Abstract
Epidemiological studies increasingly indicate that polychlorinated biphenyls (PCBs) contribute to the risk of non-Hodgkin’s lymphoma (NHL). In rodent bioassays, PCBs have long been demonstrated to be liver carcinogens, and excess tumors in the thyroid, lung, and other organs have been observed in more recent studies. Leukemias and lymphomas now classified as NHL were observed in one bioassay in which a concurrent infection was also reported. Clinical and epidemiological studies show immunosuppression and inflammation are strong risk factors for NHL, and both epidemiology and toxicology studies show that PCBs are immunosuppressive and cause inflammation. We reviewed published carcinogenesis bioassays conducted using commercial PCB products, individual congeners, and congener mixtures, with a focus on bioassay protocols and immune-related observations. Based on a mode-of-action framework for PCBs, we suggest that an immune challenge in conjunction with PCB exposure may be necessary for the observation of NHL. We conclude that the lack of concordance between human epidemiology and animal bioassays with respect to NHL may simply be the result of the bioassay methodology used, and not a difference in underlying biology. The lack of concordance should not be construed as evidence that PCBs do not contribute to the risk of NHL.
Keywords
Background
Concordance of tumor types between animals and humans has long been a concern among those with an interest in regulatory toxicology and the carcinogenic classification of chemical substances. Although the current United States Environmental Protection Agency cancer guidelines (US EPA 2005) do not require concordance of tumor sites between humans and animals to conclude that a chemical is a human carcinogen, they do require an explicit consideration of the mode of action (MOA). Both an understanding of the role of a chemical in causing a particular tumor or type of cancer and concordance of tumor types between animals and humans add to the weight of evidence of human carcinogenicity, which is demonstrated in the weight of evidence discussion that was part of the US EPA documentation for classifying trichloroethylene as a known human carcinogen (US EPA 2011). The International Agency for Research on Cancer (IARC) also considers mechanistic evidence in its evaluations of carcinogenicity (Baan et al. 2009).
Commercial mixtures of polychlorinated biphenyls (PCBs), several individual PCB congeners, and defined PCB congener mixtures cause tumors in rats and mice. Liver and bile duct tumors have been routinely observed in bioassays with multiple strains of rats and mice. Other cancers, such as non-Hodgkin’s lymphoma (NHL) and gastrointestinal and thyroid tumors, have been reported in selected studies of commercial products (Mayes et al. 1998; NCI 1978; NTP 2011; Ward 1985).
An increasing number of occupational cohort follow-up studies and population-based case control studies have shown that exposure to PCBs results in statistically significant excesses of liver and/or bile duct tumors (NTP 2011). There is also a substantial and growing body of epidemiological literature showing that exposure to PCBs increases the risk of NHL (Kramer et al., unpublished data; NTP 2011). Most of the population-based nested case-control studies show a positive relationship between exposure to PCBs and risk of NHL incidence, although a few studies do not show this relationship (NTP 2009). Several case-control studies examined both blood levels of PCBs and the presence of Epstein-Barr virus (EBV), measured as titers of antibody against the early antigen of EBV. These studies provide evidence of a significant, positive interaction between blood levels of PCBs and titers of antibodies to EBV early antigen and risk of NHL (Hardell et al. 2009; Nordstrom et al. 2000; Rothman et al. 1997).
The National Toxicology Program (NTP) classifies PCBs as reasonably anticipated to be human carcinogens (NTP 2011). The US EPA IRIS online database classifies the carcinogenicity of PCBs as Group 2B, probable human carcinogens, in an evaluation dated June 1997. In its recent evaluation, IARC classifies PCB 126, the co-planar, dioxin-like (DL) congener with the highest toxic equivalency to TCDD, as a Group 1, known human carcinogen (Baan et al. 2009). This classification is based on the complete carcinogenicity of PCB 126 in animal bioassays and the extensive evidence that PCB 126 and TCDD act via the same aryl hydrocarbon receptor (AhR)–mediated mechanism.
An understanding of the MOA can provide a useful lens through which to examine the apparent lack of concordance between the epidemiological data showing that PCBs increase NHL risk and rodent bioassays in which NHL has rarely been reported following PCB exposure. Epidemiological and clinical data indicate that immune dysregulation, particularly immunosuppression, is a very strong risk factor for NHL; congenital or acquired immunodeficiency increases NHL incidence rates fifty times or more above that of the general population (Grulich et al. 2007). There is high concordance of human and animal data showing that PCBs are immunosuppressive based on organ effects such as thymic atrophy and bone marrow hypocellularity, in addition to functional assays that show adverse impacts at doses lower than those required to induce observable loss of thymic tissue (Davis and Safe 1988). Polychlorinated biphenyls also dysregulate other aspects of the immune system, including inflammatory responses (Kerkvliet 2009).
