Abstract
To investigate the toxicity and carcinogenic potential of indole-3-carbinol (I3C), the National Toxicology Program has conducted 13-week subchronic studies in Fisher 344 rats and B6C3F1 mice, and chronic 2-year bioassays in Sprague-Dawley rats and B6C3F1 mice. While the chronic study results are not yet available, subchronic study results and short-term special evaluations of interim sacrifices in the 2-year rat bioassay are presented. F344 rats were orally gavaged ≤300 mg I3C/kg body weight 5 days a week for 13 weeks. Rats treated with ≥150 mg/kg demonstrated a dose-related dilation of lymphatics (lymphangiectasis) of the duodenum, jejunum, and mesenteric lymph nodes. Material within dilated lacteals stained positively for Oil Red O and Sudan Black, consistent with lipid. Electron microscopic evaluation confirmed extracellular lipid accumulation within the villar lamina propria, lacteals, and within villar macrophages. Analyses of hepatic and pulmonary CYP1A enzymes demonstrated dose-dependent I3C induction of CYP1A1 and 1A2. B6C3F1 mice orally gavaged ≤250 mg I3C/kg body weight did not demonstrate histopathological changes; however, hepatic CYP induction was similar to that in rats. The histopathologic changes of intestinal lymphangiectasis and lipidosis in this study share similarities with intestinal lymphangiectasia as observed in humans and dogs. However, the resultant clinical spectrum of protein-losing enteropathy was not present.
Keywords
Introduction
Indole-3-carbinol (I3C) is a naturally formed breakdown product of glucosinolate glucobrassicin, a component found in cruciferous vegetables of the Brassica genus. This genus includes Brussels sprouts, cauliflower, cabbage, kale, rape, turnips, and broccoli. Although I3C does not naturally occur in these vegetables, it does readily form from glucobrassicin via a decomposition process initiated during the simple mechanics of cutting, chewing, mashing, or cooking. Aside from being found in cruciferous vegetables, I3C is also marketed and promoted as a dietary supplement with various health claims including cancer prevention, antioxidant protection, reinforcement of the body’s immune system, and enhancement of hepatic and intestinal detoxification of chemicals (Rosen and Bryson 2004; van Poppel 1999).
Amongst other mechanisms, the protective effect of I3C against estrogen-responsive tumors is thought to be mediated in part by the modulation of cytochrome P450 (CYP)-mediated estrogen metabolism (Auborn et al. 2003). Since CYP enzymes are known to metabolize carcinogenic chemicals in various tissues, induction by I3C could potentially result in altered metabolism of carcinogens. In animal models, I3C induces the expression and activity of CYP1A1 in liver, mammary, and colon; and 1A2, 1B1, 2B1/2, and 3A in the liver (Bradfield and Bjeldanes 1984, 1987; Vang, Jensen, and Autrup 1990; Shertzer and Sainsbury 1991; Jellinck et al. 1993; Manson et al. 1998; Larsen-Su and Williams 2001; Horn et al. 2002; Crowell et al. 2006). Induction of CYP1A1 and CYP1A2 may play a critical role in the detoxification of carcinogens, inhibiting initiation in the case of mammary carcinogenesis (Grubbs et al. 1995).
Conversely, CYP1A1 is involved in the metabolic activation of procarcinogens, such as polycyclic aromatic hydrocarbons, and increased CYP1A1 induction is generally associated with toxicity via unknown mechanisms. Induction of CYP1A1 and 1A2 are characteristic responses following exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) and dioxin-like compounds, which are multisite carcinogens in rodents (National Toxicology Program [NTP] 1982a, 1982b, 2004a, 2004b, 2004c, 2004d, 2004e, 2004f, 2004g). There is mounting concern regarding the potential for carcinogenic effects as a result of I3C exposure. I3C and its condensation products are natural ligands that, like dioxins, bind with varying affinity to the aryl hydrocarbon receptor (AhR) (Bjeldanes et al. 1991; Jellinck et al. 1993; Gillner et al. 1993). Among a battery of other responsive genes, perturbation of AhR modulates CYP1A1 and 1A2 expression.
In contrast to the protective effects of I3C on tumorigenesis, several studies in laboratory animals have demonstrated that I3C has a promotional effect on neoplastic development at multiple sites in rodent initiation-promotion models of carcinogenesis (Pence, Buddingh, and Yang 1986; Kim et al. 1994, 1997; Yoshida et al. 2004). In a multiorgan rat model, I3C induced both inhibition and promotion of tumor development in the same study, depending on the target organ of interest (Stoner et al. 2002). These seemingly well-established, paradoxical effects of I3C on tumor development at multiple sites are not clearly understood.
While I3C is neither cytotoxic nor mutagenic in vitro (Reddy et al. 1983; Babich, Borenfreund, and Stern 1993), contradictory results from animal studies regarding the anticarcinogenic mechanisms of I3C have led to some concern over its widespread use as a supplement for cancer prevention (Higdon et al. 2007). Additionally, little is known about the consequence of long-term exposure to I3C. Given the potential for widespread human exposure, its ability to metabolize chemicals to their carcinogenic form through activation of cytochrome P4501A1 and 1A2, as well as the conflicting animal data, further evaluation of the toxicity and carcinogenic potential of I3C is needed (Higdon et al. 2007; Horn et al. 2002; Vang, Jensen, and Autrup 1990). Therefore, to investigate the toxicity and carcinogenic potential of I3C, the NTP has conducted 13-week subchronic studies in Fisher 344 rats and B6C3F1 mice, and chronic 2-year bioassays in Sprague-Dawley rats and B6C3F1 mice. The results of the subchronic studies and a series of short-term special evaluations conducted as interim sacrifices in the 2-year bioassays, the basis of which were the findings of the subchronic studies, are currently presented.
Materials and Methods
Chemical and Dose Formulation
Bulk indole-3-carbinol (CAS no. 700-06-1; Southern Research Lot No. SRI F52\L-2) was obtained from Chem Pacific Corporation (Baltimore, MD; Lot No. CHP801001) and supplied by Batelle Memorial Institute (Columbus, OH). It was protected from light, stored frozen at approximately –20°C, and was determined to be 99.5% pure (National Toxicology Program, Bulk Chemical Comprehensive Analysis Report: Indole-3-Carbinol, February 2004). Mazola corn oil (Red Diamond Food Service; Birmingham, AL) served as both the vehicle and the control article. The corn oil was stored in a cold room at approximately 5°C and was determined to have acceptable limits of peroxides (<3meq/kg).
Dose formulations of I3C were prepared 7 different times during the study, on week 1 and every 2 weeks thereafter, at concentrations of 3.75, 7.50, 15.0, 30.0, and 60.0 mg/mL for rats and 1.56, 3.13, 6.25, 12.5, and 25.0 mg/mL for mice. All dose formulations were determined to be within ±10% of these target concentrations. Prepared formulations were stored refrigerated in sealed amber glass containers, protected from light. Under such conditions, formulations were determined to be stable for at least 41 days, with target concentrations ranging from 105% (day 3) to 96.6% (day 21) (Weinberg et al. 2004).
Dose levels in rats and mice were selected based on previously performed National Cancer Institute (NCI) studies (Crowell et al. 2006).
Animals and Study Design
All animals were obtained from Taconic Farms, Inc. (Germantown, NY), and all experiments were conducted for the NTP at Southern Research Institute (Birmingham, AL). Animals were randomly assigned to and within control and treated groups. Both male and female F344/N rats, in addition to female B6C3F1 mice, were group-housed 5 per cage. Male B6C3F1 mice were individually housed. All female and male special study Harlan Sprague-Dawley (HSD) rats were housed up to 5 per cage. Core study HSD rats were housed up to 3 per cage. A programmatic change took place in the NTP during the conduct of the 3-month study, leading to replacement of the F344 rat with the HSD rat in NIEHS bioassays (King-Herbert and Thayer 2006; King-Herbert, Sills, and Bucher 2010). The 3-month study was not repeated in the HSD rat. All animals were housed in solid-bottom polycarbonate cages lined with irradiated hardwood chip bedding, suspended on stainless steel racks, and covered with disposable filters. All animals (both mice and rats) were housed in the same room. Environmental controls were set to maintain filtered room air at a uniform flow rate of 10–20 exchanges per hour, temperature between 65°F and 75°F, relative humidity within 35–65%, and alternating cycles of light and dark at 12-hour intervals. Control and treated groups received irradiated NTP-2000 wafer feed (Zeigler Brothers, Inc; Gardners, PA) ad libitum. Birmingham municipal water was received ad libitum via an automated watering system (Edstrom Industries, Inc; Waterford, WI). Animal husbandry and handling was conducted in concordance with NIH guidelines (Grossblatt 1996).
