Abstract
Studies of cancer chemoprevention with caffeic acid phenethyl ester (CAPE) in the resistant hepatocyte model of hepatocarcinogenesis have shown the participation of CYP drug metabolizing enzymes. To prevent neoplastic and preneoplasic lesions, we must specifically identify which CYP activities are modified in the mechanism of action of CAPE. Male Fischer-344 rats were pretreated with CAPE twelve hours before administration of diethylnitrosamine (DEN) and were sacrificed twelve hours after CAPE and twelve hours, twenty-four hours, twenty-four days, and twelve months after DEN. Other rats were treated with the CYP inhibitors α-naphthoflavone or SKF525A and sacrificed twenty-four hours and twenty-four days after DEN. Microsomes were obtained from livers to quantify protein using Western blot. Diethylnitrosamine metabolism was measured based on nitrite formation and liver histology using GGT histochemistry. Caffeic acid phenethyl ester diminished the protein levels of CYP1A2 and CYP2B1/2. The inhibition of CYP2B1/2 prevented the appearance of preneoplastic lesions. Microsomal assays demonstrated that CAPE interfered with DEN activation diminishing nitrites similar to SKF525A and probably mediated by CYP2B1/2 inhibition. A single dose of CAPE before DEN treatment reduced the appearance of tumors by 43%. These results confirmed that CAPE is a promising agent to confer chemoprotection in liver cancer and should be considered for human therapies.
Introduction
Millions of people have been treated for cancer. For many of these people, chemoprevention using natural or synthetic compounds remains the best option to prevent, retard, or revert carcinogenesis (Weinstein 1991). Liver tumors are one of the most malignant tumors. Liver cancer has an acceptable prognosis if it is detected in the early stages, whereas a late diagnosis foretells a bad prognosis (Parikh and Hyman 2007). Different compounds confer chemoprotection against liver cancer and include anti-inflammatory drugs and other compounds that have been identified from the extraction of natural products such as vegetables, seeds, and propolis. One important component from propolis is caffeic acid phenethyl ester (CAPE), which acts as a chemoprotective agent in the modified resistant hepatocyte model in different stages of disease progression with pleiotropic effects (Beltran-Ramirez et al. 2008; Carrasco-Legleu et al. 2004; Carrasco-Legleu et al. 2006; Garcia-Roman et al. 2008).
In previous work, we showed that CAPE modulated CYP activity that could be related with diethyl nitrosamine (DEN) metabolism to initiate carcinogenic activities in the resistant hepatocyte model. CAPE treatment decreased CYP1A1/2 and CYP2B1/2 mRNA expression and enzymatic activities. Simultaneously, there was a reduction of induced γ-glutamyl transpeptidase–positive (GGT+) preneoplastic lesions (Beltran-Ramirez et al. 2008; Beltran-Ramirez et al. 2010). In this model, a different mechanism for CAPE has been proposed. The inhibition of DEN activation by CYP is a well-studied phenomenon, although it is unknown whether the final effect is a result of the chemoprotective agent modifying CYP enzymes. Other natural chemoprotective agents such as garlic powder (Park et al. 2002), red pepper dihydrocapsaicin, and some components of Brussels sprouts (Koop 1992) modify CYP activities. The mechanism of action of these agents is to modify carcinogen metabolism and thus prevent the damage induced by them.
If the mechanism of action of CAPE is mediated by alterations in CYP enzymatic activities, it is important to know which of the CYP enzymes are modified. The aim of the current work was to clarify the relationship of CYP 450 with CAPE or the carcinogenic process to confirm its mechanism of action, which was related to DEN activation. Once the CAPE mechanism is well defined, we can extend the results of the current study to humans and use CAPE to develop novel therapeutics in cancer prevention.
Materials and Methods
Animals
Male Fischer-344 rats (180–200 g) were obtained from the Unit for Production of Experimental Laboratory Animals (UPEAL, Cinvestav-IPN, Mexico City, Mexico). Animals had free access to food (Formulab Diet 5008, PMI Nutrition International, St. Louis, MO, USA) and water at all times. Each rat consumed approximately 12–15 g of food and 10–15 mL of water per day. After treatment, the animals were transferred to the holding room and kept under controlled conditions of twelve hours light/twelve hours dark cycles, and 50% relative humidity at 21°C. Animal care followed institutional guidelines for the use of laboratory animals.
Animal Treatment
Animals were subjected to different treatments to test the chemoprevention of tumor appearance by CAPE or specific CYP inhibitors and to ascertain changes in protein levels and CYP-mediated activity using microsomal assays. Male Fischer-344 rats were treated according to the modified resistant hepatocyte model of Semple-Robert (Carrasco-Legleu et al. 2006). Animals were administered with one ip dose of 200 mg/kg of DEN at day 0, then, on days 7, 8, and 9 with one daily dose by gavage of 2-acethylaminofluorene, and on day 10, each animal underwent a partial hepatectomy. Some rats were pretreated with one ip dose of 20 mg/kg of CAPE twelve hours before administration of DEN and were sacrificed twelve hours after CAPE and twelve hours, twenty-four hours, twenty-four days, and twelve months after DEN.
