Abstract
Twenty-eight spontaneously occurring glial tumors (previously diagnosed as astrocytomas, oligodendrogliomas, and gliomas) and eleven granular cell tumors (GCTs) were selected for evaluation using a panel of immunohistochemistry (IHC) stains (Ricinus communis agglutinin type 1 [RCA-1], ionized calcium-binding adapter molecule 1 [Iba-1], OX-6/major immunohistocompatibility complex class II, oligodendrocytes transcription factor 2 [Olig2], glial fibrillary acidic protein [GFAP], S100 beta, glutamine synthetase, neurofilament, proliferating cell nuclear antigen). In addition, nine brain tumors from a 2-year drinking water study for acrylonitrile were obtained from the Acrylonitrile Group, Inc. Based on IHC staining characteristics, Olig2+ oligodendrogliomas were the most commonly diagnosed spontaneous tumor in these animals. Many of the spontaneous tumors previously diagnosed as astrocytomas were RCA-1+, Iba-1+ and negative for GFAP, S100beta, and glutamine synthetase; the diagnosis of malignant microglial tumor is proposed for these neoplasms. Three mixed tumors were identified with Olig2+ (oligodendrocytes) and Iba-1+ (macrophage/microglia) cell populations. The term mixed glioma is not recommended for these tumors, as it is generally used to refer to oligoastrocytomas, which were not observed in this study. GCT were positive for RCA-1 and Iba-1. All acrylonitrile tumors were identified as malignant microglial tumors. These results may indicate that oligodendrogliomas are more common as spontaneous tumors, while acrylonitrile-induced neoplasms are microglial/histiocytic in origin. No astrocytomas (GFAP, S100 beta, and/or glutamine synthetase-positive neoplasms) were observed.
Introduction
Brain tumors are rare in untreated rats; however, the brain tumor incidence in several strains of rats exceeds two to three times the incidence observed in humans and domestic animals (Maekawa and Mistsumori 1990). The highest incidence for spontaneously occurring brain tumors has been reported to be between 79 and 104 weeks of age (2.4% of male and 2.5% of female F344 rats), making this an infrequent disease of the aging rat, and astrocytomas were the most commonly identified glial tumor (Ward and Rice 1982). Experimental induction of central nervous system tumors in rodents is considered a poor model for corresponding human disease, although an increased incidence of brain tumors in chronic rat bioassays may serve as a general indicator of a carcinogenic effect (Rice and Wilbourn 2000; Krinke et al. 2000). The significance of a brain tumor response in the rat has questionable relevance to human brain tumor induction.
Difficulty often accompanies the interpretation of brain tumor induction in the rat. Small increases in the observed incidence of brain tumors in chronic bioassays have created uncertainty and have been an ongoing source of debate for many years (Rice and Wilbourn 2000; Sills et al. 1999; Koestner 1986; Koestner and Norton 1998; Solleveld et al. 1986; Swenberg 1986). Poorly characterized lesions, such as malignant reticulosis, and a lack of good immunohistochemistry (IHC) markers have further complicated matters, as it has not always been clear when to combine tumor classes to determine carcinogenicity in a given study.
Historically, diagnoses for rat brain tumors have been based on histological features due to a lack of ancillary techniques. Astrocytomas in rats have proven to be uniformly negative to staining for glial fibrillary acidic protein (GFAP), an astrocyte marker that has been useful for tumor characterization in man (Solleveld et al. 1986; Solleveld, Gorgacz, and Koestner 1991; Krinke et al. 2000). This observation has led to the supposition that rat tumors, unlike human tumors, demonstrate poor cellular differentiation and astrocytic protein expression. Granular cell tumors (GCTs) have demonstrated positive staining with reduced silver stain, lectin Ricinus communis agglutinin type 1 (RCA-1), and periodic acid-Schiff, presumably due to a high lysosomal content (Krinke et al. 2000). No previous cellular marker had been identified to stain neoplastic cells in oligodendrogliomas (Krinke et al. 2000). Most recently, spontaneously occurring astrocytomas and malignant reticulosis from rats were shown to be positive for RM-4 and (anti-CD68 antibody) ED1 (macrophage/dendritic cell markers), while lacking reactivity to GFAP and S-100 (Nagatani et al. 2009). Astrocytomas from rats treated with acrylonitrile showed variable staining for major immunohistocompatibility complex class II (MHCII) antigen, but the author was unable to further characterize what antigen-presenting cell was the cell of origin (Kolenda-Roberts and Hardisty 2010). The current study was undertaken to use current immunohistochemical antibodies and techniques to further characterize spontaneous and induced brain neoplasms in the rat.
Based on the most recent indications that rat brain tumors, particularly astrocytomas, were monocytic in origin, three available microglial/macrophage antibodies were selected for our IHC panel (Table 1). Iba-1 (ionized calcium-binding adapter molecule 1) is specifically expressed in microglia and macrophages, while RCA-1 is present in macrophages, microglia, and capillary endothelial cells. The OX-6 antibody was also included, which binds to the rat MHCII antigen. Olig2 (oligodendrocytes transcription factor 2) is expressed in oligodendrocytes and has been shown to be expressed in human oligodendrogliomas (strongly) and astrocytomas (weakly). In the central nervous system, the antibodies for GFAP and glutamine synthetase stain astrocytes and some groups of ependymal cells. S100 beta mainly stains astrocytes but is considered less astrocyte-specific than GFAP (Steiner et al. 2007). An antibody for neurofilament was used to rule out any neuronal tumors, and PCNA (proliferating cell nuclear antigen) staining was used to indicate relative cell proliferative activity in the neoplasms.
Cellular targets of the antibodies used in the immunohistochemistry panel.

Summary of typical staining characteristics of spontaneous and induced rat brain tumors.
