Abstract
Eplerenone (Inspra®) is an aldosterone receptor antagonist approved for the treatment of hypertension and heart failure after a myocardial infarction.
Introduction
Aldosterone receptor antagonists such as Inspra (eplerenone) and spironolactone have proven to be life-saving drugs for patients with heart failure. This was completely unanticipated since when spironolactone was first discovered in 1955, it was believed to be just another potassium-sparing diuretic. During this era before molecular biology, aldosterone was believed to function only in epithelial tissues such as the kidney, colon, sweat, and salivary glands to regulate fluid and electrolyte balance. In 1987, Arriza and colleagues first cloned the human mineralocorticoid receptor (MR), and complementary DNA (cDNA) probing of human tissues revealed a startling revelation—MR was detected in nonepithelial tissues such as heart, brain, and blood vessels in higher concentrations than that in classical epithelial tissues like kidney (Arriza et al. 1987). In 1990, Brilla and coworkers first hypothesized that aldosterone activation of these receptors produced deleterious effects such as myocardial fibrosis (Brilla et al. 1990). More recent work in animal models from Rocha and colleagues suggests that the primary damaging effect of aldosterone may be the induction of vascular inflammation and injury of the small arteries and arterioles in end-organ tissues, preceding a reactive and reparative fibrotic process (Rocha et al. 2002).
Irrespective of the mechanisms involved, in 1999, the RALES (Randomized Aldactone Evaluation Study) trial unequivocally proved that the blockade of aldosterone receptors with spironolactone was lifesaving when added to the standard-of-care therapy of severe heart failure patients (Pitt et al. 1999). In RALES, spironolactone reduced the risk of both morbidity and mortality among patients with severe heart failure at a dose that did not produce diuretic or hemodynamic effects. However, spironolactone even at the low doses used in RALES produces undesirable side effects since it is not completely selective for MR but also binds to androgen and progesterone receptors. In RALES, 10% of men receiving spironolactone complained of breast pain and gynecomastia compared to 1.5% in the standard-of-care group. Previous studies of spironolactone use for cirrhosis indicated that it could cause endocrine side effects such as loss of libido, menstrual irregularities, and impotence when given at high doses for longer periods of time (Greenblatt and Koch-Weser 1973). In 1987, chemists at Ciba-Geigy in Switzerland first synthesized eplerenone, an aldosterone receptor blocker with reduced potency but improved selectivity for the MR compared to spironolactone (deGasparo et al. 1987). GD Searle acquired development rights to eplerenone in 1993, and then in 1999, the company initiated the EPHESUS (
Another clinical study, EMPHASIS-HF (
Because pharmacology studies indicated a greater selectivity of eplerenone over spironolactone for the MR relative to other steroid receptors, the occurrence of prostate atrophy in dogs was unexpected in toxicity studies. This article describes the studies showing the effect and investigative strategies to understand it.
Materials and Methods
The studies described in this report were conducted at multiple locations during the mid-1990s. The original reports do not specifically indicate that the animal husbandry procedures met with existing regulatory requirements, but Institutional Animal Care and Use Committees were active at all the laboratories. All the beagle dogs used in these studies were acquired from Marshall Farms in North Rose, New York. The test article, eplerenone (code number SC-66110), was administered as neat micronized chemical powder in gelatin capsules. Doses of eplerenone were individualized based on an animal’s most recent body weight and were administered once daily. Control dogs received empty capsules. Three toxicity studies were conducted in dogs in compliance with US Food and Drug Administration Good Laboratory Practices regulations (GLP). Two investigational studies in dogs were non-GLP compliant because certain procedures were not validated, but record keeping was in accord with GLP principles.
In Vivo Studies
The dosages, numbers of animals, duration of treatment, and age of dogs at the study start for each of the
Eplerenone male dog study design.
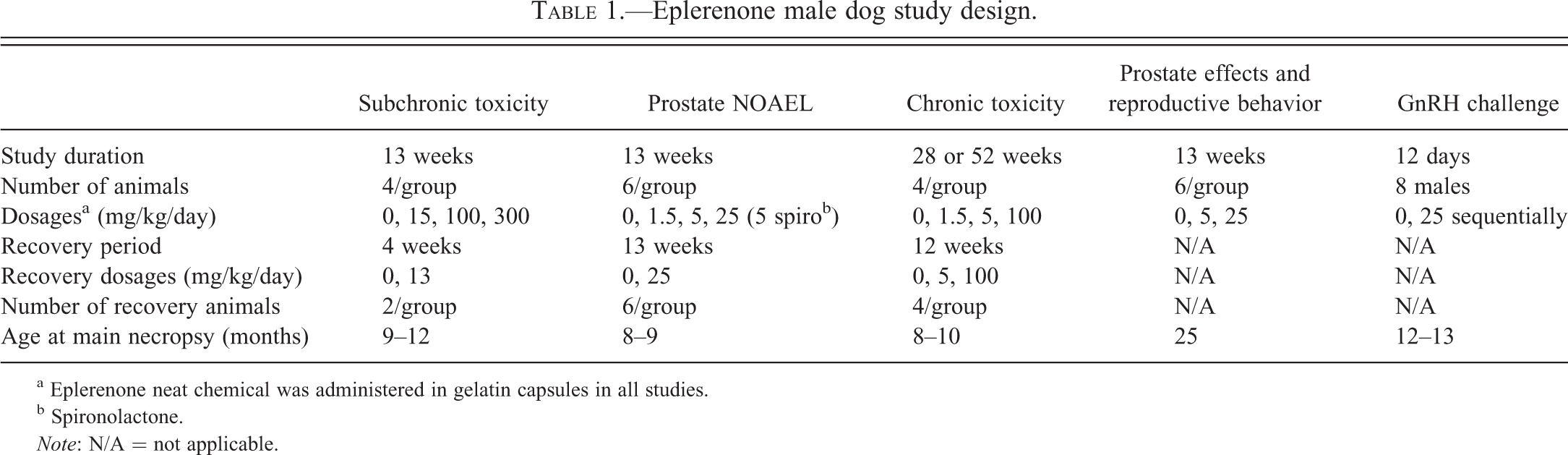
a Eplerenone neat chemical was administered in gelatin capsules in all studies.
b Spironolactone.
In a chronic toxicity study of eplerenone, some dogs were sacrificed at the end of 28 weeks and others after 52 weeks of treatment. A recovery group was treated for 52 weeks and then held without treatment for 12 weeks. In addition to the standard toxicity study design parameters, blood samples were collected and serum was analyzed for the following hormones at several intervals during the treatment and recovery periods: aldosterone, cortisol, estradiol, LH, testosterone, and DHT. Because some of these hormones exhibit pulsatile secretion, 3 blood samples were collected at 1-hr intervals beginning approximately 1 hr post dose (first sample at approximately 10 a.m.). The 3 samples per dog were pooled prior to hormone measurements. The hormonal analyses were performed and evaluated by the Animal Reproduction and Biotechnology Laboratory at Colorado State University, Fort Collins, Colorado. Details of the methods are not available.
