Abstract
The immune system functions primarily as a defense mechanism to provide protective immunity against microbial pathogens and cancer. The resulting protective responses occur through the complex interaction of tissues, cells, proteins, and molecular pathways that act in concert with other systems (e.g., nervous and endocrine) to provide the host with immunologic responses that cause pathologic processes seen primarily as inflammatory reactions. The pathologic responses can be attributed to either normal responses to infectious organisms and cancer cells, misdirected responses as in the case of hypersensitivity or autoimmune diseases, or deficient responses attributable to deficiencies or defects in components of the immune system. Pathologists need to have a basic understanding of the immune system to not only interpret findings as to their likely pathogenesis, but also to predict when the immune system may be a potential target. This review will be limited to a general overview of the basic immunologic responses and primary components involved.
General Overview
The immune system is composed of circulating cells and lymphoid organs that are categorized as primary and secondary lymphoid tissues. Primary lymphoid tissues for most mammalian species include the bone marrow and thymus, and they are the respective sites of B and T lymphocyte antigen-independent proliferation and differentiation, selection, and maturation processes before migrating to the peripheral lymphoid tissues as effector lymphocytes. The spleen, lymph nodes, tonsil, and lymphoid nodules are secondary lymphoid tissues, and they are the sites where mature B and T lymphocytes initiate immune responses and undergo antigen-dependent proliferation and activation resulting in humoral and cell-mediated immune responses to antigen. Because these lymphoid tissues can be direct targets of treatment or indirect targets through secondary stress effects, it is essential that pathologists not only accurately record any observations but also that the pathology report accurately interprets the findings in the context of all other study data to differentiate the two. The overview of the pathology of these lymphoid tissues has been extensively covered in previous publications and in a companion article in this volume of Toxicologic Pathology (Maronpot 2006). Additionally, The Society of Toxicologic Pathologists has published a Best Practice Guideline for the Routine Pathology Evaluation of the Immune system focuses on the morphologic parameters (Haley et al. 2005).
There are two categories of immune responses based on their specificity for antigen: innate (nonspecific) immunity and adaptive or acquired (specific) immunity. A basic understanding of these immune responses will facilitate the pathologist’s understanding of the toxic responses of the immune system. These two highly intertwined responses, innate and adaptive immunity, consist of cells, recognition molecules, and effector functions that provide rapid, broad responses in the case of innate immunity to slower, highly specific responses of adaptive immunity that are driven in part by the early innate responses (Table 1; Snyder 2011).
The major cells, recognition molecules, and effector mechanisms of innate and adaptive immunity.
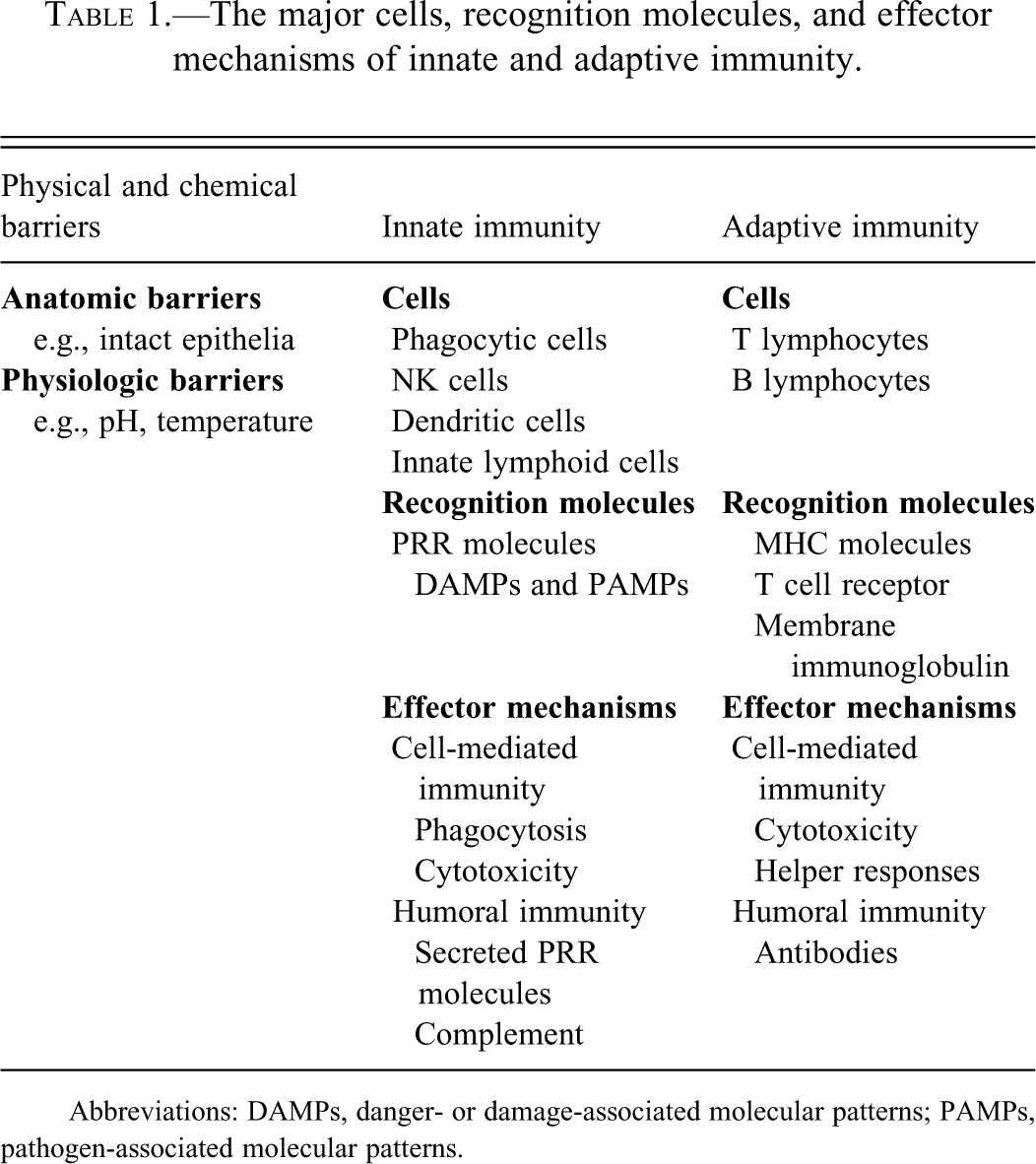
Abbreviations: DAMPs, danger- or damage-associated molecular patterns; PAMPs, pathogen-associated molecular patterns.