Acrylonitrile is a widely used chemical in the manufacture of rubber, synthetic fibers, and plastics. Chronic studies in rats have shown a significant increase in glial tumors, typically identified as astrocytomas, based on cellular morphology (Bigner et al. 1986; Johannsen and Levinska 2002a and 2002b; Quast et al. 1980). Astrocytomas from the 2-year acrylonitrile drinking-water study were evaluated with the chosen IHC panel to better characterize the neoplasms and to compare staining properties of chemically induced tumors to spontaneous tumors.
Materials and Methods
Case Selection
Data stored at the National Toxicology Program Archives were reviewed for cases of spontaneously occurring brain tumors in the rat, and the hematoxylin and eosin (H&E)-stained slides were obtained for histological review. H&E slides and paraffin blocks with sufficient tissue were chosen for immunohistochemical staining. The tumors selected included 5 oligodendrogliomas, 14 astrocytomas, 8 gliomas/mixed gliomas, and 1 case of severe gliosis that was considered an oligodendroglioma after review (this animal also had a separate GCT). Eleven GCTs from archival material were also stained. Samples from 12 acrylonitrile-treated rats (previously diagnosed as astrocytomas) were obtained, and 9 contained sufficient tissue for review. Thus, a total of 9 neoplasms from the acrylonitrile study and 39 spontaneous tumors were stained with the panel of immunohistochemical antibodies.
IHC
Five µm sections were placed onto charged slides, deparaffinized, rehydrated, and washed. Slides were placed in 1:20 citrate buffer in a decloaking chamber for 5 min under pressure, depressurized for 10 min, and allowed to cool. Endogenous peroxidase was quenched using a 15-min incubation in distilled water containing 0.3% hydrogen peroxide. Sections were blocked with serum or serum-free protein block (Olig2, glutamine synthetase, S100 beta) and stained for 30 min (mouse antihuman neurofilament primary antibody, 1:400; DAKO, Carpinteria, CA) or 60 min (anti-Olig2 rabbit polyclonal antibody at 1:1,500, Novus Biologicals, Littleton, CO; rabbit antiglutamine synthetase at 1:22,000, abcam, Cambridge, MA; mouse anti-S100 beta at 1:300, abcam; mouse antirat MHCII/OX-6 antibody at 1:150, AbD Serotec, Raleigh, NC; anti-Iba-1 rabbit polyclonal antibody at 1:2,000, Wako Chemicals, Richmond, VA). Secondary antibody was applied, and slides were developed using a Vectastain Elite ABC kit (Vector Laboratories, Burlingame, CA) and (3,3-diaminobenzidine) DAB (Biocare Medical, Concord, CA). Sections were then counterstained with hematoxylin, dehydrated, and coverslipped. For RCA-1 (1:2,000; Vector Laboratories), a decloaking step and secondary antibody were not used. For GFAP (anti-GFAP rabbit polyclonal antibody at 1:1,500; DAKO, antigen retrieval using 0.01% trypsin/Tris-HCl was used rather than a decloaking step, then after the quenching step, Background Buster (Innovex Biosciences, Richmond, CA) was applied for 25 min and a STAT-Q detection kit was used for slide development (Innovex Biosciences). PCNA was stained using a kit (Zymed, S. San Francisco, CA).
