Abstract
Gill histological changes were used as a biomarker to evaluate the health of yellowfin seabream, Acanthopagrus latus (A. latus), collected from different sites in Musa Creeks including: (A) Petrochemical, (B) Jaafari, (C) Ghazaleh, (D) Majidieh, and (E) Zangi creeks. Also Genaveh with relatively clean water was selected as reference. The gills of 60 A. latus were fixed in Bouin’s solution for 12 hr, dehydrated in a graded series of ethanol, and embedded in paraffin. Five micrometer thick sections were stained in H&E and Periodic Acid Schiff for microscopic examination. Also, the presence of gill histological alterations was assessed by the histopathologic alteration index (HAI). The gill morphological abnormalities include mucus secretion increase, debris, blood plaque, and shortening of filaments. The gill pathological changes included lamellar cells hyperplasia, aneurysm, lamellar fusion, and epithelial lifting. The HAI means were varied from moderate to severe in sites A and B, thus these sites are considered as being of low quality. Some severe pathological alterations were observed in site D, but their distribution was lower than sites A and B. The least HAI means of sites C and E demonstrated their good environmental quality. The results suggest that there is close relation between amounts of pathological alterations and environmental contamination.
Keywords
Introduction
Aquatic environments in the vicinity of industrial units are highly at risk of contamination by pollutants such as petroleum chemicals, solvents, oils, and heavy metals (Vinodhini and Narayanan 2009). Discharging large amounts of contaminations by adjacent industries into the aquatic systems leads to harmful effects on aquatic environment and organisms (Mc Glashan and Hughies 2001). Due to numerous histopathological and morphological alterations of the aquatic animals, pathological studies could be widely used as biomarkers to estimate the effects of pollutants on the health of aquatic animals such as fish exposed to cadmium (Thophon et al. 2003). The gills of the fish comprise a multifunctional organ and play a major role in the excretion of the nitrogenous waste, regulating the exchange of salt and water, respiration, and acid-base regulation; and because of direct and constant contact with the environment, they are simply affected by contaminants (Saenphet, Thaworn, and Saenphet 2009). Early researches on yellowfin seabream Acanthopagrus latus (A. latus) showed that changes in the morphology of gill filaments can be used to indicate the effect of pollutants such as mercury on the marine environment (Gregory et al. 1999).
Musa Creek, situated in the northwest of Persian Gulf and in the south of Iran, is considered a suitable place for passing the larval stages, spawning many types of aquaculture and commercial fisheries. It is also the habitat of yellowfin seabream (A. latus). Musa Creek with the different branches is also called Mahshahr Creeks. This area is contaminated with large amounts of different kinds of pollutants, especially heavy metals such as Hg, Cd, Zn, Cu, and Pb and polycyclic aromatic hydrocarbons (PAHs) discharged into it by surrounding industries located in the coastlines of some branches. Previous studies affirmed that the cast-off of the waste effluents from petrochemical and color alkali industries and oil jetty are the main source of water contamination in some parts of Musa Creeks (Faghiri 2009; Abdollah pour 2010). According to these researches, these parts are classified as low-quality waters owing to the presence of high amounts of pollutants, especially heavy metals and PAHs (Faghiri 2009; Abdollah pour 2010; Babadi 2010). On the other hand, some branches are the most supreme Creeks as they are far from pollution sources, hence they are believed to be the cleanest parts of Musa Creek (Asar 2008; Faghiri 2009; Babadi 2010). Therefore, the purpose of the present study was to determine whether histological changes in gill could be used as a biomarker to evaluate the health of yellowfin seabream, A. latus, collected from different sampling sites in Musa Creek and the water quality of these selected sites in terms of the types and prevalence of gill pathological alterations present in A. latus.
Material and Methods
Study Area
The Musa Creek, located in the south of Iran and the north of Persian Gulf, is situated between longitudes 49 and 49° 20′ E and latitudes of 30° 15′ and 30° 32′ N (Nooranian 2005). It has received much attention because various industries and petrochemical units surrounded this part of Gulf and considerable amount of waste waters are discharged daily into it.
Five different sampling sites including (A) petrochemical station, (B) Jaafari Creek, (C) Ghazaleh Creek, (D) Majidieh Creek, and (E) Zangi Creek were selected that all are situated in Musa Creek (Figure 1). Genaveh Creek, located away from the Musa Creek, was selected as a reference site because it has relatively clean water based on various research. Previous studies on the analysis of contamination amounts of different branches of Musa Creek showed that sites A, B, C, and D are the most contaminated parts of Musa Creek due to the accumulation of huge amounts of domestic and industrial, especially petrochemical, effluents; conversely, the Zangi Creek is relatively free of contaminants. Temperature, pH, and dissolved oxygen were measured before sampling at each site.
