Abstract
Bazedoxifene acetate (BZA) is a selective estrogen receptor modulator that is approved in a number of countries for the prevention and/or treatment of osteoporosis in postmenopausal women. To assess carcinogenic potential, BZA was administered
Keywords
Introduction
Corticomedullary mineralization (CM) occurs naturally in the kidneys of aging female rats, but not male rats, due to endogenous estrogens. Administration of exogenous estrogens causes CM in male rats characterized by intratubular casts, mineralized tubules, and tubulointerstial necrosis at the corticomedullary junction (Geary and Cousins 1969). Chronic progressive nephropathy (CPN) is a common spontaneous occurring renal disease of rats, which affects males more severely than females, and is the common non-neoplastic cause of death in males in 2-year carcinogenicity studies (Barthold 1979; Owen and Heywood 1986; Peter, Burek, and van Zwieten 1986; Goldstein, Tarloff, and Hook 1988; Montgomery and Seely 1990; Chandra and Frith 1993; Ettlin, Stirnimann, and Prentice 1994). The Sprague-Dawley (SD) and Fischer 344 (F344) rats are more severely affected by CPN than many other strains (Peter, Burek, and van Zwieten 1986; Montgomery and Seely 1990; Chandra and Frith 1993). In a review of non-neoplastic renal lesions in untreated S-D and F344 rats from several 2-year carcinogenicity/chronic toxicity studies, CPN occurred in 96.3% of male and 39.1% of female SD rats and in 88.8% of male and 74.5% of female F344 rats (Chandra and Frith 1993). The pathogenesis of CPN is not well understood but may be due, in part, to increased dietary protein and/or feeding high caloric diets (Goldstein, Tarloff, and Hook 1988; Montgomery and Seely 1990; Chandra and Frith 1993; Khan and Alden 2002; Seely et al. 2002).
A number of test articles evaluated in 2-year carcinogenicity studies in rats were found to exacerbate the incidence and/or severity of CPN (Hard et al. 1997; Hard 1998, 1999, 2002; Seely et al. 2002; Hard and Khan 2004; Sellers and Khan 2005; Hard et al. 2007; McGregor 2007; Hard, Betz, and Seely 2012). Several of these test articles (i.e., hydroquinone, ethyl benzene, and quercetin) have resulted in renal cell tubular hyperplasia, adenomas, and carcinomas (Hard et al. 1997; Hard 2002; Hard and Khan 2004; Hard et al. 2007; McGregor 2007; Hard, Betz, and Seely 2012). Because the mechanism of spontaneously occurring CPN is not known, there are no data to explain how these test articles exacerbate the spontaneous disease process, which provides an additional proliferative stimulus to drive tubular hyperplasia into foci of atypical tubular hyperplasia and adenomas (Hard 2002; Hard and Khan 2004). In addition, most atypical tubular hyperplasia and renal tumors are in areas of marked to severe CPN. The test articles are not generally genotoxic, do not directly cause DNA reactivity, and are associated with sustained regenerative cell proliferation within renal tubules (Hard 1998, 2002; Hard and Khan 2004). The occurrence of renal tumors in male rats under these circumstances is considered secondary to enhanced CPN, rather than a direct response of chemical exposure (Hard 1998, 2002; Hard and Khan 2004; Lock and Hard 2004; Hard et al. 2007; Hard, Betz, and Seely 2012).
Bazedoxifene acetate (BZA) is a nonsteroidal selective estrogen receptor modulator (SERM) that is approved and marketed as Conbriza® or Viviant™ in a number of European and Asian countries for the prevention and/or treatment of osteoporosis in postmenopausal women. BZA, like other SERMs, is a mixed function estrogen that demonstrates tissue-selective agonist activity on the skeletal system and lipid metabolism, while demonstrating little or no endogenous activity in the uterus through its action as an antagonist in the presence of estrogens such as estradiol. During the development of BZA, the carcinogenic potential of BZA was investigated in a 2-year study in
Materials and Methods
All animal studies were conducted with permission of the Institutional Animal Care and Use Committee at accredited Association for Assessment and Accreditation of Laboratory Animal Care research facilities.
1-, 3-, and 6-Month Repeat-Dose Toxicity Studies
Experimental Design
BZA (WAY-140424 Acetate; 1-[4-(2-Azepan-1-yl-ethoxy)-benzyl]-2-(4-hydroxy-phenyl)-3-methyl-1
Statistical Analyses
Calculations and analyses were performed separately for each gender.
Body weight, body weight gain, hematology, clinical chemistry, and organ weight data were analyzed. Jonckheere’s test was performed to test for a trend with BZA concentration, and a one-way analysis of variance (ANOVA) on the rank-transformed data was performed to test for differences among groups. If the
Genotoxicity
BZA was evaluated for its potential to induce reverse mutations in 4 strains of
BZA was evaluated for
2-Year Carcinogenicity Study
Experimental Design
BZA (WAY-140424 Acetate; 1-[4-(2-Azepan-1-yl-ethoxy)-benzyl]-2-(4-hydroxy-phenyl)-3-methyl-1
Summary of dosages and dose ratios in Sprague-Dawley rats given dietary Bazedoxifene.
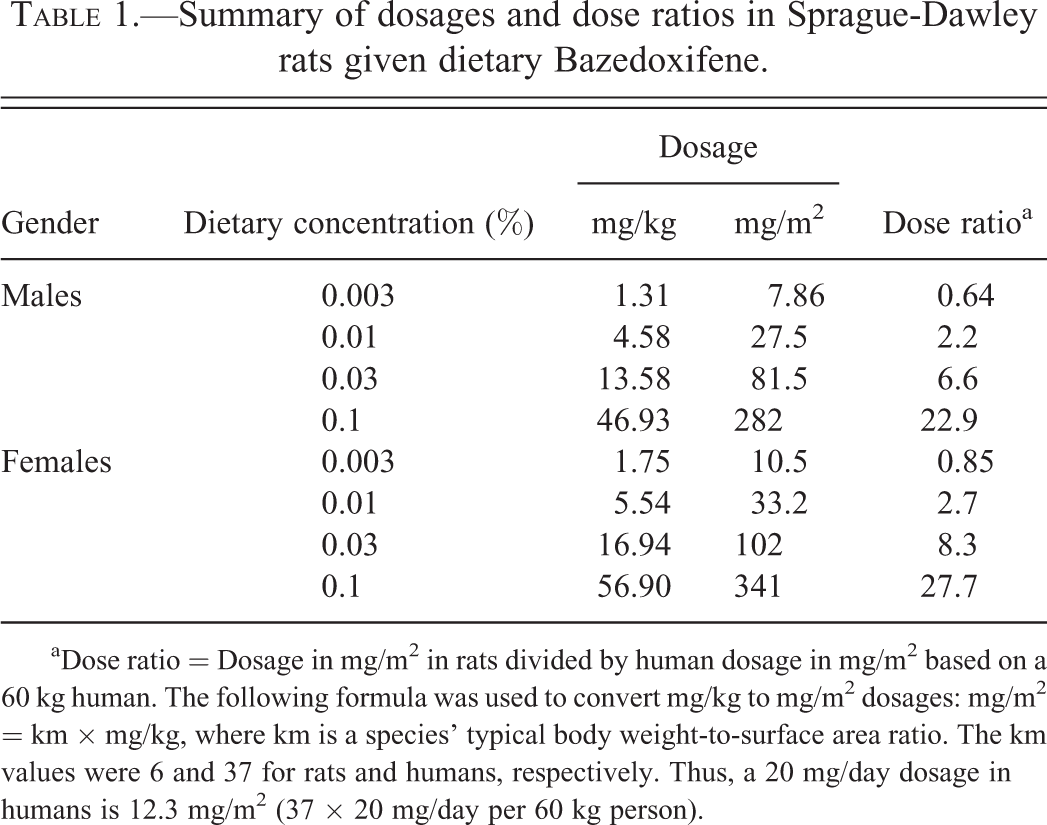
aDose ratio = Dosage in mg/m2 in rats divided by human dosage in mg/m2 based on a 60 kg human. The following formula was used to convert mg/kg to mg/m2 dosages: mg/m2 = km × mg/kg, where km is a species’ typical body weight-to-surface area ratio. The km values were 6 and 37 for rats and humans, respectively. Thus, a 20 mg/day dosage in humans is 12.3 mg/m2 (37 × 20 mg/day per 60 kg person).