Chemopreventive Effect by CAPE on Tumor Appearance
Animals were treated according to the modified resistant hepatocyte model of Semple-Robert (Carrasco-Legleu et al. 2006) with or without CAPE pretreatment, (n = 7 per group) with and without CAPE, 99% pure, obtained according to Grunberger et al. (1988). Both groups were sacrificed twelve months after the carcinogenic treatment. The liver was excised, washed in saline solution, frozen in 2-methyl butane, and stored at −80°C for GGT histochemistry (weight of the rats, 300–350 g; liver weight 9–13 g).
Assays for Protein Levels and CYP-Mediated Activity
Western Blot Assays
Five groups were used as follows: one group was untreated, two groups were treated with DEN and sacrificed twelve and twenty-four hours after the administration of DEN, and three groups were treated with CAPE twelve hours before DEN administration and sacrificed twelve hours after CAPE and twelve and twenty-four hours after DEN treatment, as described by Beltrán-Ramírez et al. (2008). Rats were sacrificed under ether anesthesia by perfusion with a physiological solution via the portal vein and processed immediately to obtain microsomes. The treatments did not affect the weight of the rat nor its liver weight. For Western blotting, a pool of four rats (180–200 g each) per group was used (liver weight 5–7 g). Microsomal protein (30 µg/lane) was separated using 10% polyacrylamide gel electrophoresis using 2× loading buffer (Sambrook and Russell 2001). Proteins were transferred onto polyvinylidene fluoride membranes, blocked overnight at 4°C with 100 mmol/L of glycine, 1% bovine serum albumin, and 5% nonfat dry milk in a solution containing phosphate-buffered saline with 1% Triton X-100. Membranes were probed with anti-rat CYP1A2 and CYP2B1/2 antibodies for one hour at room temperature followed by incubation with a horseradish peroxidase–conjugated secondary antibody for one hour at room temperature. The specific protein bands were visualized by chemiluminescence (Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA). Densitometric analysis was performed using Image J (version 1.73v) and statistics with GraphPad Prism (version 3.0, GraphPad Software, Inc., La Jolla, CA, USA).
Chemoprevention Provided by Specific CYP Inhibitors
Based on previous studies showing that CYP is involved in DEN bioactivation, two CYP inhibitors were used as follows: for CYP1A1/2 inhibition, α-naftoflavone (α-NF) was administered by gavage using two doses of 50 mg/kg for forty-eight hours and twenty-four hours before DEN, and the animals were sacrificed twenty-four days later (n = 4); for CYP2B1/2 inhibition, SKF525A was administered ip using one dose of 50 mg/kg two hours before DEN, and the animals were sacrificed twenty-four days later (n = 4 for α-NF and n = 8 for SKF525A). Animals were sacrificed under ether anesthesia. The livers were excised, washed in saline solution, frozen in 2-methyl butane, and stored at −80°C for GGT histochemistry (weight of the rats, 220–230 g; liver weight, 5–7 g).
Inhibition of DEN Metabolism In Vitro
Diethyl nitrosamine denitrozation was measured using nitrite determination. Microsomes were used for this analysis with rats that were untreated or pretreated with CAPE twelve hours before sacrifice. Microsomes from untreated rats were used as normal controls. Untreated microsomes were incubated with 50 mM DEN (as a positive control), untreated microsomes were preincubated with 50 mM CAPE or 50 mM SKF525A before the addition of DEN, and CAPE-pretreated microsomes were incubated with 50 mM DEN (n = 5). For nitrite determination, a specific colorimetric assay kit was used according to the manufacturer’s instructions (Cayman Chemical Co., Ann Arbor, MI, USA).
GGT Histochemistry
Frozen liver sections of 20 µm thickness were stained for GGT activity (Rutenburg et al. 1969). Images were captured using a digital camera (Color View 12, Soft Imaging System GmbH, Germany). Only GGT areas that were larger than 0.01 mm2 were registered and quantified using AnalySIS software (AnalySIS, Soft Imaging System GmbH, Germany).
Statistical Analysis
Data were analyzed using the one-way ANOVA test and further analyzed using the Bonferroni pairwise multiple comparison post hoc test (Klockarts et al. 1995). Analyses were done using SigmaStat software (version 3.1.1, Systat Software, Inc., Point Richmond, CA). In all tests, the level of significance was p < .05.
