Abstract
Giant cell tumor of bone (GCTB) is a common primary neoplasm of bone characterized by distinctive clinicopathological features. GCTB is exceedingly rare in nonhuman species, and it has been sporadically reported in cats, dogs, rats, and birds. This report describes a primary murine bone tumor that shares striking clinicopathological similarities with human GCTB. The neoplasm occurred in a 71-week-old C57BL/6 mouse heterozygous for the specific
Keywords
With the exception of osteomas in CF-1 and OF-1 outbred strains and osteosarcomas in nonobese diabetic (NOD) and NOD-derived mice, spontaneous primary bone tumors are exceedingly rare in most strains of laboratory mice (Charles and Turusov 1974; Kavirayani and Foreman 2010; Kavirayani, Sundberg, and Foreman 2012; Wilson, Hauser, and Ryffel 1985). Nevertheless, the recent advent of high-throughput transgenic and gene-targeting technologies has led to the development of innumerable genetically engineered mouse (GEM) models that both phenotypically and genotypically recapitulate the most salient pathobiological features of specific categories of skeletal tumors in humans (Kavirayani, Sundberg, and Foreman 2012).
Giant cell tumor of bone (GCTB) stands among those human bone tumors for which a murine equivalent has never been described neither as spontaneous lesion nor as GEM or experimentally induced model. In humans, GCTB is a benign tumor that generally arises from the meta-epiphysis of long bones (distal femur and proximal tibia in particular). GCTB accounts for approximately 5% of primary bone tumors and 20% of benign bone tumors. Females are slightly more affected than males, and the majority of patients are in their 20s to 50s (Mendenhall et al. 2006; Thomas and Skubitz 2009; Werner 2006). Most GCTBs are solitary; however, multicentric tumors do occur, especially in the distal extremities (Dhillon and Prasad 2007). Despite its benign designation, GCTB often exhibits local aggressiveness with remarkable bone destruction and invasion of adjacent soft tissues. Further underlying its unpredictable behavior, treatment by conservative local surgery is associated with a recurrence rate of approximately 30% to 60%, and a small subset of apparently benign GCTBs give rise to pulmonary metastases (Alberghini et al. 2010; Becker et al. 2008; Campanacci et al. 1987; Liu and Wang 1998; O’Donnell et al. 1994).
Histologically, GCTB consists of a mixture of round and spindle mononuclear cells, scattered among which are large multinucleated osteoclast-like giant cells. The exact histogenesis of GCTB is still controversial (Morgan et al. 2005). Although historically classified as “osteoclastoma” and currently referred to as “giant cell tumor,” osteoclast-like giant cells do not represent the neoplastic component of the neoplasm. In this regard, recent studies indicate that stromal spindle cells, the major component of the mononuclear cell population, are the true neoplastic elements of GCTB (Mendenhall et al. 2006; Salerno et al. 2008; Wülling et al. 2001; Yasko 2002; Zheng et al. 2001).
GCTB is exceedingly rare in nonhuman species, and so far it has been only sporadically described in cats, dogs, rats, and birds (Amann et al. 2007; Berg, Gliatto, and Wallace 1990; Ferreras et al. 2005; Imai et al. 1998; Trigo, Leathers, and Brobst 1983; Walsh and Rhodes 1995). This study aims at describing the pathological and immunohistochemical features of a GCTB-like lesion that occurred in a 71-week-old C57BL/6 female mouse harboring a
Tumor spectrum and frequency in a cohort of 19 C57BL/6 mice heterozygous for the specific Trp53 R172H point mutation (Lang et al. 2004).