Subchronic study (3 month)
Ten male and 10 female B6C3F1 mice were assigned to each of 6 dose groups (0, 15.6, 31.25, 62.5, 125, or 250 mg/kg body weight) based on previously described I3C studies (Crowell et al. 2006). The mice were approximately 4–5 weeks of age on day 1 and were administered I3C via oral gavage once daily, 5 days per week (excluding holidays and weekends) for 93 days. Doses were calculated using a volume of 10 mL/kg body weight. The initial dose volume was based on the animal’s body weight on day 1, whereas following dose volumes were based on the animal’s body weight from the previous week. Body weights and clinical signs of toxicity were evaluated and recorded weekly. Hematology parameters were evaluated in conjunction with terminal euthanasia on Day 94. Clinical chemistry parameters were not evaluated in mice. Cytochrome P450 (CYP) 1A1 activity in the lung and liver and CYP1A2 activity in the liver were measured at terminal sacrifice (see below). Complete necropsy examinations were performed on all animals; histopathological examinations were performed on all control and high-dose groups.
Ten male and 10 female F344 rats were assigned to each of the 6 core study and 6 special study groups with doses of 0, 18.75, 37.5, 75, 150, or 300 mg/kg body weight based on previously described I3C studies (Crowell et al. 2006). The rats were approximately 5–6 weeks of age on day 1 of the study. They were administered I3C via oral gavage once daily, 5 days per week (excluding holidays and weekends) for 93 days (core study) or 25 days (clinical pathology special study). Doses were calculated using a volume of 5 mL/kg body weight. The initial dose volume was based on the animal’s body weight on day 1, whereas following dose volumes were based on the animal’s body weight from the previous week. Body weights and clinical signs of toxicity were evaluated and recorded weekly for core study groups. Clinical chemistry parameters evaluated in rats were sorbitol dehydrogenase (SDH), alkaline phosphatase (ALP), alanine aminotransferase (ALT), creatinine kinase (CK), total protein, albumin, urea nitrogen, total bile acids, creatinine, and glucose. Clinical pathology parameters were evaluated on days 4 and 25 for special study groups and in conjunction with terminal sacrifice on day 94 for core study groups. CYP1A1 activity in the lung and liver and CYP1A2 activity in the liver were measured at terminal sacrifice for core study groups. For core study groups, full gross pathology examinations were performed on all animals; full histopathological examinations were performed on all control and high-dose groups. Histopathologic examinations for the low- and intermediate-dose groups were confined to sections of duodenum, jejunum, and mesenteric lymph node only. For special study groups, full gross pathology examinations were performed on all animals; however, histopathologic examinations were confined to sections of duodenum, jejunum, and mesenteric lymph node only. The incidence of lymphangiectatic lesions in these organs was reported as number of animals affected/number of animals in dose group. Average severity grades for lymphangiectatic lesions reported (parenthetically) were calculated by multiplying each severity grade (0–4) by the number of animals with that grade, summing the totals, and dividing by the number affected.
One-week and 4-week interim sacrifice of the 2-year study—Lipidosis evaluation
Ten male Harlan Sprague-Dawley (HSD) rats were randomly assigned to each of 4 special study dose groups (0, 75, 150, and 300 mg/kg). Five males in each dosage group were terminated at 1 and 4 weeks of treatment for lipidosis evaluation as an interim sacrifice in the 2-year study. Animals were euthanized using 100% carbon dioxide, and full gross pathology examinations were performed. Histopathologic examination was confined to sections of the proximal duodenum and proximal jejunum. Additional sections of duodenum and jejunum were harvested and frozen for histochemical analysis.
Clinical Chemistry and Hematology
Animals were anesthetized with carbon dioxide and bled from the retro-orbital sinus for evaluation of hematology (rats and mice) and clinical chemistry (rats only). Blood was collected in either serum separator tubes (clinical chemistry) and/or tubes containing EDTA (hematology).
Clinical chemistry evaluations were performed within 4 hours of collection and the following values were assessed: sorbitol dehydrogenase (SDH), alkaline phosphatase (ALP), alanine aminotransferase (ALT), creatinine kinase (CK), total protein, albumin, urea nitrogen, total bile acids, creatinine, and glucose.
Hematology analyses were performed within 6 hours of collection and included evaluation of the following parameters: erythrocyte count, hemoglobin, packed cell volume (PCV) (automated and spun hematocrit), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), platelet count, reticulocyte count, total leukocyte count, differential leukocyte count, red blood cell morphology, platelet morphology, and nucleated red blood cell count.
Pathology
Animals were euthanized on either day 94 (core study F344 rats and B6C3F1 mice), day 25 (special study F344 rats), or at 1 or 4 weeks (interim sacrifices of the 2-year bioassay in Harlan SD rats) using 100% carbon dioxide. Complete gross necropsies were performed on all animals, in which all organs and tissues were evaluated for grossly visible lesions. In all core study animals, the following organs were weighed: liver, right kidney, heart, lung, thymus, and right testis (males). Tissues were collected and fixed in 10% neutral buffered formalin. Tissues were processed, trimmed, embedded in paraffin, and sectioned to a thickness of 4–6 μm. Sections were mounted on glass microscopes slides, stained with hematoxylin and eosin, and processed for microscopic evaluation. Detailed histopathologic examinations were conducted on all animals in the control and high-dose groups from the 3-month study. This included evaluation of the following: adrenal glands, brain, clitoral glands, esophagus, eyes, femur, harderian glands, heart and aorta, small and large intestines, kidneys, liver, lungs and mainstem bronchi, lymph nodes, mammary glands with adjacent skin, muscle, nasal cavity, ovaries, pancreas, parathyroid glands, pituitary gland, preputial glands, prostate, salivary glands, seminal vesicles, spinal cord, spleen, stomach, testes and epididymis, thymus, thyroid gland, trachea, urinary bladder, and uterus, in addition to any tissue masses and/or gross lesions. For animals in the low- and intermediate-dose groups from the 3-month study, histopathologic examination was confined to the duodenum, jejunum, and mesenteric lymph node. Duodenum and jejunum were evaluated from all animals in the day-25 special study. The incidence of lymphangiectatic lesions in these organs was reported as number of animals affected/number of animals in dose group. Average severity grades for lymphangiectatic lesions reported (parenthetically) were calculated by multiplying each severity grade (0–4) by the number of animals with that grade, summing the totals, and dividing by the number affected.
Intestinal sections were transversely sectioned and evaluated for the presence of lymphangiectasis or lipidosis. Lesions were graded as follows:
Grade 1 (Minimal): Expansion of the villous lymphatic vessels with one to several dilatations with minimal disruption of villous architecture.
Grade 2 (Mild): Expansion of the villous lymphatic vessels with several to numerous small to medium-sized dilatations, with mild disruption of the villous architecture.
Grade 3 (Moderate): Expansion of the villous lymphatic vessels with several to numerous medium-sized to large dilatations, with moderate disruption of the villous architecture.
Grade 4 (Marked): Expansion of the villous lymphatic vessels with several to numerous medium-sized to large dilatations, with marked disruption of the villous architecture.
Histochemical Analysis for Lipid Accumulation
To confirm the presence of lipid within dilated lacteals in the intestinal villi, additional histochemical analyses were performed on duodenal and jejunal samples from 9 of the 1- and 4-week interim sacrifice of the 2-year study animals (2 control, 1 mid-dose, and 2 high-dose animals dosed for 1 week; 1 control, 1 mid-dose, and 2 high-dose animals dosed for 4 weeks). Tissues were cryosectioned and stained with Oil Red O or Sudan Black (lipid stains) (one section of each organ per slide).
Electron Microscopy
Samples from the jejunum of 8 rats from the 2-year interim sacrifice study (3 control and 2 high-dose animals dosed for 1 week, and 3 high-dose animals dosed for 4 weeks) were evaluated via transmission electron microscopy. Appropriate samples were collected, processed, and embedded in Spurr’s resin. Thick sections (1 μm sections stained with 1% Toluidine blue) were cut and examined by light microscopy to select the sample for electron microscopy. Thin sections (approximately 80 nm) were then cut from epoxy blocks, mounted on 200-mesh copper grids, stained with methanolic uranyl acetate and Reynold’s lead citrate, and examined on a Zeiss 900 transmission electron microscope. Five representative ultrastructure photomicrographs were taken for each sample examined.
Cytochrome P450 Enzyme Analysis
Samples from the median and left liver lobes were collected during necropsy from all core study animals. Samples were minced and pooled together, frozen, and stored at –80° C. A sample of right lung lobe was similarly collected from each animal. The frozen samples were used in preparation of microsomes and measurement of CYP1A1 in lung and liver and CYP1A2 in liver, based on the activities of 7-ethoxyresorufin-o-deethylase and acetanilide-4-hydroxylase, respectively. Microsomes were prepared by the CaCl2 aggregation method (Schenkman and Cinti 1978) and microsome protein concentrations were determined by the Lowry method (Lowry et al. 1951). Spectroflourometric methods were used to determine CYP1A1 concentrations in the liver (Waxman and Chang 1998) and lung (SOP SRI 70-34), while CYP1A2 was determined by an acetanilide-4-hydroxylase assay using an HPLC method (Hamm et al. 1998). Statistical analyses were performed using Microsoft Excel and VassarStats software.
Results
Pathology
Subchronic study (3 month)
No significant treatment-related effects were observed in either mice or rats with respect to body weight, survival, hematology, or gross lesions (data not presented). While there were statistically significant increases in relative liver weights in both mice and rats (Figure 1), no corresponding histopathologic lesion could be determined. In mice, no treatment-related histopathologic changes were noted in any of the dosage groups.