Evaluation
Slides were evaluated for relative staining intensity and distribution within the brain tumors (scale 0 to 3+) and the results were recorded. For PCNA staining, a relative percentage of positively stained nuclei were semiquantitatively evaluated by examining several medium-power fields. All stages of the cell cycle were counted, providing an estimate of growth fraction.
Results
Oligodendroglioma
Of the 16 tumors identified by IHC as oligodendrogliomas, previous diagnoses included oligodendroglioma (5), glioma/mixed glioma (6), astrocytoma (4), and gliosis (1). In these animals, the average day of death was 638 days (range of 312–898 days) and 69% of tumors were macroscopically observable at the time of necropsy. The average tumor size, as measured on the H&E slide, was 7 mm (range 3–16 mm), and they were most commonly observed in the cerebrum (88%). Histologically, tumors exhibited sharp demarcation (seen in 15 of the 16 tumors; 94%), moderate to dense cellularity (100%), and good cellular differentiation (69%). One animal also had a GCT not associated with the main mass.
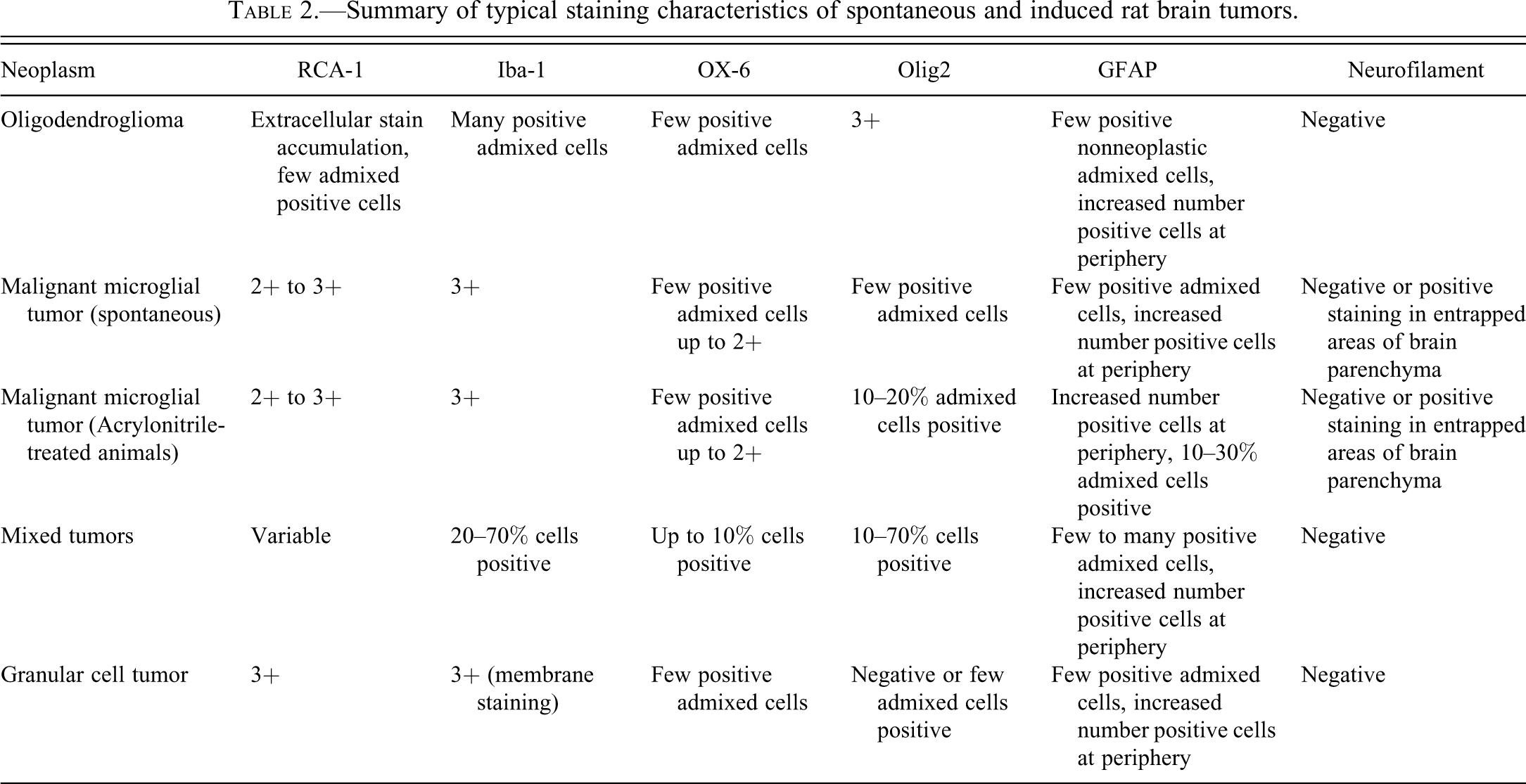
Histological features were consistent with those described in the literature for oligodendrogliomas in the rat (Gopinath 1986; Krinke et al. 2000; Newman and Mawdesley-Thomas 1974; Solleveld, Gorgacz, and Koestner 1991). The size of neoplastic cells was typically uniform, with small, round nuclei. Perinuclear halos/clear cytoplasm with fine fibrovascular stroma imparted the classic honeycomb appearance in some areas (Figures 1–8). Characteristic endothelial hyperplasia and hypertrophy (vascular garlands/glomeruloid vascular proliferations) were common features.
Spontaneous oligodendroglioma.
Anti-oligodendrocytes transcription factor 2 (Olig2) stain; 20×.
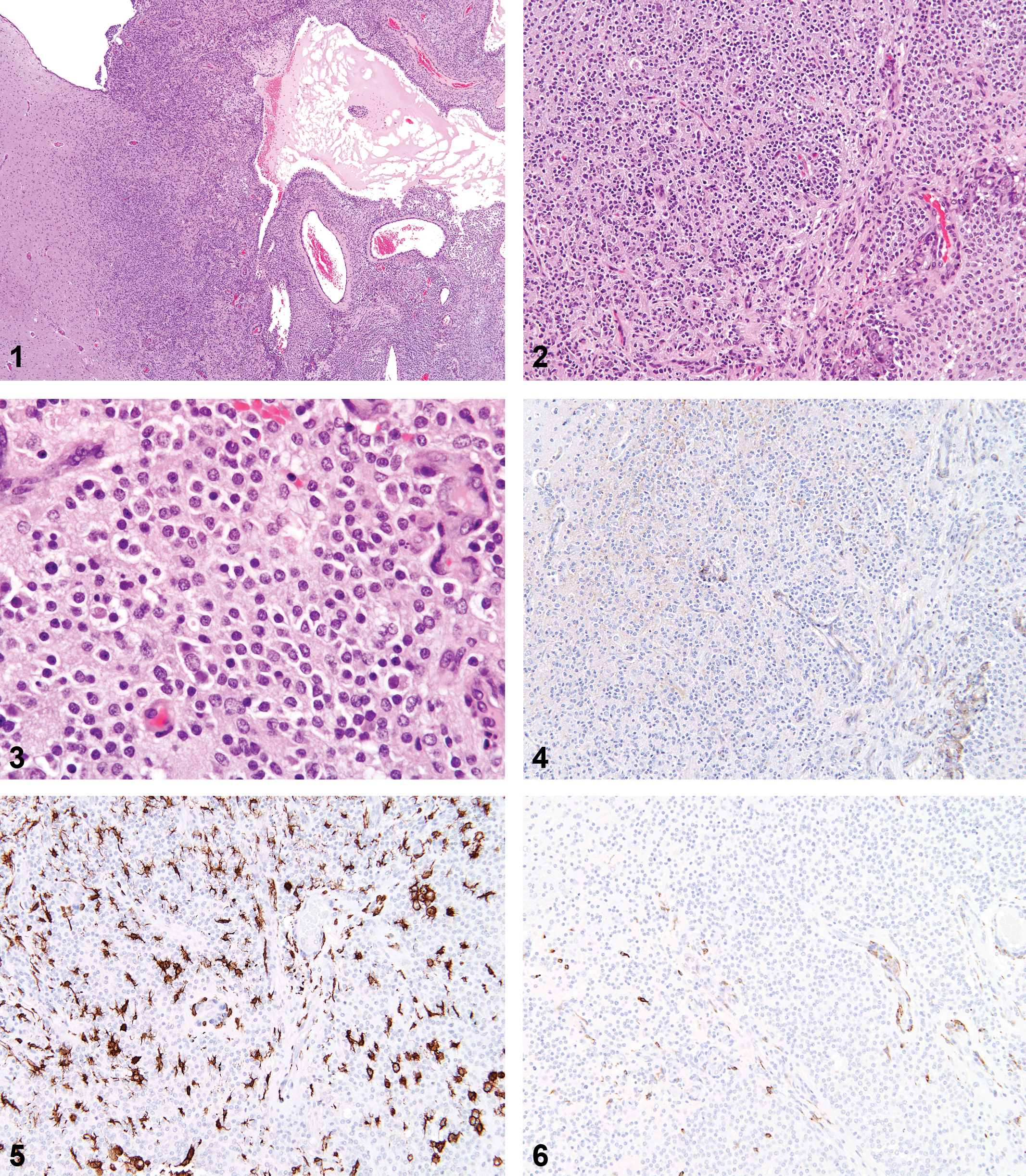
IHC results for these tumors were delineated by strong (3+) nuclear staining of neoplastic cells by the Olig2 marker. RCA-1 staining was observed as extracellular accumulation of stain around neoplastic cells, and few admixed cells were clearly positive. Iba-1 staining was typically observed in 10 to 20% of admixed nonneoplastic cells, while few cells throughout the mass were positive for OX-6 (MHCII). Neurofilament staining was negative. GFAP+/glutamine synthetase+/S100 beta+ astrocytes were observed in increased numbers at the tumor periphery, with few admixed cells or cell clusters present throughout the mass. Five of the 16 tumors (31%) were given a secondary diagnosis of reactive astrocytosis, given the extent of astrocytic cell proliferation at the tumor periphery and the number of positive cell clusters throughout the tumor. PCNA staining tended to be variable in neoplastic oligodendrocytes (10–80% positive), but positive staining within glomeruloid vascular proliferations was consistently very high (>80–90%).