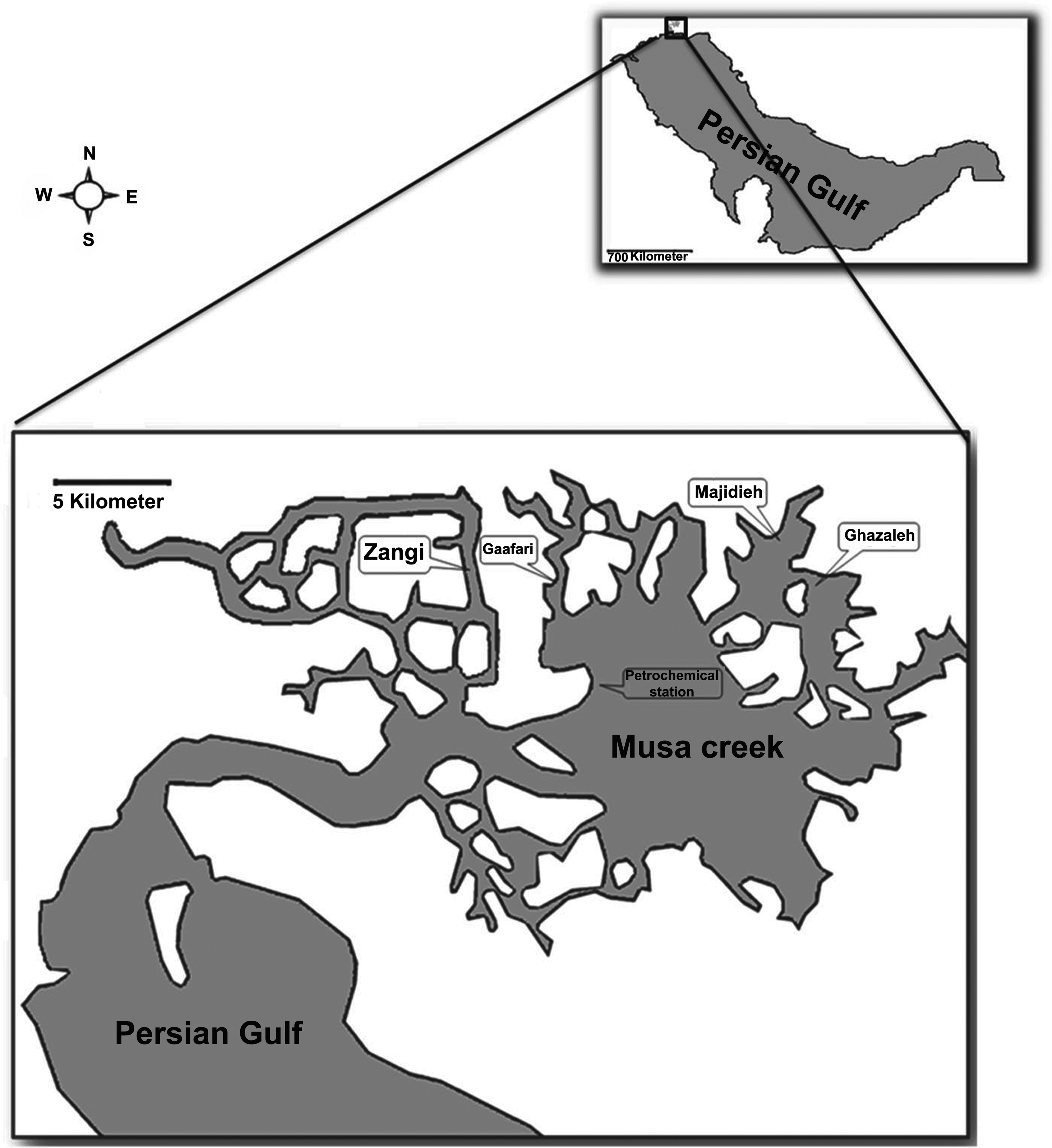
Map showing the region of sampling sites in the Musa Creek.
Sampling and Histological Study
Sixty male and female yellowfin seabream (172.9 g ± 37.3 g body weight and 20/01 cm ± 2/16 cm length) were collected from 6 sampling sites (A, B, C, D, E, and reference) during July 2012. In each site, 10 fish were anesthetized with benzocaine (0.1 g L−1) and then sacrificed by cervical section. The second gill arch of the left side of each fish was taken for morphological study using stereomicroscope (loop). Also pieces of gills, always from the second gill arch of the right side of the fish, were fixed in Bouin’s fluid for 48 hr. Then the tissue specimens were dehydrated in ascending concentrations of ethanol series, embedded in paraffin, and sectioned at 5 µm to 6 µm. The tissue sections were stained with H&E and Periodic Acid Schiff (PAS) and then microscopic evaluation was performed for histological and histometerical study.
Histometrical Study
For quantitative measurements, 5 individuals per stations and 5 slides from the gill of each were randomly selected for histometrical analysis. A total of 5 fields per slide were examined, each consisting of a portion of a gill filament and several lamellae. Lamellae epithelial thickness, lamellae length, and the number of the mucus and chloride cells were measured. All measurements were performed with light microscope using Dino lit lens (with Dino capture software, FDP2, Taiwan). All values were represented as means ± SEM. The significant difference between stations values was analyzed using analysis of variance (ANOVA) test.
