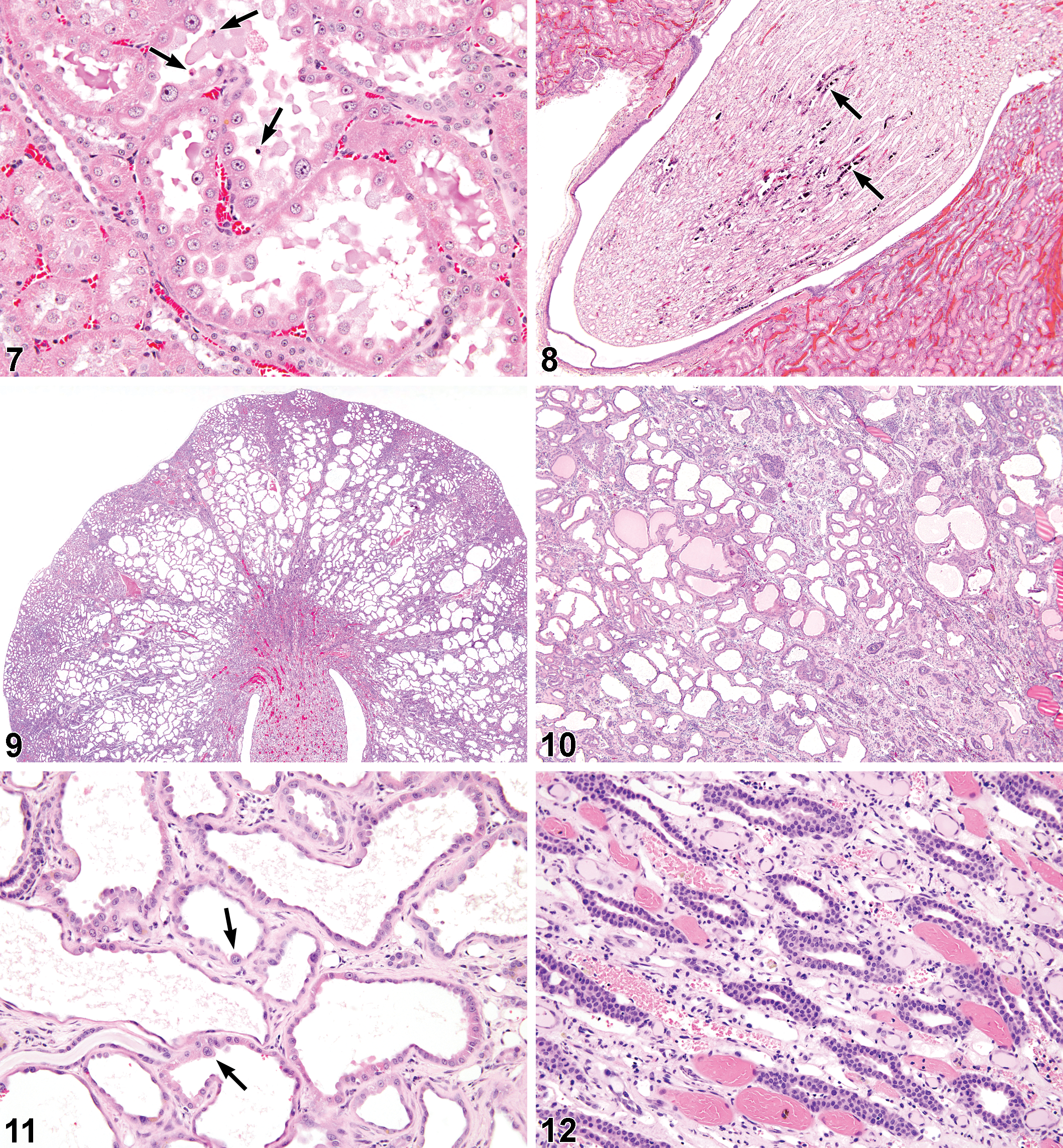
At 2,000 mg/kg, nephrosis involving the outermost S3 tubules of the OSOM affected all 8 surviving male rats and 1 rat that died during week 3 of the study. Once again, the constellation of changes included minimal to mild dilation of the S3 tubules, a moderate degree of nuclear enlargement, a low scatter of sloughed degenerate cells in the dilated S3 lumens (Figure 7), and a minimal grade of single cell degeneration in the lining epithelium of the papillary collecting ducts. Female rats had a similar pattern of nephrosis, with all 6 surviving female rats and 1 rat that died during week 13 of the study being affected, but the nephrosis was of a minimal grade, and without the collecting duct degeneration.