Results
CAPE Reduces CYP1A2 and CYP2B1/2 Protein Levels
As previously shown, CAPE modified CYP1A2 and CYP2B1/2 enzymatic activities (Beltran-Ramirez et al. 2008) and mRNA expression (Beltran-Ramirez et al. 2010) as a possible chemopreventive mechanism. To complement these results, we quantified the CYP protein after CAPE administration. Pooled microsomes (n = 4) were used to evaluate the protein quantity by Western blot analysis. Twelve and twenty-four hours after carcinogen administration, DEN-treated rats displayed CYP1A2 levels that had increased after twelve hours by 7.13% and twenty-four hours by 35.13% compared to untreated rats. The protein level of the vehicle (dimethyl sulfoxide) control showed an increment of 23.26% with respect to untreated rats. Rats that were treated with CAPE twelve hours before DEN treatment showed a protein level that had decreased by 8.83% twelve hours after DEN and 87.19% twenty-four hours after DEN, respectively, compared to untreated rats. Rats that received CAPE treatment after twelve hours showed decreased protein levels at 39.04% compared to untreated rats (Figure 1). At twelve hours and twenty-four hours after DEN administration, DEN-treated rats displayed slight increases in CYP2B1/2 at 6.09% and 4.25%, respectively, compared to untreated rats. CAPE administration twelve hours before DEN administration diminished the protein level at twelve hours and twenty-four hours after DEN treatment by 9.63% and 53.15%, respectively, compared to untreated rats. After twelve hours of CAPE administration, CAPE-treated rats displayed a decrease in the protein content of pooled microsomes of 27.64% (Figure 2). These results show that CAPE modulates CYPs at different levels, including protein expression, which could be affecting the bioactivation of carcinogens and, consequently, cancer development.
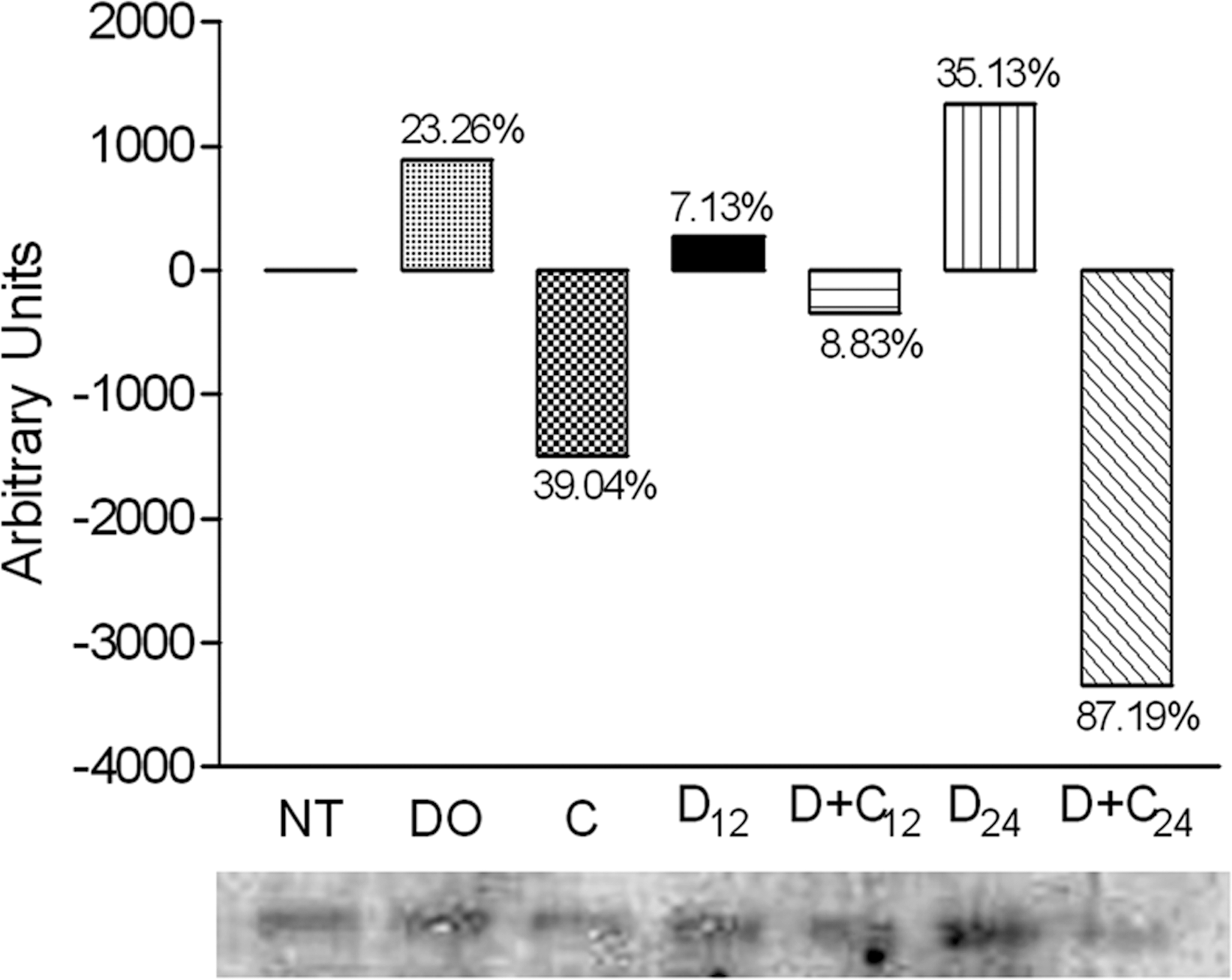
Quantitation of CYP1A2 protein levels. Protein analysis was performed using Western blotting with anti-rat CYP1A2 antibodies, from the samples pooled from four animals per treatment. Level 0 represents the rats without treatment (NT); DO, the vehicle control with DMSO; C, CAPE and sacrificed twelve hours later; D12, sacrificed twelve hours after DEN treatment; D+C12, CAPE administration twelve hours before DEN and sacrificed twelve hours after DEN administration; D24, sacrificed twenty-four hours after DEN treatment; and D+C24, CAPE administration twelve hours before DEN and sacrificed twelve hours after DEN administration.