An investigative study was designed to assess the time of onset of prostate atrophy and to determine whether this was accompanied by changes in other reproductive functions and behaviors of male dogs. This study was conducted at Johns Hopkins University under the direction of Dr. John Strandberg (Zinc 2009). Dogs were administered 0, 5, or 25 mg/kg/day of eplerenone. Based on previous studies, 25 mg/kg/day was expected to result in prostate atrophy, while 5 mg/kg/day was expected to have no prostate effect.
Prostate size was estimated for all animals using transrectal ultrasound. Measurements taken during the pre-dosing period (Weeks −6, −3, and −1) were used for assignment of dogs to treatment groups. During the treatment period, prostate ultrasound measurements were also made during weeks 1, 2, 4, 6, 9, 11, and 14. Transrectal ultrasound measurements were conducted in the transaxial orientation (to obtain the width of the prostate) and the sagittal plane (to obtain the length and depth). Prostatic volume was calculated by width (cm) × length (cm) × depth (cm). Prostatic weight was estimated using the following nomogram (Juniewicz et al. 1989): prostate volume (cm3 × 0.602) + 1.16 = estimated prostate weight (g). Assessment of libido and ejaculation was done by manual stimulation of the penis without the presence of a bitch in estrus. During the pretreatment and treatment phases, libido was assessed at every ejaculation attempt at weeks −7, −5, −3, −1, 2, 4, 6, 8, 10, and 12. Scoring was based on a number system: 0 =
Semen was collected during the pretreatment period at weeks −3, −2, −1 and biweekly during treatment at weeks 2, 4, 6, 8, 10, and 12. Ejaculation was induced by manual massage. Semen sample volume was recorded, and semen was evaluated for sperm count and motility. Attempts were made to evaluate sperm morphology, but these efforts were discontinued after determining the staining technique was inadequate. The daily sperm production and epididymal spermatozoal reserves were determined from the homogenates of testicular and epididymal tissue as previously described in dogs (Olar, Amann, and Pickett 1983).
Jugular vein blood was processed to serum to measure the concentrations of aldosterone, cortisol, testosterone, DHT, and LH. Samples were collected during pretreatment weeks −7, −6, and −2 and during treatment weeks 1, 2, 3, 4, 7, 8, and 12. The latter samples were collected at 1, 2, and 3 hr prior to dosing. Equal volumes of serum from each of the time points were pooled to minimize effects of circadian variation. A single sample was also collected 6 hr after dosing during each of weeks 1, 3, and 12; these were analyzed for cortisol, testosterone, and DHT, but sample volumes were insufficient to analyze for aldosterone or LH.
At the scheduled necropsy, the left anterior and right posterior quarters of the prostate were frozen in liquid nitrogen and used for analysis of tissue aldosterone, testosterone, and DHT concentrations. All serum and tissue hormonal analyses were performed and evaluated by the Animal Reproduction and Biotechnology Laboratory at Colorado State University, Fort Collins, Colorado. Details of the methods are not available.