Although none of the animals at the highest dose of 4,000 mg/kg survived, histopathologic evaluation of the kidneys was possible in 1 male rat sacrificed moribund at 3 days, and 1 rat dying on day 11. In the rat surviving for 3 days, there was cell degeneration (nuclear pyknosis) involving the outermost parts of the OSOM, cell debris in the lumens of Henle’s loops and collecting ducts, and the mild dilation of medullary tubules. At 11 days, the outermost band of OSOM showed tubule basophilia indicative of zonal regeneration. There were also scattered degenerate cells in this region, increased mitotic figures, and nuclear enlargement. Granular cell debris was present in the lumens of Henle’s loops and some collecting ducts.
CPN
On a severity scale of 0 to 4, nearly all male rats from the control and 250 mg/kg groups (7 in controls, 10 in 250 mg/kg group) showed a minimal (grade 1) background of CPN. There was no statistically significant increase in the incidence of CPN in female rats, and in all female rats that had CPN, the CPN was of minimal severity (grade 1). Early CPN lesions consisted of a basophilic tubule or group of tubule convolutions with crowded nuclei, associated with conspicuously thickened basement membrane, and sometimes an eosinophilic hyaline cast in the ISOM.
At 500 mg/kg, a few rats had grade 2 (mild) CPN, which may reflect a minimal increase in severity in this group. At 1,000 mg/kg, CPN had reverted to the control level of grade 1 for all male rats in this group. At the 2,000 mg/kg dose group, the average severity grade was 1.9, indicating an increase in the severity of the CPN from minimal to mild.
23-day Segment of 3-month Study
Based on MH staining and fluorescence microscopy, all male rats in the control, 250-, and 500-mg/kg dose groups had hyaline droplet accumulation, but there was a slight increase in severity in the 250- and 500-mg/kg dose groups compared to controls. However, there were no granular casts, or their precursors at the OSOM/ISOM junction in any of these animals. The 9 surviving male rats in the 1,000 mg/kg dose group were considered to have a trace of increased droplet accumulation. In the group receiving 2,000 mg/kg, the normal hyaline droplet pattern was reduced.
A minimal degree of variability in nuclear size in the outer OSOM was evident in several males in the 1,000 mg/kg dose group, but there was no definite tubule dilation affecting this zone, or papillary collecting duct change. The OSOM nephrosis, including slight S3 dilation and nuclear enlargement, was evident in all male rats at the 2,000 mg/kg dose. In addition, most of these animals had basophilic tracts of tubules running from cortex through the OSOM. These were distinct from CPN as thickening of the basement membrane was variable, and the tubules were not associated with hyaline casts in the outer or inner medulla. They were interpreted as regenerative tubules that were an early component of the nephrosis. All male rats at this dose also had minimal to mild single cell necrosis involving collecting ducts along the length of the papilla.
2-year Carcinogenicity Study
Linear Papillary Mineralization
There was a statistically significant increase in the incidence of linear papillary mineralization in the 250- and 500-mg/kg male rats compared to controls with a concurrent increase in mean severity. There was papillary mineralization in some of the female rats (though with no treatment-related increase in incidence or severity) and in 1 control male rat, but all of these were small, solitary foci of tubule mineralization in the papilla consistent with the sporadic, background lesion often seen in controls. This lesion is morphologically distinct from the lesion referred to here as linear papillary mineralization, which comprised multiple profiles of linear deposits specifically within the lumens of Henle’s limbs (Figure 8).
In contrast to the control males and all females, the majority of male rats exposed to 250 and 500 mg/kg exhibited a mild, or sometimes moderate, severity (mean grade 2.1 and 1.9, respectively) of linear papillary mineralization (Figure 8). Linear papillary mineralization was absent in male rats at the 1,000 mg/kg dose, and in all female rats.
