Abstract
The mammary gland of laboratory rodents is an important organ for the evaluation of effects of xenobiotics, especially those that perturb hormonal homeostasis or are potentially carcinogenic. Mammary gland cancer is a leading cause of human mortality and morbidity worldwide and is a subject of major research efforts utilizing rodent models. Zymbal’s, preputial, and clitoral glands are standard tissues that are evaluated in animal models that enable human risk assessment of xenobiotics. A widely accepted and utilized international harmonization of nomenclature for mammary, Zymbal’s, preputial, and clitoral gland lesions in laboratory animals will improve diagnostic alignment among regulatory and scientific research organizations and enrich international exchanges of information among toxicologists and pathologists.
Keywords
Introduction
The INHAND Project (International Harmonization of Nomenclature and Diagnostic Criteria for Lesions in Rats and Mice) is a joint initiative of the Societies of Toxicologic Pathology from Europe (ESTP), Great Britain (BSTP), Japan (JSTP), and North America (STP) to develop an internationally accepted nomenclature for proliferative and nonproliferative lesions in laboratory animals. The purpose of this publication is to provide a standardized nomenclature for classifying proliferative and nonproliferative lesions observed in the mammary, Zymbal’s, preputial, and clitoral glands of laboratory rats and mice. The diagnostic terminology and criteria described here is based in part on the standardized nomenclature for proliferative mammary gland changes (Mann et al. 1996) in rats published previously by the STP and World Health Organization (WHO). The recommended standardized nomenclature of mammary, Zymbal’s, preputial, and clitoral gland lesions presented in this document is also available electronically at the goRENI website on the Internet (http://www.goreni.org/).
While our charge and primary goal has been to provide a morphology-based terminology applicable to the rat and mouse, the extended scientific community is working toward the harmonization of nomenclature and diagnostic criteria across species and man. This is born of the genomic revolution and the one gene, one medicine concept that now permeates medicine and is in part being actualized using the genetic engineered mouse (GEM). Clearly, “harmonization” will come through genomics. The molecules that are associated with breast and other cancers in humans are also likely to be associated with cancers in the same organ in mice (and rats). The molecular biologists are proving that the entire gene expression pattern of these tumors in mice match closely that of different classes of human breast cancers. The GEM models of human breast cancer are creating tumor phenotypes that have never been seen in spontaneous or carcinogen-induced mouse mammary tumors, some of which are also morphologically similar to human cancers. An entirely new nomenclature will be required to accommodate these newly created animals. The nomenclature will need to begin with the molecule and include the microscopic anatomy. Although the “mapping” of these terms across species is not complete (Table 1), the toxicological pathology community will have increasing contact with these newly created animals. We will need to be familiar with their pathology and will need to understand their similarities to and difference from their human counterparts. For this reason, throughout this publication there has been an attempt to incorporate the emerging data from GEM models as it relates to nomenclature and diagnostic criteria.
Mapping mouse and human acinar mammary tumors.
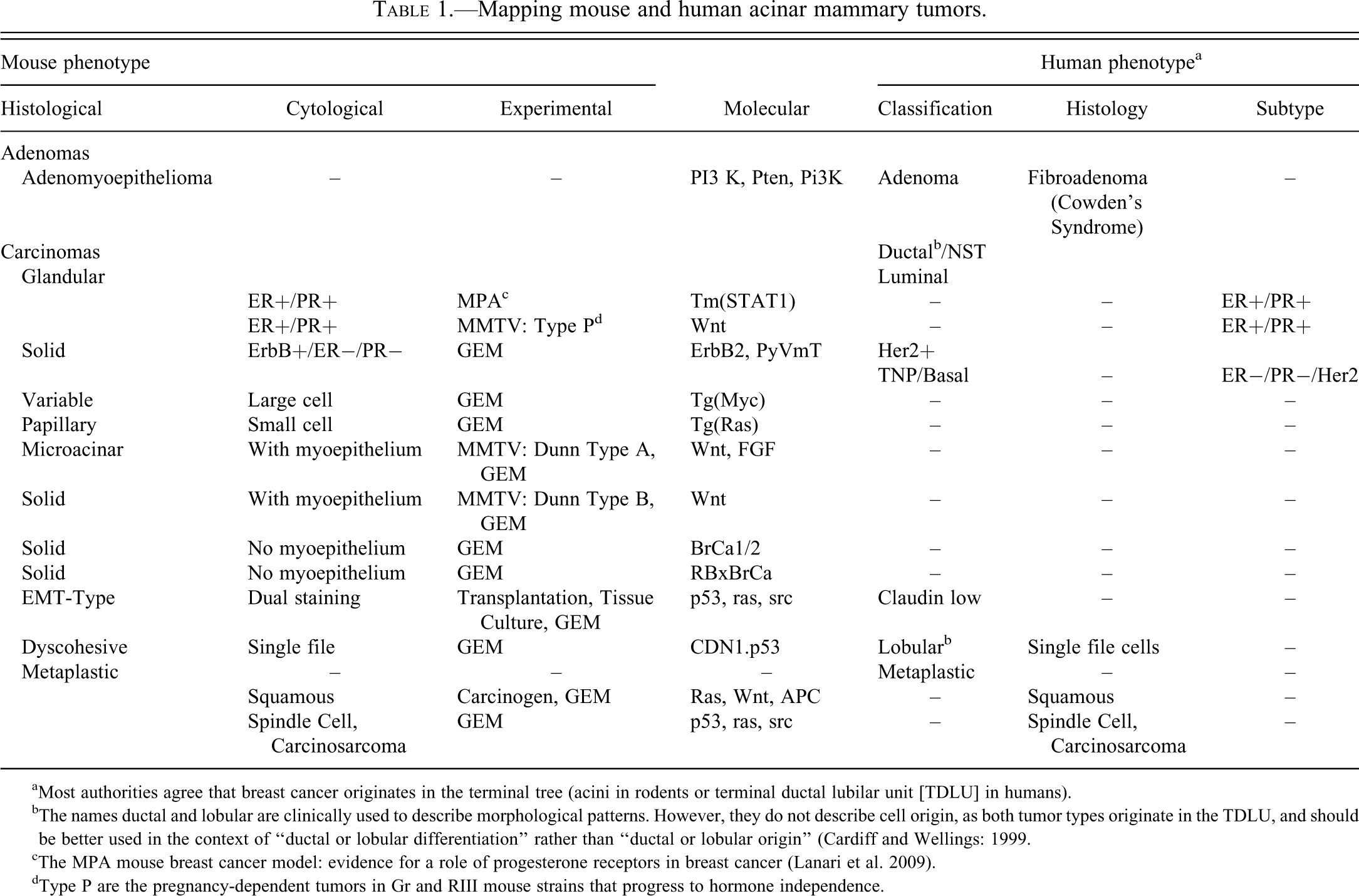
aMost authorities agree that breast cancer originates in the terminal tree (acini in rodents or terminal ductal lubilar unit [TDLU] in humans).
bThe names ductal and lobular are clinically used to describe morphological patterns. However, they do not describe cell origin, as both tumor types originate in the TDLU, and should be better used in the context of “ductal or lobular differentiation” rather than “ductal or lobular origin” (Cardiff and Wellings: 1999.
cThe MPA mouse breast cancer model: evidence for a role of progesterone receptors in breast cancer (Lanari et al. 2009).
dType P are the pregnancy-dependent tumors in Gr and RIII mouse strains that progress to hormone independence.
The mammary gland of laboratory rodents is an important organ for the evaluation of effects of xenobiotics, especially those that perturb hormonal homeostasis or are potentially carcinogenic. Mammary gland cancer is a leading cause of human mortality and morbidity worldwide (Siegel et al. 2012) and is a subject of major research efforts utilizing rodent models (Cardiff et al. 2000). Zymbal’s, preputial, and clitoral glands are standard tissues that are evaluated in animal models that enable human risk assessment of xenobiotics. A widely accepted and utilized international harmonization of nomenclature for mammary, Zymbal’s, preputial, and clitoral gland lesions in laboratory animals will decrease confusion among regulatory and scientific research organizations in different countries and will provide a common language to increase and enrich international exchanges of information among toxicologists and pathologists.
Mammary glands are standard tissues collected in all Good Laboratory Practices (GLP) and many non-GLP rodent toxicology studies. In rodent studies, all gross lesions are collected, and routinely, the inguinal (5th) mammary gland pair is dissected with or without skin and local lymph nodes from the same side in both sexes. Usually the mammary gland is trimmed in cross or transverse section. It is recommended that the mammary gland is cut in longitudinal (frontal) sections to allow for a greater chance of detecting treatment-related effects. Using frontal sectioning allows for a greater area of the mammary epithelia to be evaluated. Unless testing a specific hypothesis, toxicological pathologists will not generally use mammary gland genomic or immunohistochemistry (IHC) markers in oncogenicity studies because they are by design survey studies. However, if treatment-related changes are observed, molecular pathology techniques may be very useful in risk assessment. The assessment of the mammary gland should always be done in concert with histologic evaluation of the hypothalamic–pituitary–gonadal axis, the adrenal gland, and other end organs such as the prostate, the preputial, and the clitoral glands.
Proliferative lesions in laboratory rodents may arise from genotoxic or infectious agents or changes in hormonal homeostasis as part of the aging process or experimentally as a result of genetic engineering, but the most important proliferative mammary gland lesions are caused by exposure to potentially toxic test materials that affect hormonal homeostasis or cause cellular damage. Cellular damage from repeated exposure to toxicants induces a repair process in which the damaged tissue, if return to normal morphology is not complete, may continue to proliferate (hyperplasia or neoplasia), and/or undergo metaplasia to a different, more resistant cell type. The site of these changes is heavily dependent upon the nature of the toxicant and the type of tissue exposed.
Nonproliferative lesions in general are also associated with experimental perturbation or are a result of degenerative changes frequently associated with aging. Modern laboratory animal management practices within rodent facilities are such that spontaneous infectious processes should be infrequently encountered; thus the lesions related to infectious diseases are not described in detail in this document. For both proliferative and nonproliferative lesions, genetic engineering in an animal may also result in a model with novel and important lesions that require consistent terminology across laboratories and descriptions that aid in translation to human disease. These are described or referenced in this document.
GEM Models
Genetic engineering of mice has introduced literally hundreds of new mouse models of human breast cancer. The new models can be divided into four categories: Mouse models that (1) recapitulate spontaneous and chemically induced models; (2) develop unique genotype-specific “signature” phenotypes; (3) mimic human morphological phenotypes; and (4) simulate human genotypes or molecular phenotypes (Cardiff, Munn, and Galvez 2006).
Mouse Models that Recapitulate Spontaneous and Chemically Induced Mouse Models
Virus-induced tumors. Activation of mouse DNA by integration of mouse mammary tumor virus (MMTV) is the primary mutagenic event leading to “spontaneous” mouse mammary tumors. The original descriptions by Apolant (1906) and Haaland (1911) are based on “spontaneous” tumors induced by MMTV as well as that from Dunn (1959). Molecular analysis revealed that most of these tumors have activation of wnt, fibroblast growth factor (FGF), and/or notch. Interestingly, when mice were specifically engineered using molecular constructs containing wnt or FGF behind the MMTV-LTR promoter, they developed Type A, Type B, or Type P tumors (Sass and Dunn 1979). Equally important, activation of one oncogene, such as wnt, resulted in co-activation of the other complementary oncogene, such as FGF (van Leeuwen and Nusse 1995). MMTV is mouse-specific; therefore, while these models are useful for investigating the potential roles of different tumor suppressor and oncogenes in the pathogenesis of mammary gland neoplasms, the activation or suppression of DNA by MMTV itself is not applicable to human tumorigenesis.
Chemically induced tumors. A limited number of chemically induced tumors have been analyzed; common carcinogens such as 7,12-dimethylbenz[a]anthracene (DMBA) or 3-methylcholanthrene (MCA) result in mutational activation of the H-ras gene (Cardiff et al. 2000) frequently originating keratoacanthomas or adenosquamous carcinomas with squamous metaplasia (Currier et al. 2005). The tumors with squamous metaplasia have activation of the wnt-APC-B-Catenin pathway (Tsukamoto et al. 1988; Gaspar et al. 2009; Michaelson and Leder 2001). Other tumor types have not been analyzed.
Mouse Models that Develop Unique Genotype-specific “Signature” Phenotypes
Most GEM develop tumor phenotypes that never have been previously seen in the mouse. The key observation is that most oncogene activation results in tumors with gene-specific signature phenotypes and oncogene activation from the same pathway results in tumors with similar phenotypes (Rosner et al. 2002). For example, the myc, ras, and neu tumor phenotypes are unique and distinguishable (Cardiff et al. 2000). Tg(Myc) mice develop tumors with large pleomorphic nuclei and coarse chromatin with abundant amphophilic cytoplasm resembling a mammary counterpart to Burkitt’s lymphoma. Tg(Ras) tumors form papillary structures with small cells with oval nuclei and red cytoplasm (Sinn et al. 1987), resembling human transitional cell carcinomas of the urinary bladder. The Tg(cNeu) tumors tend to be solid nodules with intermediate sized nuclei and light pink-orange cytoplasm (Muller et al. 1988; Komitowski,Sass, and Laub 1982). With central necrosis, they resemble human comedo-carcinomas. Mutation of other molecules in the Wnt pathway, such as APC or B-catenin results in tumors with similar morphologies (Gaspar et al. 2009). These phenotypes may be primarily based on cytological characteristics, as a recent study of Tg(Myc) with different mutations share cytological characteristics but different histological patterns (Andrechek et al. 2009).
In another example, mammary tumors in Tm(PTEN−/−) mice characteristically are adenomyoepitheliomas (Stambolic et al. 2000), also characteristic of the closely related mutant PIK3 mice (Meyer et al. 2011). No molecular studies of sporadic, spontaneous adenomyoepitheliomas have been performed. The Epithelial–Mesenchymal–Transition (EMT) tumor phenotype (Cardiff 2010; Radaelli, Damonte, and Cardiff 2009) is a special type that has been previously recognized as a carcinosarcomas (Barnes et al. 2005). In GEM, EMT occurs in the context of loss of the expression of the initiating oncogene and p53 mutations (Debies et al. 2008). Such tumors have a range of phenotypes ranging from a mixed adenocarcinoma and spindle cell tumor to a pure spindle cell tumor (Damonte et al. 2007). EMT is most easily verified using the criteria of dual staining with mesenchymal and epithelial markers (Damonte et al. 2007). Some observers have speculated that these are metaplastic carcinomas. However, molecular evidence to support this hypothesis is lacking. There are numerous additional examples; further discussion is beyond the scope of this article.