Malignant Microglial Tumor
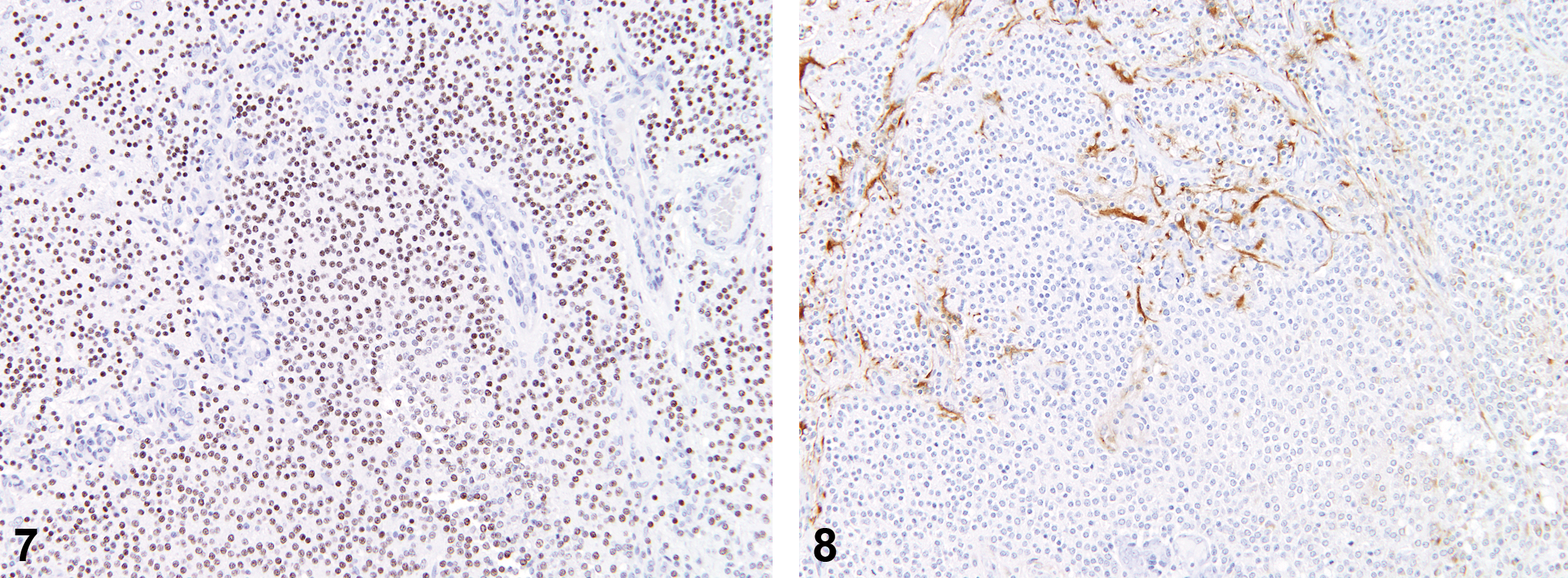
Of the nine spontaneous tumors identified as malignant microglial tumors, all nine were previously diagnosed as astrocytomas. None of these animals showed evidence of other histiocytic neoplasms/sarcomas in other organ systems. The average day of death for these animals was 608 (range 414–730 days), and the average size of the mass on the H&E slide was 6 mm (range 3–11 mm). Only 33% of the tumors were observed at necropsy, and the location was variable (cerebral cortex 67%, thalamus/hypothalamus 56%, pons/medulla 44%). Histologically, the tumors exhibited poor demarcation (seen in 6 of the 9 tumors; 67%), moderate to dense cellularity (89%), and good cellular differentiation (67%).