Nephrosis
Most of the male rats from the 250- and 500-mg/kg dose groups had some degree of nephrosis (42/50 and 46/50, respectively). In the 250-mg/kg group, the severity was minimal to mild and was characterized by minimal dilation of the S3 tubule segments with mild nuclear enlargement (including occasional karyomegaly), occasional degenerate cells in the S3 lumens, and slightly increased mitotic figures. In 2 male rats with advanced CPN, the nephrosis appeared to be more severe as well as the CPN. Only 2 females at the 250-mg/kg dose level had minimal nephrosis.
In the 500-mg/kg group, the nephrosis was more severe with a mean severity grade of 2.7. In these rats, the features of nephrosis were similar to those listed above, but tended to be slightly more severe. A few rats in this group had marked nephrosis as described below. There was minimal nephrosis in 27 of the 50 female rats.
At 1,000 mg/kg, nephrosis in the outermost OSOM was much more pronounced, with prominent S3 tubule dilation (Figures 9 and 10), low-cuboidal to hobnail-shaped cells lining the dilated S3 tubules (Figure 11), nuclear enlargement including karyomegaly, and relatively frequent pyknotic cells in the dilated S3 lumens. The severity of nephrosis was, on average, marked (grade 4) in male rats, and mild (grade 2) in female rats. The kidneys of male rats with marked nephrosis were markedly enlarged. In addition, in males only, there was a mild to moderate hyperplasia of the epithelial lining of collecting ducts in the deep medulla and papilla (Figure 12). This alteration appeared to accompany grade 3 (moderate) or 4 (marked) nephrosis.
CPN
In the control groups, the incidence of CPN in the males was 45/50 with a mean severity grade of 1.2. In the females, the incidence of CPN was 26/50 with a mean severity grade of 1.0.
In male rats at 250 mg/kg, there was a modest enhancement of CPN to a mean severity of grade 2 (mild) relative to control males (incidence was 48/50). In the females, there was a slight exacerbation of CPN with an incidence of 43/50, but the mean severity remained at grade 1 (minimal). At 500 mg/kg, the grades of CPN severity were elevated slightly, to grade 2.6 in males (incidence was 48/50), and 1.3 in females (incidence was 41/50).
At 1,000 mg/kg, CPN in male rats was mostly at grade 4 (marked), but it was difficult to distinguish from the kidney tissue involved in severe nephrosis. It was notable that no kidney showed end-stage CPN, and usually, the prominence of tubular hyaline casts that characterize severe CPN was much less than expected. There was a marked expansion of intercellular matrix, but it was uncertain whether this was strictly a component of CPN or partly contributed by the nephrosis. Islands of tubule epithelium surrounded by thickened basement membrane, often seen in very advanced CPN (Hard and Seely 2006), were more frequent and larger than usual. In female rats, the mean of CPN severity grade was elevated slightly to 1.7.