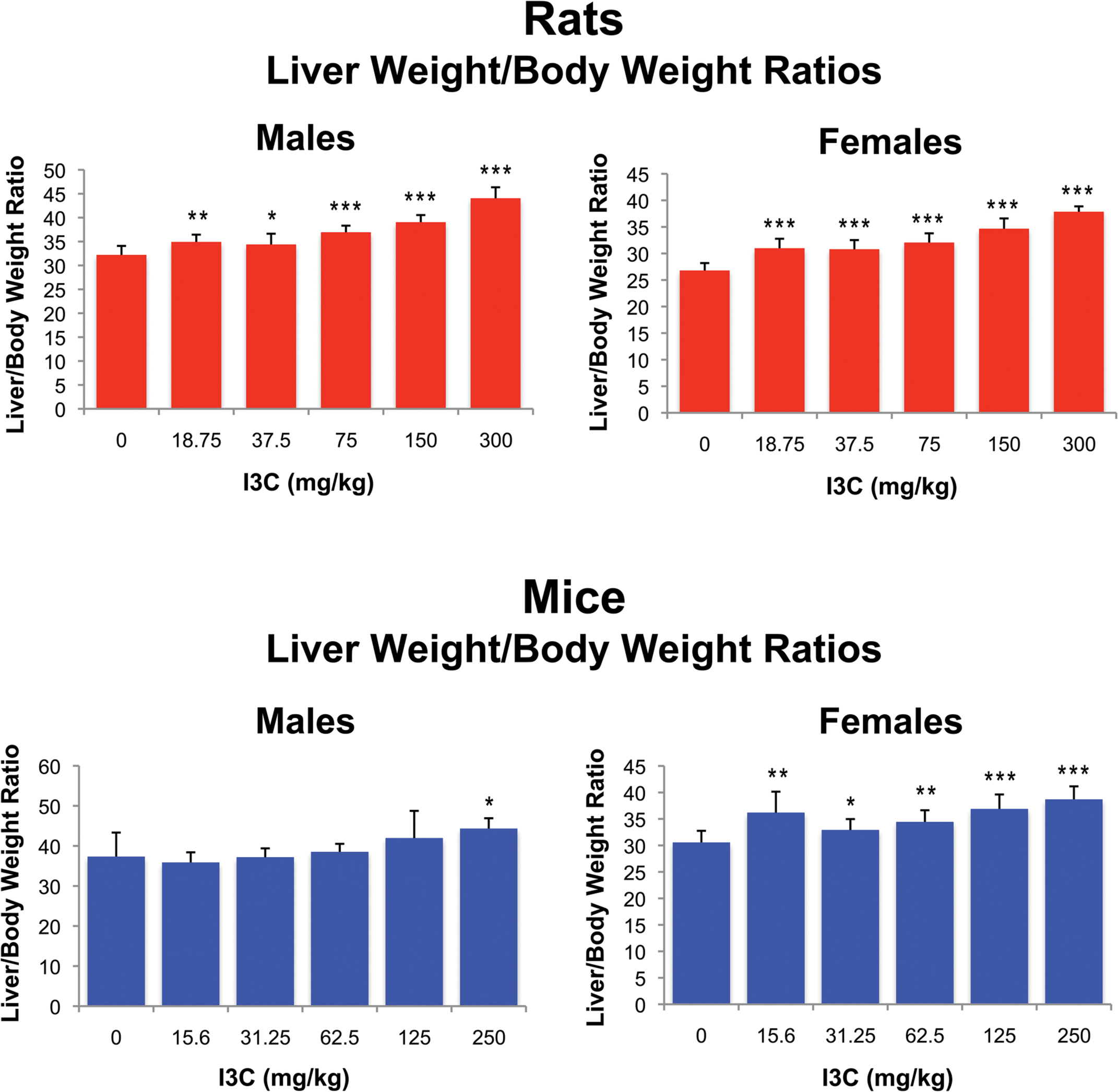
Liver weight to body weight ratios in F344 rats and B6C3F1 mice exposed to indole-3-carbinol via oral gavage. Significantly different than controls at *p < .05; **p < .001; ***p < .0001.
Rats exhibited dose-related lesions in the duodenum, jejunum, and mesenteric lymph nodes (Table 1). The intestinal lesions in rats consisted of a dose-related increase in incidence and severity of dilatation (ectasia) of lymphatic vessels in the villi (lacteals) of both the duodenum and jejunum (Figures 2a and 2b). In the duodenum, lesions were present in males treated with 75 mg/kg, and both males and females treated with 150 and 300 mg/kg I3C. Jejunal lesions were present in both males and females treated with ≥37.5 mg/kg, as well as in males treated with 18.75 mg/kg. No lesions were noted in any of the control rats. Affected lacteals were variably dilated (lymphangiectasis), lined by endothelial cells, and occasionally contained a minimal amount of lacy flocculent eosinophilic to amphophilic material. No inflammatory cells were present within the ectatic lacteals. Large macrophages with abundant vacuolated cytoplasm (resembling lipid) were occasionally present within the stroma of the affected villi adjacent the ectatic lymphatics (lipidosis).
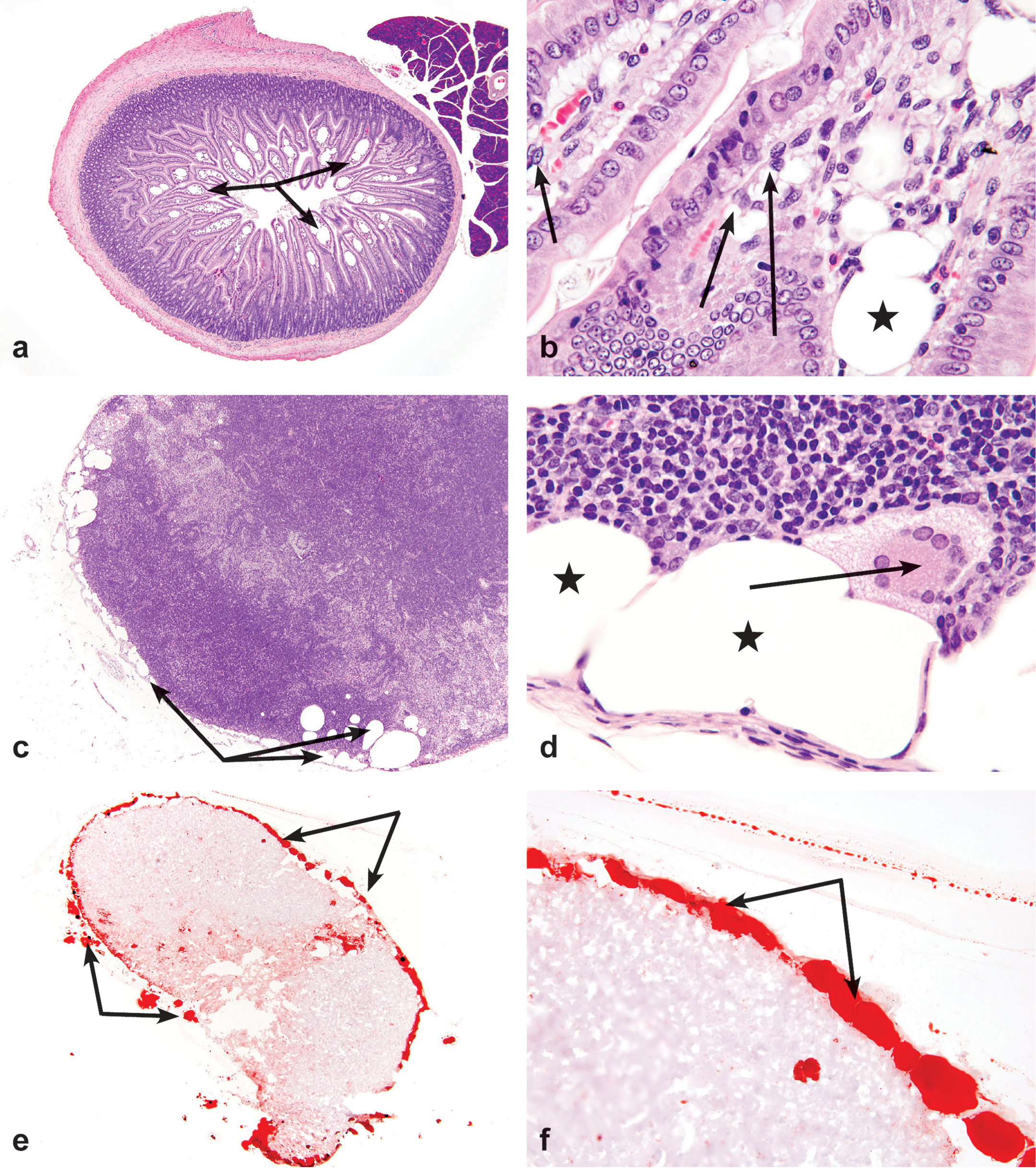
Representative photomicrographs of lesions in the small intestine and mesenteric lymph nodes of rats exposed to a 3-month treatment of 300 mg/kg indole-3-carbinol via oral gavage. (a) Duodenum; moderate ectasia of villous lacteals (lymphangiectasis) (arrows) Original objective 4X, H&E. (b) Duodenum; lymphangiectasis (asterisk). Note vacuolated macrophages within the lamina propria (arrows). Original objective 40X, H&E. (c) Mesenteric lymph node; moderate lymphangiectasis of the subcapsular sinus (arrows). Original objective 4X, H&E. (d) Mesenteric lymph node; lymphangiectasis of the subcapsular sinus (asterisks). Note vacuolated multinucleated giant cells. Original objective 40X, H&E). (e and f) Mesenteric lymph node; large aggregates and pools of red staining material within subcapsular sinuses (arrows) are consistent with excessive lipid accumulation (lipidosis). Note grade 4 lymphangiectasis. Original objectives 2X (e) and 40X (f), Oil Red O.