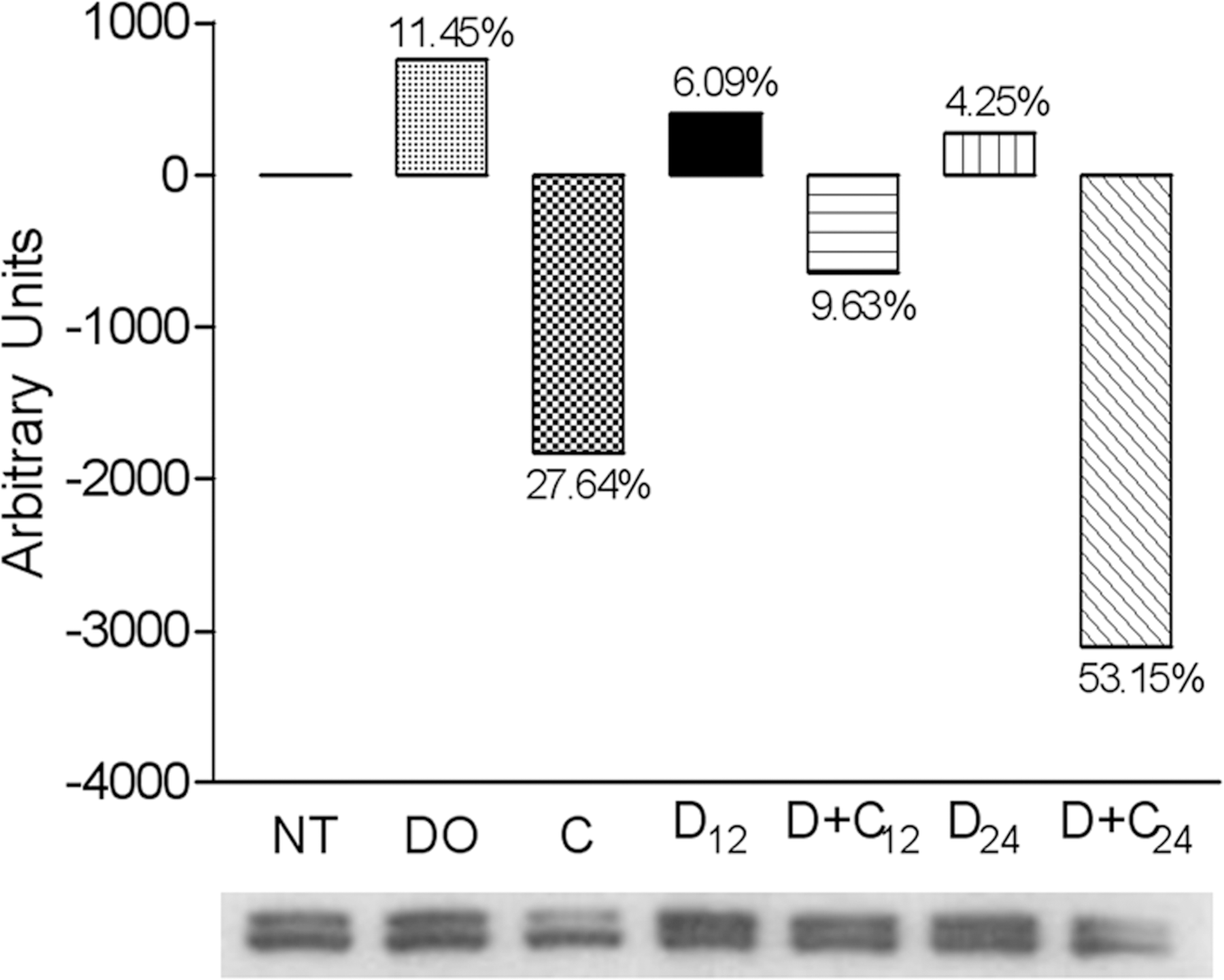
Quantitation of CYP2B1/2 protein levels. Protein determination was performed using Western blot analysis with anti-rat CYP2B1/2 antibodies, from the samples pooled from four animals per treatment. Level 0 represents the rats without treatment (NT); DO, the vehicle control with DMSO; C, CAPE treatment and sacrificed twelve hours later; D12, sacrificed twelve hours after DEN; D+C12, CAPE administration twelve hours before DEN and sacrificed twelve hours after DEN administration; D24, sacrificed twenty-four hours after DEN; and D+C24, CAPE administration twelve hours before DEN and sacrificed twelve hours after DEN administration.