In this paper, we examine the apparent lack of concordance between the human epidemiological literature with its increasing weight of evidence linking NHL and PCB exposure, and the animal bioassay literature in which NHL has rarely been reported in excess. We review data from published rodent bioassays of commercial PCB mixtures and individual congeners to determine whether there were methodological reasons that NHL has not been routinely observed in animal bioassays. Our analysis relies on identification of an MOA supported by disease-specific epidemiologic and clinical literature. The specific hypotheses examined in this paper are: (1) whether the bioassay methodology—such as animal model, doses, histopathology, and immune system challenges—would have led to the observation of NHL; and (2) whether there is previously overlooked evidence of NHL in these bioassays.
Results
Determining an MOA Using Risk Factors Identified in the NHL Clinical Literature
Non-Hodgkin’s Lymphoma is the generic term for over thirty histologically distinct tumors arising from lymphocytes. Blood cell cancer classifications have changed over time, and NHLs now include cancers that had been classified as leukemias in previous systems based on the recognition that lymphoma and leukemias, such as B-cell chronic lymphocytic leukemia and B-cell small lymphocytic lymphoma, are two manifestations of the same disease (Hartge et al. 2006; WHO 2008). Most NHLs originate in B-cells, although they also originate in T-cells and, more rarely, natural killer (NK) cells, indicating that the early steps in the neoplastic transformation can occur early in the differentiation of immune cells in the bone marrow or later in the development and maintenance of immune cell populations in the thymus and elsewhere.
Many clinical and epidemiological studies have established immunosuppression as a strong risk factor for NHL. There is also evidence that inflammation is a risk factor for NHL. The immunosuppression can be caused by many factors, including infectious agents, chemotherapy agents, genetic disorders, and various diseases and/or their treatment (Ansell and Armitage 2005; Grulich et al. 2007). Chronic infection with viruses such as human immunodeficiency virus (HIV), EBV, and human T lymphotropic virus 1 (HTLV-1) has been identified as causative or as a major contributing factor for specific lymphomas (Grulich et al. 2007). Human immunodeficiency virus enhances the risk of several B-cell lymphomas, and the risk increases with increasing immunosuppression by the virus (Grulich et al. 2007). Epstein-Barr virus is specifically associated with Burkitts lymphoma, one type of NHL. Epstein-Barr virus also has been identified in other types of NHL, although substantial immunosuppression appears to be necessary prior to the cancer’s appearance, and it is not clear if the virus plays an etiological role (Grulich et al. 2007; Hartge et al. 2006). Chronic infection with the bacterium Helicobacter pylori causes lymphomas of the mucosa-associated lymphoid tissue (MALT), which account for 8% of all B-cell lymphomas (Owens and Smith 2011). There is also evidence of NHL associated with hepatitis C and Chlamydia psittaci infections (Ansell and Armitage 2005). Other strong, well-established risk factors for NHL that result in immunosuppression are inherited immunodeficiencies and organ transplantation.
Autoimmune diseases such rheumatoid arthritis, systemic lupus erythematosus, celiac disease, and Sjogen’s syndrome are established risk factors for several types of NHL, with risk increasing with severity of disease. These diseases are associated with inflammation and are treated with immunosuppressive drugs (Hartge et al. 2006; Wang and Nieters 2010). Several studies suggest that increased risk of NHL is associated with the inflammatory disease, and not the immune-suppressive therapy used to treat it, including the observation that non-inflammatory diseases treated with high-dose steroids are not associated with increased risk of NHL (Grulich et al. 2007). In addition, studies examining genetic variations in immune-related genes and the risk of NHL have identified polymorphisms that increase the activity of two pro-inflammatory cytokines, TNF-α and lymphotoxin-α, as risk factors for several subtypes of NHL (Wang et al. 2006; Wang et al. 2009).
Comparison of Immune Effects of PCBs and TCDD in Humans and in Rodent Models
The immunosuppressive effects of PCBs have been observed in humans, wildlife, and laboratory animals (Selgrade 2007; Tryphonas 1994; Tryphonas 1995). Studies examining the impacts of PCBs in detail indicate that PCBs dysregulate both the adaptive and innate immune systems, with many observations of analogous impacts in many species, including humans. Laboratory studies have shown that both dioxin-like (DL) and non-dioxin–like (non-DL) PCB congeners have immunotoxic effects that likely act via different mechanisms. The congener studies have primarily focused on DL-PCBs, those that bind to the AhR receptor, consistent with well-known immunotoxic effects of 2,3,7,8-tetrachlorodibenzo(p)dioxin (TCDD; Kerkvliet 2009). A few recent studies have also examined the effects of non-DL PCBs (NTP 2006a). Polychlorinated biphenyls combinations intended to mimic congener mixtures commonly found in human tissues have also been tested (Davis and Safe 1989).