The increase in hyperplasia of the papilla lining reported as hyperplasia of the transitional epithelium lining of the pelvis and renal papilla (NTP 2010) was of the morphological type consistent with advanced stages of CPN. This lesion was therefore considered part of the CPN disease spectrum.
Proliferative Lesions
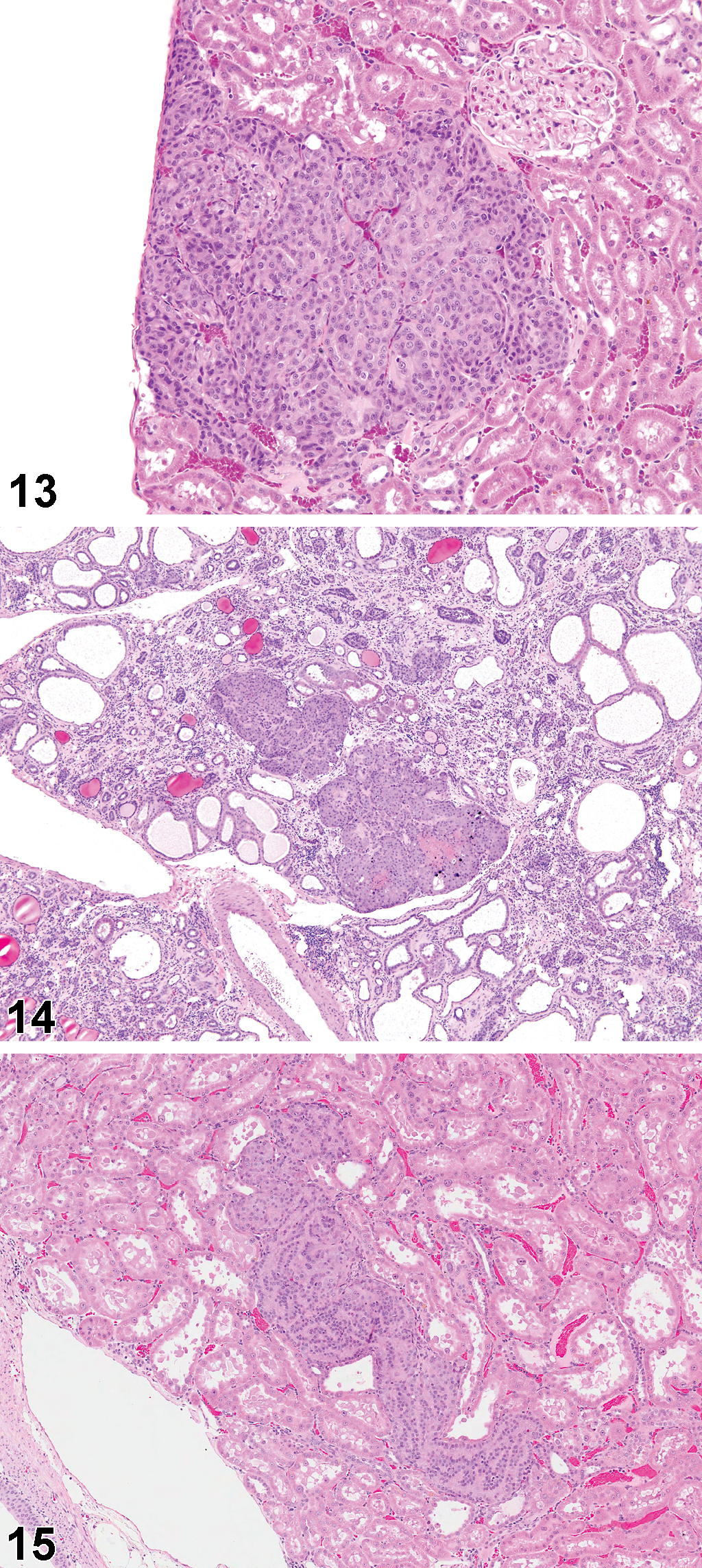
Renal tubule proliferative lesions (hyperplasia, adenomas, and carcinomas) from the original kidney sections, and subsequent step sections, were reviewed by an NTP Pathology Working Group, chaired by one of the authors (M.F.C.), and those final incidences are presented in Table 3. After combining the original and step-section data, solitary foci of renal tubule hyperplasia were observed in low incidence in all groups except the high-dose (1,000 mg/kg) males, and particularly in the low-dose (250 mg/kg) males and females. Similarly after combination of original and step-section data, adenomas and/or carcinomas were present at low incidence in all treated groups except the control males and females, but particularly evident in the low-dose and mid-dose (250 and 500 mg/kg) males, where there was up to a 30% incidence. The renal tubule tumors and precursor hyperplasia were basophilic lesions with a tendency to tubular differentiation. In the lower dose groups, small tumors or hyperplasia were often located within the cortex (Figure 13), but at higher doses, and in female rats, the lesions also appeared to involve the OSOM (Figures 14 and 15).
Discussion
β-Myrcene produced a uniquely complex spectrum of renal histopathology in F344 rats following gavage administration: hyaline droplet accumulation in the cortex at the lower doses in the 90-day study and linear papillary mineralization at the same doses in the 2-year study, and an unusual nephrosis involving the OSOM as the dose level was increased in both the 90-day and the 2-year studies. The nephrosis in the 2-year study was complicated by an apparent exacerbation of CPN with increasing dose, especially in the male rats.
At the lower doses of 250 and 500 mg/kg in the 90-day study, β-myrcene induced a mild hyaline droplet response in male rats, which was identifiable as α2u-g nephropathy (Hard et al. 1993; Swenberg and Lehman-McKeeman 1999) based on droplet staining with MH, observation under fluorescence microscopy, positive immunohistochemical labeling of the protein with antibody to α2u-g, and the presence of granular casts and/or their precursors at the junction of the outer and inner stripes of outer medulla. The development of linear papillary mineralization in the 2-year study at the same dose levels (250 and 500 mg/kg) represented the chronic histological consequence of α2u-g nephropathy.