Rats were approximately 6 to 7 weeks old at dose initiation, and males and females weighed 173 to 236 g and 130 to 187 g, respectively. Rats were housed 2 per cage (by gender and dosage group) where possible in plastic solid bottom cages with contact bedding. Animals were weighed and assigned to dosage groups by gender in a manner that minimized mean body weight differences among the dosage groups. Dosage groups were assigned to cages in a manner designed to uniformly disperse dosage groups throughout the study room. Male animals were housed in a separate room from the females. Room temperature was set at 72°F and relative humidity was 50%, and both were continuously monitored. Alternating 12-hr periods of light and dark cycles were in effect.
Evaluations for test article–related effects were based on mortality, clinical observations, palpation to detect tissue masses, body weight, food consumption, food utilization, hematology, and macroscopic and microscopic examinations. Postmortem euthanasia consisted of CO2/O2-induced anesthesia followed by exsanguinations and the creation of bilateral pneumothorax. A complete necropsy was conducted on each rat and included macroscopic examination of the external surface of the body, all orifices, the cranial, thoracic, and abdominal cavities and contents, and organs and tissues. Representative samples of the organs and tissues (except testes) were fixed in 10% neutral buffered formalin. Testes were fixed in Bouin’s solution. All fixed organs and tissues were dehydrated, embedded in paraffin, sectioned, stained with hematoxylin and eosin, and examined microscopically. Microscopic findings were classified with standard pathologic nomenclature and severities of findings were graded slight, mild, moderate, marked, or severe. Grades of severity for microscopic findings were subjective;
Pharmacokinetic Analyses
The plasma pharmacokinetics of BZA were evaluated in satellite groups of animals (9/gender/group) during week 26 (Table 2). The achieved mean BZA dosages for each dietary concentration, calculated based on mean body weights and mean food consumption values during the study, were approximately 1.31, 4.58, 13.58, and 46.93 mg/kg/day in male rats and approximately 1.75, 5.54, 16.94, and 56.90 mg/kg/day in female rats at the dietary concentrations of 0.003%, 0.01%, 0.03%, and 0.1%, respectively. On a surface area basis (mg/m2), the highest dietary concentration of 0.1% (approximately 282 mg/m2 in males and 341 mg/m2 in females) was 23 and 28 times, respectively, the clinical dose of 20 mg of BZA (approximately 12.3 mg/m2; Table 1).
Summary of exposures and exposure ratios in Sprague-Dawley rats given dietary Bazedoxifene.
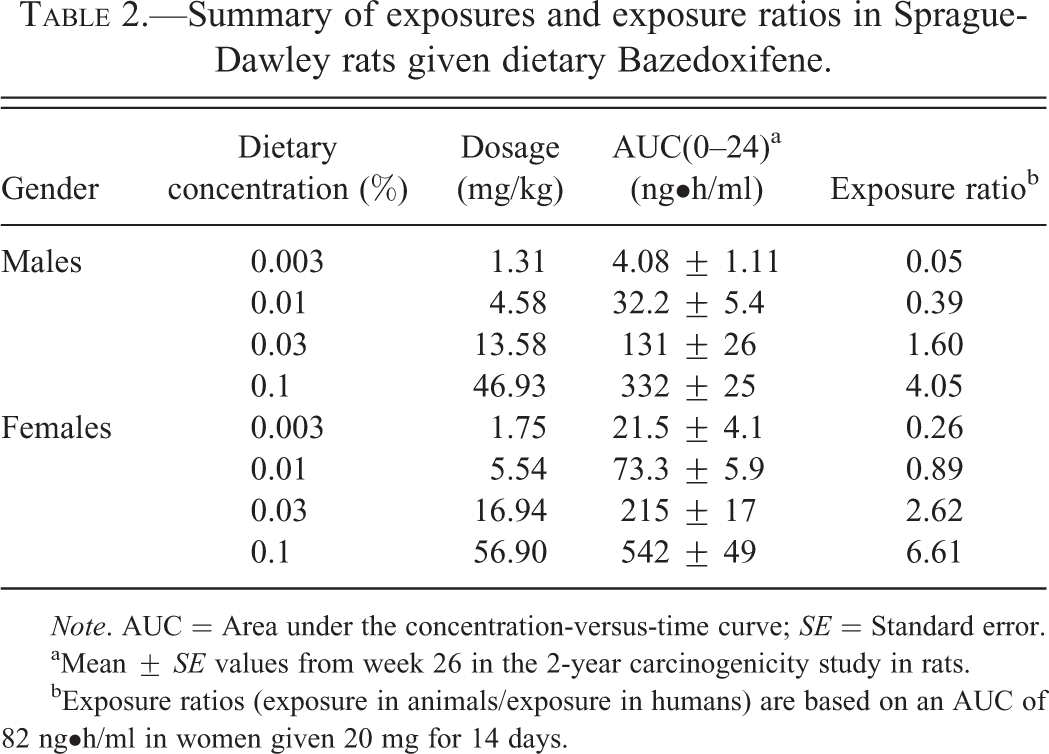
aMean ±
bExposure ratios (exposure in animals/exposure in humans) are based on an AUC of 82 ng•h/ml in women given 20 mg for 14 days.
Statistical Analyses
The two control groups were combined for descriptive statistics and statistical analyses. Calculations and analysis were performed separately for each gender.
Body Weight and Hematology Data
Body weight, body weight gain, and hematology data were analyzed using the same analyses as outlined for the 1-, 3-, and 6-month repeat-dose toxicity studies.
Food Consumption and Food Utilization Data
For each animal cage, the total weight of food measured for a given week was converted to mean gram (g) per animal per cage day by dividing the measured food consumed for the cage by the number of animal days; units for food consumption are mean g/animal/day. Food utilization was calculated as the ratio of food consumption to body weight (BW) for each animal; units for food utilization are (mean g food/animal/day)/(g BW). Descriptive statistics were calculated.