In humans, evidence of greater susceptibility to infectious disease following PCB exposure is well documented. An increase in infections of the skin and respiratory tract were observed for many years after accidental exposures to PCB-contaminated rice bran oils in Japan in 1968 (Nakanishi et al. 1985) and in Taiwan in 1979 (Lu and Wu 1985). Further observations of immunosuppressive effects included decreased serum concentrations of IgA and IgM for approximately two years after exposure and alterations of T4/T8 ratios, although the alteration was in opposite directions between the two cohorts, Yusho in Japan and Yu-Cheng in Taiwan (Lu and Wu 1985; Nakanishi et al. 1985). In the Yu-Cheng cohort, there was a low response to tuberculin tests the first year after exposure, with a recovery to the control response rate three years later, which is suggestive of temporary suppression of cellular immunity (Lu and Wu 1985). Although some of these effects may have been caused by chlorinated furans or other substances formed during the oil heating process, subsequent studies in children of occupationally exposed mothers and populations with high environmental exposure to PCBs also show immunosuppressive effects, particularly in children with prenatal and breast milk exposures. Children born to mothers who were occupationally exposed to PCBs (Kanechlor 300 and 500) in a capacitor plant in Japan were reported to experience more colds and have higher number of carious teeth (Hara 1985), a disease caused by acid-producing bacteria in the mouth (Chen and Wang 2010). Populations with high PCB exposure through the marine food chain have been studied in the Faroe Islands (Heilmann et al. 2010) and Nunavik, Québec (Dallaire et al. 2004; Dallaire et al. 2006). Children, aged five and to a lesser extent aged seven, in the Faroe Islands showed an inverse association between antibodies to tetanus and diphtheria toxins and PCB concentrations in their mother’s milk at birth and their serum concentrations at eighteen months of age. Both toxins elicit T-cell–dependent B-cell responses (Heilmann et al. 2010). Three cohorts of Inuit children residing in the Nunavik area of Québec showed a correlation between prenatal (and possibly postnatal) PCB exposure and incidence of lower respiratory tract infections. The increase in infection persisted at least until the age of five. An increased rate of otitis media for the first six months of life was also observed in the more highly PCB-exposed children in Nunavik (Dallaire et al. 2004; Dallaire et al. 2006) and in Dutch preschool children with higher current serum concentrations of PCBs (Weisglas-Kuperus et al. 2000).
The Dutch study, which was based on mothers and children in the general population, also reported that higher perinatal exposure to PCBs resulted in increased prevalence of chicken pox, altered T-lymphocyte numbers, and reduced rates of allergy and shortness of breath (Weisglas-Kuperus et al. 2000). Studies of the residents of eastern Slovakia, an area with high environmental PCB contamination, show an inverse association between PCB exposure and thymus size measured at postnatal day 4 (Park et al. 2008).
Both structural and functional immunotoxic effects have been observed in nonhuman primates orally dosed with commercial mixtures of PCBs (Tryphonas 1994; Tryphonas 1995). Nonhuman primates appear to be more sensitive than rodent models to the immunotoxic effects of PCBs (Tryphonas 1995). Exposure of rhesus monkeys to Arochlor 1248 resulted in reduced thymus size, reduced lymph nodules, inapparent germinal centers, and hypocellular bone marrow. After one year, these animals had decreased levels of antibody titers to sheep red blood cells (SRBC), a T-cell–dependent B-cell response, although the anti-tetanus toxoid titers were not affected. Oral exposure of female cynomolgus and rhesus monkeys to Arochlor 1254 for one or two years, respectively, resulted in a dose-related reduction of anti-SRBC (IgM) titers. The reduction in thymus size and the reduction in specific antibody titers (T-cell–dependent B-cell responses) are analogous to the observations in human populations, although the specificity of the antibodies differed.
Loose et al. (1978) conducted one of the early rodent studies of immune system effects of PCBs using male BALB/c mice administered Aroclor 1242 for six weeks. Observations included reduced antibody formation, increased endotoxin sensitivity, and a decreased resistance to immune challenge, all consistent with immunosuppression. Davis and Safe (1990) demonstrated suppression of the antibody response in seven- to eight-week-old male C57BL/6 N mice administered selected coplanar and non-coplanar PCBs. These and other experimental results in the mouse model reviewed by Selgrade (2007) are quite analogous to the reductions in antibody formation and increased frequency in infection observed in human populations.