Determination of Gill Histological Alteration Index (HAI)
The presence of gill histological alterations was assessed semiquantitatively by the histopathologic alteration index (HAI), which is based on the severity of the lesions (Flores-Lopes and Thomaz 2011). Classification of gill alterations was performed as scores of 0 to 3, where 0 = no alteration, 1 = slight alteration, 2 = moderate alteration, and 3 = severe alteration. The definitions slight, moderate, and severe alterations were modified from Poleksic and Mitrovic-Tutundzic (1994). Slight alteration (1) involves alterations, which do not change the normal function of the gill and healing of gill can occur with improvement of the environmental conditions; moderate alterations (2) are the most severe and harmful for the normal gill function. These elisions are reparable, but they can lead to severe alterations in the case of chronic pollution; and severe alteration (3) causes irreversible injury and recovery from such injury is not possible, even with improvement in the water quality (Table 1).
Gill histopathological changes indicating their respective stages of damage to the tissue.

Note: Stage I: do not alter the normal functioning of the tissue; Stage II: more severe; Stage III: very severe and cause irreparable damage; Flores-Lopes and Thomaz (2011) modified from Poleksic and Mitrovic-Tutundzic (1994).
A value of HAI was calculated for each animal using the following formula:
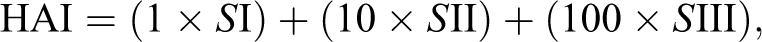
where I, II, and III relate to the number of stages of alterations 1, 2, and 3, respectively. HAI values between 0 and 10 show normal function of the organ; values between 11 and 20 indicate slight damage to the organ; values between 21 and 50 indicate moderate changes in the organ; values between 50 and 100 indicate severe lesions; and values above 100 indicate irreversible damage to the organ (Poleksic and Mitrovic-Tutundzic 1994).
Result
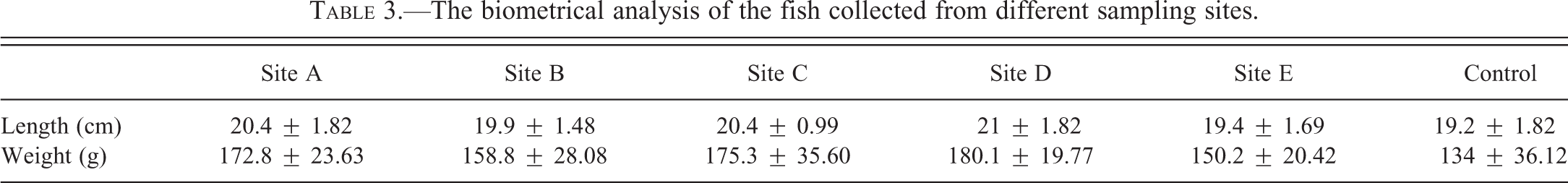
The results of water quality assessment in each site are represented in Table 2. There were no significant differences between different sampling sites for each parameter. Also biometrical analysis of the fish collected from different sampling stations is shown in Table 3. The length and weight of fish from different sites did not show significant differences.
The amounts of water physical–chemical parameters from each site.
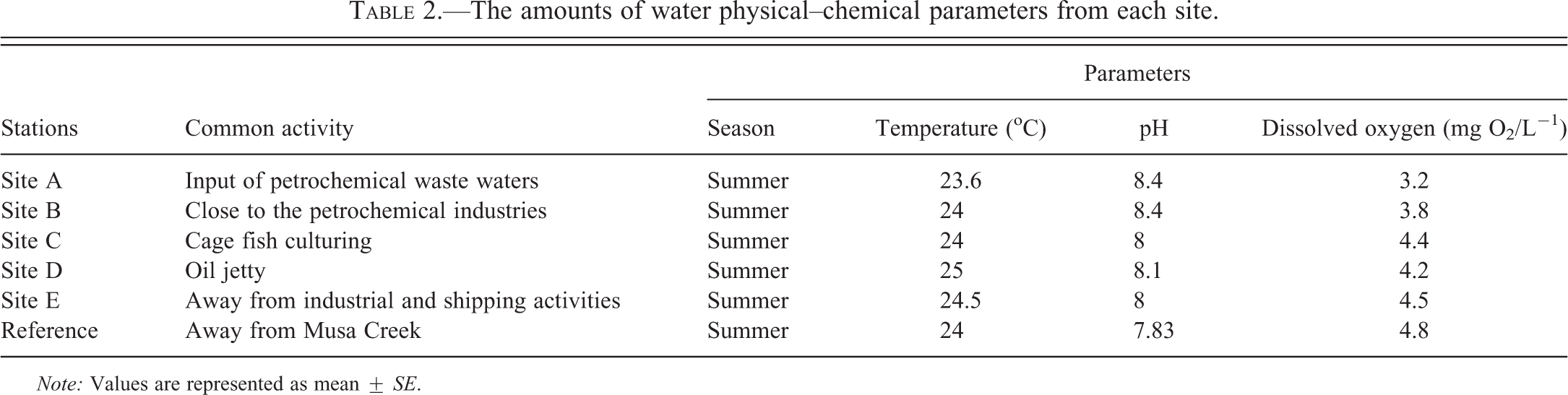
Note: Values are represented as mean ± SE.