Following the mechanical and physical barriers, innate immunity is the first line of defense in response to microbial pathogens and damaged tissues that initiate inflammatory and protective responses (Figure 1). The innate immune system has highly conserved, germ line–encoded, nonspecific (invariant) pattern recognition receptor (PRR) molecules that sense “danger” in the form of infectious agents, pathogen-associated molecular patterns (PAMPs), or danger-associated molecular patterns (DAMPs) associated with tissue damage (Kumar et al. 2009; Pedra et al. 2009; Sutterwala et al. 2007). The innate immune system is a short-lived response with no intrinsic mechanism for self-renewal or clonal expansion that directs the adaptive immune system when there is antigen persistence or continued tissue damage. The adaptive immune system is considered to be more refined, with antigen-specific (highly variant) recognition molecules expressed by T and B lymphocytes that have the capacity for clonal expansion and memory. Although this review will present basic immunologic concepts as they pertain to pathogenesis, the pathologist must understand the delineation of innate and adaptive immunity is becoming increasingly blurred as it is becoming evident that there are specialized cells, recognition molecules, and effector functions that have attributes of both innate and adaptive immunity.
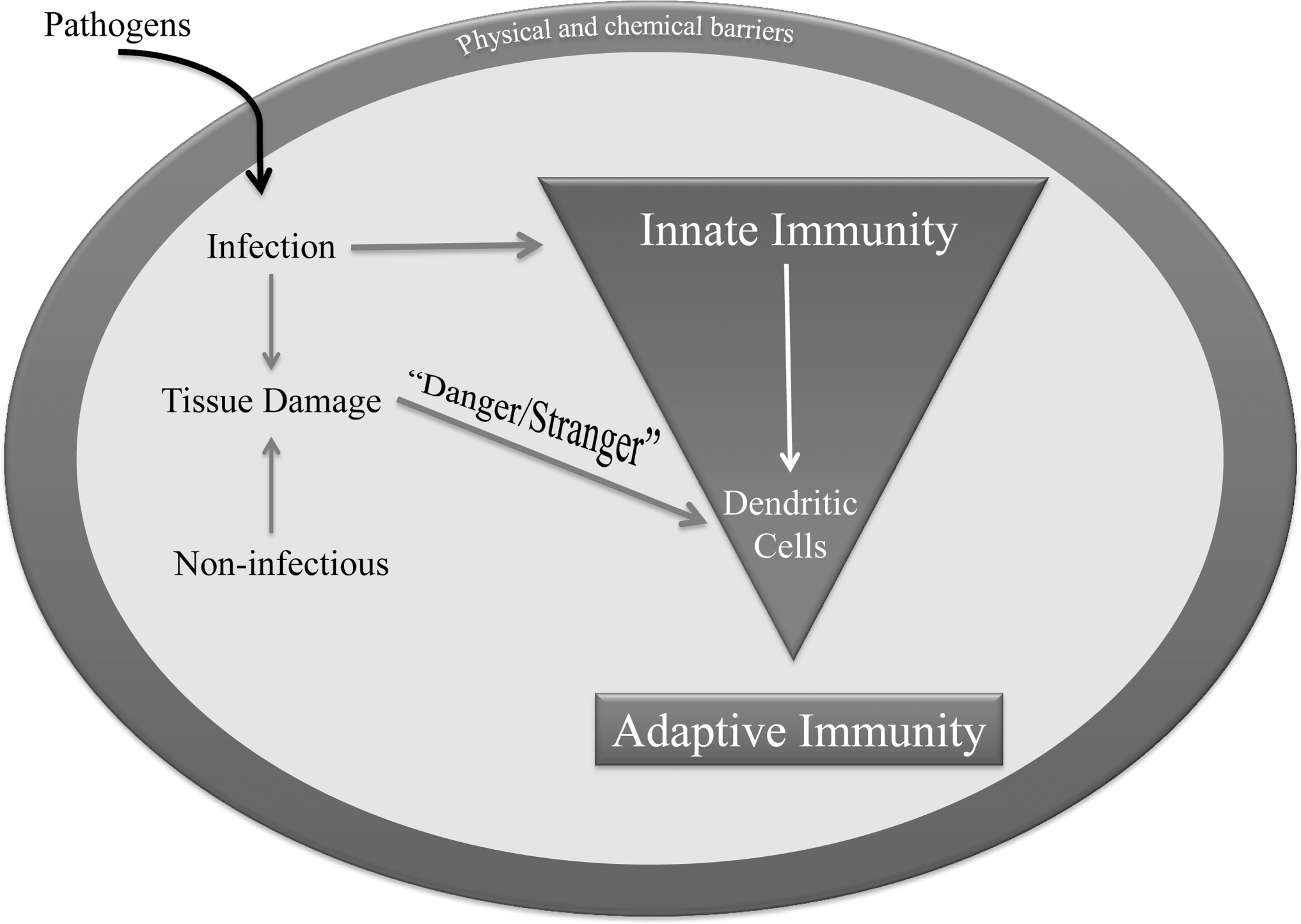
Relationship between innate and adaptive immunity responses to tissue damage.