Davis and Safe (1989) compared the relative immunosuppression of six commercial mixtures of PCBs based on the murine response to sheep red blood cell antigen. They also studied a PCB mixture reconstituted to resemble that commonly observed in human milk. Aroclor 1260 and Aroclor 1254 significantly reduced the splenic plaque-forming response at the lowest dose tested (50 mg/kg), whereas the other Aroclors did not. The relative immunotoxicity for the Aroclors in this assay was: 1260, 1254>1248>1242, 1016>1032, a consistent pattern of higher chlorinated mixtures producing more immunotoxicity. It is interesting that this pattern is similar to, but not identical to, the pattern of liver tumorigenicity reported by Brunner et al. (1997) and Mayes et al. (1998), where the potency pattern was consistent with the TCDD toxic equivalents.
Studies in rodents and nonhuman primates have shown that early life exposures to PCBs can result in persistent immunosuppression. For example, early studies showed that in utero exposure to a commercial PCB mixture (Kanechlor 500) resulted in reduction of T-helper cell activity to 20% of control levels when the mice were four weeks old (Takagi et al. 1987). More recently, using C57BL/6 mice, Arena et al. (2003) demonstrated decreases in T- and B-cells in offspring following in utero and lactational exposure to Aroclor 1242 and Aroclor 1254. Infant rhesus monkeys dosed with Aroclor 1248 for one month exhibited thymic hypoplasia and reduced hematocrit (Abrahamson and Allen 1973). Again, the results of the early life exposure studies in rodents and rhesus monkeys are analogous to the observations in several human cohorts such as the thymic hypotrophy observed in eastern Slovakia and the persistence of T-cell–dependent B-cell effects in children in the Faroe Islands. These results are consistent with the increasing understanding of the sensitivity of the developing immune system to chemical toxicants compared to the adult immune system, and the role that the AhR plays in its development (Hogaboam et al. 2008).
Polychlorinated biphenyl–associated inflammation has been observed in many animal studies, including the chronic rodent feeding studies conducted on the carcinogenicity of PCB commercial mixtures (Kimbrough et al. 1972) and both DL and non-DL PCB congeners (NTP 2006a; NTP 2006b; NTP 2006c; NTP 2006d; NTP 2010). Inflammation of DL-PCBs mediated via the innate immune system has been demonstrated in mammalian and in vitro studies examining macrophages, neutrophils, natural killer cells, and dendritic cells. These cells have pattern recognition receptors (toll-like receptors) that recognize microbial pathogens and initiate many signaling pathways, including ones that lead to the production of pro-inflammatory cytokines. A large number of genes leading to the inflammatory response contain multiple dioxin-response elements and presumably respond to the activation of AhR (Kerkvliet 2009). Non-DL PCBs are also immunotoxic. For example, chronic inflammation and bone marrow hyperplasia was observed in the NTP (2006a) bioassay of PCB 153. In summary, there is strong animal evidence that PCBs disrupt and dysregulate many parts of the immune system leading to both immunosuppression and inflammation.
Bioassay Methodologies
We reviewed the published reports of all the chronic bioassays conducted with PCBs and individual congeners to understand the methodology of the bioassays and to determine whether cancers now classified as NHL were observed or would have been detected in the bioassay. Table 1 summarizes this review.
Summary of possible immunological effects observed in PCB bioassays.