Incidence and severity of lymphangiectasis in a 3-month toxicity study of Indole-3-carbinol in male and female Fischer 344 rats.
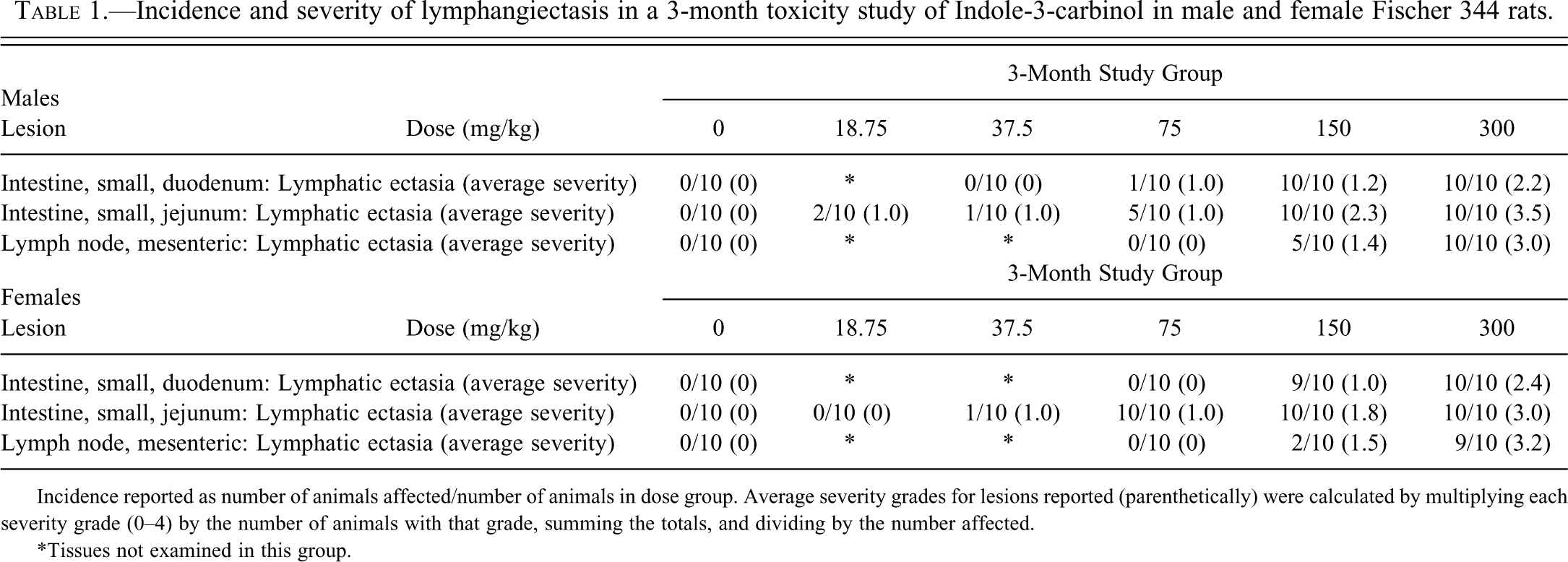
Incidence reported as number of animals affected/number of animals in dose group. Average severity grades for lesions reported (parenthetically) were calculated by multiplying each severity grade (0–4) by the number of animals with that grade, summing the totals, and dividing by the number affected.
*Tissues not examined in this group.
The mesenteric lymph nodes exhibited a similar dose-related effect, affecting male and female rats treated with 150 and 300 mg/kg I3C, as well as males treated with 18.75 and 37.5 mg/kg. Lymphangiectasis was not observed in either males or females dosed with 75.0 mg/kg. Lesions were characterized by predominately subcapsular dilatation of lymphatic vessels (Figure 2c). Occasionally cortical and paracortical or infrequently medullary lymphatic vessel dilatation was also present. Medullary sinuses often possessed increased numbers of large foamy macrophages with vacuolated cytoplasm in addition to occasional multinucleated giant cells (Figure 2d). Lesions of moderate to marked severity often disrupted nodal architecture.
Clinical chemistry parameters were not evaluated in mice. No significant abnormalities were noted in serum proteins in treated rats. Liver enzyme (SDH, ALP, ALT) alterations were noted in treated male and female rats, which were determined to be of little toxicologic significance. Statistically significant decreases in ALT levels were observed in all female rats compared to controls.
Histochemical Analysis for Lipid
Positive staining in the mesenteric lymph node for Oil Red O was characterized by pinpoint to large globular accumulations of red staining material. A diagnosis of “Present” was used only when both of the following criteria were met: (1) lymphangiectasis was present on the slide, and (2) positively stained material was present within the ectatic areas.
1- and 4-week interim sacrifice of the 2-year study
No significant gross lesions were noted in any dose group. Histopathological evaluations were confined to sections of proximal duodenum and jejunum (Table 2). A dose- and time-related increase in incidence and severity of lymphangiectasis was demonstrated, in which lesions were present in 150 and 300 mg/kg dose groups. There was also an increase in incidence and/or severity of lymphangiectasis in the 4-week exposure groups as compared to the 1-week exposure groups. Lymphangiectasis was consistently of greater incidence and severity in the sections of jejunum versus that of the duodenum. Lesions were morphologically similar to those previously described in rats from the 3-month study. There was no evidence of lymphangiectasis in any of the control or 75 mg/kg dosed rats at either sacrifice point.
Incidence and severity of lymphangiectasis in 1- and 4-week interim sacrifices in a 2-year carcinogenicity study of Indole-3-carbinol in male Harlan Sprague-Dawley rats.
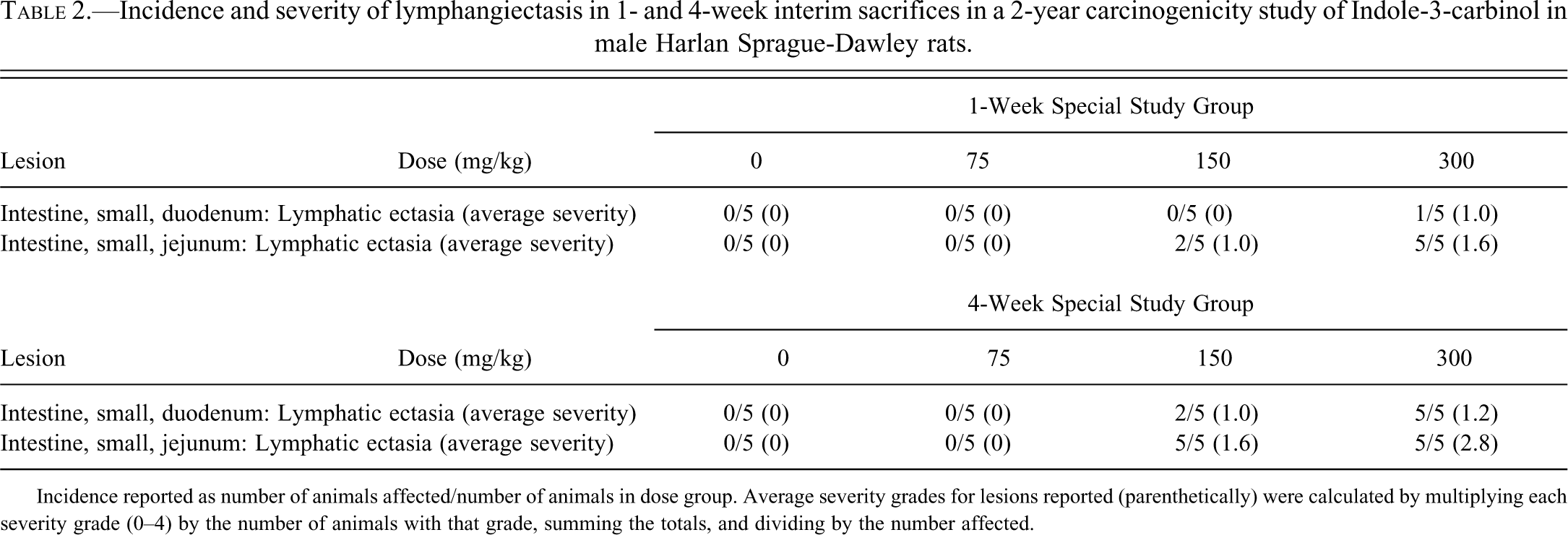
Incidence reported as number of animals affected/number of animals in dose group. Average severity grades for lesions reported (parenthetically) were calculated by multiplying each severity grade (0–4) by the number of animals with that grade, summing the totals, and dividing by the number affected.