Innate Immunity
Although the cellular component of innate immunity has historically centered on myeloid cells, mononuclear and polymorphonuclear phagocytic cells, eosinophils, dendritic cells, and lymphoid-derived natural killer (NK) cells, there is increasing evidence for inclusion of additional innate lymphoid cells that appear to be highly specialized and that also contribute to lymphoid tissue development and tissue remodeling following damage. The primary recognition molecules of the innate immune system have been an intense area of research and are currently a major focus for the pathogenesis of a number of inflammatory conditions (Asquith and Powrie 2011; Saleh and Trincheri 2011).
Innate recognition molecules provide the host with the ability to recognize “stranger and danger”–associated stimuli, pathogens, or endogenous molecules associated with tissue damage, by cells other than T and B lymphocytes, namely, antigen-presenting cells such as dendritic cells and macrophages (Kumar el al. 2009). The innate PRRs include secreted, transmembrane, and cytosolic molecules including the Toll-like receptor (TLR) family, the nucleotide-oligomerization domain–like receptor (NLR) family, and the retinoic acid–inducible gene I–like receptor family (RLR). These receptor families initiate signaling cascades that result in the production of cytokines such as type 1 interferons (IFN α and β), an important family of innate immunity cytokines, that have antiviral and immunoregulatory functions. Transmembrane (TLR1, 2, 4, 5, 6, and 11) and intracellular (TLR3, 7, 8, 9) TLRs sense unique viral nucleic acid sequences. Retinoic acid–inducible gene-1–like receptors also sense unique viral nucleic acids in the cytoplasm, and NLRs sense bacterial components in the cytoplasm.