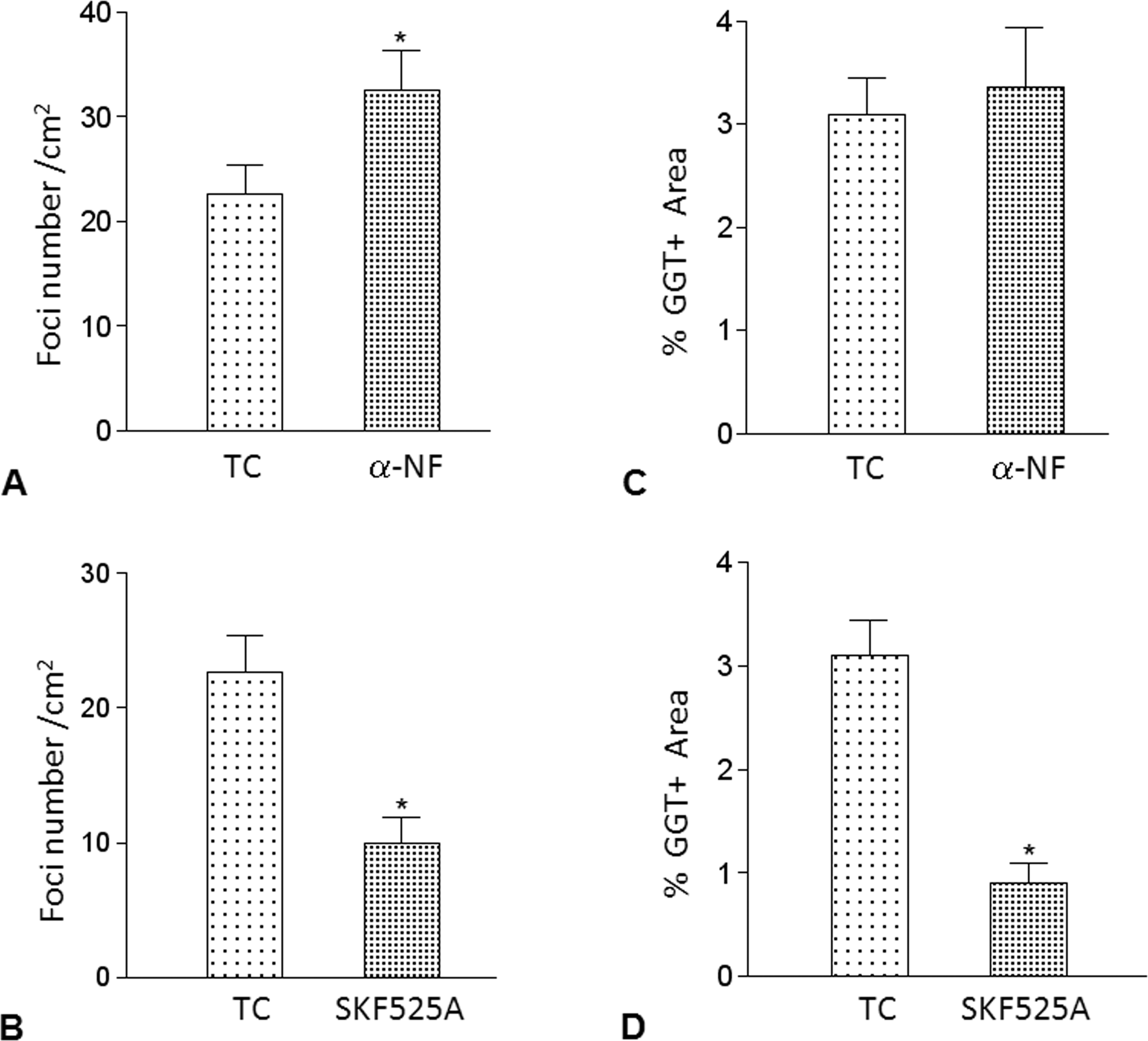
Inhibitory effect of α-NFe and SKF525A on preneoplastic lesion induction. We administered two doses of α-naphthoflavone (α-NF) twenty-four hours and forty-eight hours before DEN administration and one dose of 50 mg/kg of SKF525A two hours before DEN administration. The groups were sacrificed twenty-four days after DEN treatment (n = 4 for α-NF and n = 8 for SKF525A) (TC). We used a GGT+ histochemical technique to detect preneoplastic lesions. (A) Number of foci/cm2 with α-NF; (B) GGT-positive area with α-NF; (C) Number of foci/cm2 with SKF525A; and (D) GGT-positive area with SKF525A. *p < .05 compared with TC, Bonferroni test.