Clinically, the mouse presented with hunched posture, labored breathing, and right paraparesis associated with marked nodular swelling of the right thigh. The mouse eventually became moribound and was humanely euthanized by CO2 asphyxiation followed by atlanto-occipital dislocation. A complete necropsy was performed, and thymus, lungs, heart, spleen, liver, pancreas, kidneys, and mesenteric lymph nodes were collected and immediately fixed in 10% neutral buffered formalin (NBF). The affected hind limb was removed in toto and decalcified for 20 days in a supersaturated solution of EDTA in 10% NBF. Sampled organs/tissues were routinely processed for histological examination and embedded in paraffin blocks. Four-micrometer-thick sections obtained from these blocks were then stained with hematoxylin and eosin and evaluated under a light microscope. Serial sections from the nodular lesion in the right thigh were also obtained for further immunohistochemical analyses, picrosyrius red staining, and tartrate-resistant acid phosphatase (TRAP) activity (catalog 387A, Sigma-Aldrich, St. Louis, MO). Details concerning the panel of immunohistochemical stains applied are summarized in Table 2.
Details concerning primary antibodies and procedures used for the immunohistochemical examination of the primary murine bone tumor reported in this study.
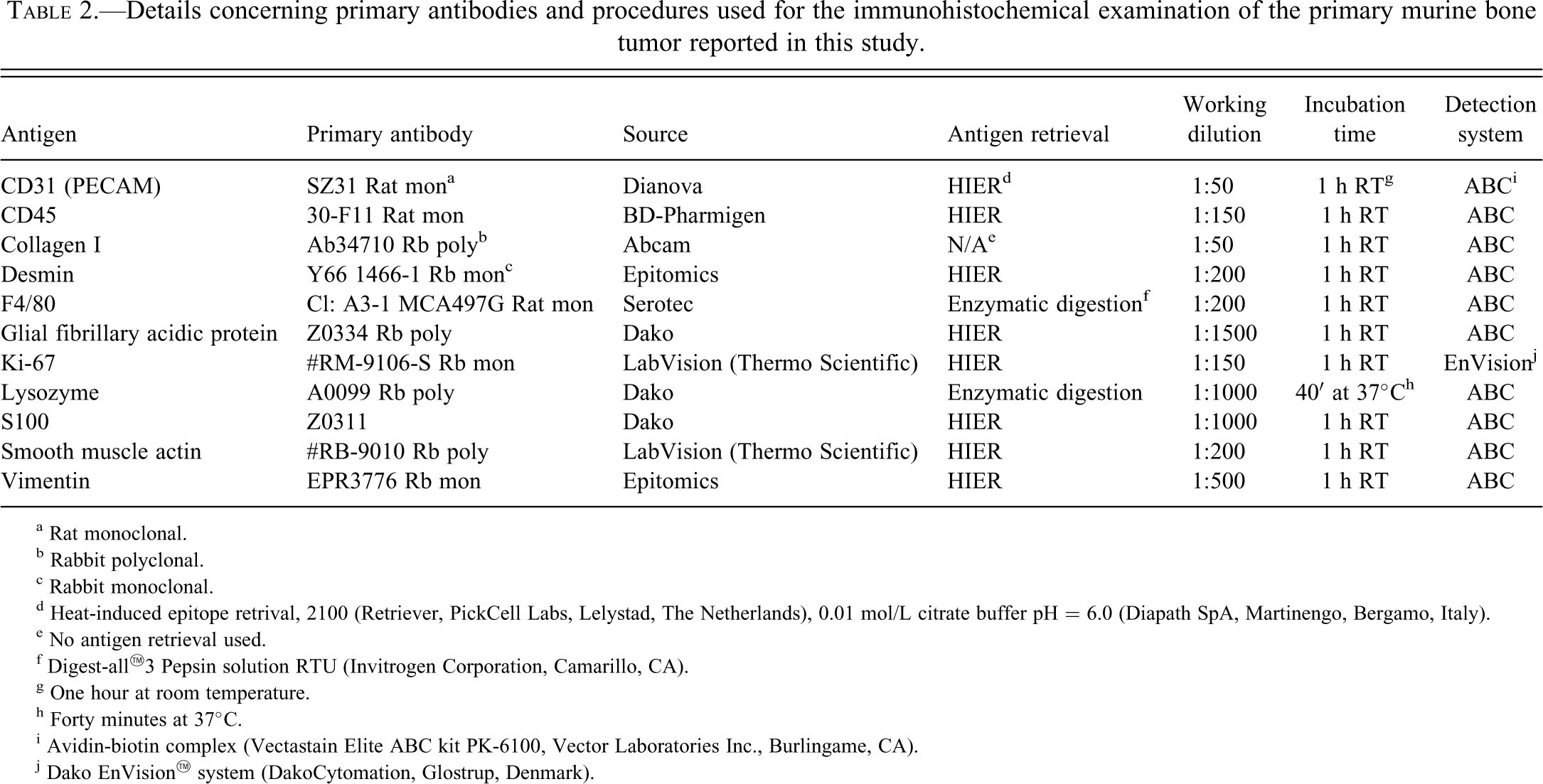
a Rat monoclonal.