The sensing of pathogens by the innate immune system PRRs also results in the production of the IL-1 family of pro-inflammatory cytokines (IL-1β, IL-18, and IL-33). The production of active forms of these cytokines is tightly regulated through post-translational mechanisms involving the conversion of pro-forms of the cytokines into active forms by caspase 1, which is highly regulated by cytosolic, multiprotein complexes, called inflammasomes, which are essential for inflammatory responses (Figure 2; Bauernfeind 2011; Khare et al. 2010). IL-1β is a pleotrophic inflammatory cytokine involved in innate and adaptive immune responses, and IL-18 is an important inducer of IFN-γ by NK cells and T lymphocytes that polarizes immune responses toward a T helper 1 (TH1) response.
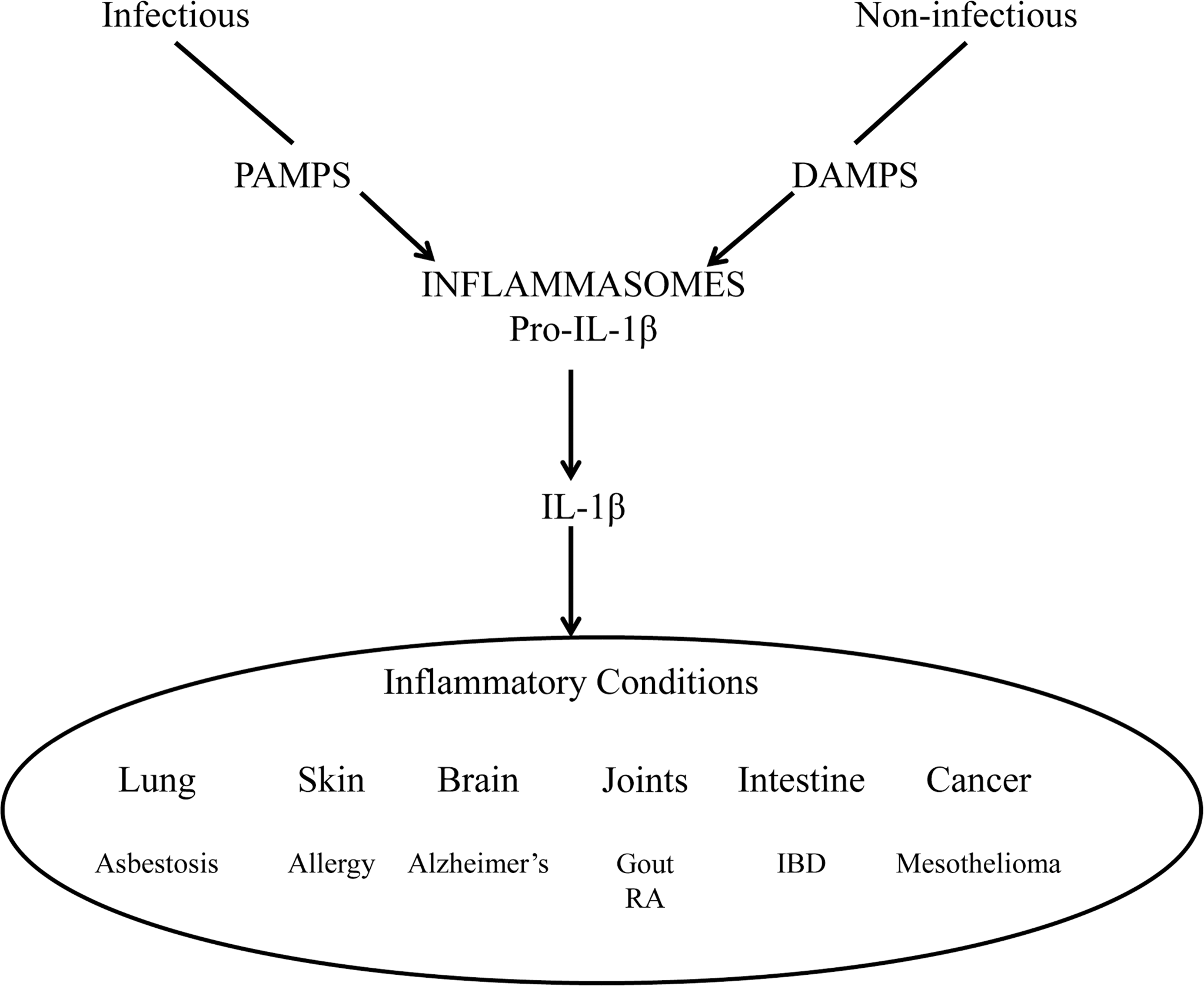
Infectious and noninfectious signaling through pathogen-associated molecular pattern and danger-associated molecular pattern molecules, respectively, resulting in activation of the inflammatory cytokine IL-1β and its association with diverse inflammatory conditions.
There is an ever-expanding family of innate lymphoid cells (ILC) that are involved in innate antimicrobial responses, lymphoid tissue formation, and tissue remodeling (Figure 3; Spits and DiSanto 2011). Natural killer cells are ILCs that are now recognized to be a phenotypically and functionally diverse population of cells whose function extends beyond their ability to recognize and kill infected or stressed cells to include other non-infection-related functions. A second ILC population includes the lymphoid tissue inducer (LTi) cells that function with stromal cells and lymphotoxin-β to induce the formation of lymph nodes during embryogenesis and the formation of lymphoid follicles in the gut and in postnatally restoring damaged lymph nodes (Spits and DiSanto 2011; Van de Pavert and Mebius 2010; Randall et al. 2008).
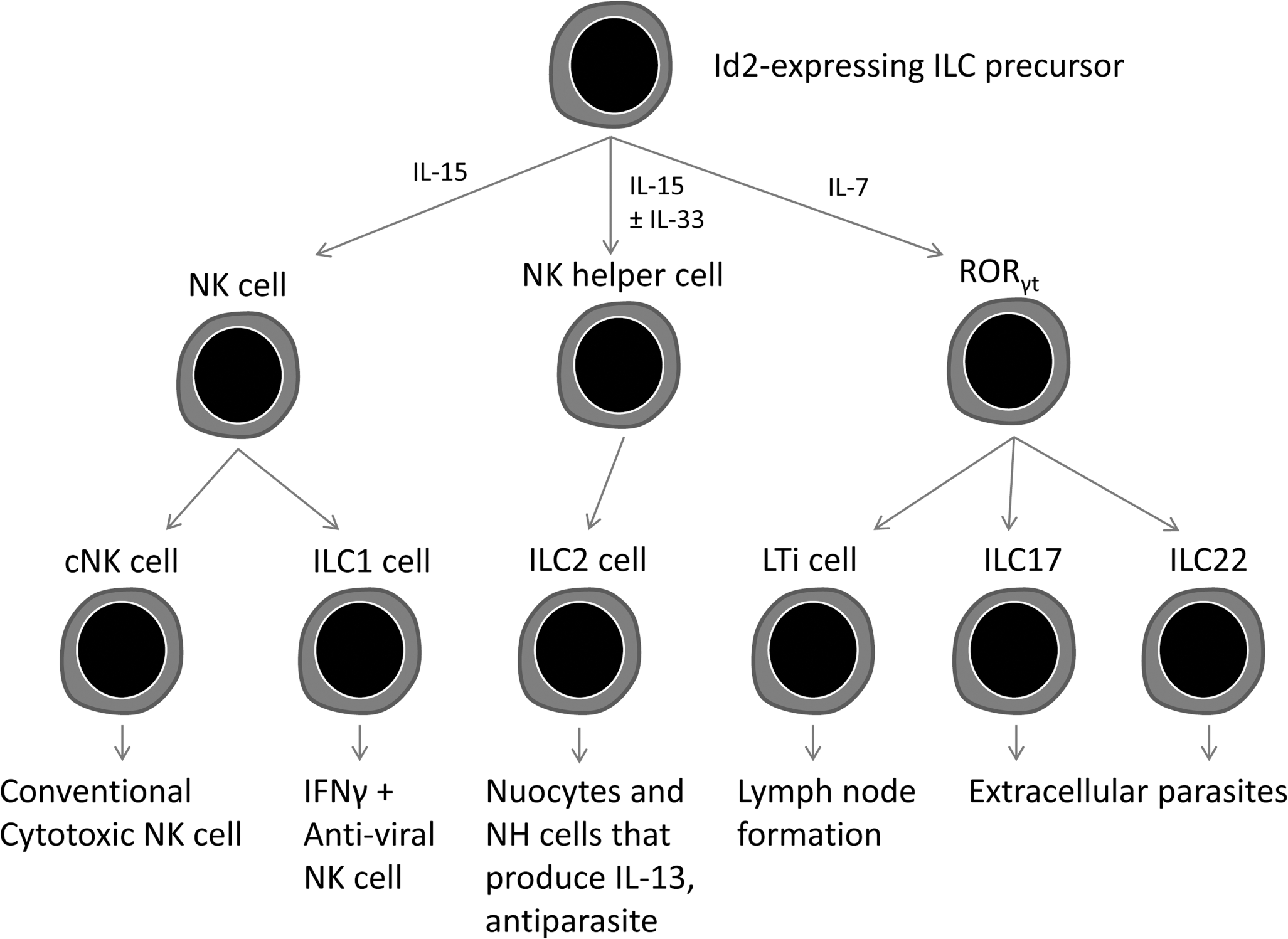
The family of innate lymphoid cells (ILC) that develop from Id2-dependent precursors into functionally distinct subsets.