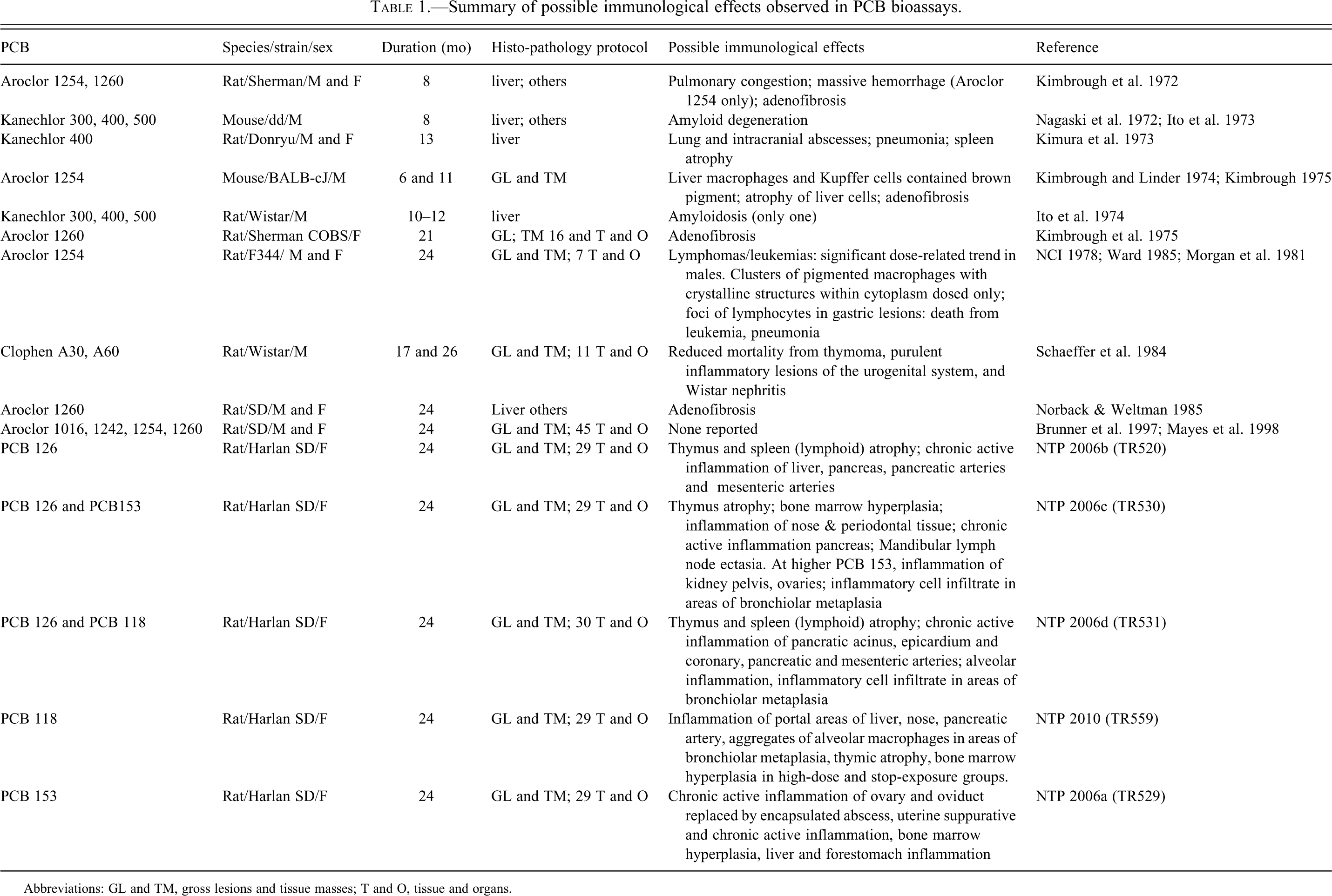
Abbreviations: GL and TM, gross lesions and tissue masses; T and O, tissue and organs.
Necropsies of experimental animals were conducted in all bioassays identified in Table 1, and prominent surface lesions were observed. The liver had grossly observable pathology: the studies reported tumors and neoplastic nodules and described the histopathology of the liver. However, lesions in inaccessible locations, such as some lymph nodes, could have been missed. The earlier bioassays, including the two conducted with mice, examined histology only in the liver and on grossly observed lesions in other tissues. Lesions that were not on the surface, even macroscopically observable lesions, would have been missed, such as the additional lesions that Kimbrough et al. (1975) observed on sectioning the liver.
The NCI (1978) bioassay and subsequent histopathological analyses of these data (Morgan et al. 1981; Ward 1985) demonstrate the importance of looking at the histopathology in addition to macroscopically observable (gross) lesions to identify neoplastic lesions. The initial study (NCI 1978) reported eighteen cases of gross lesions in the stomach, and only those lesions were examined histologically. Approximately six years after the NCI (1978) study was completed, Morgan et al. (1981) conducted histological examinations of the formalin-preserved stomachs of all of the animals and reviewed the slides prepared in the original study. In the re-analysis, additional lesions were identified, the incidence of gastric lesions was found to increase with increasing dose of Aroclor 1254, and multiple lesions were observed only in Aroclor-fed animals, not in the controls. The lesions included both metaplasia and adenocarcinoma. The additional analysis allowed the authors to conclude that Aroclor 1254 “certainly leads to the induction of intestinal metaplasia and probably leads to induction of adenocarcinoma in the glandular stomachs of F344 rats.” Ward (1985) reexamined the liver histopathology and observed that the numbers and types of hepatic lesions were significantly related to the administration of Aroclor 1254, including large, granular lymphocyte leukemia (now classified as NHL) in male rats. Ward (1985) also reported metaplasia and gastric adenocarcinoma associated with PCB exposure, and that the morphology of the metaplasia was quite different in exposed and unexposed rats.