The biometrical analysis of the fish collected from different sampling sites.
Morphological and histological analysis showed important alterations in the gills of the contaminated A. latus, which are detailed below.
Macroscopic Analysis
Gills had a normal morphological structure in control fish. Two rows of distinct and regular filaments were arranged perpendicularly on each gill arch. No lesions were observed in the gills (Figure 2).
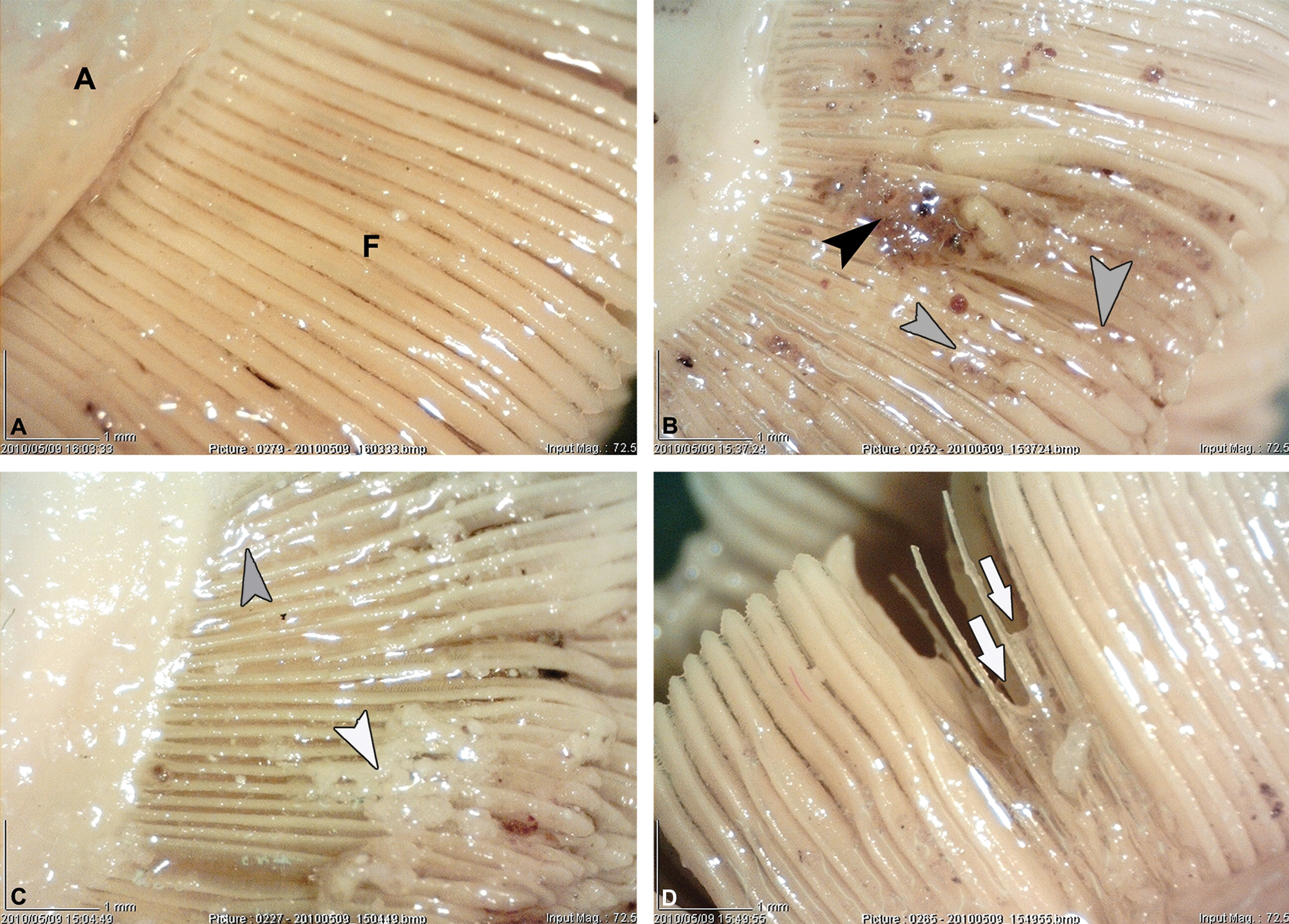
Morphological structure of the gill within the control A. latus, which possess distinct, regular filaments (F), gill arch (A), (×725); B and C. Photomicrographs of the most common morphological alterations of A. latus gills collected from different sites: Shortening of some filaments (white arrows), increase of mucus (gray arrowheads), debris on the filaments (white arrowhead), blood plaques on filaments (black arrowhead); (×725).