CYP2B Mediates DEN Bioactivity
To confirm whether the presumptive isoforms of DEN CYP metabolizing enzymes, CYP1A2 or CYP2B1/2, were involved in DEN bioactivation, we used specific inhibitors to evaluate the early effects following carcinogenic treatment, including the appearance of preneoplastic lesions at twenty-four days. We used α-NF and SKF525A to evaluate CYP1A1/2 and CYP2B1/2, respectively. Two doses of α-NF at forty-eight hours and twenty-four hours before DEN administration did not decrease the number of preneoplastic lesions twenty-four days after DEN administration. On the contrary, α-NF caused a statistically significant increase in the number of preneoplastic lesions (Figure 3A, 3B). The inhibitory CYP effect of α-NF did not interfere with the effects of DEN bioactivation. SKF525A prevented preneoplastic lesion formation, which was indicated by a 55.6% decrease in foci per cm2 and 71% in the GGT+ area (Figure 3C, 3D). Interestingly, the reduction that was observed in preneoplastic lesions with SKF525A was similar to previous observations with CAPE (Beltran-Ramirez et al. 2008). These results indicate that the CYP inhibitory effect of SKF525A interfering with CYP2B1/2 modulates DEN metabolism.
CYP2B1/2 Inhibition Diminishes DEN Bioactivation In Vitro
To obtain further evidence that CYP2B1/2 was the target of CAPE and the DEN bioactivating isoform, we used an in vitro assay to analyze the levels produced of nitrites, one of the principal metabolites of DEN activation, and nitrite derivatives from the denitrification pathway (Verna et al. 1996). In a sample of untreated rat microsomes incubated without DEN, there was no nitrite production. However, the addition of DEN triggered a substantial production of nitrites; we therefore used these levels as our reference to evaluate the effects of CAPE and SKF525A on nitrite levels produced. In microsomes from rats treated with CAPE and sacrificed twelve hours later, the nitrites produced after adding DEN diminished to 70.3% compared to the microsomes from untreated rats incubated with DEN. Thereafter, instead of microsomes that were obtained from CAPE-treated rats, this chemoprotective agent was added to untreated microsomes and later incubated with DEN, and it inhibited 68% of DEN-produced nitrites. Similarly, SKF525A was added to the microsomes of rats following DEN incubation, and we observed the inhibition of DEN-produced nitrites at 57%. These results confirm that CYP2B1/2 is important for DEN activation and that CAPE uses its chemoprotective mechanism to alter the metabolism of the carcinogen (Figure 4), which was similar to the effects of SKF525A.
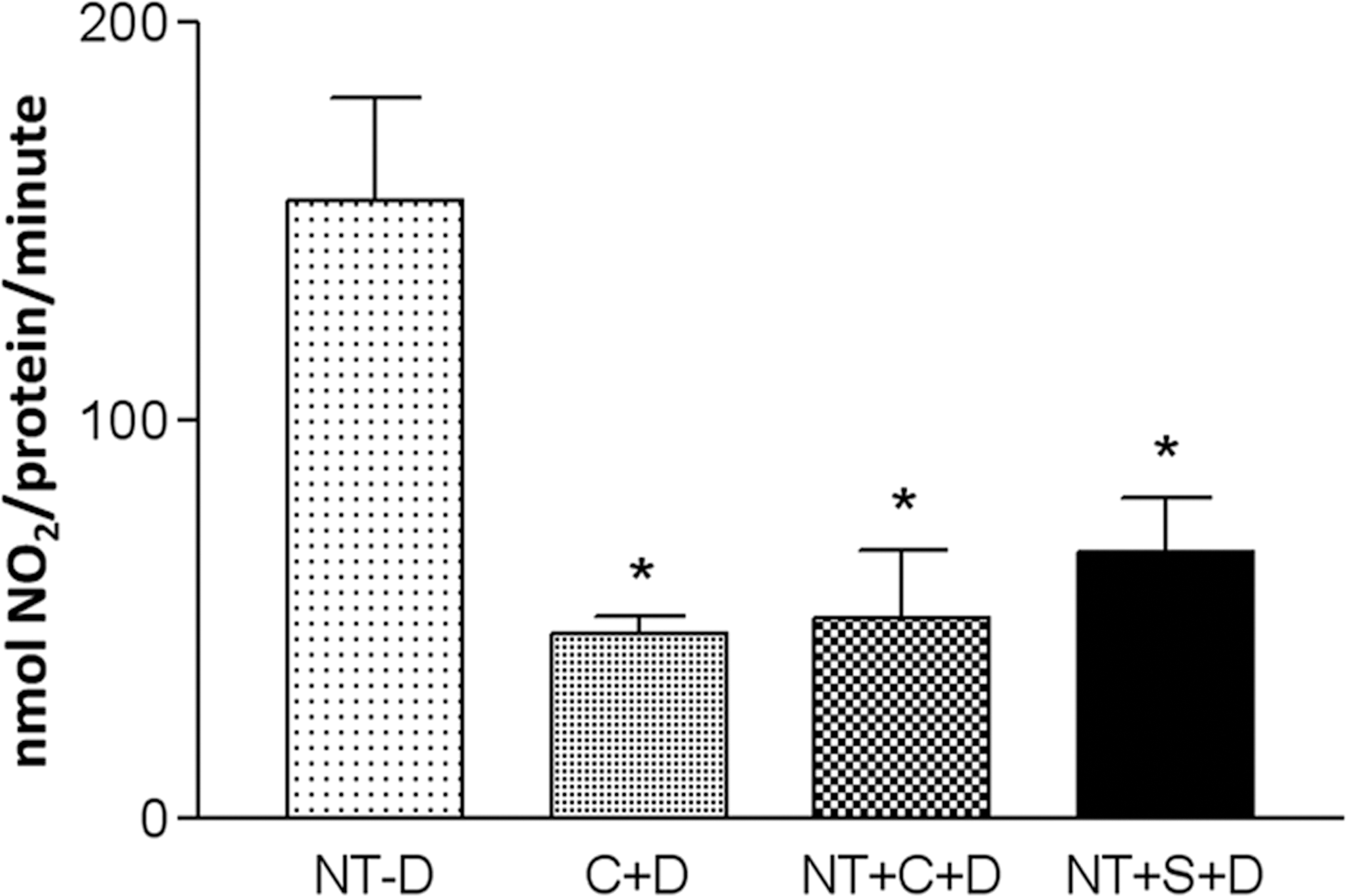