Mouse Tumors that Mimic the Histopathology of Human Tumors
With genetic engineering, numerous candidate oncogenes have been tested by targeting the mouse mammary gland using molecular modification (Borowsky 2011). Tumors that most closely resemble their human counterparts include the Tg(c-ErbB2) and Tm(CDN1−/−xp53−/−). Many Tg(c-ErbB2) tumors resemble their human counterparts that overexpress this same gene (Bouchard et al. 1989; Ursini-Siegel et al. 2007). The Tm(CDN1−/−xp53−/−) tumors have the typical single file configuration of human lobular carcinoma, which is also associated with loss of E-Cadherin (CDN1) (Derksen et al. 2006). The Tm(Stat1−/) is the first GEM model with uniformly ER+, PR+, FoxA1+ neoplasms that have experimentally proven ovarian dependence and that mimics the histopathology of some luminal A tumors (Chan et al. 2012).
Mouse Models and Molecular Classifications
The traditional morphology-based classification of human breast cancer has been published by the World Health Organization (WHO 2003). The extensive WHO classification includes criteria for all the expected diagnostic categories based on theoretical cell of origin and histological pattern. The INHAND classification generally adheres to this outline. However, the human classification includes the categories of Ductal and Lobular carcinoma which are convenient clinical designations but are not consistent with current concepts of origin (Cardiff and Wellings 1999).
The largest single category in the WHO morphological classification is “invasive ductal carcinoma not otherwise specified (NOS)” (WHO 2003). Recently, molecular classifications have been proposed for the clinical classification of human NOS or no-specific type (NST) breast cancer (Weigelt and Reis-Filho 2009). These classifications are based on hierarchical analysis of expression microarrays. The most commonly used molecular classification of NST tumors recognizes five molecular subgroups: Luminal A, Luminal B, Her2 positive, and Normal and Basal cell (Sorlie et al. 2001).
Most clinicians now find it beneficial to divide NST breast cancers into ER+/PR+, HER2+, and triple negative. This convenient clinical classification has dictated clinical management of human breast cancer over the last decade because ER and HER2 are targetable proteins. However, the triple negative tumor phenotype (TNP) was almost immediately recognized as a morphologically and clinical diverse group of human tumors (Rakha et al. 2007). The identification of “basal” biomarkers in some triple negative tumors led to a general assumption that they are synonymous. Moreover, the basal cell concept has been challenged as an untenable misinterpretation of the biology that should be discarded (Rakha, Reis-Filho, and Ellis 2008; Gusterson et al. 2005; Lavasani and Moinfar 2012). Most analyses are finding heterogeneity within any given group. A new term has been introduced, “luminobasal,” which is designed to reflect this heterogeneity (Balko et al. 2012). The above exemplifies the confusion that reigns in the current classifications of human breast cancer. The “lumping” of clinical outcomes with expression profiles circumvents the traditional morphological classifications. Only early efforts have started that match the morphology with the molecular information; therefore, the confusion will likely increase. In short, the definitive molecular classification of human breast cancer is yet to emerge.
The use of expression microarrays for clinical classifications has encouraged attempts to classify mouse mammary tumors with the same expression technology (Herschkowitz et al. 2007). Since the vast majority of mouse tumors promoted by the MMTV-LTR do not express ER, PR, or Her2, they could all fall into the “triple negative” or basal category of human NST breast cancer (Weigelt and Reis-Filho 2009). These attempts to match mouse and human molecular expression profiles have not included rigorous efforts to match histological patterns with the expression profiles. Attempts to match the human molecular categories also ignore the rich variation in GEM tumors leading to a potentially misleading grouping of genotypes. The challenge for the next iteration of INHAND will be to anticipate the movement toward molecular classifications of breast cancers and to harmonize them with the traditional structure-based classifications. At the end of this article, we include examples (Figures 40–47) of several GEM tumors.
Rat Models
Evaluation of mammary gland toxicity in rats is critical in toxicity testing. The rat in many cases may be the only toxicology animal model studied that uses sexually mature animals and is dosed at intervals ranging from a single day to a lifetime in length.
Acute to chronic bioassays conducted by industry and agencies such as the National Toxicology Program remain a standard approach for pharmaceutical safety assessment and chemical hazard identification (Davis and Fenton 2013). Industry-sponsored studies generally use the F344, Sprague-Dawley (SD), Harlan Sprague-Dawley, or Wistar rats, the latter three most common today. The importance of rat strain selection is demonstrated by their differences in susceptibility to both non-neoplastic and neoplastic changes. For example, mammary gland fibroadenomas are the most common spontaneous tumor in female SD rats, with incidences reported as high as 70% in chronic studies. In contrast, female Fischer rats have reported incidences of about 40%. This is also important in human risk assessment. Fibroadenomas are not considered a premalignant lesion in humans nor are rat mammary fibroadenomas considered predictive of carcinoma in women. In contrast, spontaneous mammary adenocarcinomas are considered relevant in studies and are at a higher spontaneous incidence in studies using the SD rat as compared to the F344 rat (Davis and Fenton 2013).
Genetic susceptibilities of the various species and strains of rodents have been exploited in the development of animal models for mammary gland cancer. For example, spontaneous mammary gland cancers in rats, generally adenocarcinomas, can be enhanced with treatment with genotoxic carcinogens like DMBA, N-nitrosomethylurea, and N-ethyl-N_nitrosourea (ENU). These induced mammary tumors are hormone-dependent and can be modulated by a number of factors including reproductive status, hormone treatment, diet, and the dose and timing of carcinogen administration (Davis and Fenton 2013).
As is the case for mice, differences in the development and physiology of the rat require understanding for accurate human risk assessment. For example, as virgin female SD and F344 rats become middle aged (from 8 to 14 months of age), normal reproductive senescence ensues and there is an increasing level of prolactin (PRL) secretion that contributes to the development of a number of spontaneous morphologic changes. These include increased secretions, duct dilation, alveolar and tubular epithelial hyperplasia, and periductal fibrosis. Xenobiotics that increase pituitary PRL secretion like dopamine receptor antagonists can also cause these rat mammary gland changes. In contrast, dopamine agonists may reduce PRL and decrease the incidence of the above histologic changes as well as the incidence of spontaneous mammary gland neoplasia. Hyperprolactinemia is not normal in postmenopausal women. Therefore, while the histologic changes of senescence in rats are a normal age-related change, histologic changes similar to those observed in women would be considered dysplastic (Davis and Fenton 2013).
Morphology
Mammary Gland
Embryologic development of the rat and mouse mammary gland has been investigated extensively and will only be briefly considered here (for reviews, see Ceriani 1970; Knight and Peaker 1982; Russo, Tewari, and Russo 1989). The male and female rat and mouse mammary gland develops from a single layer of cuboidal epithelium originating from the milk bud that forms the primitive nipple, progressing in a cephalocaudal sequence (Myers 1917; Cardiff and Wellings 1999). Islands of epithelium thicken into hillocks of cuboidal cells resting on an indistinct basal membrane; simultaneously, there is atrophy of the epithelial cells between the hillocks (Myers 1917). In the male or female fetus, mammary glands remain as rudimentary buds of epithelium without development into distinct lobules or alveoli (Knight and Peaker 1982).
In the rat and mouse, the development and function of the mammary gland is directed by numerous hormones including estrogens, androgens, progesterone, PRL, growth hormone (GH), insulin, catecholamines, and adrenocorticotropic hormone (ACTH) (Russo and Russo 1996). Androgens initiate differentiation of the male phenotype by promoting atrophy of the rudimentary buds in the male (Goldman, Shapiro, and Neumann 1976; Sourla, Martel, et al. 1998; Sourla, Flamand, et al. 1998). This atrophy of the buds is triggered by testosterone-induced condensation of the stroma (Topper and Freeman 1980). Mammary gland morphology in the male rat is altered to the morphology of the female rat if androgens are eliminated, and androgens administered to female rat fetuses cause mammary glands to appear male-like (Goldman, Shapiro, and Neumann 1976). This does not occur in the mouse, because of a loss of androgen responsiveness. PRL is also an important pituitary hormone controlling rodent mammary gland growth. While the precise roles of estrogens (E) and progesterone (P4) in the rat fetus are less clear, studies in mice and ex vivo mammary gland explants suggest that progesterone is not critical to embryonic development of mammary glands in mice and its absence in utero does not affect the potential for maturation in adulthood (Freeman and Topper 1978). Estrogens promote mammary gland development in the mouse fetus; however, E inhibits development in the rat fetus (Ceriani 1970).
The neonatal male and female rat and mouse have 6 and 5 pairs of glands, respectively, with a single, central lactiferous duct, several branching secondary ducts, and numerous tertiary ducts (Ceriani 1970; Cardiff and Wellings 1999). In the rat, the glands are distributed in pairs along the milk line, with 1 pair cervical, 2 thoracic, 1 abdominal, and 2 inguinal (Astwood, Geschickter, and Rausch 1937). Distribution in the mouse is in the neck, chest wall (2 pairs), abdominal wall, and inguinal region (Cardiff and Wellings 1999). Mammary tissue is embedded in a mass of adipocytes, pre-adipocytes, and fibroblasts, referred to as the fat pad, with a thin layer of stroma separating the epithelial cells from the fat pad (Hovey, McFadden, and Akers 1999; Imagawa et al. 2002; Silberstein 2001). For toxicology studies, it is common to include a regional lymph node in the section for which important histologic changes may be observed (Figures 1–3). In the prepubescent female rat, mammary growth is mainly influenced by GH and PRL with minimal influences of estradiol (E2) and P4 (Knight and Peaker 1982).
Mammary growth in the male and female mouse and rat during puberty is dependent on normal gonadal function, as indicated by the absence of development in ovariectomized or gonadectomized rats (Cowie and Folley 1961). While a detailed description of pubescent mammary gland development in males is not published, in females, growth of the mammary gland during puberty is characterized by differentiation of the epithelium into terminal end bud units (TEBs), rapid expansion by elongation and branching of the ducts, and hypertrophy of the fat pad (Cowie and Folley 1961; Russo, Tewari, and Russo 1989; Knight and Peaker 1982; Cardiff and Wellings 1999). New end buds form from lateral branching of mature ducts and have the potential to form lobules. The TEBs are the major hormone sensitive area of the mammary glands for sexually mature rats and mice. PRL, estrogen, and progesterone are the predominant hormones controlling rodent TEB development to lobular alveolar structures in the virgin rodent (Richards et al. 1983). In rats, these hormones are also important in influencing adult mammary gland morphology (Rudmann et al. 2005; Lucas et al. 2007). As discussed earlier, androgens contribute to the development of both the rat and the mouse mammary gland, and in the adult rat, androgens produced by the ovary effect mammary gland morphology in both the male and the female. Effects of androgens are modulated by the androgen receptor, which is upregulated with androgen stimulation as well as by the erbB family of growth factors (epidermal growth factors).
In female rats and mice, ducts, ductules, and alveoli are lined by 1 or 2 layers of epithelial cells and surrounded by myoepithelial cells. Within these epithelial structures are 3 types of epithelial cells: clear cells, dark cells, and intermediate cells. The morphologic differences between these cell populations are the result of variability in the number of ribosomes, mitochondria, lipid droplets, and secretory vacuoles (Greaves 2007). The rat mammary gland is sexually dimorphic (Figures 4 and 5; Lucas et al. 2007). In contrast to female rats, ducts in male rats are infrequent and when observed are lined by a stratified epithelium consisting of vacuolated tall cuboidal to short columnar epithelial cells. Alveoli are predominant in the male and also lined by a stratified epithelium. Imbalance of mammotrophic hormones like PRL, estrogens, and androgens may result in the male and female rat mammary gland converting to the morphologic appearance expected of the opposite sex (Rudmann et al. 2005; Lucas et al. 2007).
Degenerative Changes
Degeneration: Ductular/Alveolar Epithelium
Pathogenesis/cell of origin: Ductular, alveolar epithelial cell; myoepithelial cell.
Diagnostic features Epithelial cells swollen Epithelial vacuolization/bleb formation Loss of organization of the cell layers Dilatation (ectasia) of alveoli or ducts with accumulation of secretory material
Differential diagnoses Postmortem autolysis: Uniform dissolution of entire tissue with no change in organization or depth of cell layers Atrophy: Thinning of affected ducts or alveoli
Comment: Degenerative changes in the mammary gland are rare but can be observed with toxic agent exposure and as a consequence of aging. Surrounding tissues such as the fat pad, skin, and local lymph nodes can also undergo a variety of degenerative changes.
Single-cell Necrosis: Ductular/Alveolar Epithelium
Synonym: Apoptosis
Pathogenesis/cell of origin: Alveolar or ductular epithelium
Diagnostic features Cells usually shrunken with distinct cell membranes Membrane budding, condensed nuclei Rarely associated with inflammation
Differential diagnosis Necrosis: Cells may be swollen. Cytoplasmic eosinophilia, nuclear pyknosis/karyorhexis, cell debris, more likely associated with inflammation and thinning of epithelium
Necrosis: Ductular/Alveolar Epithelium
Pathogenesis/cell of origin: Alveolar or ductular epithelium
Diagnostic features Cellular swelling or shrinkage Cytoplasmic eosinophilia Pyknosis or karyorrhexis of nuclei Exfoliation of cells May result in thinning of epithelium May be associated with inflammation Luminal accumulations of fibrin and/or cell debris
Differential diagnoses Postmortem autolysis: Uniform dissolution of entire tissue with no change in organization or depth of cell layers Apoptosis: Cells shrunken with distinct cell membranes, membrane budding, condensed nuclei; rarely associated with inflammation Degeneration: Cell vacuolation but no inflammation or cell debris Atrophy: Thinning of cell layers but no inflammation or cell debris Inflammation: Cellular infiltrates and swelling but no exfoliation or cell debris
Comments: Mammary gland epithelial necrosis is an infrequent finding in rodents. Fat necrosis occasionally occurs in the fat pad and is usually accompanied by granulomatous inflammation and fibrosis in the rat (Boorman et al. 1990).
Basophilia: Ductular/Alveolar Epithelium
Synonym: Regeneration
Pathogenesis/cell of origin: Alveolar or ductular epithelium
Diagnostic features Normal appearing epithelial cells with basophilic cytoplasm Increased nuclear:cytoplasmic ratio Epithelial architecture may appear irregular Mitoses may be present Adjacent to or within areas of degenerating, necrotic, hyperplastic, or metaplastic epithelium
Differential diagnoses Hypertrophy/Hyperplasia: Epithelium is thickened due to increased numbers or size of cells, resulting in undulating, rugose epithelial surface and irregular arrangement of cell layers (see proliferative lesion section of this document) Neoplasia: Expansile nodule usually protruding into cavity, with cellular atypia and compression of adjacent structures (see proliferative lesion section of this document)
Comments: Basophilia is recommended as the preferred descriptive term for the lexicon. When basophilia is interpreted as evidence for regeneration (growth of cells and tissues to replace lost or damaged structures, as opposed to hypertrophy/hyperplasia), it is recommended that this interpretation be included in the text of the report (Kumar et al. 2010). Irregularity of epithelial cell arrangement may be observed in the ductular or alveolar epithelium in the process of regeneration. Degeneration, necrosis, and regeneration are often present together in mammary gland repeatedly injured by toxicants. See comments above in sections on degeneration and necrosis.