Survival Data
Survival was compared among dosage groups using nonparametric time-adjusted log-rank methods including a test for trend with BZA concentration, an overall test for any differences among the groups, and pairwise comparisons of each BZA dose group with the combined control groups.
Microscopic Pathology
Unless specified differently, all animals from all groups were always included in statistical analyses of microscopic data. The incidence of benign and malignant neoplasms was analyzed using time-adjusted Peto methods that incorporate cause of death information; both trend tests and pairwise comparisons of each BZA dose group with the combined control groups were conducted. The Peto analysis includes adjustments for time to death for animals with fatal tumors, time of first palpation for palpable tumors, and time of tumor observation for tumors that are not considered fatal or palpable. Exact
To evaluate the association between mineralization and CPN incidence in male rats, the Cochran–Mantel–Haenszel test of association was used with the presence or absence of renal tumors as the stratification variable. Also, the degree of association between the incidence of CPN and renal tumors in male rats was evaluated using the same test with the concentration of BZA as the stratification variable.
Results
Renal Findings from Subchronic and Chronic Repeat-dose Toxicity Studies
In the 1-, 3-, and 6-month repeat-dose toxicity studies in SD rats, BZA caused an increase in kidney weight, hematuria, CM, and increased tubular basophilia in male rats (Table 3). In the 6-month study, hematuria was detected in-life from weeks 4 to 26 and was confirmed with Multistix® and microscopic examination of urine sediment. CM consisted of tubules at the corticomedullary junction that were partially to completely occluded by aggregates of mineral deposits. The lining epithelium of these tubules was obliterated by the mineral deposits. Tubular basophilia consisted of renal cortical tubules, individually or in small clusters, lined by epithelial cells with bluish cytoplasm with hyalinized and thickened tubular basement membranes.
Incidence and average severities of kidney findings in repeat-dose toxicity studies in male Sprague-Dawley rats given Bazedoxifene.
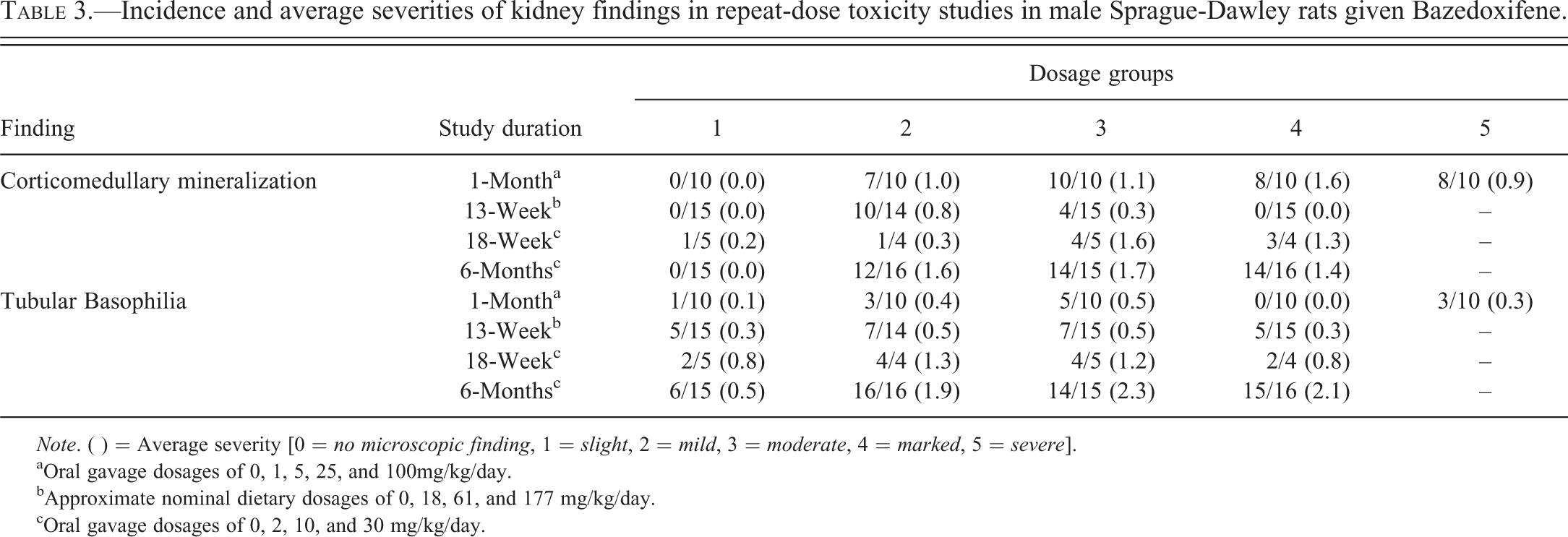
aOral gavage dosages of 0, 1, 5, 25, and 100mg/kg/day.
bApproximate nominal dietary dosages of 0, 18, 61, and 177 mg/kg/day.
cOral gavage dosages of 0, 2, 10, and 30 mg/kg/day.
Genotoxicity
BZA was not mutagenic in 3
2-Year Carcinogenicity Study
Pharmacokinetics
Exposure (based on the area under the concentration-versus-time curve [AUC]) at week 26 in male and female rats given BZA in the diet increased in an approximately dose-proportional manner and was slightly higher in females than in males. At the highest dietary concentration (0.1%), the exposure ratios were approximately 4 and 7 in male and female rats, respectively, compared with exposure (82 ng•h/ml) in women given 20 mg/day of BZA (Table 2).
Survival
The survival graph for male rats is provided in Figure 1A. In male rats administered BZA, survival was increased in all groups with a median survival time of approximately 45 days longer than controls (median survival that ranged from 720 to 729 days compared with 689 days for controls). The first death in control males occurred earlier (week 39) than in males administered BZA (week 55). Deaths in the control males were associated with pituitary gland tumors and renal disease. At the end of week 104, increased survival in males was significantly longer at all concentrations except the low dosage of 0.003%. The primary contributor to mortality in BZA-treated male rats was renal disease. There was a higher incidence of CPN in the 2 lower BZA-treated dosage groups that also had the lowest survival rate, with survival rates at the end of week 104 of 34%, 48%, 56%, 78%, and 68% at 0, 0.003%, 0.01%, 0.03%, and 0.1%, respectively.