The most common abnormalities found in the morphological structure of the gills in contaminated fish collected from different sampling sites include increase of mucus secretion, fusion of some filaments, filament disorganization, debris, and losing or shortening of some filaments (Figure 2). However, the macroscopic changes in the gill of fish collected from each sampling site are demonstrated in Table 4.
The macroscopical and histopathological changes in the gill of fish collected from each sampling site.

However, the most severe gill morphological alterations were observed in the fish collected from site C (petrochemical station). On the other hand, the samples collected from site E (Zangi station) showed the minimum morphological changes. The following model was designed to show the degree of severity of morphological changes depending on the sampling sites: site A > site B > site D > site C > site E.
Microscopic Evaluation
Series of separate and regular lamellae are sited on the upper and lower surface of each gill filament in control fish (Figure 3). Normal epithelial cells were situated along the entire length of the filaments and lamellae (Figure 3A). The chloride cells and mucous cells were sparsely distributed in the filament epithelium (Figure 3A, B).
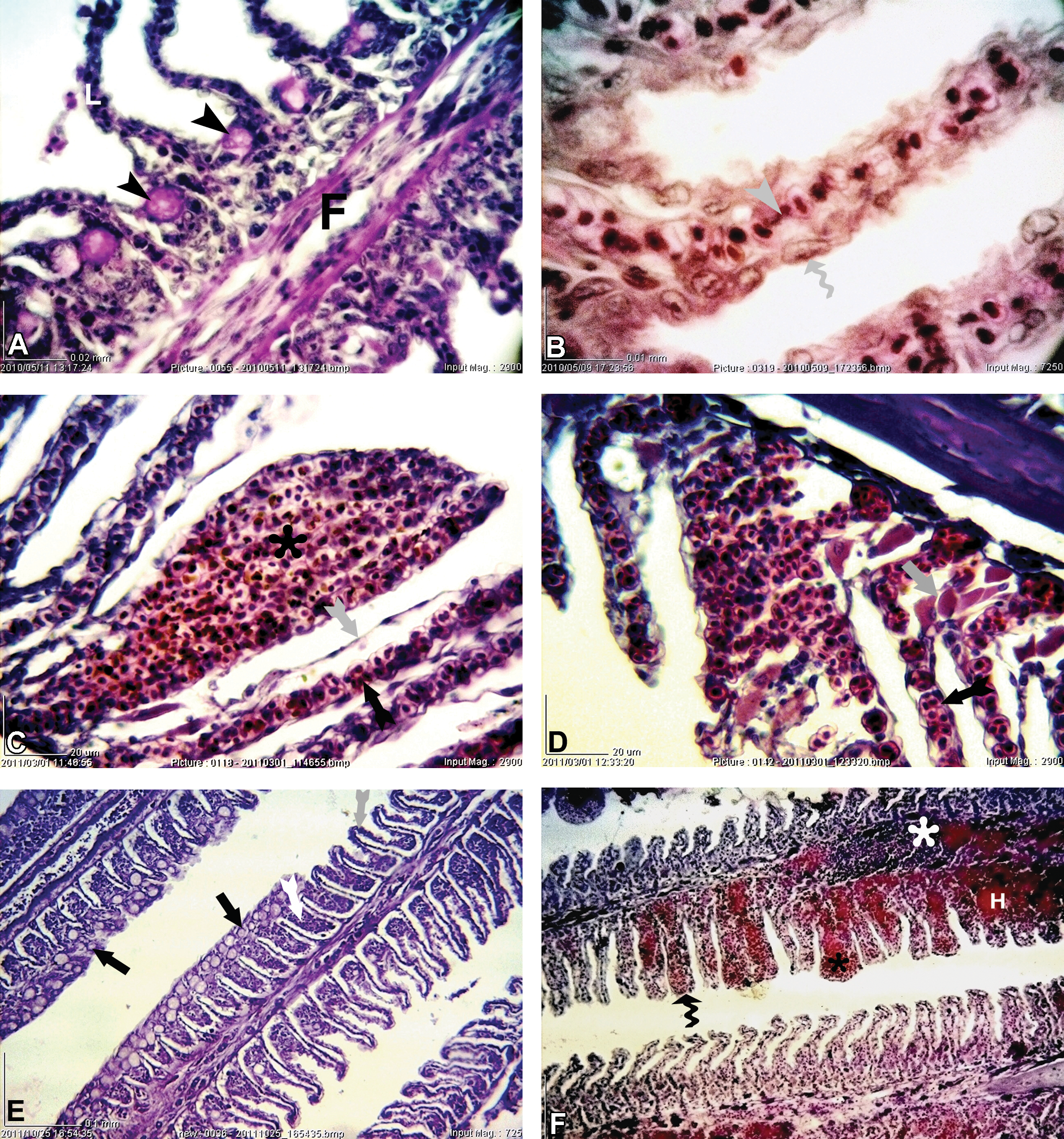
Photomicrographs of gill histological structure in A. latus collected from reference site (A and B) and polluted sites (C, D, E, and F); the filament (F), the lamellae (L), Chloride cell (white head), mucous cell (black arrowheads), pillar cell (gray arrowhead), lamellar epithelial cell (gray convoluted arrow), epithelial lifting and edema of the lamellae with enlarged sub epithelial spaces (gray forked tail arrows), lamellar aneurysm (black •) within the lamellae, telangiectasia (black forked tail arrows), increase in chloride cells (gray arrow), hemorrhage (H), leukocyte infiltration (white •), hyperplasia of the epithelial cells with fusion of the lamellae (white forked tail arrows), club shaping of gill lamellae (black convoluted arrows), increase in mucosal cells (black arrows); A (PAS; ×2,900); B (H&E ×7,250); C and D (H&E ×2,900); E and F (H&E ×725).