Inflammatory Changes
While inflammation of the mammary gland is an important cause of morbidity in large animal veterinary species, because of animal husbandry procedures, inflammation of the rodent mammary gland is uncommon. More commonly, histological changes are limited to infiltrates of foci of lymphocytes, neutrophils, macrophages, or plasma cells, alone or in combination.
Infiltrate: Lobule
Pathogenesis/cell of origin: Alveolar or ductular epithelium and associated tissues.
Diagnostic features Infiltration of a relatively pure population of lymphocytes, plasma cells, neutrophils, eosinophils, macrophages or a mixture of these cell types into the lamina propria of the lobule and associated tissues
Differential diagnoses Inflammation: Infiltrates are associated with degenerative epithelial changes, edema, hemorrhage/congestion, and/or fibrosis. Hematopoietic neoplasia: Homogenous lymphocyte population infiltrating entire tissue and other sites.
Comment: The base term infiltrate is recommended followed by the predominant cell type or “mixed” if there is not a predominant cell type. Leukocytic infiltrates consisting only of lymphocytes or plasma cells may indicate an immunologic effect of a xenobiotic but can also be observed as a spontaneous change.
Inflammation: Lobule (Figure 6)
Synonym: Mastitis
Pathogenesis/cell of origin: Alveolar or ductular epithelium and associated tissues.
Diagnostic features Acute: Vascular congestion, edema, accumulation of serous, mucous or fibrinous exudates, neutrophils, sloughed epithelial cells in tubular or alveolar lumina Chronic: Cellular infiltrate is predominantly lymphocytes, plasma cells, and macrophages; regenerative, hyperplastic, and/or metaplastic changes in the affected epithelium and fibroplasia may be present Chronic active inflammation: Cellular infiltrate is a mixture of granulocytic, lymphocytic, histiocytic cells; regenerative, hyperplastic, and/or metaplastic changes in the affected epithelium and fibroplasia may be present. Granulomatous: Cellular infiltrate is predominantly plump macrophages (epithelioid cells) that may form interlacing bundles, accompanied by lymphocytes, plasma cells, and fibrosis, depending on duration and etiologic agent. Etiologic agent may be visible, e.g., fungi, mycobacteria, or foreign bodies. Regenerative, hyperplastic, and/or metaplastic changes in the affected epithelium and fibroplasia may be present. Granulocytes may be present in the affected area, in which case the process could be described as pyogranulomatous inflammation.
Differential diagnoses Necrosis: Pyknosis, karyorrhexis of nuclei, cytoplasmic swelling or shrinkage, cellular debris, accompanied by inflammatory infiltrate
Comments: Inflammation need not be recorded with a chronicity descriptor; instead, the characteristics of the inflammation may be described in report text. In acute inflammation, migration of neutrophils into ductal and alveolar lumina produces a suppurative exudate. Eosinophils in exudate or mucosal infiltrate may indicate an immunologic or parasitic element to the inflammatory response (Kumar et al. 2010). The term chronic active inflammation implies recurrence or persistent presence of granulocytic inflammatory cells concurrent with ongoing chronic inflammation. Chronic active and granulomatous inflammations have many etiologic and morphologic similarities. Granulomatous inflammation suggests an etiologic agent which is resistant to dissolution or is immunogenic, e.g., fungi, mycobacteria, or foreign body (Kumar et al. 2009). Granulomatous inflammation is commonly seen in rats associated with rupture of a dilated duct. It is important to differentiate foreign body inflammation from other inflammatory lesions, because of the differences in their causes. Chronic inflammation may have different characteristics depending on the longevity of the lesion and the initiating cause. While it is uncommon in the rodent mammary gland, it may also occur as a result of leakage of secretory material outside the ducts.
Fibrosis: Lobule (Figure 7)
Pathogenesis/cell of origin: Fibroblasts or periductular fibroblasts.
Diagnostic features Increased thickness in the connective wall of mostly medium size ducts Usually periductular
Differential diagnosis Fibrosis in chronic inflammation
Comment: Fibrosis in periductular regions is commonly seen in aged rats and it is recommended that periductular be used as a modifier. In BALB/c mice treated with EGF, the most evident histological feature is proliferation of small and medium size ducts. This proliferation is associated with an evident increase in the interlobal connective tissue as well as periductal fibrosis (Molinolo et al. 1998).
Vascular Changes
Congestion: Lobule
Synonym: Hyperemia
Pathogenesis/cell of origin: Vasculature of mammary gland and surrounding tissue
Diagnostic features Widely dilated, blood-filled vessels
Differential diagnoses Artifact of euthanasia or necropsy procedures Postmortem autolysis: Uniform dissolution of entire tissue with lysis of red blood cells Angiectasis: Dilated vessels that distort normal architecture of the affected tissues
Comments: Congestion of the mammary gland and associated skin may be observed in rats or mice dying or killed while moribund, related to terminal pooling of blood in the dependent portions of the animal. The term congestion in the committee’s experience is generally not a useful diagnosis as an independent entity. Congestion may be part of an inflammatory or degenerative process and its description is best left as text in a report.
Edema: Fat Pad (Figure 8)
Pathogenesis/cell of origin: Vasculature of mammary gland and surrounding tissue
Diagnostic features Proteinaceous fluid in the interstitium within the fat pad or around vessels, ducts, and alveoli
Differential diagnoses Postmortem autolysis: Uniform dissolution of entire tissue with lysis of red blood cells Fibrinous exudate: Smudged pink exudate with laminar appearance of fibrils visible at high magnification Artifact of tissue processing
Hemorrhage: Lobule
Pathogenesis/cell of origin: Lumen of ducts and alveoli and associated tissues
Diagnostic features Presence of free blood in duct and alveolar lumen or in adjacent fat pad
Differential diagnoses Angiectasis: Blood present within dilated vascular lumens Artifact: A result of necropsy procedures or anesthesia protocols for euthanasia
Angiectasis: Lobule
Pathogenesis/cell of origin: Vasculature of fat pad
Diagnostic features Increased profiles of blood vessels, distorting the normal architecture of the affected tissue
Differential diagnoses Hemangioma: Blood-filled space lined with uniform endothelial cells, distorting the architecture of the affected tissue Congestion: Widely dilated, blood-filled vasculature not distorting the architecture of the affected tissue Hemorrhage: Free blood present outside vascular walls
Thrombosis: Lobule
Pathogenesis/cell of origin: Vessels of mammary gland and fat pad
Diagnostic features Amorphous pink/grey, clearly laminated mass containing leucocytes and erythrocytes Attachment to lumen of vessel is rarely visible on routine sections
Differential diagnosis Postmortem clot: Few or no leukocytes; lamination absent or very fine filaments
Comments: Thrombi are often associated with mononuclear cell leukemia or other generalized, debilitating conditions in rats.
Other Lesions
Corpora Amylacea: Ductular/Alveolar Epithelium (Figure 9)
Pathogenesis/cell of origin: Alveolar or ductular epithelium and adjacent lamina propria, lumen of alveolus or duct
Diagnostic features Small basophilic or amphophilic concretions Often laminar with mineralized areas May be associated with dilatation of ducts and alveoli
Differential diagnoses Necrosis: Other evidence of tissue damage Degeneration: Other evidence of tissue damage Mineralization: Evidence of mineralization at other sites or tissues
Comments: Corpora amylacea are seen infrequently in untreated rats and mice. Corpora amylacea that stain eosinophilic with hematoxylin and eosin may represent amyloid in mice.
Amyloid: Lobule
Pathogenesis/cell of origin: Extracellular deposits of polypeptide fragments of a chemically diverse group of glycoproteins (Solomon et al. 1999) in various tissues
Diagnostic features Lightly eosinophilic, amorphous extracellular material in submucosa or interstitium Green color using polarized light with Congo Red stain
Differential diagnoses Necrosis: Congo Red negative; other evidence of damage Degeneration: Congo Red negative Connective tissue hyaline: Congo Red negative Exudate or foreign bodies: Congo Red negative; other evidence of exudate or foreign body
Comments: Amyloidosis occurs in various tissues of aged mice of several strains (Korenaga et al. 2004) but has only been rarely described in the literature in the mammary gland of rodents (Beems, Gruys, and Spit 1978). A severity score that reflects the extent of amyloid deposits in each tissue of affected mice is not generally considered useful. It is recommended that when amyloid is observed in the mouse mammary gland, that “present” is used as a descriptor.
Dilation: Duct/Alveolus (Figures 10–12)
Synonyms: Cystic degeneration, cystic change, milk cysts, ectasia, galactocoele
Pathogenesis/cell of origin: Ducts and alveoli
Diagnostic features Dilated lumen with or without epithelial hypertrophy or hyperplasia Often luminal proteinaceous material, lipid, macrophages, or cell debris Often vacuolation of ductal/alveolar epithelial cells
Comments: Dilation may occur as a spontaneous aging change in association with mammary gland hyperplasia and metaplasia. In younger animals treated with xenobiotics, the change suggests perturbation of the hypothalamic–pituitary–gonadal axis. Severity grades and report descriptions may be used to indicate the extent of ductal and alveolar involvement and the size of the affected lumen diameters.
Pigment: Lobule (Figure 13)
Synonyms: Pigmentation, accumulation
Pathogenesis/cell of origin: Intracellular deposits of endogenous substances which often may be differentiated using special histochemical, immunohistochemical, or electron microscopic procedures
Differential diagnoses Mineralization Artifact (i.e., acid hematin)
Diagnostic features Various cell populations in the mammary gland contain intracytoplasmic endogenously derived pigments
Comments: Pigments usually represent lipofuscin, hemosiderin, or other hemoglobin breakdown products. Grading pigment changes may not be useful and it is recommended that “present” be used and the pigment described within the text of a report.
Mineralization: Fat Pad
Pathogenesis/cell of origin: Fat pad
Diagnostic features Mineral deposits in the interstitium or linear mineralization of vessels, visible on H&E sections, may be confirmed with stains for minerals Frequently accompanied by macrophages and inflammation
Comments: Mineralization of vessels in the mammary gland may be present in severe cases of chronic nephropathy of aged rats.
Growth Disturbances
Non-neoplastic
Atrophy: lobule (Figure 14)
Synonyms: Feminization (male rat)
Pathogenesis/cell of origin: Ductular and alveolar epithelial cell
Diagnostic features Male rats: If due to effects of PRL, tubular profiles are more frequent and alveolar profiles are lined by smaller, cuboidal cells with basophilic cytoplasm; alveoli have a thinned epithelium Male rats or mice: If due to effects of decreased androgens or emaciation, there are smaller and/or decreased number of alveolar profiles Female rats: Tubulo-alveolar profiles present but in decreased numbers or have thinned epithelium Tubular or alveolar lumina may appear dilated Relative amount of fat pad increased
Differential diagnoses Hypoplasia: Tubular and alveolar profiles present but in decreased numbers due to developmental anomaly Artifact: Decreased glandular tissue may result from variability in gross necropsy, trimming, and sectioning procedures.
Comments: Feminization is not a recommended term because it is syndromic, only applies to the male rat, and may be misleading because of its use in humans. In male rats, xenobiotics that increase PRL will result in atrophy of alveoli resulting in a tubulo-alveolar appearance of the gland similar to what is observed in the normal virgin female gland (Figure 15; Ose et al. 2009; Lucas et al. 2007). In both males and females, xenobiotics may perturb the hypothalamic–pituitary–gonadal axis resulting in a decrease in circulating, mammotrophic hormones resulting in glandular atrophy. Inconsistent sectioning of the mammary gland may result in the erroneous diagnosis of atrophy.
Hypertrophy/hyperplasia, alveolar and/or ductal epithelial cell: lobule (Figure 16)
Synonyms: Masculinization, virilization (female rat)
Pathogenesis/cell of origin: A decrease in the estrogen:androgen balance in the female rat may result in the tubulo-alveolar appearance of the normal female mammary gland to become more male-like (lobulo-alveolar). Presumably there may be other causes of alveolar/ductal epithelial hypertrophy and hyperplasia. Adult mouse mammary glands are not androgen responsive.
Reflect what is observed in the normal male mammary gland and include:
An increased predominance of irregular nests of alveolar epithelial cells often not associated with a clear duct versus the ductal dominant histology (so-called tubulo-alveolar) observed in a normal female mammary gland
Increased alveolar cell size and abundance of cytoplasm
Cytoplasmic basophilia and vacuolation
A pseudostratified appearance to alveolar and ductal epithelium
Increased abundance of cytoplasm
May have increased ductular or alveolar secretions
Differential diagnoses
Hyperplasia, lobuloalveolar: The distinct tubulo-alveolar architecture of the female is maintained (predominance of ducts associated with small nests of alveoli). There is a normal relationship between hyperplastic alveolar epithelial cells and associated ducts. Increase in profiles of alveoli around a duct without cytologic changes in individual alveolar or ductal cells (increased cytoplasm, vacuolation, basophilia, pseudostratification). Comments: Neither masculinization nor virilization are recommended for use because they are syndromic terms and may be misleading because of their implications in humans. In female rats, xenobiotics that cause an increase in androgens or decrease the estrogen:androgen ratio may induce an alteration in the mammary gland characterized by hypertrophy and hyperplasia of alveolar and ductal epithelium resulting in a lobulo-alveolar appearance more like the normal male mammary gland (Lucas et al. 2007; Rudmann et al. 2005).
Hyperplasia, Lobuloalveolar: Lobule (Figures 17–18)
Synonym: Pseudopregnancy
Pathogenesis/cell of origin: Mammary gland epithelium of alveoli and ducts
Diagnostic features Regular lobular architecture is maintained Normal relationship between ductal and alveolar epithelial cells, myoepithelial cells and stroma Alveolar epithelial cells form one layer and are mostly cuboidal Cells lining the ducts vary from columnar to flattened Cystic dilation may be present Alveoli and ducts may be distended with eosinophilic material as evidence of secretory activity Little or no cellular pleomorphism Focal squamous cell metaplasia may occur Diffuse: The total mammary gland is affected. The mammary fat pads are filled with glandular acini. Focal: May occur as a single or multifocal lesion. One or more lobules are affected. Lack of compression or encapsulation. Lobule enlarged due to an increase in number and size of cells in normal appearing alveoli and ducts. Hyperplastic lobules merge with the surrounding normal lobules. Usually lack of a prominent and homogenous fibrous connective tissue, but focal reactive fibrous connective tissue may occur Focal with atypia (Figures 19 and 20): One or more alveoli or ducts within a lobule are affected. Either focal, irregular proliferation of ductal or alveolar epithelium within a lobular hyperplasia or acinar, irregular proliferation of one or more alveoli or ducts within normal mammary gland tissue. Proliferation forms papillae, arches, nests extending into lumen. Small alveoli may be filled with cells and appear solid. Small areas of cellular atypia and/or pleomorphism, nuclei may be enlarged, hyperchromatic or vesicular, cytoplasm may be secretory and stain basophilic or eosinophilic. Focal with fibrous tissue proliferation: Increased amount of fibrous tissue between acini, lesion is not compressive or confluent, lobular architecture maintained.