Histological features were consistent with those that have been described in the literature for astrocytomas in the rat. Neoplastic cells had indistinct borders with round to elongated nuclei (Figures 9–16). Diffuse infiltration of the parenchyma with neuronal satellitosis, invasion of meninges, and cuffing of blood vessels were present in some tumors. In areas of degeneration/necrosis, exfoliated cells showed typical round cell phenotype, with moderate to abundant eosinophilic granular cytoplasm. Immunohistochemical staining of these tumors yielded consistent positive staining (2+ to 3+) for RCA-1 and Iba-1. Staining with OX-6 (MHCII) was variable: in some tumors, cells had strong positive staining (2+), while others had few positive cells. With staining for astrocytes (GFAP, glutamine synthetase, S100 beta), there were positive-staining cells in increased numbers along the tumor periphery, with few or clusters of admixed positively staining cells within the tumor itself. Staining for neurofilament was negative, with the exception of islands of infiltrated parenchyma. Staining for Olig2 generally showed few admixed positively staining nonneoplastic cells. PCNA staining showed high rates of cell proliferation, ranging from 50% to 90% within individual tumors.
Spontaneous microglioma.
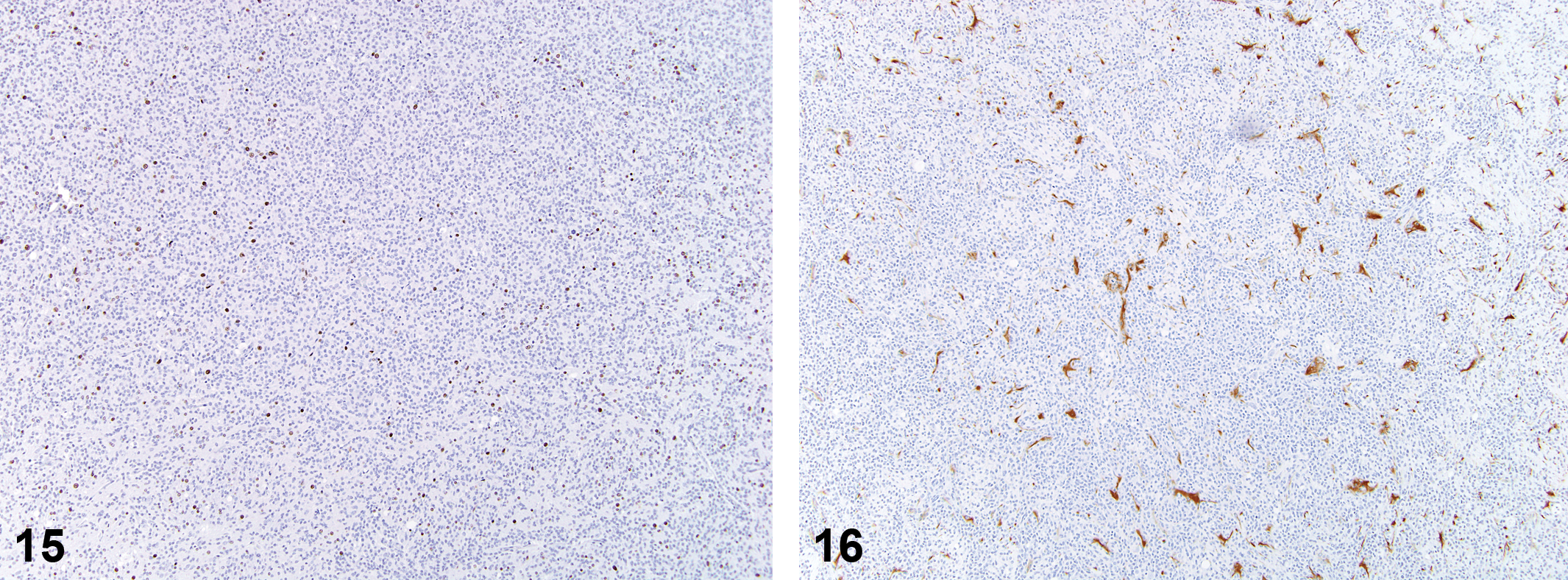
Anti-oligodendrocytes transcription factor 2 (Olig2) stain; 10×.
Mixed Tumors
Of the three tumors identified as mixed tumors, two were previously diagnosed as gliomas and one as an astrocytoma. Average day of death was 502 (range 303–844 days). For the two tumors with available information, one was visible at necropsy. Size on the H&E slide was 3–5 mm. Tumors demonstrated moderate to dense cellularity and good cellular differentiation. Histological features of the tumors showed similarities to both oligodendrogliomas and malignant microglial tumors (Figures 17–24).