The most pathological changes found in A. latus collected from different stations include hypertrophy and hyperplasia of the lamellar epithelial cells with partial or complete fusion of some lamellae, epithelial lifting and edema of the lamellae with enlarged subepithelial spaces, club-shaped gill lamellae, lamellae with the marginal channel dilated, blood congestion, lamellar aneurysm and Telangiectasia, leukocytes infiltration, and increase of chloride and goblet cells (Figure 3C, D, E, F, and Table 4).
The number of chloride and goblet cells were significantly higher in the gills of fish from site A (p < .05, Figure 4A and B). The amounts of both cells were changed according to the following pattern in different sites: site A > site B > site D > site C > site E (Figure 4A and B). The mean epithelial thickness of filaments tended to increase from site A to E as follows: site A > site B > site D > site C > site E (Figure 4C). In fish from site A, mean epithelial thickness of filaments was significantly greater than others. On the other hand, there was no significant difference between thicknesses of the filament epithelium in fish from sites C, D, and E, although they were significantly different from the control value. Also, lamellar thickness was significantly increased according to the following pattern: site A > site B > site D > site C > site E.
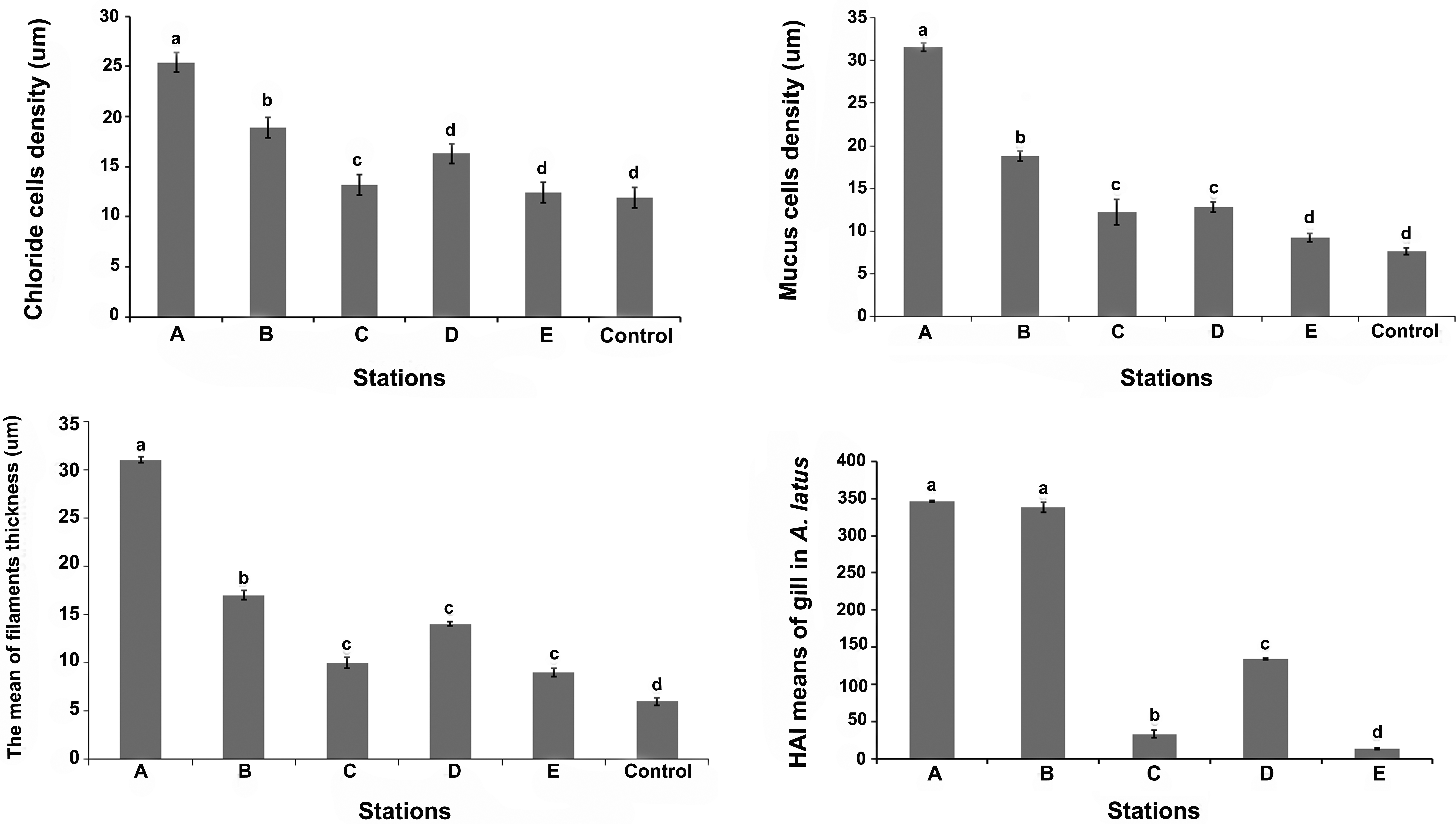
A. Changing of chloride cells density in A. latus gills in different sampling sites. B. Changing of mucus cells density in A. latus gills in different sampling sites. C. The mean of thickness of epithelial filament in different sampling sites. D. Means of HAI for the gills of A. latus collected from different sampling sites.
Higher frequencies of the most severe damages, considered to be at stages II and III in severity, were observed in the gill of fish collected from more contaminated stations (sites A and B). On the other hand, the least severe alterations (1) or no alterations in the gills (0) were found in site E.
The histopathologic alteration index (HAI) for the gills of fish collected from all sites varies from 13.33 ± 1.15 to 346.34 ± 1 (Table 5). Statistical analysis of HAI values of different sites showed that sites A and B had significantly higher HAI values (364.34 ± 1, 338.33 ± 6.65, respectively; p < .05, Figure 4D) than other sites. However, high mean of HAI was also observed at site D (134 ± 1), indicating the occurrence of severe irreparable lesions of the gills in the A. latus in these stations (A, B, and D). Therefore, based on the HAI results, these sites can be considered as having the worst environmental quality. The HAI mean value for A. latus from site C was significantly lower (33 ± 5.19) than other stations except site E (p < .05, Figure 4D). This result demonstrated the moderate changes to the organ in this site. The results showed that site E had significantly better environmental quality than others (p < .05) because of the low HAI mean (13 ± 1.15; Figure 4D), indicating slight damage to the organ.
The means of HAI in A. latus gills in different sampling sites.