A second investigative study was conducted in male beagle dogs to determine the effects of eplerenone on LH, testosterone, and DHT in response to gonadotropin-releasing hormone (GnRH) stimulation. The GnRH stimulation was used to remove the diurnal variation in hormone concentrations by maximally stimulating secretion. Each animal served as its own control. The study included a 2-week baseline period followed by a 12-day period when the dogs were administered 25 mg/kg/day of eplerenone. During the baseline period, serum was collected on days 1, 3, and 5. On day 8, GnRH (Factrel, Fort Dodge, IA) was injected intramuscularly (2.2 µg/kg). Serum was collected 15, 30, 60, and 90 min after the injections and again on days 10 and 12. Eplerenone treatment began on day 15 and continued through day 26. GnRH injections were administered again on day 22 (1 week after the initiation of eplerenone treatment) and serial serum collections were made with an identical schedule as those during the baseline period.
In Vitro Investigative Studies
To determine whether prostate atrophy could be explained by blocking the androgen receptor, competition binding assays were conducted by measuring the displacement of tritiated DHT from cytosolic fractions prepared from untreated canine prostate. Displacement was compared among eplerenone, spironolactone, and unlabeled DHT. The androgen receptor binding assay was modified from a published method (George and Noble 1984). Frozen whole canine prostates were thawed on ice, minced, and homogenized in 3 volumes of a 10-mM Tris-HCl buffer, pH 7.4, containing 1.5 mM ethylenediaminetetraacetic acid (EDTA), 10 mM sodium molybdate, 0.1% (vol/vol) monothioglycerol, and 10% glycerol (TEGMSH buffer) using a Polytron tissue homogenizer (Beckman). Following ultracentrifugation at 135,000
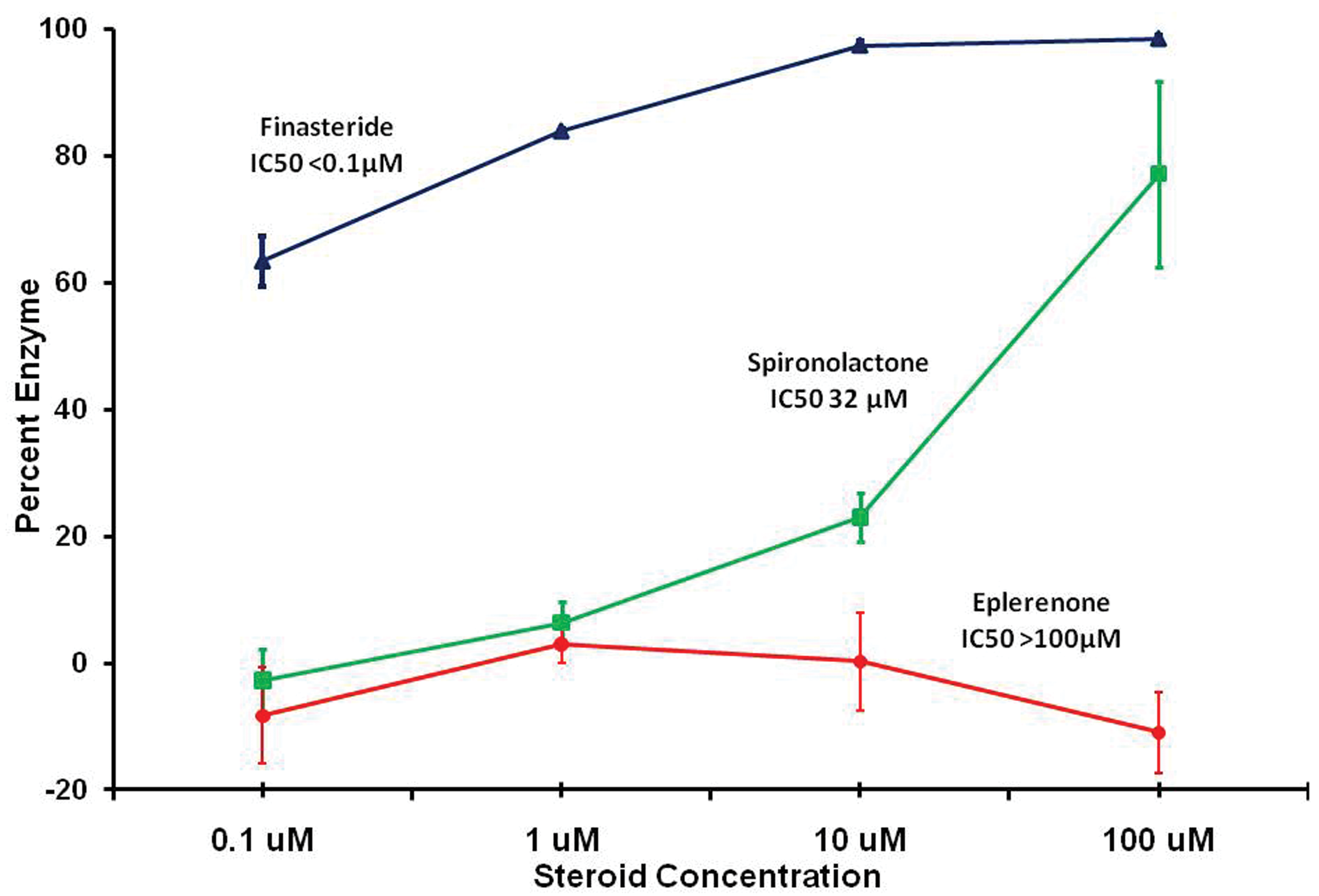
The ability of eplerenone to inhibit 5-alpha-reductase, the enzyme that converts testosterone into DHT, was also examined
Results
In the subchronic study (13 weeks of treatment, followed by a 4-week recovery period in some dogs), 2 dogs (one male and one female) administered the high dosage of 300 mg/kg/day were sacrificed during the first 4 weeks of treatment as the result of vomiting and weight loss. These dogs had electrolyte disturbances including hyponatremia (Na 136–140 mmol/L) and/or hyperkalemia (K 4.8–5.4 mmol/L) that were interpreted as being pharmacologically mediated. Aside from these 2 dogs, body weights were not affected by treatment with eplerenone and clinical signs were not evident. Slight perturbations of serum electrolytes were noted at all dosage levels, primarily early in the study (day 3). Serum aldosterone levels were markedly increased in dose-related fashion when measured during week 12. In males, for example, the control group serum aldosterone concentration was 14.7 ± 17.3 pg/mL compared with 1431 ± 742.4 pg/mL at 300 mg/kg/day of eplerenone. Aldosterone values in females were comparable to those of the males. Serum testosterone and estradiol were not affected by eplerenone. In female dogs, the NOAEL was 100 mg/kg/day based on the animal at 300 mg/kg/day that lost weight and was sacrificed early. The most notable effect of eplerenone in this study was dose-related reduction in prostate size and weight (Figures 1 and 2), at all dose levels. An NOAEL was not established for male dogs administered eplerenone in this study.
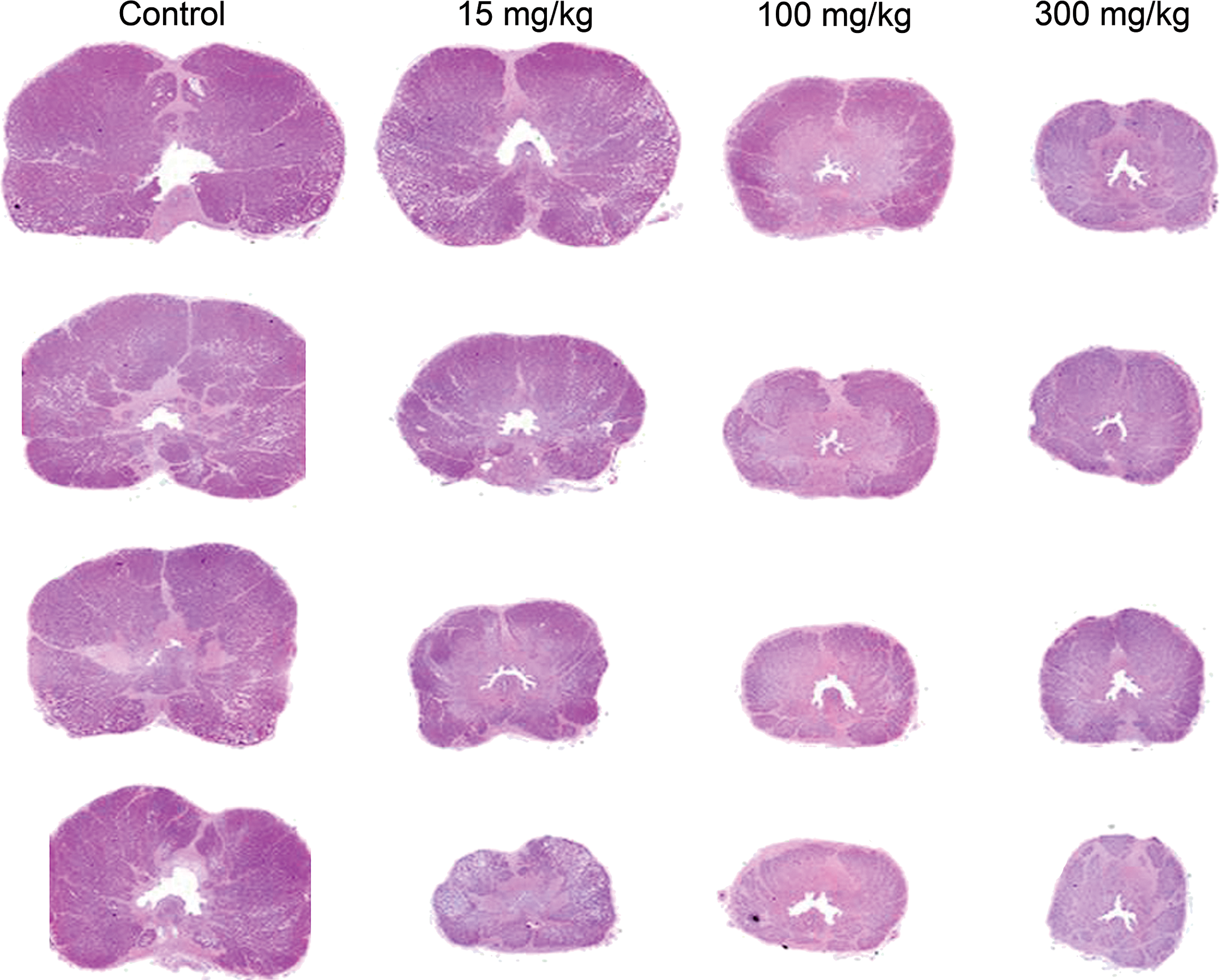
Reduced prostate size after 13 weeks of daily eplerenone treatments. Transverse sections of prostate made at the point of largest diameter.