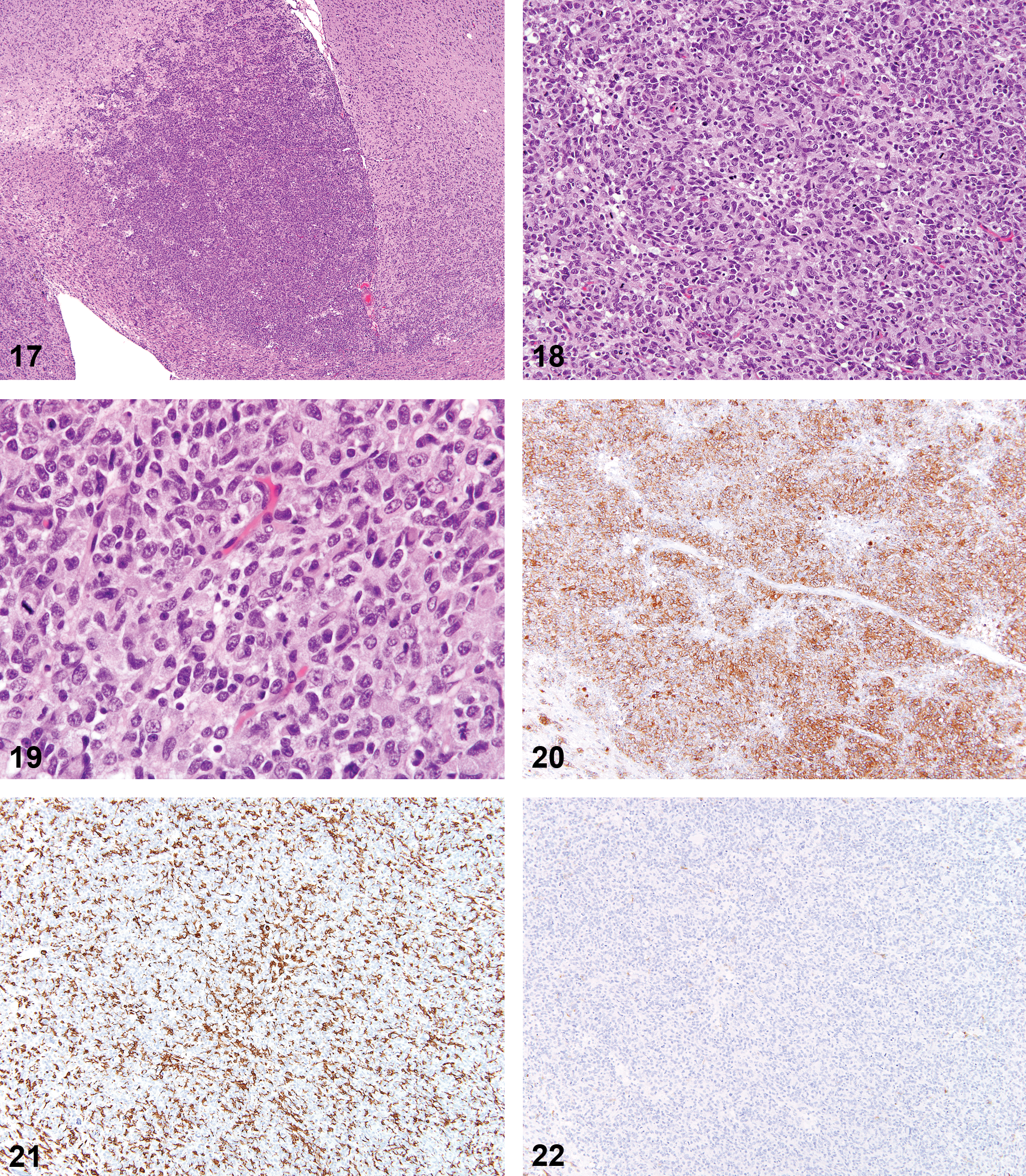
Spontaneous mixed tumor (oligodendroglial and microglial cells).
The diagnosis of mixed tumor is proposed based on the divergence of staining profiles of these tumors when compared to other oligodendrogliomas and malignant microglial tumors. Consistent positive staining with RCA-1 was not present as it was in the malignant microglial tumors. The number of Olig2+ cells was variable (5–15%, 50%, and 50–70% of cells in each of the three tumors), but so was the number of Iba-1+ cells (20–70%, 30–40%, variable with up to 2+ staining, respectively). GFAP+ staining was present along the tumor periphery and within few to clustered admixed cells, but within the third tumor roughly 20% of the cells throughout the mass were positive for GFAP. Staining for neurofilament was negative, and PCNA staining was present in most cells (50%, 60%, and >90%, respectively).
GCTs
The histologic features of these tumors were consistent with those previously described in the literature. These expansile tumors were composed of uniformly large round cells with abundant granular eosinophilic cytoplasm and were located in meningeal areas (Figures 25–28).
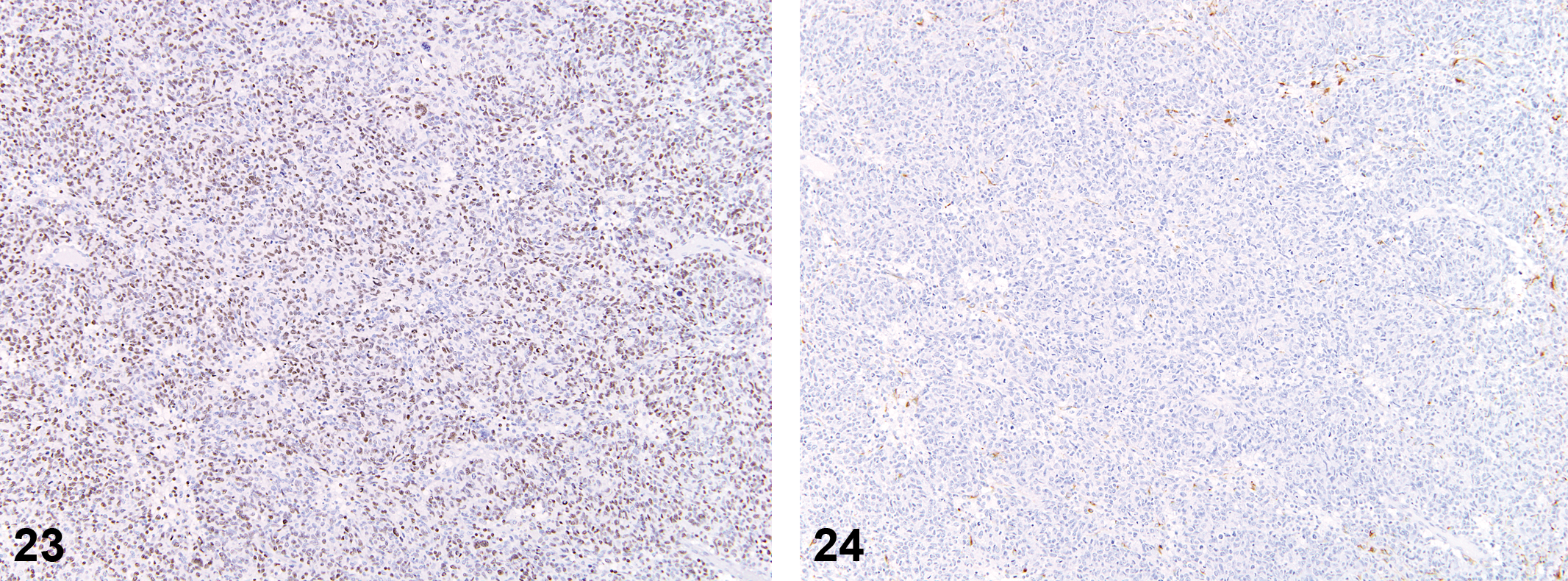
Anti-oligodendrocytes transcription factor 2 (Olig2) stain; 12.5×.