Note. HAI = histopathological alteration index.
Discussion
The gill is a sensitive target organ and the first site of pollutants action (Wood and Soivio 1991). There is only a thin wall between blood and water in the gill, which is not only essential for facilitation of the gas exchanges but also exposes the gills to different pollutants that can damage the gills by the low-concentration environmental contaminants (Wood and Soivio 1991). Therefore, the pathologic assay of gill in fish exposed to environmental pollutants has been used as a useful biomarker for evaluation of the quality of aquatic ecosystems (Poleksic and Mitrovic-Tutundzic 1994).
There is a close relation between fish and their environment; therefore, they can be readily affected by any changes in chemical and physical conditions of the ecosystem, which could be evaluated as quantifiable physiologic parameters in fish (Flores-lopes and Thomaz 2011). Generally, the histopathological alterations such as hyperplasia and hypertrophy of the epithelial cells and lamellar fusion, epithelial lifting and edema in lamellae, telangiectasia and aneurysm, increase of chloride and goblet cells, and leukocyte infiltration observed in the gill of A. latus in the present study are the responses of the fish gill to the environmental irritants. According to Thiyagarajah et al. (1996), these alterations could be due to the contaminants or infectious conditions. Oliveira et al. (1996) reported that such alterations are nonspecific and could occur as a consequence of different types of pollutants such as inorganic mercury. These pathological alterations were shown by Coutinho and Gokhale (2000) in the gills of carps (Cyprinus carpio) and tilapias (Oreochromis mossambicus) exposed to the effluents of a wastewater treatment plant. Similar histological changes have also been reported in the gills of fish exposed to sodium dodecyl sulfate (Rosety-Rodríguez et al. 2002), lead (Martinez, Nagae, and Zaia 2004), and petroleum residues (Engelhardt, Wong, and Duey 1981). Epithelial lifting, hyperplasia, and hypertrophy of the epithelial cells, which may occur due to partial fusion of lamellae, are defense mechanisms resulting in the increased distance between the aquatic environment and the blood and act as a barrier to the pollutants' entrance (Fernandes and Mazon 2003). However, these changes in the gill structure lead to oxygen uptake impairing, and fish have to increase their respiration rate by compensating (Fernandes and Mazon 2003). Aneurysms, severe pathological alteration resulting from disturbance in blood flow, could be considered as a specific reaction of the gill to the toxic substances. In a condition like this, increased blood flows in the lamellae that occur from pillar cells damage; followed by dilation of marginal canal, blood congestion, and aneurysm. This alteration occurs when fish suffer a more severe type of stress (Rosety-Rodríguez et al. 2002). Winkaler et al. (2001) also found an increased frequency of aneurysms in specimens of Astyanax fasciatus and Astyanax altiparanae captured from contaminated waters.