Differential diagnoses Hypertrophy/hyperplasia, alveolar and ductal epithelium (female rat): Male lobulo-alveolar characteristics of alveolar and tubular epithelium predominate rather than increases in number or size of normal female alveoli forming lobules around ducts Adenoma: Tumor well demarcated from the adjacent mammary tissue. The regular architecture of the gland is distorted Fibroadenoma: Well defined. Uniform structures within the epithelial and connective tissue components. Ducts and acini are surrounded by layers of proliferative fibrous tissue Adenocarcinoma: Structural or cellular pleomorphism. Detachment of epithelial cells from the basement membrane. Infiltrative growth; occurrence of distant metastasis
Comments: Diffuse hyperplasia is usually physiologic and is prominent in late gestation and during lactation. Lobular alveolar hyperplasia may result from xenobiotics that increase PRL in female rats.
Focal lesions with atypia also fit the designation of Mammary Intraepithelial Neoplasm (MIN) or Hyperplastic Alveolar Nodule (HAN; Cardiff et al. 2006). The distinction between hyperplasia and neoplasia is classically tested by serial transplantation (Cardiff and Borowsky 2010). The hyperplasia cannot be serially transplanted while the MIN/HAN are immortal and are considered premalignant. They can only survive in mammary fat pads. The immortalized MIN/HAN have a high risk of malignant transformation. Malignant cells can be transplanted into ectopic sites.
Neoplastic
Adenoma: mammary gland (Figures 21 and 22)
Pathogenesis/cell of origin: Mammary gland epithelium.
Diagnostic features Often macroscopically seen as nodule Tumor is well demarcated from the adjacent mammary tissue Compression of surrounding tissue may occur May be encapsulated The regular architecture of the gland is distorted Increase in size and variability in diameter of glandular lobules Tightly packed epithelial structures Alveoli may show varying size Epithelium may be single or double layer Epithelial cells are still attached to the basement membrane Cells are well differentiated Cells lining the alveolar structures are cuboidal Secretory activity may occur Scanty connective tissue is present Small focal areas of atypia and/or pleomorphism may be present Focal squamous cell metaplasia may occur Subtypes: the following subtypes exist, but no subtyping recommended in regulatory toxicity studies: Cystic: Simple or multiloculated cystic glandular structures Papillary: Papillary structures with fibrous core covered by cuboidal to columnar epithelium Alveolar/tubular: Round or oblong glandular structures, empty or with proteinaceous secretion
Differential diagnoses Hyperplasia: Regular lobular architecture is maintained Fibroadenoma: Well defined. Uniform structures within the epithelial and connective tissue components. Ducts and acini are surrounded by layers of proliferative fibrous tissue Adenocarcinoma: Structural or cellular pleomorphism. Detachment of epithelial cells from the basement membrane. Infiltrative growth; occurrence of distant metastasis
Fibroadenoma: mammary gland (Figure 23)
Pathogenesis/cell of origin: Mammary gland epithelium and connective tissue.
Diagnostic features Well defined and may be encapsulated Composed of proliferated glandular epithelium surrounded by layers of proliferated fibrous tissue Lobular growth pattern is present and may involve significant proportions or a total mammary gland complex Uniform structures within the epithelial and connective fibrous tissue components Diversity of structural lobular patterns resulting from various proportions of epithelial and fibrous connective tissue can occur Secretory epithelium usually composed of a single layer, but double layering may occur, focally Small focal areas of atypia and/or pleomorphism may be present Mitotic figures are rare Subtypes: The following subtypes exist, but no subtyping recommended in regulatory toxicity studies: Adenomatous: Consists mainly of the epithelial component Fibromatous: The connective tissue predominates
Differential diagnoses Hyperplasia: Regular lobular architecture is maintained Adenoma: Lacks any increased connective fibrous tissue. Tumor well demarcated from the adjacent mammary tissue. The regular architecture of the gland is distorted. Fibroma (Figure 23): Lacks any epithelial elements. Adenocarcinoma arising in fibroadenoma: Adenocarcinomatous change within fibroadenoma.
Comments: Adenocarcinomatous change within fibroadenomas should be diagnosed as “adenocarcinoma arising in fibroadenoma.”
Adenomyoepithelioma: mammary gland (Figure 24)
Pathogenesis/cell of origin: Two cell types are involved including mammary luminal epithelium and mammary myoepithelium. Studies using promoters to introduce mutant Pten or PIK3 suggest the origin is in a luminal precursor cell (Meyer et al. 2011). The WHO classification considers these tumors to be of myoepithelial origin (WHO 2003). Other authorities believe these lesions are similar too, if not identical to, human fibroadenomas and should be considered a third subtype of fibroadenoma (Borowsky 2011).
Diagnostic features Well-defined polypoid lesions Combination of acinar structures with myoepithelium and an abundant stroma with SMA positive spindle cells Acinar structures are uniform and lined by a uniform luminal type of epithelium Mitotic figures are rare
Differential diagnoses Hyperplasia: Regular lobular architecture is maintained Adenoma: Lacks any increased connective fibrous tissue. Tumor well demarcated from the adjacent mammary tissue. The regular architecture of the gland is distorted Adenocarcinoma: Structural or cellular pleomorphism. Detachment of epithelial cells from the basement membrane. Infiltrative growth; occurrence of distant metastasis
Comment: Only reported in mice.
Tumor, mixed, benign: mammary gland
Pathogenesis/cell of origin: At least two different cell types including mammary epithelium and mesenchymal cells
Diagnostic features Contains at least two different benign components of mammary tissue An epithelial component and connective tissue components other than fibrous tissue (e.g. fat, cartilage, or bone) must be present Adenolipoma type: Consists of uniform epithelial ductules or alveoli and mature adipose tissue
Differential diagnoses Fibroadenoma: Consists only of acinar and/or ductal epithelial cells and proliferating connective fibrous tissue Carcinosarcoma: Both of the components have malignant features
Comments: “Tumor, mixed, benign” summarizes all possible mixtures of benign epithelial and mesenchymal tumor components. According to well-established diagnostic criteria, “fibroadenomas” are separately classified, although they are benign mixed tumors.
Carcinoma, adenosquamous: mammary gland (Figure 25)
Synonym: Adenoacanthoma, malignant; keratoacanthoma
Pathogenesis/cell of origin: Mammary gland epithelium
Diagnostic features May be well circumscribed Differentiation is glandular and squamous. Glandular and squamous patterns are present in varying proportions Squamous areas are well differentiated Squamous cell areas cover more than 25% of the lesion Areas of gradual transition may be present from round or polygonal epithelial cells to flattened squamous cells Squamous cells exhibit intracytoplasmic keratin and pearl formation Metastasis exhibits squamous or glandular tissue
Differential diagnoses Adenocarcinoma: Squamous areas may be present, but cover not more than 25% of the lesion Carcinoma, squamous cell (skin): Only squamous differentiation is present. No evidence of glandular pattern.
Comments: Only reported in mice.
Adenocarcinoma: mammary gland (Figures 26 and 27)
Pathogenesis/cell of origin: Mammary gland epithelium
Diagnostic features Structural pleomorphism Variability in growth pattern is high and include tubular, papillary, cystic, solid, comedo, and undifferentiated subtypes (see below) Different growths pattern may be present within a single lesion Epithelium arranged in tubular or gland-like structures Loss of tubulo-alveolar pattern possible Acinar spaces show great variety in size and shape but may sometimes be regular and orderly. In wnt tumors, acinae are typically surrounded by myoepithelium Often acinar spaces are completely filled by neoplastic cells detached from basement membrane Intracystic papillary projections, solid cords, sheets, nests, or tubes with no signs of glandular differentiation may be present Acinar areas are frequently associated with cysts filled with blood, or fluid as evidence of secretory activity Secretory activity may occur Stroma within the tumor is usually scanty Areas of necrosis, ulceration, and hemorrhage may occur Neoplastic cells can be columnar or cuboidal and form one cell or several cell layers. Pronounced cellular and nuclear pleomorphism, nuclear hyperchromasia, and mitotic figures are common; nucleoli are often prominent; atypia may occur Areas of squamous differentiation of neoplastic epithelial cells may occur Squamous areas cover not more than 25% of the lesion (mouse) Invasion of adjacent tissues, muscle, or skin may be present Metastasis may occur. Most mouse mammary tumors have an expansile growth margin. Metastasis may occur by “non-invasive intravascular metastasis” Subtypes: Usually several subtypes exist in any given tumor. No subtyping recommended in regulatory toxicity studies Alveolar/tubular: Composed predominantly of alveolar or tubular structures Comedo: Multilayered epithelium surrounding central necrotic debris Cribriform: Solid sheets of neoplastic epithelial cells interrupted by round or irregularly shaped secondary lumina of variable size Cystic: Simple or multiloculated cystic glandular structures Medullary/solid: Rich in parenchymal tissue; sparse stroma Papillary: Papillary structures predominate. Scirrhous: Rich in stroma, often sclerotic, sparse epithelial components Spindle cell: Spindle-shaped cells form solid tumor structures Undifferentiated: Neoplastic epithelial cells that do not have characteristics that allow for subtyping into any of the above
Differential diagnoses Adenoma: Tumor well demarcated from the adjacent mammary tissue. The regular architecture of the gland is distorted. Epithelium single or double layered. Epithelial cells are still attached to the basement membrane Adenocarcinoma arising in fibroadenoma: Regarded as a separate entity. Adenocarcinomatous change within fibroadenoma Carcinosarcoma: Epithelial and mesenchymal neoplastic components that are both malignant Carcinoma, adenosquamous: Squamous cell areas cover more than 25% of the lesion Hyperplasia with atypia: Either focal, irregular proliferation of ductal or alveolar epithelium within a lobular hyperplasia or acinar, irregular proliferation of one or more alveoli or ducts within normal mammary gland tissue
Comments: Tumors with focal adenocarcinomatous change within adenomas are diagnosed as adenocarcinomas. The Dunn Classification has been the traditional classification of mouse mammary tumors (Cardiff and Kenney 2007). Thelma Dunn grouped tumors into Types A, B, C, and P (microacinar, ductal, and pregnancy-dependent). This classification was based on MMTV-induced “spontaneous” mammary tumors that are now known to be related to insertion activation of Wnt, Fgf, and Notch genes (Cardiff et al. 2006).
Adenocarcinoma arising in fibroadenoma: mammary gland (Figure 28A)
Pathogenesis/cell of origin: Mammary gland epithelium and connective tissue
Diagnostic features Focally adenocarcinomatous change within a well-defined primary fibroadenoma The histological pattern of the adenocarcinomatous component is variable Multiple layers of epithelial cells, pronounced pleomorphism and atypia of epithelial cells, and cellular hyperchromasia in the adenocarcinomatous areas Individual cells or nests of cells may infiltrate stroma
Differential diagnoses Fibroadenoma: Generally uniform and well-differentiated epithelial cells. Composed of proliferated glandular epithelium surrounded by layers of fibrous tissue. Only small focal areas of atypia and/or pleomorphism may occur Adenocarcinoma: Develops as primary carcinoma, whereas “adenocarcinoma arising in fibroadenoma” is a focally malignant change within a primary fibroadenoma
Comment: This tumor is observed in rats.
Sarcoma arising in fibroadenoma: mammary gland (Figure 28B)
Pathogenesis/cell of origin: Mammary gland epithelium and connective tissue
Diagnostic features Focally sarcomatous change within a well-defined primary fibroadenoma The sarcomatous part is characterized by fascicles of uniform tumor cells producing various amounts of long intertwining collagen bundles Monomorphic, fusiform spindle cells often having a characteristic “herring-bone” cellular pattern Mitotic figures may be frequent
Differential diagnoses Fibroadenoma: Only uniform and well-differentiated mesenchymal and epithelial cells. Composed of proliferated glandular epithelium surrounded by layers of proliferated fibrous tissue. No histological features of malignancy are present Fibrosarcoma: Develops as primary sarcoma, whereas “sarcoma arising in fibroadenoma” is a focally malignant change within a primary fibroadenoma
Carcinosarcoma: mammary gland (Figure 29)
Pathogenesis/cell of origin: At least two different cell types including mammary gland epithelium and mesenchymal cells
Diagnostic features This tumor is composed of epithelial cells and mesenchymal tissue elements Both epithelial and mesenchymal tissue element have malignant features Mixtures of all types of carcinomatous and sarcomatous components may be present Any element may predominate Infiltrative growth, penetration of vessels, and metastasis may occur
Differential diagnoses Tumor, mixed benign: Consists of different components, i.e., epithelial and connective tissue other than fibrous tissue (e.g., cartilage or bone). No sign of malignancy Adenocarcinoma, arising in fibroadenoma: Adenocarcinomatous change within fibroadenoma Sarcoma arising in fibroadenoma: Sarcomatous change within fibroadenoma
Comments: “Carcinosarcoma” summarizes all possible mixtures of epithelial and mesenchymal tumor cell components, where both components are malignant. According to well-established diagnostic criteria, “adenocarcinoma arising in fibroadenoma” is regarded as a separate entity. This category contains the EMT tumor phenotype that has previously been named a carcinosarcoma or a spindle cell tumor of the mammary gland (Cardiff 2010; Radaelli et al. 2009). However, these tumors have been identified as a distinct tumor arising from EMT with tumor cell loss of oncogene addiction or loss of or mutation of p53 (Damonte et al. 2007; Debies et al. 2008). The EMT tumor phenotype is characterized by a predominance of spindle cells which stain for both epithelial and mesenchymal markers (Damonte et al. 2007). The phenotype is now recognized in the human molecular classifications as a rare “Claudin Low” tumor (Herschkowitz et al. 2012).
Zymbal’s Gland
The Zymbal’s or auditory sebaceous gland is most commonly described in rats. However, a small auditory sebaceous gland is also present in mice (Seely and Boorman 1999). In rats, the gland is 3 to 5 mm in diameter and located anterior-ventral to the ear canal. It is composed of 3 to 4 lobules, each with an intralobular duct emptying into an excretory duct which empties into the ear canal. There are two smaller sebaceous glands underlying the epithelium of the ear canal which are included as part of the Zymbal’s gland by some. These are holocrine glands where the secretion is formed by mature acinar cells degenerating to become the secretory product (Haines and Eustis 1990; Nielsen 1978). Spontaneous tumors are occasionally seen and have also been induced by chemical carcinogens.