The General Electric Company (GE) sponsored bioassays of Aroclors 1016, 1242, 1254, and 1260 (Brunner et al. 1997; Mayes et al. 1998) using male and female Sprague-Dawley rats. Dose-dependent increases in liver and bile duct tumors were observed for all Aroclors, although they differed in their carcinogenic potencies with the order Aroclor 1254>1260, 1242>1016. This order is consistent with TCDD toxic equivalents of the commercial mixture. Thyroid tumors were observed in male, but not female, rats (Brunner et al. 1997; Mayes et al. 1998). A comprehensive histopathological evaluation was incorporated into its study design, suggesting that if lymphomas were present in these animals, they would have been observed in this bioassay. Similarly comprehensive histopathological evaluations were part of the study design for the individual and binary combinations of congeners tested by the NTP (NTP 2006a; NTP 2006b; NTP 2006c; NTP 2006d; NTP 2010). In addition to liver tumors, tumors of the lung, oral mucosa, and uterus, and possibly the adrenal cortex and pancreas were observed; excess lymphomas were not observed. All of the NTP bioassays were conducted only in female Sprague-Dawley rats. Exposure of animals to PCBs in the NCI, NTP, and GE bioassays began in juveniles and continued (except in the stop-exposure groups) for up to two years. The GE studies used animals certified to be free of many pathogens (Mayes et al. 1998), and the NTP protocols included procedures to enhance the use of pathogen-free animals at the start of the bioassay.
Observations That May Indicate NHL
Lymphomas/leukemias of the type now classified as NHL were observed in the bioassay conducted by the U.S. National Cancer Institute (NCI 1978), in which groups of twenty-four F344 rats were dosed with 0, 25, 50, and 100 ppm Aroclor 1254 in their feed for two years. A statistically significant trend (p = .009) for dose-related increase in leukemia/lymphoma in male rats (0 ppm 3/24; 25 ppm 2/24; 50 ppm 5/25; 100 ppm, 9/24) is reported. The first leukemias/lymphomas in the dosed groups appeared earlier than the control. However, pairwise comparison of the leukemias/lymphomas of the dosed groups with control animals was not statistically significant because of the high rate of leukemia/lymphoma in controls and the small number of animals per group. Ward (1985) notes that the leukemias/lymphomas observed in this bioassay were identified as large granular lymphocyte leukemia, considered an NHL in the current WHO classification system (WHO 2008). All leukemias/lymphomas were observed in the liver except one in the lymph node at the highest dose (NCI 1978), which raises the question of whether additional leukemias/lymphomas would have been observed if comprehensive microscopic evaluations had been conducted in other tissues.
The bioassay conditions of the NCI (1978) study were similar to those used in the GE bioassay (Brunner et al. 1997; Mayes et al. 1998) with respect to dietary levels of PCBs (50, 100 ppm), duration of the study (twenty-four months), and age of animals at the start (six to eight weeks), although the rat strains were different (Sprague-Dawley versus Fisher 344). Whereas a significant dose-related trend in leukemias/lymphomas was observed in the NCI study, no such trend was observed in the GE bioassay. It is possible that Sprague-Dawley rats are less susceptible to some immunotoxic effects such as NHL. For example, in contrast to many other studies, there was no reduction of thymus weight in rats of either sex dosed with any of the Aroclors, including at early (twenty-six-week) interim termination times in the GE study (Brunner et al. 1997; Mayes et al. 1998). Another possible explanation is that there was a lower number of leukemias/lymphomas in the control group in the NCI (1978) bioassay than in historical controls (J. Ward, personal communication), and the statistically significant dose-related trend was an anomaly, although concurrent controls are usually used as the basis for comparison. Another possible, and we argue the most likely, explanation is that an immune challenge, such as infection, is necessary for the observation of NHL in PCB-treated animals. A respiratory infection went through the colony during the NCI (1978) study, causing illness but no deaths, whereas the animals in the GE study were certified free of many pathogens. The explanation for joint action of an immunosuppressive effect and an immune challenge to cause NHL is consistent with the observations in epidemiology studies of significant, positive interaction between blood levels of PCBs and titers of antibodies to EBV early antigen (Hardell et al. 2009; Nordstrom et al. 2000; Rothman et al. 1997).