Histochemical Analysis for Lipid
Positive staining in the duodenum and/or jejunum for Oil Red O and Sudan Black was characterized by pinpoint to large globular accumulations of red or black staining material, respectively. Occasionally, large amounts of staining artifact, such as stray pools and scattered droplets of positively stained material, were common with both staining procedures, precluding an accurate assessment of severity in those cases. A diagnosis of “Present” was used only when both of the following criteria were met: (1) lymphangiectasis was present on the slide, and (2) positively stained material was present within the ectatic areas.
All sections with histologic evidence of lymphangiectasis had positively stained material within their dilated lacteals. In all jejunal sections where lymphangiectasis was present, a positive reaction for lipid was also observed by both staining modalities (Figure 3). In 8 of the 9 animals, scattered duodenal lacteals with minimal accumulations of material stained positively for one or both stains, though none of those samples exhibited evidence of lymphangiectasis. Thus, no duodenal sample met the above two criteria, despite that fact that 3 of these animals had been previously diagnosed on H&E with minimal to mild lymphangiectasis. One of those 3 animals exhibited no accumulation of positively stained material. Positive histochemical staining with Oil Red O demonstrated lipid within the dilated subcapsular sinuses (Figures 2e and 2f).
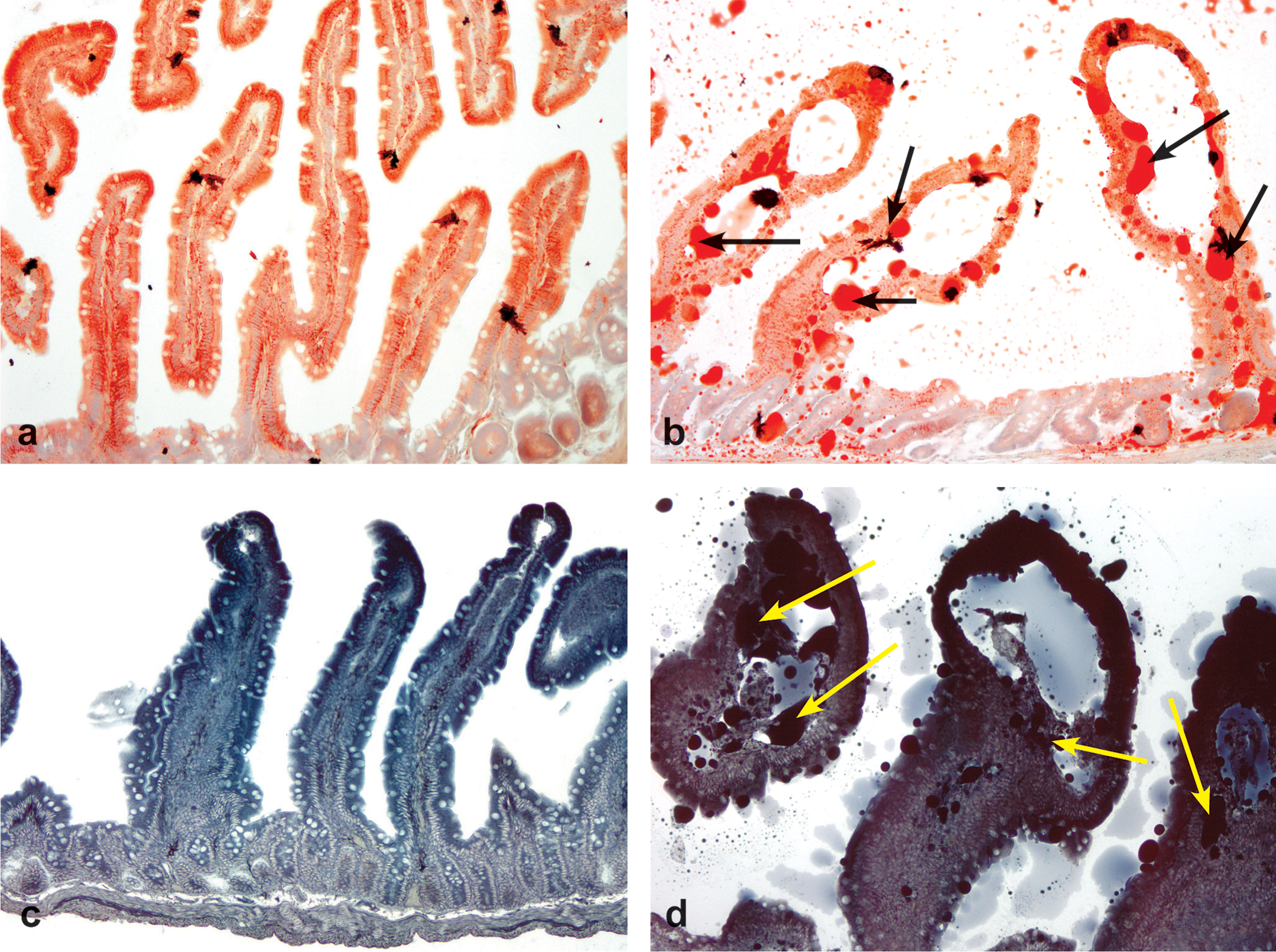
(a) Jejunum from a male control rat. Fine red stippling along mucosal epithelium and lamina propria is indicative of normal lipid accumulation associated with fat absorption. Note lack of lymphangiectasis. Original objective 10X, Oil Red O. (b) Jejunum from a male rat exposed to a 4-week treatment of 300 mg/kg indole-3-carbinol (I3C) via oral gavage. Large aggregates and pools of red staining material (arrows) are consistent with excessive lipid accumulation (lipidosis). Note grade 4 lymphangiectasis. Original objective 10X, Oil Red O. (c) Jejunum from a male control rat. Note lack of lymphangiectasis. Original objective 10X, Sudan Black. (d) Jejunum from a male rat exposed to a 4-week treatment of 300 mg/kg I3C via oral gavage. Large aggregates and pools of black staining material (arrows) are consistent with excessive lipid accumulation (lipidosis). Note grade 4 lymphangiectasis. Original objective 10X, Sudan Black.
No apparent differences were noted between the Oil Red O and Sudan Black staining in any of the treatment groups or time points. Additionally, there were no differences in lipid staining characteristics between the 1- and 4-week exposure times. Sections of mesenteric adipose tissue frequently adjacent to the sections of both duodenum and jejunum were invariably positive with both stains and served as positive internal controls.
Electron microscopy
Results of electron microscopy of 9 male rats from the 1-week and 4-week interim sacrifice study are listed in Figure 4. In control animals, the jejunal lamina propria was within normal limits and a variety of cells types were present, including macrophages, eosinophils, fibroblasts, endothelial cells, and mast cells. A single lacteal was present in one of the sections (Figure 4a).