Nitrite inhibition by caffeic acid phenethyl ester and SKF525A on diethylnitrosamine (DEN) metabolism. To detect nitrites, we used a colorimetric assay with the microsomes of untreated rats (NT) that were given DEN (NT+D). In addition, we analyzed the microsomes of rats treated with caffeic acid phenethyl ester (C) twelve hours before sacrifice that were given DEN (C+D), the microsomes of NT treated with C and DEN (NT+C+D), and microsomes of NT treated with SKF525A and DEN (NT+S+N). *p < .05 compared to NT. Bonferroni test, n = 5.
Caffeic Acid Phenethyl Ester Diminishes Tumor Incidence
With the evidence that CAPE is a good chemoprotective agent with a specific mechanism of action upon carcinogen metabolism, we evaluated chemoprevention with respect to liver cancer. For this purpose, animals were evaluated twelve months after the administration of a single dose of CAPE. Caffeic acid phenethyl ester–pretreated rats showed a reduction of 43% in tumor incidence (three of seven rats without tumors), whereas the seven rats without CAPE displayed 100% incidence of tumors. These results confirm that CAPE is a good candidate as a chemoprotective agent in liver cancer, which requires further investigation (Figure 5).

Tumor development is inhibited by caffeic acid phenethyl ester in the modified resistant hepatocyte model. We used two groups (n = 7) of rats, A, with complete treatment of the model and B, with the complete treatment of the model and 20 mg/kg of caffeic acid phenethyl ester twelve hours before diethylnitrosamine administration. Both groups were sacrificed twelve months after diethylnitrosamine administration and with complete carcinogenic treatment. The liver was excised and processed for histochemical reaction for γ-glutamyl transpeptidase detection.
Discussion
The prevention of cancer is currently more feasible than finding a cure (Yao et al. 2011). Chemoprevention has been used for many years for different diseases. However, today’s research gives us the opportunity to probe many compounds and uncover more chemoprotective agents to improve health.
There are many chemoprotective agents, including flavonoids, vitamin C, vitamin E, garlic, pepper compounds, green tea, broccoli, coffee, and propolis. Most of the agents occur as part of our diet. Chemoprotective agents regulate the expression of drug-metabolizing enzymes because their activity may contribute to the sensitivity of different individuals to develop cancer (Moon et al. 2006; Ragin et al. 2010). Confirming the effects of CAPE on a specific CYP will help us elucidate the mechanism of CAPE as a chemoprotective agent in the modified resistant hepatocyte model of hepatocarcinogenesis, which may become a referent model (Bai et al. 2011; Beltran-Ramirez et al. 2008; Beltran-Ramirez et al. 2010; Carrasco-Legleu et al. 2006).
Different effects have been observed for CAPE as a chemoprotective agent. We observed that CAPE modulated CYP enzymatic activities and mRNA expression to promote chemoprotection. To understand the alterations of these enzymes, further investigation is necessary to complement our current information (Beltran-Ramirez et al. 2008).