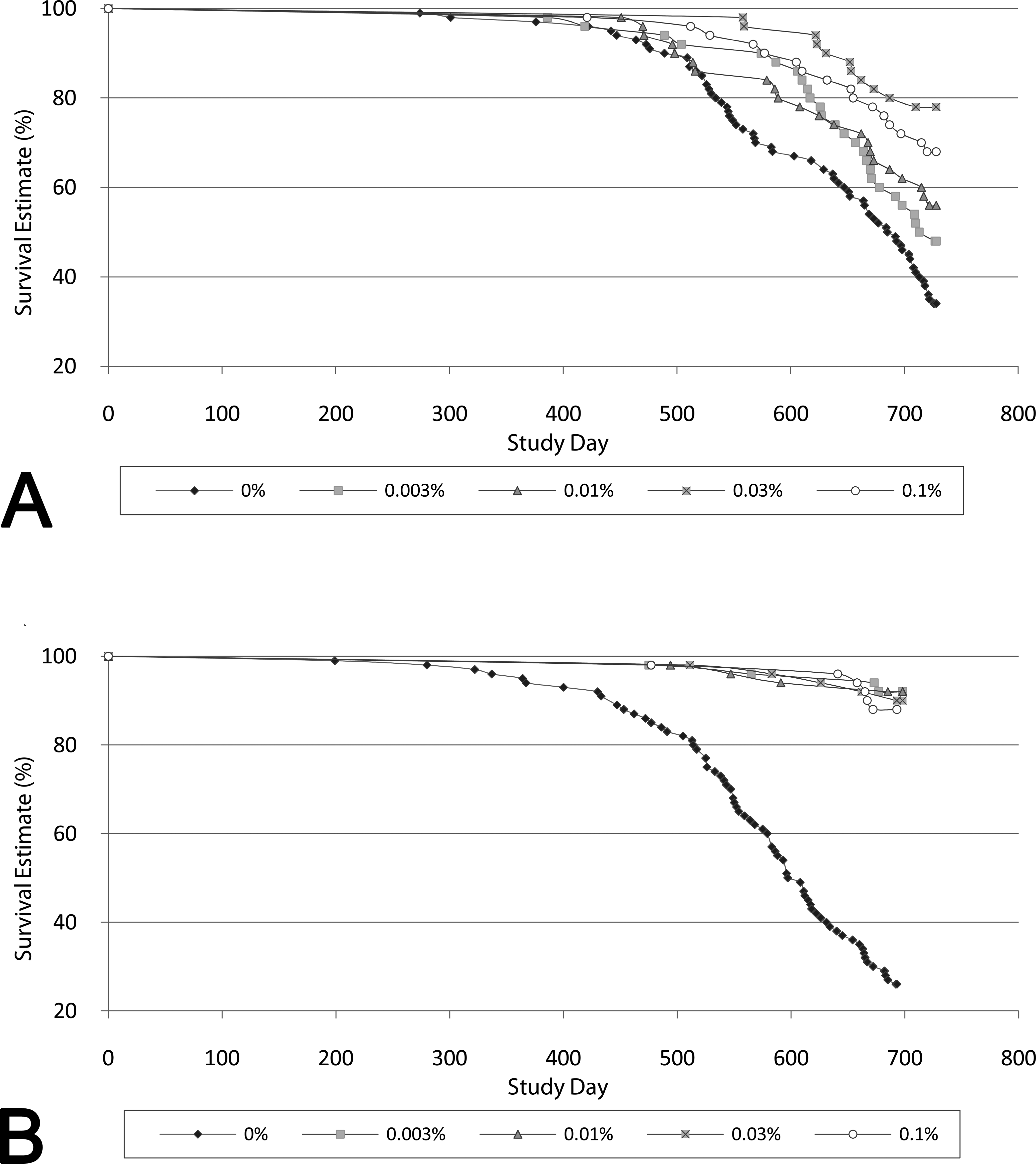
Rat survival curves: survival in male (A) and female (B) rats in a 2-year carcinogenicity study with BZA. (A) In male rats given BZA, survival was increased in all dose groups with a median survival time of approximately 45 days longer than controls. The first death in control males occurred earlier (week 39) than in males given BZA (week 55). At the end of week 104, the survival rates in males given BZA were 34%, 48%, 56%, 78%, and 68% at 0, 0.003%, 0.01%, 0.03%, and 0.1%, respectively, and significantly longer than controls at all concentrations except the low dosage of 0.003%. (B) In female rats given BZA, survival was statistically significantly increased at all dietary concentrations compared with controls, with ≥3.4 times the number of females alive at the end of the study given BZA. At the end of week 99, survival in females was 26%, 92%, 92%, 90%, and 88% at 0, 0.003%, 0.01%, 0.03%, and 0.1%, respectively. BZA = Bazedoxifene.
For comparison, the survival graph for female rats is provided in Figure 1B. Survival in females given BZA was also statistically significantly increased at all dietary concentrations compared with control females. At the end of week 99, survival was 26%, 92%, 92%, 90%, and 88% at 0, 0.003%, 0.01%, 0.03%, and 0.1%, respectively.
Body Weight and Food Consumption
Body weight differences were significantly different between treated animals and controls of both genders (Figure 2). The group mean body weights were approximately 18% to 25% less in treated male rats and 24% to 32% less in treated females than the combined control group at the end of the study. The differences were similar across all dose groups with no apparent relationship to dosage. Notably, the differences in body weight developed during the first 6 to 12 months of treatment, and did not change dramatically during the final half of the study.
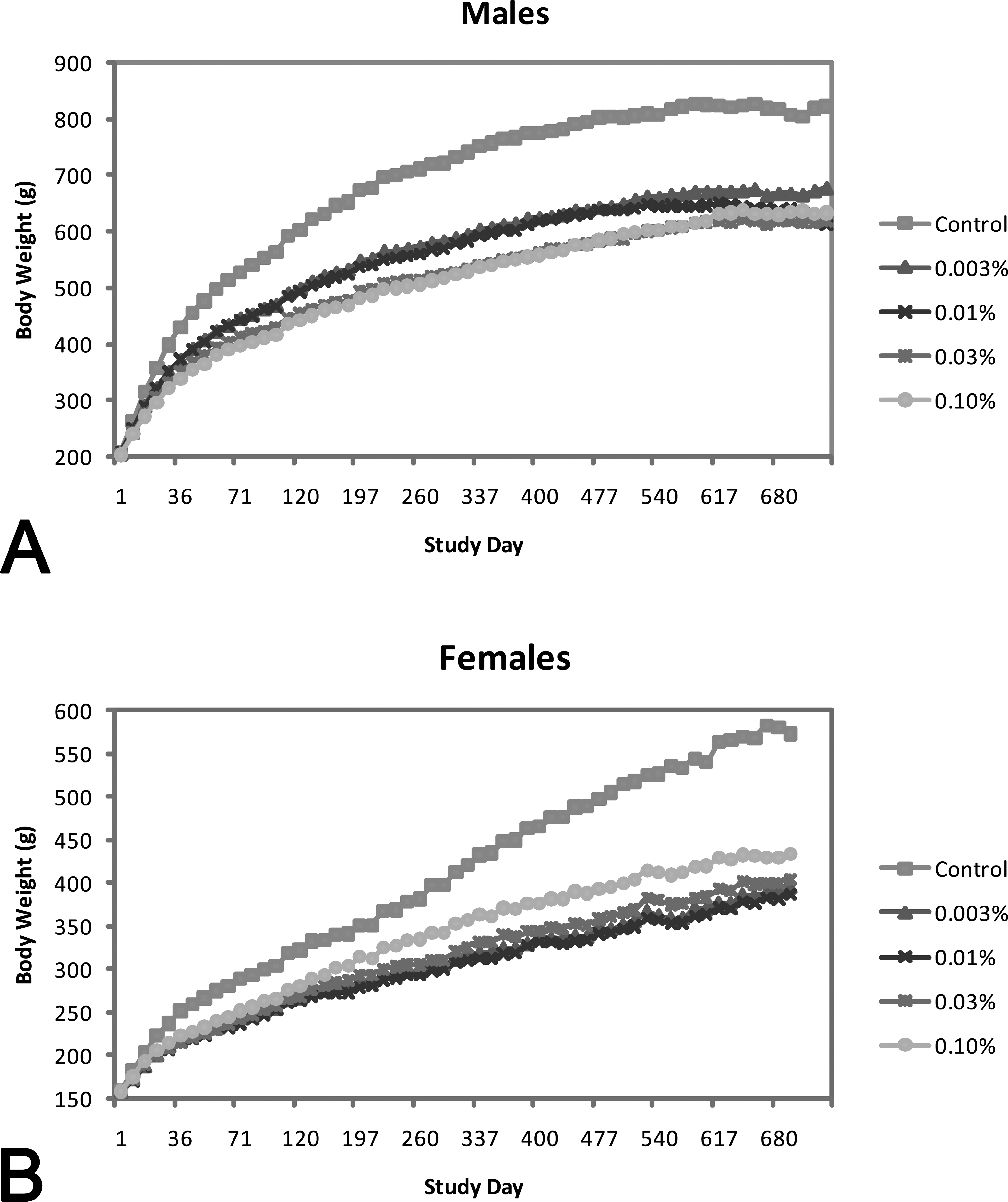
Changes in rat body weights: body weight summary in male (A) and female (B) rats in a 2-year carcinogenicity study with BZA. Group mean body weights over all evaluated intervals were consistently lower in males and females given BZA compared with control group mean body weights. At the end of the study, mean body weight was lower in males (18–25% at week 104) and females (24–32% at week 99) given BZA compared with controls. BZA = Bazedoxifene.