Degenerative, inflammatory, vascular, and other non-neoplastic changes observed in Zymbal’s gland are largely similar to those observed in the mammary gland. Specific examples of these non-neoplastic changes are displayed in Figures 30 and 31.
Degenerative Changes
Degeneration: Ductular/Sebaceous Epithelium
Pathogenesis/cell of origin: Ductular, sebaceous epithelial cell
Diagnostic features Epithelial cells swollen Epithelial vacuolization/bleb formation Loss of organization of the cell layers Dilatation (ectasia) of alveoli or ducts with accumulation of secretory material
Differential diagnoses Postmortem autolysis: Uniform dissolution of entire tissue with no change in organization or depth of cell layers Atrophy: Thinning of affected ducts or alveoli
Single-cell Necrosis: Ductular/Sebaceous Epithelium
Synonym: Apoptosis
Pathogenesis/cell of origin: Sebaceous or ductular epithelium
Diagnostic features Cells usually shrunken with distinct cell membranes Membrane budding, condensed nuclei Rarely associated with inflammation
Differential diagnoses Necrosis: Cells may be swollen. Cytoplasmic eosinophilia, nuclear pyknosis/karyorhexis, cell debris, more likely associated with inflammation and thinning of epithelium
Necrosis: Ductular/Sebaceous Epithelium
Pathogenesis/cell of origin: Sebaceous or ductular epithelium
Diagnostic features Cellular swelling or shrinkage Cytoplasmic eosinophilia Pyknosis or karyorrhexis of nuclei Exfoliation of cells May result in thinning of epithelium May be associated with inflammation Luminal accumulations of fibrin and/or cell debris
Differential diagnoses Postmortem autolysis: Uniform dissolution of entire tissue with no change in organization or depth of cell layers Single-cell Necrosis: Cells shrunken with distinct cell membranes, membrane budding, condensed nuclei; rarely associated with inflammation Degeneration: Cell vacuolation but no inflammation or cell debris Atrophy: Thinning of cell layers but no inflammation or cell debris Inflammation: Cellular infiltrates and swelling but no exfoliation or cell debris.
Basophilia: Ductular/Sebaceous Epithelium
Synonym: Regeneration
Pathogenesis/cell of origin: Sebaceous or ductular epithelium
Diagnostic features Normal appearing epithelial cells with basophilic cytoplasm Increased nuclear:cytoplasmic ratio Epithelial architecture may appear irregular Mitoses Adjacent to or within areas of degenerating, necrotic, hyperplastic, or metaplastic epithelium
Differential diagnoses Hyperplasia: Epithelium is thickened due to increased numbers of cells, resulting in undulating, rugose epithelial surface and irregular arrangement of cell layers (see proliferative lesion section of this document) Neoplasia: Expansile nodule usually protruding into cavity, with cellular atypia and compression of adjacent structures (see proliferative lesion section of this document)
Comments: Basophilia is recommended as the preferred descriptive term for the lexicon. When basophilia is interpreted as evidence for regeneration (growth of cells and tissues to replace lost or damaged structures, as opposed to hyperplasia), it is recommended that this interpretation be included in the text of the report (Kumar et al. 2010). Irregularity of epithelial cell arrangement may be observed in the ductular or sebaceous epithelium in the process of regeneration. Degeneration, necrosis, and regeneration are often present together in Zymbal’s glands repeatedly injured by toxicants.
Inflammatory Changes
Inflammation of the Zymbal’s gland is uncommon. More commonly, histological changes are limited to infiltrates of foci of lymphocytes, neutrophils, macrophages, or plasma cells, alone or in combination.
Infiltrate: Zymbal’s Gland (Figure 30)
Pathogenesis/cell of origin: Sebaceous or ductular epithelium and associated tissues
Diagnostic features Infiltration of a relatively pure population of lymphocytes, plasma cells, neutrophils, eosinophils, macrophages, or a mixture of these cell types into the lamina propria of the lobule and associated tissues
Differential diagnoses Inflammation Hematopoietic neoplasia: Homogenous lymphocyte population infiltrating entire tissue and other sites
Comment: The base term infiltrate is recommended followed by the predominant cell type or “mixed” if there is not a predominant cell type. Leukocytic infiltrates consisting only of lymphocytes or plasma cells may indicate an immunologic effect of a xenobiotic but can also be observed as a spontaneous change.
Other Changes
Dilation: Zymbal’s Gland (Figure 31)
Synonyms: Cystic degeneration, cystic change
Modifier: Ductular
Pathogenesis/cell of origin: Ducts
Diagnostic features Dilated lumen with or without epithelial hypertrophy or hyperplasia Often luminal keratin or cell debris Often associated with inflammation
Comments: Dilation is a spontaneous aging change in rodents. Severity grades and report descriptions may be used to indicate the extent of ductal and sebaceous involvement and the size of the affected lumen diameters.
Growth Disturbances
Non-neoplastic
Acinar atrophy: Zymbal’s gland
Pathogenesis/cell of origin: Acinar cells
Diagnostic features May have degeneration of sebaceous cells Acini diminished in size Consist only of a single layer of cuboidal cells or squamous cells Individual acinar cells having reduced amount of cytoplasm contain yellow-brown granular pigmentation interstitial collagen increased with scattered inflammatory cells Ducts can be dilated and filled with inspissated secretory material and inflammatory cells Only ductal dilatation and cyst formation observed in the mouse
Hyperplasia: Zymbal’s gland
Pathogenesis/cell of origin: Sebaceous cells or duct cells
Diagnostic features
Subtype: Sebaceous cell Lobular pattern retained Slight compression of adjacent tissue Affected acini enlarged/ partially fused Normal maturation sequence from periphery to center may be less apparent Cytoplasm more basophilic and less foamy than that of normal cells Nuclei can be enlarged and contain one or more prominent nucleoli Proliferation of sebaceous cells with preserved, normal architecture of sebaceous glands Can be associated with hypertrophy
Subtype: Squamous cell Focal increase in thickness of the squamous epithelium with formation of folds and short papillary projections into the duct lumen Often rete ridges form
Differential diagnoses Adenoma, sebaceous cell: Sebaceous adenomas may contain a large number of germinative (basaloid) cells. The regular architecture of sebaceous glands is distorted Papilloma, squamous cell: Arborizing pattern of papillae, stratified squamous epithelium overlies cores of connective tissue
Comments: Mostly focal; diffuse hyperplasia has been observed with administration of 3,3-dimethoxybenzidine
Neoplastic
Many Zymbal’s gland neoplasms are not large enough to be recognized by gross necropsy. They are therefore missed in studies where the Zymbal’s gland is not taken as a standard protocol organ.
Adenoma, sebaceous cell: Zymbal’s gland (Figure 32)
Pathogenesis/cell of origin: Sebaceous cells
Diagnostic features Structural resemblance to normal gland, but regular sebaceous gland architecture is not maintained Well defined but not encapsulated Lobulated structure Exophytic and endophytic growth characteristics occur Most tumors consist of a mixture of basaloid cells, cells transitional in character between basaloid cells and mature sebaceous cells Many of the tumor cells show the prominent cytoplasmic vacuolation characteristics of sebaceous gland cells There are a certain number of actively mitotic germinative (basaloid) cells which may predominate in some lobules Cystic areas are often observed derived from intense production of sebum Size equal or greater in size than an individual lobule Mitotic figures may be present Cells are usually small and nuclei are partly pyknotic
Differential diagnoses Hyperplasia, sebaceous cell: Characterized by normal architecture, an increased number of mature glandular cells, and only a small number of germinative (basaloid) cells Carcinoma, sebaceous cell subtype: Characterized by poorly differentiated sebaceous gland cells, invasive growth pattern, and cellular and nuclear atypia Carcinoma, basal cell: (Mixed) basal cell carcinomas show only few areas of sebaceous differentiation and a predominantly non-glandular pattern with a clear predominance of basaloid tumor cells
Papilloma, squamous cell: Zymbal’s gland
Pathogenesis/cell of origin: Ductal cells
Diagnostic features Arise from main duct epithelium Complex arborizing structure Stratified squamous epithelium overlies cores of connective tissue Papillary growth of cornified squamous epithelium into duct lumina No cellular atypia Usually associated with glandular proliferation
Differential diagnoses Carcinoma, squamous cell subtype: Presence of invasive growth and of cellular or nuclear atypia
Carcinoma: Zymbal’s gland
Pathogenesis/cell of origin: sebaceous cells, duct epithelium
Diagnostic features
Subtype: Sebaceous cell Often ulcerated Large irregular acini Usually ductless Can contain cystic cavities with sebum, keratin, and necrotic cells Papillary projections of squamous epithelium can occur in larger cystic cavities Central portion can show cystic changes with sebaceous material, degenerated cells, leukocytes Cells in basal layer small with hyperchromatic cytoplasm Upper layers pleomorphic One to two nucleoli in round to oval nuclei Cytoplasm light, granulated, or vacuolated Frequent mitoses Stroma shows proliferation of fibroblasts which can be pleomorphic and invade into adjacent tissue Squamous metaplasia of sebaceous epithelium possible Varying proportions of squamous and sebaceous cells
Subtype: Squamous cell May or may not have keratinization Penetration of basal lamina by invasive growth; Invasion of the dermis and striated muscle by nests or cords of squamous cells Varying cellular differentiation of squamous cells Malignancy is furthermore characterized by variation of size and staining of nuclei, atypical mitotic figures, and loss of intercellular bridges
Differential diagnoses Papilloma, squamous cell: No invasive growth and of cellular or nuclear atypia Adenoma, sebaceous cell: Absence of invasive growth and of cellular and nuclear atypia
Comment: Carcinoma as a diagnosis is sufficient for regulatory toxicology studies. Carcinomas are often mixtures of the two subtypes. Carcinomas, squamous cell subtype of the Zymbal’s gland may be difficult to differentiate from squamous cell carcinomas arising from the adjacent skin. Sebaceous cell carcinomas can be induced by various compounds including cupferron, chloroprene, 1,3-butadiene, benzene, benzidines, and aminostilbenes.
Preputial/Clitoral Gland
Preputial and clitoral glands are found in both rats and mice. They are paired modified sebaceous glands located in the inguinal region adjacent to the penis and vagina respectively (Figure 33). In males, the preputial gland empties into the preputial cavity and in females, the clitoral gland duct empties into the clitoral fossa. The growth and secretory activity of preputial and clitoral glands are regulated primarily by testosterone and the pituitary hormones, ACTH, GH, and PRL. Administration of testosterone, but not estrogens, to male or female rats causes hypertrophy and hyperplasia of acinar cells. Castration of adult male rats results in atrophy. In rats, large intracytoplasmic eosinophilic granules are a prominent feature. These granules contain pheromones (aliphatic alcohols) and beta-glucuronidase and are not present in mice. Degenerative, inflammatory, vascular, and other non-neoplastic changes observed in preputial/clitoral glands are largely similar to those observed in the mammary gland. Specific examples of these non-neoplastic changes are displayed in Figures 34 and 35. Dilated ducts containing keratin-like material with accompanying inflammatory changes is the most common lesion observed. Tumors are occasionally seen as spontaneous lesions but can be induced by a number of chemical carcinogens.
Degenerative Changes
Degeneration: Ductular/Sebaceous Epithelium
Pathogenesis/cell of origin: Ductular, sebaceous epithelial cell
Diagnostic features Epithelial cells swollen Epithelial vacuolization/bleb formation Loss of organization of the cell layers Dilatation (ectasia) of alveoli or ducts with accumulation of secretory material
Differential diagnoses Postmortem autolysis: Uniform dissolution of entire tissue with no change in organization or depth of cell layers Atrophy: Thinning of affected ducts or alveoli
Single-cell Necrosis: Ductular/Sebaceous Epithelium
Synonym: Apoptosis
Pathogenesis/cell of origin: Sebaceous or ductular epithelium
Diagnostic features Cells usually shrunken with distinct cell membranes Membrane budding, condensed nuclei Rarely associated with inflammation
Differential diagnoses Necrosis: Cells may be swollen. Cytoplasmic eosinophilia, nuclear pyknosis/karyorhexis, cell debris, more likely associated with inflammation and thinning of epithelium
Necrosis: Ductular/Sebaceous Epithelium
Pathogenesis/cell of origin: Sebaceous or ductular epithelium
Diagnostic features Cellular swelling or shrinkage Cytoplasmic eosinophilia Pyknosis or karyorrhexis of nuclei Exfoliation of cells May result in thinning of epithelium May be associated with inflammation Luminal accumulations of fibrin and/or cell debris
Differential diagnoses Postmortem autolysis: Uniform dissolution of entire tissue with no change in organization or depth of cell layers Apoptosis: Cells shrunken with distinct cell membranes, membrane budding, condensed nuclei; rarely associated with inflammation Degeneration: Cell vacuolation but no inflammation or cell debris Atrophy: Thinning of cell layers but no inflammation or cell debris Inflammation: Cellular infiltrates and swelling but no exfoliation or cell debris.
Basophilia: Ductular/Sebaceous Epithelium
Synonym: Regeneration
Pathogenesis/cell of origin: Sebaceous or ductular epithelium
Diagnostic features Normal appearing epithelial cells with basophilic cytoplasm Increased nuclear:cytoplasmic ratio Epithelial architecture may appear irregular Mitoses Adjacent to or within areas of degenerating, necrotic, hyperplastic, or metaplastic epithelium
Differential diagnoses Hyperplasia: Epithelium is thickened due to increased numbers of cells, resulting in undulating, rugose epithelial surface and irregular arrangement of cell layers (see proliferative lesion section of this document) Neoplasia: Expansile nodule usually protruding into cavity, with cellular atypia and compression of adjacent structures (see proliferative lesion section of this document)
Comments: Basophilia is recommended as the preferred descriptive term for regulatory toxicology. When basophilia is interpreted as evidence for regeneration (growth of cells and tissues to replace lost or damaged structures, as opposed to hyperplasia), it is recommended that this interpretation be included in the text of the report (Kumar et al. 2010). Irregularity of epithelial cell arrangement may be observed in the ductular or sebaceous epithelium in the process of regeneration. Degeneration, necrosis, and regeneration are often present together in preputial/clitoral glands repeatedly injured by toxicants.
Inflammatory Changes
Inflammation of the preputial or clitoral glands is uncommon. More commonly, histological changes are limited to infiltrates of foci of lymphocytes, neutrophils, macrophages, or plasma cells, alone or in combination. In male mice, inflammation of the preputial gland can be a consequence of fight wounds in gang-housed mice.
Infiltrate: Preputial/Clitoral Gland
Pathogenesis/cell of origin: Sebaceous or ductular epithelium and associated tissues
Diagnostic features Infiltration of a relatively pure population of lymphocytes, plasma cells, neutrophils, eosinophils, macrophages, or a mixture of these cell types into the lamina propria of the lobule and associated tissues
Differential diagnoses Inflammation Hematopoietic neoplasia: Homogenous lymphocyte population infiltrating entire tissue and other sites
Comment: The base term infiltrate is recommended followed by the predominant cell type or “mixed” if there is not a predominant cell type. Leukocytic infiltrates consisting only of lymphocytes or plasma cells may indicate an immunologic effect of a xenobiotic but can also be observed as a spontaneous change.