LTi cells are characterized by the transcriptional repressor Id2, the transcription factor RORγt, and IL-7. Additional populations of LTi cells have been identified that produce IL-17 and/or IL-22 that are involved in tissue repair and immunity. IL-17 is a family of inflammatory cytokines primarily involved in neutrophil recruitment and the formation of germinal centers. The majority of IL-17 is produced by sentinel innate cells, γδ T cells, invariant NK cells, lymphoid tissue inducer–like cells, NK cells, and myeloid cells associated with the lungs, skin, and intestinal mucosa, where it influences the production of antimicrobial agents and synthesis of tight-junction proteins (Cua and Tato 2010). IL-22 is a member of the IL-10 cytokine family that activates epithelial cells of the gut and skin to produce antimicrobial products.
Additional highly specific ILC cells that appear to be separate from the aforementioned cells have been identified that have characteristics of NK and LTi cells that are found primarily in inflamed mucosal tissues and produce specific cytokine profiles (e.g., ILC17 and ILC22). These ILC cells are analogous to the TH17 and TH22 subsets of CD4+ T lymphocytes of adaptive immunity.
Another unique subpopulation of ILCs is the ILC2 family of cells, which includes the natural helper cells, multi-potent progenitor type 2 cells, nuocytes, and innate type 2 helper cells (Koyasu and Moro 2011; Saenz et al. 2010). Although the four ILC2 cell types differ in their anatomical location, they share the common function of driving a TH2 response to allergens and helminth infections as a response to epithelial-derived thymic stromal lymphopoietin, IL-25, and IL-33. Not only does this functional diversification of the innate immune system result in polarized cytokine production, which is recognized as a major driving influence on the adaptive immune system, but it also has been shown to have a role in the pathogenesis of number of inflammatory conditions, such as inflammatory bowel disease, when dysregulated (Buonocore et al. 2010; Strober and Fuss 2011).
Adaptive Immunity
Adaptive immunity is composed of cell-mediated immune responses, mediated by T lymphocytes against intracellular pathogens, and humoral immunity mediated by B lymphocytes against extracellular pathogens (Figure 4). Adaptive immunity, unlike innate immunity, is characterized by antigen specificity, diversity, memory, and self–nonself recognition. Antigen and self–nonself recognition is mediated by highly specific membrane molecules. Antigen recognition by mature B and T lymphocytes is mediated by membrane-bound immunoglobulin and the T cell receptor (TCR), respectively (Haley et al. 2005). Unlike membrane-bound immunoglobulin on B lymphocytes, which can recognize intact antigens alone, TCRs can recognize only peptide fragments of antigen that are associated with major histocompatability complex (MHC) molecules. This co-receptor requirement is the basis of MHC restriction and the result of positive selection in the thymus, which is responsible for central tolerance or self-nonself recognition.