Generally, decreases (up to 21% and 32%) in mean food consumption (g/animal/day) in animals given BZA paralleled decreases in mean body weight over the first year of the study. In the second year of the study, mean food consumption in the BZA dose groups was either comparable with controls or decreased (up to 32%) in a non-dose-related manner compared with controls. Food utilization values (defined as the ratio of food consumption to body weight) indicated improved food utilization in animals given BZA over most of the study period.
Anatomic Pathology
Administration of BZA resulted in an increased incidence and severity of CM and CPN in the kidneys of male rats at all dosages compared with male controls, female controls, and female treated rats. Renal CM occurred at a high incidence (78% to 98%) in males given BZA and was not dose related (Table 4). Lesions were slight to moderate in males given 0.003% to 0.03% BZA and slight to mild in males given 0.1% BZA. Renal CM did not occur in control males.
Incidence, percentage, and severity of kidney findings in male rats given dietary Bazedoxifene for 2 years.
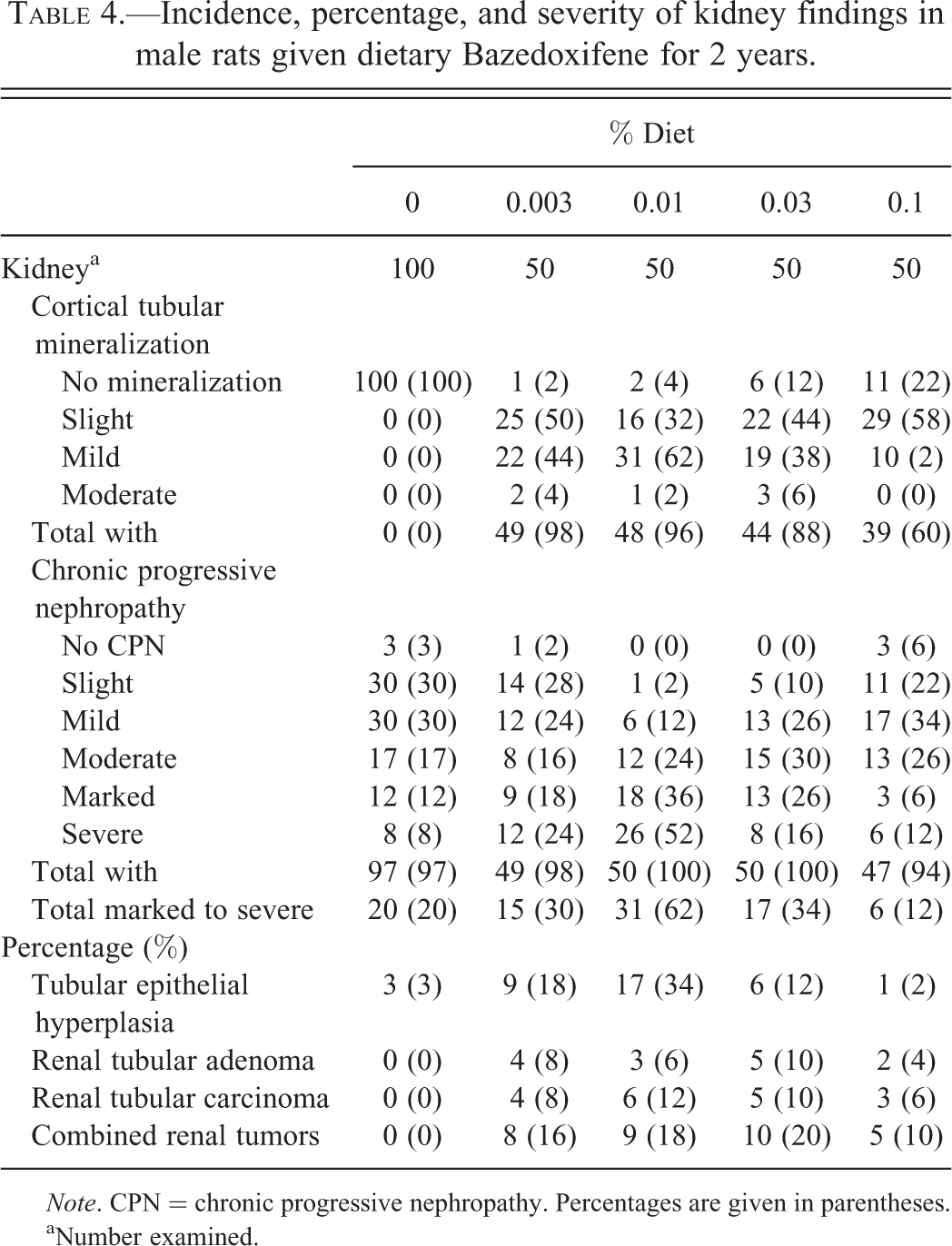
aNumber examined.
CPN consisted of cortical and medullary tubules with crowded nuclei, basophilic cytoplasm, and thickened basement membrane. Variably present were tubular casts, interstitial fibrosis, interstitial mononuclear cell infiltrates, tubular dilatation, and tubular epithelial atrophy. In advanced lesions, tubules were distended with eosinophilic proteinaceous material and glomeruli had mesangial proliferation, adhesions of glomerular tuft to Bowman’s capsule, or consisted of small sclerotic glomerular tufts that had dilated Bowman’s capsule filled with proteinaceous fluid. The percentage incidence of marked to severe CPN was 20% in control males and ranged in a non-dose-related manner (12–62% in BZA-treated males with 12% occurring in the high-dosage group; Table 4).