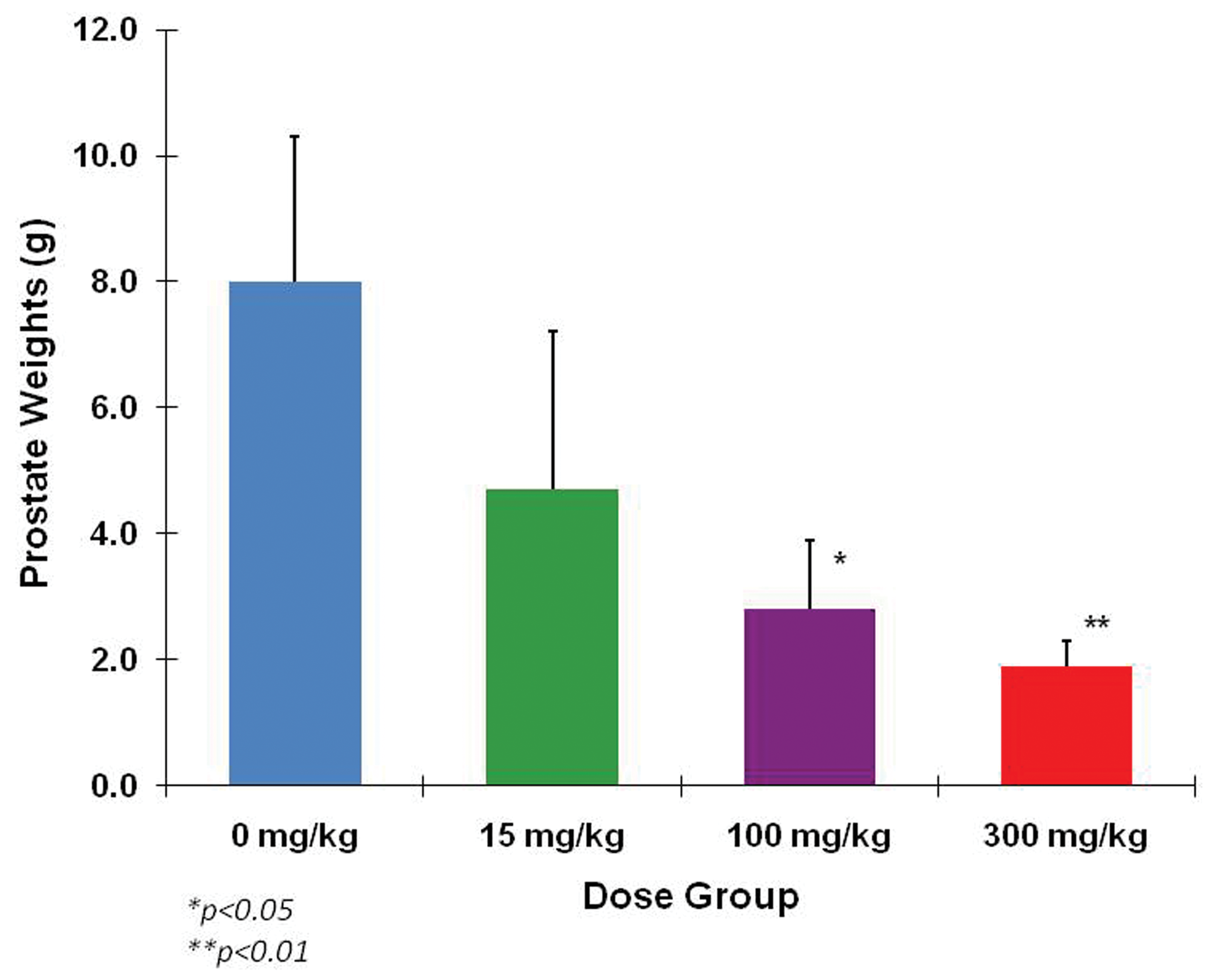
Reduced prostate weights after 13 weeks of daily eplerenone treatments. There were statistically significant weight decreases at both of the high doses but not at the low dose. Data are expressed as mean
The prostate glands were sectioned for histological examination in a transverse plane at the point of greatest diameter (Figure 1). All mid- and high-dosage males and 3 of 4 low-dosage males had varying degrees of histological change in the prostate. Prostate atrophy was reflected as a smaller cross-sectional area of the glands. The secretory cells were decreased in height and their cytoplasmic content was reduced. The connective tissue stroma was more prominent. The prostate gland in the one high-dose male that was held for a 1-month reversal period was less severely affected than the prostates of the high-dose dogs sacrificed early or at the end of the dosing period, suggesting the reversibility of the change.
A second 13-week study was conducted in male beagles to determine the NOAEL for the prostate effect. This study included 1 dose intended to cause prostate atrophy (25 mg/kg/day) and 2 doses intended to define the NOAEL (1.5 and 5 mg/kg/day). A group administered spironolactone at 5 mg/kg/day was included for comparison. Prostate weights (Figure 3) were clearly reduced by the 25 mg/kg/day dose of eplerenone and by the 5 mg/kg/day of spironolactone. The mean prostate weight at the 1.5 mg/kg/day dose of eplerenone was clearly not different from the control group and so was a definitive NOAEL. The mean prostate weight at 5 mg/kg/day of eplerenone was less than that of the control group but the difference was not statistically significant. Whether the NOAEL for the prostate effect was 1.5 or 5 mg/kg/day was uncertain at that time.