b Rabbit polyclonal.
c Rabbit monoclonal.
d Heat-induced epitope retrival, 2100 (Retriever, PickCell Labs, Lelystad, The Netherlands), 0.01 mol/L citrate buffer pH = 6.0 (Diapath SpA, Martinengo, Bergamo, Italy).
e No antigen retrieval used.
f Digest-all™3 Pepsin solution RTU (Invitrogen Corporation, Camarillo, CA).
g One hour at room temperature.
h Forty minutes at 37°C.
i Avidin-biotin complex (Vectastain Elite ABC kit PK-6100, Vector Laboratories Inc., Burlingame, CA).
j Dako EnVision™ system (DakoCytomation, Glostrup, Denmark).
Procedures involving animals and their care conformed to institutional guidelines in compliance with national (D.L.vo 116/92 and following additions) and international (EEC Council Directive 86/609, OJ L 358,1,12-12-1987; NIH Guide for the Care and Use of Laboratory Animals, U.S. National Research Council 1996) laws and policies.
At necropsy, the swelled right thigh turned out to be affected by a whitish and solid nodular mass measuring approximately 12 × 8 × 6 mm. The lesion arose from the mediocaudal aspect of the distal metaphysis of the right femur and appeared to be well demarcated from the surrounding musculofascial tissues. Additional gross changes included moderate splenomegaly and mild mesenteric lymphadenomegaly.
Microscopically, the affected portion of the femur was effaced by a densely cellular osteolytic neoplasm that, apart from few satellite nodules and focal infiltrative extensions into the surrounding soft tissues, appeared to be well demarcated, partially capsulated with an expansile growth pattern and compression atrophy of the adjacent skeletal muscles (Figure 1A). Tumors consisted of short irregular bundles of pleomorphic spindle-shaped mononuclear cells (SMCs) interspersed with numerous large multinucleated giant cells (MGCs). This latter cell population often displayed an osteoclast-like morphology with dendritic outlines, a moderate amount of dense eosinophilic cytoplasm, and up to 50 small uniform oval nuclei clustered in the center of the cell (Figure 1A, inset). Findings of cellular atypia including marked anisocytosis, anisokaryosis, karyomegaly, cytomegaly, hyperchromasia, multiple irregular nucleoli, and numerous and often bizarre mitotic figures (8-12 × hpf) were almost exclusively observed in a subset of SMCs. Individual or small aggregates of medium-sized round mononuclear cells (RMCs) morphologically consistent with activated macrophages were also scattered throughout the tumor. As highlighted by picrosyrius red stain, the neoplasm was supported by a scant amount of stroma characterized by a combination of delicate fibrovascular septa and broad bundles of fibrous connective tissue. The severe tumor-associated osteolysis was occasionally accompanied by a minimal amount of reactive woven bone formation.