Protocols that measure direct effects on the immune system were not included in any of the NCI and NTP bioassays conducted on PCB mixtures. Measures of immune function (e.g., T-cell counts, immunoglobulin levels, specific autoantibodies, and cytokine levels) are therefore not available, requiring that the interpretation of results be dependent on pathology, general clinical chemistry, and observation. In addition, the lack of systematic immune challenge precludes conditions desirable for the development and observation of immune-related effects.
Observations suggestive of immunosuppression were reported in many of the animal bioassays with commercial PCB mixtures and all of the congener studies. For example, Kimura and Baba (1973) suggested that rats exposed to Kanechlor 400 in their diet for over a year had reduced resistance to infection. Schaeffer et al. (1984) observed that Wistar rats fed Clophens in their diet for approximately two years experienced fewer deaths from thymomas, Wistar nephritis, and purulent inflammatory lesions of the urogenital system. All of these effects are indicative of a suppressed immune system. Although no lymphomas were reported in these studies, it is not clear that they would have been observed because of the limitations of the experimental protocol as previously described. In the NCI study, the contributing causes of death in the high-dose males were leukemia (5) and pneumonia (2) in addition to liver, intestinal, and brain lesions or tumors. For intermediate-dose males, most of the early deaths were caused by leukemia (four) and pneumonia (six; Ward 1985). Although these deaths were attributed to natural causes, the immunosuppressive effects of PCBs could have played a role.
The animal bioassays also indicate that PCB exposure causes an inflammatory response. The comprehensive histological evaluation of the stomachs of rats exposed to Aroclor 1254 in the NCI (1978) study led to the observation that one-half of the gastric lesions were adjacent to aggregates of lymphocytes, which Morgan et al. (1981) suggested may stem from an immune response to inflammation of the gastric mucosa or to altered cellular antigens. This finding is consistent with other studies of rats, such as Kimbrough et al. (1972), in which administration of Arochlor 1254 led to inflammation and ulceration of the glandular stomach. These observations are also consistent with the frequent observation of adenofibrosis (Kimbrough et al. 1972; Kimbrough et al. 1975; Kimbrough and Linder 1974; Norback and Weltman 1985). Adenofibrosis, as seen under a light microscope, is described as glandular structures lined by mucin-secreting columnar or cuboidal cells and surrounded by layers of connective tissue (Kimbrough 1975; Norback and Weltman 1985). Electron microscope observations identify adenofibrosis as intestinal metaplasia with goblet cells, enterochromaffin cells, and Paneth cells (Norback and Weltman 1985). Paneth cells play a role in immune function/host defense in the intestine. Recent evidence indicates that they play a role in inflammatory bowel diseases, such as Crohn’s disease and ulcerative colitis (Bevins and Salzman 2011; Gersemann et al. 2011), which are known risk factors for NHL. These observations can also be viewed in the epidemiological context that 12% to 18% of extranodal NHL is categorized as MALT B-cell lymphoma (Wu et al. 2009). Approximately 85% of all MALT lymphoma cases in humans occur in the stomach (Owens and Smith 2011), with fewer in the gastrointestinal tract.
The NTP studies of individual DL congeners PCB 126 and PCB 118 and the non-DL congener 153 clearly show the ability of both DL and non-DL PCB congeners to cause chronic, active inflammation (NTP 2006a; NTP 2006b; NTP 2006c; NTP 2006d; NTP 2010). Chronic exposure to the DL PCBs, either alone or in combination with PCB 153, resulted in thymus and spleen atrophy, which are expressions of immunotoxicity and immunosuppression that are hallmarks of TCDD toxicity. Tetrachlorodibenzo(p)dioxin-induced thymic atrophy has been shown to depend on ligand binding to the AhR receptor (Staples et al. 1998; Tryphonas 1994). Chronic exposure to PCB 153 alone resulted in bone marrow hyperplasia, which could be indicative of immunosuppression, and chronic inflammation of the ovary, oviduct, and uterus. The inflammatory effects on the ovary, oviduct, and uterus were not observed in the studies of PCB 126 and PCB 118 alone (NTP 2006b; NTP 2010). In a binary study, these effects were most clearly observed at the higher ratio of PCB 153 to PCB 126 (NTP 2006c). Since PCB 153 does not act through the AhR receptor, these inflammatory effects and other immunodysregulation must occur by some other mechanism.