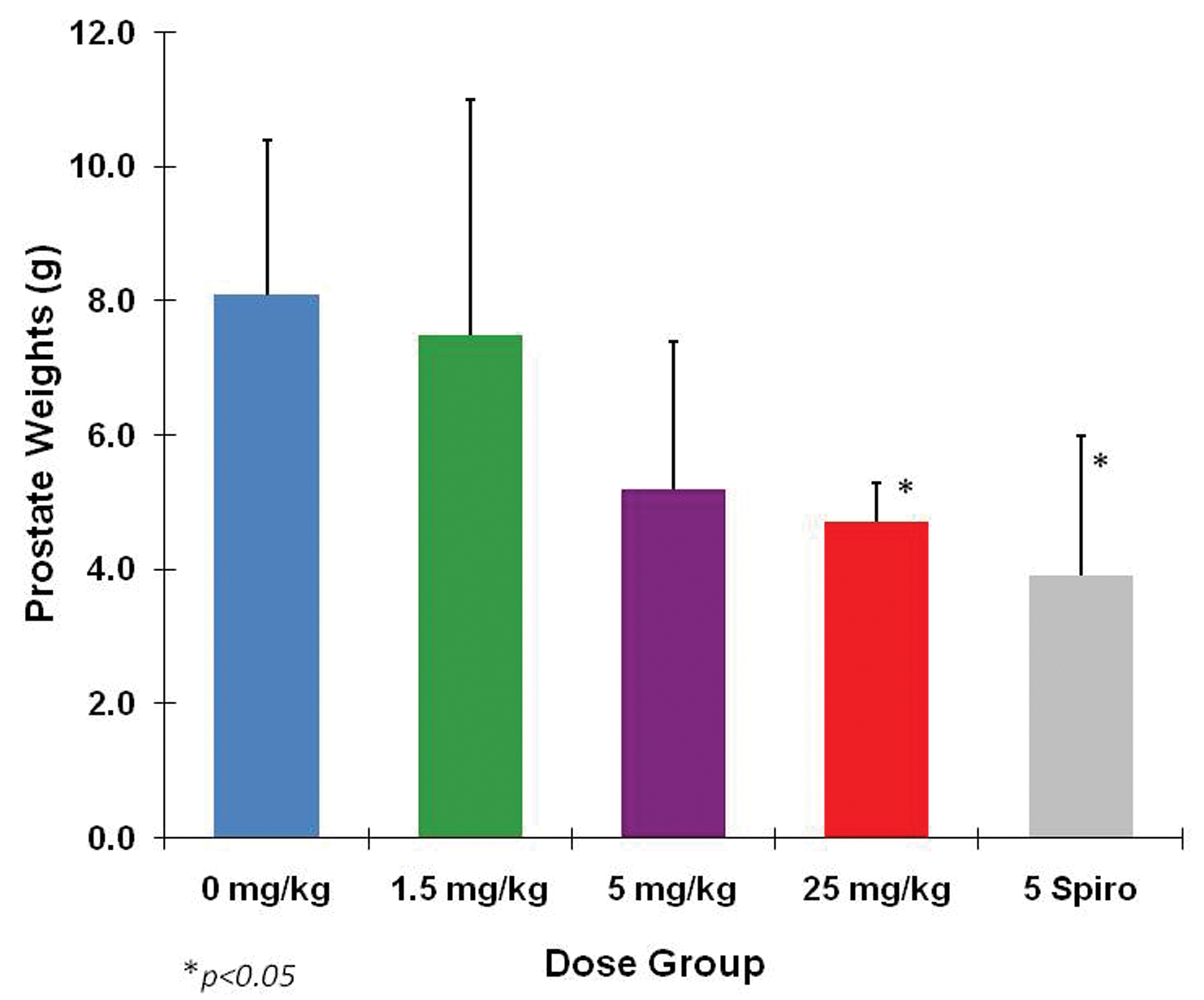
Prostate weights from a 13-week NOAEL study with spironolactone as a comparator. Weights at 25 mg/kg eplerenone and 5 mg/kg spironolactone were statistically significantly reduced. The decrease in weights in the 5 mg/kg eplerenone group, although not statistically significant, was suspicious. However, it was the 5 mg/kg spironolactone group that demonstrated the greatest effect. Data are expressed as mean
In the chronic toxicity study, the male and female beagles were administered 1.5, 5, or 100 mg/kg/day of eplerenone. Animals were sacrificed after 6 or 12 months of treatment and after 12 months of treatment followed by 3 months of recovery. For female dogs, the NOAEL remained at 100 mg/kg/day as it had been in the 13-week study. For males, prostate atrophy was again the primary adverse effect, occurring at 100 mg/kg/day by the end of 6 months (Figure 4). It was clear in this study that 5 mg/kg/day was the NOAEL. Prostate atrophy at 100 mg/kg/day was fully reversible within 3 months after the cessation of eplerenone treatment. Serum aldosterone was markedly elevated throughout the treatment period at 100 and 5 mg/kg/day and returned to control levels by the end of the recovery period (Figure 5). Other hormones, including cortisol, estradiol, LH, testosterone, and DHT, were not affected.
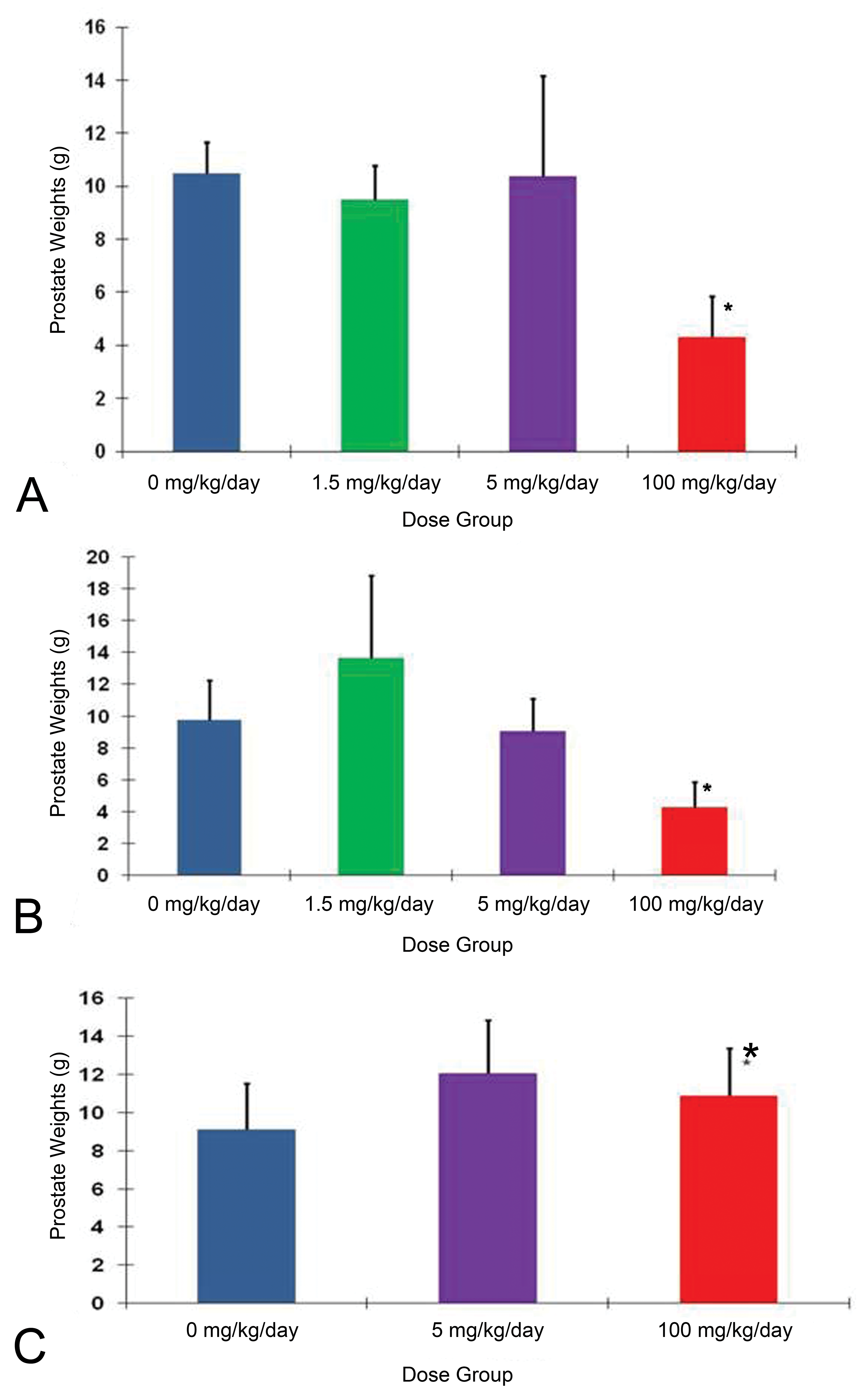
Prostate weights from 1-year eplerenone study. A, Prostate weights at 6 months’ interim sacrifice. B, Prostate weights at 1-year sacrifice. C, Prostate weights after 13 weeks recovery period. Clearly, there was no effect in the 5 mg/kg/day group. *Effects seen at 100 mg/kg/day did not progress between 6 months and 1 year and were fully reversible within 3 months after cessation of treatment. Data are expressed as mean