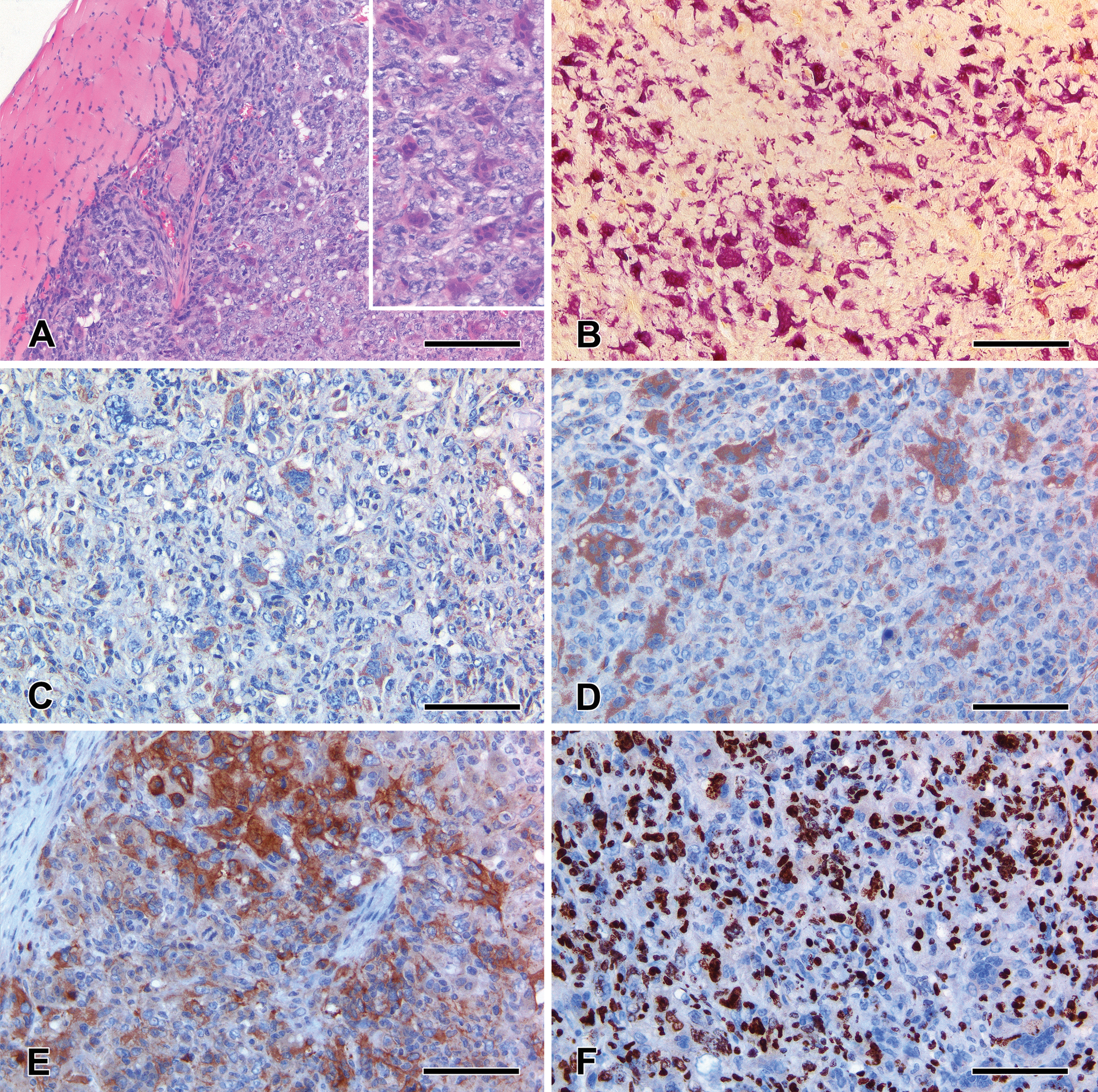
Giant cell tumor of bonelike lesion in a 71-week-old Trp53 mutant female mouse. Densely cellular heterogeneous tumor that compresses and focally infiltrates the adjacent skeletal muscles; hematoxylin and eosin (H&E) stain, scale bar 200 µm (A). Note the morphology of the scattered intratumoral osteoclast-like multinucleated giant cells; H&E stain, scale bar 100 µm (A, inset). An elevated number of tartrate-resistant acid phosphatase (TRAP)–positive osteoclast-like multinucleated giant cells populate the neoplasm; TRAP histochemistry, scale bar 200 µm (B). Marked CD45 and lysozyme immunoreactivity in the osteoclast-like multinucleated giant cells and in scattered round mononuclear cells morphologically consistent with activated macrophages; note the unstained pleomorphic spindle-shaped mononuclear cells; CD45 and lysozyme immunoperoxidase staining, scale bar 100 µm (C and D, respectively). Scattered bundles of spindle-shaped mononuclear cells are markedly immunoreactive for smooth muscle actin (SMA), SMA immunoperoxidase staining, scale bar 100 µm (E). Ki-67 nuclear expression in the vast majority of pleomorphic spindle-shaped mononuclear cells; note the unstained osteoclast-like multinucleated giant cell; Ki-67 immunoperoxidase staining, scale bar 100 µm (F).