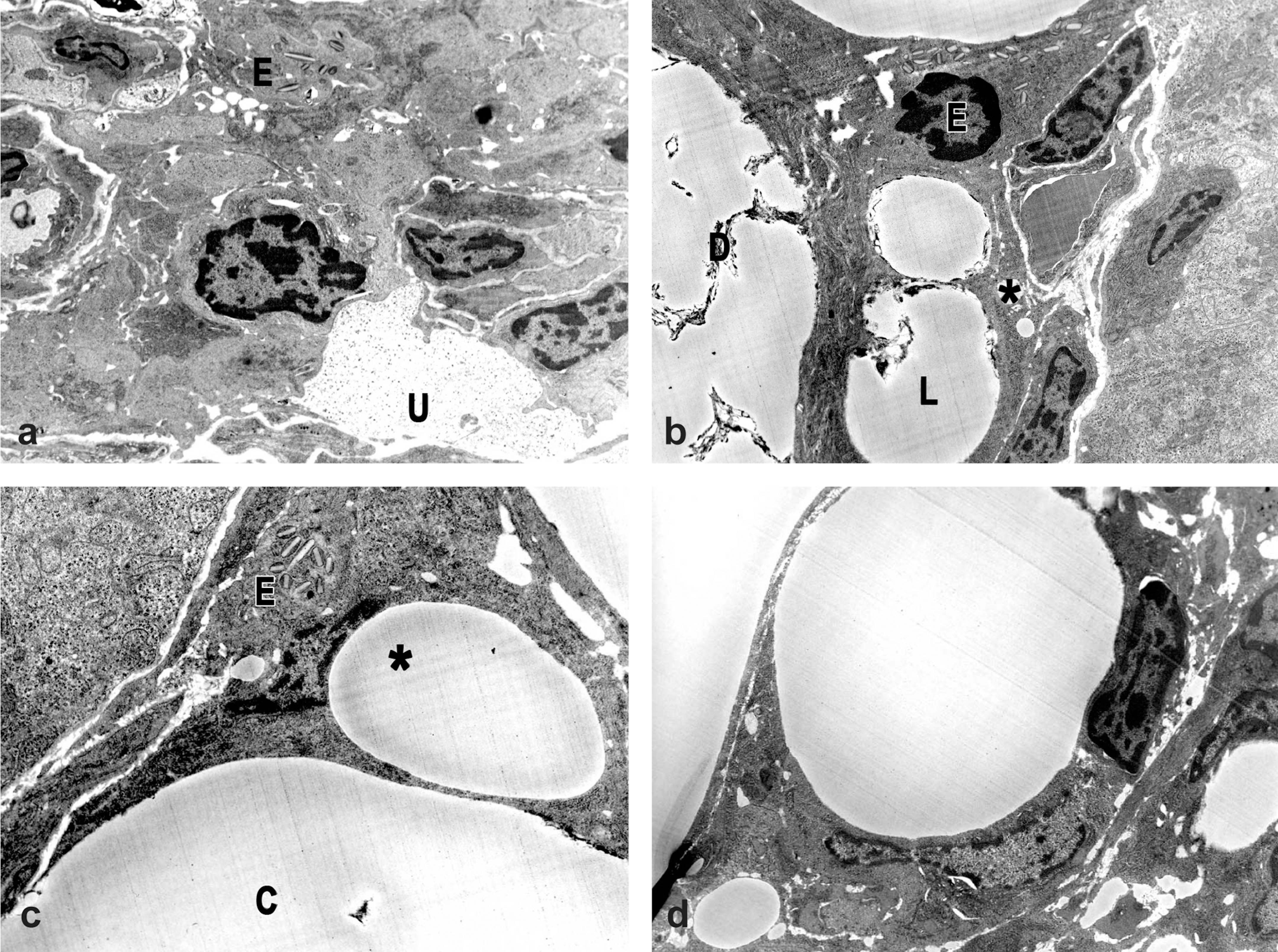
Electron microscopy. (a) Jejunum from a control male rat. Note mucus droplet (U) and eosinophil (E). 10,800X. (b) Jejunum from a male rat exposed to a 1-week treatment of 300 mg/kg indole-3-carbinol (I3C) via oral gavage. Note large extracellular lipid droplets (L), small intracellular lipid droplets (*), membranous debris (D), and eosinophil (E). 10,800X. (c) Jejunum from a male rat exposed to a 1-week treatment of 300 mg/kg I3C via oral gavage. Large droplets appear to be within lacteals, while smaller droplets appear to be intracellular. Note eosinophil (E), lipid in lacteal (C), and smaller droplet indenting the endothelial nucleus (*). 10,800X. (d) Jejunum from a male rat exposed to a 4-week treatment of 300 mg/kg I3C via oral gavage. Note presence of one large and several smaller lipid droplets that appear to be intracellular within macrophages. 10,800X.
After 1 week of treatment with 300 mg/kg I3C, lipid droplets were present within both the lacteals and the lamina propria (Figures 4b and 4c), similar to that previously reported as intestinal lipidosis by Visscher, Robison, and Hartman (1980). Larger droplets appeared to be located extracellularly and were often associated with membranous debris (lipidosis). Smaller droplets appeared to be within the cytosol, although the affected cell type could not be definitively determined.
The electron microscopic appearance of the jejunum was similar at the 1-week and 4-week exposure times at 300 mg/kg I3C: large lipid droplets accumulated in the lacteals and lamina propria and appeared to be within the extracellular matrix, and often had adjacent membranous debris (Figure 4b). At the 4-week exposure it was possible to identify macrophages as the cell type in which the smaller lipid droplets were present based on correlation to light microscopy findings.
Cytochrome P450 (CYP) Enzyme Analysis
In rats, hepatic CYP1A1 activity was dose-dependently increased in males and females with a maximal induction of 109- and 81-fold, respectively, at 300 mg/kg (Figure 5). Compared to controls, there was statistically significant enzyme induction in all dose groups except 18.75 mg/kg. Hepatic CYP1A2 activity was increased in a dose-dependent manner with a maximal induction of approximately 5-fold in both males and females. Compared to controls, enzyme induction was statistically significant in all dose groups of female rats and in all dose groups of male rats except 18.75 mg/kg.
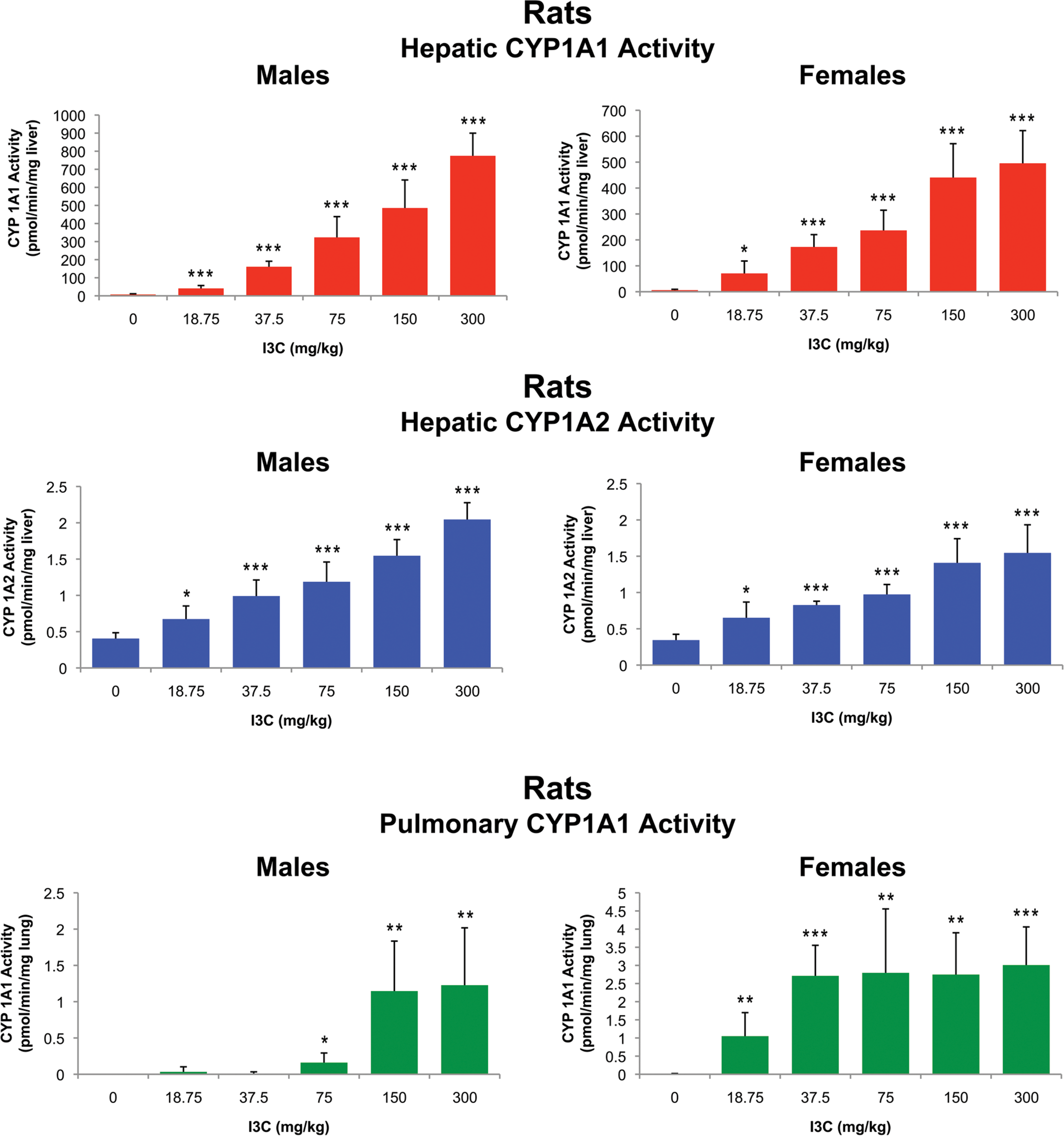
Hepatic cytochrome P450 (CYP) 1A1 and 1A2, and pulmonary CYP1A1 activities of Fisher 344 rats exposed to indole-3-carbinol via oral gavage. Significantly different from controls at *p < .05; **p < .001; ***p < .0001.
In mice, hepatic CYP1A1 and 1A2 activities were maximally increased 2- to 4-fold (Figure 6). Compared to controls, statistically significant increases in enzyme induction were noted in males and females at the 125 and 250 mg/kg dose groups and males in the 62.5 mg/kg dose group.