The 11 GCTs showed strong (3+) uniform cytoplasmic staining with IHC for RCA-1 and strong membrane staining for Iba-1. Few admixed cells stained positively for OX-6 (MHCII) or GFAP, and most tumors were uniformly negative for S100 beta. Staining for neurofilament was negative. Almost all neoplastic cells were positive for PCNA (>90% of cells).
CNS Neoplasms from Acrylonitrile-treated Animals
Of the twelve animals from acrylonitrile-treated rats (previously diagnosed as astrocytomas), nine samples contained sufficient tissue for review. All nine were identified as malignant microglial tumors based on immunohistochemical staining. Histologic features were similar to those described for spontaneous malignant microglial tumors (Figures 29–36).
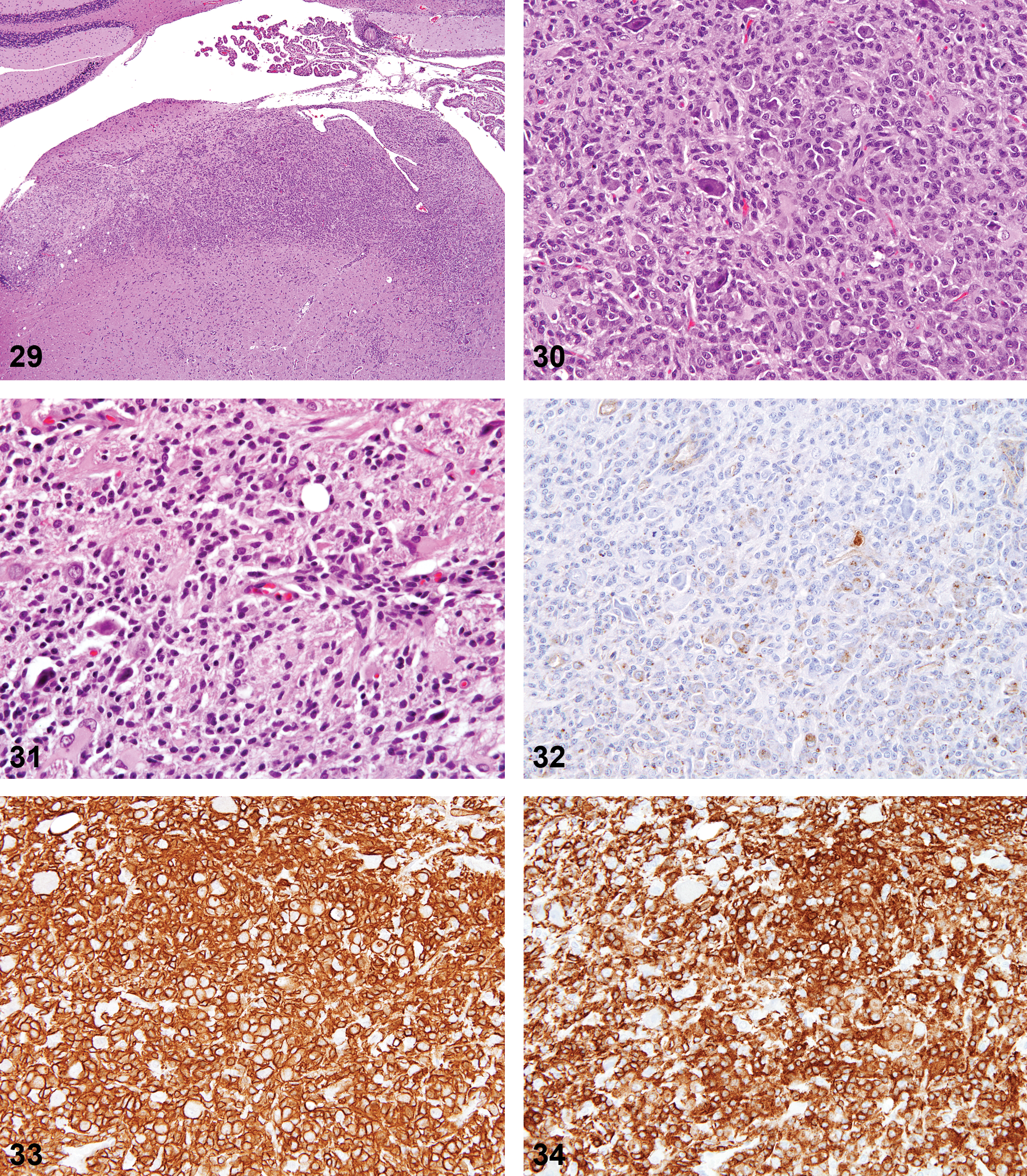
Acrylonitrile-induced microglioma.