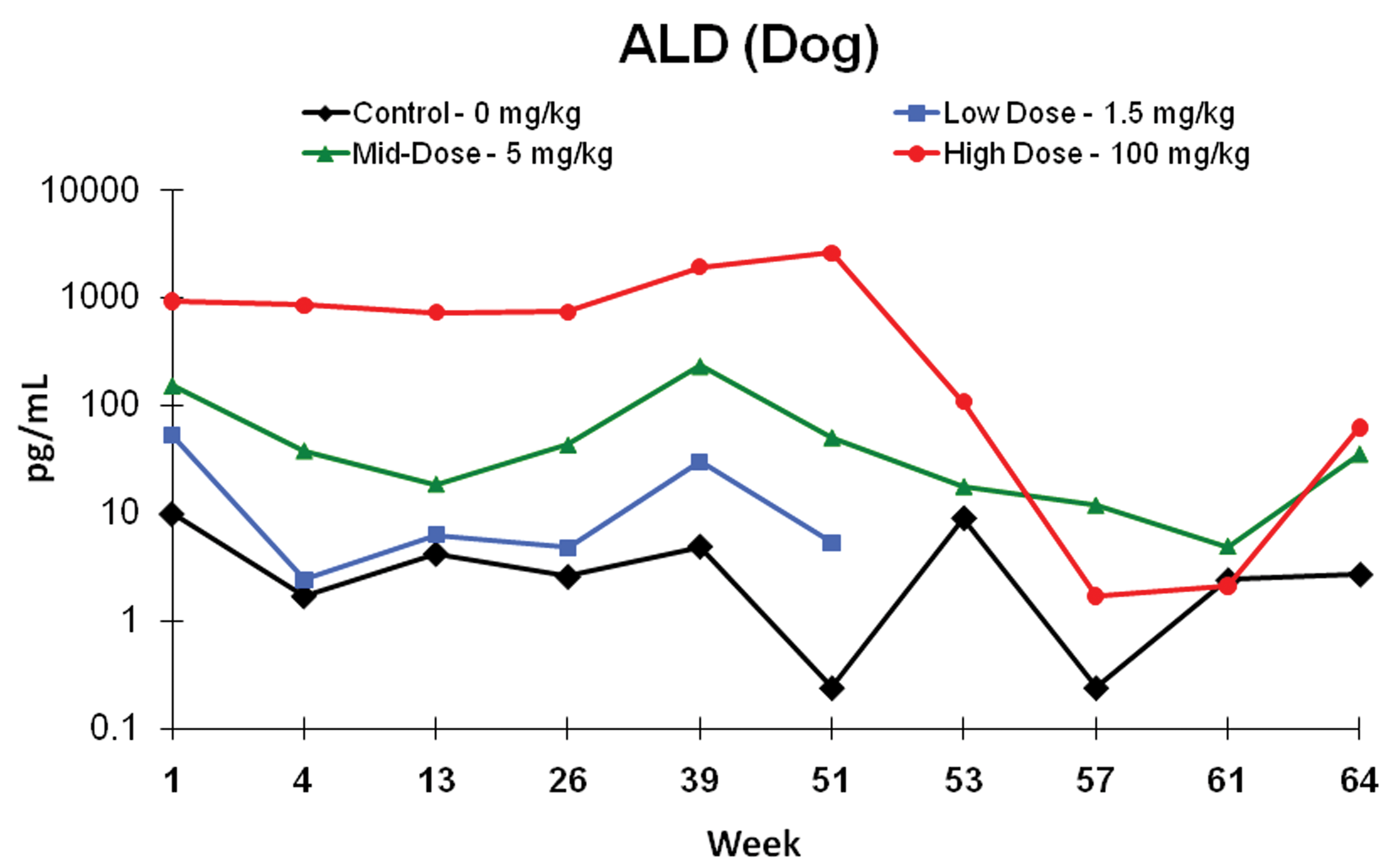
Aldosterone levels from 1-year eplerenone study Dose-related increases were evident throughout the treatment period and declined during the recovery phase. Data are expressed as mean ± standard deviation (
Because of the consistent effects on the prostate, it was important to determine whether there was any effect on male dog reproductive behavior and semen. This concern led to the design of the progressive prostate effects and reproductive behavior study at Johns Hopkins. For this study, fully mature male beagles (25 months of age at necropsy) were used. The eplerenone doses were selected to provide one dose that would likely produce prostate atrophy (25 mg/kg/day) and another dose that would not (5 mg/kg/day). Intrarectal ultrasound measurements were introduced in the nomogram described earlier to calculate the estimated prostate weights of the dogs during the course of 13 weeks of treatment. The estimated prostate weights of dogs receiving 25 mg/kg/day began to decline within 2 weeks of the start of treatment and continued declining throughout the treatment period (Figure 6A), whereas the estimated prostate weights of the 5 mg/kg/day were always comparable to the control group. The actual prostate weights at necropsy reflected the ultrasound data (Figure 6B). The 25 mg/kg/day dose group had a small decrease in semen volume but not in other measures of semen quality. Serum aldosterone was dose-relatedly increased by eplerenone but other hormones (cortisol, testosterone, DHT, and LH) were not affected. Furthermore, eplerenone treatment did not affect the libido of the dogs even when prostate atrophy was present.