A detailed summary of the histochemical and immunohistochemical features observed in SMCs, MGCs, and RMCs is reported in Table 3. Osteoclast-like MGCs exhibited prominent tartrate-resistant acid phosphatase (TRAP) activity (Figure 1B) and consistent immunohistochemical expression of vimentin and histiocytic markers including CD45 (common leukocyte antigen) and lysozyme (Figures 1C and D). Few scattered MGCs also displayed weak immunoreactivity for smooth muscle actin (SMA). No TRAP activity or lysozyme and CD45 immunohistochemical expression was identified in SMCs. This latter cell population showed marked and diffuse vimentin immunoreactivity. A subset of SMCs was also strongly positive for SMA (Figure 1E). Variable degrees of vimentin, CD45, lysozyme, and F4/80 expression were observed in the intratumoral aggregates of RMCs morphologically consistent with activated macrophages. Ki-67 immunohistochemistry revealed a remarkable number of positive nuclei within the SMC population, whereas a negligible level of Ki-67 expression was evident in MGCs and RMCs (Figure 1F). SMCs, MGCs, and RMCs were largely negative for CD31/PECAM, collagen I, glial fibrillary acidic protein, and S100 immunohistochemistry.
Summary of the histochemical and immunohistochemical phenotype displayed by spindle-shaped mononuclear cells, multinucleated giant cells, and round mononuclear cells populating the murine giant cell tumor of bonelike lesion examined in this study.
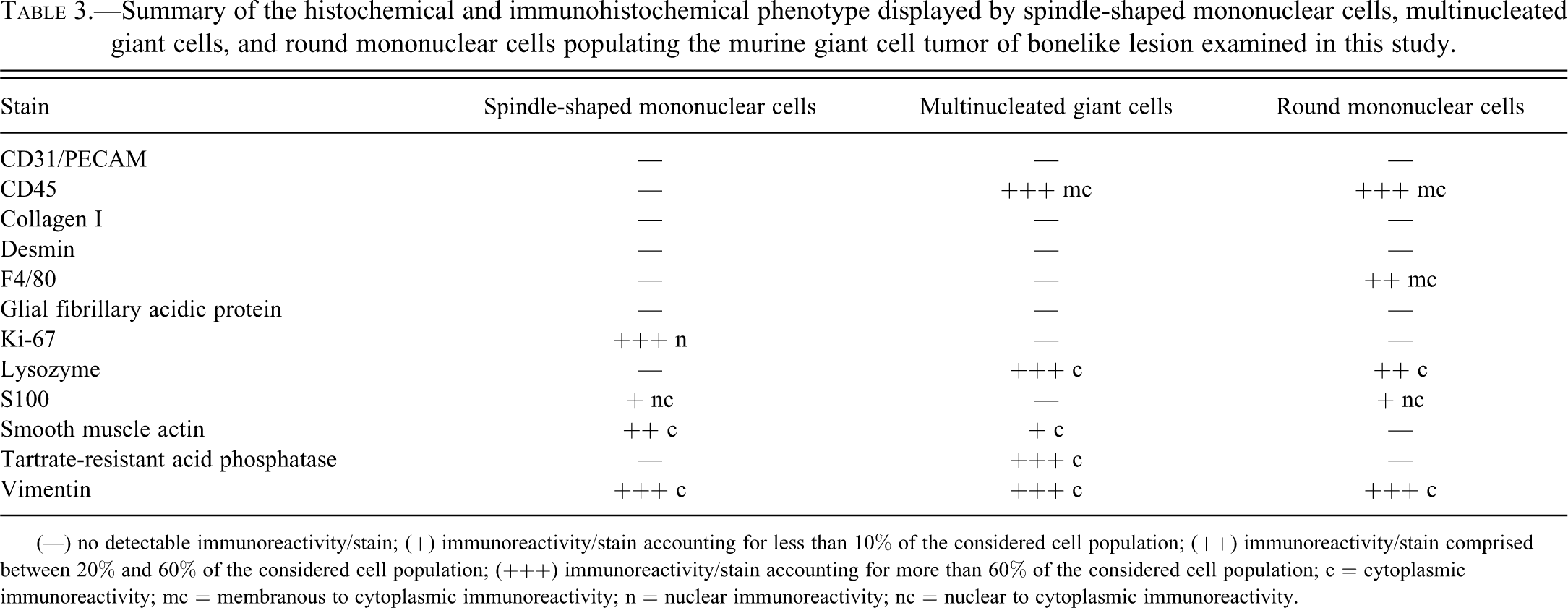
(—) no detectable immunoreactivity/stain; (+) immunoreactivity/stain accounting for less than 10% of the considered cell population; (++) immunoreactivity/stain comprised between 20% and 60% of the considered cell population; (+++) immunoreactivity/stain accounting for more than 60% of the considered cell population; c = cytoplasmic immunoreactivity; mc = membranous to cytoplasmic immunoreactivity; n = nuclear immunoreactivity; nc = nuclear to cytoplasmic immunoreactivity.
Additional histopathologic changes observed in the other organs/tissues included splenic low-grade marginal zone lymphoma and marked sinus histiocytosis of the mesenteric lymph nodes.
This report describes a primary murine bone tumor that shares striking clinicopathological similarities with human GCTB. The neoplasm presented as a mono-ostotic nodular proliferation arising from the distal femoral metaphysis. Interestingly, femoral metaphysis is one of the most common sites of origin for solitary GCTB in humans (Mendenhall et al. 2006). In this context, it should be also recalled that the only GCTB so far described in a laboratory rodent (i.e., a 98-week-old male F344 rat) arose from the distal end of the femur (Imai et al. 1998). Based on the morphological and immunohistological results obtained, the bone tumor described in this study appears to be composed of the same heterogeneous cell populations typically observed in human GCTB.