Renal cell tubular adenomas occurred in 0 of 100 control males (0%) and 4, 3, 5, and 2, of 50 males (4–10%) at 0.003%, 0.01%, 0.03%, and 0.1%, respectively (Table 4). Renal cell tubular carcinomas occurred in none of the 100 control males (0%) and 4, 6, 5, and 3 of 50 males (6–12%) at 0.003%, 0.01%, 0.03%, and 0.1%, respectively (Table 4). Renal tubular hyperplasia occurred in 3 of 100 control males (3%) and 9, 17, 6, and 1 of 50 males (2–34%) at 0.003%, 0.01%, 0.03%, and 0.1%, respectively (Table 4).
There was a statistically significant correlation between CM and marked to severe CPN (
Incidence, percentage, and severity of chronic progressive nephropathy in female rats given dietary Bazedoxifene for 2 years.
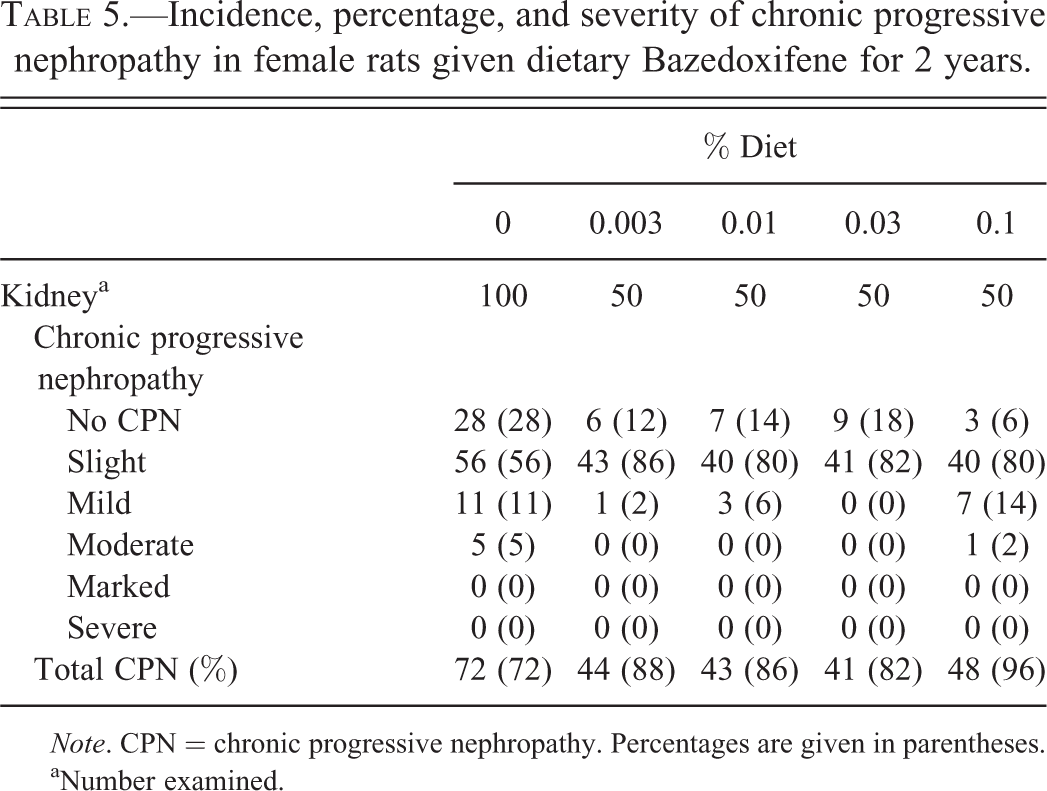
aNumber examined.
Statistical Analyses
To better understand the relationship between renal CM, CPN, and renal tumors in male rats in the 2-year carcinogenicity study, 2 statistical analyses were conducted. The first analysis was conducted to determine whether there was an association (correlation) between the degree of mineralization and the percentage of animals with marked to severe CPN. Male rats from the 2-year carcinogenicity study were categorized by 3 variables: severity of CPN (none to moderate vs. marked to severe), degree of mineralization (no to slight mineralization vs. mild to moderate mineralization), and presence or absence of renal tumors. Table 6 shows the number of male rats that fell into each of the 8 categories formed by these 3 variables. The Cochran–Mantel–Haenszel test of association was used to determine whether the percentage of male rats with marked to severe CPN was greater for rats with mild to moderate mineralization than those with no to slight mineralization. The presence or absence of renal tumors was used as a stratification variable in this analysis. This result was highly, statistically significant (
Renal corticomedullary mineralization and chronic progressive nephropathy in male rats given dietary Bazedoxifene for 2 years.

aData presented as number of males.
The second statistical analysis was carried out to determine whether there was an association between CPN and presence or absence of renal tumors. Male rats from the 2-year carcinogenicity study were categorized by 3 variables: treatment (control or BZA-treated), severity of CPN (no to moderate vs. marked to severe), and presence or absence of renal tumors. Table 7 shows the number of male rats that fell into each of the 8 categories formed by these 3 variables. For male rats given BZA, the incidence of renal tumors for animals with no to moderate CPN is 9% whereas the incidence for males with marked to severe CPN is 29%. The association between CPN severity and tumor incidence was evaluated using the Cochran–Mantel–Haenszel test of association with treatment as the stratification variable. This result of this test was highly statistically significant (
Renal tumor incidence and chronic progressive nephropathy severity in male rats given dietary Bazedoxifene for 2 years.
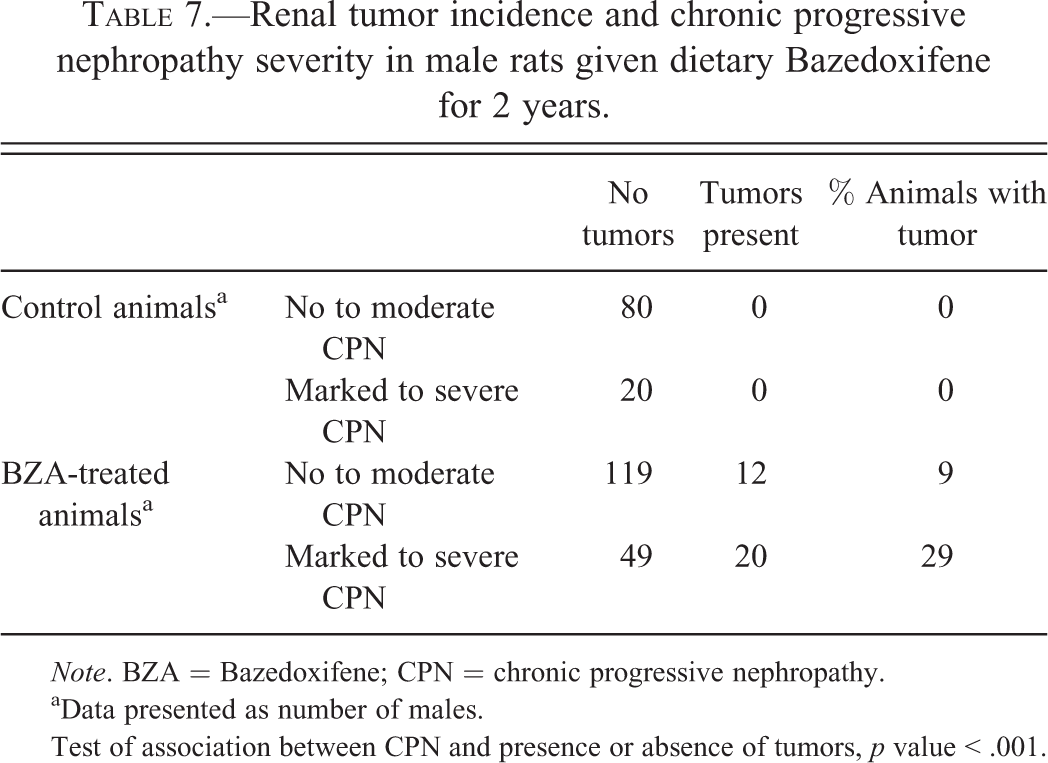
aData presented as number of males.