Inflammation: Preputial/Clitoral Gland (Figure 34)
Pathogenesis/cell of origin: Sebaceous or ductular epithelium and associated tissues
Modifiers: Neutrophilic, eosinophilic, lymphocytic, plasmacytic, histiocytic, or a combination of the aforementioned
Diagnostic features Acute: Vascular congestion, edema, accumulation of serous, mucous, or fibrinous exudates, neutrophils, sloughed epithelial cells in tubular lumens Chronic: Cellular infiltrate is predominantly lymphocytes, plasma cells, and macrophages; regenerative, hyperplastic, and/or metaplastic changes in the affected epithelium and fibroplasia may be present Chronic active inflammation: Cellular infiltrate is a mixture of granulocytic, lymphocytic, histiocytic cells; regenerative, hyperplastic, and/or metaplastic changes in the affected epithelium and fibroplasia may be present Granulomatous: Cellular infiltrate is predominantly plump macrophages (epithelioid cells) which may form interlacing bundles, accompanied by lymphocytes, plasma cells, and fibrosis, depending on duration and etiologic agent. Etiologic agent may be visible, e.g., fungi, mycobacteria, or foreign bodies. Regenerative, hyperplastic, and/or metaplastic changes in the affected epithelium and fibroplasia may be present. Granulocytes may be present in the affected area, in which case the process could be described as pyogranulomatous inflammation
Differential diagnoses Necrosis: Pyknosis, karyorrhexis of nuclei, cytoplasmic swelling or shrinkage, cellular debris, accompanied by inflammatory infiltrate
Comments: Inflammation need not be recorded with a chronicity or cell type descriptor; instead, the characteristics of the inflammation are best described in report text. In acute inflammation, migration of neutrophils into ductal lumens produces a suppurative exudate. The term chronic active inflammation implies recurrence or persistent presence of granulocytic inflammatory cells concurrent with ongoing chronic inflammation. Granulomatous inflammation is commonly seen in rats associated with rupture of dilated ducts containing keratin-like material.
Other Changes
Dilation: Preputial/Clitoral Gland (Figure 35)
Synonyms: Cystic degeneration, cystic change
Modifier: Ductular
Pathogenesis/cell of origin: Ducts
Diagnostic features Dilated lumen with or without epithelial hypertrophy or hyperplasia Often luminal keratin or cell debris Often associated with inflammation
Comments: Dilation is a spontaneous aging change in rodents. Severity grades and report descriptions may be used to indicate the extent of ductal involvement and the size of the affected lumen diameters.
Growth Disturbances
Non-neoplastic
Atrophy: Preputial/Clitoral Gland
Pathogenesis/cell of origin: Acinar cells
Diagnostic features Acini diminished in size Increased degeneration of sebaceous cells Consist only of a single layer of cuboidal cells or squamous cells Individual acinar cells having reduced amount of cytoplasmic contain yellow-brown granular pigmentation Interstitial collagen more prominent with scattered inflammatory cells Ducts can be dilated and filled with inspissated secretory material and inflammatory cells
Hyperplasia: Preputial/Clitoral Gland
Hyperplasia
Modifier: Acinar; Ductular
Pathogenesis/cell of origin: Acinar/ductular epithelium
Diagnostic features
Acinar: Hyperplastic acinar cells are similar to normal acinar epithelium Central round nucleus with one or rarely two large nucleoli Condensation of heterochromatin on the nuclear envelope Cytoplasm contains small vacuoles and typical eosinophilic hyalinized granules Focal hyperplasia, foci are surrounded by dark flat basal cells Foci show lobulation similar to normal glands
Ductular: May occur focal, multifocal, or generalized Acanthosis and hyperkeratosis of ductular squamous epithelium frequently secondary to inflammation Epithelium consists of 3 to 7 flat layers of squamous epithelium
Differential diagnoses Adenoma: Well circumscribed; usually solid growth, loss of normal structure Squamous cell papilloma: Compression of surrounding duct structure and/or destruction of ducts, complex arborizing structure
Comments: This lesion occurs spontaneously, mainly in older animals. It may be misdiagnosed as a mammary gland lesion especially if anatomical location at necropsy is misleading. In older mice, this lesion is associated with dilatation of ducts and chronic inflammation. The lesion may be unilateral or bilateral. It is not considered a pre-neoplastic change in these older mice but rather a reactive hyperplasia, and there is no evidence of progression. Squamous cell hyperplasia is also associated with squamous cell papilloma but has not been studied as precursor for neoplasia.
Neoplastic
Many preputial or clitoral gland neoplasms are not large enough to be recognized by gross necropsy. They are therefore missed in studies, where preputial and clitoral glands are not taken as standard protocol organs.
Adenoma: Preputial/Clitoral Gland
Pathogenesis/cell of origin: Acinar cells
Diagnostic features Masses on lower abdominal wall near genitalia Often bilateral, macroscopically firm, tan, and multilobulated Well circumscribed Compression may occur Capsule may be present Normally the general microarchitecture maintained, but cells are disorganized Usually solid growth mimicking normal glands Differentiation toward both sebaceous and squamous cells Eosinophilic granules in tumor cells Stratified acinar epithelial formations with dark-staining sebaceous cells with little cellular or structural atypia Necrosis minimal Low degree of atypia and few mitosis No invasive growth Cystadenomas contain cystic cavities filled with necrotic material
Differential diagnoses Carcinoma, mixed cell subtype: Mixture of sebaceous and squamous cells; invasion of surrounding tissue (skin, mammary gland); cellular atypia, frequent mitoses Carcinoma, squamous cell: Derived from ductal cells, no acinar involvement, rare Hyperplasia: Uniformity of cells (glandular or ductal tissue), no capsule
Comment: Spontaneous adenomas are extremely rare. Cystic and papillary growth variants as seen in the rat have not been described in the mouse.
Papilloma, Squamous Cell: Preputial/Clitoral Gland
Pathogenesis/cell of origin: Ductal cells
Diagnostic features Arise from main duct epithelium Complex arborizing structure Stratified squamous epithelium overlies cores of connective tissue Papillary growth of squamous epithelium into duct lumina, can be keratinizing or non-keratinizing No cellular atypia Usually associated with glandular proliferation
Differential diagnosis Carcinoma squamous cell (skin): Destruction of basal membrane; invasion into surrounding acinar tissue; cellular atypia and disorientation; frequent mitosis Adenoma acinar cell: Shows sebaceous differentiation Hyperplasia: Uniformity of cells (ductal tissue), no capsule
Adenocarcinoma: Preputial/Clitoral Gland (Figures 36–39)
Synonyms: Cystadenocarcinoma
Pathogenesis/cell of origin: Acinar cells
Diagnostic features Larger than adenomas, skin over tumors often ulcerated No regular border, infiltration of subcutaneous tissue Can be surrounded by thick fibrous capsule that is often infiltrated by acinar cells Loss of typical acinar structure Irregular acinar cell nests surrounded by thin connective tissue Some differentiation to squamous cells may occur Small sometimes cystic duct-like structures lined with squamous epithelium can be present Cystic cavities can contain keratin and necrotic material Nuclear/cellular atypia may occur High mitotic activity Subtypes: Solid, cystic, papillary, papillary cystic, and mixed cell types are described but subtyping is not recommended in regulatory toxicity studies
Solid No regular border; infiltrates the subcutaneous tissue Forms nests and loses the typical acinar structure Mitotic activity is high The duct system in glands containing solid adenocarcinomas shows squamous cell hyperplasia, dilatation, and is filled with inflammatory cells and necrotic cell debris
Cystic Contains multifocal cystic cavities of different sizes. These cavities represent dilated lumina of acini or small ducts Cysts frequently filled with necrotic material and inflammatory cells
Papillary The acinar cells form irregular papillary structures within grossly enlarged glands The mitotic activity is usually high In some areas, a basal cell involvement (basal cell hyperplasia) is noticeable
Papillary/cystic Has the same cellular composition as the cystic adenocarcinomas The difference is a formation of papillary tumor parts which grow into the cystic cavities
Mixed cell Mixture of sebaceous and squamous cells
Differential diagnoses Adenoma/Cystadenoma: Well circumscribed; usually solid growth more regular structure of acini, no invasion or atypia
Tumor, Basal Cell, Malignant: Preputial/Clitoral Gland
Synonym: Basal cell carcinoma
Pathogenesis/cell of origin: Basal cells
Diagnostic features Shows darker basophilic staining cells with elongated to oval nuclei and a high mitotic rate Cells are arranged in circumscribed nests with bundles of mesenchymal tissue between them Acinar cells and ducts are located around these tumors
Differential diagnoses Carcinoma, squamous cell (skin): Non-keratinizing squamous cells with dark stained cytoplasm; many mitotic figures Tumor, basal cell, malignant basal cell tumor of the skin: Lacks adjacent Preputial/Clitoral glandular structures and eosinophilic granules; anatomical location helpful
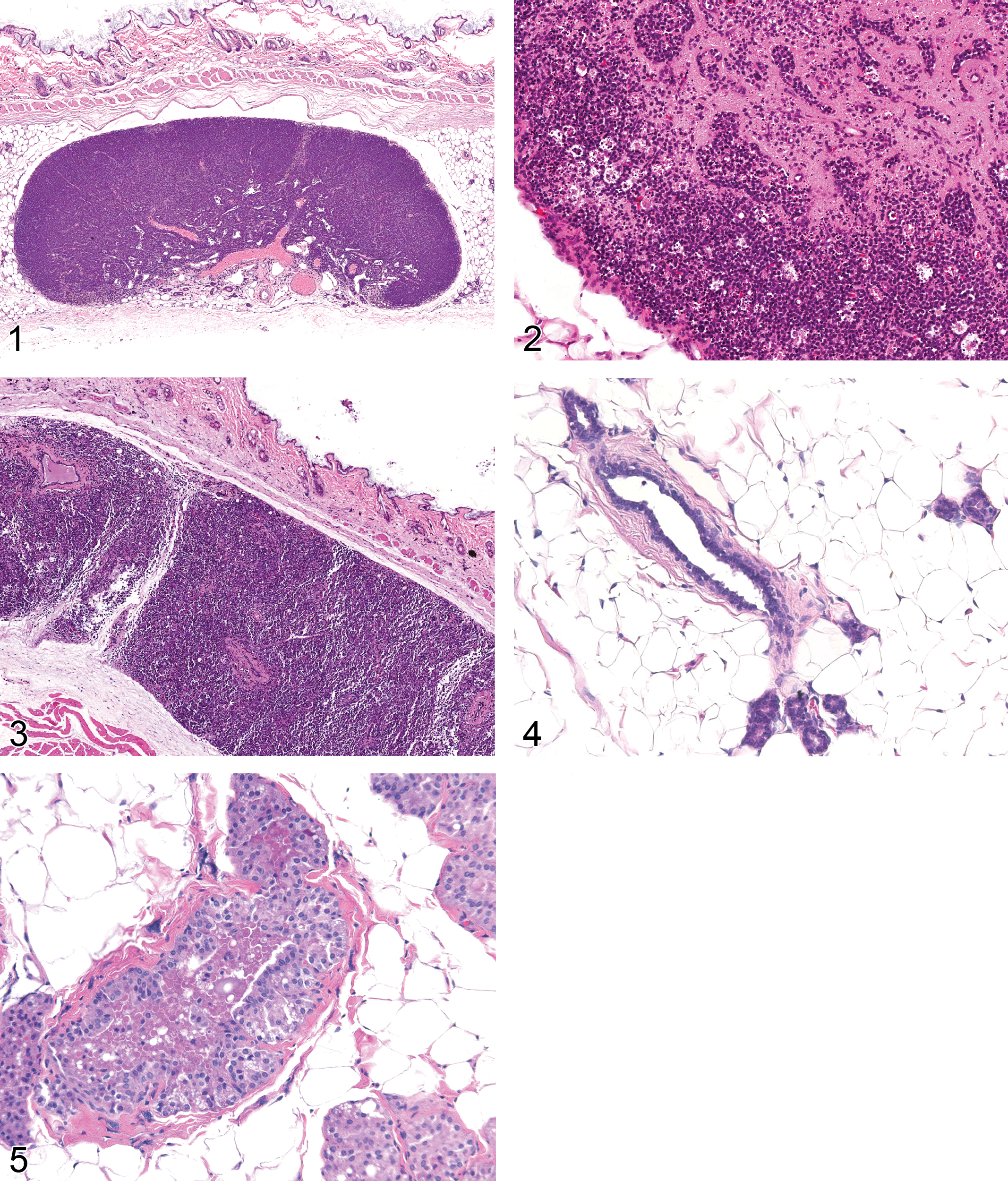
Normal mammary gland (inguinal) lymph node (female rat).