SMCs were the principal cell type observed in the murine GCTB-like lesion. Consistent with the putative origin of human SMCs from multipotent mesenchymal stem cells (Robinson, Segal, and Nevo 2002-2003; Roux et al. 2002; Werner 2006; Wülling et al. 2001; Wülling, Delling, and Kaiser 2003), murine SMCs displayed an indefinite mesenchymal histogenesis being positive only for vimentin and, to a lesser degree, SMA. It is now well established that SMCs represent the true neoplastic component of GCTB. The same could be assumed also for the bone tumor described in this report as features of cellular atypia, aberrant mitosis, and abnormally high proliferative activity were limited to the SMC component. Similar anaplastic changes and increased proliferative activity are well documented also in human GCTB, where they are considered hallmarks of sarcomatous transformation and “dedifferentiation,” carrying a dismal prognosis in term of metastatic potential (Bertoni, Bacchini, and Staals 2003; Forsyth and Hogendoorn 2003; Reid, Banerjee, and Sciot 2002). SMA immunoreactivity in the murine GCTB-like lesion further supports this view since the expression of this marker has been specifically reported in primary GCTBs with high recurrence and/or metastatic potential (Alberghini et al., 2010). The exact biological significance of SMA expression by SMCs from GCTB is still unclear and a matter of controversy. It may indicate the acquisition of a myofibroblastic phenotype, as suggested by some studies (Imai et al. 1998; Watanabe et al. 1997). However, other authors report that myofibroblastic differentiation in SMCs is unlikely as they lack key structures typical of both reactive and transformed myofibroblasts, including fibronexus (Watanabe et al. 2000). It must be also considered that transformed multipotent mesenchymal stem cells (the progenitor cells SMCs are supposed to derive from) have been demonstrated to express SMA and display a myofibroblastic-like phenotype under the influence of a wide plethora of oncogenic stimuli (Burns et al. 2008; Jeong et al. 2011). Malignant transformation in GCTB has been also frequently correlated with p53 mutations, and in this context, it should be emphasized that the affected mouse was genetically manipulated to carry a point mutant p53 allele (Oda et al. 2001; Wu et al. 1997).