Test of association between CPN and presence or absence of tumors,
Discussion
In the 2-year rat carcinogenicity study, BZA administration resulted in a reduction and a delayed onset in total tumor burden in both male and female rats. Survival rates were enhanced due to decreased pituitary and mammary tumors and decreased body weight gain in BZA-treated animals compared with controls (Wright et al. 2012). In male rats only, an increase in renal tubular tumors was observed. The higher incidence of renal tumors in male rats given BZA was attributed to test article–induced CM, associated with clinical hematuria, and tubular basophilia in the subchronic and chronic studies. Prolonged administration of BZA resulted in further CM related renal tubular injury and exacerbation of naturally occurring CPN in aged male rats that led to proliferative changes and tumor formation in the 2-year carcinogenicity study (Figure 3). These findings were consistent with a non-genotoxic mechanism, unique to male rats.
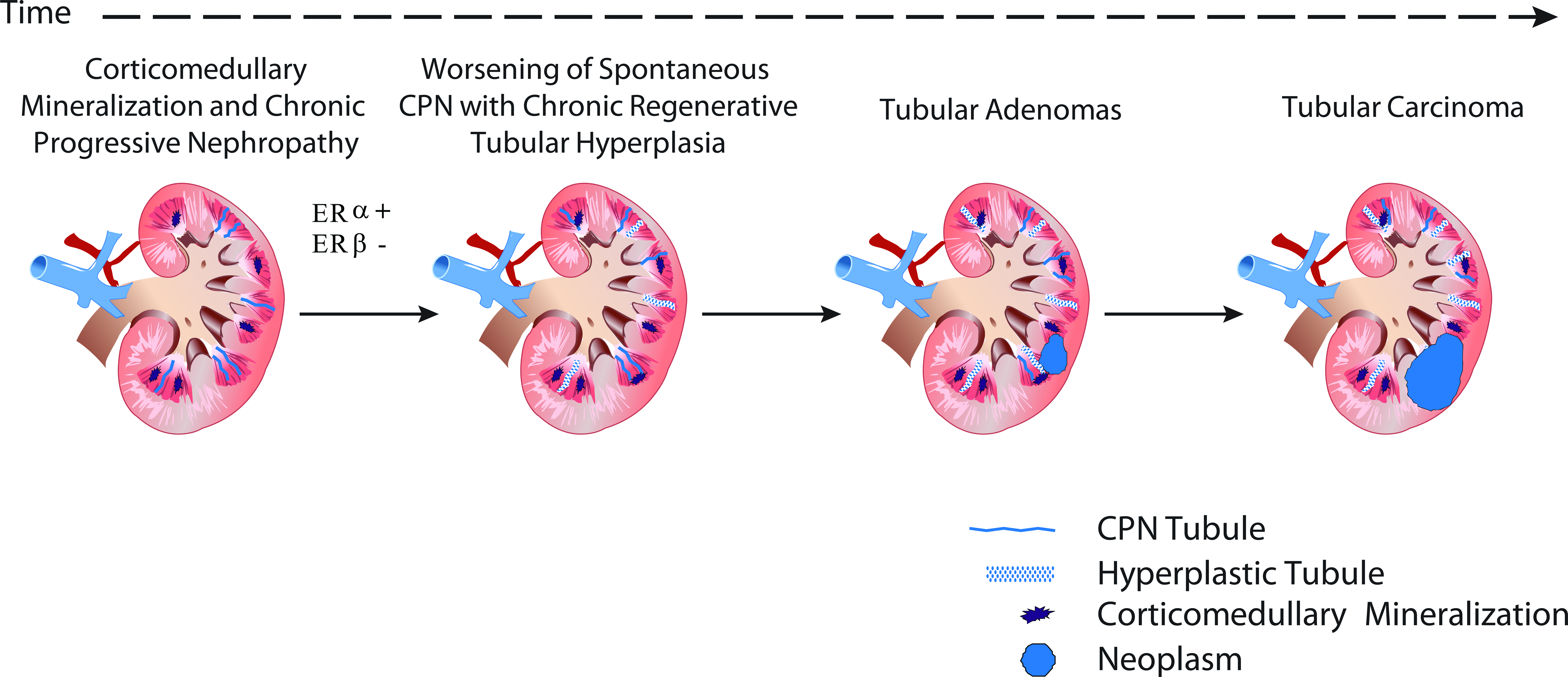
Proposed mechanism of renal tubular proliferative lesions in male rats given Bazedoxifene. Bazedoxifene, through its estrogenic (by ERα) action on the male rat kidney, causes tubular injury with hematuria and mineralization of corticomedullary tubules with exacerbation of CPN. Sustained renal tubular degeneration and repair combined with increased survival resulted in increased incidence of renal tubular hyperplasia and neoplasia. CPN = chronic progressive nephropathy; ER = estrogen receptor.
CM is a known estrogen-related effect unique to the male rat kidney (Geary and Cousins 1969); therefore, CM in male rats administered BZA was attributed to its estrogenic effects. Whether BZA functions as an agonist or antagonist in the rat kidney is unknown. Because estrogens normally cause CM in female rats, and this effect occurs in male rats given BZA, it is presumed that BZA elicits agonistic activity in the rat kidney. The primary estrogen receptor subtype expressed in the rat kidney is estrogen receptor α (Kuiper et al. 1997, 1998), and concentrations of estrogen receptor in male rat kidneys are approximately twice that found in female rat kidneys (Hagenfeldt and Eriksson 1988). Increases in estradiol occurred in female rats given BZA or raloxifene, and the source is the ovarian follicles (Wright et al. 2013). The absence of ovaries in male rats precludes similar increases in estradiol in male rats given BZA or raloxifene. Thus, increased endogenous estradiol in female rats given BZA or raloxifene likely competes for binding sites at the renal estradiol receptor, thus decreasing the toxicologic effects of the SERM in the kidneys. Because male rats do not respond to SERMs with an increase in endogenous estradiol, we propose BZA or raloxifene is able to bind to renal estradiol receptors relatively unopposed by endogenous estradiol with resulting tubular mineralization and exacerbation of CPN, tubular hyperplasia, and eventual neoplasia.