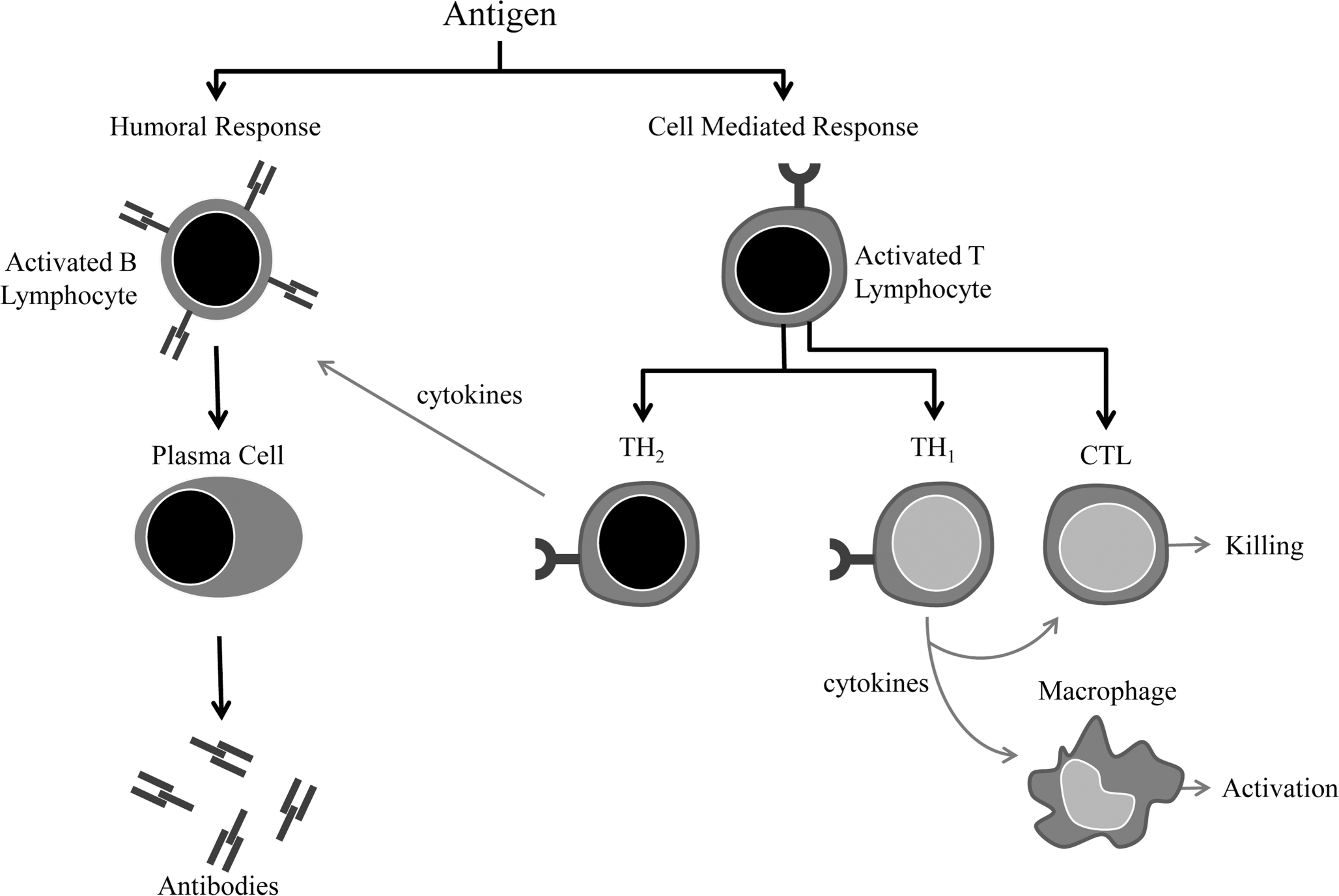
General overview of the humoral and cell-mediated arms of adaptive immunity.
T Lymphocytes
In most species, the majority of T lymphocytes express αβ TCRs, and their activation results in adaptive immune responses. T cell receptors are linked to the CD3 molecule, which is invariant, and although they do not bind antigen, they do function in signal transduction. T lymphocytes require two signals for activation: signal one is provided by the TCR and MHC-antigen complex and the CD4 or CD8 MHC complex, and signal two is provided by CD28, an accessory molecule on the T lymphocyte, and its ligand B7, expressed on antigen-presenting cells. When T lymphocytes receive both signals, they are induced to express IL-2 receptors and produce IL-2, resulting in clonal expansion of a population of T lymphocytes of the same antigen specificity that then differentiate into populations of effectors and memory cells. The non-polymorphic, accessory molecules CD4 and CD8 functionally subdivide T lymphocytes into CD8+ cytotoxic (TC) lymphocytes and CD4+ helper (TH) lymphocytes, which are further classified into populations based on transcription factors and cytokine profiles, as depicted in Figure 5 (2011).
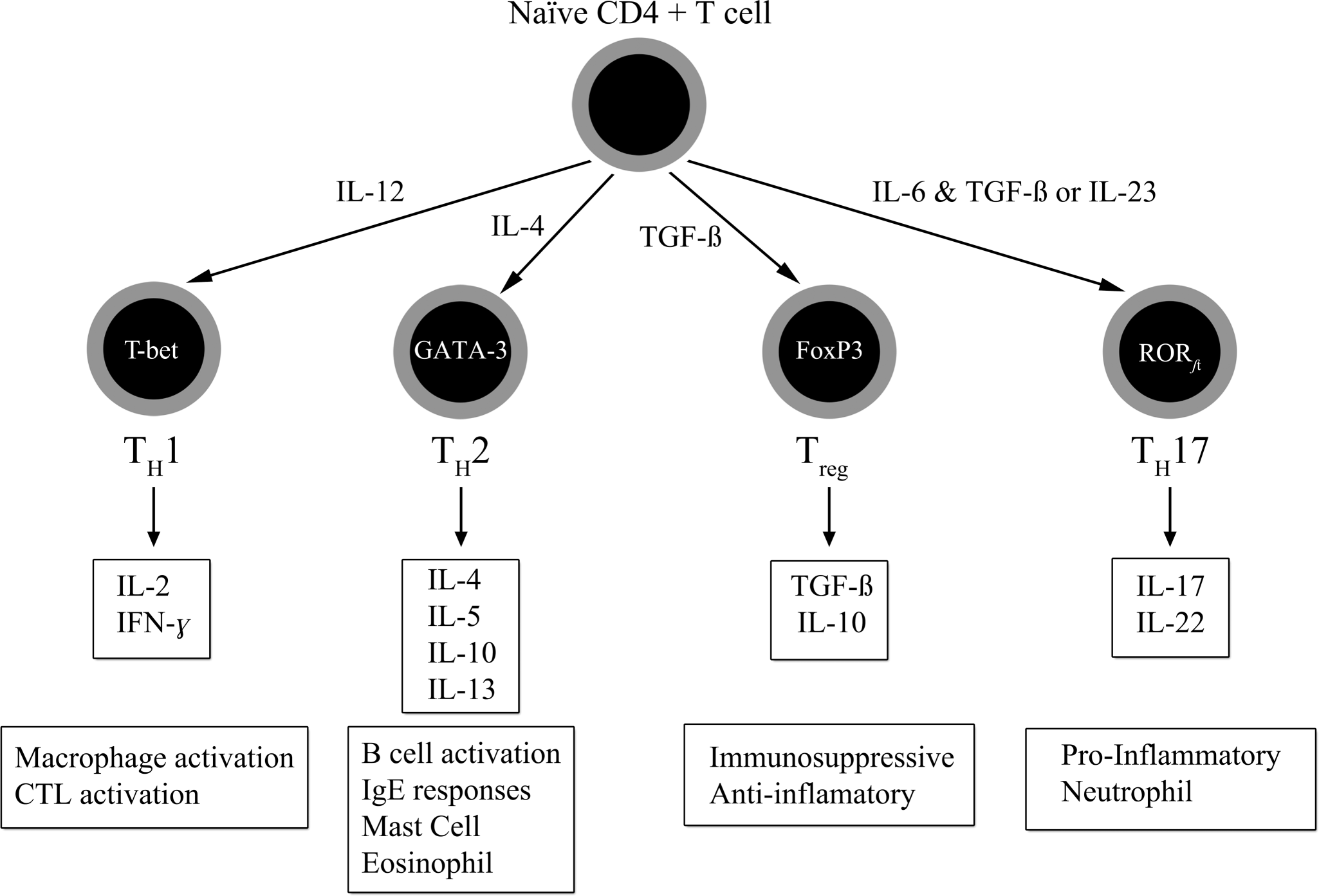
Functional classification of the major T helper lymphocyte subsets.
Differentiation of naïve CD4+ T lymphocytes into TH1 cells is dependent on antigen recognition by the TCR and MHC class II molecules and IL-12. TH1 responses are involved in the pathogenesis of cell-mediated immune responses, primarily mediated by Type VI hypersensitivity reactions and activation of TC cells and macrophages through their production of IFN-γ.
Differentiation of naïve CD4+ T lymphocytes into TH2 cells is dependent on antigen recognition by the TCR and MHC class II molecules and IL-4. TH2 responses are involved primarily in B lymphocyte activation and antibody responses involved in the pathogenesis of Type I hypersensitivity reactions, including allergy and asthma, and in defense of helminth infections primarily mediated through the production of IL-4, IL-5, and IL-6.
Another functionally distinct subpopulation of CD4+ T lymphocytes is the regulatory T lymphocyte (Treg ). Treg cells function to suppress the response of self-reactive CD4 cells that have escaped the negative selection process in the thymus. They are distinguished from other CD4 T lymphocytes by the expression of CD25 on the cell surface. Like TH1 and TH2 lymphocytes, Treg lymphocyte differentiation is driven by cytokine environments; however, where TH1 and TH2 lymphocyte activation occurs through transcriptional activators T-bet and GATA-3, respectively, Treg lymphocytes are activated through the transcriptional repressor FoxP3. TGF-β and IL-21 drive the differentiation of naïve CD4+ cells into TH17 cells that produce IL-17, IL-17F, IL-22, IL-6, and TNF-α.