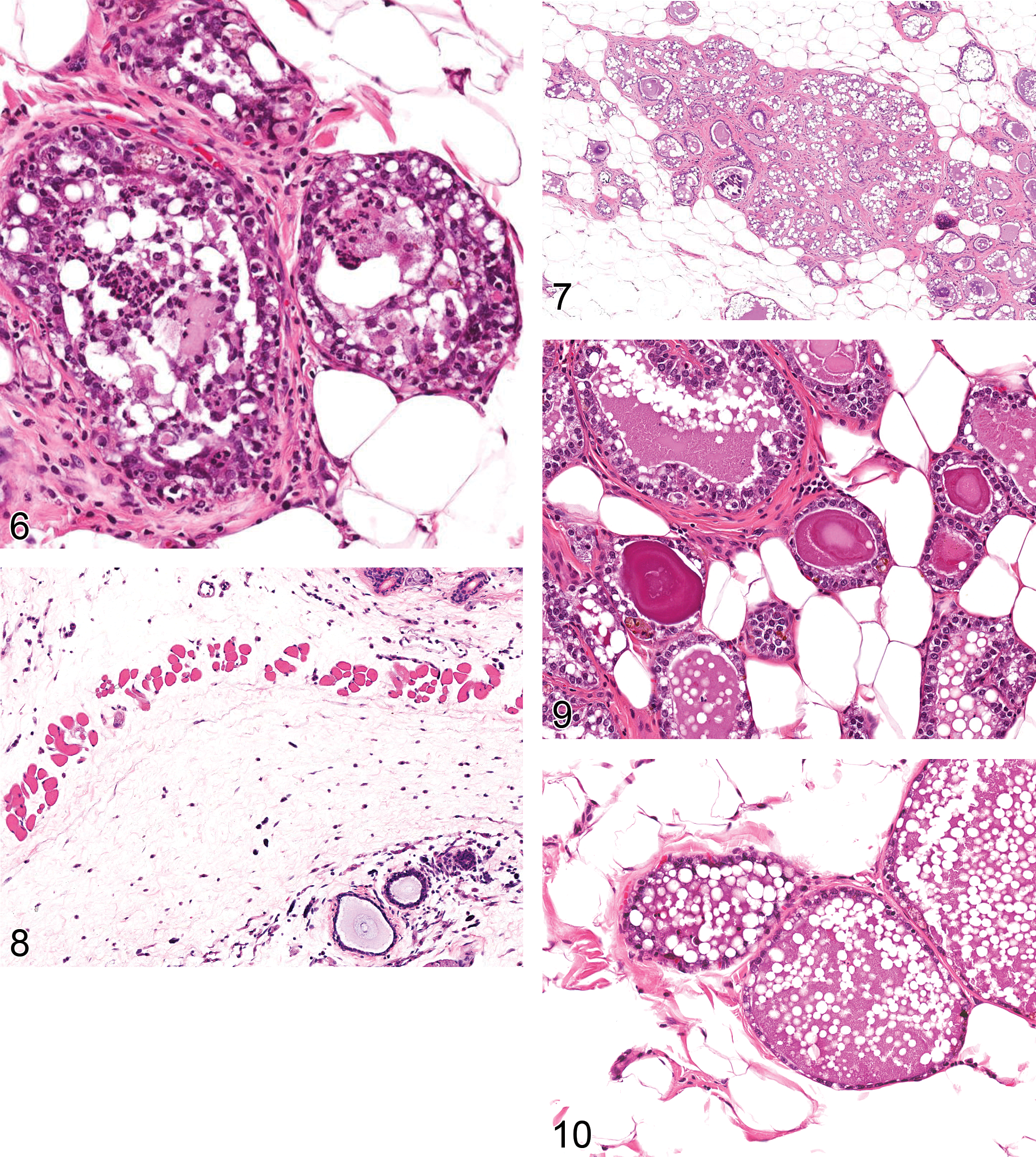
Mammary gland: acute inflammation (female rat).
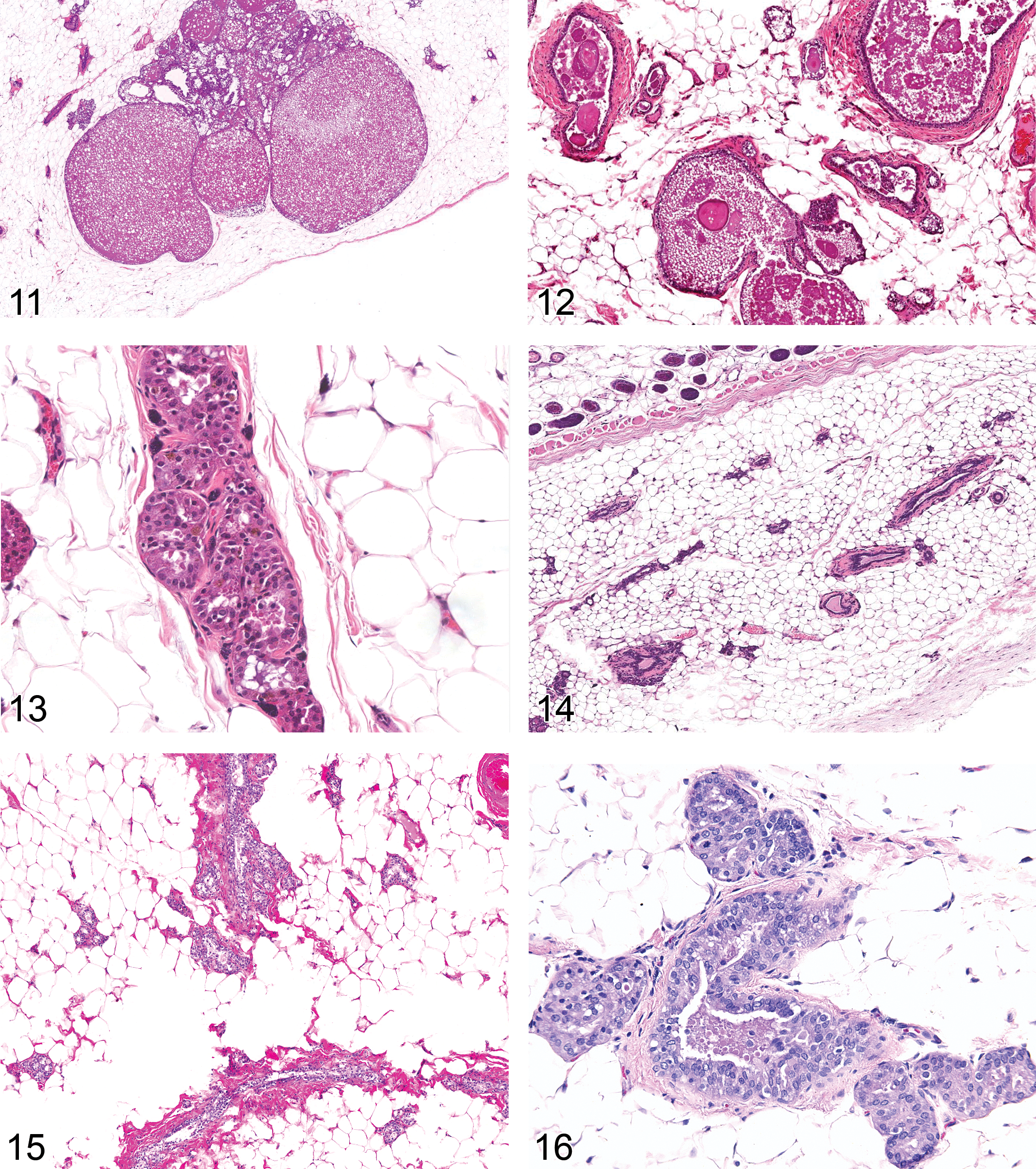
Mammary gland: ductal and alveolar dilation with secretions and/or vacuolation (female rat).
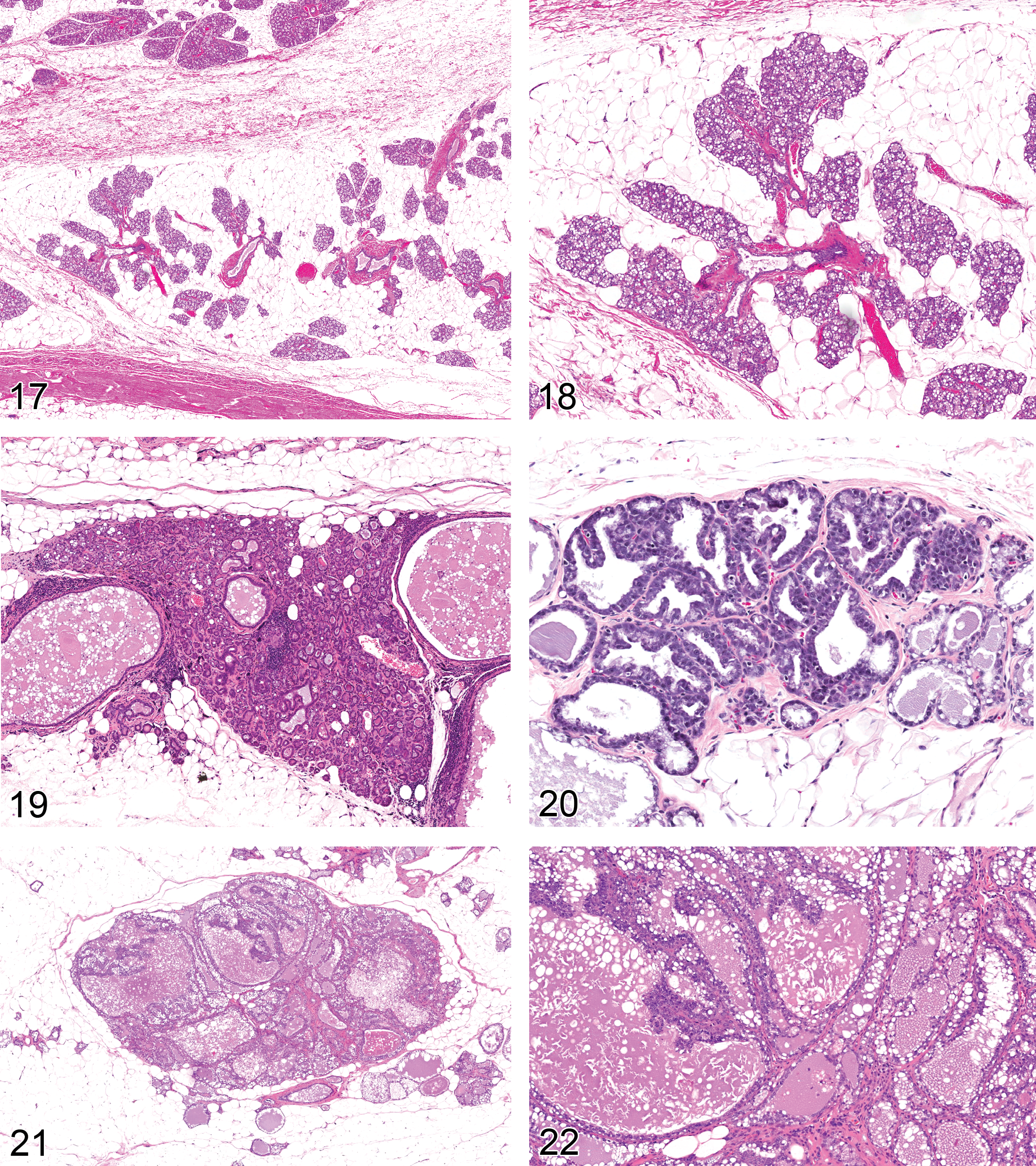
Mammary gland: hyperplasia, lobuloalveolar (female rat).
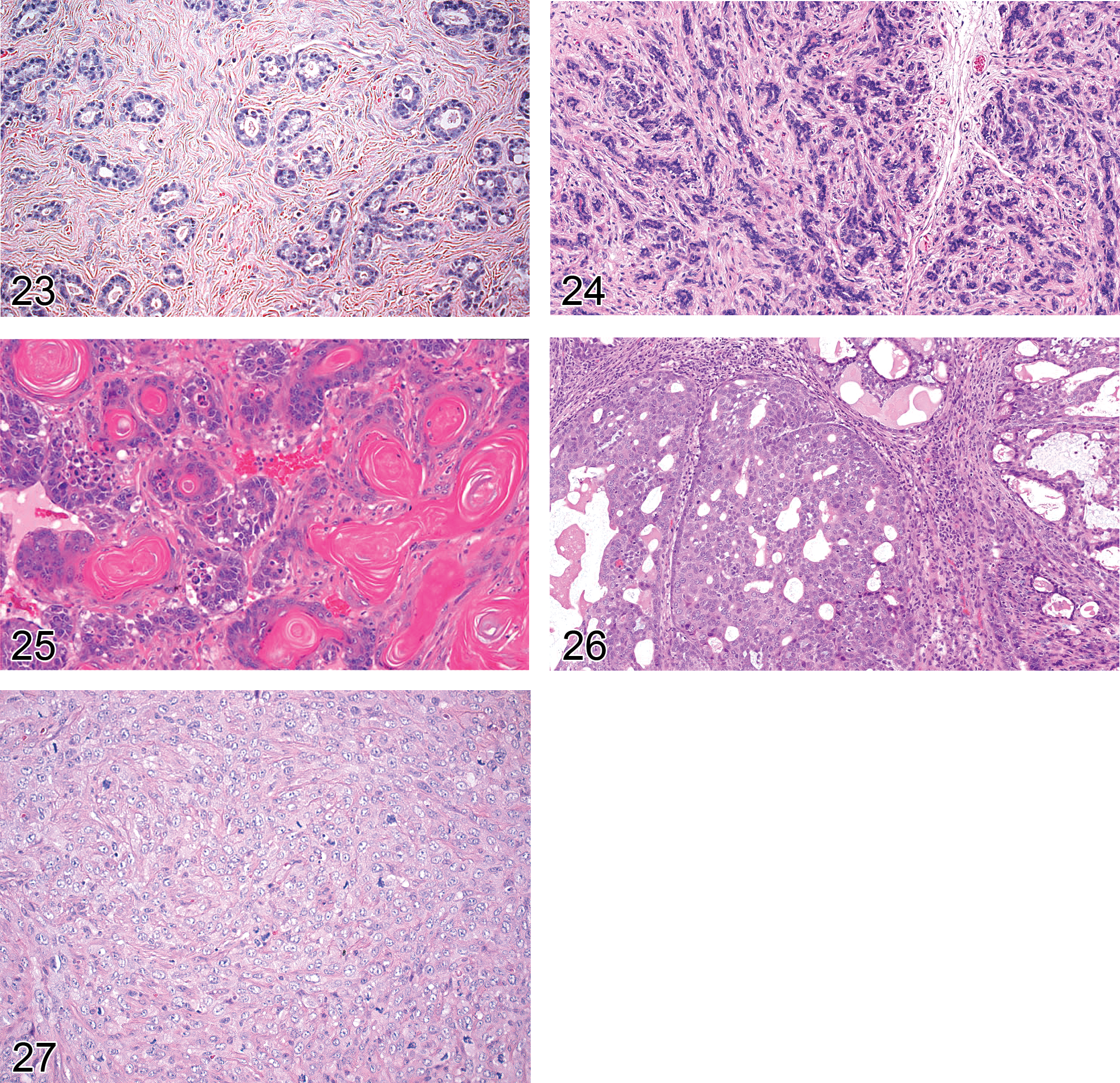
Mammary gland: fibroadenoma (female rat).