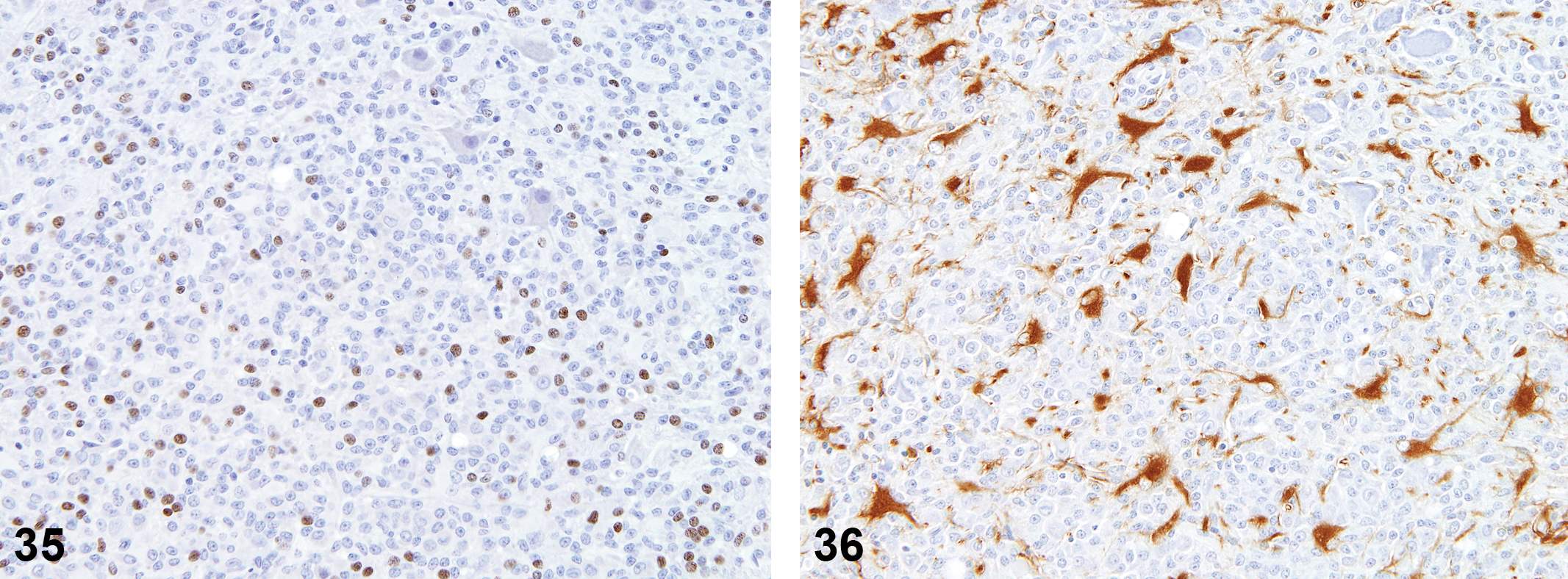
Anti-oligodendrocytes transcription factor 2 (Olig2) stain; 16×.
Staining profiles for these neoplasms were consistent with those of the spontaneous malignant microglial tumors, but there was a mild increase in the subpopulations of GFAP+ and Olig2+ cells throughout the tumors.
Discussion
Attempts to develop a working protocol for oligodendrocyte-specific protein using two different antibodies were unsuccessful, but the nuclear marker Olig2 proved straightforward to use and was valuable to the diagnosis of oligodendrogliomas. While reactive astrocytosis was a feature in some tumors, based on the literature and outcome of this study, GFAP and the other astrocyte markers (glutamine synthetase, S100 beta) are not crucial stains in the ultimate determination of the cell of origin for rat brain tumors. For the diagnosis of malignant microglial tumors, mixed tumors and GCTs, RCA-1, and Iba-1 (rather than OX-6/MHCII) proved to work well and provide significant staining in neoplastic cells.
Of the non-GCT spontaneous glial tumors, oligodendrogliomas (16 of the 28; 57%) were the most commonly observed tumor and were easily demonstrated by IHC using the Olig2 marker. Nine tumors (32%) previously diagnosed as astrocytomas stained positively for Iba-1 and RCA-1, indicating a monocytic cell of origin (macrophage/microglia). Given the absence of any histiocytic sarcomas in other organs in these animals, the diagnosis of malignant microglial tumor is proposed for these neoplasms, both to identify the relevant cell type and to reduce confusion with a small glial tumor (microglioma). GFAP+ astrocytomas and neuronal tumors were not observed in this study.
The staining properties of the mixed tumors were unexpected, as mixed brain tumors are presumed to contain subpopulations of astrocytic and oligodendroglial cells, rather than our observed mix of oligodendroglial and microglial cells. The diagnostic term mixed glioma is generally used in the current literature to refer to oligoastrocytomas, which would preclude its use for mixed tumors in the rat brain. Based on the current study, it cannot be determined whether these results indicate an oligodendroglioma with a secondary inflammatory or other cellular response or if these neoplasms truly represent a mixed tumor response.
The GCTs stained most intensely for lectin RCA-1 and Iba-1. Based on the current findings, GCTs are a separate histiocytic neoplasm that derive from a different subset of macrophage, presumably associated with the meninges.
All of the acrylonitrile-treated rats with sufficient tumor samples were identified as malignant microglial tumors (9 of the 9). The staining properties (RCA-1+, Iba-1, OX-6) were similar to those observed in the spontaneous malignant microglial tumors, but there was a mild increase in subpopulations of Olig2+ and GFAP+ cells throughout the tumors.
The exact cell of origin for the malignant microglial tumors is not clear from the current study; however, these neoplasms appear to represent a unique primary neoplastic site for a cell of monocytic/macrophage origin. The available IHC markers for rat histiocytic cells currently limit our ability to further characterize the subsets affected in these tumors. No histiocytic tumors at other sites in the body were found for any of the affected animals.
While currently there are no immunohistochemical markers that can distinguish between microglia and blood monocytes, microglia have been characterized by cellular activity and cell surface morphology as a distinct population of cells (Giulian et al. 1995). Resting microglia typically lack MHCII expression (Sedgwick et al. 1993), so the variability in OX-6 staining and lack of other tumor sites in the body of affected rats supports this diagnosis.
As small increases in the incidence of gliomas in chronic rat bioassays have been difficult to interpret, the use of IHC and potential redesignation of these tumors as malignant microglial tumors may further call into question the relevance of these tumors to human disease. The most common human brain tumors are astrocytomas (adults) and medulloblastomas (children), and the experimental induction of malignant microglial tumors in rats may represent a species-specific manifestation.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article has been funded in whole or in part with Federal funds from the National Institute of Environmental Health Sciences, National Institutes of Health, Department of Health and Human Services, under Contract No. N01-ES-00005.