A fourth subpopulation of CD4+ cells is the TH17 lymphocytes, whose differentiation and expansion is dependent on TGF-β, IL-6, IL-1, and IL-23 (Iwakura et al. 2011; Moissec et al. 2009). The expression of IL-23 receptor by TH17 is dependent on the transcription factor RORγt. TH17 lymphocytes, through the production of chemokines IL-17 and IL-22, induce the recruitment of monocytes and neutrophils to sites of inflammation, among other functions, as discussed previously in the innate immunity section. IL-17 is a family of cytokines designated IL-17A –F; IL-17A and F are involved in acute responses on the nature of four to eight hours after infection.
Although the concept of polarized immune responses greatly facilitates our understanding of the pathogenesis of a number of inflammatory diseases, it also suggests that the immune response has tremendous plasticity that is driven by the antigen and the elicited cytokine environment. Additional populations of T lymphocytes that express γδ TCR also develop in the thymus and migrate to the skin, intestine, mammary gland, and reproductive organs as intraepithelial lymphocytes (IELs). The γδ T lymphocyte can recognize native antigen in the absence of MHC binding and have limited diversity for antigen recognition. It has also been reported that γδ T lymphocytes are an important source of IL-17. This is a brief, overly simplified description of a complex network of highly specific lymphocyte subpopulations that have been shown to be important in initiating and maintaining appropriate and inappropriate immune responses.
B Lymphocytes
B lymphocytes develop in primary lymphoid organs and function in secondary lymphoid tissues as antigen recognition cells, and they are a component of the lymphoid follicle. Activation of B lymphocytes by antigen leads to the formation of secondary lymphoid follicles with germinal centers. Like the T lymphocyte, the B lymphocyte contains accessory molecules, co-stimulatory signals, and cytokines for activation. Naïve B lymphocytes, those not yet activated by antigen, circulate through the blood and lymph nodes via the high endothelial venules, and they have a lifespan of days to weeks. B lymphocytes that encounter antigen and help—in the form of co-stimulatory molecules and cytokines from T lymphocytes of the same antigen specificity in the paracortex of the lymph node—develop into low-affinity, antibody-producing plasma cells that reside in the medullary regions of the lymph node.