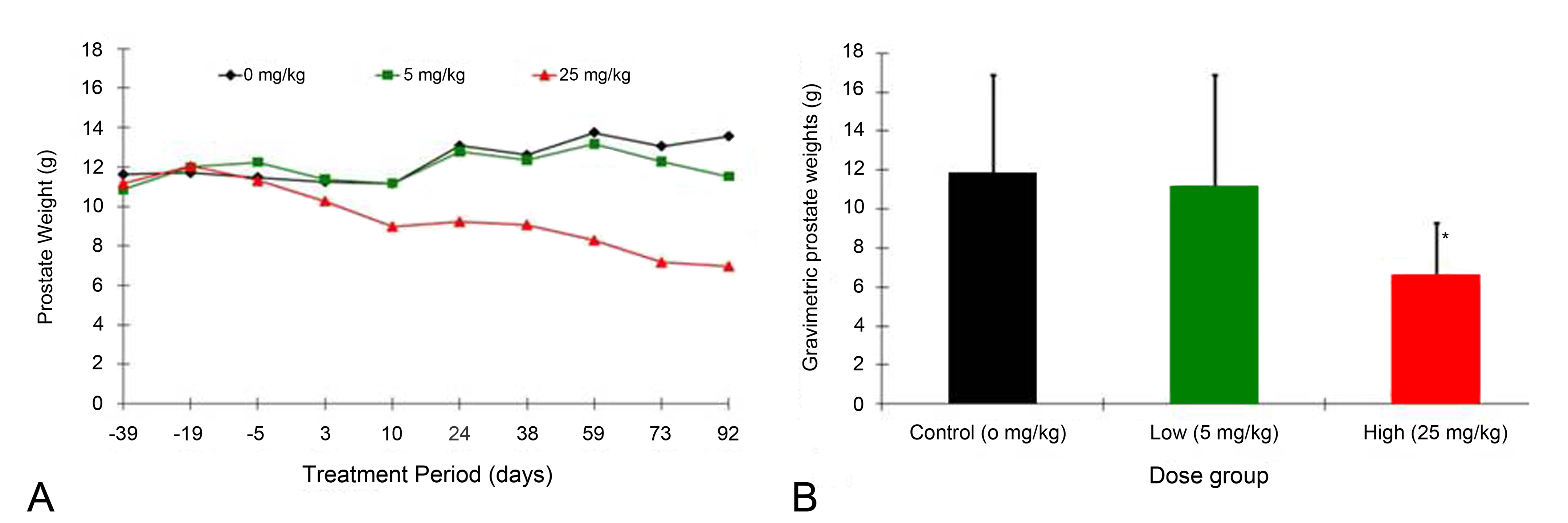
Prostate weights from the 13-week reproductive behavioral/function study. A, Prostate weights estimated by intrarectal ultrasound derived from ultrasound measurements of the gland and converted into weight via a nomogram. B, Prostate weights at necropsy. Prostate atrophy at 25 mg/kg/day was evident by ultrasound (5A) as early as day 10 posttreatment and was statistically significantly at *
The GnRH stimulation study showed no difference in hormone responses from baseline in the same dogs after they had been administered 25 mg/kg/day of eplerenone for 1 week (Figure 7). Thus, the dogs’ ability to synthesize and secrete these hormones was not impaired by eplerenone.
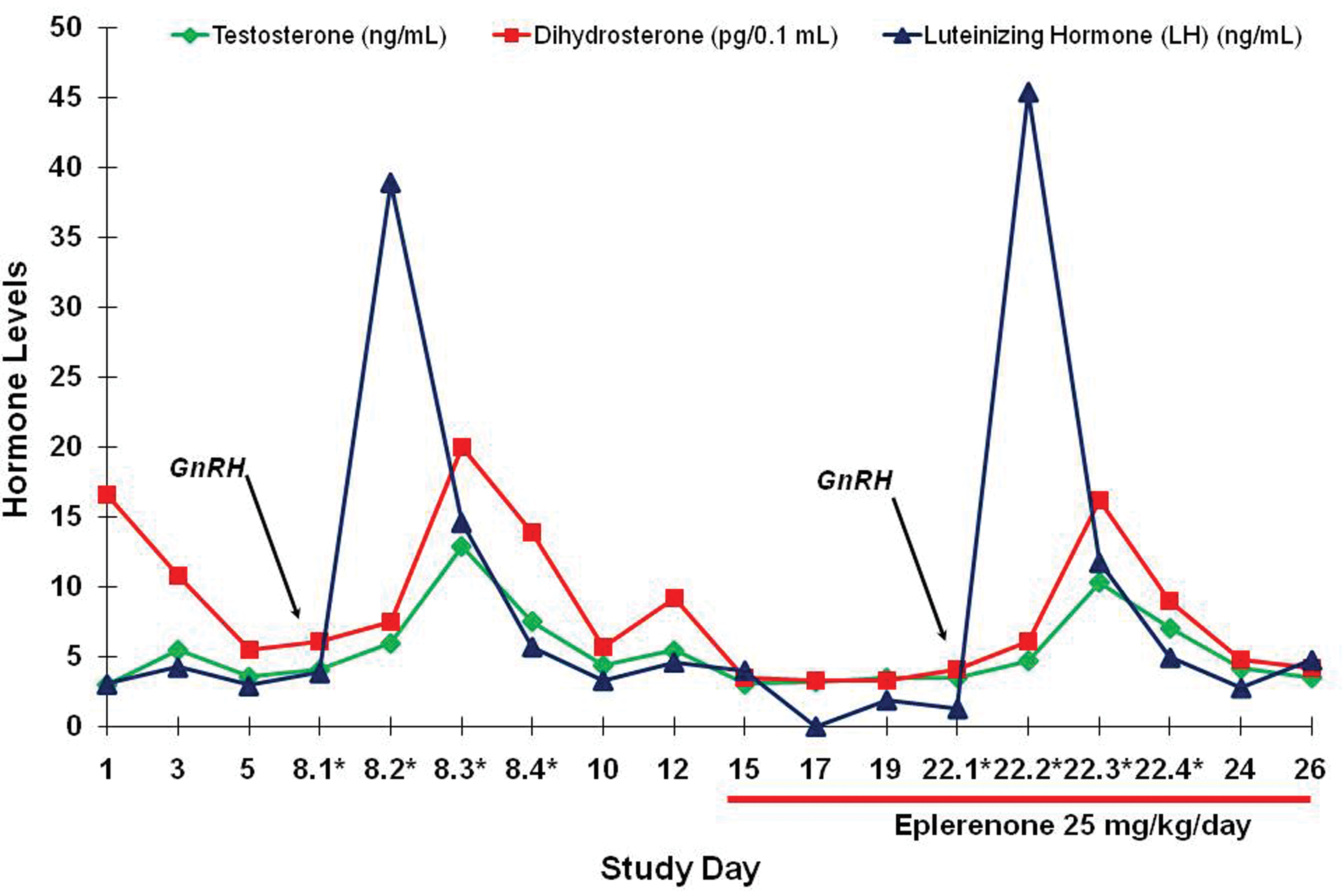
Gonadotropin-releasing hormone (GnRH) stimulation test. Dogs demonstrated similar responses pre- and posttreatment of eplerenone.