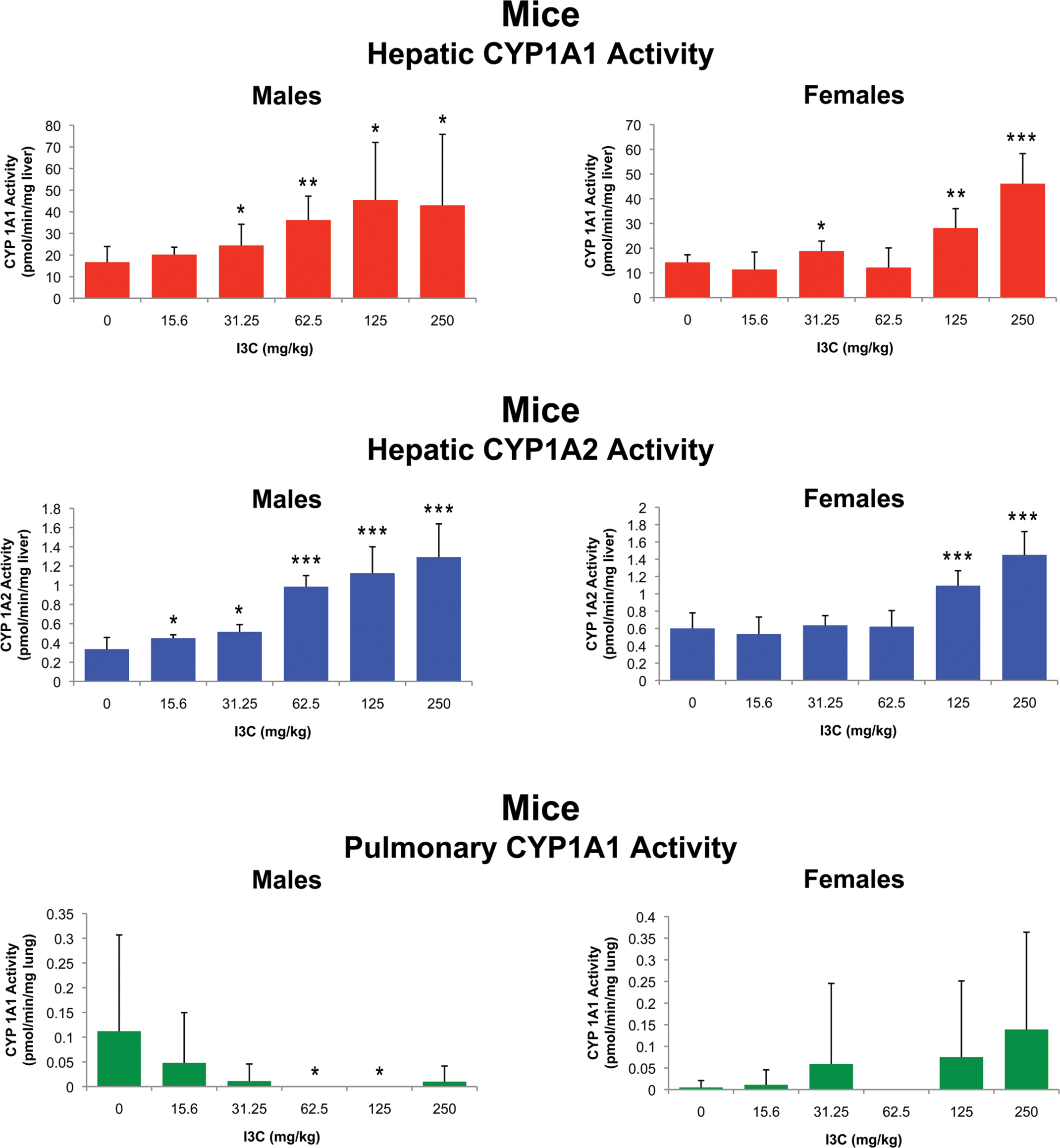
Hepatic cytochrome P450 (CYP) 1A1 and 1A2, and pulmonary CYP1A1 activities of B6C3F1 mice exposed to indole-3-carbinol via oral gavage. Significantly different from controls at *p < .05; **p < .001; ***p < .0001.
In rats, dose-dependent increases of pulmonary CYP1A1 activity were noted in both sexes (Figure 5), although the changes were much smaller than those observed in the liver. In the control animals, lung CYP1A1 activity was undetectable in all male rats and 2/10 female rats. In males, there was a maximal enzyme induction approximately 37-fold higher than the lowest dose group. Activity for female rats in the highest dose group was 501-fold higher than the controls, and 3-fold higher than the lowest dose group. Compared to controls, enzyme induction was statistically significant for male rats in the two highest dose groups (100 and 300 mg/kg) and for females at all doses except the lowest dose group (18.75mg/kg).
In mice, no statistically significant increases in pulmonary CYP1A1 enzyme activity were observed in either sex (Figure 6). Statistically significant decreases in pulmonary CYP1A1 enzymes were noted in the 62.5 and 125 mg/kg dose groups. However, due to a lack of significant decrease in the 250 mg/kg group, and therefore no clear dose-response relationship, these changes were determined to be of no biological significance.
Discussion
Treatment-related histopathological findings due to exposure to indole-3-carbinol (I3C) via oral gavage were limited to the rats, and consisted of lymphangiectasis and lipidosis in the duodenum, jejunum, and mesenteric lymph nodes. Lesions consisted of minimal to marked dilatation of the duodenal and jejunal villous lacteals, in addition to the subcapsular lymphatics of the mesenteric lymph nodes. Additionally, the subcapsular sinuses and cortical, paracortical, and medullary lymphatics of those lymph nodes were occasionally affected. Positive histochemical staining with Oil Red O and/or Sudan Black confirmed that the material in these dilated lacteals was lipid. In 8 of the 9 special study animals, scattered duodenal lacteals stained positively for one or both stains, though none of those samples exhibited evidence of lymphangiectasis. This finding highlights the variability of comparing formalin-fixed paraffin-embedded hematoxylin and eosin stained sections results with those from snap frozen Oil Red O stained sections, demonstrating the impact of sample handling on morphology. Occasionally, lipid-laden macrophages and multinucleated giant cells were also present within the stroma of affected villi and within dilated lymphatics and stroma of mesenteric lymph nodes. Electron microscopy revealed that while the smaller lipid droplets were intracellular within the intestinal epithelial cells, larger droplets were present extracellularly, both within the lamina propria and within the lacteals. These larger droplets were sometimes accompanied by nearby membranous debris. In some instances, lipid droplets were clearly identified within macrophages.
Lesions exhibited dose-related increases in incidence and severity. Additionally, increases in incidence and/or severity were also noted in animals with a 4-week exposure to I3C compared to those with a 1-week exposure. In both 1- and 4-week exposure groups, lesions were more common and more severe in the jejunum than the duodenum. The highest dose group without any observable effects was 75 mg/kg for both the 1- and 4-week exposures groups.
During the process of normal lipid absorption, resynthesis, and transport, the upper third of the intestinal villus is the most active; thus, small amounts of lipid within this region are considered normal (Visscher, Robison, and Hartman 1980). In our current study, all animals in all dose groups exhibited a fine stippling pattern within this region when histochemical stains for lipid (Oil Red O and Sudan Black) were applied. This stippling pattern corresponded electron microscopically with the presence of small intracellular lipid droplets.
For a more detailed description of the biochemistry of lipid transport, the authors refer to the review by Friedman and Nylund (1980). Briefly, the lipid digestion, absorption, and transport process begins following consumption of a meal. Cholecystokinin release is stimulated by the transit of digesta through the proximal duodenum, particularly fatty acids, proteins, and hydrogen ions (Cummings and Overduin 2007; Friedman and Nylund 1980; Zerbe and Washabau 2000). Among its myriad physiologic effects, cholecystokinin stimulates the release of pancreatic lipase, cholesterol esterase, and phospholipase A2 (Bruss 2008; Zerbe and Washabau 2000). Dietary lipids partially digested by gastric lipase are further digested by pancreatic lipase and emulsified and incorporated into micelles composed of bile acids, monoglycerides, free fatty acids, phospholipids, cholesterol, and fat-soluble vitamins (Bruss 2008; Evans and Duncan 2003; Stockham and Scott 2002). The enzymatic attack on the surface of the micelle results in the release of fatty acids, mono- and diglycerides, and cholesterol (Bauer 2000; Bruss 2008). These products become incorporated within the brush border of duodenal villous epithelial cells (enterocytes) (Bruss 2008; Evan and Duncan 2003; Stockham and Scott 2002). During what is commonly called “triglyceride resynthesis,” the enterocytes resynthesize triglycerides, primarily triacylglycerol, from monoglycerides and fatty acids (Bruss 2008; Stockham and Scott 2002). Enterocytes assemble triglycerides, apolipoproteins, phospholipids, and cholesterol esters into large, tryglyceride-rich lipoproteins called “chylomicra” (Stockham and Scott 2002; Evans and Duncan 2003). These chylomicra are assembled within the endoplasmic reticulum of the enterocyte, packaged in the Golgi, and extruded through the basolateral membrane into the villar lamina propria (Bruss 2008; Friedman and Nylund 1980). Chylomicra then move through the lamina propria to the central lacteal. Smaller lacteals often fused, accounting for the progressively larger areas of positive Oil Red O and/or Sudan Black staining noted in our study toward the lacteals and the mucosal base. The pathway of normal lipid absorption and transport as described above is compared to that found after treatment with I3C.
It is speculated that in the current study there is impairment in the process of lipid transport through the lacteals. The processes of lipid uptake and delivery to the lamina propria appear to be occurring appropriately. However, accumulation within the lacteals and the extracellular lamina propria results in lymphatic telangiectasia, or lymphangiectasis, and extracellular and extralymphatic lipid accumulation, or lipidosis, respectively.