Other antigen-activated B lymphocytes clonally expand and form germinal centers where, under cytokine influence from T lymphocytes, they can undergo further development and refinement for antigen specificity (e.g., isotype switching, affinity maturation, and memory cell development) and develop into long-lived (months) plasma cells residing in the bone marrow. For the pathologist, understanding the specific anatomical location of these functions enhances his or her interpretation of lymph node findings. An activated lymphoid follicle has readily identifiable dark and light zones that correspond to areas of B lymphocyte proliferation and affinity maturation, and memory B cells, plasma cell development and isotype switching, respectively. The mantle region at the periphery of the lymphoid follicle is where mature naïve B lymphocytes reside.
Immunotoxicity
Immunotoxicity can be seen as immunosuppression or immunoenhancement (Figure 6).
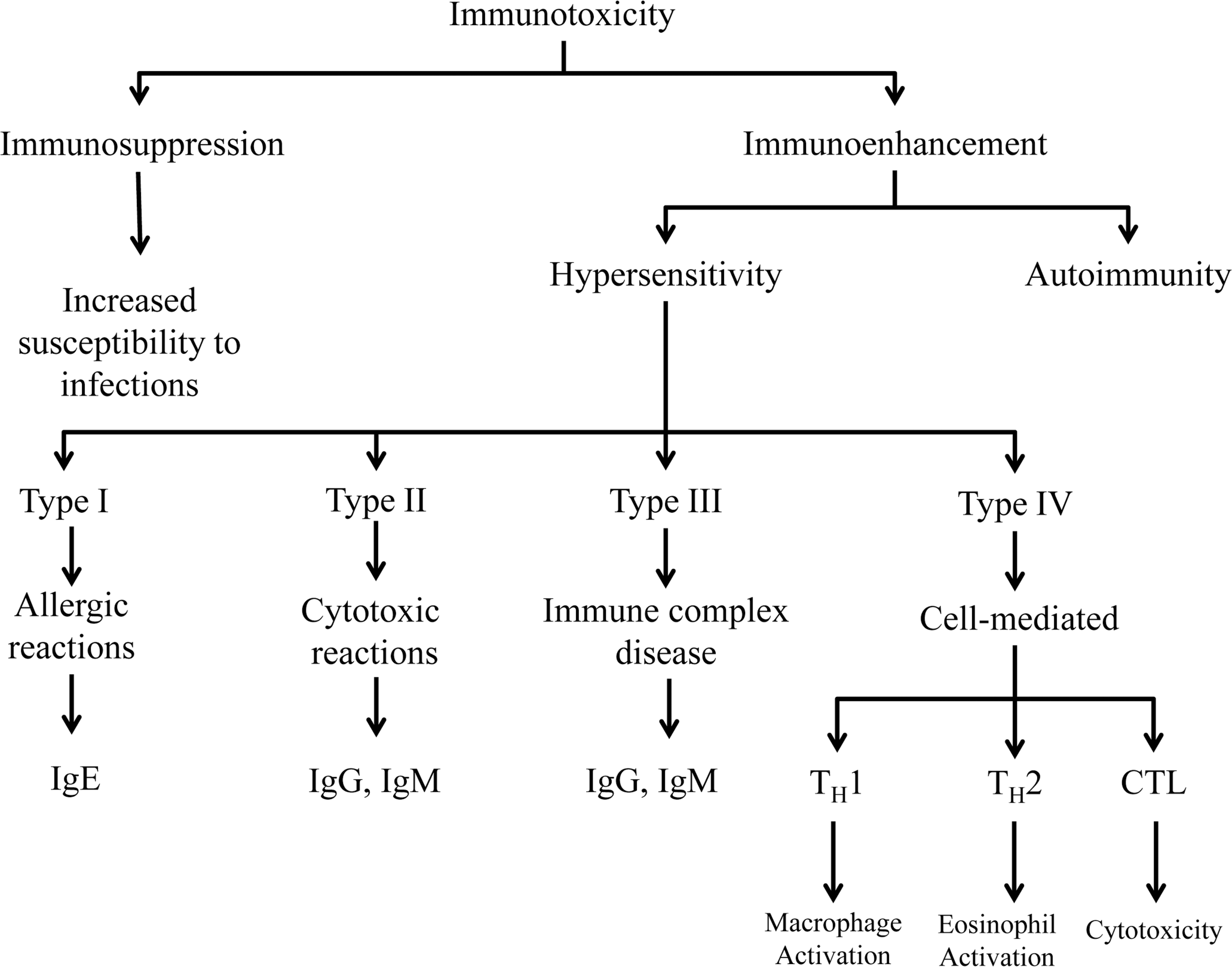
Overview of immunoenhancement and immunosuppression associated with immunotoxicity and the underlying pathogenesis.
Immunosuppression occurs when there is an inability to develop a normal immune response, and it is most often associated with an increased susceptibility to infections. Immunoenhancement refers to an inappropriate immune response as a result of autoimmunity or hypersensitivity. Biomarkers of immunotoxicity typically focus on individual components or products of innate and adaptive immunity or on specific aspects of cellular activation (e.g., cytokine production, cell proliferation). Even with the use of high-throughput assays, the complexity of the immune system with regard to diverse tissues and cells involved, as well as the redundancy and pleotrophic nature of the responses, makes interpretation of the data problematic. Some of these specific topics are covered in related manuscripts in this symposium issue.
Footnotes
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article. The authors received no financial support for the research and/or authorship of this article.