Discussion
For PCBs, as for most chemicals, bioassay methodology is developed in the context of models of carcinogenesis. The dominant model in the 1970s, when PCB testing in animals began, was the initiation-promotion model, in which mutagens were considered initiating agents and all others as potential promoting agents. The chronic bioassays were conducted to ascertain whether PCBs were “complete carcinogens,” that is, whether or not they had both initiating and promoting activities. The objective of the most recent chronic bioassays of individual PCB congeners conducted by the NTP was to test their potency relative to TCDD (toxic equivalency factor), and thus they were based on the model of carcinogenesis that DL-PCBs act, like TCDD, through the AhR. In accordance with current protocols, they were conducted in a disease-free environment.
Non-Hodgkin’s lymphomas are tumors of the cells of the immune system, and the MOA almost certainly involves immunosuppression and possibly inflammation. Kerkvliet (2009) has pointed out that, particularly for chemicals that act through the AhR receptor, the effects of exposure are context dependent because of the many different immune-related cells that express AhR. Tetrachlorodibenzo(p)dioxin, for example, affects cells that are responding to immunological stimuli; it has little effect on resting cells (Kerkvliet 2009). Thus, for cancers with an immune system MOA, it may be necessary to incorporate an immune challenge, such as a viral infection, into the bioassay protocol in order for the cancer to develop.
Effects indicative of immune dysfunction were reported in most of the bioassays with commercial PCB mixtures and all of the more recent congener bioassays. However, the NCI (1978) bioassay was the only one that indicated a statistically significant trend for leukemia/lymphoma with increasing dose. We suggest that the concurrent respiratory infection during this bioassay presented an immune challenge that was lacking in other bioassays, and that it led to the observation of NHL. This experimental condition is the most consistent with the observations in epidemiology studies of significant, positive interaction between blood levels of PCBs and titers of antibodies to the EBV virus (Hardell et al. 2009; Nordstrom et al. 2000; Rothman et al. 1997). The impact of an immune challenge during exposure to carcinogens acting via an immune system MOA deserves further study.
The early bioassays of PCBs focused on the liver and would have missed all but gross lesions in other tissues. Other studies conducted histopathological evaluations only on tissues with macroscopic lesions, and they would have missed lymphomas in both the lymph nodes and extranodal locations. This was actually the case, as demonstrated by the reevaluation of the NCI (1978) study by Morgan et al. (1981) and Ward (1985).
The animal bioassays that incorporated lifetime exposure to PCBs to evaluate carcinogenicity were conducted only with rats, as shown in Table 1. Although both rats and mice have been validated for immunotoxicity testing (Germolec 2004), the developing and adult mouse immune system has been far more intensively studied and compared in detail with the human immune system (Hogaboam et al. 2008), and the NTP protocol for immunomodulation testing specifies the use of the mouse (NTP 2009). In addition, a few studies using TCDD have suggested that mice may be more sensitive to some end points of immunosuppression than rats (Smialowicz et al. 1994; Smialowicz et al. 1996). Based on these considerations, the mouse appears to be a more appropriate animal model than the rat for identifying cancers of the immune system, or cancers with an immune system MOA such as NHL.
We conclude that the published bioassays of PCBs provide evidence of dose-related immune system dysfunction, including immunosuppression and inflammation, which are risk factors and part of the putative MOA for NHL. Non-Hodgkin’s lymphoma itself was observed in the NCI (1978) bioassay with a statistically significant dose-related trend, and the data strongly suggest that additional NHL lesions would have been detected if a more complete histological examination had been conducted. The weaknesses of methodologies used to conduct the animal bioassay of PCBs, including the lack of intentional immune challenge in the bioassays, the limited histopathological protocols of the early studies, and the use of a possibly less sensitive rat model rather than mouse model in the recent bioassays together suggest that NHL may have been observed if different experimental choices had been made. We conclude that the lack of concordance between human epidemiology and animal bioassays may simply be the result of the bioassay methodology used, and not a difference in underlying biology. The lack of concordance should not be construed as evidence that PCBs do not contribute to the risk of NHL.
Footnotes
Acknowledgments
Shira Kramer and others at Epidemiology International provided the impetus for this work and helpful comments. Whitney Cowell provided technical assistance. We thank Dr. Jerrold Ward for his review and comments on an early draft of this paper and the peer reviewer for comments that strengthened it. This work was funded by Allen Stewart, P.C., although the sponsor had no control over the design, interpretation, writing, or publication of this work.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Harlee Strauss is employed by H. Strauss Associates, Inc., a for-profit consulting company. Wendy Heiger-Bernays is on the faculty of the Boston University School of Public Health. Manuscript preparation was funded by Allen Stewart, P.C., a law firm. The funder had no control over the design, interpretation, writing, or publication of this work. No other work has been conducted for this law firm by either Dr. Strauss or Dr. Heiger-Bernays.