Chemically induced intestinal lipidosis has previously been reported in rats. Indeed, the process of lipid absorption through the small intestinal lymphatics was first observed in rats by Henriques and Hansen in 1900 (Friedman and Nylund 1980). Experimental administration of puromycin (Friedman and Cardell 1972) and ethionine (Hyams et al. 1966) resulted in abundant lipid accumulation within intestinal epithelial cells. Similarly, oral administration of 2,6-di-tert-butylamino-3-acetyl-4-methylpyridine (SaH 51-055), a glucose transport inhibitor with antiobesity indications, resulted in the accumulation of lipid droplets within intestinal epithelial cells (duodenum > jejunum), in addition to the intercellular spaces and lamina propria of the small intestinal villi (Visscher, Robison, and Hartman 1980). In this instance, no structural changes were noted in the epithelial cells or lacteals. Visscher, Robison, and Hartman (1980) proposed that the phenomenon was related to abnormalities in the triglyceride resynthesis process or chylomicron synthesis, resulting ultimately in the formation of abnormal chylomicra that coalesced and were subsequently phagocytosed. Alteration in any component of chylomicra formation (lipid, protein, and/or carbohydrate) could result in the synthesis of abnormal chylomicra. As SaH 51-055 is a glucose transport inhibitor, it is plausible that the sugar moiety of the chylomicra could have been altered. An alternative theory is that abnormal protein synthesis within the intestinal epithelial cells could have resulted in an inadequate protein coating of the chylomicra resulting in their coalescence within the intercellular spaces. A study conducted by Koo and Turk (1977) demonstrated the importance of chylomicron protein coating. In their study, zinc-deficient rats had impaired protein synthesis resulting in lipid coalescence within the Golgi apparatus. Additional alternative theories involve chylomicron assembly: inhibition of long chain fatty acid breakdown and/or assembly, or inhibition of esterification leading to defective assembly. Fatty acids such as linoleic acid don’t require esterification for transport, but cholesterol reesterification is critical for these processes (Lubbers et al. 2011; Friedman and Nylund 1980).
Lymphangiectasis in veterinary species is defined as dilatation of normally developed lymphatic vessels (Maxie and Robinson 2007; Soto et al. 2010). This abnormality is most commonly diagnosed as intestinal lymphangiectasia, commonly attributed as the cause of protein-losing enteropathy in canines (Brown, Baker, and Barker 2007). Affected animals present similarly to human patients, with diarrhea and weight loss, and variable hypoproteinemia, hypocholesterolemia, lymphopenia, and hypocalcemia (Peterson and Willard 2003; Kull et al. 2001; Brown, Baker, and Barker 2007). In companion animals, where significant hypoalbuminemia may not be detected, intestinal lymphangiectasia is confirmed or discovered through histopathologic evaluation of endoscopic biopsies (Peterson and Willard 2003).
Intestinal lymphangiectasia in humans is defined as a disorder of lymphatics in the mucosa and submucosa of the small intestine, characterized by marked telangiectasis of the lymphatic vessels (Shapiro 1971). Intestinal lymphangiectasia is subdivided into primary intestinal lymphangiectasia (PIL) and secondary intestinal lymphangiectasia (Makhija et al. 2004). PIL, also known as Waldmann's disease, is a rare disease in humans characterized by dilated lymphatics in the duodenum, jejunum, or mesentery (Buohnik et al. 2000; Koo et al. 2005). It is congenital or idiopathic, and has been linked to several syndromes including Milroy’s, Von Recklinghausen, Turner, Klippel Trenaunay, Hennekam, and Noonan’s (Keberle et al. 2000; Suresh et al. 2009). An immunohistochemical study by Hokari et al. (2008) identified several growth factors and receptors significant in lymphangiogenesis and lymphangiectasia, namely VEGF-C and -D, FOXC2, SOX18, Prox 1, and VEGFR3 and LYVE-1, respectively, the latter two of which were specific and sensitive markers for lymphatics. Macdonald et al. (2010) recently found an association between PIL and severity of small intestinal angiodysplasia. The structure of lymphatics in the lacteals, with their discontinuous basal lamina and thin walls, do not lend themselves to competency under duress of dysplastic change (Dobbins 1966, Macdonald et al. 2010).
PIL primarily affects children, and is commonly diagnosed at 2–3 years of age (Mazzie et al. 2003; Freeman and Nimmo 2011). Clinically, these patients may manifest with severe, frequently asymmetric edema of the lower extremities, ascities, chylous pleural effusions, steatorrhea, hypoproteinemia, hypoalbuminemia, hypogammaglobulinemia, trace metal deficiency, elevated fecal α-1 antitrypsin, and/or lymphocytopenia (Shapiro 1971; Hokari et al. 2008; Kim et al. 2009; Mazzie et al. 2003; Braamskamp, Dolman, and Tabbers 2010). Studies have also shown that individuals with endoscopically or histologically discovered duodenal lymphangiectasia may be clinically asymptomatic (Kim et al. 2009).
Though the cause of PIL may not be known, patients have responded to supplementation with medium chain triglycerides and administration of somatostatin analogues such as octreotide (Filik et al. 2004; Makhija et al. 2004). Although no definite mechanism for the purported efficacy of the latter has been determined, several have postulated alterations in the autonomic nervous system and thus contraction of lymphatic vasculature, a normal rhythmic process which propels lymph from the apical villar to deep mucosal lymphatics, may be responsible for the response to octreotide (Sari, Baris, and Dalgic 2009). However, the experiments of Makhija et al. dispute these theories (Makhija et al. 2004; Benoit et al. 1989; von der Weid 2001).
Secondary intestinal lymphangiectasia has myriad etiologies including abdominal or retroperitoneal neoplasia, Tropheryma whipplei infection (Whipple’s disease), chronic congestive heart failure, constrictive pericarditis, retroperitoneal fibrosis, abnormal intestinal rotation, systemic lupus erythematosus, iatrogenic lymphatic vascular trauma during surgery, and enteric infections including mesenteric nodal tuberculosis (Makhija et al. 2004; Salvia et al. 2001; Mazzie et al. 2003; Freeman and Nimmo 2011).
No toxic or neoplastic histologic lesions were noted in the liver of any of these animals despite statistically significant increases in liver weights, which was predominantly seen in female rats and mice. Interestingly, the findings reported by Crowell et al. (2006) demonstrate histologic lesions at a lower maximum dose than was given in the current studies. One possibility for this discrepancy is the use of different rat strains; Virus-Antibody Free (VAF) and non-VAF CD rats were used in the Crowell et al. studies; the current studies employed Fischer 344 and Harlan Sprague-Dawley rats.
Decreased ALT was observed in female F344 rats treated with I3C compared to concurrent controls. While increased serum ALT is a commonly-utilized surrogate of hepatocellular damage, decreased ALT implicates decreased synthesis (Solter, Liu, and Guzman 2000). Decreased ALT levels might has been attributed to starvation, malnutrition, or malabsorption, however, body weights and total protein levels were not significantly different between groups in this study. Waner and Nyska (1991) illustrate various causes for decreased ALT in laboratory species, such as loss of the fat-soluble vitamin B6. These possibilities were not pursued in this study. As such, the biological significance of this decrease remains unknown.
The magnitude of CYP1A1 induction (in the ng/kg range) was equivalent to that observed in the 2-year carcinogenicity study of TCDD in which both neoplastic and nonneoplastic lesions had been induced (NTP 1982a, 1982b; NTP 2006a, 2006b, 2006c, 2006d, 2006e, 2006f, 2006g). Therein lies a point of great interest: quite unlike the observations in the dioxin-like compound (DLC) studies, the potent CYP inducer I3C caused no discernable lesions in the liver or thyroid. Kim et al. showed I3C acts as a promoter, increasing incidences of liver and thyroid tumors in rats post-initiation (Kim et al. 1997). Studies show similar CYP induction between DLCs and I3C, yet neither lymphangiectasis nor intestinal lipidosis was observed in the DLC studies (NTP 1982a, 1982b; NTP 2006a, 2006b, 2006c, 2006d, 2006e, 2006f, 2006g). These data suggest a metabolite specific to I3C, not observed in testing the DLCs, is responsible for the unique intestinal lesions observed in the I3C studies. Due to the presence of the unique intestinal lesion, and the lack of lesions expected from this potent CYP inducer, further studies are required to elucidate the specific metabolite(s) and mechanisms responsible for the lesions observed. This information is necessary for thorough assessment of the possible spectrum of effects of I3C. The histopathologic changes of intestinal lymphangiectasis observed in this study share morphologic similarities with intestinal lymphangiectasia as observed in humans and dogs. However, the resultant clinical spectrum of protein-losing enteropathy (hypoalbuminemia, hypocholesterolemia, lymphocytopenia, hypogammaglobulinemia, edema, etc.) was not present. Additionally, the intestinal lipidosis observed in rats (extracellular lipid accumulation demonstrated by electron microscopy) is not described as a feature of the various lymphangiectatic syndromes in humans.
Footnotes
Acknowledgments
We thank Drs. June Dunnick and Arun Pandiri of the National Toxicology Program for their thorough review of this article and Beth Mahler and Eli Ney for extensive assistance with figures.