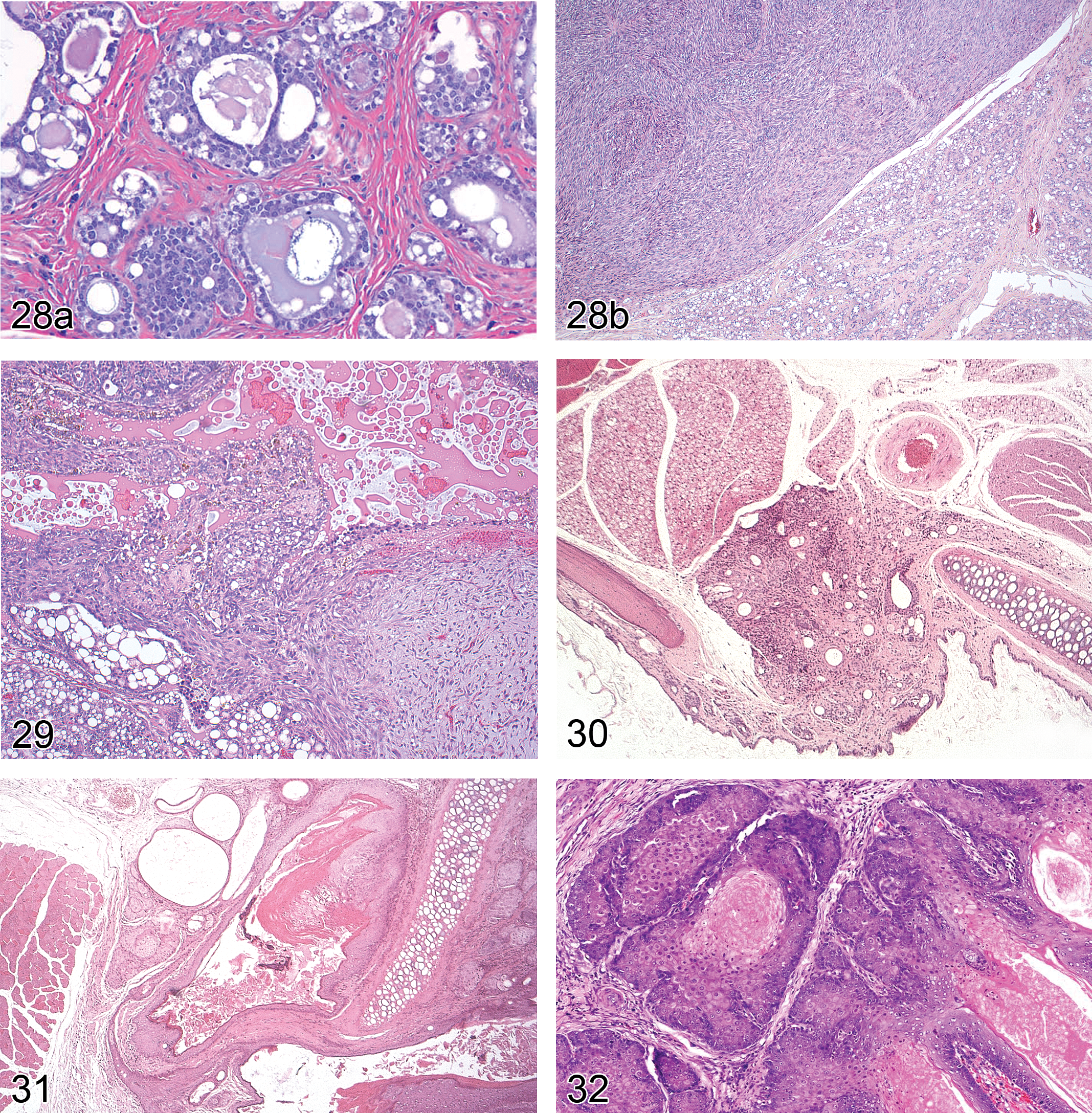
(A). Mammary gland: adenocarcinoma arising in fibroadenoma (female rat). (B). Mammary gland: sarcoma arising in fibroadenoma (female mouse).
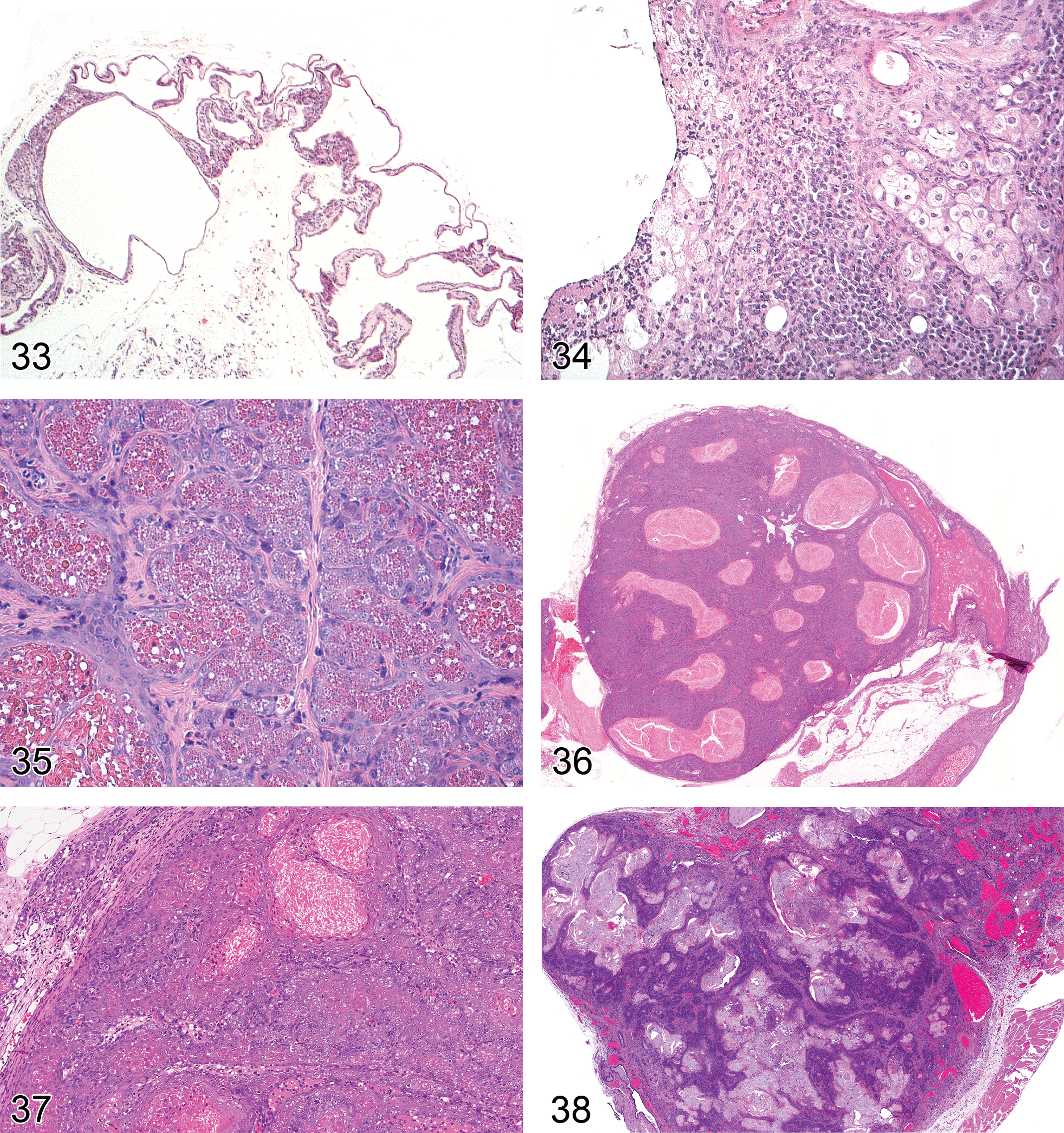
Preputial gland: acinar atrophy and dilation of ducts (mouse).
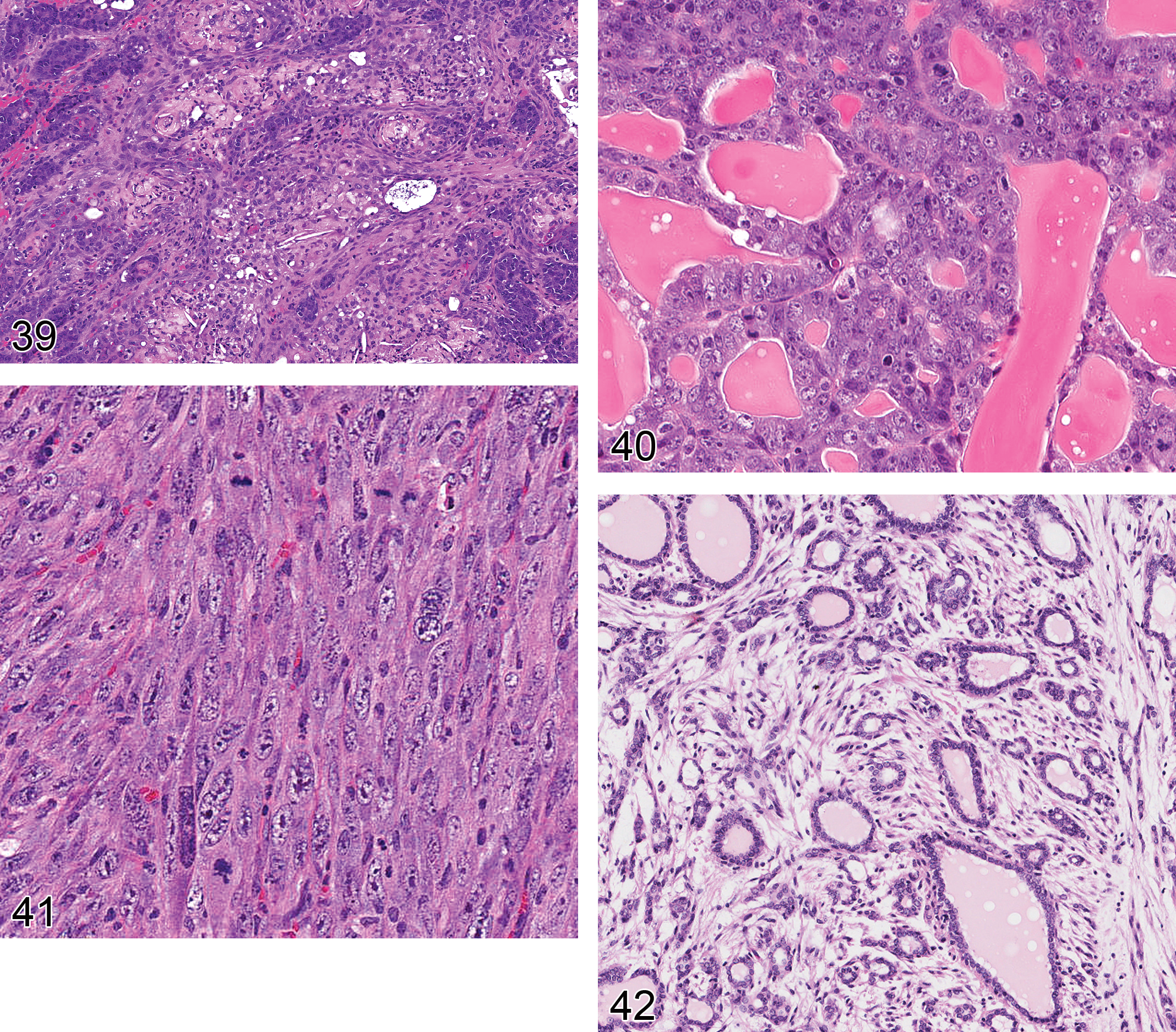
Clitoral gland: adenocarcinoma (rat).
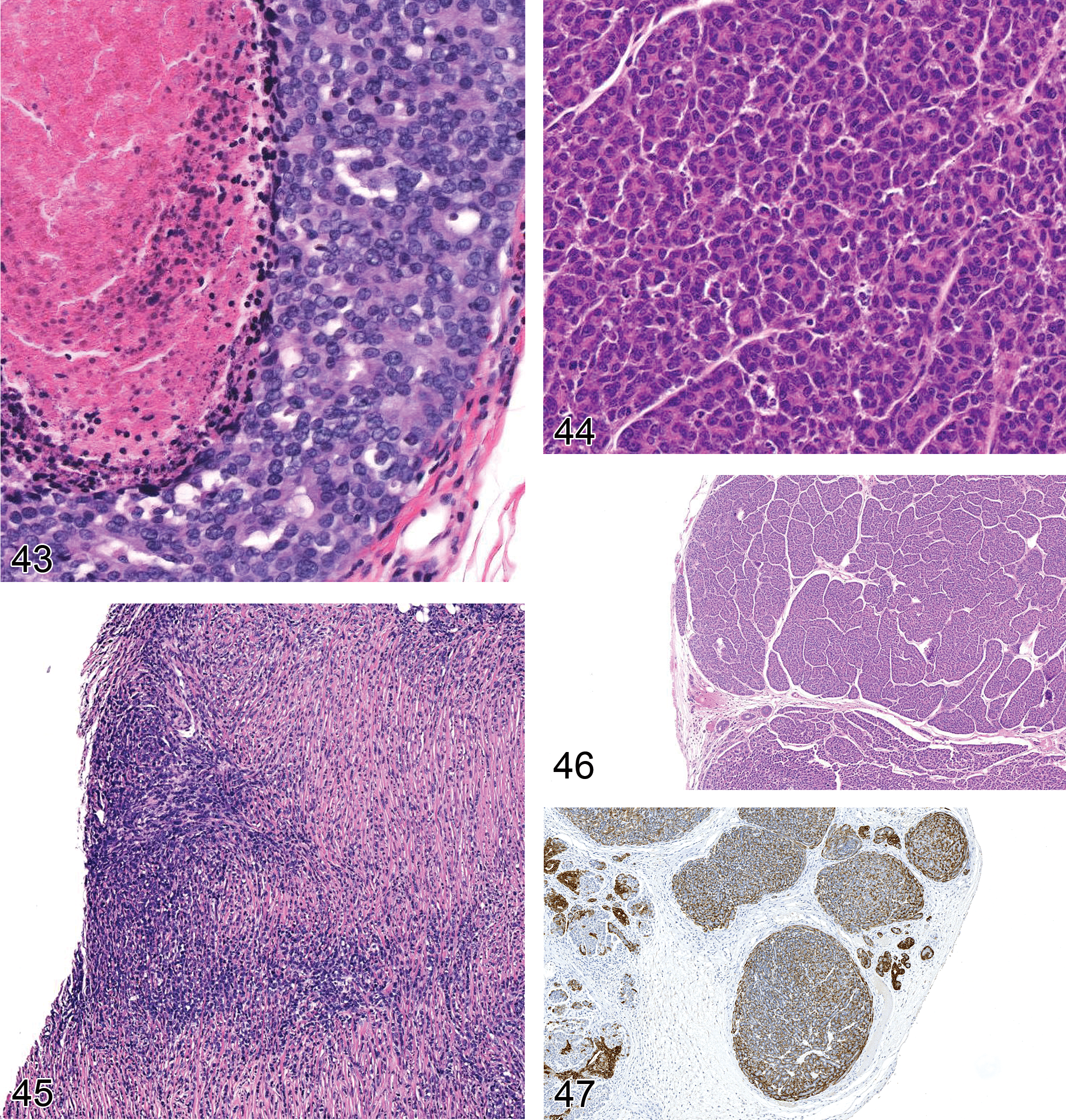
NST carcinoma with comedo pattern (Human).
Footnotes
Acknowledgments
The authors wish to express their appreciation for the excellent advice and assistance of the staffs of the Cellular and Molecular Pathology Branch at National Toxicology Program/National Institute of Environmental Health Sciences and the Fraunhofer Institute of Toxicology and Experimental Medicine in Hannover, the members of the ESTP, STP, JSTP, and BSTP who are participating in the INHAND project, and pathologists and other professional scientists worldwide whose work contributed to this document.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article .