Hyaline droplets at 90 days and linear papillary mineralization at 2 years (evidence of α2u-g nephropathy) were absent in the kidneys of male rats exposed to the highest doses of β-myrcene in each case (2,000 mg/kg in the 90-day study and 1,000 mg/kg in the 2-year study). The cause of this striking difference from the lower dose groups was not explained. However, it could be speculated that exposure to increasing concentrations of β-myrcene may have progressively suppressed hepatic synthesis of α2u-g, which is the origin of circulating α2u-g in male rats (Roy et al. 1983).
Commencing at the mid-dose level (500 mg/kg), a second pathological lesion developed in the 90-day study, involving S3 segments of proximal tubule of the outer part of the OSOM. This nephrosis became more pronounced with increasing dose and with increased exposure duration (i.e., in the 2-year study). Early deaths occurring up to 11 days at the 2,000 mg/kg dose level indicated that the nephrosis commenced as a necrotic change involving the epithelium of the outermost parts of the S3 tubules in the OSOM, followed by tubule regeneration in the same zone. The reason for the slight dilation of this tubule region, occurring at both the 90-day and the 2-year time points, could not be determined, but the epithelial lining of the outer S3 tubules became more distorted with cellular abnormality, including nuclear enlargement and some karyomegaly, with increasing dose and length of study.
The responses of the male and female rats to β-myrcene differed in that α2u-g nephropathy was restricted to the males, but the nephrosis was common to both sexes. However, the nephrosis was generally not as severe in the females as in the males, a difference that was particularly noticeable at the high dose (2,000 mg/kg in the 90-day study and 1,000 mg/kg in the 2-year study).
It is reasonable to deduce that the main cause for cortical adenomas at the lower doses in male rats was α2u-g nephropathy, while at the highest dose, where hyaline droplet accumulation was absent in the 90-day study, tumors were probably associated with the nephrosis. Adenomas developmentally linked with exacerbated CPN are most commonly found in end-stage CPN (Hard et al., 2012) when almost the entire kidney is affected by CPN, and the kidney is misshapen due to the prominence of hyaline casts. These characteristics were not observed in the β-myrcene-treated male rats suggesting that CPN was not necessarily involved in the pathogenesis of β-myrcene-associated tumors.
The mode of action whereby renal tubule tumors develop as a consequence of α2u-g nephropathy in male rats has been determined to be a species- and sex-specific phenomenon (Baetcke et al. 1991; International Agency for Research on Cancer [IARC] 1999) of no relevance for extrapolation to humans for hazard and risk assessment purposes. However, this judgment pertains only if female rat kidneys are lesion-free, and when there are no other renal changes of toxicity in males (Swenberg and Lehman-McKeeman 1999). The dose-related occurrence of a form of nephrosis in both male and female rats exposed to β-myrcene by oral gavage, and the development of renal tubule tumors in males and possibly in females (in the absence of α2u-g nephropathy at the highest male dose), suggests that an additional mechanism other than α2u-g nephropathy exists for β-myrcene-induced renal carcinogenesis.
Footnotes
Acknowledgments
This work was supported by the Division of the National Toxicology Program of the National Institutes of Health (NIH), National Institute of Environmental Health Sciences (NIEHS), Research Triangle Park, NC. However, the statements, opinions, or conclusions contained herein do not necessarily represent the statements, opinions, or conclusions of NIEHS, NIH, or the U.S. Government. Dr Hard’s efforts in this project were supported, in part, by Federal funds from the NIEHS, NIH, under contract NO1-ES-95435 to Experimental Pathology Laboratories Inc., Research Triangle Park, NC. The authors wish to thank Ms. Natasha Clayton and Mr. David Olson for preparing the immunohistochemical stains, and Drs. David Malarkey and Kyathanahalli Janardhan for their review of this article.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received the following financial support for the research, authorship, and/or publication of this article The authors received government funding from the Division of the National Toxicology Program, National Institute of Environmental Health Sciences, National Institutes of Health, Department of Health and Human Services.