MGCs observed in the murine GCTB-like lesion confirmed their actual osteoclast differentiation exhibiting prominent TRAP activity and consistent immunohistochemical expression for markers of the monocyte/macrophage lineage including CD45 and lysozyme. Both CD45 and lysozyme have been previously reported to specifically identify the MGCs populating GCTB (Aqel, Pringle, and Horton 1988; Bouropoulou, Kontogeorgos, and Manika 1985; Ferreras et al, 2005; Fornasier et al. 1996; Morgan et al. 2005; Roux et al. 2002).
Immunohistochemical examination of the murine GCTB-like lesion also highlighted the presence of multiple intratumoral aggregates of activated macrophages in the form of RMCs with variable degrees of vimentin, CD45, lysozyme, and F4/80 expression. It has been widely documented that GCTB is populated by a variable number of ovoid mononuclear cells phenotypically consistent with activated monocytes/macrophages (Forsyth and Hogendoorn 2003; Mendenhall et al. 2006; Reid, Banerjee, and Sciot 2002; Thomas and Skubitz 2009; Werner 2006). Beside the expression of typical lineage-specific markers (e.g. CD68), GCTB-associated monocytes/macrophages have been demonstrated to represent osteoclast precursors that, in response to a paracrine-juxtacrine stimulus likely generated by SMCs, are recruited in the site of tumor development and differentiate into osteoclasts. In this context, GCTB-associated osteoclastogenesis has been delineated at the molecular level, highlighting how the interaction between receptor activator of nuclear factor κ B and its ligand expressed on recruited monocyte/macrophages and SMCs, respectively, is a key to the formation and maturation of osteoclasts (Roux et al. 2002; Werner 2006; Zheng et al. 2001). Other humoral factors released by SMCs such as stromal cell derived factor-1, macrophage chemoattractant protein-1, and macrophage colony-stimulating factor proved to play a critical role in the production, recruitment, and fusion of osteoclast precursors (Liao et al. 2005; Miyamoto et al. 2000; Zheng et al. 1998).
From a pathobiological perspective, it is unclear whether the same molecular mechanisms underlying GCTB in humans are also responsible for the development of the primary murine bone tumor described in this study. For this reason, the neoplasm has been cautiously defined as GCTB-like lesions on a morphophenotypic basis.
The main differential diagnoses considered for this case of murine GCTB-like lesion were rhabdomyosarcoma, histiocytic sarcoma, and giant cell-rich osteosarcoma. However, since both MGCs and SMCs failed to express immunohistochemical markers indicative of muscular (i.e., desmin), histiocytic (i.e., F4/80), and osteoblastic (i.e., collagen I) differentiation, these alternative diagnostic hypotheses were ruled out. In addition, standard histology combined with collagen I and picrosyrius red stain did not evidence any intratumoral accumulation of extracellular matrix compatible with osteoid. In this regard, the complete lack of intratumoral deposits of osteoid is generally regarded as an important diagnostic feature to differentiate GCTB from giant cell-rich osteosarcoma (Negrin et al. 2006; Sato et al. 1996).
In conclusion, the unusual primary murine bone tumor described in this study has been putatively classified as GCTB since it recapitulates many of the morphological and immunohistological features of the equivalent human condition. The accurate definition of this rare spontaneous tumor in mice may eventually stimulate the design and conduction of experiments aimed at establishing an animal model that may serve as a tool to dissect the molecular mechanisms underlying the onset and progression of GCTB and to test novel therapeutic strategies. This case report should also reinforce the awareness of the possible occurrence of previously undescribed bone tumor phenotypes in GEM models in which the function of crucial gatekeeper tumor suppressor genes (e.g.,
Footnotes
Acknowledgments
We gratefully acknowledge Drs. Marco Losa, Camilla Recordati, and Vittoria Castiglioni of the Mouse & Animal Pathology Lab for their assistance with immunohistological procedures.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