The shortening of gill filaments and lamellae observed in A. latus in the current study is similar to the responses made by other contaminants, such as mercury (Jagoe et al. 1993). Abnormal increase of chloride cells in the interlamellar spaces is consistent with altered ion flux at the gills, as previously reported by Jagoe, Faivre, and Newman (1996). Several studies reported that the increase of these cells in fish is caused by a number of pollutants such as mercury and cadmium (Perry, Goss, and Laurent 1992; Jagoe, Faivre, and Newman 1996). Many water-dissolved contaminants have been reported to increase the number of goblet cells leading to considerable increase of mucus secretion, as observed in the gills of A. latus. This alteration can act as a barrier to reduce absorption of toxicants, but it can also result in the increase of blood–gas diffusion distance and gas exchange reduction (Saenphet, Thaworn, and Saenphet 2009). Camargo and Martinez (2007) observed alterations such as dilation of the marginal channel, hyperplasia of the secretory cells, and increase of mucus secretion in Prochilodus lineatus caged in an urban stream. Similar changes were also reported by Mohamed (2009) in the gills of Tilapia zillii and Solea vulgaris obtained from Lake Qarun, Egypt.
According to the results, the highest frequencies of gill pathological alterations were found in A. latus collected from A and B stations and the means of HAI were significantly higher in these sites compared to others (p < .05). HAI has a close association with degradation of environmental quality, particularly chemical pollution. Therefore, the sampling sites with the highest frequencies of alterations and HAI means seem to be more contaminated. The following pattern was identified for different sites on the basis of the means of HAI: site A > site B > site D > site C > site E.
As stated by previous studies, the discard of petrochemical effluents from adjacent industries is the main source of water contamination in Musa Creeks (Faghiri 2009; Abdollah pour 2010). According to the Faghiri (2009) and Abdollah pour (2010), the waters of site A receive a large load of contaminated effluent with petroleum chemical and heavy metals from petrochemical and color alkali industries, which surround this part of Creek. On the other hand, high similarity was found in the chemical contents and concentrations between A and B stations due to their proximity and high water exchange between them. The result showed that HAI means were within the ranges varied from moderate to severe (II and III) in both sites A and B. Therefore in accordance with the results, these sites could be considered as being of low quality because of high frequency of pathological changes (particularly moderate and severe alterations) and an increased HAI mean found in these stations. Other investigators also classified these sites as being of low quality due to the finding of elevated amounts of several pollutant agents such as heavy metals and PAHs (Faghiri 2009; Abdollah pour 2010; Babadi 2010).
Although some moderate and severe pathological alterations (such as aneurysm) were observed in site D (Majidieh), the distribution of such alterations and the mean of HAI were considerably lower than sites A and B (p < .05). However, the elevated HAI mean in this site reveals increase in the degradation of its water quality. The least HAI means detected in sites C (Ghazaleh) and E (Zangi) demonstrated good environmental quality. As indicated by prior studies, sites C, D, and E are the most supreme Creeks of Musa Creek and receive the least polluted effluents from the pollution sources (Asar 2008; Faghiri 2009; Babadi 2010). It should be noted that site E is frequently believed to be the cleanest part of Persian Gulf (Musa Creek).
Flores-Lopes and Thomaz (2011) also reported that the most frequencies of severe pathological alterations in Astyanax fasciatus and Cyanocharax alburnus were found in the waters of Guaiba, which receives a large amount of pollutants via the branch rivers of lago Guaiba. It receives high loads of domestic and industrial wastes discharged into lower parts of the rio dos Sinos. Also, Camargo and Martinez (2007) showed that histopathological alterations of gill, liver, and kidney of Prochilodus lineatus were higher in regions contaminated with the most amount of urban stream.
Thus, the results obtained in the present work reinforce the importance of the addition of histopathologic analysis in programs for the evaluation of water quality as well as the use of different methods of evaluating environmental alterations and degradations of anthropic origin that can compromise the quality of the ecosystems, all together serving as tools for a more precise evaluation of environmental quality of the region to be studied in programs of environmental monitoring.
In conclusion, the present study indicates that morphological and histopathological alterations are useful biomarkers for screening the environmental contamination and field assessment. The results of the present research suggest that the morphological and histopathological alterations of the gill in A. latus were caused by the different stressors found in Musa Creek and there is a close relation between amounts of these alterations and environmental contamination.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