The principal CYP isoforms CYP1A1/2, CYP2B1/2, and CYP2E1 mediate DEN bioactivation. In our carcinogenic model, CAPE acted as a chemoprotective agent without CYP2E1 modification (Bellec et al. 1996; Yamazaki, Inui et al. 1992). Therefore, we concluded that CYP2E1 was not related to the chemoprotective activities of CAPE (data not shown). Caffeic acid phenethyl ester decreases CYP1A2 mRNA (Beltran-Ramirez et al. 2010) and activity (Beltran-Ramirez et al. 2008), but not to the same magnitude that was observed in this work. The alcoholic extract of propolis diminishes the enzymatic activity of CYP1A1/2, which inhibits the carcinogen benzo[a]pyrene (Jeng et al. 2000). We cannot discard the hypothesis that CYP1A1/2 may metabolize DEN in our model. Despite our conclusions that CAPE modulated CYP1A1/2 mRNA, protein, and enzymatic activity, the administration of a specific CYP1A1/2 inhibitor did not affect the appearance of preneoplastic lesions. These results indicate that CYP1A1/2 inhibition does not affect DEN metabolism and does not participate in the chemoprotective activity of CAPE.
Previous reports have suggested that CYP2B1/2 participates in DEN metabolism (Bellec et al. 1996; Verna et al. 1996; Yamazaki, Inui et al. 1992; Yamazaki, Oka et al. 1992) and are in agreement with our results. Because CAPE decreased CYP2B1/2 at different levels, we used an inhibitor of this isoform to confirm whether these modifications were related to DEN inhibition. Administration of SKF525A, a specific inhibitor of CYP2B1/2, mimicked CAPE’s effects and decreased preneoplastic lesions. These results indicate that CYP2B1/2 inhibition interferes with DEN metabolism and that the progression of carcinogenesis is not effective. These results provide information for the mechanism of CAPE-mediated chemoprotection, which was not previously reported in liver cancer.
Although CAPE decreased DEN activation, further investigation was required using an in vitro assay. Activated DEN produces two principal metabolites, ethyl carbonium ion and nitrites (Verna et al. 1996). In our experiments, CAPE reduced nitrites when administered before or simultaneously with DEN, showing immediate CYP2B1/2 inhibition. These results resemble the effect of SKF525A, which reduces DEN activation to the same degree as CAPE, demonstrating that the chemoprotective agent modifies CYP2B1/2 activity while altering the process of carcinogenesis.
CYP2B1/2 is a subfamily that is expressed at low levels under normal conditions. Age is not a factor that modifies these CYPs (Imaoka et al. 1991). These CYPs are drug inducible and are major participants in cancer progression, suggesting that CYP activity is altered when DEN is administered as a carcinogen. However, the preincubation of microsomes with CAPE before the addition of DEN decreased the DEN-metabolizing activity of these CYPs, which shows that CAPE directly acts on the enzyme.
CYP2B plays an important role in several injury processes such as cancer, smoking damage by nicotine, and hormonal alterations (Khokhar et al. 2010). Although the participation of CYP2B in cancer development has not been elucidated, there are several other factors such as activation of the constitutive androstane receptor (CAR) and NF-kappaB induction (Price et al. 2008; Zordoky and El-Kadi 2009). Some compounds such as phenobarbital, pyrethrins, and several nonmutagenic carcinogens induce CAR-dependent CYP2B expression and activity. The activation of CAR results in a pleiotropic response that includes the stimulation of CYP2B isoforms, liver hypertrophy, increased cell proliferation, and the inhibition of apoptosis. Prolonged treatment with phenobarbital and phyrethrins results in the formation of altered hepatic foci and liver tumors (Elcombe et al. 2010; Kamino et al. 2011; Tien and Negishi 2006). In these cases, CYP2B isoforms promote cancer, which is consistent with the results observed in our work.
CYP2B1/2 is related to the detoxification of the metabolite derivatives, which are mediated by DEN activation, and decreased proliferation of precancerous cells in the liver cancer model (Arellanes-Robledo et al. 2010; Salcido-Neyoy et al. 2009). With other animal models such as the LEC rat, which is a mutant strain that displays hereditary hepatitis with severe jaundice and liver cancer, a decrease in CYP2B1/2 protein correlates to disease progression. Although there are no indications for the involvement of carcinogens or drug metabolism, CYP2B1/2 diminution is important to induce liver damage (Sugiyama et al. 1989). Each CYP isoform may participate in separate mechanisms to mediate cancer progression.
Our most important demonstration is that CAPE blocks the progression of liver carcinogenesis by reducing tumor incidence by 43% at twelve months following the administration of a single dose of CAPE in our cancer model.
In conclusion, a single dose of CAPE inhibits the hepatocarcinogenic process, modifying DEN bioactivation, which is reflected in early stages by decreasing preneoplastic lesions and in later stages by reducing tumor incidence. This compound is an excellent option to prevent cancer development, especially in high-risk patients. Further studies are needed for a detailed study of the effects of this chemoprotective agent in treatments to prevent or reverse the effects of cancer.
Footnotes
Acknowledgments
We would like to acknowledge the excellent animal technical support of UPEAL-Cinvestav from M. V. Z. María Antonieta López-López, Rafael Leyva Muñoz, Manuel Flores-Cano, Ricardo Gaxiola-Centeno, and UPEAL Chairman Dr. Jorge Fernández.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the Consejo Nacional de Ciencia y Tecnología (grant no. 39525-M).