Renal CM in BZA-treated male rats was characterized by the presence of mineral within the lumen of renal tubules at the corticomedullary junction. Mineralization results from precipitation of calcium phosphate microcrystals initially in proximal tubule brush borders. The crystals subsequently move down the nephron, enlarging by further mineralization and collection of cellular debris. Eventually, the crystals block the tubule, forming characteristic deposits around the corticomedullary junction of the kidney. As a consequence of tubular obstruction, there is injury to the nephron both proximal and distal to the deposit (Al-Modhefer et al. 1986). Mineralization with concurrent tubular basophilia was not evident in female rats. The results of these nonclinical studies with BZA suggests that BZA injured tubular epithelium via intraluminal mineralization and this process exacerbated spontaneously occurring CPN to an advanced stage of severity (marked to severe) compared with controls. The increased cell proliferation in affected tubules resulting from chronic injury, when combined with the already proliferative activity of spontaneous CPN, led to renal tubular hyperplasia and renal tumors in a manner described in the literature (Hard 1998; Hard and Khan 2004; Lock and Hard 2004; Hard et al. 2007; Hard, Betz, and Seely 2012). A strong statistically significant correlation was established between CM, marked and severe CPN, and the occurrence of renal adenomas and carcinomas. Based on the unique occurrence of CM in male rats given BZA and its association with marked to severe CPN and renal tumors, CM is the fundamental toxic effect on the male rat kidney that resulted in renal tumors. Similar findings of an increased incidence and severity of cortical tubular mineralization, CPN, and renal tumors occurred in the kidneys of male rats given the SERM raloxifene (CDER 1997).
The mechanism for the formation of renal tumors in male rats given BZA was considered epigenetic and was associated with marked to severe CPN. This correlation is well documented in published scientific literature (Williams 1992; Hard 1998, 1999; Capen et al. 1999; Elcombe et al. 2002; Hard, Betz, and Seely 2012). This assessment is in agreement with the observation that non-genotoxic carcinogens are more specific than genotoxic carcinogens in their tumorigenic potential, as they frequently induce tumors in a single species, gender, and organ (Lima and Van der Laan 2000). In rodent models of estrogen-induced renal tumor renal neoplasia, such as the male Syrian hamsters (Hacker, Bannasch, and Liehr 1988) or Eker rats (Wolf et al. 1998), there is little evidence that estrogen directly results in renal epithelial tumors in rats. In the hamster model, renal tumors are of mesenchymal in origin and thought to result from stimulation of an estrogen-responsive interstitial stem cell, not though, to occur in the rat (Hacker, Bannasch, and Liehr 1988; Liehr et al. 1987; Li and Li 1990). Rat renal tumors could be induced with estrogens in the Eker but were associated with significant nephrotoxicity, and moreover, control wild-type rats concomitantly administered estrogen did not develop any proliferative lesions (Wolf et al. 1998). Collectively, Syrian hamster and Eker rat data support our assessment that the renal epithelial proliferative lesions in male rats administered BZA are the result of prolonged renal tubular injury rather a direct hormonal effect.
Increases in renal tubular tumors in male rats administered BZA were also related to decreased body weight and increased survival. It is well documented that reduction in caloric intake and reduction in body weight increases survival and reduces chronic nephropathy in both SD and F344 rats (Geary and Cousins 1969; Keenan et al. 2000; Nold et al. 2001). Furthermore, when estrogenic compounds are given to rats, there is generally a dose-dependent decrease in body weight and food consumption (Wallen, Belanger, and Wittnich 2001; Eckel 2011). Reduction in body weight and food consumption has been proven to increase survival in ad lib fed rats. Much of this survival benefit is a result of a decrease or delay of naturally occurring renal disease, which is a major cause of mortality (Shiau et al. 1998). This may also account for the higher survival in groups administered BZA, as body weight decreases were also more pronounced with resultant increased longevity. Similarly raloxifene also demonstrated a dose-dependent increase in survival, that inversely correlated to the severity of tubular mineralization and CPN, and also had renal tumors in male rats at the high dose (CDER 1997). This relationship between body weight, CPN, and survival helps explain the dose-response relationship when male rats are administered estrogen or SERMs. The exogenous estrogen or SERM results in prolonged tubular cell injury with mineralization and regenerative proliferation, while the decrease in body weight and food consumption confers some benefit in reducing the severity of CPN. Thus, at lower doses, renal disease is more severe and progresses faster, resulting in lower survival. At a higher dose, renal damage occurs but is less severe or proceeds more slowly as a result of the benefit of lower body weight and prolonged survival. With increases in survival, there is increased time for the proliferative features of CPN, exacerbated by chronic tubular injury from mineralization, to progress from hyperplastic tubules to neoplasia.
Many chemicals are known to exacerbate the severity of CPN, and this interaction between chemical and CPN can result in an increased incidence of renal tumors in 2-year carcinogenicity studies (English et al. 1994; Hard 1999; Hard et al. 2007; Hard, Betz, and Seely 2012). Review of the pathologic entities associated with chronic renal failure in humans indicates that this rodent disease has no human counterpart (Hard, Johnson, and Cohen 2009). Because CPN is a rat-specific disease, increases in male renal tubular tumors from exacerbation of CPN by a test article in carcinogenicity studies has been postulated to have no relevance for extrapolation in human risk assessment (Hard and Khan 2004; Hard, Betz, and Seely 2012).
The finding of BZA-related renal tumors in male rats is consistent with criteria defining renal tumors in male rats secondary to enhanced CPN, rather than a direct response of chemical exposure (Hard 1998, 2002; Hard and Khan 2004; Lock and Hard 2004; Hard et al. 2007; Hard, Betz, and Seely 2012). These criteria are (1) the chemical is non-genotoxic, (2) male rats are more severely affected than females, (3) the chemical must exacerbate CPN to advanced lesions or end-stage kidney disease, (4) there is a slight to modest increase in renal tumors with a preponderance of adenomas of small size, (5) the tumors are primarily present in kidneys with the greatest CPN severity, (6) the preneoplastic foci are restricted to CPN affected kidney, and (7) there is no evidence of renal cellular injury in kidney unaffected with CPN.
In conclusion, administration of the SERM, BZA, to male rats for 2 years exacerbated spontaneously occurring CPN, and this interaction with advanced CPN led to renal tubular hyperplasia and neoplasia in male rats. The proposed mechanism was a test article–related exacerbation of CPN due to an estrogenic mineralizing effect of the SERM on renal tubules in male rats leading to advanced CM. The injury to the tubules exacerbated proliferative changes that occurred with spontaneous CPN, resulting in renal tubular hyperplasia and neoplasia in male rats.
Footnotes
Abbreviations
Acknowledgments
The authors would like to thank Debra Kirchner for managing the conduct of the 2-year rat carcinogenicity study, Peter Mann for serving as the study pathologist, Arthur Carter for providing statistical support of the 2-year carcinogenicity study, Dan Minck for providing project support, and Gerald Fisher for guidance with study interpretations.
Authors’ Notes
Carol A. Thompson’s contribution to the project occurred prior to joining Abbott Laboratories.