An
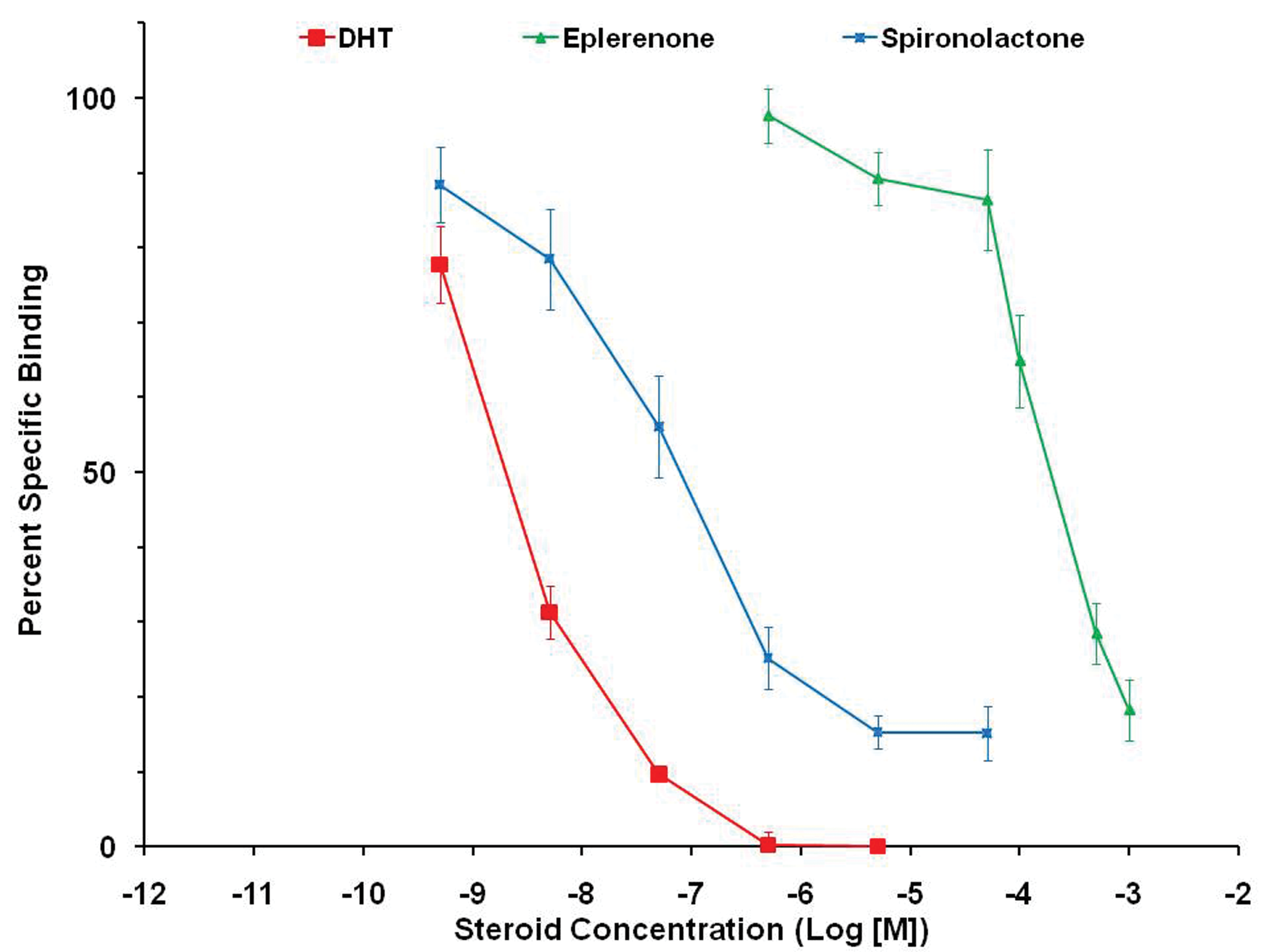
Comparative binding of eplerenone and spironolactone at the androgen receptor compared to dihydrotestosterone ([DHT] positive control). Spironolactone is 1,000 times more potent (left of eplerenone).
Discussion
Eplerenone was studied in the Beagle dog in a series of studies that ranged from 2 weeks at 100 mg/kg/day, 13 weeks at dosages up to 300 mg/kg/day, and 1 year at dosages up to 100 mg/kg/day. There were no deaths or overt clinical signs in dogs at dosages up to 100 mg/kg/day for up to 1 year (AUC multiples for both sexes averaged 20×). Two 13-week toxicity studies in Beagles demonstrated dose-related prostate atrophy at dosages ≥15 mg/kg/day. One of the studies also showed prostate atrophy with spironolactone at 5 mg/kg/day. The NOAEL for eplerenone was established as 5 mg/kg/day. The degree of prostate atrophy did not increase between 13 weeks and 52 weeks of administration. The prostate atrophy was reversible even after 1 year of treatment at 100 mg/kg/day.
Decreased weight of the dog prostate gland was the most sensitive indicator of eplerenone’s side effects in any species, that is, effects not mediated by its intended antagonism of the MR. Histological evaluation of the prostate glands showed atrophy of the epithelial and stromal components. This atrophy tended to be most prominent in the central (periurethral) portion of the gland, which is known to be the portion most sensitive to growth factors in dogs and men (Juniewicz et al. 1994; Monti et al. 1998). The histological appearance of the affected prostates presented no unique features. The degree of atrophy corresponded to the organ weight changes. Since organ weight is a more objective measure than histological assessment, the weight of the prostate is considered to be the definitive measurement of eplerenone’s effects on this organ.
Studies of the mechanisms of prostate atrophy were undertaken in dogs and with dog prostate tissue
The hypothesis that eplerenone or the secondary rise in serum aldosterone might interfere with the 5-alpha-reductase enzyme to reduce the production of DHT from testosterone is not supported by the
The available evidence supports a conclusion that the mechanism for prostate size reduction in dogs is by blockade of androgen receptors at suprapharmacological concentrations of eplerenone. An
Although aldosterone in dogs was somewhat more potent than eplerenone in binding to androgen receptors
It is concluded that mild antiandrogenic effects can occur at dosages of eplerenone that produce suprapharmacological exposure concentrations. Dogs were much more sensitive to the antiandrogenic effects of eplerenone than other species such as rats and mice. The reasons for the species differences in susceptibility were not investigated. Humans are unlikely to achieve plasma levels of eplerenone that could block androgen receptors. Spironolactone has greater antiandrogenic activity than eplerenone both
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
Acknowledgments
The authors thank the many scientists who contributed to the work described in the article including (from Searle/Pharmacia/Pfizer) James Andress, Chyung Cook, William Kelce, Arthur Roth, Peter Senese, Anita White, Clynn Wilker, Ann Zaizer, (from Johns Hopkins University) John Strandberg, Julie Crone, (from Covance) Mark Walker, and Borg Ulland.
